Abstract
Infectious diseases are the tip of the iceberg in the economic burden of the developing countries, due to the resistance of the pathogens to antibiotics and the lack of vaccines. The vaccines have become a big challenge in the last decades, where the attention has been focused on scientific challenges such as new vaccine development and adjuvants or delivery systems. The classical vaccines were developed from live-attenuated or killed organisms, such as influenza, smallpox, and BCG, as well as subunits such as Hepatitis B. The attenuated vaccines carry the risk of regaining their pathogenicity under immunosuppression conditions. The development of subunit vaccines without risk are considered as an essential need in combination with adequate delivery systems to obtain desired cell and humoral immune responses against infectious diseases. In the last decades, the use of nanoparticles as a delivery system in vaccines has received special attention to improve vaccine efficacy. These nanoparticles could be composed of lipids, metal and nonmetal inorganics, several polymers, and virus-like particles, which have been tested in research; some of them have already been approved for human and animal use. The characteristics of the nanoparticles have allowed targeting desired antigen-presenting cells to improve immunization strategies to induce protection. The main characteristics of the nanoparticles are to protect the antigens from early proteolytic degradation, control antigen release, and help antigen uptake and processing by antigen-presenting cells, and they should be safe for human and veterinary use. In addition, the nanoparticles could be modified in their physicochemical properties to target specific cells and improve vaccine efficacy. This chapter focuses on the nanoparticle-based vaccine formulations and the approaches used to realize efficient delivery of vaccines in order to induce host protective immunity against infectious diseases.
Keywords: Antigen-presenting cell, Dendritic cell (DC), Lipid, Liposome, Nanoparticle (NP), Poly lactic-co-glycolic acid, Polymer, Vaccine, Vaccine delivery, Virus-like particle
1. Chapter outline
1.1. Introduction
Nanomedicine has seen increased interest by the medical community in recent years, as it has enabled modification of engineering devices for delivery to and interaction with cell environments. In particular, this technology has enabled advances in the design of delivery systems for drugs and vaccines [1]. Nanoparticles (NPs) are now being engineered from different biodegradable materials, including natural and synthetic polymers (poly[lactic-co-glycolic] acid (PLGA) and polylactic acid (PLA)), metals (gold, copper oxide, aluminum oxide, zinc oxide, iron oxide, and silver), or lipids (phosphatidylserine, phosphatidylcholine, and cholesterol) [2].
NPs and microparticles (MPs) have been widely used to deliver drugs, especially cytotoxic drugs or immune-suppression treatments for transplantation. These NPs are effective in the distribution of medications to target specific organs and control drug delivery. These drug carriers not only transport drugs to sites of cancer or other target diseased organs, preventing damage to healthy cells, but also protect the drug from degradation [3]. Some of the metal nanoparticles (e.g., gold, silver, or PLGA) themselves may have dual functions, working both as carriers and as targeted delivery systems using the membranes of cancer cells, which contain tumor-associated markers. In a recent study, Fang et al. used a membrane from mouse melanoma cancer cells in the outer layer of PLGA nanoparticles. These NPs were stable and persisted in the structure during cellular endocytosis, activating the maturation of dendritic cells (DCs) and presenting especially to T cells that have TCR binding to gp100 and inducing the production of IFN-γ. Moreover, the authors found that the PLGA covering membrane had receptors that allow interaction with cancer cells and delivery of the drug [4], [5]. Furthermore, the NPs were used to deliver drugs to the specific site of organ transplantation to prevent rejection. Systemic immunosuppression is a high risk for recipients of organ transplantation, as it uses high doses of immunosuppression agents that make them more susceptible to infections and in some cases cause death. Previous studies found that diabetic mice were transplanted with islets into the eye to prevent islet graft immune rejection in vivo using rapamycin MPs. The islets transplanted with the immunosuppressed drug MPs survived more than 1 month compared with the control (empty MPs), which rejected the islets in the second week [6].
Work in last few decades has increased our knowledge of infectious diseases and the mechanisms of evasion of immune responses. However, new variants of antibiotic-resistant pathogenic microorganisms have emerged and are becoming a challenge for designing new vaccines and adjuvants. Until now vaccines have been developed from live-attenuated microorganisms or killed pathogens (first-generation vaccines) [7], DNA vaccines (third-generation vaccines), subunit vaccines [8], and synthetic peptides (second-generation vaccines). The last three vaccine types eliminate the risk of developing the disease, but they must be used in conjunction with an adequate adjuvant or delivery system (Table 7.1 ). The combination of the vaccine with the adjuvant or delivery system should be safe, stable, and have the ability to induce long-lived memory B and T cell responses, preferably with a single dose and a maximum of two doses and be free from strict storage requirements [9]. DNA and RNA vaccines are safe but need a second boost with recombinant protein or DNA from another vector. NPs have become an alternative to targeting vaccine delivery to immune cells, improving vaccine efficacy with slow release, easy antigen uptake, and induction of humoral and cellular responses [8].
Table 7.1.
Types of vaccines.
| Vaccine | Constituent | Examples |
|---|---|---|
| Live-attenuated vaccines | Whole pathogen | Measles, mumps, rubella, rotavirus, smallpox, chickenpox, yellow fever |
| Inactivated vaccines | Whole pathogen | Rabies, flu, polio, hepatitis A |
| Subunit, recombinant, polysaccharide, and conjugate vaccines | Part of the pathogen | Hepatitis B, whooping cough, Hib (Haemophilus influenzae type b) disease, human papillomavirus, shingles, meningococcal disease, pneumococcal disease |
| Toxoid vaccines | Toxin | Diphtheria, tetanus |
| Future of vaccines | DNA | Research studies |
NPs have played an important role in the activation of antigen-presenting cells (APCs), especially DCs, which may determine vaccine efficacy. Although there is some cytotoxic effect of the NPs [10], [11], the risk is low compared with the benefits of vaccine delivery [12]. In this chapter, we will summarize the different nanocarrier-based vaccine formulations that achieve the desired host immunity against infectious diseases and cancer, and at the end, we will discuss on the limitations of the respective carriers.
1.2. Immune responses after vaccination
DCs are specialized APCs that coordinate the innate and adaptive immune responses to induce “talking” by chemokines to start the defense against infectious diseases. There are three subtypes: plasmacytoid, myeloid, and follicular DCs. Depending on the subtype of DC, the cytokine profile will be different, and the responses may be protective or not. The interaction and delivery of antigens and adjuvants to DCs is a research priority, in order to optimize the humoral and cellular vaccine responses [13]. Follicular DCs are the key to activating naïve T and B cells to initiate the adaptive immune response and the induction of long-lived memory cells. Lymphocyte activation is started after recognition of the antigen by the T cell receptor (TCR) and B cell receptor (BCR). The recognition of a specific antigen is associated with reorganization of costimulatory surface proteins such as CD40 ligand, CD28, CD4, or CD8 in T cells and CD40, CD80, and CD86 in APCs. The early molecular events that underlie the formation of the synapse are highly coordinated and tightly controlled. The B cells spread over the antigen- APC- C3 or FcR or CR, rapidly, preventing antigen internalization and phagocytosis by DCs and their posterior presentation to MHC class I or class II. Before the antigen is processed and presented along with MHC class II molecules to the TCR, antigen–MHC complexes mediate the recruitment of naïve CD4+ T helper cells. The DCs present the antigen–MHC class II complex to the TCR, and the T cells differentiate into one of the subtypes of CD4+ T helper cells (Th). The interaction of B cell MHC class II–antigen complexes with the TCR of CD4+ T cells is necessary for full B cell activation and production of antibodies [14]. The DCs are the principal target for delivery of the vaccine and hence for NP, although other APCs, such as macrophages, B cells, and lung epithelial cells, can present the antigen to T cells. Unlike DCs, macrophages induce high lysosomal activity after phagocytosis to enhance antigen presentation and thus effector immune responses. Nanotechnology has been improving antigen delivery, depending on the size, charge, and type of NP that can enter through receptors, such as Toll-like receptors (TLRs), pinocytosis, phagocytosis, or special receptor targets. The entry of DNA vaccines using different types of NPs is shown in Fig. 7.1 [5].
