Abstract
Immunological effector cells must be sensitive to the antigens or environmental signals that indicate that a pathogen is present. To this end, a group of cells known as the professional antigen‐presenting cells have the ability to educate T, B and NK cells as to the fingerprints of specific infections. The most adept of these cells are a closely related family termed dendritic cells (DC). A subset of these act as peripheral sentinels, specializing in the uptake, processing and presentation of antigenic material combined with an ability to detect a wide variety of ‘danger’ signals. These ‘danger’ or activation signals induce profound changes in dendritic cell physiology, facilitating the efficient stimulation of both adaptive and innate immunity. In the present review, a number of recent advances in the understanding of DC biology are discussed. These advances offer insights into the pathogenesis of a wide variety of diseases and point towards future strategies for immunotherapy.
Keywords: autoimmunity, cancer, dendritic cell, immunotherapy, persistent viral infection, transplantation
Introduction
Dendritic cells (DC) are now recognized as essential regulators of both innate and acquired arms of the immune system. 1 Dendritic cells, in addition to their distinctive morphology, have a number of phenotypic and functional characteristics that make them formidable APC (Table 1). Dendritic cells bear sole responsibility for the stimulation of virgin T lymphocytes, a property that distinguishes them from all other APC (e.g. B cells). 2 , 3 The DC are also essential accessory cells in the generation of primary antibody responses 4 and are powerful enhancers of NK cell cytotoxicity. 5 Conversely, DC are also involved in the maintenance of tolerance to antigens, with DC in the thymus contributing towards shaping of the T cell repertoire by deleting autoreactive lymphocytes. 6 , 7 As a consequence of this heterogeneity in vivo, it is now acknowledged that DC are a family of cells, with each subset exerting control over a different area of immunity. In terms of origin, DC are bone marrow derived cells originating from both myeloid and lymphoid precursors. 8 , 9 Examples of DC in vivo include the epidermal Langerhans cell, the interstitial DC (found in the heart, lungs, liver and other organs), the veiled DC found in afferent lymph, interdigitating DC (found in T cell‐rich areas of lymphoid tissue) and thymic DC. Follicular DC (FDC) are an exception to this group because they are believed to be involved in the long‐term maintenance of B cell memory by retaining immune complexes; these cells differ markedly from the aforementioned group and will not be discussed here (reviewed by Tew et al. 10 ).
Table 1.
Properties of dendritic cells
| 1. | Stellate morphology and cytoplasmic processes giving rise to the characteristic ‘conker’ appearance when immature. Maturing DC develop complex veils (in afferent lymph). |
| 2. | Rapid motility, as demonstrated by time lapse photography. |
| 3. | Efficient stimulation of autologous and allogeneic T cells (naïve and mature); DC are approximately 100‐fold better at stimulating an allogeneic MLR than bulk leucocyte culture. The DC are unique in their ability to stimulate naïve/virgin T cells, unlike other APC, such as B cells, which only activate memory T cells. |
| 4. | Adept environmental antigen sampling. Fluid phase endocytosis estimated at 100 times the DC volume per hour for in vitro‐generated DC. Ultrastructural analysis reveals the presence of many fluid‐filled endosomes. Antigen sampling is also by receptor‐mediated uptake, for example Fc receptors (FcγRII/CD32, FcγRI/CD64), complement receptors (C3bi/CD11b) and C‐type lectin receptors (MMR, DEC‐205). |
| 5. | Dendritic cells are distinct from macrophages in that they do not express CD16 or macrophage characteristic antimicrobial enzymes (e.g. lysozyme and myeloperoxidase). |
| 6. | High surface density of antigen presentation molecules, for example MHC‐I and MHC‐II (expression 10–100‐fold greater than on other APC, e.g. B cells). |
| 7. | Mature DC have a high surface density of accessory/costimulatory molecules (CD40, ICAM‐1/CD54, ICAM‐3/CD50, LFA‐3/CD58, B7‐1/CD80 and B7‐2/CD86). |
| 8. | Production of large quantities of IL‐12 after treatment with activation signals, including CD40L or LPS. |
| 9. | An ability to cross prime antigen into the MHC‐I presentation pathway: protein from the exogenous milieu can be presented to CD8+ T cells. This allows specific class‐I mediated immunity to be generated without the DC becoming infected. Cross priming is therefore of prime importance in the generation of CTL responses against pathogens that infect non‐haemopoietic cells. |
dC, dendritic cell;
ICAM, intercellular adhesion molecule‐1;
LFA, leucocyte functional antigen.
The main function of Langerhans and interstitial type DC is as immunological sentinels, being strategically placed to detect invading microbes. Sentinel DC remain dormant until signals in the extracellular milieu (derived either from microbes or distressed bystander cells) induce a rapid change in function, also known as ‘activation’. Activation induces a number of important changes in DC (Figure 1), not least of which is migration into local lymphoid tissue where they communicate antigenic information to lymphocytes. Recent clarification of the biochemistry of activation signals and the effects of DC activation have prompted research into the part played by activated DC in the pathogenesis of disease and whether DC‐modulating immunotherapies can be used in the treatment of a wide range of diseases, including autoimmunity, allergy, immunodeficiency, transplant rejection, persistent viral infection and cancer.
Figure 1.
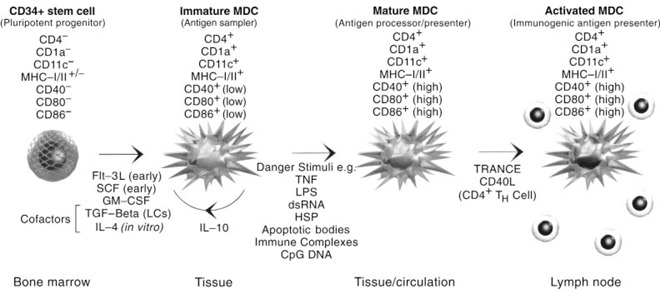
Dendritic cell life history can be subdivided into a number of phases, all with discrete cellular functionality. Transition between phases is mediated by diverse signals and is accompanied by changes in expression patterns of many surface markers and secreted factors. MDC, myeloid dendritic cell; SCF, stem cell factor; GM‐CSF, granulocyte–macrophage colony stimulating factor; TGF, transforming growth factor; HSP, heat shock protein.
Properties of dendritic cells
Dendritic cells were initially characterized on the basis of their distinctive morphology, with numerous cytoplasmic processes giving rise to a stellate appearance. As a consequence, the DC has a high surface area permitting intimate contact with a large number of surrounding cells. As proof of this point, in vitro DC form large spherical aggregates with lymphocytes, and experimentally, only one mature DC is required to stimulate 100–3000 T cells. Dendritic cells also possess an array of mechanisms for sampling antigen. These include macropinocytosis, where fluid from the extracellular milieu is taken up into pinocytic vesicles and antigen is concentrated by expelling excess water via channels called aquaporins. 11 The DC also expresses a repertoire of receptors for efficient receptor‐mediated antigen uptake. Additional receptors expressed by DC include FcγRII (CD32), FcγRI (CD64), 12 , 13 FcεRI 14 and the C3 bi complement receptors (CD11b), 15 which increase the efficiency of immune complex endocytosis. Dendritic cells express C‐type lectin receptors, including the macrophage mannose receptor and DEC‐205, which bind bacterial carboydrates. 16 , 17 Dendritic cells also express markedly higher levels of antigen presentation molecules than any other cell (e.g. CD1a, MHC‐I and MHC‐II). In addition to this, DC have a high surface density of accessory/costimulatory molecules, including intercellular adhesion molecule (ICAM)‐1/CD54, ICAM‐3/CD50, leucocyte functional antigen (LFA)‐3/CD58, B7–1/CD80 and B7–2/CD86 (Figure 2), which facilitate both the interaction with, and stimulation of, lymphocytes. 18 , 19
Figure 2.
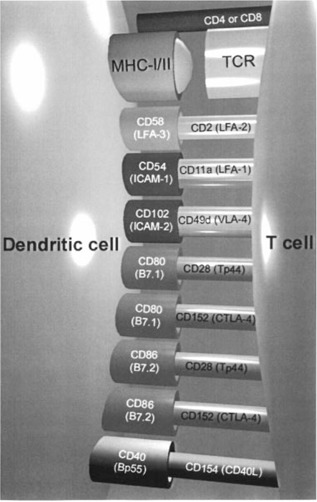
Interaction of dendritic cells with T lymphocytes. Antigen is presented as peptide MHC class‐I/II complexes (signal 1). T lymphocytes are activated by the presence of costimulatory molecules, which communicate that the presented antigen is associated with a ‘threat’ (signal 2). Absence of these secondary signals induces tolerance towards the presented peptide. LFA, leucocyte functional antigen; ICAM, intercellular adhesion molecule; VLA, very late antigen.
An additional noteworthy property of DC is that exogenous antigen (e.g. immune complexes) can be ‘cross primed’ into the MHC class I presentation pathway. This pathway would normally only present antigen from the endogenous compartment. The DC are thought to possess a specific mechanism that allows fluid from an endocytic vesicle to gain direct access to the cytoplasm. In essence, this allows the DC to present new peptides to CD8+ T lymphocytes without themselves being infected or damaged. 20 , 21
Dendritic cell activation
Throughout evolution, the presence of a pathogen has been accompanied by a series of unique markers. For example, this may be dsRNA for influenza virus or LPS in the case of gram‐negative bacteria. The DC are sensitive to a wide range of these stimuli that serve not only to activate innate immunity via the release of chemokines and proinflammatory mediators, but also trigger DC migration towards local lymphoid tissue in order to generate antigen‐specific (adaptive) immunity.
