Abstract
Sterilization and disinfection are the basic components of hospital infection control activities. Every day, a number of hospitals are performing various surgical procedures. Even more number of invasive procedures are being performed in different health care facilities. The medical device or the surgical instrument that comes in contact with the sterile tissue or the mucus membrane of the patient during the various processes is associated with increased risk of introduction of pathogens into the patient's body. Moreover, there is chance of transmission of infection from patient to patient; from patient or to health care personnel, and vice versa; or from the environment to the patient through the improper sterilized or disinfected devices. Hence, medical personnel, laboratory people and the health care providers should have better knowledge regarding these techniques to prevent the spread of these pathogens.
Keywords: Bundle approach, Disinfection techniques, Health care–associated infections, Medical equipment, Sterilization techniques
Background
Sterilization and disinfection are the basic components of hospital infection control activities. Every day, a number of hospitals are performing various surgical procedures. Even more number of invasive procedures are being performed in different health care facilities. The medical device or the surgical instrument that comes in contact with the sterile tissue or the mucus membrane of the patient during the various processes is associated with increased risk of introduction of pathogens into the patient’s body. Moreover, there is chance of transmission of infection from patient to patient; from patient or to health care personnel, and vice versa [e.g., hepatitis B virus (HBV)]; or from the environment to the patient (e.g., Pseudomonas aeruginosa, Acinetobacter spp.) through the improper sterilized or disinfected devices. A number of outbreaks and infections were reported in the hospital setup because of improperly sterilized devices.1, 2 Many infections were reported throughout the world due to use of contaminated endoscopes.3, 4, 5, 6, 7 Hence, adequate decontamination techniques for medical and surgical devices are needed in all the health care facilities. The health care providers are equally responsible for the reduction and elimination of such infections. Every hospital should have its own guidelines of sterilizing and disinfecting items on the basis of their intended use of medical devices and associated infections. Currently, there is an increase in frequency of newly emerging and multidrug-resistant pathogens in all health care centers for which only few/if any treatments are available. Hence, the medical personnel, laboratory people, and health care providers should have better knowledge regarding these techniques to prevent the spread of these pathogens.
There is no uniform definition or standards for sterilization, disinfection, and cleaning. However, the Clinical and Laboratory Standards Institute has defined some minimum inhibitory concentration testing–based recommendations, which are currently standardized by the International Organization of Standardization.8 The norms were first established in 2004, and later in 2010, new guidelines were published.
Sterilization: Sterilization is defined as a process of complete elimination or destruction of all forms of microbial life (i.e., both vegetative and spore forms), which is carried out by various physical and chemical methods. Technically, there is reduction ≥106 log colony forming units (CFU) of the most resistant spores achieved at the half-time of a regular cycle.
Chemical sterilant: These are chemicals used for a longer duration (3–12 h) to destroy all forms of microbes, e.g., peracetic acid (PAA) (0.2%), glutaraldehyde (≥2.4%), ortho-phthalaldehyde (OPA) (0.55%), and hydrogen peroxide (7.5%).
Disinfection: Disinfection is defined as a process of complete elimination of vegetative forms of microorganisms except the bacterial spores from inanimate objects. Technically, there is reduction of ≥103 log CFU of microorganisms by this method without spores.
High-level disinfectant (HLD): Used for shorter duration and able to kill 106 log microorganisms except spores, e.g., glutaraldehyde (≥2.0%), OPA (0.55%), hydrogen peroxide (7.5%), hypochlorite (650–675 ppm), and hypochlorous acid (400–450 ppm).
Intermediate level disinfectant (ILD): These disinfectants act against Mycobacterium tuberculosis and are mainly used for noncritical items contaminated with blood/body fluids.
Low-level disinfectant (LLD): LLDs are used to remove the vegetative form of bacteria, few fungi, and some enveloped viruses from the noncritical items, e.g., 3% hydrogen peroxide, quaternary ammonium compound, diluted glutaraldehyde, phenolics, etc.
Decontamination and cleaning: Decontamination is the process of removal of pathogenic microorganisms from objects so that they are safe to handle. Cleaning is defined as removal of visible soil (e.g., organic and inorganic materials) from the surfaces and objects. Technically, it achieves minimum reduction of ≥1 log CFU of microorganisms.
Antisepsis: Antisepsis is a process of removal of germs from the skin. When it is related to the patient’s skin, it means disinfection of living tissue or skin. When it is related to the health care worker, it means reduction or removal of transient microbe from the skin.
Germicide: It is the agent that destroys germs. It includes both antiseptics and disinfectants. The type of microorganism is identified from the prefix (e.g., virucide, fungicide, bactericide, sporicide, and tuberculocide).
Classification of Medical Devices and/or Equipment
The principal goal of cleaning, disinfection, and sterilization is to reduce the number of microorganisms on the device to such a level that the probability of transmission of infection will be nil. The risk of transmission of infection depends on the type of device. For example, in case of blood pressure–measuring cuff, the device that comes in contact with the skin carries least risk of transmission of disease. On the other hand, devices that come in contact with the neural tissue of a patient suffering from Creutzfeldt–Jakob disease (CJD) have high risk of transmission of infection.9, 10, 11 To reduce the potential risks of transmission of infection through different devices, Earle H. Spaulding in 1968 proposed a classification to define the desired level of antimicrobial killing for different devices.12 He categorized the devices into three categories depending on the potential risk of transmission of infectious agents: critical, semicritical, and noncritical (Table 59.1 ).
Table 59.1.
Spaulding’s Classification of Devices12
| Device/Item | Definition | Risk of Infection | Example | Reprocessing Procedure |
|---|---|---|---|---|
| Critical | Medical device that is intended to enter a normally sterile environment, sterile tissue, or the vasculature | High | Surgical instrument, cardiac catheter, implants, needle, ultrasound probes used in sterile body cavity | Sterilization by steam, plasma, or ethylene oxide |
| Semicritical | Devices that are intended to come in contact with the mucous membrane or nonintact skin | High/intermediate | Flexible endoscope, respiratory therapy equipment, manometry probes, diaphragm-fitting rings, laryngoscope blades | Sterilization desirable, high-level disinfectants |
| Noncritical | Devices come in contact with intact skin | Low | Blood pressure cuff, stethoscope | Intermediate or low-level disinfectant |
This simple classification of Spaulding’s needs to be revised, as it did not address the devices that come in contact with the mucous membrane (e.g., endoscope), biopsy forceps touching the breach sterile tissue, heat-sensitive items, and prions. The Centers for Disease Control and Prevention (CDC) in 1991 proposed an additional category to Spaulding’s classification as “environmental surfaces” to represent the surfaces that usually do not come in contact with patient.13 Environmental surfaces can be further subgrouped as clinical contact surfaces (medical equipment or high-touch surfaces) and housekeeping surfaces. CDC defines clinical contact surfaces as the areas that act like reservoirs of microorganisms, e.g., hands of health care workers. High-touch surfaces such as telephone, light switch board, bedrails, computer, door handle, and medical equipment like ventilator, X-ray machines, and hemodialysis machines are the contacting equipment that subsequently contact the patient. CDC had issued guidelines for hand washing and hospital environmental control. Different LLDs and ILDs that can be used to disinfect the clinical contact surfaces were approved by the Environmental Protection Agency (EPA). The housekeeping surfaces such as walls, floor, and sinks carry very low risk of transmission of infection. So, disinfection of such surfaces is less frequent in comparison to the previous one.