Figure 7.2.
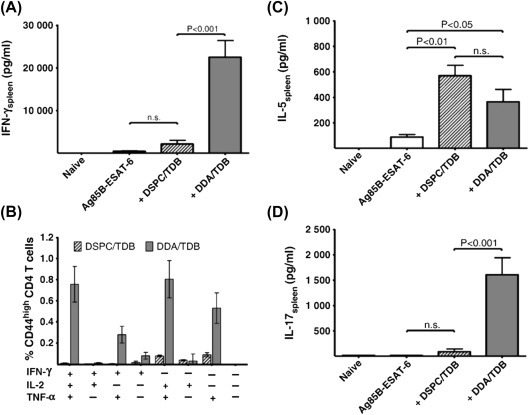
Immune responses in mice 3 weeks after the last of three immunizations with 2 μg of Ag85B–ESAT-6 alone (white) or in combination with DSPC–TDB (dashed) or DDA–TDB (gray). (A) IFN-γ responses in the spleen. (B) Frequency of CD44high T cells in response to each of eight possible cytokine subsets of IFN-γ, IL-2, and/or TNF-α. (C) IL-5 responses in the spleen. (D) IL-17 responses in the spleen.
Adapted from Henriksen-Lacey M, Christensen D, Bramwell VW, Lindenstrøm T, Agger EM, Andersen P, et al. Liposomal cationic charge and antigen adsorption are important properties for the efficient deposition of antigen at the injection site and ability of the vaccine to induce a CMI response. J Control Release 2010;145(2):102–8 with permission.
Figure 7.1.
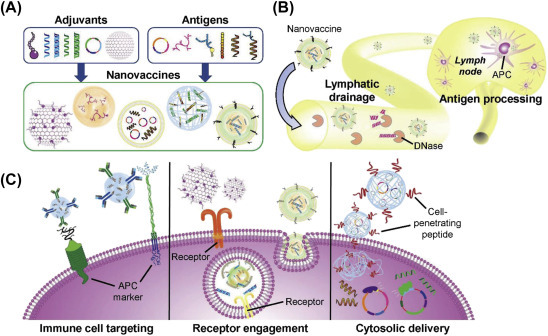
Advantages of NPs for vaccine design. (A) Various combinations of adjuvants and antigens can be formulated using NP platforms such as liposomes, emulsions, nanogels, and other substances. (B) Nanovaccines can access the lymphatic drainage system for lymph node delivery while protecting their cargoes from environmental degradation. Once at the lymph nodes, the nanocarriers deliver their cargoes to APCs for immune processing. (C) Nanovaccine properties can be tuned to efficiently deliver their cargoes for maximum immune activation. For example, NPs can be modified to target specific subsets of immune cells. They can also be delivered to specific intracellular compartments, where receptors for immune pathways can be triggered.
From Kroll AV, Jiang Y, Zhou J, Holay M, Fang RH, Zhang L. Biomimetic nanoparticle vaccines for cancer therapy. Adv Biosyst 2019:1800219 with permission.
1.3. Types of NPs used to deliver vaccines
Until now there have been various types of NPs, composed of gold, dendrimers, carbon, polymers, and liposomes, that have been used to deliver vaccines. All can stimulate the production of cytokines and antibody responses [15], [16], [17], [18], [19]. The cargo is not limited to vaccines, and it is possible to add adjuvants and immune stimulatory molecules, including silica and iron, TLR agonist, and cytokines, to improve immunogenicity [20], [21]. Several vaccines have been tested on the different types of NPs (Table 7.2 ).
Table 7.2.
List of antigens delivered by nanocarriers for the treatment of different diseases in vaccine research.
| Antigen | Nanocarrier used | Disease |
|---|---|---|
| Against Bacterial Infection | ||
| Antigenic protein | Poly(D,L-lactic-co-glycolic acid) nanospheres | Anthrax |
| DNA encoding T cell epitopes of Esat-6 and FL | Chitosan nanoparticle | Tuberculosis |
| Mycobacterium lipids | Chitosan nanoparticle | Tuberculosis |
| Polysaccharides | Liposomes | Pneumonia |
| Bacterial toxic and parasitic protein | Liposomes | Cholera and malaria |
| Fusion protein | Liposomes | Helicobacter pylori infection |
| Antigenic protein | Nanoemulsion | Cystic fibrosis |
| Antigenic protein | Nanoemulsion | Anthrax |
| Mycobacterium fusion protein | Liposome | Tuberculosis |
| Against Viral Infection | ||
| Antigenic protein | Chitosan nanoparticles | Hepatitis B |
| Viral protein | Gold nanoparticles | Foot and mouth disease |
| Membrane protein | Gold nanoparticles | Influenza |
| Viral plasmid DNA | Gold nanoparticles | HIV |
| Tetanus toxoid | Poly(D,L-lactic-co-glycolic acid) nanospheres | Tetanus |
| Hepatitis B surface antigen | Poly(D,L-lactic-co-glycolic acid) nanospheres | Hepatitis B |
| Hepatitis B surface antigen | Alginate-coated chitosan nanoparticle | Hepatitis B |
| Live virus vaccine | Chitosan nanoparticles | Newcastle disease |
| Capsid protein | VLPs | Norwalk virus infection |
| Capsid protein | VLPs | Norwalk virus infection |
| Influenza virus structural protein | VLPs | Influenza |
| Nucleocapsid protein | VLPs | Hepatitis |
| Fusion protein | VLPs | Human papilloma virus |
| Multiple proteins | VLPs | Rotavirus |
| Virus proteins | VLPs | Blue tongue virus |
| Enveloped single protein | VLPs | HIV |
| Viral protein | Polypeptide nanoparticles | Corona virus for severe acute respiratory syndrome (SARS) |
| Against Parasitic Infection | ||
| Merozoite surface protein | Iron oxide nanoparticles | Malaria |
| Epitope of Plasmodium berghei circumsporozoite protein. | Polypeptide nanoparticles | Rodent malarial parasitic infection |
| Surface protein from Eimeria falciformis sporozoites | ISCOMs | Diarrhea |
From Pati R, Shevtsov M, Sonawane A. Nanoparticle vaccines against infectious diseases. Front Immunol 2018;9. adapted with permission.
