Abstract
Background
The treatment of distal (below the knee) deep vein thrombosis (DVT) is not clearly established. Distal DVT can either be treated with anticoagulation, or monitored with close follow‐up to detect progression to the proximal veins (above the knee), which requires anticoagulation. Proponents of this monitoring strategy base their decision to withhold anticoagulation on the fact that progression is rare and most people can be spared from potential bleeding and other adverse effects of anticoagulation.
Objectives
To assess the effects of different treatment interventions for people with distal (below the knee) deep vein thrombosis (DVT).
Search methods
The Cochrane Vascular Information Specialist searched the Cochrane Vascular Specialised Register, CENTRAL, MEDLINE, Embase and CINAHL databases and World Health Organization International Clinical Trials Registry Platform and ClinicalTrials.gov trials registers to 12 February 2019. We also undertook reference checking to identify additional studies.
Selection criteria
Randomised controlled trials (RCTs) for the treatment of distal DVT.
Data collection and analysis
Two review authors independently selected trials and extracted data. We resolved disagreements by discussion. Primary outcomes of interest were recurrence of venous thromboembolism (VTE), DVT and major bleeding and follow up ranged from three months to two years. We performed fixed‐effect model meta‐analyses with risk ratio (RRs) and 95% confidence intervals (CIs). We assessed the certainty of the evidence using GRADE.
Main results
We identified eight RCTs reporting on 1239 participants. Five trials randomised participants to anticoagulation for up to three months versus no anticoagulation. Three trials compared anticoagulation treatment for different time periods.
Anticoagulant compared to no intervention or placebo for distal DVT treatment
Anticoagulation with a vitamin K antagonist (VKA) reduced the risk of recurrent VTE during follow‐up compared with participants receiving no anticoagulation (RR 0.34, 95% CI 0.15 to 0.77; 5 studies, 496 participants; I2 = 3%; high‐certainty evidence), and reduced the risk of recurrence of DVT (RR 0.25, 95% CI 0.10 to 0.67; 5 studies, 496 participants; I2 = 0%; high‐certainty evidence). There was no clear effect on risk of pulmonary embolism (PE) (RR 0.81, 95% CI 0.18 to 3.59; 4 studies, 480 participants; I2 = 0%; low‐certainty evidence). There was little to no difference in major bleeding with anticoagulation compared to placebo (RR 0.76, 95% CI 0.13 to 4.62; 4 studies, 480 participants; I2 = 26%; low‐certainty evidence). There was an increase in clinically relevant non‐major bleeding events in the group treated with anticoagulants (RR 3.34, 95% CI 1.07 to 10.46; 2 studies, 322 participants; I2 = 0%; high‐certainty evidence). There was one death, not related to PE or major bleeding, in the anticoagulation group.
Anticoagulation for three months or more compared to anticoagulation for six weeks for distal DVT treatment
Three RCTs of 736 participants compared three or more months of anticoagulation with six weeks of anticoagulation. Anticoagulation with a VKA for three months or more reduced the incidence of recurrent VTE to 5.8% compared with 13.9% in participants treated for six weeks (RR 0.42, 95% CI 0.26 to 0.68; 3 studies, 736 participants; I2 = 50%; high‐certainty evidence). The risk for recurrence of DVT was also reduced (RR 0.32, 95% CI 0.16 to 0.64; 2 studies, 389 participants; I2 = 48%; high‐certainty evidence), but there was probably little or no difference in PE (RR 1.05, 95% CI 0.19 to 5.88; 2 studies, 389 participants; I2 = 0%; low‐certainty evidence). There was no clear difference in major bleeding events (RR 3.42, 95% CI 0.36 to 32.35; 2 studies, 389 participants; I2 = 0%; low‐certainty evidence) or clinically relevant non‐major bleeding events (RR 1.76, 95% CI 0.90 to 3.42; 2 studies, 389 participants; I2 = 1%; low‐certainty evidence) between three months or more of treatment and six weeks of treatment. There were no reports for overall mortality or PE and major bleeding‐related deaths.
Authors' conclusions
Our review found a benefit for people with distal DVT treated with anticoagulation therapy using VKA with little or no difference in major bleeding events although there was an increase in clinically relevant non‐major bleeding when compared to no intervention or placebo. The small number of participants in this meta‐analysis and strength of evidence prompts a call for more research regarding the treatment of distal DVT. RCTs comparing different treatments and different treatment periods with placebo or compression therapy, are required.
Plain language summary
Treatment of below‐the‐knee deep vein thrombosis
Background
Venous thromboembolism (VTE) is a condition in which a blood clot forms in a vein, most commonly in the deep veins of the legs or pelvis. This is known as deep vein thrombosis, or DVT. The blood clot can dislodge and travel in the blood, particularly to the pulmonary arteries. This is known as pulmonary embolism, or PE. The term VTE includes both DVT and PE.
Distal DVT (also known as isolated distal DVT, calf DVT or below‐the‐knee DVT) occurs when the blood clot develops inside the leg veins (below the knee). The extension of the clot in proximal (above the knee) veins and the migration of a clot to the lungs (PE) are the most common complications. The best treatment of distal DVT is not clearly established. Distal DVT can either be treated with anticoagulation (medicines that help prevent blood clots), with or without additional use of compression stockings, or no medications can be given, and monitoring with repeat ultrasounds can be performed to see if the clots grow, which requires anticoagulation. The main side effect of anticoagulation medication is the increased risk of bleeding.
Study characteristics and key results
We identified eight randomised controlled trials (clinical studies where people are randomly put into one of two or more treatment groups) reporting on 1239 participants. Five of these trials randomised participants to anticoagulation for up to three months compared with no anticoagulation. Three trials compared anticoagulation treatment for different time periods.
Our review demonstrated that in participants with distal DVT compared with no anticoagulation or placebo (pretend treatment), anticoagulation reduced the risk of recurrence of VTE. There were similar results for the recurrence of DVT, while there was no clear effect on risk of PE. This benefit was seen at the expense of an increase in clinically relevant non‐major bleeding, but not major bleeding.
In a direct comparison of treatment duration, anticoagulation for three months or more was superior to a shorter course lasting up to six weeks, showing a reduced risk of recurrence of VTE and DVT with no clear difference in major bleeding and clinically relevant non‐major bleeding.
Reliability of the evidence
For the comparison anticoagulation versus no anticoagulation or placebo, the reliability of the evidence was high for recurrence of VTE, DVT, and clinically relevant non‐major bleeding, and low for PE and major bleeding. For the comparison anticoagulation for three months or more versus six weeks, the reliability of the evidence was high for recurrence of VTE and DVT; and low for PE, major bleeding and clinically relevant non‐major bleeding, The reliability of the evidence was downgraded because of variation (or imprecision) of the results due to small numbers of events.
Conclusion
Our review found a benefit for people with distal DVT treated with anticoagulation therapy with little or no clear difference in major bleeding events, although there was an increase in clinically relevant non‐major bleeding when compared with no treatment or placebo. The small number of participants in this meta‐analysis and strength of evidence suggests more research regarding the treatment of distal DVT is needed. Randomised controlled trials comparing different treatments and different treatment periods with placebo or compression therapy are required.
Summary of findings
Background
Description of the condition
Venous thromboembolism (VTE) is the obstruction of any deep vein by a blood clot or the embolism of pulmonary arteries after thrombus migration. The pathophysiology of venous thromboses are described by Virchow's Triad as hypercoagulability, haemodynamic changes (as stasis or turbulence) and endothelial injury. VTE is comprised of deep vein thrombosis (DVT) or pulmonary embolism (PE), or both, and can occur spontaneously. The incidence of VTE in mostly white populations is between 100 per 100,000 person‐years and 200 per 100,000 person‐years (Heit 2015; White 2003). Of these, it is estimated that 45 per 100,000 person‐years to 117 per 100,000 person‐years are due to DVT (without PE) and 29 per 100,000 person‐years to 78 per 100,000 person‐years are due to PE (with or without DVT) (Heit 2015). After anticoagulant discontinuation, recurrent VTE occurs in approximately 7.4% of people in one year, rising to 30.4% of people in 10 years (Cushman 2007; Heit 2015; White 2003). There are many risk factors for VTE, including periods of inactivity, dehydration, hospitalisation, trauma, surgery, clotting disorders and previous thrombosis, superficial vein thrombosis, pregnancy, oral combined hormonal contraceptives, malignancy, obesity, smoking and advancing age (Anderson 2003; NICE 2018).
DVT describes the formation of thrombus in the deep veins in the legs, and can be divided into three types:
distal DVT (also known as isolated distal DVT, calf DVT or below the knee DVT), involving the infrapopliteal venous system;
femoropopliteal DVT, involving the proximal leg veins up to the inguinal ligament; and
iliofemoral DVT involving proximal DVT that extends above the inguinal ligament.
DVT is also sometimes categorised as provoked or unprovoked, that is, the DVT is caused by a known risk factor or not. Risk factors may be transient or persistent and the presence of risk factors and the type of risk factor influence the risk of recurrent VTE.
DVT can also occur in the upper extremities (arms) but this is not the focus of this Cochrane Review and has been addressed in Feinberg 2017.
Clinical presentation of DVT of the lower limbs may be associated with localised pain, swelling and erythema, and the later occurrence of post‐thrombotic syndrome (PTS) (leg pain, tenderness, leg fatigue, persistent swelling, erythema, pigmentation or ulceration). Distal DVT is a common form of DVT but the precise incidence of distal DVT is unknown. It is estimated that it affects 0.1% of people per year (48 per 100,000) and represents about one‐third to one‐half of lower extremity DVT (Kesieme 2011). Distal DVT, particularly if left untreated, may recur or extend to the proximal veins, so increasing the risk for complications, including PE and PTS. Although rare, symptomatic PE can present as a complication of isolated distal DVT, with shortness of breath, pain on inspiration, tachycardia and right heart overload. If untreated, it can lead to circulatory collapse and death. After anticoagulation treatment there is a lower annualised incidence of VTE recurrence in people with isolated DVT compared with people with proximal DVT, but a similar incidence of PE recurrence (Galanaud 2014).
The standard treatment for VTE is anticoagulation therapy (Kearon 2016), which aims to interrupt the extension of the thrombus in proximal veins and reduce the risk for VTE recurrence (as DVT or PE). Anticoagulant drugs have the risk of bleeding as a complication of the treatment. The severity of bleeding is a reason for discontinuing the treatment and major bleeding episodes mostly require transfusion, endoscopic or angiographic intervention or surgery. Some major bleedings are fatal, and people with comorbidities are more prone to bleedings and most often interrupt the treatment (Di Nisio 2016).
Description of the intervention
Clinical guidelines provide recommendations for treatment of DVT and VTE in different settings (Kearon 2016; NICE 2012; Streiff 2016). In general, anticoagulation is the recommended treatment of choice. The recommended initial treatment is with either a direct oral anticoagulant (DOAC; with or without initial parenteral anticoagulation as indicated) or a parenteral anticoagulant in conjunction with a vitamin K antagonist (VKA). Long‐term therapy (usually anticoagulation for a minimum of three months) is indicated to treat acute VTE. Then extended‐duration anticoagulation, often referred to as 'indefinite therapy' when no anticipated stop date exists, may be used among select patients for the secondary prevention of thrombosis. In some cases of VTE associated with an increased risk for morbidity or mortality, initial thrombolysis may be appropriate (Kearon 2016; NICE 2012; Watson 2016). Evidence and guidance do not recommend using compression stockings routinely for the prevention of PTS (Kahn 2014; Kearon 2016). In people with VTE in whom anticoagulation is inappropriate, or in those who develop further pulmonary emboli despite adequate anticoagulation, temporary intravenous catheter filters can be placed to prevent further embolisation (NICE 2012).
The best treatment for people with distal DVT is not clearly established. Distal DVT can be treated with anticoagulation, for example, a form of heparin or fondaparinux transitioned to a VKA, a low molecular weight heparin (LMWH) or a DOAC. Distal DVT can also be managed expectantly with close follow‐up to detect progression to the proximal veins requiring anticoagulation. Proponents of this strategy base their decision to withhold anticoagulation on the fact that progression is rare and most people can be spared from potential bleeding and other adverse effects of anticoagulation.
To date, only a limited number of studies have reported on treatment of distal DVT using VKAs (Horner 2014; Lagerstedt 1985; Nielsen 1994; Pinede 2001), and some on LMWHs such as nadroparin (Righini 2016; Schwarz 2010). The CACTUS trial reported that anticoagulation with "nadroparin was not superior to placebo in reducing the risk of proximal extension or venous thromboembolic events in low‐risk outpatients with symptomatic calf DVT, but did increase the risk of bleeding" (Righini 2016). However, in one systematic review of randomised controlled trials (RCTs) and cohort studies, Lim 2017 concluded that in a low‐risk population, the risk for recurrence of DVT or the extension of the distal DVT in a proximal deep vein was higher when a no treatment approach was performed compared with treatment with an anticoagulant drug.
How the intervention might work
Compression may improve venous function by preventing venous stasis, which in turn may prevent progression of the distal DVT to the proximal veins.
Where no treatment is given, close observation with ultrasound is required for two weeks (Fleck 2017; Kearon 2012; Kearon 2016), so anticoagulation can be started selectively in those few patients with documented proximal progression of the thrombus.