Recommendation of Preferred Methods for Various Medical Devices
The antimicrobial spectra of different methods are different from each other (Fig. 59.1 ). Hence, health care personnel should have adequate knowledge for the selection and recommendation of different sterilization and disinfection methods (Table 59.2, Table 59.3 ). A brief knowledge about the compatibility, toxicity, odor, and irritability due to various agents/methods is essential and useful for achieving adequate decontamination.
Figure 59.1.
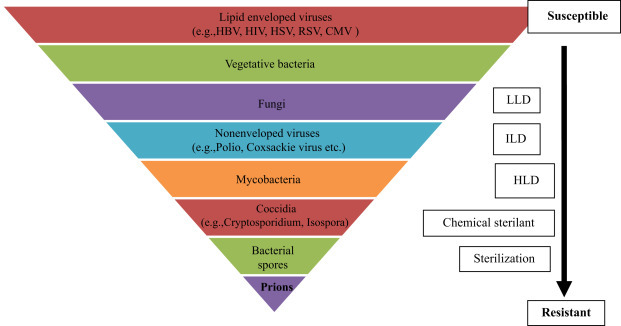
Increasing order of resistance of microorganisms to sterilization and different levels of disinfectants.
Table 59.2.
Different Methods of Sterilization With Examples
| Methods of Sterilization | Example | Target | Application |
|---|---|---|---|
| High temperature | Steam, dry heat | All forms of microbes (vegetative and spore) |
|
| Low temperature | Ethylene oxide gas, hydrogen peroxide, ozone, gas plasma, gaseous chlorine dioxide, ionizing radiation, pulsed light | Gram-positive bacteria, gram-negative bacteria, mycobacteria, lipid-enveloped viruses, large nonenveloped viruses, spores, cyst, trophozoite, coccidia |
|
| Liquid chemicals | Chemical sterilants | Prions (resistant to any form of sterilization) |
|
| Others | Filtration |
|
Table 59.3.
Different Methods of Disinfection With Examples
| Methods of Disinfection | Example | Target | Application |
|---|---|---|---|
| HLD | |||
| Heat | Pasteurization (∼50 min) | All vegetative forms of microorganisms |
|
| Chemicals | Chemical sterilants | Gram-positive bacteria, gram-negative bacteria, mycobacteria, lipid-enveloped viruses, large nonenveloped viruses, spores, cyst, trophozoite, coccidian |
|
| ILD | |||
| Chemicals | EPA-registered with tuberculocidal activity (e.g., chlorine-based products, phenolics) | Gram-positive bacteria, gram-negative bacteria, enveloped and nonenveloped viruses, mycobacteria |
|
| LLD | |||
| Chemicals | EPA registered but without tuberculocidal activity (e.g., chlorine-based products, phenolics, quaternary ammonium compounds, 70–90% alcohol) | Vegetative forms of bacteria, lipophilic viruses and some fungi |
|
EPA, Environmental Protection Agency; GI, gastrointestinal; HLD, high-level disinfectant; ILD, intermediate-level disinfectant; LLD, low-level disinfectant.
The various chemicals used for the process of antisepsis or skin disinfection are chloroxylenols, anilides, hexachloraphene, polymeric biguanides, alexidine, diamidines, and triclosan.
Sterilization, Disinfection, and Cleaning in the Health Care Facilities
Sterilization, disinfection, and cleaning in health care facilities include disinfection and cleaning of environmental surfaces with/without cleaning and reprocessing the medical equipment. The former includes mainly the noncritical items such as surfaces, floors, and high-contact surfaces (sinks, telephones, switches board, bed railings, trolleys etc.). It is observed that regular cleaning of all these housekeeping surfaces dramatically reduces the transmission of the infection.
Recommendation for the Cleaning and Decontamination of Environmental Surfaces14
-
•
The space for cleaning and other work should be clearly demarcated and separated by walls.
-
•
The hospital staffs should be properly trained regarding the cleaning and decontamination practices of hospital surfaces.
-
•
The staffs should wear personal prophylactic equipment (PPE), i.e., gowns, gloves, masks, and boots. There must be separate area for removing PPE.
-
•
Fresh preparation of detergents or disinfectants should be made every day and used with the appropriate dilution as per the manufacturer instruction.
-
•
Wet mopping of floors should be encouraged as dry mopping generates dust aerosols.
-
•
Avoid using HLD for environmental surfaces of the hospital and offices. Mopping of the hospital surfaces should be done using detergent. Table tops and counters should also be cleaned regularly by detergent only.
-
•
Mopping of high-risk areas such as the intensive care units (ICUs), burn wards, transplant units, isolation wards, operation theaters (OTs), and dialysis machines should be done using HLD instead of detergent. Cleaning by vacuum pump and use of high-efficiency particulate air (HEPA) filters for the exhaust are preferred in these places.
Recommendation for Blood Spill on the Surface15, 16, 17, 18, 19, 20
-
•
Cleaning and decontamination of the hospital surfaces having spilled blood is done as per the recommendation of Occupational Safety and Health Administration/World Health Organization/CDC.
-
•
ILD or disinfectants with tuberculocidal activity should be used for blood spill in the hospital surfaces.
-
•
For decontamination of small amount of blood spills (<10 mL), sodium hypochlorite solution is used with a dilution of 1:100. For spill >10 mL, sodium hypochlorite with 1:10 dilution is used for the first application. The organic matter should be cleaned with absorbent material, and final disinfection may be done using sodium hypochlorite solution with 1:100 dilution.
Cleaning and Disinfection of Medical Instruments15, 16, 17, 21, 22, 23, 24, 25, 26, 27, 28
The cleaning and disinfection of medical equipment depends on their physical nature, character of the material it is made up of, lumen size, etc. Thorough cleaning is preferred before the use of the disinfectants as cleaning effectively removes majority of the microbes from the equipment.
-
•
Staffs should be properly educated and trained regarding the cleaning procedure, physical and chemical nature of the instruments, nature of disinfectants, etc. All the staffs during the process should use PPE.
-
•
Dry organic materials are difficult to remove from the instrument. Hence, drying should be avoided by immersing the equipment in the detergent or disinfectant solution prior to cleaning. The soaked matter can be cleaned by manual scrubbing and rubbing with brush or automated scrubber and thoroughly washed with water under pressure. Avoid prolonged or overnight soaking of the devices.
-
•
The time of exposure, and concentration of the detergent or disinfectant, should be properly maintained as mentioned in the literature. Too low concentration may not work effectively to remove the organic materials or microorganisms.
-
•
The pH of the disinfectant should be properly obtained as per the manufacturer’s instruction. Delicate articles should be processed in neutral pH.
-
•
Enzymes like proteases may be added to the solution to fasten the cleaning action. Enzymatic cleaners with neutral pH are preferred to avoid the damage of the articles. For example, in case of flexible endoscope, neutral pH detergent with enzymatic action is preferred. A new nonenzyme product [hydrogen peroxide based, US Food and Drug Administration (FDA) cleared] has been found to be very effective as cleaning agent.
Cleaning and Reprocessing of Patient Care Equipment14
Patients care equipment are divided into three categories (critical, semicritical, and noncritical) depending on the intended use and risk of transmission of infection. The cleaning and reprocessing protocol for each category are detailed in Table 59.4, Table 59.5, Table 59.6 .
Table 59.4.
