Abstract
Tumor associated carbohydrate antigens (TACAs) are a class of attractive antigens for the development of anti-cancer immunotherapy. Besides monoclonal antibodies and vaccines, chimeric antigen receptor (CAR) T cells and bispecific antibodies (BsAbs) targeting TACA are exciting directions to harness the power of the immune system to fight cancer. In this review, we focus on two TACAs, i.e., the GD2 ganglioside and the mucin-1 (MUC1) protein. The latest advances in CAR T cells and bispecific antibodies targeting these two antigens are presented. The roles of co-stimulatory molecules, structures of the sequences for antigen binding, methods for CAR and antibody construction, as well as strategies to enhance solid tumor penetration and reduce T cell exhaustion and death are discussed. Furthermore, approaches to reduce “on target, off tumor” side effects are introduced. With further development, CAR T cells and BsAbs targeting GD2 and MUC1 can become powerful agents to effectively treat solid tumor.
Keywords: Bispecific antibody, cancer, CAR T cell, GD2 ganglioside, immunotherapy, mucin-1
1. Introduction
Cancer is a leading cause of death worldwide. The global cancer burden, which has been estimated as 18.1 million new cases and 9.6 million deaths in 2018, is predicted to rise to 29.5 million new incidences in 2040 [1,2]. The alarming increase in cancer burden accentuates the urgent need to develop innovative approaches for more effective and less toxic cancer treatments complementing conventional therapeutics such as chemotherapy, radiotherapy, surgery and palliative care. Cancer immunotherapy holds great promises in cancer treatment, which was named as the Breakthrough of the Year in 2013 by Science [3].
Tumor associated carbohydrate antigens (TACAs) are a class of attractive antigens for anti-cancer immunotherapy development [4,5]. Multiple TACAs are over-expressed on the surface of cancer cells compared to those on normal cells [6]. In addition, TACAs can be found at high levels on many types of cancer, rendering them intriguing targets for potential broad spectrum anti-cancer immunotherapy [7]. Tremendous efforts have been devoted to the development of anti-cancer vaccines targeting TACAs with innovative designs of the antigen structures, the carrier moieties to deliver the antigens to the immune system, as well as incorporation of immune activation elements including adjuvants and cytotoxic T cell epitopes into the vaccine constructs [8–11]. Besides vaccines, chimeric antigen receptor (CAR) T cells and bispecific antibodies (BsAbs) have emerged as appealing immunotherapeutic strategies to combat cancer, as evident from multiple products approved by the FDA [12]. Recently, great progresses have been achieved in the development of CAR T cells and BsAbs targeting two TACAs, i.e., gangliosides GD2 and glycoprotein mucin-1 (MUC1). In this review, we will discuss the advances and challenges in these areas to stimulate further development.
2. GD2 CAR T cells: going beyond the anti-GD2 monoclonal antibodies
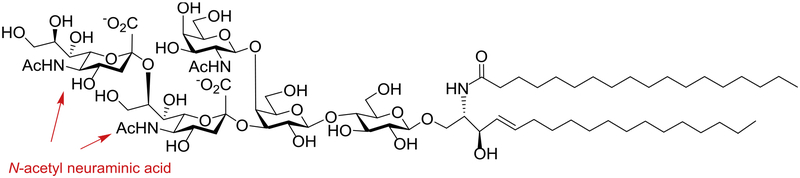
Disialoganglioside GD2 is an N-acetyl neuraminic acid containing glycolipid antigen composed of five monosaccharides anchored to the lipid bilayer of plasma membrane through a ceramide lipid (Figure 1). Normal tissues have low expression levels of GD2 [13], but in various types of cancers such as neuroblastoma, small-cell lung cancer, melanoma, glioma, and sarcomas, the expression of this weakly immunogenic antigen can reach 107 molecules per cell [8,14,15]. Furthermore, GD2 has been discovered as a potential biomarker for the purported breast cancer stem cells [16]. GD2 can induce tyrosine phosphorylation leading to activation of a variety of kinase pathways, resulting in increased proliferation, cell migration and invasion of cancer cells. Knockdown of GD2 levels abrogated tumor formation in vivo [16]. With its high expression on tumor cells and importance in tumor development, GD2 was ranked as one of the top antigenic targets for cancer [14,17].
Figure 1.
Structure of the GD2 ganglioside.
Monoclonal antibodies against GD2 have achieved clinical success in cancer treatment. Early generation of anti-GD2 cancer immunotherapy utilized murine monoclonal antibodies, 14G2a and 3F8, in patients with melanoma, neuroblastoma, and osteosarcoma [14]. However, due to the mouse origin of these antibodies, significant human anti-mouse antibodies were induced, limiting the dose that can be administered and reducing their anti-tumor efficacy. In addition, administration of these antibodies are associated with side effects such as pain, fever, hypertension and urticarial reactions, which limit their wide applications. To overcome these challenges, human-mouse chimeric anti-GD2 monoclonal antibodies have been generated [14,18]. ch14.18 consisting of the variable regions of murine 14G2a mAb fused with the constant regions of human IgG1, has higher antibody-dependent cell-mediated cytotoxicity and longer half-life compared to 14G2a. Further development of humanized anti GD2 antibody, hu3F8 and hu14.18K322A with a K322A mutation of the Fc region to prevent complement fixation, improved their half-lives and tolerable dose. However, in clinical trials, similar side effects as m3F8, including pain and hypertension, were observed.
Adoptive transfer of CAR T cells is a promising immunotherapy strategy to treat cancer in an MHC-independent manner. CAR T cells are designed by linking the single-chain variable fragment (scFv) of a monoclonal antibody with the T cell receptor (TCR) ζ-chain transmembrane and cytoplasmic regions. Further development of CAR Ts by the addition of costimulatory signal (CD28) for full activation of these cells has led to 2nd generation of CAR Ts and the third generation includes additional signaling domain (CD27, 4–1BB or OX40) aimed at improving proliferation, survival and cytokine release from the cells. With the high anti-tumor efficacy in acute lymphoblastic leukemia, two anti-CD19 CAR T cell based immunotherapies (KYMRIAH and CARTA) have been approved by FDA. However, applications of CAR T therapy in solid tumors have met with limited successes. Some of the main barriers of CAR T cell therapy include: limited ability of CAR T cells to proliferate, inefficient trafficking of CAR T cells to tumor tissues, limited T cell extravasation into solid tumor, and suppressive tumor microenvironment that dampens T cell proliferation and cytokine production[19]. With the importance and targetability of GD2 for solid tumors, anti GD2 CAR T cell immunotherapy has been investigated in various types of tumors.
2.1. Building co-stimulatory signals into GD2 CAR T cells
One of the first CAR T cells products tested in children was an anti-GD2 CAR containing only the CD3ζ endodomain but no costimulatory domain [20]. In preclinical models, Rossig et al. demonstrated that GD2 was a viable CAR T cell target for neuroblastoma. However, the culture of these first generation CAR T cells could not be maintained for longer than 8 weeks upon stimulation with GD2+ tumor cells. In addition, these CAR T cells failed to proliferate when incubated with GD2+ tumor cells presumably due to a lack of co-stimulatory signals upon antigen binding. Further investigations of GD2 CAR T focused on CAR constructs incorporating endodomain CD28 and OX40 together with the anti-GD2 scFv [21].
The importance of costimulatory signals has been studied in a clinical trial in eleven individuals with neuroblastoma, who had Epstein-Barr virus (EBV)-associated malignancies [22]. EBV-specific cytotoxic T lymphocytes (CTLs) or bulk T cells activated with anti-CD3 antibodies (ATCs) were transduced with a CAR specific for GD2 antigen. Equal numbers of GD2 CAR-CTLs and CAR-ATCs were injected into patients. Persistence of the EBV specific GD2 CAR-CTLs was detectable beyond 6 weeks, which were almost two times longer compared to the GD2 CARATCs. These results highlight the importance of costimulation of native antigen receptor on CARCTLs, when these cells engaged EBV antigens on professional antigen-presenting cells through their native receptors. Four of the eight patients (50%) with evaluable tumors had shown tumor necrosis or regressions. A subsequent study with 19 patients including the original 11 patients for up to 4 years showed that although both CAR-CTLs and CAR-ATCs were low or undetectable beyond 6 weeks, the continued presence of even low levels of CAR–T cells was associated with a significantly longer time to disease progression. Three patients achieved complete remission out of the 11 patients with active disease at the time of infusion. The proportion of CD4+ helper cells and central memory cells present in infused CAR T cells correlated with the long term persistence of CAR T cells [23].
