Abstract
Cow’s milk is consumed by most North American children, yet the relationships between the volume and fat content of cow’s milk consumed and childhood fracture risk are unclear. Our primary objectives in this study were to evaluate whether the volume or fat content of cow’s milk consumed at 1–3 years of age was associated with the risk of fracture between 3 and 10 years of age. This was a prospective cohort study of 2,466 children enrolled in Toronto, Ontario, Canada, between 2008 and 2016. The primary exposure was volume of cow’s milk consumed between the ages of 1 and 3 years, and the secondary exposure was average percentage of milk fat consumed by each child during the same period. The primary outcome was a parental report of child fracture at ages 3–10 years. In the primary and secondary adjusted analyses, no association between milk volume and fracture risk (adjusted relative risk = 1.04, 95% confidence interval: 0.87, 1.26) or between milk-fat content and fracture risk (adjusted relative risk = 1.05, 95% confidence interval: 0.84, 1.31) was observed. In this study, we did not identify a protective association of early childhood cow’s milk volume or fat consumption with fracture risk in later childhood. Future prospective research is needed to understand whether cow’s milk is beneficial for fracture prevention through the life course.
Keywords: childhood, cow’s milk, fracture, fracture risk, nutrition, prepubescence, prospective studies
Abbreviations
- BMI
body mass index
- CI
confidence interval
- SD
standard deviation
- TARGet Kids!
The Applied Research Group for Kids
One third of children fracture a bone by the end of childhood (1, 2). During the last 2 decades, the frequency of childhood fractures has increased by one third, causing significant hardship and pain (3, 4). Childhood fracture has been associated with an increased risk of adolescent and adulthood fractures, as well as osteoporosis later in life (1, 3, 5, 6).
Several risk factors for childhood fracture have been identified, including older age, male sex, high and low levels of physical activity, greater adiposity, and nutritional factors such as sugar-sweetened beverage consumption and milk avoidance (2, 5–12). Observational studies have identified an increased risk of fractures among children who do not consume cow’s milk (7, 13, 14). However, children who do not consume cow’s milk may do so for various reasons, including food allergy, personal preference, or underlying illness, which may themselves cause increased fracture risk (15–17).
Researchers who have evaluated the association between volume of cow’s milk consumed and fracture risk in childhood have reported inconsistent findings (7, 18, 19). In a 2015 systematic review and meta-analysis of observational studies, Händel et al. (7) aimed to identify early-life dietary patterns which may contribute to fracture risk among healthy children. They found that the relationship between cow’s milk consumption and fracture risk in children was unclear and identified the need for prospective cohort studies to evaluate the temporal relationship between early childhood nutritional factors, such as cow’s milk consumption, and childhood fracture.
The fat content of cow’s milk may also influence fracture risk. Recent research found that low-fat milk consumption was associated with lower vitamin D stores and higher risk of childhood obesity (20), both of which may increase risk of childhood fracture (6, 11, 21–23). It has also been hypothesized that vitamin D and calcium may support optimal bone growth and reduce fracture risk (6, 7, 24) and that absorption of these substances may be enhanced when consumed with dietary fat (20, 25, 26).
We hypothesized that a higher volume of cow’s milk and higher milk-fat content consumed in early childhood would be protective against fracture in later childhood. To address this, we designed a prospective cohort study of milk consumption in early childhood and fracture risk in later childhood. The primary objective of this study was to evaluate whether volume of cow’s milk consumed at 1–3 years of age was associated with risk of fracture between 3 and 10 years of age. Secondary objectives included exploring whether milk fat consumed at 1–3 years of age was associated with risk of fracture between 3 and 10 years of age and whether milk-fat content modified the relationship between milk volume and fracture risk.
METHODS
Study design
A prospective cohort study was conducted through a practice-based research network, The Applied Research Group for Kids (TARGet Kids!). TARGet Kids! is a collaboration between the University of Toronto’s Faculty of Medicine and clinicians in the University’s departments of Paediatrics and Family and Community Medicine (27).
For this study, parents of children between 1 and 3 years of age were recruited by trained research assistants at a primary health-care visit made between September 2008 and December 2016 at one of 9 primary health-care clinics in Toronto, Ontario, Canada, and followed prospectively. Children who had conditions affecting growth (e.g., failure to thrive, cystic fibrosis), chronic illness (excluding asthma), or severe developmental delay were excluded.
Exposures and outcomes
Cow’s milk volume and fat content were measured at the first TARGet Kids! visit between 1 and 3 years of age, which was defined as the exposure window. This window was chosen to be consistent with a 2015 systematic review and meta-analysis (7) and with recommendations from the American Academy of Pediatrics (28) and the National Institutes of Health (29), which recommend consumption of whole cow’s milk starting at 1 year of age. Bone fracture was measured at the last TARGet Kids! visit made between 3 and 10 years of age, which defined the outcome window. This window was chosen because childhood fracture may be more sensitive to nutritional factors than adolescent fracture (7) and is known to be predictive of later fracture (4, 8). Informed consent was obtained from parents of participating children, and ethical approval was obtained from the Research Ethics Board of The Hospital for Sick Children and St. Michael’s Hospital.
Trained research assistants collected data from parents at each participating medical practice using a standardized data collection instrument adapted from the Canadian Community Health Survey (30). Anthropometric measurements were taken using standardized techniques.
The primary exposure variable was volume of cow’s milk consumed by each child during the exposure window. Milk volume was determined by asking a parent the question, “How many 250-mL cups of milk does your child currently have in a typical day?”. Children who did not consume cow’s milk were included in the analysis as consuming 0 cups per day. For children who consumed more than one type of milk fat, the average milk-fat content of all types of milk consumed was calculated.
