Abstract
Charcot-Marie-Tooth (CMT) disease is an inherited peripheral motor and sensory neuropathy. The disease is divided into demyelinating (CMT1) and axonal (CMT2) neuropathies, and although we have gained molecular information into the details of CMT1 pathology, much less is known about CMT2. Due to its clinical and genetic heterogeneity, coupled with a lack of animal models, common underlying mechanisms remain elusive. In order to gain an understanding of the normal function of genes associated with CMT2, and to draw direct comparisons between them, we have studied the behavioural, cellular and molecular consequences of mutating nine different genes in the nematode Caenorhabditis elegans (lin-41/TRIM2, dyn-1/DNM2, unc-116/KIF5A, fzo-1/MFN2, osm-9/TRPV4, cua-1/ATP7A, hsp-25/HSPB1, hint-1/HINT1, nep-2/MME). We show that C. elegans defective for these genes display debilitated movement in crawling and swimming assays. Severe morphological defects in cholinergic motors neurons are also evident in two of the mutants (dyn-1 and unc-116). Furthermore, we establish methods for quantifying muscle morphology and use these to demonstrate that loss of muscle structure occurs in the majority of mutants studied. Finally, using electrophysiological recordings of neuromuscular junction (NMJ) activity, we uncover reductions in spontaneous postsynaptic current frequency in lin-41, dyn-1, unc-116 and fzo-1 mutants. By comparing the consequences of mutating numerous CMT2-related genes, this study reveals common deficits in muscle structure and function, as well as NMJ signalling when these genes are disrupted.
Introduction
First identified in 1886, Charcot-Marie-Tooth (CMT) disease is the most common inherited peripheral neuropathy with an estimated prevalence of 1 in 2,500 individuals [1, 2]. Common clinical features of CMT include slowly progressive distal muscle weakness and atrophy, distal sensory impairment, foot deformities, secondary steppage gait and mobility impairment [1, 3]. There is currently no cure for CMT, and patients frequently suffer lifelong disabilities. While CMT can be inherited in an autosomal dominant, autosomal recessive or X-linked manner, it is predominantly (~90% of cases) inherited autosomal-dominantly [4]. The disease is typically divided into two main categories: type 1 CMT (CMT1), also known as demyelinating neuropathy, affects the myelin sheath surrounding the axon resulting in decreased nerve conduction velocities (less than 38 m/s); whereas axonal Type 2 CMT (CMT2) affects the nerve axon and normally does not affect the speed of nerve conduction [1, 4, 5].
CMT subtype 2A (CMT2A) is the most prevalent among CMT2 and accounts for approximately 20% of CMT2 cases [6]. However, more than 20 genetic loci have been associated with CMT2, with each gene encoding a different protein [7]. These proteins are associated with an eclectic mix of cellular functions. For instance, the causative gene for CMT2A, MFN2, encodes mitofusin 2, a GTPase that regulates outer mitochondrial membrane fusion [8, 9]. Conversely, HSPB1, the gene involved in CMT2F, encodes a heat shock protein with molecular chaperone functions [10]. Other genes are involved in protein translation (GARS, AARS, MARS, HARS), osmotic regulation (TRPV4), signalling pathways and cell adhesion (LRSAM1), neuroprotection (TRIM2), and sodium/potassium-transport (ATP1A1). The age and mechanisms of onset, disease progression and severity also vary from one CMT2 subtype to another, and from patient to patient within the same subtype [7]. Due to its genetic and clinical heterogeneity coupled with the relative neoteric discovery of causal genes, it remains to be determined how mutations in a vast range of proteins with seemingly distinct functions all lead to CMT2. This has hindered the development of therapeutics for the disease, as has the relative scarcity of CMT2 animal models [6].
C. elegans has emerged as one of the most widely used animal model systems to address questions regarding cellular and molecular aspects of human disease in vivo. It is also one of the few available animal models that are suitable for conducting cost-effective, rapid drug screening due to its ease of culturing, small body size (~1 mm in length), high brood size (~300 offspring), and short generation time (~3 days to adulthood) [11]. Furthermore, its conserved musculature features, transparent body and ease of genetic manipulation make C. elegans a highly desirable model organism to study cellular morphology and human diseases [12]. Approximately 60–80% of human genes have an orthologue in the C. elegans genome, and 42% of these genes are disease-related [11]. While C. elegans is an established model to study other neurodegenerative diseases such as amyotrophic lateral sclerosis, Alzheimer’s disease, Parkinson’s disease, Huntington’s disease and spinal muscular atrophy, only a handful of the many CMT subtypes have been studied in the nematode [11, 13–17].
To advance our understanding of the function of genes associated with CMT2, we have used C. elegans to assess the cellular and behavioural consequences of loss-of-function or null mutations in nine orthologous/paralogous genes associated with CMT2. We selected the closest C. elegans orthologues of the human genes based on sequence homology and the information available for each gene. Specifically, we studied crawling and swimming behaviour, both individually and within populations, examined the morphology and function of cholinergic motor neurons and body wall muscles, and analyzed neuromuscular junction transmission in these animals. We have also devised methodology for quantitatively measuring muscle cell area and the length of muscle filaments in order to non-subjectively determine and compare the extent of muscle defects. Our study reveals common deficiencies in muscle structure and function, and consequent locomotion difficulties amongst our mutants. Strikingly, our study also reveals shared deficits in cholinergic neurons, as well as a prevailing site of dysfunction at the neuromuscular junction in the genes associated with the most severe movement and muscle phenotypes. Using C. elegans for comprehensive analyses of the orthologous genes involved in nine different CMT2 subtypes, our study reveals novel insights into the functions of the CMT2-related genes, identifies common and diverse sites of defects when the genes are mutated, and paves the way for future studies aimed at defining the precise function of these genes.
Results
Individual CMT2-associated mutants exhibit movement defects
The genes associated with CMT2 investigated in this study, along with their encoded proteins, are listed in Table 1 and S1 Table. In addition to the genetic and clinical heterogeneity, our understanding of CMT2 is further complicated by numerous different alleles of the same gene being associated with each subtype. For example, more than 100 different mutations in the MFN2 gene have been identified in CMT2A patients [18]. Moreover, these individual mutations affect the encoded mitofusin 2 protein in different ways: some have no effect on protein function, some are gain-of-function, and others are loss-of-function that block its function in mitochondrial fusion [19, 20]. To expand our global understanding of the cellular roles for genes implicated in CMT2, instead of generating strains that carry individual patient-specific mutations, we have assessed the consequences of genetic recessive null or loss-of-function alleles. As defective locomotion is a major symptom in CMT2 patients, we first sought to test whether loss-of-function alleles in the orthologous genes in C. elegans would result in similar behavioural phenotypes. We explored two different forms of locomotion displayed by C. elegans: swimming in liquid and crawling on solid media. Both of these activities involve molecular mechanisms conserved with humans, including depletion of energy, muscle fatigue and increased mitochondrial oxidation in muscle cells [12].
Table 1. List of genes associated with CMT2 explored in this study.
| C. elegans gene | Human gene | CMT subtype | Predominant mode of inheritance [7] |
|---|---|---|---|
| lin-41 (ma104) | TRIM2 | CMT2R | Autosomal recessive |
| dyn-1(ky51) | DNM2 | CMT2M | Autosomal dominant |
| unc-116(e2310) | KIF5A | Yet to be classified | Autosomal dominant |
| fzo-1(cjn020) | MFN2 | CMT2A | Autosomal dominant, autosomal recessive |
| osm-9(ok1677) | TRPV4 | CMT2C | Autosomal dominant |
| cua-1(gk107) | ATP7A | Yet to be classified | X-linked |
| hsp-25(tm700) | HSPB1 | CMT2F | Autosomal dominant |
| hint-1(ok972) | HINT1 | Yet to be classified | Autosomal recessive |
| nep-2(ok2846) | MME | CMT2T | Autosomal recessive |
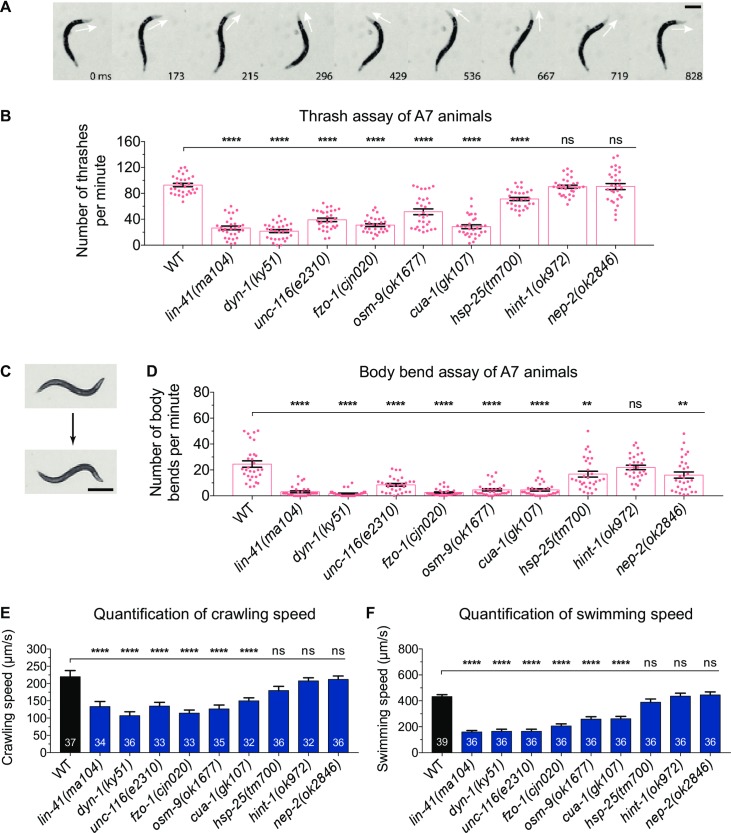
To assess swimming behaviour, we performed thrash assays. Single worms were placed in liquid and the number of thrashes calculated per minute, with a single thrash defined as a complete cycle of maximum bend on one side of the body (Fig 1A). We quantified thrash rates in four synchronized ages: the first larval stage 1 (L1), the final larval stage 4 (L4), 3-day old adult (A3) and 7-day old adult (A7). Mutations in seven of the nine CMT2-associated genes led to progressive deteriorations in locomotion, especially in the older animals (Fig 1B and S1A Fig). Interestingly, the onset of the defect varied between different genetic mutations. Worms carrying mutations in dyn-1, unc-116 or cua-1 presented lower thrash rates across all ages tested compared to wild-type, whereas the swimming ability of lin-41(ma104) and osm-9(ok1677) mutants were only significantly reduced in adult animals. Similarly, adult worms lacking functional FZO-1 also displayed significantly decreased thrash rates. However, the younger L4-stage worms were not different to the wild-type, even though L1 fzo-1(cjn020) worms were found to be defective. The age-dependent trend was also absent in hsp-25(tm700) animals, as only the L4 and A7 worms had significantly lower thrash rates. Importantly, introduction of the wild-type version of each gene into the corresponding mutant strain led to a significant rescue of the defect in all seven strains displaying a reduction in thrash rates (S2A Fig). Surprisingly, mutation of hint-1 or nep-2 did not reduce the thrash rates at any age tested. The hint-1(ok972) allele deletes the entire hint-1 gene, while the nep-2(ok2846) allele is a 362 bp deletion across the second and third exons of nep-2. Thus, although we expect both to strongly affect gene function, our analyses suggest that neither hint-1 nor nep-2 is required for normal swimming behaviour in C. elegans.
Fig 1. Motility assays in C. elegans mutants.
(A) An animal performing a single thrash in liquid culture. The head begins facing dorsal, moves ventral and then returns to dorsal. Arrows points to direction of the head; time in milliseconds displayed at bottom relative to the first image. Scale bar represents 250 µm. (B) Number of thrashes per minute calculated for 7-day old adult (A7) N2 wild-type (WT) and the nine mutant strains shown. See S1A Fig for thrash assays performed across different ages. (C) An animal undergoing one complete body bend on nematode growth media (NGM) agar. One body bend is defined as the maximum bending of the section behind the pharynx in the opposite direction, from top to bottom picture. Scale bar represents 500 µm. (D) Quantification of number of body bends of WT and CMT2 mutant A7 animals crawling on solid NGM agar. Number of body bends across different ages is included in S1B Fig. Each dot in (B) and (D) represents a single animal (n ≥ 30). (E) The rates of crawling on solid NGM agar and, (F) swimming in M9 liquid for 3-day old adult WT and nine CMT2 mutant worms. The swimming and crawling velocity were calculated using WormLab software; n values are within each bar. One-way ANOVA with Dunnett’s post hoc tests were used to compare swimming and crawling behaviour between WT and mutant animals in (B), (D), (E) and (F). Data is represented as mean ± S.E.M. **P < 0.01, ***P < 0.001, ****P < 0.0001, ns = not significant.
