Abstract
Bioactive coatings which support the adhesion of late-outgrowth peripheral blood endothelial progenitor cells (EOCs) are actively being investigated as a means to promote rapid endothelialization of “off-the-shelf,” small-caliber arterial graft prostheses following implantation. In the present work, we evaluated the behavior of EOCs on thromboresistant graft coatings based on the collagen-mimetic protein Scl2-2 and poly(ethylene glycol) (PEG) diacrylate. Specifically, the attachment, proliferation, migration, and phenotype of EOCs on PEG-Scl2-2 hydrogels were evaluated as a function of Scl2-2 concentration (4, 8, and 12 mg/mL) relative to human umbilical vein endothelial cells (HUVECs). Results demonstrate the ability of each PEG-Scl2-2 hydrogel formulation to support EOC and HUVEC adhesion, proliferation, and spreading. However, only the 8 and 12 mg/mL PEG-Scl2-2 hydrogels were able to support stable EOC and HUVEC confluence. These PEG-Scl2-2 formulations were therefore selected for evaluation of EOC and HUVEC phenotype relative to PEG-collagen hydrogels. Cumulatively, both gene and protein level data indicated that 8 mg/mL PEG-Scl2-2 hydrogels supported similar or improved levels of EOC maturation relative to PEG-collagen controls based on evaluation of CD34, VEGFR2, PECAM-1, and VE-Cadherin. The 8 mg/mL PEG-Scl2-2 hydrogels also appeared to support similar or improved levels of EOC homeostatic marker expression relative to PEG-collagen hydrogels based on von Willebrand factor, collagen IV, NOS3, thrombomodulin, and E-selectin assessment. Combined, the present results indicate that PEG-Scl2-2 hydrogels warrant further investigation as “off-the-shelf” graft coatings.
Keywords: endothelial progenitor cells, cell adhesion, migration, phenotype, endothelialization, vascular grafts, coatings
1. INTRODUCTION
In the United States alone, approximately one in three adults suffer from at least one type of cardiovascular disease (CVD).1,2 In most patients, the onset and subsequent progression of CVD is associated with plaque formation within small-caliber arteries with inner diameters less than 6 mm.3 Diseased small-caliber vessels are primarily treated using autologous artery or vein grafts.3,4 However, in over 10% of patients, suitable autologous vessels are not available due to trauma, disease, or previous surgeries.4 In addition, grafts fabricated from synthetic materials generally have high, long-term failure rates in small-diameter applications, due in part to associated platelet activation and intimal hyperplasia.5,6 As such, post-implantation endothelialization of the graft lumen is believed to be critical to the longer-term success of “off the shelf”, small-caliber prostheses.7 However, the extent of graft endothelialization following implantation in vascular patients is generally limited to ~10 mm from each graft-host anastomosis, leaving substantial portions of the graft surface without an endothelial lining.8 These results in human patients are in in contrast to often extensive graft endothelialization observed in animal models. Therefore, a significant need exists for approaches which support the endothelialization of small-caliber arterial prostheses, particularly for grafts intended for emergency or “off-the-shelf” applications.
Graft endothelialization requires a source of autologous endothelial cells (ECs) or endothelial progenitor cells (EPCs) that can be obtained at high purity and at high yields – ideally through minimally invasive procedures. Excised jugular or saphenous vein9 or liposuctioned fat10 are currently the most common sources of autologous ECs. However, venous tissue harvest can be quite invasive and ECs derived from fat are often contaminated with undesired cell types.11 In contrast, endothelial progenitor cells (EPCs) can be obtained from an essentially non-invasive procedure in which peripheral blood mononuclear cells (MNCs) are harvested and then stimulated to differentiate into EC-like cells ex vivo. Distinct EPC subpopulations can be obtained from MNCs depending on the method of ex vivo culture.12–17 Specifically, “early outgrowth” EPCs can be sorted by flow cytometry from MNC fractions and obtained at relatively high levels.18–22 These isolated EPCs are generally specified as CD34+/CD133+/VEGFR2+, display several EC markers, and produce high levels of angiogenic factors.23 Unfortunately, “early outgrowth” EPCs have limited capacity to proliferate and also display markers and phagocytic capacities typical of macrophages, characteristics which are generally considered undesirable for endothelial applications.24
In contrast, “late outgrowth” EPCs are confined to the CD34+/CD133−/CD146+ MNC fraction25 and are significantly more scarce than “early outgrowth” EPCs.26,27 That said, “late outgrowth” EPCs (hereafter referred to as EOCs) can be expanded ex vivo at high yields,13,14 and EOCs have been demonstrated to endothelialize denuded vessels when injected in vivo in a rabbit model.28 EOCs also express normal EC surface markers,13,14 exhibit typical EC function,29 but do not express monocytic character.30 In fact, EOCs have been shown to display EC and integrin marker profiles similar to that of human umbilical vein endothelial cells (HUVECs),25,31,32 an EC type that has been widely examined for vascular graft applications.25,31,32 In addition, recent work indicates that EOCs isolated from patients with significant coronary artery disease (CAD) exhibit similar expansion capacity, adhesion, and angiogenic potential as EOCs from healthy individuals.33 Thus, blood could potentially be drawn from a CAD patient receiving an “off-the-shelf” vascular graft, and patient-specific EOCs could then be isolated, expanded, and injected in vivo to populate the implanted graft.
In vascular graft designs focused on EOC-based endothelialization, the graft lumen is generally coated with extracellular matrix (ECM) molecules and/or antibodies to EOC surface markers in order to enhance EOC adhesion to the luminal surface.34–43 In the case of ECM-based coatings, collagen,44 fibronectin43 and fibrin39 coatings have each been employed due to the relatively high expression levels of α5, αv, and α2 integrin subunits on EOC surfaces.25,33,45 However, collagen, fibronectin, and fibrin – and certain derivative peptides (eg. RGD) – also support platelet aggregation.46–49 An ideal vascular graft coating would intrinsically resist platelet aggregation and activation while promoting rapid EOC adhesion, proliferation, confluence, and phenotypic maturation.
Recently, a biomaterial platform has been developed based on the collagen-mimetic protein Scl2-2.50,51 Scl2-2 proteins contain the GXY-repeats characteristic of native collagen and spontaneously assemble into stable triple-helical structures of ~120 kDa.52 However, in contrast to collagen, Scl2-2 resists significant platelet aggregation52 and contains only a single known cell adhesion motif — GFPGER.52,53 The GF/LPGER adhesion motif is a structural analog of GF/LOGER (O; hydroxyproline), an adhesion site in native collagen which binds α1β1 and α2β1 integrins,9 with GLPGER having previously been shown to bind α2β1 integrins when presented in a triple helical context.53 Furthermore, although native collagen (which contains many cell binding sites in addition to GF/LOGER) can induce thrombosis, platelet interaction with the GF/LOGER site alone is not sufficient to initiate platelet activation.54–56 Consistent with these cumulative observations, recent studies of GFPGER within the triple helical context of Scl2-2 indicate that Scl2-2 supports cell adhesion via engagement of α1 and α2 integrin subunits57 but does not significantly stimulate platelet aggregation.52
As opposed to native collagen, Scl2-2 does not require post-translational modification to assemble into physiologically-stable triple helical structures and therefore can be readily expressed recombinantly in bacterial systems.52 The capacity for bacterial expression also allows significant potential for cost effective scale-up of Scl2-2 production. This is in contrast to the relatively high scale-up costs associated with solid phase synthesis of the lengthy GFOGER-containing peptides (~37 mers) capable of taking on the stable triple helical structures required for full integrin engagement.58–60 The recent conjugation of acrylate-functionalized Scl2-2 proteins into poly(ethylene glycol) (PEG) diacrylate hydrogels — which intrinsically resist protein adsorption and cell adhesion — has extended the potential biomedical applications Scl2-2 proteins.57,61,62 The functionalized Scl2-2 proteins maintain a triple helical structure and confer selective bioactivity to the resulting PEG-Scl2-2 hydrogels.57 In addition, PEG-Scl2-2 hydrogels have been shown to maintain the resistance to the platelet adhesion and activation characteristic of pure PEG hydrogels61 and the resistance to platelet aggregation associated with pure Scl2-2 proteins.52 Furthermore, previous studies have reported that the parent Scl2 protein is cytocompatible and does not elicit an immune response in SJL/J and Arc mice.1 Given the relatively high expression of α2β1 integrin by EOCs25,33,45 and the need for thromboresistance in vascular graft coatings, Scl2-2 proteins may hold promise for use in small-caliber graft applications.
The primary goal of the current study was to evaluate the capacity of PEG-Scl2-2 hydrogel coatings to support desired EOC behaviors – namely cell adhesion, migration, and phenotypic maturation.63 Three Scl2-2 concentrations — 4, 8, and 12 mg/mL — were selected for examination based on previous studies with bovine aortic endothelial cells.57,61,62 PEG hydrogels containing 4 mg/mL of collagen I — which is roughly equivalent to 4 mg/mL Scl2-2 in terms of integrin α1/α2-I domain binding affinity52 — were analyzed as internal controls. Collagen I was selected as a control over collagen IV, which also contains the GF/LOGER motif, to allow improved comparison with previous Scl2-2 studies.54,56,57 Results were compared against HUVECs due to the phenotypic similarity between EOCs and HUVECs noted in previous studies25,31,32 and due to the widespread use of HUVECs in vascular graft literature.64–68
2. MATERIALS AND METHODS
2.1. Polymer Synthesis and Scl2-2 Expression and Functionalization
2.1.1. PEG diacrylate (PEGDA) synthesis
Twenty four grams of 3.4 kDa PEG diol (Fluka) dissolved in anhydrous dichloromethane were reacted with acryloyl choride using a 1:4 molar ratio in the presence of trimethylamine (1:2 molar ratio).69 The reaction was carried out at 4 °C for 12 h under argon. To remove hydrochloric acid byproduct, the reaction mixture was washed with 2 M K2CO3 and separated into aqueous and organic phases. The lower phase (organic) was then dried with anhydrous MgSO4 and precipitated in ice cold diethyl ether. The resulting slurry was then filtered to retrieve the PEGDA. The conversion of the terminal hydrogel groups to acrylate groups was confirmed by proton nuclear magnetic resonance to be > 95%.
