Abstract
Mycoplasma pneumonia of swine (MPS) lung lesions and immunogenic properties were compared between a Landrace line that was genetically selected for reduced incidence of pulmonary MPS lesions, and a non‐selected Landrace line. The MPS‐selected Landrace line showed significantly lower degrees of pulmonary MPS lesions compared with the non‐selected Landrace line. When changes in immunity before and after vaccination were compared, the percentage of B cells in the peripheral blood of the MPS‐selected Landrace line was significantly lower than that of the non‐selected line. Furthermore, the concentration of growth hormone and the mitogen activity of peripheral blood mononuclear cells in the MPS‐selected Landrace line showed significantly (P < 0.05) lower increases after vaccination than the non‐selected line. Conversely, the concentration of peripheral blood interferon (IFN)‐γ and salivary immunoglobulin A (IgA) after Mycoplasma hyopneumoniae vaccination was significantly higher in the MPS‐selected Landrace line than in the non‐selected line. Gene expression of toll‐like receptor (TLR)2 and TLR4 was significantly higher in the MPS‐selected Landrace line in immune tissues, with the exception of the hilar lymph nodes. The present results suggest that peripheral blood IFN‐γ, salivary IgA TLR2, and TLR4 are important immunological factors influencing the development of MPS lesions.
Keywords: disease resistance, immunogenic properties, mycoplasma pneumonia lesions, pig
Introduction
Mycoplasma pneumonia of swine (MPS) is a global endemic chronic respiratory disease in the swine industry. This disease, which is induced mainly by Mycoplasma hyopneumoniae infection, causes sizeable economic losses due to increased veterinary costs and product loss (Thacker et al. 1999; Maes et al. 2008). M. hyopneumoniae is located in the lumina of the respiratory tract, as well as the mucosal surface of the trachea, bronchi and bronchioles (Blanchard et al. 1992); it can propagate on tracheal and bronchial surfaces after infection. In MPS, the bronchial and bronchiolar epithelia show a loss of cilia from the epithelial cells and sloughing of epithelial cells into the airway lumina (Redondo et al. 2009). The primary clinical sign is a sporadic, dry, nonproductive cough (Thacker et al. 1999). Destruction of the cilia by M. hyopneumoniae increases the morbidity to other pathogens such as Pasteurella multocida, porcine respiratory virus, reproductive syndrome virus, porcine circovirus type 2, and porcine respiratory coronavirus (Ciprian et al. 1988; Thacker et al. 1999; Kim et al. 2003; Maes et al. 2008).
Several strategies for the control of infectious diseases in swine are commonly employed, such as improved housing conditions, use of antimicrobial agents and vaccination. However, these measures cannot completely eradicate the involved pathogens or inhibit their transmission. Further, the residue of the antibiotic in livestock products and antibacterial substances in domesticated animals is heavily regulated in Japan (The Ministry of Agriculture, Forestry and Fisheries, Japan 2014). Therefore, safer and more efficient ways to control swine chronic respiratory diseases are necessary.
From the point of view of genetic animal breeding, selection for disease resistance is very useful to ultimately minimize the need for medication. For example, Kadowaki et al. (2012) have improved the Landrace breed population by selecting for resistance to MPS and for greater meat production at the Miyagi Prefecture Animal Industry Experiment Station. Previously, we have reported the immune responsiveness of this MPS‐selected Landrace line to sheep red blood cells (SRBCs) (Katayama et al. 2011) and M. hyopneumoniae vaccine sensitization (Shimazu et al. 2013, 2014). However, the relationship between MPS lung lesions and immune responsiveness has not been reported. Therefore, the present study compared the MPS disease resistance of MPS‐selected Landrace pigs and non‐selected Landrace pigs by assessing their lung lesions and immunogenic characteristics.
Materials and Methods
Experimental animals
All experimental animals were maintained in a conventional pig barn at Miyagi University School of Food, Agricultural and Environmental Sciences. To confirm repeatability, two experiments were conducted. In experiment 1, we used 12 castrated Landrace pigs (average age, 16 weeks), which were selected for lower levels of MPS lesions for five generations at the Miyagi Prefecture Animal Industry Experiment Station (Kadowaki et al. 2012), and 12 non‐selected castrated Landrace pigs from the Research and Development Center, Nippon Meat Packers, Inc., Hokkaido, Japan. In experiment 2, we used six MPS‐selected (as describe above) castrated Landrace pigs (average 17 weeks of age), and six non‐selected castrated Landrace pigs from the Fukushima Livestock Experiment Station.
All experimental animal procedures were approved by Tohoku University and Miyagi University regulations concerning the protection and care of experimental animals.
Experimental design
In experiment 2 as well as experiment 1, animals of each line were bred in the same group and allowed to acclimatize for about 1 month before the experiment began. Pigs were raised in a crude, vinyl‐roofed, open‐air barn with a bedding of rice husks. The bedding was neither changed or cleaned, nor was the excrement removed during the experiment, in order to provide stressful conditions with a bad environment. During the experimental period, M. hypneumoniae vaccine (HYORESP®; Merial Japan Ltd., Tokyo, Japan), which consisted of an inactivated, adjuvant whole‐cell preparation, was injected intramuscularly twice, once on day −7 and again, on day 0, after blood collection. Thus, day 0 was defined as the day when the second dose of vaccine was administered. Immune functional analyses of the samples were conducted on days −7, 0, 2, 7 and 14. Blood samples were collected via jugular venipuncture into vacuum tubes containing heparin and ethylenediaminetetraacetic acid (EDTA: Terumo, Tokyo, Japan). At each sampling time, salivary samples were collected from the oral cavity with a cotton swab. In order to confirm the presence of pathogenic M. hyopneumoniae, we sampled the nasal mucus of each animal at each blood sampling time. On day 14, all pigs were killed by stunning at the anatomy facilities of Tohoku University, and immune tissues were collected. At this time, pulmonary MPS lesions were confirmed and measured by the veterinarian. In experiment 1, half of the pigs (six MPS‐selected and six non‐selected) were killed on day 14, and the remaining pigs remained until they reached a body weight of approximately 110 kg (average age, 23 weeks), when they were transported to the slaughterhouse (day 30 in this experimental period) to be slaughtered at the Miyagi Prefecture meat market center. After slaughter, pulmonary MPS lesions were examined. Blood samples were not collected from these animals on day 14.