While swimming is an energy-demanding exercise, worms crawling on a solid surface face greater mechanical resistance than those swimming in liquid [12]. To quantify crawling capacity, we manually counted the number of body bends of individual worms crawling on NGM agar over a three-minute time frame (Fig 1C and 1D). Similar to the swimming deficiencies, all but one of the mutant strains displayed a considerably lower number of body bends over time compared to wild-type (Fig 1D and S1B Fig). Mutation of dyn-1 or fzo-1, led to the largest reductions in crawling rate, with younger worms presenting 60–80% reductions and older worms registering an average of just 2 body bends per minute compared to 25 in the wild-type. Unlike thrashing, the crawling ability of lin-41(ma104) mutants was impaired from birth, and drastically worsened in adulthood. In contrast, while unc-116(e2310), osm-9(ok1677) and cua-1(gk107) worms experienced significantly lower body bending rates across all ages, the decline was more gradual. Mutations in both hint-1 and nep-2 again had minimal effects on animal movement, although a significant reduction was observed in nep-2(ok2846) mutants at the oldest age analyzed. Importantly, this reduction in nep-2 mutants was rescued by introducing the wild-type version of the gene into these animals (S2B Fig). This indicates that while nep-2 may not be important for swimming behaviour, the same cannot be said for crawling behaviour on solid agar.
Next, we quantified swimming and crawling speeds using an automated worm tracking program (WormLab), which we used to simultaneously track and analyze multiple worms. We focused on A3 worms as the locomotion defect of most mutants became prominent at this age. Similar to the thrash and body bend assays, the majority of mutants displayed at least 30–40% declines in swimming and crawling speeds compared to wild-type worms (Fig 1E and 1F). Overall, these data establish that C. elegans carrying loss-of-function mutations in CMT2-associated genes experience defects in locomotion.
Populations of CMT2-associated mutants experience declines in motility over time
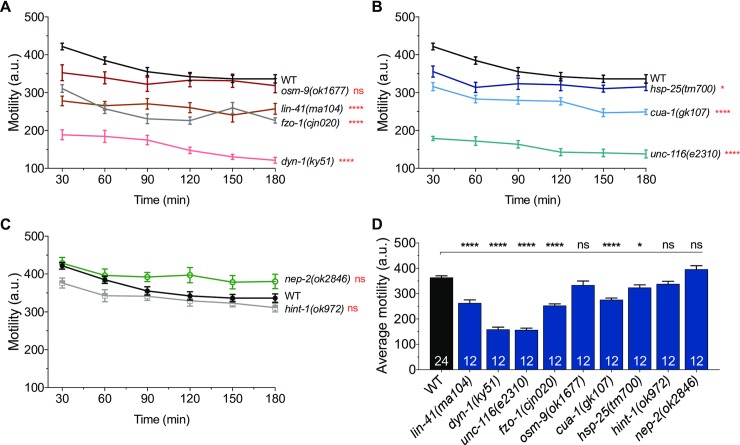
We next investigated whether locomotor defects were also observable on a population level. We recorded the motility of 30–50 worms from each group for three hours using an automated movement detection instrument (WMicrotracker). The WMicrotracker system works by detecting the interference of infrared microbeams as a result of animal movement [21]. Beam disruption is registered and converted into an activity count. To obtain consistently reproducible data, we optimized methods to generate synchronized populations of worms (S3 Fig, see Methods). During this optimization, we found that having 30–50 worms per well was optimal to prevent overcrowding, which we observed to inversely affect motility.
From the WMicrotracker screens, the wild-type populations of worms started with a motility count of about 420 arbitrary units (au), but slowly became less motile, reaching a plateau of around 330 au over the final hour of analysis (Fig 2A–2C). The average motility of wild-type worms during the 3-hour experiments was 365 au (Fig 2D). In stark contrast, both dyn-1(ky51) and unc-116(e2310) mutants consistently recorded motility counts below 200 au (Fig 2A, 2B and 2D). Populations of lin-41(ma104), fzo-1(cjn020) and cua-1(gk107) animals also experienced significantly reduced movement, registering between 220 and 330 au throughout the experiments (Fig 2A, 2B and 2D). Worms lacking hsp-25 had a slight (~12%), but statistically significant reduction in motility, whereas mutation of osm-9, hint-1 or nep-2 had no effect on movement (Fig 2A–2D). In summary, these population-based motility assays revealed comparable movement defects to what was observed with individual worms in our previous locomotion assays. These automated, time-effective WMicrotracker assays could prove highly advantageous for drug screening approaches to identify compounds that are able to reverse the movement defects in these animals.
Fig 2. High-throughput analysis of swimming behaviour in animal populations.
(A-C) Activity count graphs from WMicrotracker experiments. Motility of swimming animals (30–50 animals per well, 6 wells per genotype per experiment) was measured over a 3 h period in duplicate runs (n = 12 wells). Locomotor activity was compared between wild-type (WT) animals and the mutant strains indicated. (D) Summary and comparison of the average activity count for individual genotypes. The number of wells analyzed is reported in each bar. Data is represented as mean ± S.E.M. *P < 0.05, ****P < 0.0001, ns = not significant from one-way ANOVA with Dunnett’s post hoc tests; a.u. arbitrary units.
Loss-of-function mutations in CMT2-related genes results in defective cholinergic motor neuron morphology
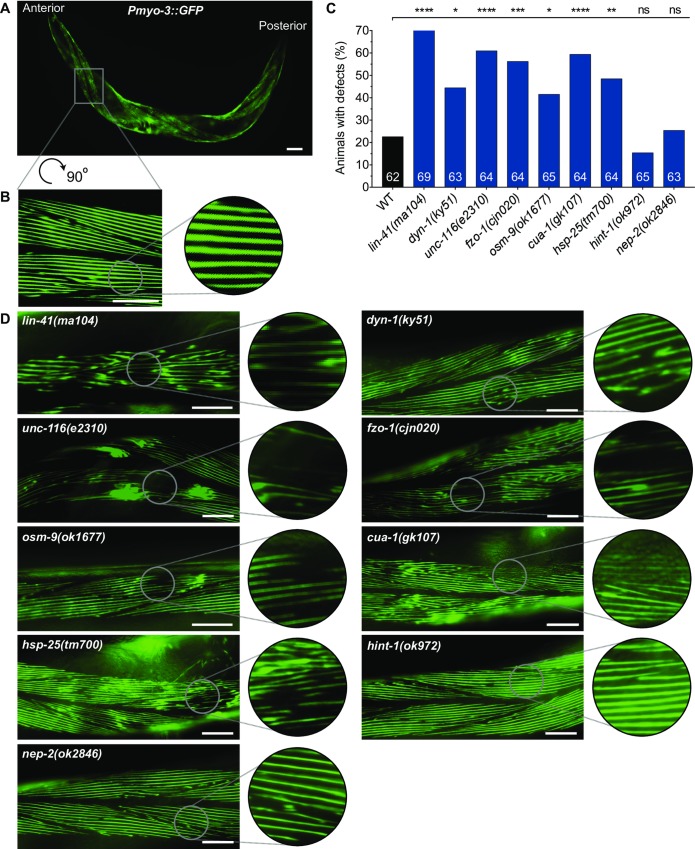
Degeneration of peripheral axons of motor neurons is one of the key features of CMT2. In mammals, these neurons are cholinergic in nature and extend axons from the spinal cord to the periphery where they innervate skeletal muscle. In C. elegans, there are 17 classes of cholinergic motor neurons, where four of these, VA, VB, DA and DB, are known to innervate body wall muscles that are essential for locomotion [22, 23]. In this study, we focused on the axonal morphology of C. elegans cholinergic motor neurons, DA and DB, which innervate dorsal wall muscles. Unfortunately, we could not accurately identify VA and VB neurons due to their overlapping positions with the other cholinergic neurons on the ventral side of the worm. We examined the commissures of DA and DB neurons in laterally-positioned A3 stage wild-type and CMT2 mutant worms using a cholinergic-specific, green fluorescent protein (GFP)-expressing transgene [vsIs48(Punc-17::GFP)] (Fig 3A). Worms were qualified as defective or not based on the presence of one or more specific defects in the DA or DB neurons: absent commissure (missing), axonal guidance defect (guidance), visible breakage along the commissure (break), and/or short neurite extending less than half the body width (short) (Fig 3B).
Fig 3. Morphological analysis of cholinergic motor neurons.

(A) Full body image of a 3-day old adult animal carrying the vsIs48(Punc-17::gfp) transgene, which produces GFP expression in all cholinergic neurons. VNC = ventral nerve cord. (B) Images (top) and schematics (bottom) of specific morphological phenotypes: absent commissure (missing), wayward commissure that failed to connect with the dorsal nerve cord (guidance), a gap or break within a commissure (break), and short neurite (short). The phenotypes are highlighted with the dashed boxes. (C) Quantification of morphological defects in WT and CMT2-associated mutant animals. Animals were scored visually for defects. Bar represents percentage of defective animals; n values are shown in each bar. Chi-square test with false discovery rate were performed to compare defects between WT and mutant animals. *P < 0.05, ****P < 0.0001. (D) Proportion of specific morphological defects in unc-116(e2310), and (E) dyn-1(ky51) animals. Scale bar represents 40 µm in (A) and 8 µm in (B).
Of the nine genes studied, mutation of unc-116 caused the most severe axonal defects in DA and DB motor neurons, with half of the worms displaying defects (Fig 3C). Nearly all of these defective worms had misguided commissures, and more than half presented with stunted neurite growth (Fig 3D). This trend was also observed in dyn-1 mutants, the second most defective amongst the nine genes, with guidance and short commissure defects again emerging as the most common types of defects (80% and 33% respectively; Fig 3E). While mutation of nep-2 saw an increase in DA/DB defect to 20%, this was not statistically significant (P > 0.05) (Fig 3C). Worms carrying mutations in the remaining six genes did not exhibit substantial morphological defects in DA and DB neurons, suggesting that neuronal degeneration may not be a major driver of the movement defects in these animals. However, in addition to the 4 classes of cholinergic motor neurons, the C. elegans body wall muscles are also innervated by GABAergic motor neurons not present in mammals. It is possible that degeneration and/or dysfunction of these neurons may play a role in the locomotion impairments.
CMT2-associated mutants display defective body wall muscles
Due to axonopathy, the skeletal muscles of CMT2 patients gradually undergo denervation, leading to muscle atrophy and weakness [7]. Hence, we next sought to characterize the morphology of muscle cells in our loss-of-function mutants. C. elegans possesses 95 obliquely striated body wall muscle cells, equivalent to mammalian skeletal muscles, that are arranged as staggered pairs in four longitudinal quadrants along the length of the worm (Fig 4A and 4B) [24, 25]. Individual muscle cells are connected intercellularly through gap junctions, allowing electrical signals to be transmitted rapidly between cells. Like motor neurons, functional body wall muscles are essential for C. elegans locomotion, including crawling and swimming [24].
Fig 4. Visual scoring of body wall muscle defects.
(A) A single 3-day old adult animal carrying the stEx30(Pmyo-3::gfp::myo-3 + rol-6(su1006)) transgene, which drives GFP-tagged myosin heavy chain in the body wall muscles. Animals with stEx30 also possess a rolling phenotype that facilitates visualization of muscle cells that flank the dorsal and ventral sections of the body. (B) 90 o rotated and expanded view of muscle area within dashed box in (A). (C) Categorical scoring of 3-day old adult WT and mutant animals, all carrying stEx30, for abnormalities in muscle fibres. Data is represented as the percentage of defective animals; n values are shown in each bar. Defects are compared between WT and CMT2 mutant animals using chi-square analysis with false discovery rate. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns = not significant. (D) Representative close-up muscle fibre images of individual CMT2 mutant animals. Scale bar represents 60 µm in (A), and 25 µm in (B) and (D).
