Abstract
Fusarium asiaticum belongs to one of the phylogenetical subgroups of the F. graminearum species complex and is epidemically predominant in the East Asia area. The life cycle of F. asiaticum is significantly regulated by light. In this study, the fungal blue light receptor white collar complex (WCC), including FaWC1 and FaWC2, were characterized in F. asiaticum. The knockout mutants ΔFawc1 and ΔFawc2 were generated by replacing the target genes via homologous recombination events. The two mutants showed similar defects in light-induced carotenoid biosynthesis, UV-C resistance, sexual fruiting body development, and the expression of the light-responsive marker genes, while in contrast, all these light responses were characteristics in wild-type (WT) and their complementation strains, indicating that FaWC1 and FaWC2 are involved in the light sensing of F. asiaticum. Unexpectedly, however, the functions of Fawc1 and Fawc2 diverged in regulating virulence, as the ΔFawc1 was avirulent to the tested host plant materials, but ΔFawc2 was equivalent to WT in virulence. Moreover, functional analysis of FaWC1 by partial disruption revealed that its light–oxygen–voltage (LOV) domain was required for light sensing but dispensable for virulence, and its Zinc-finger domain was required for virulence expression but not for light signal transduction. Collectively, these results suggest that the conserved fungal blue light receptor WCC not only endows F. asiaticum with light-sensing ability to achieve adaptation to environment, but it also regulates virulence expression by the individual component FaWC1 in a light-independent manner, and the latter function opens a way for investigating the pathogenicity mechanisms of this important crop disease agent.
Keywords: photobiology, transcription factor, White collar complex, Fusarium asiaticum, virulence
1. Introduction
Fusarium head blight (FHB), which is usually caused by the pathogen Fusarium graminearum, is known as a global problem devastating small grain cereal crops [1]. Phylogenetic species recognition has revealed that F. graminearum sensu lato comprises at least 15 biogeographically structured and phylogenetically distinct species, all of which are known as the Fusarium graminearum species complex, or FGSC [2,3,4,5,6,7]. Among the FGSC, Fusarium asiaticum belongs to one sub-lineage and is the predominant FHB agent species in East Asia [8]; it is especially prevalent in the wheat production zones of the Yangtze-Huaihe valley in China [9]. Besides being commonly associated with FHB, F. asiaticum has also been found to cause postharvest rot on asparagus spears and produce 3A-DON mycotoxin during host infection [10].
Usually, introgression of the disease resistance genes identified in natural sources into elite cultivars represents a reliable route for plant disease management [11]. However, plant sources for FHB resistance are unfortunately limited, and no fully resistant cultivars are yet available [12]. Controlling FGSC-caused diseases will benefit from an in-depth understanding of how the pathogens infect and spread inside the host. The availability of the F. graminearum genome [1] has greatly stimulated the research activity on identification of functional genes as well as pathogenicity factors of this phytopathogen [13,14,15]. However, the molecular mechanism of development and virulence regulation is less known in F. asiaticum than in F. graminearum, although there is an ongoing trend in which F. asiaticum becomes more aggressive and devastating than F. graminearum in the East Asian area [6,14,16].
Light is an important environmental factor that can extensively influence varied aspects of most living organisms on earth [17]. Filamentous fungi can use light as a general signal for regulating development, metabolism, sexual or asexual reproduction, and other life processes to adapt to a specific ecological niche [18,19,20]. At the molecular level, light is sensed by fungal photoreceptors, leading to activating or suppressing the transcription of photoresponsive genes, which are furthermore considered to result in the accumulation of light-sensing responses.
Light signaling is most extensively studied in the model species Neurospora crassa, in which carotenoid biosynthesis and morphological development, including the formation of asexual spores and protoperithecia, are notably regulated by blue light [21,22,23]. The analysis of blind mutants revealed that the white-collar 1 protein (WC-1), a transcription regulator which contains a light–oxygen–voltage (LOV) domain to bind the flavin chromophore, and the WC-2, a second transcription regulator without a chromophore-binding domain, can form a heterodimer called white-collar complex (WCC) to positively regulate the light-induced genes. Besides N. crassa, the molecular components for blue light sensing appeared to be widely conserved in the fungal genomes of Ascomycetes, Mucoromycetes, and Basidiomycetes. Moreover, genes under the control of the WCC can be either light responsive or not light responsive, and WC-1 and WC-2 can also have individual functions besides acting cooperatively as the WCC [24,25].
Many fungal species are causing detrimental diseases to mammals and plants, since the outcomes of all the epidemic diseases on earth can be determined by the triangular interactions among host–pathogen–environment [26]. Whether light signaling in fungi is involved in determining the disease outcomes has attracted considerable research attempts to characterize the photoreceptor functions in pathogenic fungi. However, the WCC regulatory circuit demonstrates functional variation among different species of fungal pathogens, and the significance of fungal light-sensing capacity for virulence expression is concluded on a case-by-case basis [27,28,29,30]. Moreover, despite these functional studies of the WCC orthologs in varied phytopathogenic fungi, the following questions remain mysterious: whether sensing light (or the absence of light) by phytopathogenic fungi is essential for pathogenicity, and how to exclude light-independent functions of WCC orthologs when evaluating their contribution in determining fungal pathogenicity.
In this paper, the cloning and characterization of the Fawc1 and Fawc2 genes in F. asiaticum demonstrate that both Fawc1 and Fawc2 are involved in light sensing and regulating pleiotropic fungal development processes, suggesting that FaWC1 and FaWC2 function cooperatively as the WCC to fulfill the photo receptor tasks. However, it is Fawc1 but not Fawc2 that is required for virulence. Functional domain analysis of FaWC1 reveals that the LOV and Zinc-finger domains are independently required for light sensing and virulence, respectively. These findings not only expand the knowledge of fungal photobiology but also provide novel insights about the mechanisms for diverged functions of WCC components in determining fungal virulence in a light-independent manner.
2. Materials and Methods
2.1. Fungal Strains and Culture Conditions
The wild-type strain EXAP-08 was previously isolated from postharvest asparagus spears with serious rot symptoms and characterized as F. asiaticum, and it is used as the recipient strain for genetic modifications in this study. The wild-type strain and the resulting mutants are listed in Table 1, and all the fungal strains were purified by single spore isolation and were stored as spore suspension in 20% glycerol at −80 °C. The fungal cultures were grown on complete medium (CM) (10 g glucose, 2 g peptone, 1 g yeast extract, 1 g casamino acids, nitrate salts, trace elements, 0.01% of vitamins, 10 g agar and 1 L water, pH 6.5) for mycelial growth, carboxymethyl cellulose (CMC) medium [31] for conidiation, and carrot agar medium [32] for sexual development. For light-responsive phenotypic analyses, plates were incubated for 3–7 days at 25 °C as indicated, either under illumination (20 W m−2 white light obtained with fluorescent bulbs) or under dark conditions.
Table 1.
F. asiaticum strains used in this study. LOV: Light–oxygen–voltage, ZnF: Zinc finger.
| Name | Genotype | Reference |
|---|---|---|
| EXAP-08 | Wild type | [10] |
| ∆Fawc1 | Knockout mutant, Fawc1::Hyg | This study |
| ∆Fawc2 | Knockout mutant, Fawc2::Hyg | This study |
| ∆Fawc1-C | Fawc-1 complemented transformant of ΔFawc-1 | This study |
| ∆Fawc2-C | Fawc-2 complemented transformant of ΔFawc-2 | This study |
| ∆Fawc1-C∆LOV | LOV domain deletion mutant | This study |
| ∆Fawc1-C∆ZnF | ZnF domain deletion mutant | This study |
2.2. Sequence Analysis of Fawc1 and Fawc2 of F. Asiaticum
The amino acid sequences of WC-1and WC-2 of Neurospora crassa were used to blast against the Fusarium graminearum genome at the Ensembl Fungi database (http://fungi.ensembl.org/index.html). The obtained target sequences were used as references to design primers to amplify the corresponding orthologs in F. asiaticum wild-type strain EXAP-08. The amplified products were subcloned to T-vector and subsequently sequenced. The obtained sequences of Fawc1 and Fawc2 were further aligned with their orthologs from other fungal species retrieved from publically available databases by DNAMAN version 5.2.2 (LynnonBiosoft Company, Pointe-Claire, QC, Canada).
