Key Concepts
-
•
Community-acquired pneumonia (CAP) is the most common cause of admission of adults in the USA.
-
•
Diagnosis of community-acquired pneumonia is relatively easy in previously healthy patients but may be challenging in those with underlying cardiopulmonary disease or in the elderly.
-
•
The highest yield for diagnostic testing for CAP etiology is in the critically ill and those with risk factors for drug-resistant pathogens.
-
•
The majority of hospitalized CAP patients can be treated with either a respiratory fluoroquinolone or cephalosporin/macrolide combination.
-
•
Alternative antibiotic treatment should be based on presence of multiple risk factors for drug-resistant pathogens (i.e. healthcare-associated pneumonia), specific risks (e.g. travel or zoonotic risks), or unique syndromes (e.g. toxin-mediated community-acquired MRSA syndrome).
-
•
Decisions regarding initial placement in an intensive care unit (ICU) of tenuous CAP patients should be based on the number of minor physiologic factors and laboratory abnormalities associated with risk of subsequent deterioration.
-
•
Number of deaths by pneumonia is decreasing in the world linked to child vaccination of pneumococcus and large-scale use of antibiotics in India/China.
Introduction
Captain of the men of death …
Sir William Osler MD
Community-acquired pneumonia (CAP) is one of the most underappreciated medical illnesses in the USA. The combination of pneumonia and influenza is the ninth leading cause of death overall and the most common cause of infectious death in the USA, causing an estimated 50 000 deaths in 2010.1 This number is likely an underestimate because many deaths caused directly by CAP are coded as sepsis, for which pneumonia is the most common source,2 or attributed to an underlying condition (such as cancer and Alzheimer's disease), for which pneumonia is the terminal event. For example, the proximate cause of death in >40% of patients with dementia is pneumonia.3 Lower respiratory tract infection remains the leading cause of infectious death in the world as well, exceeding deaths from tuberculosis, human immunodeficiency virus (HIV) and malaria combined.4
CAP is the most common reason for hospital admission of adults in the USA.5 It is a common cause of severe complications, including septic shock,2 acute respiratory distress syndrome (ARDS) and acute renal failure. Even in survivors, hospital admission for CAP has been associated with increased subsequent mortality and with accelerated cognitive decline.
CAP is also costly, with the estimated annual cost of CAP in the USA being $10.8 billion.6, 7 Indirect costs are also substantial: CAP is a major cause of work days and days of school lost to illness.
The mortality rate from CAP has changed very little until the last decade. Two factors likely have contributed to this recent decrease: widespread use of conjugate pneumococcal vaccines in children, with a beneficial effect of herd immunity on adults,8 and more extensive management of adults according to guideline-recommended antibiotic therapy.9
Diagnosis
Diagnosis of classic CAP is not difficult in patients without underlying cardiopulmonary disease. A triad of (1) evidence of infection (fever or chills, leukocytosis) with (2) signs or symptoms localizing to the respiratory system (cough, increased sputum production, shortness of breath, chest pain, abnormal pulmonary exam with crackles, signs of consolidation, or finding of a pleural effusion), accompanied by (3) a new or changed radiographic infiltrate, usually accurately defines a patient with CAP. In patients with lung cancer, pulmonary fibrosis or other chronic infiltrative lung diseases, and congestive heart failure (CHF),10 the diagnosis of CAP can be very difficult. Atypical presentations also complicate diagnosis. Confusion may be the only presenting symptom in the elderly, leading to delay in diagnosis.11
The differential diagnosis of CAP (Table 28-1 ) results from either noninfectious inflammatory disorders that also cause radiographic infiltrates or concurrent non-lower respiratory tract infection with other causes of infiltrate. Viral upper respiratory tract infection (URI) in association with worse CHF is probably the most common pneumonia mimic, given the frequency of both disorders.
TABLE 28-1.
Differential Diagnosis of Community-Acquired Pneumonia
| Abnormal CXR | Normal CXR |
|---|---|
| Congestive heart failure* | Acute exacerbation of COPD |
| Aspiration pneumonitis | Influenza |
| Pulmonary infarction | Acute bronchitis |
| Acute exacerbation of pulmonary fibrosis | Pertussis |
| Acute exacerbation of bronchiectasis | Asthma* |
| Acute eosinophilic pneumonia | |
| Hypersensitivity pneumonitis | |
| Pulmonary vasculitis | |
| Cocaine-induced (‘crack lung’) | |
CXR, chest radiograph.
Fever or other signs of infection due to concomitant upper respiratory infection.
Radiology
Radiographic infiltrates may also be subtle: an individual radiologist may miss infiltrates in up to 15% of cases and two radiologists reading the same chest radiograph disagree in 10% of cases.12 Computed tomography (CT) detects alveolar infiltrates in a not inconsequential number of patients with manifestations of CAP but normal chest radiographs.13 The inflammatory reaction caused by antibiotic-induced bacterial lysis and aggressive fluid resuscitation will often unmask these otherwise radiographically occult infiltrates on a subsequent chest radiograph.
The most difficult radiographic challenge is detection of acute pneumonia in the setting of chronic lung disease, such as pulmonary fibrosis, pneumoconioses, bronchiectasis, cystic fibrosis and even emphysema or CHF. Comparison to chest radiographs at a time of clinical stability is very important for these cases. If not available, other clinical manifestations, including use of biomarkers,10 may be required to avoid excessive antibiotic therapy.
The pattern of radiographic infiltrates is occasionally helpful in the differential diagnosis of etiology. Cavitary CAP (Figure 28-1 ) has a limited differential diagnosis (Table 28-2 ), although it varies somewhat by geographic location. Conversely, even though viral or atypical bacterial pneumonia more commonly cause diffuse interstitial infiltrates, this pattern is not distinctive enough to guide antibiotic therapy.
Figure 28-1.
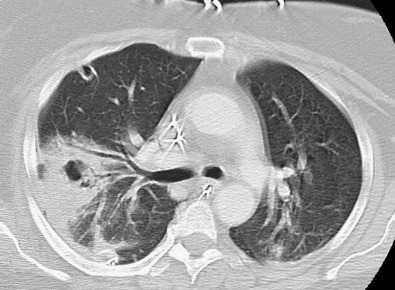
Cavitary pneumonia in an otherwise healthy young adult caused by methicillin-resistant Staphylococcus aureus (MRSA).
TABLE 28-2.
Differential Diagnosis of Cavitary/Necrotizing CAP in Non-immunocompromised Patients
|
Radiographic pattern is also associated with prognosis. Initial presence of bilateral infiltrates is consistently associated with greater mortality and need for ICU care.9 A rapid increase in radiographic infiltrates, whether due to uncontrolled infection or development of ARDS, in the initial 24–48 hours, is also associated with antibiotic failure and need for ICU care.14 Conversely, presence of a pleural effusion has been associated with better prognosis.
