Abstract
This study was conducted to determine the distribution of antimicrobial resistance among Escherichia coli isolated from feces of healthy dromedary camels in Kenya. A total of 162 fecal samples were cultivated for E. coli. Samples were also subcultivated to detect E. coli with extended-spectrum ß-lactamases (ESBLs). Antimicrobial susceptibility testing (AST) was performed by disk diffusion using a panel of 16 antimicrobials. In addition, isolates were screened for the presence of the plasmid-mediated colistin resistance genes mcr-1 to mcr-5.
Samples from 20 (12.4%) of the camels contained antimicrobial resistant (AMR) E. coli, and 85% of the AMR isolates were multidrug resistant (MDR). The highest frequency of resistance was observed to tetracycline (11.7%), followed by ampicillin and streptomycin (both 10.5%), and sulfamethoxazole/trimethoprim (9.9%). Two (1.2%) of the isolates showed intermediate resistance to cefazolin and streptomycin, respectively. All the isolates were susceptible to amoxycillin/clavulanic acid, ciprofloxacin, fosfomycin, aztreonam and kanamycin, and 86.4% of the isolates were susceptible to all 16 antimicrobials used in this study. The prevalence of fecal carriage of ESBL producing E. coli was 0.6%. PCR and amplicon sequencing showed that the ESBL producer belonged to E. coli phylogenetic group A, sequence type (ST) 48, and harbored blaCTX-M-15. None of the isolates contained mcr genes. The results indicate that dromedary camels in Kenya may be reservoirs of AMR E. coli, including ESBL producers, that could potentially be transmitted to humans by direct contact or via the food chain.
Keywords: Kenya, Dromedary camels, Escherichia coli, Antimicrobial resistance, ESBL
Introduction
Antimicrobial resistance (AMR) represents one of the major threats to public health, affecting the well-being of humans, animals, and the environment [1]. AMR is a challenge that requires international and multidisciplinary cooperation using a One Health approach [2]. Within this approach, targeted research is essential to identify reservoirs of AMR and demonstrate potential transmission routes within and between the human-animal-environmental interfaces.
Escherichia coli, natural inhabitants of the intestinal tracts of humans and mammals, potentially represent a major reservoir of AMR and play an important role in acquisition and the dissemination of AMR mechanisms [3]. Livestock and food producing animals have emerged as important hosts for AMR E. coli. Fecal carriage of AMR E. coli poses a potential health hazard, either through direct transmission of bacteria between animal hosts and humans, or indirectly through transmission of resistance genes by means of horizontal transfer [4]. The widely disseminated resistance to critically important third generation cephalosporins, and the recently emerged resistance to the last resort antimicrobial colistin are of particular concern to both human and animal health alike [5], [6], [7].
The number of reports of AMR E. coli isolated from healthy livestock, including strains harboring plasmid-mediated resistance genes such as extended-spectrum ß-lactamase (ESBL) genes belonging to the blaCTX-M group, or colistin resistance mcr genes [4], [8], [9], [10], is increasing worldwide. By contrast, for Africa, information on the prevalence of AMR in healthy husbandry animals is still scarce and there is a lack of a molecular studies of the clonal lineages of E. coli associated with the spread of bla ESBLs and other resistance genes [11,12]. In East Africa, the one-humped Arabian camel (Camelus dromedarius) is one of the most important livestock species among nomadic pastoral communities [13,14]. Whilst camels are acknowledged as a reservoir for pathogens such as Coxiella burnetii, Brucella spp., the hepatitis E virus, and Middle East respiratory syndrome coronavirus [15], [16], [17], [18], data on AMR in healthy camels in Africa are very restricted [19].
In response to this knowledge gap, this study aimed to evaluate the distribution of antimicrobial resistance among Escherichia coli isolated from camels in Eastern Africa by screening fecal samples of a herd of healthy animals kept under extensive husbandry systems on a ranch in Laikipia, Kenya. In addition, the prevalence of E. coli harboring bla ESBL and mcr genes was investigated.