1.4. Liposomes
Liposomes are the second-most common type of NP, and they self-assemble in water under special conditions. They are composed of lipids, which have a hydrophilic head and a hydrophobic tail that maintain hydrophilic inner and outer membranes, in lamellar lipid bilayers or in multilayers that simulate vesicles found within cells [22]. Liposomes can induce cellular responses or humoral responses, depending on the charge, size, and lipid composition. A previous study showed that charge had the main role in activation of the cellular or humoral immune responses. The authors' approach was to investigate the importance of the antigen–liposome interaction in immunogenicity and depot formation. They used subunit antigens Ag85B–ESAT-6 (pI = 4.9) from TB, and “CTH1” (pI = 9.0), from Chlamydia vaccines and a model antigen, lysozyme (pI = 11). The injection of cationic dimethyldioctadecylammonium bromide (DDA): trehalose 6,6′-dibehenate (TDB) liposomes with associated Ag85B–ESAT-6 form a deposit at the site of injection. By contrast, the anion disappears immediately. The cationic liposomes induce the infiltration of monocytes at the site of immunization, possibly mediated by depot formation. These liposomes induce strong T cell proliferation and differentiation through Th1 and Th17 responses, although the authors could not find DCs. However, DCs are known to migrate to the secondary lymph nodes to activate the T and B cells, which the authors did not analyze in this manuscript [23]. Previous studies have shown that DDA induces proinflammatory cytokines and chemokines after infiltration of monocytes and macrophages in vivo [24]. One of the advantages of liposomes is mimicking properties of the pathogens, inducing humoral and cellular immune responses. The antigen’s presentation to APCs depends on the membrane characteristics, size, and ligand-receptor binding. Liposomes could induce Th2 responses if the lipids are unsaturated whereas saturated lipids promoted Th1-type immune responses [25], [26]. Liposomes could be modified according to the needs of delivery to produce Th1, Th2, Th17, or Tfh immune responses. Cationic liposomes can increase the uptake of subunit vaccines as synthetic peptides and recombinant proteins; this can be explained by the interaction between positively charged liposome with the APCs that have a negative charge in their membrane [26], [27]. Another modification is the pH-sensitive fusogenic liposomes, which are stable in neutral pH 7.4, but in acidic conditions, the antigen is released and presented by MHC class I or II. These liposomes containing phosphatidylethanolamine and amphiphilic stabilizers allow PE-containing liposomes to form aggregates, due to the poor hydration of their headgroups, which can explain their high affinity to adhere to cell membranes [28], [29].
Another study designed a unique structure composed of inter-bilayer-crosslinked multilamellar vesicles (ICMVs), which is stable in the extracellular environment but rapidly released in endosomes/lysosomes, thereby enhancing vaccine immune responses. The ICMVs carried the antigen OVA mixed with the adjuvant monophosphoryl lipid A (MPLA). This mixture amplified vaccine responses and upregulated the costimulatory cells on splenic and bone marrow DCs. In addition, splenic DCs incubated with the OVA+ vaccine MPLA triggered proliferation of naïve OT-1 CD8+ T cells in vitro, suggesting that the ICMVs enhanced cross-presentation of the antigen. The same results were obtained in vivo after the vaccination of mice with the mixture, and the ICMVs elicited robust antibody titers that were ∼1000-fold greater than simple liposomes. These results could be attributed to activation enhancement of DCs and antigen cross presentation [30]. The first use of liposomes as a delivery system for a malaria vaccine was in the 1980s. Ballou et al. synthetized peptides derived from the repetitive region of the circumsporozoite (CS) protein of Plasmodium falciparum sporozoites. The synthetic peptides were conjugated to keyhole limpet hemocyanin (KLH) proteins and were incorporated into liposomes. Immunized mice and rabbits produced antibodies against the repeat region of the protein with biologic activity correlated with protection [31]. RTS, S is the only vaccine against malaria that is in phase three clinical trials in Africa. The RTS, S vaccine has the central repeat region of CSP, and T cell epitopes localized in the C-terminal region are fused to hepatitis B surface antigen (HBsAg) and expressed in Saccharomyces cerevisiae yeast. This virus-like particle (VLP) vaccine also contains MPLA and QS-21, which enhances humoral and CD4+ T cell responses in the first 4 years, with a level of protection of 18%–36%. Protection decreased rapidly thereafter, with negative efficacy in some children. The problem is not RTS, S nor the adjuvant system, as both induce potent immune responses. However, the CSP part of RTS, S is an antigenic polymorphism, and T cell epitopes present on the CS protein that are incorporated into the vaccine are also polymorphic. To address this problem, the newest generation of this vaccine is coformulated with Matrix-M, a saponin-based liposomal adjuvant [32]. However, the vaccine protection against P. falciparum has not been achieved due to the short-lived memory cells and the evasion mechanisms of the parasite [33]. The first malaria vaccine was tested in 1967 in animals using Plasmodium bergheiś radiation-attenuated sporozoites, with a 100% of protection [34]. Hoffman et al. reported in 2002 that 10 human volunteers were vaccinated with irradiated sporozoites of P. falciparum strain NF54, and all of them were protected between 2 to 9 weeks [35]. This vaccine will be used to vaccinate 360,000 children a year in three African countries (Ghana, Malawi, and Kenya). The vaccine could prevent 4 in 10 cases according to previous clinical trials.
1.5. Virus-like particles
Virus-like particles (VLPs) are composed of a self-assembling viral membrane maintaining viral surface proteins. VLPs can be modified to express additional proteins of other microorganisms, which could be engineered by fusion of the proteins with membrane antigens or by endogenous expression of other antigens [36]. Gardasil is a four-component VLP-type vaccine specific against HPV. It contains the L1 major capsid protein of HPV types 6, 11, 16, and 18 and is administered along with an aluminum adjuvant. This vaccine was administered to 1158 women and was followed up for incidence of persistent associated infection for 35 months. The efficacy was 100% for preventing clinical disease. The HPV combination vaccine was immunogenic, inducing the production of long-lived antibodies [37]. However, some problems with the vaccine in teenage girls were reported. The adverse events included dermatologic/mucosa-allergic reaction (25%), rash (22%), and local/injection-site reaction (20%). However, some serious adverse events following immunization were reported (7.5% of reports), including two incidents of anaphylaxis, two seizures, one incident of thrombocytopenia, and one death [38].
1.6. Metal and nonmetal inorganic NPs
Inorganic metal NPs are frequently used in drug delivery and bioimaging, especially in treating cancer patients. DNA vaccines are more stable and protected from degradation when carried by a gold, silica, or silver NP delivery system. The covalent attachment of Chito6 to GNPs increases the NP molecular weight, enhancing DNA binding and stability without compromising DNA release and transfer. Chito6–GNP–DNA (HBsAg) complexes induce effective antibody and T cell responses after immunization of BALB/c mice. By contrast, naked DNA-primed HBsAg induces antibodies after a series of four immunizations with 10 mg of naked DNA. HBsAg-specific CD8+ T cells eliminated P815/BALB target cells that had been sensitized with an HBsAg CTL epitope peptide in vitro. These chimeric NPs, employing a minimal amount of DNA, induce effective immune responses when compared with naked DNA [39].