It is hardly surprising that intact, viable microbes, such as influenza virus or mycobacteria, can be observed to activate DC. 22 , 23 However, the question remains as to which individual molecules mediate DC activation and via which receptors. Factors specific to bacteria, such as LPS and, more surprisingly, CpG motifs in DNA, have been shown to activate DC. 24 , 25 Lipopolysaccharide‐induced activation is mediated by CD14 acting in concert with the recently described Toll‐like receptors (TLR) 2 and 4. 26 , 27 However, the receptor that mediates DNA‐induced activation still remains to be identified. The ligation of Fc receptors during the uptake of immune complexes has also been shown to induce the maturation of dendritic cells. 28 A further interesting observation is that several DC‐activating factors (including LPS, TNF‐α and CD40L) are able to rescue immature DC from apoptosis. 29
The second group of DC‐activating signals is derived from distressed or dying cells. The phenomenon of Langerhans cell migration into tissue culture media after in vitro culture of skin explants for 24 h is thought to reflect inflammatory stimuli resulting from skin damage promulgating DC migration. 30 Cells, as a consequence of infection, will release pools of pro‐activating factors, which will indirectly activate DC. The action of pro‐inflammatory cytokines (TNF‐α) and prostaglandins (PGE2) on immature DC produces a maturation event characterized by an increase in the levels of membrane HLA‐DR (MHC‐II) and a reduction in the propensity of the cells to take up soluble antigen. 31 In addition, DC that encounter apoptotic cells undergo maturation in vitro; this maturation involves autocrine/paracrine secretion of IL‐1β and TNF‐α and is thought to be mediated by αVβ5 integrin and CD36. 32 , 33
One of the primary events after activation is the release of chemokines, such as RANTES, monocyte chemotactic protein (MCP)‐1, macrophage inflammatory protein (MIP)‐1α, and MIP‐1, 34 which not only attract new mononuclear cells (including DC precursors) but also serve to activate NK cells, supporting the inference that the DC is an important bridge between the acquired and innate immune systems. Other chemokine‐related phenomena that accompany infection include a reduction in the chemotactic response of DC towards MIP‐1α, MIP‐1β, MIP‐3β, RANTES, MCP‐3 and fMLP 35 and an increase in the chemotactic response towards chemokines stromal cell‐derived factor (SDF)‐1 (CXCR4) and monocyte‐derived chemokine (MDC), CCR4. 36 Chemokines play an important role in targeting DC to lymphoid tissue. The chemokine receptor CCR7 is gradually up‐regulated after activation. The ligands for this receptor, 6Ckine and MIP‐3β, are chemoattractants for DC and are produced primarily in the T cell‐rich parafollicular areas of lymphoid tissue. 37 , 38
For the Langerhans cell, activation is also accompanied by the loss of specific markers, such as cutaneous lymphocyte antigen (CLA) and Birbeck granules, along with altered surface expression of cell adhesion molecules that facilitate movement into the afferent lymph. For example, Langerhans cells express E‐cadherin, which mediates homotypic interactions with keratinocytes. The production of this molecule is down‐regulated during maturation. 39 , 40 MHC class II molecules, stored within vesicles termed MIIC, are transported to the cell surface and there is a concomitant increase in surface half‐life of both MHC class I and MHC class II molecules. 41 Activation‐induced changes serve to change the immature DC, which are adept at antigen sampling, into DC dedicated to the presentation of antigen. Immediately after exposure to an activation signal, such as TNF‐α, there is a short‐term increase in antigen uptake followed by an almost complete cessation in uptake preceding migration into afferent lymphatics. 42 , 43
In summary, DC activation is accompanied by a number of changes, including increased expression of costimulatory and antigen presentation molecules, a decrease in the propensity to capture soluble antigen, the increased stability of MHC class I and II molecules, changes in chemokine ligand and receptor expression and translocation into lymphoid tissue.
Dendritic cells in lymphoid tissue
Once inside the lymphoid tissue, the DC interacts with T, B and NK cells. The unique ability of DC to attract and stimulate naïve T cells at this point is still poorly understood; however, it appears that DC secrete a C‐C chemokine (DC‐CK1) that selectively attracts these cells, 44 while both DC and activated B cells produce a C‐C chemokine ABCD‐1 that attracts mature T cells. 45 Dendritic cells, by virtue of their high levels of surface MHC‐II and costimulator expression, readily cluster CD4+ T helper cells. Two different subsets of dendritic cells have recently been characterized that control the fate of naïve T helper cells. 46 One subset, designated DC1, secretes large quantities of IL‐12 that promotes the development of a Th1‐type phenotype, which is important in the generation of immunity to intracellular parasites. Another subset, the DC2, promotes a Th2 pattern of cytokine production via the release of an as yet uncharacterized factor. The Th2 responses are important in the generation of immunity to extracellular infections and also in allergic responses. The interaction of the CD4/TCR complex with DC1 MHC‐II has significant effects on the cytokine production of DC, one of the most important being enhancement of IL‐12 production. Expression of CD40L by T helper cells further stimulates the production of IL‐12 and IL‐15 from DC. 47 , 48 The secretion of these Th1‐type cytokines by DC is important in enhancing the generation of cytotoxic T cells from naïve CD8+ T cells. The CD40/40L pathway has many other important functions in the context of DC. The interaction of DC‐expressed CD40 with CD40L on NK cells has also been shown to enhance NK cell cytotoxicity and IFN‐γ production. 5
Dendritic cells also interact both directly and indirectly with B cells. As alluded to previously, one of the first observations made regarding DC was that they are essential accessory cells for the generation of primary antibody responses. The addition of DC to a culture of B cells promotes an LFA‐1‐dependent clustering, proving that DC/B cell communication is not mediated solely via Th cell intermediates. 49 , 50 The DC stimulate the proliferation of CD40‐activated B cells and also enhance maturation into IgM‐producing plasma cells. 51 , 52 This differentiation of naïve B cells into IgM‐producing plasma cells is dependent on DC secretion of IL‐12. 53 In the presence of IL‐10 and TGF‐β, DC have also been observed to promote immunoglobulin class switching towards IgA. 54
The final stage in the DC life cycle is apoptosis, mediated either by T lymphocytes or NK cells. This process makes way for the next wave of DC migrating from local tissues into the afferent lymph 55 , 56 and provides an explanation as to why DC are never found in the efferent lymph. In summary, immature DC are activated by a number of environmental signals that are associated with cellular stress or the presence of a microbe. These signals are transduced by the DC into costimulators, cytokines and chemokines, which activate innate and acquired immunity.
Dendritic cells and infectious disease
Dendritic cells are commonly the first immunological cells to encounter foreign organisms. Not surprisingly, DC play an important role in the generation of protective immunity towards intracellular parasites. 57 , 58 However, as a consequence, DC function may also be subverted as part of the life cycle of a pathogen. A number of viruses use molecules expressed by DC as receptors; examples include CD4, CCR5 and CXCR4 (HIV), 59 CD13 (coronavirus and cytomegalovirus) 60 , 61 and CD46 (measles virus; MV). 62 The most extensively studied example of DC involvement in infection is HIV. This lentivirus can remain latent in DC and exploits the trafficking of DC towards lymphoid tissue as a strategy to enhance the infection of permissive CD4+ lymphocytes. 63 , 64 The HIV appears to be activated in DC by CD40 ligation or the presence of Th cells. The activation status of the DC themselves is thought to have an impact on viral replication, with immature and cutaneous DC supporting productive infection of macrophage tropic virus, while mature DC are able to transport HIV but appear unable to replicate both T cell and macrophage tropic strains of virus. 65 A further interesting observation is that viruses derived from infected DC carry T cell‐specific factors that make them highly infectious. 66 The mechanisms used by HIV to subvert antigen presentation by DC undoubtedly have direct parallels in other viral infections. For example, activation of MV‐infected DC with CD40L or with CD4+ T cells results in a profound viral replicative event, which ultimately leads to cell death. 67 The aforementioned activation of DC by CD40L occurs primarily in the lymph node and it appears that this mechanism will prevent both MV‐ and HIV‐infected DC from successfully presenting antigen to T cells. 68
Mechanisms for interfering directly with antigen presentation, such as those used by adenoviruses, EBV and CMV, may also prevent DC from effectively modulating CTL responses. 69 For example, many human herpesviruses, including EBV (HHV4), encode a viral homologue of IL‐10, which will suppress the ability of professional APC such as the DC to produce effective CTL responses. 70 Human cytomegalovirus (HCMV) causes severe morbidity in immunocompromised patients and again subverts immunosurveillance using a latent ‘protein‐free’ replicative stage. Human cytomegalovirus remains latent in the CD33+ myeloid progenitors of monocytes and dendritic cells. Reactivation has been observed when these cells are treated with cytokines that promote differentiation of DC (TNF‐α, IL‐4 and granulocyte–macrophage colony stimulating factor (GM‐CSF)). Reactivation under the influence of these signals would be an ideal mechanism for interfering with antigen presentation. The inadequate antigen‐presenting ability of Langerhans cells that express hepatitis C virus genes is thought to reflect active interference with antigen presentation by an indeterminate mechanism, perhaps involving perturbed IL‐12 production. 71
The picture for other intracellular parasites is somewhat similar. Effective Th1‐type immune responses are essential for the generation of immunity towards parasites such as Histoplasma, Leishmania and Mycobacterium spp. 72 , 73 , 74 It appears that DC secretion of Th1‐promoting cytokines, such as IL‐12, is inhibited by infection with these parasites. 75 Other microbes, such as the sexually transmitted intracellular pathogen Chlamydia spp., have been detected in DC, 76 although the mechanisms that afford this organism relative freedom from immunosurveillance still remain to be clarified. A Salmonella sp. was recently discovered to both infect and survive within DC and, as with HIV, this may be an important mechanism for disseminating the disease away from mucosal sites. 77 In summary, many organisms use DC as bases for the evasion of immunosurveillance. Subversion of DC antigen presentation and activation is a newly recognized phenomenon, which helps to explain the persistent nature of some infections.