Material and methods
In a previous study, the occurrence of Shiga toxin-producing Escherichia coli (STEC) among grazing dromedaries from Kenya was investigated [20]. Fecal samples had been collected between during January and February 2017 from camels located in a ranch in the northern Laikipia county in Kenya. Fecal matter was collected by rectal palpation using one glove per animal. Although efforts were made to select both genders of camels in the herd, a truly random sampling was not possible due to logistical constraints, for example compliance of the animals. Samples were placed in sterile containers, cooled for transport and stored at −80 °C until analyses. For this study, 162 samples (from 116 females, 45 males and one of unknown gender) were analyzed.
Each fecal sample was enriched at a 1:10 ratio in Enterobacteriaceae enrichment (EE) broth (Becton, Dickinson, Heidelberg, Germany) for 24 h at 37 °C. For nonselective isolation of E. coli, the enrichment was spread on RAPID'E. coli agar (Biorad, Munich, Germany). One E. coli colony randomly selected from each agar plate was collected for further analysis. For selective isolation of ESBL producing Enterobacteriaceae, one loopful of each of the enrichment cultures was streaked onto Brilliance ESBL agar plates (Oxoid, Hampshire, UK). Plates were incubated under aerobic conditions for 24 h at 37 °C.
Antimicrobial susceptibility testing (AST)
Antimicrobial susceptibility testing was performed using the disk-diffusion method according to the guidelines of the Clinical and Laboratory Standards Institute [21]. Antibiotics included ampicillin (AM), amoxicillin with clavulanic acid (AMC), azithromycin (AZM), cefazolin (CZ), cefepime (FEP), cefotaxime (CTX), chloramphenicol (C), ciprofloxacin (CIP), fosfomycin (FOS), gentamicin (G), kanamycin (K), nalidixic acid (NA), nitrofurantoin (F/M), streptomycin (S), sulfamethoxazole/trimethoprim (SXT), and tetracycline (TE) (Becton Dickinson, Allschwil, Switzerland). Results were interpreted according to CLSI performance standards [21]. For azithromycin, an inhibition zone of ≤ 12 mm was interpreted as resistant. Isolates displaying resistance to three or more classes of antimicrobials (counting ß-lactams as one class) were defined as multidrug-resistant (MDR).
Detection of antimicrobial resistance genes
DNA was extracted by a standard heat lysis protocol [22] and analysed by PCR for the presence of antimicrobial resistance genes. Synthesis of primers and DNA custom sequencing were carried out by Microsynth (Balgach, Switzerland). Purification of amplification products was performed using a PCR purification kit (Qiagen, Courtaboeuf, France).
The identification of bla ESBL genes in ESBL producing E. coli was established by PCR and sequencing as described previously [23], [24], [25]. In brief, specific published primers were used to search for ß-lactamase encoding genes belonging to bla TEM, bla SHV [26], and the three bla CTX − M alleles belonging to groups 1, 2 and 9, as described by Woodford et al. [24], using amplification conditions as described previously [24,26].
Purified amplicons were custom sequenced using primers form up- and downstream bla CTX − M flanking regions as noted by Geser at al. [23].
Screening by PCR for mcr-1 through mcr-5 was undertaken as described by Rebelo et al. [27], using a multiplex PCR protocol that specifically amplifies the fragments of the five mcr genes, which range in size from 320 bp (mcr-1) to 1644 bp (mcr-5) [27].
Nucleotide sequences were analysed with CLC Main Workbench 6.6.1. Database searches were performed using the BLASTN program of NCBI (http://www.ncbi.nlm.nih.gov/blast/).
Phylogenetic characterization of ESBL producing E. coli
E. coli can be classified into one of the eight phylogroups A, B1, B2, C, D, E, F (E. coli sensu stricto), or Escherichia clade I, whereby commensal strains mainly belong to phylogroups A and B1, and virulent extraintestinal E. coli strains to group B2 and, to a lesser extent, to group D [28]. Phylogenetic grouping was performed by using quadruplex PCR as described by Clermont et al. [28]. In brief, the presence or absence of the four gene targets arpA, chuA, yjaA and TspE4.C2 was established, using primers and amplification conditions described previously [28]. Phylogroup assignment was carried out according to the scheme described by Clermont et al. [28].
