Abstract
General self-efficacy (GSE), the expectation that one is able to perform a behavior successfully, may differentiate those who are able to successfully utilize self-care symptom management strategies (SCSMS). This subanalysis (n=569) of an international 12 site longitudinal randomized controlled trial (RCT) (n=775), investigated GSE as an important factor determining symptom burden, SCSMS, engagement with the provider, and medication adherence over time, and identified differences in those with high and low GSE ratings concerning these variables. Parametric and nonparametric repeated-measures tests were employed to assess GSE and the perceived effectiveness of SCSMS for anxiety, depression, diarrhea, fatigue, nausea, and neuropathy. Symptom burden, engagement with the provider, and antiretroviral adherence were analyzed with regard to GSE. Our data indicated that there were differences in the perceived symptom burden over time of HIV infected individuals by GSE. Those individuals with higher GSE had fewer symptoms and these symptoms were perceived to be less intense than those experienced by the low GSE group. There were few meaningful differences in the SCSMS used by those with high versus low GSE other than the use of illicit substances in the low GSE group. The low GSE group was also significantly (p=< 0.001) less engaged with their healthcare providers. Given the difference in substance use by perceived GSE, and the importance of engagement with the healthcare provider, more attention to the resolution of the concerns of those with low GSE by healthcare providers is warranted.
Introduction
General self-efficacy (GSE), the expectation that “one can successfully perform a behavior,” has been of interest to investigators for decades.1 Bandura noted that “the freedom to exert some control over one's life” is an aspect of agent causality (p. 7). He stated that “perceived self-efficacy is not a measure of the skills one has but a belief about what one can do under different sets of conditions with whatever skills one possesses” (p. 37).2 Sherer and Adams observed that self-efficacy is influenced by past experience, suggesting that the level of GSE reflects success or lack thereof in dealing with previous life challenges, and is attained in the process of socialization from child to adult.3 The individual carries this reservoir of experience forward to the challenges posed by new situations.4
Bandura and others noted the importance of assessing self-efficacy for discrete areas as a performance component is involved. Although instruments have been developed to measure such performance areas as coping self-efficacy,5,6 HIV treatment adherence self-efficacy,7 and HIV-self-efficacy,8 there are other performance areas where instruments are not available. In those instances, and to obtain a more global perspective, a GSE instrument may be a suitable approach to examine the relationships of interest, in this case GSE and the perceived effectiveness of self-care symptom management strategies (SCSMS) and symptom burden. In this study, we explored the relationship between GSE and the SCSMS selected by people living with HIV infection.
Management of symptoms and side effects
Both the symptoms related to HIV/AIDS and the side effects of antiretroviral (ARV) therapy, whether physical or psychological, affect not only quality of life but the willingness and ability of HIV-infected individuals to maintain the level of adherence required for suppression of HIV.9 Whereas some infected individuals will contact their healthcare provider (HCP) about various symptoms, others will seek to remediate the conditions through a process of self-care management using the advice of family members, friends, and other HIV-infected individuals. Those individuals who have reported their symptoms to their provider may also engage in self-care management, particularly if the symptoms persist.
A series of studies have investigated the self-care strategies engaged in by HIV-infected individuals for anxiety, depression, diarrhea, fatigue, nausea, and neuropathy.10–12 These strategies subsequently were organized into a self-care symptom management manual and compared with a manual containing information on nutrition based on the World Health Organization HIV/AIDS Nutrition Guide.13 Although the self-care symptom management manual was found to have a higher helpfulness rating than the nutrition manual, there is a paucity of information regarding whether attributes of the individual affect self-care symptom management, including whether ARV adherence and the relationship with the HCP differs for those with higher general self-efficacy.
Relationship with the HCP has been shown to be associated with better treatment adherence.14,15 Bakken and colleagues found that the characteristic of the patient/client associated with less engagement with the care provider was past or present injection drug use.16 Age, ethnicity, and gender, however, were not found to have statistically significant relationships with engagement with the provider. Bakken and colleagues did not examine the psychological attributes of the patient in this study. Such characteristics may be key to understanding the relationship with the provider as well as with self-care symptom management, symptom burden, and ARV adherence.
The importance of an “informed, activated patient” and a ‘”prepared, proactive practice team” are emphasized by Seaton in his discussion of the chronic care model.17 He observes that HIV/AIDS is considered a chronic disease but is nonetheless distinct. The factors that make HIV/AIDS unique include the complexity of ARV adherence, the lack of immediate feedback as to health status as is available for diabetes, the discrimination accompanying an HIV diagnosis, and the stigma of disease related to perceived culpability for disease acquisition and its' communicability. To produce an informed, activated patient, Seaton advocated self-management education, which he contrasted with traditional health and illness education. Where self-management education emphasized problem solving skills, health education has as its focus “disease specific information and technical skills” (p. 4).
The assumption that provision of health education in and of itself is sufficient to produce the desired effect assumes that the rational person will engage in the necessary behavior based on the education provided. Such an assumption fails to ascertain what the patient has heard in the communication and what obstacles might impede achieving the necessary behavior. Traditional health education also neglects to identify similar behaviors with which the patient was successful in achieving the desired outcome. Furthermore, such an approach does not assess the patient's general self-efficacy in achieving desired outcomes.
