Abstract
Treponema denticola is a spirochete that is etiologic for periodontal diseases. This bacterium is one of two periodontal pathogens that have been shown to have a complete three step enzymatic pathway (GTSP) that catabolizes glutathione to H2S. This pathway may contribute to the tissue pathology seen in periodontitis since diseased periodontal pockets have lower glutathione levels than healthy sites with a concomitant increase in H2S concentration. In order to be able to demonstrate that glutathione catabolism by the GTSP is critical for the pathogenic potential of T. denticola, allelic replacement mutagenesis was used to make a deletion mutant (Δggt) in the gene encoding the first enzyme in the GTSP. The mutant cannot produce H2S from glutathione since it lacks gamma-glutamyltransferase (GGT) activity. The hemolytic and hemoxidation activities of wild type T. denticola plus glutathione are reduced to background levels with the Δggt mutant and the mutant has lost the ability to grow aerobically when incubated with glutathione. The Δggt bacteria with glutathione cause less cell death in human gingival fibroblasts (hGFs) in vitro than do wild type T. denticola and the levels of hGF death correlate with the amounts of H2S produced. Importantly, the mutant spirochetes plus glutathione make significantly smaller lesions than wild type bacteria plus glutathione in a mouse back lesion model that assesses soft tissue destruction, a major symptom of periodontal diseases. Our results are the first to prove that T. denticola thiol-compound catabolism by its gamma-glutamyltransferase can play a significant role in the in the types of host tissue damage seen in periodontitis.
Keywords: Treponema denticola, ggt mutant, Periodontal disease, Gingival fibroblast death, Glutathione catabolism
Introduction
Periodontal diseases are among the most prevalent human bacterial infections; roughly half of the adult population in the United States suffers from mild to severe periodontitis [1,2]. Although the symptoms are often mild, periodontitis can cause severe pain and tooth loss from connective tissue damage [3]. This disease is due to a complex mixture of inflammation and other host responses to polymicrobial infections [4–8]. Treponema denticola is an anaerobic spirochete that is considered to be a periodontal pathogen since its levels increase in the subgingival microbiome of periodontally-diseased patients [5,9,10], relative to healthy individuals, and it can cause disease symptoms (soft tissue destruction and alveolar bone loss) in animal models [11–13]. Dozens of virulence determinants have been identified in this bacterium [14,15] including potential roles for metabolism and metabolic end-products in virulence [15]. One such metabolic pathway, and end product, that may play a role in T. denticola pathogenesis is the glutathione three-step pathway which metabolizes glutathione into H2S. These two thiol compounds are of particular interest because their levels differ in the gingival crevicular fluid (GCF) of healthy versus periodontally-diseased individuals. Specifically, the levels of glutathione, the predominant thiol compound in human GCF, are lower in the GCF of periodontitis patients [16–18]. This decrease in glutathione levels may contribute to the disease process since glutathione is an antioxidant that can moderate host cell damage [19–21] and since glutathione can also reduce the inflammatory response [22,23]. On the other hand, H2S is present at high levels in pockets associated with periodontitis [24–27] and thus could play a role in periodontal pathology. For example, H2S can induce apoptosis in human periodontal epithelial, ligament and fibroblast cells [28–30] consistent with the finding that the proportion of apoptotic cells in gingival tissue is higher in periodontitis patients [31–33]. H2S has also been shown to cause hemoxidation and hemolysis of human red blood cells, and its activity is enhanced if H2S is generated from cysteine by the T. denticola cystalysin enzyme [34]. Finally, the high levels of the reducing agent H2S could play a role in creating the low levels of oxygen found in diseased periodontal pockets [35,36]. Low oxygen conditions are necessary for the growth of many periodontal pathogens, which are often anaerobes [5,7]. Taken together, these results suggest that alterations in the levels of glutathione and H2S in the periodontal pocket will play a critical role in the extent of tissue/bone damage seen in diseased patients.
Thus, one distinct possibility is that the catabolism of glutathione into hydrogen sulfide by periodontal bacteria plays a significant role in periodontal disease symptomology. Although numerous oral bacteria can generate H2S from cysteine or methionine [37–39], T. denticola and Fusobacterium nucleatum are the only well-established periodontal pathogens that have been shown to be able to make H2S from glutathione [40–42]. We, and others, have identified, purified and characterized the enzymes in a three-step-pathway (GTSP) for glutathione metabolism in T. denticola. Initially, glutathione is cleaved into Cys-Gly and glutamate by gamma-glutamyltransferase (GGT) [40,43]. Then, Cys-Gly is degraded into glycine and cysteine by cysteinylglycinase [44]. Finally, cysteine is hydrolyzed by cystalysin [45–51], releasing ammonia, pyruvate and H2S. Since T. denticola can, by itself, convert glutathione into H2S [40] and since H2S has been shown, as described above, to have toxic effects on host cells in vitro, it is reasonable to hypothesize that the GTSP pathway will play a key role in periodontal pathology. However, this has not been proven. In this report, experiments are described which definitively test the role of the T. denticola GTSP in functional assays related to periodontitis symptoms. We have constructed a ggt-deletion strain and demonstrated that this mutant T. denticola no longer makes H2S from glutathione. This mutant was then used to show that the GTSP is required for maximal induction of host cell death when human gingival fibroblasts are incubated with T. denticola, for hemoxidation and hemolysis of erythrocytes by this spirochete and for this bacterium to grow aerobically. Most importantly, the mutant plus glutathione formed significantly smaller lesions than wild type T. denticola plus glutathione in a mouse back lesion model of soft tissue destruction, an in vivo manifestation of periodontal disease.
Materials and Methods
Bacterial growth conditions.
Unless otherwise noted, Treponema denticola strain ATCC 33520 was grown in GM-1 medium [52] which contains 1.5% heat-inactivated rabbit serum and 6 mM L-cysteine. The bacteria were incubated in a Coy anaerobic chamber (5% CO2, 10% H2, 85% N2). Before use, the culture purity was checked by dark-field microscopy at a magnification of ×500.
Construction of ggt mutant in T. denticola.
We followed the allelic replacement protocol of Xu et al [53] to construct a ggt mutant in T. denticola. First, PCR and standard recombinant DNA procedures were used to construct plasmid pGGT:erm, in which an erythromycin resistance cassette [54] replaced part of the ggt coding region. The ermres gene was flanked by an ~1.1 kb fragment of genomic DNA that is normally upstream of the ggt gene and by an ~1.0 kb fragment of genomic DNA that contained the last four-fifths of the ggt gene and some downstream sequences. The plasmid was linearized by Xho1 and electroporated into T. denticola [53]. After electroporation, the cells were incubated overnight in 2 ml GM-1 broth without erythromycin and then plated on TYGVS containing 0.85% sea plaque agarose with 40 μg/ml erythromycin. Antibiotic resistant colonies, which appeared after 7–12 days, were tested by PCR for the presence of the ermres gene in the ggt locus. PCR fragments containing the junctions between the ermres gene and the T. denticola genome were sequenced from the isolate used in these studies.
Measuring enzyme activities in T. denticola.
Gamma-glutamyl transferase activity (GGT) in T. denticola cells was assayed as described [55] with minor modifications. Briefly, two day bacterial cultures were harvested, suspended in PBS buffer (to one-fourth of original volume) and sonicated. After removing large particles by centrifugation at 14,000 × g for 10min, the soluble material was used for assessing enzyme activity. One ml of soluble material (equal to 4 ml bacterial culture) was added to 1 ml reaction mixture containing 2 mM β-mercaptoethanol, 20 mM Tris buffer at pH 8.0 and 2 mM Na-γ-glutamyl–4-nitroaniline (GNA). After incubation for 2 hr at 37°C, the absorption of each sample at OD 405 nm was determined.
Cysteinylglycinase activity was measured using L-Leu-p-NA as substrate [56]. One ml of concentrated whole bacterial isolate, prepared as described for the GGT assay, was added to 1 ml reaction mixture containing 50 mM Tris-HCl (pH 8.0), 0.2 mM MnCl2 and 2 mM L-Leu-p-NA (final concentrations). The reaction was incubated at 37°C for 60 min before being stopped by cooling on ice. The concentration of the reaction product was measured by the absorbance at OD405nm.
Cystalysin activity was measured as the production of ammonia from L-cysteine, as described by Chu et al [48]. Briefly, the reaction mixture consisted of soluble cell fraction material, prepared as described above, which was incubated with L-cysteine (2 mM) for 60 min at 37°C. The samples were then filtered through an Amicon Centricon filter (10-kDa cutoff) and the material in the flow-through was measured for ammonia using the method of Bauer et al [57].
Determination of H2S production by T. denticola.
Hydrogen sulfide produced by T. denticola, after growth in various conditions, was assayed as described by Siegel [58] with some modifications. Briefly, after removal of bacterial cells by centrifugation, a portion (180 μl) of the supernatant was removed to a fresh tube for H2S determination. Ten μl of 0.02 M N,N-dimethyl-p-phenylenediamine sulfate in 7.2 N HCl and 10 μl of 0.3 M FeCl3 in 1.2 N HCl were added sequentially to the tube. After color development for 30 min at room temperature, the absorbance at 620 nm was measured. The sulfide concentration was determined from an Na2S standard curve.
Hemoxidation and hemolytic analyses.