2.1.2. Expression, purification and characterization of Scl2-2 protein
The E. coli BL21 (Novagen) strain was used to express the recombinant Scl2-2 protein.52 After cell lysis, the released protein was separated from the lysate by affinity chromatography using a HisTrap HP column followed by a HiTrap Q column (GE Healthcare).52 Scl2-2 purity was qualitatively confirmed by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE) and Coomassie Blue staining.57 Final purification of the Scl2-2 protein was achieved by dialysis against double deionized water for 48 h. The final purified product was then lyophilized and stored at -80 °C until use.
2.1.3. Acrylate-derivatization of the Scl2-2 protein
In order to covalently link the Scl2-2 protein into the structure of PEGDA hydrogels, the Scl2-2 protein was acrylate-derivatized using PEG-succinimidyl valerate (ACRL-PEG-SVA, 3.4 kDa; Laysan Bio). Briefly, Scl2-2 trimer and ACRL-PEG-SVA were reacted at a molar ratio of 1:6 for 2 h in phosphate buffered saline (PBS, pH 7.4; Life Technologies). Immediately following the derivation reaction, the resulting ACRL-PEG-Scl2-2 product was used for the fabrication of the PEG-Scl2-2 hydrogels. As a control, rat tail collagen I (Life Technologies) was also reacted with ACRL-PEG-SVA for 2 h at a 1:6 molar ratio in 50 mM sodium bicarbonate buffer, pH 8.5.70 The resulting products were then dialyzed against double deionized water for 48 h. Following purification, the acrylate-derivatized collagen was lyophilized and stored at -80 ºC until use. The incorporation of acrylate groups within the Scl2-2 and collagen I was confirmed by Fourier Transform Infrared Spectroscopy following previously described methods.57 Both acrylate-derivatized and unmodified Scl2-2 were also analyzed by circular dichroism as previously described.57 Results from the circular dichroism analyses are shown in Supplementary Figure 1.
2.2. Fabrication of PEG-Scl2-2 and PEG-collagen (PEG-coll) Hydrogels
PEG-Scl2-2 hydrogels were prepared to achieve a final PEGDA concentration of 10% (w/w) and three distinct levels of acrylate-derivatized Scl2-2 protein: 4 mg/mL, 8 mg/mL and 12 mg/mL. An additional control formulation comprised of 10% (w/w) PEGDA and 4 mg/mL acrylate-derivatized collagen I in 20 mM acetic acid was used as a positive control. Hydrogels were cured in 0.5 mm thick rectangular glass molds by exposure for 6 min to longwave UV radiation (~6 mW/cm2, 365 nm) in the presence of 2.6 mg/mL Irgacure 2959 (Sigma).71 Resulting hydrogels were sterilized by immersion in 70% (v/v) ethanol for 1 h and subsequently washed with a series of graded ethanol solutions for 20 min each (70%, 50%, 20% and 0% in PBS). The hydrogels were then immersed in PBS containing 1% PSA (PSA: 10,000 U/mL penicillin, 10,000 mg/L streptomycin and 25 mg/L amphotericin; Mediatech) for 12 h at room temperature. Unifrom 8 mm discs were cored from the hydrogel slabs and were placed in PBS within separate wells of a 48-well plate for seeding with EOCs or HUVECs.
2.3. EOC and HUVEC Culture
Cryopreserved HUVECs were obtained from Lonza, Inc., whereas EOCs were obtained through a protocol approved by the Duke University Institutional Review Board. In brief, blood was collected from a consenting 60 year old female patient with advanced CAD who was undergoing left heart catheterization at Duke University Medical Center. Late-outgrowth EOCs were isolated and expanded from the blood as previously described27,33 and were characterized by flow cytometry as positive for CD31 (PECAM-1) and CD105 and negative for CD133, CD14, and CD45.33 The EOCs derived from this CAD patient have previously been shown to have similar proliferative capacity, surface antigen expression, and function as EOCs derived from healthy patients.33
Both EOCs and HUVECs were maintained in EBM-2 medium (Lonza, Inc.) supplemented with EGM-2 SingleQuots, 10% fetal bovine serum (FBS; Altanta Biologicals), and 1% PSA (Mediatech). HUVECs were used at passage 5–7 for all experiments. EOCs were proliferative and exhibited healthy morphology through passage 10.33 However, the EOCs were utilized between passage 7–9 in the present studies.
2.4. Flow Cytometry Analysis of Integrin Subunits
Cell expression of the integrin subunits α1 and α2 was assessed by flow cytometry using the Agilent 2100 Bioanalyzer microfluidic system. Briefly, EOCs or HUVECs were resuspended at 200,000 cells per mL, placed on ice, and exposed to 2 μM Calcein AM (Life Technologies) in staining buffer (1% BSA in PBS) for 30 min. The cells were then pelleted by centrifugation and washed twice with staining buffer, after which they were resuspended in 100 μL staining buffer containing 10 μg/mL of appropriate primary antibody (integrin α1: clone FB12, integrin α2: clone P1E6; Millipore), or IgG control. After incubation for 30 min, the cells were centrifuged and the pellet was rinsed twice with 1 mL of staining buffer. Secondary antibody (Alexafluor 647 conjugated donkey anti-mouse IgG; Jackson Immunoresearch) was then applied at 10 μg/mL in staining buffer for 30 min. Finally, the cell pellet was rinsed twice with 1 mL staining buffer, resuspended at 1x106 cells/mL in loading buffer, and then applied to the flow cytometry microfluidic chip (Agilent 2100 Bioanalyzer) according to the manufacturer’s instructions. For each antibody and cell type, flow assessment was run in triplicate. Flow cytometry for the β1 integrin subunit was also conducted and is shown in Supplementary Figure 2.
2.5. Assessment of Adherent Cell Number and Surface Coverage
EOCs and HUVECs were seeded at 5,000 cells/cm2 onto the surfaces of the various hydrogel discs. After 24 h and 72 h post-seeding, hydrogel discs were gently rinsed with PBS and fixed with a 10% formalin solution for 30 min. The discs were then exposed to DAPI (Life Technologies) and rhodamine-phalloidin (Life Technologies) to allow visualization of the nuclei and cytoskeletal F-actin filaments of adherent cells. A total of 10 randomly selected regions in 6 distinct hydrogel discs were them imaged for each hydrogel formulation using a Zeiss Axiovert 200M fluorescence microscope and a 10X objective. All images were obtained using consistent microscope, lamp, and camera settings, including light intensity and exposure time.
To quantify the number of adherent cells (number of cells/mm2), the total number of cell nuclei stained with DAPI in each image field was manually counted and then normalized to the area of the imaged field. To quantify the fraction of the imaged area covered by cells, each rhodamine channel image was first loaded into Image J software as an 8-bit digital grayscale image. A threshold on gray levels was then selected in order to separate lighter-appearing cells from the darker background. In this process, pixels with grayscale values above the selected threshold were assigned a new grayscale value of “255” (white), and pixels with grayscale values below this threshold were assigned a new grayscale value of “0” (black). A median filter was then applied to the modified images to remove non-cell debris appearing as “white” following thresholding. Remaining white “non-cell” regions were removed manually using the ImageJ “Paintbrush” tool. “Fraction area coverage” was subsequently calculated for each image as the total white area divided by the total imaged area.
2.6. Cell Migration on Hydrogels
To assess cell migration rate, EOCs or HUVECs were seeded at 5,000 cells/cm2 and were allowed to attach and spread for 24 h. For each hydrogel formulation, cell migration was then assessed by monitoring the position of 110–290 cells from at least three randomly-selected regions of 6 different hydrogel discs for 55 min at 5-min intervals. The Manual Tracking Plug-In in Image J was utilized to track the movement of individual cell centroids within each acquired image series. Monitored cells maintained at least 100 μm distance from surrounding cells throughout the image series. Positional information was used to calculate the mean-square displacement (MSD, <D2>) of each tracked cell at each 5-min time increment, i, over the monitoring period, t:
where N is the total number of 5-min time intervals within the time t, and di is the square of cell displacement during time increment, i.72 The cell speed, S, and persistence time, P, were then determined by fitting the MSD (<D2>) and the time interval, t, to the persistent random walk equation using nonlinear least squares regression analysis:73,74
2.7. Cell Phenotype at Confluence on Hydrogels
For characterization of cell phenotype, EOCs or HUVECs were seeded at 10,000 cell/cm2 on hydrogels containing 4, 8, or 12 mg/mL Scl2-2. Twenty four hours following confluence (defined herein as greater than 90% cell surface coverage), adherent cells were lysed for gene expression and protein analyses relative to 4 mg/mL PEG-collagen (PEG-58coll) controls.
2.7.1. Extraction of mRNA and gene expression analyses
mRNA was extracted from confluent cell layers using the Dynabeads mRNA direct kit (Life Technologies).71 Briefly, the cell culture media was carefully removed from the samples. Following media removal, the hydrogels discs were rinsed with 200 μL of PBS for 5 min and then incubated with 330 μL of lysis buffer (provided with the kit) at room temperature for 10 min. PolyA-mRNA in the lysis buffer was subsequently harvested using 20 μL of oligo(dT)25 magnetic beads. Following rinsing steps, the polyA-mRNA was released from the beads in 100 μL of a 10 mM Tris-HCl solution by heating the beads to 80 ºC for 2 min. The polyA-mRNA was stored at -80 ºC until the time of analysis.
Gene expression analyses were performed using a StepOne real-time PCR system (Life Technologies) and the SuperScript III Platinum One-Step qRT-PCR kit (Life Technologies). Verified qPCR primers (OriGene) targeting mRNA encoding for human CD34, VEGR2, EphrinB2, EphB4, VE-Cadherin, von Willebrand factor (vWF), NOS3, and RPL-32 were utilized (Table 1). Approximately 16 ng of polyA-mRNA and 5 μL of a 1 μM primer solution were combined into a total reaction volume of 25 μL. Forty amplification cycles were monitored using SYBR Green as a reporter and ROX dye as a passive reference. A threshold value for the fluorescence signal was identified in the exponential phase of amplification using the StepOne software v2.0. The fractional cycle value at the intersection between the amplification curve for a particular gene product and this threshold line was recorded as the Ct (threshold cycle) of that gene. Melting temperature analysis was performed to verify the desired product. To calculate the relative expression of the genes of interest, the ΔΔCt method was employed using utilizing RPL-32 as the reference gene and the HUVEC PEG-coll hydrogels as the reference sample.75
Table 1.
