Abstract
Background
Human infection with avian influenza H5N1 is an emerging infectious disease characterised by respiratory symptoms and a high fatality rate. Previous studies have shown that the human infection with avian influenza H5N1 could also target organs apart from the lungs.
Methods
We studied post-mortem tissues of two adults (one man and one pregnant woman) infected with H5N1 influenza virus, and a fetus carried by the woman. In-situ hybridisation (with sense and antisense probes to haemagglutinin and nucleoprotein) and immunohistochemistry (with monoclonal antibodies to haemagglutinin and nucleoprotein) were done on selected tissues. Reverse-transcriptase (RT) PCR, real-time RT-PCR, strand-specific RT-PCR, and nucleic acid sequence-based amplification (NASBA) detection assays were also undertaken to detect viral RNA in organ tissue samples.
Findings
We detected viral genomic sequences and antigens in type II epithelial cells of the lungs, ciliated and non-ciliated epithelial cells of the trachea, T cells of the lymph node, neurons of the brain, and Hofbauer cells and cytotrophoblasts of the placenta. Viral genomic sequences (but no viral antigens) were detected in the intestinal mucosa. In the fetus, we found viral sequences and antigens in the lungs, circulating mononuclear cells, and macrophages of the liver. The presence of viral sequences in the organs and the fetus was also confirmed by RT-PCR, strand-specific RT-PCR, real-time RT-PCR, and NASBA.
Interpretation
In addition to the lungs, H5N1 influenza virus infects the trachea and disseminates to other organs including the brain. The virus could also be transmitted from mother to fetus across the placenta.
Introduction
A pandemic outbreak of human infection with avian influenza H5N1 currently poses a potentially serious health threat worldwide. Since the outbreak of infection with avian influenza H5N1 virus in 2003, WHO has reported 277 laboratory confirmed cases in ten countries with a mortality rate of about 60%.1 So far the virus has spread only from animals to human beings. However, human-to-human transmission potentiated by viral genomic mutation and reassortment of genomic subunits could be imminent. Recently, the first cases of probable human-to-human transmission have been reported.2, 3 The H5N1 influenza A virus is a negative-stranded RNA virus in which the genome consists of eight segments encoding ten viral proteins including haemagglutinin, neuraminidase, polymerase proteins, and nucleoprotein.4
Little is known about the specific effects in organs and cells targeted by the virus. The infection initially seemed to be restricted to the lungs, but later reports5, 6 have suggested that influenza A H5N1 could disseminate beyond the lungs. For various reasons (eg, religion), full autopsies of H5N1-infected human cases can often not be obtained. Accordingly, only a few reports2, 5, 7, 8, 9 have described histopathology and virus distribution in H5N1 cases. Studies using in-situ hybridisation to detect viral genomic sequences in target cells have not been reported thus far.
We present clinicopathological data from H5N1 autopsies of two unrelated Chinese cases, as well as the histopathological changes and pattern of infection in the placenta and fetus from one of the patients, who was pregnant at the time of death. To gain further insight into the tissue tropism of influenza A H5N1 virus, we used in-situ hybridisation and immunohistochemistry to analyse viral localisation in various organs. Reverse transcription (RT) PCR, real-time RT-PCR, and nucleic acid sequence-based amplification (NASBA) H5 detection assays were also done to detect viral RNA in tissue samples, as well as strand-specific RT-PCR.
Methods
Patients
The clinical data of patient 1 have previously been published in detail.10 A 24-year-old Chinese woman from China's Anhui province who was 4 months pregnant presented with a 6-day history of fever, cough, and dyspnoea. 2 weeks before admission, she had handled birds, several of which had died. On admission, she was lymphopenic, confused, and irritable, had bilateral infiltration on chest radiograph, and substantially reduced oxygen saturation. She was placed on a ventilator and treated with antibiotics, corticosteroids (hydrocortisone 400 mg on day 6 and day 7, and methylprednisolone 160 mg on day 8 and 240 mg on day 9), and fluids, but died 62 h after admission, 9 days after the onset of symptoms. No antiviral treatment was given.
Patient 2 was a 35-year-old Chinese man from China's Jiangxi province who had a 6-day history of fever and productive cough. He had participated in selling birds, of which several had died. On admission, chest radiographs showed evidence of pneumonia, and laboratory results showed abnormally increased hepatic-associated and cardiac-associated enzymes and lymphopenia. He was treated with antibiotics. Corticosteroids were given initially with methylprednisolone 40 mg on day 10 and then 120 mg per day for 17 days. Antiviral treatment was given, including rimantadine 100 mg twice daily on days 10 and 11 and then oseltamivir 150 mg per day from day 11 for 10 days. After admission, he became increasingly irritable and confused, followed by lowered consciousness. He developed respiratory failure and mechanical ventilation was initiated. In a sputum culture, several gram-positive microorganisms and Candida albicans were isolated. Antifungal drugs were added to his treatment. He developed multiple organ failure and died 27 days after the onset of symptoms.
The Chinese Centre for Disease Control and Prevention confirmed human infection with avian influenza H5N1 in both patients. RT-PCR detected H5N1 viral sequences in nasal swabs and nasopharyngeal aspirates, which were obtained on day 6 of illness for patient 1 and day 10 for patient 2. Viruses were isolated from the nasopharyngeal aspirate cultures, and designated as influenza A/Anhui/1/2005 virus10 in patient 1 and A/Jiangxi/1/2005 virus in patient 2. The haemagglutinin genes of viruses in patient 1 (GenBank accession number: DQ371928)10 and patient 2 (webappendix)11 were sequenced. The receptor-binding sites of both viruses were identical to those of previous H5N1 isolates.12 H5N1 viruses isolated from both patients were susceptible to both the M2 inhibitors amantadine and rimantadine, and the neuraminidase inhibitors oseltamivir and zanamivir.
Both cadavers were stored at 4°C and underwent autopsy about 18–20 h after death. The autopsies were done following conventional protocols and strict safety procedures.13 Tissue samples from all major organs and tissues were taken and fixed in 4% formalin. The brain of patient 1 was not available for investigation.
Immunohistochemistry
Immunohistochemistry was done on the basis of the technique of Lin and colleagues, with antigen retrieval by a standard technique.14, 15 To detect viral antigen, tissue slides of 4 μm thickness were incubated with mouse monoclonal antibodies to nucleoproteins and haemagglutinin. Furthermore, monoclonal antibodies to the following cell markers were used: CD68 (for macrophages), CD3 (T lymphocytes), CD20 (B lymphocytes), CD8 (cytotoxic T cells), S100 (dendritic cells), cytokeratin AE1/AE3 (epithelial cells), surfactant protein A (type II pneumoncytes), tubulin-β (ciliated epithelial cells), placental alkaline phosphatase (PLAP, syncytiotrophoblasts), E-cadherin (cytotrophoblasts),16 neurofilament (neurons), neuron-specific enolase (neurons), and factor VIII (endothelial cells, webtable).17 For controls, we used unrelated antibodies in place of the primary antibody.
In-situ hybridisation
For the development of probes, we used haemagglutinin (GenBank accession number DQ100556) and nucleoprotein gene sequences (DQ100560) of the H5N1 A/black-headed gull/Qinghai/1/2005 virus, which was recently isolated from a migratory bird at China's Qinghai lake.18 Plasmids were generated by cloning of the full haemagglutinin gene (1779 bp) and full nucleoprotein gene (1565 bp) into a plasmid vector PGEM-T (Promega, Madison, WI, USA) yielding pGEM-HA for haemagglutinin and pGEM-NP for nucleoprotein. Both plasmids were linearised with appropriate restriction enzymes. Two sense and two antisense RNA probes were prepared by in-vitro transcription with T7 and Sp6 RNA polymerase (Promega) in the presence of digoxigenin-UTP (Roche Diagnostics, Penzberg, Germany). Since H5N1 is a negative-stranded RNA virus, sense probes were defined as the probes that detect the viral RNA (negative-stranded), whereas antisense probes detected mRNA and complement RNA (cRNA), which are both positive-stranded.
Briefly, before hybridisation, all solutions were prepared with diethyl pyrocarbonate (DEPC)-treated water.19 After deparaffinisation and rehydration, tissue sections of 4 μm thickness were treated with proteinase K digestion or microwave heating. Tissue sections were then incubated with a hybridisation cocktail containing 50 μg/mL of one of the four sense and antisense probes at 45°C for 16 h. All sense and antisense probes were applied separately on consecutive tissue sections. After blocking with horse serum (1:100), sections were incubated with alkaline phosphatase-labelled digoxigenin antibody (1:500, Roche Diagnostics, Penzberg, Germany) for 1 h, and the reaction products were colourised with nitroblue tetrazolium/5-bromo-4-choloro-3-indolyl phosphate (Promega).