Hemoxidation (HeO) activity was assessed as described by Leahy and Smith [59] with some modifications [45]. Briefly, 2 ml of 2% (vol/vol) sheep red blood cells (RBCs), after being washed twice to remove soluble hemoglobin, was added to 2 ml of T. denticola cell sonicates containing 6 mM glutathione. Samples were incubated at 37°C with slow rotation (90 rpm) and aliquots were removed at 0, 1, 2, 4, 6, and 8 hrs. The formation of methemoglobin (or sulfhemoglobin) in the RBCs was then determined [60], where 1 HeO unit is defined as 20% methemoglobin (or sulfhemoglobin) formation from the RBCs.
Hemolytic activity (HeA) was determined using sheep red blood cells as described by Chu and Holt [45] with some modifications. Briefly, 2 ml of 2% (vol/vol) sheep RBCs, after being washed twice to remove soluble hemoglobin, was added to 2 ml of T. denticola cell sonicates containing 6 mM glutathione. Samples were incubated at 37°C with slow rotation (90 rpm) and aliquots of the supernatants were removed at various times The amount of hemoglobin (or its oxidized form) released from the RBCs was measured as the optical density of each sample at OD 560nm. The percentage (%) hemolysis was calculated relative to the amount of hemoglobin released from the same number of RBCs lysed by hypotonic distilled water (100% lysis).
Testing aerobic growth of T. denticola.
The ability of T. denticola strains to grow in aerobic conditions was assessed as detailed previously, with some modifications [36]. Bacterial cultures that had been grown anaerobically to an optical density of approximately 0.26 (about 4.9 × 108 cells/ml) were diluted to a starting OD660nm of 0.09 by inoculation into a set of 10 ml tubes each containing six ml (final volume) of basic GM-1 medium with all supplements except L-cysteine. Glutathione, Cys-Gly, or L-cysteine were then added separately to a final concentration of 6 mM. After incubation at 37°C for 48 hrs in aerobic conditions without shaking, 2 ml from each sample was diluted into 4 ml of fresh media of the same composition as in each sample’s original tube. After incubation at 37°C for an additional 48 hrs in aerobic conditions without shaking, bacterial growth in each tube was determined spectrophotometrically at OD660nm.
Evaluating induction of host cell death by T. denticola.
Primary cultures of human gingival fibroblast cells (hGFs) were established from biopsies of gingiva from healthy individuals, with the fully informed consent of the donors and using de-identified samples, following published methods [61,62]. Briefly, gingival tissues were sliced into small pieces and put into tissue culture plates with DF10 media that contained 100 U/ml penicillin and 100mg/ml streptomycin. The samples were grown at 37 °C in 5% CO2. Once cells grew, the cultures were expanded and stocks were frozen in liquid nitrogen. HGF cells between passages 10–16 were used for all experiments. To prepare cells for cell death experiments, the hGFs were grown in T25 flasks containing 50 ml DMEM/10% fetal bovine serum at 37°C in a 5% CO2 incubator [30]. When the hGFs reached ~80% confluency, wild type T. denticola or Δggt mutant bacteria were added to different flasks to a final bacterial concentration of 5 × 108 cells in 50 ml for each flask. Glutathione (final concentration of 2 mM) was added to some flasks. Control flasks contained hGF cells plus glutathione without bacteria. After 24 hours, hGF cell death was quantified by annexinV/propidium iodide staining followed by flow cytometry, as described in Zhang et al [30]. This hGF cell death experiment was done four times (biological replicates). H2S levels were also measured, as described in a previous section, in the supernatant of the cultures from the sample set of one experiment.
Assessing lesion formation by wild type and Δggt T. denticola in the presence of glutathione.
Four groups (8 mice/group) of BALB/c mice were infected subcutaneously, as described by Kesavalu et al [63], on the back with 5 × 1010 of (i) wild type T. denticola, (ii) wild type bacteria and glutathione (16 mM), (iii) Δggt mutant cells, (iv) Δggt mutant cells and glutathione (16 mM). The bacteria were grown with glutathione only if they were to be used for the “and glutathione” inoculations. A fifth group of mice (control) was challenged with media plus glutathione (16 mM) alone. After challenge, the animals were monitored for symptoms of infection. Subcutaneous abscess and/or necrotic skin lesion sizes (length and width) were measured daily for 15 days and the area was determined and expressed in square millimeters. The maximum lesion size for each mouse was used to generate the mean lesion size shown for each group of mice. All mouse studies were done following protocols approved by the UTHSCSA Institutional Animal Care and Use Committee (IACUC).
Statistical analyses.
The data for enzyme activities, aerobic growth, hGF cell death and mouse lesion formation are expressed as means ± standard deviations. For the hGF cell death and murine lesion formation experiments, the statistical significance was evaluated for independent experiments using the Tukey’s multiple comparison test. Differences between samples were considered statistically significant if the P value was <0.05.
Results
A Δggt mutant specifically lacks gamma-glutamyl transferase activity and does not produce H2S from glutathione.
In order to evaluate the biological role of the three-steppathway for glutathione metabolism (GTSP), a T. denticola mutant in the ggt gene was created by allelic replacement mutagenesis [53]. One erythromycin-resistant strain was characterized by sequencing PCR products generated from the region around the ggt gene. The sequencing showed that the ermF-ermAM ermres cassette had replaced the genomic DNA segment encoding amino acids 17–36 of T. denticola GGT (Gen Bank EMB43424.1). As expected, the rest of the DNA around the ermres cassette matched the genomic sequence of T. denticola strain ATCC 33520 [http://www.homd.org/index.php].
The sequence analysis, indicating that the disrupted ggt gene should not produce GGT, was corroborated by testing the mutant strain for GGT activity. As predicted, the Δggt cells had no GGT activity above background whereas wild type bacteria had significant levels of GGT (Fig. 1A). These data also indicate that T. denticola does not have a second, unidentified ggt gene. As expected, the activities or expression of the two other enzymes in the GTSP, CGase and cystalysin, were not affected by the Δggt mutation (Figs. 1B and 1C).
Fig. 1. GTSP enzyme activity levels in wild type T. denticola and its isogenic Δggt mutant.
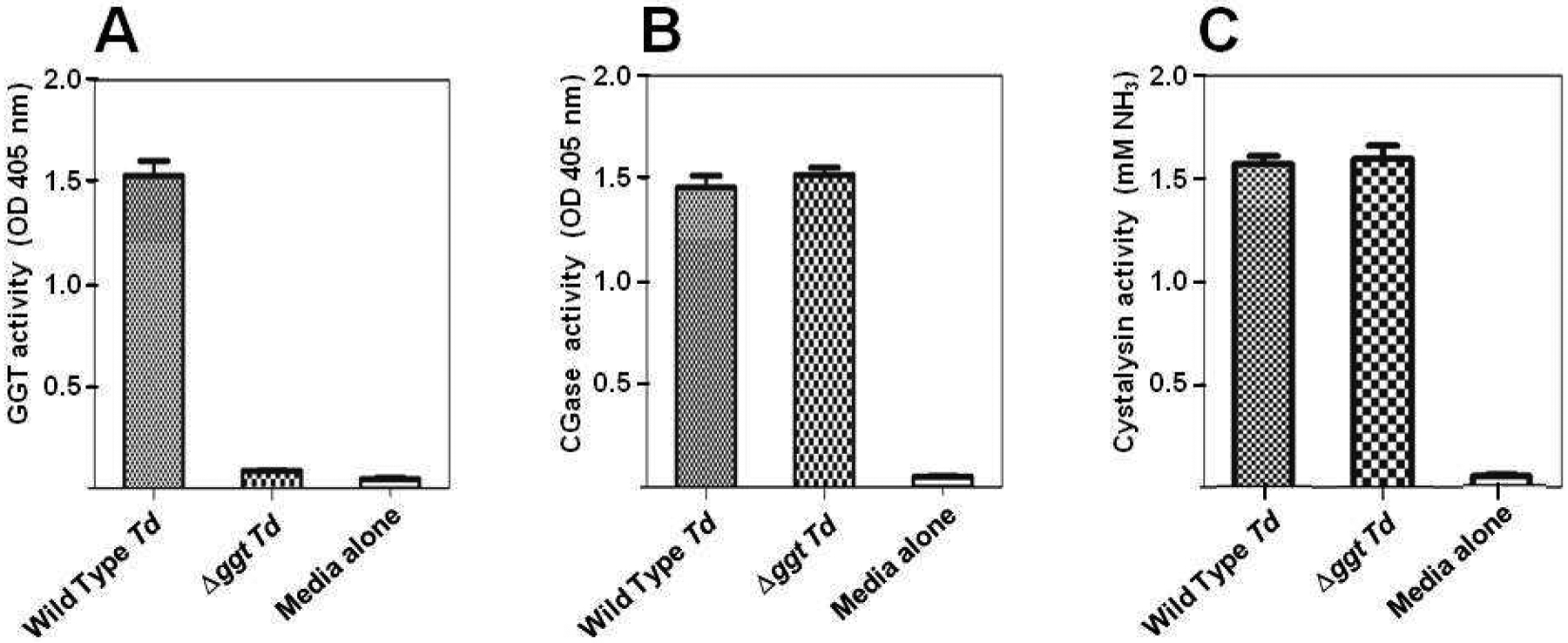
The levels of enzyme activity for each of the three GTSP proteins were measured in equal amounts of clarified extract from wild type and mutant (Δggt) T. denticola (Td) cells. The Media alone control contained buffer with no bacteria. (A) The gamma-glutamyl transferase activity was assayed using GNA as the substrate. The levels of the resulting product were determined by the absorption of each sample at 405 nm. (B) Cysteinylglycinase activity was measured using L-Leu-p-NA as the substrate. The levels of the resulting product, 4-nitroaniline, were determined by the absorption of each sample at 405 nm. (C) Cystalysin activity was assessed using L-cysteine as the substrate. The resulting levels of NH3 were determined as detailed in Materials and Methods. Each experiment was done three times. The bars indicate standard deviations of the mean.