Examination of pulmonary MPS lesions
After slaughter, the pulmonary MPS lesions were confirmed and scored according to Goodwin's lung score (Goodwin 1972) by a trained veterinarian.
Analysis of immune cells
The total number of white blood cells (WBCs) in 1 μL of peripheral blood was measured using an autohemolytic counter (Celltec; Nihon Koden, Tokyo, Japan). The percentage and number of each type of immune cell were measured by flow cytometry assay. Heparin‐treated swine blood was mixed with 2% dextran‐phosphate‐buffered saline (PBS) to allow for erythrocyte sedimentation and blood leukocyte enrichment into the supernatant (Genovesi et al. 1989; Yang & Parkhouse 1996). The upper phase was retained, and the cells were washed before a brief resuspension in hypotonic ammonium chloride solution to remove erythrocytes. Cells were stained with fluorescent antibodies in PBS containing 2% (v/v) fetal bovine serum (FBS) and 0.01% (w/v) sodium azide, and subjected to flow cytometry analysis. Cells were labeled with the following primary antibodies: biotin conjugated‐CD3ε (PPT3), phycoerythrin (PE)‐CD8α (73‐21‐11), fluorescein isothiocyanate (FITC)‐CD4α (74‐12‐4), FITC‐SWC3a (CD172a: 74‐22‐15), PE‐CD21 (BB6‐11C9.6), and unconjugated CD25 (K23.3B2, mouse immunoglobulin G1 (IgG1); AbD Serotec, Oxford, UK). The binding of unlabeled CD25 monoclonal antibody and biotin‐conjugated CD3ε was visualized using phycoerythrin (PE)‐goat F(ab′)2 anti‐mouse IgG1 and streptavidin PE/Cy5 (Bio Legend, San Diego, CA, USA), respectively. All antibodies were obtained from Beckman Coulter (Fullerton, CA, USA), unless otherwise stated. The stained cells were analyzed using a FACSCalibur™ fluorescence‐activated cell sorter (BD Biosciences, Franklin Lakes, NJ, USA) equipped with CellQuest software. Data were analyzed using FlowJo software (Tree Star, Ashland, OR, USA).
M. hyopneumoniae‐specific IgG
Blood samples were collected from the cervical veins of the animals. The serum was separated by centrifugation (1600 × g for 10 min at 4°C), divided into aliquots, and stored at −20°C until analysis. A 96‐well M. hyopneumoniae‐specific plate was incubated with M. hyopneumoniae vaccine (HYORESP®; Merial Japan Ltd., Tokyo, Japan) for 1 h at room temperature. Nonspecific binding of the antigen or antibody to the plate was blocked by treatment with Block Ace (UK‐B40; DS Pharma Biomedical Co., Ltd., Osaka, Japan) for 2 h at room temperature. A 200‐fold dilution of swine serum was then exposed to the plate; pooled swine serum was used as a standard (incubated for 2 h at 30°C). After washing with Tris buffered saline (T6664; Sigma, Tokyo, Japan), alkaline phosphatase‐labeled Rabbit‐Poly Anti‐Pig IgG (H+L) Antigen (Rockland Immunochemicals Inc., Philadelphia, PA, USA) was added, and the samples were incubated for 24 h at 5°C to detect M. hyopneumoniae‐specific IgG. After washing to remove nonspecific binding, fluorescence intensity was measured at 405 nm at 5‐min intervals after the reaction with the enzyme substrate solution.
Salivary enzyme‐linked immunosorbent assay (IgA)
Salivary samples were collected from the oral cavity of animals by using cotton swabs. The saliva was separated by centrifugation (at 1600 × g for 10 min at 4°C), divided into sterilized microtubes (WATSON CO., LTD., Tokyo, Japan), and stored at −80°C until analysis. A 96‐well plate was incubated with affinity‐purified goat anti‐pig IgA coating antibody (A100‐102A; Bethyl Laboratories, Inc., Montgomery, TX, USA) for 1 h at room temperature. Nonspecific binding of the antigen or antibody to the plate was blocked by treatment with Postcoat Solution (Bethyl Laboratories, Inc.) for 0.5 h at room temperature. A 500‐fold dilution of swine saliva was then exposed to the plate. Pig reference serum (RS10‐107‐3) was used as a standard (incubated for 1 h at room temperature). After washing with Tris buffered saline, horseradish peroxidase (HRP)‐conjugated goat anti‐pig IgA detection antibody (A100‐102P) was added, and the samples were incubated for 1 h at room temperature to detect the IgA. After 15 min of incubation with the enzyme substrate solution, the reaction was stopped by a stop solution, and the fluorescence intensity was measured at 450 nm.