2.3. Generation of Mutants and Complementation Strains in F. asiaticum
The flanks 5′ and 3′ of Fawc1 and Fawc2 were amplified from EXAP-08 genomic DNA, and the hph was amplified from plasmid p22. Overlap PCR was performed to obtain the 5′-Fawc1-hph-Fawc1-3′ gene knockout cassettes by mixing an equimolar ratio of 5′-Fawc1, hph, and Fawc1-3′ as templates, and the 5′-Fawc2-hph-Fawc2-3′ cassette was prepared in a similar way. The resulted products were separated by gel electrophoresis and then recovered by a gel extraction kit. The purified gene knockout cassettes (2 µg for each cassette) were subjected to protoplast transformation of the wild-type strain EXAP-08 according to the reported method [33]. To screen for the correct mutants of ∆Fawc1 and ∆Fawc2, the transformants grown on selective media containing 75 µg/mL hygromycin B were purified and subjected to genomic DNA extraction via the standard CTAB protocol. Site specific primers were used to carry out PCR assay to screen for the knockout mutants.
To generate the complementation strains, wild-type Fawc1 and Fawc2 connected with their 1.5 kb 5′- and 1 kb 3′-flanking sequences were amplified and subsequently cloned into the flu6 plasmid that contains the geneticin (G418) resistance gene, resulting in flu6-Fawc1-com and flu6-Fawc2-com expression vectors. Then, the complementation vectors were transformed into the protoplasts of the corresponding gene deletion mutants. CM containing 100 µg/mL of G418 was used to select the successful transformants. To generate mutant strains carrying truncated FaWC1 that lacked either LOV or ZnF domains, the Fawc1∆LOV and Fawc1∆ZnF fragments with deletion of the coding regions for LOV and ZnF domains, respectively, were generated by overlap PCR, and the resulted products were gel-purified and cloned into the flu6 plasmid, leading to the expression vectors flu6-Fawc1∆LOV and flu6-Fawc1∆ZnF, which were delivered into ∆Fawc1 mutant to generate the ∆Fawc1-C∆LOV and ∆Fawc1-C∆ZnF strains. All the primers used in the mutant generation and diagnosis PCR reactions are listed in supplementary Table S1.
2.4. Extraction of RNA and Quantitative RT-PCR Analysis
For gene expression analysis, samples were prepared as follows: aliquots of 200 μL conidia suspension (106/mL) were inoculated on solid CM medium with cellophane overlays and incubated at 23 °C. The mycelium samples were harvested after either 48 h culture in total darkness, or by ending the 48 h culture with light illumination of indicated time duration. Total RNA was extracted using QIAGEN Reagent (Germany), and 1 μg of each RNA sample was used for reverse transcription with the Prime Script™ RT reagent Kit (Perfect Real Time) (TakaRa Biotechnology, Co., Dalian, China). The real-time PCR amplifications were conducted in a CFX96TM Real-Time System (BIO-BAD, Inc., USA) using TakaRa SYBR Premix Ex Taq (TakaRa Biotechnology, Co., Dalian, China). For each sample, the expression of the β-tubulin gene was used as an internal reference. There were three replicates for each sample. The experiment was repeated three times. The gene expression levels were calculated using the 2−∆∆Ct method.
2.5. Growth and Development Phenotyping
Phenotypic differences between the mutant strains and wild type were analyzed by culturing them on CM agar medium in dark and light conditions. Colony morphology was recorded by photographs at four days after inoculation. To assess the self-fertility of each strain, mycelium plugs of 5 mm diameter were excised from the edge of 4-day-old colonies and then inoculated on carrot agar medium. The Petri dishes were put under constant light for four days when the mycelia overgrew the petri dish; then, we added 1 mL of Tween-20 in the petri dish and overwhelmed the aerial mycelia by the back of a spoon. Afterwards, the plates were transferred to near-UV light for homothallic sexual development induction. After two months, the plates were checked for perithecium formation and maturation and photo recorded via a stereomicroscope equipped with a CCD digital camera (SMC168, Moticam Pro 205B Motic, Xiamen, China). For ascospore observation, a very small brick of perithecium was picked off, pressed on a glass slide under a cover glass, and then observed using a microscope (AE30, Motic, Xiamen, China).
2.6. Carotenoid Measurement Assay
Mycelium samples grown in liquid CM medium (23 °C, 150 rpm) for three days were collected for lyophilization. For each strain, 200 mg of lyophilized mycelial biomass was added in 750 µL of hexane and 500 µL of methyl alcohol; then, it was homogenized in a Tissuelyser-24 with stainless-steel beads (Jingxin, Ltd., Shanghai, China). The resulted suspensions were centrifuged at 12,000 rpm for 10 min. The supernatant was used to determine the absorbance value (A) at 445 nm. Carotenoid contents of the samples were quantified via the equation: X(mg/100 g) = , in which “y”, “Af”, and “g” represent the volume of the extraction buffer, average absorption coefficient of the carotenoid molecular (2500), and the weight of the sample, respectively.
2.7. UV Sensitivity Assay
Five microliters of serial diluted conidial suspensions (106, 105, 104 conidia/mL) from cultures grown in liquid straw media was point-inoculated onto CM agar medium. The plates were exposed to UV-C light (provided by UVB HL-2000 Hybridizer, peaking at 245 nm, 0.2 kJ/m2) and then allowed to recover in dark or under white light for two days at 23 °C. The UV sensitivity of each strain was determined by comparing the survival of colonial cultures grown in light versus dark after UV exposure.
2.8. Virulence Assay
Fungal conidia were harvested from CMC medium and were suspended in liquid minimum Gamborg B5 medium (or GB5, Coolaber, China, containing 0.6 g·L−1 GB5 salts, 10 mM glucose, pH 7.0), and the concentration was adjusted to 1 × 106 conidia/mL. The infection assay was conducted according to the reported method [14] with modifications as follows: the wheat (Triticum aestivum cultivars Zhongyuan 98–68) coleoptiles grown for two days were cut with a scissor and inoculated with 5 µL conidial suspension, and then they were kept in a transparent box at 23 °C under a 12 h/12 h light cycle and humid condition. The infected samples were photographed every day, and the lesion areas were calculated using the ImageJ software.
2.9. Statistical Analysis
The data obtained in this study were analyzed with ANOVA followed by Duncan’s multiple range tests (p < 0.05) for means comparison with the use of SPSS 17.0.