Autoimmunity
Dendritic cells have been studied in a number of common autoimmune conditions. Despite the diverse nature and tissue distribution of autoimmune diseases, DC share certain common characteristics irrespective of disease and sites from which they are recovered. These include increased numbers of tissue DC, particularly in a perivascular distribution, DC infiltration at the earliest stage of disease and an altered DC phenotype. Early DC infiltration contributes to the recruitment of other immune cells.
Rheumatoid arthritis (RA) is a chronic destructive autoimmune disease of poorly defined pathogenesis with a prevalence approaching 1%. For almost two decades, the helper T cell has been considered to be the mediator of the immune response leading to joint destruction. This is based on the strong HLA DR4 association with RA, on histological features of the synovium and on the trend towards a response in RA with T cell‐modulating therapies. However, the primacy of T cells in RA has been challenged and several authors have investigated DC expression and function in RA. Increased numbers of DC have been shown in both the peripheral blood, synovial fluid exudates 78 , 79 and synovial tissues in patients with RA. 80
It has been argued that RA is due to DC presentation of endogenous self peptides. 81 This is a feasible hypothesis, but a more important question is: what factors result in synovial DC activation? Both TNF‐α and GM‐CSF are abundant in the synovium, which could contribute to activation, as could mechanical factors such as trauma. Pro‐inflammatory mediators, such as TNF‐α, could be up‐regulated in the synovium by rheumatoid‐factor auto‐antibodies directed against the Fc moiety of immunoglobulins, which could lead to macrophage activation by complement. The plethora of cellular and molecular abnormalities reported in RA could therefore be secondary to the ravages of chronic inflammation rather than being of primary pathogenic importance. Anti‐TNF‐α therapy is an exciting development in the treatment of RA. The sustained immunomodulatory effect of anti‐TNF therapy in early RA could be due to modulation of DC function down‐ regulating both costimulatory signals and DC trafficking. 82
Seronegative polyarthritis includes the related conditions ankylosing spondylitis (AS), reactive arthritis (ReA), psoriatic arthritis and undifferentiated arthritis. 83 These diverse clinical entities are interrelated clinically by spinal inflammation and at an immunological level by the presence of the MHC class I molecule HLA B27. In the HLA B27 transgenic rat model of AS there is evidence that DC play a critical function, because disease can be transferred by bone marrow cells presumed to be DC. 84
Putative activation signals for seronegative arthritis have been identified. Bacteria may preferentially home to the joint tissues and activate an immune response. 85 , 86 Another signal could be TNF‐α, which can induce similar disease phenotypes in experimental models to human AS. 87 The synovial inflammation seen in these conditions could be related to pro‐inflammatory cytokines released from the adjacent joint capsule. Direct mechanical trauma to the tissues could also serve to initiate cell‐mediated immunity in these conditions.
Dendritic cell infiltration is an early feature of islet cell autoimmunity in diabetes mellitus 88 and contributes to local lymphoid tissue formation in the islets cells. 89 What could the signal for DC activation be in autoimmune diabetes? The destructive processes are directed against the pancreatic β cell. A viral tropism for the β cell is the one mechanism that has been postulated to initiate autoimmunity. Clues to DC activation signals for endocrine disease in general come from studies in thyroid disease, which show that DC infiltration is preceded by metabolic abnormalities in the thyroid gland itself, 90 suggesting that some types of autoimmunity are due to primary abnormalities in the tissue targeted.
Psoriasis is a common skin condition with a prevalence of about 2%. The skin has a rich source of Langerhans cells, which are thought to be important in the pathogenesis of inflammation at that site. Streptococcal bacteria and IFN‐γ have both been implicated in the pathogenesis of psoriasis 91 and will also influence the activation status of DC. A recently identified cutaneous danger signal may be IL‐1. This is based on experiments that show that mechanical stressing of keratinocytes results in liberation of high concentrations of IL‐1, 92 which is noteworthy because the most common sites of psoriasis are the elbows and knees, where the skin is subject to considerable trauma and stretching.
Knowledge of the central role of DC in autoimmune disease is important for both determining the mechanism of action of currently available therapies and for the development of future therapeutic strategies.
Not all studies on DC support an activated DC phenotype in autoimmune disease, with some studies showing that expression of costimulatory molecules is down‐regulated at the sites of disease. 93 , 94 , 95 This could be due to an immunomodulatory response at these sites to reduce the severity of inflammation. However, these changes have been interpreted as representing a primary defect in DC function that could theoretically allow auto‐aggressive T cell clones to emerge during thymic selection. For such a contention to be supported, DC abnormalities would need to be demonstrated prior to the chronic phases of the respective diseases.
Cancer
The cell‐mediated arm of the immune system plays an important role in the detection and elimination of malignant cells. T lymphocyte responses to tumour cells will require initial antigen presentation by professional APC such as the DC. Dendritic cell infiltration into a tumour has for many years been linked to increased patient survival and reduction in the number of metastases in a variety of malignancies (including endometrial, 96 gastric 97 and lung 98 cancers). This positive prognostic indicator is tempered by the observation that DC isolated from cancer patients show an impaired ability to present antigen as a product of decreased surface expression of the costimulators CD80 and CD86. 99 , 100
It also appears that many sporadic tumours actively exploit immunosuppressive pathways to interfere with antigen presentation. Tumour‐derived IL‐10 acts directly on tissue DC to prevent maturation and therefore subsequent immunogenic presentation to T lymphocytes. 101 Increased expression of FasL on tumour cells may also induce apoptosis in DC and T cell effectors. 102 , 103 Vascular endothelial growth factor (VEGF) is produced by a majority of carcinomas and has been shown to inhibit maturation of DC within the tumour and to impair differentiation of haemopoietic progenitors into DC. 104 An in vitro study using a renal cell carcinoma cell line has shown that factors are released that inhibit the differentiation of dendritic cells from CD34+ progenitors. Interleukin‐6 and macrophage (M)‐CSF have been subsequently identified as the factors mediating this inhibition via the down‐regulation of the GM‐CSF receptor. 105 , 106 These mechanisms represent parodies of the normal processes intended to establish tolerance between a tissue and the immune system. The role of the DC in the pathogenesis of sporadic cancers is still poorly understood.
Transplantation
There is a great deal of interplay between the acquired and innate arms of the immune system during allotransplantation. However, it is the action of cytotoxic T cells that determine whether a graft survives or is rejected. The DC, with its ability to control primary T lymphocyte responses, will therefore initiate attack on an allograft by the presentation of alloantigen. Tissue damage during transplantation will ‘stress’ the host and graft tissues to thresholds that induce DC maturation via the release of, for example, the pro‐inflammatory cytokines TNF‐α and IL‐1β. Activated alloantigen‐presenting DC will then migrate away from the graft to present antigen primarily in host lymphoid tissue. 107 The presentation of peptide by donor DC in the context of foreign MHC molecules will activate both helper (CD4+) and cytotoxic (CD8+) T cells. Cytotoxic T cells generated against alloantigen will then move into the circulation and eventually initiate an attack on graft cells expressing the specific peptides that were presented by the DC.
The role of the DC in graft rejection is exemplified by the observation that graft survival can be dramatically increased by DC depletion. 108 In addition, infusions of costimulator‐negative DC are able to prolong the life of similar allografts by inducing T cell hyporesponsiveness. 109 Indeed, the paucity of corneal allotransplant rejection is thought to represent the low level of DC within this tissue. Modulation of cytokines that directly affect the maturation of DC will also influence graft survival; for example, blockade of CD40 signalling in DC has been shown to prolong cardiac graft survival. 110 In a recent study on the biology of the cytokine IL‐17, it was shown that not only does this factor induce the functional maturation of DC, but a novel antagonist to IL‐17 has been shown to promote the survival of cardiac grafts. 111 , 112
Therapeutic applications
The observation that DC play a role in the progression and severity of many diseases has led many researchers to investigate how modulation of DC function could be used therapeutically. The power of the activated DC could be used in cancer or intracellular parasite infection to redirect an immune response towards defective (malignant) or infected cells that have evaded immunosurveillance (Figure 3). Alternatively, DC maturation could be suppressed to alleviate the symptoms of autoimmunity and allotransplant rejection.
Figure 3.