Research with other disease populations as well as those with HIV/AIDS has demonstrated that higher self-efficacy affects health status,18 levels of depression and active coping,19 patient health behaviors,20 self-management by kidney transplant recipients,21 HIV disease progression,22 and medication adherence.23
We explored the relationship of GSE with the use of SCSMS, symptom burden, ARV adherence, and engagement with the provider by a subanalysis of a data set developed as an international, longitudinal randomized controlled trial (RCT) that examined the usefulness of a self-care symptom manual.13
The conceptual model depicted in Fig. 1 portrays the relationships of interest to this study, which are specified by the research question, “Is perceived general self-efficacy related to self-care management strategies, perceived symptom burden, antiretroviral medication adherence, and engagement with the provider over time for persons living with HIV infection?”
FIG. 1.
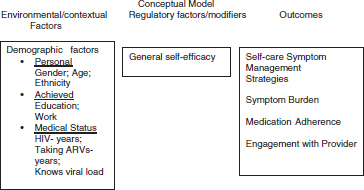
Conceptual model depicting relationships of environmental/contextual factors, regulatory factors, and outcomes.
Methods
Design
A descriptive, repeated measures design was used for this secondary analysis of data from an existing study.13
Sample
This study utilized data from the experimental arm of an RCT in which persons living with HIV infection received the HIV/AIDS Self Care Symptom Management Manual. There were a total of 775 participants in the experimental arm of the parent study and 569 completed the general self-efficacy scale, and therefore became the sample for this analysis.
Protection of human subjects
The coordinating site for this study was the University of California–San Francisco, whose investigators obtained overall approval for this research as well as San Francisco site approval. In order to participate in the study, each site obtained approval from their Protection of Human Subjects Committee. As this was a clinical trial, a Data Safety and Monitoring Board was established at the University of California–San Francisco to monitor the performance and safety of the trial. Certificates of confidentiality were also obtained as required by each site's Human Subjects Review Committee. This clinical trial was registered with the National Institutes of Health (NIH) at (ClinicalTrials.gov).
Instruments
Seven instruments were used for this analysis.
1. Demographic Questionnaire
A 13 item self-report demographic questionnaire was used to collect demographic and illness background information. This questionnaire has been used in prior research by members of the International HIV/AIDS Nursing Research Network.
2. GSE Scale
is a 10 item scale designed to assess optimistic self-beliefs about coping with a variety of life's demands.24 Originally developed in German, the GSE Scale is available in 29 languages and has been widely used with participants from numerous countries. Responses range from 1 (not at all true) to 4 (exactly true). Cronbach's α ranged from 0.76 to 0.90.
3. Engagement with HCP
is a 13 item scale designed to assess how individuals rate their interactions with their HCP.16 A four point scale (1=always true and 4=never true) measured each item. A low score indicated greater provider engagement. Cronbach's α reliability estimate was 0.96.
4. HIV Self-Care Symptom Management Survey
(HIV-SCSMS) is composed of two parts.25 The first part assesses the frequency, intensity, and impact of depression, anxiety, nausea, neuropathy, diarrhea, and fatigue. The second part of the instrument examines the use of self-care behaviors, querying whether the behavior is used (yes/no), frequency of use (daily, weekly, monthly) and effectiveness of the strategy (1=not at all effective to 10=very effective).
5. Revised AIDS Clinical Trials Group (ACTG) Reasons for Nonadherence to Medications
(ACTGrev) is a self-report measure of reasons for missing medications that was revised through a randomized clinical trial on adherence.26,27 The revised scale consists of two factors with a total of nine items (pill-taking problems [5 items] and forgetfulness [4 items]) using a four point Likert-type scale ranging from 0=never to 3=often. A higher score indicates less adherence to medication regimens. Cronbach's α for the two subscales and the total scale ranged from 0.8 to 0.9.
6. The Revised Sign and Symptom Checklist for HIV
(SSC-HIV). This 72 item checklist asks respondents to rate frequently experienced HIV symptoms on a Likert scale where 1=mild, 2=moderate, and 3=severe.28 Part 1 consists of 45 items clustering into 11 factors with a total score and reliability estimates ranging from 0.76 to 0.91. Part 2 consists of 19 HIV-related symptoms that do not cluster into factor scores but may be of clinical interest. Part 3 is composed of 8 gynecological items. Cronbach's α reliability scores ranged from 0.85 to 0.90 for each of the factors and 0.92 for the items in Parts 1 and 2.28
7. HIV/AIDS Targeted Quality of Life Instrument
(HAT-QoL) is a 34 item HIV-specific quality of life measurement assessing nine dimensions including overall function, life satisfaction, health worries, financial worries, medication worries, HIV mastery, disclosure, provider trust, and sexual function.29–31 All dimensions are scored so that the final dimension score is transformed to a linear 0–100 scale where 0 is the worst possible score and 100, the best possible score.