3. Results
3.1. The Orthologs of WC-1 and WC-2 in F. asiaticum
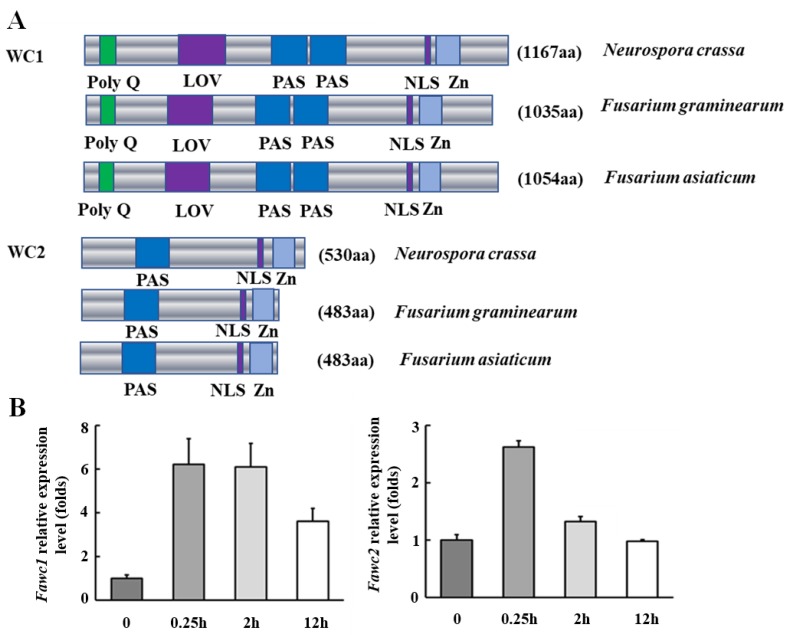
Since F. asiaticum belongs to a sub-lineage of the Fusarium graminearum species complex, the orthologous genes of wc1 and wc2 in F. asiaticum, namely Fawc1 and Fawc2, were identified as follows: BLASTP search of the F. gaminearum genome in Ensembl Fungi database, using the amino acid sequences of Neurospora crassa WC-1 (NCU02356) and WC-2 (NCU00902) proteins [34] as queries, resulted in the corresponding orthologs namely FgWC1 (FGSG_07941) and FgWC2 (FGSG_00710), respectively. The corresponding genes and their flanking sequences were used as references to design specific primers to amplify the open reading frame (ORF) sequences of their orthologs, Fawc1 and Fawc2, in F. asiaticum strain EXAP-08 via the Pfu DNA polymerase. Sequencing of the cloned PCR products indicated that Fawc1 (KX905081.1) and Fawc2 (MT019868) of F. asiaticum showed high similarity to Fgwc1 (99.05%) and Fgwc2 (99.39%) of F. graminearum. The deduced amino acid sequences of FaWC1 and FaWC2 were analyzed via the SMART online tool, and the results indicated that FaWC1 possessed a Zn-finger DNA-binding domain as well as three PAS domains, of which the N-terminal most PAS domain should be the special subclass called the LOV domain (for light, oxygen, and voltage) being responsible for binding the chromophore molecule of flavin adenine dinucleotide (FAD), while the FaWC2 just essentially contained a single PAS domain and a Zn-finger DNA binding domain. In general, FaWC1 and FaWC2 show high similarity with their orthologs in F. graminearum and N. crassa (Figure 1A). Gene expression analysis showed that both Fawc1 and Fawc2 were induced to peak levels by 15 min light exposure, while the long period (12 h) of illumination caused less induction of the transcription levels of these two genes (Figure 1B).
Figure 1.
Identification of two photoreceptor genes, Fawc1 and Fawc2, in F. asiaticum. (A). Schematic demonstration of the domains of WC1 and WC2 orthologs from N. crassa, F. graminearum, and F. asiaticum. Accessions of the amino acid sequences are as follows: WC1 (NCU02356); WC2 (NCU00902); FgWC1 (FGSG_07941); FgWC2 (FGSG_00710); FaWC1 (KX905081.1); FaWC2 (MT019868). Domains of these photoreceptor proteins were analyzed via the SMART online tool (http://smart.embl-heidelberg.de/). PAS: Per-period circadian protein; Arnt: Ah receptor nuclear translocator protein; Sim: Single-minded protein, NLS: Nuclear Location Singal, Zn: Zinc finger binding to DNA consensus sequence. (B). Transcript levels of Fawc1 and Fawc2 are regulated by light. The horizontal axis indicates light treatment time. The bars present mean values ± SD of three replicate samples.
3.2. Generation and Characterization of the ∆Fawc1 and ∆Fawc2 Mutants
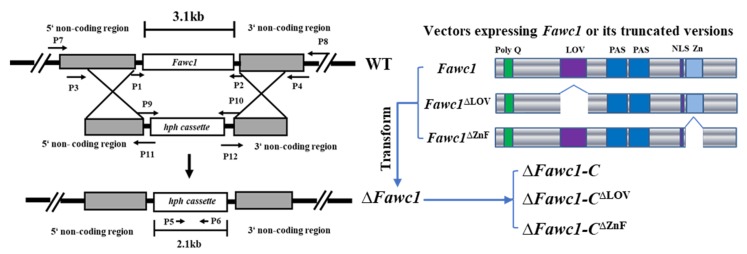
To reveal the functions of Fawc1 and Fawc2, homologous recombination cassettes used for gene knockout purposes were created as shown in Figure 2. After protoplast transformation, the transformants with hygromycin resistance successfully grew up on the selection medium. To characterize the knockout mutants of ∆Fawc1 and ∆Fawc2 in which the hygromycin resistance cassette (hph) had correctly replaced each target gene, PCR assays with the specific primer pairs were performed with the genomic DNA, and the correct transformants for each gene were selected for further analysis. To verify that the Fawc1 or Fawc2 was completely knocked out, reverse transcription (RT)-PCR was applied, revealing that the transcripts of Fawc1 and Fawc2 were present in the EXAP-08 wild-type strain but absent in the mutants of ∆Fawc1 and ∆Fawc2, respectively. To confirm the functions of these two genes, the wild-type Fawc1 and Fawc2 connected with their native promoters, and terminators were transformed into ∆Fawc1 and ∆Fawc2 to obtain the complementation strains. As FaWC1 possesses both signal input and output domains, the LOV and zinc finger (ZnF) domains, respectively, the truncated versions of FaWC1, lacking either LOV or ZnF domains (Figure 2), were expressed in the ∆Fawc1 mutant to explore the functions of these domains in light signaling and other life aspects of F. asiaticum. All the fungal strains used in this study and their genotypes are listed in Table 1.
Figure 2.
Generation of the transgenic mutants of F. asiaticum. Schematic diagrams of homologous recombination occurred between the replacement vector carrying the hygromycin resistance marker (hph) and the target gene (Fawc1) of F. asiaticum strain EXAP-08, resulting in the knockout mutant (∆Fawc1). Meanwhile, the wild-type Fawc1 or its truncated versions, Fawc1∆LOV and Fawc1∆Zn, were amplified from genomic DNA of EXAP-08 and cloned into the flu6 plasmid, and then transformed into the ∆Fawc1 mutant, resulting in the complementation strain ∆Fawc1-C, and ∆Fawc1-C∆LOV and ∆Fawc1-C∆ZnF mutant strains. The knockout mutant ∆Fawc2 and its complementation strain ∆Fawc1-C were generated via a similar strategy.
3.3. The Marker Responses to Light Signal Are Mediated by WCC and Dependent on LOV but not ZnF Domain of FaWC1 in F. asiaticum
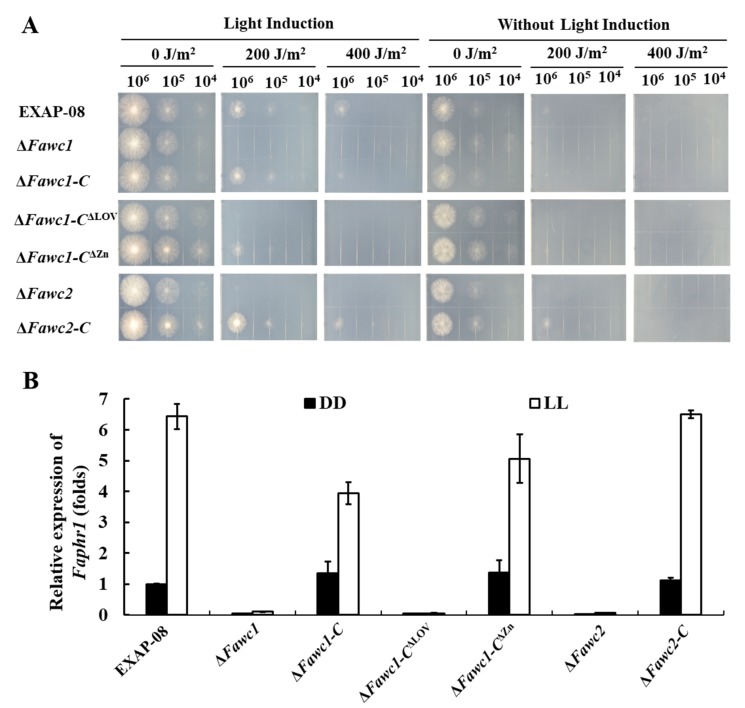
As fungi commonly sense light to indicate the presence of deleterious ultraviolet (UV) radiation, the light-induced survivability through UV damage is recognized as a marker response to light signal in fungi [35,36]. In the present study, the survival of serially diluted spores of all the tested strains was similarly poor if the fungal cultures were kept in the dark after UV-C treatment. However, when white light illumination was applied, the wild type (WT) could survive much better from UV-C irradiation than those cultures in the dark, but no recovery of survival spores was observed with ∆Fawc1 and ∆Fawc2. The complementation strains, ∆Fawc1-C and ∆Fawc2-C, demonstrated similar photoreactivation levels as the WT, indicating that the defect in the light-induced UV-C resistance in the ∆Fawc1 and ∆Fawc2 mutants is indeed caused by the loss of these two genes (Figure 3A). Interestingly, the ∆Fawc1-C∆LOV showed similar UV-C susceptibility as the ∆Fawc1 and ∆Fawc2 mutants, while in contrast, the ∆Fawc1-C∆ZnF restored UV-C tolerance to the WT level. Gene expression analysis showed that the transcript level of the photolyase gene Faphr1 was significantly induced by light in WT, ∆Fawc1-C, ∆Fawc2-C, and ∆Fawc1-C∆ZnF strains; however, no remarkable change of Faphr1 expression as influenced with light has been observed in the ∆Fawc1, ∆Fawc2, and ∆Fawc1-C∆LOV strains (Figure 3B).