As a positive control, we used brain tissue samples of a black-headed gull, for which H5N1 infection of the brain was confirmed by viral isolation.18 We used lung tissues from a mouse infected with H9N2 influenza virus as a negative control. Negative controls also included an unrelated antisense probe against the fragment of the polymerase gene (R1AB) of the severe acute respiratory syndrome-associated coronavirus (SARS-CoV),20 as well as H5N1 in-situ hybridisation probes to tissues (including lung and tracheal) obtained from seven adults who died from infectious lung diseases other than H5N1 influenza (four, SARS; one, purulent bronchitis; two, pneumonia), one adult who died from a non-infectious disease (gastric ulcer), one pregnant woman who died from an amniotic embolism, and one aborted fetus.
We identified the cell types infected by the virus with double labelling by combining in-situ hybridisation for viral genomic sequences and immunohistochemistry for one of the cell-associated markers. To identify placental cells and cerebral neurons containing viral sequences or antigens, consecutive sections adjacent to the sections used for in-situ hybridisation or immunohistochemistry were immunostained with CD68, E-cadherin, PLAP, neurofilament, or neuron-specific enolase. Tissues from the pharynx, nose and paranasal sinuses, and lymph nodes other than hilar nodes were not available for investigation.
On preliminary investigation of lung tissues, we noted a contrast between the extent of histological damage and the limited number of positive pneumocytes (patient 1) or absence of positive pneumocytes (patient 2) shown by in-situ hybridisation and immunohistochemistry studies. Therefore, we did extensive pulmonary sampling for further histological assessment to ensure that the results were representative.
Expected localisation of signals from in-situ hybridisation and immunohistochemistry
After receptor-mediated endocytosis of the virus, the polymerase proteins, nucleoprotein, and encapsidated RNA segments migrate to the nucleus of the infected cell. In the nucleus, RNA segments are transcribed into mRNA and cRNA. mRNA is subsequently translated to viral proteins (eg, haemagglutinin and nucleoprotein) in the cytoplasm. Newly synthesised nucleoprotein is transported back to the nucleus. cRNAs function as antigenomic templates for the production of progeny RNA segments in the nucleus. Assembly of progeny gene segments and proteins occurs in the cytoplasm.4
Therefore, sense and antisense signals after in-situ hybridisation for nucleoprotein and haemagglutinin could be seen in both the nucleus and cytoplasm of infected cells, as well as immunohistochemical signals for nucleoprotein in the nucleus and for haemagglutinin in the cytoplasm. Since antisense probes hybridise to mRNA and cRNA, a positive signal would probably indicate active viral replication.
RT-PCR, real-time RT-PCR, and NASBA
RNA was extracted from deparaffinised tissue samples, or directly extracted from formalin-fixed tissues after overnight incubation with proteinase K (10 μg/μL, Ameresco, Cleveland, OH, USA). We did RNA extraction with Trizol (Invitrogen, CA, USA), and PCR as previously described.21 The haemagglutinin gene of the H5N1 virus was detected with H5for as the forward primer and H5rev as the reverse primer for H5 gene amplification (panel ). Reamplification was done for specific samples with the same set of primers if no band was seen in gel electrophoresis. We obtained negative controls for RT-PCR from uninfected tissues (eg, lung, brain, placenta, and intestine) from three patients who died from non-infectious diseases. RT-PCR for glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was assessed in parallel as an internal control.
Panel. Nucleotide sequences of primers used in RT-PCR, strand-specific RT-PCR, and NASBA.
H5for
5′ gTg AYA ATg AAT gYA Tgg AA 3′
H5rev
5′ CCA IAA AgA YAg ACC AgC TA 3′
tag-H5for
5′ TCT Agg Tgg CgA gTA AgT gAY AAT gAA TgY ATg gAA 3′
tag-H5rev
5′ TCT Agg Tgg CgA gTA ACC AIA AAg AYA gAC CAg CTA 3′
Tag
5′ TCT Agg Tgg CgA gTA A 3′
NASBA capture probe
DIG-GCRAGTTCYCTAGCACTGGCAAT
NASBA detection probe
Biotin-GATGCAAGGTCGCATATGAG
To detect viral RNA in fetal tissues, real-time RT-PCR (H5 avian influenza virus Nucleic Acid Amplification Fluorescent Quantitative Detection Kit, PG Biotech, Shenzhen, China) was used as recommended by the manufacturer and the national standards of the People's Republic of China.
Strand-specific RT-PCR was undertaken on the basis of the technique of Yue and co-workers,22 with minor modification. Briefly, two-part reactions were used. First, the RT reaction was done in the presence of tagged primer, tag-H5for or tag-H5rev (panel). A third of cDNA products then underwent PCR with primers Tag/H5rev or Tag/H5for. We reamplified under the same PCR conditions for a specific sample if no band was seen after gel analysis of the first PCR reaction.
Virus-specific RNA was also detected with the avian influenza virus (H5 subtype) NASBA diagnostic test kit (MP version, Hong Kong DNA Chips, Hong Kong). The test was done as previously described23 with H5-specific capture and detection probes (panel). Absorbance of the amplified product was measured at 405 nm by an ELISA plate reader (Bio-Rad, Hercules, CA, USA). A negative control from the test kit was also included for H5 detection. The study was approved by the internal review board and ethics committee of the Peking University Health Science Centre.
Role of the funding source
The sponsor of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Results
The microscopic features of both patients were similar, apart from more extensive fibroproliferative changes in the lungs of patient 2. Both lungs of patient 1 showed features of diffuse alveolar damage (figure 1A ) and focal desquamation of epithelial cells into alveolar spaces without evidence of type II pneumocyte hyperplasia. In the lungs of patient 2, patchy foci of consolidated bronchopneumonia and areas of fibrosis were seen. We found variable numbers of macrophages in the alveoli (especially in patient 2), and moderate numbers of scattered neutrophils and rare lymphocytes in the interstitial spaces. Both patients had substantially depleted lymphoid tissue in the spleen, lymph nodes, and mucosal lymphoid tissue in the gastrointestinal tract. The liver in both patients had spotty necrosis. In both patients, we detected very low numbers of macrophages with haemophagocytosis in the spleen, lymph nodes, and liver. The kidneys showed extensive tubular necrosis. Other organ systems showed no pronounced histological changes, apart from hypertrophy in the thymus of patient 2.
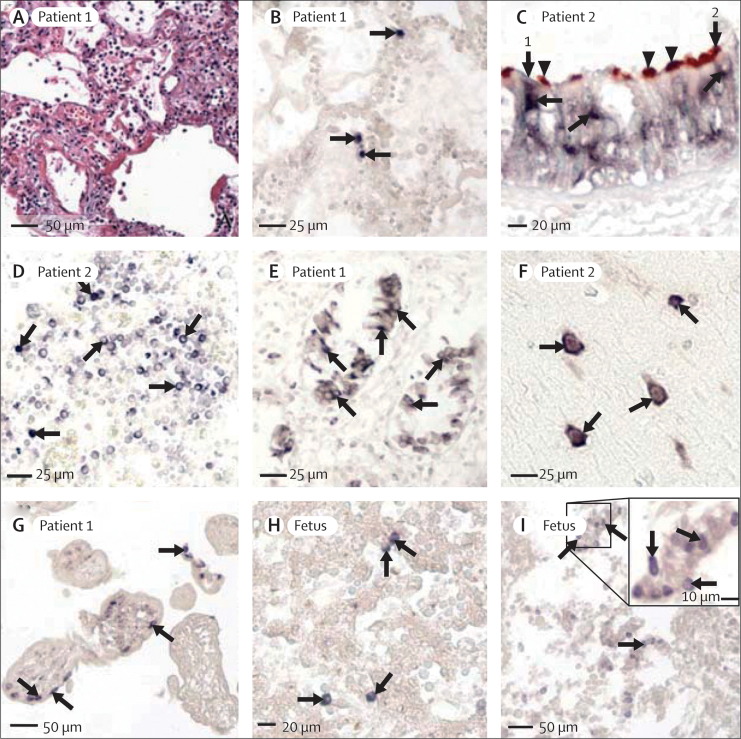
Figure 1.
