- I.
- II.
- III.
- IV.
I. Introduction
Pigs are efficient converters of feed grains into valuable animal protein. As a result the swine industry provides about 25% of the energy and 9% of the protein that human beings obtain from animal sources (Pond, 1983). The industry annually provides about 30–35 kg of high-quality protein for each person in the United States. To increase production efficiency, innovative management practices have been instituted and biologicals are used to reduce disease-related losses. Moreover, swine production has changed from a large number of small farms to a relatively small number of large operations. Because of the intensive nature of these production units, losses due to contagious disease have been magnified, especially those manifested in the respiratory, reproductive, and enteric systems. To prevent diseases, swine are routinely vaccinated against common pathogens that are responsible for significant mortality, morbidity, and reduced weight gain. Some of the live vaccines, used, for example, against the viral diseases, are rotavirus, transmissible gastroenteritis virus, pseudorabies virus, and parvovirus. In spite of the availability of effective vaccines for some diseases, novel pathogens (e.g., the porcine respiratory and reproductive syndrome virus) continue to emerge and some of the attenuated virus (e.g., pseudorabies virus) vaccines can become latent.
Although the economic impact of swine diseases varies, significant losses due to infectious agents still occur. While current information on disease-related losses is not available, according to a 1986 report of the Committee on CSRS Animal Health Research Programs, major disease-related losses attributed to respiratory infections were $400 million, to reproductive disorders were $200 million, and to enteric infections were $214 million annually in 1976. In this regard, three distinct swine diseases that are viral in origin are briefly described next.
Aujeszky's disease is caused by the herpesvirus (pseudorabies virus) and is responsible for significant economic losses to the swine industry. This disease is contagious and is characterized by encephalomyelitis and inflammation of the upper respiratory tract. Mortality can reach 100% in piglets under 2 weeks of age. The respiratory form of the disease is common in growing and adult pigs. In pregnant sows, abortion, mummification of fetuses, or stillbirths can occur depending on the stage of pregnancy. Recovered or subclinically infected pigs continue to shed virus leading to persistent herd infection. The annual cost of pseudorabies for swine producers was more than $21 million in the mid-1980s (Miller et al., 1996). Most vaccines do provide clinical protection against disease but do not prevent shedding or multiplication of the virus. Thus, some animals remain carriers for variable periods and become a source of infection for susceptible animals. Therefore, it is important for vaccination to prevent or reduce virus shedding to the extent that transmission to other susceptible animals is reduced. To attenuate the virus further, several genetically engineered deletion mutants have been developed and evaluated for their ability to reduce virus shedding. The impact of such vaccines is not yet fully known. Several countries are attempting to eradicate pseudorabies virus infection in their swine populations with or without the use of vaccines. However, in certain regions feral swine harboring latent virus can still be a potential source of infection for the domestic swine. Eradication of pseudorabies virus from such a population is practically impossible.
Diarrheal disease is a common and significant problem among neonatal pigs. Economic losses in the United States due to neonatal diarrhea are estimated in excess of $200 million annually. Similar problems are encountered in other countries, such as Australia. Here, Mullan et al. (1994) estimated a loss of $260 to $330 per breeding sow in the ensuing 12 months after infection with transmissible gastroenteritis virus (TGEV). According to the National Animal Monitoring System of USDA, TGE cost the pork industry located in Iowa alone $10 million annually in 1987 and 1988 (Hill, 1989). TGEV causes a highly contagious enteric disease affecting pigs of all ages. In case of neonatal pigs, TGE is characterized by severe diarrhea, vomiting, and mortality approaching 100%. This disease is caused by a coronavirus that is shed in feces and nasal secretions. Current vaccines consisting of attenuated or inactivated TGEV are inadequate (Saif and Jackwood, 1989; Saif and Wesley, 1992).
In recent years an economically important emerging pathogen, porcine respiratory and reproductive syndrome (PRRS) virus, has been responsible for significant losses to the swine industry. Clinical signs of the disease range from inapparent infection to severe losses of more than 20% pig production and can occur in all types of swine production systems (Becker and Schwartz, 1996). PRRS virus strains of variable virulence cause reproductive and respiratory tract disease. Modified live virus vaccines against PRRS are available although a considerable amount of evidence suggests that vaccines are clearly not the entire solution to the PRRS problem. A severe form of PRRS recently emerged in Iowa, despite vaccination (Halbur and Bush, 1997).
II. Conventional Vaccines
The greatest triumph in the history of disease prevention and eradication came toward the end of the eighteenth century when Edward Jenner introduced inoculation against smallpox. Jenner's work has led to the development of many human and animal vaccines and the rapid advancement of the sciences of immunology, virology, and vaccinology. Vaccines represent an important tool for the prevention or eradication of diseases. In this regard, the greatest achievement in this century has been the eradication of smallpox.
Prevention of diseases through vaccination has been shown to be extremely beneficial, not only in reducing mortality and morbidity but also in reducing the cost of animal production. In addition to providing protection to vaccinated animals, vaccines also reduce the spread of infection. Due to their relatively low cost, vaccines are popular instruments of disease prevention that offer an important form of investment for the long-range success of the animal industry. The impact of vaccines for the prevention and control of diseases is becoming increasingly recognized and research on all aspects of vaccination has increased considerably in recent years.