The secondary exposure variable was the type of milk fat consumed by each child during the exposure window. This variable was measured by asking a parent the question, “Please specify your child’s diet for the past 3 days: skim, 1%, 2%, or whole milk.” For subjects who consumed more than one type of milk fat, the mean value was calculated by averaging the fat content of each type of milk consumed (20).
The primary outcome was 1 or more fractures experienced during the outcome window. It was measured by asking the question, “Has your child ever broken a bone?”. The response was categorized as yes or no (31).
Other variables
Covariates which might confound the relationship between cow’s milk consumption and fracture risk in childhood were determined a priori by means of a literature review (see Figure 1). These variables were measured during the exposure window using questions adapted from the Canadian Community Health Survey (30) and included maternal ethnicity (12, 32), household income (33, 34), child age, child sex (1, 2, 6), child body mass index (BMI) z score (6, 35, 36), child sugar-sweetened beverage consumption (37, 38), and child multivitamin and vitamin D supplementation (11, 39).
Figure 1.
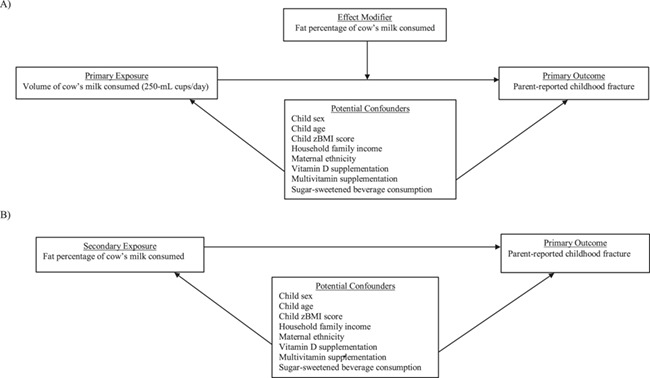
Directed acyclic graphs (DAGs) of relationships between variables in a study of early-childhood cow’s milk consumption and fracture risk, Toronto, Ontario, Canada, 2008–2016. A) DAG for the primary exposure with an effect modifier; B) DAG for the secondary exposure variable. zBMI score, body mass index z score.
Each child’s weight was measured with a Seca precision digital scale (±0.025%; Seca GmbH & Co. AG, Hamburg, Germany), and height was obtained using a Seca stadiometer. BMI was calculated as weight (kg) divided by the square of height (m2) (40). BMI z score was derived using the World Health Organization growth standards, which are age- and sex-standardized and are believed to represent children’s optimal growth (41, 42). Sugar-sweetened beverage consumption was quantified by asking a parent the question, “How many 250-mL cups of sugar-sweetened beverages does your child currently have in a typical day?”. Multivitamin and vitamin D supplementation were determined by asking the question, “Does your child take any vitamins or supplements regularly (vitamin D, multivitamin, etc.)?”. Annual household family income was reported by the parent. Maternal ethnicity was geographically categorized (43) to be consistent with the work of Wren et al. (12), who prospectively examined the influence of ethnicity on childhood fracture.
Demographic and nutritional factors, including sugar-sweetened beverage consumption, multivitamin supplementation, and vitamin D supplementation, were measured during the exposure window. Other covariates which may be associated with fracture risk, including age, BMI z score, outdoor free play time, and household family income, were measured during the outcome window.
Statistical analysis
Population characteristics were determined using mean values and proportions for the exposure variables, the outcome variable, and covariates. A modified Poisson regression model was used to evaluate the relationship between milk consumption during the exposure window and parental report of a fracture during the outcome window. Poisson regression was modified using a sandwich estimator, as recommended by McNutt et al. (44) and Zou et al. (45), to avoid overestimated relative risk estimates, which can occur when Poisson regression is applied to binomial data. Results were exponentiated to derive relative risks and 95% confidence intervals.
To minimize the possibility of biased R2 values, which can occur through data-generated model-building (46), we included all covariates listed above in the adjusted model, regardless of statistical significance. We used the same methods for the secondary analysis to evaluate the relationship between milk-fat consumption during the exposure window and parental report of a fracture during the outcome window (45).
To explore possible effect modification by milk-fat consumption, an interaction term for interaction between milk volume and milk fat was added to the primary model. The interaction was tested at an α = 0.05 significance level using a likelihood ratio test.
Multicollinearity was evaluated using the variance inflation factor, which was less than 5 for all covariates (47). We tested for nonlinearity in both the primary and secondary models by using restricted cubic splines (47). Missing data appeared to be missing at random and were handled using multiple imputation (48). Multiple imputation was conducted using all covariates and exposure and outcome data. All variables had less than 10% missing data (3.1% and 0.6% missing data for the primary and secondary exposures, respectively). All analyses were run on 50 imputed data sets, and results were then pooled for interpretation. R, version 3.31 (R Foundation for Statistical Computing, Vienna, Austria), and RStudio, version 0.99.903 (R Foundation for Statistical Computing), were the software packages used (49).