In-situ hybridisation locating gene sequences of H5N1 viral antigens (nucleoprotein and haemagglutinin)
Signals seen with nitroblue tetrazolium/5-bromo-4-choloro-3-indolyl phosphate (purple-blue) and immunohistochemical signals with 3-amino-9-ethylcarbazole (double labelling, brown-red). No counterstaining done unless stated otherwise. (A) Lung tissue showing severe damage, hyaline membrane formation, and oedema (by haematoxylin and eosin staining), by contrast with limited number of cells positive for in-situ hybridisation in lung tissue of patient 1 (figure 1B). (B) Positive signals (with nucleoprotein sense probe) in nuclei of isolated pneumocytes (arrows). (C) Double labelling of in-situ hybridisation (with nucleoprotein antisense probe) and immunohistochemistry (with tubulin-β antibody; brown, arrowheads) showing dark-blue viral genomic sequences (arrows) in cytoplasm of tubulin-negative non-ciliated cell (arrow 1) and tubulin-positive ciliated cell (arrow 2) in trachea. No signal seen in nuclei (lightly counterstained with methyl-green). (D) Positive signals (with nucleoprotein sense probe) in several mononuclear cells in lymph node (arrows, lightly counterstained with methyl-green). (E) Positive signals (with nucleoprotein antisense probe) in mucosal epithelial cells of small intestine (arrows, lightly counterstained with methyl-green). (F) Positive signals (with nucleoprotein sense probe) in brain cells (arrows) from left parietal lobe (lightly counterstained with methyl-green), mainly located in cytoplasm and confirmed to be neurons from immunostaining for neurofilament or neuron-specific enolase (webfigures 1O and 1P). (G) Positive signals (with nucleoprotein antisense probe) in large mononuclear cells with morphological features of cytotrophoblasts in periphery of chorionic villus (arrows). Cells confirmed as cytotrophoblasts (webfigure 4D). No positive signals noted in any syncytiotrophoblasts. (H) Positive signals (with nucleoprotein sense probe) in fetal liver cells (arrows), confirmed as Kupffer cells (webfigure 1F). (I) Positive signals (with haemagglutinin sense probe) in bronchiolar epithelial cells in fetal lung tissue (arrows).
The placenta showed development appropriate for the length of gestation. We saw scattered foci of syncytiotrophoblast necrosis, sometimes with associated dystrophic calcification. Whether this finding was induced virally or was the sole result of maternal shock is unclear. Acute necrotising deciduitis was detected focally, and regarded as consistent with maternal shock.
Fetal tissues mostly showed no specific histopathological findings, and development was also consistent with gestational age. However, sections of fetal lung showed oedema and very small numbers of scattered interstitial neutrophils, which raised the possibility of mild acute interstitial pneumonitis, although this appearance was notably less severe than that seen in patients 1 and 2 (webfigure 1).
Sense and antisense probes for in-situ hybridisation detected viral genomes focally in tissue samples from various organs (table 1 ). The two sets of probes (for haemagglutinin and nucleoprotein) generated identical staining results. In both autopsies, the trachea, small intestines, and lymph nodes showed positive signals of in-situ hybridisation, although pneumocytes were positive only in patient 1. In the lungs, sense probes hybridised in the nuclei of pneumocytes (figure 1B), whereas antisense probes hybridised in both the cytoplasm and nuclei. In all other organs with positive signals, both sense and antisense were present mainly in the cytoplasm of infected cells.
Table 1.
Results of in-situ hybridisation and immunohistochemistry in selected organs and cell types
|
Patient 1 |
Patient 2 |
Fetus |
||||
|---|---|---|---|---|---|---|
| ISH* | IHC† | ISH | IHC | ISH | IHC | |
| Trachea | +/+ | +/+ | +/+ | +/+ | .. | .. |
| Bronchi | −/− | −/− | −/− | −/− | +/+ | +/+ |
| Alveolar pneumocytes | +/+ | +/+ | −/− | −/− | +/+ | +/+ |
| Lymph nodes | +/+‡ | .. | +/+‡ | +/+‡ | .. | .. |
| Spleen | −/− | −/− | −/− | −/− | .. | .. |
| Heart | −/− | −/− | −/− | −/− | −/− | −/− |
| Endothelial cells | −/− | −/− | −/− | −/− | −/− | −/− |
| Hepatocytes | −/− | −/− | −/− | −/− | −/− | .. |
| Kupffer cells | −/− | −/− | −/− | −/− | +/+ | .. |
| Kidneys | −/− | −/− | −/− | −/− | −/− | .. |
| Small intestine | +/+ | −/− | +/+ | −/− | −/− | .. |
| Brain | .. | .. | +/+ | +/+ | .. | .. |
| Syncytiotrophoblasts | −/− | −/− | n/a | n/a | n/a | n/a |
| Cytotrophoblasts | +/+ | +/+ | n/a | n/a | n/a | n/a |
| Hofbauer cells | +/+ | +/+ | n/a | n/a | n/a | n/a |
| Circulating mononuclear cells | −/− | −/− | −/− | +/+ | +/+ | +/+ |
ISH=in-situ hybridisation. IHC=immunohistochemistry. n/a=not applicable. Plus sign=positive. Minus sign=negative.
Results presented for sense/antisense probes (identical results for nucleoprotein and haemagglutinin).
Results presented for nucleoprotein/haemagglutinin signals (nucleoprotein mainly detected in nucleus and haemagglutinin in cytoplasm).
Includes putative T lymphocytes.
In the respiratory tract, we detected positive signals in tracheal epithelial cells and alveolar epithelial cells (figures 1B and 1C, webfigure 1). However, only an estimated 10–20% of epithelial cells in the trachea and about 5% of epithelial cells in the alveoli showed positive signals. Both bronchi and bronchioles were negative. Double labelling combining in-situ hybridisation and immunohistochemistry with tubulin-β antibody showed that both ciliated (tubulin-β-postive) cells and non-ciliated (tubulin-β-negative) cells of the trachea had viral sequences (figure 1C, webfigure 2). We also found putative basal cells to be infected. Double labelling with antibodies for cytokeratin and surfactant protein A showed that the positive alveolar cells were type II pneumocytes (webfigures 1C and 1D). In-situ hybridisation showed no positive staining in endothelial cells, macrophages, lymphocytes, fibroblasts, or any other cell type in the lungs or blood. However, we found positive viral signals in the cytoplasm of mononuclear cells in hilar lymph nodes (figure 1D). Double labelling and consecutive sections showed that cells positive for in-situ hybridisation were T lymphocytes (ie, positive for CD3 and negative for CD68, CD20, and S100, webfigure 3). Additionally, positive signals of intracytoplasmic viral sequences were present in mucosal epithelial cells of the small intestine (>50% in some intestinal segments, figure 1E). We also detected viral sequences in the cytoplasm (and to a much lesser extent in the nuclei) of brain cells from patient 2 (figure 1F, webfigure 1). Double labelling with neural markers neurofilament or neuron-specific enolase showed that these H5N1-positive cells were neurons (webfigures 1O and 1P). Table 2 shows the topographic distribution of cells with positive signals from in-situ hybridisation. No positive signals were seen in the heart, liver, spleen, kidneys, oesophagus, bone marrow, or stomach.
Table 2.
Results of in-situ hybridisation, RT-PCR, and NASBA for H5 detection assay in selected brain regions of patient 2
|
ISH* |
RT-PCR |
NASBA-based H5 detection |
|||
|---|---|---|---|---|---|
| Sense | Antisense | Optical density (405 nm) | Result | ||
| Parietal lobe | + | + | + | 2·424 | + |
| Frontal lobe | − | − | + | 2·499 | + |
| Hippocampus | + | + | + | 2·521 | + |
| Occipital lobe | + | + | + | 2·312 | + |
| Internal capsule | + | + | + | 2·290 | + |
| Temporal lobe | + | + | + | 2·466 | + |
| Amygdala | + | + | + | 1·919 | + |
| Thalamus | + | + | + | 2·295 | + |
| Mesencephalon | − | − | − | 1·490 | + |
| Pons | + | + | + | 2·031 | + |
| Medulla oblongata | − | − | − | 2·469 | + |
ISH=in-situ hybridisation.
Signals obtained with sense and antisense probes were mainly located in cytoplasm of infected neurons.
Placental tissue samples showed a large number of infected cells in chorionic villi (webfigure 4). Most positive cells were localised in the connective tissue core of these villi. These cells were confirmed (by labelling with monoclonal antibodies to CD68 and PLAP in consecutive sections) to be Hofbauer cells but not syncytiotrophoblasts. A subgroup of the cells with positive signals was found in the periphery of chorionic villi (figure 1G), which morphologically resembled cytotrophoblastic cells and were confirmed by immunostaining with E-cadherin antibody on consecutive sections. No positive signal was seen in syncytiotrophoblasts.
In the fetus, in-situ hybridisation identified viral sequences in the lungs, circulating mononuclear cells (webfigure 1), and mononuclear cells in the liver (figure 1H). The latter cells were identified as macrophages (Kupffer cells) by double labelling with antibody to CD68 (webfigure 1). Both sense and antisense probes were positive, mainly in the nuclei of pneumocytes and in a few detached epithelial cells of the bronchi (figure 1I). In Kupffer cells and circulating mononuclear cells, sense and antisense probes showed positive signals in both the cytoplasm and nucleus. The fetal lungs appeared to have more positively stained cells than did the maternal lungs.
The specificity of in-situ hybridisation was established by the results with the negative and positive controls (webfigure 1). The brain sections of the black-headed gull showed almost all neurons with positive signals for in-situ hybridisation, indicating the high detecting sensitivity of this technique.