Data analysis
The analysis for this study used parametric and nonparametric repeated-measures tests for GSE and the perceived effectiveness by the participant of SCSMS for the selected symptoms of anxiety, depression, diarrhea, fatigue, nausea, and neuropathy. We grouped the self-care strategies for all six previously mentioned symptoms into categories of strategies for each symptom. Symptom burden was assessed by examining symptom frequencies with the SSC-HIV-rev. Demographic variables were also examined. Engagement with the provider and the health and medication worry dimensions from the HAT-QoL were analyzed with regard to self-efficacy. The GSE scale was split above and below the median (median=30) to create high (HSE) and low (LSE) self-efficacy groups. For both self efficacy groups, the symptom intensity mixed model growth estimations were computed by testing the unconditional means model to each individual's initial symptom intensity status at baseline (within person) and its associated variation from the mean symptom intensity value.
Results
The mean age for the sample participants (n=569) who completed the self-efficacy scale was 42.8 years (SD=9.6) and the gender composition was 38% (n=218) female, 59.7% (n=340) male, and 2% (n=11) transgender. The participants were 39.9.1% (n=227) African/African American, 17.9% (n=102) Caucasian, 34.3% (n=195) Hispanic, 1.8% (n=10) Asian/ Pacifica Islander, and 5.3% (n=30) self-described as “Other.” Most participants, 39% (n=222) had a high school education, 30.4% (n=173) had a grade school education, and 30.6 % (n=174) had college or post-high school education. Of this sample, 24.5% (n=139) worked for pay. The HIV illness characteristics indicate that the mean number of years living with HIV were 14.8 years (SD=6.4), and that 42% (n=240) had an AIDS diagnosis, 66.8% (n=370) reported a comorbid illness, and 72.9% (n=412) were presently taking ARV medications (data not shown).
To explore the impact of GSE on self-care symptom management, participants were divided into high and low GSE groups with 44.8% (n=252) in the high GSE group and 55.2% (n=317) of the participants in the low GSE group. An examination of the differences by GSE category of the sociodemographic characteristics of the sample indicates that the sample is significantly different by gender (p=0.02), ethnicity (p=0.03), education (p=0.007), whether respondents work for pay (p=0.038), and whether they know their viral load (p=0.006). There are no significant differences by age, years that individuals were HIV positive, whether they are taking ARV medications presently, or the numbers of years taking antiretroviral ARV medications (see Table 1).
Table 1.
Demographic Characteristics (n=569) of Those Completing the General Self-Efficacy Instrument
| High self efficacy group (HSE) n=253 | Low self efficacy group (LSE) n=316 | χ2, or t | p Value | |
|---|---|---|---|---|
| Gender | Male to female only | 0.02 | ||
| Male | 176 (52%) | 164 (48%) | ||
| Female | 84 (38%) | 134 (62%) | χ2 (1,558)=5.07 | |
| Transgender | 5 (2%) | 6 (2%) | ||
| Age mean (SD) | 44.0 (9.8) | 42.7 (8.8) | t (548)=1.61 | 0.10 |
| Ethnicity | Black/Non-black only | 0.03 | ||
| Black | 89 (35%) | 138 (44%) | ||
| Non-black | χ2 (1,569)=4.80 | |||
| Hispanic | 97 (38%) | 98(31%) | ||
| White | 54 (17%) | 48 (19%) | ||
| Other (Asian, Native American, others) | 24 (8%) | 21 (8%) | ||
| Highest education level | χ2 (4,569)=13.9 | 0.007 | ||
| Grade school | 66 (26%) | 107 (34%) | ||
| High school | 92 (36%) | 130 (41%) | ||
| College (Associate) | 57 (22%) | 54 (17%) | ||
| College (Bachelors) | 26 (10%) | 18 (6%) | ||
| Masters/higher | 13 (9%) | 6 (2%) | ||
| Work for pay | χ2 (1,568)=14.32 | 0.038 | ||
| Yes | 73 (28%) | 66 (21%) | ||
| No | 182 (71%) | 247 (79%) | ||
| Years HIV+mean (SD) | 10.1 (6.4) | 9.5 (6.4) | t (548)=1.22 | 0.22 |
| Taking ARVs now | χ2 (1,565)=2.1 | 0.15 | ||
| Yes | 192 (76%) | 220 (70%) | ||
| No | 61 (24%) | 92 (30%) | ||
| Years taking ARVs mean (SD) | 7.1 (5.1) | 6.7 (5.1) | t (212)=0.598 | 0.55 |
| Does not know HIV VL | 89 (36%) | 129 (42%) | χ2 (2,559)=10.1 | 0.006 |
| Knows VL undetectable | 109 (44%) | 96 (31%) | ||
| Knows detectable VL value | 51 (20%) | 85 (27%) |
ARV, antiretroviral medication; VL, viral load.
Strategies for self-care symptom management for the six symptoms (anxiety, depression, diarrhea, fatigue, nausea, neuropathy) differed depending upon the symptom although there were a number of commonalities. The categories of activities/thoughts, medications, and substance use were general strategies used for all of the symptoms. Other self-care strategies, those for diarrhea for example, also included foods to eat; and for another symptom the category foods to avoid, a category that was specific to that symptom, was included. Table 2 depicts the data for activities/thoughts, the only general strategy category that was significant for all symptoms.
Table 2.