Figure 3.
Effect of light on UV-C resistance. (A). Serial dilutions of all strains were point-inoculated onto complete agar medium (CM). After the UV irradiation of indicated dosages, the plates were incubated for one day in light (left) or darkness (right). (B). The relative expression level of the deduced photolyase gene Faphr1 in wild-type and mutant strains as influenced by light. DD, samples cultured for 48 h in darkness; LL, samples experienced 47 h culture in darkness followed by one hour of light illumination. The bars present mean values ± SD of three replicate samples.
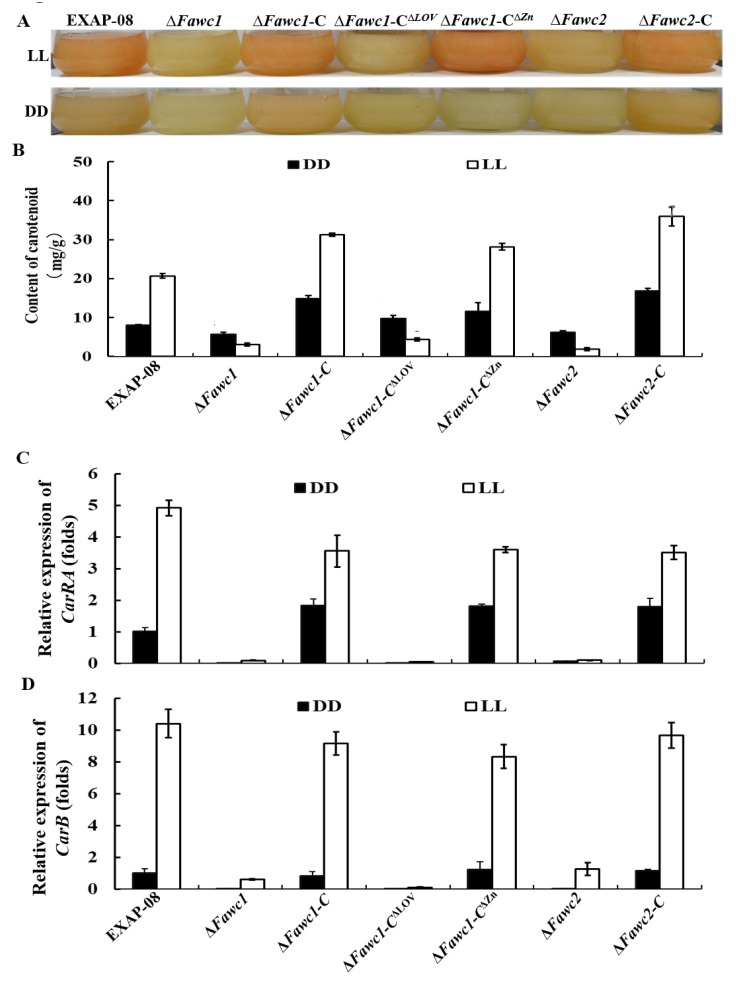
Another marker response to the light signal in fungi is pigment production [18,37]. The WT strain could produce significantly more orange-colored carotenoid pigment in constant light compared to dark condition after growth in liquid complete medium (CM) for three days (Figure 4A). In contrast, there was no observable orange pigment accumulated by ∆Fawc1 and ∆Fawc2 in light and dark conditions (Figure 4A). Similarly, the ∆Fawc1-C∆LOV mutant, which lacks the LOV domain of FaWC1, showed similar pigmentation phenotypes as the ∆Fawc1 and ∆Fawc2 mutants. However, the ∆Fawc1-C∆ZnF with truncating the ZnF domain of FaWC1 demonstrated enhanced carotenogenesis in response to light, which was similar to the WT strain. Quantitative measurement assay also showed that in darkness, all strains produced basic carotenoid levels, and light treatment caused a significant increment of carotenoid accumulation in WT, ∆Fawc1-C, ∆Fawc2-C, and ∆Fawc1-C∆ZnF strains, but light failed to alter the pigmentation behavior in ∆Fawc1, ∆Fawc2-C, and ∆Fawc1-C∆LOV strains (Figure 4B). Gene expression analysis showed that the transcript levels of the carotenoid biosynthetic genes CarRA and CarB [37,38] were up-regulated in light versus dark condition in the WT, ∆Fawc1-C, ∆Fawc2-C, and ∆Fawc1-C∆ZnF strains. Contrarily, the expression of CarRA and CarB could not be induced by light in the ∆Fawc1, ∆Fawc2, and ∆Fawc1-C∆LOV strains (Figure 4C,D).
Figure 4.
Effect of light on carotenogenesis. (A). Carotenoid pigment accumulation of the tested strains cultured in liquid CM for four days under constant light (LL) or darkness (DD). (B). Measurement of carotenoid contents in the mycelium of each strain harvested from the liquid shaking culture in (A). (C) and (D). Relative expression levels of deduced carotenoid biosynthesis genes CarRA and CarB in wild-type and mutant strains under light (LL) or darkness (DD). The bars in B, C, and D present mean values ± SD of three replicate samples.
Collectively, the above data suggest that both FaWC1 and FaWC2 are responsible for light signaling to induce the marker responses, including carotenoid accumulation and UV damage tolerance. Moreover, the LOV and ZnF domains of FaWC1 are required and dispensable, respectively, for mediating the light responses in F. asiaticum.