To sustain the survival of GD2 CAR T cells, additional stimulatory molecule such as interleukin-15 (IL15) has been incorporated generating the GD2 CAR.15 T cells [24]. IL15 provided the survival signals to GD2 CAR.15 T cells while they were in circulation, and costimulation when the cells reached the tumor site. The GD2 CAR.15 T cells expressed lower amounts of programmed cell death protein-1 (PD-1), an immune checkpoint protein, and could proliferate and persist longer in vitro and in vivo even in the absence of exogenous cytokines or antigen support. GD2 CAR.15 T cells were more effective in controlling tumor growth with a lower number of total infused T cells, and in improving survival after rechallenge experiments. Further clinical trials ( NCT03294954) for GD2 CAR.15 T cells are ongoing.
Besides IL15, another CAR T design incorporated the CD27 costimulatory domains [25], which can augment survival and tumor killing activities of T cells [26]. These CAR T cells showed long term persistence with 30% of infused CAR T cells lasting up to 180 days. Two patients had markedly tumor regression and the 1-year overall survival rate reached 74%.
2.2. Enhancing homing and penetration of solid tumor by GD2 CAR T cells
An important factor in dictating CAR T efficacy is the ability of these cells to reach solid tumor sites. To aid in trafficking and homing of CAR T cells, chemokine receptor can be incorporated [27]. While earlier finding confirmed the presence of C-C chemokine receptor type 2 (CCR2) on human activated T cells and T cell migration in response to C-C motif ligand 2 (CCL2) chemokine, CCR2 expression was deficient in CAR T cells due to culture conditions and the usage of anti-CD28 antibody, which reduced the levels of CCR2 following TCR activation. In this regard, modified GD2 CAR T cells expressing CCR2b was able to improve the homing and expansion of T cells at tumor site in vivo by day 14. This increased frequency of T cells at the tumor site correlated with reduced tumor growth. Hence CCL2 chemokine concentration at tumor site can be exploited to attract effector cells to tumor environment.
In addition to incorporation of chemokine receptor, modification of the tumor microenvironment and vascularity with anti-angiogenic agents such as anti-vascular endothelial growth factor antibody (Bevacizumab, BEV) can improve the trafficking of CAR T cells to tumor [28]. In a mouse neuroblastoma (NB) model, administration of anti-GD2 CAR T cells alone failed to lead to any increases in survival compared to the control group receiving non-transduced T cells. To address this, the combination of BEV and anti-GD2 CAR T was tested. While injection of BEV at a relatively low dose (2 mg/kg) did not significantly affect the number of micro-blood vessels in tumor tissues, addition of BEV to anti-GD2 CAR T cell treatment regime led to enhanced migration and infiltration to inner tumor core by these T cells, which was confirmed by double CD31/CD3 immunostaining. The T cells produced interferon-γ and reprogrammed the tumor microenvironment, with the mice receiving the combination therapy showing modest improvements in survival. Interestingly, NB cells were found to upregulate the levels of PD-L1. As GD2-CAR T cells in tumors were found to express programmed cell death protein-1 (PD-1), this study suggested the interactions of PD-1 with its ligand PD-L1 may limit the anti-tumor efficacy.
Once reaching tumor tissues, the ability of CAR-T cells to penetrate stroma-rich solid tumors is critical for antitumor effects. To facilitate tissue penetration, extracellular matrix (ECM), including the heparan sulfate proteoglycans (HSPGs) need to be degraded. The effect of in vitro culture of T lymphocytes on HSPG degrading heparanase enzyme (HPSE) has been investigated [29]. In a Matrigel - based cell invasion assay, briefly activated T cells showed 1.5 times higher invasion of ECM compared to freshly isolated resting T cells. In contrast, long-term ex vivo–expanded T cells had 3 times lower invasion than the freshly isolated T cells. Mechanistic studies showed that the freshly isolated and briefly activated T cells retained the expression of active HPSE, while in long-term expanded T cells, the enzyme was not detected by either Western blotting or immunofluorescence. Building on this knowledge, CAR T cells were engineered to express HPSE, which exhibited improved capability of ECM degradation and deeper tumor penetration. While both HPSE modified and un-modified GD2-CAR T cells had similar efficiencies in lysing GD2+ human cell lines in vitro, HPSE expressing GD2-CAR T cells provided significantly improved protection to mice from GD2+ tumor cell induced death.
2.3. Combination of GD2 CAR T therapy with checkpoint blockade and chemotherapy
Improvement of GD2 CAR T cell therapy was investigated in combination with anti-PD-1 blockade to reduce T cell exhaustion and improve survival. In vitro study showed that repeated antigen stimulation reduced the percentage of viable GD2 CAR T cells [30]. This activation induced cell death (AICD) of CAR T cells is dependent on the level of antigen expression since CAR T cell survival was higher in cell lines with lower GD2 expression. The AICD process could be potentially reversed with anti-PD-1 blockade, as treatment with the anti-PD-1 mAb was able to restore CAR T-cell survival similar to the level observed for CAR T cells cultured with GD2− cell lines. Interestingly, while siRNA knockdown of PD-1 enhanced the CAR T-cell viability, PD-L1 was found to have a more complex role. PD-L1 signaling via PD-1 caused inhibition or deletion of activated PD-1+ T cells [31,32], whereas signaling via CD80 stimulated naive T cells [33]. Furthermore, PD-L1 could mediate reverse signaling upon binding to PD-1 within PD-L1+ T cells and promote their survival. SiRNA knockdown of PD-L1 significantly reduced cell survival. Following the detailed in vitro study, the GD2 CAR T construct was administrated to four metastatic melanoma patients. The GD2-CAR population declined in all patients beyond day 28 post-infusion, and failed to persist in two patients. These observations suggested that CAR T cells were also being depleted in vivo. Based on the in vitro data, combination of PD-1 blockade with CAR T therapy may help to augment the efficacy and persistence of CAR T cells.
GD2-CAR T cell treatment can be combined with chemotherapy to enhance efficacy. Heczey et. al [34] reported their clinical results in NB treatment, with three cohorts receiving GD2-CAR T, GD2-CAR T with cyclophosphamide and fludarabine (Cy/Flu) prior to cell infusions, and GD2-CAR T co-administered with Cy/Flu and PD-1 inhibitor respectively. GD2-CAR T and Cy/Flu treatment led to superior T cell expansion and higher levels of IL15 in the blood. Co-administering a PD-1 inhibitor did not make any differences in expansion, persistence of the cells, or circulating cytokine levels in this report. In contrast, other CAR T studies demonstrated beneficial clinical outcome when combined with PD-1 inhibitors [35–37], which may be due to variations in cancer types, antigen targets and timing/duration of PD-1 inhibitor treatment. Thus, careful optimization is necessary for each treatment protocol.
2.4. Reengineering of the GD2 CAR
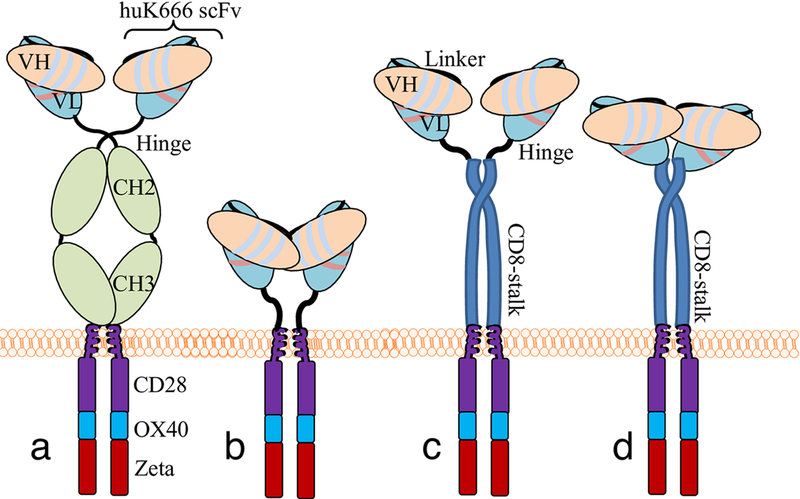
As many anti-GD2 mAbs originated from mice, administration of CAR T cells based on these antibodies can potentially generate anti-idiotype or anti-mouse antibodies recognizing the CAR and causing immune rejections. Thomas et.al [38] modified GD2-CAR T to incorporate humanized scFv of anti-GD2 mAb. The humanized CAR T had the same efficiency in proliferation and cytokine release as its murine counterpart. In addition, the effect of spacer between the ScFv and CD28 on CAR function was investigated. The inclusion of IgG Fc domain (Figure 2a) showed optimal efficiency compared to CAR T comprising of the hinge alone (Figure 2b), the hinge attached to the stalk of CD8a (Figure 2c) or the CD8a stalk alone (Figure 2d). These results showed the flexibility and binding ability to target antigen can play an important role in CAR T activation and cytokine release.