To understand the consequences of mutating the CMT2-related genes on muscle morphology, we used a transgene [stEx30(Pmyo-3::GFP)] to fluorescently label myosin heavy chain filaments within the body wall muscles (Fig 4A and 4B). We assessed A3 stage worms for defects in filament integrity, scoring for loss of the distinct striations, GFP clumping that may signify the accumulation of cellular debris, and the presence of gaps between the filaments. Categorical scoring revealed that mutation of lin-41 resulted in the highest proportion (~70%) of defective animals, with most displaying irregularly organized filaments or large gaps between the filaments (Fig 4C and 4D). This was followed by unc-116(e2310), cua-1(gk107), and fzo-1(cjn020) mutants, with greater than 55% of animals found to be defective in each of these backgrounds. The muscular organization appeared slightly better in these mutant backgrounds compared to lin-41 mutants, although deterioration of filament lattices resulting in large gaps contributed to the high proportions of defects. GFP clumping was particularly prominent in unc-116(e2310) mutant animals. In comparison, the significant but smaller extent of muscle cell degeneration and deposits in hsp-25(tm700), dyn-1(ky51) and osm-9(ok1677) mutants meant that only 40–50% of animals were scored defective. Animals lacking hint-1 or nep-2 were not significantly different from the wild-type, which surprisingly displayed structural defects in about 20% of animals. This may be due to aging-associated decline of muscle structure [26].
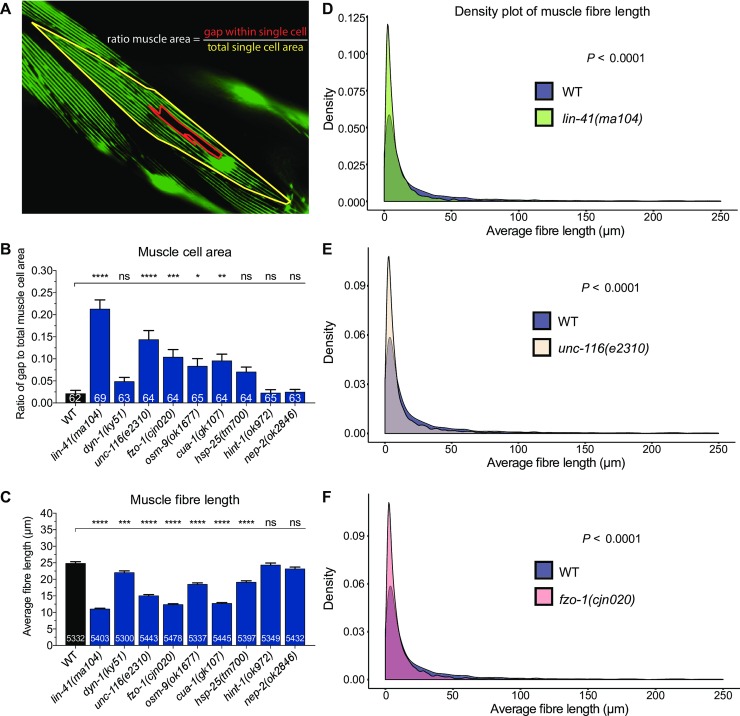
With clear structural collapse of the myofilaments in the majority of mutant animals, we next sought to quantify these morphological changes in more detail. To the best of our knowledge, no protocols exist for quantitatively assessing morphological changes in C. elegans muscles, with qualitative scoring typically used to compare muscle defects. As such, we applied innovative but simple approaches with freely available tools to quantify two aspects of the body wall muscle phenotypes: cell area and myosin filament length. For the body wall muscle area, we calculated the total area of a single cell and the area of gaps within that selected cell using the polygon tool in Fiji software. The ratio of gap to total cell area was then calculated and compared between wild-type and mutant animals (Fig 5A). In line with our categorical scoring results, the lin-41(ma104) mutant animals displayed the highest level of defects, with a 0.21 area ratio (Fig 5B). This ratio was in stark contrast to that of wild-type, hint-1(ok972) and nep-2(ok2846) mutants, where the ratio was 10-fold lower at approximately 0.02. The ratio of gaps in the muscle cells of unc-116, fzo-1, osm-9, and cua-1 mutants were also significantly increased compared to the wild-type (Fig 5B). In contrast, and despite a considerably high number of dyn-1(ky51) and hsp-25(tm700) worms being scored as defective in our categorical scoring (Fig 4C), the gaps within the muscle cells pertaining to structural deficits were inconsequential, resulting in no significant differences to the wild-type (Fig 5B).
Fig 5. Quantification of body wall muscle area and fibre length in 3-day old adults.
(A) Example image of how the muscle area ratio (gap to total cell area) was calculated. Gaps left by degenerating muscle fibres (red-lined area) and the total area of a single muscle cell (yellow-lined area) were drawn and calculated in Fiji using the polygon selection tool. (B) Comparison of gap to total muscle cell area ratio between WT and CMT2 mutants. (C) Measurement of individual fibre length using a combination of Fiji skeletonization and ilastik segmentation. The protocol is illustrated in S4 Fig. Only images with at least one complete visible oblique muscle cell were included for analysis. Fibres that recorded 0 µm or longer than 250 µm were excluded. (D-F) Density plot of muscle fibre length compared between WT and lin-41(ma104), unc-116(e2310), and fzo-1(cjn020). Density plots of other CMT2 mutants can be found in S5 Fig. Bar represents mean ± S.E.M in (B) and (C). Number of animals and fibres analyzed is listed in each bar in (B) and (C), respectively. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns = not significant from one-way ANOVA with Dunnett’s post hoc tests for multiple comparison in (B) and (C). F test was used for variance comparison between WT and mutants in (D) to (F), with significance set at P < 0.05. All animals expressed the stEx30 transgene.
Next, we used the ilastik image segmentation software to calculate the length of myofilaments (S4A Fig). We exploited the ‘suggest features’ and ‘live update’ functions in the training window to help us determine if the labelling had been sufficient. Once satisfactorily segmented, the images were exported as PNG binary images, and opened in Fiji software (S4B Fig). Default thresholding was then applied to the PNG image before skeletonization, a Fiji plugin, was implemented to filter out the border pixels (S4C Fig). Following skeletonization, detailed analysis of the length of each filament was performed using the “Analyze Skeleton” plugin. Any fibre measurement of 0 µm was omitted, as this was not physiologically possible, and any measurement greater than 250 µm was also excluded as we calculated this to be the maximum reasonable length for a filament. Using this analysis, we observed significantly lower average fibre lengths in seven of the nine genetic mutations (Fig 5C). In particular, the fibre lengths of lin-41(ma104), fzo-1(cjn020) and cua-1(gk107) worms were more than 50% shorter compared to wild-type, while hint-1(ok972) and nep-2(ok2846) worms were again unchanged. Mutations in osm-9 and hsp-25 led to a moderate 6 µm decrease in average filament length compared to wild-type, while the larger muscle gaps in unc-116(e2310) mutants further reduced the mean filament lengths by 10 µm. Despite the low ratio of gap to total muscle cell area in dyn-1(ky51) mutants, the average fibre length was significantly shortened by 3 µm in these animals (Fig 5C).
We further analyzed the morphological differences in muscle structure by comparing the variances of filament length using density plots. In wild-type animals, the average filament length was more widely distributed compared to most of the mutants, with a higher proportion of longer filaments evident. In contrast, lin-41(ma104), unc-116(e2310) and fzo-1(cjn020) mutants had significantly smaller distributions of filament lengths (Fig 5D–5F). All of the remaining mutants except for nep-2(ok2846) also displayed smaller ranges of filament lengths (S5 Fig). In summary, we optimized quantitative methods to study the structural deficits in the body wall muscles and found that mutations in genes associated with CMT2 led to increases in the ratio of gaps to total cell area, and considerably shorter myosin fibres. These methods help to reduce potential bias typical of phenotypic scoring and introduce reliable means for quantifying muscle cell morphology. Moreover, our results suggest that muscle degeneration and/or abnormal muscle development could be a prominent feature associated with the mutation of the CMT2-associated genes in C. elegans.
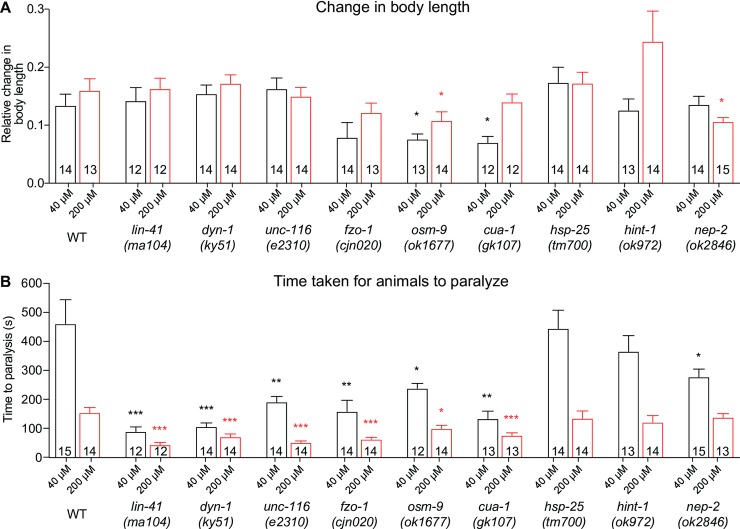
Loss of CMT2-associated genes impacts muscle function
Next, we questioned if the morphological defects in the body wall muscles caused functional deficits. To specifically analyze the functionality of the body wall muscles, we exposed A3 stage worms to different concentrations of levamisole (40 µM and 200 µM). Levamisole is a potent agonist of nicotinic acetylcholine receptors, which are located postsynaptically on muscle cell membranes, and is commonly used to analyze the contractility of the body wall muscles. We measured the body length before and after levamisole treatment, as well as the time taken for the worms to completely paralyze. There were only negligible differences compared to the wild-type for most of the mutants in the relative change in body length (Fig 6A), signifying minimal changes in muscle contraction and body stiffness. However, this was not the case for osm-9(ok1677), cua-1(gk107) and surprisingly, nep-2(ok2846), which all demonstrated significantly reduced changes in body length in response to levamisole (Fig 6A). It is important to note that lin-41(ma104) and unc-116(e2310) mutants have a shorter body length on average compared to wild-type and the other mutants, which could affect the response of these animals to levamisole. Strikingly however, when we calculated the time taken for the animals to reach complete paralysis, all mutants except for hsp-25(tm700) and hint-1(ok972) were completely paralyzed by 40 µM in less than two-thirds of the time required by wild-type worms (Fig 6B). The wild-type and mutant animals were all paralyzed more quickly when exposed to a higher concentration of levamisole, but lin-41, dyn-1, unc-116, fzo-1, osm-9 and cua-1 mutants again displayed significantly faster responses. Curiously, the shorter time taken for some of the mutants to become fully paralyzed does not directly correlate with our results on locomotor impairment and the presence of structural defects within the body wall muscles. This indicates that muscle morphology and function are not strictly dependent. It may also indicate that the mutations affect nicotinic acetylcholine receptor signalling within the muscles in different ways, only some of which culminate in deficits in the specific behavioural paradigms we have tested. In summary, the phenotypic characterization and levamisole-induced contractions of body wall muscles show that loss-of-function mutations in gene associated with CMT2 impair muscle morphology and function, likely contributing to decreases in mobility.
Fig 6. Quantification of body contraction in response to levamisole.
(A) Relative change in body length of 3-day old adult wild-type (WT) and mutant animals following the application of different concentrations of levamisole (40 μM or 200 μM). The original and contracted body lengths were measured before levamisole administration and when the animals had been paralyzed by levamisole, respectively. (B) Bar graph of the time taken for the animals to reach full paralysis following the addition of 40 μM or 200 μM levamisole. Bars represent mean ± S.E.M, n values are within each bar. We used one-way ANOVA with Dunnett’s post hoc tests to compare WT and CMT2 mutant animals within each concentration. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns = not significant.