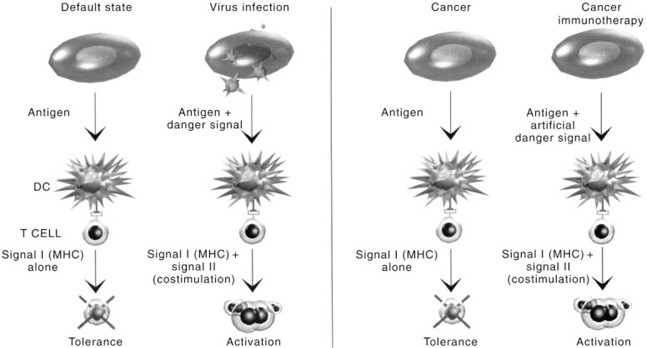
Dendritic cell immunotherapy of cancer. A state of immunological tolerance usually exists between the tumour and the host. Infusions of activated, tumour antigen‐loaded dendritic cells (DC) stimulate the proliferation of antigen‐specific anti‐tumour CTL.
Advances in the extraction and in vitro culture of DC have been a major driving force behind the recent increased interest in these cells and have facilitated the inclusion of these powerful adjuvants in therapeutic trials. Refined laboratory protocols are now available for either the generation of DC from a number of readily available sources 113 or for the direct isolation of DC from mixed cell populations. 114 Cells that have been found to yield DC, after culture in lineage‐ restricting cocktails of cytokines, include CD34+ stem cells 115 , 116 , 117 , 118 and CD14+ monocytes. 119 , 120 , 121 CD14+ monocytes are perhaps the most readily available precursors used to generate human DC, because they constitute some 7–8% of human PBMC. The simplest method of obtaining DC is by allowing PBMC to adhere to tissue culture dishes for 2 h. After several washes, the adherent fraction will be enriched for CD14+ monocytes. Immature DC can then be produced using GM‐CSF + IL‐4/IL‐13. The GM‐CSF has the effect of restricting differentiation towards a myelomonocytic lineage, while IL‐4 or IL‐13 inhibits the development of macrophages. 122 The overall result is a forced differentiation towards a DC lineage. Cells obtained from in vitro culture with high doses of GM‐CSF and IL‐4/IL‐13 resemble immature DC, in that they have a high affinity for soluble antigen and express lower levels of costimulatory molecules than the activated form. 123 Subsequent addition of TNF‐α, LPS or CD40L to a culture of these DC promotes maturation and activation, as would be expected from the discussion of DC life history presented earlier. High‐purity DC cultures can also be obtained by immunomagnetic selection. 124 Contaminating leucocyte subsets can be depleted with a cocktail of antibodies against T cells (CD3), B cells (CD19), NK cells (CD56), monocytes (CD14) and macrophages (CD16), leaving a fraction enriched for DC. Alternatively, DC can be positively selected from mixed cultures using antibodies against antigens that have DC‐restricted expression, for example CD83 125 or CMRF‐44. 126
Dendritic cell‐based immunotherapy of cancer has been demonstrated in a number of studies using murine tumours. Established malignancies ranging from melanoma 127 to mammary carcinoma 128 have all been successfully regressed using infusions of antigen‐loaded, activated DC. The generation of specific tumour‐specific CTL requires the introduction of antigen into the DC, thus providing a target for immunity. The DC are able to present antigen that has been introduced by a variety of methods, including plasmid, 129 mRNA, 130 peptide 131 (eluted and synthetic), cell lysate 132 and recombinant viral vectors, including adenovirus 133 and vaccinia virus. 134
Data relating to human DC clinical trials are scarce, but certain studies have reported encouraging results. One study has used DC pulsed with tumour lysate or peptide for the treatment of patients with metastatic melanoma. 135 Of the 16 patients immunized, five had objective responses (30%) with two subjects experiencing considerable disease regression. Malignant melanoma, due to its intrinsic antigenicity, is an ideal target for a DC‐based immunotherapy. Of the published studies on the use of DC for the treatment of prostate cancer, one of most promising cites that nine of 33 patients (27%) responded to immunization. 136 Hsu et al. treated patients suffering from follicular B cell lymphoma with DC and tumour‐specific idiotype protein. This resulted in a marked antitumour immune responses in all four patients. 137 For B cell lymphoma, the presence of a truly tumour‐specific antigen is the ideal in terms of a DC‐based immunotherapy, because it will reduce autoimmune complications arising from coexpression of antigens on normal tissue. For example, patients immunized against melanoma‐derived antigens (e.g. MART‐1) sometimes experienced an autoimmune vitelligo resulting from destruction of normal melanocytes by autoreactive CTL. 135 It is suspected that DC immunotherapy of solid tumours will be considerably less efficacious than for melanoma or leukaemia, perhaps due in part to the higher incidence of HLA antigen dysregulation in these tumours and the paucity of good tumour‐restricted antigens. Further increases in our understanding of how DC are activated will allow us to associate more ‘danger’ with the tumour antigens under investigation.
Culturing DC ex vivo for tumour immunotherapy represents a considerable challenge. One alternative may be to use infusions of factors that directly promote DC differentiation, in an effort to increase the level of antitumour immunosurveillance. A case in point is the haemopoietic growth factor Flt‐3. This growth factor has been shown to increase the numbers of DC and monocytes in peripheral blood without producing the serious side effects that are a consequence of using other cytokines/growth factors as adjuvants (e.g. IL‐2, IFN‐γ). 138 Flt‐3 is able to induce protective antitumour immunity in some animal models, 139 , 140 which is thought to be a consequence of increased presentation of tumour antigens combined with increased NK cell activity. 141 The synergy of a DC growth‐promoting factor, such as Flt‐3 ligand, with a DC activation signal, such as CD40L or tumour necrosis factor‐related activation‐induced cytokine (TRANCE), may provide additional benefits by promoting CTL generation from an already expanded pool of DC.
With respect to persistent viral disease, DC function could be modulated to reactivate silent T cells and lessen the effects of relapse. Diseases that may be amenable to this form of intervention include viral hepatitis (HBV and HCV), papillomavirus infection and chronic HCMV infection. Optimizing DC therapies for persistent viral disease is hampered by a lack of appropriate animal models. However, it is encouraging to note that in a murine model of chronic hepatitis, where mice are transgenic for hepatitis B surface antigen (HBsAg), tolerance to HBsAg can be broken by immunization with cytokine‐activated DC. 142 It is inferred from the earlier discussion of DC life history that, for immunotherapy of cancer and viral disease, DC activated by members of the tumour necrosis family of ligands (e.g. TNF‐α or CD40L) will be the best candidates for breaking immunological tolerance. 143 , 144
In situations where intracellular parasites interfere with DC function, there may be some opportunity to correct the defects. For example, DC secretion of IL‐12 is inhibited by Leishmania sp. and prevents an effective Th1‐type immune response, which is crucial for parasite clearance. Infusions of parasite‐pulsed DC, in combination with IL‐12 or other Th1‐promoting cytokines, may help to reduce parasite burden by repolarizing immunity. It has been suggested that a possible therapy for HIV could involve infusions of IL‐12‐transfected DC in an attempt to counter the suppression of Th1 immunity that is a characteristic of this infection.
Conversely, T lymphocytes could be anergized by direct suppression of DC activation for the treatment of autoimmune diseases and for reducing the severity of allotransplant rejection. Enhancement of the ability of DC to tolerize T lymphocytes could be accomplished in a number of ways, for example by pretreatment of DC with maturation‐suppressing factors such as IL‐10 and TGF‐β. 145 One recent study has shown that infusions of Flt‐3 ligand can increase the survival time of skin allografts when administered in combination with a factor such as anti‐CD40L, which will prevent DC activation. 146 In a further landmark study, it has been found that DC transfected with CD95L could induce antigen‐specific tolerance after being pulsed with a peptide to which they had previously been sensitized. 147 This observation provides proof that it may also be possible to delete autoreactive T cells from the repertoire using modified DC.
Conclusions
In the present review, we have described how an in‐depth understanding of DC biology has provided insights into the coordination of both innate and adaptive immunity. The importance of these cells in a number of disease states is also being gradually revealed. It is therefore realistic that in the near future, once the lineage, danger and tolerance paradigms have been fully resolved, it will be possible to modulate DC function for considerable therapeutic benefit.
Acknowledgements
We would like to offer our sincere thanks to Stephen Richards, Richard Jones and Peter Cotterrell for their constructive comments regarding the manuscript. Laboratory work is supported by grants from Yorkshire Cancer Research, The Arnold Tunstall Fellowship and The Medical Research Council.