Figure 2.
Schematic demonstration of various GD2 CAR constructs. a) The CAR includes the hinge region as well as the Fc domain (CH2 and CH3); b) CAR without the Fc domain; c) CAR with the hinge attached to the stalk of CD8a; and d) CAR with CD8a stalk only without the hinge region. (Image adapted from [38])
Effective targeting of GD2 specific CAR T cells is a challenging task. Richman et.al.[39] showed by mutation of the aspartic acid residue 101 in the heavy chain of CAR scFv to lysine (E101HK), the affinity of GD2-CAR T cells could be significantly improved towards various GD2+ cell lines. In a mouse liver cancer model, histologic analysis of livers of mice treated with this construct showed less than 1% tumor cell foci compared to >95% in control and parent GD2 CAR T cell treated groups highlighting the efficacy of these engineered CAR T cells. However, all the mice treated developed severe neurotoxicity associated with T cell infiltration and proliferation into their brains presumably due to the expression of GD2 in brain tissues and the “on-target, off-tumor tissue” toxicity.
Enhancing the safety profile of CAR T cell therapy while achieving high anti-cancer efficacy of GD2 CAR T cells requires further cellular engineering. One potential cause for toxicity is the recognition of Fc on CAR-T by cells with Fcγ receptors. Since IgG Fc is the natural ligand for high affinity Fcγ receptors (CD64), mutation of the Fc domain, which is commonly used as the spacer between the antigen recognition domain and the intracellular signaling domain in CAR structure (Figure 2a), significantly reduced the off target toxicity of GD2-CAR T cells by preventing the engagement of CD64 expressing myeloid cells with CAR T cells [38].
An alternative approach to avoid the “on-target, off-tumor” effect is to build in inducible suicide genes. The inducible Caspase 9 (iCasp9) suicide gene was incorporated into the CAR of GD2 CAR T cells [38,40]. The iCasp9 was not toxic at basal expression levels. Administration of the small molecule dimerizer drug to the cells induced the expression of iCasp9, resulting in rapid onset of apoptosis in transduced cells. Activated cells expressing the CAR were preferentially killed, providing a safety switch. Similarly, the iCasp9 suicide gene has been applied to another type of GD2 CAR T, i.e., GD2 CAR.15 T cells, enhancing the safety profile of the construct [24].
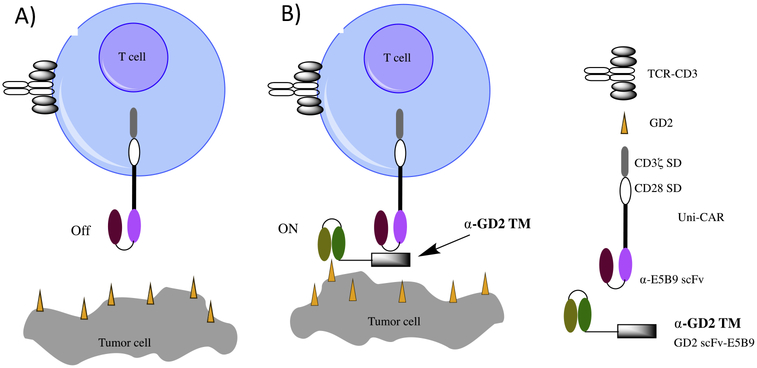
Mitwasi et al. [41] designed a new type CAR T, termed universal CAR T (UniCAR), whose activation is dependent on a target module (TM), and can be potentially turned on and off via dosing of TM. The scFv binding domain of UniCAR recognized a short peptide epitope of 10 amino acid derived from the human nuclear autoantigen (5B9). The UniCAR cells were inactive after infusion since the target epitope did not exist on the cell surface. A TM was synthesized by fusing the 5B9 antigen to GD2 binding scFv (Figure 3). In the presence of the TM, UniCARs were able to specifically target GD2+ tumor in a TM-dependent manner. At effector to target cell ratio of 5:1, only less than 0.1 nM of GD2-TMs was needed for uniCAR T to mediate the lysis of tumor cells. No killings were observed in control cell lines or in the absence of TMs. In contrast to other safety switches such as modification with inducible suicide genes, the uniCAR T cells were unique that the activities could be modulated rapidly via dosing of a target module.
Figure 3.
Construction of UniCAR and TM. A) UniCAR cells do not recognize tumor cells in the absence of TM due to the lack of receptor on T cells towards tumor antigens. B) Upon addition of the TM comprised of the conjugate of anti-GD2 scFv and E5B9, the UniCAR can bind with the TM through E5B9, thus gaining the abilities to recognize GD2+ tumor cells.
3. MUC1 CAR T cells
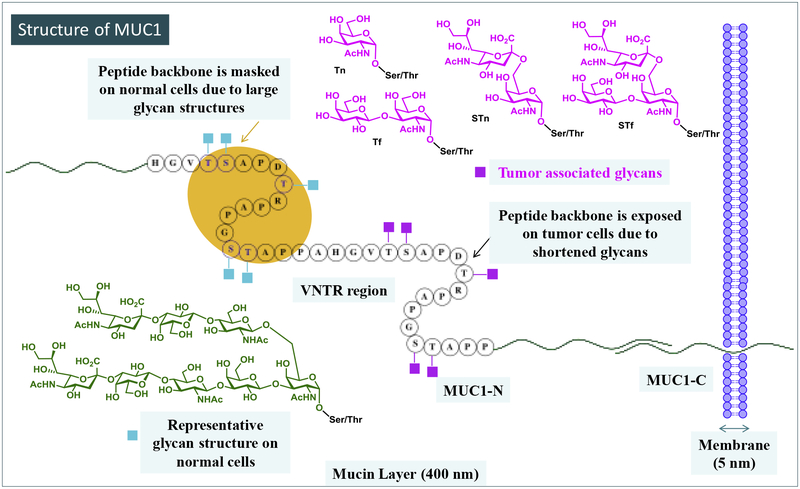
The human mucin 1 (MUC1) are high molecular weight glycoproteins expressed on the epithelial cell layers in the lung, breast, pancreas, kidney, ovary, colon, and other tissues to provide protection of these cells, which are exposed to external environments [42]. MUC1 consists of two subunits, MUC1-N and MUC1-C, which form a stable non-covalent complex at the cell surface. MUC1-N contains a variable number of 20 amino acid tandem repeats (VNTR) that are heavily glycosylated on serine or threonine residues of each VNTR in normal cells (Figure 4). However, on cancer cells, owing to altered glycosyltransferase expression, tumor-associated MUC1 is decorated by a preponderance of shorter glycans, including Thomsen-nouveau (Tn), sialyl Tn (STn), Thomsen-Friedenreich (Tf), and sialyl-T (STf) (Figure 4). Under-glycosylation of MUC1 unmasks cryptic epitopes within the extracellular domain, enabling tumor-selective binding by antibodies.
Figure 4.
Schematic demonstration of MUC1 glycoprotein structure. MUC1 is composed of a heterodimer of MUC1-N linked non-covalently with the transmembrane MUC1-C. MUC1-N contains a variable number of 20 amino acid VNTRs that are heavily glycosylated on serine or threonine residues of each VNTR in normal cells shielding the protein backbone for immune recognition. However, tumor associated MUC1 are hypoglycosylated exposing the protein backbone.