1.7. Polymeric NPs
Biodegradable polymers are of significant interest in the delivery of drugs and vaccines against infectious diseases. These polymers consist of either natural or synthetic monomers that are biodegradable, are nonimmunogenic, have low cytotoxicity, and are easy to prepare. There are several polymers, such as chitosan, PLGA, polyethylene glycol (PEG), polycaprolactone, and dextran used as delivery systems [40]. PLGA is approved for human use by the US Food and Drug Administration (FDA) and European Medicines Agency (EMA), while PLGA has been used to deliver drugs for long periods. DCs play an important role in the activation of adaptive immune responses, in which B cells and T cells activate and differentiate into subpopulations that determine which humoral or cellular immune responses eliminate the microorganism. Cruz et al. studied the delivery of NPs and MPs to DCs to create a biocompatible and biodegradable slow-release vaccine and effectively target the cells. The NPs and MPs (PLGA–PEG) were loaded with tetanus toxoid peptide–FITC linked to a Lys–Lys cathepsin cleavage site and an anti-DC-SIGN antibody. The DCs targeted PLGA-based vaccine NPs but not MPs. The NPs were efficiently taken up with the help of anti-DC-SIGN antibodies and induced proliferation of T cells (Fig. 7.3 ). However, the MPs were taken up for all APCs, including the DCs (Fig. 7.4 ). New research is needed to understand the biology, antigen processing, and presentation needed to induce long-lived memory B and T cells [41].
Figure 7.3.
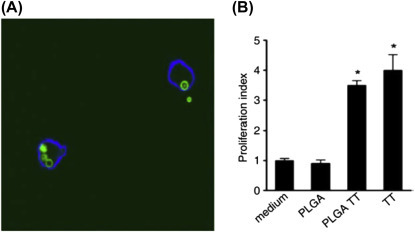
Uptake of PLGA MPs with FITC–TT peptide by DCs results in antigen presentation. DCs were incubated with FITC–TT containing MPs (green; light gray in printed version) for 1 h to confirm uptake by human DCs. Cells were analyzed by confocal laser scanning microscopy. The cell surface was visualized by MHC class II staining (blue; white in print version). The image represents the middle focal plane of the DCs, with the iris set at 2 nm (A). Presentation of PLGA-encapsulated FITC–TT peptide was studied by culturing DCs for 18 h in culture medium alone, medium supplemented with empty PLGA MPs (PLGA), or with 0.1 μg of FITC–TT peptide encapsulated within PLGA MPs (PLGA TT). In addition, DCs were pulsed with 1 μM TT peptide as a positive control for antigen presentation (TT). Subsequently, autologous TT-responsive peripheral blood lymphocytes were added. After 3 days, cellular responses were assessed in a proliferation assay. Data are mean proliferation indices ± SD relative to medium control for experiments performed in triplicate. Significant differences from medium control according to ANOVA and Dunnett's test:∗P < .01.
From Cruz LJ, Tacken PJ, Fokkink R, Joosten B, Stuart MC, Albericio F, et al. Targeted PLGA nano-but not microparticles specifically deliver antigen to human dendritic cells via DC-SIGN in vitro. J Control Release 2010;144(2):118–26 with permission.
Figure 7.4.
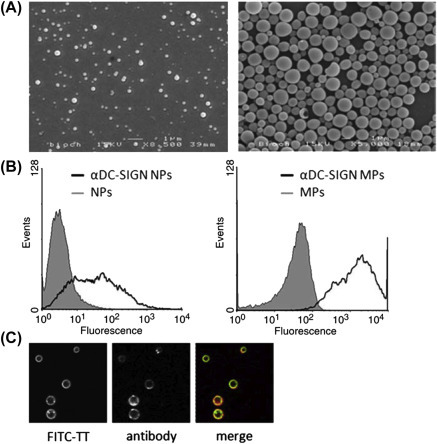
Antibodies were introduced on the surface of PLGA NPs and MPs. The morphology of NPs (A, left panel) and MPs (A, right panel) with PEG–lipids was analyzed by scanning electron microscopy. The presence of the antibodies on the PLGA particle surface was confirmed by flow cytometry. The NPs and MPs were stained with fluorescence-labeled secondary antibodies and analyzed on a flow cytometer (B). PLGA MPs were mounted on glass slides and analyzed by confocal laser scanning microscopy to visualize antibodies present on the particle surface. FITC–TT peptide was detected as a green (light gray in print version) fluorescent ring surrounding the PLGA particles (see also Fig. 7.2A). Antibodies on the particle surface were detected by secondary antibody staining with Alexa 647-labeled anti-human IgG. The images represent the middle focal plane of particles and show split channels of the FITC signal (FITC–TT), the Alexa 647 signal (antibody), and a merged image showing the antibody in red (dark gray in print version) and the FITC–TT peptide in green (white in print version) (C).
From Cruz LJ, Tacken PJ, Fokkink R, Joosten B, Stuart MC, Albericio F, et al. Targeted PLGA nano-but not microparticles specifically deliver antigen to human dendritic cells via DC-SIGN in vitro. J Control Release 2010;144(2):118–26 with permission.
Botulism is a lethal neuroparalytic disease produced by Clostridium botulinum toxins (A-H). Ruwona et al. demonstrated that cationic PLGA NPs can carry plasmid DNA encoding the BoNT heavy-chain (Hc) fragment and that its product is nontoxic. Immunized mice produced high titers of antibodies after 5 to 9 weeks (Fig. 7.5 ).
Figure 7.5.

Serum anti-BoNT/C-Hc50 IgG (A), IgG1 (B), and IgG2a (C) induced by intramuscular immunization of mice with pVax/opt-BoNT/C-Hc50 (20 μg/mouse), alone (pBoNT/C) or coated on cationic PLGA NPs (pBoNT/C-NP). SKH-1 Elite mice (n = 5) were dosed in weeks 0, 2, 4, and 8. Control mice received PBS only. Blood samples were collected in week 5 (day 35) and week 9 (day 63). Data are mean ± SD (n = 5). ∗P < .05 compared with control group, #P < .05 compared with pBoNT/C only, and ξp < .05, day 35 versus day 63.
Ruwona TB, Xu H, Li J, Diaz-Arévalo D, Kumar A, Zeng M, et al. Induction of protective neutralizing antibody responses against botulinum neurotoxin serotype C using plasmid carried by PLGA nanoparticles. Hum Vaccines Immunother 2016;12(5):1188–92 with permission.
After four immunizations, specific IgG1 had decreased, but IgG2a had increased, compared with the first immunization. After challenge, 100% of the mice vaccinated with PLGA–pVax/opt-BoNT/C-Hc50 survived, while only 80% of the mice immunized with naked plasmid were protected (Fig. 7.6 ) [17].
Figure 7.6.
Protective immunity against BoNT/C challenge in immunized mice. SKH-1 Elite mice (n = 5) were dosed in weeks 0, 2, 4, and 8 with pVax/opt-BoNT/C-Hc50 plasmid (20 μg per mouse), plasmid alone (pBoNT/C), or plasmid coated onto PLGA NPs (pBoNT/C–NP), with control mice receiving PBS only and challenged in week 12 with 100 ± MLD50 of BoNT/C toxin.