Specific Activities and Thoughts Mentioned for Each Symptom
| Activities/thoughts | Anxiety | Depression | Diarrhea | Fatigue | Nausea | Neuropathy |
|---|---|---|---|---|---|---|
| Talk with family/friends | X | X | X | |||
| Talk with HCP | X | X | ||||
| Talk with others with HIV | X | X | ||||
| Attend support groups | X | X | ||||
| Denial/Try not to think | X | X | ||||
| Cry | X | |||||
| Stay alone | X | |||||
| Talk myself through it | X | |||||
| Watch TV | X | |||||
| Play cards | X | |||||
| Read | X | |||||
| Cook | X | |||||
| Avoid negative or annoying thing | X | |||||
| Go out to work | X | |||||
| Treat myself with special foods | X | |||||
| Keep busy | X | |||||
| Draw | X | |||||
| Listen to music | X | |||||
| Don't dwell on it | X | |||||
| Get enough sleep at night | X | |||||
| Take frequent breaks | X | |||||
| Adjust social activities | X | |||||
| Not get stressed out | X | |||||
| Nap during the day | X | |||||
| Lie down and rest | X | |||||
| Take a deep breath | X | |||||
| Rub the stomach | X | |||||
| Get fresh air | X | |||||
| Take a hot bath or shower | X | X | ||||
| Stay off your feet | X |
HCP, healthcare provider.
A comparison of these SCSMS by GSE group indicates that there were significant differences in the use of prescribed or over-the-counter medications and substance use. When the effectiveness of these strategies was considered, only substance use was significantly different (p=0.04) in the two GSE groups. Substance use was higher in the low GSE group as was the use of the other strategies in the low GSE group. The high GSE group was more likely to be engaged with their HCP (p=0.001 and adherent to their medications (p=0.001) (see Table 3).
Table 3.
Self-Care Symptom Management Strategy by Self-Efficacy
| Bivariate analyses–self care symptom management strategy type used | ||||
|---|---|---|---|---|
| Self care symptom management category n=569 | High self efficacy group (HSE) | Low self efficacy group (LSE) | Value | p |
| Thoughts and activities frequency – (mean rank) | 272.86 | 299.37 | ZU=1.91 | p=0.056 (CI=0.050–0.062) |
| Taking prescribed and OTC medications therapies frequency – (mean rank) | 271.92 | 300.13 | ZU=2.17 | p=0.030 |
| Exercise frequency – (mean rank) | 281.10 | 292.69 | ZU=0.870 | p=0.38 |
| Complementary therapies frequency – (mean rank) | 282.46 | 291.59 | ZU=0.687 | p=0.49 |
| Substance use frequency–(mean rank) | 271.78 | 300.25 | ZU=0.025 | p=0.025 |
| Thoughts and activities effectiveness – (mean rank) | 276.01 | 296.82 | ZU=1.51 | p=0.13 |
| Taking prescribed and OTC medications therapies effectiveness – (mean rank) | 278.37 | 294.91 | ZU=1.25 | p=0.21 |
| Exercise effectiveness–(mean rank) | 285.92 | 288.78 | ZU=0.22 | p=0.83 |
| Complementary therapies effectiveness–(mean rank) | 286.66 | 288.06 | ZU=0.20 | p=0.82 |
| Substance use effectiveness-(mean rank) | 273.67 | 298.71 | ZU=1.98 | p=0.04 |
| HIV assessment tool (HAT) | ||||
| HIV control over illness (taking/not taking ART) | 75.34 (SD=26.0) | 56.70 (SD=29.9) | t=4.20 | p≤0.001 |
| Engagement with health care provider (HCP) | ||||
| HCP total score (lower score=improved engagement) | 14 | 17 | t= | p≤0.001 |
| Symptom frequency (SSC-HIV rev) total score (range 0–64) | ||||
| Baseline | 19.40 (SD=17.9) | 23.0 (SD=19.5) | F=5.90 | p=0.015 |
| Assessment 1 | 17.37 (SD=15.8) | 20.62 (SD=19.8) | ||
| Assessment 2 | 14.02 (SD=13.9) | 17.97 (SD=18.5) | ||
ZU, Mann–Whitney nonparametric statistical test; ART, antiretroviral therapy.
Analysis of change over time
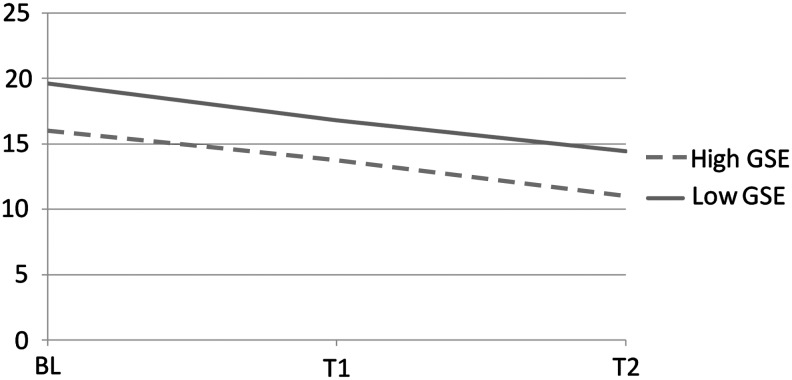
When symptom burden, measured by symptom frequency is considered, the high GSE group had a lower symptom frequency at baseline and maintained an advantage in the additional two assessments, although the symptom frequency declined for both groups over time. These differences are significant at the (p=0.015) level (see Fig. 2).