Electrophysiological recordings uncover neuromuscular junction defects
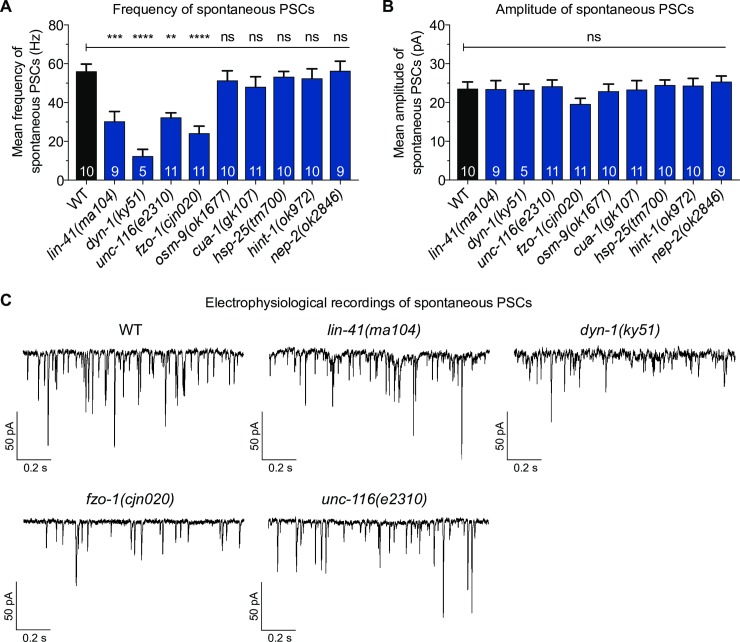
At the neuromuscular junction (NMJ), where axons innervate muscle cells, the axon terminals of motor neurons release acetylcholine to bind and activate postsynaptic nicotinic acetylcholine receptors to initiate muscle contraction. Compared to body wall muscles and neurons, the specific function of the NMJ is rarely studied in CMT animal models, even though loss of NMJ function has been reported in CMT2D mice models [27, 28]. It has also been suggested that NMJ dysfunction is length-dependent because the distal extremities are more severely affected than proximal limbs in CMT2 patients [29]. Therefore, we next questioned if dysfunctional NMJ signalling may be a common deficit associated with mutating CMT2-associated genes, and whether it also contributed to the progressive decay in locomotion we observed in our C. elegans mutants (S1A Fig and S1B Fig). To analyze NMJ activity, we performed patch clamp electrophysiological recordings on the NMJs in the intact ventral nerve cord of A3 stage animals. We focused on two properties of NMJ activity: the frequency and the amplitude of post-synaptic currents (PSCs). PSCs are generated when postsynaptic muscle receptors are activated by spontaneous acetylcholine release from presynaptic motor axon terminals. PSC frequency reflects the rate of spontaneous neurotransmitter release from presynaptic neurons. The PSC amplitude is further dependent on postsynaptic membrane integrity and cholinergic receptor activity. Hence, normal PSC frequency, equivalent to that seen in wild-type worms, relies heavily on the activity of presynaptic motor neurons, whereas the functionality of both presynaptic motor neurons and postsynaptic body wall muscles could affect the amplitude of PSCs [30].
As shown in Fig 7A and 7C, the frequency of spontaneous PSCs in lin-41(ma104), dyn-1(ky51), unc-116(e2310) and fzo-1(cjn020) animals was between 2- and 5-fold lower than the wild-type. The lower PSC frequencies of unc-116(e2310) and dyn-1(ky51) correlates with the morphological defects identified in the cholinergic motor neurons (Fig 3C–3E). Although we did not see a change in cholinergic motor neuron morphology as a result of the lin-41 and fzo-1 mutations, we did not analyze all cholinergic (or other classes of) motor neurons that may display degenerative phenotypes in these backgrounds. Nonetheless, the decrease in spontaneous PSC frequencies correlate with the potent deficits in both crawling and swimming, as well as the levamisole-induced muscle contractions, suggesting that presynaptic deficits likely play a major role in the movement defects and shorter response time to levamisole. In contrast, despite the numerous body wall muscle defects seen earlier, none of the mutant worms exhibited a significant decrease in mean amplitude of spontaneous PSCs (Fig 7B and 7C). This could mean that the quantity, distribution and positioning of postsynaptic receptors are sufficient to offset the degeneration observed in some muscle cells. Nevertheless, our results provide strong evidence linking several genes implicated in CMT2 with robust NMJ signalling.
Fig 7. Electrophysiological recordings from the neuromuscular junction (NMJ).
(A) Bar graph of the frequency of spontaneous postsynaptic currents (PSCs) recorded from 3-day old adult WT and CMT2 mutant animals. (B) Bar graph of the amplitude of spontaneous PSCs recorded from 3-day old adult WT and CMT2 mutant animals. (C) Sample traces of spontaneous PSCs recorded from ventral cord NMJs of individual worms. All recordings were made at a holding potential of –60 mV. All animals carried the vsIs48 transgene. Bars represent mean ± S.E.M, n values are within each bar in (A) and (B). One-way ANOVA with Dunnett’s post hoc tests were used for comparison between WT and CMT2 mutant animals in (A) and (B). **P < 0.01, ***P < 0.001, ****P < 0.0001, ns = not significant.
Discussion
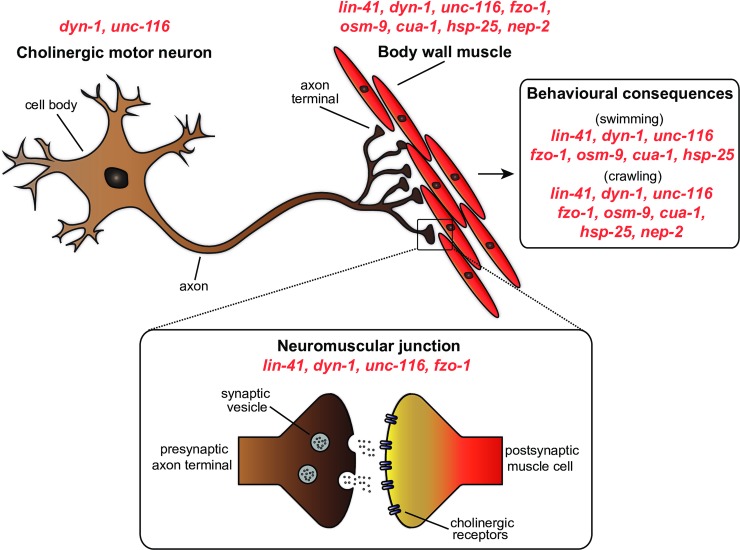
Through the targeting of CMT2-related genes in C. elegans, our study identifies new insights into the importance of these genes for cellular function and animal behaviour. A summary of the main findings in this study is provided in Fig 8. Overall, our analysis of CMT2-associated genes in C. elegans revealed many common characteristics including difficulties moving, as well as functional declines in neuronal and muscle function. Further studies to precisely dissect the commonalities between the many causal genes and disease phenotypes will be important for a full understanding of their roles in CMT2.
Fig 8. Summary of the sites at which mutation of CMT2-associated genes affect cellular function and animal behaviour in C. elegans.
Mutations in dyn-1 and unc-116 led to defects in cholinergic motor neuron morphology, while mutations in lin-41, dyn-1, unc-116, fzo-1, osm-9, cua-1, hsp-25, hint-1 and nep-2 resulted in irregularities in the body wall muscle. Neuromuscular junction dysfunction occurred as a result of lin-41, dyn-1, unc-116 and fzo-1 mutations, whereas lin-41, dyn-1, unc-116, fzo-1, osm-9, cua-1, hsp-25 and nep-2 mutants experienced motility impairments in swimming and/or crawling.
Shared physical characteristics between C. elegans and human patients following mutations in CMT2-associated genes
As CMT2 progresses, difficulty moving becomes the biggest obstacle for patients, mainly due to the loss of motor neurons, weakened muscles and limb deformities [7]. Locomotion analysis of the loss-of-function mutants in our study revealed that C. elegans also experienced similar physical debilitation. In particular, worms carrying loss-of-function mutations in the orthologous genes associated with CMT2A (fzo-1/MFN2), CMT2C (osm-9/TRPV4), CMT2F (hsp-25/HSPB1), CMT2M (dyn-1/DNM2), CMT2R (lin-41/TRIM2) and unclassified-CMT2s/motor neuropathies (unc-116/KIF5A, cua-1/ATP7A) experienced declines in swimming and crawling ability as they aged. We speculated that these movement defects were a result of weakening body wall muscle and motor neuron structure, as these underlying impairments are also reported in CMT2 patients. Our hypothesis was first supported by the high proportion of DA/DB cholinergic motor neuron defects, especially in the absence of functional unc-116 and dyn-1 genes. The kinesin-1 heavy chain UNC-116/KIF5A is a microtubule-dependent motor protein expressed in neurons and muscle, which is required for the transport and localization of mitochondria and lysosomes, and for regulating neuronal polarity. Downregulation of kinesin-1 has been reported to reverse the axon-dendrite organization, leaving behind disordered neurites without distinct dendrites or axons [31]. Similarly, dysmorphic neuronal structure is observed with loss of the DYN-1/dynamin-2 GTPase. DYN-1 displays overlapping cellular expression with UNC-116 and is involved in microtubule bundle processing and vesicular trafficking [32–37]. However, human patients with mutations in dynamin-2 have presented contradicting neuronal features, with some exhibiting neuronal degeneration but not others [38–41]. This could be due to the precise position of the mutations, with sequence changes within Pleckstrin homology domain and C-terminal proline-rich domain shown to induce more severe neuronal loss [42].
Our morphological analysis of body wall muscles revealed structural breakdown of myosin heavy chain filaments, again supporting the role of body wall muscle health on habitual locomotion. However, muscle biopsies are not often performed in patients, and accessible records show varied muscle descriptions among different subtypes of CMT2 [43–46]. Nevertheless, and consistent with our findings, many CMT2 patients carrying mutations in the orthologous genes for fzo-1, unc-116, lin-41, hsp-25 and osm-9 have been reported to exhibit muscular atrophy and myopathy especially in the distal limbs [1, 47–52]. As listed in S1 Table, the functions of the genes and encoded proteins vary greatly between the subtypes, from outer mitochondrial membrane fusion (FZO-1) to osmotic sensory receptors (OSM-9) and ubiquitin ligases involved in gene regulation (LIN-41). Providing further complications, genes such as cua-1/ATP7A are also involved in other diseases. Mutation of ATP7A, which encodes a copper transporter, was identified as causative for CMT2 less than a decade ago [53], but has been associated with the neurodegenerative Menkes disease since 1993 [54]. In hereditary neuropathy, missense mutations in ATP7A occur in a locus that results in distinct neuronal loss without causing the severe copper deficiency characteristic of Menkes disease [53]. Interestingly, mutation of another gene with similar copper-binding functions, SCO2, was reported to cause a different type of axonal CMT with similar symptoms, thus highlighting the possible role of copper homeostatic imbalance in axonal neuropathy [55]. Conversely, studies on CMT2M patients with DNM2 mutations mainly report normal muscle morphology, but occasional reports have described destabilization of skeletal muscle structure, further underlining the complexity of disease mechanisms [44, 45]. We also observed prominent GFP aggregation in the myosin filaments of some of the mutants, especially unc-116/KIF5A, which suggests underlying cellular toxicity, although the exact molecular mechanism behind this phenotype remains to be determined [26, 56]. Previously research has demonstrated that the destabilization of myosin fibres in C. elegans muscles result from the loss of functional filamentous actin [57]. As such, it would be intriguing to explore whether this is the cause of the muscle defects we observed in the current study, and therefore if the CMT2-associated genes impact filamentous actin.
From the investigation of muscle function using levamisole, we found that some of the animals lacking CMT2-associated gene function experienced paralysis far more quickly than wild-type animals. This observation was somewhat unexpected, as the time taken to reach paralysis should be longer if the body wall muscles are impaired because less receptors would be available on damaged postsynaptic membranes for levamisole to bind. However, due to structural defects and presynaptic deficits, an increase in the number and sensitivity of postsynaptic receptors to agonists may occur as a compensatory mechanism for a reduced number of endogenous agonists at the NMJ. Hence, the addition and abundance of exogenous levamisole could activate the waiting postsynaptic receptors and thereby explain the shorter paralysis times observed by some of the mutants.
The need for viable CMT2 animal models
In order to better understand the underlying pathophysiology of CMT2, effective animal models are required. Unfortunately, the complexity of the disease has contributed to the scarcity of working animal models. Hence, numerous attempts to generate accurate animal models of CMT2, especially in rodents, have not been successful. For instance, constitutive DNM2 knockout (DNM2-/-) is embryonic lethal in mice, while heterozygous DNM2+/- mice do not present any CMT2-like phenotypes [58, 59]. DNM2-deficient mice expressing a floxed DNM2 allele were found to be viable, but comprehensive analyses have not been performed, with only myelinated peripheral neurons analyzed [60]. Similarly, constitutive knockout in mice of KIF5A, the gene responsible for a number of neuromuscular disorders including CMT2, hereditary spastic paraplegia, and amyotrophic lateral sclerosis, results in lethality soon after birth. Karle et al. overcame this lethality with selective neuronal knockout of KIF5A, but insights into other cellular consequences such as muscle or NMJ phenotypes could not be made [61]. Mouse models have been generated for specific CMT2A mutations, including T105M and R94W, which have revealed important insights into the cellular mechanism of disease and therapeutic targets [6, 62–65]. However, further complicating our understanding of the disease is the finding of different patient-specific mutations having alternative (and sometimes opposing) effects on protein function [19, 20]. Furthermore, our findings on HINT1 mutants largely mirrored those observed by Seburn and colleagues, where mice lacking functional HINT1 were phenotypically normal, indicating the need for a more effective animal model for HINT1-related disease [66]. Through detailed studies of muscle morphology, however, we could identify subtle changes in worms lacking hint-1 that might impact behaviour in older animals or with additional genetic or environmental impacts.