Treatment with any anticoagulation limits the extension of the thrombus and decreases the risk for recurrence and complications. The anticoagulant effect of VKAs is achieved by decreasing the production of coagulation factors from the liver. LMWHs indirectly inhibit factors IIa and Xa; and synthetic pentasaccharides (including fondaparinux) are selective indirect factor Xa inhibitors. Among the new oral anticoagulants, dabigatran is a selective direct thrombin inhibitor, while rivaroxaban, apixaban and edoxaban are selective factor Xa inhibitors (Kesieme 2011).
Why it is important to do this review
Distal DVT represents a separate issue from proximal DVT for several reasons, including the initial clinical decision to observe or treat; and if anticoagulation is given, there is debate on the appropriate treatment length. To date, existing Cochrane Reviews on this topic have not provided subgroup analyses to deal with these issues, as included RCTs either did not involve participants with isolated distal DVTs, or did not report distal DVT separately from proximal DVT (Brandao 2017; Middeldorp 2014; Robertson 2015; Robertson 2017). Therefore, it is currently unclear which treatment for distal DVT is most effective and safe. Due to ongoing controversy, some people may be denied anticoagulation based on relatively small and possibly underpowered trials. There is a need for a standardised treatment approach for distal DVT in daily clinical practice. We aimed to identify existing RCT evidence for treatment of distal DVT and to determine the most effective and safe treatment options. Our review first determined if there is a need to anticoagulate; and second, what is the most appropriate length of anticoagulation.
Objectives
To assess the effects of different treatment interventions for people with distal (below the knee) deep vein thrombosis (DVT).
Methods
Criteria for considering studies for this review
Types of studies
We included randomised control trials (RCTs).
Types of participants
We included adults (aged 18 years or older) with distal (also known as below the knee or calf) DVT. Diagnosis of a first or recurrent DVT was made using venography or ultrasonography.
Types of interventions
We included studies which use anticoagulants (including LMWHs, DOACs, VKAs, synthetic pentasaccharides), or compression therapy for the treatment of distal DVT, of any dose or treatment duration. We planned to include the following comparisons:
any anticoagulant versus no intervention or placebo;
any anticoagulant versus any anticoagulant;
any compression therapy versus no intervention or placebo;
any anticoagulant versus any compression therapy;
compression therapy versus no compression therapy.
No intervention or placebo may have included serial ultrasound observations.
We listed all treatment groups in the Characteristics of included studies table.
Types of outcome measures
Outcomes were measured at the end of the intervention and the follow‐up period, as reported by the individual studies, usually between three and six months from recruitment. We include long‐term results (one to two years), where available.
Primary outcomes
Recurrence of VTE: defined as DVT recurrence in the calf veins, or progression of DVT to proximal veins (e.g. popliteal or femoral vein) or PE, provided that these were objectively diagnosed with venography or ultrasonography for DVT and pulmonary angiography, computed tomography or V/Q scan for PE. We considered any episode of DVT after the first episode, including propagation of distal DVT, as recurrence.
Major bleeding: defined as clinically overt and associated with a fall in haemoglobin level of 20 g/L or more; transfusion of two or more units of red cells; retroperitoneal, intracranial, occurred in a critical site, or contributed to death.
Secondary outcomes
Recurrence of DVT, which included DVT recurrence in the calf veins, progression of DVT to proximal veins (e.g. popliteal or femoral vein); objectively diagnosed with venography or ultrasonography. We considered any episode of DVT after the first episode, including propagation of distal DVT, as recurrence.
PE: objectively diagnosed with pulmonary angiography, computed tomography or V/Q scan.
Clinically relevant non‐major bleeding, defined as overt bleeding not meeting the criteria for major bleeding but associated with medical intervention, unscheduled contact with a physician, interruption or discontinuation of study treatment, or associated with any other discomfort such as pain or impairment of activities of daily life.
Overall mortality.
Mortality related to PE or major bleeding.
Post‐thrombotic syndrome (PTS).
Resolution of symptoms.
Search methods for identification of studies
Electronic searches
The Cochrane Vascular Information Specialist conducted systematic searches of the following databases for RCTs and controlled clinical trials without language, publication year or publication status restrictions:
the Cochrane Vascular Specialised Register via the Cochrane Register of Studies (CRS‐Web searched from inception to 12 February 2019);
the Cochrane Central Register of Controlled Trials (CENTRAL) Cochrane Register of Studies Online (CRSO 2019, Issue 1);
MEDLINE (Ovid MEDLINE Epub Ahead of Print, In‐Process & Other Non‐Indexed Citations, Ovid MEDLINE Daily and Ovid MEDLINE) (1946 onwards) (searched from 1 January 2017 to 12 February 2019);
Embase Ovid (from 1974 onwards) (searched from 1 January 2017 to 12 February 2019);
CINAHL EBSCO (from 1982 onwards) (searched from 1 January 2017 to 12 February 2019).
The Information Specialist modelled search strategies for other databases on the search strategy designed for MEDLINE. Where appropriate, they were combined with adaptations of the highly sensitive search strategy designed by Cochrane for identifying RCTs and controlled clinical trials (as described in the Cochrane Handbook for Systematic Reviews of Interventions Chapter 6, Lefebvre 2011). Search strategies for major databases are provided in Appendix 1.
The Information Specialist searched the following trials registries on 12 February 2019:
the World Health Organization International Clinical Trials Registry Platform (who.int/trialsearch);
ClinicalTrials.gov (clinicaltrials.gov).
Searching other resources
We searched the reference lists of relevant articles to identify additional trials.
Data collection and analysis
Selection of studies
Two review authors (GIK, SS) independently screened titles and abstracts of retrieved studies for relevance. We retrieved full‐text articles of all potentially eligible studies. Two review authors (GIK, SS) independently reviewed each full‐text article for eligibility. Any discrepancy was resolved by discussion or by consulting a third review author (SKK). We illustrated the study selection process in a PRISMA diagram (Liberati 2009). We listed all articles excluded after full‐text assessment in the Characteristics of excluded studies table and provided the reasons for their exclusion. Where studies had multiple publications, we collated the reports of the same study so that each study, rather than each report, was the unit of interest for the review, and such studies had a single identifier with multiple references.
Data extraction and management
Two review authors (GIK, SS) independently extracted relevant data on primary and secondary outcomes and transfer the information to data collection sheets. We resolve discrepancies by consulting with a third review author (SKK) and transferred additional general information on each included study, including numbers, gender, mean age of participants, and types of interventions, to the Characteristics of included studies table.
Assessment of risk of bias in included studies
Two review authors (GIK, SS) independently assessed each included study for risk of bias according to the following criteria: random sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias) and other bias, as recommended by the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). We judged all included studies as having low, high or unclear risk of bias based on these criteria. We resolved disagreements by discussion with a third review author (SKK).
Measures of treatment effect
We used Review Manager 5 to calculate the measures of treatment effect (Review Manager 2014). We assessed dichotomous data by calculating risk ratios (RRs) with 95% confidence intervals (CIs). We assessed continuous data by calculating the mean difference (MD) and standard deviation (SD) with corresponding 95% CI. We used standardised mean differences (SMD) with 95% CIs to combine outcomes from trials that used different scales.
Unit of analysis issues
Each individual participant was the unit of analysis.
Dealing with missing data
We recorded missing and unclear data. If possible, we performed all analyses using an intention‐to‐treat approach, that is, we analysed all participants and their outcomes within the groups to which they were allocated, regardless of whether they received the intervention or were assessed for the outcome. If necessary, we contacted study authors to request missing data.
Assessment of heterogeneity
We assessed interstudy heterogeneity using a forest plot. We calculated Chi2 and I2 statistics to measure the amount of heterogeneity. I2 values less than 50% indicate low heterogeneity, I2 values between 50% and 75% indicate moderate heterogeneity, and I2 values greater than 75% indicate significant heterogeneity (Higgins 2011). We performed subgroup analyses to explore sources of heterogeneity.
Assessment of reporting biases
We planned to assess reporting bias by using funnel plots, when a meta‐analysis included more than 10 studies (Higgins 2011).
Data synthesis
We used a fixed‐effect model for meta‐analysis when we observed no or low heterogeneity. In cases of moderate/significant heterogeneity (I2 greater than 50%), we planned to use a random‐effects model. If we had identified substantial clinical, methodological or statistical heterogeneity across included trials, we planned not to report pooled results from the meta‐analysis but instead use an alternative approach to data synthesis (McKenzie 2019).
Subgroup analysis and investigation of heterogeneity
Where possible and when sufficient data were available, we performed subgroup analysis by examining:
different types of anticoagulants;
different types of compression;
treatment length (short‐term about six weeks, or long‐term, about three to six months) as suggested by previous research and the American College of Chest Physicians and the International Union of Angiology guidelines (Kearon 2016; Nicolaides 2013);
dose of anticoagulant;
provoked or unprovoked DVT;
number of thrombosed calf veins (one versus multiple).
Sensitivity analysis
We planned to carry out sensitivity analysis by excluding RCTs with a high risk for bias in any domain displayed in the 'Risk of bias' graph and repeat the analyses.
Summary of findings and assessment of certainty of the evidence
We prepared 'Summary of findings' tables using the GRADEpro Guideline Development Tool (www.gradepro.org), to present the main findings of the review (Atkins 2004). The population consisted of people with distal DVT. We included the following outcomes that are considered essential for decision‐making in our 'Summary of findings' tables:
recurrence of VTE;
major bleeding;
recurrence of DVT;
PE;
clinically relevant non‐major bleeding;
overall mortality;
mortality related to PE.
We create one 'Summary of findings' table for each comparison considered to be of clinical relevance. We evaluated the certainty of the evidence using the GRADE approach (GRADEpro GDT 2015). We assigned one of four levels of quality: high, moderate, low or very low, based on overall risk of bias, directness of the evidence, inconsistency of results, precision of the estimates and risk of publication bias as previously described (Higgins 2011). We explained the downgrading of the evidence in the footnotes. We did not stratify into risk groups or use external information for the assumed risk of the comparison group.
Results
Description of studies
Results of the search
See Figure 1.
1.
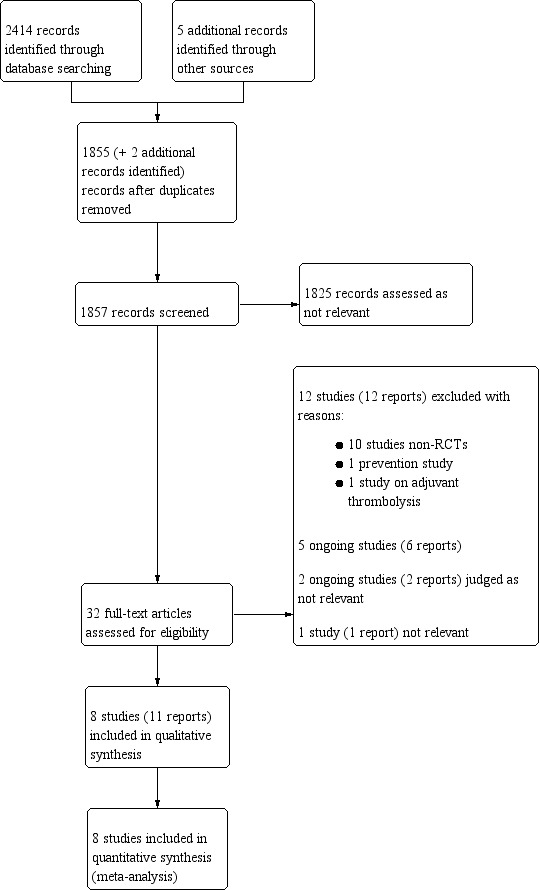
Study flow diagram.
We identified eight studies (11 records) which met the inclusion criteria (Ferrara 2006; Horner 2014; Lagerstedt 1985; Nielsen 1994; Pinede 2001; Righini 2016; Schulman 1995; Schwarz 2010). We excluded 15 studies (15 records) (Dentali 2017; Donadini 2017; Galanaud 2017; Ho 2016; Leizorovicz 2003; McBane 2018; Musil 2000; NCT00816920; NCT01234064; NCT01252420; NCT02746185; Pegoraro 2016; Schulman 1986; Schwarz 2001; Utter 2016). In addition, we identified five ongoing studies (six records) (EUCTR2005‐004235‐21‐IT; NCT02722447; NCT03368313; NCT03590743; UMIN000028105).
Included studies
We included eight studies (11 records) that met the inclusion criteria, reporting outcomes on 1239 participants (Ferrara 2006; Horner 2014; Lagerstedt 1985; Nielsen 1994; Pinede 2001; Righini 2016; Schulman 1995; Schwarz 2010). Three studies compared VKA treatment for six weeks versus 12 weeks or more (Ferrara 2006; Pinede 2001; Schulman 1995). From the remaining five studies, three studies compared anticoagulants versus no intervention or placebo for three months (Horner 2014; Lagerstedt 1985; Nielsen 1994), and two studies compared anticoagulants versus placebo for up to six weeks (Righini 2016; Schwarz 2010). One trial that enrolled postoperative patients studied provoked DVT (Ferrara 2006), and two trials that enrolled outpatients with a first episode of isolated calf DVT studied unprovoked DVT (Horner 2014; Righini 2016). The remaining five trials enrolled populations with provoked and unprovoked DVT without providing details for the subgroups (Lagerstedt 1985; Nielsen 1994; Pinede 2001; Schulman 1995; Schwarz 2010). See Characteristics of included studies table for further details. Two included studies had more than one reference (Horner 2014; Righini 2016).