Ruwona TB, Xu H, Li J, Diaz-Arévalo D, Kumar A, Zeng M, et al. Induction of protective neutralizing antibody responses against botulinum neurotoxin serotype C using plasmid carried by PLGA nanoparticles. Hum Vaccines Immunother 2016;12(5):1188–92 with permission.
Modifications of polymeric NPs have been used to deliver synthetic peptides or recombinant proteins to dendritic cells and macrophages. Salvador et al., modified PLGA MS, 50:50 lactide–glicolide ratio, to produce cationic NPs using polyethylene imine (PEI), and BSA as antigen. In addition, the NPs were modified encapsulating monophosphoryl lipid A (MPLA) or polyinosinic-polycytidilic acid poly(I:C) and α-galactosyl ceramide to address their adjuvant effect. Coumarin 6 was incorporated into the oily phase for the preparation of fluorescent NPs to evaluate in vitro assays. The NPs were tested in vitro using monocytes and dendritic cells, and mice were immunized with the different NP modifications to determine the immune responses in vivo. The cationic NPs showed a noticeable enhancement of their uptake by the monocytes, MDCs, and PDCs in comparison to the classical NPs. In addition, the cationic NPs increased immunostimulatory effect since they induced the production of antibodies and Th1 cell responses producing IFN-γ [12].
1.8. NP-investing companies and clinical trials
The interest of companies in nanoparticle-based vaccine delivery dramatically increased in the last decades. Pevion Biotech Ltd., a Swiss company founded in 2002, developed the virosome-based technology platforms to make efficient and safe prophylactic/therapeutic vaccine candidates. For Candida albicans that can cause vulvovaginal mucosal infections, Pevion used the aspartyl-proteinase (Sap2), which is an immunodominant antigen and virulence factor; this recombinant protein was assembled with virosomes (PEV7). The evaluation in mouse model and the initial clinical trial on women showed that this candidate vaccine, intravaginally administered, has a therapeutic potential for the treatment of recurrent candidiasis [42]. In addition, this company tested a malaria vaccine in Africa. This vaccine had combination of FFM ME-TRAP+PEV3A (AMA-1) and was tested in phase I/IIa clinical trials. Although the vaccine did not show sterile protection, it induced responses on blood stage parasites and lower rates of parasite growth in human volunteers vaccinated with PEV3A, compared to unvaccinated controls [43]. GlaxoSmithKline plc is working to improve the coformulated RTS, S vaccine using Matrix-M, a saponin-based liposomal adjuvant [32]. Novavax is developing respiratory syncytial virus (RSV) F nanoparticle vaccine with aluminum. The purpose of the reported study was to evaluate the safety and efficacy of maternally transferred antibodies in preventing RSV in infants. The vaccinated healthy third-trimester pregnant women showed significant protection to newborn children from RSV challenge and reduced pulmonary inflammation, and the vaccine was safe and effective for maternal and adult vaccination [44]. Furthermore, this company is working in recombinant trivalent nanoparticle influenza vaccine with matrix M-1. The vaccine can induce responses to one or more conserved HA head and stem epitopes. The antibody responses are able to neutralize against both homologous and heterologous strains [45].
The nanoparticles have been used in recent years in only a few of advanced clinical trials. The vaccine against RSV infection has three studies using RSV F nanoparticle vaccine with aluminum in phase 2 and 3 trials. The first clinical study of RSV was RSV F Dose-Ranging Study in Women, which started October 2013 and finished May 2016. The study showed that all formulations were well tolerated, without treatment-related serious adverse events. Antibodies anti-F IgG and palivizumab-competitive antibody responses were correlated and increased after both doses, while microneutralization assays increased significantly after the first dose, then plateaued [46]. The second study was RSV F Vaccine Maternal Immunization Study in Healthy Third-trimester Pregnant Women, which started September 2014 and finished June 2017. This study demonstrated that the vaccine is safe to infants and pregnant women. The third clinical study is in phase three trials and will finish in July 2019. The vaccine is safe and effective for infants and pregnant women [44]. The study “Evaluation of the Safety and Immunogenicity of a Recombinant Trivalent Nanoparticle Influenza Vaccine with Matrix M-1 Adjuvant (NanoFlu)” was used to test protection in people older than age 60, but no results have been publicly released. The other study is the phase two trial entitled “Dose and Formulation Confirmation of Quad-NIV in Older Adults.” In this study, 1375 subjects were randomized to seven treatment groups to receive the vaccine or an active comparator. Table 7.3 summarizes the clinical trials [47]: phase I–IV clinical trials/vaccines/nanoparticles.
Table 7.3.
Phase I and III clinical trials with vaccines delivery by nanoparticles.
| Title | Infectious diseases | ClinicalTrials.gov identifier: | Phase |
|---|---|---|---|
| Evaluation of the safety and immunogenicity of a recombinant trivalent nanoparticle influenza vaccine with matrix M-1 adjuvant (NanoFlu) | Influenza | NCT03293498 | I and II |
| Dose and formulation confirmation of Quad-NIV in older adults | Influenza | NCT03658629 | II |
| RSV F dose–ranging study in women | Respiratory syncytial virus infections | NCT01960686 | II |
| RSV F vaccine maternal immunization study in healthy third-trimester pregnant women. | Respiratory syncytial virus infections | NCT02247726 | II |
| A study to determine the safety and efficacy of the RSV F vaccine to protect infants via maternal immunization | Respiratory syncytial virus infections | NCT02624947 | III |
1.9. Nanocytotoxicity
Engineered NPs have revolutionized the delivery of drugs, monoclonal antibodies, and vaccines to target cells. NPs have been designed from metals and nonmetals, polymeric materials, lipids, VLPs, and bioceramics, giving distinctive physicochemical and electrical characteristics that specifically interact with a targeted cell or organ. However, NPs can enter easily into the human body by crossing lipid bilayers and may interact with sensitive organs [48]. The clearance and excretion of NPs is mediated through the mononuclear phagocytic system, the renal urinary system, and by biliary clearance. Macrophages phagocytize the NPs and keep them in the secondary lymph organs and/or liver using the Mertk (Mer) receptor (the same receptor tyrosine kinase family as Axl and Tyro-3), which is responsible for promoting apoptotic cell engulfment and supports platelet aggregation and clot stability in vivo [49]. In addition, Kupffer cells, which are liver macrophages, sequester 100-nm nanoparticles [50]. When the particles are ≤5 nm in diameter, they are rapidly cleared from the circulatory system via renal filtration [51]. On the other hand, some studies have shown that metallic NPs (MeNPs) have effects on innate immunity. Specifically, in vitro assays showed that MeNPs have some cytotoxicity and genotoxicity and interfere with cytokine production and gene expression due to receptor modifications. The response of the innate immune system to threats is mediated by inflammation and the production of cytokines, chemokines, free radicals (nitric oxide) [52], and other reactive oxygen species (ROS). In this process, there is an accumulation of oxidized glutathione (GSSG), generating stress from the proinflammatory signal (involving TLRs) on the MeNPs and causing cell death and cancer [53]. Other studies have shown that adenovirus VLPs combine with a gene to control Gelsinger's ammonia metabolism (encoding ornithine transcarbamylase), which invades all the organs and induces severe reactions that can lead to death [54]. Other severe side reactions have been reported as carcinogenesis or germline alterations observed in animal models, in which the VLPs can become widely dispersed in the body, and the viral vector could end up in the ovaries and testes [55]. Eudragit is a drug delivery polymer: poly (ethyl acrylate-co-methyl methacrylate-cotrimethylammonioethyl methacrylate chloride) 1:2:0.1. It has been reported that using NR8383 macrophages cocultured with NP/ERS, the NPs were close to the inner membrane of the mitochondria, as observed by transmission electronic microscopy. The authors also analyzed genes responsible for mitochondrial function by microarray and found that Opa1 was reduced in expression. This gene helps to maintain network morphology and dynamics and in the regulation of the signaling pathways for cell death, and NP/ERS-treated cells reduced glutathione (GSH), stimulating ROS production. Due to unbalancing of oxidant–antioxidant homeostasis and changes of proteins implied in activation and autophagy, the mitochondria decay through phagophore and autophagosome formation, and mitophagy can occur [11].