Multilocus sequence typing (MLST) of ESBL producing E. coli
For multilocus sequence typing of E. coli isolates, internal fragments of the seven housekeeping genes adk, fumC, gyrB, icd, mdh, purA, and recA were amplified by PCR, using primers and conditions described by Wirth et al. [29]. Amplicons were sequenced and allele numbers were obtained by comparing with corresponding alleles available in the E. coli MLST database website (https://enterobase.warwick.ac.uk), to determine MLST types and clonal complexes (CCs).
Results and discussion
By nonselective cultivation, 162 E. coli isolate were obtained for AST. Thereof, 140 (86.4%) were fully susceptible to all 16 antimicrobials used in this study, and two (1.2%) showed intermediate resistance to cefazolin and streptomycin, respectively (supplementary Table 1). All isolates were susceptible to amoxycillin/clavulanic acid, ciprofloxacin, fosfomycin, aztreonam and kanamycin (supplementary Table 1).
AMR E. coli were isolated from 20 (12.4%) of the samples. Resistance to tetracycline was detected most frequently (11.7%), followed by ampicillin and streptomycin (both 10.5%), and sulfamethoxazole/trimethoprim (9.9%). Resistance to all other antimicrobial agents were detected at rates between 0% and 1.2%. MDR was identified in 17 (85%) of the 20 resistant E. coli isolates, corresponding to 10.5% of the total of 162 E. coli. The distribution of antimicrobial resistance among the 162 isolates is shown in Fig. 1 , and the resistance profiles of the AMR E. coli are summarized in Table 1 .
Fig. 1.
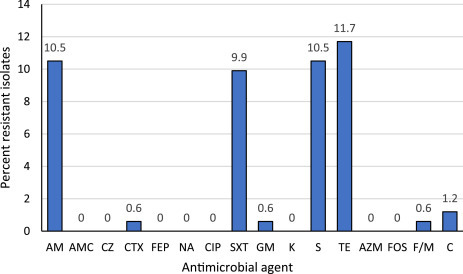
Distribution of antimicrobial resistance among 162 Escherichia coli isolated from feces of healthy camels in Kenya. Numbers above columns indicate percentage of isolates.
AM, ampicillin; AMC, amoxicillin/clavulanic acid, AZM, azithromycin; C, chloramphenicol; CIP, ciprofloxacin; CTX, cefotaxime; CZ, cefazolin; FEP, cefepime; F/M, nitrofurantoin; FOS, fosfomycin; GM, gentamicin; K, kanamycin; NA, nalidixic acid; S, streptomycin; SXT, sulfamethoxazole/trimethoprim; TE, tetracycline.
Table 1.
Antimicrobial resistance profiles of 20 Escherichia coli isolates with AMR isolated from 162 fecal samples of dromedary camels in Kenya.
| No. (%) of AMR isolates (n = 20) | Resistance profile | Prevalence (%) among fecal samples (n = 162) |
|---|---|---|
| 14 (70) | AM, SXT, S, TE | 8.6 |
| 2 (10) | AM, SXT, S, C, TE | 1.2 |
| 1 (5) | AM, S, TE | 0.6 |
| 1 (5) | CTX | 0.6 |
| 1 (5) | F/M, TE | 0.6 |
| 1 (5) | GM, TE | 0.6 |
AM, ampicillin; CTX, cefotaxime; F/M, nitrofurantoin; C, chloramphenicol; GM, gentamicin; S, streptomycin; SXT, sulfamethoxazole/trimethoprim; TE, tetracycline.
There is a scarcity of previous data on AMR in camels in Africa to compare our findings with. However, a previous study from Tunisia showed that among E. coli isolated from fecal samples of camel calves, the ampicillin/tetracycline resistance phenotype was most prevalent, corresponding to the most commonly used antibiotics in that country [30]. For Kenya, recent reports on the consumption of antimicrobials in animals are not available, however, the tetracycline and sulfamethoxazole/trimethoprim resistance phenotypes observed in this study correlate with data reported from 1997 to 2001 that show that tetracyclines, sulfonamides and trimethoprim are the most frequently used antimicrobials in food-producing animals in Kenya [31]. Notably, the AST results for the antimicrobials were interpreted using breakpoints relevant for the administration in humans and thus may not be reflective of susceptibility when applied to camels. Nevertheless, the results presented in this study provide the possibility to monitor trends in antimicrobial susceptibility and to identify emerging resistance. Accordingly, quantitative data such as the zone diameter values provided in supplementary Table 1 could be helpful.