Cleaning and Reprocessing of Critical Patient Care Items
| Objects | Mode of Reprocessing |
|---|---|
| Catheters (cardiac, arterial, urine) | Sterile, single use |
| Needles | Disposable, single use |
| Implantable devices | Sterile, single use |
| Intravascular devices | Sterile, single use |
| Surgical instruments | Heat sterilization in autoclaves |
| Arterial pressure transducers | Sterilize by heat/low-temperature (H2O2)/ethylene oxide |
| Diagnostic ultrasound | Sterilization/HLD as per manufacturer’s instruction |
| Heart–lung oxygenator surfaces | Heat/low-temperature sterilization |
| Hemodialysis and plasmapheresis | Heat/low-temperature sterilization |
| Neurological test needles | Disposable, if using reusable: Sterilize with heat/steam/sterilize by EtO |
EtO, ethylene oxide; HLD, high-level disinfectant.
Table 59.5.
Cleaning and Reprocessing of Semicritical Patient Care Items
| Items | Methods |
|---|---|
| Anesthetic equipment (airways, endotracheal tubes, etc.) | Preferably sterilization, HLD may be used as an alternative |
| Respiratory equipment | HLD |
| Laryngoscopes and its blade | HLD with liquid germicides/disinfectants |
| Endoscopes | Heat sterilization/low-temperature sterilization/HLD |
| Nebulizer and nebulizer cups | Clean and disinfect |
| Respiratory therapy equipment | HLD |
| Resuscitation accessories | Heat disinfect/wash with detergent and hot water |
HLD, high-level disinfectant.
Table 59.6.
Cleaning of Noncritical Patient Care Items
| Items | Method |
|---|---|
| Ambu bag and mask | Clean with detergent, dry, and thermally disinfect |
| Blood pressure apparatus and cuff | Disinfect the cuff with 70–90% alcohol/other LLDs |
| Cloth appliances | If reusable, clean with detergent and water, dry and disinfect with 70% alcohol |
| Doppler | Head of the Doppler should be wiped with 70% alcohol |
| Bed and bed rails | LLD |
| Gowns | Single use in case of disposable, if reusable, use 0.5% bleaching powder, dry in sun/cloth drier |
| High-touch surface | Clean with 70–90% alcohol twice a day |
| IV stand, IV monitoring pumps | Clean with detergent and water and dry |
| Surgical mask | Disposable/discard as per manufacturer’s recommendation |
| Stethoscope | Clean the bell with 70–90% alcohol |
| Thermometer | After every use wipe with 70–90% alcohol |
| Trolleys | Wipe with 70–90% alcohol every day |
| Walls, wash basin | clean with detergent and water |
IV, intravenous; LLD, low-level disinfectant.
Reprocessing of the Critical Items
Devices that come in contact with the sterile parts of the body are included in critical items category. They carry the highest risk of transmission of infection. Hence, sterilization is the method of choice for the reprocessing of these items (heat stable). The FDA has approved ethylene oxide (EtO), plasma sterilization, and liquid sterilization with glutaraldehyde or PAA in heat-sensitive items.22, 23 All packed sterile items should be kept with proper precaution to avoid environmental contamination.
Reprocessing of Semicritical Items
Items that come in contact with the mucous membrane of the skin are included in this category. These items should be processed by either heat sterilization or HLD after cleaning (Table 59.5).
All the semicritical items should be rinsed with sterile water or alcohol. Forced air drying after the rinsing process drastically reduces the rate of contamination. It is found that cleaning also reduces the transmission of infection in human immunodeficiency virus (HIV)–contaminated instruments. Items are found to be germ free when soaked in 2% glutaraldehyde for 20 min after the cleaning process. OPA, glutaraldehyde, and automated process using PAA are the three disinfectants commonly used for the reprocessing of endoscopes.
Reprocessing of Noncritical Items
Items that come in contact with the intact skin are included within noncritical items. These include clothing, floors, high-touch surfaces, furniture, baths, bed pans, weighing scale, brushes, beddings, crockery, earphones, mobiles, and trolleys. The risk of transmission of infection with these items is observed to be the lowest. However, they contribute to the transmission of infection in indirect way. For example, methicillin-resistant Staphylococcus aureus (MRSA), and vancomycin-resistant enterococci (VRE) are commonly isolated with the patient’s belonging and can be easily transmitted to other patient by health care worker’s hand causing infection. These items do not need sterilization; however, they should be regularly cleaned and disinfected with LLD to decrease the transmission of infective organisms (Table 59.6).
Reprocessing of Respiratory Apparatus and Endoscopes15, 21, 24, 29, 30, 31, 32, 33, 34
Respiratory apparatus such as ventilators, humidifiers, nebulizers, pulmonary screening devices, anesthetic equipment, laryngoscope and its blade, and suction equipment are most important in the ICU setup because of its association with the risk of transmission of infection. Proper cleaning and infection preventive measures should be followed while handling these instruments as they are highly associated with the transmission of infection from one patient to other. They come in contact with the mucous membrane of the body, are included in the semicritical item category, and are sterilized or disinfected with the HLD.
Ventilators
Ventilators are important sources of hospital-acquired infection. This artificial airway is associated with increased chance of aspiration of the bacteria causing infection. Mechanical ventilators are directly not associated with the infection, but their internal circuits (includes the filter, tubing, humidifier, etc.) and the fluids are the potential source of infection. As per the CDC guidelines, the permanent circuits should be replaced with sterile ones, when there is visible soiling or mechanical obstruction. It has been also seen that changing interval of tubing at 7, 14, and 30 days drastically reduces the transmission of infection. In case of detachable circuits, it should be dismantled, cleaned, and disinfected.
Routine Ventilator Care
-
•
Clean the equipment regularly and cover the ventilator, when it is not in use.
-
•
Use sterile water to fill the humidifier as tap water causes introduction of microorganisms like Burkholderia cepacia and Legionella spp.
-
•
Use PPE, and mask during handling these equipment. Discard all disposals and perform hand hygiene after each handling.
-
•
Tubings of the ventilator are infected with the secretion of the patient. The condensate from the inspiratory lines may spill to the tracheobronchial tree of the patient or into the nebulizer while handling, changing, or manipulating the ventilator circuit.
-
•
The effluent from the ventilator may contaminate the environment and can reenter through ventilator to the patient’s airway increasing the chance of infection.
-
•
Select HEPA filters for both the inspiratory and expiratory limbs of ventilator circuits.
-
•
Do not allow the condensate to drain back into the patient’s airway or back to into humidifiers.
-
•
Change the disposable parts of the ventilator after each use, and decontamination should be done after 48 h for the reusable items.
-
•
Clean the visible soiling; sterilize the parts with autoclaving/low-temperature sterilization.
-
•
Infant ventilators should be sterilized with EtO.
-
•
Toxic residues should be removed after each cycle of sterilization by flushing with air and oxygen.
Humidifiers
For proper humidification, use sterile water in place of tap water to fill the humidifiers. The fluid should be dispensed aseptically without entering or touching by hand. On the other hand, heat–moisture exchanger (HME) can be used. It absorbs the heat and humidity from the expired air of the patient and stores it. During the inhalation process, the cold dry gas entering to the ventilator absorbs this heat and moisture, thereby reducing the formation of condensate. HME should be exchanged in case of gross contamination, mechanical dysfunction, or in between patients. Humidifiers can be cleaned using 70–90% alcohol. No antiseptic should be added to the water used for the humidifier.
Nebulizer
Use sterile water for nebulization. The remaining fluid or medication should be handled aseptically. After every use, the nebulizer cap should be made dry or flushed with 70–90% alcohol before filling with water. The mouthpiece and mask should also be cleaned with warm water and dried before every use.