Pathophysiology
Pneumonia results from the proliferation of microbial pathogens, most commonly bacteria, but occasionally by viruses, fungi, parasites and other infectious agents, in the alveoli and the host's response to those pathogens. The latter is critically important since recent data have demonstrated the presence of a normal bacterial microbiome in the alveoli.
Bacterial Invasion
Infection of the lower respiratory tract can occur at each level, with a varying proportion of viral and bacterial etiologies at each level, and can be confused with pneumonia. Respiratory bronchiolitis due to respiratory syncytial virus (RSV) in children is a classic example of confusion between CAP and more proximal level infection, with the increased secretions and airway narrowing due to RSV airway infection leading to a radiographic infiltrate from atelectasis. Conversely, infection can progress through the entire respiratory tract, such as influenza URI followed by cough and wheezing from tracheobronchitis, culminating in hypoxemia and infiltrates from influenza pneumonia.
Classically, pneumonia is thought to result from introduction of pathogens into the lower respiratory tract through four pathways. Aspiration from the oropharynx is likely the most common for bacterial pneumonia. Small-volume aspiration occurs frequently during sleep (especially in the elderly) and in patients with decreased levels of consciousness. Viruses and tuberculosis are inhaled as contaminated droplets. Rarely, pneumonia occurs via hematogenous spread (e.g., from tricuspid endocarditis) or by contiguous extension from an infected pleural or mediastinal space.
Recognition that a normal flora exists at the alveolar level of the lung, rather than the distal lung being sterile, raises an alternative mechanism for development of bacterial pneumonia. The normal lung microbiome is similar to that of the normal oropharynx, predominantly streptococci (including the pneumococcus) but also including Haemophilus, Mycoplasma and other CAP pathogens, but at significantly lower concentrations.15, 16 CAP may therefore result from a perturbation in the normal balance, for example a viral URI, resulting in disruption of the balance and outgrowth of a specific species. This hypothesis is very consistent with the frequent association between antecedent or concomitant viral infection and bacterial CAP.
Host Defenses
For pneumonia to occur, lung host defenses must be overcome. The normal lung host defenses are formidable, given that the lung represents the greatest amount of surface area in contact with the external environment and is therefore routinely exposed to infectious micro-organisms. As a result, the lungs and entire respiratory tract have effective and redundant host defense mechanisms in order to respond to this infectious challenge.
Mechanical factors are critically important for inhaled pathogens; the hairs and turbinates of the nares and the branching architecture of the tracheobronchial tree trap microbes on the airway lining, where mucociliary clearance and local antibacterial factors either clear or kill potential pathogens. The gag reflex and cough play major roles in protection from aspiration challenges.
By adhering to mucosal cells of the oropharynx, normal flora prevent attachment of pathogenic bacteria and thereby decrease risk of aspirating these more virulent bacteria. Disruption of the normal microbiome of both oropharynx and lung by antibiotics, viruses, or other factors not only leads to increased risk of pneumonia but also predisposes to more antibiotic-resistant pathogens.
When these mechanical barriers are overcome or when the micro-organisms are small enough to be directly inhaled to the alveolar level, resident alveolar macrophages are extremely efficient at clearing and killing pathogens. Macrophages are assisted by the alveolar epithelial cells, which produce proteins (e.g., surfactant proteins A and D) with opsonic properties or direct antibacterial or antiviral activity. Once engulfed by the macrophage, the pathogens – even if they are not killed – are eliminated via either the mucociliary elevator or the lymphatics.
Only when the capacity of the alveolar macrophages to ingest or kill the micro-organisms is exceeded does clinical pneumonia become manifest. In that situation, the alveolar macrophages initiate the inflammatory response to bolster lower respiratory tract defenses.
Localizing infection to the alveolar space is an important but underappreciated component of host immunity. Factors preventing bacteremia and defending the vascular space are poorly understood. Even the presence of bacterial DNA in peripheral blood appears to correlate with mortality and organ dysfunction.17 Clearly, preformed antibody is important, since the most incontrovertible evidence of pneumococcal vaccine efficacy is prevention of invasive disease, including bacteremia.18 Ability to opsonize bacteria is also important since deficiencies in mannose-binding lectin and complement are also associated with increased bacteremia and invasive pneumococcal disease.19, 20 Splenic clearance of opsonized bacteria is also important for the pneumococcus and other encapsulated bacteria.
Clinical Manifestations
The host inflammatory response, rather than simply proliferation of micro-organisms, triggers the clinical syndrome of pneumonia. Release of inflammatory mediators, such as interleukin (IL)-1 and tumor necrosis factor (TNF), results in fever. Chemokines, such as IL-8 and granulocyte colony-stimulating factor, stimulate bone marrow release of neutrophils and homing to the lung, producing both peripheral leukocytosis and increased purulent secretions. Erythrocytes crossing the alveolar–capillary membrane in the stage of red hepatization result in hemoptysis. Inflammatory mediators released by macrophages and the newly recruited neutrophils cause an alveolar capillary leak equivalent to that demonstrated for ARDS, although initially localized in pneumonia. Radiographic infiltrates and rales detectable on auscultation are a direct result of the alveolar–capillary leak syndrome. Hypoxemia results from alveolar filling but may be exacerbated by paralysis of the hypoxemic vasoconstriction that would normally occur with fluid-filled alveoli by some bacterial pathogens. Increased respiratory drive as part of the systemic inflammatory response syndrome leads to respiratory alkalosis. Decreased compliance due to capillary leak, hypoxemia, increased respiratory drive, increased secretions and occasionally infection-related bronchospasm all lead to dyspnea and, if severe enough, respiratory failure.
Immunocompromise
People with no recognizable defect in any component of host defense can develop CAP. However, the more severe the manifestations and the less virulent the pathogen, the more likely some component of the host defense is deficient. Genetic deficiencies in every component of host defense have been described and the list of primary immunodeficiency syndromes increases yearly.21 Extrinsic factors such as cigarette smoke, alcohol intoxication and particulate matter inhalation can contribute. However, the most important risks are age and co-morbid illnesses, such as diabetes mellitus, CHF, emphysema, cirrhosis and liver failure.22 Even in cases of overt immunocompromise, such as neutropenia from chemotherapy, leukemia and HIV disease, the usual CAP pathogens are still important although the differential of etiologies becomes much larger.
Pathology
The series of pathologic changes seen in classic lobar bacterial pneumonia is described in Table 28-3 . The gray hepatization phase corresponds with successful containment of the infection and improvement in gas exchange, with restoration of the normal hypoxic vasoconstrictor response. This classic pattern does not apply to pneumonia of all etiologies, especially viral or Pneumocystis pneumonia. If microaspiration is the underlying mechanism, a bronchopneumonia pattern is seen and the corresponding phases may not occur.