The NPs are characterized for their size (<100 nm) with greater surface area per mass compared with larger-sized particles of the same chemistry causing NPs more active biologically. The physicochemical properties of the material could increase the uptake by the antigen-presenting cells, but they can go to other tissues that can generate adverse biological effects such as apoptosis or necrosis. There are some materials that can be degraded easily. However, the NPs (PLGA) had been modified to release antigens slowly to immunize only once and to increase the efficiency of protective adaptive immune responses. Nevertheless, intranasal immunization with NPs had been reported to cause lung injury through oxidative stress, which induced the production of cytotoxic cellular responses and inflammatory cytokines. Another problem with the NPs is the aggregation that may block the blood vessels in the host. Modifications of polymeric NPs with PEI have been used to prevent the recognition to other cells such as epithelial cells in the lung. Other technologies could be used as the single-chain fragment region of antibodies binding to NPs, which recognizes the specific receptor on dendritic cells or macrophages that may deliver the antigens to target organ. Other modifications in the NPs have been used as PLGA covered with PEG (PLGA–PEG) and formulated with tetanus toxoid peptide–FITC linked to a Lys–Lys cathepsin cleavage site and an anti-DC-SIGN antibody, which help the delivery to DCs but not macrophages, with enhanced efficient proliferation of T cells [56]. Negatively charged NPs prevent the activation of the immune system and induction of immunotolerance, therefore preventing the effect of exacerbated inflammatory responses. Nanoparticles can also be engineered to be porous to increase the diffusion of intracellular proteases, resulting in earlier processing of APCs and presentation T cells [57]. In addition, some NPs carrying antigen and adjuvant in the same NP are less efficient to induce the immune response than the ones with adjuvant separated from antigens in different NPs. This may because of the competing effect of coactivation in several pathways [58].
2. Conclusion
NPs are widely explored in vaccine delivery and targeting APCs, especially DCs. Despite advances in immunology, it is critical to understand the mechanisms of entry of NPs into cells and activation of the adaptive immune response and related toxicity. Some approaches inhibit the cytokine and chemokine cascade with DCs and T and B cells. NPs have great potential in immunology, although in-depth study of toxicity is necessary. In addition, NPs require molecules, such as antibodies or ligands, that bind to receptors on target cells to ensure that NPs only adhere to those cells. Any modification should be tested in cell lines or animals to prevent side effects or death. The synthesis of NPs can be shifted to methods that generate reproducible nanoparticles in terms of size (<10 nm, allowing elimination by the kidney), shape, and composition (biodegradable materials) to reduce cytotoxicity.
References
- 1.Shah M.A.A., Ali Z., Ahmad R., Qadri I., Fatima K., He N. DNA mediated vaccines delivery through nanoparticles. J Nanosci Nanotechnol. 2015;15(1):41–53. doi: 10.1166/jnn.2015.9603. [DOI] [PubMed] [Google Scholar]
- 2.Pati R., Shevtsov M., Sonawane A. Nanoparticle vaccines against infectious diseases. Front Immunol. 2018;9 doi: 10.3389/fimmu.2018.02224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.He Q., Gao Y., Zhang L., Zhang Z., Gao F., Ji X. A pH-responsive mesoporous silica nanoparticles-based multi-drug delivery system for overcoming multi-drug resistance. Biomaterials. 2011;32(30):7711–7720. doi: 10.1016/j.biomaterials.2011.06.066. [DOI] [PubMed] [Google Scholar]
- 4.Fang R.H., Hu C.-M.J., Luk B.T., Gao W., Copp J.A., Tai Y. Cancer cell membrane-coated nanoparticles for anticancer vaccination and drug delivery. Nano Lett. 2014;14(4):2181–2188. doi: 10.1021/nl500618u. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kroll A.V., Jiang Y., Zhou J., Holay M., Fang R.H., Zhang L. Biomimetic nanoparticle vaccines for cancer therapy. Adv Biosyst. 2019:1800219. doi: 10.1002/adbi.201800219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fan Y., Zheng X., Ali Y., Berggren P.-O., Loo S.C.J. Local release of rapamycin by microparticles delays islet rejection within the anterior chamber of the eye. Sci Rep. 2019;9(1):3918. doi: 10.1038/s41598-019-40404-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sridhar S., Brokstad K., Cox R. Influenza vaccination strategies: comparing inactivated and live attenuated influenza vaccines. Vaccines. 2015;3(2):373–389. doi: 10.3390/vaccines3020373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lugade A.A., Bharali D.J., Pradhan V., Elkin G., Mousa S.A., Thanavala Y. Single low-dose un-adjuvanted HBsAg nanoparticle vaccine elicits robust, durable immunity. Nanomed Nanotechnol Biol Med. 2013;9(7):923–934. doi: 10.1016/j.nano.2013.03.008. [DOI] [PubMed] [Google Scholar]
- 9.Levine M.M., Sztein M.B. Vaccine development strategies for improving immunization: the role of modern immunology. Nat Immunol. 2004;5(5):460–464. doi: 10.1038/ni0504-460. Epub 2004/04/30. [DOI] [PubMed] [Google Scholar]
- 10.Eidi H., Joubert O., Attik G., Duval R.E., Bottin M.C., Hamouia A. Cytotoxicity assessment of heparin nanoparticles in NR8383 macrophages. Int J Pharm. 2010;396(1–2):156–165. doi: 10.1016/j.ijpharm.2010.06.006. Epub 2010/06/15. [DOI] [PubMed] [Google Scholar]
- 11.Eidi H., Joubert O., Nemos C., Grandemange S., Mograbi B., Foliguet B. Drug delivery by polymeric nanoparticles induces autophagy in macrophages. Int J Pharm. 2012;422(1–2):495–503. doi: 10.1016/j.ijpharm.2011.11.020. Epub 2011/11/29. [DOI] [PubMed] [Google Scholar]
- 12.Salvador A., Sandgren K.J., Liang F., Thompson E.A., Koup R.A., Pedraz J.L. Design and evaluation of surface and adjuvant modified PLGA microspheres for uptake by dendritic cells to improve vaccine responses. Int J Pharm. 2015;496(2):371–381. doi: 10.1016/j.ijpharm.2015.10.037. Epub 2015/10/18. [DOI] [PubMed] [Google Scholar]