FIG. 2.
Symptom intensity by self-efficacy.
For both GSE groups, the symptom intensity mixed model growth estimations were computed by testing the unconditional means model to each individual's initial symptom intensity status at baseline (within person) and its associated variation from the mean symptom intensity value (see Table 4, Model A). The model has no growth parameter. In the initial model (Model A), there was a significant decrease in symptoms in this subsample over the 3 month period. This model showed the symptom intensity intercept value to be 0.92 antilog (-0.079) with significant variation in an individual's initial status of symptom intensity scores at baseline (0.005) and in within-person change (0.003). This was calculated to be 0.63 (0.005/0.005+0.003), indicating that almost two-thirds of the total variation in symptom intensity was attributable to differences among the participants. This value also indicated that there was large residual autocorrelation as expected in repeated measures analyses.
Table 4.
Multilevel Analysis of Change Over Time–Dependent Variable (DV)=Log of Total Symptom
| Unconditional growth model | Growth model with GSE 2 groups | Growth model with all level 2 predictors and covariates | |||||
|---|---|---|---|---|---|---|---|
| Model A | Model B | Model C | Interpretation | ||||
| Estimate | S E | Estimate | S E | Estimate | S E | Model C | |
| Intercept | −0.079 | 0.009 | −0.090a | 0.011 | 1.206a | 0.039 | There is significant variation about the intercept in symptom intensity |
| Time | 1.377 | 0.020 | 1.399a | 0.027 | −0.067a | 0.012 | There is a significant decrease in symptom intensity in both groups over the 3-month period |
| Black | 0.095b | 0.042 | Black individuals had a significantly higher symptom intensity rating at baseline compared with other ethnicities | ||||
| Female | 0.021 | 0.042 | Females had a significantly higher symptom intensity rating at baseline compared with male and transgender individuals | ||||
| [gse2grp=0.00] | 0.137a | 0.034 | 0.087b | 0.042 | Individuals in the low self efficacy group had a significantly higher symptom intensity at baseline compared with the high GSE group | ||
| Activities_eff | 0.008b | 0.004 | Individuals who rated activities effectiveness higher had increased symptom intensity scores at baseline | ||||
| Activities_used | 0.008 | 0.004 | Individuals who used more symptom management activities had increased symptom intensity scores at baseline | ||||
| [gse2grp=1.00]a activities_useda black | −0.010b | 0.004 | Black individuals in the high selfefficacy group used significantly fewer activities compared with non-black individuals at baseline | ||||
| [gse2grp=1.00]a timea activities_useda black | 0.001 | 0.003 | Black individuals in the high self- efficacy group remained constant over 3 months in using fewer activities compared with non-black individuals, but not significantly fewer activities | ||||
| [gse2grp=0.00]a activities_used | 0.010 | 0.006 | The low self efficacy group did not use more strategies at baseline compared with the high self efficacy group | ||||
| [gse2grp=0.00]a activities_eff | −0.008 | 0.006 | The low self efficacy group did not rate strategy effectiveness differently at baseline compared with the high self efficacy group | ||||
| Model fit statistics | |||||||
| −2 log likelihood | 1996.0 | 1409.9 | 1219.2 | The model fit improved with the addition of explanatory level 2 variables | |||
| Akaike information criterion (AIC) | 1986.0 | 1397.9 | 1255.2 | ||||
| Schwarz's Bayesian criterion (BIC) | 2023.2 | 1441.0 | 1347.8 | ||||
| Residuals analysis | |||||||
| Residual in model | 0.003 | 0.013 | 0.090 | 0.005 | 0.088 | 0.005 | |
| Residual variance - intercept | 0.005 | 0.0009 | 0.132 | 0.012 | 0.104 | 0.010 | 21% of the variance has been explained from model B with the addition of the additional level 2 variables |
| Residual variance - time | 0.195 | 0.087 | 0.004 | 0.003 | 0.002 | 0.003 | 50% of the variance in time is explained by the addition of level 2 variables |
p≤0.001, bp≤0.05.
Next, the addition of the GSE group variable to obtain the individual growth trajectories from baseline as well as the between-person variation in trajectories was estimated. In this model, the intercept symptom intensity value was 0.81 antilog (0.09) and again, over time on average, whereas both groups decreased in the symptom intensity scores, the low GSE group had a significantly higher increase in the intercept for symptom intensity 1.11 antilog (-0.09+0.137) that continued over the 3-month period. The variances showed significant individual variation about the individual trajectories (0.09) as well as significant between-person variability in initial status (0.132) and the rate of change (0.004). The addition of the self-efficacy group variable increased the variation about the initial intercept and explained little of the variation. However, the smaller goodness of fit statistics relative to Model A showed that the model was a better fit. The estimates of variation from these two unconditional models were used for the subsequent model comparison to assess any further reduction in variance, and improved model fit by the addition of level 2 variables (self-efficacy and other descriptive variables) in describing the sample and to reduce variation. Model C shows the final model that tested the effects of adding additional independent variables for the use of self-management activities and demographic data, and the modeled interactions of these variables, to assess the effects of symptom intensity change over time. Whereas 21% of the variance in Model B was explained by the addition of level 2 variables, 50% of the variance in GSE over time was explained in Model C (see Table 4). Black individuals had a significantly higher intensity rating at baseline compared with other ethnicities and those with high GSE scores, and used significantly fewer activities for self-care management than those used by non-black individuals at baseline.