TABLE 28-3.
Pathologic Phases of Classic Lobar Pneumonia
| Exudative |
|
| Red Hepatization |
|
| Gray Hepatization |
|
| Resolution |
|
Epidemiology
CAP occurs in every ecological niche in the world from the Arctic regions to deserts to jungle, although the most frequent pathogens may vary. Table 28-4 lists geographic and zoonotic considerations for etiology. A general seasonal pattern occurs, with higher rates in the winter/rainy season, tracking most closely with respiratory viruses such as influenza and RSV.
TABLE 28-4.
Zoonoses and Geographic Considerations
| Travel | Pathogen(s) | Exposure | Pathogen(s) |
|---|---|---|---|
| Ohio/Mississippi/St Lawrence river valleys | Histoplasma capsulatum | Bird or bat dung | Histoplasma capsulatum |
| Southwestern USA |
Coccidioides spp. Hantavirus Yersinia pestis |
Pet birds | Chlamydophila psittaci |
| Upper Midwest USA woods | Blastomyces | Rabbits | Francisella tularensis |
| South East Asia |
Burkholderia pseudomalleoli Avian influenza Acinetobacter spp. |
Exposure to sheep, goats, parturient cats | Coxiella burnetii |
| Hotel or cruise ship stay in last 2 weeks | Legionella spp. | Sick dogs | Blastomyces |
CAP occurs in all ages but incidence and mortality are greatest in the extremes of age.23 In infants, lack of humoral immunity to common pathogens such as influenza, RSV and Streptococcus pneumoniae is the major factor. In the elderly, a senescent host immune system and high frequency of co-morbid illnesses play the greatest role. Females are slightly more likely to develop CAP while males are more likely to die from CAP.
In the USA, 80% of CAP patients are treated as outpatients. Of hospitalized patients, 15–20% require ICU monitoring or interventions.
Etiology
The major etiologies of CAP are listed in Table 28-5 . By far, the most common bacterial etiology is Strep. pneumoniae. The actual proportion caused by viruses is difficult to determine since the majority of detections are from the upper respiratory tract, and it is unclear whether the virus present in the oropharynx is causing the pneumonia, predisposed to a superinfection bacterial pneumonia, or is simply an innocent bystander. This dilemma is most obvious for human rhinovirus detection in adults.
TABLE 28-5.
Common Etiologies of CAP*
| BACTERIAL |
|
| VIRAL |
|
| FUNGAL |
|
SARS, severe acute respiratory syndrome; MERS, Middle East respiratory syndrome.
Roughly order of frequency.
Association with CAP but unclear causality.
Less common etiologies are usually associated with specific geographic areas or exposure to specific zoonoses (see Table 28-4). Occasionally, more chronic pulmonary infections can masquerade as acute CAP (Table 28-6 ) and should be considered in endemic areas and if the time course is more indolent.24
TABLE 28-6.
Chronic Pulmonary Infections that May Present as Acute Pneumonia
|
Healthcare-Associated Pneumonia (HCAP)
Concern has been raised about community-onset pneumonia caused by pathogens usually associated with hospital-acquired pneumonia or even ventilator-associated pneumonia, including methicillin-resistant Staphylococcus aureus (MRSA) and multidrug resistant (MDR) gram-negative pathogens.9, 25, 26, 27 Several community-onset pneumonia syndromes at risk for more drug-resistant pathogens can be defined (Table 28-7 ). In the USA, transfer of hospitalized patients to long-term ventilator-weaning facilities or acute rehabilitation institutes, rather than completing their recovery in an acute care hospital, does not decrease their risk of the typical hospital-acquired pathogens. These patients have previously been lumped together with those in lower-risk settings such as nursing homes and chronic dialysis units. An episode of aspiration weeks prior to presentation without intervening medical attention is the classic predisposition for anaerobic pneumonia, often complicated by empyema as well. Otherwise, anaerobes play a minor role in usual CAP.
TABLE 28-7.
Community-Onset Pneumonia Syndromes in Special Populations
| Syndrome | Examples |
|---|---|
| Hospital-acquired | Recent discharge, long-term weaning facilities, rehabilitation institutes |
| Healthcare-associated | Nursing homes, chronic hemodialysis |
| Immunocompromised | Chemotherapy, HIV disease, transplant, acute leukemia/lymphoma |
| Aspiration | Severe alcoholism, seizure disorder, stroke |
HCAP was proposed as a discrete entity with the goal of identifying those patients who were more likely to receive initially inappropriate antibiotic therapy, and have an associated higher mortality risk.27, 28 While early observational studies of culture-positive cases suggest improved outcome from broad-spectrum antibiotic therapy in persons with HCAP risk factors,27, 28 prospective studies using the same definition find lower rates of antibiotic-resistant pathogens and many culture-negative cases.26, 29, 30 Even more concerning were reports of adverse outcomes among persons with HCAP risk factors treated with broad-spectrum antibiotic therapy.26, 31
Rather than using the original definition derived from healthcare-associated bacteremia, a prospective multicenter study identified six independent risk factors (Table 28-8 ) for pneumonia caused by pathogens resistant to the usual inpatient antibiotic regimens recommended by Infectious Diseases Society of America (IDSA)/ American Thoracic Society (ATS) guidelines.26 While the risk factors were similar to the original, the incidence of drug-resistant pathogens was not significantly increased until three or more risk factors are present. A separate analysis specifically for MRSA found that presence of one MRSA-specific risk factor (prior MRSA infection/colonization, chronic hemodialysis, or heart failure) and another pneumonia-specific risk factor may warrant MRSA coverage (but not dual anti-pseudomonal antibiotics). Importantly, this new definition would result in significantly fewer patients receiving broad-spectrum antibiotics than the original HCAP definition.9
TABLE 28-8.
Criteria for Healthcare-Associated Pneumonia (HCAP)
| Original Criteria9 | Pneumonia-Specific Criteria26 |
|---|---|
| Hospitalization for ≥2 days in previous 90 days | Hospitalization for ≥2 days in previous 90 days |
| Nursing home or extended care facility residents | Antibiotics in previous 90 days |
| Chronic home infusion therapy | Non-ambulatory status |
| Chronic dialysis within 30 days | Tube feedings |
| Home wound care | Immunocompromise |
| Family member with MDR pathogen |
Gastric acid suppressive agents |
| Immunosuppressive disease/therapy* | |
Not included in original criteria but frequently included in many HCAP studies.