RESULTS
Exposure data were available on 4,461 TARGet Kids! participants at 1–3 years of age, and outcome data were available at ages 3–10 years for the 2,466 children who were included in the analysis. At the time of analysis, 1,738 children were younger than 3 years of age and 257 were older than 10 years of age (Figure 2). The mean age of participants during the exposure window was 1.6 (standard deviation (SD), 0.6) years, and 1,137 (46%) were female. Participants consumed a mean of 1.9 (SD, 1.2) 250-mL cups of cow’s milk per day during the exposure window, and the milk they consumed had a mean fat content of 2.4% (SD, 0.9) (Table 1). The mean age of the participants during the outcome window was 5.4 (SD, 1.8) years, with a mean duration of follow-up of 3.8 years. By the end of follow-up, 153 (6.2%) children had experienced at least 1 bone fracture, and 20 (13.1%) of those children had experienced 2 or more bone fractures.
Figure 2.
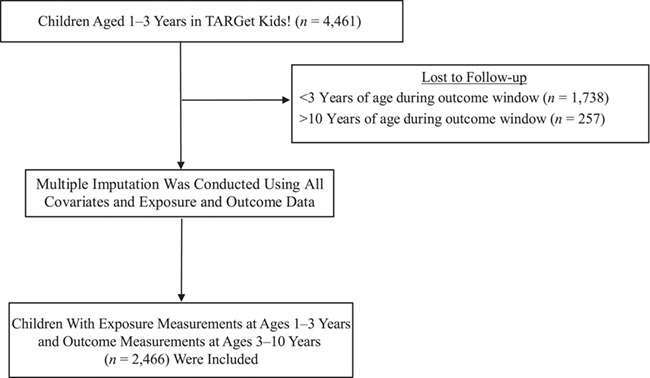
Selection of TARGet Kids! cohort participants for a study of early-childhood cow’s milk consumption and fracture risk, Toronto, Ontario, Canada, 2008–2016. At exposure measurement (ages 1–3 years), 76 (3.1%) children were missing data on volume of cow’s milk consumed and 14 (0.6%) children were missing data on milk-fat content, and therefore data were imputed for analysis. At outcome measurement (ages 3–10 years), 10 (0.4%) children were missing data on fracture, and those data were also imputed for analysis. TARGet Kids!, The Applied Research Group for Kids.
Table 1.
Characteristics of Participants with Follow-up Data in a Study of Early-Childhood Cow’s Milk Consumption and Fracture Risk, Toronto, Ontario, Canada, 2008–2016
| Measure | All Children (n = 2,466) | Category of Cow’s Milk Consumption, 250-mL cups/day | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0 (None) (n = 220) | <2 (n = 636) | 2 (n = 893) | >2 (n = 641) | |||||||
| No. | % | No. | % | No. | % | No. | % | No. | % | |
| Age, monthsa | 64.69 (22.66) | 25.25 (8.34) | 28.11 (7.30) | 28.47 (6.93) | 27.13 (7.22) | |||||
| Female sex | 1,137 | 46 | 106 | 48 | 293 | 46 | 426 | 48 | 283 | 44 |
| Cow’s milk consumption, 250-mL cups/daya | 1.88 (1.15) | 0.00 (0.00) | 0.86 (0.23) | 2.00 (0.00) | 3.37 (0.63) | |||||
| Cow’s milk-fat content, %a | 2.40 (0.86) | 0.00 (0.00) | 2.36 (0.92) | 2.40 (0.86) | 2.49 (0.82) | |||||
| Consumption of sugar-sweetened beverages, 250-mL cups/daya | 0.04 (0.26) | 0.02 (0.22) | 0.03 (0.23) | 0.03 (0.20) | 0.03 (0.23) | |||||
| Self-reported household family income, Can$/year | ||||||||||
| <30,000 | 75 | 3 | 10 | 5 | 12 | 2 | 21 | 3 | 25 | 4 |
| 30,000–79,999 | 294 | 13 | 34 | 17 | 79 | 14 | 97 | 12 | 73 | 12 |
| 80,000–149,999 | 651 | 29 | 55 | 27 | 176 | 30 | 234 | 28 | 171 | 29 |
| ≥150,000 | 1,253 | 55 | 102 | 51 | 311 | 54 | 480 | 58 | 327 | 55 |
| Child multivitamin supplementation (yes) | 649 | 27 | 59 | 28 | 177 | 29 | 226 | 27 | 169 | 27 |
| Child vitamin D supplementation (yes) | 1,250 | 53 | 118 | 55 | 345 | 56 | 458 | 54 | 300 | 48 |
| Maternal ethnicity | ||||||||||
| European | 1,659 | 72 | 138 | 69 | 449 | 76 | 618 | 73 | 413 | 70 |
| East Asian, South Asian, or Southeast Asian | 344 | 15 | 33 | 16 | 83 | 14 | 119 | 14 | 91 | 15 |
| Arab, African-American, or Latin American | 181 | 8 | 16 | 8 | 32 | 5 | 68 | 8 | 57 | 10 |
| Other | 105 | 5 | 14 | 7 | 26 | 4 | 36 | 4 | 27 | 5 |
| Bone fractureb | 153 | 6 | 17 | 8 | 31 | 5 | 59 | 7 | 41 | 6 |
a Values are expressed as mean (standard deviation).
b Bone fracture was measured during the outcome window at 3–10 years of age.