Anesthetic Equipment
Anesthetic equipment such as face mask, ambu bag, tubings, and endotracheal tubes should be regularly cleaned. In patients suspected of tuberculosis, disposable face mask and tubings should be used. Ambu bag should be kept covered to avoid exposure to dust particles. The bags should be changed in case of visible soiling or secretion.
Pulmonary Screening Device
Do not routinely sterilize or disinfect the internal machinery of pulmonary function test machines. They should be wiped and disinfected with HLD in between patients. All the screening devices (inspiratory force manometer, tidal volume/vital capacity devices, and peak flow meters) should be discarded after single use.
Reprocessing of Endoscopes29, 35
Endoscopes are very useful tools for diagnostic as well as therapeutic processes. Many outbreaks are reported with the contaminated endoscopes due to faulty reprocessing processes. Reprocessing of endoscopes remains the most challenging task in the health care facilities. Although the incidence of infection associated with the contaminated endoscope is quite low (∼1 in 1.8 million procedures), its frequency is found higher in comparison to other devices. The bioburden depends on the body cavity it is intended to visualize. For example, endoscopes used for gastrointestinal tract harbor 105 CFU–1010 CFU/mL compared with 6.4 × 104 CFU/mL in bronchoscopes. It also depends on the material it is made up of. Most of the flexible endoscopes (e.g., flexible bronchoscope, gastroscope, duodenoscope, sigmoidoscopes) are found to be heat sensitive and unable to withstand many chemicals. Flexible endoscopes have multiple channels, small lumen, as well as honeycombed and blind ends, which are very difficult to clean. The endoscopes can acquire contamination from the patients, hospital environment, or water supply. It has been noticed that procedures such as endoscopic retrograde cholangiopancreatography (ERCP) are associated with many iatrogenic infection.
When the endoscopes touch the sterile tissue, they are classified as critical items and sterilization or HLD is the ideal procedure for reprocessing. Endoscopes coming in contact with the mucus membrane are classified as semicritical items, and HLD should be used for the reprocessing. With proper cleaning, the level of bioburden seems to be decreased by 4–6 log10. Many reports showed that with proper cleaning decontamination could be achieved from HIV also. Few advice the application of 20% glutaraldehyde for 20 min after the cleaning step. The FDA has approved a list of HLDs/sterilants for reprocessing endoscopes. The following norms should be followed by the staffs to reduce the faults during reprocessing.
-
•
Each person who reprocesses should be properly trained, use PPE (gowns, gloves, goggles, face mask, etc.) during the process
-
•
They should be properly educated about the chemical and biological hazards and comply with the manufacturer’s instruction.
-
•
Determine which process of sterilization is suitable for your endoscope. If it is heat stable, use autoclave/low-temperature sterilization methods such as hydrogen peroxide gas plasma or PAA, whereas liquid sterilants/HLDs can be used for heat-sensitive scopes.
In general, reprocessing of the scopes includes five steps after a leak testing: cleaning, disinfection, rinsing, drying, and storing.
Cleaning
Cleaning can be done by completely immersing the scopes in the detergent/disinfectant solution. All the components should be dismantled before immersion. The internal and external surface of the scopes should be gently cleaned with the help of brush or soft cloth. Brushes should be applied to the orifices and internal surfaces to remove the organic residues. All the items should be cleaned thoroughly, and the disinfectant/detergent should be discarded after cleaning. Determine whether the scope can be suitable for automatic washer with sterilizers. These systems contain PAA/hydrogen peroxide plasma-based (HPP), which are highly effective for killing vegetative and spore forms. Hence, precleaning step is not required with these systems.
Disinfection
First, the endoscope and its accessories should be completely immersed into the disinfectant/sterilant. All the channels should be profused with the disinfectant. The air pockets should be removed for adequate contact of the sterilant with surfaces. The time of exposure, concentration, and temperature should be maintained as per the instruction. The disinfectants that should not be used for endoscopes are chlorine compounds, iodoforms, quaternary ammonium compounds, phenols, and alcohols. OPA for 12 min was found to be more advantageous than glutaraldehyde. Low-temperature sterilization can be achieved with EtO, but it is very lengthy, toxic, and expensive.
Rinse and Dry
After the treatment with the sterilant, the endoscopes should be thoroughly washed with sterile water/filtered water to remove all the residual chemicals. The scopes should be dried flushing 70–90% alcohol and forced air. Drying should be done after disinfection and before storage to reduce the contamination.
Store
The disinfected endoscopes should be dried, capped, and kept vertically for the prevention of contamination. If automated washer is used, regular maintenance and disinfection of automated washer should be done. Protocol should be developed to know whether the endoscopes are properly cleaned and disinfected or not. After the reprocessing, it can be used up to for 1/2 weeks with the maintenance of proper storage condition.
Risk of Nonendoscopic Transmission of Infection
There are many issues regarding nonendoscopic transmission of various infections.36 There were many outbreaks due to the mishandling of various parts of endoscopes [e.g., intravenous (IV) tubings, needles, or syringes]. There is no consensus on the microbiological finding especially when the numbers of isolated bacteria are very small/isolation of environmental contaminants like coagulase-negative Staphylococcus aureus or Bacillus sp. Alfa et al. have given <100 CFU/mL as the benchmark of a reprocessed endoscope channel. It has been also observed that pathogens like M. tuberculosis and P. aeruginosa are increasingly resistant to the commonly used HLD, i.e., glutaraldehyde. Now many rapid test methods such a strip detecting the residual protein, hemoglobin carbohydrate within the channels, and bioluminescence-based tests are available. Along with the new technology of HLDs such as improved hydrogen peroxide, improved automated endoscopic reprocessor are also introduced, e.g., EVOTECH (FDA cleared). The EVOTECH machine reduces the manual cleaning process. All the scopes are put inside it after cleaning, and it checks all the leaks, disinfects, and promotes drying by alcohol flush.
Processing of Other Scopes
Laparoscope is one of the popular equipment that enters into the sterile space. It is rigid, easy to clean, and disinfect. Hence the risk of transmission of infection is also found less. It is disinfected with the HLDs. The gastrointestinal endoscopes such as duodenoscopes are used in the ERCP process. They are commonly found to be associated with introduction of iatrogenic infection. All the gastrointestinal endoscopes must be reprocessed after every patient because of high bioburden. Since majority of them are heat sensitive, they should be sterilized with HPP or PAA. Similarly, bronchoscope should be reprocessed depending upon the material used. In general, the flexible bronchoscopes are sterilized with HPP or PAA, and rigid bronchoscopes are sterilized with steam sterilization. All other flexible scopes such as the thoracoscope, laryngoscope, sinuscope, and esophagoscope are sterilized with HLDs.
Specific Issues
Cleaning and Disinfecting Surface and Floors37, 38, 39, 40
In general, routine cleaning and disinfection of the hospital surface and floors should be done using EPA-registered hospital detergent/disinfectant. These procedures are very important in places like the ICU, OTs, and emergency rooms, where there is likely chance of getting contamination with the blood/other body fluids. Routine environmental disinfection remains crucial particularly in case of respiratory syncytial virus infection, severe acute respiratory syndrome complex, norovirus infection, clusters of Clostridium difficile or infection with MRSA. Disinfection should be done daily for all areas with patients in contact isolation (patients infected with MRSA). Norovirus infection is highly contagious, hence items contaminated with norovirus infection vomit should be immediately disinfected with concentrated bleach or oxygen-related compound is needed.