Mitogenicity assay
Peripheral blood samples from day −7 (nonstimulated) and day 0 (7 days after the first M. hyopneumoniae vaccination) were used for this assay. Peripheral blood mononuclear cells (PBMCs) were isolated using density gradient centrifugation from individual porcine blood samples and washed with PBS and Roswell Park Memorial Institute 1640 medium (Sigma, Tokyo, Japan), supplemented with 2% FBS, 100 mg/mL streptomycin, and 100 U/mL penicillin. Next, these cells were placed in a 96‐well microplate (Corning Costar Japan, Tokyo, Japan; 2 × 105 cells/well) and stimulated with M. hyopneumoniae (2 × 106 cells/well; M. hyopneumoniae J (ATCC 25934), a non‐pathogenic strain inactivated with formalin), lipopolysaccharide (LPS; 20 µg/well LPS from Escherichia coli O55:B5 prepared using phenol extraction followed by gel‐filtration chromatography; Sigma), or concanavalin A (ConA; 2 µg/well). The cells were then maintained in an atmosphere containing 5% CO2 at 37°C. After a 32‐h incubation, the cells were radiolabeled with 9.25 kBq/well methyl‐[3H]‐thymidine (GE Healthcare, Tokyo, Japan) and incubated for a further 16 h. The cells were then harvested with a glass fiber filter (PerkinElmer Japan, Kanagawa, Japan). The methyl‐[3H]‐thymidine incorporation was counted in a liquid scintillation counter (Beckman Instruments, Palo Alto, CA, USA). Subsequent results, presented as stimulation index (SI), were calculated using the following equation:
For a comparison of SI before and after vaccination samples (relative SI), the following equation was applied:
Cytokines in peripheral blood by ELISA and messenger RNA (mRNA) expression analysis by real‐time PCR
Blood samples were collected from the cervical veins of the animals. The serum was separated by centrifugation (1600 × g for 10 min at 4°C), divided into aliquots, and stored at −20°C until analysis. The concentrations of interleukin (IL)‐10, IL‐13, IL‐17, interferon (IFN)‐γ and tumor necrosis factor (TNF)‐α in serum samples were determined using commercially available ELISA kits according to the manufacturers’ instructions (IL‐10, IFN‐γ and TNF‐α, Invitrogen, Carlsbad, CA, USA and IL‐13 and IL‐17, Bethyl Laboratories). Procedure of real‐time quantitative PCR analysis to expression levels of mRNAs in blood was previously described by Shimazu et al. (2014).
Analysis of toll‐like receptor (TLR) and cytokine mRNA expression by real‐time PCR
In experiment 2, the expression levels of mRNAs from total blood leukocytes, and from immune tissues (Peyer's patch (Pp), the mesenteric lymph node (MLN), the hilar lymph node (HLN), the spleen, and pulmonary alveolar macrophages (PAMs)) were analyzed by real‐time quantitative PCR. EDTA‐treated peripheral blood samples (250 μL) were mixed with 750 μL of TRIzol LS Reagent (Invitrogen, Carlsbad, CA, USA) for total RNA extraction. PAMs were collected from the bronchoalveolar lavage fluid by centrifugation. Total RNA in PAMs and immune tissues was extracted by mixing with 1 ml of TRIzol Reagent and crushing with beads. The total RNA concentration extracted from each sample was measured using a NanoDrop 2000c spectrophotometer (Thermo Fisher Scientific, Wilmington, DE, USA), and 1 µg of total RNA was subjected to reverse transcription. The complementary DNAs (cDNAs) were synthesized using a PrimeScript RT reagent kit with genomic DNA (gDNA) Eraser (Takara, Tokyo, Japan), according to the manufacturer's recommendations. Real‐time quantitative PCR was performed using a Thermal Cycler Dice® Real Time System II (Takara) and SYBR® Premix Ex Taq II (Tli RNaseH Plus; Takara). The conditions for PCR amplification were as follows: 30 s at 95°C, followed by 40 cycles of 5 s each at 95°C, and finally, 30 s at 60°C. Expression of β‐actin was used to normalize for the variations in total cDNA levels from sample to sample. The primer sequences used were as previously described (Moue et al. 2008).
Growth hormone analysis
Plasma concentrations of growth hormone (GH) were measured by double‐antibody radioimmunoassay. Porcine GH (AFP‐10888C; National Hormone and Peptide Program, Harbor‐UCLA Medical Center, Torrance, CA, USA) was used as the radio‐iodinated antigen and standard. Porcine GH antiserum (AFP‐422801; National Hormone and Peptide Program) was used at a dilution of 1:6 × 105; the detection limit was 0.0361 ng/tube. The intra‐assay coefficient of variation for GH was < 10%.
Statistical analysis
Data was analyzed using the SAS MIXED procedure (SAS Cary, NC, USA). The fixed effects were the pig lines (selected Landrace and non‐selected Landrace) and the day of the experiment; the random effect was the animals. We compared the mean change in these two lines “after” vaccination. P‐values less than 0.05 were considered statistically significant.
Results
Some results of experiment 1 have been previously reported (Shimazu et al. 2013, 2014). Therefore, to confirm the reproducibility of the immunity traits that had been reported by experiment 1, the results obtained in experiment 2 are mainly described.
There was no significant difference in the average daily gain of MPS‐selected Landrace and non‐selected Landrace lines in experiments 1 and 2 (data not shown).