Since the Δggt strain does not have GGT activity, this mutant should not be able to produce H2S from glutathione via the GTSP, unless there is an alternate catabolic pathway that can carry out the same overall reaction. To rule out this possibility, the production of H2S from three substrates was examined in wild type and mutant cells. As seen previously [40], wild type T. denticola was able to make H2S from glutathione, Cys-Gly or L-cysteine (Fig. 2). However, the Δggt mutant could not catabolize glutathione into H2S (Fig. 2), although it made normal levels of H2S from Cys-Gly and L-cysteine. This proves that this disruption of the ggt gene inactivates the first step in the glutathione three enzyme glutathione catabolism pathway and that the GTSP is the only way for T. denticola to produce H2S from glutathione.
Fig. 2. Production of H2S from wild type T. denticola and its isogenic Δggt mutant.
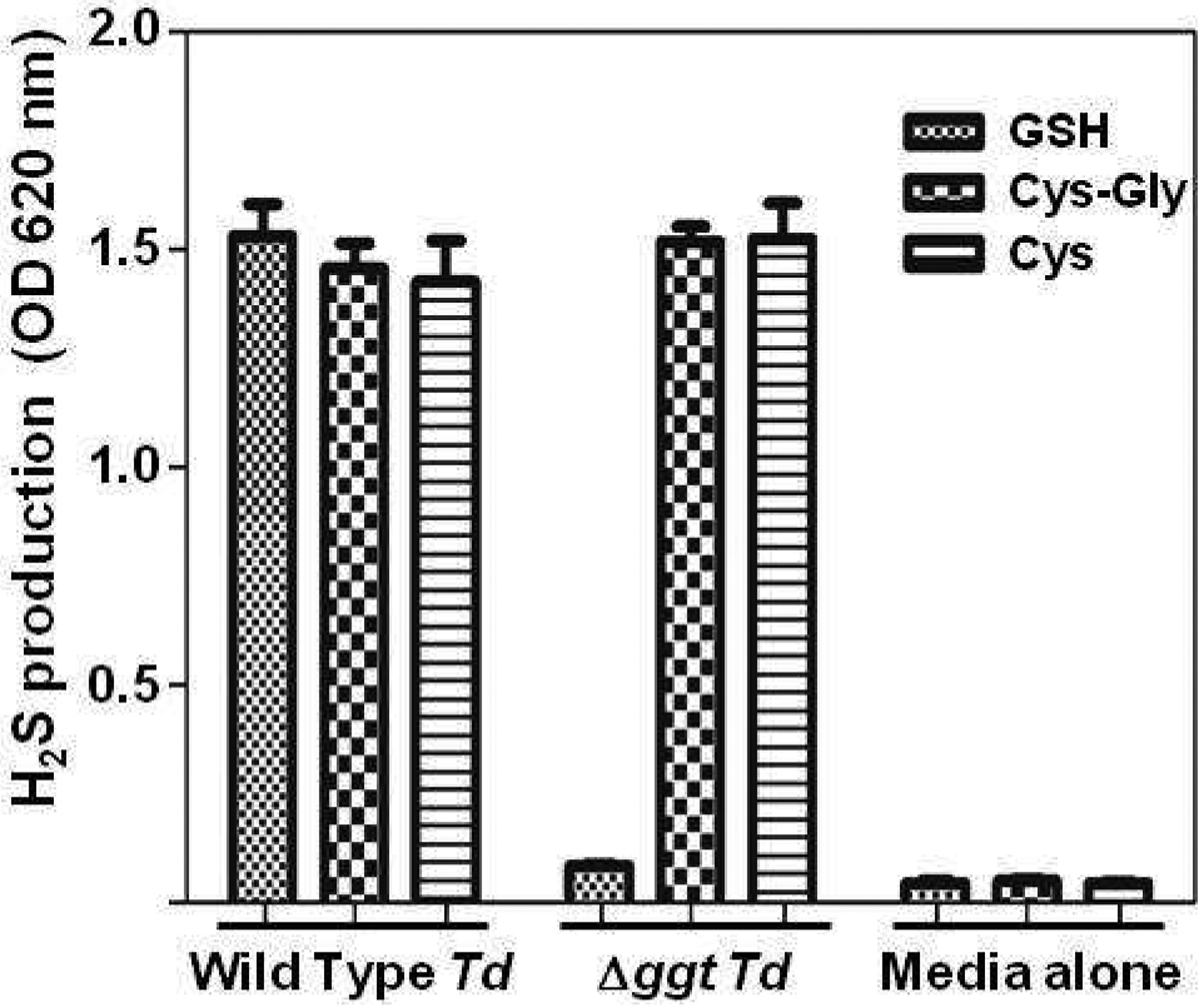
Parental T. denticola (wild type Td) and its isogenic Δggt mutant were grown anaerobically to an OD620 nm of 0.320. The indicated substrates, GSH (glutathione), Cys-Gly (L-cysteine-L-glycine), or Cys (L-cysteine) were then added individually to each sample. H2S production (as OD at 620 nm) was measured 1 hr later. Control = media alone with no bacteria. Each experiment was done three times. Error bars indicate standard deviations of the mean.
The T. denticola Δggt mutant has decreased hemolytic and hemoxidative activity.
T. denticola has been shown to be able to lyse red blood cells and oxidize hemoglobin to methemoglobin in the presence of the GTSP substrates glutathione, Cys-Gly or L-cysteine [40]. This suggests, but does not prove, that the GTSP plays a key role in these hemolytic (HeA) and hemoxidative (HeO) functions. To assess the relative contribution of the GTSP to the HeA and HeO activities of T. denticola, their levels were measured in the Δggt mutant. Wild type T. denticola had high levels of HeA and HeO activity when glutathione was added, but the amount of activity in the Δggt cells was the same as in the media alone controls (Fig. 3). When Cys-Gly or L-cysteine were used as substrates in the assays instead of glutathione, the Δggt mutant and wild type strain had similar HeA and HeO activities (data not shown) indicating that the GGT protein does not play a direct role in hemolysis and hemoxidation. These results demonstrate that the GTSP has a major role in the ability of T. denticola to hemoxidize and lyse erythrocytes and support the hypothesis that H2S production is needed for these functions.
Fig. 3. Hemoxidation and hemolytic activity in wild type T. denticola and the Δggt mutant.