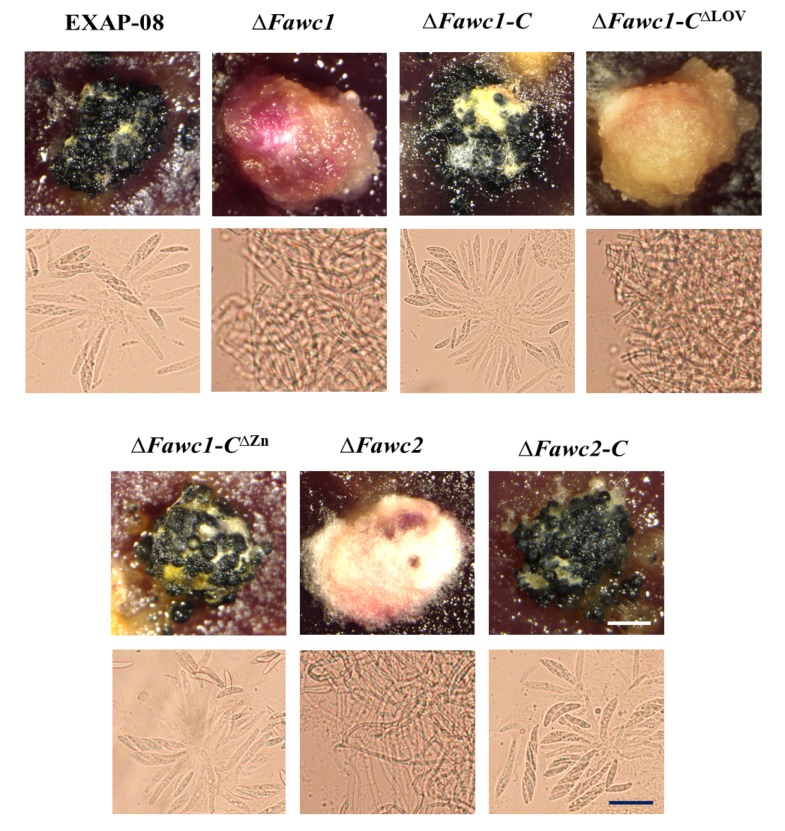
3.4. Perithecia Maturation and Ascospore Development of F. asiaticum Are Regulated by WCC Photoreceptor
Sexual reproduction is usually vital for the dissemination of fungal pathogens in their lifecycles, and the near-UV light is known to induce the perithecia maturation and ascospore formation in FGSC [39]. However, two independent studies reported inconsistent effects of the WCC photoreceptor on the sexual development of F. graminearum, which was probably due to the difference of the wild-type background strains used in each study [30,40]. The present work with F. asiaticum showed that the WT strain was able to form mature perithecia, in which the sexual ascospores could be found apparently. In contrast, the ∆Fawc1 and ∆Fawc2 mutants could produce a comparable amount of perithecia as WT, but these mutants’ perithecia failed to develop into the black-pigmented mature stage, being deficient in ascospore formation (Figure 5). Re-introducing the wild-type Fawc1 and Fawc2 into the corresponding gene deletion mutants had fully recovered their perithecia maturation and ascospore formation abilities, suggesting that these WCC photo receptor components, FaWC1 and FaWC2, are indeed required for sexual reproduction development in this fungus. Additionally, the ∆Fawc1-C∆ZnF and ∆Fawc1-C∆LOV demonstrated phenotypes in sexual development as WT and ∆Fawc1, respectively, indicating that it should be the LOV domain, but not the ZnF domain, that is required for mediating the light signal to regulate the sexual reproduction of F. asiaticum.
Figure 5.
Regulation of Fawc1 and Fawc2 on the sexual reproduction development processes of F. asiaticum. The perithecium formed by each strain on carrot agar medium was observed via stereomicroscope. Via picking up the perithecia and pressing them on glass slides for microscopic analysis, mature ascospores were observed from the perithecia of EXAP-08, ∆Fawc1-C, ∆Fawc1-C∆ZnF, and ∆Fawc2-C strains, while in contrast, only mycelium biomass could be found in the immature perithecium of ∆Fawc1, ∆Fawc2, and ∆Fawc1-C∆LOV mutant strains.
3.5. FaWC1 and FaWC2 Play Different Roles in Regulating Virulence Expression
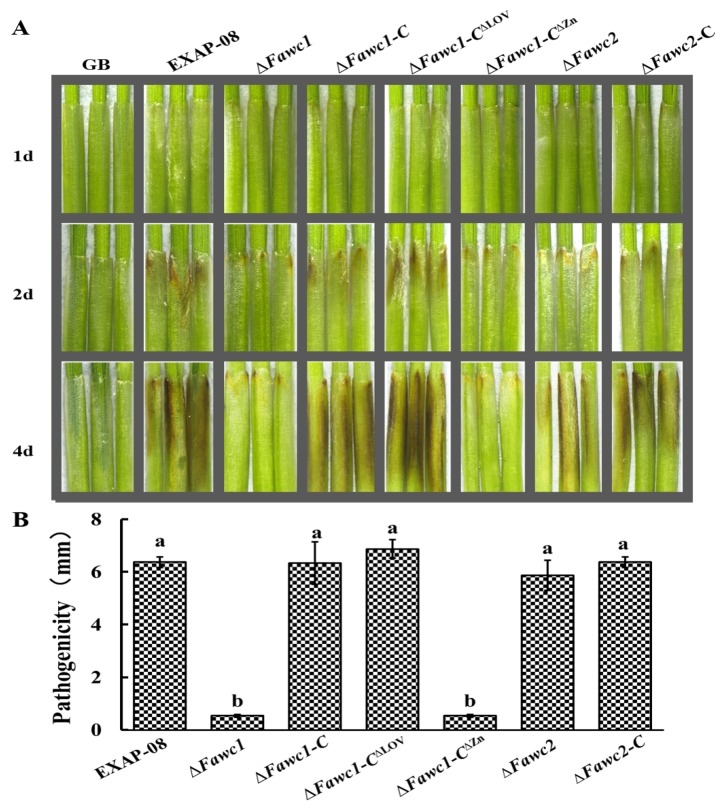
In the infection assay with wheat coleoptiles, inoculation with the WT caused apparent brown rot symptom in the host plant materials (Figure 6). In contrast, the ∆Fawc1 mutant showed more than 80% reduction in pathogenicity in comparison with WT. Meanwhile, the complementation strain ∆Fawc1-C had a recovered pathogenicity level similar to that of the WT strain, suggesting that FaWC1 is involved in regulating the pathogenicity of F. asiaticum. However, the mutant with the deletion of Fawc2 caused equivalent disease severity to the WT. These data suggested that FaWC1 and FaWC2 played independently different roles in regulating the pathogenicity of this fungus.
Figure 6.
Virulence assay on wheat confirms that Fawc1 regulate the virulence of F. asiaticum in a light-independent manner. (A). Wheat coleoptiles were inoculated with 5 µL conidial suspensions and were kept humid inside a plastic box. Fungal strains for test include the wild-type EXAP-08, ∆Fawc1, ∆Fawc1-C, ∆Fawc1-C∆LOV, ∆Fawc1-C∆ZnF, ∆Fawc2, and ∆Fawc2-C. Photographs were taken at four days post inoculation. (B). Statistical analysis of lesion sizes caused by each fungal genotype. Different letters represent a significant difference at p < 0.05. The bars present mean values ± SD (n = 20).
In order to assess whether and how light signaling via the WCC pathway could be involved in fungal pathogenicity expression, the mutant strains lacking the LOV and ZnF domains of FaWC1 were further analyzed in the host infection assay. Unexpectedly, the ∆Fawc1-C∆LOV mutant showed similar pathogenicity as the WT and complementation strains. While in contrast, the pathogenicity of the ∆Fawc1-C∆ZnF mutant was similar to the ∆Fawc1 mutant, being significantly reduced compared to WT (Figure 6). Consequently, it can be concluded that the FaWC1 LOV domain, which is required for sensing light signals, exerts no influence on pathogenicity; on the other hand, the ZnF domain of FaWC1 is involved in regulating pathogenicity, although this domain is dispensable for mediating light signals in F. asiaticum.
4. Discussion
Light is a strong environment cue orchestrating numerous biological processes in various organisms. Understanding the photobiology mechanisms in fungi can be useful in designing feasible strategies to decrease the detrimental effects of fungi while enhancing those qualities that are beneficial. Most known fungal responses to light are mediated by the well-conserved fungal blue light receptor white collar complex (WCC) [41,42]. In this paper, we functionally investigated the WCC orthologs, Fawc1 and Fawc2, in the plant pathogenic species F. asiaticum.
The FaWC1 and FaWC2 characterized in F. asiaticum strain EXAP-08 showed high similarity to their counterparts in F. graminearum sensu stricto, although they are distinct from each other in geographic, metabolic, and pathogenic phenotypes [9,16,43,44]. The FaWC1 contains a LOV domain for binding the flavin chromophore, a nuclear localization domain (NLS), a Zinc finger domain (ZnF) for DNA binding and transcription factor functions, and a PAS domain for protein–protein interaction. The FaWC2 also contains a PAS domain, NLS domain, and ZnF domain. All these protein domain characteristics of FaWC1 and FaWC2 are highly similar to WC-1 and WC-2 of N. crassa, respectively. Light generally increases cellular metabolism and at the same time causes significant oxidative stress to the organism, and to deal with this stress, protective pigments, e.g., carotenoids, are made [45]. Stimulated carotenogenesis as influenced with light treatment is a common photoresponsive phenotype among filamentous fungi [25], including several Fusarium species. Experiments in this study showed a clear dependence on WCC for light-induced carotenoid biosynthesis in F. asiaticum.