- 13.Palucka K., Banchereau J., Mellman I. Designing vaccines based on biology of human dendritic cell subsets. Immunity. 2010;33(4):464–478. doi: 10.1016/j.immuni.2010.10.007. Epub 2010/10/30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Batista F.D., Harwood N.E. The who, how and where of antigen presentation to B cells. Nat Rev Immunol. 2009;9(1):15–27. doi: 10.1038/nri2454. [DOI] [PubMed] [Google Scholar]
- 15.Herzog C., Hartmann K., Künzi V., Kürsteiner O., Mischler R., Lazar H. Eleven years of Inflexal® V—a virosomal adjuvanted influenza vaccine. Vaccine. 2009;27(33):4381–4387. doi: 10.1016/j.vaccine.2009.05.029. [DOI] [PubMed] [Google Scholar]
- 16.Kumar R., Ledet G., Graves R., Datta D., Robinson S., Bansal G.P. Potent functional immunogenicity of Plasmodium falciparum transmission-blocking antigen (Pfs25) delivered with nanoemulsion and porous polymeric nanoparticles. Pharm Res. 2015;32(12):3827–3836. doi: 10.1007/s11095-015-1743-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ruwona T.B., Xu H., Li J., Diaz-Arévalo D., Kumar A., Zeng M. Induction of protective neutralizing antibody responses against botulinum neurotoxin serotype C using plasmid carried by PLGA nanoparticles. Hum Vaccines Immunother. 2016;12(5):1188–1192. doi: 10.1080/21645515.2015.1122147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Salvador A., Igartua M., Hernández R.M., Pedraz J.L. Designing improved poly lactic-co-glycolic acid microspheres for a malarial vaccine: incorporation of alginate and polyinosinic–polycytidilic acid. J Microencapsul. 2014;31(6):560–566. doi: 10.3109/02652048.2014.885608. [DOI] [PubMed] [Google Scholar]
- 19.Wang G., Pan L., Zhang Y., Wang Y., Zhang Z., Lü J. Intranasal delivery of cationic PLGA nano/microparticles-loaded FMDV DNA vaccine encoding IL-6 elicited protective immunity against FMDV challenge. PLoS One. 2011;6(11):e27605. doi: 10.1371/journal.pone.0027605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Roberts R.A., Eitas T.K., Byrne J.D., Johnson B.M., Short P.J., McKinnon K.P. Towards programming immune tolerance through geometric manipulation of phosphatidylserine. Biomaterials. 2015;72:1–10. doi: 10.1016/j.biomaterials.2015.08.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mohanan D., Slütter B., Henriksen-Lacey M., Jiskoot W., Bouwstra J.A., Perrie Y. Administration routes affect the quality of immune responses: a cross-sectional evaluation of particulate antigen-delivery systems. J Control Release. 2010;147(3):342–349. doi: 10.1016/j.jconrel.2010.08.012. [DOI] [PubMed] [Google Scholar]
- 22.Smith J.D., Morton L.D., Ulery B.D. Nanoparticles as synthetic vaccines. Curr Opin Biotechnol. 2015;34:217–224. doi: 10.1016/j.copbio.2015.03.014. [DOI] [PubMed] [Google Scholar]
- 23.Henriksen-Lacey M., Christensen D., Bramwell V.W., Lindenstrøm T., Agger E.M., Andersen P. Liposomal cationic charge and antigen adsorption are important properties for the efficient deposition of antigen at the injection site and ability of the vaccine to induce a CMI response. J Control Release. 2010;145(2):102–108. doi: 10.1016/j.jconrel.2010.03.027. [DOI] [PubMed] [Google Scholar]
- 24.Korsholm K.S., Petersen R.V., Agger E.M., Andersen P. T-helper 1 and T-helper 2 adjuvants induce distinct differences in the magnitude, quality and kinetics of the early inflammatory response at the site of injection. Immunology. 2010;129(1):75–86. doi: 10.1111/j.1365-2567.2009.03164.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Foged C., Arigita C., Sundblad A., Jiskoot W., Storm G., Frokjaer S. Interaction of dendritic cells with antigen-containing liposomes: effect of bilayer composition. Vaccine. 2004;22(15–16):1903–1913. doi: 10.1016/j.vaccine.2003.11.008. [DOI] [PubMed] [Google Scholar]
- 26.Taneichi M., Ishida H., Kajino K., Ogasawara K., Tanaka Y., Kasai M. Antigen chemically coupled to the surface of liposomes are cross-presented to CD8+ T cells and induce potent antitumor immunity. J Immunol. 2006;177(4):2324–2330. doi: 10.4049/jimmunol.177.4.2324. [DOI] [PubMed] [Google Scholar]
- 27.Joseph A., Itskovitz-Cooper N., Samira S., Flasterstein O., Eliyahu H., Simberg D. A new intranasal influenza vaccine based on a novel polycationic lipid—ceramide carbamoyl-spermine (CCS): I. Immunogenicity and efficacy studies in mice. Vaccine. 2006;24(18):3990–4006. doi: 10.1016/j.vaccine.2005.12.017. [DOI] [PubMed] [Google Scholar]
- 28.Abdul Ghaffar K., Kumar Giddam A., Zaman M., Skwarczynski M., Toth I. Liposomes as nanovaccine delivery systems. Curr Top Med Chem. 2014;14(9):1194–1208. doi: 10.2174/1568026614666140329232757. [DOI] [PubMed] [Google Scholar]
- 29.Simões S., Moreira J.N., Fonseca C., Düzgüneş N., de Lima M.C.P. On the formulation of pH-sensitive liposomes with long circulation times. Adv Drug Deliv Rev. 2004;56(7):947–965. doi: 10.1016/j.addr.2003.10.038. [DOI] [PubMed] [Google Scholar]
- 30.Moon J.J., Suh H., Bershteyn A., Stephan M.T., Liu H., Huang B. Interbilayer-crosslinked multilamellar vesicles as synthetic vaccines for potent humoral and cellular immune responses. Nat Mater. 2011;10(3):243. doi: 10.1038/nmat2960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ballou W.R., Rothbard J., Wirtz R.A., Gordon D.M., Williams J.S., Gore R.W. Immunogenicity of synthetic peptides from circumsporozoite protein of Plasmodium falciparum. Science. 1985;228(4702):996–999. doi: 10.1126/science.2988126. [DOI] [PubMed] [Google Scholar]
- 32.Ssemaganda A., Giddam A.K., Zaman M., Skwarczynski M., Toth I., Stanisic D.I. Induction of Plasmodium-specific immune responses using liposome-based vaccines. Front Immunol. 2019;10:135. doi: 10.3389/fimmu.2019.00135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rénia L., Goh Y.S. Malaria parasites: the great escape. Front Immunol. 2016;7(463) doi: 10.3389/fimmu.2016.00463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nussenzweig R.S., Vanderberg J., Most H., Orton C. Protective immunity produced by the injection of x-irradiated sporozoites of Plasmodium berghei. Nature. 1967;216(5111):160–162. doi: 10.1038/216160a0. Epub 1967/10/14. [DOI] [PubMed] [Google Scholar]