Lung lesion scores
First, we confirmed M. hyopneumoniae DNA in nasal swabs from all pigs used in the two experiments (data not shown), confirming that this bacterium is indigenous in pigs. In experiment 1 and experiment 2, we found that MPS lung lesion scores were significantly lower in the MPS‐selected Landrace line than in the non‐selected Landrace line (Fig. 1). Two non‐selected Landrace lines were introduced from a different farm, with a different genetic background and breeding environment. The MPS‐selected line also showed significantly lower lung lesion scores than either of these two non‐selected lines.
Figure 1.
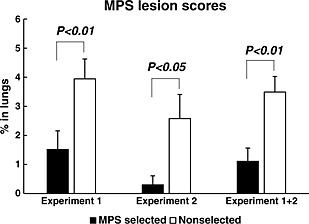
The pulmonary mycoplasma pneumonia of swine (MPS) lesions of lungs from MPS‐selected (black bars) and non‐selected Landrace pigs (white bars) were confirmed by slaughter, and the scores were measured following Goodwin's lung score (1972). The results of experiment 1, experiment 2, and integrate of 1 and 2 are shown from left to right, respectively. Statistical differences are indicated as the comparison between the two lines on the same day. Error bars indicate the standard error.
Analysis of immune cells in peripheral blood
In experiment 2, there was no significant difference in the number of WBCs in 1 μL of the peripheral blood between MPS‐selected Landrace and non‐selected Landrace lines (data not shown). However, the percentage of B cells decreased as in the previous result (experiment 1, Shimazu et al. 2013) after M. hyopneuoniae vaccination, and was significantly higher in the non‐selected Landrace line than in the MPS‐selected Landrace line on days 2 and 7 after vaccination (Fig. 2A). We did not observe any significant difference in the numbers of any other immune cells between the MPS‐selected Landrace and non‐selected Landrace lines.
Figure 2.
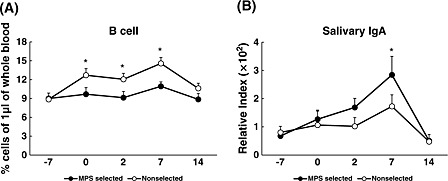
The average percent of B cells in 1 μL of peripheral blood, and the concentration of salivary immunoglobulin A (IgA) obtained from mycoplasmal pneumonia of swine (MPS)‐selected ● (closed circles) and non‐selected ○ (open circles) Landrace pigs. Blood samples were collected on 5 days (−7, 0, 2, 7 and 14). MPS vaccination was conducted on days −7 and 0 after blood sampling on those days. Statistical differences are indicated as the comparison between the two lines on the same day. Error bars indicate the standard error. (Asterisk mark indicates a significant difference at P < 0.05).
M. hyopneumoniae‐specific IgG production and salivary IgA production
Although there was no significant difference in M. hyopneumoniae vaccine‐specific IgG production in peripheral blood from the MPS‐selected line and non‐selected line in experiment 1 (Shimazu et al. 2014), in experiment 2, IgG tended to be more suppressed in the MPS‐selected line than in the non‐selected line (data not shown). In addition, in experiment 2, the IgA concentration in salivary glands 7 days after the second vaccination increased in both lines, and the increase was significantly greater in the MPS‐selected line than in the non‐selected line (Fig. 2B).
Mitogenicity assay
Cell proliferation activity between the MPS‐selected and non‐selected Landrace lines before and after vaccination was compared. In preset studies, we stimulated PBMCs harvested from these two lines with M. hyopneumoniae, LPS, and ConA to examine their proliferative abilities. In the present study (experiment 2), the non‐selected line showed significantly higher SI values stimulation with M. hyopneumoniae before and after the MPS vaccination, and with LPS before the MPS vaccination (Fig. 3A,B). However, no statistical difference for relative mitogen activity, which is calculated by dividing the SI value after vaccination by the SI value before vaccination, was observed (Fig. 3C).
Figure 3.
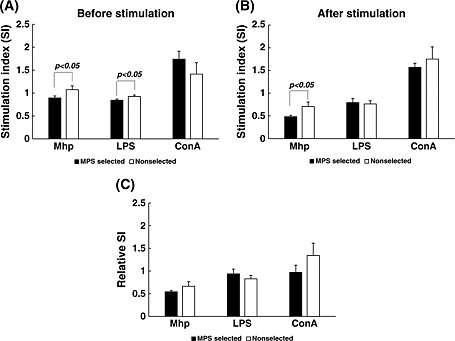
Mitogen activity of peripheral blood mononuclear cells (PBMCs) from mycoplasma pneumonia of swine (MPS)‐selected (black bars) and non‐selected Landrace pigs (white bars). Mitogen activity was analyzed in PBMCs obtained from the two lines before and after sensitization with MPS vaccine. These cells were stimulated with Mycoplasma hyopneumoniae (2 × 106 cells/well), or lipopolysaccharide (LPS; 20 µg/well), or concanavalin A (ConA; 2 µg/well). The activities before (A) and after vaccination (B) are shown as values of stimulation index (SI). Relative SI, which is calculated by dividing the SI value after vaccination by the value before vaccination, is also shown (C).
The mRNA expression and concentration of cytokines in peripheral blood
In this study, the IFN‐γ mRNA expression was significantly increased in the MPS‐selected line at days 0 and 7 after vaccination compared with the non‐selected line (Fig. 4). No significant difference in the mRNA expressions of any other cytokines between the MPS‐selected Landrace and non‐selected Landrace lines were observed (data not shown). The concentration of IFN‐γ measured was not possible to show; because measurements by the ELISA kit indicated the value below the detection limit.
Figure 4.
