A novel coronavirus (2019-nCoV) was identified as the cause associated with emerging pneumonia (COVID-19) detected in Wuhan on January 7, 2020. Because the number of patients has risen rapidly worldwide, COVID-19 has become a thorny international public health event. As of Mar 24, 2020, China has cumulatively diagnosed 81,747 cases and 147 new cases, while the number of cases in other countries has grown rapidly to a total of 291,070 confirmed cases and 22,027 new cases identified every day, as of this writing. Emerging studies suggest that COVID-19 preferentially afflicts the elderly, particularly those with chronic comorbidities (1,2). However, the clinical profiles of COVID-19 in refractory heart failure patients is unknown. This paper reports the clinical features in a group of end-stage heart failure patients with COVID-19, providing strong evidence of cardiac injury by the virus.
This study was approved by the institutional review board of Union Hospital, Tongj Medical College. All hospitalized patients in the authors’ department were screened for 2019-nCoV infection by nucleic acid test and chest computed tomography scans. Demographic information and clinical, biochemical, and radiological characteristics and treatment and outcome data were retrieved from electronic medical records. According to China Centers for Disease Control protocol, duplex reverse transcription-polymerase chain reaction or serum antibody test was performed to detect 2019-nCoV infection in throat swabs or blood samples. If respiratory samples tested positive by both open reading frame 1aboratory gene and nucleocapsid protein gene, the case was considered laboratory-confirmed. Presumed hospital-related transmission was suspected if hospitalized patients in the same wards became infected in a certain time period.
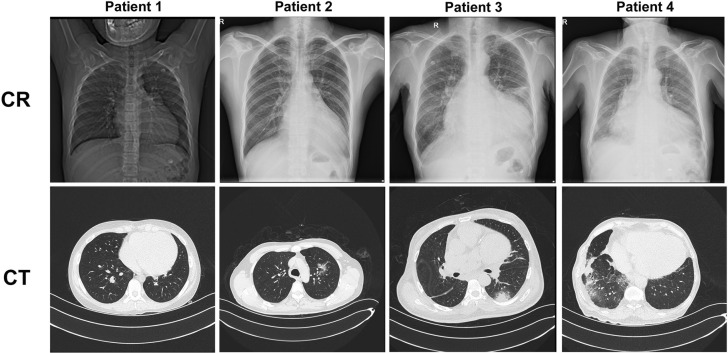
Four hospitalized patients with severe heart failure were retrospectively included who had been infected with COVID-19 between January 7, 2020, and March 15, 2020, in the authors’ department. All patients were transferred to an isolation ward since the diagnosis was confirmed or highly suspected. Three patients were suspected of having received hospital-related transmission because they were once in the same ward. None of the patients had fever during the illness, and they had just mild cough or fatigue at the time of diagnosis. Significantly enlarged left ventricle (Figure 1 ) and reduced left ventricular ejection fraction were observed in 4 patients, and all had New York Heart Association functional class IV. Interestingly, Patient #1 was had negative results for 2 consecutive nucleic acid tests but positive result for serum antibodies (immunoglobulin M [IgM] 69.12 AU/ml). Only 2 patients had typical ground-glass imaging changes on lung computed tomography scans (Figure 1). With exacerbations, Patient #3 and #4 were transferred to intensive care units, and both died 10 days after the first positive nucleic acid test result. Three patients had elevated troponin I (TNI) in the later period, especially in Patients #3 and #4, TNI increased significantly a few days before death. Moreover, the levels of C-reactive protein (CRP) and brain natriuretic peptide in patients 3 and 4 were significantly higher than those in the remaining 2 patients. It is also worth mentioning that testing results for patient 2 turned positive again after 2 consecutive negative test results for nucleic acid. The detailed information and treatment of patients are shown in Table 1 .
Figure 1.
Chest Radiography and Computed Tomography
Chest radiography and computed tomography of 4 end-stage heart failure patients with COVID-19.
Table 1.