3.1. MUC1 epitope structures for CAR T cell targeting
Multiple mAbs are available against the MUC1-N domain. A representative example is AS1402, which has been evaluated in phase 1 clinical trial for breast cancer patients [43]. However, the result showed that the addition of AS1402 to chemotherapy had the same efficacy compared to chemotherapy treatment alone [44]. As MUC1-N is known to be shed from cancer cell surface into circulation, the extracellular pool of MUC1-N in plasma is considered a major barrier for antibody-dependent cellular toxicity against MUC1-N. In addition to passive immunity using anti-MUC1 mAbs, vaccines targeting tumor associated MUC1 have been evaluated, including L-BLP25 and PANVAC-V, which have been tested in late-stage clinical trials for the treatment of breast cancer [43,45–47]. However, no successful anti-MUC1 vaccines are available yet. As an alternative to mAbs and vaccines, anti-MUC1 CAR T cells have been investigated.
One of the key considerations in designing anti-MUC1 CAR T cells is the epitope structure targeted by the CAR. As MUC1 is a large glycoprotein, there are many potential sequences with MUC1 that can be targeted. Early MUC1 CAR T design utilized scFvs derived from anti-MUC1 mAb SM3 [48]. While SM3 recognized well the unglycosylated PDTR peptide within the VNTR region of MUC1-N, it bound poorly with glycopeptides such as the STf antigen present on many types of cancer cells. As a result, the SM3 MUC1 CAR T cells did not respond strongly to MUC1 expressing tumor cells. Nevertheless, MUC1 CAR T cells expressing a mutated analog of SM3 to improve MUC1 binding, were evaluated in a first-in-human study by direct injection into tumor lesions [49]. Reduction of MUC1 expression due to apoptosis and necrosis of tumor cells were observed in solid tumors treated with these CAR T cells, providing new options for improved CAR T therapy.
To overcome the low MUC1 affinity of SM3 mAb, another anti-MUC1 mAb, HMFG2 was utilized to construct the CAR [48]. HMFG2 has higher affinities to MUC1 than SM3 and can bind STf bearing MUC1 glycopeptides. In order to reduce the steric hindrance posted by immobilized MUC1, an IgD hinge was inserted into the CAR yielding a potent receptor containing a fused CD28/OX40/CD3ζ endodomain, so that the Fab regions can engage antigen in virtually any orientations. These HMFG2-CD28-OX40-CD3ζ CAR T cells proliferated upon encountering the MUC1 antigen, mediated production of proinflammatory cytokines, and killed MUC1+ tumor cells. When tested in a preclinical model, these CAR T cells led to a significant delay of tumor growth [48,50,51].
A potential drawback of the HMFG2 antibody is that besides the glycosylated form of MUC1, HMFG2 also recognizes unglycosylated MUC1 peptide, which raises concerns of possible auto-immunity due to binding with unglycosylated MUC1 in normal tissues. Anti-MUC1 mAb 5E5 is an antibody that has been shown to have high affinities for the Tn glycosylated MUC1 (MUC1-Tn) with little cross-reactivity with the non-glycosylated MUC1–60-mer peptide or normal human tissues, supporting the more cancer specific expression of Tn-glycopeptide epitopes [52]. With the knowledge of high tumor selectivity by 5E5, Posey et. al. developed MUC1-Tn CAR T cells using 5E5, which recognized multiple tumor cell lines expressing MUC1-Tn [53]. Increasing the expression of enzymes in tumor cells that converts Tn to other glycans reduced the levels of MUC1-Tn on tumor cells. As a result, the 5E5 CAR T cell induced cytotoxicity to these cells were significantly suppressed, which may be concerning for cancer treatment due to the heterogeneity of MUC1 glycosylation patterns on cancer cells. Interestingly, when evaluated in xenograft model of human pancreatic cancer studies, 5E5 CAR T cells provided complete protection to tumor bearing mice, suggesting that 5E5 driven CAR T cells are able to detect low levels of antigen targets, and antigenic diversity does not preclude tumor eradication [53].
Another example of CAR-T design against the exposed peptide backbone of aberrantly glycosylated MUC1 is to use scFv derived from anti-MUC1 mAb (TAB004) for recognition of STAPPVHNV peptide sequence within the VNTR region. The scFv was fused to CD28 and CD3ζ T cell intracellular signaling molecule to generate the CAR T cells, which were evaluated in triple-negative breast cancer (TNBC) [54]. MUC1 CAR T cell mediated tumor cell lysis correlated with MUC1 expression levels in vitro without significant lysis of normal breast epithelial tissues, suggesting their high tumor selectivities. Treatment of HCC70 tumor-bearing mice with this construct dramatically reduced the tumor growth from as early as 4-days post T cell injection, which lasted up to 57 days. However, the tumor started to progress faster after about 60 days post treatment compared to the control group. Histological analysis showed that MUC1 expression levels were similar in both treatment groups, suggesting MUC1 tumor antigen down-regulation to avoid immune clearance by MUC1 CAR T cells is likely not a major contributing factor to tumor escape. Instead, an approximate two-fold increase of PD-1 expression on tumor cells was observed after CAR T cell treatment, which may be a major reason causing the exhaustion of CAR T cells. Increased lysis potency by MUC1 CAR T cells with pretreatment of an anti-PD1 antibody suggested the potential beneficial combination of CAR T cells with an anti-PD1 antibody for future evaluations.
3.2. Building costimulatory signals into MUC1 based CAR T cells and dual targeting of tumor cells
To enhance the anti-tumor efficacy and reduce tumor escape from the immune pressure, an attractive strategy is to combine multiple types CAR T cells. This combinational CAR T cell therapy has been investigated for the treatment of non-small-cell lung cancer [55]. Prostate stem cell antigen (PSCA) is frequently expressed in non-small-cell lung cancer along with MUC1 antigen. MUC1 and PSCA CAR T cells were constructed based on HMFG2 and humanized 1G8 antibodies respectively. When administered individually, MUC1 and PSCA CAR T cells significantly reduced tumor growth in a mouse non-small-cell lung cancer model compared to mock groups receiving non-tumor targeting CAR T cells. Combining the MUC1 and PSCA CAR T cells led to further reduction of tumor sizes, highlighting the power of this combinatorial approach.
Costimulatory signals such as CD28 and CD3ζ are critical to sustain CAR T cell proliferation, enhance cytotoxicity, and resist AICD. However, as the CAR T cells become more powerful, toxicities have emerged [56,57], which are often attributed to “on target, off tumor” effects due to the presence of tumor – associated antigens in healthy tissues off tumor sites. CAR T cells are commonly designed with both CD28 and CD3ζ on the same gene (also known as cis arrangement). Upon CAR engagement, CD28 and CD3ζ are both activated with CD3ζ signaling eliciting cytotoxicity and interferon-γ production, and CD28 creating signal 2 to promote T-cell proliferation and interleukin (IL)-2 production.
One novel design to increase the specificity of CARs and improve their activity and safety is to physically separate the two stimulatory signals by incorporating them into 2 distinct CARs specific for 2 different antigens (trans signaling). In this regard, Wilkie and his coworkers engineered CAR T cells to simultaneously target ErbB2 and MUC1, two common breast cancer associated antigens [58]. The resulting CAR T, called ITH, coexpresses the ErbB2 specific CAR using scFv of anti ErbB2 mAb followed by CD3ζ signaling domain (Iz1) and MUC1 specific CARs based on scFv of anti MUC1 mAb, HMFG2, coupled to CD28 domain (HDF28). The two transgenes were co-expressed in equal amounts by separating the inserts with a Thosea asigna (T2A) peptide. The ITH construct could deliver complementary signals required for proliferation and lead to greater expansion of T cells compared to control CAR Ts with CD28 or CD3ζ alone. ITH CAR T cells showed similar cytolytic efficiency as anti-ErbB2 CAR T cells against multiple breast cancer cell lines with varying levels of ErbB2 and MUC1 expression.
Deeper analysis of ITH CAR T cells was performed. It was notable that the production of IL-2 by ITH CARs was much lower compared to the CARs with cis fused CD28 + CD3ζ endodomain. Possible reasons included the conformational changes of scFv induced after binding of the first antigen to its corresponding scFv, which may hinder the optimal binding of the second scFv with its respective antigen, posing steric challenge for simultaneous targeting of both target antigens [59]. Alternatively, co-stimulatory signaling from the ITH CAR T cells may be either less efficient or distinct from that provided by CARs containing a fused CD28 + CD3ζ endodomain.