For the primary analysis, in both unadjusted and adjusted models, there was no statistically significant association between the volume of cow’s milk consumed at 1–3 years of age and the risk of 1 or more parent-reported child fractures between 3 and 10 years of age (per daily 250-mL cup of cow’s milk, the unadjusted relative risk was 1.05 (95% confidence interval (CI): 0.91, 1.18) and the adjusted relative risk was 1.04 (95% CI: 0.87, 1.26)) (Table 2). For the secondary analysis, neither unadjusted nor adjusted models identified a statistically significant association between the fat content of cow’s milk consumed at 1–3 years of age and fracture risk between 3 and 10 years of age (per 1% increment of milk fat, the unadjusted relative risk was 0.98 (95% CI: 0.79, 1.17) and the adjusted relative risk was 1.05 (95% CI: 0.84, 1.31)) (Table 2). There was no evidence of nonlinear relationships between cow’s milk volume or cow’s milk fat and risk of 1 or more fractures (Figures 3 and 4). Cow’s milk-fat consumption did not modify the relationship between milk volume and risk of fracture (P = 0.24).
Table 2.
Risk of Fracture During Follow-up (Ages 3–10 Years) According to Volume of Cow’s Milk Consumed and Milk-Fat Concentration at Baseline (Ages 1–3 Years) in an Adjusted Poisson Regression Model, Toronto, Ontario, Canada, 2008–2016
| Measure | Unadjusted Results | Adjusted Results a | ||||
|---|---|---|---|---|---|---|
| RR | 95% CI | P Value | RR | 95% CI | P Value | |
| Cow’s milk volume (per 250-mL cup/day) | 1.05 | 0.91, 1.18 | 0.52 | 1.04 | 0.87, 1.26 | 0.65 |
| Cow’s milk-fat content (per 1% increase in milk fat) | 0.98 | 0.79, 1.17 | 0.87 | 1.05 | 0.84, 1.31 | 0.66 |
Abbreviations: CI, confidence interval; RR, relative risk.
a Results were adjusted for child sex, child age (months), household family income (Can$/year), maternal ethnicity (see Table 1), child body mass index z score, multivitamin supplementation, vitamin D supplementation, and sugar-sweetened beverage consumption.
Figure 3.
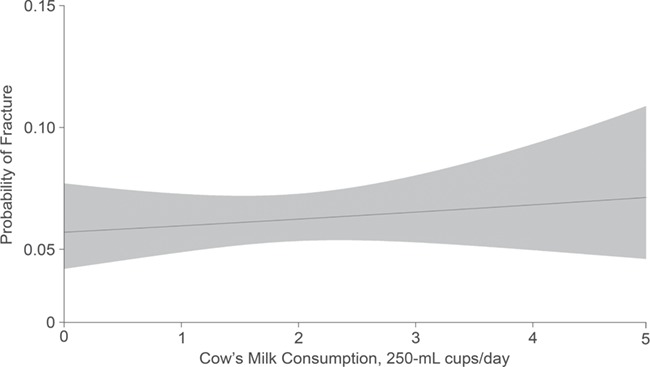
Relationship between cow’s milk intake and fracture risk among children from the TARGet Kids! cohort, Toronto, Ontario, Canada, 2008–2016. Results were adjusted for child sex, child age (in months), household family income, maternal ethnicity, child body mass index z score, multivitamin supplementation, vitamin D supplementation, and sugar-sweetened beverage consumption. The probability of fracture was determined during the outcome window at 3–10 years of age. Volume of cow’s milk consumed was measured during the exposure window at 1–3 years of age. The gray shaded area represents the 95% confidence intervals. TARGet Kids!, The Applied Research Group for Kids.
Figure 4.
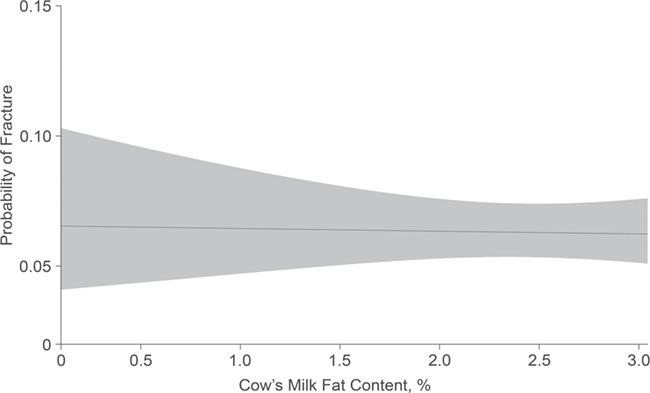
Relationship between cow’s milk-fat content and fracture risk among children from the TARGet Kids! cohort, Toronto, Ontario, Canada, 2008–2016. Results were adjusted for child sex, child age (in months), household family income, maternal ethnicity, child body mass index z score, multivitamin supplementation, vitamin D supplementation, and sugar-sweetened beverage consumption. The probability of fracture was determined during the outcome window at 3–10 years of age. Cow’s milk-fat content was measured during the exposure window at 1–3 years of age. The gray shaded area represents the 95% confidence intervals. TARGet Kids!, The Applied Research Group for Kids.
DISCUSSION
In this study, we used prospectively collected data from a large cohort of healthy urban children to evaluate the relationship of cow’s milk volume and fat content consumed in early childhood with fracture risk in later childhood. Neither milk volume nor the fat content of milk consumed in early childhood was associated with lower fracture risk in later childhood. Given that cow’s milk is consumed by over 80% of North American children on a daily basis (50) and that childhood fracture is a risk factor for subsequent fractures, as well as osteoporosis later in life (1, 3, 5, 6), the relationship between cow’s milk consumption and fracture risk is of both clinical and public health importance. To our knowledge, no other prospective cohort studies have examined the relationship between volume of cow’s milk consumed or milk-fat content and fracture risk in children.