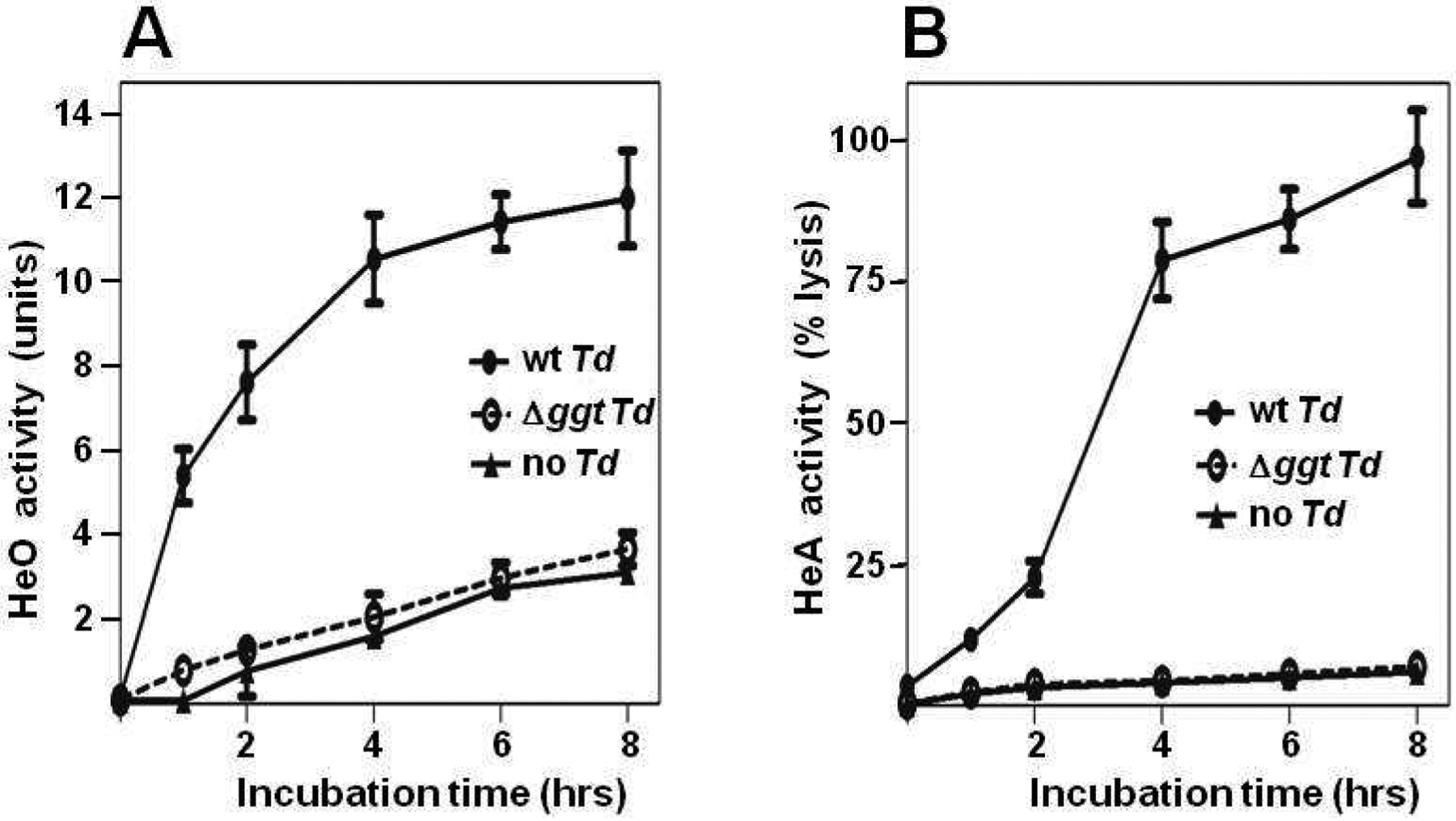
Material from sonicates of wild type T. denticola (wt Td) cells or Δggt mutant cells were added to sheep red blood cells (RBCs) in media with glutathione. Control (no Td) = red blood cells in media with glutathione but no bacteria. (A) Aliquots of RBCs were removed at the indicated times and hemoxidation activity (HeO), the formation of methemoglobin (or sulfhemoglobin) in the RBCs, was measured. One HeO unit is defined as 20% methemoglobin (or sulfhemoglobin) formation from the RBCs. (B) Aliquots of sample supernatants were removed at the indicated times. Hemolytic activity (HeA) was assayed by measuring the amount of hemoglobin (or its oxidized form) released from the RBCs which was determined by the optical density of the RBC supernatant at 560 nm. The percentage hemolysis was calculated relative to the amount of hemoglobin released from RBCs by hypotonic lysis, which was set as 100%.
T. denticola does not grow aerobically without GGT.
The growth rates of the Δggt mutant and wild type cells were the same when T. denticola, an obligate anaerobe, was grown anaerobically in GM-1 media with L-cysteine. This was not surprising since the mutant still has cystalysin activity and thus can produce pyruvate, which is used by T. denticola to increase its growth [40]. If the bacteria were grown anaerobically in GM-1 media with glutathione instead of L-cysteine, the Δggt cells grew more slowly and reached a lower final cell density than wild type cells since the mutant cannot produce pyruvate from glutathione (data not shown). Interestingly, even though T. denticola is an obligate anaerobe, it can grow in aerobic conditions when glutathione or L-cysteine are included in the media [36]. The ability to grow aerobically correlates with the production of H2S by T. denticola, leading to the hypothesis that the organism’s GTSP is required for aerobic growth. To test this inference, the aerobic growth of wild type and Δggt cells were compared. As expected, wild type T. denticola grew aerobically when incubated with glutathione, Cys-Gly or L-cysteine (Fig. 4). The Δggt cells also grew aerobically when the media was supplemented with Cys-Gly or L-cysteine but not when glutathione was the only supplement added (Fig. 4). To determine why the mutant cells could not grow aerobically with glutathione, the levels of oxygen and H2S were measured in the samples that had been grown with glutathione. The media with wild type T. denticola had high levels of H2S (2.1 ± 0.090 mM) whereas the media with the mutant spirochetes had significantly (p<0.001) less H2S (0.07 ± 0.02 mM). Conversely, wild type T. denticola samples had low levels of dissolved oxygen (0.05 ± 0.006 mM) and the samples from the mutant bacteria had significantly (p<0.001) higher levels of dissolved oxygen (2.1 ± 0.115 mM). These results prove that the GTSP is essential for T. denticola to grow aerobically and reinforce the concept that it is the production of H2S by the GTSP that leads to the reduction of oxygen levels allowing this anaerobe to grow “aerobically”.
Fig. 4. Testing aerobic growth of wild type and mutant T. denticola.
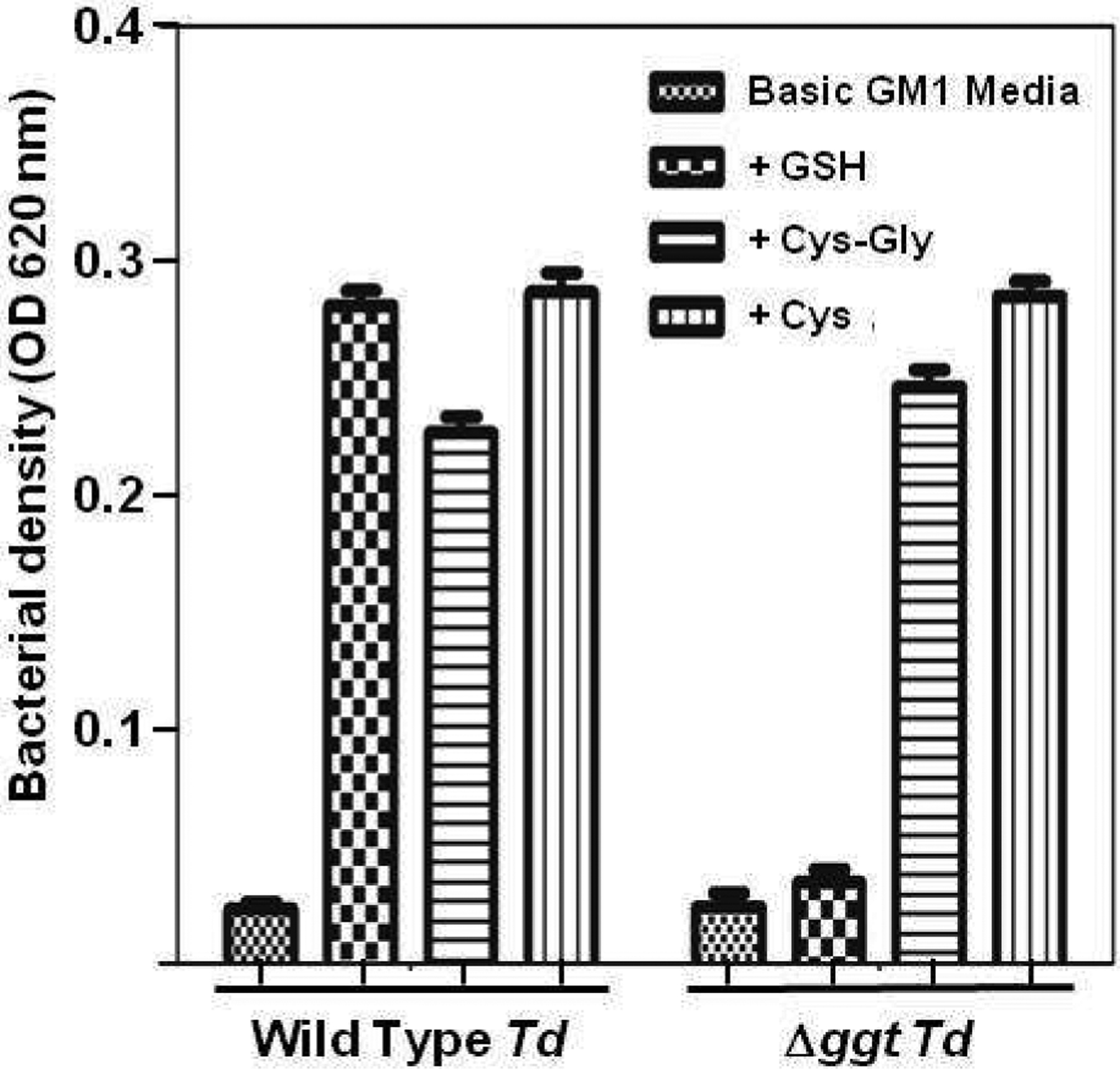
Wild type T. denticola and the isogenic Δggt mutant were inoculated, to an initial OD620nm of 0.09, into basic GM-1 media without L-cysteine. Glutathione (GSH), L-cysteine-L-glycine (Cys-Gly) and L-cysteine (Cys) were added individually to different tubes as indicated and the samples were incubated aerobically. After 48 hr each sample was diluted three-fold into fresh media with the same additions and incubated aerobically for another 48 hr. Growth was then measured as the absorbance of each sample at O.D620nm. Basic GM-1 media alone (without Cys, GSH or CysGly) was used as a negative control. Each experiment was done three times. Error bars indicate standard deviations of the mean.
Catabolism of glutathione to H2S by the T. denticola GTSP induces gingival fibroblast cell death.