Excluded studies
We excluded 15 studies (15 reports) due to: retrospective study design (Dentali 2017; Donadini 2017; Ho 2016; Utter 2016), prospective single‐arm study design (McBane 2018), observational nature of the studies (Galanaud 2017; NCT00816920; NCT01252420), cohort studies (Pegoraro 2016; Schwarz 2001), prevention study (Leizorovicz 2003), testing of adjuvant thrombolysis (Schulman 1986), ongoing prevention study (NCT01234064), ongoing study for VTE in cancer (NCT02746185), and irrelevant (Musil 2000). See Characteristics of excluded studies table for further details.
Ongoing studies
We identified five ongoing studies (EUCTR2005‐004235‐21‐IT; NCT02722447; NCT03368313; NCT03590743; UMIN000028105). See Characteristics of ongoing studies table for details of the ongoing studies.
Risk of bias in included studies
See Figure 2; Figure 3. With the exception of performance bias, risk of bias domains were mostly low risk.
2.

Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
3.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Allocation
The randomisation method was unclear in two included RCTs (Ferrara 2006; Nielsen 1994).
We identified an unclear risk for allocation concealment in two included RCTs (Ferrara 2006; Schwarz 2010).
Blinding
There was a high risk of performance bias in seven studies because of their open‐label randomisation (Ferrara 2006; Horner 2014; Lagerstedt 1985; Nielsen 1994; Pinede 2001; Schulman 1995; Schwarz 2010). Only one study was double‐blind and judged at low risk of performance bias (Righini 2016).
One study reported outcome assessors were blinded (Righini 2016). The remaining studies did not blind outcome assessors, but we judged that the outcome measures reported by the studies this review (recurrent VTE, major bleeding, recurrent DVT, PE, clinically relevant non‐major bleeding, overall mortality and mortality related to PE and major bleeding) were not likely to be influenced by a lack of blinding of outcome assessors (Ferrara 2006; Horner 2014; Lagerstedt 1985; Nielsen 1994; Pinede 2001; Schulman 1995; Schwarz 2010).
Incomplete outcome data
There was no incomplete outcome data reporting.
Selective reporting
We identified no findings of selective reporting.
Other potential sources of bias
There was no other potential source of bias.
Effects of interventions
Summary of findings for the main comparison. Anticoagulant compared to no intervention or placebo for distal deep vein thrombosis treatment.
| Anticoagulant compared to no intervention or placebo for distal DVT treatment | ||||||
|
Patient or population: people with distal DVT Setting: hospital Intervention: anticoagulant Comparison: no intervention or placebo | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with no intervention or placebo | Risk with anticoagulant | |||||
|
Recurrence of VTE (follow‐up: 3 months) |
Study population | RR 0.34 (0.15 to 0.77) | 496 (5 studies) | ⊕⊕⊕⊕ Higha,b |
— | |
| 91 per 1000 | 31 per 1000 (14 to 70) | |||||
| Major bleeding (follow‐up: 3 months) | Study population | RR 0.76 (0.13 to 4.62) | 480 (4 studies) | ⊕⊕⊝⊝ Lowc |
— | |
| 8 per 1000 | 6 per 1000 (1 to 38) |
|||||
|
Recurrence of DVT (follow‐up: 3 months) |
Study population | RR 0.25 (0.10 to 0.67) | 496 (5 studies) | ⊕⊕⊕⊕ Higha,b |
— | |
| 79 per 1000 | 20 per 1000 (8 to 53) | |||||
|
PE (follow‐up: 3 months) |
Study population | RR 0.81 (0.18 to 3.59) | 480 (4 studies) | ⊕⊕⊝⊝ Lowc |
— | |
| 12 per 1000 | 10 per 1000 (2 to 44) | |||||
|
Clinically relevant non‐major bleeding (follow‐up: 3 months) |
Study population | RR 3.34 (1.07 to 10.46) | 322 (2 studies) | ⊕⊕⊕⊕ Higha,b |
— | |
| 18 per 1000 | 61 per 1000 (19 to 190) | |||||
| Overall mortality (follow‐up: 3 months) | Study population |
RR 3.2 (0.13 to 77.69) |
430 (3 studies) |
⊕⊕⊝⊝ Lowc |
1 death reported in the anticoagulant group. 0 deaths reported in the no intervention or placebo group. | |
| 0 per 1000 | 0 per 1000 (0 to 0) |
|||||
| Mortality related to PE (follow‐up: 3 months) | See comment | Not estimable | 430 (3 studies) |
See comment | 0 PE‐related deaths reported. | |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; DVT: deep vein thrombosis; PE: pulmonary embolism; RR: risk ratio; VTE: venous thromboembolism. | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
aDowngraded one level as total number of events was fewer than 300. bUpgraded one level due to large treatment effect. cDowngraded two levels for imprecision due to very few events and 95% CIs include both appreciable benefit and appreciable harm.
Summary of findings 2. Anticoagulation for three months or more compared to anticoagulation for six weeks for distal deep vein thrombosis treatment.
| Anticoagulation for 3 months or more compared to anticoagulation for 6 weeks for distal DVT treatment | ||||||
|
Patient or population: people with distal DVT Setting: hospital Intervention: VKA for ≥ 3 months Comparison: VKA for 6 weeks | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with anticoagulation for 6 weeks | Risk with anticoagulation for ≥ 3 months | |||||
|
Recurrence of VTE (follow‐up: 16 weeks to 24 months) |
Study population | RR 0.42 (0.26 to 0.68) | 736 (3 studies) | ⊕⊕⊕⊕ Higha,b |
— | |
| 135 per 1000 | 57 per 1000 (35 to 92) | |||||
|
Major bleeding (follow‐up: 16 weeks to 15 months) |
Study population | RR 3.42 (0.36 to 32.35) | 389 (2 studies) | ⊕⊕⊝⊝ Lowc |
— | |
| 5 per 1000 | 17 per 1000 (2 to 161) | |||||
|
Recurrence of DVT (follow‐up: 16 weeks to 15 months) |
Study population | RR 0.32 (0.16 to 0.64) | 389 (2 studies) | ⊕⊕⊕⊕ Higha,b |
— | |
| 144 per 1000 | 46 per 1000 (23 to 92) | |||||
|
PE (follow‐up: 16 weeks to 15 months) |
Study population | RR 1.05 (0.19 to 5.88) | 389 (2 studies) | ⊕⊕⊝⊝ Lowc |
— | |
| 10 per 1000 | 10 per 1000 (2 to 59) | |||||
|
Clinically relevant non‐major bleeding (follow‐up: 16 weeks to 15 months) |
Study population | RR 1.76 (0.90 to 3.42) | 389 (2 studies) | ⊕⊕⊝⊝ Lowc |
— | |
| 60 per 1000 | 105 per 1000 (54 to 204) | |||||
| Overall mortality | See comment | See comment | — | See comment | The included studies did not report on overall mortality. | |
|
Mortality related to PE (follow‐up: 15 months) |
See comment | Not estimable | 197 (1 study) |
See comment | 1 included study reported there were no cases of death from PE. | |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; DVT: deep vein thrombosis; PE: pulmonary embolism; RR: risk ratio; VTE: venous thromboembolism. | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
aDowngraded one level as total number of events was fewer than 300. bUpgraded one level due to large treatment effect. cDowngraded two levels for imprecision due to very few events and 95% CIs include both appreciable benefit and appreciable harm.
Five RCTs compared anticoagulation therapy versus placebo (Horner 2014; Lagerstedt 1985; Nielsen 1994; Righini 2016; Schwarz 2010). Three RCTs compared treatment with VKAs for six weeks versus three months or more (Schulman 1995; Ferrara 2006; Pinede 2001).
We found no relevant studies for compression therapy versus no intervention or placebo, anticoagulation versus compression therapy and compression therapy versus no compression therapy.
Any anticoagulant versus no intervention or placebo
Five RCTs compared anticoagulation therapy versus no intervention or placebo (Horner 2014; Lagerstedt 1985; Nielsen 1994; Righini 2016; Schwarz 2010). The studies used different forms of anticoagulation therapy: unfractionated heparin (Lagerstedt 1985; Nielsen 1994), nadroparin (Righini 2016; Schwarz 2010), and dalteparin (Horner 2014).
Primary outcomes
Recurrence of venous thromboembolism
Five RCTs compared the effect of anticoagulation therapy versus no intervention or placebo, for the rates of recurrence of VTE: two RCTs for up to six weeks (RR 0.63, 95% CI 0.23 to 1.69; 2 studies, 359 participants; I2 = 0%) (Righini 2016; Schwarz 2010), and the remaining three RCTs for three months (RR 0.13, 95% CI 0.02 to 0.65; 3 studies, 137 participants; I2 = 0%) (Horner 2014; Lagerstedt 1985; Nielsen 1994). The overall recurrence of VTE was lower in the anticoagulation group compared with the no intervention or placebo group (RR 0.34, 95% CI 0.15 to 0.77; 5 studies, 496 participants; I2 = 3%; high‐certainty evidence). The test for subgroup differences showed no clear difference in recurrence of VTE between the subgroups anticoagulation for three months and anticoagulation for up to six weeks (P = 0.10) (Analysis 1.1).
1.1. Analysis.
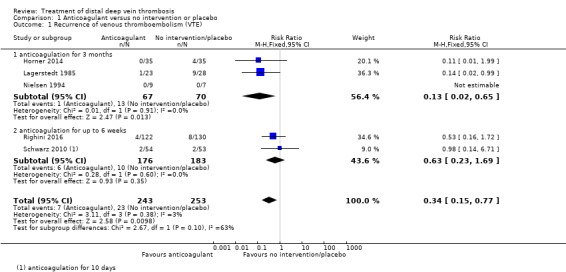
Comparison 1 Anticoagulant versus no intervention or placebo, Outcome 1 Recurrence of venous thromboembolism (VTE).
Major bleeding
The meta‐analysis showed there was little to no difference in major bleeding events between anticoagulation therapy and no intervention or placebo (RR 0.76, 95% CI 0.13 to 4.62; 4 studies, 480 participants; I2 = 26%; low‐certainty evidence; Analysis 1.2).
1.2. Analysis.

Comparison 1 Anticoagulant versus no intervention or placebo, Outcome 2 Major bleeding.
Secondary outcomes
Recurrence of deep vein thrombosis
Results for recurrence of DVT showed a similar pattern to the results for recurrence of VTE. The overall recurrence of DVT was lower in the anticoagulation group compared with the no intervention or placebo group (RR 0.25, 95% CI 0.10 to 0.67; 5 studies, 496 participants; I2 = 0%; high‐certainty evidence). The risk of recurrence of DVT in the subgroup of three months of anticoagulation was RR 0.09 (95% CI 0.01 to 0.71; 3 studies, 137 participants; I2 = 0%) and the subgroup of up to six weeks of anticoagulation was RR 0.46 (95% CI 0.14 to 1.48; 2 studies, 359 participants; I2 = 0%). The test for subgroup differences showed no clear difference in recurrence of DVT between the subgroups anticoagulation for three months and anticoagulation for up to six weeks (P = 0.18) (Analysis 1.3)
1.3. Analysis.
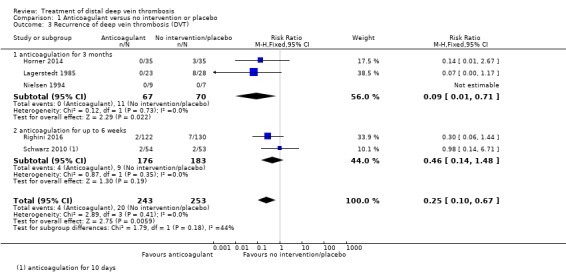
Comparison 1 Anticoagulant versus no intervention or placebo, Outcome 3 Recurrence of deep vein thrombosis (DVT).
Pulmonary embolism
We identified no clear difference in PE between the anticoagulation group compared with the no intervention or placebo group (RR 0.81, 95% CI 0.18 to 3.59; 4 studies, 480 participants; I2 = 0%; low‐certainty evidence). The risk of PE in the subgroup of three months of anticoagulation was RR 0.37 (95% CI 0.04 to 3.42; 2 studies, 121 participants; I2 = 0%) and in the subgroup up to six weeks of anticoagulation was RR 2.13 (95% CI 0.20 to 23.21; 2 studies, 359 participants; I2 = 0%) (Analysis 1.4).
1.4. Analysis.
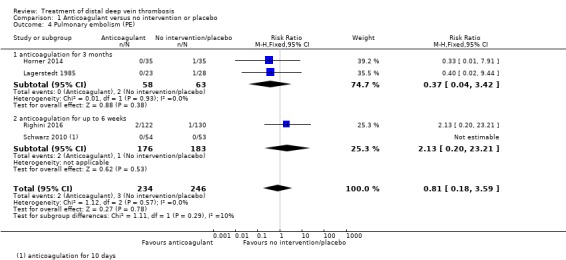
Comparison 1 Anticoagulant versus no intervention or placebo, Outcome 4 Pulmonary embolism (PE).
Clinically relevant non‐major bleeding
Clinically relevant non‐major bleeding events were increased in the anticoagulation group compared with the no intervention or placebo group (RR 3.34, 95% CI 1.07 to 10.46; 2 studies, 322 participants; I2 = 0%; high‐certainty evidence) (Analysis 1.5).
1.5. Analysis.
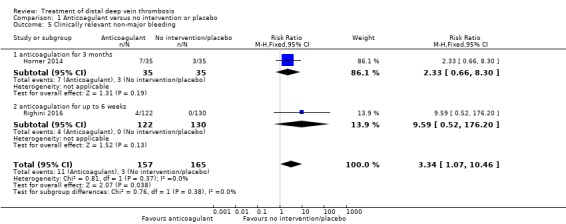
Comparison 1 Anticoagulant versus no intervention or placebo, Outcome 5 Clinically relevant non‐major bleeding.