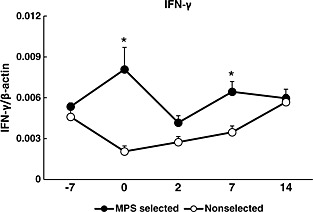
Mean and SE of the gene expression of interferon (IFN)‐γ in peripheral blood from mycoplasma pneumonia of swine (MPS)‐selected ● (closed circles) and non‐selected Landrace ○ (open circles) pigs. Blood samples were collected on 5 days (−7, 0, 2, 7 and 14). MPS vaccination was conducted on days −7 and 0 after blood sampling on those days. Statistical differences are indicated as the comparison between the two lines on the same day. The expression of IFN‐γ is shown as value of relative index (RI), which is calculated by dividing the expression levels of IFN‐γ by the expression levels of β‐actin. (Asterisk mark indicates a significant difference at P < 0.05).
TLR and cytokine mRNA expression in immune tissues
In experiment 2, mRNA expression of TLR2 and TLR4 in Pps, MLNs and spleens, and TLR4 in PAMs, were significantly higher in the MPS‐selected Landrace line than in the non‐selected line. However, there were no significant differences in the mRNA expression levels of TLR2 and TLR4 in HLNs, or that of TLR2 in PAMs, between MPS‐selected and non‐selected lines. On the other hand, the mRNA expressions of IL‐6 in Pps and MLNs, and IL‐10 in spleens, Pps and MLNs were significantly higher in the MPS‐selected line than in the non‐selected line. There were no significant differences in expression of IL‐6 in the spleens, HLNs and PAMs, or IL10 in the HLNs and PAMs, between the MPS‐selected line and the non‐selected line (Fig. 5).
Figure 5.
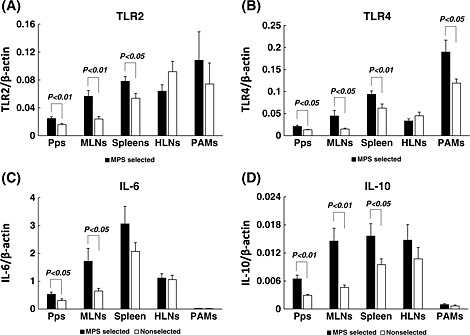
Mean and SE of the gene expression of toll‐like receptors (TLRs) and cytokines in immune tissues from mycoplasma pneumonia of swine (MPS)‐selected (black bars) and non‐selected Landrace (white bars) pigs. Pps, Peyer's patch, MLNs, mesenteric lymph nodes; HLNs, hilar lymph nodes; spleens; and PAMs, pulmonary alveolar macrophages. The expression of TLR2 (A), TLR4 (B), interleukin (IL)‐6 (C) and IL‐10 (D) are shown as values of relative index (RI), which is calculated by dividing the expression levels of TLRs and cytokines by the expression levels of β‐actin.
Growth hormone analysis
In experiment 1, the decrease in the concentration of GH was significantly greater in the MPS‐selected Landrace line than in the non‐selected Landrace line after the first vaccination (day 0) (Shimazu et al. 2014). Similarly, in experiment 2, GH concentration significantly decreased in the MPS‐selected Landrace line; however, it increased in the non‐selected Landrace line on days 7 and 14 after the second vaccination (Fig. 6).
Figure 6.
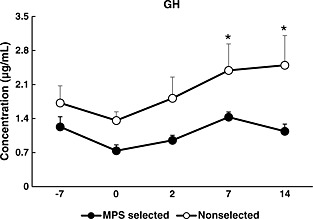
Changes in the concentrations of growth hormone (GH) in plasma samples obtained from mycoplasma pneumonia of swine (MPS)‐selected ● (closed circles) and non‐selected ○ (open circles) Landrace pigs. Blood samples were collected on 5 days (−7, 0, 2, 7 and 14). MPS vaccination was conducted on days −7 and 0 after blood sampling on those days. Statistical differences are indicated as the comparison between the two lines on the same day. Error bars indicate the standard error. (Asterisk mark indicates a significant difference at P < 0.05).
Discussion
Previous reports have clarified the immune responses of the MPS‐selected Landrace line and the non‐selected line to SRBCs (Katayama et al. 2011) and M. hyopneumoniae vaccination (Shimazu et al. 2013, 2014). In the present study, to confirm the repeatability of the observed effects on immune response, we compared both MPS resistance and immune response to M. hyopneumoniae vaccination in the MPS‐selected Landrace line with non‐selected Landrace lines. These non‐selected Landrace lines used in two experiments were genetically different lines.
First, we observed a significant difference in MPS lung lesion scores between the MPS‐selected Landrace line and two non‐selected Landrace lines. As expected, lung lesion scores were significantly lower in the MPS‐selected Landrace line than in the two non‐selected Landrace lines. This result confirms the resistance to MPS of this selected line.
In experiments 1 and 2, various immune traits were measured. Therefore, we calculated the correlations between the MPS lung lesion and the each immune trait to clarify which immune traits were related to the MPS lung lesion. However, we could not get any significant correlations. Nevertheless, many significant differences in immune traits between the MPS‐selected line and non‐selected line were observed in both experiments.