H2S has been shown to cause apoptosis in human gingival fibroblast (hGF) cells and periodontal ligament cells [30]. Thus, it is reasonable to postulate that catabolism of glutathione by the T. denticola GTSP, which produces H2S, will induce host cell death. To assess this possibility, wild type and Δggt cells were incubated with hGF cells, in the presence or absence of glutathione. After 24 hours, Annexin V and propidium iodide staining was used to measure cell death. Dead hGFs include early apoptotic cells (Annexin V positive/propidium iodide low), late apoptotic cells (V+/propidium iodide high) and necrotic hGFs (propidium iodide high) (Fig. 5A). Gingival fibroblasts incubated with wild type T. denticola and glutathione had significantly more cell death than hGFs grown with wild type bacteria without glutathione (Fig. 5B). This induction required the GTSP since Δggt cells with glutathione induced much less host cell death (12%) than did wild type bacteria with glutathione (52%). These results correlated well (Pearson correlation coefficient = 0.97) with the levels of H2S in the same samples (Fig. 5C). For example, the media from hGFs incubated with mutant T. denticola and glutathione had 0.13 mM H2S, whereas wild type bacteria produced 1.56 mM H2S from glutathione. The results demonstrate that the GTSP plays a major role in the ability of T. denticola to induce cell death in human gingival fibroblasts and strongly argue that it is the production of H2S from glutathione that leads to gingival fibroblast death.
Figure 5. Effect of T. denticola ggt gene deletion on death of hGF cells.
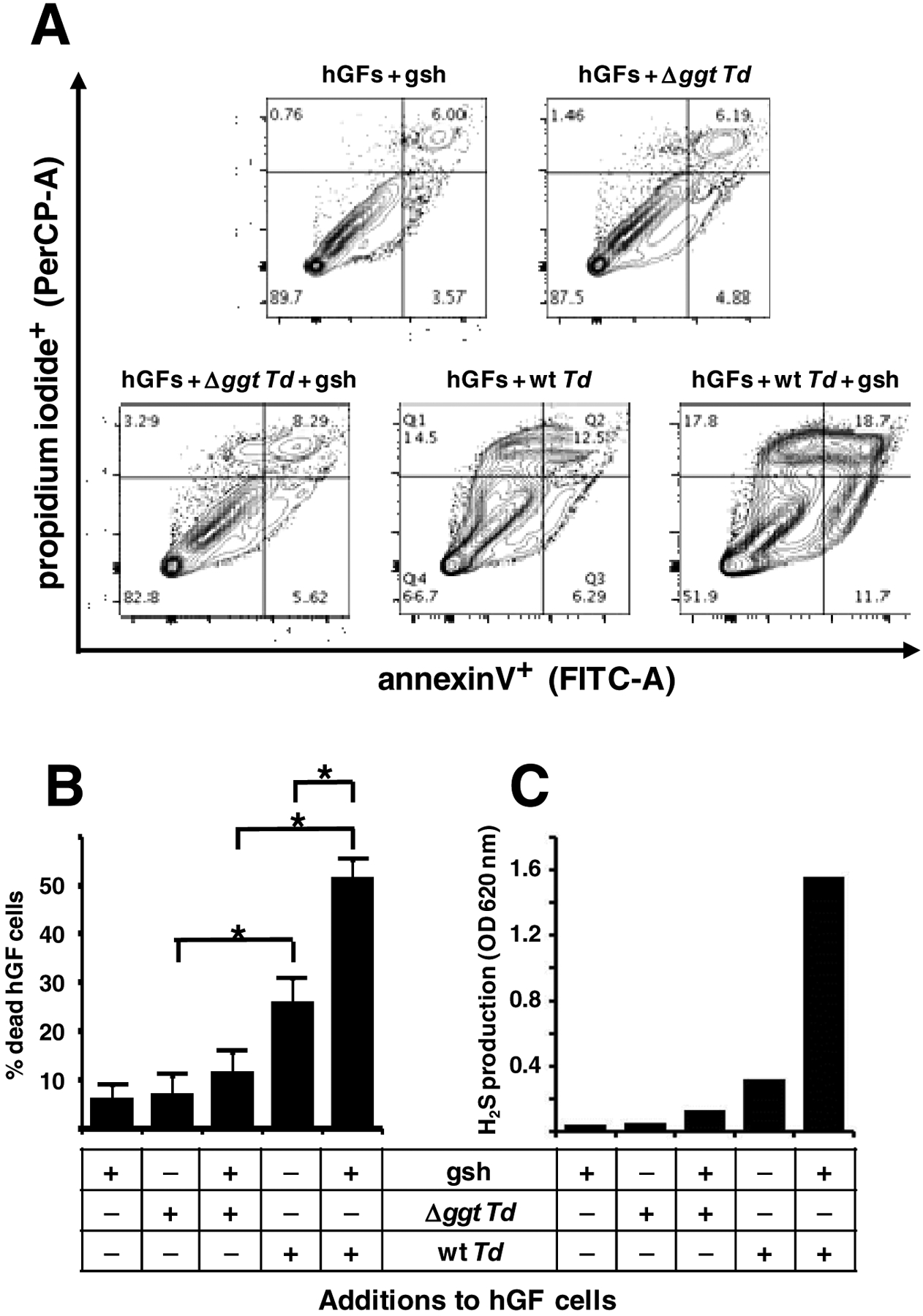
Wild type T. denticola (wt Td) or Δggt mutant bacteria and glutathione (gsh) were added in various combinations to different tissue culture flasks of human gingival fibroblast cells (hGFs) at near80% confluency. After 24 hours incubation, cell death (apoptosis plus necrosis) of hGFs was measured by flow cytometry. The experiment was done four times (biological replicates). The levels of H2S in the media were also measured in one experiment. (A) The hGF cells from the samples indicated were stained with annexin V and propidium iodide followed by flow cytometry analysis (30). Annexin V levels were measured with a FITC-A filter set and propidium iodide staining was measured with a PerCP-A filter set. The results of one representative experiment (of four) are shown. (B) Quantification of the four flow cytometry plots, where % death of hGF cells in the indicated samples equals the percentage of cells that stained with Annexin V, propidium iodide, or both. (C) H2S levels in the culture supernatants of the indicated samples. *, p = 0.001 per Tukey’s test analysis.
The T. denticola Δggt mutant makes smaller lesions in a mouse back abscess model.
Since physiologically relevant concentrations of glutathione (2mM) were used in the experiments showing that GGT had a major role in the ability of T. denticola to cause host cell death in vitro, we hypothesized that the GTSP would play a similarly important role in causing host tissue damage in vivo. Thus, a murine back abscess model [63] was used to assess the role of the GTSP in soft tissue destruction, one of the hallmarks of periodontal disease. Animals were challenged with wild type T. denticola or the Δggt mutant, with or without glutathione being added, and the necrotic skin lesion sizes were measured over time. Importantly, mice challenged with Δggt bacteria and glutathione had significantly (p = 0.0007) smaller lesions (113.1 mm2) than the animals inoculated with wild-type T. denticola plus glutathione (148.6 mm2) (Fig. 6). This result proves our hypothesis that GTSP catabolism of glutathione by T. denticola plays a role in soft tissue destruction in vivo, most likely because of its production of H2S.
Figure 6. Effect of T. denticola ggt gene deletion on murine back lesion size.
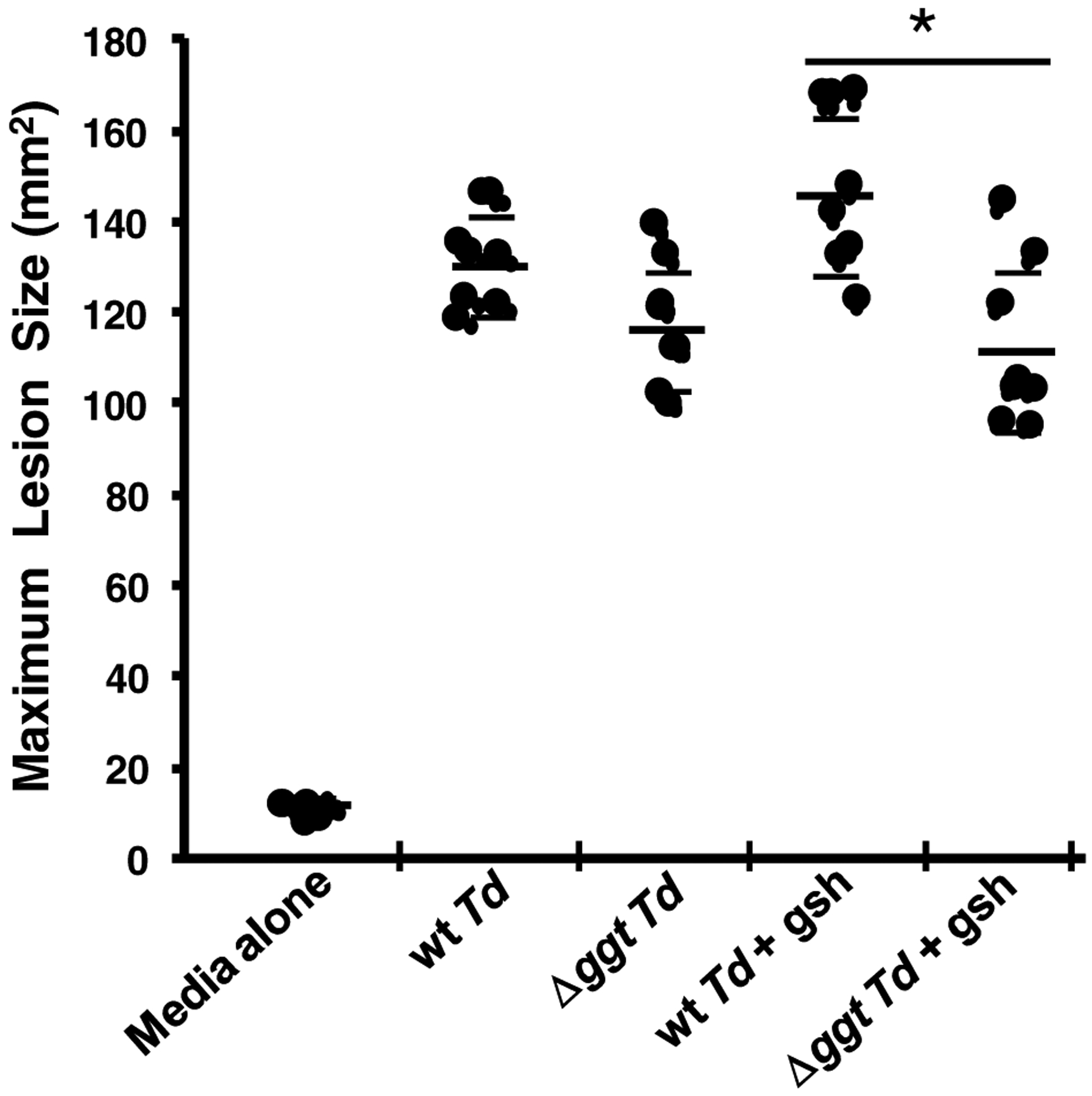
Wild type T. denticola (wt Td) or Δggt mutant bacteria with or without glutathione (gsh) were injected subcutaneously into the backs of mice. Control mice were injected with media alone (no bacteria, no glutathione) The size of each lesion was measured as its area (mm2) daily for 15 days. The maximum lesion size for each mouse was used to generate the mean lesion size shown as a longer, thicker bar for each group of mice (eight mice per group). The thinner, shorter bars delimit the standard deviations of the mean for each group of mice. *, p = 0.0007 per Tukey’s test analysis.