The traditional approach to vaccine development includes both modified live (attenuated) and inactivated vaccines. In many instances attenuation has been attempted by serial passage of the virulent virus in an in vitro cell culture system. Live attenuated vaccines are developed by trying to establish the balance between maximum immunogenicity and minimum virulence for the host. Because the genetic makeup of such vaccine strains is not known, in many cases there is the risk of reversion to virulence under field conditions. Efficacy of vaccines is usually determined by the protective immune response in the host. Because immunity can be broken by an overwhelming challenge, no vaccine can be claimed as “perfect.” Recovery from a natural infection usually leads to a strong and long-lasting protection against reinfection by the same pathogen. Although a vaccine that is completely safe normally does not induce as strong an immunity as the natural infection, to be efficacious it should induce an appreciable degree of protective immunity in the host when exposed to a reasonable natural challenge.
Attenuated modified live vaccines are generally preferred since they provide strong, long-lasting immunity and are more easily produced than inactivated vaccines. However, as mentioned earlier, they often pose the threat of reversion to virulence and transmissibility to other species and often are required to be maintained at a correct passage level. Moreover, the same vaccine produced by different biological manufacturers may have a varied potential for reversion to virulence. Furthermore, it is probable that in vivo recombination either between different vaccine strains and/or field strains, for example, pseudorabies virus, will result in the generation of a strain with greater virulence than the original vaccine strain(s). These concerns are unwarranted when using inactivated or subunit vaccines; however, frequent administration is required and, therefore, application of killed vaccines becomes more expensive than the use of live vaccines. Although in designing a vaccine the main emphasis is to protect the host against the disease, it is also important to consider the duration of immunity, lack of any adverse effects, ease of administration, and low cost. The increasing number of vaccines against common and emerging pathogens has made their individual administration impractical. In this regard, the biological companies have realized for a long time the advantages of combined vaccines that will protect against several diseases. Because multi-antigenic vaccines require a relatively lower number of injections, the cost of packaging, storage, delivery equipment, and labor are reduced.
The problems associated with the use of current vaccines can be overcome by the development of a new generation of live vaccines in which only the protective antigen(s) of a pathogen is presented to the immune system of the host and chances of reversion to virulence are eliminated. In such a vaccine the beneficial properties of both live and killed vaccines can be retained.
III. Recombinant Virus Vectored Vaccines
The availability of molecular methods and knowledge enable us to overcome the limitations of traditional approaches in vaccine development. Using modern genetic engineering techniques, it is possible to isolate, identify, and sequence important genes of pathogenic organisms and place them into new vectors in which they can be faithfully expressed. Both bacterial and viral vectors can be used for the expression of foreign genes. Bacteria are easy to manipulate and can provide a high level of expression, but in bacteria glycosylation, proteolytic processing, and subunit assembly of eukaryotic proteins may not occur properly. Consequently, the use of such vectors may not result in the production of an authentic protein(s). Therefore, the genomes of both small and large viruses, for example, baculoviruses, adenoviruses, herpesviruses, and poxviruses, have been manipulated for expression of foreign proteins. The genomes of the large-sized viruses (e.g., poxviruses and herpesviruses) are difficult to alter but have the capacity to express a significant amount of foreign genetic material (i.e., several foreign genes). When considering herpesviruses as vaccine vectors, however, it is important to consider their potential for delayed persistence and oncogenesis. On the other hand the small-sized viruses (e.g., adenoviruses) have the limitation that their genomes can accommodate only a small amount of foreign genetic material without the virus becoming defective in replication.
A. Vaccinia and Avianpox Viruses
Although poxviruses have been of concern for many years by virtue of their impact on human and animal health, the recent increasing interest in these viruses stems from their usefulness as viral vectors (Moss and Flexner, 1987). Extensive experience obtained with the use of vaccinia virus as a live vaccine, as well as its wide host range, large size genome capable of accommodating a substantial amount of foreign DNA and inability to induce oncogenic transformation have been some of the important features favoring its potential use as a vector for immunization against important pathogens. Additionally, genetically altered vaccinia viruses maintain their infectivity after insertion of foreign genes, and induce both humoral and cellular immunity. Vaccinia vectored vaccines are relatively inexpensive to produce and easy to administer.
Since the first demonstration in 1982 of the ability of vaccinia virus to express an inserted herpes simplex virus thymidine kinase (TK) gene (Panicali and Paoletti, 1982), a large variety of foreign genes have been expressed by recombinant vaccinia viruses. When those genes encode for antigens responsible for eliciting an immune response, the resultant recombinant viruses have been shown to elicit protective immunity in animals against the respective pathogens. Thus, when foreign genes are expressed under vaccinia virus regulation, the proteins are produced in a native state. Because the genome of vaccinia virus has the ability to accept up to 25,000 bp of inserted foreign DNA, more than 10 foreign genes could conceivably be expressed by a single live recombinant virus (Flexner and Moss, 1997). Thus the demonstrated and potential success with vaccinia virus as a gene expression vector has kept its popularity unchallenged for so many years. However, because of its wide host range, there has been reluctance in accepting vaccinia-vectored vaccines due to postvaccination complications in humans (Gurvich, 1992), which were observed during the use of vaccinia for the eradication of smallpox. Consequently, this technology has been applied to host-specific avianpox viruses (e.g., fowlpox and canarypox viruses). Use of such vectors with their greatly restricted replicative ability provides a safety advantage for the host as well as those who come in contact with them.