Among the 20 camels with AMR E. coli, 17 (85%) were female, and three (15%) were male, amounting to 14.6% of the 116 female and 6.7% of the 45 male camels included in this study, respectively. Accordingly, fecal carriage of MDR E. coli was confirmed for 12.9% of the female and 6.7% of the male camels, respectively. The relatively high prevalence of AMR isolates among female camels is noteworthy. The frequency of ampicillin, streptomycin and sulfamethoxazole/trimethoprim resistance may be reflective of the usage of antimicrobials commonly used to treat mastitis in camels. In contrast to cattle, mastitis in camels is treated systemically with penicillin/aminoglycoside or trimethoprim–sulfamethoxazole [32]. However, for this study, no data on the use of antimicrobials was available, and further studies would be required in order to correlate the use of antimicrobials with the fecal carriage of AMR in camels.
By selective cultivation, one ESBL producing E. coli was detected, amounting to 0.6% of the 162 samples. Molecular characterization assigned the isolate to phylogenetic group A and sequence type (ST) 48 and identified the ESBL-encoding gene bla CTX − M-15.
The ESBL producer was identified in a fecal sample of a female camel, corresponding to a fecal carriage rate of 0.6% among the 162 camels and 0.9% of the female camels.
ESBL producing E. coli from feces of healthy camels have very rarely been described in Africa. So far, only one CTX-M-1 producing E. coli isolated from a food producing dromedary camel in Tunisia has been described [19]. While CTX-M-1 is a livestock associated ESBL, CTX-M-15, which was identified in E. coli ST48 in this study, has emerged worldwide as one of the most important ESBLs in human medicine [10]. Although the phylogenetic group A and the sequence type of E.coli ST48 does not indicate high pathogenicity [28,29], it is to be noted that CTX-M-15 producing E. coli ST48 have been reported to cause nosocomial urinary tract infections in Belgium [33] and have been detected among human isolates in hospitals in Nigeria [34]. Furthermore, E. coli ST48 belongs to clonal complex 10 which is associated globally with the dispersion of CTX-M type ESBLs [35].
Although the prevalence of ESBL producing E. coli among the camels analyzed in this study is low, this observation is important, since such isolates could be transmitted to humans. Indeed, ESBL producing Enterobacteriaceae have been detected previously in raw and in fermented camel milk in Kenya [36]. Fecal carriage of ESBL producers in camels may contribute to such contaminations in non-industrialized milk production.
The emergence of plasmid-mediated colistin resistance is most probably related to the use of colistin in animal husbandry [6]. Monitoring colistin resistance is important, since the mcr gene has been detected in food producing animals worldwide, including Africa [37]. In our study, screening for plasmid-mediated colistin resistance genes mcr-1 through mcr-5 remained negative for all isolates (data not shown). The absence of plasmid-mediated colistin resistance confirms previous reports from Tunisia and suggests that camels do not play a major role as a reservoir for mcr harboring E. coli [38]. However, although we screened for all known variants at the time of analysis, novel mcr genes are emerging globally, and it cannot be excluded that variants including mcr-6 through mcr-8 may have been missed in this study. A further limitation of this study is the sampling scheme which may have led to bias associated with operational circumstances, e.g., sampling of more manageable animals. This may have resulted in an overrepresentation of samples originating from female camels.
In conclusion, our results indicate that dromedary camels in Kenya may be reservoirs of E. coli exhibiting resistance to antimicrobials commonly used in humans and animal medicine. Furthermore, camels may carry ESBL producing E. coli that could potentially be transmitted to humans by direct contact or via the food chain.
CRediT authorship contribution statement
Magdalena Nüesch-Inderbinen: Formal analysis, Writing - original draft. Patrick Kindle: . Melinda Baschera: Data curation, Formal analysis, Investigation. Anne Liljander: Data curation, Investigation. Jörg Jores: Project administration, Writing - review & editing. Victor Max Corman: Project administration, Writing - review & editing. Roger Stephan: Supervision, Visualization, Writing - review & editing.
Declaration of Competing Interest
None to declare.