In addition to the unclassified CMT2s in this study, there have previously been no working animal models to explore the function of genes involved in CMT2C, CMT2R and CMT2T. In contrast to rodents, C. elegans knockouts are mostly viable, and mutation of the CMT2-associated genes in this study produced phenotypes that could be related to human patients. In summary, this study of both behavioural and cellular consequences of mutating CMT2-associated genes in C. elegans is an important step forward in the establishment of reliable animal models to understand disease mechanisms. This is especially true for the unclassified CMT2 subtypes, as well as CMT2C, CMT2R and CMT2T, for which no animal models have been generated.
C. elegans as an animal model for high-throughput drug screening
High-throughput screening (HTS) of small compounds on whole animals has been gaining momentum in recent years in bid to identify effective therapeutics that could improve disease symptoms. The main advantage of using whole animals in HTS is that the biological target does not need to be identified, as compounds can be screened based on phenotypic changes in motility or lifespan. This is a huge benefit for human diseases with poorly understood biological mechanisms such as CMT2. While rodents are highly favoured as disease models due to their closer similarities with humans, using rodents in HTS is not only extremely costly, but also time-consuming. In addition to the advantages associated with ease of culturing, small size, high progeny rate, and rapid life cycle, the C. elegans genome can be readily manipulated, and its transparent body makes visualization and analysis of cellular structures simpler than other animal models [11]. For these reasons, the nematode has been increasingly used as a tool for HTS.
One of the earliest large-scale, liquid-based compound screenings in C. elegans took place more than a decade ago, and focused on identifying novel antimicrobials and anti-ageing drugs [67]. Following the success of early HTS, a number of novel therapeutics that reversed the symptoms of a range of human diseases has since been identified in automated HTS assays using C. elegans. For instance, fluphenazine was discovered from HTS in C. elegans to be efficacious in reducing the accumulation of misfolded proteins responsible for α1-antitrypsin deficiency (ATS). The finding was subsequently replicated and validated in mammalian cell lines and a mouse model of the disease [68]. In a different study, Sleigh and colleagues modelled spinal muscular atrophy in C. elegans and combined the automated phenotyping system with HTS to identify three compounds, 4-aminopyridine (potassium channel blocker), gaboxadol (GABAA receptor agonist), and N-acetylneuraminic acid (monosaccharide), that could improve motility in the mutant worms [69]. More recently, similar restoration of movement by a number of drug classes (including neuroleptics) isolated from HTS in nematode models of amyotrophic lateral sclerosis and autism spectrum disorders was also discovered [70, 71]. In the present study, we utilized the automated movement tracker WMicrotracker to detect the motility of worms in a high-throughput manner (96-wells per run). Consistent with manual and individual movement assays, a number of CMT2-associated genetic mutants recorded a reduction in movement rate in the WMicrotracker. This phenotypic impairment has opened new doors of opportunity for high-throughput screening of small molecules on C. elegans CMT2 mutants, where compounds that successfully rescue the movement defect could be developed into effective therapeutics for the currently incurable disease.
Beyond qualitative scoring: Building the foundation for quantifying muscle morphology
The transparency of C. elegans allows for simple visualization and analysis of underlying cellular and organelle structures and processes. However, despite the large number of studies that have explored cellular defects as a result of genetic mutations, no reliable tools or measurements for quantifying changes in cellular morphology in C. elegans have been developed. Instead, categorical scoring has been the method of choice for defining defects. In 2014, Rizk and colleagues first optimized a “segmentation and quantification of subcellular shapes (SQUASSH)” protocol that enabled researchers to distinguish and quantify cellular and subcellular structures or shapes from fluorescence-based microscopy images [72]. Previously, we optimized the parameters in the SQUASSH segmentation ImageJ macro for a detailed, non-subjective assessment of mitochondrial morphology in both muscle and neuronal cells of C. elegans [73]. In this previous research, we also included movement assays, with the outcomes consistent with the current study, whereby animals carrying the fzo-1(cjn020) mutation experienced decreased movement compared to wild-type animals.
In the present study, we further improved on quantitative methods to non-subjectively analyze muscle defects. Using a combination of Fiji and ilastik segmentation software [74, 75], we were able to measure the muscle cell area and myosin fibre lengths for comparison with our qualitative scoring. From visual analysis of muscle cells, we saw conspicuous structural breakdown and irregularities of the myosin fibres especially in lin-41, unc-116, fzo-1, osm-9 and cua-1 mutants, which correlated with the lack of normal movement from these animals. Further examination of individual myosin fibre lengths and gaps using quantitative approaches also saw significant defects in the same mutants, thus validating our methodology. The lack of difference in the gap to total muscle cell area ratio and average fibre length between wild-type, hint-1 and nep-2 mutants was also consistent with the phenotypic scoring results, further supporting the viability of the quantitative measurements. Surprisingly, however, comparison of the relative density and variance of muscle fibre length revealed a sizeable difference between wild-type and all mutants except nep-2, although these differences may not necessarily lead to behavioural defects. Our results imply that comprehensive computational-based analyses can detect subtle changes in phenotypic variation that cannot be identified by visual means. Thus, by using Fiji and ilastik segmentation to quantify muscle cell morphology, we have taken a step forward in improving quantitative methodology to measure cellular phenotypic changes in C. elegans, and reduced the potential bias accompanying categorical scoring.
The neuromuscular junction as a target for therapeutics
To our knowledge, this study is the first to report a reduction in the frequency of spontaneous PSCs in C. elegans carrying mutations in genes associated with CMT2A, CMT2M, CMT2R, and the uncharacterized CMT2 associated with mutations in unc-116. Our findings suggest that these genes are important for robust NMJ function. The presence of presynaptic cholinergic neuron defects and/or postsynaptic body wall muscle degeneration in these mutant animals further supports the notion of NMJ deficits, which are likely to further compound the motility impairments identified in the mutant animals. Deficits in NMJ function have previously been noted for other CMT classes, including CMT1A and CMT4 [76–78]. Furthermore, Spaulding, Sleigh and colleagues first demonstrated that synaptic defects could be a contributing factor to the early symptoms of axonal CMT [28, 79]. NMJ loss was observed in the proximal limb muscles in a CMT2D mouse model, which led to muscle weakness and restricted locomotion. It is also intriguing to note that administration of physostigmine, a reversible inhibitor of acetylcholinesterase which acts at the synapse, improved the movement of CMT2D mice, thus supporting the role of NMJ as a biological target for the development of CMT2 therapeutics [28]. Further studies assessing the effects of physostigmine and other synaptically-acting drugs on NMJ function and motility in CMT2 animal models will be important for delineating the involvement of the NMJ in CMT2 pathophysiology. Taken together, the presence of NMJ abnormalities in animals lacking CMT2-asscoiated genes suggests that the NMJ could be a major site in the disease pathology, and additional studies looking into the properties of NMJ in the presence of CMT2 genetic mutations will provide a greater understanding of the role of NMJ stability in CMT2 pathophysiology and as a therapeutic target.
Limitations of the current study
As the aim of our study was to compare the function of genes associated with CMT2 by analysing loss-of-function mutations, it is important to point out its limitations. Firstly, as CMT2 is typically inherited in an autosomal dominant manner, to build upon our study, and possibly provide more relevant disease models, it will be important to generate C. elegans models carrying patient-specific mutations in the orthologous genes. These could then be compared to our study and used to determine if the mutations are loss-of-function in nature, or if they affect the genes in other ways. Furthermore, rescue of the defects in the C. elegans mutants using the orthologous human genes could be carried out to determine if the genes function similarly between the species. This could also facilitate the generation of disease models, as patient-specific version of the genes could be introduced and compared to worms carrying the wild-type human genes.
In terms of quantitative measurement of muscle morphology, while a number of steps have been taken to greatly reduce subjective bias, it has not been eliminated completely. For example, to visualize the muscle fibres we used an extrachromosomal array (stEx30) that results not only in GFP-tagged myosin, but also a rolling phenotype. Due to the unpredictability of the rolling phenotype (i.e. extent of rolling and direction), imaging a consistent area of fibres or the same muscle cells across animals is extremely challenging. Generation of a transgenic strain without the rolling phenotype and the use of simple strategies to roll the worms prior to imaging will solve this issue. Worms that are sufficiently immobilized by anesthetics will maintain a straight body, and a cover slip can be used to gently slide over the worms until they lay on their lateral side. To optimize our methods further, microfluidic devices could be used to straighten the animals and ensure entire muscle fibres on one of the lateral sides can be visualized [80].
Conclusions
We have shown that C. elegans carrying loss-of-function mutations in genes associated with CMT2 share many disease characteristics with human patients. This study has further highlighted the advantages of C. elegans as a reliable animal model to study neuropathies, and through the development of the first quantitative methods for analyzing muscle defects has laid the foundation for future comprehensive studies not just involving the C. elegans body wall muscles, but also potentially other cellular types. Our study of nine orthologous genes associated with CMT2 has revealed common sites of deficiencies when these genes are lacking (Fig 8), with functional loss at the NMJ and within the musculature most prominent. Our extensive characterization of CMT2-associated genetic mutants in C. elegans provides valuable insights and tools for understanding the common pathways underlying the heterogeneous neuropathy, and for future high-throughput drug screening approaches aimed at identifying novel therapeutics for the currently incurable and debilitating disease.
Materials and methods
Generation and maintenance of C. elegans strains
Maintenance, crosses, and other genetic manipulations were all performed via standard procedures [81]. A full list of strains used in this study is shown in S1 Table. CRISPR/Cas-9 approach was used to generate the fzo-1(cjn020) deletion allele, with nucleotides 25–2654 of the fzo-1 locus removed [73]. The Pcua-1::cua-1::cua-1 3`UTR DNA rescue plasmid was generated by SunyBiotech corporation. Hermaphrodites were used for all experiments and were grown at 20°C on nematode growth medium (NGM) plates (0.25% peptone, 51 mM NaCl, 25 mM KH2PO4, 5 µg/ml cholesterol, 1 mM CaCl2, 1 mM MgSO4, 2% agar) seeded with OP50 Escherichia coli.
Swimming assays
Random individual synchronized worms (larvae stage 1, larvae stage 4, 3-day old adult and 7-day old adult) were transferred to an unseeded 60 mm plate at room temperature to remove leftover OP50, then individual worms were transferred to a 10 μL droplet of M9 buffer (22 mM KH2PO4, 42 mM NaH2PO4, 86 mM NaCl, 1 mM MgSO4) [81]. Each worm was left to acclimatize for 15 seconds, and the number of thrashes per minute was counted using a hand counter under an Olympus SZ51 microscope. Experiments were performed blinded to the genotype. The total time window from worm picking to end of recording for each worm was 70–80 seconds. A total of 10 animals were used for each replicate, with three replicates performed. For analysis of swimming speed, videos of 5 worms per replicate swimming in 1 mL of M9 buffer on single unseeded 35 mm plate were recorded. These videos were then converted in Fiji (version 2.0.0) [74] to 30 fps frame rate in .avi format and swimming speed of animals within field of view was quantified using WormLab software (version 3.1.0, MBF Bioscience). The settings for swimming assays were adjusted according the protocol provided on the WormLab website [82]. Recordings were performed in triplicates or more, depending on the number of animals within the field of view. Data of any worm that went out of focus during recording was discarded. All images and videos were taken with Leica Microsystems M80/MC190 camera microscope. All assays were performed at room temperature.