Overall mortality
Two studies reported no deaths in each of the study arms (Horner 2014; Schwarz 2010), and one study reported one death from cancer in the nadroparin group (Righini 2016) (RR 3.20, 95% CI 0.13 to 77.69; 3 studies, 430 participants; I2 = 0%; low‐certainty evidence). The remaining two studies did not report on mortality (Lagerstedt 1985; Nielsen 1994) (Analysis 1.6).
1.6. Analysis.

Comparison 1 Anticoagulant versus no intervention or placebo, Outcome 6 Overall mortality.
Mortality related to pulmonary embolism or major bleeding
There were no cases of mortality related to PE or major bleeding in three studies (Horner 2014; Righini 2016; Schwarz 2010). The remaining two studies did not report on mortality related to PE or major bleeding (Lagerstedt 1985; Nielsen 1994).
Post‐thrombotic syndrome
There were no reports of PTS in four studies included in the quantitative synthesis (Horner 2014; Lagerstedt 1985; Nielsen 1994; Schwarz 2010). An additional publication of an included study (Righini 2016) reported on PTS (Galanaud 2018). A minimum of one year after randomisation, participants were assessed for PTS. Galanaud 2018 reported PTS was present in 54/178 (29%) of participants and was moderate or severe in 13/54 (24%) of cases. There was no clear difference in rates of PTS in the nadroparin compared with the placebo group (RR 0.90, 95% CI 0.58 to 1.41; 1 study, 178 participants).
Resolution of symptoms
There were no reports for resolution of symptoms in the studies included in the quantitative synthesis (Horner 2014; Lagerstedt 1985; Nielsen 1994; Righini 2016; Schwarz 2010), but one additional publication of an included study (Righini 2016) did report on pain (Righini 2019). Righini 2019 reported no clear difference in the mean visual analogue pain scale (VAS) reduction between 106 participants treated with therapeutic nadroparin and 109 treated with placebo (after 1 week: 2.6 (SD 2.4) with nadroparin versus 2.3 (SD 2.0) with placebo; after 6 weeks: 4.4 (SD 2.8) with nadroparin versus 4.0 (SD 2.4) with placebo). The use of compression stockings was associated with a reduction in pain.
Any anticoagulation versus any anticoagulation: three months or more of vitamin K antagonist versus six weeks of vitamin K antagonist
Three RCTs compared treatment with VKAs for three months or more versus six weeks (Ferrara 2006; Pinede 2001; Schulman 1995). Stratified data for Schulman 1995 were only available for recurrent VTE.
Primary outcomes
Recurrence of venous thromboembolism
The incidence of recurrence of VTE was lower for the three months or more treatment period compared with the six weeks' treatment period (5.8% with 3 months or more versus 13.9% with 2 weeks; RR 0.42, 95% CI 0.26 to 0.68; 3 studies, 736 participants; I2 = 50%; high‐certainty evidence) (Analysis 2.1).
2.1. Analysis.

Comparison 2 Anticoagulant versus any anticoagulant: three months or more of vitamin K antagonist (VKA) versus six weeks of VKA, Outcome 1 Recurrence of venous thromboembolism (VTE).
Major bleeding
There was no clear difference in major bleeding events between the three months or more and six weeks' treatment period (RR 3.42, 95% CI 0.36 to 32.35; 2 studies, 389 participants; I2 = 0%; low‐certainty evidence) (Analysis 2.2).
2.2. Analysis.

Comparison 2 Anticoagulant versus any anticoagulant: three months or more of vitamin K antagonist (VKA) versus six weeks of VKA, Outcome 2 Major bleeding.
Secondary outcomes
Recurrence of deep vein thrombosis
The risk for recurrence of DVT was reduced for the three months or more treatment period compared with the six weeks' treatment period (RR 0.32, 95% CI 0.16 to 0.64; 2 studies, 389 participants; I2 = 48%; high‐certainty evidence) (Analysis 2.3).
2.3. Analysis.

Comparison 2 Anticoagulant versus any anticoagulant: three months or more of vitamin K antagonist (VKA) versus six weeks of VKA, Outcome 3 Recurrence of deep vein thrombosis (DVT).
Pulmonary embolism
There was probably little or no difference in recurrent PE between three months or more and six weeks' treatment period (RR 1.05, 95% CI 0.19 to 5.88; 2 studies, 389 participants; I2 = 0%; low‐certainty evidence) (Analysis 2.4).
2.4. Analysis.
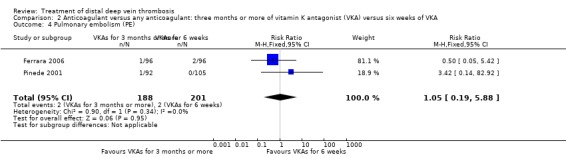
Comparison 2 Anticoagulant versus any anticoagulant: three months or more of vitamin K antagonist (VKA) versus six weeks of VKA, Outcome 4 Pulmonary embolism (PE).
Clinically relevant non‐major bleeding
There was no clear difference in clinically relevant non‐major bleeding events between the three months or more and the six weeks' treatment period (RR 1.76, 95% CI 0.90 to 3.42; 2 studies, 389 participants; I2 = 1%; low‐certainty evidence) (Analysis 2.5).
2.5. Analysis.

Comparison 2 Anticoagulant versus any anticoagulant: three months or more of vitamin K antagonist (VKA) versus six weeks of VKA, Outcome 5 Clinically relevant non‐major bleeding.
Overall mortality
There were no reports of overall mortality in included studies for people with distal DVT (Ferrara 2006; Pinede 2001; Schulman 1995).
Mortality related to pulmonary embolism or major bleeding
Two included studies reported no deaths related to major bleeding (Ferrara 2006; Pinede 2001). One study reported no cases of mortality related to PE (Pinede 2001).
Post‐thrombotic syndrome
There were no reports of PTS (Ferrara 2006; Pinede 2001; Schulman 1995).
Resolution of symptoms
There were no reports of resolution of symptoms (Ferrara 2006; Pinede 2001; Schulman 1995).
Discussion
Summary of main results
Our review has demonstrated that in people with distal DVT compared with no anticoagulation or placebo, anticoagulation reduces the risk of VTE recurrence. The results showed a clear effect for a three‐month course of anticoagulation, but not for a treatment lasting up to six weeks, likely as a result of a type II error due to the small number of events, or it may represent a true difference between a shorter or longer course of therapy. There were similar results for the outcome measure of recurrence of DVT, while there was no clear effect on risk of PE, again, likely as a result of a type II error. This benefit was obtained at the expense of clinically relevant non‐major bleeding, but not major bleeding. In a direct comparison of treatment duration, anticoagulation for three months or more was superior to a shorter course lasting up to six weeks, in terms of reduced risk for recurrence of VTE and DVT with no clear difference in major bleeding and clinically relevant non‐major bleeding.
Overall completeness and applicability of evidence
The number of VTE events, as clearly shown in the corresponding forest plots, is relatively small and this may prompt further trials. However, the magnitude and direction of the effect is consistent in the subgroup of Analysis 1.1, but also in Analysis 2.1, suggesting that three months of anticoagulation may be more beneficial than a course of shorter duration.
Furthermore, most RCTs included in this review excluded recurrent and cancer‐associated distal DVT, which makes any extrapolation to these groups unsafe; in view of the high‐risk nature of these groups, it is plausible to accept that a minimum of three months of anticoagulation, if not longer, would be also required in these high‐risk situations. Certainly, RCTs on extended treatment (secondary prevention) would be required, ideally testing VKAs and DOACs in prophylactic and therapeutic doses, against placebo. Therapies associated with a low risk for bleeding, such as the prophylactic dose of DOACs may well be tested in secondary prevention of other distal DVTs thought to be high‐risk conditions, such as unprovoked DVT.
All but one RCT used a VKA in the investigational arm (following an initial course of heparin), which has the implication that our results apply only to this type of anticoagulant. Further RCTs with LMWHs or DOACs are required.
One of the included studies gave anticoagulation for 10 days only (Schwarz 2010), while the remainder of the studies used anticoagulation treatment for six weeks or more.
This review identified no studies using compression for the treatment of distal DVT. RCTs using compression would be required to make a statement regarding this intervention as either a stand‐alone treatment or adjunct to anticoagulant therapy.
Finally, our findings may not apply to people with single vein postoperative distal DVT in view of the findings of one RCT included in this review (Ferrara 2006), where there was no benefit of a three‐month course compared with the shorter six‐week course. The same may also apply to people with thrombosis limited in extent to the soleal or gastrocnemius veins, pending confirmatory results.
Quality of the evidence
We observed a low risk of bias for most attributes, with the exception of blinding of participants and personnel (performance bias) due to the open‐label design of most RCTs. Nevertheless, the results of the present meta‐analysis update are generally consistent with a low amount of heterogeneity in almost all comparisons. However, the inclusion of RCTs only in this review must be viewed as a strong point.
Using the GRADE assessment, the certainty of evidence for recurrence of VTE and DVT was high; and for PE was low for the comparison of anticoagulation versus placebo and for the comparison of anticoagulation for six weeks versus three months.
The certainty of the evidence for the comparison of anticoagulation versus placebo was low for major bleeding and high for clinically relevant non‐major bleeding. The certainty of the evidence for major bleeding and clinically relevant non‐major bleeding for the comparison of anticoagulation for six weeks versus three months was low..
The certainty of the evidence was downgraded because of imprecision due to small number of events and wide confidence intervals.
Potential biases in the review process
The review authors made a conscious effort to identify all potentially relevant trials for inclusion in the present review. Nevertheless, publication bias still could have limited the validity of our results.
This review set out to assess only RCTs. Although five of the eight RCTs were published before 2010, the reporting of the study methodology was mostly adequate.
The review assessed recurrence of VTE, defined as DVT recurrence in the calf veins, or progression of DVT to proximal veins (e.g. popliteal or femoral vein) or PE, provided that these were objectively diagnosed with venography or ultrasonography for DVT and pulmonary angiography, computed tomography or V/Q scan for PE, as the primary outcome measure. Recurrence of DVT, which included DVT recurrence in the calf veins or progression of DVT to proximal veins, was included as a secondary outcome measure, but in future updates, data permitting, we will add symptomatic proximal DVT and clinically important VTE (proximal DVT and symptomatic PE) as additional important outcomes.
Agreements and disagreements with other studies or reviews
The results presented here agree with one systematic meta‐analysis of 20 case‐control or cohort studies and RCTs that included 2936 people with calf DVT (Franco 2017). Franco 2017 demonstrated a reduction in recurrent VTE rates in people who received anticoagulation compared to those who did not receive anticoagulation (either therapeutic or prophylactic, odds ratio (OR) 0.50, 95% CI 0.31 to 0.79), without an increase in the risk of major bleeding (OR 0.64, 95% CI 0.15 to 2.73). PE rates were also lower with anticoagulation than in the control group (OR 0.48, 95% CI 0.25 to 0.91; 1997 participants). There was a lower rate of recurrent VTE in people who received more than six weeks of anticoagulation in comparison to those who received six weeks of anticoagulation (OR 0.39, 95% CI 0.17 to 0.90; 4 studies, 1136 participants). In comparison, our meta‐analysis included only RCTs known to provide a higher level of evidence than case‐control studies.
Authors' conclusions
Implications for practice.
Our review suggests a benefit for people with distal deep vein thrombosis (DVT) treated with anticoagulation therapy using VKA with little or no difference in major bleeding although there was an increase in clinically relevant non‐major bleeding when compared with no intervention or placebo. This evidence may be used in daily practice for treatment of distal DVT and influence decisions made in guidelines for treatment of DVT, considering the certainty of evidence included in this review.
Implications for research.
The small number of participants of this review prompts for more extended research in distal DVT. More randomised control trials comparing different treatments and different treatment periods with placebo or compression therapy are required to support these results. An interesting endpoint for future randomised control trials is the discrimination of symptomatic or asymptomatic recurrence of DVT or venous thromboembolism.
Acknowledgements
The review authors and the Cochrane Vascular editorial base are grateful to the following peer reviewers for their time and comments: Scott M Stevens, University of Utah, USA; Shannon M Bates, McMaster University, Canada; Dee Shneiderman, USA.