It has been argued that nutritional factors may play a larger role in childhood fracture than adolescent or adulthood fracture (7). In adolescence, when peak height velocity is reached, bone length supersedes bone mineralization while strenuous physical activity increases, creating a scenario where bones may be more susceptible to fracture (4, 51). Children’s bones are less dense, not as rigid, and not exposed to the same forces as adolescent bones (51). Therefore, in childhood, fractures may be more likely to result from underlying causes such as nutritional factors or high-impact mechanisms (4).
In a 2008 systematic review of randomized controlled trials which focused on dietary calcium intake and bone mineralization among children aged 4–17 years, Huncharek et al. (24) found that increasing dietary calcium from dairy products resulted in increased total body and lumbar spine bone mineral density at 12–48 months of follow–up. However, bone mineralization is a proxy measure for fracture, and the authors concluded that further research is needed to determine whether dairy-food consumption in childhood translates beyond bone mineralization to decreased childhood fracture risk (24). Händel et al.’s 2015 systematic review and meta-analysis of observational studies (7), which aimed to evaluate nutritional factors that were associated with childhood fractures, included 18 observational studies, of which 2 were case-control studies that evaluated volume of cow’s milk consumed and fracture risk in healthy children aged 2–13 years (37, 39). The authors concluded that insufficient evidence was available to evaluate the association of cow’s milk consumption with childhood fracture risk (7). Lanou et al. (18) reviewed the literature on the effects of dietary calcium, dairy products, and bone health in children. The authors reported a positive relationship between consumption of dairy products and measures of bone health among children in 3 of 11 studies. One of those studies was cross-sectional and 2 were randomized controlled trials, neither of which measured the functional outcome of bone fracture. The authors concluded that “scant evidence supports nutrition guidelines focused specifically on increasing milk or other dairy product intake for promoting child and adolescent bone mineralization” (18, p. 736).
A relationship between cow’s milk-fat consumption and fracture risk has been hypothesized but little studied (52). It has been argued that calcium and vitamin D absorption, which are believed to support bone health and reduce fracture risk (6, 7, 24), may be enhanced if calcium and vitamin D are consumed with dietary fat (20, 25, 26). Further, consumption of cow’s milk fat has been associated with lower adiposity, which is believed to be protective against fractures (6, 20, 22, 23). However, in a case-control study, Chan et al. (53) examined fat consumption and fracture risk in children and did not find an association between fat intake and fracture. Similarly, in the present study, we did not identify a relationship between milk-fat consumption and childhood fracture risk.
This study had a number of strengths, including the prospective design involving a large, culturally diverse cohort of healthy urban children. Since cow’s milk consumption was measured in early childhood and bone fractures were measured up to 10 years of age, directionality in the hypothesized relationships was accounted for. Further, detailed questionnaire and anthropometric data allowed for adjustment for numerous potential risk factors which may have confounded a relationship between milk consumption and fracture risk.
This study also had several limitations, including the use of parent-reported measures, which may be susceptible to recall, social desirability, or misclassification bias. Social desirability bias may change the directionality of the relationship between cow’s milk and fracture if parents overreport cow’s milk consumption to more closely reflect current milk fat and volume guidelines. Misclassification bias and recall bias may affect the reliability of the responses and could affect the relationships in either direction. While numerous potential confounders were included in the statistical models, residual confounding is a possibility. Although cow’s milk is the main dietary source of calcium and vitamin D for most North American children, we were unable to account for other dietary sources of calcium because of data limitations (50). We were also unable to determine the mechanism of fracture, since some types of fractures may be more sensitive to nutritional factors (i.e., low-impact mechanisms). Another potential limitation is the risk of cointerventions. For example, children with multiple fractures may have received nutritional counseling which altered subsequent fracture risk. Although our study sample was relatively large, many children in our cohort were not yet 3 years of age at the time of analysis, limiting our ability to detect a small effect. Lastly, although the TARGet Kids! population is ethnically diverse, it may not be representative of all urban children.
In the present study, neither the volume nor the fat content of cow’s milk consumed in early childhood was associated with fracture in later childhood. These findings support a growing body of literature which suggests that efforts aimed at increasing cow’s milk consumption in childhood may not result in childhood fracture prevention (18, 52, 54). Future research in young children is needed to understand whether cow’s milk consumption in childhood offers benefits for fracture prevention through the life course.
ACKNOWLEDGMENTS
Author affiliations: Department of Nutritional Sciences, Faculty of Medicine, University of Toronto, Toronto, Ontario, Canada (Riley M. Allison, Catherine S. Birken, Andrew Howard, Marry L’Abbe, Jonathon L. Maguire); Department of Pediatrics, St. Michael’s Hospital, Toronto, Ontario, Canada (Riley M. Allison, Marie-Elssa Morency, Jonathon L. Maguire); Li Ka Shing Knowledge Institute, St. Michael’s Hospital, Toronto, Ontario, Canada (Riley M. Allison, Gerald Lebovic, Marie-Elssa Morency, Jonathon L. Maguire); Department of Pediatrics, The Hospital for Sick Children, Toronto, Ontario, Canada (Catherine S. Birken, Andrew Howard, Mary L’Abbe, Jonathon L. Maguire); Division of Pediatric Medicine, The Hospital for Sick Children, Toronto, Ontario, Canada (Catherine S. Birken, Andrew Howard, Mary L’Abbe, Jonathon L. Maguire); Child Health Evaluative Sciences, The Hospital for Sick Children, Peter Gilan Centre for Research and Learning, Toronto, Ontario, Canada (Riley M. Allison, Catherine S. Birken, Andrew Howard, Mary L’Abbe, Jonathon L. Maguire); and Institute of Health Policy and Management and Evaluation, School of Public Health, University of Toronto, Toronto, Ontario, Canada (Catherine S. Birken, Gerald Lebovic, Jonathon L. Maguire).