Distribution of immunohistochemical staining (table 1) was consistent with that of in-situ hybridisation, apart from the absence of viral antigens in the intestines. Positive staining for nucleoprotein and haemagglutinin was detected in pneumocytes (figure 2A ) and cytotrophoblasts and Hofbauer cells in the placenta (figure 2B) in patient 1; as well as in tracheal epithelial cells (figure 2C), the brain (figure 2D), and T lymphocytes in hilar lymph-node tissue (figure 2E) in patient 2. The fetus showed positive staining in bronchial epithelial cells, pneumocytes (figure 2F), and circulating mononuclear cells. Nucleoprotein was mainly detected in the nucleus and haemagglutinin in the cytoplasm. Negative controls validated the specificity of the immunohistochemistry protocol (webfigure 5).
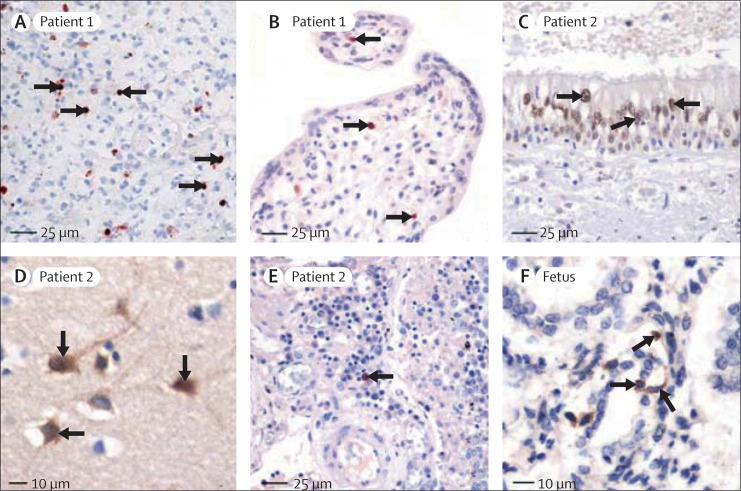
Figure 2.
Immunohistochemical staining for H5N1 viral antigens (nucleoprotein, haemagglutinin)
Positive signals seen with diaminobenzidine (brown; Zymed Laboratories, San Francisco, CA, USA) or 3-amino-9-ethylcarbazole (red-brown; Sigma, St Louis, MO, USA). Slides counterstained with haematoxylin. (A) Positive staining (with nucleoprotein antibody) in nuclei and cytoplasm of pneumocytes (arrows). (B) Positive signals (with nucleoprotein antibody) in nuclei of mononuclear cells (arrows) with morphological features of macrophages in core of chorionic villus. Immunostaining in adjacent sections indicate cells to be Hofbauer cells (webfigure 4). (C) Positive signals (with nucleoprotein antibody) in nuclei of epithelial cells of trachea (arrows). (D) Positive staining (with nucleoprotein antibody) in cytoplasm and nuclei of neurons (arrows) from hippocampus. (E) Positive staining (with haemagglutinin antibody) in mononuclear cell in lymph node (arrow). (F) Positive staining (with haemagglutin antibody) in cytoplasm of pneumocytes (arrows) in fetal lung tissue.
All organs tested showed positive RT-PCR results, apart from the lymph nodes of patient 2 (Table 2, Table 3 , figure 3 ). All non-paraffin-embedded samples were positive for H5 expression. However, paraffin-embedded tissues only showed H5 expression after reamplification of the RT-PCR products. NASBA showed positive results on both types of samples without the need of reamplification.
Table 3.
Results of RT-PCR, strand-specific RT-PCR, real-time RT-PCR, and NASBA for H5 detection in specific tissue samples
|
RT-PCR and strand-specific RT-PCR |
Real-time RT-PCR |
NASBA-based H5 detection assay |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
Patient 1 |
Patient 2 |
Fetus |
Patient 1 |
Patient 2 |
|||||||
| +/− | − | + | +/− | − | + | Optical density (405 nm)* | Result | Optical density (405 nm)* | Result | ||
| Lungs | Y | Y | N | Y | N | Y | Y | 2·579 | Y | 0·961 | Y |
| Trachea | .. | .. | .. | Y | Y | Y | .. | .. | n/a | 1·236 | Y |
| Intestines | Y | Y | N | Y | Y | Y | N | 2·277 | Y | 2·112 | Y |
| Brain | .. | .. | .. | Y | Y | Y | .. | .. | n/a | 2·424 | Y |
| Heart | Y | N | Y | Y | N | N | N | 2·415 | Y | 2·555 | Y |
| Spleen | Y | Y | N | Y | N | N | .. | 1·326 | Y | 1·442 | Y |
| Liver | Y | N | N | Y | N | N | Y | 2·176 | Y | 2·225 | Y |
| Kidneys | Y | Y | N | Y | N | N | N | 2·371 | Y | 1·587 | Y |
| Lymph node | .. | .. | .. | N | Y | N | .. | .. | n/a | 1·107 | Y |
| Placenta | Y | Y | Y | n/a | n/a | n/a | n/a | 1·653 | Y | n/a | n/a |
+/−, −, and + represent total, negative-stranded, and positive-stranded RNA, respectively. Y=positive result. N=negative result. n/a=not applicable.
Samples regarded as H5-positive if absorbance higher than 0·45.
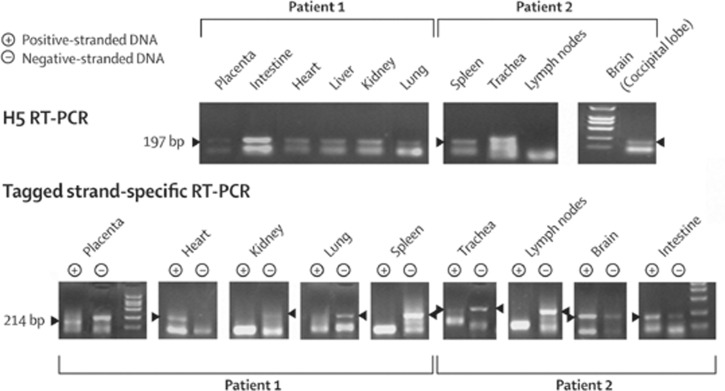
Figure 3.
H5 RT-PCR and tagged strand-specific RT-PCR on selected organs
No positive band in RT-PCR seen for lymph nodes in patient 2.
With real-time RT-PCR, viral RNA was detected in the lungs and liver of the fetus (table 3). GAPDH was an internal control for successful RNA extraction in these assays. Positive-stranded RNA was detected in the heart and placenta of patient 1 and in the lungs, trachea, intestines, and brain of patient 2 (table 3, figure 3). The specificities of the RT-PCR and NASBA were further confirmed by use of the negative controls.
Discussion
Our comprehensive investigation of the tissue tropism of H5N1 influenza virus, based on two adult autopsies and one fetal autopsy, focuses on the localisation of viral genomic sequences and antigens. We present evidence suggesting that the virus disseminates beyond the respiratory system. In addition to the lungs, viral sequences and antigens were found in the cerebral neurons and lymphocytes.
Presence of viral sequences and antigens in the CNS is consistent with the recent isolation of H5N1 virus from cerebrospinal fluid of a boy who died from encephalitis6 with neurological symptoms commonly seen in patients with H5N1 influenza (Gao Zh, unpublished), including the two cases in this study. Brain neurons were found to be infected by the virus. We also saw regional variations in positive neuronal distribution and negative neurons next to positive neurons. Possible reasons might include differing densities of the avian influenza virus receptor in human beings, differences in blood supply pathways and nerve connections that allow virus-target cell contact, and differing viral loads and viral replication stages. The detection of positive-stranded RNA by RT-PCR and in-situ hybridisation could indicate active viral replication in the brain. The virus could reach the CNS by penetrating the blood-brain barrier or by invading the afferent fibres of the olfactory, vagal, trigeminal, and sympathetic nerves after replicating in the respiratory mucosa, as has been shown in animals.24
Our data imply that the virus also infects and actively replicates in the small intestines, which is consistent with previous studies.5 The origin of infection in the intestines could be blood-borne, which is lent support by previous studies isolating live H5N1 virus from the serum6 and plasma.25 However, ingestion of infected respiratory secretion cannot be excluded as a possible route of infection, since H5N1 influenza viruses maintain sialidase activity despite the low pH in the upper digestive tract.26 Although in-situ hybridisation, NASBA, and RT-PCR detected viral RNA in the intestines, immunohistochemistry for viral antigens was negative. This discrepancy is consistent with the findings of Uiprasertkul and colleagues,5 although the reason is still unclear.