Discussion
The construction of a defined ggt deletion mutant (Δggt) in T. denticola has allowed us to show definitively that the catabolism of glutathione is necessary for several virulence activities of the bacterium in vitro. The hemolytic and hemoxidation activities of wild type T. denticola plus glutathione are absent in the Δggt mutant and the mutant cannot grow aerobically when incubated with glutathione whereas wild type T. denticola can. Most relevant to a role for the T. denticola GTSP in tissue damage in periodontal disease is the fact that the Δggt bacteria with glutathione cause less cell death in human gingival fibroblasts (hGFs) in vitro than do wild type spirochetes. Since the levels of hGF death correlate with the amounts of H2S produced in these studies, the results are consistent with the hypothesis that it is the production of H2S from glutathione by the GTSP of T. denticola that leads to gingival fibroblast death.
Surprisingly, there were higher levels of H2S (0.32 mM) when wild type T. denticola was incubated with hGFs without exogenous glutathione than in the sample incubated with the Δggt mutant without glutathione (0.05 mM H2S) (Fig. 5C). This correlated with the significant increase in hGF cell death seen with wild type bacteria without glutathione (26%) versus the mutant samples without glutathione (7%) (Fig. 5B). This outcome implies that there is endogenous glutathione that the wild type spirochetes can use to make H2S which then causes cell death. In fact there are two potential sources for endogenous glutathione in all of the samples: the fetal bovine serum (FBS) added to the media and the hGF cells. Although the amount of glutathione in FBS is not routinely measured, the literature clearly shows that FBS can contain glutathione [64,65] although the amount can vary as much as thirty-fold depending upon the lot of FBS [66]. In addition, host cells can generate glutathione from the basic amino acids that are present in the DMEM component of the growth media. Although most of the host-cell-generated glutathione is retained inside the cell, where it serves essential functions in antioxidant defense and the regulation of metabolic functions [67], some is secreted. The amount secreted is typically in the μM range [68,69]. The amount of glutathione made by hGF cells has not been measured, but it could be higher than seen in other host cells since hGFs could be a source for the higher levels of glutathione (0.5 – 2.5 mM) found in gingival crevicular fluid [16,69]. Thus, clearly there will be some endogenous glutathione that can be used by the GTSP of wild type T. denticola to generate H2S in the absence of exogenously added glutathione.
Another unexpected result, since we have shown that the Δggt mutant does not make H2S from glutathione, is the observation that the addition of glutathione to hGF cells and Δggt spirochetes led to an increase in H2S levels (from 0.052 to 0.13 mM, Fig 5C) and more cell death (from 7% to 12%, Fig. 5B). Although this increase in cell death was not statistically significant, all four paired samples had the same pattern. The rise in cell death seen when glutathione was added to hGF cells and Δggt spirochetes would appear to be an additive effect from two other ways that H2S, which leads to increased cell death, was being generated in this experiment. First, the hGF cells by themselves (no bacteria) can make H2S (0.045 mM) in the presence of glutathione (Fig. 5C). Second, Δggt mutant bacteria can make H2S (0.052 mM) in the absence of glutathione (Fig. 5C); this H2S is presumably being made from L-cystine (a dimer of cysteine), which is present in the media, by the still-present cystalysin in Δggt T. denticola. Since these two means of making H2S are independent of each other, they should produce an additive amount of H2S (0.045 mM + 0.052 = 0.097 mM H2S). Indeed, 0.097 mM H2S is close to the 0.13 mM H2S seen in the sample with hGF cells plus the Δggt mutant and glutathione. This increase in H2S concentration presumably leads to the increased level (12%) of host cell death seen when hGF cells, the Δggt mutant and glutathione are added together (Fig. 5B), as opposed to separately. A possible alternate explanation for the 7% cell death seen when mutant bacteria are added to hGF cells in the absence of glutathione is that other T. denticola molecules can cause host cell death. This is certainly a possibility since the T. denticola Sip protein has been shown to induce apoptosis in human T cells [70] and outer membrane extracts from T. denticola can increase apoptosis in porcine endothelial cells [71]. However, we do not think these other T. denticola virulence factors contribute much to hGF cell death in our experiments since the correlation between H2S levels and the percentage of cell death is so tight (Pearson correlation coefficient = 0.97) in our data (Fig. 5).
Importantly, the in vitro results demonstrating that glutathione catabolism by T. denticola is involved in the pathogenic potential of the organism were confirmed in vivo. Specifically, the mutant spirochetes plus glutathione made significantly smaller lesions than wild type bacteria plus glutathione in a mouse back lesion model that assesses soft tissue destruction, a major symptom of periodontal diseases. These results are the first to prove that T. denticola thiol-compound catabolism can play a critical role in vivo in the types of host tissue damage seen in periodontitis. Somewhat unexpectedly, wild type T. denticola appeared (p=0.26) to induce larger lesions (132.5 mm2) than ggt mutant spirochetes (118 mm2) even in the absence of exogenous glutathione. The most likely explanation is that wild type bacteria can make H2S from the low levels of glutathione found in blood serum (1 – 4 μM as opposed to 0.5 – 2.5 mM in gingival crevicular fluid) [16,69] and cause tissue damage even without exogenous glutathione.
Even though our results demonstrate that the GTSP is important for T. denticola’s ability to cause soft tissue destruction, lesions still formed in mice challenged with the Δggt mutant, which cannot make H2S from glutathione (Fig. 6). This data indicates that either (1) there is enough endogenous cysteine in mice to generate H2S using the still-functional T. denticola cystalysin, and/or, (2) there are other proteins or metabolites produced by T. denticola that could also contribute to host tissue damage. In fact, the latter is likely to be true; dozens of potential T. denticola virulence factors have been identified in various in vitro experiments (14,15,72). However, only a handful of these non-GTSP proteins have been tested for their pathogenic potential in animal models. Deletion mutants in the genes encoding two T. denticola proteases have been tested for their virulence in the mouse back abscess model. A mutant in dentilisin, which alters the structural organization of T. denticola’s outer sheath, formed lesions that were 25–40% smaller than the lesions made by wild type spirochetes (73). The cysteine protease dentipain is also important for soft tissue destruction since a T. denticola dentipain mutant made 33% smaller lesions than wild type bacteria, although the difference is only seen at later time points (days 4–6) postinoculation (74). Bian et al (75) have characterized a mutant in the cyclic di-GMP binding protein (TDE0214) in vitro and in vivo. The Δtde0214 strain was less motile than wild type and did not form biofilms very well. When tested in the murine back abscess model, the lesions made by Δtde0214 bacteria were 25% the size of those made by wild type T. denticola. The T. denticola neuraminidase TDE0471 removes sialic acid from host serum proteins and plays a role in bacterial virulence in vivo; a TDE0471 deletion mutant formed lesions in the back abscess model that were half the size of lesions made by wild type spirochetes (76). Finally, Miller et al (77) assessed the effect of a T. denticola mutant in FhbB, a surface-exposed lipoprotein that binds to the complement system regulatory protein factor H and blocks dentilisin-mediated cleavage of factor H. In the mouse back abscess model, the ΔfhbB mutant had lesions that were 50% the size of those made by wild type T. denticola. Thus, multiple molecules/pathways, including the generation of H2S by the catabolism of glutathione, which we have demonstrated here, contribute to the soft tissue destruction caused by T. denticola. The relative contributions of the various T. denticola virulence determinants to lesion formation can be evaluated only by constructing combinations of virulence protein mutants and comparing them directly to each other in the back abscess model.