Funding for the TARGet Kids! Collaboration was provided by the Canadian Institutes of Health Research (CIHR) Institute of Human Development, Child and Youth Health (grant MOP-106532); by the CIHR Institute of Nutrition, Metabolism and Diabetes; and by the St. Michael’s Hospital Foundation. The Pediatric Outcomes Research Team was supported by a grant from The Hospital for Sick Children Foundation. J.L.M. received an unrestricted research grant for a completed investigator-initiated study from the Dairy Farmers of Canada (2011–2012), and the Ddrops Company (Woodbridge, Ontario, Canada) provided nonfinancial support (vitamin D supplements) for an investigator-initiated study on vitamin D and respiratory tract infections (2011–2015).
We thank all of the participating families for their time and involvement in TARGet Kids! and are grateful to all medical practitioners who are currently involved in the TARGet Kids! practice-based research network.
The TARGet Kids! Collaborators—Co-Lead Investigators: Catherine S. Birken, Jonathon L. Maguire; Advisory Committee: Ronald Cohn, Eddy Lau, Andreas Laupacis, Patricia C. Parkin, Michael Salter, Peter Szatmari, Shannon Weir; Science Review and Management Committees: Laura N. Anderson, Cornelia M. Borkhoff, Charles Keown-Stoneman, Christine Kowal, Dalah Mason; Site Investigators: Murtala Abdurrahman, Kelly Anderson, Gordon Arbess, Jillian Baker, Tony Barozzino, Sylvie Bergeron, Dimple Bhagat, Gary Bloch, Joey Bonifacio, Ashna Bowry, Caroline Calpin, Douglas Campbell, Sohail Cheema, Elaine Cheng, Brian Chis, Evelyn Constantin, Karoon Danayan, Paul Das, Anh Do, Kathleen Doukas, Anne Egger, Allison Farber, Amy Freedman, Sloane Freeman, Sharon Gazeley, Charlie Guiang, Dan Ha, Curtis Handford, Laura Hanson, Leah Harrington, Sheila Jacobson, Lukasz Jagiello, Gwen Jansz, Paul Kadar, Tara Kiran, Holly Knowles, Bruce Kwok, Sheila Lakhoo, Margarita Lam-Antoniades, Eddy Lau, Denis Leduc, Fok-Han Leung, Alan Li, Patricia Li, Jessica Malach, Roy Male, Vashti Mascoll, Aleks Meret, Elise Mok, Rosemary Moodie, Maya Nader, Katherine Nash, Sharon Naymark, James Owen, Michael Peer, Kifi Pena, Marty Perlmutar, Navindra Persaud, Andrew Pinto, Michelle Porepa, Vikky Qi, Nasreen Ramji, Noor Ramji, Danyaal Raza, Alana Rosenthal, Katherine Rouleau, Janet Saunderson, Vanna Schiralli, Michael Sgro, Hafiz Shuja, Susan Shepherd, Barbara Smiltnieks, Cinntha Srikanthan, Carolyn Taylor, Suzanne Turner, Fatima Uddin, Meta van den Heuvel, Joanne Vaughan, Thea Weisdorf, Sheila Wijayasinghe, Peter Wong, Ethel Ying, Elizabeth Young, Michael Zajdman; Research Team: Farnaz Bazeghi, Vincent Bouchard, Marivic Bustos, Charmaine Camacho, Dharma Dalwadi, Christine Koroshegyi, Tarandeep Malhi, Sharon Thadani, Julia Thompson, Laurie Thompson; Project Team: Mary Aglipay, Imaan Bayoumi, Sarah Carsley, Katherine Cost, Karen Eny, Theresa Kim, Laura Kinlin, Jessica Omand, Shelley Vanderhout, Leigh Vanderloo; Applied Health Research Centre: Christopher Allen, Bryan Boodhoo, Olivia Chan, David W. H. Dai, Judith Hall, Peter Juni, Gerald Lebovic, Karen Pope, Kevin Thorpe; Mount Sinai Services Laboratory: Rita Kandel, Michelle Rodrigues, Hilde Vandenberghe.
This work was presented at the 2018 meeting of the Pediatric Academic Societies (Toronto, Ontario, Canada) on May 7, 2018, and the 95th Annual Conference of the Canadian Paediatric Society (Quebec City, Quebec, Canada) on May 30, 2018.
The funding agencies played no role in study design; the collection, analysis, and interpretation of data; the writing of the report; or the decision to submit the article for publication.
Conflict of interest: none declared.