Another common reason for fungi to sense light should be to survive through the environmental stress posed by sunlight, especially the radiation of ultraviolet (UV), which can cause DNA damage or induce the accumulation of toxic reactive oxygen species (ROS) [45]. The ability to sense less harmful wavelengths of visible light can help the organisms to anticipate the emergence of, and promote tolerance to, the harmful UV. This premise has been shown among diverse fungal species through functional study with the orthologs of blue light receptor WCC [46]. It is the photoinduction of DNA repair enzymes, photolyases, and UV endonucleases, by the conserved blue light signaling pathway (WCC) that should be mainly responsible for the light-induced UV resistance in varied fungi [40,47,48,49,50]. In the present study, this reliable discipline has been confirmed in F. asiaticum in which the loss of Fawc1 or Fawc2 resulted in a deficiency of light-induced expression of photolyase gene (Faphr1) and consequently hypersensitivity to UV. Taken together, the two marker responses to light signals, including the expression of pigment synthesis and DNA repair enzymes to protect the fungal cells from the damage caused by UV during sunlight exposure, are indeed mediated cooperatively by FaWC1 and FaWC2 in F. asiaticum.
Fungal development can also be regulated by light signals. According to varied reports, light signals should have specific roles in certain species including Aspergillus nidulans [51], Cercospora zeae-maydis [27], Botrytis cinerea [29], and Alternaria alternate [52]. A key factor required in the regulation of fungal reproductive development events is the velvet complex composed of VeA, VelB, and LaeA [53], and the interactions between photoreceptors and velvet proteins have been demonstrated in A. nidulans [54]. As for the FGSC, the development of perithecia and production of ascospores are recognized to be important for the success of these pathogens in their disease cycle [55]. As found in the F. graminearum strain Z-3639, WCC should be required for the normal maturity of perithecia during sexual development [40]. However, another group later reported that WCC could negatively regulate sexual development in another strain of F. graminearum; the Z-3643 [30]. In the present study, the perithecial maturation and ascospore formation of F. asiaticum are dependent on the presence of the WCC. Consequently, the role of the conserved light receptors in regulating fungal reproductive development could be varied case by case.
Besides fungal development and metabolism, the involvement of light receptors in fungal pathogenesis is increasingly attracting research attention [20]. The orthologs of WCC have been recognized in several pathogenic species. However, functional differences have also been reported among them [27,28,29,30,40,52]. For example, in Magnaporthe oryzae, constant light suppresses disease development, which is mediated via MGWC-1 [28]. In the maize leaf pathogen C. zeae-maydis, the WC-1 ortholog Crp-1 is required for stomata tropism and for appressorium and lesion formation, implying that Crp-1 positively contributed to its virulence [27]. While in the necrotrophic pathogen B. cinerea, the WCC and its direct target BcLTF1, a light-responsive transcription factor homologous to the SUB1 in N. crassa, are required to cope with the oxidative stress that is caused by either excessive exposure to light or arising during host invasion, and thus required for achieving full virulence under excessive light [29].The present study with F. asiaticum shows that the deletion of Fawc1 resulted in decreased virulence. This is in contrast with the reports of F. graminearum, in which the WCC seems to be not involved in regulating virulence [30,40]. Even in those fungal species in which WCC are involved in determining virulence expression, the ways are quite different from each other; for example, WCC orthologs are involved in affecting the infection activities of B. cinerea, C. zeae-maydis, and M. oryzae in a light-dependent manner [27,28,29]. While in contrast, WCC are required for the full virulence of C. neoformans and F. oxysporum in mammals in a light-independent way [20,47,49]. Since the WCC proteins possess domains for both signal input (LOV domain) and output (Zn-finger domain), it occurs in several fungi that the WCC orthologs have both light-dependent and independent regulatory roles; for example, in Trichoderma reesei, the WCC photoreceptors, BLR1 and BLR2, play a role in the alteration of carbohydrate metabolic functions and the transport of compounds with distinct, both positive and negative targets in darkness [25].
To address the issue that whether the light signal and light-perceiving ability by fungi can affect pathogenicity, we have further revealed that the LOV domain of FaWC1 is required, while the ZnF domain is dispensable for inducing carotenoid synthesis and UV tolerance. These findings imply that the WCC-mediated light signaling in F. asiaticum does rely on the LOV domain for perceiving light signals, while the ZnF domain of FaWC1 seems to be not involved in regulating the downstream light responses. This phenomenon can be interpreted according to what has been found in several other fungi that indicates that the WC-2 ZnF domain is indeed responsible for the light signal output to regulate the expression of downstream genes [56]. More importantly, the ZnF domain of FaWC1 should have its downstream targets, which are not for light signaling but most likely for pathogenicity regulation. These findings have raised two open questions for future study: (1) to identify the light-independent targets of FaWC1 that are involved in pathogenicity expression; and (2) to resolve the upstream mechanisms that can orchestrate the photoreceptor to well balance between sensing light and regulating light-independent pathogenicity behaviors.
5. Conclusions
In the present study with F. asiaticum, the conserved WCC photoreceptor orthologs, namely FaWC1 and FaWC2, have been characterized. Both FaWC1 and FaWC2 are required for perceiving light signals to regulate UV resistance, secondary metabolism, and sexual reproduction. However, FaWC1 and FaWC2 performed diverged roles in virulence expression. Based on the genetics data, we conclude that the WCC orthologs in F. asiaticum are responsible for light signaling to adapt to the environmental niche; meanwhile, moonlighting functions of the individual component FaWC1 are required for regulating pathogenicity in a light-independent manner.
Acknowledgments
We thank Weihua Tang (Institute of Plant Physiology and Ecology, Chinese Academy of Sciences, Shanghai, China) for kindly providing plasmid pflu6.
Supplementary Materials
The following are available online at https://www.mdpi.com/2076-2607/8/3/365/s1, Table S1: All the primers used in the mutant generating and diagnosis PCR reactions.
Author Contributions
Conceptualization, P.Z. and L.X.; methodology, P.Z., Y.J. and Y.T.; software, Y.T.; validation, P.Z., Y.T. and L.X.; formal analysis, P.Z.; investigation, Y.T., P.Z., Z.L., Y.Q., L.H., N.Z. and Y.W.; resources, P.Z. and L.X.; data curation, Y.T. and P.Z.; writing—original draft preparation, Y.T. and P.Z.; writing—review and editing, Y.T., P.Z., H.N., Y.J. and L.X.; visualization, Y.T. and P.Z.; supervision, P.Z. and Y.J. and L.X.; project administration, P.Z. and L.X.; funding acquisition, P.Z. and L.X. All authors have read and agreed to the published version of the manuscript.
Funding
This research was financially supported by the National Key Research and Development Program of China (2016YFD0400105), National Natural Science Foundation of China (31972121), Shanghai Municipal Science and Technology Commission (18391901400), and Shanghai Municipal Agricultural and Rural Committee (2019-02-08-00-02-F01146).