- 35.Hoffman S.L., Goh L.M., Luke T.C., Schneider I., Le T.P., Doolan D.L. Protection of humans against malaria by immunization with radiation-attenuated Plasmodium falciparum sporozoites. J Infect Dis. 2002;185(8):1155–1164. doi: 10.1086/339409. [DOI] [PubMed] [Google Scholar]
- 36.Strable E., Finn M. Chemical modification of viruses and virus-like particles. Viruses Nanotechnol Springer. 2009:1–21. doi: 10.1007/978-3-540-69379-6_1. [DOI] [PubMed] [Google Scholar]
- 37.Villa L.L., Costa R.L., Petta C.A., Andrade R.P., Ault K.A., Giuliano A.R. Prophylactic quadrivalent human papillomavirus (types 6, 11, 16, and 18) L1 virus-like particle vaccine in young women: a randomised double-blind placebo-controlled multicentre phase II efficacy trial. Lancet Oncol. 2005;6(5):271–278. doi: 10.1016/S1470-2045(05)70101-7. [DOI] [PubMed] [Google Scholar]
- 38.Harris T., Williams D.M., Fediurek J., Scott T., Deeks S.L. Adverse events following immunization in Ontario's female school-based HPV program. Vaccine. 2014;32(9):1061–1066. doi: 10.1016/j.vaccine.2014.01.004. [DOI] [PubMed] [Google Scholar]
- 39.Zhou X., Zhang X., Yu X., Zha X., Fu Q., Liu B. The effect of conjugation to gold nanoparticles on the ability of low molecular weight chitosan to transfer DNA vaccine. Biomaterials. 2008;29(1):111–117. doi: 10.1016/j.biomaterials.2007.09.007. [DOI] [PubMed] [Google Scholar]
- 40.Wen R., Umeano A.C., Chen P., Farooqi A.A. Polymer-based drug delivery systems for cancer. Crit Rev Ther Drug Carrier Syst. 2018;35(6):521–553. doi: 10.1615/CritRevTherDrugCarrierSyst.2018021124. Epub 2018/10/16. [DOI] [PubMed] [Google Scholar]
- 41.Cruz L.J., Tacken P.J., Fokkink R., Joosten B., Stuart M.C., Albericio F. Targeted PLGA nano-but not microparticles specifically deliver antigen to human dendritic cells via DC-SIGN in vitro. J Control Release. 2010;144(2):118–126. doi: 10.1016/j.jconrel.2010.02.013. [DOI] [PubMed] [Google Scholar]
- 42.De Bernardis F., Graziani S., Tirelli F., Antonopoulou S. Candida vaginitis: virulence, host response and vaccine prospects. Med Mycol. 2018;56(Suppl_1):S26–S31. doi: 10.1093/mmy/myx139. [DOI] [PubMed] [Google Scholar]
- 43.Thompson F.M., Porter D.W., Okitsu S.L., Westerfeld N., Vogel D., Todryk S. Evidence of blood stage efficacy with a virosomal malaria vaccine in a phase IIa clinical trial. PLoS One. 2008;3(1):e1493. doi: 10.1371/journal.pone.0001493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Blanco J.C., Pletneva L.M., McGinnes-Cullen L., Otoa R.O., Patel M.C., Fernando L.R. Efficacy of a respiratory syncytial virus vaccine candidate in a maternal immunization model. Nat Commun. 2018;9(1):1904. doi: 10.1038/s41467-018-04216-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Smith G., Liu Y., Flyer D., Massare M.J., Zhou B., Patel N. Novel hemagglutinin nanoparticle influenza vaccine with Matrix-M™ adjuvant induces hemagglutination inhibition, neutralizing, and protective responses in ferrets against homologous and drifted A (H3N2) subtypes. Vaccine. 2017;35(40):5366–5372. doi: 10.1016/j.vaccine.2017.08.021. [DOI] [PubMed] [Google Scholar]
- 46.August A., Glenn G.M., Kpamegan E., Hickman S.P., Jani D., Lu H. A Phase 2 randomized, observer-blind, placebo-controlled, dose-ranging trial of aluminum-adjuvanted respiratory syncytial virus F particle vaccine formulations in healthy women of childbearing age. Vaccine. 2017;35(30):3749–3759. doi: 10.1016/j.vaccine.2017.05.045. [DOI] [PubMed] [Google Scholar]
- 47.Medicine NUSNLo. Clinical trials. Gov.
- 48.Pourmand A., Abdollahi M. Current opinion on nanotoxicology. Daru. 2012;20(1):95. doi: 10.1186/2008-2231-20-95. Epub 2013/01/29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sather S., Kenyon K.D., Lefkowitz J.B., Liang X., Varnum B.C., Henson P.M. A soluble form of the Mer receptor tyrosine kinase inhibits macrophage clearance of apoptotic cells and platelet aggregation. Blood. 2007;109(3):1026–1033. doi: 10.1182/blood-2006-05-021634. Epub 2006/10/19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Devadasu V.R., Bhardwaj V., Kumar M.N. Can controversial nanotechnology promise drug delivery? Chem Rev. 2013;113(3):1686–1735. doi: 10.1021/cr300047q. Epub 2013/01/02. [DOI] [PubMed] [Google Scholar]
- 51.Tang S., Chen M., Zheng N. Sub-10-nm Pd nanosheets with renal clearance for efficient near-infrared photothermal cancer therapy. Small. 2014;10(15):3139–3144. doi: 10.1002/smll.201303631. Epub 2014/04/15. [DOI] [PubMed] [Google Scholar]
- 52.Stuehr D.J., Nathan C.F. Nitric oxide. A macrophage product responsible for cytostasis and respiratory inhibition in tumor target cells. J Exp Med. 1989;169(5):1543–1555. doi: 10.1084/jem.169.5.1543. Epub 1989/05/01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Petrarca C., Clemente E., Amato V., Pedata P., Sabbioni E., Bernardini G. Engineered metal based nanoparticles and innate immunity. Clin Mol Allergy. 2015;13(1):13. doi: 10.1186/s12948-015-0020-1. Epub 2015/07/17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Marshall E. Gene therapy death prompts review of adenovirus vector. Science. 1999;286(5448):2244–2245. doi: 10.1126/science.286.5448.2244. [DOI] [PubMed] [Google Scholar]
- 55.Boyce N. Nature Publishing Group; 2001. Trial halted after gene shows up in semen. [DOI] [PubMed] [Google Scholar]
- 56.Cruz J.C., Pfromm P.H., Tomich J.M., Rezac M.E. Conformational changes and catalytic competency of hydrolases adsorbing on fumed silica nanoparticles: II. Secondary structure. Colloids Surf B Biointerfaces. 2010;81(1):1–10. doi: 10.1016/j.colsurfb.2010.06.005. Epub 2010/07/20. [DOI] [PubMed] [Google Scholar]
- 57.Bookstaver M.L., Tsai S.J., Bromberg J.S., Jewell C.M. Improving vaccine and immunotherapy design using biomaterials. Trends Immunol. 2018;39(2):135–150. doi: 10.1016/j.it.2017.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Mohsen M.O., Gomes A.C., Cabral-Miranda G., Krueger C.C., Leoratti F.M., Stein J.V. Delivering adjuvants and antigens in separate nanoparticles eliminates the need of physical linkage for effective vaccination. J Control Release. 2017;251:92–100. doi: 10.1016/j.jconrel.2017.02.031. [DOI] [PubMed] [Google Scholar]