Body bends
Individual synchronized worms (larvae stage 1, larvae stage 4, 3-day old adult and 7-day old adult) were gently placed on unseeded 60 mm NGM plates and left to acclimatize for 3–5 minutes at room temperature. The number of body bends, defined as the maximum bend of the part of worm just behind the pharynx from one end to the opposite direction in a forward sinusoidal pattern, was manually counted for 3 minutes under the Olympus SZ51 microscope [83]. Reverse and omega bends were included in the count, but reverse bend in the same direction was not included. The number of body bends per minute was then averaged. The total time window from worm picking to end of recording for each worm was 6 to 8 minutes. To quantify crawling speed, videos of 5 worms per replicate crawling on a single unseeded 35 mm plate were recorded and converted in Fiji (version 2.0.0) [74] to 15 fps frame rate in .avi format. Crawling speed of the worms within field of view in the converted video was then quantified using WormLab software (version 3.1.0, MBF Bioscience). The settings for crawling assay were based on the protocol on the WormLab website [82]. Recordings were performed in triplicates or more. Data of any worm that went out of focus during the recording was discarded. All images and videos were taken with a Leica Microsystems M80/MC190 camera microscope. All assays were performed blinded to genotype at room temperature.
Optimization of worm synchronization for WMicrotracker
Large population of age-synchronized worms was prepared according to the Solis and Petrascheck protocol, with mild modifications (S3 Fig) [84]. To obtain large numbers of synchronized, gravid adults were first bleached to release the eggs, which were incubated overnight for hatching [84]. Hatched L1 worms were then plated the next day and left to grow until they reached adulthood. Previous studies have used fluorodeoxyuridine (FUdR) as an inhibitor of DNA synthesis to prevent adult worms from reproducing. However, since FUdR can affect C. elegans stress responses and lifespan [85, 86], as well as eliciting responses after metabolism in E. coli (the food source for C. elegans) [87], we instead manually separated adult worms from their progeny until they reached the 3-day old adult stage for WMicrotracker experiments. The separation was achieved by washing the plate containing adult worms and progenies into a tube and letting the adult worms precipitate by gravity. Each day until the day of experiment (3-day old adults), the worms were washed and separated from their progenies through gravitational precipitation onto new seeded plates. The synchronized A3 worms were then plated into a 96-well plate (30–50 worms per well and at least 6 wells per group per experiment) and movement measured using the WMicrotracker instrument.
WMicrotracker assay
On the day of experiment, synchronized 3-day old worms were initially washed off the plates with M9 buffer into micro-centrifuge tubes, and subsequent washing of the worms in the tubes were performed at least twice to remove residual OP50 and offspring. 100 μL of 30–50 worms were then plated into each well of 96-well plate, 6 wells per genotype across two replicate experiments. The plate was then placed in the WMicrotracker-One™ instrument (PhylumTech) and analyzed for 3 hours at room temperature.
Fluorescence imaging
The strains expressing the stEx30(Pmyo-3::gfp::myo-3 + rol-6(su1006)) transgene were used for analysis of body wall muscles, while the vsIs48(Punc-17::gfp) transgene was used to visualize cholinergic neurons. Worms were immobilized with 0.05% tetramisole hydrochloride solution and mounted on 4% agarose pads on glass slides. Imaging of both body wall muscle and cholinergic neuron was carried out using Zeiss Axio Imager M2 microscope on 3-day old adult worms at 400 X magnification. For body wall muscle, the images were captured from the upper or lower part of the worm, excluding the extreme anterior and posterior regions, and regions adjacent to the vulva. For cholinergic neurons, only the dorsal neurons were analyzed. Both the defects of body wall muscle and cholinergic motor neurons were scored categorically as defective or not defective.
Quantitative measurements of body wall muscle cell area and fibre length
To quantify body wall muscle cell area, we made sure that every image taken included at least one complete muscle cell (yellow enclosed region in Fig 5A) [88]. All images were taken from similar body areas in the mutant strains and wild-type controls. In all instances, body wall muscles were imaged from either the mid region between the pharynx and vulva, or the mid region between the vulva and anus. Using the polygon selection in Fiji (version 2.0.0), we measured the area size of a muscle cell and the gaps within each cell. The ratio of the gaps relative to the total muscle cell area was then calculated. For this analysis, 60 or more worms were imaged and analyzed. For muscle fibre (myosin filament) length, 5 to 11 body wall muscle images (.tif format converted from .czi in Fiji version 2.0.0) of each mutant strain were first segmented in ilastik (version 1.3.0) [75] (S4 Fig). During segmentation, the individual muscle fibres were carefully “trained” or classified to separate them from the unwanted background. Each segmented image was then exported (output data type unassigned 8-bit, range renormalized from 0,1 to 0,255) as a viewable binary image in .png format. The .png files were next threshold-adjusted (default setting) and skeletonized in Fiji (version 2.0.0, Skeletonize 2D/3D plugin). The lengths of the ‘skeletons’, or individual myosin filaments, were then analyzed using the Fiji Analyze Skeleton 2D/3D plugin (S4C Fig). Filaments that recorded 0 or more than 250 µm (the maximum reasonable length for a filament) were excluded. Muscle cells with sections that were out of focus were also not included in the analysis.
Quantification of muscle contraction using levamisole
To quantify levamisole-induced body contraction, animals were first incubated in 10 μL drug-free solution (140 mM NaCl, 5 mM KCl, 5 mM CaCl2, 5 mM MgCl2, 11 mM dextrose, and 5 mM HEPES; 330 mOsm, pH adjusted to 7.2) to measure the original body length. This was followed by measurement of body length after the addition of 10 μL of 80 μM or 400 μM levamisole solution to obtain a final concentration 40 μM or 200 μM levamisole. The time taken for the worms to reach full paralysis after being incubated in levamisole solution was recorded. Worm body was monitored under Olympus MVX10 microscope equipped with XM10 CCD camera. The cellSens software (version 1.7, Olympus) was used to calculate the length of the worm body.
Electrophysiology
Whole-cell patch-clamp recordings of NMJs were performed on 3-day old adult animals at room temperature under a 60 X water immersion lens with an EPC-10 amplifier and the Patchmaster software (HEKA) [30, 89]. Voltages were clamped at −60 mV. Recordings of spontaneous PSCs from each worm were analyzed for the frequency and amplitude of spontaneous PSCs using MiniAnalysis (Synaptosoft, Inc.). Recording pipettes (4–6 MΩ) were pulled from borosilicate glass and fire polished. The pipette solution contained 120 mM KCl, 20 mM KOH, 4 mM MgCl2, 10 mM HEPES, 0.25 mM CaCl2, 36 mM sucrose, 5 mM EGTA, and 4 mM Na2ATP (315 mOsm; pH adjusted to 7.2). The bath solution contained 140 mM NaCl, 5 mM KCl, 5 mM CaCl2, 5 mM MgCl2, 11 mM dextrose, and 5 mM HEPES (330 mOsm; pH adjusted to 7.2).
Statistical analysis
Statistical analysis and graphs were performed/generated using Prism 7 (GraphPad Software) or R version 3.2.2 (R Core Team) [90]. Student’s t-test was used to compare two groups, and one-way ANOVA was used for comparing means of more than two groups followed by Dunnett's multiple comparison post hoc test. F test was used to compare variances. Chi-square test (chisq.post.hoc R package), controlled for false discovery rate, was used to compare categorical data. Significance was set at P < 0.05 unless stated otherwise.
Supporting information
(A) Thrash rates compared between wild-type (WT) and the nine mutant strains, across three different ages, larval stage 1 (L1), larval stage 4 (L4), 3-day old adults (A3) and 7-day old adults (A7). (B) Rate of body bends of WT and CMT2 mutant animals quantified across the same ages as in (A). Each dot in (A) and (B) represents a single animal (n ≥ 30). One-way ANOVA with Dunnett’s post hoc tests were used to compare rates of thrash or body bend between WT and mutant animals in (A) and (B). Data is represented as mean ± S.E.M. **P < 0.01, ***P < 0.001, ****P < 0.0001, ns = not significant.
(PDF)
(A) Thrash rates compared between 7-day old adult non-transgenic mutant strains and their transgenic siblings. All transgenic animals carry fosmids containing the corresponding wild-type version of the gene, except for fzo-1, for which a Pmyo-3::fzo-1::unc-54 3`UTR was used, and cua-1, which expresses a Pcua-1::cua-1::cua-1 3’UTR plasmid. Data is represented as mean ± S.E.M, with symbols showing individual animals. (B) Rate of body bends compared between 7-day old adult non-transgenic nep-2 mutants and their transgenic siblings expressing wild-type copies of nep-2 from a fosmid. For (A) and (C), *P < 0.05, ****P < 0.0001 from Student’s t-tests.
(PDF)
Schematic of the workflow, from synchronization via bleaching and washing, to plating of synchronized 3-day old adult animals in a 96-well plate for experimentation.
(PDF)
(A) Original muscle segment must contain at least one full visible oblique muscle cell. (B) Distinct myosin fibre is classified and segregated from the unwanted background (in asterisk) in ilastik. (C) Skeletonization of image in Fiji. The image was skeletonized to filter out the border pixels, leaving behind only the skeletal remnants that become the topological representations of the original fibres. Measurement of each fibre was performed following skeletonization. Fibres that were 0 µm or more than 250 µm were excluded. Scale bar represents 25 µm.
(PDF)
(A-F) The variance of all except for nep-2(ok2846) was statistically different compared to wild-type (WT). Statistics were performed using F test for variances, significance set at P ≤ 0.05. Experiments were performed on 3-day old adult animals that carried stEx30 transgene.