Appendices
Appendix 1. Database searches February 2019
| Source | Search strategy | Hits retrieved |
| Cochrane Vascular Specialised Register via the Cochrane Register of Studies | #1 MESH DESCRIPTOR Pulmonary Embolism EXPLODE ALL AND INREGISTER #2 MESH DESCRIPTOR Thromboembolism EXPLODE ALL AND INREGISTER #3 MESH DESCRIPTOR Thrombosis EXPLODE ALL AND INREGISTER #4 MESH DESCRIPTOR Venous Thromboembolism EXPLODE ALL AND INREGISTER #5 MESH DESCRIPTOR Venous Thrombosis EXPLODE ALL AND INREGISTER #6 (vein* or ven*) adj thromb* AND INREGISTER #7 blood adj3 clot* AND INREGISTER #8 deep vein thrombosis AND INREGISTER #9 lung adj3 clot* AND INREGISTER #10 PE or DVT or VTE AND INREGISTER #11 peripheral vascular thrombosis AND INREGISTER #12 post‐thrombotic syndrome AND INREGISTER #13 pulmonary embolism AND INREGISTER #14 pulmonary adj3 clot* AND INREGISTER #15 thrombus* or thrombopro* or thrombotic* or thrombolic* or thromboemboli* or thrombos* or embol* or microembol* AND INREGISTER #16 venous thromboembolism AND INREGISTER #17 #1 OR #2 OR #3 OR #4 OR #5 OR #6 OR #7 OR #8 OR #9 OR #10 OR #11 OR #12 OR #13 OR #14 OR #15 OR #16 #18 below the knee AND INREGISTER #19 cDVT AND INREGISTER #20 ICMVT AND INREGISTER #21 IDDVT AND INREGISTER #22 infrapopliteal deep veins AND INREGISTER #23 calf AND INREGISTER #24 distal AND INREGISTER #25 #18 OR #19 OR #20 OR #21 OR #22 OR #23 OR #24 #26 #25 AND #17 |
360 |
| CENTRAL via CRSO | #1 MESH DESCRIPTOR Pulmonary Embolism EXPLODE ALL TREES 890 #2 MESH DESCRIPTOR Thromboembolism EXPLODE ALL TREES 1784 #3 MESH DESCRIPTOR Thrombosis EXPLODE ALL TREES 4275 #4 MESH DESCRIPTOR Venous Thromboembolism EXPLODE ALL TREES 479 #5 MESH DESCRIPTOR Venous Thrombosis EXPLODE ALL TREES 2413 #6 (((vein* or ven*) adj thromb*)):TI,AB,KY 8171 #7 (blood adj3 clot*):TI,AB,KY 3981 #8 (deep vein thrombosis):TI,AB,KY 3534 #9 (lung adj3 clot*):TI,AB,KY 7 #10 (PE or DVT or VTE):TI,AB,KY 5333 #11 (peripheral vascular thrombosis):TI,AB,KY 0 #12 (post‐thrombotic syndrome):TI,AB,KY 133 #13 (pulmonary embolism):TI,AB,KY 2206 #14 (pulmonary adj3 clot*):TI,AB,KY 10 #15 (thrombus* or thrombopro* or thrombotic* or thrombolic* or thromboemboli* or thrombos* or embol* or microembol*):TI,AB,KY 23686 #16 (venous thromboembolism):TI,AB,KY 2471 #17 #1 OR #2 OR #3 OR #4 OR #5 OR #6 OR #7 OR #8 OR #9 OR #10 OR #11 OR #12 OR #13 OR #14 OR #15 OR #16 30248 #18 (below the knee):TI,AB,KY 173 #19 cDVT:TI,AB,KY 2 #20 ICMVT:TI,AB,KY 1 #21 IDDVT:TI,AB,KY 4 #22 (infrapopliteal deep veins):TI,AB,KY 0 #23 calf:TI,AB,KY 1451 #24 distal:TI,AB,KY 8172 #25 #18 OR #19 OR #20 OR #21 OR #22 OR #23 OR #24 9724 #26 #17 AND #25 901 |
901 |
| MEDLINE (Ovid MEDLINE Epub Ahead of Print, In‐Process & Other Non‐Indexed Citations, Ovid MEDLINE Daily and Ovid MEDLINE) 1946 to present 2017, 2018 and 2019 only |
1 Pulmonary Embolism/ 2 Thromboembolism/ 3 Thrombosis/ 4 exp Venous Thromboembolism/ 5 exp Venous Thrombosis/ 6 ((vein* or ven*) adj thromb*).ti,ab. 7 (blood adj3 clot*).ti,ab. 8 deep vein thrombosis.ti,ab. 9 (lung adj3 clot*).ti,ab. 10 (PE or DVT or VTE).ti,ab. 11 peripheral vascular thrombosis.ti,ab. 12 post‐thrombotic syndrome.ti,ab. 13 pulmonary embolism.ti,ab. 14 (pulmonary adj3 clot*).ti,ab. 15 (thrombus* or thrombopro* or thrombotic* or thrombolic* or thromboemboli* or thrombos* or embol* or microembol*).ti,ab. 16 venous thromboembolism.ti,ab. 17 or/1‐16 18 "below the knee".ti,ab. 19 cDVT.ti,ab. 20 ICMVT.ti,ab. 21 IDDVT.ti,ab. 22 "infrapopliteal deep veins".ti,ab. 23 calf.ti,ab. 24 distal.ti,ab. 25 or/18‐24 26 17 and 25 27 randomized controlled trial.pt. 28 controlled clinical trial.pt. 29 randomized.ab. 30 placebo.ab. 31 drug therapy.fs. 32 randomly.ab. 33 trial.ab. 34 groups.ab. 35 or/27‐34 36 exp animals/ not humans.sh. 37 35 not 36 38 26 and 37 39 (2017* or 2018* or 2019*).ed. 40 38 and 39 41 from 40 keep 1‐244 |
244 |
| Embase 2017, 2018 and 2019 only | 1 lung embolism/ 2 thromboembolism/ 3 thrombosis/ 4 exp venous thromboembolism/ 5 exp vein thrombosis/ 6 ((vein* or ven*) adj thromb*).ti,ab. 7 (blood adj3 clot*).ti,ab. 8 deep vein thrombosis.ti,ab. 9 (lung adj3 clot*).ti,ab. 10 (PE or DVT or VTE).ti,ab. 11 peripheral vascular thrombosis.ti,ab. 12 post‐thrombotic syndrome.ti,ab. 13 pulmonary embolism.ti,ab. 14 (pulmonary adj3 clot*).ti,ab. 15 (thrombus* or thrombopro* or thrombotic* or thrombolic* or thromboemboli* or thrombos* or embol* or microembol*).ti,ab. 16 venous thromboembolism.ti,ab. 17 or/1‐16 18 "below the knee".ti,ab. 19 cDVT.ti,ab. 20 ICMVT.ti,ab. 21 IDDVT.ti,ab. 22 "infrapopliteal deep veins".ti,ab. 23 calf.ti,ab. 24 distal.ti,ab. 25 or/18‐24 26 17 and 25 27 randomized controlled trial/ 28 controlled clinical trial/ 29 random$.ti,ab. 30 randomization/ 31 intermethod comparison/ 32 placebo.ti,ab. 33 (compare or compared or comparison).ti. 34 ((evaluated or evaluate or evaluating or assessed or assess) and (compare or compared or comparing or comparison)).ab. 35 (open adj label).ti,ab. 36 ((double or single or doubly or singly) adj (blind or blinded or blindly)).ti,ab. 37 double blind procedure/ 38 parallel group$1.ti,ab. 39 (crossover or cross over).ti,ab. 40 ((assign$ or match or matched or allocation) adj5 (alternate or group$1 or intervention$1 or patient$1 or subject$1 or participant$1)).ti,ab. 41 (assigned or allocated).ti,ab. 42 (controlled adj7 (study or design or trial)).ti,ab. 43 (volunteer or volunteers).ti,ab. 44 trial.ti. 45 or/27‐44 46 26 and 45 47 (2017* or 2018* or 2019*).em. 48 46 and 47 |
747 |
| CINAHL 2017, 2018 and 2019 only | S42 S40 AND S41 S41 EM 2017 OR EM 2018 OR EM 2019 S40 S26 AND S39 S39 S27 OR S28 OR S29 OR S30 OR S31 OR S32 OR S33 OR S34 OR S35 OR S36 OR S37 OR S38 S38 MH "Random Assignment" S37 MH "Single‐Blind Studies" or MH "Double‐Blind Studies" or MH "Triple‐Blind Studies" S36 MH "Crossover Design" S35 MH "Factorial Design" S34 MH "Placebos" S33 MH "Clinical Trials" S32 TX "multi‐centre study" OR "multi‐center study" OR "multicentre study" OR "multicenter study" OR "multi‐site study" S31 TX crossover OR "cross‐over" S30 AB placebo* S29 TX random* S28 TX trial* S27 TX "latin square" S26 S17 AND S25 S25 S18 OR S19 OR S20 OR S21 OR S22 OR S23 OR S24 S24 TX distal S23 TX calf S22 TX infrapopliteal deep veins S21 TX IDDVT S20 TX ICMVT S19 TX cDVT S18 TX below the knee S17 S1 OR S2 OR S3 OR S4 OR S5 OR S6 OR S7 OR S8 OR S9 OR S10 OR S11 OR S12 OR S13 OR S14 OR S15 OR S16 S16 TX venous thromboembolism S15 TX thrombus* or thrombopro* or thrombotic* or thrombolic* or thromboemboli* or thrombos* or embol* or microembol* S14 TX pulmonary n3 clot* S13 TX pulmonary embolism S12 TX post‐thrombotic syndrome S11 TX peripheral vascular thrombosis S10 TX PE or DVT or VTE S9 TX lung n3 clot* S8 TX deep vein thrombosis S7 TX blood n3 clot* S6 TX ((vein* or ven*) n thromb*) S5 (MH "Venous Thrombosis+") S4 (MH "Venous Thromboembolism") S3 (MH "Thrombosis") S2 (MH "Thromboembolism") S1 (MH "Pulmonary Embolism") |
50 |
| Clinicaltrials.gov | below the knee OR distal OR calf | Interventional Studies | Venous Thromboembolism OR Venous Thrombosis OR Pulmonary Embolism OR deep vein thrombosis OR DVT | 94 |
| ICTRP Search Portal | below the knee OR distal OR calf | Venous Thromboembolism OR Venous Thrombosis OR Pulmonary Embolism OR deep vein thrombosis OR DVT | 18 |
Data and analyses
Comparison 1. Anticoagulant versus no intervention or placebo.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Recurrence of venous thromboembolism (VTE) | 5 | 496 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.34 [0.15, 0.77] |
| 1.1 anticoagulation for 3 months | 3 | 137 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.13 [0.02, 0.65] |
| 1.2 anticoagulation for up to 6 weeks | 2 | 359 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.63 [0.23, 1.69] |
| 2 Major bleeding | 4 | 480 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.76 [0.13, 4.62] |
| 2.1 anticoagulation for 3 months | 2 | 121 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.24 [0.01, 4.80] |
| 2.2 anticoagulation for up to 6 weeks | 2 | 359 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.20 [0.13, 77.69] |
| 3 Recurrence of deep vein thrombosis (DVT) | 5 | 496 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.25 [0.10, 0.67] |
| 3.1 anticoagulation for 3 months | 3 | 137 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.09 [0.01, 0.71] |
| 3.2 anticoagulation for up to 6 weeks | 2 | 359 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.46 [0.14, 1.48] |
| 4 Pulmonary embolism (PE) | 4 | 480 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.81 [0.18, 3.59] |
| 4.1 anticoagulation for 3 months | 2 | 121 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.37 [0.04, 3.42] |
| 4.2 anticoagulation for up to 6 weeks | 2 | 359 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.13 [0.20, 23.21] |
| 5 Clinically relevant non‐major bleeding | 2 | 322 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.34 [1.07, 10.46] |
| 5.1 anticoagulation for 3 months | 1 | 70 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.33 [0.66, 8.30] |
| 5.2 anticoagulation for up to 6 weeks | 1 | 252 | Risk Ratio (M‐H, Fixed, 95% CI) | 9.59 [0.52, 176.20] |
| 6 Overall mortality | 3 | 430 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.20 [0.13, 77.69] |
| 6.1 anticoagulation for 3 months | 1 | 70 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6.2 anticoagulation for up to 6 weeks | 2 | 360 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.20 [0.13, 77.69] |
| 7 Mortality related to PE | 3 | 430 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.1 anticoagulation for 3 months | 1 | 70 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.2 anticoagulation for up to 6 weeks | 2 | 360 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8 Mortality related to major bleeding | 3 | 430 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8.1 anticoagulation for 3 months | 1 | 70 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8.2 anticoagulation for up to 6 weeks | 2 | 360 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9 Post‐thrombotic syndrome (PTS) | 1 | 178 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.58, 1.41] |
1.7. Analysis.

Comparison 1 Anticoagulant versus no intervention or placebo, Outcome 7 Mortality related to PE.
1.8. Analysis.

Comparison 1 Anticoagulant versus no intervention or placebo, Outcome 8 Mortality related to major bleeding.
1.9. Analysis.

Comparison 1 Anticoagulant versus no intervention or placebo, Outcome 9 Post‐thrombotic syndrome (PTS).
Comparison 2. Anticoagulant versus any anticoagulant: three months or more of vitamin K antagonist (VKA) versus six weeks of VKA.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Recurrence of venous thromboembolism (VTE) | 3 | 736 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.42 [0.26, 0.68] |
| 2 Major bleeding | 2 | 389 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.42 [0.36, 32.35] |
| 3 Recurrence of deep vein thrombosis (DVT) | 2 | 389 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.32 [0.16, 0.64] |
| 4 Pulmonary embolism (PE) | 2 | 389 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.05 [0.19, 5.88] |
| 5 Clinically relevant non‐major bleeding | 2 | 389 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.76 [0.90, 3.42] |
| 6 Mortality related to PE | 1 | 197 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7 Mortality related to major bleeding | 2 | 389 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
2.6. Analysis.

Comparison 2 Anticoagulant versus any anticoagulant: three months or more of vitamin K antagonist (VKA) versus six weeks of VKA, Outcome 6 Mortality related to PE.
2.7. Analysis.

Comparison 2 Anticoagulant versus any anticoagulant: three months or more of vitamin K antagonist (VKA) versus six weeks of VKA, Outcome 7 Mortality related to major bleeding.