In-situ hybridisation and immunohistochemistry detected viral sequences and antigens in lymphocytes in the lymph nodes, and fetal macrophages in the placenta. Circulating mononuclear cells in the fetus and macrophages in the liver were found to harbour viral sequences. Previous in-vitro experiments have shown infection of macrophages by H5N1,27, 28 and ex-vivo experiments have shown that the virus attaches to alveolar macrophages in human lung tissue.29 In addition to viraemia, infected immune cells could also carry the virus to extrapulmonary organs, which has been thought to participate in the pathogenesis of SARS.20
The virus localised to type II pneumocytes in the respiratory tract, which has also been reported previously.5, 29, 30 However, with double labelling, we found viral sequences and antigens in both ciliated and non-ciliated epithelial cells of the trachea (figure 1C), contrasting with previous in-vivo and ex-vivo studies.5, 29 In cultures of human tracheobronchial epithelial cells, H5N1 influenza viruses have been reported to infect mainly ciliated cells, which express mainly avian influenza virus receptors (α-2,3-linked sialic acids), although a limited number of non-ciliated cells (<20% of all infected cells) have also been reported to be infected.31, 32 Some studies have detected only human influenza virus receptors (α-2,6-linked sialic acids) on non-ciliated cells,32 whereas others also have found avian influenza virus receptors in these cells, albeit to a lesser extent.31, 33 Changes in receptor-binding properties of A/Anhui/1/2005 and A/Jiangxi/1/2005 viruses could, in theory, also account for the infection of non-ciliated cells. However, preliminary tests have not revealed any substantial changes in the receptor-binding sites of either virus, compared with previous H5N1 isolates.12
Notably, only a few scattered epithelial cells in the lungs were found to harbour the virus, contrasting with the severe and widespread histopathological changes in the lungs. Since this contrast was unexpected, lung tissue was sampled and analysed extensively, but the number of cells with viral localisation was consistently low in both patients. With the technique's very high detection sensitivity (close to 100%), the percentages of positive epithelial cells recorded in this study could be reasonable estimates of H5N1-infected cells. In view of the low number of infected cells in patient 1 and the absence of cells with positive signals after in-situ-hybridisation in patient 2, direct viral injury to the epithelial cells of the respiratory tract is, in our view, unlikely to cause such severe pathological changes. The lack of histopathological changes in the brain, despite our findings indicating active viral replication in the region, also suggests that viral replication might not be specifically pathogenic. Recent in-vitro and in-vivo studies have indicated that hyperinduction of cytokines and chemokines could take part in the pathogenesis of H5N1 influenza.5, 7, 8, 27, 28, 34
Despite the high number of infected cells in the fetal respiratory system, we saw no evidence of severe damage to the fetal lungs, which greatly contrasts with the extensive damage found in the adult lungs. The absence of severe pulmonary damage (ie, high numbers of infected cells) in the fetus probably indicated an immunological naive status, which would be expected to result in low concentrations of the cytokines or chemokines to which the fetal lung tissues were exposed, and thus reduce or eliminate their induction of tissue damage. This theory is supported by in-vitro experiments27 showing that H5N1-infected neonatal macrophages express much lower amounts of chemokines than H5N1-infected adult macrophages.
Although the intracellular distribution pattern of immunohistochemical signals conformed to our expectations, it did not for signals from in-situ hybridisation. Probes hybridised mainly in the nuclei of pneumocytes and in the cytoplasm of other organs. In mice infected with H5N1 influenza virus, nucleoprotein sense and antisense probes have also hybridised mainly in the cytoplasm of infected cells, although the reason for this finding is unclear.24
RNA analysis with RT-PCR and NASBA assays showed that H5-specific RNA was present in all tissues examined apart from the lymph nodes of patient 2, for which only NASBA showed positive result. This result could be due to the higher sensitivity of NASBA than that of RT-PCR. In fact, RT-PCR needed reamplification of the PCR products on the paraffin-embedded samples, which indicated a lower detecting sensitivity than NASBA.
RT-PCR and NASBA results were generally consistent with those of in-situ hybridisation and immunohistochemistry. However, viral RNA was also seen in viscera and some regions of the brain that showed negative results for both in-situ hybridisation and immunohistochemistry. A similar discrepancy has also been reported in a SARS study,35 which was attributed either to very low copy numbers of RNA and protein in these organs that might not be detectable or to false-positive RT-PCR results. False-positive results might be caused by the presence of virus in blood perfusing the organs without actual viral replication in the tissue parenchyma.35 Detection of positive-stranded RNA in the lung, heart, intestines, placenta, brain, and trachea in our study could imply that viral replication occurs in these organs. The absence of corresponding negative-stranded RNA in the lung and heart could be due to a lower detecting sensitivity of RT-PCR for negative strands than for positive strands.
This study has shown the capacity for human vertical transmission of the H5N1 virus. Transplacental transmission of the H5N1 virus warrants careful investigation, since maternal infections with common human influenza virus are generally thought not to affect the fetus.36 A sero-epidemiological study showed no evidence of transplacental transmission in pregnant women with human influenza infection.37 Our placenta autopsy showed viral genomic sequences in cytotrophoblasts and resident macrophages; furthermore, the virus infected the fetus. Viraemia has been reported in avian influenza virus infections,6, 25 which is by contrast with the rare occurrence of viraemia in human influenza virus infections.38 Therefore, the likelihood of virus reaching the uterus and placenta is probably higher in avian influenza than in human influenza.
The vertical transmission route of avian influenza virus could be similar to that of human cytomegalovirus, which also targets cytotrophoblasts and Hofbauer cells.39 Two possible routes of transplacental transmission have been suggested:39 transcytosis across syncytiotrophoblasts to cytotrophoblasts in chorionic villi, or via infection of invasive cytotrophoblasts in the uterine wall (which could be infected after contact with maternal blood). These infected cells subsequently transmit the virus to the anchoring chorionic villi and could then be transmitted to Hofbauer cells that enter the fetal circulation. The presence of viral sequences and antigens in cytotrophoblasts, Hofbauer cells, and circulating fetal mononuclear cells supports this theory.
We detected viral sequences in cytotrophoblasts of chorionic villi but not in syncytiotrophoblasts. Differences in virus receptor expression could explain why cytotrophoblasts are susceptible to avian influenza virus infections but not human influenza virus infections. Both syncytiotrophoblasts and cytotrophoblasts have been found to lack α-2,6-linked sialic acids,40 but whether the placenta expresses α-2,3-linked sialic acids is unknown. The relative number of infected cells in the fetal lungs, as detected by in-situ hybridisation and immunohistochemistry, was substantially higher than in the two adults, which could be explained by the dominance of avian-influenza-virus receptors over human-influenza-virus receptors in the bronchial and alveolar epithelia during the pseudoglandular stage of lung histiogenesis (up to the 20th gestational week).41
Despite the long duration of the disease and antiviral treatment in patient 2, viral sequences and antigens were detected in the post-mortem tissues. This finding is different from a previous study.7 The delayed clearance of viral antigens and sequences could be due to the immunosuppressive effect of the high-dose corticosteroids with which the patient had been treated for a long period before death. Since viral cultures were not done on post-mortem tissues, whether the detection of antigens and sequences indicates active viral replication is unclear. Positive results with in-situ hybridisation and RT-PCR have been reported in patients with SARS who died late in the course of disease.20, 35, 42 However, these positive RT-PCR results have been ascribed to the presence of small amounts of residual genome, rather than to active viral replication.35
We have shown that the H5N1 virus spreads beyond the lungs, infecting both ciliated and non-ciliated epithelial cells of the trachea, the placenta, T lymphocytes in lymph nodes, and cerebral neurons. We also report evidence of transplacental transmission, resulting in infection of fetal organs. These newly obtained data are important in the clinical, pathological, and epidemiological investgation of human H5N1 infection, and have implications for public-health and health-care providers.
Acknowledgments
Acknowledgments
We thank Hongquan Shao and Ning Li for their assistance with the autopsies; Lu Yao, Ruishu Deng, and Ruiqi Xue for their assistance in the experiments; and Ting Zhang for helping with the photos. This study is supported partly by the Lifu Educational Foundation, National Basic Research Program (973) of China (grant no 2005CB523003), National Natural Science Foundation of China (grant no 30599431), and awards from the National Institute of Allergy and Infectious Diseases, National Institutes of Health. CK is supported by grants from the Prins Bernhard Cultuurfonds (Wassink-Hesp Fonds and Kuitse Fonds), the Netherlands.
DIG=digoxin.
Contributors
JG initiated, designed, and coordinated the study, analysed the results, and took part in the writing of the manuscript. ZX took part in the autopsies, tissue collection, and routine pathology. ZhG was responsible for the clinical management and clinical data analysis. JLiu took part in the probe-design molecular biology, viral test, and the writing of the manuscript. JY did the immunohistochemistry and in-situ hybridisation. CK took part in the study design, result and literature analysis, and writing of the manuscript. LTL took part in the RT-PCR and NASBA, result analysis, and writing of the manuscript. JLu did the RT-PCR and NASBA. ZiG took part in the autopsies, tissue collection, routine pathology, and clinical data analysis. BZ did the molecular pathology, in-situ hybridisation, and probe design. MAM took part in the routine pathology, clinical data analysis, and writing of the manuscript. ML took part in the autopsies, tissue collection, and routine pathology. VMA did the fetoplacental pathology and molecular pathology. EG took part in routine pathology and tissue processing. ACHY designed and coordinated the RT-PCR and NASBA study, analysed the results, and took part in the writing of the manuscript. WIL had overall responsibility for the study design, and took part in the writing of the manuscript.