Inactivation of Emerging/Resistant Bacteria
Emerging pathogens and multidrug-resistant bacteria are one of the major concerns in medical facilities. Emergence of antibiotic resistance in certain bacteria, e.g., multidrug-resistant M. tuberculosis, MRSA, VRE, extended spectrum of β-lactamases, and metallo-β-lactamase producers are also important causes of various infection. Except prions, most of the aforementioned pathogens are susceptible to different sterilization and disinfection procedures (see list of FDA-approved chemicals in Table 59.7 ). Hence, standard sterilization methods and EPA-registered disinfectants should be used for all the equipment, devices, and surfaces irrespective of the patient status (known to be infected/not infected) to prevent the transmission of these infections. A sporicidal germicide such as glutaraldehyde/OPA/PAA/glutaraldehyde with phenol/H2O2 is required for the proper elimination of C. difficile spores.
Table 59.7.
Disinfectants Approved by the FDA for Processing Medical Devices14
| Chemical Sterilants (Sporicidal) Claiming Sterilization | ||
|---|---|---|
| Name of Disinfectant | Use-Dilution | Contact Time |
| Glutaraldehyde | ≥2.4% | 10 h at 20–25°C 7.5 h at 35°C |
| Glutaraldehyde with phenol/phenate | 1.12%/1.93%, 0.95%/1.64% | 12 h at 25°C |
| Hydrogen peroxide with peracetic acid | 7.35%/0.23%, 1.0%/0.08%, 8.3%/7% | 3 h at 20°C, 5 h at 25°C |
| Hydrogen peroxide | 7.5% | 6 h at 20°C |
| Peracetic acid | 0.2% | Only cleared for use with STERIS system, 12 min at 50–55°C |
| HLDs | ||
| Glutaraldehyde | ≥2.0% | 5 min at 35−37.8°C to 90 min at 25°C |
| Ortho-phthalaldehyde | 0.55% | 12 min at 20°C, 5 min at 50°C |
| 0.6% | 12 min at 20°C | |
| Hydrogen peroxide | 7.5% | 30 min at 20°C |
| Hydrogen peroxide and peracetic acid | 1.0%/0.08% | 25 min at 20°C |
| 7.35%/0.23% | 15 min at 20°C | |
| 8.3%/7% | 5 min at 20°C | |
| Hypochlorite and hypochlorous acid | 650–675 ppm | 10 min at 25°C |
| 400–450 ppm | 10 min at 30°C | |
| Glutaraldehyde and phenol/phenate | 1.121%/1.93% | 20 min at 25°C |
| Glutaraldehyde + isopropyl alcohol | 3.4%/26% | 10 min at 20°C |
FDA, US Food and Drug administration; HLD, high-level disinfectant.
Disinfection of Human Immunodeficiency Virus, Hepatitis B Virus, Hepatitis C Virus, and Tuberculosis-Contaminated Devices
All the equipment, devices, and surfaces should be treated in the same way as infected regardless of the whether the patient is known to be infected with HIV, HBV, HCV, or M. tuberculosis. These should be sterilized or disinfected with HLDs approved by the EPA or FDA (see Table 59.7).
Among the LLDs, isopropyl alcohol is one of the common disinfectants used for small surface areas at a concentration of 60–95%. It has no action on spores, but it is active against bacteria, viruses, fungi, and M. tuberculosis. Iodophore and phenolic compounds are ILDs, which are used to disinfect various surgical and medical instruments. The exposure time is about 10 min. They were found to be inactive against bacterial spores.
Special Precaution for Inactivation of Creutzfeldt–Jakob Disease13, 23, 29, 30
CJD has become a major crisis for the medical field because of lack of curative treatment. CJD is a highly infectious disease caused by the infection by prions (altered protein fragment). High-risk tissues include the neurological tissues such as the brain, spinal cord, and eye (retina, optic nerve). Prions are resistant to the routine sterilization methods (includes heat sterilization and chemical sterilants). There are special guidelines for the sterilization and decontamination of the instruments exposed to patients with CJD.
Risk assessment of the patient is done with the following criteria.
-
•
Known or suspected case of CJD or other transmissible spongiform encephalopathy
-
•
Unusual progressive dementia with myoclonus and ataxia
-
•
Family history of CJD/Gerstmann–Sträussler–Scheinker disease/fatal familial insomnia, dura matter transplant
-
•
Cadaver-derived pituitary hormone injection, etc.
All the critical and semicritical devices exposed to the aforementioned surfaces are considered as infective and should be disinfected with proper care. Sodium hydroxide (1N NaOH, 121°C for 30 min) along with steam sterilization is used. Instruments that do not tolerate this above method, must be cleaned twice, treated with various chemicals such as PAA, iodophors, 3% sodium dodecyl sulfate, or 6M urea and autoclaved at 121°C for 30 min. The noncritical items are difficult to clean and should be discarded.
Health Care–Associated Infections
Definition13, 41, 42
Hospital-acquired infection or nosocomial infection (NI) is defined as the localized or systemic infection acquired during the hospital care due to the adverse reactions to the presence of an infectious agent(s) or its toxin, which was neither present nor in incubating period during the time of admission. On the other hand, it can be defined as the infection that appeared after 48 h of admission to the hospital. Over 1.4 million people acquire infection from the hospital worldwide.43, 44 In developed countries, the incidence of NI varies from 5% to 25%.45, 46 The incidence of NI in pediatric ICU is estimated to be about 6.1–29.6%.14 Across the world, the incidence of NI (1%) was found lowest in the Netherlands.14
Nowadays, health care–associated infection (HAI) has become a major arm of NI, which indirectly reflects the colonized pathogens or exposure to variety of infectious agent through contaminated devices. The infection is usually mild, but sometimes it may become severe and life threatening. Majority of infection occur in the ICUs, nurseries, and surgical units. Additionally, with the increase in the length of stay, the treatment procedures become very expensive. In Europe, HAIs contribute approximately 16 million extra stays in the hospital and 37,000 deaths per year.47 The incidence of HAI varies from 5.1% to 11.6% in developing countries in comparison to 7.1% in Europe.47 Death rate varies from infection to infection. For example, HAI due to blood stream infection and ventilator-associated pneumonia increase the death rate as well as the extra cost due to hospital stay. It was estimated that death rate due to HAI is more than death rate due to AIDS, breast cancer, and road traffic accidents. HAI is considered as the eighth most common cause of death in the United States. The mortality rate also varies depending on the patient population and health care setting.
Common Health Care–Associated Infections48
HAIs can be transmitted by the medical devices or the unclean hands of the health care workers. The infection caused by devices (such as catheter, ventilator) are catheter-related blood stream infection (CR-BSI), catheter-associated urinary tract infection (CA-UTI), ventilator-associated pneumonia (VAP), etc. Infection due to C. difficile can be transmitted among the patients due to mal hand hygiene practice of the health care worker. The various bacterial pathogens causing HAI are MRSA, VRE, vancomycin-resistant Staphylococcus aureus, M. tuberculosis, Acinetobacter spp., P. aeruginosa, B. cepacia, C. difficile, Clostridium sordellii, Escherichia coli, and Klebsiella spp. Infections due to hepatitis virus, HIV, influenza virus, and norovirus are the common infections acquired in the hospital settings.