Genes assessed in evaluating shifts in EOC and HUVEC phenotype.
| Function | Gene Marker | Primer sequence Forward 5′-3 (F), Reverse 5′-3′ (R) |
|---|---|---|
| Reference Gene | RPL-32 | F: ACAAAGCACATGCTGCCCAGTG R: GCTCGCAGTACGACTACACTGAC |
| Late Outgrowth Endothelial Progenitor Cell Markers | CD34 | F: AAGGCTGGGTGAAGACCCTTA R: TGAATGGCCGTTCTGGAAGT |
| VEGFR2 | F: GGAACCTCACTATCCGCAGAGT R: CCAAGTTCGTCTTTTCCTGGGC |
|
| Arterial/Venous Endothelial Cell Markers | EphrinB2 | F: GCAAGTTCTGCTGGATCAACCAG R: GCTGTTGCCGTCTGTGCTAGAA |
| EphB4 | F: GAAAAGGAAGTGCCCAACA R: CTGGCAAGGGAGTCACACT |
|
| Mature Endothelial Cell Marker | VE-Cadherin | F: GAAGCCTCTGATTGGCACAGTG R: TTTTGTGACTCGGAAGAACTGGC |
| Regulators of Vascular Homeostasis | vWF | F: CCTTGAATCCCAGTGACCCTGA R: GGTTCCGAGATGTCCTCCACAT |
| NOS3 | Proprietary (Qiagen) |
2.7.2. Quantification of protein production
Based on the gene expression results, a subset of hydrogel formulations were further analyzed by western blot or protein multiplex assay. Specifically, cell phenotype was assessed by measuring the protein levels of PECAM-1, thrombomodulin (TM), E-Selectin, and Collagen IV (Col IV) (Table 2). For each target protein analyzed by western blot, an amount of cell lysate representing a similar number of cells (~150 ng DNA as measured by the Quant-iT™ PicoGreen® dsDNA Reagent (Life Technologies)) was utilized. Samples were first concentrated using a 3,000 MWCO Amicon filter units (Millipore) and then denatured by the addition of β-mercaptoethanol and sample heating at 95 °C for 10 min. The concentrated protein lysates were loaded into the wells of a 10% polyacrylamide gel and separated via SDS-PAGE. After electrophoresis, proteins were transferred to a nitrocellulose membrane (Thermo Scientific), washed twice with double distilled water and blocked with a solution of 5% bovine serum albumin (BSA; Fisher Scientific) in TBST (25 mM Tris-HCl, pH 7.5, 137 mM NaCl, 0.1% Tween 20) for 1 h at room temperature. Following blocking, the membranes were incubated for 12 h at 4 °C with primary antibodies for PECAM-1 (clone EP3095; Abcam), TM (clone H-11; Santa Cruz Biotechnology), or E-selectin (clone D7; Santa Cruz Biotechnology) diluted in TBST containing 5% BSA and 0.05% NaN3. Corresponding horse radish peroxidase (HRP)-conjugated or alkaline phosphatase (AP)-conjugated secondary antibody (Jackson Immunoresearch) diluted in TBST solution containing 5% BSA was applied to the membrane for 1 h at room temperature. Bound secondary antibody was detected using Luminol reagent (Santa Cruz Biotechnology) or AP chemiluminescent solution (Invitrogen). The signal intensity of the reporting molecule was monitored using a Chemidoc XRS system (Biorad). The integrated optical density of each target band was measured using Adobe Photoshop and subsequently normalized to sample DNA content.
Table 2.
Proteins assessed in evaluating shifts in EOC and HUVEC phenotype.
| Function | Protein Marker |
|---|---|
| Endothelial Cell Marker | PECAM-1 |
| Mature Endothelial Cell Marker | Col IV |
| Regulators of Vascular Homeostasis | TM |
| E-selectin |
The protein levels of Col IV were measured from the cell lysates using a human premixed magnetic bead analyte kit (R&D Systems) and a MAGPIX detection system (Luminex). Briefly, samples were loaded into a 96-well plate, after which magnetic bead suspensions, detection antibodies, and streptavidin-phycoerythrin were added to sample wells. Sample concentrations of Col IV were obtained on the basis of a mean fluorescence intensity (MFI) relative to a standard curve. The resulting measures were then normalized by sample DNA content.
2.8. Statistical Analyses
A two-factor analysis of variance (ANOVA) was utilized to detect significant differences caused by the following main effects: cell type (EOC and HUVEC) and formulation (4, 8, and 12 mg/mL PEG-Scl2-2 and 4 mg/mL PEG-coll hydrogels). For comparisons among more than two formulations, significant differences were detected utilizing a Tukey’s post-hoc test. In cases of a significant interaction between factors (cell type and formulation), differences among formulations were determined with a one way ANOVA with a Tukey post-hoc for each cell type separately. In these instances, differences were not claimed between EOC and HUVECs. All analysis was done with SPSS software with p < 0.050 considered significant. All data are reported as average ± standard error of the mean with a sample size of at least 3–4 independent specimens.
3. RESULTS
In the following study, EOC adhesion, proliferation, migration, and phenotype on PEG-Scl2-2 hydrogels containing varying concentrations of Scl2-2 were assessed relative to PEG-coll hydrogels, and results were compared against corresponding data for HUVECs.
3.1. Integrin Subunit Expression
As previously noted, Scl2-2 proteins support interactions with integrin α1 and α2 subunits.52,57 Thus, EOCs and HUVECs were assessed for their expression of integrin α1 and α2 via flow cytometry. Figure 1 shows the percentage of cells with positive staining for a particular integrin subunit relative to their respective negative controls. Consistent with previous literature,45 ~98% of EOCs and HUVECs expressed the integrin subunit α2. In contrast, only 7–9% of EOCs and HUVECs expressed the α1 integrin subunit. This lower level of integrin α1 expression by EOCs and HUVECs relative to integrin α2 is also in agreement with previous literature.25,31,32,37 These data – coupled with the fact that pure PEG-based hydrogels do not enable significant cell attachment in the absence of specifically-incorporated adhesion ligands61 – indicate that initial EOC and HUVEC attachment to the PEG-Scl2-2 hydrogels may be mediated primarily via integrin subunit α2.
Figure 1.
Integrin α1 and α2 subunit assessment by flow cytometry for EOCs and HUVECs. Gray filled curves represent negative controls. Black lined curves represent test samples.
3.2. Number of Adherent Cells and Extent of Surface Coverage
EOCs and HUVECs were seeded onto PEG-Scl2-2 hydrogel surfaces at 5,000 cells/cm2, and the number of adherent cells and their surface coverage were analyzed at 24 h and 72 h of culture. Representative images of EOCs and HUVECs on PEG-Scl2-2 hydrogels are shown in Figure 2 and Supplementary Figure 3 (at lower magnification) to allow for qualitative comparison of adherent cell number across hydrogel formulations. Quantitative assessment at 24 h indicated that the average adherent cell number on 8 mg/mL PEG-Scl2-2 hydrogels was ~30% greater than on 4 mg/mL PEG-Scl2-2 hydrogels for both EOCs and HUVECs (p < 0.001 and p = 0.044, respectively; Figure 3). For HUVECs, the level of adherent cells at 24 h also increased as Scl2-2 concentration increased from 8 mg/mL to 12 mg/mL (p = 0.021). By 72 h of culture, the number of adherent EOCs and HUVEC on each PEG-Scl2-2 hydrogel had increased significantly from the 24 h adhesion levels (p < 0.001; Figure 3), indicating that Scl2-2 surfaces were able to support stable cell attachment and proliferation. Furthermore, both cell types continued to display a ~30% greater number of adherent cells on 8 mg/mL PEG-Scl2-2 hydrogels relative to 4 mg/mL PEG-Scl2-2 hydrogels at 72 h of culture (EOC: p < 0.001; HUVEC: p < 0.001). However, no increase in the average adherent cell number was observed for either cell type at 72 h when Scl2-2 levels were further increased from 8 mg/mL to 12 mg/mL.
Figure 2.
Representative images of EOCs and HUVECs stained with phalloidin and counterstained with DAPI on PEG-Scl2-2 and PEG-coll hydrogels at 24 h and 72 h. The scale bar in each image series equals 200 μm and applies to all images in the series.
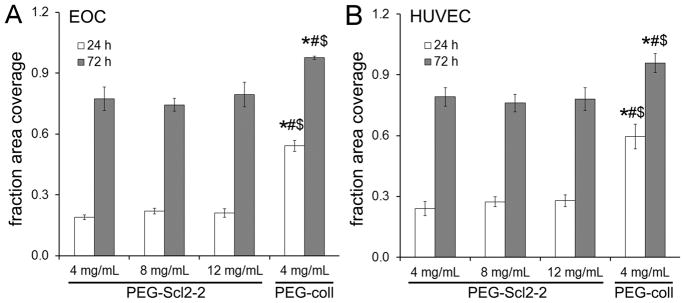
Figure 3.
Quantitative assessment of the surface cell density of (A) EOCs and (B) HUVECs at 24 h and 72 h. Measurements are expressed as mean ± standard error of mean. Results were obtained from n = 6 independent samples per formulation with 10 images per sample. All values at 24 h for a given cell type were significantly different from the corresponding 72 h values. * significantly different from the 4 mg/mL PEG-Scl2-2 hydrogel at the corresponding time point, p < 0.05; # significantly different from the 8 mg/mL PEG-Scl2-2 hydrogel at the corresponding time point, p < 0.05; $ significantly different from the 12 mg/mL PEG-Scl2-2 hydrogel at the corresponding time point, p < 0.05.