Highlights.
T. denticola lacking γ-glutamyl transferase (Δggt) cannot make H2S from glutathione
The T. denticola Δggt mutant has decreased hemolytic and hemoxidative activity
T. denticola does not grow aerobically without γ-glutamyl transferase activity
Without GGT, T. denticola does not fully induce cell death in gingival fibroblasts
The T. denticola Δggt mutant makes smaller lesions in a mouse back abscess model
Acknowledgments
We thank David Cappelli for helpful discussions about the animal experiments, Yangming Xiao for his advice in analyzing the FACS data, and Robert Danso for reviewing our statistical analyses.
Funding
This work was supported by a National Institutes of Health grant (1RO1DE023532) from the NIH/NIDCR to L.C.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflict of interest
On behalf of all authors, the corresponding author, Dr. Kolodrubetz, states that there is no conflict of interest.
References
- 1.Eke PI, Dye BA, Wei L, Thornton-Evans GO, Genco RJ. 2012. Prevalence of periodontitis in adults in the United States: 2009 and 2010. J Dent Res 91:914–920. [DOI] [PubMed] [Google Scholar]
- 2.Philstrom BL, Michalowicz BS, Johnson NW. 2005. Periodontal diseases. Lancet 366:1809–1820. [DOI] [PubMed] [Google Scholar]
- 3.Armitage GC. 2004. Periodontal diagnoses and classifications of periodontal diseases. Periodontol 2000 34:9–21. [DOI] [PubMed] [Google Scholar]
- 4.Van Dyke TE. 2008. The management of inflammation in periodontal disease. J Periodontol 79:1601–1608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Socransky SS, Haffajee AD, Cugini MA, Smith C, Kent RL Jr. 1998. Microbial complexes in subgingival plaque. J Clin Periodontol 25:134–44. [DOI] [PubMed] [Google Scholar]
- 6.Brogden KA, Guthmiller JM, Taylor CE. 2005. Human polymicrobial infections. Lancet 365:253–255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Socransky SS, Haffajee AD. 2005. Periodontal microbial ecology. Periodontol 2000 38:135–187. [DOI] [PubMed] [Google Scholar]
- 8.Darveau RP. 2010. Periodontitis: a polymicrobial disruption of host homeostasis. Nature Rev Microbiol 8:481–490. [DOI] [PubMed] [Google Scholar]
- 9.Ellen RP, Galimanas VB. 2005. Spirochetes at the forefront of periodontal infections. Periodontol 2000, 38:13–32. [DOI] [PubMed] [Google Scholar]
- 10.Takeuchi Y, Umeda M, Sakamoto M, Benno Y, Huang Y, Ishikawa I. 2001. Treponema socranskii, Treponema denticola, and Porphyromonas gingivalis are associated with severity of periodontal tissue destruction. J Periodontol 72:1354–1363. [DOI] [PubMed] [Google Scholar]
- 11.Kesavalu L, Sathishkumar S, Bakthavatchalu V, Matthews C, Dawson D, Steffen M, Ebersole J. 2007. Rat model of polymicrobial infection, immunity, and alveolar bone resorption in periodontal disease. Infect Immun 75:1704–1712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lee SF, Andrian E, Rowland E, Marquez IC. 2009. Immune response and alveolar bone resorption in a mouse model of Treponema denticola infection. Infect Immun 77:694–698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ebersole JL, Kesavalu L, Schneider SL, Machen RL, Holt SC. 1995. Comparative virulence of periodontopathogens in a mouse abscess model. Oral Dis 1:115–128. [DOI] [PubMed] [Google Scholar]
- 14.Ishihara K 2010. Virulence factors of Treponema denticola. Periodontol 2000 54:117–135. [DOI] [PubMed] [Google Scholar]
- 15.Dashper S, Seers C, Tan K, Reynolds E. 2011. Virulence factors of the oral spirochete Treponema denticola. J Dent Res 90:691–703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chapple IL, Brock G, Eftimiadi C, Matthews JB. 2002. Glutathione in gingival crevicular fluid and its relation to local antioxidant capacity in periodontal health and disease. J Clin Pathol Mol Pathol 55:367–373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Grant MM, Brock GR, Matthews JB, Chapple IL. 2010. Crevicular fluid glutathione levels in periodontitis and the effect of non-surgical therapy. J Clin Periodontol 37:17–23. [DOI] [PubMed] [Google Scholar]
- 18.Savita AM, Sarun E, Arora S, Krishnan S. 2015. Evaluation of glutathione level in gingival crevicular fluid in periodontal health, in chronic periodontitis and after nonsurgical periodontal therapy: A clinicobiochemical study. Contemp Clin Dent 6:206–210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Townsend DM, Tew KD, Tapiero H. 2003. The importance of glutathione in human disease. Biomed Pharmacother 57:145–155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sies H 1999. Glutathione and its role in cellular functions. Free Rad Bíol Medn 27:916–921. [DOI] [PubMed] [Google Scholar]
- 21.Pastore A, Federici G, Bertini E, Piemonte F. 2003. Analysis of glutathione: implication in redox and detoxification. Clin Chim Acta 333:19–39. [DOI] [PubMed] [Google Scholar]
- 22.Paolicchi A, Dominici S, Pieri L, Maellaro E, Pompella A. 2002. Glutathione catabolism as a signaling mechanism. Biochem Pharmacol 64:1027–1035 [DOI] [PubMed] [Google Scholar]
- 23.Ghezzi P 2011. Role of glutathione in immunity and inflammation in the lung. Int. J. Gen. Medn 4:105–113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Persson S 1992. Hydrogen sulfide and methyl mercaptan in periodontal pockets. Oral Microbiol Immunol 7:378–379. [DOI] [PubMed] [Google Scholar]
- 25.Rizzo AA. 1967. The possible role of hydrogen sulfide in human periodontal disease. 1. Hydrogen sulfide production in periodontal pockets. Periodontics 5:233–236. [PubMed] [Google Scholar]
- 26.Horowitz A, Folke LE. 1973. Hydrogen sulfide production in the periodontal environment. J Periodontol 44:390–395. [DOI] [PubMed] [Google Scholar]
- 27.Morhart RE, Mata LJ, Sinskey AJ, Harris RS. 1970. A microbiological and biochemical study of gingival crevice debris obtained from Guatemalan Mayan Indians. J Periodontol 11:644–649. [DOI] [PubMed] [Google Scholar]
- 28.Murata T, Yaegaki K, Qian W, Herai M, Calenic B, Imai T, Sato T, Tanaka T, Kamoda T, Ii H. 2008. Hydrogen sulfide induces apoptosis in epithelial cells derived from human gingiva. J Breath Res 2: 017007. [DOI] [PubMed] [Google Scholar]
- 29.Yaegaki K, Qian W, Murata T, Imai T, Sato T, Tanaka T, Kamoda T. 2008. Oral malodorous compound causes apoptosis and genomic DNA damage in human gingival fibroblasts. J Periodontal Res 43:391–399. [DOI] [PubMed] [Google Scholar]
- 30.Zhang Z, Dong Z, Chu L. 2009. H2S induces apoptosis of periodontium cells. J Perio Res 45:71–78. [DOI] [PubMed] [Google Scholar]
- 31.Gamonal J, Bascones A, Acevedo A, Blanco E, Silva A. 2001. Apoptosis in chronic adult periodontitis analyzed by in situ DNA breaks, electron microscopy, and immunohistochemistry. J Periodontol 72:517–525. [DOI] [PubMed] [Google Scholar]
- 32.Jarnbring F; Somogyi E; Dalton J; Gustafsson A; Klinge B. 2002. Quantitative assessment of apoptotic and proliferative gingival keratinocytes in oral and sulcular epithelium in patients with gingivitis and periodontitis. J Clin Periodontol 29:1065–1071. [DOI] [PubMed] [Google Scholar]
- 33.Listyarifah D, Al-Samadi A, Salem A, Syaify A, Salo T, Tervahartiala T, Grenier D, Nordström DC, Sorsa T, Ainola M. 2017. Infection and apoptosis associated with inflammation in periodontitis: An immunohistologic study. Oral Dis 23:1144–1154. [DOI] [PubMed] [Google Scholar]
- 34.Chu L, Ebersole JL, Holt SC. 1999. Hemoxidation and binding of the 46-kDa cystalysin of Treponema denticola leads to a cysteine-dependent hemolysis of human erythrocytes. Oral Microbiol Immunol 14:293–303. [DOI] [PubMed] [Google Scholar]
- 35.Mettraux GR, Gusberti FA, Graf H. 1984. Oxygen tension (pO2) in untreated human periodontal pockets. J Periodontol 55:516–521. [DOI] [PubMed] [Google Scholar]
- 36.Lai Y, Chu L. 2008. A novel mechanism for aerobic growth of anaerobic Treponema denticola. Appl Environ Microbiol 74: 73–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Claesson R, Edlund MB, Persson Carlsson J. 1990. The production of volatile sulfur compounds by various Fusobacterium S species. Oral Microbiol Immunol 5:137–142. [DOI] [PubMed] [Google Scholar]
- 38.Persson S, Claesson R, Carlsson J. 1989. The capacity of subgingival microbiotas to produce volatile sulfur compounds in human serum. Oral Microbiol Immunol 4:169–172. [DOI] [PubMed] [Google Scholar]