Conflict of interest statement
We declare that we have no conflict of interest.
Web Extra Material
In-situ hybridisation (ISH) with sense and antisense probes to haemagglutinin and nucleoprotein, double labelling, consecutive sections, and positive and negative controls
ISH signals seen with nitroblue tetrazolium/5-bromo-4-choloro-3-indolyl phosphate (purple-blue) and immunohistochemical signals with 3-amino-9-ethylcarbazole (double labelling; brown-red). (A) Haematoxylin and eosin staining of fetal lung tissue showing little or no damage, by contrast with severe damage in adult cases. (B) Positive ISH signals (with nucleoprotein antisense probe) in trachea, localising to cytoplasm of epithelial cells (arrows), with no evidence of nuclear staining, counterstained with methyl-green. (C) Double labelling combining ISH (with nucleoprotein sense probe) and antibodies for cytokeratin (AE1/AE3) showing both brown-red intracytoplasmic signals (cytokeratin, arrowheads) and dark-blue signals for viral genome (arrows) in nuclei of the same lung cells, establishing that infected cells are epithelial cells. (D) Double labelling with ISH (with nucleoprotein sense probe) and surfactant antibody showing dark-blue nuclear stain (H5N1 viral genome) and brown-red cytoplasmic signal of surfactant (type II pneumocytes) in one lung cell (arrows). (E) Positive ISH signals (with haemagglutinin sense probe) in mononuclear cells (arrows) in one chamber of fetal heart. (F) Double labelling (with nucleoprotein sense probe and CD68 antibody, brown-red) showing positive ISH and CD68 staining in same fetal liver cells (arrows), identifying them as Kupffer cells. (G) No positive ISH signal in H5N1-infected lungs with use of unrelated probe (antisense probe against fragment of polymerase gene [R1AB] of SARS-CoV). (H) No positive ISH signal (with nucleoprotein antisense probe) in lungs of patient with viral infectious pneumonia who died of respiratory failure (SARS-CoV infection). (I) No positive ISH signals (nucleoprotein antisense probe) in placenta of a pregnant woman who died of amniotic fluid embolism. Counterstained with methyl-green (negative control). (J) No positive ISH signal in trachea with use of unrelated probe. (K) No positive ISH signal (with nucleoprotein antisense probe) in trachea of a patient who died from purulent bronchitis. (L) No positive signal (with nucleoprotein antisense probe) in lungs of a mouse infected with H9N2 virus. (M) Brain tissue of black-headed gull was confirmed as infected with H5N1 by viral isolation and used as a positive control for probes, showing positive ISH signals with nucleoprotein antisense probe in cytoplasm of neurons. (N) Immunohistochemistry with neurofilament antibody (arrows) on consecutive section from webfigure 1M showing identical cells (numbered arrows) to be positive for H5N1 and neurofilament (ie, confirming cells to be neurons). (O) Positive ISH signals (with nucleoprotein antisense probe) in brain cells. (P) Immunohistochemistry of consecutive section to webfigure 1O with neurofilament antibodies, showing ISH-positive cells to be brain neurons. (Q) No positive ISH signal in brain with use of unrelated probe (antisense probe against fragment of polymerase gene [R1AB] of SARS-CoV). (R) ISH signals on brain tissue mainly located in cytoplasm of neurons, with slight staining of nuclei. (S) Section from webfigure 1R counterstained with Hoechst stain to identify nuclei of neurons (light blue); positive ISH signals for H5N1 mainly localised in cytoplasm.
Double labelling combining in-situ hybridisation (ISH, with nucleoprotein antisense probe) and immunohistochemistry (with tubulin-β antibody) on tracheal tissue from patient 2
ISH signals seen with nitroblue tetrazolium/5-bromo-4-choloro-3-indolyl phosphate (purple-blue) and immunohistochemical signals with 3-amino-9-ethylcarbazole (brown-red). (A) Positive ISH signals detected in cytoplasm (arrow) but not in nucleus of tubulin-negative, non-ciliated cell. Positive tubulin staining seen on apical surface of ciliated cells (arrowheads). (B) Both tubulin-positive signals (arrowheads) and ISH-positive signals (arrows) seen in ciliated cells. ISH signals seen in cytoplasm (arrows) but not nuclei of infected cells. (C) Viral genomic sequences (arrows) seen in cytoplasm of non-ciliated columnar epithelial cells (*) and tubulin-positive ciliated epithelial cells (arrowheads). No ISH signals seen in nuclei. (D) Double labelling showing tubulin-negative non-ciliated cell with positive intracytoplasmic ISH signal (small arrow) and tubulin-positive uninfected cell (arrowhead) with no signal. Two ISH-positive putative basal cells are also seen (large arrows).
Double labelling of lymphocytes of hilar lymph node
In-situ hybridisation (ISH) signals seen with nitroblue tetrazolium/5-bromo-4-choloro-3-indolyl phosphate (purple-blue) and immunohistochemistry (IHC) signals with 3-amino-9-ethylcarbazole (brown-red). (A,B) Double labelling combining ISH (with haemagglutinin probe) and CD3 IHC. In some cells, both signals appear to colocalise in the same cells (arrows), in cytoplasm, suggesting a small proportion of lymphocytes in lymph node to be infected by H5N1 virus. (C,D) Double labelling with ISH (with haemagglutinin probe) and CD20 IHC. ISH-positive cells are not positive for CD20 (arrows), suggesting cells not to be B lymphocytes. (E,F) Double labelling with ISH (with haemagglutinin probe) and CD68 IHC. Morphologically, ISH-positive cells do not resemble macrophages or monocytes, with different distribution patterns. Positive ISH signals (arrows) only seen in small cells that are not positive for CD68. (G,H) Double labelling with ISH (with haemagglutinin probe) and S100 IHC. Morphologically, ISH-positive cells do not resemble dendritic cells, with different distribution patterns and often located in different lymph node areas. Positive ISH signals (arrows) only seen in small cells that are not positive for S100.
In-situ hybridisation (ISH) and immunohistochemistry (IHC) on consecutive sections of placenta
ISH signals seen with nitroblue tetrazolium/5-bromo-4-choloro-3-indolyl phosphate (purple-blue) and IHC signals seen with diamenobenzodin (brown). (A) Placenta with haematoxylin and eosin staining. (B) Positive ISH signals seen in cytoplasm of cells localised in core of chorionic villus. Cells morphologically resemble macrophages. (C) Positive ISH signals seen in cytoplasm of cells in periphery of chorionic villus. Cells line the inner layer but not outer layer of trophoblast barrier, therefore are probably cytotrophoblasts but not syncytiotrophoblasts. (D) Immunostaining with E-cadherin antibody (arrows) on consecutive sections confirms some ISH-positive cells to be cytotrophoblasts but not syncytiotrophoblasts. (E,F) Immunohistochemistry with monoclonal antibodies to PLAP (brown). Positive staining is distributed throughout outer layer of chorionic villi, implying that ISH-positive cells seen in webfigure 4C are not syncytiotrophoblasts. (G,H) Distribution of CD68-positive cells is similar to that of ISH-positive cells seen in webfigure 4B, implying fetal macrophages (Hofbauer cells) to be infected with H5N1 virus.
Negative controls for immunohistochemistry
No positive cells seen in any negative controls. (A) Tracheal tissue on which primary monoclonal antibody to nucleoprotein replaced with PBS. (B) Immunostaining with monoclonal antibody to nucleoprotein on tracheal tissue from patient not infected with H5N1. (C) Lung tissue on which primary monoclonal antibody to nucleoprotein replaced with PBS. (D) Immunostaining with monoclonal antibody to haemagglutinin on lung tissue from patient infected with SARS-CoV. (E) Immunostaining on lymph-node tissue on which primary antibody replaced with PBS. (F) Immunostaining with monoclonal antibody to haemagglutinin on brain tissue from patient infected with SARS-CoV. (G) Immunostaining with monoclonal antibody to haemagglutinin on lung tissue from aborted fetus from a healthy mother. (H) Immunostaining with monoclonal antibody to nucleoprotein on brain cortex tissue from patient not infected with H5N1.
Sources, dilutions, and incubation times of primary monoclonal antibodies used in immunohistochemistry
References
- 1.WHO Cumulative number of confirmed human cases of avian influenza A/(H5N1) reported to WHO. http://www.who.int/csr/disease/avian_influenza/country/cases_table_2007_03_01/en/index.html (accessed March 1, 2007).