Besides the MUC1 and ErbB2 combination for ITH CAR T cells, other dual targeting CAR T cells with CAR pairs such as prostate antigens PSMA/PSCA and mesothelin/folate receptor have been evaluated in vitro and in vivo [60,61]. These studies showed that co-transduced trans-signaling CAR T cells could destroy tumors expressing both antigens, without affecting those with either antigen alone. Thus, a dual targeting, trans-signaling CAR approach can enhance the anti-cancer efficacy of CAR T cells while minimizing off tumor side effects against normal tissues bearing single antigen.
4. Bispecific Antibodies targeting GD2 and MUC1
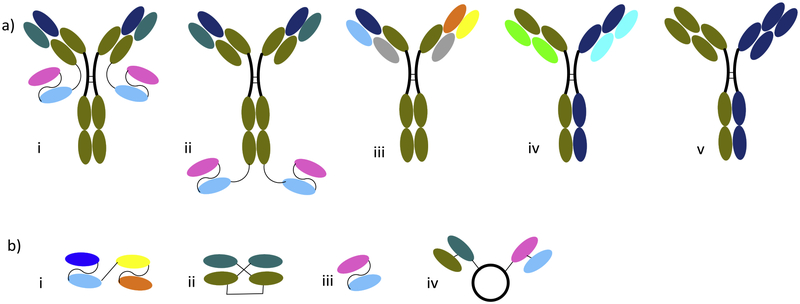
Bispecific antibodies (BsAbs) are engineered antibodies that can simultaneously engage two unique epitopes, such as ligands, receptors, and cytokines. While there are many different designs of BsAbs in development, they can be generally classified into two categories: immunoglobulin G (IgG)-like molecules and non-IgG-like molecules [62]. With two heavy and two light chains, the overall geometry of IgG like BsAbs resemble those of IgG (Figure 5a). However, unlike traditional IgGs, the two chains of BsAbs recognize two distinct epitopes. The IgG like BsAbs contain the Fc domain, which can mediate effector functions such as antibody-dependent cell-mediated cytotoxicity, complement-dependent cytotoxicity, and antibody-dependent cellular phagocytosis. In comparison, the non-IgG like BsAbs lack the entire Fc domain (Figure 5b). These BsAbs can include chemically linked Fabs, various types of bivalent and trivalent single-chain variable fragments (scFvs) with different epitope specificities.
Figure 5.
Schematic representation of various BsAb formats. a) IgG-like BsAbs: i) and ii) IgG-scFv, iii) triomab, iv) quadroma, and v) half molecule exchange format. And b) non-IgG-like BsAb: i) tandem scFv, ii) dual-affinity re-targeting antibody, iii) bi-nanobody, iv) scFv-human serum albumin-scFv.
BsAbs may improve tumor targeting specificity by binding two tumor-associated antigens. Furthermore, BsAbs can be designed to engage immune system effector cells, usually through CD3 on cytotoxic T cells or CD16 on natural killer cells. This can bring the immune cells into close proximity with cancer cells and lead to the killing of cancer cells through perforin/granzyme-mediated non-MHC-restricted specific antitumor cytotoxicity [63–66].
4.1. GD2 targeting BsAbs
The most common TACA based BsAbs target GD2. One type of BsAbs was produced by chemically conjugating anti-CD3 (OKT3) and anti-GD2 (3F8) antibodies through a heterobifunctional linker [67]. This BsAb could coat expanded T cells in vitro, and help direct the activated T cells to neuroblastoma cells expressing GD2 antigens. The BsAb treatment could kill GD2 positive cells more effectively than T cells or 3F8 antibody treatment alone. Furthermore, it is highly promising that compared to intravenous injection of 3F8 mAb alone, the dose required for 3F8BsAb was 200 times lower for similar efficacy. This is a major advantage, as the lower dose needed can potentially reduce the dose-limiting pain caused by 3F8 binding to peripheral nerve fibers.
A second type anti-GD2 BsAb, Ektomab, was built in a quadroma-based format. One binding arm of Ektomab is from mouse IgG2a ME361, which can recognize ganglioside GD2 albeit with a lower affinity compared to 3F8. The other binding arm is from rat IgG2b recognizing CD3 on human T cells. Significant lytic activity of Ektomab could be observed down to 40 ng/ml. However, both murine and rat components could induce neutralizing antibodies in humans, which diminish their effects rendering the need for higher doses.
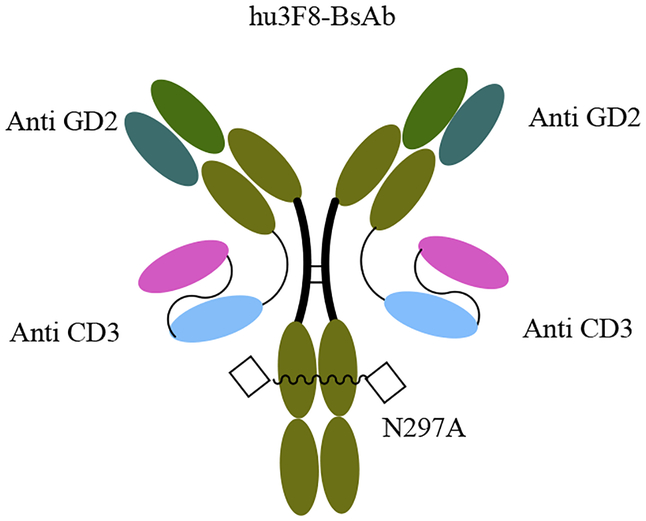
To reduce anti-mouse/rat antibodies generated in humans, the first humanized IgG-scFv BsAb was developed for targeting GD2. In this work, the scFv of humanized mouse anti CD3 was attached to the carboxy end of anti GD2 light chain antibody (Figure 6) [68]. The BsAb has the same binding affinity for GD2 as parental IgG but CD3 binding was significantly reduced compared to parental anti-CD3 mAb. Using humanized anti GD2 and CD3 antibodies lowered the amounts of neutralizing antibodies generated when administered in humans. The Hu3f8-BsAb was highly potent against GD2+ tumor with cytotoxicity at femtomolar concentrations and greater than 105 fold selectivity over normal tissues. In addition, asparagine 297 of the Fc was mutated to alanine to remove the Fc glycosylation, which was proposed to reduce the risk of cytokine storm syndrome by preventing Fc receptor mediated binding. The Hu3F8-BsAb effectively reduced tumor growth in humanized mouse model with a high safety profile. Mechanistic studies showed that besides T cells, monocytes also play critical roles in sustaining T-cell infiltration of tumor stroma, survival, or proliferation, and contributing significantly to the exceptional antitumor effect of Hu3f8-BsAb.
Figure 6.
Schematic demonstration of the Hu3F8-BsAb structure.
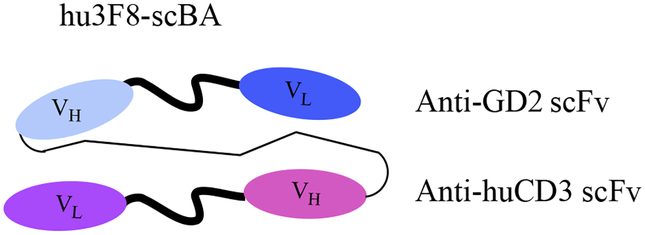
A non-IgG like anti-GD2 BsAb was designed by a tandem fusion of single chain variable fragment of Hu3F8 to anti-CD3 antibody HuOKT3-scFv with a 15-residue linker (GGGGS)3 producing the hu3F8-scBA (Figure 7) [69]. This engineered BsAb contained the human anti GD2 (Hu3F8-scFv) rather than the murine anti-GD2 mAb 5F11 in earlier generation of the BsAb, resulting in a 13 fold higher affinity for GD2. This higher affinity led to stronger T cell activation and cytokine release in vitro. T cell cytotoxicity assays against GD2 expressing human cancer cells showed that Hu3F8-scFv was highly potent with EC50 values in the femtomolar range, which was up to 5,000-fold stronger than the 5F11 version of the BsAb. Hu3f8-scBA showed significant suppression of tumor growth in DKO mice with human neuroblastoma/melanoma xenografts. One drawback of Hu3F8-scFv is that this antibody has lower thermal stability, which needs to be improved for future clinical applications. Despite the decrease in thermal stability, this study highlighted that even relatively modest increases in antigen affinity could lead to substantial enhancement of the functional properties of the BsAb.
Figure 7.
scFv-based bispecific antibody format of Hu3F8-scBA (VH is the heavy chain of the variable region, and the VL is the light chain of the variable region).