In spite of limited basic information about the genome of these viruses, remarkable progress has been made in the development of avianpox virus vectored vaccines. Some key events that led to these early successes were (1) the continuous use of live fowlpox virus vaccines by the poultry industry for more than 50 years to prevent fowlpox in chicken and turkeys, (2) the effective substitution of heterologous vaccinia virus promoters in lieu of homologous avianpox virus promoters (Tripathy and Wittek, 1990; Schnitzlein and Tripathy, 1990) in the development of recombinant fowlpox virus vectors, (3) the ability of primary as well as a permanent cell line of avian origin to support the growth of fowlpox virus, and (4) the availability of several genes encoding for specific proteins from poultry pathogens. Consequently, several recombinant fowlpox viruses expressing specific proteins from a variety of avian pathogens were created. Immunization of susceptible birds with such recombinants resulted in the development of specific antibodies and enabled protection to subsequent challenge with the respective virulent pathogen (Tripathy, 1996). These developments have resulted in the licensing of a recombinant fowlpox virus vaccine expressing the hemagglutinin of Newcastle disease virus. Additionally, it has been shown that avianpox viruses (e.g., fowlpox and canarypox viruses) expressing foreign antigens can induce an immune response in mammalian hosts (Taylor et al., 1988, 1991) without causing a productive infection.
B. Swinepox Virus as Vaccine Vector
Before considering swinepox virus as an expression vector for genes from swine pathogens, it is necessary to mention that several other viruses have been engineered to express genes encoding for protective antigens of swine pathogens. For example, Tuboly et al. (1994) showed that baculovirus-expressed spike protein of the transmissible gastroenteritis virus was capable of inducing TGEV-specific antibodies of the IgG class in pigs. A recombinant pseudorabies virus expressing the enveloped glycoprotein E1 of hog cholera virus provided protection against both pseudorabies and hog cholera (classical swine fever) viruses (van Zijl et al., 1991). A highly attenuated strain of vaccinia virus was developed by Tartaglia et al. (1992) and used as a vector for the expression of pseudorabies virus proteins, which were evaluated for protection (Brockmeier et al., 1993, 1997; Mengeling et al., 1994).
Successes with vaccinia and avianpox viruses as expression vectors provided further impetus to consider using swinepox virus for the expression of antigens from swine pathogens. Like vaccinia virus, swinepox virus is brick-shaped and its genome consists of a double-stranded DNA of approximately 175 kb (Massung and Moyer, 1991a). It is the only distinct member of the genus Suipoxvirus that has worldwide distribution. As with vaccinia and fowlpox virus, modification of swinepox virus into an expression vector is practical due to certain biological attributes. These include its host specificity, mild pathogenicity, thermo stability, and low transmissibility (Tripathy et al., 1981; House and House, 1992; Tripathy, 1993). The restricted replicative ability of swinepox virus with its attenuated nature provides a safety advantage not only to the recipient but also to nonvaccinated contacts.
In the case of modified viral vectors, the main safety concern has been whether the vector is virulent and capable of producing clinical disease in the host. The development, production, and application of a conventional vaccine is based on several factors: its safety, efficacy, and cost as well as the seriousness of the disease. Because swinepox virus infection is so mild and occurs so rarely, a need to develop a vaccine against it was never realized. Only isolated cases of swinepox have been reported in recent years (Olffumi et al., 1981; Borst et al., 1990) and genetic and antigenic differences of those viruses are not known. Unlike vaccinia virus, very limited basic or applied research had been done on this virus in recent years. However, due to the recent interest in poxviruses as vaccine vectors, some studies on the molecular biology of this virus have been conducted (Massung and Moyer, 1991a,b).
Because of the large size of its genome, like vaccinia virus, swinepox virus can accommodate a significant amount of foreign genetic material with the possibility of developing polyvalent vaccines. A cell line of porcine origin that will support the growth of the swinepox virus has been available for many years. As with other poxvirus vectored vaccines, the antigens expressed by swinepox virus should be properly processed and glycosylated. Finally, the recombinant swinepox virus vectored vaccines would be safer than conventional live vaccines since they will only contain a minor portion of the genome of the foreign pathogen.
So far the only nonessential locus that has been identified in the swinepox virus genome is the TK gene (Schnitzlein and Tripathy, 1991). Since studies with other poxviruses including fowlpox virus (Tripathy and Schnitzlein, 1991; Beard et al., 1991) have shown that following insertional inactivation of TK gene, the recombinant viruses become less pathogenic than the parent virus, the resulting recombinant swinepox virus should become less virulent than the parent virus. Thus, the swine industry would benefit from a new generation of recombinant swinepox virus vectored vaccines because of some desirable features of this virus.