Infections in Operating Rooms and Intensive Care Units
Source
The infections in operating rooms (ORs) mainly appear as surgical site infections (SSIs). The route of entry of the pathogens can be either air born or by contact of the contaminated instruments and hands of health care provider.49, 50 The majority of SSIs occur due to transfer of bacteria from air to the wound and less frequently with the contact. The bacteria from the patient get into the air and later settle down on the surface of the floor/instruments/health care provider’s hand, etc. Ventilation of the OTs dramatically reduce the incidence of postoperative infections. It is also observed that the number of airborne infection increases with the number of members and movements inside the OT setup. The patients, in the ICU, have severe illness, impaired defense mechanism, and on the invasive devices (e.g., ventilators, IV catheter, urinary catheter).14 Extreme of age, multiple trauma, burn, and abdominal surgery are the commonest risk factors for the patients to acquire NI during their stay. Prolong hospitalization, immobilization, overcrowding, low patient–nurse ratio, and poor infection control practices enhance the infection rate in the ICUs.14 Common infections that occur in the ICUs are VAP, CR-BSI, CR-UTI, SSI, C. difficile–associated diarrhea, etc. Device-related infections are most common among all.
Measures to Reduce Health Care–Associated Infection
SSIs are the commonest HAI infections that occur in the OT due to faulty preventive protocols. These infections are difficult to treat and life threatening also. Hence, recommendation of guidelines and application of the methods are required to prevent SSIs in the ORs.
Design of Operating Room14
Designing the ORs is a complex task and should be done as per the recommendations. At least one anesthesiologist should be in the team while designing ORs. The aim is to give maximum benefit to the patients arriving to the ORs for various diagnostic, therapeutic, and palliative procedures. The ORs should be separated from the main hospitals. The floors and walls should be smooth and covered with antistatic material, so that lesser amount of dust particles can be absorbed. Broadly, the ORs can be described in three sections. From outer to inner disposal zone, clean zone, and aseptic zone. The innermost protective zone should have maximum negative pressure and least bacteriological count in comparison to other zones. The inner negative pressure allows the free flow of germ-free air from outside to inside.
-
1.
Outer/disposal zone: outer disposal area of the ORs.
-
2.
Clean zone: a semirestricted area contains store room, anesthetic room, recovery room, scrub room, and rest room for the staffs. The exit of the clear zone should be through the outer zone.
-
3.
Aseptic zone: it should be restricted to the working team. It includes the OT and the area for storage of sterile equipment.
Ventilation and Cleaning of the Operating Rooms
In the ORs, air flow system or ventilation removes the majority of airborne bacteria. ORs should be designed properly for adequate ventilation. The air flow system is composed of ventilation, direction of air flow, maintenance of pressure, air filtration, etc. Clean filtered air and adequate ventilation of indoor air are the two key factors in reduction of airborne infection. Ventilation helps in maintenance of the temperature and humidity. The OR’s temperature should be kept 1°C lesser than the outside and maintained in between 18 and 24°C. About 40–60% of relative humidity should be maintained inside the ORs for the comfort of the staff. Ventilation causes exchange of air, and thereby removes all the bacteria released from the patient during the operative procedure and dilutes the anesthetic gases from the ORs. The direction of air flow should remain from the ORs to the main corridor. It has been observed that turbulent air flow exchanges the air more efficiently than low-velocity unidirectional flow. The anesthetic gases should be removed other than the main exhaust. Filters with an efficiency of 20–40% are adequate at the entry side. HEPA filters, which can remove particles of size >0.3 μm with efficiency of >99.97% are used as the second-line filters near the ORs, ICUs, burn wards etc. Air from the HEPA filters flow unidirectionally (vertically or horizontally) causing few airborne bacteria at the operation site. Exponential laminar air flow has been designed to overcome the limitation. The recommended air change rates (ACHs) in ORs and recovery room are 20 ACH/h and 12–15 ACH/h, respectively. In ultraclean ventilated theaters, the minimum ACH should be 37 ACH/h.
Cleaning of the OT complex should be done either with vacuum or by wet mopping. Try to keep the surfaces and floors dry, when the room is in use. The equipment inside the room should be covered during the cleaning process. All the inanimate objects (like tables, chairs, trolleys, sink door handler, etc.) should be cleaned with an EPA-approved LLD detergent. Mopping should be regularly done at the beginning of each day before the OT process is going to start. The OT tables, trolley tops, lamps, etc., should be cleaned between use for each patient using LLD.
Microbiological Sampling
Routine bacteriological surveillance of ORs should be done for monitoring the presence of various airborne pathogens. The rate of microbial contamination in a well-designed, properly filtered, ventilated, and disinfected OT is much less. In conventionally ventilated theaters, microbiological sampling is done in the following way. Close all the doors of the ORs and keep empty for 1 h. Using an air sampler, >250 L [recommended amount is 1 m3 air (100 L)] air should be exposed to the no-selective culture medias. At least two samples should be taken per OR. The colony count should not exceed 50–150 CFU/m3. As per the UK guidelines, with a 5-min exposure, the standard bacterial count should not exceed 35 bacterial and/or fungal particles per cubic metre of ventilating air. Similarly, the Geneva guidelines takes the standard of 25 CFU/m3 for an empty OR and 180 CFU/m3 for 5-min exposure. The working condition of HEPA filters should be checked first. In properly functioning HEPA filters, the amount of airborne microbial contamination is very low. In unidirectional air flow zone, microbiological sampling is done from the four corners of the perimeter zone, one from the center and four from the inner corners of the inner zone. This zone should not contain more than 0.5 CFU/m3.
Health Care–Associated Infections in the Intensive Care Units and Its Prevention
NIs or HAIs are a major cause of morbidity and mortality in the medical setup. The patients in the ICUs are observed to be more ill, receiving multiple high antibiotics, on multiple devices, and exposed to multiple antibiotic-resistant colonizers. The incidence of HAIs is found to be five to six times higher in the ICUs than in the general wards. The overall rate of ICU infection varies from 5% to 35% of which approximately 25% are hospital-acquired infections. The most common infections observed in the ICU setup are ventilator-associated infection (VAP), CR-BSI, CA-UTI, SSI, C. difficile-associated diarrheas, etc. Among these infections device-related infection (VAP, CA-BSI, CA-UTI) constitute 80%. Various risk factors for the development of infections are divided into host-related and hospital-related factors. The host-related factors include severity of diseases, extreme of ages, immunocompromised conditions (malignancy/transplant/organ failure/HIV infection), burn injury, trauma, and extensive surgeries. The factors related to the hospital or treatment are device related, treatment on immunosuppressive drugs, multiple blood transfusion, hemodialysis, parenteral nutrition, prolong immobilization, etc. Since most of the infections are caused by the colonizers, the common pathogens isolated in these infections are MRSA, VRE, ESBL-producing gram-negative bacteria, Stenotrophomonas maltophilia, fluconazole-resistant Candida spp., etc.
HAIs in the ICUs can be prevented by the application of recommended preventive measures. The HAI rate has been reduced to one-third with proper applicability of infection control programs. One should have adequate knowledge about the risk factors, source of infection, type, and causation of infections to plan and establish the preventive strategies.
Bundle Approach for Reducing Infection
Bundle approach is an evidence-based group approach of preventive measures, which is found to be more effective when executed together. Studies confirm that combined interventions result in better outcome than individual. These bundle approaches are small (three to five), straightforward practices performed in groups. These are mainly applied for the NIs such as CR-BSI, VAP, CA-UTI, and SSI. However, the rate of reduction of these infections also depends on the baseline rate of infection of that health care center, staff adherence to the tactics, and the preventive measures chosen for the bundle.