References
- 1. Landin LA. Fracture patterns in children. Analysis of 8,682 fractures with special reference to incidence, etiology and secular changes in a Swedish urban population 1950–1979. Acta Orthop Scand Suppl. 1983;202:1–109. [PubMed] [Google Scholar]
- 2. Cooper C, Dennison EM, Leufkens HG, et al. Epidemiology of childhood fractures in Britain: a study using the General Practice Research Database. J Bone Miner Res. 2004;19(12):1976–1981. [DOI] [PubMed] [Google Scholar]
- 3. Goulding A. Childhood fractures: time to implement strategies to reduce these events. Int Congress Ser. 2007;1297:3–14. [Google Scholar]
- 4. Ryan LM, Teach SJ, Searcy K, et al. Epidemiology of pediatric forearm fractures in Washington, DC. J Trauma. 2010;69(4 suppl):S200–S205. [DOI] [PubMed] [Google Scholar]
- 5. Yeh FJ, Grant AM, Williams SM, et al. Children who experience their first fracture at a young age have high rates of fracture. Osteoporos Int. 2006;17(2):267–272. [DOI] [PubMed] [Google Scholar]
- 6. Clark EM. The epidemiology of fractures in otherwise healthy children. Curr Osteoporos Rep. 2014;12(3):272–278. [DOI] [PubMed] [Google Scholar]
- 7. Händel MN, Heitmann BL, Abrahamsen B. Nutrient and food intakes in early life and risk of childhood fractures: a systematic review and meta-analysis. Am J Clin Nutr. 2015;102(5):1182–1195. [DOI] [PubMed] [Google Scholar]
- 8. Goulding A, Grant AM, Williams SM. Bone and body composition of children and adolescents with repeated forearm fractures. J Bone Miner Res. 2005;20(12):2090–2096. [DOI] [PubMed] [Google Scholar]
- 9. Goulding A. Risk factors for fractures in normally active children and adolescents. Med Sport Sci. 2007;51:102–120. [DOI] [PubMed] [Google Scholar]
- 10. Manias K, McCabe D, Bishop N. Fractures and recurrent fractures in children; varying effects of environmental factors as well as bone size and mass. Bone. 2006;39(3):652–657. [DOI] [PubMed] [Google Scholar]
- 11. Anderson LN, Heong SW, Chen Y, et al. Vitamin D and fracture risk in early childhood: a case-control study. Am J Epidemiol. 2017;185(12):1255–1262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Wren TA, Shepherd JA, Kalkwarf HJ, et al. Racial disparity in fracture risk between white and nonwhite children in the United States. J Pediatr. 2012;161(6):1035–1040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Goulding A, Rockell JE, Black RE, et al. Children who avoid drinking cow’s milk are at increased risk for prepubertal bone fractures. J Am Diet Assoc. 2004;104(2):250–253. [DOI] [PubMed] [Google Scholar]
- 14. Konstantynowicz J, Nguyen TV, Kaczmarski M, et al. Fractures during growth: potential role of a milk-free diet. Osteoporos Int. 2007;18(12):1601–1607. [DOI] [PubMed] [Google Scholar]
- 15. Monti G, Libanore V, Marinaro L, et al. Multiple bone fractures in an 8-year-old child with cow’s milk allergy and inappropriate calcium supplementation. Ann Nutr Metab. 2007;51(3):228–231. [DOI] [PubMed] [Google Scholar]
- 16. Henriksen C, Eggesbø M, Halvorsen R, et al. Nutrient intake among two-year-old children on cow’s milk-restricted diets. Acta Paediatr. 2000;89(3):272–278. [PubMed] [Google Scholar]
- 17. Heyman MB, Committee on Nutrition . Lactose intolerance in infants, children, and adolescents. Pediatrics. 2006;118(3):1279–1286. [DOI] [PubMed] [Google Scholar]
- 18. Lanou AJ, Berkow SE, Barnard ND. Calcium, dairy products, and bone health in children and young adults: a reevaluation of the evidence. Pediatrics. 2005;115(3):736–743. [DOI] [PubMed] [Google Scholar]
- 19. Rizzoli R. Dairy products, yogurts, and bone health. Am J Clin Nutr. 2014;99(5 suppl):1256S–1262S. [DOI] [PubMed] [Google Scholar]
- 20. Vanderhout SM, Birken CS, Parkin PC, et al. Relation between milk-fat percentage, vitamin D, and BMI z score in early childhood. Am J Clin Nutr. 2016;104(6):1657–1664. [DOI] [PubMed] [Google Scholar]
- 21. Kim SJ, Ahn J, Kim HK, et al. Obese children experience more extremity fractures than nonobese children and are significantly more likely to die from traumatic injuries. Acta Paediatr. 2016;105(10):1152–1157. [DOI] [PubMed] [Google Scholar]
- 22. Paulis WD, Silva S, Koes BW, et al. Overweight and obesity are associated with musculoskeletal complaints as early as childhood: a systematic review. Obes Rev. 2014;15(1):52–67. [DOI] [PubMed] [Google Scholar]
- 23. Kessler J, Koebnick C, Smith N, et al. Childhood obesity is associated with increased risk of most lower extremity fractures. Clin Orthop Relat Res. 2013;471(4):1199–1207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Huncharek M, Muscat J, Kupelnick B. Impact of dairy products and dietary calcium on bone-mineral content in children: results of a meta-analysis. Bone. 2008;43(2):312–321. [DOI] [PubMed] [Google Scholar]
- 25. Dawson-Hughes B, Harris SS, Lichtenstein AH, et al. Dietary fat increases vitamin D-3 absorption. J Acad Nutr Diet. 2015;115(2):225–230. [DOI] [PubMed] [Google Scholar]
- 26. Bullamore JR, Wilkinson R, Gallagher JC, et al. Effect of age on calcium absorption. Lancet. 1970;2(7672):535–537. [DOI] [PubMed] [Google Scholar]
- 27. Carsley S, Borkhoff CM, Maguire JL, et al. Cohort profile: The Applied Research Group for Kids (TARGet Kids!). Int J Epidemiol. 2015;44(3):776–788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Gidding SS, Dennison BA, Birch LL, et al. Dietary recommendations for children and adolescents: a guide for practitioners. Pediatrics. 2006;117(2):544–559. [DOI] [PubMed] [Google Scholar]