Acknowledgments
Acknowledgments
This work was partly supported by the University of Zurich and the Swiss Federal Office of Public Health, Division communicable Disease. Additional funds were received from the German Ministry of Economic Cooperation (Contract No. 81195004). Anne Liljander was supported by the Centre for International Migration and Development. We thank Katrin Zurfluh for her technical help.
Ethics statement
The sample collection was performed in accordance with the United Kingdom's Animals (Scientific Procedures) Act 1986 that contains guidelines and codes of practice for the housing and care of animals used in scientific procedures, and with the Kenyan legislation for animal experimentation. The procedure was approved of by the institutional animal care and use committee (IACUC reference number 2014.08).
Disclosures
The authors report no conflict of interest. All authors have read and approved the final article.
Editor: Dr. B. Gyampoh
Footnotes
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.sciaf.2020.e00274.
Appendix. Supplementary materials
References
- 1.World Health Organization (WHO) WHO Press; Geneva, Switzerland: 2014. Antimicrobial Resistance: Global Report on Surveillance.https://apps.who.int/iris/bitstream/handle/10665/112642/9789241564748_eng.pdf;jsessionid=E748F6CB85FB55891F79FA5AAB5E7296?sequence=1 Available at. Accessed 29. MAY 2019. [Google Scholar]
- 2.McEwen S.A., Collignon P.J. Antimicrobial resistance: a one health perspective. Microbiol. Spectr. 2018;6 doi: 10.1128/microbiolspec.ARBA-0009-2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rolain J.M. Food and human gut as reservoirs of transferable antibiotic resistance encoding genes. Front. Microbiol. 2013;4:173. doi: 10.3389/fmicb.2013.00173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Aarestrup F.M. The livestock reservoir for antimicrobial resistance: a personal view on changing patterns of risks, effects of interventions and the way forward. Philos. Trans. R Soc. Lond. B Biol. Sci. 2015;370 doi: 10.1098/rstb.2014.0085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.World Health Organization (WHO). Critically Important Antimicrobials for Human Medicine - 5th Rev. Available athttps://www.who.int/foodsafety/areas_work/antimicrobial-resistance/cia/en/. Accessed 29 MAY 2019.
- 6.Poirel L., Nordmann P. Emerging plasmid-encoded colistin resistance: the animal world as the culprit. J. Antimicrob. Chemother. 2016;71(8):2326–2327. doi: 10.1093/jac/dkw074. [DOI] [PubMed] [Google Scholar]
- 7.Paterson D.L., Harris P.N. Colistin resistance: a major breach in our last line of defence. Lancet Infect. Dis. 2016;16:132–133. doi: 10.1016/S1473-3099(15)00463-6. [DOI] [PubMed] [Google Scholar]
- 8.Nüesch-Inderbinen M., Stephan R. Epidemiology of extended-spectrum β-lactamase-producing Escherichia coli in the human-livestock environment. Curr. Clin. Microbiol. Rep. 2016;3:1–9. [Google Scholar]
- 9.Schwarz S., Johnson A.P. Transferable resistance to colistin: a new but old threat. Antimicrob. Chemother. 2016;71:2066–2070. doi: 10.1093/jac/dkw274. 2016. [DOI] [PubMed] [Google Scholar]
- 10.Cantón R., González-Alba J.M., Galán J.C. CTX-M enzymes: origin and diffusion. Front. Microbiol. 2012;3:110. doi: 10.3389/fmicb.2012.00110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Founou L.L., Amoako D.G., Founou R.C., Essack S.Y. Antibiotic resistance in food animals in Africa: a systematic review and meta-analysis. Microb. Drug Resist. 2018;24:648–665. doi: 10.1089/mdr.2017.0383. [DOI] [PubMed] [Google Scholar]
- 12.Alonso C.A., Zarazaga M., Ben Sallem R., Jouini A., Ben Slama K., Torres C. Antibiotic resistance in Escherichia coli in husbandry animals: the African perspective. Lett. Appl. Microbiol. 2017;64:318–334. doi: 10.1111/lam.12724. [DOI] [PubMed] [Google Scholar]
- 13.Schwartz H.J., Dioli M. 1st ed. A pictorial guide to diseases, health care, and management; Berlin: 1992. The One-Humped Camel (Camelus dromedarius) in Eastern Africa. [Google Scholar]
- 14.V. Benka, Human-Wildlife Conflict, Interspecies Disease, and Justice in a Wild-Life Rich Region in Kenya University of Michigan. Master's thesis. Available athttps://deepblue.lib.umich.edu/bitstream/handle/2027.42/96190/benka%20thesis%20final.pdf?sequence=1. Accessed 29 Nov 2018.