- 2.Ungchusak K, Auewarakul P, Dowell SF. Probable person-to-person transmission of avian influenza A (H5N1) N Engl J Med. 2005;352:333–340. doi: 10.1056/NEJMoa044021. [DOI] [PubMed] [Google Scholar]
- 3.Normile D. Human transmission but no pandemic in Indonesia. Science. 2006;312:1855. doi: 10.1126/science.312.5782.1855b. [DOI] [PubMed] [Google Scholar]
- 4.Horimoto T, Kawaoka Y. Pandemic threat posed by avian influenza A viruses. Clin Microbiol Rev. 2001;14:129–149. doi: 10.1128/CMR.14.1.129-149.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Uiprasertkul MP, Puthavathana K, Sangsiriwut P. Influenza A H5N1 replication sites in humans. Emerg Infect Dis. 2005;11:1036–1041. doi: 10.3201/eid1107.041313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.De Jong MD, Van Cam B, Qui PT. Fatal avian influenza A (H5N1) in a child presenting with diarrhea followed by coma. N Engl J Med. 2005;352:686–691. doi: 10.1056/NEJMoa044307. [DOI] [PubMed] [Google Scholar]
- 7.To KF, Chan PKS, Chan KF. Pathology of fatal human infection associated with avian influenza A H5N1 virus. J Med Virol. 2001;63:242–246. doi: 10.1002/1096-9071(200103)63:3<242::aid-jmv1007>3.0.co;2-n. [DOI] [PubMed] [Google Scholar]
- 8.Peiris JSM, Yu WC, Leung CW. Re-emergence of fatal human influenza A subtype H5N1 disease. Lancet. 2004;363:617–619. doi: 10.1016/S0140-6736(04)15595-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chotpitayasunondh T, Ungchusak K, Hanshaoworakul W. Human disease from influenza A (H5N1), Thailand. Emerg Infect Dis. 2004;11:201–209. doi: 10.3201/eid1102.041061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shu Y, Yu H, Li D. Lethal avian influenza A (H5N1) infection in a pregnant woman in Anhui Province, China. N Engl J Med. 2006;354:1421–1422. doi: 10.1056/NEJMc053524. [DOI] [PubMed] [Google Scholar]
- 11.School of Basic Medical Sciences. Peking University HA sequence of the virus isolated from case 2 (A/Jiangxi/1/2005 virus) http://www.chinapathology.org/lancet/supplement_1_HA_sequence.doc (accessed Aug 29, 2007).
- 12.Chen H, Smith GJ, Li KS. Establishment of multiple sublineages of H5N1 influenza virus in Asia: implications for pandemic control. Proc Natl Acad Sci USA. 2006;103:2845–2850. doi: 10.1073/pnas.0511120103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Li L, Gu J, Shi X. Biosafety level 3 laboratory for autopsies of patients with severe acute respiratory syndrome: principles, practices, and Prospects. Clin Infect Dis. 2005;41:815–821. doi: 10.1086/432720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lin Y, Shen X, Yang RF. Identification of an epitope of SARS-coronavirus nucleocapsid protein. Cell Res. 2003;13:141–145. doi: 10.1038/sj.cr.7290158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gu J, Forte M, Hance H. Microwave fixation, antigen retrieval, and accelerated immunocytochemistry. Cell Vision. 1994;1:76–77. [Google Scholar]
- 16.Brown LM, Lacey HA, Baker PN. E-cadherin in the assessment of aberrant placental cytotrophoblast turnover in pregnancies complicated by pre-eclampsia. Histochem Cell Biol. 2005;124:499–506. doi: 10.1007/s00418-005-0051-7. [DOI] [PubMed] [Google Scholar]
- 17.School of Basic Medical Sciences. Peking University Sources, dilutions, and incubation times of primary antibody used in immunohistochemistry. http://www.chinapathology.org/lancet/suppl_2__antibodies_(new).doc (accessed Aug 29, 2007).
- 18.Liu J, Xiao H, Lei F. Highly pathogenic H5N1 influenza virus infection in migratory birds. Science. 2005;309:1206. doi: 10.1126/science.1115273. [DOI] [PubMed] [Google Scholar]
- 19.Zhang QL, Ding YQ, Hou JL. Detection of severe acute respiratory syndrome (SARS)-associated coronavirus RNA in autopsy tissues with in situ hybridization. Di Yi Jun Yi Da Xue Xue Bao. 2003;23:1125–1127. [PubMed] [Google Scholar]
- 20.Gu J, Gong E, Zhang B. Multiple organ infection and the pathogenesis of SARS. J Exp Med. 2005;202:415–424. doi: 10.1084/jem.20050828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yuen KY, Chan PK, Peiris M. Clinical features and rapid viral diagnosis of human disease associated with avian influenza A H5N1 virus. Lancet. 1998;351:467–471. doi: 10.1016/s0140-6736(98)01182-9. [DOI] [PubMed] [Google Scholar]
- 22.Yue C, Genersch E. RT-PCR analysis of deformed wing virus in honeybees (Apis mellifera) and mites (Varroa destructor) J Gen Virol. 2005;86:3419–3424. doi: 10.1099/vir.0.81401-0. [DOI] [PubMed] [Google Scholar]
- 23.Lau LT, Banks J, Aherne R. Nucleic acid sequence-based amplification methods to detect avian influenza virus. Biochem Biophys Res Commun. 2004;313:336–342. doi: 10.1016/j.bbrc.2003.11.131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Park CH, Ishinaka M, Takada A. The invasion routes of neurovirulent A/Hong Kong/483/97 (H5N1) influenza virus into the central nervous system after respiratory infection in mice. Arch Virol. 2002;147:1425–1436. doi: 10.1007/s00705-001-0750-x. [DOI] [PubMed] [Google Scholar]
- 25.Chutinimitkul S, Bhattarakosol P, Srisuratanon S. H5N1 influenza A virus and infected human plasma. Emerg Infect Dis. 2006;12:1041–1043. doi: 10.3201/eid1206.060227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Takahashi T, Suzuki Y, Nishinaka D. Duck and human pandemic influenza A viruses retain sialidase activity under low pH conditions. J Biochem. 2001;130:279–283. doi: 10.1093/oxfordjournals.jbchem.a002983. [DOI] [PubMed] [Google Scholar]
- 27.Zhou J, Law HKW, Cheung CY. Differential expression of chemokines and their receptors in adult and neonatal macrophages infected with human or avian influenza viruses. J Infect Dis. 2006;194:61–70. doi: 10.1086/504690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cheung CY, Poon LLM, Lau AS. Induction of proinflammatory cytokines in human macrophages by influenza A (H5N1) viruses: a mechanism for the unusual severity of human disease? Lancet. 2002;360:1831–1837. doi: 10.1016/s0140-6736(02)11772-7. [DOI] [PubMed] [Google Scholar]
- 29.Van Riel D, Munster VJ, de Wit E. H5N1 virus attachment to lower respiratory tract. Science. 2006;312:399. doi: 10.1126/science.1125548. [DOI] [PubMed] [Google Scholar]
- 30.Shinya K, Ebina M, Yamada S. Avian flu: influenza virus receptors in the human airway. Nature. 2006;440:435–436. doi: 10.1038/440435a. [DOI] [PubMed] [Google Scholar]
- 31.Thompson CI, Barclay WS, Zambon MC. Infection of human airway epithelium by human and avian strains of influenza a virus. J Virol. 2006;80:8060–8068. doi: 10.1128/JVI.00384-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Matrosovich MN, Matrosovich TY, Gray T. Human and avian influenza viruses target different cell types in cultures of human airway epithelium. Proc Natl Acad Sci U S A. 2004;101:4620–4624. doi: 10.1073/pnas.0308001101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zhang L, Bukreyev A, Thompson CI. Infection of ciliated cells by human parainfluenza virus type 3 in an in vitro model of human airway epithelium. J Virol. 2005;79:1113–1124. doi: 10.1128/JVI.79.2.1113-1124.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.de Jong MD, Simmons CP, Thanh TT. Fatal outcome of human influenza A (H5N1) is associated with high viral load and hypercytokinemia. Nat Med. 2006;12:1203–1207. doi: 10.1038/nm1477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Nicholls JM, Butany J, Poon LL. Time course and cellular localization of SARS-CoV nucleoprotein and RNA in lungs from fatal cases of SARS. PLoS Med. 2006;3:e27. doi: 10.1371/journal.pmed.0030027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ramphal R, Donnelly WH, Small PA. Fatal influenzal pneumonia in pregnancy: Failure to demonstrate transplacental transmission of influenza virus. Am J Obstet Gynecol. 1980;138:347–348. doi: 10.1016/0002-9378(80)90265-3. [DOI] [PubMed] [Google Scholar]
- 37.Irving WL, James DK, Stephenson T. Influenza virus infection in the second and third trimesters of pregnancy: a clinical and seroepidemiological study. BJOG. 2000;107:1282–1289. doi: 10.1111/j.1471-0528.2000.tb11621.x. [DOI] [PubMed] [Google Scholar]
- 38.Mori I, Nagafuji H, Matsumoto K. Use of the polymerase chain reaction for demonstration of influenza virus dissemination in children. Clin Infect Dis. 1997;24:736–737. [PubMed] [Google Scholar]
- 39.Fisher S, Genbacev O, Maidji E. Human cytomegalovirus infection of placental cytotrophoblasts in vitro and in utero: implications for transmission and pathogenesis. J Virol. 2000;74:6808–6820. doi: 10.1128/jvi.74.15.6808-6820.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kaneko Y, Yamamoto H, Colley KJ. Expression of Gal beta 1,4GlcNAc alpha 2,6-sialyltransferase and alpha 2,6-linked sialoglycoconjugates in normal human and rat tissues. J Histochem Cytochem. 1995;43:945–954. doi: 10.1177/43.9.7642967. [DOI] [PubMed] [Google Scholar]