C. Procedure for Creation of Recombinant Swinepox Virus
The basic recombinant DNA techniques used to construct vaccinia virus and fowlpox virus recombinants which have been modified and extended to swinepox virus are briefly described below.
The first step in the production of recombinant viruses is to create plasmids that can direct the insertion of the foreign transcriptional unit(s) into the virus genome. Insertion occurs by homologous recombination and thus requires that the foreign DNA be flanked by a contiguous virus genomic region. In the case of vaccinia and fowlpox virus recombinants, the most commonly used insertion site for foreign genes is the TK gene of these viruses. In this regard, the TK gene of swinepox virus has been identified and sequenced (Schnitzlein and Tripathy, 1991; Feller et al., 1991). This gene (TK) is nonessential for virus replication. The development of swinepox virus as a recombinant vector capable of inducing immunity against various swine pathogens requires that the inserted foreign gene(s) be expressed. Because the transcriptional machinery of the virus will not recognize host—cell promoters, foreign genes much be linked to poxvirus promoters.
Because of the unique and conserved nature of poxvirus transcriptional regulatory elements, two defined vaccinia virus promoters (P11 and P7.5) have been predominantly used in the creation of recombinant vaccinia and avianpox viruses. As with the distantly related fowlpox virus (Tripathy and Wittek, 1990; Schnitzlein and Tripathy, 1990), both promoters are also recognized by swinepox virus. Generally the vaccinia virus late P11 promoter is used to regulate transcription of the marker gene, whereas expression of the other gene (encoding the protective antigen) is controlled by the early–late promoter P7.5. These transcriptional units are positioned immediately adjacent to each other and are flanked by virus DNA sequences to ensure insertion into the virus genome.
Once the plasmid has been generated, it is transfected into pig kidney (PK-15) cells that have been previously infected with swinepox virus. Recombinants are generated by homologous recombination between the replicating swinepox virus genomes and the transfected plasmid as shown schematically in Fig. 1 . Because more than 99% of the progeny from such an infection retain the parental genome, a procedure for the screening/selection of the recombinant progeny is required. One method of identification utilizes the Escherichia coli lacZ gene as a marker. In this case, a chromogenic substrate, 5-bromo–4chloro-3 indolylβ-D-galactosidase (X-gal), which is converted to a blue compound by the action of the expressed enzyme (β-galactosidase) is then used to identify the virus plaques produced by the recombinant virus in the progeny against a background of colorless plaques generated by nonrecombinant viruses. Alternatively, recombinants carrying the E. coli xanthine-guanine phosphoribosyl transferase gene (gpt) as a marker can be selected due to their resistance to mycophenolic acid (Boyle and Coupar, 1988). In addition, recombinant viruses can be identified by plaque hybridization using a DNA probe specific for the inserted foreign gene. Thymidine kinase-deficient (TK–) cells have been used effectively in the selection of recombinant viruses in which foreign genes were introduced within the TK gene. Since a TK– cell line of swine origin which will allow selective growth of the recombinant TK– swinepox virus is not available, inclusion of the marker gene becomes important to facilitate selection/screening of the recombinant virus.
Fig. 1.
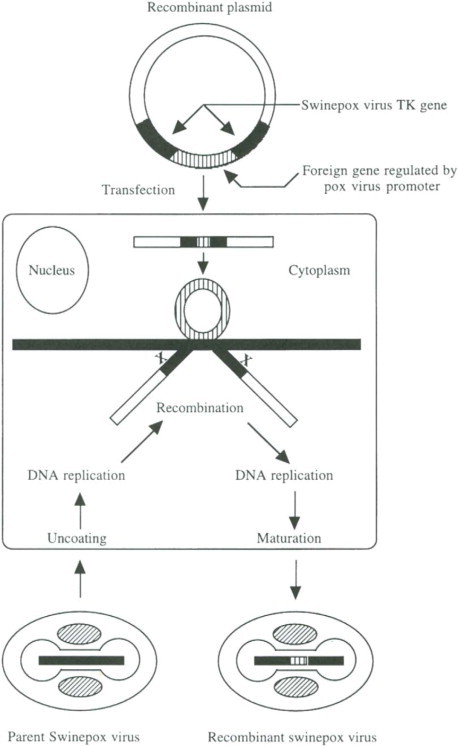
Schematic representation of generation of recombinant swinepox virus.
Regardless, of the identification procedure, virus stocks are prepared from plaques after no wild-type virus can be detected in two consecutive rounds of infection. The production of authentic antigen by the recombinant swinepox virus is verified by using specific antibodies against the respective protein(s) in an immunoprecipitation assay or by detecting immunofluorescence on the surface of cells infected with the recombinant viruses (Fig. 2 ).
Fig. 2.