Discussion
As was noted in the results, demographic characteristics make a difference with regard to general self-efficacy as evidenced by our finding that females, blacks, those who do not work, individuals with a high school education or less, and those who don't know their viral load were all more likely to be in the low GSE group. Demographic characteristics were also significant with regard to symptom intensity, with both blacks and females having higher symptom intensity at baseline. This raises the question about whether GSE has an impact on access to care or the attention of the HCP, or whether lack of such access and attention result in lower GSE. It is plausible that there is a recursive effect, with self-efficacy affecting the interaction with the HCP and the interaction with the HCP affecting self-efficacy. Our results indicate a significant (p=< 0.001) difference in engagement with the HCP by GSE with those who had lower GSE experiencing less engagement with their providers. Engagement with the provider has been found to be important both to ARV adherence and retention in care.32,33 Self-efficacy may provide yet another indicator as to those who will require greater attention by providers.
GSE also made a difference with regard to symptom burden. The high GSE group had lower symptom intensity than did the low GSE group at baseline, and over the three time periods. Again, engagement with the provider may be part of the explanation for this finding. Nonetheless, there is a decline in symptom frequency over the three time periods for both the high and low GSE groups. Whether this is a result of the attention obtained by participation in the study could not be determined with the data at hand.
Symptom intensity did have an impact on the use of SCSMS. Individuals who used more strategies had increased symptom intensity at baseline, suggesting that intensity led these individuals to experiment with a number of approaches to reduce their discomfort. Further, individuals who rated the effectiveness of these strategies higher also had increased symptom intensity at baseline. It should be noted that individuals in the low GSE group had higher symptom intensity at baseline compared with the high GSE group.
Our data indicated that the high and low GSE groups differed with regard to the number and type of SCSMS utilized. However, black individuals in the high GSE group used significantly fewer activities compared with non-blacks, suggesting that race may be less important than other factors for individuals with high GSE. Whether this indicates the ability to be more selective and efficacious in determining which strategies to use requires further study.
Another difference between high and low GSE groups in the type of strategies used included the categories of taking prescribed and over-the-counter medications and substance use. The effectiveness of substance use was the only category of strategies that differed between the two GSE groups. This raises questions about the perceived inadequacy of the treatment of symptoms. Given that substance use has been found to have a negative relationship with adherence, more attention to the treatment of symptoms is required. Our data indicated a significant (p=< 0.001) difference between the high and low GSE groups regarding adherence to ARV medications, underscoring the importance of addressing substance use and ARV therapy in low GSE individuals. GSE was considered a modifiable factor by Arnsten and colleagues.34 This suggests that interventions to enhance GSE may be developed. Further, given our findings that the high GSE group had significantly better engagement with the provider than did the low GSE group, the importance of the provider cannot be overemphasized. The provider is only part of the equation, however, and attention to the empowerment of the patient, a far more complex issue, requires further attention as well.35
Our data indicate that there are differences in the perceived symptom burden over time of HIV-infected individuals by perceived GSE. Those individuals with higher GSE had fewer symptoms, and these symptoms were perceived to be less intense than those experienced by the low GSE group.
Perceived GSE was related to the type, frequency, and effectiveness of the SCSMS used by HIV-infected individuals. And whereas there were no differences in the high and low GSE groups in the frequency of use of the thoughts/activities, exercise, and complementary therapies, there were significant differences in the use of prescribed and over-the-counter medications and substance use. However, only substance use was significantly different in effectiveness in the two GSE groups. Ultimately, the gold standard in the self-care management of symptoms is the effectiveness of the strategies selected. Given the difference in substance use by perceived GSE and the importance of engagement with the HCP, more attention to the resolution of the concerns of those with low GSE by HCP is warranted.
Our results showed that individuals in the low GSE group were more likely to use substances as a SCSMS. Our data did not indicate, however, whether these individuals as compared with those in the high GSE group were more likely to use substances recreationally. Such a distinction would be helpful for making recommendations. Our data cannot inform us as to whether recreational use led to the serendipitous finding of symptom relief.
This study would have benefited by having included instruments measuring performance self-efficacy so as to ascertain the degree of association with GSE. Such instruments might include adherence self-efficacy and condom self-efficacy. GSE was divided into high and low groups using the median. Other approaches to determining group membership, including using a high, medium, and low group categorization, may enhance our understanding of GSE, particularly when combined with performance-related measures.
This study has demonstrated the significance of GSE in identifying individuals who are more likely to use substance use as a SCSMS. And although Bandura and others indicate that GSE must be specified with regard to a particular activity such as adherence self-efficacy, it may be that those with high GSE are more likely to have higher adherence self-efficacy. It is plausible that for those activities that don't require special skills or training, GSE may provide the foundation of confidence necessary to incorporate the behaviors required to achieve adherence. Further research to investigate the relationship of GSE with a range of specified self-efficacy behaviors will enhance our understanding of the role of self-efficacy in healthcare outcomes. Additional study on the use of substances for symptom management is also warranted. Would such use occur if symptoms were managed more effectively? Although the common-sense response might be in the affirmative, further research would refine our understanding of this question.