CR-BSI is an important cause of mortality and morbidity in patients admitted to the ICUs. The number increases with the increase in handling, duration of insertion, number of manipulation, number of lumens, etc. Studies have proved that a large proportion of infections are preventable by applying the various control measures. All the elements of the bundle must be executed at the same point of time. These include hand washing, full-barrier precaution during the insertion of central line catheter, cleaning of the skin with chlorhexidine solution every time, frequent infusion of heparin or heparinlike substance through total parenteral nutrition to prevent the formation of fibrin, removal of unnecessary catheters, and avoidance femoral site catheterization.
VAP is another important cause of ICU-acquired infection. It not only causes mortality but also is responsible for substantial cost of treatment. Various recommendations for the prevention of VAP include (1) appropriate cleaning, disinfection, and sterilization of ventilator equipment, (2) maintenance of ventilator circuits, and (3) routine care of patients requiring ventilation.14 The cleaning, disinfection, and sterilization of the equipment have been discussed earlier in the chapter. The preventive measures taken for the patients to reduce the rate of VAP are use of orotracheal intubation, noninvasive ventilation, minimization of the duration of ventilation. The patient should be maintained in semirecumbent position (30–45 degrees elevation of head of the bed). Regular oral care should be performed. CA-UTI is another common cause of infection in the ICUs14. Staff must be trained enough to differentiate asymptomatic bacteriuria from infection due to catheterization. Regular education regarding repeated hand washing before and after catheterization, maintenance of a closed system, maintaining free urine flow, and securing the catheter position correctly should be given to the health care workers for the prevention of these infections. SSI is a very common and life-threatening infection during ICU stays.14 There are various evidence-based guidelines recommended by the CDC. Preoperative patient preparation, management of the colonizers, correct surgical technique, adequate antimicrobial prophylaxis, adequate sterilization and disinfection of the environmental surfaces, proper aseptic techniques, as well as washing surgical hands before and after the operative procedure are various preventive measures that can be taken to reduce the incidence of SSIs.
About one-third of the infections in the ICUs can be prevented by implementation of recommended preventive measures. Apart from education of the health care personnel and standard precaution measures, other precautions like contact precaution, airborne precaution, and droplet precaution should be taken to prevent various infections. Antibiotic-associated diarrhea due to C. difficile, and infection due to E. coli (O157:H7), Shigella spp., hepatitis A, rotavirus, etc., can be transmitted by contact with infected patients or items. Similarly infections transmitted by droplet such as tuberculosis, varicella, measles, viral hemorrhagic fever, and influenza can be prevented by taking adequate precaution. Use of antibiotics should be restricted in the ICU setup. Minimum precautions like heating the water before use, regular cleaning, and maintenance of water tanks can be done to prevent water-borne infections.
Conclusion
To conclude, sterilization, disinfection, and cleaning are the mainstay of hospital infection control activities. The selection and application of different methods should be done as per the recommendation and according to the manufacturer’s instruction. Hand hygiene remains the cornerstone of all infection control activities.
References
- 1.Sopwith W., Hart T., Garner P. Preventing infections from reusable medical equipment: a systematic review. BMC Infect Dis. 2002;2:4. doi: 10.1186/1471-2334-2-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rutala W.A., Waber D.J. New disinfection and sterilization methods. Emerg Infect Dis. 2001;7(2):348–353. doi: 10.3201/eid0702.010241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Biron F., Verrier B., Peyramond D. Transmission of the human immunodeficiency virus and hepatitis C virus. N Engl J Med. 1997;337:348–349. doi: 10.1056/NEJM199707313370515. [DOI] [PubMed] [Google Scholar]
- 4.Gillespie T.G., Hogg L., Budge E., Duncan A., Coia J.E. Mycobacterium chelonae isolated from rinse water within an endoscope washer-disinfector. J Hosp Infect. 2000;45:332–334. doi: 10.1053/jhin.2000.0782. [DOI] [PubMed] [Google Scholar]
- 5.Schelenz S., French G. An outbreak of multidrug-resistant Pseudomonas aeruginosa infection associated with contamination of bronchoscopes and an endoscope washer-disinfector. J Hosp Infect. 2000;46:23–30. doi: 10.1053/jhin.2000.0800. [DOI] [PubMed] [Google Scholar]
- 6.Spach D.H., Silverstein F.E., Stamm W.E. Transmission of infection by gastrointestinal endoscopy and bronchoscopy. Ann Intern Med. 1993;118:117–128. doi: 10.7326/0003-4819-118-2-199301150-00008. [DOI] [PubMed] [Google Scholar]
- 7.Srinivasan A., Wolfenden L.L., Song K., Mackie T., Hartsell L., Jones H.D. An outbreak of Pseudomonas aeruginosa infections associated with flexible bronchoscopes. N Engl J Med. 2003;348:221–227. doi: 10.1056/NEJMoa021808. [DOI] [PubMed] [Google Scholar]
- 8.Rutala W.A., Weber D.J. Guideline for disinfection and sterilization of prion-contaminated medical instruments. Infect Control Hosp Epidemiol. 2010;31:107–117. doi: 10.1086/650197. [DOI] [PubMed] [Google Scholar]
- 9.Belay E.D., Schonberger L.B. The public health impact of prion diseases. Annu Rev Public Health. 2005;26:191–212. doi: 10.1146/annurev.publhealth.26.021304.144536. [DOI] [PubMed] [Google Scholar]
- 10.Collins S.J., Lawson V.A., Masters C.L. Transmissible spongiform encephalopathies. Lancet. 2004;363:51–61. doi: 10.1016/S0140-6736(03)15171-9. [DOI] [PubMed] [Google Scholar]
- 11.Weissmann C. Birth of a prion: spontaneous generation revisited. Cell. 2005;122:165–168. doi: 10.1016/j.cell.2005.07.001. [DOI] [PubMed] [Google Scholar]
- 12.Spaulding E.H. Chemical disinfection of medical and surgical materials. In: Lawrence C., Block S.S., editors. Disinfection, sterilization, and preservation. Lea & Febiger; Philadelphia (PA): 1968. pp. 517–531. [Google Scholar]
- 13.Favero M.S., Bond W.W. Chemical disinfection of medical and surgical materials. In: Block S.S., editor. Disinfection, sterilization, and preservation. 5th ed. Lippicott Williams & Wilkins; Philadelphia (PA): 2001. pp. 881–917. [Google Scholar]
- 14.Mathur P. 1st ed. Lippicott Williams & Wilkins; Philadelphia (PA): 2010. Hospital acquired infections prevention and control. [Google Scholar]
- 15.Ratula W.A., Weber J.A. CDC; Atlanta (GA): 2008. The healthcare infection control practices advisory committee (HICPAC). Guidelines for disinfection and sterilization in health care facilities. [Google Scholar]
- 16.CDC . U.S. Department of Health and Human services, CDC; Atlanta (GA): 2003. Guidelines for environmental infection control in health-care facilities. Recommendations of CDC and the healthcare infection control practices advisory committee (HICPAC) [PubMed] [Google Scholar]
- 17.WHO . WHO; Geneva (Switzerland): 2002. Guidelines on prevention and control of hospital associated infections. South East Asian Region. [Google Scholar]
- 18.Occupational safety and health administration Occupational exposure to bloodborne pathogens: final rule. Fed Regist. 1991;56:64003–64182. [PubMed] [Google Scholar]
- 19.Centres for Disease Control Recommendations for prevention of HIV transmission in health-care settings. Morb Mort Wkly Rep. 1987;36:S3–S18. [Google Scholar]
- 20.Environmental protection agency. EPA list U.S. Lists A, B, C, D, E, ad F: EPA registered disinfectants, sanitizers and sterilants. Available at: http://WWW.Epa.gov/oppad001/chemregindex.htm.