(PDF)
(DOCX)
Acknowledgments
We thank Joe Byrne for generating the fzo-1(cjn020) strain; Marina Kennerson and Ebru Boslem for comments on the manuscript, and members of the Neumann lab for valuable discussions and input. Some strains were provided by the CGC, which is funded by NIH Office of Research Infrastructure Programs (P40 OD010440). The authors thank WormBase for its wealth of information on C. elegans, and acknowledge Monash Micro Imaging, Monash University, for the provision of instrumentation, training and technical support. We are also grateful to Ruidong Xiang for his help and advice on R programming.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
This work was supported by Charcot-Marie-Tooth Association of Australia research grants (2015 and 2018), and National Health and Medical Research Council Project Grant 1099690 awarded to BN. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Barisic N, Claeys KG, Sirotković-Skerlev M, Löfgren A, Nelis E, De Jonghe P, et al. Charcot-Marie-Tooth disease: A clinico-genetic confrontation. Ann Hum Genet. 2007;72(3):416–41. [DOI] [PubMed] [Google Scholar]
- 2.Barreto LCLS, Oliveira FS, Nunes PS, de França Costa IM, Garcez CA, Goes GM, et al. Epidemiologic study of Charcot-Marie Tooth disease: A systematic review. Neuroepidemiology. 2016;46(3):157–65. 10.1159/000443706 [DOI] [PubMed] [Google Scholar]
- 3.Reilly MM, Murphy SM, Laurá M. Charcot‐Marie‐Tooth disease. J Peripher Nerv Syst. 2011;16(1):1–14. 10.1111/j.1529-8027.2011.00324.x [DOI] [PubMed] [Google Scholar]
- 4.d’Ydewalle C, Benoy V, Van Den Bosch L. Charcot-Marie-Tooth disease: Emerging mechanisms and therapies. Int J Biochem Cell Biol. 2012;44:1299–304. 10.1016/j.biocel.2012.04.020 [DOI] [PubMed] [Google Scholar]
- 5.Shy ME, Patzkó Á. Axonal Charcot–Marie–Tooth disease. Curr Opin Neurol. 2011;24:475–83. 10.1097/WCO.0b013e32834aa331 [DOI] [PubMed] [Google Scholar]
- 6.Cartoni R, Martinou J. Role of mitofusin 2 mutations in the physiopathology of Charcot–Marie–Tooth disease type 2A. Exp Neurol. 2009;218:268–73. 10.1016/j.expneurol.2009.05.003 [DOI] [PubMed] [Google Scholar]
- 7.Bird TD. Charcot-Marie-Tooth neuropathy type 2 1998. September 24 [Updated 2016 Apr 14]. 1993–2018. In: GeneReviews® [Internet] [Internet]. Seattle (WA): University of Washington, Seattle; Available from: https://www.ncbi.nlm.nih.gov/books/NBK1285/. [Google Scholar]
- 8.Chandhok G, Lazarou M, Neumann B. Structure, function, and regulation of mitofusin-2 in health and disease. Biol Rev Camb Philos Soc. 2018;92(2):933–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Züchner S, Mersiyanova IV, Muglia M, Bissar-Tadmouri N, Rochelle J, Dadali EL, et al. Mutations in the mitochondrial GTPase mitofusin 2 cause Charcot-Marie-Tooth neuropathy type 2A. Nat Genet. 2004;36:449 10.1038/ng1341 https://www.nature.com/articles/ng1341#supplementary-information. [DOI] [PubMed] [Google Scholar]
- 10.Ackerley S, James PA, Kalli A, French S, Davies KE, K T. A mutation in the small heat-shock protein HSPB1 leading to distal hereditary motor neuronopathy disrupts neurofilament assembly and the axonal transport of specific cellular cargoes. Hum Mol Genet. 2006;15(2):347–54. 10.1093/hmg/ddi452 [DOI] [PubMed] [Google Scholar]
- 11.Markaki M, Tavernarakis N. Modeling human diseases in Caenorhabditis elegans. Biotechnol J. 2010;5:1261–76. 10.1002/biot.201000183 [DOI] [PubMed] [Google Scholar]
- 12.Laranjeiro R, Harinath G, Burke D, Braeckman BP, Driscoll M. Single swim sessions in C. elegans induce key features of mammalian exercise. BMC Biology. 2017;15(30):1–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ma L, Zhao Y, Chen Y, Cheng B, Peng A, Huang K. Caenorhabditis elegans as a model system for target identification and drug screening against neurodegenerative diseases. Eur J Pharmacol. 2018;819:169–80. 10.1016/j.ejphar.2017.11.051. 10.1016/j.ejphar.2017.11.051 [DOI] [PubMed] [Google Scholar]
- 14.Li J, Le W. Modeling neurodegenerative diseases in Caenorhabditis elegans. Exp Neurol. 2013;250:94–103. 10.1016/j.expneurol.2013.09.024. 10.1016/j.expneurol.2013.09.024 [DOI] [PubMed] [Google Scholar]
- 15.Silverman GA, Luke CJ, Bhatia SR, Long OS, Vetica AC, Perlmutter DH, et al. Modeling molecular and cellular aspects of human disease using the nematode Caenorhabditis elegans. Pediatr Res. 2009;65:10 10.1203/PDR.0b013e31819009b0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Safka Brozkova D, Deconinck T, Beth Griffin L, Ferbert A, Haberlova J, Mazanec R, et al. Loss of function mutations in HARS cause a spectrum of inherited peripheral neuropathies. Brain. 2015;138(8):2161–72. 10.1093/brain/awv158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vester A, Velez-Ruiz G, McLaughlin HM, Program NCS, Lupski JR, Talbot K, et al. A loss-of-function variant in the human histidyl-tRNA synthetase (HARS) gene is neurotoxic in vivo. Hum Mutat. 2013;34(1):191–9. Epub 10/11. 10.1002/humu.22210 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Iapadre G, Morana G, Vari MS, Pinto F, Lanteri P, Tessa A, et al. A novel homozygous MFN2 mutation associated with severe and atypical CMT2 phenotype. European Journal of Paediatric Neurology. 2018;22(3):563–7. 10.1016/j.ejpn.2017.12.020. 10.1016/j.ejpn.2017.12.020 [DOI] [PubMed] [Google Scholar]
- 19.Detmer SA, Chan DC. Complementation between mouse Mfn1 and Mfn2 protects mitochondrial fusion defects caused by CMT2A disease mutations. The Journal of Cell Biology. 2007;176(4):405 10.1083/jcb.200611080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.El Fissi N, Rojo M, Aouane A, Karatas E, Poliacikova G, David C, et al. Mitofusin gain and loss of function drive pathogenesis in <em>Drosophila</em> models of CMT2A neuropathy. EMBO reports. 2018;19(8):e45241 10.15252/embr.201745241 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Simonetta SH, Golombek DA. An automated tracking system for Caenorhabditis elegans locomotor behavior and circadian studies application. Journal of Neuroscience Methods. 2007;161(2):273–80. 10.1016/j.jneumeth.2006.11.015. 10.1016/j.jneumeth.2006.11.015 [DOI] [PubMed] [Google Scholar]
- 22.Altun ZF, Hall DH. Nervous System, General Description 2009. Available from: http://www.webcitation.org/5qMVQWHl2.
- 23.White JG, Southgate E, Thomson JN, Brenner S. The structure of the nervous system of the nematode Caenorhabditis elegans. Philos Trans R Soc Lond B Biol Sci. 1986;314(1165):1–340. 10.1098/rstb.1986.0056 [DOI] [PubMed] [Google Scholar]
- 24.Altun ZF, Hall DH. Muscle system, somatic muscle 2009. Available from: 10.3908/wormatlas.1.7 [DOI]
- 25.Gieseler K, Qadota H, Benian GM. Development, structure, and maintenance of C. elegans body wall muscle 2005. In: WormBook: The Online Review of C elegans Biology [Internet] [Internet]. Pasadena (CA): WormBook; Available from: https://www.ncbi.nlm.nih.gov/books/NBK426064/. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Meissner B, Warner A, Wong K, Dube N, Lorch A, McKay SJ, et al. An integrated strategy to study muscle development and myofilament structure in Caenorhabditis elegans. PLoS Genet. 2009;5(6):e1000537 10.1371/journal.pgen.1000537 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chandhok G, Soh MS. Presynaptic deficits at neuromuscular junctions: A specific cause and potential target of axonal neuropathy in type 2 Charcot–Marie–Tooth disease. J Neurosci. 2016;36(31):8067–69. 10.1523/JNEUROSCI.1515-16.2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Spaulding EL, Sleigh JN, Morelli KH, Pinter MJ, Burgess RW, Seburn KL. Synaptic deficits at neuromuscular junctions in two mouse movels of Charcot-Marie-Tooth type 2d. Journal of Neuroscience. 2016;36(11):3254–67. 10.1523/JNEUROSCI.1762-15.2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Saporta MA. Cellular reprogramming and inherited peripheral neuropathies: perspectives and challenges. Neural Regen Res. 2015;10(6):894–6. 10.4103/1673-5374.158345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liu J, Zhang B, Lei H, Feng Z, Liu J, Hsu A, et al. Functional aging in the nervous system contributes to age-dependent motor activity decline in C. elegans. Cell Metab. 2013;18(3):392–402. 10.1016/j.cmet.2013.08.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yan J, Chao DL, Toba S, Koyasako K, Yasunaga T, Hirotsune S, et al. Kinesin-1 regulates dendrite microtubule polarity in Caenorhabditis elegans. eLIFE. 2013;2:e00133 10.7554/eLife.00133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Clark SG, Shurland DL, Meyerowitz EM, Bargmann CI, van der Bliek AM. A dynamin GTPase mutation causes a rapid and reversible temperature-inducible locomotion defect in C. elegans. Proc Natl Acad Sci U S A. 1997;94(19):10438–43. 10.1073/pnas.94.19.10438 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Consortium TU. UniProt: a worldwide hub of protein knowledge. Nucleic Acids Res. 2019;47(D1):D506–15. 10.1093/nar/gky1049. 10.1093/nar/gky1049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Göhre V, Vollmeister E, Bölker M, Feldbrügge M. Microtubule-dependent membrane dynamics in Ustilago maydis: Trafficking and function of Rab5a-positive endosomes. Commun Integr Biol. 2012;5(5):485–90. 10.4161/cib.21219 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Harris TW, Arnaboldi V, Cain S, Chan J, Chen WJ, Cho J, et al. WormBase: a modern model organism information resource. Nucleic Acids Res. 2019. 10.1093/nar/gkz920 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hyun-Ok S, Jungsoo L, Yon Ju J, Meenakshi D, Jeong Hoon C, Byung-Jae P, et al. Calcineurin regulates coelomocyte endocytosis via DYN-1 and CUP-4 in Caenorhabditis elegans. Mol Cells. 2010;30(3):255–62. Epub 09/30. 10.1007/s10059-010-0116-x [DOI] [PubMed] [Google Scholar]
- 37.Thompson HM, Skop AR, Euteneuer U, Meyer BJ, McNiven MA. The large GTPase dynamin associates with the spindle midzone and is required for cytokinesis. Curr Biol. 2002;12(24):2111–7. 10.1016/s0960-9822(02)01390-8 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Berciano J, García A, Gallardo E, Peeters K, Pelayo-Negro AL, Álvarez-Paradelo S, et al. Intermediate Charcot–Marie–Tooth disease: an electrophysiological reappraisal and systematic review. J Neurol. 2017;264(8):1655–77. 10.1007/s00415-017-8474-3 [DOI] [PubMed] [Google Scholar]
- 39.Claeys KG, Züchner S, Kennerson M, Berciano J, Garcia A, Verhoeven K, et al. Phenotypic spectrum of dynamin 2 mutations in Charcot-Marie-Tooth neuropathy. Brain. 2009;132(Pt 7):1741–52. Epub 06/05. 10.1093/brain/awp115 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Haberlová J, Mazanec R, Ridzoň P, Baránková L, Nürnberg G, Nürnberg P, et al. Phenotypic Variability in a Large Czech Family with a Dynamin 2–Associated Charcot-Marie-Tooth Neuropathy. J Neurogenet. 2011;25(4):182–8. 10.3109/01677063.2011.627484 [DOI] [PubMed] [Google Scholar]
- 41.Jeub M, Bitoun M, Guicheney P, Kappes-Horn K, Strach K, Druschky KF, et al. Dynamin 2-related centronuclear myopathy: Clinical, histological and genetic aspects of further patients and review of the literature. Clin Neuropathol. 2008;27(6):430–8. 10.5414/npp27430 [DOI] [PubMed] [Google Scholar]
- 42.Chandhok G, Lazarou M, Neumann B. Structure, function, and regulation of mitofusin-2 in health and disease. Biol Rev Camb Philos Soc. 2017. 10.1111/brv.12378 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ericson U, Ansved T, Borg K. Charcot-Marie-Tooth disease–muscle biopsy findings in relation to neurophysiology. Neuromuscular Disorders. 1998;8(3):175–81. 10.1016/S0960-8966(98)00018-2. [DOI] [PubMed] [Google Scholar]
- 44.González-Jamett AM, Baez-Matus X, Olivares MJ, Hinostroza F, Guerra-Fernández MJ, Vasquez-Navarrete J, et al. Dynamin-2 mutations linked to Centronuclear Myopathy impair actin-dependent trafficking in muscle cells. Sci Rep. 2017;7(1):4580 10.1038/s41598-017-04418-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Renwick NM, Goldfarb LG, Sambuughin N, Hinshaw JE, Toro C, Platonov FA. Progressive neuromuscular syndromes linked to dynamin-2 mutations. J Mult Scler. 2016;3(1):1000163. [Google Scholar]
- 46.Duis J, Dean S, Applegate C, Harper A, Xiao R, He W, et al. KIF5A mutations cause an infantile onset phenotype including severe myoclonus with evidence of mitochondrial dysfunction. Annals of neurology. 2016;80(4):633–7. Epub 2016/08/24. 10.1002/ana.24744 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Guinto CO, Diarra S, Diallo S, Cissé L, Coulibaly T, Diallo SH, et al. A novel mutation in KIF5A in a Malian family with spastic paraplegia and sensory loss. Ann Clin Transl Neurol. 2017;4(4):272–5. 10.1002/acn3.402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Landouré G, Zdebik A, Martinez TL, Burnett BG, Stanescu HC, Inada H, et al. Mutations in TRPV4 cause Charcot-Marie-Tooth disease type 2C. Nat Genet. 2010;42(2):170–4. 10.1038/ng.512 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Rouzier C, Bannwarth S, Chaussenot A, Chevrollier A, Verschueren A, Bonello-Palot N, et al. The MFN2 gene is responsible for mitochondrial DNA instability and optic atrophy 'plus' phenotype. Brain. 2012;135(1):23–34. [DOI] [PubMed] [Google Scholar]