Notably, the numbers of adherent EOCs and HUVECs on the PEG-coll controls at both 24 h and 72 h were ~2-fold greater than on the PEG-Scl2-2 hydrogels (p < 0.001; Figure 3). At 24 h of culture, this difference in adherent cell number observed between the PEG-Scl2-2 and PEG-coll surfaces was also reflected in assessments of percent cell surface coverage (Figure 4). Specifically, the levels of EOC and HUVEC surface coverage on the PEG-Scl2-2 hydrogels at 24 h were 2.3- to 2.6-fold lower, respectively, than on the PEG-coll hydrogels (p < 0.001). By 72 h of culture, however, only a 1.3-fold difference in surface coverage was observed between the PEG-Scl2-2 and the PEG-coll hydrogels for either EOCs or HUVECs (Figure 4). In addition, similar levels of fraction area coverage were observed for both cell types irrespective of Scl2-2 concentration at 72 h. This occurred despite the lower adherent cell number present on the 4 mg/mL PEG-Scl2-2 hydrogels relative to 8 mg/mL and 12 mg/mL PEG-Scl2-2 hydrogels. In short, HUVECs and EOCs displayed increased spreading on the lower concentration Scl2-2 hydrogels at 72 h of culture, resulting in similar degrees of surface coverage across Scl2-2 formulations (as can be seen visually in Figure 2).
Figure 4.
Quantitative assessment of the fraction of surface covered by (A) EOCs and (B) HUVECs at 24 h and 72 h of culture. Measurements are expressed as mean ± standard error of the mean. For each hydrogel type, n = 5–6 independent samples were analyzed. All values at 24 h for a given cell type were significantly different from the corresponding 72 h values. * significantly different from the 4 mg/mL PEG-Scl2-2 hydrogel at the corresponding time point, p < 0.05; # significantly different from the 8 mg/mL PEG-Scl2-2 hydrogel at the corresponding time point, p < 0.05; $ significantly different from the 12 mg/mL PEG-Scl2-2 hydrogel at the corresponding time point, p < 0.05.
3.3. Cell Average Surface Migration Speed
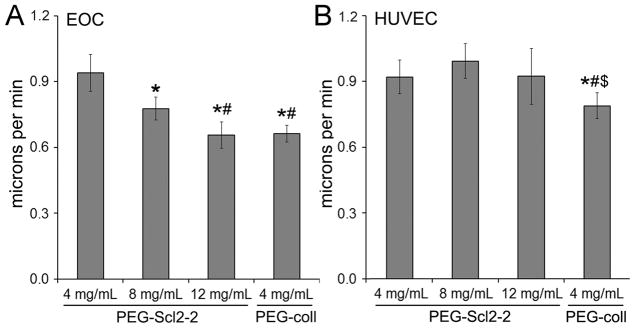
Cell migration over the graft luminal surface is also critical to achieving post-implantation graft endothelialization. Previous studies have shown that the average speed of cell migration on a surface displays a biphasic dependence on the adhesion ligand density presented by that surface.76,77 Specifically, the average cell migration speed generally increases with increasing adhesion ligand density through a maximum, after which further increases in ligand density result in a decrease in cellular migration capacity.76,77 Thus, EOC and HUVEC migration rates on PEG-Scl2-2 hydrogels were analyzed as a function of Scl2-2 concentration with the goal of identifying a PEG-Scl2-2 formulation which supports an appropriate balance of adhesion strength and migration speed (Figure 5). For EOCs, the 4 mg/mL PEG-Scl2-2 hydrogels were associated with the highest average migration speed (~0.94 μm/min) of the formulations assayed. Furthermore, average EOC migration speeds decreased monotonically as Scl2-2 concentration increased from 4 mg/mL to 8 mg/mL to 12 mg/mL (p < 0.001), with the average EOC migration speed on the 12 mg/mL PEG-Scl2-2 hydrogels approaching that observed on PEG-coll controls (~0.66 μm/min). In contrast, the average migration speed of HUVECs on PEG-Scl2-2 hydrogels (~0.92 – 0.99 μm/min) did not display a clear dependence on Scl2-2 concentration. However, each PEG-Scl2-2 hydrogel type supported a higher average HUVEC migration speed than the PEG-coll control (p < 0.042).
Figure 5.
Influence of Scl2-2 concentration on the migration speed of (A) EOCs and (B) HUVECs. For each formulation, n = 110–290 cells were analyzed per cell type. Measurements are expressed as mean ± standard error of mean. * significantly different from the 4 mg/mL PEG-Scl2-2 hydrogel, p < 0.05; # significantly different from the 8 mg/mL PEG-Scl2-2 hydrogel, p < 0.05.
3.4. Cell Confluence and Phenotypic Marker Expression
A successful graft coating not only must support initial EOC attachment, but also must support EC marker expression and the formation of a confluent cell layer. Relative to “early-outgrowth” EPCs, EOCs demonstrate a number of phenotypic similarities to HUVECs – a fully committed EC type and lack monocytic characteristics.25,31,32 Thus, a panel of markers associated with EC phenotypic maturation and activation were examined for confluent EOCs on the various hydrogel surfaces.
3.4.1. Gene expression analyses
To evaluate cell phenotype on the PEG-Scl2-2 surfaces, EOCs and HUVECs were seeded at an initial density of 10,000 cells/cm2. Cells on all surfaces achieved confluence by 72 h. However, confluent EOC and HUVEC layers on the 4 mg/mL PEG-Scl2-2 hydrogels proved to be unstable. Although the HUVEC layers delaminated at a later time-point than the EOC layers, both HUVEC and EOC cell layers delaminated from the 4 mg/mL PEG-Scl2-2 hydrogels within several hours of confluence (data not shown). As a result, only the 8 mg/mL and 12 mg/mL PEG-Scl2-2 formulations were included in the gene expression analyses (Table 1) and were evaluated relative to PEG-coll controls.
At 24 h post-confluence, both EOCs and HUVECs expressed mRNA for CD34, a cell surface glycoprotein associated not only with hematopoietic cells but also with EPCs and committed vascular ECs (Figure 6A).25,31,32,63,78 As anticipated, both HUVECs and EOCs expressed CD34, with EOC CD34 mRNA levels being 10–14 fold greater than for HUVECs (p < 0.001).31 Notably, however, EOC CD34 mRNA levels on PEG-Scl2-2 hydrogels were ~30% lower than on the PEG-coll control (p = 0.033). Gene expression levels of VEGFR2 – the receptor subtype considered to be the primary mediator of the mitogenic and chemotactic effects of VEGF observed in adult ECs79 – were ~70% greater on PEG-Scl2-2 hydrogels relative to PEG-coll controls for both cell types (p < 0.001; Figure 6B). In contrast, mRNA levels for VE-cadherin – a primary EC intercellular junction protein – were similar across cell types and hydrogel formulations (Figure 6D).
Figure 6.
Relative gene expression of the endothelial markers (A) CD34, (B) VEGFR2, (C) EphrinB2:EphB4, (D) VE-Cadherin, (E) vWF, and (F) NOS3 of confluent EOCs and HUVECs cultured on PEG-Scl2-2 hydrogels and PEG-coll controls. For each cell type, n = 3–6 independent samples were analyzed for each hydrogel formulation. Measurements are expressed as mean ± standard error of the mean. * significantly different from the 8 mg/mL PEG-Scl2-2 hydrogel, p < 0.05; # significantly different from the 12 mg/mL PEG-Scl2-2 hydrogel, p < 0.05; + significant difference between cell types.
Given the goal of small-caliber arterial graft endothelialization, the relative expression of EphrinB2 to EphB4 was examined to gain initial insight into the capacity of PEG-Scl2-2 hydrogels to support EOC progression toward an arterial EC phenotype (Figure 6C). In early arteriovenous differentiation, the transmembrane signaling molecule EphrinB2 is specifically expressed in arterial ECs, and its tyrosine kinase receptor EphB4 is specifically expressed in venous ECs prior to the onset of circulation.80,81 A higher ratio of EphrinB2:EphB4 during early EC commitment has thus been correlated with increased progression toward an arterial (versus venous) EC phenotype.82 Although the HUVEC EphrinB2:EphB4 gene expression ratio did not vary with hydrogel formulation, EOCs on the 12 mg/mL PEG-Scl2-2 hydrogels displayed an increased EphrinB2:EphB4 ratio relative to the PEG-coll control (p = 0.005). Furthermore, gene expression of vWF – a blood glycoprotein generally associated with an increased risk of thrombosis – was ~40% lower on both PEG-Scl2-2 hydrogel formulations relative to PEG-coll controls for both EOCs and HUVECs (p < 0.005; Figure 6E).
Collectively, the above data indicate that the examined PEG-Scl2-2 hydrogels may support improved EOC maturation relative to PEG-coll hydrogels, although there appeared to be limited differences between the 8 mg/mL and 12 mg/mL PEG-Scl2-2 hydrogels. When NOS3 gene expression was evaluated, however, differences in the gene expression supported by the Scl2-2 formulations were observed. Notably, HUVEC and EOC mRNA levels for NOS3 – an enzyme that plays a critical role in vascular homeostasis and thromboresistance – were over 50% lower on the 12 mg/mL PEG-Scl2-2 formulation relative to both the 8 mg/mL PEG-Scl2-2 hydrogel and the PEG-coll control (p < 0.005 for all; Figure 6F). These differences in NOS3 expression – coupled with the fact that EOC migration was significantly reduced on the 12 mg/mL relative to the 8 mg/mL PEG-Scl2-2 formulation – led us to focus our final protein level analyses on the 8 mg/mL PEG-Scl2-2 hydrogels and the PEG-coll controls.
3.4.2. Protein level analyses
To further compare EOC and HUVEC phenotypic differences on the 8 mg/mL PEG-Scl2-2 hydrogels relative to PEG-coll controls, the following additional markers were examined at the protein level using either western blotting or magnetic bead multiplexing: PECAM-1 (an endothelial intercellular cell junction protein), Col IV (a basement membrane protein that plays a key role in regulating EC behavior), TM (a protein with anti-coagulant activity expressed by ECs),75,83 and E-selectin (an endothelial-leukocyte adhesion protein expressed following EC activation with proinflammatory cytokines).