Characteristics of 4 Hospitalized End-Stage Heart Failure Patients Infected With COVID-19
| Patient #1 | Patient #2 | Patient #3 | Patient #4 | |
|---|---|---|---|---|
| Demographics | ||||
| Age, yrs | 11 | 38 | 57 | 67 |
| Sex | Male | Male | Male | Male |
| DiagNosis | Myocarditis, 10 yrs after TOF operation | DCM, moderate MI | DCM, severe MI, severe AI | Severe AS and AI, severe MI |
| LVEDD (cm) | 5.5 | 7.2 | 10.8 | 8.5 |
| LVEF (%) | 22 | 26 | 22 | 30 |
| NYHA functional class | IV | IV | IV | IV |
| Heart failure course | 1 month | 2 yrs | 5 yrs | 6 yrs |
| Comorbidities | Tachycardia | Ventricular arrhythmia | Diabetes, COPD | Hypertension, diabetes, ventricular premature beat |
| Confirmation of COVID-19 | 12-Mar | 19-Feb | 10-Feb | 14-Feb |
| Symptoms at onset | Poor appetite, fatigue | Cough | Chest tightness, cough | Cough, sputum |
| Laboratory findings | ||||
| White blood cell count (Normal range: 3.5-9.5 g/l) | 8.48 | 5.56 | 5.40 | 12.18 |
| Lymphocyte count (Normal range: 1.1-3.2 g/l) | 2.63 | 1.40 | 0.67 | 0.52 |
| Lymphocyte% (Normal range: 20%-50%) | 31.0 | 25.2 | 12.4 | 4.3 |
| CRP (Normal range: 8.0 mg/l) | <3.14 | <3.14 | 143 | 103 |
| First BNP (Normal range: 100 pg/ml) | 250.4 | 2,085.7 | 82,22.1 | 4,450.0 |
| Latest BNP (Normal range: 100 pg/ml) | 109.8 | 603.0 | 20,700 | >5,000 |
| First TNI (Normal range: 26.2 ng/l) | 48.6 | 9.5 | 143.5 | 71.2 |
| Latest TNI (Normal range: 26.2 ng/l) | 3.8 | 71.3 | 3,749.6 | 1,518.2 |
| Plasma albumin (Normal range: 35-55 g/l) | 38.0 | 41.9 | 33.7 | 26.5 |
| D-dimers (Normal range: 0.5 mg/l FEU) | 0.22 | 0.23 | 0.47 | 20.0 |
| 2019nCoV nucleic acid test (throat swabs) | Twice negative | Twice positive | Twice positive | Twice positive |
| 2019nCoV antibody (10 AU/ml) | IgM 69.12 | No | No | No |
| CT findings | No abNormalities | Mild infectious lesions in bilateral lung | Patchy dense shadow and ground-glass changes in the lower lobe of bilateral lung | Multiple ground-glass changes in bilateral pulmonary zone |
| Anti-heart failure drugs | ||||
| Beta-blocker | Yes | Yes | Yes | Yes |
| Diuretics | Yes | Yes | Yes | Yes |
| Sacubitril valsartan sodium tablets | No | Yes | Yes | Yes |
| Recombinant human BNP | No | No | Yes | Yes |
| Intravenous inotropes | Yes | No | Yes | Yes |
| Anti-COVID-19 treatment | ||||
| Ribavirin | No | No | Yes | No |
| Antibiotic | No | Yes | Yes | Yes |
| Abidol | No | No | Yes | No |
| Interfero-α | Yes | Yes | Yes | Yes |
| Glucocorticoids | No | No | No | No |
| Intravenous immune globin | No | Yes | No | No |
| Oxygen supply | Yes | Yes | Yes | Yes |
| Clinical course | ||||
| Intensive unit care | No | No | Yes | Yes |
| ARDS | No | No | Yes | Yes |
| Mechanical ventilation | No | No | No | No |
| Outcome | Under therapy | Under therapy | Death | Death |
ARDS = acute respiratory distress syndrome; BNP = brain natriuretic peptide; CRP = C-reactive protein; CT = computed tomography; FEU = fibrinogen equivalent units; IgM = immuNoglobulin M; LVEDD = left ventricular end-diastolic diameter; LVEF = left ventricular ejection fraction; NYHA = New York Heart Association; TNI = troponin I.
This study reported for the first time 4 end-stage heart failure patients who were infected with COVID-19, 2 with severe presentation and the others mild. These patients showed some similar characteristics as described in previous reports (3). For instance, all 4 patients were male, consistent with previous findings that higher percentages of infection were found in men than in women. In addition, critically ill COVID-19 patients with heart failure also had typical lymphopenia and significantly increased CRP levels.
Patients with end-stage heart failure seemed to have a high mortality rate after infection with pneumonia. Older age, more comorbidities, poor general condition, and severe myocardial injury may be risk factors. The most novel finding was that the TNI levels of the 2 critically ill patients were 20-fold increased, indicating myocardial injury. Although there have been previous reports of myocardial damage in COVID-19 patients (4,5), those reports chose mainly nonspecific indicators such as creatine kinase-MB (CK-MB) and lactate dehydrogenase (LDH), which could be confounded by many other factors in clinic. In addition, CK-MB and LDH were not, in fact, significantly increased in those reports. The present findings provided definitely stronger evidence of myocardial injury by COVID-19.
The exact mechanism of myocardial injury caused by 2019-nCoV is not completely clear, but through present and previous findings, it is clear that 2019-nCoV infection can cause myocardial injury and is closely related to disease progression. The study was limited by small sample size. Longitudinal studies in a larger cohort of heart failure patients would help to understand the prognosis of the disease.
Footnotes
Please note: †Drs. Dong and Cai contributed equally to this work. The authors have reported that they have no relationships relevant to the contents of this paper to disclose.
The authors attest they are in compliance with human studies committees and animal welfare regulations of the authors’ institutions and Food and Drug Administration guidelines, including patient consent where appropriate. For more information, visit the JACC: Heart Failureauthor instructions page.
References
- 1.Wang D., Hu B., Hu C. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020 Feb 7 doi: 10.1001/jama.2020.1585. [E-pub ahead of print.] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yang X., Yu Y., Xu J. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020 Feb 24 doi: 10.1016/S2213-2600(20)30079-5. [E-pub ahead of print.] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Guan W.J., Ni Z.Y., Hu Y. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020 Feb 28 doi: 10.1056/NEJMoa2002032. [E-pub ahead of print.] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chen N., Zhou M., Dong X. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;15:507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wu C., Chen X., Cai Y. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020 Mar 13 doi: 10.1001/jamainternmed.2020.0994. [E-pub ahead of print.] [DOI] [PMC free article] [PubMed] [Google Scholar]