- 29. MedlinePlus, US National Library of Medicine Feeding patterns and diet—children 6 months to 2 years. Bethesda, MD: National Library of Medicine; 2015. https://medlineplus.gov/ency/patientinstructions/000713.htm. Accessed August 15, 2017. [Google Scholar]
- 30. Statistics Canada Canadian Community Health Survey—annual component (CCHS). Ottawa, ON, Canada: Health Canada; 2016. https://www.statcan.gc.ca/eng/survey/household/3226. Accessed August 16, 2017. [Google Scholar]
- 31. Clark EM, Ness AR, Bishop NJ, et al. Association between bone mass and fractures in children: a prospective cohort study. J Bone Miner Res. 2006;21(9):1489–1495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Wetzsteon RJ, Hughes JM, Kaufman BC, et al. Ethnic differences in bone geometry and strength are apparent in childhood. Bone. 2009;44(5):970–975. [DOI] [PubMed] [Google Scholar]
- 33. Stark AD, Bennet GC, Stone DH, et al. Association between childhood fractures and poverty: population based study. BMJ. 2002;324(7335):457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Lyons RA, Delahunty AM, Heaven M, et al. Incidence of childhood fractures in affluent and deprived areas: population based study. BMJ. 2000;320(7228):149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Jones IE, Williams SM, Goulding A. Associations of birth weight and length, childhood size, and smoking with bone fractures during growth: evidence from a birth cohort study. Am J Epidemiol. 2004;159(4):343–350. [DOI] [PubMed] [Google Scholar]
- 36. Bialo SR, Gordon CM. Underweight, overweight, and pediatric bone fragility: impact and management. Curr Osteoporos Rep. 2014;12(3):319–328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Ma D, Jones G. Soft drink and milk consumption, physical activity, bone mass, and upper limb fractures in children: a population-based case-control study. Calcif Tissue Int. 2004;75(4):286–291. [DOI] [PubMed] [Google Scholar]
- 38. Petridou E, Karpathios T, Dessypris N, et al. The role of dairy products and non alcoholic beverages in bone fractures among schoolage children. Scand J Soc Med. 1997;25(2):119–125. [DOI] [PubMed] [Google Scholar]
- 39. Ryan LM, Teach SJ, Singer SA, et al. Bone mineral density and vitamin D status among African American children with forearm fractures. Pediatrics. 2012;130(3):e553–e560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Mei Z, Grummer-Strawn LM, Pietrobelli A, et al. Validity of body mass index compared with other body-composition screening indexes for the assessment of body fatness in children and adolescents. Am J Clin Nutr. 2002;75(6):978–985. [DOI] [PubMed] [Google Scholar]
- 41. Pietrobelli A, Faith MS, Allison DB, et al. Body mass index as a measure of adiposity among children and adolescents: a validation study. J Pediatr. 1998;132(2):204–210. [DOI] [PubMed] [Google Scholar]
- 42. World Health Organization WHO Child Growth Standards: Length/Height-for-Age, Weight-for-Age, Weight-for-Length, Weight-for-Height and Body Mass Index-for-Age. Methods and Development Geneva, Switzerland: World Health Organization; 2006. http://www.who.int/childgrowth/standards/technical_report/en/. Accessed August 15, 2017. [Google Scholar]
- 43. Omand JA, Carsley S, Darling PB, et al. Evaluating the accuracy of a geographic closed-ended approach to ethnicity measurement, a practical alternative. Ann Epidemiol. 2014;24(4):246–253. [DOI] [PubMed] [Google Scholar]
- 44. McNutt LA, Wu C, Xue X, et al. Estimating the relative risk in cohort studies and clinical trials of common outcomes. Am J Epidemiol. 2003;157(10):940–943. [DOI] [PubMed] [Google Scholar]
- 45. Zou G. A modified Poisson regression approach to prospective studies with binary data. Am J Epidemiol. 2004;159(7):702–706. [DOI] [PubMed] [Google Scholar]
- 46. LR DB. Statistical Analysis With Missing Data. 2nd ed. Hoboken, NJ: John Wiley & Sons, Inc.; 2002. [Google Scholar]
- 47. Chan BKC. Biostatistics for Epidemiology and Public Health Using R. 1st ed. New York, NY: Springer Publishing Company; 2013. [Google Scholar]
- 48. Klebanoff MA, Cole SR. Use of multiple imputation in the epidemiologic literature. Am J Epidemiol. 2008;168(4):355–357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. R Core Team R: A Language and Environment for Statistical Computing Vienna, Austria: R Foundation for Statistical Computing; 2018. http://www.R-project.org. Accessed March 3, 2017. [Google Scholar]
- 50. Garriguet D. Beverage consumption of children and teens. Health Rep. 2008;19(4):17–22. [PubMed] [Google Scholar]
- 51. Rauch F, Neu C, Manz F, et al. The development of metaphyseal cortex—implications for distal radius fractures during growth. J Bone Miner Res. 2001;16(8):1547–1555. [DOI] [PubMed] [Google Scholar]
- 52. Michaëlsson K, Wolk A, Langenskiöld S, et al. Milk intake and risk of mortality and fractures in women and men: cohort studies. BMJ. 2014;349: g6015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Chan GM, Hess M, Hollis J, et al. Bone mineral status in childhood accidental fractures. Am J Dis Child. 1984;138(6):569–570. [DOI] [PubMed] [Google Scholar]
- 54. Weinsier RL, Krumdieck CL. Dairy foods and bone health: examination of the evidence. Am J Clin Nutr. 2000;72(3):681–689. [DOI] [PubMed] [Google Scholar]