For both HUVECs and EOCs, PECAM-1 levels were 1.4-to-2.1 fold higher on 8 mg/mL PEG-Scl2-2 hydrogels relative to PEG-coll controls (p = 0.008; Figure 7). In addition, Col IV deposition by EOCs and HUVECs was 17.8-fold and 7.3-fold greater, respectively, on 8 mg/mL PEG-Scl2-2 hydrogels relative to PEG-coll hydrogels (p < 0.005). In contrast, no differences could be distinguished in the levels of the anti-coagulant protein TM across formulations for either EOCs or HUVECs. For the EC activation marker E-selectin, no difference in EOC E-selectin levels was noted with formulation although the 8 mg/mL PEG-Scl2-2 hydrogel was associated with a ~ 63% reduction in HUVEC E-selectin production (p = 0.013). Together, the protein data suggest that 8 mg/mL PEG-Scl2-2 formulation supports EC maturation while maintaining similar levels of cell quiescence/activation relative to PEG-coll controls. Representative western blot images are shown in Supplementary Figure 4.
Figure 7.
Relative protein expression of the endothelial markers PECAM-1, Col IV, TM, and E-selectin by confluent EOCs and HUVECs cultured on PEG-Scl2-2 hydrogels and PEG-coll controls. For each cell type, n = 3–6 independent samples were analyzed for each hydrogel formulation. Measurements are expressed as mean ± standard error of the mean. * significantly different from the 8 mg/mL PEG-Scl2-2 hydrogel, p < 0.05; # significantly different from the 12 mg/mL PEG-Scl2-2 hydrogel, p < 0.05; + significant difference between cell types.
4. DISCUSSION
One approach to promote rapid endothelialization of “off-the-shelf” vascular prostheses involves utilizing bioactive luminal coatings to support EOC adhesion, proliferation, and differentiation.34–43 As previously noted, PEG-Scl2-2 hydrogels have been shown to maintain the resistance to the platelet adhesion and activation characteristic of pure PEG hydrogels61 and the resistance to platelet aggregation52 and activation associated with pure Scl2-2 proteins.61 Thus, we investigated the effect of hydrogels containing 4 mg/mL, 8 mg/mL, or 12 mg/mL of Scl2-252,61 on EOC responses relative to 4 mg/mL collagen-containing controls (roughly equivalent to the 4 mg/mL PEG-Scl2-2 formulation in terms of integrin α1/α2-I domain binding affinity).52 The EOC results were also compared against data for HUVECs, an commited EC type commonly employed in graft pre-endothelialization procedures.84
To enable EOC-based endothelialization following implantation, PEG-Scl2-2 coatings must support EOC adhesion, proliferation, and confluence. The combined cell adhesion/proliferation data (Figure 3) indicated that increasing Scl2-2 levels from 4 mg/mL to 8 mg/mL resulted in a moderate increase in initial cell adhesion. However, no further increase in initial cell attachment was observed with a further increase in Scl2-2 levels to 12 mg/mL, and Scl2-2 concentration did not appear to impact the rate of cell proliferation. Furthermore, the percent surface coverage by EOCs and HUVECs on the PEG-Scl2-2 hydrogels increased from 20–28% at 24 h to 74–79% at 72 h and did not display a dependence on cell type or Scl2-2 concentration. Nevertheless, differences in ability of the various PEG-Scl2-2 formulations to support confluence (> 95% surface coverage) were observed. Specifically, the 4 mg/mL PEG-Scl2-2 surfaces were unable to support stable EOC or HUVEC confluence, while the 8 mg/mL and 12 mg/mL PEG-Scl2-2 surfaces both allowed for the formation of stable, confluent cell layers for both cell types.
The present cell adhesion and surface coverage results appear to compare favorably with results from several previously published EOC endothelialization strategies.36,37,39 For instance, Camci-Unal et al. recently reported that methacrylated hyaluronic acid hydrogels with conjugated methacrylate-heparin supported ~75% coverage for EOCs and ~35% coverage for HUVECs.37 Furthermore, these surfaces retained less than 18% and 14% of initially seeded EOCs and HUVECs, respectively, after 72 h of culture. That said, the cell number on all PEG-Scl2-2 surfaces was significantly lower than that on the PEG-coll controls at both 24 h and 72 h for both cell types. These collagen versus Scl2-2 results are consistent with the fact that native collagen provides a range of cell-interaction/binding motifs aside from GFOGER,85,86 and thus is likely to be able to support cell adhesion through a broader array of pathways.
As with cell adhesion and proliferation, cell migration can significantly enhance the process of graft endothelialization.87 While the average HUVEC migration speed did not display a dependence on Scl2-2 concentration, the average migration speed for EOCs decreased from ~0.94 to ~0.66 μm/min as Scl2-2 levels increased from 4mg/mL to 8 mg/mL (p < 0.005). The difference in the dependence of HUVEC and EOC migration rates on Scl2-2 concentration likely stems from the cell-type specific, biphasic interplay between average cell migration speed and surface adhesion ligand density.76 Notably, both the 4 mg/mL and 8 mg/mL Scl2-2 formulations supported significantly higher average EOC and HUVEC migration speeds than the collagen controls.
To evaluate the potential of PEG-Scl2-2 hydrogels to support phenotypic maturation, we assessed EOC and HUVEC phenotype at confluence on the various hydrogel formulations. Cumulatively, both gene and protein level data indicated that 8 mg/mL PEG-Scl2-2 hydrogels supported similar or improved levels of EOC maturation relative to PEG-coll controls (i.e. ↓ CD34, ↑ VEGFR2, ↑ PECAM-1, ↔ VE-Cadherin). The 8 mg/mL PEG-Scl2-2 hydrogels also appeared to support similar or improved levels of EOC homeostatic marker expression relative to PEG-coll hydrogels (i.e. ↓ vWF, ↑ Col IV, ↔ NOS3, ↔ TM, ↔ E-selectin). Furthermore, EOCs on PEG-Scl2-2 hydrogels displayed a higher EphrinB2:EphB4 ratio relative to EOCs on PEG-coll hydrogels. These EphrinB2:EphB4 ratio results may suggest increased EOC progression toward an arterial (versus venous) EC phenotype on PEG-Scl2-2 hydrogels, although further phenotypic and functional assessments would be required for definitive statements to be made. However, an increased arterial phenotype would be desirable for EOC use in coronary artery applications, as important functional differences are known to exist between arterial and venous ECs.88–91 Finally, although Col IV is a natural component of the arterial basement membrane, it is possible – if confluence of endothelial layer is disrupted – that the Col IV deposited by the EOCs and HUVECs could be exposed to blood and induce thrombus formation. That said, Col IV is considered to be among the less blood-reactive collagens92 and has previously been utilized to reduce thrombus formation through its ability to enhance endothelial cell recruitment.93
Collectively, the results from the current study indicate that PEG-Scl2-2 hydrogel formulations can be identified which promote intermediate levels of EOC adhesion and migration, while also supporting EOC phenotypic maturation and quiescence. Future work will extend the development of PEG-Scl2-2 coatings to incorporate the effects of dynamic flow conditioning on EOC behavior and layer stability. Furthermore, a broader panel of EC markers will be assessed in evaluating EOC phenotypic changes, including the development of key functional behaviors (eg. endothelial layer barrier function).
Supplementary Material
Supplementary Figure 1. Integrin β1 subunit assessment by flow cytometry for EOCs and HUVECs. Gray filled curves represent negative controls. Black lined curves represent test samples.
Supplementary Figure 2. (A) Circular dischroism wavelength scans of Scl2-2 and acrylate-derivatized Scl2-2 (termed “fScl2-2”) demonstrate the presence of a peak at 220 nm, indicative of a triple helical structure. (B) Circular dichroism thermal transition results for Scl2-2 and fScl2-2. These circular dichroism data are consistent with previous literature results for Scl2-2.57
Supplementary Figure 3. Representative, lower magnification images of EOCs and HUVECs stained with phalloidin and counterstained with DAPI on PEG-Scl2-2 and PEG-coll hydrogels at 24 h and 72 h. The scale bar in each image series equals 200 μm and applies to all images in the series.
Supplementary Figure 4. Representative western blot images of PECAM-1, thrombomodulin (TM), and E-selectin from EOCs and HUVECs cultured on 8 mg/mL PEG-Scl2-2 and 4 mg/mL PEG-coll hydrogels for 72 h.
Acknowledgments
We would like to acknowledge the NIH R01 EB013297, NIH R03 EB0152167, NIH R21 EB020978, and the NSF DMR CAREER Award 1346807 for funding.
Footnotes
DISCLOSURES
Drs. Magnus Höök and Brooke Russell are affiliated with ECM Technologies, a start-up that utilizes bacterial-derived proteins as a platform for designing collagens for biomedical needs.