Community-Acquired MRSA CAP
The MRSA identified in patients with HCAP risk factors is likely a hospital-acquired strain. However, in the USA a specific USA300 strain of MRSA causes CAP in previously healthy patients, specifically without HCAP or other risk factors for MDR pathogens.32, 33 Many of the characteristic presenting features of this MRSA strain (Table 28-9 ), as well as the methicillin-sensitive variant, are a result of exotoxin production.32 The Panton–Valentine Leukocidin (PVL) gene is an efficient marker of toxigenic strains but is not the main exotoxin involved in the increased lethality.33 The USA300 strain is increasingly being found in hospital-acquired MRSA infections, blurring some of the epidemiologic distinctions.
TABLE 28-9.
Clinical Features Suggesting Community-Acquired MRSA Pneumonia
| Cavitary infiltrate or necrosis | Neutropenia |
| Rapidly increasing pleural effusion | Erythematous skin rash |
| Gross hemoptysis (not just blood-streaked) | Skin pustules |
| Concurrent influenza | Young, previously healthy |
| Severe CAP in summer months | |
Determination of Etiology
While the diagnosis of CAP is relatively straightforward, determination of etiology is very difficult.9 Even with aggressive use of currently available diagnostic tests, the etiology remains unknown in >50% of cases.
A complete history of travel, pets and hobbies is critical for suspicion of the less common pathogens (see Table 28-4), as well as CAP mimics (see Table 28-1). Unfortunately, diagnosis of many of these pathogens requires acute and convalescent serology or tests sent to a reference laboratory, making most treatment empirical.
In general, the greater the likelihood of unusual bacterial pathogens, the greater the yield of diagnostic tests. Patients with severe CAP requiring ICU admission34 and/or HCAP risk factors26 started on broad-spectrum antibiotics have the clearest indication for extensive diagnostic testing, including attempts at obtaining sputum culture. The yield of testing is higher in the critically ill CAP patient, possibly because endotracheal intubation allows direct sampling of the lower respiratory tract. Other indications and the corresponding appropriate tests are listed in Table 28-10 .9
TABLE 28-10.
Indications for More Aggressive Diagnostic Testing in Cap9
| ICU admission | Cirrhosis/severe chronic liver disease* |
| HCAP risk factors26 | Severe chronic obstructive lung disease† |
| Failure of outpatient antibiotic therapy | Asplenia (anatomic and functional)* |
| Cavitary infiltrates on presentation | Recent travel (within 2 weeks)‡ |
| Leukopenia | Positive Legionella or pneumococcal urinary antigen test† |
| Active alcohol abuse | Pleural effusion |
Mainly blood cultures.
Mainly respiratory sample.
Legionella urinary antigen.
Biomarkers have been used in an attempt to differentiate viral from bacterial pneumonia. The best validated is procalcitonin (PCT).10, 35 This pro-hormone is elevated in uncontrolled bacterial infections and actively suppressed by the interferon response induced in many viral pneumonias. However, PCT may be low in atypical pathogen CAP as well and is clearly elevated in severe viral CAP, such as seen in the 2009–2010 influenza A pandemic, with or without evidence of superimposed bacterial pneumonia. C-reactive protein (CRP) is more nonspecific than PCT in CAP but may be a better predictor of treatment failure.14
Treatment
Almost every antibiotic approved by the US Food and Drug Administration in the past four decades has an indication for CAP. In general, keys to appropriate therapy are adequate coverage of Strep. pneumoniae and the atypical bacterial pathogens (Mycoplasma, Chlamydophila, Legionella). The recommended regimens from the IDSA/ATS guideline are listed in Table 28-11 .9 European guidelines differ in that β-lactam antibiotics (typically amoxicillin) remain the recommended agent for mild–moderate CAP.36, 37 A recent study from the Netherlands suggests that a strategy of empirical treatment for moderately severe CAP with β-lactam monotherapy is noninferior to either β-lactam–macrolide combination therapy or fluoroquinolone monotherapy.38 The primary factors to discriminate among the antibiotic options, therefore, should be local resistance patterns in community organisms, recent antibiotic use, which increases the risk of class resistance,39 and cost.
TABLE 28-11.
IDSA/ATS Recommended Empirical Antibiotic Therapy9
| Disposition | Recommended Class | Typical Examples |
|---|---|---|
| Outpatient | Macrolide | Azithromycin 500 mg po once, then 250 mg q day |
| Doxycycline | Clarithromycin 500 mg po BD | |
| Recent oral antibiotics | Change antibiotic class Consider: Fluoroquinolone Amoxicillin ± clavulanate |
|
| Non-ICU inpatient |
Respiratory fluoroquinolone | Moxifloxacin 400 mg q day or |
| Levofloxacin 750 mg po q day | ||
| or | Ceftriaxone 1–2 g q day or | |
| β-lactam and macrolide |
Ampicillin–sulbactam 2 g iv q8h plus | |
| Azithromycin 500 mg q day | ||
| ICU patient | Ceftriaxone plus Azithromycin or Respiratory fluoroquinolone |
|
Since outpatient treatment failure is rare and the guideline-compliant therapy covers 90% of etiologies in hospitalized patients, deviation from these guidelines should have appropriate justification. Presence of risk factors for MDR (see Table 28-8) or zoonotic/geographic-specific pathogens (see Table 28-4) may justify alternative empirical coverage but should be accompanied by aggressive attempts at diagnosis, in order to appropriately de-escalate broader-spectrum antibiotic therapy.26, 40 Quality improvement projects consistently show that as compliance with IDSA/ATS guideline antibiotics increases, mortality rates and length of stay decrease.41, 42 Conversely, continuing broad-spectrum antibiotics for CAP patients without documented MDR pathogens is associated with excess mortality.26, 31
Macrolides appear to have beneficial effects in excess of their coverage of atypical pathogens, especially in the more severely ill patient.43, 44 These benefits may be due to immunomodulatory effects on the host, less cell lysis-induced cytokine release, or inhibition of bacterial virulence factors, such as biofilms, quorum sensing and toxin production.
CA-MRSA would require specific coverage since the regimens in Table 28-11 have inadequate MRSA coverage. For patients with HCAP-MRSA risk factors, linezolid has a 15% better clinical response rate than vancomycin.45 Because manifestations of the USA300 strain of CA-MRSA CAP are disproportionately exotoxin-mediated (see Table 28-7),32 treatment with antibiotics that suppress toxin production, such as linezolid or clindamycin (added to vancomycin), are preferred and have been associated with lower mortality.33 Ceftaroline, the only antibiotic approved for CAP recently, has MRSA activity as well.
One of the most critical elements of treatment is early initiation of appropriate antibiotic therapy after the diagnosis of CAP has been made. The first dose should be given in the emergency department (ED) to allow closer monitoring of the initial response and to assure that the initial dose is given promptly.9 Timing of the first dose is even more important when the patient presents with septic shock; the goal should be initial antibiotic within the first hour.46
For uncomplicated bacterial CAP, the usual duration of treatment should be 5–7 days. Certain pathogens, such as Legionella, may require up to 2 weeks of therapy. Conversion to an equivalent oral agent is appropriate whenever the patient is clinically improving and able to tolerate food.