- 39.Persson S, Edlund MB, Claesson R, Carlsson. 1990. The formation of hydrogen sulfide and methyl mercaptan by oral bacteria. Oral Microbiol Immunol 5:195–201. [DOI] [PubMed] [Google Scholar]
- 40.Chu L, Dong Z, Xu X, Cochran DL, Ebersole JL. 2002. Role of glutathione metabolism of Treponema denticola in bacterial growth and virulence expression. Infect Immun 70:1113–1120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Carlsson J, Larsen JT, Edlund MB. 1993. Peptostreptococcus micros has a uniquely high capacity to form hydrogen sulfide from glutathione. Oral Microbiol Immunol 8:42–45. [DOI] [PubMed] [Google Scholar]
- 42.Carlsson J, Larsen JT, Edlund MB. 1993. Utilization of glutathione (L-gamma-glutamyl-Lcysteinylglycine) by Fusobacterium nucleatum subspecies nucleatum. Oral Microbiol Immunol 9:297–300. [DOI] [PubMed] [Google Scholar]
- 43.Chu L, Xu X, Dong Z, Cappelli D, Ebersole JL. 2003. Role for recombinant γ glutamyltransferase from Treponema denticola in glutathione metabolism. Infect Immun 71:335–342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Chu L, Lai Y, Xu X, Eddy S, Yang S, Song L, Kolodrubetz D. 2008. A 52-kDa leucyl aminopeptidase from Treponema denticola is a cysteinylglycinase that mediates the second step of glutathione metabolism. J Biol Chem 283:19351–19358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chu L, Holt SC. 1994. Purification and characterization of a 45 kDa hemolysin from Treponema denticola ATCC 35404. Microb Pathog 16:197–212. [DOI] [PubMed] [Google Scholar]
- 46.Chu L, Burgum A, Kolodrubetz D, Holt SC. 1995. 46 kDa hemolysin gene from Treponema denticola encodes a novel hemolysin homologous to aminotransferases. Infect Immun 63:4448–4455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chu L, Ebersole JL, Kurzban GP, Holt SC. 1997. Cystalysin, a 46-kilodalton cysteine desulfhydrase from Treponema denticola, with hemolytic and hemoxidative activities. Infect Immun 65:3231–3238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chu L, Ebersole JL, Kurzban GP, Holt SC. 1999. Cystalysin, a 46-kDa L-cysteine desulfhydrase from Treponema denticola: biochemical and biophysical characterization. Clin Infect Dis 28:442–450. [DOI] [PubMed] [Google Scholar]
- 49.Kurzban GP, Chu L, Ebersole JL, Holt SC. 1999. Sulfhemoglobin formation in human erythrocytes by cystalysin, an L-cysteine desulfhydrase from Treponema denticola. Oral Microbiol Immunol 14:153–164. [DOI] [PubMed] [Google Scholar]
- 50.Krupka HI, Huber R, Holt SC, Clausen T. 2000. Crystal structure of cystalysin from Treponema denticola: a pyridoxal 5’-phosphate-dependent protein acting as a haemolytic enzyme. EMBO J 19:3168–3178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Bertoldi M, Cellini B, Clausen T, Voltattorni CB. 2002. Spectroscopic and kinetic analyses reveal the pyridoxal 5’-phosphate binding mode and the catalytic features of Treponema denticola cystalysin. Biochemistry. 41:9153–9164. [DOI] [PubMed] [Google Scholar]
- 52.Blakemore RP, Canale-Parola E. 1976. Arginine catabolism by Treponema denticola. J Bacteriol 128:616–622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Xu X, Kolodrubetz D. 2002. Construction and analysis of hemin binding protein mutants in the oral pathogen Treponema denticola. Res Microbiol 153:569–577. [DOI] [PubMed] [Google Scholar]
- 54.Li H, Ruby J, Charon N, Kuramitsu H. 1996. Gene inactivation in the oral spirochete Treponema denticola: construction of an flgE mutant. J Bacteriol 178, 3664–3667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Makinen P-L, Makinen KK. 1997. gamma-Glutamyltransferase from the outer cell envelope of Treponema denticola ATCC 35405. Infect Immun 65:685–691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Morty RE, Morehead J. 2002. Cloning and characterization of a leucyl aminopeptidase from three Leishmania species. J Biol Chem 277:26057–26065. [DOI] [PubMed] [Google Scholar]
- 57.Bauer JD, Ackermann PG, Toro P. 1974. Clinical laboratory methods, p. 399–401. The C. V. Mosby Company, St. Louis, Mo. [Google Scholar]
- 58.Siegel LM. 1965. A direct microdetermination for sulfide. Anal Biochem 11:126–132. [DOI] [PubMed] [Google Scholar]
- 59.Leahy T, Smith R. 1960. Note on methemoglobin determination. Clin Chem 6:148–152. [PubMed] [Google Scholar]
- 60.Chu L, Kennell W, Holt SC. 1994. Characterization of hemolysis and hemoxidation activities by Treponema denticola. Microb Pathog 16:183–195. [DOI] [PubMed] [Google Scholar]
- 61.Oates TW, Mumford JH, Carnes DL, Cochran DL. 2001. Characterization of proliferation and cellular wound fill in periodontal cells using an in vitro wound model. J Periodontol 72:324–330. [DOI] [PubMed] [Google Scholar]
- 62.Stanley CM, Wang Y, Pal S, Klebe RJ, Harkless LB, Xu X, Chen Z, Steffensen B. 2008. Fibronectin fragmentation is a feature of periodontal disease sites and diabetic foot and leg wounds and modifies cell behavior. J Periodontol 79:861–875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kesavalu L, Holt SC, Ebersole JL. 1999. Environmental modulation of oral treponeme virulence in a murine model. Infect Immun 67:2783–2789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Bump EA, Reed DJ. 1977. A unique property of fetal bovine serum: High levels of proteinglutathione mixed disulfides. In Vitro 13:115–118. [DOI] [PubMed] [Google Scholar]
- 65.Livesey JC, Reed DJ. 1984. Measurement of glutathione-protein mixed disulfides. Int J Radiation Oncology Biol Phys 10:1507–1510. [DOI] [PubMed] [Google Scholar]
- 66.Hoffeld JT, Oppenheim JJ. 1980. The capacity of fetal calf serum to support a primary antibody response in vitro is determined, in part, by its reduced glutathione content. Cellular Immunol 53:325–332. [DOI] [PubMed] [Google Scholar]
- 67.Aquilano K, Baldelli S, Ciriolo MR. 2014. Glutathione: new roles in redox signaling for an old antioxidant. Front. Pharmacology 5:1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bannai S, Tsukeda H. 1979. The export of glutathione from human diploid cells in culture. J Biol Chem 254:3444–3450. [PubMed] [Google Scholar]
- 69.Chapple ILC, Matthews JB. 2007. The role of reactive oxygen and antioxidant species in periodontal tissue destruction. Periodontology 2000 43:160–232. [DOI] [PubMed] [Google Scholar]
- 70.Lee W, Pankoski L, Zekavat A, Shenker BJ. 2004. Treponema denticola immunoinhibitory protein induces irreversible G1 arrest in activated human lymphocytes. Oral Microbiol Immunol 19:144–149. [DOI] [PubMed] [Google Scholar]
- 71.Bernardini C, Gaibani P, Zannoni A, Vocale C, Bacci ML, Piana G, Forni M, Sambri V. 2010. Treponema denticola alters cell vitality and induces HO-1 and Hsp70 expression in porcine aortic endothelial cells. Cell Stress Chaperones 15:509–516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Visser MB, Ellen RP. 2011. New insights into the emerging role of oral spirochaetes in periodontal disease. Clin Microbiol Infect 17:502–512. [DOI] [PubMed] [Google Scholar]
- 73.Ishihara K, Kuramitsu HK, Miura T, Okuda K. 1998. Dentilisin activity affects the organization of the outer sheath of Treponema denticola. J Bacteriol 180:3837–3844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ishihara K, Wawrzonek K, Shaw LN, Inagaki S, Miyamoto M, Potempa J. 2010. Dentipain, a Streptococcus-pyogenes-IdeS-protease homologue, is a novel virulence factor of Treponema denticola. Biol Chem 391:1047–1055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Bian J, Liu X, Cheng Y, Li C. 2013. Inactivation of cyclic di-GMP binding protein TDE0214 affects the motility, biofilm formation, and virulence of Treponema denticola. J Bacteriol 195:3897–3905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Kurniyati K, Zhang W, Zhang K, Li C. 2013. A surface-exposed neuraminidase affects complement resistance and virulence of the oral spirochaete Treponema denticola. Molec Microbiol 89:842–856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Miller DP, Oliver LD Jr, Tegels BK, Reed LA, O’Bier NS, Kurniyati K, Faust LA, Lawson CK, Allard AM, Caimano MJ, Marconi RT. 2016. The Treponema denticola FhbB protein is a dominant early antigen that elicits FhbB variant-specific antibodies that block factor H binding and cleavage by dentilisin. Infect Immun 84:2051–2058. [DOI] [PMC free article] [PubMed] [Google Scholar]