References
- 1. Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature 1998; 392: 245–52. [DOI] [PubMed] [Google Scholar]
- 2. Croft M, Duncan DD, Swain SL. Response of naive antigen‐ specific CD4+ T cells in vitro: Characteristics and antigen presenting requirements. J. Exp. Med. 1992; 176: 1431–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Ronchese F, Hausmann B. B lymphocytes in vivo fail to prime naive T cells but can stimulate antigen‐experienced T lymphocytes. J. Exp. Med. 1993; 177: 679–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Inaba K, Steinman RM, Van Voorhis WC, Muramatsu S. Dendritic cells are critical accessory cells for thymus dependent antibody responses in mouse and man. Proc. Natl Acad. Sci. USA 1983; 80: 6041–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Kitamura H, Iwakabe K, Yahata T et al The natural killer T (NKT) cell ligand alpha‐galactosylceramide demonstrates its immunopotentiating effect by inducing interleukin (IL)‐12 production by dendritic cells and IL‐12 receptor expression on NKT cells. J. Exp. Med. 1999; 189: 1121–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Brocker T. MHC class II expression on dendritic cells is necessary and sufficient for survival of CD4+ T cells. J. Exp. Med. 1997; 186: 1223–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Brocker T, Riedinger M, Karjalainen K. Targeted expression of MHC class II molecules demonstrates that dendritic cells can induce negative but no positive selection of thymocytes in vivo. J. Exp. Med. 1997; 185: 541–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Thomas R, Lipsky PE. Human peripheral blood dendritic cell subsets. Isolation and characterisation of precursor and mature antigen‐presenting cells. J. Immunol. 1994; 153: 4016–28. [PubMed] [Google Scholar]
- 9. Ardavin C, Wu L, Li CL, Shortman K. Thymic dendritic cells and T cells develop simultaneously in the thymus from a common precursor population. Nature 1993; 362: 761–3. [DOI] [PubMed] [Google Scholar]
- 10. Tew JG, Wu J, Qin D, Helm S, Burton GF, Szakal AK. Follicular dendritic cells and presentation of antigen and costimulatory signals to B cells. Immunol. Rev. 1997; 156: 39–52. [DOI] [PubMed] [Google Scholar]
- 11. Norbury CC, Chambers BJ, Prescott AR, Ljunggren HG, Watts C. Constitutive macropinocytosis allows TAP‐dependent major histocompatibility complex class I presentation of exogenous soluble antigen by bone marrow‐derived dendritic cells. Eur. J. Immunol. 1997; 27: 280–8. [DOI] [PubMed] [Google Scholar]
- 12. Fanger NA, Wardwell K, Shen L, Tedder TF, Guyre PM. Type I (CD64) and type II (CD32) Fc gamma receptor‐mediated phagocytosis by human blood dendritic cells. J. Immunol. 1996; 157: 541–8. [PubMed] [Google Scholar]
- 13. Fanger NA, Viogtlaender D, Liu C et al Characterisation of expression, cytokine regulation, and effector function of the high affinity IgG receptor Fc gamma RI (CD64) expressed on human blood dendritic cells. J. Immunol. 1997; 158: 3090–8. [PubMed] [Google Scholar]
- 14. Maurer D, Fiebiger E, Reininger B et al Fc Epsilon receptor I on dendritic cells delivers IgE bound multivalent antigens into a cathepsin S‐dependent pathway of MHC class II presentation. J. Immunol. 1998; 161: 2731–9. [PubMed] [Google Scholar]
- 15. Green I, Stingl G, Shevach EM, Katz SI. Antigen presentation and allogeneic stimulation by Langerhans cells. J. Invest. Dermatol. 1980; 75: 44–5. [DOI] [PubMed] [Google Scholar]
- 16. Sallusto F, Cella M, Danieli C, Lanzavecchia A. Dendritic cells use macropinocytosis and the mannose receptor to concentrate macromolecules in the major histocompatibility complex class II compartment: Downregulation by cytokines and bacterial products. J. Exp. Med. 1995; 182: 389–400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Jiang W, Swiggard WJ, Heufler C et al The receptor DEC‐205 expressed by dendritic cells and thymic epithelial cells is involved in antigen processing. Nature 1995; 375: 151–5. [DOI] [PubMed] [Google Scholar]
- 18. Unger J, Phares W. Differential gene expression in cultured human Langerhans cells in response to phagocytic stimulation. Adv. Exp. Med. Biol. 1997; 417: 475–80. [DOI] [PubMed] [Google Scholar]
- 19. Engel P, Tedder TF. New CD from the B cell section of the fifth international workshop on human leukocyte differentiation antigens. Leuk. Lymphoma 1994; 13: 61–4. [DOI] [PubMed] [Google Scholar]
- 20. Shen Z, Reznikoff G, Dranoff G, Rock KL. Cloned dendritic cells can present exogenous antigens on both MHC class I and class II molecules. J. Immunol. 1997; 158: 2723–30. [PubMed] [Google Scholar]
- 21. Brossart P, Bevan MJ. Presentation of exogenous protein antigens on major histocompatibility complex class I molecules by dendritic cells: Pathway of presentation and regulation by cytokines. Blood 1997; 90: 1594–9. [PMC free article] [PubMed] [Google Scholar]
- 22. Henderson RA, Watkins SC, Flynn JL. Activation of human dendritic cells following infection with mycobacterium tuberculosis. J. Immunol. 1997; 159: 635–43. [PubMed] [Google Scholar]
- 23. Cella M, Salio M, Sakakibara Y, Langen H, Julkunen I, Lanzavecchia A. Maturation activation, and protection of dendritic cells induced by double stranded RNA. J. Exp. Med. 1999; 189: 821–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. De Smedt T, Pajak B, Muraille E et al Regulation of dendritic cell numbers and maturation by lipopolysaccharide in vivo. J. Exp. Med. 1996; 184: 1413–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Sparwasser T, Koch ES, Vabulas RM et al Bacterial DNA and immunostimulatory CpG oligonucleotides trigger maturation and activation of murine dendritic cells. Eur. J. Immunol. 1998; 28: 2045–54. [DOI] [PubMed] [Google Scholar]
- 26. Yang RB, Mark MR, Gray A et al Toll‐like receptor‐2 mediates lipopolysaccharide‐induced cellular signalling. Nature 1998; 395: 284–8. [DOI] [PubMed] [Google Scholar]
- 27. Poltorak A, He X, Smirnova I et al Defective LPS signaling in C3H/Hej and C57BL/10ScCr mice: Mutations in Tlr4 gene. Science 1998; 282: 2085–8. [DOI] [PubMed] [Google Scholar]
- 28. Regnault A, Lankar V, Lacabanne A et al FcγR‐mediated induction of dendritic cell maturation and MHC class I‐restricted antigen presentation after immune complex internalisation. J. Exp. Med. 1999; 189: 371–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Ludewig B, Graf D, Gelderblom HR, Becker Y, Kroczek RA, Pauli G. Spontaneous apoptosis of dendritic cells is efficiently inhibited by TRAP (CD40‐ligand) and TNF‐alpha, but strongly enhanced by interleukin‐10. Eur. J. Immunol. 1995; 25: 1943–50. [DOI] [PubMed] [Google Scholar]
- 30. Lukas M, Stossal H, Hefel L et al Human cutaneous dendritic cells migrate through dermal lymphatic vessels in a skin organ culture model. J. Invest. Dermatol. 1996; 106: 1293–9. [DOI] [PubMed] [Google Scholar]
- 31. Jonuleit H, Kuhn U, Muller G et al Pro‐inflammatory cytokines and prostaglandins induce maturation of potent immunostimulatory dendritic cells under fetal calf serum free conditions. Eur. J. Immunol. 1997; 27: 3135–42. [DOI] [PubMed] [Google Scholar]
- 32. Rovere P, Vallinoto C, Bondanza A et al Bystander apoptosis triggers dendritic cell maturation and antigen‐presenting function. J. Immunol. 1998; 161: 4467–71. [PubMed] [Google Scholar]
- 33. Albert ML, Pearce SF, Francisco LM et al Immature dendritic cells phagocytose apoptotic cells via alpha beta5 and CD36, and cross‐present antigens to cytotoxic T lymphocytes. J. Exp. Med. 1998; 188: 1359–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Sallusto F, Palermo B, Lenig D et al Distinct patterns and kinetics of chemokine production regulate dendritic cell function. Eur. J. Immunol. 1999; 29: 1617–25. [DOI] [PubMed] [Google Scholar]
- 35. Sozzani S, Allavena P, D'Amico G et al Differential regulation of chemokine receptors during dendritic cell maturation: A model for their trafficking properties. J. Immunol. 1998; 161: 1083–6. [PubMed] [Google Scholar]
- 36. Delgado E, Finkel V, Baggiolini M, Mackay CR, Steinman RM, Granelli‐Piperno A. Mature dendritic cells respond to SDF‐1, but not to several beta‐chemokines. Immunobiology 1998; 198: 490–500. [DOI] [PubMed] [Google Scholar]
- 37. Lin CL, Suri RM, Rahdon RA, Austyn JM, Roake JA. Dendritic cell chemotaxis and transendothelial migration are induced by distinct chemokines and are regulated on maturation. Eur. J. Immunol. 1998; 28: 4114–22. [DOI] [PubMed] [Google Scholar]