- 15.DePuy W., Benka V., Massey A., Deem S.L., Kinnaird M., O'Brien T., et al. Vol. 11. EcoHealth; Kenya: 2014. pp. 429–433. (Q Fever Risk Across a Dynamic, Heterogeneous Landscape in Laikipia County). [DOI] [PubMed] [Google Scholar]
- 16.Ducrotoy M., Bertu W.J., Matope G., Cadmus S., Conde-Álvarez R., Gusi A.M., et al. Brucellosis in sub-saharan Africa: current challenges for management, diagnosis and control. Acta Trop. 2017;165:179–193. doi: 10.1016/j.actatropica.2015.10.023. [DOI] [PubMed] [Google Scholar]
- 17.Rasche A., Saqib M., Liljander A.M., Bornstein S., Zohaib A., Renneker S., et al. Hepatitis E virus infection in dromedaries, north and east Africa, United Arab Emirates, and Pakistan, 1983-2015. Emerg. Infect. Dis. 2016;22:1249–1252. doi: 10.3201/eid2207.160168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jores J. UK; 2015. Middle East Respiratory Syndrome-Coronavirus in Camels: An Overview For Sub-Saharan and North Africa. Evidence on Demand. [DOI] [Google Scholar]
- 19.Ben Sallem R., Ben Slama K., Sáenz Y., Rojo-Bezares B., Estepa V., Jouini A., et al. Prevalence and characterization of extended-spectrum beta-lactamase (ESBL)- and CMY-2-producing Escherichia coli isolates from healthy food-producing animals in Tunisia. Foodborne Pathog. Dis. 2012;9:1137–1142. doi: 10.1089/fpd.2012.1267. [DOI] [PubMed] [Google Scholar]
- 20.Baschera M., Cernela N., Stevens M.J., Liljander A., Jores J., Corman V.M., et al. Shiga toxin-producing Escherichia coli (STEC) isolated from fecal samples of African dromedary camels. One Health. 2019;7:100087. doi: 10.1016/j.onehlt.2019.100087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Clinical and Laboratory Standards Institute (CLSI) 27th ed. CLSI; Wayne: 2017. Performance Standards For Antimicrobial Susceptibility Testing. Supplement M100S. [Google Scholar]
- 22.Sambrook J., Russell D.W. Cold Spring Harbor Laboratory Press; New York: 2001. Molecular Cloning: A Laboratory Manual (3-Volume Set) [Google Scholar]
- 23.Geser N., Stephan R., Korczak B.M., Beutin L., Hächler H. Molecular identification of extended-spectrum-β-lactamase genes from enterobacteriaceae isolated from healthy human carriers in Switzerland. Antimicrob. Agents Chemother. 2012;56:1609–1612. doi: 10.1128/AAC.05539-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Woodford N., Fagan E.J., Ellington M.J. Multiplex PCR for rapid detection of genes encoding CTX-M extended-spectrum (beta)-lactamases. J. Antimicrob. Chemother. 2006;57(1):154–155. doi: 10.1093/jac/dki412. [DOI] [PubMed] [Google Scholar]
- 25.Zurfluh K., Nüesch-Inderbinen M., Morach M., Berner A.Z., Hächler H., Stephan R. Extended-spectrum ß-lactamase-producing-Enterobacteriaceae in vegetables imported from the Dominican Republic, India, Thailand and Vietnam. Appl. Environ. Microbiol. 2015;81:3115–3120. doi: 10.1128/AEM.00258-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pitout J.D., Thomson K.S., Hanson N.D., Ehrhardt A.F., Moland E.S., Sanders C.C. Beta-lactamases responsible for resistance to expanded-spectrum cephalosporins in Klebsiella pneumoniae, Escherichia coli, and Proteus mirabilis isolates recovered in South Africa. Antimicrob. Agents Chemother. 