- 50.Ylikallio E, Pöyhönen R, Zimon M, De Vriendt E, Hilander T, Paetau A, et al. Deficiency of the E3 ubiquitin ligase TRIM2 in early-onset axonal neuropathy. Hum Mol Genet. 2013;22(15):2975–83. 10.1093/hmg/ddt149 [DOI] [PubMed] [Google Scholar]
- 51.Houlden H, Laura M, Wavrant-De Vrièze F, Blake J, Wood N, Reilly MM. Mutations in the HSP27 (HSPB1) gene cause dominant, recessive, and sporadic distal HMN/CMT type 2. Neurology. 2008;71(21):1660–8. 10.1212/01.wnl.0000319696.14225.67 [DOI] [PubMed] [Google Scholar]
- 52.Evgrafov OV, Mersiyanova I, Irobi J, Van Den Bosch L, Dierick I, Leung CL, et al. Mutant small heat-shock protein 27 causes axonal Charcot-Marie-Tooth disease and distal hereditary motor neuropathy. Nat Genet. 2004;36(6). [DOI] [PubMed] [Google Scholar]
- 53.Kennerson ML, Nicholson GA, Kaler SG, Kowalski B, Mercer JF, Tang J, et al. Missense mutations in the copper transporter gene ATP7A cause X-linked distal hereditary motor neuropathy. Am J Hum Genet. 2010;86(3):343–52. 10.1016/j.ajhg.2010.01.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Vulpe C, Levinson B, Whitney S, Packman S, Gitschier J. Isolation of a candidate gene for Menkes disease and evidence that it encodes a copper-transporting ATPase. Nat Genet. 1993;3(1):7–13. 10.1038/ng0193-7 [DOI] [PubMed] [Google Scholar]
- 55.Rebelo AP, Saade D, Pereira CV, Farooq A, Huff TC, Abreu L, et al. SCO2 mutations cause early-onset axonal Charcot-Marie-Tooth disease associated with cellular copper deficiency. Brain. 2018;141(3):662–72. 10.1093/brain/awx369 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Link CD, Fonte V, Hiester B, Yerg J, Ferguson J, Csontos S, et al. Conversion of green fluorescent protein into a toxic, aggregation-prone protein by C-terminal addition of a short peptide. J Biol Chem. 2006;281(3):1808–16. 10.1074/jbc.M505581200 [DOI] [PubMed] [Google Scholar]
- 57.Baird NA, Douglas PM, Simic MS, Grant AR, Moresco JJ, Wolff SC, et al. HSF-1-mediated cytoskeletal integrity determines thermotolerance and life span. Science. 2014;346(6207):360–3. 10.1126/science.1253168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zhao M, Maani N, Dowling JJ. Dynamin 2 (DNM2) as cause of, and modifier for, human neuromuscular disease. Neurotherapeutics. 2018;15(4):966–75. 10.1007/s13311-018-00686-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Cowling BS, Chevremont T, Prokic I, Kretz C, Ferry A, Coirault C, et al. Reducing dynamin 2 expression rescues X-linked centronuclear myopathy. J Clin Invest. 2014;124(3):1350–63. 10.1172/JCI71206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sidiropoulos PN, Miehe M, Bock T, Tinelli E, Oertli CI, Kuner R, et al. Dynamin 2 mutations in Charcot-Marie-Tooth neuropathy highlight the importance of clathrin-mediated endocytosis in myelination. Brain. 2012;135(5):1395–411. [DOI] [PubMed] [Google Scholar]
- 61.Karle KN, Möckel D, Reid E, Schöls L. Axonal transport deficit in a KIF5A (-/-) mouse model. Neurogenetics. 2012;13(2):169–79. 10.1007/s10048-012-0324-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Feely SM, Laura M, Siskind CE, Sottile S, Davis M, Gibbons VS, et al. MFN2 mutations cause severe phenotypes in most patients with CMT2A. Neurology. 2011;76(20):1690–6. 10.1212/WNL.0b013e31821a441e [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Bannerman P, Burns T, Xu J, Miers L, Pleasure D. Mice hemizygous for a pathogenic mitofusin- 2 allele exhibit hind limb/foot gait deficits and phenotypic perturbations in nerve and muscle. PLoS One. 2016;11(12):e0167573 10.1371/journal.pone.0167573 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Strickland AV, Rebelo AP, Zhang F, Price J, Bolon B, Silva JP, et al. Characterization of the mitofusin 2 R94W mutation in a knock‐in mouse model. J Peripher Nerv Syst. 2014;19(2):152–64. 10.1111/jns5.12066 [DOI] [PubMed] [Google Scholar]
- 65.Rocha AG, Franco A, Krezel AM, Rumsey JM, Alberti JM, Knight WC, et al. MFN2 agonists reverse mitochondrial defects in preclinical models of Charcot-Marie-Tooth disease type 2A. Science. 2018;360(6386):336 10.1126/science.aao1785 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Seburn KL, Morelli KH, Jordanova A, Burgess RW. Lack of neuropathy-related phenotypes in Hint1 knockout mice. J Neuropathol Exp Neurol. 2014;73(7):693–701. 10.1097/NEN.0000000000000085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.O'Reilly LP, Luke CJ, Perlmutter DH, Silverman GA, Pak SC. C. elegans in high-throughput drug discovery. Adv Drug Deliv Rev. 2013;69–70:247–53. 10.1016/j.addr.2013.12.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Li J, Pak SC, O'Reilly LP, Benson JA, Wang Y, Hidvegi T, et al. Fluphenazine reduces proteotoxicity in C. elegans and mammalian models of alpha-1-antitrypsin deficiency. PloS One. 2014;9(1):e87260–e. 10.1371/journal.pone.0087260 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Sleigh JN, Buckingham SD, Esmaeili B, Viswanathan M, Cuppen E, Westlund BM, et al. A novel Caenorhabditis elegans allele, smn-1(cb131), mimicking a mild form of spinal muscular atrophy, provides a convenient drug screening platform highlighting new and pre-approved compounds. Hum Mol Genet. 2011;20(2):245–60. 10.1093/hmg/ddq459 [DOI] [PubMed] [Google Scholar]
- 70.Patten SA, Aggad D, Martinez J, Tremblay E, Petrillo J, Armstrong GA, et al. Neuroleptics as therapeutic compounds stabilizing neuromuscular transmission in amyotrophic lateral sclerosis. JCI insight. 2017;2(22):e97152 10.1172/jci.insight.97152 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Schmeisser K, Fardghassemi Y, Parker JA. A rapid chemical-genetic screen utilizing impaired movement phenotypes in C. elegans: Input into genetics of neurodevelopmental disorders. Exp Neurol. 2017;293:101–14. 10.1016/j.expneurol.2017.03.022 [DOI] [PubMed] [Google Scholar]
- 72.Rizk A, Paul G, Incardona P, Bugarski M, Mansouri M, Niemann A, et al. Segmentation and quantification of subcellular structures in fluorescence microscopy images using Squassh. Nat Protoc. 2014;9(3):586–96. 10.1038/nprot.2014.037 [DOI] [PubMed] [Google Scholar]
- 73.Byrne JJ, Soh MS, Chandhok G, Vijayaraghavan T, Teoh J-S, Crawford S, et al. Disruption of mitochondrial dynamics affects behaviour and lifespan in Caenorhabditis elegans. Cellular and Molecular Life Sciences. 2019:1–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, et al. Fiji: an open-source platform for biological-image analysis. Nat Methods. 2012;9(7):676–82. 10.1038/nmeth.2019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Sommer C, Strähle C, Köthe U, Hamprecht FA, editors. Ilastik: Interactive learning and segmentation toolkit. Eighth IEEE International Symposium on Biomedical Imaging (ISBI); 2011; Chicago, IL, USA: Proceedings.
- 76.Scurry AN, Heredia DJ, Feng CY, Gephart GB, Hennig GW, Gould TW. Structural and functional abnormalities of the neuromuscular junction in the Trembler-J homozygote mouse model of congenital hypomyelinating neuropathy. J Neuropathol Exp Neurol. 2016;75(4):334–46. 10.1093/jnen/nlw004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Cipriani S, Phan V, Médard JJ, Horvath R, Lochmüller H, Chrast R, et al. Neuromuscular junction changes in a mouse model of Charcot-Marie-Tooth Disease Type 4C. Int J Mol Sci. 2018;19(12):E4072 10.3390/ijms19124072 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ang ET, Schäfer R, Baltensperger R, Wernig A, Celio M, Oliver SS. Motor axonal sprouting and neuromuscular junction loss in an animal model of Charcot-Marie-Tooth disease. J Neuropathol Exp Neurol. 2010;69(3):281–93. 10.1097/NEN.0b013e3181d1e60f [DOI] [PubMed] [Google Scholar]
- 79.Sleigh JN, Grice SJ, Burgess RW, Talbot K, Cader MZ. Neuromuscular junction maturation defects precede impaired lower motor neuron connectivity in Charcot-Marie-Tooth type 2D mice. Human Molecular Genetics. 2014;23(10):2639–50. 10.1093/hmg/ddt659 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Gupta BP, Rezai P. Microfluidic approaches for manipulating, imaging, and screening C. elegans. Micromachines. 2016;7(7):123 10.3390/mi7070123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Brenner S. The genetics of Caenorhabditis elegans. Genetics. 1974;77(1):71–94. Epub 1974/05/01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.MBF Bioscience. WormLab®4 user guide: MBF Bioscience, Williston, VT USA; 2018. Available from: https://www.mbfbioscience.com/help/wormlab4/Content/tutorials.htm.
- 83.Koelle M. Behaviour. 2006. In: WormBook [Internet]. The C. elegans Research Community. Available from: http://www.wormbook.org/chapters/www_behavior/behavior.html#sec7_1.
- 84.Solis GM, Petrascheck M. Measuring Caenorhabditis elegans life span in 96 well microtiter plates. J Vis Exp. 2011;49:2496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Van Raamsdonk JM, Hekimi S. FUdR causes a twofold increase in the lifespan of the mitochondrial mutant gas-1. Mech Ageing Dev. 2011;132(10):519–21. 10.1016/j.mad.2011.08.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Feldman N, Kosolapov L, Ben-Zvi A. Fluorodeoxyuridine improves Caenorhabditis elegans proteostasis independent of reproduction onset. PLoS One. 2014;9(1):e85964 10.1371/journal.pone.0085964 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.García-González AP, Ritter AD, Shrestha S, Andersen EC, Yilmaz LS, Walhout AJM. Bacterial metabolism affects the C. elegans response to cancer chemotherapeutics. Cell. 2017;169(3):e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Teoh JS, Soh MS, Byrne JJ, Neumann B. Quantitative approaches for studying cellular structures and organelle morphology in Caenorhabditis elegans. J Vis Exp. 2019;149:e59978 10.3791/59978 (2019). [DOI] [PubMed] [Google Scholar]
- 89.Richmond JE, Jorgensen EM. One GABA and two acetylcholine receptors function at the C. elegans neuromuscular junction. Nat Neurosci. 1999;2(9):791–7. 10.1038/12160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.R Core Team. R: A language and environment for statistical computing: R Foundation for Statistical Computing, Vienna, Austria; 2014. Available from: http://www.R-project.org/. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(A) Thrash rates compared between wild-type (WT) and the nine mutant strains, across three different ages, larval stage 1 (L1), larval stage 4 (L4), 3-day old adults (A3) and 7-day old adults (A7). (B) Rate of body bends of WT and CMT2 mutant animals quantified across the same ages as in (A). Each dot in (A) and (B) represents a single animal (n ≥ 30). One-way ANOVA with Dunnett’s post hoc tests were used to compare rates of thrash or body bend between WT and mutant animals in (A) and (B). Data is represented as mean ± S.E.M. **P < 0.01, ***P < 0.001, ****P < 0.0001, ns = not significant.
(PDF)
(A) Thrash rates compared between 7-day old adult non-transgenic mutant strains and their transgenic siblings. All transgenic animals carry fosmids containing the corresponding wild-type version of the gene, except for fzo-1, for which a Pmyo-3::fzo-1::unc-54 3`UTR was used, and cua-1, which expresses a Pcua-1::cua-1::cua-1 3’UTR plasmid. Data is represented as mean ± S.E.M, with symbols showing individual animals. (B) Rate of body bends compared between 7-day old adult non-transgenic nep-2 mutants and their transgenic siblings expressing wild-type copies of nep-2 from a fosmid. For (A) and (C), *P < 0.05, ****P < 0.0001 from Student’s t-tests.
(PDF)
Schematic of the workflow, from synchronization via bleaching and washing, to plating of synchronized 3-day old adult animals in a 96-well plate for experimentation.
(PDF)
(A) Original muscle segment must contain at least one full visible oblique muscle cell. (B) Distinct myosin fibre is classified and segregated from the unwanted background (in asterisk) in ilastik. (C) Skeletonization of image in Fiji. The image was skeletonized to filter out the border pixels, leaving behind only the skeletal remnants that become the topological representations of the original fibres. Measurement of each fibre was performed following skeletonization. Fibres that were 0 µm or more than 250 µm were excluded. Scale bar represents 25 µm.
(PDF)
(A-F) The variance of all except for nep-2(ok2846) was statistically different compared to wild-type (WT). Statistics were performed using F test for variances, significance set at P ≤ 0.05. Experiments were performed on 3-day old adult animals that carried stEx30 transgene.
(PDF)
(DOCX)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.