References
- 1.Peng YY, Yoshizumi A, Danon SJ, Glattauer V, Prokopenko O, Mirochnitchenko O, Yu Z, Inouye M, Werkmeister JA, Brodsky B, et al. A Streptococcus pyogenes derived collagen-like protein as a non-cytotoxic and non-immunogenic cross-linkable biomaterial. Biomaterials. 2010;31(10):2755–61. doi: 10.1016/j.biomaterials.2009.12.040. Epub 2010 Jan 6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rubin BG, McGraw DJ, Sicard GA, Santoro SA. New RGD analogue inhibits human platelet adhesion and aggregation and eliminates platelet deposition on canine vascular grafts. Journal of vascular surgery. 1992;15(4):683–692. [PubMed] [Google Scholar]
- 3.Burkel W. The challenge of small diameter vascular grafts. Medical progress through technology. 1987;14(3–4):165–175. [PubMed] [Google Scholar]
- 4.Hoshi RA, Van Lith R, Jen MC, Allen JB, Lapidos KA, Ameer G. The blood and vascular cell compatibility of heparin-modified ePTFE vascular grafts. Biomaterials. 2013;34(1):30–41. doi: 10.1016/j.biomaterials.2012.09.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sarkar S, Sales KM, Hamilton G, Seifalian AM. Addressing thrombogenicity in vascular graft construction. J Biomed Mater Res B Appl Biomater. 2007;82(1):100–8. doi: 10.1002/jbm.b.30710. [DOI] [PubMed] [Google Scholar]
- 6.Xue L, Greisler HP. Biomaterials in the development and future of vascular grafts. J Vasc Surg. 2003;37(2):472–80. doi: 10.1067/mva.2003.88. [DOI] [PubMed] [Google Scholar]
- 7.Zilla P, Deutsch M, Meinhart J. Endothelial cell transplantation. Semin Vasc Surg. 1999;12(1):52–63. [PubMed] [Google Scholar]
- 8.Noh I, Goodman SL, Hubbell JA. Chemical modification and photograft polymerization upon expanded poly(tetrafluoroethylene) Journal of Biomaterials Science, Polymer Edition. 1998;9:407–426. doi: 10.1163/156856298x00532. [DOI] [PubMed] [Google Scholar]
- 9.Tiwari A, Salacinski HJ, Hamilton G, Seifalian AM. Tissue engineering of vascular bypass grafts: role of endothelial cell extraction. Eur J Vasc Endovasc Surg. 2001;21(3):193–201. doi: 10.1053/ejvs.2001.1316. [DOI] [PubMed] [Google Scholar]
- 10.Jarrell BE, Williams SK, Stokes G, Hubbard FA, Carabasi RA, Koolpe E, Greener D, Pratt K, Moritz MJ, Radomski J, et al. Use of Freshly Isolated Capillary Endothelial-Cells for the Immediate Establishment of a Monolayer on a Vascular Graft at Surgery. Surgery. 1986;100(2):392–399. [PubMed] [Google Scholar]
- 11.Arts CHP, Joosten PPAH, Blankensteijn JD, Staal FJT, Ng PYY, Heijnen-Snyder GJ, Sixma JJ, Verhagen HJM, de Groot PG, Eikelboom BC. Contaminants from the transplant contribute to intimal hyperplasia associated with microvascular endothelial cell seeding. European Journal of Vascular and Endovascular Surgery. 2002;23(1):29–38. doi: 10.1053/ejvs.2001.1532. [DOI] [PubMed] [Google Scholar]
- 12.Gulati R, Jevremovic D, Peterson TE, Chatterjee S, Shah V, Vile RG, Simari RD. Diverse origin and function of cells with endothelial phenotype obtained from adult human blood. Circulation Research. 2003;93(11):1023–1025. doi: 10.1161/01.RES.0000105569.77539.21. [DOI] [PubMed] [Google Scholar]
- 13.Hur J, Yoon CH, Kim HS, Choi JH, Kang HJ, Hwang KK, Oh BH, Lee MM, Park YB. Characterization of two types of endothelial progenitor cells and their different contributions to neovasculogenesis. Arteriosclerosis Thrombosis and Vascular Biology. 2004;24(2):288–293. doi: 10.1161/01.ATV.0000114236.77009.06. [DOI] [PubMed] [Google Scholar]
- 14.Ingram DA, Mead LE, Tanaka H, Meade V, Fenoglio A, Mortell K, Pollok K, Ferkowicz MJ, Gilley D, Yoder MC. Identification of a novel hierarchy of endothelial progenitor cells using human peripheral and umbilical cord blood. Blood. 2004;104(9):2752–2760. doi: 10.1182/blood-2004-04-1396. [DOI] [PubMed] [Google Scholar]
- 15.Lin Y, Weisdorf D, Solovey A, Hebbel RP. Origins of circulating endothelial cells and endothelial outgrowth from blood. Journal of Clinical Investigation. 2000;105(1):71–77. doi: 10.1172/JCI8071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shi Q, Rafii S, Wu MHD, Wijelath ES, Yu C, Ishida A, Fujita Y, Kothari S, Mohle R, Sauvage LR, et al. Evidence for circulating bone marrow-derived endothelial cells. Blood. 1998;92(2):362–367. [PubMed] [Google Scholar]
- 17.Sieveking DP, Buckle A, Celermajer DS, Ng MKC. Strikingly different angiogenic properties of endothelial progenitor cell subpopulations - Insights from a novel human angiogenesis assay. Journal of the American College of Cardiology. 2008;51(6):660–668. doi: 10.1016/j.jacc.2007.09.059. [DOI] [PubMed] [Google Scholar]
- 18.Lambiase PD, Edwards RJ, Anthopoulos P, Rahman S, Meng YG, Bucknall CA, Redwood SR, Pearson JD, Marber MS. Circulating humoral factors and endothelial progenitor cells in patients with differing coronary collateral support. Circulation. 2004;109(24):2986–92. doi: 10.1161/01.CIR.0000130639.97284.EC. [DOI] [PubMed] [Google Scholar]
- 19.Massa M, Rosti V, Ferrario M, Campanelli R, Ramajoli I, Rosso R, De Ferrari GM, Ferlini M, Goffredo L, Bertoletti A, et al. Increased circulating hematopoietic and endothelial progenitor cells in the early phase of acute myocardial infarction. Blood. 2005;105(1):199–206. doi: 10.1182/blood-2004-05-1831. [DOI] [PubMed] [Google Scholar]
- 20.Scheubel RJ, Zorn H, Silber RE, Kuss O, Morawietz H, Holtz J, Simm A. Age-dependent depression in circulating endothelial progenitor cells in patients undergoing coronary artery bypass grafting. J Am Coll Cardiol. 2003;42(12):2073–80. doi: 10.1016/j.jacc.2003.07.025. [DOI] [PubMed] [Google Scholar]
- 21.Schmidt-Lucke C, Rossig L, Fichtlscherer S, Vasa M, Britten M, Kamper U, Dimmeler S, Zeiher AM. Reduced number of circulating endothelial progenitor cells predicts future cardiovascular events: proof of concept for the clinical importance of endogenous vascular repair. Circulation. 2005;111(22):2981–7. doi: 10.1161/CIRCULATIONAHA.104.504340. [DOI] [PubMed] [Google Scholar]
- 22.Werner N, Kosiol S, Schiegl T, Ahlers P, Walenta K, Link A, Bohm M, Nickenig G. Circulating endothelial progenitor cells and cardiovascular outcomes. N Engl J Med. 2005;353(10):999–1007. doi: 10.1056/NEJMoa043814. [DOI] [PubMed] [Google Scholar]
- 23.Urbich C, Aicher A, Heeschen C, Dernbach E, Hofmann WK, Zeiher AM, Dimmeler S. Soluble factors released by endothelial progenitor cells promote migration of endothelial cells and cardiac resident progenitor cells. J Mol Cell Cardiol. 2005;39(5):733–42. doi: 10.1016/j.yjmcc.2005.07.003. [DOI] [PubMed] [Google Scholar]
- 24.Rehman J, Li JL, Orschell CM, March KL. Peripheral blood “endothelial progenitor cells” are derived from monocyte/macrophages and secrete angiogenic growth factors. Circulation. 2003;107(8):1164–1169. doi: 10.1161/01.cir.0000058702.69484.a0. [DOI] [PubMed] [Google Scholar]
- 25.Tura O, Skinner EM, Barclay GR, Samuel K, Gallagher RCJ, Brittan M, Hadoke PWF, Newby DE, Turner ML, Mills NL. Late Outgrowth Endothelial Cells Resemble Mature Endothelial Cells and are Not Derived from Bone Marrow. Stem Cells. 2013;31(2):338–348. doi: 10.1002/stem.1280. [DOI] [PubMed] [Google Scholar]
- 26.Yoder MC, Mead LE, Prater D, Krier TR, Mroueh KN, Li F, Krasich R, Temm CJ, Prchal JT, Ingram DA. Redefining endothelial progenitor cells via clonal analysis and hematopoietic stem/progenitor cell principals. Blood. 2007;109(5):1801–9. doi: 10.1182/blood-2006-08-043471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hirschi KK, Ingram DA, Yoder MC. Assessing identity, phenotype, and fate of endothelial progenitor cells. Arterioscler Thromb Vasc Biol. 2008;28(9):1584–95. doi: 10.1161/ATVBAHA.107.155960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gulati R, Jevremovic D, Witt TA, Kleppe LS, Vile RG, Lerman A, Simari RD. Modulation of the vascular response to injury by autologous blood-derived outgrowth endothelial cells. Am J Physiol Heart Circ Physiol. 2004;287(2):H512–7. doi: 10.1152/ajpheart.00063.2004. [DOI] [PubMed] [Google Scholar]
- 29.Timmermans F, Plum J, Yoder MC, Ingram DA, Vandekerckhove B, Case J. Endothelial progenitor cells: identity defined? J Cell Mol Med. 2009;13(1):87–102. doi: 10.1111/j.1582-4934.2008.00598.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Timmermans F, Van Hauwermeiren F, De Smedt M, Raedt R, Plasschaert F, De Buyzere ML, Gillebert TC, Plum J, Vandekerckhove B. Endothelial outgrowth cells are not derived from CD133(+) cells or CD45(+) hematopoietic precursors. Arteriosclerosis Thrombosis and Vascular Biology. 2007;27(7):1572–1579. doi: 10.1161/ATVBAHA.107.144972. [DOI] [PubMed] [Google Scholar]
- 31.Cheng CC, Chang SJ, Chueh YN, Huang TS, Huang PH, Cheng SM, Tsai TN, Chen JW, Wang HW. Distinct angiogenesis roles and surface markers of early and late endothelial progenitor cells revealed by functional group analyses. Bmc Genomics. 2013:14. doi: 10.1186/1471-2164-14-182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yoder MC. Defining Human Endothelial Progenitor Cells. Experimental Hematology. 2010;38(9):S111–S111. [Google Scholar]