From the results of experiment 1 (Shimazu et al. 2013, 2014) and 2, and previous results (Katayama et al. 2011), the possibility that the humoral immunity which is characterized by B cells and their activities were suppressed in the MPS‐selected line against vaccination was suggested. Compared with the non‐selected Landrace line, the percentage of B cells in peripheral blood (experiments 1 and 2) and their IgG secretions (experiment 2) and the proliferative abilities of PBMCs (experiment 1) were significantly decreased in the MPS‐selected Landrace line. Furthermore, the same result was confirmed in our previous study that the SRBC‐specific IgG production was decreased in the MPS‐selected line (Katayama et al. 2011). In addition, previous studies have indicated that increased IgG production in serum is not directly involved in the clinical resistance to mycoplasma pneumonia (Thacker et al. 1998; Chen et al. 2006). These results also indicate that increased antibody‐specific IgG production most likely contributes to MPS lung lesion severity during natural infection.
In association with the suppression of IgG in the MPS‐selected Landrace line, the secretion of GH in peripheral blood was significantly suppressed in experiment 2 and experiment 1 (Shimazu et al. 2014). B cells are the major immune cells that express both GH and GH receptors (Hattori et al. 2001; Hattori 2009). GH promotes lymphocyte survival and proliferation (Jeay et al. 2002; Borghetti et al. 2006). Therefore, the decrease in B cell percentage, GH secretion and proliferative ability of PBMCs in the MPS‐selected line suggests that these traits correlate with each other. Donahue et al. (2006) had reported that the secretion of GH was increased in male rats and female rats during diestrus exposed to a stressor. From this, the MPS‐selected Landrace line may be less susceptible to stress than the non‐selected Landrace line. However, whether the increase and decrease of GH is involved in MPS resistance is unclear.
Contrary to the decrease in the B cells and their IgG secretion, the salivary IgA secretion was increased in the MPS‐selected Landrace line. Peripheral blood IgG and saliva IgA are produced by different tissues or organs. The IgG and monomeric type of IgA are known to be produced in lymph node, spleen and bone marrow, and then secreted in blood. The dimeric type of IgA secreted in saliva is mainly produced in submucous‐lymph tissues, and then secreted in tears, saliva, sweat, colostrum (Parham 2009). The mucosal immune system is a first defense line of inner body surfaces and IgA is essential in this surface defense system. Secretion of IgA is critical for protection against infectious pathogens (Phalipon et al. 2002; Brandtzaeg 2003; Tlaskalová‐Hogenová et al. 2004). Increased IgA secretion may also play a protective role against M. hyopneumoniae infections in the upper respiratory tract (Woolard et al. 2004). The activation of IgA‐dependent mucosal immunity may be one of the reasons the lung lesions were reduced in the MPS‐selected line.
On the other hand, natural immunity and cell‐mediated immunity, which are characterized by granular leukocyte percentage and INF‐γ protein and mRNA expressions, showed significantly higher in the MPS‐selected Landrace line than that in two non‐selected Landrace lines. Increased IFN‐γ expression eliminates bacteria and parasites by activating macrophages and killer T cells (Romagnani 1992; Thacker et al. 2000; Jones et al. 2002; Woolard et al. 2004), and also plays an important role in pulmonary defense mechanisms against mycoplasma respiratory disease (Woolard et al. 2004; Rodriguez et al. 2007). Furthermore, cell‐mediated immunity is important for controlling MPS (Thacker et al. 2000; Chen et al. 2006) and may be one of the reasons for the increased resistance to MPS in the MPS‐selected line.
In the present study (experiment 2), the mRNA expressions of TLR2, TLR4, IL‐6 and IL‐10 of the Pps, MLNs, spleens and PAMs were significantly higher in those immune tissues obtained from the MPS‐selected line than that of the non‐selected line. These differences in immune response suggest a genetically correlated response according to MPS selection. By recognizing microbe‐associated molecular patterns, the TLR family plays a critical role in host defense and in the development of inflammation. TLR2 recognizes ligands from Gram‐positive bacteria, whereas TLR4 responds to endotoxin from Gram‐negative bacteria. TLRs are also involved in the responses to lung injury (Jiang et al. 2005) and to M. hyopneumoniae infection (Chu et al. 2005; Wu et al. 2008; Love et al. 2010). IL‐6 is a primary cytokine responsible for acute inflammation (Thacker 2006) and M. hyopneumoniae clearance in lungs (Pietsch et al. 1994; Wu et al. 2008). IL‐10 is an endogenous regulator that is critical for chemokine expression in acute lung inflammation (Shanley et al. 2000). These results indicate that interactions between these cytokines and TLRs may contribute to the balance of the inflammatory and immune responses in the lungs of the MPS‐selected line.
In conclusion, the MPS‐selected Landrace line shows strong resistance against MPS due to the strength of its cell‐mediated and mucosal immune responses, as described above. Furthermore, our research indicates that immune system components such as IFN‐γ, TLR2, TLR4 and salivary IgA may be working synergistically against MPS. In present studies, cytokine and TLR levels in mucosal tissues, PAMs and salivary IgA were measured only in experiment 2. Therefore, it is necessary to confirm the repeatability of these results. In addition, although the MPS‐selected Landrace line showed a strong resistance against MPS, further studies to confirm whether this MPS‐selected line has resistance to other respiratory chronic diseases are warranted.
Acknowledgments
This study was supported by a grant from the Program for Promotion of Basic and Applied Research for Innovations in Bio‐oriented Industry to Dr. K. Suzuki.
Borjigin, L. , Shimazu, T. , Katayama, Y. , Li, M. , Satoh, T. , Watanabe, K. , Kitazawa, H. , Roh, S.‐g. , Aso, H. , Katoh, K. , Uchida, T. , Suda, Y. , Sakuma, A. , Nakajo, M. , and Suzuki, K. (2016) Immunogenic properties of Landrace pigs selected for resistance to mycoplasma pneumonia of swine. Anim Sci J, 87: 321–329. doi: 10.1111/asj.12440.