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Cuomo C.A., Güldener U., Xu J.R., Trail F., Turgeon B.G., Pietro A.D., Walton J.D., Ma L.J., Baker S.E., Rep M., et al. The Fusarium graminearum genome reveals a link between localized polymorphism and pathogen specialization. Science. 2007;317:1400–1402. doi: 10.1126/science.1143708. [DOI] [PubMed] [Google Scholar]
- 2.O’Donnell K., Kistler H.C., Tacke B.K., Casper H.H. Gene genealogies reveal global phylogeographic structure and reproductive isolation among lineages of Fusarium graminearum, the fungus causing wheat scab. PNAS. 2000;97:7905–7910. doi: 10.1073/pnas.130193297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.O’Donnell K., Ward T.J., Geiser D.M., Kistler H.C., Aoki T. Genealogical concordance between the mating type locus and seven other nuclear genes supports formal recognition of nine phylogenetically distinct species within the Fusarium graminearum clade. Fungal Genet. Biol. 2004;41:600–623. doi: 10.1016/j.fgb.2004.03.003. [DOI] [PubMed] [Google Scholar]
- 4.O’Donnell K., Ward T.J., Aberra D., Kistler H.C., Aoki T., Orwig N., Kimura M., Bjørnstad Å., Klemsdal S.S. Multilocus genotyping and molecular phylogenetics resolve a novel head blight pathogen within the Fusarium graminearum species complex from Ethiopia. Fungal Genet. Biol. 2008;45:1514–1522. doi: 10.1016/j.fgb.2008.09.002. [DOI] [PubMed] [Google Scholar]
- 5.Starkey D.E., Ward T.J., Aoki T., Gale L.R., Kistler H.C., Geiser D.M., Suga H., Tóth B., Varga J., O’Donnell K. Global molecular surveillance reveals novel Fusarium head blight species and trichothecene toxin diversity. Fungal Genet. Biol. 2007;44:1191–1204. doi: 10.1016/j.fgb.2007.03.001. [DOI] [PubMed] [Google Scholar]
- 6.Yli-Mattila T., Gagkaeva T., Ward T.J., Aoki T., Kistler H.C., O’Donnell K. A novel Asian clade within the Fusarium graminearum species complex includes a newly discovered cereal head blight pathogen from the Russian Far East. Mycologia. 2009;101:841–852. doi: 10.3852/08-217. [DOI] [PubMed] [Google Scholar]
- 7.Sarver B.A.J., Ward T.J., Gale L.R., Broz K., Kistler H.C., Aoki T., Nicholson P., Carter J., O’Donnell K. Novel Fusarium head blight pathogens from Nepal and Louisiana revealed by multilocus genealogical concordance. Fungal Genet. Biol. 2011;48:1096–1107. doi: 10.1016/j.fgb.2011.09.002. [DOI] [PubMed] [Google Scholar]
- 8.Zhang H., Van der Lee T., Waalwijk C., Chen W., Xu J., Xu J., Zhang Y., Feng J. Population analysis of the Fusarium graminearum species complex from wheat in China show a shift to more aggressive isolates. PLoS ONE. 2012;7:e31722. doi: 10.1371/journal.pone.0031722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhang X., Ma H., Zhou Y., Xing J., Chen J., Yu G., Sun X., Wang L. Identification and genetic division of Fusarium graminearum and Fusarium asiaticum by species-specific SCAR markers. J. Phytopathol. 2014;162:81–88. doi: 10.1111/jph.12155. [DOI] [Google Scholar]
- 10.Zhu P., Wu L., Liu L., Huang L., Wang Y., Tang W., Wu L. Fusarium asiaticum: An Emerging Pathogen Jeopardizing Postharvest Asparagus Spears. J. Phytopathol. 2013;161:696–703. doi: 10.1111/jph.12120. [DOI] [Google Scholar]
- 11.Steiner B., Kurz H., Lemmens M., Buerstmayr H. Differential gene expression of related wheat lines with contrasting levels of head blight resistance after Fusarium graminearum inoculation. Theor. Appl. Genet. 2009;118:753–764. doi: 10.1007/s00122-008-0935-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rawat N., Pumphrey M.O., Liu S.X., Zhang X.F., Tiwari1 V.K., Ando K., Trick H.N., Bockus W.W., Akhunov E., Anderson J.A., et al. Wheat Fhb1 encodes a chimeric lectin with agglutinin domains and a pore-forming toxin-like domain conferring resistance to Fusarium head blight. Nat. Genetics. 2016;48:1576–1580. doi: 10.1038/ng.3706. [DOI] [PubMed] [Google Scholar]
- 13.Ma L.J., Van der Does H.C., Borkovich B.A., Coleman J.J., Daboussi M.J., Pietro A.D., Dufresne M., Freitag M., Grabherr M., Henrissat B., et al. Comparative genomics reveals mobile pathogenicity chromosomes in Fusarium. Nature. 2010;464:367–373. doi: 10.1038/nature08850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang X.W., Jia L.J., Zhang Y., Jiang G., Li X., Zhang D., Tang W.H. In planta stage-specific fungal gene profiling elucidates the molecular strategies of Fusarium graminearum growing inside wheat coleoptiles. Plant Cell. 2012;24:5159–5176. doi: 10.1105/tpc.112.105957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Taylor R.D., Saparno A., Blackwell B., Anoop V., Gleddie S., Tinker N.A., Harriset L.J. Proteomic analyses of Fusarium graminearum grown under mycotoxin-inducing conditions. Proteomics. 2008;8:2256–2265. doi: 10.1002/pmic.200700610. [DOI] [PubMed] [Google Scholar]
- 16.Zhang H., Zhang Z., Van der Lee T., Chen W.Q., Xu J., Xu J.S., Yang L., Yu D., Waalwijk C., Feng J. Population genetic analyses of Fusarium asiaticum populations from barley suggest a recent shift favoring 3ADON producers in southern China. Phytopathology. 2010;100:328–336. doi: 10.1094/PHYTO-100-4-0328. [DOI] [PubMed] [Google Scholar]
- 17.Bahn Y.S., Xue C., Idnurm A., Rutherford J.C., Heitman J., Cardenas M.E. Sensing the environment: Lessons from fungi. Nat. Rev. Microbiol. 2007;5:57–69. doi: 10.1038/nrmicro1578. [DOI] [PubMed] [Google Scholar]
- 18.Tisch D., Schmoll M. Light regulation of metabolic pathways in fungi. Appl. Microbiol. Biotechnol. 2010;85:1259–1277. doi: 10.1007/s00253-009-2320-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Heintzen C., Loros J.J., Dunlap J.C. The PAS protein VIVID defines a clock-associated feedback loop that represses light input, modulates gating, and regulates clock resetting. Cell. 2001;104:453–464. doi: 10.1016/S0092-8674(01)00232-X. [DOI] [PubMed] [Google Scholar]
- 20.Idnurm A., Crosson S. The photobiology of microbial pathogenesis. PLoS Pathog. 2009;5:e1000470. doi: 10.1371/journal.ppat.1000470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ballario P., Vittorioso P., Magrelli A., Talora C., Cabibbo A., Macino G. White collar-1, a central regulator of blue light responses in Neurospora, is a zinc finger protein. EMBO J. 1996;15:1650–1657. doi: 10.1002/j.1460-2075.1996.tb00510.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Harding R.W., Melles S. Genetic Analysis of Phototropism of Neurospora crassa Perithecial Beaks Using White Collar and Albino Mutants. Plant Physiol. 1983;72:996–1000. doi: 10.1104/pp.72.4.996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lauter F.R., Russo V.E. Blue light induction of conidiation-specific genes in Neurospora crassa. Nucleic Acids. Res. 1991;19:6883–6886. doi: 10.1093/nar/19.24.6883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Schmoll M., Tian C., Sun J., Tisch D., Glass N.L. Unravelling the molecular basis for light modulated cellulase gene expression - the role of photoreceptors in Neurospora crassa. BMC Genomics. 2012;13:127. doi: 10.1186/1471-2164-13-127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tisch D., Schmoll M. Targets of light signalling in Trichoderma reesei. BMC Genomics. 2013;14:657. doi: 10.1186/1471-2164-14-657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Scholthof K.B.G. The disease triangle: Pathogens, the environment and society. Nat. Rev. Microbiol. 2007;5:152–156. doi: 10.1038/nrmicro1596. [DOI] [PubMed] [Google Scholar]