- 33.Stroncek JD, Grant BS, Brown MA, Povsic TJ, Truskey GA, Reichert WM. Comparison of endothelial cell phenotypic markers of late-outgrowth endothelial progenitor cells isolated from patients with coronary artery disease and healthy volunteers. Tissue Engineering Part A. 2009;15(11):3473–3486. doi: 10.1089/ten.tea.2008.0673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Allen JB, Khan S, Lapidos KA, Ameer GA. Toward Engineering a Human Neoendothelium with Circulating Progenitor Cells. Stem Cells. 2010;28(2):318–328. doi: 10.1002/stem.275. [DOI] [PubMed] [Google Scholar]
- 35.Hinds MT, Ma M, Tran N, Ensley AE, Kladakis SM, Vartanian KB, Markway BD, Nerem RM, Hanson SR. Potential of baboon endothelial progenitor cells for tissue engineered vascular grafts. Journal Of Biomedical Materials Research Part A. 2008;86A(3):804–812. doi: 10.1002/jbm.a.31672. [DOI] [PubMed] [Google Scholar]
- 36.Camci-Unal G, Aubin H, Ahari AF, Bae H, Nichol JW, Khademhosseini A. Surface-modified hyaluronic acid hydrogels to capture endothelial progenitor cells. Soft Matter. 2010;6(20):5120–5126. doi: 10.1039/c0sm00508h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Camci-Unal G, Nichol JW, Bae H, Tekin H, Bischoff J, Khademhosseini A. Hydrogel surfaces to promote attachment and spreading of endothelial progenitor cells. J Tissue Eng Regen Med. 2013;7(5):337–347. doi: 10.1002/term.517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wang X, Cooper S. Adhesion of endothelial cells and endothelial progenitor cells on peptide-linked polymers in shear flow. Tissue Engineering Part A. 2013;19(9–10):1113–1121. doi: 10.1089/ten.tea.2011.0653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sreerekha PR, Krishnan LK. Cultivation of endothelial progenitor cells on fibrin matrix and layering on dacron/polytetrafluoroethylene vascular grafts. Artificial organs. 2006;30(4):242–249. doi: 10.1111/j.1525-1594.2006.00211.x. [DOI] [PubMed] [Google Scholar]
- 40.Stroncek J, Ren L, Klitzman B, Reichert W. Patient-derived endothelial progenitor cells improve vascular graft patency in a rodent model. Acta Biomaterialia. 2012;8(1):201–208. doi: 10.1016/j.actbio.2011.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kang T-Y, Lee JH, Kim BJ, Kang J-A, Hong JM, Kim BS, Cha HJ, Rhie J-W, Cho D-W. In vivo endothelization of tubular vascular grafts through in situ recruitment of endothelial and endothelial progenitor cells by RGD-fused mussel adhesive proteins. Biofabrication. 2015;7(1):015007. doi: 10.1088/1758-5090/7/1/015007. [DOI] [PubMed] [Google Scholar]
- 42.Lee K-W, Johnson NR, Gao J, Wang Y. Human progenitor cell recruitment via SDF-1α coacervate-laden PGS vascular grafts. Biomaterials. 2013;34(38):9877–9885. doi: 10.1016/j.biomaterials.2013.08.082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wijelath ES, Rahman S, Murray J, Patel Y, Savidge G, Sobel M. Fibronectin promotes VEGF-induced CD34+ cell differentiation into endothelial cells. J Vasc Surg. 2004;39(3):655–660. doi: 10.1016/j.jvs.2003.10.042. [DOI] [PubMed] [Google Scholar]
- 44.Shirota T, Yasui H, Shimokawa H, Matsuda T. Fabrication of endothelial progenitor cell (EPC)-seeded intravascular stent devices and in vitro endothelialization on hybrid vascular tissue. Biomaterials. 2003;24(13):2295–2302. doi: 10.1016/s0142-9612(03)00042-5. [DOI] [PubMed] [Google Scholar]
- 45.Deb A, Skelding KA, Wang S, Reeder M, Simper D, Caplice NM. Integrin profile and in vivo homing of human smooth muscle progenitor cells. Circulation. 2004;110(17):2673–2677. doi: 10.1161/01.CIR.0000139842.15651.B2. [DOI] [PubMed] [Google Scholar]
- 46.Beumer S, Heijnen HF, MJIJ, Orlando E, de Groot PG, Sixma JJ. Platelet adhesion to fibronectin in flow: the importance of von Willebrand factor and glycoprotein Ib. Blood. 1995;86(9):3452–60. [PubMed] [Google Scholar]
- 47.Farndale RW. Collagen-induced platelet activation. Blood Cells Mol Dis. 2006;36(2):162–5. doi: 10.1016/j.bcmd.2005.12.016. [DOI] [PubMed] [Google Scholar]
- 48.Jirouskova M, Dyr JE, Suttnar J, Holada K, Trnkova B. Platelet adhesion to fibrinogen, fibrin monomer, and fibrin protofibrils in flowing blood -- the effect of fibrinogen immobilization and fibrin formation. Thromb Haemost. 1997;78(3):1125–31. [PubMed] [Google Scholar]
- 49.Noel S, Hachem A, Merhi Y, De Crescenzo G. Development of a Polyester Coating Combining Antithrombogenic and Cell Adhesive Properties: Influence of Sequence and Surface Density of Adhesion Peptides. Biomacromolecules. 2015;16(6):1682–94. doi: 10.1021/acs.biomac.5b00219. [DOI] [PubMed] [Google Scholar]
- 50.Xu Y, Keene D, Bujnicki J, Höök M, Lukomski S. Scl1 and Scl2 proteins form collagen-like triple helices. Biol Chem. 2002;277(30):27312–8. doi: 10.1074/jbc.M201163200. [DOI] [PubMed] [Google Scholar]
- 51.Han R, Zwiefka A, Caswell C, Xu Y, Keene D, Lukomska E, Zhao Z, Höök M, Lukomski S. Assessment of collagen-like sequences derived from Scl1 and Scl2 proteins as a source of recombinant GXY polymers. Applied Microbiology and Biotechnology. 2006;72(1):109–115. doi: 10.1007/s00253-006-0387-5. [DOI] [PubMed] [Google Scholar]
- 52.Seo N, Russell BH, Rivera JJ, Liang X, Xu X, Afshar-Kharghan V, Höök M. An Engineered α1 Integrin-binding Collagenous Sequence. Journal of Biological Chemistry. 2010;285(40):31046–31054. doi: 10.1074/jbc.M110.151357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Humtsoe JO, Kim JK, Xu Y, Keene DR, Hook M, Lukomski S, Wary KK. A streptococcal collagen-like protein interacts with the alpha2beta1 integrin and induces intracellular signaling. J Biol Chem. 2005;280(14):13848–57. doi: 10.1074/jbc.M410605200. Epub 2005 Jan 12. [DOI] [PubMed] [Google Scholar]
- 54.Knight CG, Morton LF, Onley DJ, Peachey AR, Ichinohe T, Okuma M, Farndale RW, Barnes MJ. Collagen-platelet interaction: Gly-Pro-Hyp is uniquely specific for platelet Gp VI and mediates platelet activation by collagen. Cardiovasc Res. 1999;41(2):450–7. doi: 10.1016/s0008-6363(98)00306-x. [DOI] [PubMed] [Google Scholar]
- 55.Farndale RW, Slatter DA, Siljander PRM, Jarvis GE. Platelet receptor recognition and cross-talk in collagen-induced activation of platelets. Journal of Thrombosis and Haemostasis. 2007;5:220–229. doi: 10.1111/j.1538-7836.2007.02521.x. [DOI] [PubMed] [Google Scholar]
- 56.Munnix ICA, Gilio K, Siljander PRM, Raynal N, Feijge MAH, Hackeng TM, Deckmyn H, Smethurst PA, Farndale RW, Heemskerk JWM. Collagen-mimetic peptides mediate flow-dependent thrombus formation by high- or low-affinity binding of integrin alpha 2 beta 1 and glycoprotein VI. Journal of Thrombosis and Haemostasis. 2008;6(12):2132–2142. doi: 10.1111/j.1538-7836.2008.03167.x. [DOI] [PubMed] [Google Scholar]
- 57.Cosgriff-Hernandez E, Hahn MS, Russell B, Wilems T, Munoz-Pinto D, Browning MB, Rivera J, Hook M. Bioactive hydrogels based on Designer Collagens. Acta Biomater. 2010;6(10):3969–77. doi: 10.1016/j.actbio.2010.05.002. Epub 2010 May 11. [DOI] [PubMed] [Google Scholar]
- 58.Knight CG, Morton LF, Peachey AR, Tuckwell DS, Farndale RW, Barnes MJ. The collagen-binding A-domains of integrins alpha(1)beta(1) and alpha(2)beta(1) recognize the same specific amino acid sequence, GFOGER, in native (triple-helical) collagens. Journal of Biological Chemistry. 2000;275(1):35–40. doi: 10.1074/jbc.275.1.35. [DOI] [PubMed] [Google Scholar]
- 59.Reyes CD, Garcia AJ. Engineering integrin-specific surfaces with a triple-helical collagen-mimetic peptide. Journal of Biomedical Materials Research Part A. 2003;65A(4):511–523. doi: 10.1002/jbm.a.10550. [DOI] [PubMed] [Google Scholar]
- 60.Khew ST, Zhu XH, Tong YW. An integrin-specific collagen-mimetic peptide approach for optimizing Hep3B liver cell adhesion, proliferation, and cellular functions. Tissue engineering. 2007;13(10):2451–2463. doi: 10.1089/ten.2007.0063. [DOI] [PubMed] [Google Scholar]
- 61.Browning MB, Dempsey D, Guiza V, Becerra S, Rivera J, Russell B, Hook M, Clubb F, Miller M, Fossum T, et al. Multilayer vascular grafts based on collagen-mimetic proteins. Acta Biomater. 2012;8(3):1010–21. doi: 10.1016/j.actbio.2011.11.015. Epub 2011 Nov 20. [DOI] [PubMed] [Google Scholar]
- 62.Browning MB, Guiza V, Russell B, Rivera J, Cereceres S, Hook M, Hahn MS, Cosgriff-Hernandez EM. Endothelial cell response to chemical, biological, and physical cues in bioactive hydrogels. Tissue Eng Part A. 2014;20(23–24):3130–41. doi: 10.1089/ten.TEA.2013.0602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Urbich C, Dimmeler S. Endothelial progenitor cells characterization and role in vascular biology. Circulation Research. 2004;95(4):343–353. doi: 10.1161/01.RES.0000137877.89448.78. [DOI] [PubMed] [Google Scholar]