References
- Blanchard B, Vena MM, Cavalier A, Lannic J, Gouranton J, Kobisch M. 1992. Electron microscopic observation of the respiratory tract of SPF piglets inoculated with Mycoplasma hyopneumoniae. Veterinary Microbiology 30, 329–341. [DOI] [PubMed] [Google Scholar]
- Borghetti P, De Angelis E, Saleri R, Cavalli V, Cacchioli A, Corradi A, et al 2006. Peripheral T lymphocyte changes in neonatal piglets: relationship with growth hormone (GH), prolactin (PRL) and cortisol changes. Veterinary Immunology and Immunopathology 110, 17–25. [DOI] [PubMed] [Google Scholar]
- Brandtzaeg P. 2003. Role of secretory antibodies in the defence against infections. International Journal of Medical Microbiology 293, 3–15. [DOI] [PubMed] [Google Scholar]
- Chen AY, Fry SR, Forbes FJ, Daggard G, Mukkur TK. 2006. Evaluation of the immunogenicity of the P97R1 adhesin of Mycoplasma hyopneumoniae as a mucosal vaccine in mice. Journal of Medical Microbiology 55, 923–929. [DOI] [PubMed] [Google Scholar]
- Chu HW, Jeyaseelan S, Rino JG, Voelker DR, Wexler RB, Campbell K, et al 2005. TLR2 signaling is critical for Mycoplasma pneumoniae‐induced airway mucin expression. The Journal of Immunology 174, 5713–5719. [DOI] [PubMed] [Google Scholar]
- Ciprian A, Pijoan C, Cruz T, Camacho J, Tortora J, Colmenares G, et al 1988. Mycoplasma hyopneumoniae increases the susceptibility of pigs to experimental Pasteurella multocida pneumonia. Canadian Journal of Veterinary Research 52, 434. [PMC free article] [PubMed] [Google Scholar]
- Donahue CP, Kosik KS, Shors TJ. 2006. Growth hormone is produced within the hippocampus where it responds to age, sex, and stress. Proceedings of the National Academy of Sciences 103, 6031–6036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Genovesi EV, Knudsen RC, Gerstner DJ, Card DM, Martins CL, Quintero JC, et al 1989. In vitro induction of swine peripheral blood monocyte proliferation by the fibroblast‐derived murine hematopoietic growth factor CSF‐1. Veterinary Immunology and Immunopathology 23, 223–244. [DOI] [PubMed] [Google Scholar]
- Goodwin RFW. 1972. Isolation of Mycoplasma suipneumoniae from the nasal cavities and lungs of pigs affected with enzootic pneumonia or exposed to this infection. Research in Veterinary Science 13, 262–267. [PubMed] [Google Scholar]
- Hattori N. 2009. Expression, regulation and biological actions of growth hormone (GH) and ghrelin in the immune system. Growth Hormone & IGF Research 19, 187–197. [DOI] [PubMed] [Google Scholar]
- Hattori N, Saito T, Yagyu T, Jiang BH, Kitagawa K, Inagaki C. 2001. GH, GH receptor, GH secretagogue receptor, and ghrelin expression in human T cells, B cells, and neutrophils. The Journal of Clinical Endocrinology & Metabolism 86, 4284–4291. [DOI] [PubMed] [Google Scholar]
- Jeay S, Sonenshein GE, Postel‐Vinay MC, Kelly PA, Baixeras E. 2002. Growth hormone can act as a cytokine controlling survival and proliferation of immune cells: new insights into signaling pathways. Molecular and Cellular Endocrinology 188, 1–7. [DOI] [PubMed] [Google Scholar]
- Jiang D, Liang J, Fan J, Yu S, Chen S, Luo Y, et al 2005. Regulation of lung injury and repair by Toll‐like receptors and hyaluronan. Nature Medicine 11, 1173–1179. [DOI] [PubMed] [Google Scholar]
- Jones HP, Tabor L, Sun X, Woolard MD, Simecka JW. 2002. Depletion of CD8+ T cells exacerbates CD4+Th cell‐associated inflammatory lesions during murine mycoplasma respiratory disease. Journal of Immunology 168, 3493–3501. [DOI] [PubMed] [Google Scholar]
- Kadowaki H, Suzuki E, Kojima‐Shibata C, Suzuki K, Okamura T, Onodera W, et al 2012. Selection for resistance to swine mycoplasmal pneumonia over 5 generations in Landrace pigs. Livestock Science 147, 20–26. [Google Scholar]
- Katayama M, Fukuda T, Okamuara T, Suda Y, Suzuki E, Uenishi H, et al 2011. Immunophenotype characterization for swine selected line, which is resistant for the mycoplasma pneumonia. Asian‐Australasian Journal of Animal Sciences 24, 889–897. [Google Scholar]
- Kim J, Chung HK, Chae C. 2003. Association of porcine circovirus 2 with porcine respiratory disease complex. The Veterinary Journal 166, 251–256. [DOI] [PubMed] [Google Scholar]
- Love W, Dobbs N, Tabor L, Simecka JW. 2010. Toll‐like receptor 2 (TLR2) plays a major role in innate resistance in the lung against murine Mycoplasma. PLoS One 5, e10739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maes D, Segales J, Meyns T, Sibila M, Pieters M, Haesebrouck F. 2008. Control of Mycoplasma hyopneumoniae infections in pigs. Veterinary Microbiology 126, 297–309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moue M, Tohno M, Shimazu T, Kido T, Aso H, Saito T, et al 2008. Toll‐like receptor 4 and cytokine expression involved in functional immune response in an originally established porcine intestinal epitheliocyte cell line. Biochimica et Biophysica Acta (BBA)‐General Subjects 1780, 134–144. [DOI] [PubMed] [Google Scholar]