Influenza Treatment
Treatment of influenza pneumonia has not been prospectively studied specifically. Experience during the 2009–10 pandemic and retrospective analysis47 suggest that antivirals should be used if a patient has a radiographic infiltrate, no matter the duration of symptoms. The potential for oseltamivir-resistant strains should be monitored from CDC and local health department information as each influenza season progresses. The major issue is whether antibiotics are always needed for influenza CAP, with no clear data or consensus. For a full description of the use of antiviral therapy, see Chapter 154.
Other Management
Disposition
The major determinant of the cost of CAP care is the physician's decision to hospitalize. Of CAP patients who present to the ED, 40–60% are admitted,22, 48, 49 with considerable variability in admission for patients with similar clinical characteristics. Use of scoring systems, such as the Pneumonia Severity Index (PSI)22 and the CURB-65 Score50 that were developed specifically to guide admission decisions, result in fewer admissions of low acuity patients with no increase in adverse outcomes.48 PSI is a complex score, requiring formal scoring or electronic decision support whereas CURB-65 (confusion, uremia, respiratory rate, blood pressure, age >65 years) is both easy to remember and calculate, although not as well validated as PSI. Both scores are valid for analysis of groups of CAP admissions, but admission of low score patients is legitimate, for both objective reasons (e.g. low arterial saturations) and subjective (e.g. unreliable home support, concern regarding compliance).
Decisions regarding initial ICU placement of tenuous CAP patients probably have the greatest potential impact on mortality. Patients transferred to the ICU within 48 hours of initial admission to a general medical service have higher mortality than those with an obvious need for ICU care (mechanical ventilation or hypotension requiring vasopressors) at the time of admission.49, 51, 52 The fraction of hospitalized pneumonia patients admitted to the ICU also varies widely (5–20%) depending on hospital and health system characteristics.49, 53, 54, 55
The IDSA/ATS guidelines suggest that presence of >3 of a group of nine minor criteria (Table 28-12 ) warrant consideration for ICU admission.9 Other scores to predict clinical deterioration with similar parameters have also been developed and validated.53, 54, 55 For each, the probability of need for invasive ventilatory or vasopressor therapy increases with increasing number of criteria or points, with a threshold score around three to consider ICU admission. All these ICU admission scores are overly sensitive, resulting in substantially more ICU admissions if followed rigidly.9, 49 The most appropriate use of these scores may be to focus attention on patients with high scores while still in the ED. A quality-improvement study demonstrated that increased attention in the ED to patients with >3 IDSA/ATS minor criteria resulted in decreased mortality (23.4% to 5.7%) and fewer floors to ICU transfers (32.0% to 14.8%) without significantly increasing direct ICU admissions.49
TABLE 28-12.
Minor Criteria for Consideration of ICU Admission for Severe CAP
| IDSA/ATS Criteria9 | Other Criteria53–55 |
|---|---|
| Confusion | Lactic acidosis |
| Uremia (BUN >20 mg/dL) | pH <7.30–7.35 |
| Tachypnea (RR >30/min) | Low albumin |
| Bilateral radiographic infiltrates | Hyponatremia (<130 mEq/L) |
| Severe hypoxemia (P/F <250) | Leukocytosis >20 x109/L |
| Thrombocytopenia | Hypoglycemia |
| Hypotension requiring aggressive fluid resuscitation | |
| Hypothermia | |
| Leukopenia | |
BUN, blood urea nitrogen; RR, respiratory rate; P/F, PaO2/FiO2ratio.
Pleural Effusion
A new pleural effusion in a patient admitted with CAP should always prompt concern for empyema or complicated parapneumonic effusion (generally pleural fluid pH <7.2). Early diagnosis by thoracentesis, placement of a chest tube and use of tissue plasminogen activator combined with DNAase can prevent the need for surgical intervention in the majority of cases.56 Management of pleural effusions in patients with CHF and intermittent pleural effusions is less straightforward but thoracentesis in all unclear situations is warranted.
Adjunctive Treatment
Some patients benefit from aerosolized β-agonist bronchodilators for wheezing or other bronchial hygiene maneuvers for difficult expectoration. Patients with viral lower respiratory tract infections occasionally require anticholinergic aerosols to control nonproductive cough.
Use of systemic corticosteroids in CAP patients who have no other indication, e.g. asthma or COPD exacerbation associated with pneumonia, remains controversial. In moderate disease, a potential benefit of shortening hospitalization is counterbalanced by an increased risk of superinfection.57 In severe viral pneumonia, either SARS or the 2009–2010 influenza pandemic,58 steroid use was associated with worse outcomes.
Exacerbation of Co-Morbid Illnesses
As mentioned, CAP can exacerbate underlying chronic illnesses such as asthma and COPD, diabetes mellitus and CHF. Up to 15–20% of patients admitted with pneumococcal CAP can have a new cardiovascular diagnosis during the acute hospitalization, including acute myocardial infarction, atrial fibrillation and other arrhythmias, or CHF.59 Destabilization of co-morbid illness is more likely to cause hospital readmission than complications of CAP or its treatment.
Prevention
The main CAP preventive measures are vaccination and smoking cessation.9 Even among patients without obstructive lung disease, smokers are at increased risk of pneumococcal CAP.
Influenza Vaccination
Two forms of influenza vaccine are available – intramuscular inactivated influenza vaccine and intranasal live-attenuated cold-adapted influenza vaccine. The latter is contraindicated in immunocompromised patients. Specific vaccine components are reassessed yearly based on the main circulating strains in the opposite hemisphere. In the event of an influenza outbreak, unprotected patients at risk from complications should be vaccinated immediately and given chemoprophylaxis with oseltamivir for 2 weeks, at which time vaccine-induced antibody levels should be sufficiently protective.
Pneumococcal Vaccine
A pneumococcal polysaccharide vaccine (PPV23) and a protein conjugate vaccine (PCV13) are both available in the USA. The vaccine efficacy of PPV23 has been questionable, particularly in the elderly and other at-risk populations. Administration of the protein conjugate vaccine to children has led to an overall decrease in the prevalence of antimicrobial-resistant pneumococci and in the incidence of invasive pneumococcal disease among both children and adults.18, 60 However, vaccination may result in replacement of vaccine serotypes with nonvaccine serotypes, as was seen with serotypes 19A and 35B after introduction of the original 7-valent conjugate vaccine.61 The 13-valent conjugate vaccine is now also recommended for the elderly and for younger immunocompromised patients (see also Chapter 177).
![]() References available online at
expertconsult.com
.
References available online at
expertconsult.com
.