The finding that individuals in the low GSE group were more likely to utilize substances as a SCSMS indicates that symptoms are not being adequately addressed by HCP. Whether this is because individuals with low GSE do not present their concerns and complaints, or that these individuals are not queried about their concerns, the result is that they do not receive the care required. It is possible that those with low GSE have a greater range of issues that need to be addressed than those with higher GSE, and that therefore only the most significant, as perceived by the HCP, are treated in the limited time available. The constraints of time may ultimately prove to be penny wise and pound foolish.
Author Disclosure Statement
No competing financial interests exist.
References
- 1.Bandura A. Self-efficacy: Toward a unifying theory of behavioral change. Psychol Rev. 1977;84:191–215. doi: 10.1037//0033-295x.84.2.191. [DOI] [PubMed] [Google Scholar]
- 2.Bandura A. Self-efficacy: The Exercise of Control. New York, N.Y.: W.H. Freeman; 1997. [Google Scholar]
- 3.Sherer M. Adams CH. Construct validation of the self-efficacy scale. Psychol Rep. 1983;53:899–902. [Google Scholar]
- 4.Sherer M. Maddux JE. Mercandante B. Prentice–Dunn S, Jacobs B, Rogers RW. The self-efficacy scale: Construction and validation. Psychol Rep. 1982;51:663–671. [Google Scholar]
- 5.Chesney MA. Neilands TB. Chambers DB. Taylor JM. Folkman S. A validity and reliability study of the coping self-efficacy scale. Br J Health Psychol. 2006;11:421–437. doi: 10.1348/135910705X53155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kamau TM. Olson VG. Zipp GP. Clark MA. Coping self-efficacy as a predictor of adherence to antiretroviral therapy in men and women living with HIV in Kenya. AIDS Patient Care STDs. 2011;25:557–561. doi: 10.1089/apc.2011.0125. [DOI] [PubMed] [Google Scholar]
- 7.Johnson MO. Neilands TB. Dilworth SE. Morin SF. Remien RH. Chesney MA. The role of self-efficacy in HIV treatment adherence: validation of the HIV treatment adherence self-efficacy scale (HIV-ASES) J Behav Med. 2007;30:359–370. doi: 10.1007/s10865-007-9118-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shively M. Smith TL. Bormann J. Gifford AL. Evaluating self-efficacy for HIV disease management skills. AIDS Behav. 2002;6:371–379. [Google Scholar]
- 9.Mannheimer SB. Wold N. Gardner EM, et al. Mild-to-moderate symptoms during the first year of antiretroviral therapy worsen quality of life in HIV-infected individuals. Clin Infect Dis. 2008;46:941. doi: 10.1086/528859. [DOI] [PubMed] [Google Scholar]
- 10.Corless IB. Bunch EH. Kemppainen JK, et al. Self-care for fatigue in patients with HIV. Oncol Nurs Forum. 2002;29:60–69. doi: 10.1188/02.ONF.E60-E69. [DOI] [PubMed] [Google Scholar]
- 11.Kemppainen J. Eller LS. Bunch E, et al. Strategies for self-management of HIV-related anxiety. AIDS Patient Care STDs. 2006;18:597–607. doi: 10.1080/09540120500275726. [DOI] [PubMed] [Google Scholar]
- 12.Nicholas PK. Kemppainen JK. Canaval GE, et al. Symptom management and self-care for peripheral neuropathy in HIV/AIDS. AIDS Patient Care STDs. 2007;19:179–189. doi: 10.1080/09540120600971083. [DOI] [PubMed] [Google Scholar]
- 13.Wantland DJ. Holzemer WL. Moezzi S, et al. A randomized controlled trial testing the efficacy of an HIV/AIDS symptom management manual. J Pain Symptom Manage. 2008;36:235–246. doi: 10.1016/j.jpainsymman.2007.10.011. [DOI] [PubMed] [Google Scholar]
- 14.Demmer C. Relationship with health care provider and adherence to HIV medications. Psychol Rep. 2003;93:494–496. doi: 10.2466/pr0.2003.93.2.494. [DOI] [PubMed] [Google Scholar]
- 15.Schneider J. Kaplan SH. Greenfield S. Li W. Wilson IB. Better physician—patient relationships are associated with higher reported adherence to antiretroviral therapy in patients with HIV infection. J Gen Intern Med. 2004;19:1096–1103. doi: 10.1111/j.1525-1497.2004.30418.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bakken S. Holzemer WL. Brown MA, et al. Relationships between perception of engagement with health care provider and demographic characteristics, health status, and adherence to therapeutic regimen in persons with HIV/AIDS. AIDS Patient Care STDs. 2000;14:189–197. doi: 10.1089/108729100317795. [DOI] [PubMed] [Google Scholar]
- 17.Seaton R. Providing HIV/AIDS care in a changing environment: Self-management and the chronic care model. HRSA Careaction. Jan, 2006. pp. 1–8. [PubMed]