- Parham P. 2009. The Immune System, 3rd edn. Garland Science. Part of Taylor & Francis Group LLC, New York, NY. [Google Scholar]
- Phalipon A, Cardona A, Kraehenbuhl JP, Edelman L, Sansonetti PJ, Corthésy B. 2002. Secretory component: a new role in secretory IgA‐mediated immune exclusion in vivo. Immunity 17, 107–115. [DOI] [PubMed] [Google Scholar]
- Pietsch K, Ehlers S, Jacobs E. 1994. Cytokine gene expression in the lungs of BALB/c mice during primary and secondary intranasal infection with Mycoplasma pneumoniae. Microbiology 140, 2043–2048. [DOI] [PubMed] [Google Scholar]
- Redondo E, Masot AJ, Fernandez A, Gazquez A. 2009. Histopathological and immunohistochemical findings in the lungs of pigs infected experimentally with Mycoplasma hyopneumoniae. Journal of Comparative Pathology 140, 260–270. [DOI] [PubMed] [Google Scholar]
- Rodriguez F, Quesada O, Poveda JB, Fernandez A, Lorenzo H. 2007. Immunohistochemical detection of Interleukin‐12 and Interferon gamma in pigs experimentally infected with Mycoplasma hyopneumoniae. Journal of Comparative Pathology 136, 79–82. [DOI] [PubMed] [Google Scholar]
- Romagnani S. 1992. Induction of TH1 and TH2 responses: a key role for the ‘natural’ immune response? Immunology Today 13, 379–381. [DOI] [PubMed] [Google Scholar]
- Shanley TP, Vasi N, Denenberg A. 2000. Regulation of chemokine expression by IL‐10 in lung inflammation. Cytokine 12, 1054–1064. [DOI] [PubMed] [Google Scholar]
- Shimazu T, Borjigin L, Katayama Y, Li M, Satoh T, Watanabe K, et al 2013. Immunological characterization of peripheral blood leukocytes using vaccine for mycoplasmal pneumonia of swine (MPS) in swine line selected for resistance to MPS. Animal Science Journal 84, 683–692. [DOI] [PubMed] [Google Scholar]
- Shimazu T, Borjigin L, Katayama Y, Li M, Satoh T, Watanabe K, et al 2014. Genetic selection for resistance to mycoplasmal pneumonia of swine (MPS) in the Landrace line influences the expression of soluble factors in blood after MPS vaccine sensitization. Animal Science Journal 85, 365–373. [DOI] [PubMed] [Google Scholar]
- Thacker EL. 2006. Lung inflammatory responses. Veterinary Research 37, 469–486. [DOI] [PubMed] [Google Scholar]
- Thacker EL, Halbur PG, Ross RF, Thanawongnuwech R, Thacker BJ. 1999. Mycoplasma hyopneumoniae potentiation of porcine reproductive and respiratory syndrome virus‐induced pneumonia. Journal of Clinical Microbiology 37, 620–627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thacker EL, Thacker BJ, Boettcher TB, Jayappa H. 1998. Comparison of antibody production, lymphocyte stimulation and production induced by four commercial Mycoplasma hyopneumoniae bacterins. Swine Health and Production 6, 107–114. [Google Scholar]
- Thacker EL, Thacker BJ, Kuhn M, Hawkins PA, Waters WR. 2000. Evaluation of local and systemic immune responses induced by intramuscular injection of a Mycoplasma hyopneumoniae bacterin to pigs. American Journal of Veterinary Research 61, 1384–1389. [DOI] [PubMed] [Google Scholar]
- The Japanese Ministry of Agriculture, Forestry and Fisheries . 2014. About the antibacterial substance used for the domestic animal [homepage on the internet]. Japanese Ministry of agricultural Forestry and Fisheries, Tokyo, Japan; [cited 11 November 2014]. Available from URL: http://www.maff.go.jp/j/syouan/tikusui/yakuzi/koukinzai.html
- Tlaskalová‐Hogenová H, Štěpánková R, Hudcovic T, Tučková L, Cukrowska B, Lodinová‐Žádníková R, et al 2004. Commensal bacteria (normal microflora), mucosal immunity and chronic inflammatory and autoimmune diseases. Immunology Letters 93, 97–108. [DOI] [PubMed] [Google Scholar]
- Woolard MD, Hodge LM, Jones HP, Schoeb TR, Simecka JW. 2004. The upper and lower respiratory tracts differ in their requirement of IFN‐γ and IL‐4 in controlling respiratory mycoplasma infection and disease. The Journal of Immunology 172, 6875–6883. [DOI] [PubMed] [Google Scholar]
- Wu Q, Martin RJ, LaFasto S, Efaw BJ, Rino JG, Harbeck RJ, et al 2008. Toll‐like receptor 2 down‐regulation in established mouse allergic lungs contributes to decreased mycoplasma clearance. American Journal of Respiratory and Critical Care Medicine 177, 720–729. [DOI] [PubMed] [Google Scholar]
- Yang H, Parkhouse RM. 1996. Phenotypic classification of porcine lymphocyte subpopulations in blood and lymphoid tissues. Immunology 89, 76–83. [DOI] [PMC free article] [PubMed] [Google Scholar]