Key References
- Americam Thoracic Society, Infectious Diseases of America Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2005;171(4):388–416. doi: 10.1164/rccm.200405-644ST. [DOI] [PubMed] [Google Scholar]
- Attridge R.T., Frei C.R., Restrepo M.I. Guideline-concordant therapy and outcomes in healthcare-associated pneumonia. Eur Respir J. 2011;38:878–887. doi: 10.1183/09031936.00141110. [DOI] [PubMed] [Google Scholar]
- Charles P.G., Wolfe R., Whitby M. SMART-COP: a tool for predicting the need for intensive respiratory or vasopressor support in community-acquired pneumonia. Clin Infect Dis. 2008;47:375–384. doi: 10.1086/589754. [DOI] [PubMed] [Google Scholar]
- Charlson E.S., Bittinger K., Haas A.R. Topographical continuity of bacterial populations in the healthy human respiratory tract. Am J Respir Crit Care Med. 2011;184:957–963. doi: 10.1164/rccm.201104-0655OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dean N.C., Bateman K.A., Donnelly S.M. Improved clinical outcomes with utilization of a community-acquired pneumonia guideline. Chest. 2006;130:794–799. doi: 10.1378/chest.130.3.794. [DOI] [PubMed] [Google Scholar]
- Fine M.J., Auble T.E., Yealy D.M. A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med. 1997;336:243–250. doi: 10.1056/NEJM199701233360402. [DOI] [PubMed] [Google Scholar]
- Griffin M.R., Zhu Y., Moore M.R. U.S. hospitalizations for pneumonia after a decade of pneumococcal vaccination. N Engl J Med. 2013;369:155–163. doi: 10.1056/NEJMoa1209165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim H.F., Phua J., Mukhopadhyay A. IDSA/ATS minor criteria aided pre-ICU resuscitation in severe community-acquired pneumonia. Eur Respir J. 2014;43:852–862. doi: 10.1183/09031936.00081713. [DOI] [PubMed] [Google Scholar]
- Maisel A., Neath S.X., Landsberg J. Use of procalcitonin for the diagnosis of pneumonia in patients presenting with a chief complaint of dyspnoea: results from the BACH (Biomarkers in Acute Heart Failure) trial. Eur J Heart Fail. 2012;14:278–286. doi: 10.1093/eurjhf/hfr177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mandell L.A., Wunderink R.G., Anzueto A. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44(Suppl. 2):S27–S72. doi: 10.1086/511159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Musher D.M., Rueda A.M., Kaka A.S. The association between pneumococcal pneumonia and acute cardiac events. Clin Infect Dis. 2007;45:158–165. doi: 10.1086/518849. [DOI] [PubMed] [Google Scholar]
- Rahman N.M., Maskell N.A., West A. Intrapleural use of tissue plasminogen activator and DNase in pleural infection. N Engl J Med. 2011;365:518–526. doi: 10.1056/NEJMoa1012740. [DOI] [PubMed] [Google Scholar]
- Rello J., Lisboa T., Lujan M. Severity of pneumococcal pneumonia associated with genomic bacterial load. Chest. 2009;136:832–840. doi: 10.1378/chest.09-0258. [DOI] [PubMed] [Google Scholar]
- Shindo Y., Ito R., Kobayashi D. Risk factors for drug-resistant pathogens in community-acquired and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2013;188:985–995. doi: 10.1164/rccm.201301-0079OC. [DOI] [PubMed] [Google Scholar]
References
- 1.Murphy S.L., Xu J., Kochanek K.D. Deaths: Preliminary data for 2010. Nat Vital Stat Rep. 2012;60(4):1–51. [PubMed] [Google Scholar]
- 2.Angus D.C., Linde-Zwirble W.T., Lidicker J. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med. 2001;29:1303–1310. doi: 10.1097/00003246-200107000-00002. [DOI] [PubMed] [Google Scholar]
- 3.Mitchell S.L., Teno J.M., Kiely D.K. The clinical course of advanced dementia. N Engl J Med. 2009;361:1529–1538. doi: 10.1056/NEJMoa0902234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.World Health Organization The top 10 causes of death. www.who.int/mediacentre/factsheets/fs310/en/index.html Fact sheet No. 310 updated June 2011. Geneva: WHO.; Available.
- 5.Pfuntner A., Wier L.M., Stocks C. Most frequent conditions in U.S. hospitals, 2011: Statistical brief No. 162. Healthcare Cost and Utilization Project (HCUP) statistical briefs. 2013. www.ncbi.nlm.nih.gov/pubmed/24228292 Rockville, MD: Agency for Healthcare Research and Quality; Available.
- 6.Angus D.C., Marrie T.J., Obrosky D.S. Severe community-acquired pneumonia: use of intensive care services and evaluation of American and British Thoracic Society Diagnostic criteria. Am J Respir Crit Care Med. 2002;166:717–723. doi: 10.1164/rccm.2102084. [DOI] [PubMed] [Google Scholar]
- 7.Pfuntner A., Wier L.M., Steiner C. Costs for hospital stays in the United States, 2011: Statistical brief No. 168. Healthcare Cost and Utilization Project (HCUP) statistical briefs. 2013. www.ncbi.nlm.nih.gov/pubmed/24455786 Rockville, MD: Agency for Healthcare Research and Quality; Available.