- 27.Kim H., Ridenour J.B., Dunkle L.D., Bluhm B.H. Regulation of stomatal tropism and infection by light in Cercospora zeae-maydis: Evidence for coordinated host/pathogen responses to photoperiod? PLoS Pathog. 2011;7:e1002113. doi: 10.1371/journal.ppat.1002113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kim S., Singh P., Park K., Park S., Friedman A., Zheng T., Lee Y.H., Lee K. Genetic and molecular characterization of a blue light photoreceptor MGWC-1 in Magnaporth oryzae. Fungal Genet. Biol. 2011;48:400–407. doi: 10.1016/j.fgb.2011.01.004. [DOI] [PubMed] [Google Scholar]
- 29.Canessa P., Schumacher J., Hevia M.A., Tudzynski P., Larronodo L.F. Assessing the effects of light on differentiation and virulence of the plant pathogen Botrytis cinerea: Characterization of the White Collar Complex. PLoS ONE. 2013;8:e84223. doi: 10.1371/journal.pone.0084223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kim H., Kim H.K., Lee S., Yun S.H. The white collar complex is involved in sexual development of Fusarium graminearum. PLoS ONE. 2015;10:e0120293. doi: 10.1371/journal.pone.0120293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Xu Y.B., Li H.P., Zhang J.B., Song B., Chen F.F., Duan X.J., Xu H.Q., Liao Y.C. Disruption of the chitin synthase gene CHS1 from Fusarium asiaticum results in an altered structure of cell walls and reduced virulence. Fungal Genet. Biol. 2010;47:205–215. doi: 10.1016/j.fgb.2009.11.003. [DOI] [PubMed] [Google Scholar]
- 32.Leslie J.F., Summerell B.A. Fusarium laboratory workshops-A recent history. Mycotoxin Res. 2006;22:73–74. doi: 10.1007/BF02956766. [DOI] [PubMed] [Google Scholar]
- 33.Desmond O.J., Manners J.M., Stephens A.E., Maclean D.J., Schenk P.M., Gardiner D.M., Munn A.L., Kazan K. The Fusarium mycotoxin deoxynivalenol elicits hydrogen peroxide production, programmed cell death and defence responses in wheat. Mol. Plant Pathol. 2008;9:435–445. doi: 10.1111/j.1364-3703.2008.00475.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Crosthwaite S.K., Dunlap J.C., Loros J.J. Neurospora wc-1 and wc-2: Transcription, photoresponses, and the origins of circadian rhythmicity. Science. 1997;276:763–769. doi: 10.1126/science.276.5313.763. [DOI] [PubMed] [Google Scholar]
- 35.Thoma F. Light and dark in chromatin repair: Repair of UV- induced DNA lesions by photolyase and nucleotide excision repair. EMBO J. 1991;18:6585–6598. doi: 10.1093/emboj/18.23.6585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Corrochano L.M. Light in the Fungal World: From Photoreception to Gene Transcription and Beyond. Annu. Rev. Genet. 2019;53:1–22. doi: 10.1146/annurev-genet-120417-031415. [DOI] [PubMed] [Google Scholar]
- 37.Avalos J., Pardo-Medina J., Parra-Rivero O., Ruger-Herreros M., Rodríguez-Ortiz R., Hornero-Méndez D., Limón M.C. Carotenoid Biosynthesis in Fusarium. J. Fungi. 2017;3:39. doi: 10.3390/jof3030039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Prado M.M., Prado-Cabrero A., Fernández-Martín R., Avalos J. A gene of the opsin family in the carotenoid gene cluster of Fusarium fujikuroi. Curr. Genet. 2004;46:47–58. doi: 10.1007/s00294-004-0508-6. [DOI] [PubMed] [Google Scholar]
- 39.Tschanz A.T., Horst R.K., Nelson P.E. The Effect of Environment on Sexual Reproduction of Gibberella Zeae. Mycologia. 1976;68:327–340. doi: 10.1080/00275514.1976.12019914. [DOI] [Google Scholar]
- 40.Kim H., Son H., Lee Y.W. Effects of light on secondary metabolism and fungal development of Fusarium graminearum. J. Appl. Microbiol. 2014;116:380–389. doi: 10.1111/jam.12381. [DOI] [PubMed] [Google Scholar]
- 41.Chen C.H., Ringelberg C.S., Gross R.H., Dunlap J.C., Loros J.J. Genome-wide analysis of light-inducible responses reveals hierarchical light signalling in Neurospora. EMBO J. 2009;28:1029–1042. doi: 10.1038/emboj.2009.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wu C., Yang F., Smith K.M., Peterson M., Dekhang M., Zhang Y., Zucker J., Bredeweg E.L., Mallappa C., Zhou X., et al. Genome-wide characterization of light-regulated genes in Neurospora crassa. Genes. 2014;4:1731–1745. doi: 10.1534/g3.114.012617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Van der Lee T., Zhang H., van Diepeningenc A., Waalwijk C. Biogeography of Fusarium graminearum species complex and chemotypes: A review. Food Addit. Contam. 2015;32:453–460. doi: 10.1080/19440049.2014.984244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Backhouse D. Global distribution of Fusarium graminearum, F. asiaticum and F. boothii from wheat in relation to climate. Eur. J. Plant Pathol. 2014;139:161–173. doi: 10.1007/s10658-013-0374-5. [DOI] [Google Scholar]
- 45.Fuller K.K., Loros J.J., Dunlap J.C. Fungal photobiology: Visible light as a signal for stress, space and time. Curr. Genet. 2015;61:275–288. doi: 10.1007/s00294-014-0451-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yu Z., Fischer R. Light sensing and responses in fungi. Nat. Rev. Microbiol. 2019;17:25–36. doi: 10.1038/s41579-018-0109-x. [DOI] [PubMed] [Google Scholar]
- 47.Zhu P., Idnurm A. The contribution of the White Collar complex to Cryptococcus neoformans virulence is independent of its light-sensing capabilities. Fungal Genet. Biol. 2018;121:56–64. doi: 10.1016/j.fgb.2018.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Berrocal-Tito G.M., Esquivel-Naranjo E.U., Horwitz B.A., Herrera-Estrella A. Trichoderma atroviride PHR1, a fungal photolyase responsible for DNA repair, autoregulates its own photoinduction. Eukaryot Cell. 2007;6:1682–1692. doi: 10.1128/EC.00208-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ruiz-Roldán M.C., Garre V., Guarro J., Mariné M., Roncero M.I.G. Role of the white collar 1 photoreceptor in carotenogenesis, UV resistance, hydrophobicity, and virulence of Fusarium oxysporum. Eukaryot Cell. 2008;7:1227–1230. doi: 10.1128/EC.00072-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Verma S., Idnurm A. The Uve1 endonuclease is regulated by the white collar complex to protect cryptococcus neoformans from UV damage. PLoS Genet. 2013;9:e1003769. doi: 10.1371/journal.pgen.1003769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Bayram Ö., Braus G.H., Fischer R., Rodriguez-Romero J. Spotlight on Aspergillus nidulans photosensory systems. Fungal Genet. Biol. 2010;47:900–908. doi: 10.1016/j.fgb.2010.05.008. [DOI] [PubMed] [Google Scholar]
- 52.Pruβ S., Fetzner R., Seither K., Herr A., Pfeiffer E., Metzler M., Lawrence C.B., Fischer R. Role of the Alternaria alternata blue-light receptor LreA (white-collar 1) in spore formation and secondary metabolism. Appl. Environ. Microbiol. 2014;80:2582–2591. doi: 10.1128/AEM.00327-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bayram Ö., Krappmann S., Ni M., Bok J.W., Helmstaedt K., Valerius O., Braus-Stromeyer S., Kwon N.J., Keller N.P., Yu J.H., et al. VelB/VeA/LaeA Complex Coordinates Light Signal with Fungal Development and Secondary Metabolism. Science. 2008;320:1504–1506. doi: 10.1126/science.1155888. [DOI] [PubMed] [Google Scholar]
- 54.Purschwitz J., Müller S., Kastner C., Schöser M., Haas H., Espeso E.A., Atoui A., Calvo A.M., Fischer R. Functional and physical interaction of blue- and red-light sensors in Aspergillus nidulans. Curr. Biol. 2008;18:255–259. doi: 10.1016/j.cub.2008.01.061. [DOI] [PubMed] [Google Scholar]
- 55.Trail F., Xu H., Loranger R., Gadoury D. Physiological and environmental aspects of ascospore discharge in Gibberella zeae (anamorph Fusarium graminearum) Mycologia. 2002;94:181–189. doi: 10.1080/15572536.2003.11833223. [DOI] [PubMed] [Google Scholar]
- 56.Wang B., Zhou X., Loros J.J., Dunlap J.C. Alternative Use of DNA Binding Domains by the Neurospora White Collar Complex Dictates Circadian Regulation and Light Responses. Mol. Cell Biol. 2015;36:781–793. doi: 10.1128/MCB.00841-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.