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Ferrara 2006.
| Methods | Open, randomised, controlled clinical trial | |
| Participants | Number: 192 participants Group 1: 68 participants, involved a single collecting vein. 24 participants had undergone orthopaedic surgery, 22 abdominal surgery and 22 urological surgery. The thrombus was present in a single collecting vein in 48 of these participants and in a single muscular vein in 20. This group was subdivided into 2 subgroups of 34 participants (called 1A and 1B); in the 1A subgroup, the treatment was continued for 12 weeks, and in the 1B subgroup, it was continued for 6 weeks. Group 2: 124 participants involved > 1 collecting vein. 35 participants had undergone orthopaedic surgery, 45 abdominal surgery and 44 urological surgery. 89 of these participants with distal DVT in > 1 vessel presented with thrombotic lesions in 2 vessels and 35 participants had thrombotic lesions in 3 vessels. This group was divided into 2 subgroups of 62 participants (called 2A and 2B); in the 2A subgroup, treatment was continued for 12 weeks, and in the 2B subgroup, it was continued for 6 weeks. Age (range; years): 44–71, group 1; 48–72, group 2 Gender (M/F): 23/45, group 1; 41/83, group 2 |
|
| Interventions | VKAs for 12 weeks vs VKAs for 6 weeks. All participants were treated with nadroparin calcium at daily doses of 200 IU anti‐Xa per kg bodyweight, given in 2 doses. Contemporarily, sodium warfarin was administered, and nadroparin was stopped until prothrombin activity had stabilised at about 30–40% with an INR of 2–3. Heparin treatment was stopped in all participants after 5 or 6 days. |
|
| Outcomes | Extension of calf DVT to proximal veins, symptomatic PE and major bleeding | |
| Notes | People with cancer, inherited coagulopathies (factor V Leiden, G20120 mutation in prothrombin, hyperhomocystinaemia, dysplasminogenaemia, deficits in vitamin C or protein S, dysfibrinogenaemia), hyperviscosity syndromes or antiphospholipid antibodies, or absolute or relative contraindications for heparin or anticoagulant treatment were excluded. Funding/support: not reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Insufficient information about the sequence generation process. |
| Allocation concealment (selection bias) | Unclear risk | Insufficient information about the allocation concealment. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Open‐label study. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | No blinding of outcome assessment but the review authors judged that the outcome measurement was unlikely to be influenced by lack of blinding. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing data. |
| Selective reporting (reporting bias) | Low risk | The published report included all expected outcomes. |
| Other bias | Low risk | The study appeared free of other sources of bias. |
Horner 2014.
| Methods | Randomised, open‐label trial 93 assessed for eligibility and 23 excluded 0 lost to follow‐up |
|
| Participants | Number: 70 participants 35 allocated to therapeutic anticoagulation and 35 allocated to conservative management Age (mean ± SD; years): 60.9 ± 17.8, therapeutic anticoagulation; 59.8 ± 17.9, conservative management Gender (M/F): 9/26, therapeutic anticoagulation; 15/20, conservative management |
|
| Interventions | Dalteparin/VKA for 3 months vs no anticoagulation. Intervention group: initially given a subcutaneous therapeutic dose of dalteparin with phased transition to an oral VKA for a total of 3 months. All participants were followed up in a dedicated anticoagulant clinic for INR monitoring, with target INR 2.5 (range: 2.0–3.0). Control group: given anti‐inflammatory medication and paracetamol for symptomatic relief, but received no anticoagulation. All participants regardless of treatment allocation were referred for fitted grade 2 compression stockings. |
|
| Outcomes | Proximal propagation with or without symptoms, symptomatic PE, VTE‐related sudden death or major bleeding | |
| Notes | People with previous VTE were excluded. Funding/support: "This work was funded by the College of Emergency Medicine and supported by the National Institute for Health Research through the comprehensive local research network." |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Via a web‐based platform with an externally generated randomisation sequence in variable permuted block sizes. Participants were randomised in a 1:1 allocation ratio. |
| Allocation concealment (selection bias) | Low risk | Central allocation via a web‐based platform. Quote: "Randomization occurred following written informed consent, such that allocation concealment was maintained until the absolute pint of inclusion." |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Open‐label study. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | No blinding of outcome assessment but the review authors judged that the outcome measurement was unlikely to be influenced by lack of blinding. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing data. |
| Selective reporting (reporting bias) | Low risk | Published report included all expected outcomes. |
| Other bias | Low risk | Study appeared free of other sources of bias. |
Lagerstedt 1985.
| Methods | Randomised, open label trial 52 randomised, 1 excluded and lost follow‐up |
|
| Participants | Number: 51 participants 23 received warfarin and 28 control group with no anticoagulation Age (mean ± SD; years); 65.0 ± 14.4, warfarin group; 60.9 ± 12.4, control group Gender (M/F): 14/9, warfarin group; 15/13, control group |
|
| Interventions | UFH/warfarin for 3 months vs no anticoagulation All participants received a 5‐day course of sodium heparin intravenously, 500–600 IU/kg/day in 6 divided doses. Intervention: participants receiving oral anticoagulation were started on warfarin as soon as a diagnosis was confirmed by phlebography. The aim was to achieve an INR of 2.5–4.2. Heparin treatment was extended for 1–2 days if the therapeutic level was not reached on the 5th day. Control: no anticoagulation All participants were asked to wear graded compression stocking during the study |
|
| Outcomes | VTE recurrence, clinical or revealed (or both) on imaging | |
| Notes | People with previous VTE, PE, thrombi extending into popliteal vein, malignancy were excluded. Funding/support: quote: "The study was supported in part by grants from the National Organisation against Heart and Lung Diseases." |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Used sealed envelopes. |
| Allocation concealment (selection bias) | Low risk | Used sealed envelopes. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Open‐label study. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | No blinding of outcome assessment but the review authors judged that the outcome measurement was unlikely to be influenced by lack of blinding. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing data. |
| Selective reporting (reporting bias) | Low risk | Published report included all expected outcomes. |
| Other bias | Low risk | Study appeared free of other sources of bias. |
Nielsen 1994.
| Methods | Randomised, open‐label trial | |
| Participants | Number; 16 participants 9 received anticoagulant treatment and 7 received no anticoagulant treatment Age: not specified for participants with distal DVT Gender: not specified for participants with distal DVT |
|
| Interventions | UFH/phenprocoumon for 3 months vs no anticoagulation. Intervention: sodium heparin administered intravenously. Treatment initiated by bolus injection of 10,000 IU, followed by continuous infusion (20,000 IU of heparin in 500 mL of 5% dextrose) with APTT target at 1.5–2.5. Phenprocoumon was given from the 3rd day. Heparin treatment was continued for ≥ 6 days or until INR had reached 2.0–4.3. Control: participants were treated with phenylbutazone 200 mg 3 times at the day of admission and then 100 mg 3 times daily for the following 9 days. All participants were actively mobilised from the day of admission and wore graduated compressing stockings. |
|
| Outcomes | Propagation or development of new VTE | |
| Notes | People with clinical symptoms of PE were excluded. Study protocol involved 90 participants with DVT; 16 were diagnosed with distal DVT. Funding/support: not reported. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Insufficient information about sequence generation process. |
| Allocation concealment (selection bias) | Low risk | Random allocation. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Open‐label study. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | No blinding of outcome assessment but the review authors judged that the outcome measurement was unlikely to be influenced by lack of blinding. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing data. |
| Selective reporting (reporting bias) | Low risk | Published report included all expected outcomes. |
| Other bias | Low risk | Study appeared free of other sources of bias. |
Pinede 2001.
| Methods | Open‐label, randomised, controlled trial | |
| Participants | Number: 197 participants 105 received 6 weeks of anticoagulation treatment and 92 received 12 weeks of anticoagulation treatment Age, % > 60 years age: 38.5%, 6‐week treatment group; 27.0%, 12‐week treatment group Gender: not specified for distal thrombosis |
|
| Interventions | VKAs for 6 weeks vs 12 weeks. All participants received fluindione, a VKA with a long half‐life. During the acute phase, physicians were free to use either intravenous or subcutaneous UFH or 1 or 2 daily injections of LMWH. The recommended duration of heparin therapy was ≥ 5 days and until an anticoagulation value within the INR target range of 2.0–3.0. Graduated compression stockings were systematically recommended for all participants with DVT. |
|
| Outcomes | Recurrent VTE and haemorrhage | |
| Notes | Exclusion criteria: pregnancy, breastfeeding, previous VTE, vena cava filter implantation, surgical thrombectomy, free‐floating thrombus in the inferior vena cava lumen, evolutive cancer or malignant haematological disease, known biological thrombophilia, severe PE (defined by an amputation of > 50% of vascularisation), PE treated by thrombolysis (considered a marker of severe PE), myocardiopathy or other diseases justifying prolonged anticoagulation therapy, and liver insufficiency. The data came from a larger study which included people with proximal and distal DVT. Funding/support; quote: "The DOTAVK study was financially supported by the French Ministry of Health (HCL‐PHRC n°93‐95.010), Hospices Civils de Lyon, Conseil Régional Rhône‐Alpes, and the Association pour la recherche et l’évaluation thérapeutique (APRET)." |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated allocation schedule in blocks of 4. |
| Allocation concealment (selection bias) | Low risk | Random allocation via centralised computer. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Open‐label study. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | No blinding of outcome assessment but the review authors judged that the outcome measurement was unlikely to be influenced by lack of blinding. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing data. |
| Selective reporting (reporting bias) | Low risk | Published report included all expected outcomes. |
| Other bias | Low risk | Study appeared free of other sources of bias. |
Righini 2016.
| Methods | Randomised, double‐blind, placebo‐controlled trial (746 participants assessed for eligibility, 487 excluded, 3 lost to follow‐up, 4 withdrew consent) | |
| Participants | Number; 259 participants 126 received nadroparin and 133 received placebo Age (median (IQR); years): 52 (17), nadroparin group; 53 (17), placebo group Gender (M/F): 62/64, nadroparin group; 70/63, placebo group |
|
| Interventions | Nadroparin for 6 weeks vs placebo Within 36 hours of their qualifying ultrasound, participants were randomised. Intervention: nadroparin 171 UI/kg daily administered by the participant subcutaneously for 42 days Control: identically packaged placebo injections (sterilised saline 0.9%) administered by the participant subcutaneously for 42 days Participants were prescribed and instructed to use graduated elastic compression stockings (30 mmHg) daily. |
|
| Outcomes | Extension of calf DVT to proximal veins, contralateral proximal DVT, symptomatic PE, PTS, pain | |
| Notes | People with previous VTE or cancer were excluded. Funding/support: quote: "This study was supported by a grant from the Swiss National Science Foundation (number 3200B0–104172), a grant from the Programme Hospitalier de Recherche Clinique (French Ministry of Health [PHRC] 20051409), a grant from the Canadian Institutes of Health Research (MOP‐119524), and a grant from the 2007 International Society on Thrombosis and Haemostasis (ISTH) Presidential Fund. GLG holds a Faculty of Medicine Department of Medicine Chair on Diagnosis of Venous Thromboembolism and a Clinician Scientist Award from the Heart and Stroke Foundation of Ontario. Nadroparin and placebo were provided by GlaxoSmithKline France and Aspen. Stockings were provided to Canadian patients by Sigvaris." |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Random allocation of each patient was centrally determined by an independent research clinical centre using the ORTA/VISTA software of the European Organisation for Research and Treatment of Cancer (EORTC) and its web interface, with allocation stratified by study centre. Variable block sizes were used." |
| Allocation concealment (selection bias) | Low risk | Quote: "Sequentially numbered boxes including 42 syringes were delivered to the pharmacy of the enrolling centres. All boxes were tamper‐proof, equal in weight, and similar in appearance. Using an EORTC web‐based interface, the investigator received by email the number of the box that had to be delivered to the patient." |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "All study personnel, participants, and outcome assessors (central adjudication committee) were masked to group assignment." Comment: double‐blind |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "All study personnel, participants, and outcome assessors (central adjudication committee) were masked to group assignment." Comment: double‐blind |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing data. |
| Selective reporting (reporting bias) | Low risk | Published reports included all expected outcomes. |
| Other bias | Low risk | Study appeared free of other sources of bias. |
Schulman 1995.
| Methods | Randomised, open‐label trial | |
| Participants | Number: 347 participants 176 participants received anticoagulation treatment for 6 weeks and 171 participants received anticoagulation treatment for 6 months Age: not stratified by distal thrombosis Gender: not stratified by distal thrombosis |
|
| Interventions | VKA for 6 months vs 6 weeks The initial treatment with UFH or LMWH administered intravenously or subcutaneously for ≥ 5 days, until a prothrombin time within the target range had been achieved. Participants with DVT were provided with a graded compression stocking and instructed to wear it on the affected leg during the day for ≥ 1 year. Oral anticoagulation with warfarin sodium or dicumarol was usually started at the same time as heparin. Oral anticoagulation was targeted to an INR of 2.0–2.85. |
|
| Outcomes | Recurrent VTE at 2 years | |
| Notes | People with previous VTE or cancer were excluded. The data came from a larger study which included people with proximal and distal DVT. We only had stratified data for the outcome recurrent VTE at 2 years. Funding/support: quote: "Supported by grants from Swedish Heart Lung Foundation, the Swedish Society of Medicine, the Karolinska Institute, Skandia, Trygg–Hansa, Triolab, Karolinska Hospital, Nycomed, and Stago." |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated allocation. |
| Allocation concealment (selection bias) | Low risk | Central allocation. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Open‐label study. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | No blinding of outcome assessment but the review authors judged that the outcome measurement was unlikely to be influenced by lack of blinding. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing data. |
| Selective reporting (reporting bias) | Low risk | Published report included all expected outcomes. |
| Other bias | Low risk | Study appeared free of other sources of bias. |
Schwarz 2010.
| Methods | Randomised, open‐label trial | |
| Participants | Number; 107 participants 54 received nadroparin and 53 received compression therapy Age (mean ± SD years): 54 ± 15, nadroparin group; 57 ± 14, compression therapy group Gender (M/F): 24/30, nadroparin group; 15/38, compression therapy group |
|
| Interventions | Nadroparin for 10 days vs no anticoagulation Intervention: 180 antiXa u/kg nadroparin once daily for 10 days plus compression therapy with graduated class‐II‐calf stockings for 3 months. Control: compression therapy alone with graduated class‐II‐calf stockings for 3 months. |
|
| Outcomes | Progression into the deep veins and clinical PE | |
| Notes | People with previous VTE were excluded. Funding/support; "Supported by Sanofi Synthelabo, Berlin, Germany." |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomisation table. |
| Allocation concealment (selection bias) | Unclear risk | Insufficient information about allocation concealment. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Open‐label study. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | No blinding of outcome assessment but the review authors judged that the outcome measurement was unlikely to be influenced by lack of blinding. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No missing data. |
| Selective reporting (reporting bias) | Low risk | Published report included all expected outcomes. |
| Other bias | Low risk | Study appeared free of other sources of bias. |
APTT: activated partial thromboplastin time; DVT: deep vein thrombosis; F: female; INR: international normalised ratio; IQR: interquartile range; LMWH: low molecular weight heparin; M: male; PE: pulmonary embolism; PTS: post‐thrombotic syndrome; SD: standard deviation; UFH: unfractionated heparin; VKA: vitamin K antagonist; VTE: venous thromboembolism.