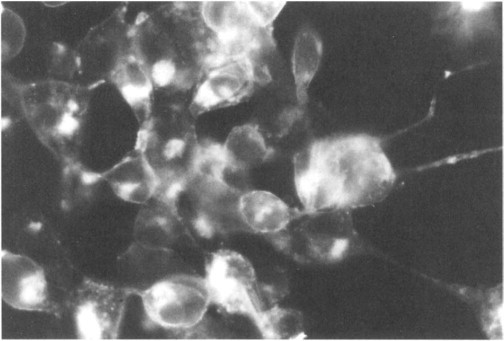
PK-15 cells infected with recombinant swinepox virus expressing gIII glycoprotein of pseudorabies virus. Specific immunofluorescence is observed following reaction with antibody against gIII glycoprotein of pseudorabies virus.
D. Recombinant Swinepox Virus Vaccines
Using the above-mentioned protocols, recombinant swinepox viruses expressing the gp50 or gIII glycoprotein of pseudorabies virus have been generated (Tripathy et al., 1994). These enveloped glycoproteins were chosen due to their importance in the induction of protective immunity. Of the several pseudorabies virus glycoproteins that have been identified, gIII and gp50 have been shown to induce antibodies that neutralize the virus (Marchioli et al., 1987; Zuckermann et al., 1990) and also provide protection from subsequent lethal challenge (Marchioli et al., 1987). Genes encoding these glycoproteins were separately inserted into the swinepox virus genome.
To evaluate the protective ability of recombinant swinepox viruses expressing pseudorabies virus genes, susceptible pigs were vaccinated with either of the two recombinants or a mixture of both. All animals vaccinated with the recombinant viruses were protected when challenged with virulent pseudorbies virus. In contrast, either mortality occurred or severe clinical disease developed in those swine mock vaccinated or inoculated with unaltered swinepox virus (Tripathy et al., 1994). All animals injected with either the unaltered swinepox virus or recombinant swinepox virus developed local lesions at the site of inoculation which regressed within 7–10 days. No secondary lesions were observed in any of the animals. Moreover, the primary lesions resulting from recombinant swinepox virus infection regressed faster than those produced by the parental unmodified swinepox virus. This phenomenon has been observed with other poxviruses in which their TK gene has been insertionally inactivated. Further evidence of protection by recombinant swinepox virus expressing glycoproteins gp50 and gp63 of pseudorabies virus has been reported by van der Leek et al. (1994).
These studies indicate that the swinepox virus vectored recombinant vaccines are capable of expressing the foreign genes to a level that will induce protective immunity. The host specificity of such recombinants should favor their use as ideal immunizing agents for swine.
E. Future of Swinepox Virus Vectored Vaccines
Vaccination against diseases is carried out to limit the economic losses caused by mortality and morbidity and growth retardation in infected animals. The ultimate aim, however, if possible, is to eradicate the disease by regular use of attenuated or inactivated vaccines. In spite of the regular use of conventional vaccines, significant losses do still occur. Therefore, new or improved vaccines are needed for many current and emerging infectious swine diseases which contribute to unnecessary mortality and morbidity and significant economic loss.
A new approach to vaccine development is the expression of genes of foreign pathogens using live attenuated viral vaccines as vectors. In this regard, large size viruses such as pox and herpesviruses have been very promising. However, as mentioned earlier, herpesviruses have the potential of reactivation from latency resulting in recurrent infections and possible shedding of the virus. It is possible that in certain instances in vivo recombination of different vaccine strains may result in the generation of recombinants with virulence greater than that of involved vaccine strains. Vaccinia virus vectored vaccines, on the other hand, have proved to be highly successful immunizing agents, but regular application of such live recombinant vaccinia viruses has been controversial because of minor complications associated with the vaccination program against smallpox. Therefore, the development of live vaccines that are self-restricted, which cannot be transmitted from vaccinated animals to contact animals, and which are not released into the environment would be ideal. Limited experimental studies with TK-inactivated swinepox virus indicate that such live swinepox virus monovalent or polyvalent vaccine vectors can be created.
Lack of adequate local immunity by parenterally administered vaccines against those pathogens whose portal of entry is respiratory or gastrointestinal tract has been realized for a long time. Indeed vaccinia vectored vaccines administered parenterally stimulate protective levels of serum IgG antibody and cellular immunity but do not induce mucosal immunity associated with IgA antibody. Because oral immunization with recombinant vaccinia virus containing the rabies glycoprotein gene provided protection against rabies (Rupprecht et al., 1986), field trials are being conducted with an oral bait for vaccinating wild animals for the control of rabies. In this case, immunization probably occurs due to viral replication in the tonsils. Safety and efficacy of oral vaccination of raccoons by feeding raccoonpox virus containing rabies virus glycoprotein in sponge-baits has been described by Esposito et al. (1988). Enteric immunization of mice with recombinant vaccinia virus containing the influenza virus hemagglutinin gene induced mucosal IgA antibody, serum IgG antibody, and cell-mediated immunity (Meitin et al., 1994). Similarly, mice immunized intragastrically with recombinant vaccinia virus containing the hemagglutinin and nucleoprotein genes of influenza virus were protected against influenza (Bender et al., 1996).