BsAbs can be applied in combinatorial therapy to enhance the effectiveness of the therapy and address immune suppression encountered. Deppisch et. al. constructed a BsAb antibody termed Surek, which targets both GD2 and the CD3 receptor on murine T cells with the Fc region consisting of mouse IgG2a and rat IgG2b isotypes recognizable by Fc receptors [70]. The combination of mouse IgG2a and rat IgG2b isotypes can facilitate the purification of quadroma bispecific antibodies by affinity chromatography using protein A taking advantage of the differential affinities of various IgG isotypes with protein A. The Surek was able to successfully recruit T cells to tumor tissues. However, detailed analysis showed that cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), another immune checkpoint protein, was upregulated in these redirected T cells, which led to the addition of an anti-CTLA-4 mAb for treatment. The combined Surek and anti-CTLA-4 mAb regime increased the overall survival of mice challenged with B78-D14 melanoma to 90% compared to the Surek alone (60%), while only 20% of the mice in the group receiving anti-CTLA-4 mAb alone survived the tumor. However, the survival advantage was more modest in another melanoma model B16-EpCAM.
Besides the direct effect of Surek on tumor cells, Surek could induce tumor-specific humoral immune responses [16], enhancing tumor protection in vivo. Combined administration of Surek with CTLA-4 blockade improved humoral immunity against cancer, which correlated with increased serum titers of melanoma-reactive IgG2a antibodies. The combinatorial vaccination markedly increased the number of memory CD4+ T cells [70]. Survival rate of mice challenged with tumor three weeks after immunization receiving both Surek and anti-CTLA-4 antibodies was 40% better than the group immunized with Surek only.
4.2. MUC1 based BsAbs
Compared to GD2 based BsAbs, there are far fewer studies on MUC1 based BsAbs. Two BsAbs, i.e., MUC1 × CD3 and MUC1 × CD28 were constructed [71]. These two BsAbs reacted well with MUC1+ tumor cells, and in the presence of interleukin-12 activated killer cells, were able to exhibit significant cytotoxicity to tumor cells. The cytotoxicity by MUC1 × CD3 BsAb alone was similar to those when the MUC1 × CD3 and MUC1 × CD28 BsAbs were combined. When evaluated in a mouse tumor model, MUC1 × CD3 BsAb was able to significantly slow down the growth of TFK-1, a type of human bile duct carcinoma.
In addition to T cells, natural killer cells (NK cells) are another type of effector cells capable of killing infected or malignant cells. NK cell mediated antibody-dependent cellular cytotoxicity plays a major role in antibody mediated targeted cancer therapy, including anti-MUC1 and anti-GD2 monoclonal antibody treatment [72–74]. As an alternative to T cell recruiting BsAbs, bispecific antibody capable of binding with NK cells has been investigated for TACAs. A new non-IgG like BsAb was designed by linking single domain anti-MUC1 and anti-CD16 antibodies through a short dipeptide linker. This BSAb could be efficiently expressed in E. coli, and recruit NK cells to bestow potent killing of MUC1 expressing tumor cells. In mouse xenograft tumor models, the BsAb provided significant protection from human colon cancer LS174 growth [75].
4.3. CAR T cells vs BsAbs
A direct comparison study of GD2 targeting BsAb (Hu3F8 × anti CD3) vs CAR T cells was reported by Cheung and coworkers [76]. When incubated with GD2 expressing tumor cells in vitro, a majority of CAR T cells with high density receptors were depleted while T cells stimulated with BsAb survived. Upregulations of PD1 and LAG3 marker expression were seen in both groups after stimulation with target cells suggesting that over-expression of PD1 was not the exhaustion signal in this study and blocking of PD1 did not improve the CAR T cell survival. CAR T cell exhaustion was observed in all generated CAR T cells with different affinities, indicating that the density of CAR on the cell surface rather than the affinity of the CAR itself may be the important determinant of the GD2 CAR T cell depletion. BsAb in conjunction with untransduced T cells showed superior antitumor activity in vivo in human melanoma xenograft model. While both CAR T cells and BsAb recruited T cells penetrated into the tumor sites, the number of tumor infiltrating lymphocytes (TIL) was greater with untransduced T cells plus BsAb treatment compared to the CAR T cell group, which can be the result of higher proliferation of T cells in presence of BsAb. The TIL showed equal percentage of CD4+ and CD8+ T cell population for BsAb group versus CAR T group with almost all the CAR T cells expressing CD8+. This absence of CD4+ T cell may have compromised the therapeutic response of CAR T cells [76].
5. Conclusions and perspectives.
With the overexpression on a wide range of cancer cells, TACAs are attractive targets for immunotherapy development. Anti-TACA CAR T cells and BsAbs are exciting directions for TACA based immunotherapy.
While the concept of CAR T cells can be relatively straightforward, multiple parameters need to be established for effective CAR T therapy. Early studies of GD2 CAR T cells demonstrated that it is important to build in costimulatory domains such as CD28 and OX40 into the T cells in order to maintain the abilities of cells to proliferate, as well as to reduce T cell exhaustion and AICD. Other stimulatory molecules including IL15 and CD27 have been incorporated into GD2 CAR T cells, which enabled long term persistence of the cells in human patients, and led to improved clinical outcome. Co-administration of immune checkpoint blockade through anti-PD-1 antibody may help further augment the efficacy and persistence of GD2 CAR T cells.
To overcome low penetration of CAR T cells into solid tumor tissues, GD2 CAR T therapy has been combined with anti-angiogenic agents as well as heparanase for digestion of extracellular matrix. This led to significantly improved protection of mice from GD2+ tumor induced death.
As GD2 CAR T cells become more potent, the “on target, off tumor” binding of T cells to low level of GD2 expressed in normal tissues can cause serious side effects including neurotoxicity. To address this challenge, CAR engineering can be performed to reduce Fc receptor binding to CAR. Furthermore, innovative research has been established to introduce inducible suicide gene or design target modules to turn on and turn off the CAR T responses when necessary.
Similar to GD2 CAR T cells, the design of effective MUC1 CAR T cells required built-in co-stimulatory signals to sustain CAR T cell proliferation, enhance cytotoxicity, and resist AICD. Another major consideration for MUC1 CAR T cells is the MUC1 epitope to be targeted as MUC1 glycoprotein contains many potential antigenic sites. Early generations of the cells were based on antibodies recognizing the unglycosylated MUC1 peptides in the VNTR region of MUC1-N. While in preclinical models, these CAR T cells delayed tumor growth, there were concerns regarding potential auto-immunity. To address this issue, MUC1 CAR T cells were developed targeting the more tumor specific MUC1-Tn glycopeptides, which recognized multiple tumor cell lines. When evaluated in xenograft model of human pancreatic cancer studies, these CAR T cells provided complete protection to tumor bearing mice.
As an alternative to CAR T cells, bispecific antibodies have shown great promises in anti-cancer therapy. The most common type of TACA based BsAbs targets GD2 and CD3 on T cells. Engineering of the BsAbs include humanization of the non-human origin scFv to reduce the level of neutralization antibodies, and Fc mutation to reduce the risk of cytokine storm syndrome. These BsAbs can direct activated T cells to GD2 expressing tumor tissues, leading to significant tumor lysis even with femtomolar concentrations of the BsAbs.
A direct comparison study has been performed to compare GD2 BsAbs and CAR T cells. Administration of BsAbs in conjunction with untransduced T cells were found to lead to longer survival of activated T cells. Furthermore, BsAbs-T cells provided more effective tumor protection in tumor models. The superiority of BsAbs-T cells could be partly attributed to the presence of CD4+ helper T cells in BsAbs-T cells, while the infused CAR T cells were almost exclusively CD8+ T cells.
Building on the results from the reported CAR T cell and BsAb studies, a promising direction to explore is to test novel combination therapies. As cancer cells may mutate to downregulate the levels of targeted antigen under immune pressure, constructs that can potentially bind with multiple types of antigens may help reduce the chances of tumor escape and enhance treatment efficacy. Other combinations can include the addition of checkpoint inhibition such as anti-PD1 and anti-CTLA4 mAbs, agents that can enhance the access and penetration of immune cells to solid tumor, as well as chemotherapeutic agents. In order to guide such studies, it is critical to gain a more thorough understanding of the tumor microenvironment and factors in suppressing the immune responses. These approaches may overcome the inhibitory signals in tumor microenvironment, high interstitial fluid pressure, compressed vasculature and dense fibrotic tissue surrounding solid tumors and increase the delivery of immunotherapeutic agents to solid tumors.