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Dentali 2017 | Retrospective study |
| Donadini 2017 | Retrospective cohort study |
| Galanaud 2017 | Observational, prospective study |
| Ho 2016 | Retrospective study |
| Leizorovicz 2003 | Prevention study |
| McBane 2018 | Prospective single‐arm study |
| Musil 2000 | Not relevant |
| NCT00816920 | Observational study |
| NCT01234064 | Ongoing prevention study |
| NCT01252420 | Observational study |
| NCT02746185 | Ongoing study on VTE treatment in people with cancer |
| Pegoraro 2016 | Multicentre cohort study – abstract only |
| Schulman 1986 | Study of adjuvant thrombolysis |
| Schwarz 2001 | Cohort study – non‐RCT |
| Utter 2016 | Retrospective cohort study |
RCT: randomised controlled trial; VTE: venous thromboembolism.
Characteristics of ongoing studies [ordered by study ID]
EUCTR2005‐004235‐21‐IT.
| Trial name or title | Calf vein thrombosis 6 weeks treatment with LMWH vs warfarin |
| Methods | Randomised open‐label trial |
| Participants | 800 men and women aged ≥ 18 years with distal DVT |
| Interventions | LMWH 8000 IU (sc) Warfarin 5 mg (oral) |
| Outcomes | Not reported in trial record |
| Starting date | Not reported in trial record |
| Contact information | Study sponsor: Azienda Ospedaliera Arcispedale S. Maria Nuova |
| Notes | EUCTR record reported limited information According to EUCTR record study completed. The study authors have confirmed that data are not yet available. |
NCT02722447.
| Trial name or title | Rivaroxaban for the treatment of symptomatic isolated distal deep vein thrombosis |
| Methods | Randomised, clinical trial, quadruple masked (participant, care provider, investigator, outcomes assessor) |
| Participants | 1100 men and women aged ≥ 18 years, with objective diagnosis of isolated distal DVT |
| Interventions | Rivaroxaban vs placebo |
| Outcomes |
|
| Starting date | January 2017 |
| Contact information | Walter Ageno, Associate Professor of Medicine, Università degli Studi dell'Insubria, Italy |
| Notes |
NCT03368313.
| Trial name or title | Non‐elastic compression garment therapy versus control for deep vein thrombosis |
| Methods | Randomised, single‐blind (outcome assessor) trial |
| Participants | 120 men and women aged 18–80 years with acute DVT of lower extremity involving femoropopliteal or ilofemoral segment, or both |
| Interventions | Compression device vs no compression |
| Outcomes |
|
| Starting date | 1 May 2018 |
| Contact information | Eduardo Matta, MD, Pró Circulação, Brazil |
| Notes |
NCT03590743.
| Trial name or title | Calf deep vein thrombosis treatment trial |
| Methods | Randomised, double‐blind, placebo‐controlled |
| Participants | 250 men and women aged ≥ 18 years, with confirmed acute calf vein thrombosis confined to either the deep (posterior tibial, anterior tibial, or peroneal) or muscular (gastrocnemius or soleal) veins |
| Interventions | Apixaban vs placebo |
| Outcomes |
|
| Starting date | 19 February 2019 |
| Contact information | Robert D McBane, Mayo Clinic, USA |
| Notes |
UMIN000028105.
| Trial name or title | Evaluation of rivaroxaban for distal deep vein thrombosis – a single center, randomized, open‐label, assessor‐blind, parallel group, exploratory study |
| Methods | Randomised, open‐label, with blinded assessors |
| Participants | 150 men and women aged ≥ 20 years, who have been newly diagnosed with distal DVT by CUS |
| Interventions | Rivaroxaban vs physical treatment only Rivaroxaban: 15 mg tablets orally administered after meal twice daily for 21 days after starting treatment (initial treatment period). Subsequently, dosage reduced to 15 mg tablet once daily for 69 days (maintenance period). If there is a bleeding risk in the initial treatment period, dosage can be reduced to 15 mg tablet once daily according to judgement of physician. In addition, physical therapy including wearing an elastic stocking or an elastic wrap shall be thoroughly instructed. Physical treatment: physical therapy including wearing an elastic stocking or an elastic wrap. |
| Outcomes | Primary outcome
Secondary outcomes
|
| Starting date | 2 April 2018 |
| Contact information | Masaaki Ito, Mie University Graduate School of Medicine, Japan |
| Notes |
CUS: compression ultrasonography; DVT: deep vein thrombosis; LMWH: low molecular weight heparin; PE: pulmonary embolism; PTS: post‐thrombotic syndrome; sc: subcutaneous; VEINES‐QOL/Sym: VEnous INsufficiency Epidemiological and Economic Study – Quality Of Life/Symptoms; VTE: venous thromboembolism.
Differences between protocol and review
None.
Contributions of authors
GIK: drafted the protocol; selected which studies to include, extracted data from studies, entered data into Review Manager 5, carried out and interpreted the analysis, drafted the final review and will update the review.
SKK: drafted the protocol; obtained copies of studies, interpreted the analysis, drafted the final review and will update the review.
CB: contributed to the protocol; interpreted the analysis, drafted the final review and will update the review.
SS: contributed to the protocol; selected which studies to include, extracted data from studies, interpreted the analysis, drafted the final review and will update the review.
KK: contributed to the protocol; interpreted the analysis, drafted the final review and will update the review.
Sources of support
Internal sources
No sources of support supplied
External sources
-
Chief Scientist Office, Scottish Government Health Directorates, The Scottish Government, UK.
The Cochrane Vascular editorial base is supported by the Chief Scientist Office.
Declarations of interest
GIK: none.
SKK: has received a consulting fee or honorarium from Bayer, Pfizer, Sanofi and Leo. There are no known conflicts of interest with this review.
CB: has received payments from Medtronic, Orzone and Bolton Medical as part of a contract for consultancy and for lectures related to this contract. He has no conflict of interest related to the submitted work. Unrelated to this work, he has received payment for travel and conference fees from Medtronic, Gore, Bolton Medical and Vascutek.
SS: none.
KK: none.
New
References
References to studies included in this review
Ferrara 2006 {published data only}
- Ferrara F, Meli F, Amato C, Cospite V, Raimondi F, Novo G. Optimal duration of treatment in surgical patients with calf venous thrombosis involving one or more veins. Angiology 2006;57:418‐23. [DOI] [PubMed] [Google Scholar]
Horner 2014 {published data only}
- Horner D, Hogg K, Body R, Nash MJ, Baglin T, Mackway‐Jones K. The Anticoagulation of Calf Thrombosis (ACT) project: results from the randomized controlled external pilot trial. Chest 2014;146(6):1468‐77. [DOI: 10.1378/chest.14-0235] [DOI] [PubMed] [Google Scholar]
- Horner D, Hogg K, Body R, Nash MJ, Mackway‐Jones K. The Anticoagulation of Calf Thrombosis (ACT) project: study protocol for a randomized controlled trial. Trials 2012;13:31. [DOI] [PMC free article] [PubMed] [Google Scholar]
Lagerstedt 1985 {published data only}
- Lagerstedt C, Olsson CG, Fagher BO, Oqvist BW, Albrechtsson U. Need for long‐term anticoagulant treatment in symptomatic calf‐vein thrombosis. Lancet 1985;2(8454):515‐8. [DOI] [PubMed] [Google Scholar]
Nielsen 1994 {published data only}
- Nielsen HK, Husted SE, Krusell LR, Fasting H, Charles P, Hansen HH, et al. Anticoagulant therapy in deep venous thrombosis. A randomized controlled study. Thrombosis Research 1994;73:215‐26. [DOI] [PubMed] [Google Scholar]
Pinede 2001 {published data only}
- Pinede L, Ninet J, Duhaut P, Chabaud S, Demolombe‐Rague S, Durieu I, et al. Comparison of 3 and 6 months of oral anticoagulant therapy after a first episode of proximal deep vein thrombosis or pulmonary embolism and comparison of 6 and 12 weeks of therapy after isolated calf deep vein thrombosis. Circulation 2001;103(20):2453‐60. [DOI: 10.1161/01.CIR.103.20.2453] [DOI] [PubMed] [Google Scholar]
Righini 2016 {published data only}
- Galanaud J‐P, Righini M, Lecollen L, Douillard A, Robert‐Ebadi H, Pontal D, et al. Long‐term risk of post‐thrombotic syndrome after symptomatic distal deep‐vein thrombosis: the cactus‐PTS study. Research and Practice in Thrombosis and Haemostasis 2018;2(Suppl 1):328‐9. [DOI: 10.1002/rth2.12125] [DOI] [Google Scholar]
- Righini M, Galanaud JP, Guenneguez H, Brisot D, Diard A, Faisse P, et al. Anticoagulant therapy for symptomatic calf deep vein thrombosis (CACTUS): a randomised, double‐blind, placebo‐controlled trial. Lancet Haematology 2016;3(12):556‐62. [DOI: ] [DOI] [PubMed] [Google Scholar]
- Righini M, Robert‐Ebadi H, Glauser F, Blondon M, Ouvry P, Diamand J‐M, et al. Effect of anticoagulant treatment on pain in distal deep vein thrombosis: an ancillary analysis from the cactus trial. Journal of Thrombosis and Haemostasis 2019;17(3):507‐10. [DOI] [PubMed] [Google Scholar]
Schulman 1995 {published data only}
- Schulman S, Rhedin A, Lindmarker P, Carlsson A, Lafrars G, Nicol P, et al. A comparison of six weeks with six months of oral anticoagulant therapy after a first episode of venous thromboembolism. New England Journal of Medicine 1995;332(25):1661‐5. [DOI] [PubMed] [Google Scholar]
Schwarz 2010 {published data only}
- Schwarz T, Buschmann L, Beyer J, Halbritter K, Rastan A, Schellong S. Therapy of isolated calf muscle vein thrombosis: a randomized, controlled study. Journal of Vascular Surgery 2010;52(5):1246‐50. [DOI: 10.1016/j.jvs.2010.05.094] [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Dentali 2017 {published data only}
- Dentali F, Pegorara A, Barco S, Minno MN, Mastroiacovo D, Pomero F, et al. Clinical course of isolated deep vein thrombosis in patients with active cancer: a multicenter cohort study. Journal of Thrombosis and Haemostasis 2017;15(9):1757‐63. [DOI] [PubMed] [Google Scholar]
Donadini 2017 {published data only}
- Donadini MP, Dentali F, Pegoraro S, Pomero F, Brignone C, Guasti L, et al. Long‐term recurrence of venous thromboembolism after short‐term treatment of symptomatic isolated distal deep vein thrombosis: a cohort study. Vascular Medicine 2017;22(6):518‐24. [DOI] [PubMed] [Google Scholar]
Galanaud 2017 {published data only}
- Galanaud JP, Sevestre M‐A, Pernod G, Genty C, Richelet S, Kahn SR, et al. Long‐term outcomes of cancer‐related isolated distal deep vein thrombosis: the OPTIMEV study. Journal of Thrombosis and Haemostasis 2017;15(5):907‐16. [DOI] [PubMed] [Google Scholar]
Ho 2016 {published data only}
- Ho P, Lim HY, Chua CC, Sleeman M, Tacey M, Donnan G, et al. Retrospective review on isolated distal deep vein thrombosis(IDDVT) – a benign entity or not?. Thrombosis Research 2016;142:11‐6. [DOI] [PubMed] [Google Scholar]
Leizorovicz 2003 {published data only}
- Leizorovicz A, Goldhaber SZ, Olsson CG, Cohen AT, Weitz JI, Turpie AG. Mortality is increased in patients with proximal, but not distal, deep vein thrombosis in medically ill patients entered in the PREVENT trial. Blood 2003;102:165a. [Google Scholar]
McBane 2018 {published data only}
- McBane R, Bott‐Kitslaar D, Casanegra A, Froehling D, Bartlett M, Lenz C, et al. Rivaroxaban and apixaban for treatment of acute isolated calf deep vein thrombosis – a single center prospective study. Research and Practice in Thrombosis and Haemostasis 2018;2(Suppl 1):339. [Google Scholar]
Musil 2000 {published data only}
- Musil D. Regression of deep venous thrombosis. Vnitrni Lekarstvi 2000;46(1):16‐8. [PubMed] [Google Scholar]
NCT00816920 {published data only}
- NCT00816920. Natural history of isolated deep vein thrombosis of the calf (CALTHRO). clinicaltrials.gov/ct2/show/NCT00816920 (first received 5 January 2009).