In natural cases of swinepox in neonatal piglets, lesions in the mouth and respiratory tract have been observed indicating that the virus can replicate in sites other than skin. Additionally, antibodies in the sera and antibodies of IgA class in the intestinal contents of swine infected orally with swinepox virus have been observed providing further evidence of virus multiplication after oral administration (Tuboly et al., 1993). This study suggests that swinepox virus can be a potential vector for the expression of antigens from enteric and respiratory viral agents of swine. Use of an oral vaccine for enteric pathogens would not only be convenient for mass administration through the drinking water but less expensive than conventional vaccines. Additionally, swinepox virus could be used in the construction of a multivalent recombinant vaccine because of its ability to accommodate a large amount of foreign genetic material. Alternatively, combined recombinant vaccines expressing different antigens could be used to protect against multiple pathogens. Whether oral or intranasal administration of recombinant swinepox virus vaccine expressing antigens of enteric/respiratory pathogens would induce a desirable protective immune response needs to be determined. If such an effective recombinant vaccine is ever developed, the swine industry would benefit tremendously.
Like avianpox viruses, swinepox virus has never been isolated from any other host than the pig. Interestingly, host-restricted recombinant avianpox viruses have been shown to express foreign antigens in mammalian hosts without producing a productive infection. In a similar manner, if recombinant swinepox viruses containing foreign antigens were able to express them optimally in hosts other than swine without a productive infection, this virus could become a potential candidate for vectored vaccines for a wide range of species including man.
IV. Summary
Several small and large viruses (e.g., adenovirus, poxvirus, and herpesviruses) have been investigated as vaccine vectors. Each viral system has its advantages and disadvantages. One major advantage for viral vector vaccines is their ability to elicit a protective cell-mediated immunity as well as a humoral response to the antigen delivered by the vector. One major problem to using recombinant viruses as vaccines is the pathogenic potential of the parent virus. Therefore, it is important that along with the optimal expression of the foreign genes and ability to provide protection, the pathogenicity of the vector virus must be reduced during genetic manipulation without affecting its multiplication.
The requirements to develop a viral vector, for example, swinepox virus, are a cell culture system that will support the growth of the virus, a suitable nonessential region(s) in the virus genome for insertion of foreign DNA so that virus replication is not affected, a foreign gene(s) that encodes for an immunogenic protein of a swine pathogen, strong transcriptional regulatory elements (promoters) necessary for optimal expression of the foreign genes, a procedure for delivering the foreign gene(s) into the nonessential locus, and a convenient method of distinguishing the recombinant viruses from the parent wild-type virus.
Using this methodology, recombinant swinepox virus vaccines expressing pseudorabies virus antigens have been developed and shown to provide protection against challenge. These studies and evidence of local infection of the oral tract by swinepox virus indicate its potential as a recombinant vector for providing immunity against various swine pathogens including those that infect the respiratory and gastrointestinal tracts.
Acknowledgments
The author would like to thank D. William M. Schnitzlein for helpful suggestions and for Fig. 1.
References
- Beard C.W., Schnitzlein W.M., Tripathy D.N. Protection of chickens against highly pathogenic avian influenza (H5N2) virus by recombinant fowlpox viruses. Avian Dis. 1991;35:356–359. [PubMed] [Google Scholar]
- Becker B.J., Schwartz K.J. Winter. Iowa State University Veterinarian; Ames: 1996. pp. 69–78. (Survey of Porcine Reproductive and Respiratory Syndrome). [Google Scholar]
- Bender B.S., Rowe C.A., Taylor S.F., Wyatt L.S., Moss B., Small P.A., Jr. Oral immunization with a replication-deficient recombinant vaccinia virus protects mice against influenza. J. Virol. 1996;70:6418–6424. doi: 10.1128/jvi.70.9.6418-6424.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borst G.H.A., Kimman T.G., Gielkens A.L.J., van der Kamp J.S. Four sporadic cases of congenital swinepox. Vet. Rec. 1990;127:61–63. [PubMed] [Google Scholar]