A critical factor in the success of immunotherapy is in minimizing potential toxicity and side effects, especially considering many tumor antigens can also be found in normal tissues, albeit at low levels. To reduce the “on target, off tumor” side effects, further investigation for characterization of TACA structures in solid tumor tissues can lead to the discovery of unique TACA derivatives. For example, the expression of the 9-O-acetylated GD2 gangliosides has been reported to be restricted to tumor tissues, while they are not found in normal tissues [77]. Furthermore, compared to normal cells, tumors tend to contain more N-glycolyl neuraminic acid [78,79]. These rarer structures may provide more specific targets for anti-cancer immunotherapy to enhance the safety of the treatment.
Acknowledgements
We are grateful to the National Institutes of Health (R01 CA225105, R01AI146210), Michigan State University Foundation, the Michigan Economic Development Corporation through the MTRAC program, the department of chemistry, Michigan State University for partial financial support.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- [1].World Health Organisation, Latest global cancer data, Int. Agency Res. Cancer (2018) 13–15. https://www.iarc.fr/wp-content/uploads/2018/09/pr263_E.pdf (accessed November 3, 2019).
- [2].International Agency for Research on Cancer, Cancer Tomorrow, World Heal. Organ (2019)1–2.https://gco.iarc.fr/tomorrow/graphic-line?type=0&population=900&mode=populatio&sex=0&cancer=39&age_group=value&apc_male=0&apc_female=0 (accessed November 3, 2019).
- [3].Couzin-Frankel J, Breakthrough of the year 2013. Cancer immunotherapy., Science. 342 (2013) 1432–3. doi: 10.1126/science.342.6165.1432. [DOI] [PubMed] [Google Scholar]
- [4].Hakomori SI, Tumor-associated carbohydrate antigens defining tumor malignancy: Basis for development of anti-cancer vaccines, in: Adv. Exp. Med. Biol, 2001: pp. 369–402. doi: 10.1007/978-1-4615-1267-7_24. [DOI] [PubMed] [Google Scholar]
- [5].Buskas T, Thompson P, Boons GJ, Immunotherapy for cancer: Synthetic carbohydrate-based vaccines, Chem. Commun (2009) 5335–5349. doi: 10.1039/b908664c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Jing X, Liang H, Hao C, Yang X, Cui X, Overexpression of MUC1 predicts poor prognosis in patients with breast cancer, Oncol. Rep 41 (2019) 801–810. doi: 10.3892/or.2018.6887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Danishefsky S, Allen J, From the laboratory to the clinic: A retrospective on fully synthetic carbohydrate-based anticancer vaccines frequently used abbreviations are listed in the appendix., Angew. Chem. Int. Ed. Engl 39 (2000) 836–863. doi: 10.1002/(sici)1521-3773(20000303)39:5<836::aid-anie836>3.0.co;2-i. [DOI] [PubMed] [Google Scholar]
- [8].Yin Z, Huang X, Recent development in carbohydrate based anticancer vaccines, J. Carbohydr. Chem 31 (2012) 143–186. doi: 10.1080/07328303.2012.659364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Wei MM, Wang YS, Ye XS, Carbohydrate-based vaccines for oncotherapy, Med. Res. Rev 38 (2018) 1003–1026. doi: 10.1002/med.21493. [DOI] [PubMed] [Google Scholar]
- [10].Feng D, Shaikh AS, Wang F, Recent advance in tumor-associated carbohydrate antigens (TACAs)-based antitumor vaccines, ACS Chem. Biol 11 (2016) 850–863. doi: 10.1021/acschembio.6b00084. [DOI] [PubMed] [Google Scholar]
- [11].Guo Z, Wang Q, Recent development in carbohydrate-based cancer vaccines, Curr. Opin. Chem. Biol 13 (2009) 608–617. doi: 10.1016/j.cbpa.2009.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Riley RS, June CH, Langer R, Mitchell MJ, Delivery technologies for cancer immunotherapy, Nat. Rev. Drug Discov 18 (2019) 175–196. doi: 10.1038/s41573-018-0006-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Lloyd KO, Old LJ, Human monoclonal antibodies to glycolipids and other carbohydrate antigens: dissection of the humoral immune response in cancer patients, Cancer Res. 49 (1989) 3445–3451. https://cancerres.aacrjournals.org/content/49/13/3445.full-text.pdf (accessed October 31, 2019). [PubMed] [Google Scholar]
- [14].Sait S, Modak S, Anti-GD2 immunotherapy for neuroblastoma, Expert Rev. Anticancer Ther 17 (2017) 889–904. doi: 10.1080/14737140.2017.1364995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Hung J-T, Yu AL, GD2-Targeted Immunotherapy of Neuroblastoma, in: Neuroblastoma, Elsevier, 2019: pp. 63–78. doi: 10.1016/b978-0-12-812005-7.00004-7. [DOI] [Google Scholar]
- [16].Battula VL, Shi Y, Evans KW, Wang RY, Spaeth EL, Jacamo RO, Guerra R, Sahin AA, Marini FC, Hortobagyi G, Mani SA, Andreeff M, Ganglioside GD2 identifies breast cancer stem cells and promotes tumorigenesis, J. Clin. Invest 122 (2012) 2066–2078. doi: 10.1172/JCI59735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Cheever MA, Allison JP, Ferris AS, Finn OJ, Hastings BM, Hecht TT, Mellman I, Prindiville SA, Viner JL, Weiner LM, Matrisian LM, The prioritization of cancer antigens: A national cancer institute pilot project for the acceleration of translational research, Clin. Cancer Res 15 (2009) 5323–5337. doi: 10.1158/1078-0432.CCR-09-0737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Gillies SD, Lo KM, Wesolowski J, High-level expression of chimeric antibodies using adapted cDNA variable region cassettes, J. Immunol. Methods 125 (1989) 191–202. doi: 10.1016/0022-1759(89)90093-8. [DOI] [PubMed] [Google Scholar]
- [19].D’Aloia MM, Zizzari IG, Sacchetti B, Pierelli L, Alimandi M, CAR-T cells: The long and winding road to solid tumors review-article, Cell Death Dis. 9 (2018). doi: 10.1038/s41419-018-0278-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Rossig C, Bollard CM, Nuchtern JG, Merchant DA, Brenner MK, Targeting of GD2-positive tumor cells by human T lymphocytes engineered to express chimeric T-cell receptor genes, Int. J. Cancer 94 (2001) 228–236. doi: 10.1002/ijc.1457. [DOI] [PubMed] [Google Scholar]
- [21].Richards RM, Sotillo E, Majzner RG, CAR T cell therapy for neuroblastoma, Front. Immunol 9 (2018) 2380. doi: 10.3389/fimmu.2018.02380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Pule MA, Savoldo B, Myers GD, Rossig C, Russell HV, Dotti G, Huls MH, Liu E, Gee AP, Mei Z, Yvon E, Weiss HL, Liu H, Rooney CM, Heslop HE, Brenner MK, Virus-specific T cells engineered to coexpress tumor-specific receptors: Persistence and antitumor activity in individuals with neuroblastoma, Nat. Med 14 (2008) 1264–1270. doi: 10.1038/nm.1882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Louis CU, Savoldo B, Dotti G, Pule M, Yvon E, Myers GD, Rossig C, Russell HV, Diouf O, Liu E, Liu H, Wu MF, Gee AP, Mei Z, Rooney CM, Heslop HE, Brenner MK, Antitumor activity and long-term fate of chimeric antigen receptor-positive T cells in patients with neuroblastoma, Blood. 118 (2011) 6050–6056. doi: 10.1182/blood-2011-05-354449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Chen Y, Sun C, Landoni E, Metelitsa L, Dotti G, Savoldo B, Eradication of neuroblastoma by T cells redirected with an optimized GD2-specific chimeric antigen receptor and interleukin-15, Clin. Cancer Res 25 (2019) 2915–2924. doi: 10.1158/1078-0432.CCR-18-1811. [DOI] [PubMed] [Google Scholar]