- 64.McKee JA, Banik SS, Boyer MJ, Hamad NM, Lawson JH, Niklason LE, Counter CM. Human arteries engineered in vitro. EMBO reports. 2003;4(6):633–638. doi: 10.1038/sj.embor.embor847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Lu YC, Song W, An D, Kim BJ, Schwartz R, Wu MM, Ma ML. Designing compartmentalized hydrogel microparticles for cell encapsulation and scalable 3D cell culture. Journal of Materials Chemistry B. 2015;3(3):353–360. doi: 10.1039/c4tb01735h. [DOI] [PubMed] [Google Scholar]
- 66.Park AR, Park YH, Kim HJ, Kim MK, Kim SG, Kweon H, Kundu SC. Tri-layered silk fibroin and poly-E >-caprolactone small diameter vascular grafts tested in vitro and in vivo. Macromolecular Research. 2015;23(10):924–936. [Google Scholar]
- 67.Schneider KH, Aigner P, Holnthoner W, Monforte X, Nurnberger S, Runzler D, Redl H, Teuschl AH. Decellularized human placenta chorion matrix as a favorable source of small-diameter vascular grafts. Acta Biomaterialia. 2016;29:125–134. doi: 10.1016/j.actbio.2015.09.038. [DOI] [PubMed] [Google Scholar]
- 68.Shalumon KT, Deepthi S, Anupama MS, Nair SV, Jayakumar R, Chennazhi KP. Fabrication of poly (L-lactic acid)/gelatin composite tubular scaffolds for vascular tissue engineering. International Journal of Biological Macromolecules. 2015;72:1048–1055. doi: 10.1016/j.ijbiomac.2014.09.058. [DOI] [PubMed] [Google Scholar]
- 69.Munoz-Pinto DJ, Qu X, Bansal L, Hayenga HN, Hahn J, Hahn MS. Relative impact of form-induced stress vs. uniaxial alignment on multipotent stem cell myogenesis. Acta Biomater. 2012;8(11):3974–81. doi: 10.1016/j.actbio.2012.06.044. [DOI] [PubMed] [Google Scholar]
- 70.Munoz-Pinto DJ, Jimenez-Vergara AC, Gelves LM, McMahon RE, Guiza-Arguello V, Hahn MS. Probing vocal fold fibroblast response to hyaluronan in 3D contexts. Biotechnology and bioengineering. 2009;104(4):821–831. doi: 10.1002/bit.22436. [DOI] [PubMed] [Google Scholar]
- 71.Munoz-Pinto DJ, Jimenez-Vergara AC, Gharat TP, Hahn MS. Characterization of sequential collagen-poly (ethylene glycol) diacrylate interpenetrating networks and initial assessment of their potential for vascular tissue engineering. Biomaterials. 2015;40:32–42. doi: 10.1016/j.biomaterials.2014.10.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Stokes CL, Lauffenburger DA, Williams SK. Migration of individual microvessel endothelial cells: stochastic model and parameter measurement. J Cell Sci. 1991;99(Pt 2):419–30. doi: 10.1242/jcs.99.2.419. [DOI] [PubMed] [Google Scholar]
- 73.Reinhart-King CA, Dembo M, Hammer DA. Cell-Cell Mechanical Communication through Compliant Substrates. Biophysical Journal. 95(12):6044–6051. doi: 10.1529/biophysj.107.127662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.DiMilla P, Stone J, Quinn J, Albelda S, Lauffenburger D. Maximal migration of human smooth muscle cells on fibronectin and type IV collagen occurs at an intermediate attachment strength. The Journal of Cell Biology. 1993;122(3):729–737. doi: 10.1083/jcb.122.3.729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Livak KJ, Schmittgen TD. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods. 2001;25(4):402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 76.Peyton SR, Putnam AJ. Extracellular matrix rigidity governs smooth muscle cell motility in a biphasic fashion. Journal of Cellular Physiology. 2005;204(1):198–209. doi: 10.1002/jcp.20274. [DOI] [PubMed] [Google Scholar]
- 77.Charras G, Sahai E. Physical influences of the extracellular environment on cell migration. Nat Rev Mol Cell Biol. 2014;15(12):813–24. doi: 10.1038/nrm3897. [DOI] [PubMed] [Google Scholar]
- 78.Yang JJ, Ii M, Kamei N, Alev C, Kwon SM, Kawamoto A, Akimaru H, Masuda H, Sawa Y, Asahara T. CD34(+) Cells Represent Highly Functional Endothelial Progenitor Cells in Murine Bone Marrow. Plos One. 2011;6(5) doi: 10.1371/journal.pone.0020219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Waltenberger J, Claesson-Welsh L, Siegbahn A, Shibuya M, Heldin CH. Different signal transduction properties of KDR and Flt1, two receptors for vascular endothelial growth factor. J Biol Chem. 1994;269(43):26988–95. [PubMed] [Google Scholar]
- 80.Adams RH, Wilkinson GA, Weiss C, Diella F, Gale NW, Deutsch U, Risau W, Klein R. Roles of ephrinB ligands and EphB receptors in cardiovascular development: demarcation of arterial/venous domains, vascular morphogenesis, and sprouting angiogenesis. Genes & Development. 1999;13(3):295–306. doi: 10.1101/gad.13.3.295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Wang HU, Chen ZF, Anderson DJ. Molecular distinction and angiogenic interaction between embryonic arteries and veins revealed by ephrin-B2 and its receptor Eph-B4. Cell. 1998;93(5):741–753. doi: 10.1016/s0092-8674(00)81436-1. [DOI] [PubMed] [Google Scholar]
- 82.Salvucci O, Tosato G. Essential Roles of EphB Receptors and EphrinB Ligands in Endothelial Cell Function and Angiogenesis. Guidance Molecules in Cancer and Tumor Angiogenesis. 2012;114:21–57. doi: 10.1016/B978-0-12-386503-8.00002-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Malek AM, Jackman R, Rosenberg RD, Izumo S. Endothelial expression of thrombomodulin is reversibly regulated by fluid shear stress. Circulation Research. 1994;74(5):852–60. doi: 10.1161/01.res.74.5.852. [DOI] [PubMed] [Google Scholar]
- 84.de Mel A, Punshon G, Ramesh B, Sarkar S, Darbyshire A, Hamilton G, Seifalian AM. In situ endothelialization potential of a biofunctionalised nanocomposite biomaterial-based small diameter bypass graft. Biomed Mater Eng. 2009;19(4–5):317–31. doi: 10.3233/BME-2009-0597. [DOI] [PubMed] [Google Scholar]
- 85.Castanos-Velez E, Biberfeld P, Patarroyo M. Extracellular matrix proteins and integrin receptors in reactive and non-reactive lymph nodes. Immunology. 1995;86(2):270. [PMC free article] [PubMed] [Google Scholar]
- 86.Tulla M. Collagen receptor integrins: evolution, ligand binding selectivity and the effect of activation. University of Jyväskylä; 2007. [Google Scholar]
- 87.Dixit P, Hern-Anderson D, Ranieri J, Schmidt CE. Vascular graft endothelialization: Comparative analysis of canine and human endothelial cell migration on natural biomaterials. J Biomed Mater Res. 2001;56(4):545–555. doi: 10.1002/1097-4636(20010915)56:4<545::aid-jbm1126>3.0.co;2-v. [DOI] [PubMed] [Google Scholar]
- 88.Ott I, Keller U, Knoedler M, Götze KS, Doss K, Fischer P, Urlbauer K, Debus G, von Bubnoff N, Rudelius M. Endothelial-like cells expanded from CD34+ blood cells improve left ventricular function after experimental myocardial infarction. The FASEB journal. 2005;19(8):992–994. doi: 10.1096/fj.04-3219fje. [DOI] [PubMed] [Google Scholar]
- 89.Zhang Q, Kandic I, Barfield JT, Kutryk MJ. Coculture with Late, but Not Early, Human Endothelial Progenitor Cells Up Regulates IL-1β Expression in THP-1 Monocytic Cells in a Paracrine Manner. Stem cells international. 2013;2013 doi: 10.1155/2013/859643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.He TR, Peterson TE, Holmuhamedov EL, Terzic A, Caplice NM, Oberley LW, Katusic ZS. Human endothelial progenitor cells tolerate oxidative stress due to intrinsically high expression of manganese superoxide dismutase. Arteriosclerosis Thrombosis and Vascular Biology. 2004;24(11):2021–2027. doi: 10.1161/01.ATV.0000142810.27849.8f. [DOI] [PubMed] [Google Scholar]
- 91.Cui XF, Lu YW, Lee V, Kim D, Dorsey T, Wang QJ, Lee Y, Vincent P, Schwarz J, Dai GH. Venous Endothelial Marker COUP-TFII Regulates the Distinct Pathologic Potentials of Adult Arteries and Veins. Scientific Reports. 2015;5 doi: 10.1038/srep16193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Farndale RW, Sixma JJ, Barnes MJ, de Groot PG. The role of collagen in thrombosis and hemostasis. J Thromb Haemost. 2004;2(4):561–73. doi: 10.1111/j.1538-7836.2004.00665.x. [DOI] [PubMed] [Google Scholar]
- 93.Heo Y, Shin YM, Lee YB, Lim YM, Shin H. Effect of immobilized collagen type IV on biological properties of endothelial cells for the enhanced endothelialization of synthetic vascular graft materials. Colloids Surf B Biointerfaces. 2015;134:196–203. doi: 10.1016/j.colsurfb.2015.07.003. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Figure 1. Integrin β1 subunit assessment by flow cytometry for EOCs and HUVECs. Gray filled curves represent negative controls. Black lined curves represent test samples.
Supplementary Figure 2. (A) Circular dischroism wavelength scans of Scl2-2 and acrylate-derivatized Scl2-2 (termed “fScl2-2”) demonstrate the presence of a peak at 220 nm, indicative of a triple helical structure. (B) Circular dichroism thermal transition results for Scl2-2 and fScl2-2. These circular dichroism data are consistent with previous literature results for Scl2-2.57
Supplementary Figure 3. Representative, lower magnification images of EOCs and HUVECs stained with phalloidin and counterstained with DAPI on PEG-Scl2-2 and PEG-coll hydrogels at 24 h and 72 h. The scale bar in each image series equals 200 μm and applies to all images in the series.
Supplementary Figure 4. Representative western blot images of PECAM-1, thrombomodulin (TM), and E-selectin from EOCs and HUVECs cultured on 8 mg/mL PEG-Scl2-2 and 4 mg/mL PEG-coll hydrogels for 72 h.