1998;42:1350–1354. doi: 10.1128/aac.42.6.1350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rebelo A.R., Bortolaia V., Kjeldgaard J.S., Pedersen S.K., Leekitcharoenphon P., Hansen I.M., et al. Multiplex PCR for detection of plasmid-mediated colistin resistance determinants. Euro Surveill. 2018;23:29–39. doi: 10.2807/1560-7917.ES.2018.23.6.17-00672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Clermont O., Christenson J.K., Denamur E., Gordon D.M. The Clermont Escherichia coli phylo-typing method revisited: improvement of specificity and detection of new phylo-groups. Environ. Microbiol. Rep. 2013;5:58–65. doi: 10.1111/1758-2229.12019. [DOI] [PubMed] [Google Scholar]
- 29.Wirth T., Falush D., Lan R., Colles F., Mensa P., Wieler L.H., et al. Sex and virulence in Escherichia coli: an evolutionary perspective. Mol. Microbiol. 2006;60:1136–1151. doi: 10.1111/j.1365-2958.2006.05172.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bessalah S., Fairbrother J.M., Salhi I., Vanier G., Khorchani T., Seddik M.M., et al. Antimicrobial resistance and molecular characterization of virulence genes, phylogenetic groups of Escherichia coli isolated from diarrheic and healthy camel-calves in Tunisia. Comp. Immunol. Microbiol. Infect. Dis. 2016;49:1–7. doi: 10.1016/j.cimid.2016.08.008. [DOI] [PubMed] [Google Scholar]
- 31.Mitema E.S., Kikuvi G.M., Wegener H.C., Stohr K. An assessment of antimicrobial consumption in food producing animals in Kenya. J. Vet. Pharmacol. Therap. 2001;24:385–390. doi: 10.1046/j.1365-2885.2001.00360.x. [DOI] [PubMed] [Google Scholar]
- 32.Jilo K., Galgalo W., Mata W. Camel mastitis: a review. MOJ Ecol. Environ. Sci. 2017;2:00034. [Google Scholar]
- 33.Smet A., Martel A., Persoons D., Dewulf J., Heyndrickx M., Claeys G. et al. characterization of extended-spectrum β-lactamases produced by Escherichia coli isolated from hospitalized and nonhospitalized patients: emergence of CTX-M-15-producing strains causing urinary tract infections. Microb. Drug Resist. 2010;16:129–134. doi: 10.1089/mdr.2009.0132. [DOI] [PubMed] [Google Scholar]
- 34.Aibinu I., Odugbemi, Koenig T., Ghebremedhin W. B. Sequence type ST131 and ST10 complex (ST617) predominant among CTX-M-15-producing Escherichia coli isolates from Nigeria. Clin. Microbiol. Infect. 2012;18:E49–E51. doi: 10.1111/j.1469-0691.2011.03730.x. [DOI] [PubMed] [Google Scholar]
- 35.Naseer U., Sundsfjord A. The CTX-M conundrum: dissemination of plasmids and Escherichia coli clones. Microb. Drug Resist. 2011;17:83–97. doi: 10.1089/mdr.2010.0132. [DOI] [PubMed] [Google Scholar]
- 36.Njage P.M., Dolci S., Jans C., Wangoh J., Lacroix C. Ampicillin resistance and extended spectrum β-lactamases in enterobacteriaceae isolated from raw and spontaneously fermented camel milk. Afr. J. Microbiol. Res. 2012;6:1446–1452. [Google Scholar]
- 37.Grami R., Mansour W., Mehri W., Bouallègue O., Boujaâfar N., Madec J.Y., et al. Impact of food animal trade on the spread of mcr-1-mediated colistin resistance, tunisia, July 2015. Euro Surveill. 2016;21 doi: 10.2807/1560-7917.ES.2016.21.8.30144. [DOI] [PubMed] [Google Scholar]
- 38.Rhouma M., Bessalah S., Salhi I., Thériault W., Fairbrother J.M., Fravalo P. Screening for fecal presence of colistin-resistant Escherichia coli and mcr-1 and mcr-2 genes in camel-calves in southern Tunisia. Acta Vet. Scand. 2018;60:35. doi: 10.1186/s13028-018-0389-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