- 8.Griffin M.R., Zhu Y., Moore M.R. U.S. hospitalizations for pneumonia after a decade of pneumococcal vaccination. N Engl J Med. 2013;369:155–163. doi: 10.1056/NEJMoa1209165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mandell L.A., Wunderink R.G., Anzueto A. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44(Suppl. 2):S27–S72. doi: 10.1086/511159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Maisel A., Neath S.X., Landsberg J. Use of procalcitonin for the diagnosis of pneumonia in patients presenting with a chief complaint of dyspnoea: results from the BACH (Biomarkers in Acute Heart Failure) trial. Eur J Heart Fail. 2012;14:278–286. doi: 10.1093/eurjhf/hfr177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Waterer G.W., Kessler L.A., Wunderink R.G. Delayed administration of antibiotics and atypical presentation in community-acquired pneumonia. Chest. 2006;130:11–15. doi: 10.1378/chest.130.1.11. [DOI] [PubMed] [Google Scholar]
- 12.Albaum M.N., Hill L.C., Murphy M. Interobserver reliability of the chest radiograph in community-acquired pneumonia. Chest. 1996;110:343–350. doi: 10.1378/chest.110.2.343. [DOI] [PubMed] [Google Scholar]
- 13.Self W.H., Courtney D.M., McNaughton C.D. High discordance of chest X-ray and computed tomography for detection of pulmonary opacities in ED patients: implications for diagnosing pneumonia. Am J Emerg Med. 2013;31:401–405. doi: 10.1016/j.ajem.2012.08.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Arancibia F., Ewig S., Martinez J.A. Antimicrobial treatment failures in patients with community-acquired pneumonia: causes and prognostic implications. Am J Respir Crit Care Med. 2000;162:154–160. doi: 10.1164/ajrccm.162.1.9907023. [DOI] [PubMed] [Google Scholar]
- 15.Charlson E.S., Bittinger K., Haas A.R. Topographical continuity of bacterial populations in the healthy human respiratory tract. Am J Respir Crit Care Med. 2011;184:957–963. doi: 10.1164/rccm.201104-0655OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Erb-Downward J.R., Thompson D.L., Han M.K. Analysis of the lung microbiome in the ‘healthy’ smoker and in COPD. PLoS ONE. 2011;6:e16384. doi: 10.1371/journal.pone.0016384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rello J., Lisboa T., Lujan M. Severity of pneumococcal pneumonia associated with genomic bacterial load. Chest. 2009;136:832–840. doi: 10.1378/chest.09-0258. [DOI] [PubMed] [Google Scholar]
- 18.Whitney C.G., Farley M.M., Hadler J. Decline in invasive pneumococcal disease after the introduction of protein-polysaccharide conjugate vaccine. N Engl J Med. 2003;348:1737–1746. doi: 10.1056/NEJMoa022823. [DOI] [PubMed] [Google Scholar]
- 19.Skattum L., van Deuren M., van der Poll T. Complement deficiency states and associated infections. Mol Immunol. 2011;48:1643–1655. doi: 10.1016/j.molimm.2011.05.001. [DOI] [PubMed] [Google Scholar]
- 20.Garcia-Laorden M.I., Sole-Violan J., Rodriguez de Castro F. Mannose-binding lectin and mannose-binding lectin-associated serine protease 2 in susceptibility, severity, and outcome of pneumonia in adults. J Allergy Clin Immunol. 2008;122:368–374. doi: 10.1016/j.jaci.2008.05.037. 74 e1-2. [DOI] [PubMed] [Google Scholar]
- 21.Waterer G.W., Wunderink R.G. Genetic susceptibility to pneumonia. Clin Chest Med. 2005;26:29–38. doi: 10.1016/j.ccm.2004.10.002. [DOI] [PubMed] [Google Scholar]
- 22.Fine M.J., Auble T.E., Yealy D.M. A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med. 1997;336:243–250. doi: 10.1056/NEJM199701233360402. [DOI] [PubMed] [Google Scholar]
- 23.Marston B.J., Plouffe J.F., File T.M., Jr Incidence of community-acquired pneumonia requiring hospitalization. Results of a population-based active surveillance Study in Ohio. The Community-Based Pneumonia Incidence Study Group. Arch Intern Med. 1997;157(15):1709–1718. [PubMed] [Google Scholar]
- 24.Chang D.C., Anderson S., Wannemuehler K. Testing for coccidioidomycosis among patients with community-acquired pneumonia. Emerg Infect Dis. 2008;14:1053–1059. doi: 10.3201/eid1407.070832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Americam Thoracic Society, Infectious Diseases of America Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2005;171(4):388–416. doi: 10.1164/rccm.200405-644ST. [DOI] [PubMed] [Google Scholar]
- 26.Shindo Y., Ito R., Kobayashi D. Risk factors for drug-resistant pathogens in community-acquired and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2013;188:985–995. doi: 10.1164/rccm.201301-0079OC. [DOI] [PubMed] [Google Scholar]
- 27.Kollef M.H., Shorr A., Tabak Y.P. Epidemiology and outcomes of health-care-associated pneumonia: results from a large US database of culture-positive pneumonia. Chest. 2005;128:3854–3862. doi: 10.1378/chest.128.6.3854. [DOI] [PubMed] [Google Scholar]
- 28.Micek S.T., Kollef K.E., Reichley R.M. Health care-associated pneumonia and community-acquired pneumonia: a single-center experience. Antimicrob Agents Chemother. 2007;51:3568–3573. doi: 10.1128/AAC.00851-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Labelle A.J., Arnold H., Reichley R.M. A comparison of culture-positive and culture-negative health-care-associated pneumonia. Chest. 2010;137:1130–1137. doi: 10.1378/chest.09-1652. [DOI] [PubMed] [Google Scholar]
- 30.Chalmers J.D., Taylor J.K., Singanayagam A. Epidemiology, antibiotic therapy, and clinical outcomes in health care-associated pneumonia: a UK cohort study. Clin Infect Dis. 2011;53:107–113. doi: 10.1093/cid/cir274. [DOI] [PubMed] [Google Scholar]
- 31.Attridge R.T., Frei C.R., Restrepo M.I. Guideline-concordant therapy and outcomes in healthcare-associated pneumonia. Eur Respir J. 2011;38:878–887. doi: 10.1183/09031936.00141110. [DOI] [PubMed] [Google Scholar]
- 32.Gillet Y., Vanhems P., Lina G. Factors predicting mortality in necrotizing community-acquired pneumonia caused by Staphylococcus aureus containing Panton–Valentine leukocidin. Clin Infect Dis. 2007;45:315–321. doi: 10.1086/519263. [DOI] [PubMed] [Google Scholar]
- 33.Sicot N., Khanafer N., Meyssonnier V. Methicillin resistance is not a predictor of severity in community-acquired Staphylococcus aureus necrotizing pneumonia – results of a prospective observational study. Clin Microbiol Infect. 2013;19:E142–E148. doi: 10.1111/1469-0691.12022. [DOI] [PubMed] [Google Scholar]
- 34.van der Eerden M.M., Vlaspolder F., de Graaff C.S. Comparison between pathogen directed antibiotic treatment and empirical broad spectrum antibiotic treatment in patients with community acquired pneumonia: a prospective randomised study. Thorax. 2005;60:672–678. doi: 10.1136/thx.2004.030411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Christ-Crain M., Stolz D., Bingisser R. Procalcitonin guidance of antibiotic therapy in community-acquired pneumonia: a randomized trial. Am J Respir Crit Care Med. 2006;174:84–93. doi: 10.1164/rccm.200512-1922OC. [DOI] [PubMed] [Google Scholar]