NCT01234064 {published data only}
- NCT01234064. Graduated Compression Stockings (GCS) pilot substudy. clinicaltrials.gov/ct2/show/NCT01234064 (first received 4 November 2010).
NCT01252420 {published data only}
- NCT01252420. Two weeks of low molecular weight heparin for distal vein thrombosis (TWISTER). clinicaltrials.gov/ct2/show/NCT01252420 (first received 3 December 2010).
NCT02746185 {published data only}
- NCT02746185. Cancer associated thrombosis, a pilot treatment study using rivaroxaban (CASTA‐DIVA). clinicaltrials.gov/ct2/show/NCT02746185 (first received 21 April 2016).
Pegoraro 2016 {published data only}
- Pegoraro S, Nisio M, Barco S, Minno MN, Mastroiacovo D, Pomero F, et al. Clinical history of cancer patients with isolated distal deep vein thrombosis: a multicenter cohort study. Italian Journal of Medicine 2016;10(Suppl 2):87. [DOI] [PubMed] [Google Scholar]
Schulman 1986 {published data only}
- Schulman S, Granqvist SA, Juhlin‐Dannfelt A, Lockner D. Long‐term sequelae of calf vein thrombosis treated with heparin or low‐dose streptokinase. Acta Medica Scandinavica 1986;219(4):349‐57. [DOI] [PubMed] [Google Scholar]
Schwarz 2001 {published data only}
- Schwarz T, Schmidt B, Beyer J, Schellong SM. Therapy of isolated calf muscle vein thrombosis – results of a cohort study. Annals of Hematology 2001;80(Suppl 1):A59. [Google Scholar]
Utter 2016 {published data only}
- Utter GH, Dhillon TS, Salcedo ES, Shouldice DJ, Reynolds CL, Humphries MD, et al. Therapeutic anticoagulation for isolated calf deep vein thrombosis. JAMA Surgery 2016;151(9):e161770. [DOI] [PubMed] [Google Scholar]
References to ongoing studies
EUCTR2005‐004235‐21‐IT {published data only}
- EUCTR2005‐004235‐21‐IT. Calf vein thrombosis 6 weeks‐treatment with LMWH vs warfarin. clinicaltrialsregister.eu/ctr‐search/trial/2005‐004235‐21/IT (Date first received 12 January 2007).
- Ghirarduzzi A, Camporese G, Siragusa S, Imberti D, Bucherini E, Landini F, et al. A randomized, prospective, open‐label study on distal vein thrombosis (low‐molecular‐weight heparin vs. warfarin for 6 weeks): the todi study. Journal of Thrombosis and Haemostasis 2009;7(Suppl 2):abstract no: PP‐WE‐403. [Google Scholar]
NCT02722447 {published data only}
- NCT02722447. Rivaroxaban for the treatment of symptomatic Isolated distal deep vein thrombosis (RIDTS). clinicaltrials.gov/ct2/show/NCT02722447 (first received 30 March 2016).
NCT03368313 {published data only}
- NCT03368313. Non‐elastic compression garment therapy versus control for deep vein thrombosis. clinicaltrials.gov/ct2/show/NCT03368313 (first received 11 December 2017).
NCT03590743 {published data only}
- NCT03590743. Calf deep vein thrombosis treatment trial. clinicaltrials.gov/ct2/show/NCT03590743 (first received 18 July 2018).
UMIN000028105 {published data only}
- UMIN000028105. Evaluation of rivaroxaban for distal deep vein thrombosis – a single center, randomized, open‐label, assessor‐blind, parallel group, exploratory study. upload.umin.ac.jp/cgi‐open‐bin/ctr_e/ctr_view.cgi?recptno=R000032126 (first received 6 July 2017).
Additional references
Anderson 2003
- Anderson FA, Spencer FA. Risk factors for venous thromboembolism. Circulation 2003;107(23 Suppl 1):I‐9. [DOI] [PubMed] [Google Scholar]
Atkins 2004
- Atkins D, Best D, Briss PA, Eccles M, Falck‐Ytter Y, Flottorp S, et al. Grading quality of evidence and strength of recommendations. BMJ 2004;328(7454):1490‐4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Brandao 2017
- Brandao GM, Junqueira DR, Rollo HA, Sobreira ML. Pentasaccharides for the treatment of deep vein thrombosis. Cochrane Database of Systematic Reviews 2017, Issue 12. [DOI: 10.1002/14651858.CD011782.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Cushman 2007
- Cushman M. Epidemiology and risk factors for venous thrombosis. Seminars in Hematology 2007;44(2):62‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Di Nisio 2016
- Nisio M, Ageno W, Rutjes AW, Pap AF, Büller HR. Risk of major bleeding in patients with venous thromboembolism treated with rivaroxaban or with heparin and vitamin K antagonists. Thrombosis and Haemostasis 2016;115(2):424‐32. [DOI: ] [DOI] [PubMed] [Google Scholar]
Feinberg 2017
- Feinberg J, Nielsen EE, Jakobsen JC. Thrombolysis for acute upper extremity deep vein thrombosis. Cochrane Database of Systematic Reviews 2017, Issue 12. [DOI: 10.1002/14651858.CD012175.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Fleck 2017
- Fleck D, Albadawi H, Wallace A, Knuttinen G, Naidu S, Oklu R. Below‐knee deep vein thrombosis (DVT): diagnostic and treatment patterns. Cardiovascular Diagnosis and Therapy 2017;7(Suppl 3):S134‐9. [DOI: 10.21037/cdt.2017.11.03] [DOI] [PMC free article] [PubMed] [Google Scholar]
Franco 2017
- Franco L, Giustozzi M, Agnelli G, Becattini C. Anticoagulation in patients with isolated distal deep vein thrombosis: a meta‐analysis. Journal of Thrombosis and Haemostasis 2017;15(6):1142‐54. [DOI] [PubMed] [Google Scholar]
Galanaud 2014
- Galanaud JP, Sevestre MA, Genty C, Kahn SR, Pernod G, Rolland C, et al. Incidence and predictors of venous thromboembolism recurrence after a first isolated distal deep vein thrombosis. Journal of Thrombosis and Haemostasis 2014;12(4):436‐43. [DOI: ] [DOI] [PubMed] [Google Scholar]
Galanaud 2018
- Galanaud JP, Righini M, Collen L, Douillard A, Robert‐Ebadi H, Pontal D, et al. Long‐term risk of post‐thrombotic syndrome after symptomatic distal deep‐vein thrombosis: the CACTUS‐PTS study. Research and Practice in Thrombosis and Haemostasis 2018:rth2.12125. [DOI: 10.1002/rth2.12125] [DOI] [PubMed]
GRADEpro GDT 2015 [Computer program]
- McMaster University (developed by Evidence Prime). GRADEpro GDT. Version accessed 30 January 2019. Hamilton, ON: McMaster University (developed by Evidence Prime), 2015.
Heit 2015
- Heit JA. Epidemiology of venous thromboembolism. Nature Reviews Cardiology 2015;12:464‐74. [DOI: 10.1038/nrcardio.2015.83] [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration. Available from handbook.cochrane.org.
Kahn 2014
- Kahn SR, Shapiro S, Wells PS, Rodger MA, Kovacs MJ, Anderson DR, et al. Compression stockings to prevent post‐thrombotic syndrome: a randomised placebo‐controlled trial. Lancet 2014;383(9920):880‐8. [DOI] [PubMed] [Google Scholar]
Kearon 2012
- Kearon C, Akl EA, Comerota AJ, Prandoni P, Bounameaux H, Goldhaber SZ, et al. Antithrombotic therapy for VTE disease: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence‐based clinical practice guidelines. Chest 2012;141 (2 Suppl):e419S‐e496S. [DOI: 10.1378/chest.11-2301] [DOI] [PMC free article] [PubMed] [Google Scholar]
Kearon 2016
- Kearon C, Akl EA, Ornelas J, Blaivas A, Jimenez D, Bounameaux H, et al. Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. Chest 2016;149(2):315‐52. [DOI] [PubMed] [Google Scholar]
Kesieme 2011
- Kesieme E, Kesieme C, Jebbin N, Irekpita E, Dongo A. Deep vein thrombosis: a clinical review. Journal of Blood Medicine 2011;2:59‐69. [DOI: 10.2147/JBM.S19009] [DOI] [PMC free article] [PubMed] [Google Scholar]
Lefebvre 2011
- Lefebvre C, Manheimer E, Glanville J. Chapter 6: Searching for studies. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Liberati 2009
- Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta‐analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Medicine 2009;6:e1000100. [DOI] [PMC free article] [PubMed] [Google Scholar]
Lim 2017
- Lim MS, Ariyarajah A, Oldmeadow C, Hall A, Enjeti AK. A systematic review and meta‐analysis comparing anticoagulation versus no anticoagulation and shorter versus longer duration of anticoagulation for treatment of isolated distal deep vein thrombosis. Seminars in Thrombosis and Hemostasis 2017;43(8):836‐48. [DOI: 10.1055/s-0037-1604085] [DOI] [PubMed] [Google Scholar]
McKenzie 2019
- McKenzie JE, Brennan SE. Chapter 12: Synthesizing and presenting findings using other methods. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al, editor(s). Cochrane Handbook for Systematic Reviews of Interventions version 6.0 (updated July 2019). Cochrane, 2019. Available from training.cochrane.org/handbook.
Middeldorp 2014
- Middeldorp S, Prins MH, Hutten BA. Duration of treatment with vitamin K antagonists in symptomatic venous thromboembolism. Cochrane Database of Systematic Reviews 2014, Issue 8. [DOI: 10.1002/14651858.CD001367.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
NICE 2012
- National Institute for Health and Care Excellence. Venous thromboembolic diseases: the management of venous thromboembolic diseases and the role of thrombophilia testing. nice.org.uk/guidance/cg144 (accessed September 2015).
NICE 2018
- National Institute for Health and Care Excellence. Venous thromboembolism in over 16s: reducing the risk of hospital‐acquired deep vein thrombosis or pulmonary embolism (NG89). nice.org.uk/guidance/ng89 (accessed August 2018). [PubMed]
Nicolaides 2013
- Nicolaides AN, Fareed J, Kakkar AK, Comerota AJ, Goldhaber SZ, Hull R, et al. Prevention and treatment of venous thromboembolism: international consensus statement. International Angiology 2013;32(2):111‐260. [PubMed] [Google Scholar]
Review Manager 2014 [Computer program]
- Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Righini 2019
- Righini M, Robert‐Ebadi H, Glauser F, Blondon M, Ouvry P, Diamand JM, et al. Effect of anticoagulant treatment on pain in distal deep vein thrombosis: an ancillary analysis from the cactus trial. Journal of Thrombosis and Haemostasis 2019;17(3):507‐10. [DOI: 10.1111/jth.14387] [DOI] [PubMed] [Google Scholar]
Robertson 2015
- Robertson L, Kesteven P, McCaslin JE. Oral direct thrombin inhibitors or oral factor Xa inhibitors for the treatment of deep vein thrombosis. Cochrane Database of Systematic Reviews 2015, Issue 6. [DOI: 10.1002/14651858.CD010956.pub2] [DOI] [PubMed] [Google Scholar]
Robertson 2017
- Robertson L, Strachan J. Subcutaneous unfractionated heparin for the initial treatment of venous thromboembolism. Cochrane Database of Systematic Reviews 2017, Issue 2. [DOI: 10.1002/14651858.CD006771.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Streiff 2016
- Streiff MB, Agnelli G, Connors JM, Crowther M, Eichinger S, Lopes R. Guidance for the treatment of deep vein thrombosis and pulmonary embolism. Journal of Thrombosis and Thrombolysis 2016;41(1):32‐67. [DOI: 10.1007/s11239-015-1317-0] [DOI] [PMC free article] [PubMed] [Google Scholar]
Watson 2016
- Watson L, Broderick C, Armon MP. Thrombolysis for acute deep vein thrombosis. Cochrane Database of Systematic Reviews 2016, Issue 11. [DOI: 10.1002/14651858.CD002783.pub4] [DOI] [PMC free article] [PubMed] [Google Scholar]
White 2003
- White RH. The epidemiology of venous thromboembolism. Circulation 2003;107(23 Suppl 1):I‐4‐I‐8. [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
Kirkilesis 2019
- Kirkilesis G, Kakkos SK, Bicknell C, Salim S, Kakavia K. Treatment of distal deep vein thrombosis. Cochrane Database of Systematic Reviews 2019, Issue 9. [DOI: 10.1002/14651858.CD013422] [DOI] [PMC free article] [PubMed] [Google Scholar]