- 38. Kellermann SA, Hudak S, Oldham ER, Liu YJ, McEvoy LM. The CC chemokine receptor‐7 ligands 6Ckine and macrophage inflammatory protein‐3 beta are potent chemoattractants for in vitro and in vivo derived dendritic cells. J. Immunol. 1999; 162: 3859–64. [PubMed] [Google Scholar]
- 39. Tang A, Amagai M, Granger LG, Stanley JR, Udey MC. Adhesion of epidermal Langerhans cells to keratinocytes mediated by E‐cadherin. Nature 1993; 361: 82–5. [DOI] [PubMed] [Google Scholar]
- 40. Cumberbatch M, Dearman RJ, Kimber I. Langerhans cells require signals from both tumour necrosis factor alpha and interleukin 1 beta for migration. Adv. Exp. Med. Biol. 1997; 417: 125–8. [DOI] [PubMed] [Google Scholar]
- 41. Pierre P, Turley SJ, Gatti E et al Developmental regulation of MHC class II transport in mouse dendritic cells. Nature 1997; 388: 787–92. [DOI] [PubMed] [Google Scholar]
- 42. Sallusto F, Lanzavecchia A. Efficient presentation of soluble antigen by cultured dendritic cells is maintained by granulocyte/macrophage colony‐stimulating factor plus interleukin 4 and down regulated by tumour necrosis factor alpha. J. Exp. Med. 1994; 179: 1109–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Schuler G, Romani N, Steinman RM. A comparison of murine epidermal Langerhans cells with spleen dendritic cells. J. Invest. Dermatol. 1985; 85: S99–106. [DOI] [PubMed] [Google Scholar]
- 44. Adema GJ, Hartgers F, Verstraten R et al A dendritic‐cell‐derived C‐C chemokine that preferentially attracts naive T cells. Nature 1997; 387: 713–17. [DOI] [PubMed] [Google Scholar]
- 45. Schaniel C, Pardali E, Sallusto F et al Activated murine B lymphocytes and dendritic cells produce a novel CC chemokine which acts selectively on activated T cells. J. Exp. Med. 1998; 188: 451–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Rissoan M, Soumelis V, Kadowaki N et al Reciprocal control of T helper cell and dendritic cell differentiation. Science 1999; 283: 1183–6. [DOI] [PubMed] [Google Scholar]
- 47. Cella M, Scheidegger D, Palmer‐Lehmann K, Lane P, Lanzavecchia A, Alber G Ligation of CD40 on dendritic cells triggers production of high levels of interleukin‐12 and enhances T cell stimulatory capacity: T‐T help via APC activation. J. Exp. Med. 1996; 184: 747–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Kuniyoshi JS, Kuniyoshi CJ, Lim AM et al Dendritic cell secretion of IL‐15 is induced by recombinant huCD40LT and augments the stimulation of antigen‐specific cytolytic T cells. Cell. Immunol. 1999; 193: 48–58. [DOI] [PubMed] [Google Scholar]
- 49. Kushnir N, Liu L, MacPherson GG. Dendritic cells and resting B cells form clusters in vitro and in vivo: T cell independence, partial LFA‐1 dependence, and regulation by cross‐linking surface molecules. J. Immunol 1998; 160: 1774–81. [PubMed] [Google Scholar]
- 50. Bjorck P, Flores‐Romo L, Liu YL. Human interdigitating dendritic cells directly stimulate CD40‐activated naive B cells. Eur. J. Immunol. 1997; 27: 1266–74. [DOI] [PubMed] [Google Scholar]
- 51. Fayette J, Durand I, Bridon JM et al Dendritic cells enhance the differentiation of naive B cells into plasma cells in vitro. Scand. J. Immunol. 1998; 48: 563–70. [DOI] [PubMed] [Google Scholar]
- 52. Dubois B, Barthelemy C, Durand I, Liu YL, Caux C, Briere F. Towards a role of dendritic cells in the germinal center reaction: Triggering of B cell proliferation and isotype switching. J. Immunol. 1999; 162: 3428–36. [PubMed] [Google Scholar]
- 53. Dubois B, Massacrier C, Vanbervliet B et al Critical role of IL‐12 in dendritic cell‐induced differentiation of naive B lymphocytes. J. Immunol. 1998; 161: 2223–31. [PubMed] [Google Scholar]
- 54. Fayette J, Dubois B, Vandenabeele S et al Human dendritic cells skew isotype switching of CD40‐activated naive B cells towards IgA1 and IgA2. J. Exp. Med. 1997; 185: 1909–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Winzler C, Rovere P, Rescigno M et al Maturation stages of mouse dendritic cells in growth factor‐dependent long term cultures. J. Exp. Med. 1997; 185: 317–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Parajuli P, Nishioka Y, Nishimura N et al Cytolysis of human dendritic cells by autologous lymphokine‐activated killer cells: Participation of both T cells and NK cells in the killing. J. Leukoc. Biol. 1999; 65: 764–70. [DOI] [PubMed] [Google Scholar]
- 57. Ludewig B, Ehl S, Karrer U, Odermatt B, Hengartner H, Zinkernagel RM. Dendritic cells efficiently induce protective antiviral immunity. Virology 1998; 72: 3812–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Flohe SB, Bauer C, Flohe S, Moll H. Antigen‐pulsed epidermal Langerhans cells protect susceptible mice from infection with the intracellular parasite Leishmania major. Eur. J. Immunol. 1998; 28: 3800–11. [DOI] [PubMed] [Google Scholar]
- 59. Baluvelt A. The role of skin dendritic cells in the initiation of human immunodeficiency virus infection. Am. J. Med. 1997; 102: 16–20. [DOI] [PubMed] [Google Scholar]
- 60. Yeager CL, Ashmun RA, Williams RK et al Human aminopeptidase N is a receptor for human coronavirus 229E. Nature 1992; 357: 420–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Soderberg C, Giugni TD, Zaia JA, Larsson S, Wahlberg JM, Moller E. CD13 (human aminopeptidase N) mediates human cytomegalovirus infection. J. Virol. 1993; 67: 6576–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Schnorr JJ, Xanthakos S, Keikavoussi P, Kampgen E, ter Meulen V, Schneider‐Schaulies S. Induction of maturation of human blood dendritic cell precursor by measles virus is associated with immunosuppression. Proc. Natl Acad. Sci. USA 1997; 94: 5326–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Masurier C, Salomon B, Guettari N et al Dendritic cells route human immunodeficiency virus to lymph nodes after vaginal or intravenous administration to mice. J. Virol. 1998; 72: 7822–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Granelli‐Piperno A, Finkel V, Delgado E, Steinman RM. Virus replication begins in dendritic cells during the transmission of HIV‐1 from mature dendritic cells to T cells. Curr. Biol. 1999; 14: 21–9. [DOI] [PubMed] [Google Scholar]
- 65. Granelli‐Piperno A, Delgado E, Finkel V, Paxton W, Steinman RM. Immature dendritic cells selectively replicate macrophage tropic (M‐tropic) human immunodeficiency virus type 1, while mature cells efficiently transmit both M‐ and T‐tropic virus to T cells. J. Virol. 1998; 72: 2733–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Frank I, Kacani L, Stoiber H et al Human immunodeficiency virus type 1 derived from cocultures of immature dendritic cells with autologous T cells carries T‐cell‐specific molecules on its surface and is highly infectious. J. Virol. 1999; 73: 3449–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Fugier‐Vivier I, Servat‐Delprat C, Rivailler P, Rissoan MC, Lui YL, Rabourdin‐Combe C. Measles suppresses cell‐mediated immunity by interfering with the survival and functions of dendritic and T‐cells. J. Exp. Med. 1997; 186: 813–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Grosjean I, Caux C, Bella C et al Measles virus infects human dendritic cells and blocks their allostimulatory properties for CD4+ T‐cells. J. Exp. Med. 1997; 186: 801–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Wiertz EJHJ, Mukherjee S, Ploegh HL. Viruses use stealth technology to escape from host immune system. Mol. Med. Today 1997; 3: 116–23. [DOI] [PubMed] [Google Scholar]
- 70. Suzuki T, Tahara H, Narula S, Moore KW, Robbins PD, Lotze MT. Viral interleukin 10 (IL‐10), the human herpes virus 4 cellular IL‐10 homologue, induces local anergy to allogenic and syngeneic tumours. J. Exp. Med. 1995; 182: 477–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Hiasa Y, Horiike N, Akbar SM et al Low stimulatory capacity of lymphoid dendritic cells expressing hepatitis C virus genes. Biochem. Biophys. Res. Commun. 1998; 249: 90–5. [DOI] [PubMed] [Google Scholar]
- 72. Zhou P, Seder RA. CD40 ligand is not essential for induction of type 1 cytokine responses or protective immunity after primary or secondary infection with histoplasma capsulatum. J. Exp. Med. 1998; 187: 1313–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Schopf LR, Bliss JL, Lavigne LM, Chung CL, Sypek JP, Wolf SF. Interleukin‐12 is capable of generating an antigen‐specific Th1‐type response in the presence of an ongoing infection‐driven Th2‐type response. Infect. Immun. 1999; 67: 2166–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Thompson‐Snipes L, Skamene E, Radzioch D. Acquired resistance but not innate resistance to mycobacterium bovis bacillus Calmette‐Guerin is compromised by interleukin‐12 ablation. Infect. Immun. 1998; 66: 5268–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Ahuja SS, Mummidi S, Malech HL, Ahuja SK. Human dendritic cell (DC)‐based anti‐infective therapy: Engineering DCs to secrete functional IFN‐gamma and IL‐12. J. Immunol. 1998; 161: 868–76. [PubMed] [Google Scholar]