- 41.Cerna A, Janega P, Martanovic P. Changes in sialic acid expression in the lung during intrauterine development of the human fetus. Acta Histochem. 2002;104:339–342. doi: 10.1078/0065-1281-00669. [DOI] [PubMed] [Google Scholar]
- 42.Farcas GA, Poutanen SM, Mazzulli T. Fatal severe acute respiratory syndrome is associated with multiorgan involvement by coronavirus. J Infect Dis. 2005;191:193–197. doi: 10.1086/426870. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
In-situ hybridisation (ISH) with sense and antisense probes to haemagglutinin and nucleoprotein, double labelling, consecutive sections, and positive and negative controls
ISH signals seen with nitroblue tetrazolium/5-bromo-4-choloro-3-indolyl phosphate (purple-blue) and immunohistochemical signals with 3-amino-9-ethylcarbazole (double labelling; brown-red). (A) Haematoxylin and eosin staining of fetal lung tissue showing little or no damage, by contrast with severe damage in adult cases. (B) Positive ISH signals (with nucleoprotein antisense probe) in trachea, localising to cytoplasm of epithelial cells (arrows), with no evidence of nuclear staining, counterstained with methyl-green. (C) Double labelling combining ISH (with nucleoprotein sense probe) and antibodies for cytokeratin (AE1/AE3) showing both brown-red intracytoplasmic signals (cytokeratin, arrowheads) and dark-blue signals for viral genome (arrows) in nuclei of the same lung cells, establishing that infected cells are epithelial cells. (D) Double labelling with ISH (with nucleoprotein sense probe) and surfactant antibody showing dark-blue nuclear stain (H5N1 viral genome) and brown-red cytoplasmic signal of surfactant (type II pneumocytes) in one lung cell (arrows). (E) Positive ISH signals (with haemagglutinin sense probe) in mononuclear cells (arrows) in one chamber of fetal heart. (F) Double labelling (with nucleoprotein sense probe and CD68 antibody, brown-red) showing positive ISH and CD68 staining in same fetal liver cells (arrows), identifying them as Kupffer cells. (G) No positive ISH signal in H5N1-infected lungs with use of unrelated probe (antisense probe against fragment of polymerase gene [R1AB] of SARS-CoV). (H) No positive ISH signal (with nucleoprotein antisense probe) in lungs of patient with viral infectious pneumonia who died of respiratory failure (SARS-CoV infection). (I) No positive ISH signals (nucleoprotein antisense probe) in placenta of a pregnant woman who died of amniotic fluid embolism. Counterstained with methyl-green (negative control). (J) No positive ISH signal in trachea with use of unrelated probe. (K) No positive ISH signal (with nucleoprotein antisense probe) in trachea of a patient who died from purulent bronchitis. (L) No positive signal (with nucleoprotein antisense probe) in lungs of a mouse infected with H9N2 virus. (M) Brain tissue of black-headed gull was confirmed as infected with H5N1 by viral isolation and used as a positive control for probes, showing positive ISH signals with nucleoprotein antisense probe in cytoplasm of neurons. (N) Immunohistochemistry with neurofilament antibody (arrows) on consecutive section from webfigure 1M showing identical cells (numbered arrows) to be positive for H5N1 and neurofilament (ie, confirming cells to be neurons). (O) Positive ISH signals (with nucleoprotein antisense probe) in brain cells. (P) Immunohistochemistry of consecutive section to webfigure 1O with neurofilament antibodies, showing ISH-positive cells to be brain neurons. (Q) No positive ISH signal in brain with use of unrelated probe (antisense probe against fragment of polymerase gene [R1AB] of SARS-CoV). (R) ISH signals on brain tissue mainly located in cytoplasm of neurons, with slight staining of nuclei. (S) Section from webfigure 1R counterstained with Hoechst stain to identify nuclei of neurons (light blue); positive ISH signals for H5N1 mainly localised in cytoplasm.
Double labelling combining in-situ hybridisation (ISH, with nucleoprotein antisense probe) and immunohistochemistry (with tubulin-β antibody) on tracheal tissue from patient 2
ISH signals seen with nitroblue tetrazolium/5-bromo-4-choloro-3-indolyl phosphate (purple-blue) and immunohistochemical signals with 3-amino-9-ethylcarbazole (brown-red). (A) Positive ISH signals detected in cytoplasm (arrow) but not in nucleus of tubulin-negative, non-ciliated cell. Positive tubulin staining seen on apical surface of ciliated cells (arrowheads). (B) Both tubulin-positive signals (arrowheads) and ISH-positive signals (arrows) seen in ciliated cells. ISH signals seen in cytoplasm (arrows) but not nuclei of infected cells. (C) Viral genomic sequences (arrows) seen in cytoplasm of non-ciliated columnar epithelial cells (*) and tubulin-positive ciliated epithelial cells (arrowheads). No ISH signals seen in nuclei. (D) Double labelling showing tubulin-negative non-ciliated cell with positive intracytoplasmic ISH signal (small arrow) and tubulin-positive uninfected cell (arrowhead) with no signal. Two ISH-positive putative basal cells are also seen (large arrows).
Double labelling of lymphocytes of hilar lymph node
In-situ hybridisation (ISH) signals seen with nitroblue tetrazolium/5-bromo-4-choloro-3-indolyl phosphate (purple-blue) and immunohistochemistry (IHC) signals with 3-amino-9-ethylcarbazole (brown-red). (A,B) Double labelling combining ISH (with haemagglutinin probe) and CD3 IHC. In some cells, both signals appear to colocalise in the same cells (arrows), in cytoplasm, suggesting a small proportion of lymphocytes in lymph node to be infected by H5N1 virus. (C,D) Double labelling with ISH (with haemagglutinin probe) and CD20 IHC. ISH-positive cells are not positive for CD20 (arrows), suggesting cells not to be B lymphocytes. (E,F) Double labelling with ISH (with haemagglutinin probe) and CD68 IHC. Morphologically, ISH-positive cells do not resemble macrophages or monocytes, with different distribution patterns. Positive ISH signals (arrows) only seen in small cells that are not positive for CD68. (G,H) Double labelling with ISH (with haemagglutinin probe) and S100 IHC. Morphologically, ISH-positive cells do not resemble dendritic cells, with different distribution patterns and often located in different lymph node areas. Positive ISH signals (arrows) only seen in small cells that are not positive for S100.
In-situ hybridisation (ISH) and immunohistochemistry (IHC) on consecutive sections of placenta
ISH signals seen with nitroblue tetrazolium/5-bromo-4-choloro-3-indolyl phosphate (purple-blue) and IHC signals seen with diamenobenzodin (brown). (A) Placenta with haematoxylin and eosin staining. (B) Positive ISH signals seen in cytoplasm of cells localised in core of chorionic villus. Cells morphologically resemble macrophages. (C) Positive ISH signals seen in cytoplasm of cells in periphery of chorionic villus. Cells line the inner layer but not outer layer of trophoblast barrier, therefore are probably cytotrophoblasts but not syncytiotrophoblasts. (D) Immunostaining with E-cadherin antibody (arrows) on consecutive sections confirms some ISH-positive cells to be cytotrophoblasts but not syncytiotrophoblasts. (E,F) Immunohistochemistry with monoclonal antibodies to PLAP (brown). Positive staining is distributed throughout outer layer of chorionic villi, implying that ISH-positive cells seen in webfigure 4C are not syncytiotrophoblasts. (G,H) Distribution of CD68-positive cells is similar to that of ISH-positive cells seen in webfigure 4B, implying fetal macrophages (Hofbauer cells) to be infected with H5N1 virus.
Negative controls for immunohistochemistry
No positive cells seen in any negative controls. (A) Tracheal tissue on which primary monoclonal antibody to nucleoprotein replaced with PBS. (B) Immunostaining with monoclonal antibody to nucleoprotein on tracheal tissue from patient not infected with H5N1. (C) Lung tissue on which primary monoclonal antibody to nucleoprotein replaced with PBS. (D) Immunostaining with monoclonal antibody to haemagglutinin on lung tissue from patient infected with SARS-CoV. (E) Immunostaining on lymph-node tissue on which primary antibody replaced with PBS. (F) Immunostaining with monoclonal antibody to haemagglutinin on brain tissue from patient infected with SARS-CoV. (G) Immunostaining with monoclonal antibody to haemagglutinin on lung tissue from aborted fetus from a healthy mother. (H) Immunostaining with monoclonal antibody to nucleoprotein on brain cortex tissue from patient not infected with H5N1.
Sources, dilutions, and incubation times of primary monoclonal antibodies used in immunohistochemistry