- Boyle D.B., Coupar B.E.H. A dominant selectable marker for the construction of recombinant poviruses. Gene. 1988;65:123–128. doi: 10.1016/0378-1119(88)90424-6. [DOI] [PubMed] [Google Scholar]
- Brockmeier S.L., Larger K.M., Tartaglia J., Riviere M., Paoleti E., Mengeling W.L. Vaccination of pigs against pseudorabies with highly attenuated vaccinia (NYVAC) recombinant viruses. Vet. Microbiol. 1993;38:41–58. doi: 10.1016/0378-1135(93)90074-h. [DOI] [PubMed] [Google Scholar]
- Brockmeier S.L., Larger K.M., Mengeling W.L. Successful pseudorabies vaccination in maternally immune piglets using recombinant vaccinia virus vaccines. Res. Vet. Sci. 1997;62:281–285. doi: 10.1016/s0034-5288(97)90205-8. [DOI] [PubMed] [Google Scholar]
- Esposito J., Knight J.C., Shaddock J.H., Novembre F.J., Baer G.M. Successful oral rabies vaccination of raccoons with raccoon poxvirus recombinants expressing rabies virus glycoprotein. Virology. 1988;165:313–316. doi: 10.1016/0042-6822(88)90692-7. [DOI] [PubMed] [Google Scholar]
- Feller J.A., Massung R.F., Turner P.C., Gibbs E.P.J., Bockamp E.O., Beloso A., Talavera A., Vinuela E., Moyer R.W. Isolation and molecular characterization of the swinepox virus thymidine kinase gene. Virology. 1991;183:578–585. doi: 10.1016/0042-6822(91)90986-l. [DOI] [PubMed] [Google Scholar]
- Flexner C., Moss B. Vaccinia virus as a live vector for expression of immunogens. In: Levine M.M., Woodrow G.C., Kaper J.B., Cobon G.S., editors. New Generation of Vaccines. 2nd rev. expanded ed. Dekker; New York: 1997. pp. 297–314. [Google Scholar]
- Gurvich E.B. The age-dependent risk of postvaccination complications in vaccines with smallpox vaccine. Vaccine. 1992;10:96–97. doi: 10.1016/0264-410x(92)90023-d. [DOI] [PubMed] [Google Scholar]
- Halbur P.G., Bush E. Update on abortion storms and sow mortality. Rep.-Swine Health Prod. 1997;5:73. [Google Scholar]
- Hill H.T. Preventing epizootic TGE from becoming enzootic TGE. Vet. Med. 1989;84:432–436. [Google Scholar]
- House J.A., House C.A. In: Diseases of Swine. 7th ed. Leman A.D., Straw B.E., Mengeling W.L., D'Allaire S., Taylor D.J., editors. Iowa State Univ. Press; Ames: 1992. pp. 358–361. [Google Scholar]
- Marchioli C.C., Yancey R.J., Petrovskis E.A., Timmins J.G., Post L.E. Evaluation of pseudorabies virus glycoprotein gp50 as a vaccine for Aujeszky's disease in mice and swine: Expression by vaccinia virus and Chinese hamster ovary cells. J. Virol. 1987;61:3977–3982. doi: 10.1128/jvi.61.12.3977-3982.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Massung R.F., Moyer R.W. The molecular biology of swinepox virus. I. A characterization of the viral DNA. Virology. 1991;180:347–354. doi: 10.1016/0042-6822(91)90039-e. [DOI] [PubMed] [Google Scholar]
- Meitin C.A., Bender B.S., Small P.A., Jr. Enteric immunization of mice against influenza with recombinant vaccinia. Proc. Natl. Acad. Sci. USA. 1994;91:11187–11191. doi: 10.1073/pnas.91.23.11187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mengeling W.L., Brockmeier S.L., Larger K.M. Evaluation of a recombinant vaccinia virus containing pseudorabies (PR) virus glycoprotein genes gp50, gII and gIII as a PR vaccine for pigs. Arch. Virol. 1994;134:259–269. doi: 10.1007/BF01310565. [DOI] [PubMed] [Google Scholar]
- Miller G.Y., Tsai J.S., Foster D.L. Benefit-cost analysis of the national pseudorabies virus eradication program. J. Am. Vet. Med. Assoc. 1996;208:208–213. [PubMed] [Google Scholar]
- Moss B., Flexner C. Vaccinia virus expression vectors. Annu. Rev. Immunol. 1987;5:305–324. doi: 10.1146/annurev.iy.05.040187.001513. [DOI] [PubMed] [Google Scholar]
- Mullan B.P., Davies G.T., Cutler R.S. Simulation of the economic impact of transmissible gastroenteritis on commercial pig production in Australia. Aust. Vet. J. 1994;71:151–154. doi: 10.1111/j.1751-0813.1994.tb03370.x. [DOI] [PubMed] [Google Scholar]
- Olffumi B.E., Ayoade G.O., Ikede B.O., Akpavie S.O. Swinepox in Nigeria. Vet. Rec. 1981;109:278–280. doi: 10.1136/vr.109.13.278. [DOI] [PubMed] [Google Scholar]
- Panicali D., Paoletti E. Construction of poxviruses as cloning vectors: Insertion of the thymidine kinase gene from herpes simplex virus into the DNA of infectious vaccinia virus. Proc. Natl. Acad. Sci. USA. 1982;79:4927–4931. doi: 10.1073/pnas.79.16.4927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pond W.G. Modern pork production. Sci. Am. 1983;248:96–103. [Google Scholar]
- Rupprecht C.E., Wiktor T.J., Johnston D.N., Hamir A.N., Dietzchold B., Wunner, W.H., Glickman L.T., Koprowski H. Oral immunization and protection of raccoons (Procyon lotor) with a vacciniarabies glycoprotein recombinant virus vaccine. Proc. Natl. Acad. Sci. USA. 1986;83:7947–7950. doi: 10.1073/pnas.83.20.7947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saif L.J., Jackwood D.J. Enteric virus vaccines: Theoretical considerations, current status, and future approaches. In: Saif L.J., Theil K., editors. Viral Diarrheas of Man and Animals. CRC Press; Boca Raton, FL: 1989. pp. 313–329. [Google Scholar]