- 76. Ojcius DM, Bravo de Alba Y, Kanellopoulos JM et al Internalisation of Chlamydia by dendritic cells and stimulation of Chlamydia‐specific T cells. J. Immunol 1998; 160: 1297–303. [PubMed] [Google Scholar]
- 77. Marriott I, Hammond TG, Thomas EK, Bost KL. Salmonella efficiently enter and survive within cultured CD11c+ dendritic cells initiating cytokine expression. Eur. J. Immunol. 1999; 29: 1107–15. [DOI] [PubMed] [Google Scholar]
- 78. Zvaifler NJ, Steinman RM, Kaplan G, Lau LL, Rivelis M. Identification of immunostimulatory dendritic cells in the synovial effusions of patients with rheumatoid arthritis. J. Clin. Invest. 1985; 76: 789–800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Harding B, Knight SC. The distribution of dendritic cells in the synovial fluids of patients with arthritis. Clin. Exp. Immunol. 1986; 63: 594–600. [PMC free article] [PubMed] [Google Scholar]
- 80. Thomas R, Davis LS, Lipsky PE. Rheumatoid synovium is enriched in mature antigen‐presenting dendritic cells. J. Immunol. 1994; 152: 2613–23. [PubMed] [Google Scholar]
- 81. Thomas R, Lipsky PE. Presentation of self peptides by dendritic cells: Possible implications for the pathogenesis of rheumatoid arthritis. Arthritis Rheum. 1996; 39: 183–90. [DOI] [PubMed] [Google Scholar]
- 82. Camussi G, Lupia E. The future role of anti‐tumour necrosis factor (TNF) products in the treatment of rheumatoid arthritis. Drugs 1998; 55: 613–20. [DOI] [PubMed] [Google Scholar]
- 83. McGonagle D, Gibbon W, Emery P. Classification of inflammatory arthritis by enthesitis. Lancet 1998; 352: 1137–40. [DOI] [PubMed] [Google Scholar]
- 84. Breban M, Hammer RE, Richardson JA, Taurog JD. Transfer of the inflammatory disease of HLA‐B27 transgenic rats by bone marrow engraftment. J. Exp. Med. 1993; 178: 1607–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Schulz LC, Schaening U, Pena M, Hermanns W. Borderline‐tissues as sites of antigen deposition and persistence – A unifying concept of rheumatoid inflammation? Rheumatol. Int. 1985; 5: 221–7. [DOI] [PubMed] [Google Scholar]
- 86. Heeg K, Sparwasser T, Lipford GB, Hacker H, Zimmermann S, Wagner H. Bacterial DNA as an evolutionary conserved ligand signalling danger of infection to immune cells. Eur. J. Clin. Microbiol. Infect. Dis. 1998; 17: 464–9. [DOI] [PubMed] [Google Scholar]
- 87. Crew MD, Effros RB, Walford RL, Zeller E, Cheroutre H, Brahn E. Transgenic mice expressing a truncated Peromyscus leucopus TNF‐alpha gene manifest an arthritis resembling ankylosing spondylitis. J. Interferon Cytokine Res. 1998; 18: 219–25. [DOI] [PubMed] [Google Scholar]
- 88. Dahlen E, Dawe K, Ohlsson L, Hedlund G. Dendritic cells and macrophages are the first and major producers of TNF‐alpha in pancreatic islets in the nonobese diabetic mouse. J. Immunol. 1998; 160: 3585–93. [PubMed] [Google Scholar]
- 89. Ludewig B, Odermatt B, Landmann S, Hengartner H, Zinkernagel RM. Dendritic cells induce autoimmune diabetes and maintain disease via de novo formation of local lymphoid tissue. J. Exp. Med. 1998; 188: 1493–501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Simons PJ, Delemarre FG, Jeucken PH, Drexhage HA. Pre‐autoimmune thyroid abnormalities in the biobreeding diabetes‐prone (BB‐DP) rat: A possible relation with the intrathyroid accumulation of dendritic cells and the initiation of the thyroid autoimmune response. J. Endocrinol. 1998; 157: 43–51. [DOI] [PubMed] [Google Scholar]
- 91. Fierlbeck G, Rassner G, Muller C. Psoriasis induced at the injection site of recombinant interferon gamma. Results of immunohistologic investigations. Arch. Dermatol. 1998; 126: 351–5. [PubMed] [Google Scholar]
- 92. Lee RT, Briggs WH, Cheng GC, Rossiter HB, Libby P, Kupper T. Mechanical deformation promotes secretion of IL‐1 alpha and IL‐1 receptor antagonist. J. Immunol. 1997; 159: 5084–8. [PubMed] [Google Scholar]
- 93. Jansen A, van Hagen M, Drexhage HA. Defective maturation and function of antigen‐presenting cells in type 1 diabetes. Lancet 1995; 345: 491–2. [DOI] [PubMed] [Google Scholar]
- 94. Takahashi K, Honeyman MC, Harrison LC. Impaired yield, phenotype, and function of monocyte‐derived dendritic cells in humans at risk for insulin‐dependent diabetes. J. Immunol. 1998; 161: 2629–35. [PubMed] [Google Scholar]
- 95. Stagg AJ, Breban M, Hammer RE, Knight SC, Taurog JD. Defective dendritic cell (DC) function in a HLA‐B27 transgenic rat model of spondyloarthropathy (SpA). Adv. Exp. Med. Biol. 1995; 378: 557–9. [DOI] [PubMed] [Google Scholar]
- 96. Coppola D, Fu L, Nicosia SV, Kounelis S, Jones M. Prognostic significance of p53, bcl‐2, vimentin, and S100 protein‐positive Langerhans cells in endometrial carcinoma. Hum. Pathol. 1998; 29: 455–62. [DOI] [PubMed] [Google Scholar]
- 97. Tsujitani S, Kakeji Y, Maehara Y, Sugimachi K, Kaibara N. Dendritic cells prevent lymph node metastasis in patients with gastric cancer. In Vivo 1993; 7: 233–7. [PubMed] [Google Scholar]
- 98. Zeid NA, Muller HK. S100 positive dendritic cells in human lung tumours associated with cell differentiation and enhanced survival. Pathology 1993; 25: 338–43. [DOI] [PubMed] [Google Scholar]
- 99. Gabrilovich DI, Corak J, Ciernik IF, Kavanaugh D, Carbone DP. Decreased antigen presentation by dendritic cells in patients with breast cancer. Clin. Cancer Res. 1997; 3: 483–90. [PubMed] [Google Scholar]
- 100. Chaux P, Moutet M, Faivre J, Martin F, Martin M. Inflammatory cells infiltrating human colorectal carcinoma express HLA‐II but not B7‐1 and B7‐2 costimulatory molecules of the T‐cell activation. Lab. Invest. 1996; 74: 975–83. [PubMed] [Google Scholar]
- 101. Wittke F, Hoffmann R, Buer J et al Interleukin 10 (IL‐10): An immunosuppressive factor and independent predictor in patients with metastatic renal cell carcinoma. Br. J. Cancer 1997; 79: 1182–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Strand S, Hofmann WJ, Hug H et al Lymphocyte apoptosis induced by CD95 (APO‐1/Fas) ligand‐expressing tumor cells – A mechanism of immune evasion? Nat. Med. 1999; 2: 1361–6. [DOI] [PubMed] [Google Scholar]
- 103. Gratas C, Tohma Y, Barnas C, Taniere P, Ohgaki H. Up‐ regulation of Fas (APO‐1/CD95) ligand and down‐regulation of Fas expression in human esophageal cancer. Cancer Res. 1998; 58: 2057–62. [PubMed] [Google Scholar]
- 104. Gabrilovitch DI, Chen HL, Girgis KR et al Production of vascular endothelial growth factor by human tumors inhibits the functional maturation of dendritic cells. Nat. Med. 1996; 2: 1096–103. [DOI] [PubMed] [Google Scholar]
- 105. Ishida T, Oyama T, Carbone DP, Gabrilovich DI. Defective function of Langerhans cells in tumour‐bearing animals is the result of defective maturation from hemopoietic progenitors. J. Immunol. 1998; 161: 4842–51. [PubMed] [Google Scholar]
- 106. Menetrier‐Caux C, Montmain G, Dieu MC et al Inhibition of the differentiation of dendritic cells from CD34 (+) progenitors by tumor cells: Role of interleukin‐6 and macrophage colony‐stimulating factor. Blood 1998; 92: 4778–91. [PubMed] [Google Scholar]
- 107. Richters CD, Van Gelderop E, du Pont JS, Hoekstra MJ, Kreis RW, Kamperdijk EW. Migration of dendritic cells to the draining lymph node after allogenic or congenic rat skin transplantation. Transplantation 1999; 67: 828–32. [DOI] [PubMed] [Google Scholar]
- 108. Oding KA, Halliday GM, Muller HK. Enhanced survival of skin grafts depleted of Langerhans’ cells by treatment with dimethylbenzanthracene. Immunology 1987; 62: 379–85. [PMC free article] [PubMed] [Google Scholar]
- 109. Fu F, Li Y, Lu L et al Costimulatory molecule deficient dendritic cell progenitors induce T cell hyporesponsiveness in vitro and prolong the survival of vascularised cardiac allografts. Transplant. Proc. 1997; 29: 1310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Lu L, Li W, Fu F et al Blockade of the CD40‐CD40 ligand pathway potentiates the capacity of donor‐derived dendritic cell progenitors to induce long‐term cardiac allograft survival. Transplantation 1997; 64: 1808–15. [DOI] [PubMed] [Google Scholar]