- 21.Rutala W.A., Weber D.J. Disinfection and sterilization in health care facilities: what clinicians need to know. Clin Infect Dis. 2004;39:702–709. doi: 10.1086/423182. [DOI] [PubMed] [Google Scholar]
- 22.WHO . WHO; Geneva (Switzerland): 2003. Practical guidelines for infection control in health care facilities. [Google Scholar]
- 23.Laboratory centres for the Disease control Bureau of infectious Diseases . Laboratory centre for Disease control Bureau of Infectious Diseases; Ottawa (Canada): 1998. Infection control guidelines. Communicable disease report. [Google Scholar]
- 24.Widmer A.F., Frei R. Decontamination, disinfection and sterilization. In: Pealler M.A., editor. the clinical microbiology laboratory in infection detection, prevention and control. 3rd ed. 2001. [Google Scholar]
- 25.Ayeliff G.A.J., Fraise A.P., Geddes A.M., Michell K. 4th ed. Arnold; New York: 2000. Control of hospital infection. [Google Scholar]
- 26.Roberts C.G. Studies on the bioburden on medical devices and the importance of cleaning. In: Rutala W.A., editor. Disinfection, sterilization and antisepsis: principles and practices in healthcare facilities. Association for Professional in Infection Control and Epidemiology; Washington (DC): 2001. pp. 63–69. [Google Scholar]
- 27.Hutchisson B., LeBlanc C. The truth and consequences of enzymatic detergents. Gastroenterol Nurs. 2005;28:372–376. doi: 10.1097/00001610-200509000-00003. [DOI] [PubMed] [Google Scholar]
- 28.Alfa M.J., Jackson M.A. A new hydrogen peroxide-based medical-device detergent with germicidal properties: comparison with enzymatic cleaners. Am J Infect Control. 2001;29:168–177. doi: 10.1067/mic.2001.113616. [DOI] [PubMed] [Google Scholar]
- 29.Communicable Diseases Network Austrelia, the National Public Health Partnership and Austrelian Health Ministers’ Advisory Council . Communicable Diseases Network Australia, The National Public Health Partnership and Austrelian Health, Minister’s Advisory Council; Canberra (Austrelia): January 2004. Infection control guidelines for the prevention of transmission of infectious diseases in the healthcare setting. [Google Scholar]
- 30.Rutala W.A. APIC guideline for selection and use of disinfectants. AJIC Am J Infect Control. 1996;24:313–342. doi: 10.1016/s0196-6553(96)90066-8. [DOI] [PubMed] [Google Scholar]
- 31.Schembre D.B. Infectious complications associated with gastrointestinal endoscopy. Gastrointest Endosc Clin N Am. 2000;10:215–232. [PubMed] [Google Scholar]
- 32.Nelson D.B., Muscarella L.F. Current issues in endoscope reprocessing and infection control during gastrointestinal endoscopy. World J Gastroenterol. 2006;12:3953–3964. doi: 10.3748/wjg.v12.i25.3953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Nelson D.B., Jarvis W.R., Rutala W.A., Foxx-Orenstein A.E., Isenberg G., Dash G.R. Multi society guideline for reprocessing flexible endoscopes. Society for health care epidemiology of America. Infect Control Hosp Epidemiol. 2003;24:532–537. doi: 10.1086/502237. [DOI] [PubMed] [Google Scholar]
- 34.Culver D.A., Gordon S.M., Meheta A.C. Infection control in the bronchoscopy suite. Am J Resp Crit Care Med. 2003;24:532–537. doi: 10.1164/rccm.200208-797CC. [DOI] [PubMed] [Google Scholar]
- 35.MHRA. Medicine and health care products manual. Available at: http://mhra.gov.uk/home/idcplg.
- 36.Rutal W.A., Weber D.J. New developments in reprocessing semicritical items. Am J Infect Control. 2013;41:S60–S66. doi: 10.1016/j.ajic.2012.09.028. [DOI] [PubMed] [Google Scholar]
- 37.Dancer S.J. Importance of the environment in the methicillin-resistant Staphylococcus aureus acquisition: the case for hospital cleaning. Lancet Infect Dis. 2008;8:101–103. doi: 10.1016/S1473-3099(07)70241-4. [DOI] [PubMed] [Google Scholar]
- 38.Gamage B., Moore D., Copes R., Yassi A., Bryce E. Protecting HCWs from SARS and other respiratory pathogens: a review of the infection control literature. Am J Infect. 2005;33:114–121. doi: 10.1016/j.ajic.2004.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hall C.B., Douglas R.G. Modes of transmission of respiratory syncytial virus. J Pediatr. 1981;99:100–103. doi: 10.1016/s0022-3476(81)80969-9. [DOI] [PubMed] [Google Scholar]
- 40.Khanna N., Goldenberger D., Graber P., Battegay M., Widmer A.F. Gastroenteritis outbreak with norovirus in a Swiss university hospital with a newly identified virus strain. Lancet. 2003;55:131–136. doi: 10.1016/s0195-6701(03)00257-3. [DOI] [PubMed] [Google Scholar]
- 41.Garner J.S., Jarvis W.R., Emori T.G., Horan T.C., Hughes J.M. CDC definitions for nosocomial infections. Am J Infect Control. 1998;16:128–140. doi: 10.1016/0196-6553(88)90053-3. [DOI] [PubMed] [Google Scholar]
- 42.CDC . 2004. CDC definitions for nosocomial infections.http://www.ncbi.nlm.nih.gov/pubmed/2841893 Available at: [Google Scholar]
- 43.WHO . WHO; Geneva: 2002. Guidelines on prevention and control of hospital associated infections. World Health Organization. South East Asian Region. [Google Scholar]
- 44.Weinstein R.A. Nosocomial infection update. Emerg Infect Dis. 1998;4:416–420. doi: 10.3201/eid0403.980320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.WHO . WHO; Geneva (Switzerland): 2005. World Alliance for patient safety. The global patient safety challenge 2005–2006 ’Clean care is safer care’.http//www.who.int/gpsc/en/ Available at: [Google Scholar]
- 46.Vincent J.L. Nosocomial infections in adult intensive care units. Lancet. 2003;361:2068–2077. doi: 10.1016/S0140-6736(03)13644-6. [DOI] [PubMed] [Google Scholar]
- 47.Jakab Z. V International conference on patient’s safety, Health care associated infection and antimicrobial resistance.Spain. June 2010. Prevention of health care-associated-infections (HAI) and antimicrobial resistance (AMR) in Europe. pdf. [Google Scholar]
- 48.Vincent J.L., Bihari D.J., Suter P.M., Bruining H.A., White J., Nicholas-Chanion M.H. The prevalence of nosocomial infection in intensive care units in Europe. Results of the European Prevalence of Infection in Intensive care (EPIC) study. EPIC International Nosocomial Infection Control Committee. JAMA. 1995;274:639–644. [PubMed] [Google Scholar]
- 49.Whyte W., Hambraeus A., Laurell G., Hoborn J. The relative importance of routes and sources of wound contamination during general surgery. II. Nonairborne. J Hosp Infect. 1991;18:93–107. doi: 10.1016/0195-6701(91)90154-z. [DOI] [PubMed] [Google Scholar]
- 50.Whyte W., Hambraeus A., Laurell G., Hoborn J. The relative importance of routes and sources of wound contamination during general surgery. II. Airborne. J Hosp Infect. 1992;22:41–54. doi: 10.1016/0195-6701(92)90129-a. [DOI] [PubMed] [Google Scholar]