- Saif L.J., Wesley . Transmissible gastroenteritis. In: Leman A.D., Straw B.E., Mengeling W.L., D'Allaire S., Taylor D.J., editors. Diseases of Swine. 7th ed. Iowa State University Press; Ames: 1992. pp. 362–386. [Google Scholar]
- Schnitzlein W.M., Tripathy D.N. Utilization of vaccinia virus promoters by fowlpox virus recombinants. Anim. Biotechnol. 1990;2:164–174. [Google Scholar]
- Schnitzlein W.M., Tripathy D.N. Identification and nucleotide sequences of the thymidine kinase gene of swinepox virus. Virology. 1991;181:727–732. doi: 10.1016/0042-6822(91)90907-s. [DOI] [PubMed] [Google Scholar]
- Tartaglia J., Perkus M.E., Taylor J., Norton E.K., Audonnet J.-C., Cox W.I., Davis S.W., Van der Hoeven J., Meigner B., Riviere M., Languet B., Paoletti E. NYVAC: A highly attenuated strain of vaccinia virus. Virology. 1992;188:217–232. doi: 10.1016/0042-6822(92)90752-b. [DOI] [PubMed] [Google Scholar]
- Taylor J., Weinberg R., Languet B., Desmetire P., Paoletti E. Recombinant fowlpox virus inducing protective immunity in non-avian species. Vaccine. 1988;6:497–503. doi: 10.1016/0264-410x(88)90100-4. [DOI] [PubMed] [Google Scholar]
- Taylor J., Trimarchi C., Weinberg R., Languet B., Guillemin F., Dsmettre P., Paoletti E. Efficacy studies on a canarypox-rabies recombinant virus. Vaccine. 1991;9:190–193. doi: 10.1016/0264-410x(91)90152-v. [DOI] [PubMed] [Google Scholar]
- Tripathy D.N. Swinepox. Curr. Vet. Ther. 1993;3:482–483. [Google Scholar]
- Tripathy D.N. Vol. II. 1996. Fowlpox virus vectored vaccines for control of poultry diseases; pp. 497–503. (Proc. World's Poult. Cong., 20th, New Delhi, India). [Google Scholar]
- Tripathy D.N., Schnitzlein W.M. Expression of avian influenza hemagglutinin gene by recombinant fowlpox virus. Avian Dis. 1991;35:186–191. [PubMed] [Google Scholar]
- Tripathy D.N., Wittek R. Regulation of foreign gene in fowlpox virus by a vaccinia virus promoter. Avian Dis. 1990;34:218–220. [PubMed] [Google Scholar]
- Tripathy D.N., Hanson L.E., Crandell R.A. Poxviruses of veterinary importance, diagnosis of infections. In: Krustak E., editor. Comparative Diagnosis of Viral Diseases. Academic Press; New York: 1981. pp. 267–346. Chapter 6. [Google Scholar]
- Tripathy D.N., Schnitzlein W.M., Zuckermann F.A. Recombinant swinepox viruses expressing pseudorabies virus genes protect swine against virulent pseudorabies virus. Abstr. Int. Conf. Proxviruses and Iridoviruses, 10th; Banff, Alberta, Canada ; 1994. p. 191. April 30–May 5, 1994. [Google Scholar]
- Tuboly T., Nagy E., Derbyshire J.B. Potential viral vectors for the stimulation of mucosal antibody responses against enteric viral antigens in pigs. Res. Vet. Sci. 1993;54:345–350. doi: 10.1016/0034-5288(93)90133-Z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tuboly T., Nagy E., Dennis J.R., Derbyshire J.B. Immunogenicity of the S protein of transmissible gastroenteritis virus expressed in baculovirus. Arch. Virol. 1994;137:55–67. doi: 10.1007/BF01311173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Leek M.L., Feller J.A., Sorensen G., Isaacson W., Adams C.L., Borde D.J., Pfeiffer N., Tran T., Moyer R.W., Gibbs E.P.J. Evaluation of swinepox virus as a vaccine vector in pigs using an Aujeszky's disease (pseudorabies) virus gene insert coding for glyoproteins gp50 and gp63. Vet. Rec. 1994;134:13–18. doi: 10.1136/vr.134.1.13. [DOI] [PubMed] [Google Scholar]
- van Zijl M., Wensvoort G., deKluyver E., Hulst M., van der Gulden H., Gaielkens A., Berns A., Moorman R. Live attenuated pseudorabies virus expressing envelope glycoprotein E1 of hog cholera virus protects swine both against pseudorabies and hog cholera. J. Virol. 1991;65:2761–2765. doi: 10.1128/jvi.65.5.2761-2765.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuckermann F.A., Zsak L., Mettenleiter T.C., Ben-Porat T. Pseudorabies virus glycoprotein III is a major target antigen for murine and swine virus-specific cytotoxic T lymphocytes. J. Virol. 1990;64:802–812. doi: 10.1128/jvi.64.2.802-812.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
Uncited References
- Committee on CSRS Animal Health Research Programs, Board of Agriculture, National Research Council . National Academy Press; Washington, DC: 1986. p. 26. (Animal Health Research Programs of the Cooperative State Research Service, Strengths, Weaknesses, and Opportunities). [Google Scholar]
- Massung R.F., Moyer R.W. The molecular biology of swinepox virus. II. The infectious cycle. Virology. 1991;180:355–364. doi: 10.1016/0042-6822(91)90040-i. [DOI] [PubMed] [Google Scholar]