- 111. Antonysamy MA, Fanslow WC, Fu F et al Evidence for a role of IL‐17 in organ allograft rejection: IL‐17 promotes the functional differentiation of dendritic cell progenitors. J. Immunol. 1999; 162: 577–84. [PubMed] [Google Scholar]
- 112. Antonysamy MA, Fanslow WC, Fu F et al Evidence for a role of IL‐17 in alloimmunity: A novel IL‐17 antagonist promotes heart graft survival. Transplant. Proc. 1999; 31: 93. [DOI] [PubMed] [Google Scholar]
- 113. Gluckman JC, Canque B, Chapuis F, Rozenwajg M. In vitro generation of human dendritic cells and cell therapy. Cytokines Cell. Mol. Ther. 1997; 3: 187–96. [PubMed] [Google Scholar]
- 114. Williams L, Egner W, Hart DNJ. Isolation and function of human dendritic cells. Int. Rev. Cytol. 1994; 153: 41–103. [DOI] [PubMed] [Google Scholar]
- 115. Strunk D, Rappersberger K, Egger C et al Generation of human dendritic cell/Langerhans cells from circulating CD34+ hematopoietic progenitor cells. Blood 1996; 78: 1292–302. [PubMed] [Google Scholar]
- 116. Szabolcs P, Moore MAS, Young JW. Expansion of immunostimulatory dendritic cells among the myeloid progeny of human CD34+ bone marrow precursors cultured with c‐kit ligand, granulocyte‐macrophage colony stimulating factor, and TNF‐alpha. J. Immunol. 1995; 154: 5851–61. [PubMed] [Google Scholar]
- 117. Santiago‐Schwarz F, Belilos E, Diamond B, Carson SE. TNF in combination with GM‐CSF enhances the differentiation of neonatal cord blood stem cells into dendritic cells and macrophages. J. Leukoc. Biol. 1992; 52: 274–81. [PubMed] [Google Scholar]
- 118. Bender A, Sapp M, Schuler G, Steinman RM, Bhardwaj N. Improved methods for the generation of dendritic cells from nonproliferating progenitors in human blood. J. Immunol. Meth. 1996; 196: 121–35. [DOI] [PubMed] [Google Scholar]
- 119. Zhou LJ, Tedder TF. CD14+ Blood monocytes can differentiate into functionally mature CD83+ dendritic cells. Proc. Natl Acad. Sci. USA 1996; 93: 2588–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Pickl WF, Majdic O, Kohl P et al Molecular and functional characteristics of dendritic cells generated from highly purified CD14+ peripheral blood monocytes. J. Immunol. 1996; 157: 3850–9. [PubMed] [Google Scholar]
- 121. Chapuis F, Rosenzwajg M, Yagello M, Ekman M, Biberfeld P, Gluckman JC. Differentiation of human dendritic cells from monocytes in vitro. Eur. J. Immunol. 1997; 27: 431–41. [DOI] [PubMed] [Google Scholar]
- 122. Szabolics P, Avigan D, Gezelter S et al Dendritic cells and macrophages can mature independently from a human bone marrow‐derived, post colony‐forming unit intermediate. Blood 1996; 87: 4520–30. [PubMed] [Google Scholar]
- 123. Lardon F, Snoeck HW, Berneman ZN et al Generation of dendritic cells from bone marrow progenitors using GM‐CSF, TNF‐alpha and additional cytokines: Antagonistic effects of IL‐4 and IFN‐gamma and selective involvement of TNF‐alpha receptor‐1. Immunology 1997; 91: 553–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Simon JC, Dittmar HC, de Roche R, Wilting J, Christ B, Schopf E. Rapid purification of human Langerhans cells using paramagnetic microbeads. Exp. Dermatol. 1995; 4: 155–61. [DOI] [PubMed] [Google Scholar]
- 125. Xhou LJ, Tedder TF. Human blood dendritic cells selectively express CD83, a member of the immunoglobin superfamily. J. Immunol. 1995; 154: 3821–35. [PubMed] [Google Scholar]
- 126. Fearnley DB, Mclellan AD, Mannering SI, Hock BD, Hart DN. Isolation of human blood dendritic cells using the CMRF‐44 monoclonal antibody: Implications for studies on antigen‐ presenting cell function and immunotherapy. Blood 1997; 89: 3708–16. [PubMed] [Google Scholar]
- 127. Wang J, Saffold S, Cao X, Krauss J, Chen W. Eliciting T cell immunity against poorly immunogenic tumors by immunisation with dendritic cell‐tumor fusion vaccines. J. Immunol. 1998; 161: 5516–24. [PubMed] [Google Scholar]
- 128. DeMatos P, Abdel‐Wahab Z, Vervaert C, Hester D, Seigler H. Pulsing of dendritic cells with cell lysates from either B16 melanoma or MCA‐106 fibrosarcoma yields equally effective vaccines against B16 tumours in mice. J. Surg. Oncol. 1998; 68: 79–91. [DOI] [PubMed] [Google Scholar]
- 129. Philip R, Brunette E, Ashton J et al Transgene expression in dendritic cells to induce antigen‐specific cytotoxic T cells in healthy donors. Cancer Gene Ther. 1998; 5: 236–46. [PubMed] [Google Scholar]
- 130. Nair SK, Boczkowki D, Morse M, Cumming RI, Lyerly HK, Gilboa E. Induction of primary carcinoembryonic antigen (CEA)‐specific cytotoxic T lymphocytes in vitro using human dendritic cells transfected with RNA. Nat. Biotechnol. 1998; 16: 364–9. [DOI] [PubMed] [Google Scholar]
- 131. Gabrilovich DI, Nadaf S, Corak J, Berzofsky JA, Carbone DP. Dendritic cells in anti tumor immune responses. II. Dendritic cells grown from bone marrow precursors, but not mature DC from tumor‐bearing mice, are effective antigen carriers in the therapy of established tumors. Cell. Immunol. 1996; 170: 111–19. [DOI] [PubMed] [Google Scholar]
- 132. DeMatos P, Abdel‐Wahab Z, Vervaert C, Seigler HF. Vaccination with dendritic cells inhibits the growth of hepatic metastases in B6 mice. Cell. Immunol. 1998; 185: 65–74. [DOI] [PubMed] [Google Scholar]
- 133. Butterfield LH, Jilani SM, Chakraborty NG et al Generation of melanoma‐specific cytotoxic T lymphocytes by dendritic cells transduced with a MART‐1 adenovirus. J. Immunol. 1998; 161: 5607–13. [PubMed] [Google Scholar]
- 134. Bronte V, Carroll MW, Goletz TJ et al Antigen expression by dendritic cells correlates with the therapeutic effectiveness of a model recombinant poxvirus tumor vaccine. Proc. Natl Acad. Sci. USA 1997; 94: 3183–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135. Nestle FO, Alijagic S, Gilliet M et al Vaccination of melanoma patients with peptide or tumor lysate‐pulsed dendritic cells. Nat. Med. 1998; 4: 328–32. [DOI] [PubMed] [Google Scholar]
- 136. Tjoa BA, Simmons SJ, Bowes VA et al Evaluation of phase I/II clinical trials in prostate cancer with dendritic cells and PSMA peptides. Prostate 1998; 36: 39–44. [DOI] [PubMed] [Google Scholar]
- 137. Hsu FJ, Benike C, Fagnoni F et al Vaccination of patients with B‐cell lymphoma using autologous antigen‐pulsed dendritic cells. Nat. Med. 1996; 2: 52–8. [DOI] [PubMed] [Google Scholar]
- 138. Maraskovsky E, Brasel K, Teepe M et al Dramatic increase in the numbers of functionally mature dendritic cells in Flt3 ligand‐treated mice: multiple dendritic cell subpopulations identified. J. Exp. Med. 1996; 184: 1953–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139. Shurin MR, Pandharipande PP, Zorina TD et al FLT3 ligand induces the generation of functionally active dendritic cells in mice. Cell. Immunol. 1997; 179: 174–84. [DOI] [PubMed] [Google Scholar]
- 140. Lynch DH, Andreasen A, Marakovsky E, Whitmore J, Miller RE, Schuh JC. Flt3 ligand induces tumour regression and antitumor immune responses in vivo. Nat. Med. 1997; 3: 625–31. [DOI] [PubMed] [Google Scholar]
- 141. Fernandez NC, Lozier A, Flamant C et al Dendritic cells directly trigger NK cell function: Cross‐talk relevant in innate anti‐tumour immune responses in vivo. Nat. Med. 1999; 5: 405–11. [DOI] [PubMed] [Google Scholar]
- 142. Shimizu Y, Guidotti LG, Fowler P, Chisari FV. Dendritic cell immunisation breaks cytotoxic T lymphocyte tolerance in hepatitis B virus transgenic mice. J. Immunol. 1998; 161: 4520–9. [PubMed] [Google Scholar]
- 143. Mackey MF, Gunn JR, Maliszewsky C, Kikutani H, Noelle RJ, Barth RJ. Dendritic cells require maturation via CD40 to generate protective antitumour immunity. J. Immunol. 1998; 161: 2094–8. [PubMed] [Google Scholar]
- 144. Labeur MS, Roters B, Pers B et al Generation of tumor immunity by bone marrow‐derived dendritic cells correlates with dendritic cell maturation stage. J. Immunol. 1999; 162: 168–75. [PubMed] [Google Scholar]
- 145. Lee WC, Zhong C, Qian S et al Phenotype, function, and in vivo migration and survival of allogenic dendritic cell progenitors genetically engineered to express TGF‐β. Transplantation 1998; 66: 1810–17. [DOI] [PubMed] [Google Scholar]
- 146. Markees TG, Phillips NE, Gordon EJ et al Prolonged skin allograft survival in mice treated with Flt3‐ligand‐induced dendritic cells and anti‐CD154 monoclonal antibody. Transplant. Proc. 1999; 31: 884–5. [DOI] [PubMed] [Google Scholar]
- 147. Matsue H, Matsue K, Walter M, Okumura K, Yagita H, Takashima A. Induction of antigen‐specific immunosuppression by CD95L cDNA‐transfected ‘killer’ dendritic cells. Nat. Med. 1999; 5: 930–7. [DOI] [PubMed] [Google Scholar]