- 36.Lim W.S., Baudouin S.V., George R.C. BTS guidelines for the management of community acquired pneumonia in adults: update 2009. Thorax. 2009;64(Suppl.3):iii1–iii55. doi: 10.1136/thx.2009.121434. [DOI] [PubMed] [Google Scholar]
- 37.Wiersinga W.J., Bonten M.J., Boersma W.G. SWAB/NVALT (Dutch Working Party on Antibiotic Policy and Dutch Association of Chest Physicians) guidelines on the management of community-acquired pneumonia in adults. Neth J Med. 2012;70:90–101. [PubMed] [Google Scholar]
- 38.Postma D.F., van Werkhoven C.H., van Elden L. Antibiotic treatment strategies for community-acquired pneumonia in adults. N Engl J Med. 2015;372:1312–1323. doi: 10.1056/NEJMoa1406330. [DOI] [PubMed] [Google Scholar]
- 39.Vanderkooi O.G., Low D.E., Green K. Predicting antimicrobial resistance in invasive pneumococcal infections. Clin Infect Dis. 2005;40:1288–1297. doi: 10.1086/429242. [DOI] [PubMed] [Google Scholar]
- 40.Wunderink R.G. Community-acquired pneumonia versus healthcare-associated pneumonia: the returning pendulum. Am J Respir Crit Care Med. 2013;188:896–898. doi: 10.1164/rccm.201308-1536ED. [DOI] [PubMed] [Google Scholar]
- 41.Capelastegui A., Espana P.P., Quintana J.M. Improvement of process-of-care and outcomes after implementing a guideline for the management of community-acquired pneumonia: a controlled before-and-after design study. Clin Infect Dis. 2004;39:955–963. doi: 10.1086/423960. [DOI] [PubMed] [Google Scholar]
- 42.Dean N.C., Bateman K.A., Donnelly S.M. Improved clinical outcomes with utilization of a community-acquired pneumonia guideline. Chest. 2006;130:794–799. doi: 10.1378/chest.130.3.794. [DOI] [PubMed] [Google Scholar]
- 43.Sligl W.I., Asadi L., Eurich D.T. Macrolides and mortality in critically ill patients with community-acquired pneumonia: a systematic review and meta-analysis. Crit Care Med. 2014;42:420–432. doi: 10.1097/CCM.0b013e3182a66b9b. [DOI] [PubMed] [Google Scholar]
- 44.Metersky M.L., Ma A., Houck P.M. Antibiotics for bacteremic pneumonia: improved outcomes with macrolides but not fluoroquinolones. Chest. 2007;131:466–473. doi: 10.1378/chest.06-1426. [DOI] [PubMed] [Google Scholar]
- 45.Wunderink R.G., Niederman M.S., Kollef M.H. Linezolid in methicillin-resistant Staphylococcus aureus nosocomial pneumonia: a randomized, controlled study. Clin Infect Dis. 2012;54:621–629. doi: 10.1093/cid/cir895. [DOI] [PubMed] [Google Scholar]
- 46.Kumar A., Roberts D., Wood K.E. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 2006;34:1589–1596. doi: 10.1097/01.CCM.0000217961.75225.E9. [DOI] [PubMed] [Google Scholar]
- 47.McGeer A., Green K.A., Plevneshi A. Antiviral therapy and outcomes of influenza requiring hospitalization in Ontario, Canada. Clin Infect Dis. 2007;45:1568–1575. doi: 10.1086/523584. [DOI] [PubMed] [Google Scholar]
- 48.Yealy D.M., Auble T.E., Stone R.A. Effect of increasing the intensity of implementing pneumonia guidelines: a randomized, controlled trial. Ann Intern Med. 2005;143:881–894. doi: 10.7326/0003-4819-143-12-200512200-00006. [DOI] [PubMed] [Google Scholar]
- 49.Lim H.F., Phua J., Mukhopadhyay A. IDSA/ATS minor criteria aided pre-ICU resuscitation in severe community-acquired pneumonia. Eur Respir J. 2014;43:852–862. doi: 10.1183/09031936.00081713. [DOI] [PubMed] [Google Scholar]
- 50.Lim W.S., van der Eerden M.M., Laing R. Defining community acquired pneumonia severity on presentation to hospital: an international derivation and validation study. Thorax. 2003;58:377–382. doi: 10.1136/thorax.58.5.377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Renaud B., Santin A., Coma E. Association between timing of intensive care unit admission and outcomes for emergency department patients with community-acquired pneumonia. Crit Care Med. 2009;37:2867–2874. doi: 10.1097/CCM.0b013e3181b02dbb. [DOI] [PubMed] [Google Scholar]
- 52.Restrepo M.I., Mortensen E.M., Rello J. Late admission to the ICU in patients with community-acquired pneumonia is associated with higher mortality. Chest. 2010;137:552–557. doi: 10.1378/chest.09-1547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Charles P.G., Wolfe R., Whitby M. SMART-COP: a tool for predicting the need for intensive respiratory or vasopressor support in community-acquired pneumonia. Clin Infect Dis. 2008;47:375–384. doi: 10.1086/589754. [DOI] [PubMed] [Google Scholar]
- 54.Renaud B., Labarere J., Coma E. Risk stratification of early admission to the intensive care unit of patients with no major criteria of severe community-acquired pneumonia: development of an international prediction rule. Crit Care. 2009;13:R54. doi: 10.1186/cc7781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Espana P.P., Capelastegui A., Gorordo I. Development and validation of a clinical prediction rule for severe community-acquired pneumonia. Am J Respir Crit Care Med. 2006;174:1249–1256. doi: 10.1164/rccm.200602-177OC. [DOI] [PubMed] [Google Scholar]
- 56.Rahman N.M., Maskell N.A., West A. Intrapleural use of tissue plasminogen activator and DNase in pleural infection. N Engl J Med. 2011;365:518–526. doi: 10.1056/NEJMoa1012740. [DOI] [PubMed] [Google Scholar]
- 57.Meijvis S.C., Hardeman H., Remmelts H.H. Dexamethasone and length of hospital stay in patients with community-acquired pneumonia: a randomised, double-blind, placebo-controlled trial. Lancet. 2011;377:2023–2030. doi: 10.1016/S0140-6736(11)60607-7. [DOI] [PubMed] [Google Scholar]
- 58.Brun-Buisson C., Richard J.C., Mercat A. Early corticosteroids in severe influenza A/H1N1 pneumonia and acute respiratory distress syndrome. Am J Respir Crit Care Med. 2011;183:1200–1206. doi: 10.1164/rccm.201101-0135OC. [DOI] [PubMed] [Google Scholar]
- 59.Musher D.M., Rueda A.M., Kaka A.S. The association between pneumococcal pneumonia and acute cardiac events. Clin Infect Dis. 2007;45:158–165. doi: 10.1086/518849. [DOI] [PubMed] [Google Scholar]
- 60.Griffin M.R., Zhu Y., Moore M.R. Hospitalizations for pneumonia after a decade of pneumococcal vaccination. N Engl J Med. 2013;369:155–163. doi: 10.1056/NEJMoa1209165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ampofo K., Pavia A.T., Chris S. The changing epidemiology of invasive pneumococcal disease at a tertiary children's hospital through the 7-valent pneumococcal conjugate vaccine era: a case for continuous surveillance. Pediatr Infect Dis J. 2012;31:228–234. doi: 10.1097/INF.0b013e31823dcc72. [DOI] [PMC free article] [PubMed] [Google Scholar]