- 18.Cross MJ. March LM. Lapsley HM. Byrne E. Brooks PM. Patient self-efficacy and health locus of control: Relationships with health status and arthritis-related expenditure. Rheumatology. 2006;45:92–96. doi: 10.1093/rheumatology/kei114. [DOI] [PubMed] [Google Scholar]
- 19.Luszczynska A. Scholz U. Schwarzer R. The general self-efficacy scale: Multicultural validation studies. J Psychol. 2005;139:439–457. doi: 10.3200/JRLP.139.5.439-457. [DOI] [PubMed] [Google Scholar]
- 20.Jerant A. Kravitz RL. Azari R, et al. Training residents to employ self-efficacy-enhancing interviewing techniques: Randomized controlled trial of a standardized patient intervention. J Gen Intern Med. 2009;24:606–613. doi: 10.1007/s11606-009-0946-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Weng LC. Dai YT. Huang HL. Chiang YJ. Self-efficacy, self-care behaviours and quality of life of kidney transplant recipients. J Adv Nurs. 2010;66:828–838. doi: 10.1111/j.1365-2648.2009.05243.x. [DOI] [PubMed] [Google Scholar]
- 22.Ironson G. Hayward HS. Do positive psychosocial factors predict disease progression in HIV-1: A review of the evidence. Psychosom Med. 2008;70:546–554. doi: 10.1097/PSY.0b013e318177216c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Barclay TR. Hinkin CH. Castellon SA, et al. Age-associated predictors of medication adherence in HIV-positive adults: Health beliefs, self-efficacy, and neurocognitive status. Health Psychol. 2007;26:40–49. doi: 10.1037/0278-6133.26.1.40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Schwarzer RE. Self-efficacy: Thought Control of Action. Washington, DC: Hemisphere Publishing Corporation; 1992. [Google Scholar]
- 25.Holzemer WL. Hudson A. Kirksey KM. Jane Hamilton M. Bakken S. The revised sign and symptom check-list for HIV (SSC-HIVrev) J Assoc Nurses AIDS Care. 2001;12:60–70. doi: 10.1016/s1055-3290(06)60263-x. [DOI] [PubMed] [Google Scholar]
- 26.Chesney MA. Ickovics JR. Chambers DB, et al. Self-reported adherence to antiretroviral medications among participants in HIV clinical trials: the AACTG adherence instrument. AIDS Patient Care STDs. 2000;12:255–266. doi: 10.1080/09540120050042891. [DOI] [PubMed] [Google Scholar]
- 27.Holzemer WL. Bakken S. Portillo CJ, et al. Testing a nurse-tailored HIV medication adherence intervention. Nurs Res. 2006;55:189–197. doi: 10.1097/00006199-200605000-00005. [DOI] [PubMed] [Google Scholar]
- 28.Nicholas PK. Voss J. Wantland D, et al. Prevalence, self-care behaviors, and self-care activities for peripheral neuropathy symptoms of HIV/AIDS. Nurs Health Sci. 2010;12:119–126. doi: 10.1111/j.1442-2018.2009.00505.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Holmes WCMSCE. Shea JA. A new HIV/AIDS-targeted quality of life (HAT-QoL) instrument: development, reliability, and validity. Med Care. 1998;36:138–154. doi: 10.1097/00005650-199802000-00004. [DOI] [PubMed] [Google Scholar]
- 30.Holmes WC. Shea JA. Two approaches to measuring quality of life in the HIV/AIDS population: HAT-QoL and MOS-HIV. Qual Life Res. 1999;8:515–527. doi: 10.1023/a:1008931006866. [DOI] [PubMed] [Google Scholar]
- 31.Holzemer WL. Bakken Henry S. Portillo CJ. Miramontes H. The client adherence profiling-intervention tailoring (CAP-IT) intervention for enhancing adherence to HIV/AIDS medications: a pilot study. J Assoc Nurses AIDS Care. 2000;11:36–44. doi: 10.1016/s1055-3290(06)60420-2. [DOI] [PubMed] [Google Scholar]
- 32.Mallinson RK. Rajabiun S. Coleman S. The provider role in client engagement in HIV care. AIDS Patient Care STDs. 2007;21:77–84. doi: 10.1089/apc.2007.9984. [DOI] [PubMed] [Google Scholar]
- 33.Tobias C. Cunningham WE. Cunningham CO. Pounds MB. Making the connection: the importance of engagement and retention in HIV medical care. AIDS Patient Care STDs. 2007;21:3–8. doi: 10.1089/apc.2007.9992. [DOI] [PubMed] [Google Scholar]
- 34.Arnsten JH. Li X. Mizuno Y, et al. Factors associated with antiretroviral therapy adherence and medication errors among HIV-infected injection drug users. J Acquir Immune Defic Syndr. 2007;36(Supplement 2):S64–S71. doi: 10.1097/QAI.0b013e31815767d6. [DOI] [PubMed] [Google Scholar]
- 35.Johnson MO. The shifting landscape of health care: Toward a model of health care empowerment. Am J Public Health. 2011;101:265–270. doi: 10.2105/AJPH.2009.189829. [DOI] [PMC free article] [PubMed] [Google Scholar]