- [25].You F, Jiang L, Zhang B, Lu Q, Zhou Q, Liao X, Wu H, Du K, Zhu Y, Meng H, Gong Z, Zong Y, Huang L, Lu M, Tang J, Li Y, Zhai X, Wang X, Ye S, Chen D, Yuan L, Qi L, Yang L, Phase 1 clinical trial demonstrated that MUC1 positive metastatic seminal vesicle cancer can be effectively eradicated by modified Anti-MUC1 chimeric antigen receptor transduced T cells, Sci. China Life Sci 59 (2016) 386–397. doi: 10.1007/s11427-016-5024-7. [DOI] [PubMed] [Google Scholar]
- [26].Song DG, Ye Q, Poussin M, Harms GM, Figini M, Powell DJ, CD27 costimulation augments the survival and antitumor activity of redirected human T cells in vivo, Blood. 119 (2012) 696–706. doi: 10.1182/blood-2011-03-344275. [DOI] [PubMed] [Google Scholar]
- [27].Craddock JA, Lu A, Bear A, Pule M, Brenner MK, Rooney CM, Foster AE, Enhanced tumor trafficking of GD2 chimeric antigen receptor T cells by expression of the chemokine receptor CCR2b, J. Immunother 33 (2010) 780–788. doi: 10.1097/CJI.0b013e3181ee6675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Bocca P, Di Carlo E, Caruana I, Emionite L, Cilli M, De Angelis B, Quintarelli C, Pezzolo A, Raffaghello L, Morandi F, Locatelli F, Pistoia V, Prigione I, Bevacizumab-mediated tumor vasculature remodelling improves tumor infiltration and antitumor efficacy of GD2-CAR T cells in a human neuroblastoma preclinical model, Oncoimmunology. (2017). doi: 10.1080/2162402X.2017.1378843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Caruana I, Savoldo B, Hoyos V, Weber G, Liu H, Kim ES, Ittmann MM, Marchetti D, Dotti G, Heparanase promotes tumor infiltration and antitumor activity of CAR-redirected T lymphocytes, Nat. Med 21 (2015) 524–529. doi: 10.1038/nm.3833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Gargett T, Yu W, Dotti G, Yvon ES, Christo SN, Hayball JD, Lewis ID, Brenner MK, Brown MP, GD2-specific CAR T cells undergo potent activation and deletion following antigen encounter but can be protected from activation-induced cell death by PD-1 blockade, Mol. Ther 24 (2016) 1135–1149. doi: 10.1038/mt.2016.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Gibbons RM, Liu X, Pulko V, Harrington SM, Krco CJ, Kwon ED, Dong H, B7-H1 limits the entry of effector CD8+ T cells to the memory pool by upregulating bim, Oncoimmunology. 1 (2012) 1061–1073. doi: 10.4161/onci.20850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Latchman YE, Liang SC, Wu Y, Chernova T, Sobel RA, Klemm M, Kuchroo VK, Freman GJ, Sharpe AH, PD-L1-deficient mice show that PD-L1 on T cells, antigen-presenting cells, and host tissues negatively regulates T cells, Proc. Natl. Acad. Sci. U. S. A 101 (2004) 10691–10696. doi: 10.1073/pnas.0307252101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Rowe JH, Johanns TM, Ertelt JM, Way SS, PDL-1 blockade impedes T Cell expansion and protective immunity primed by attenuated listeria monocytogenes, J. Immunol 180 (2008) 7553–7557. doi: 10.4049/jimmunol.180.11.7553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Heczey A, Louis CU, Savoldo B, Dakhova O, Durett A, Grilley B, Liu H, Wu MF, Mei Z, Gee A, Mehta B, Zhang H, Mahmood N, Tashiro H, Heslop HE, Dotti G, Rooney CM, Brenner MK, CAR T cells administered in combination with lymphodepletion and PD-1 Inhibition to patients with neuroblastoma, Mol. Ther 25 (2017) 2214–2224. doi: 10.1016/j.ymthe.2017.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].John LB, Kershaw MH, Darcy PK, Blockade of PD-1 immunosuppression boosts CAR T-cell therapy, Oncoimmunology. 2 (2013) e26286. doi: 10.4161/onci.26286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Cherkassky L, Morello A, Villena-Vargas J, Feng Y, Dimitrov DS, Jones DR, Sadelain M, Adusumilli PS, Human CAR T cells with cell-intrinsic PD-1 checkpoint blockade resist tumor-mediated inhibition, J. Clin. Invest 126 (2016) 3130–3144. doi: 10.1172/JCI83092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Chong EA, Melenhorst JJ, Lacey SF, Ambrose DE, Gonzalez V, Levine BL, June CH, Schuster SJ, PD-1 blockade modulates chimeric antigen receptor (CAR)-modified T cells: Refueling the CAR, Blood. 129 (2017) 1039–1041. doi: 10.1182/blood-2016-09-738245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Thomas S, Straathof K, Himoudi N, Anderson J, Pule M, An optimized GD2-targeting retroviral cassette for more potent and safer cellular therapy of neuroblastoma and other cancers, PLoS One. 11 (2016). doi: 10.1371/journal.pone.0152196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Richman SA, Nunez-Cruz S, Moghimi B, Li LZ, Gershenson ZT, Mourelatos Z, Barrett DM, Grupp SA, Milone MC, High-Affinity GD2-specific CAR T cells induce fatal encephalitis in a preclinical neuroblastoma model, Cancer Immunol. Res 6 (2018) 36–46. doi: 10.1158/2326-6066.CIR-17-0211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Gargett T, Brown MP, The inducible caspase-9 suicide gene system as a “safety switch” to limit on-target, off-tumor toxicities of chimeric antigen receptor T-cells, Front. Pharmacol 5 (2014) 235. doi: 10.3389/fphar.2014.00235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Mitwasi N, Feldmann A, Bergmann R, Berndt N, Arndt C, Koristka S, Kegler A, Jureczek J, Hoffmann A, Ehninger A, Cartellieri M, Albert S, Rossig C, Ehninger G, Pietzsch J, Steinbach J, Bachmann M, Development of novel target modules for retargeting of UniCAR T cells to GD2 positive tumor cells, Oncotarget. 8 (2017) 108584–108603. doi: 10.18632/oncotarget.21017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Apostolopoulos Vasso PXX, Xiu Fen Hu, Pouniotis Dodie Stephanie, MUC1: a molecule of many talents, Curr. Trends Immunol 6 (2004) 39–55. [Google Scholar]
- [43].Kufe DW, MUC1-C oncoprotein as a target in breast cancer: Activation of signaling pathways and therapeutic approaches, Oncogene. 32 (2013) 1073–1081. doi: 10.1038/onc.2012.158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Ibrahim NK, Yariz KO, Bondarenko I, Manikhas A, Semiglazov V, Alyasova A, Komisarenko V, Shparyk Y, Murray JL, Jones D, Senderovich S, Chau A, Erlandsson F, Acton G, Pegram M, Randomized phase II trial of letrozole plus Anti-MUC1 antibody AS1402 in hormone receptor-positive locally advanced or metastatic breast cancer, Clin. Cancer Res 17 (2011) 6822–6830. doi: 10.1158/1078-0432.CCR-11-1151. [DOI] [PubMed] [Google Scholar]
- [45].Steentoft C, Migliorini D, King TR, Mandel U, June CH, Posey AD, Glycan-directed CAR-T cells, Glycobiology. 28 (2018) 656–669. doi: 10.1093/glycob/cwy008. [DOI] [PubMed] [Google Scholar]
- [46].Hossain MK, Wall KA, Immunological evaluation of recent MUC1 glycopeptide cancer vaccines, Vaccines. 4 (2016) 25. doi: 10.3390/vaccines4030025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Taylor-Papadimitriou J, Burchell JM, Graham R, Beatson R, Latest developments in MUC1 immunotherapy, Biochem. Soc. Trans 46 (2018) 659–668. doi: 10.1042/BST20170400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [48].Wilkie S, Picco G, Foster J, Davies DM, Julien S, Cooper L, Arif S, Mather SJ, Taylor-Papadimitriou J, Burchell JM, Maher J, Retargeting of human T cells to tumor-associated MUC1: the evolution of a chimeric antigen receptor, J. Immunol 180 (2008) 4901–4909. doi: 10.4049/jimmunol.180.7.4901. [DOI] [PubMed] [Google Scholar]
- [49].You F, Jiang L, Zhang B, Lu Q, Zhou Q, Liao X, Wu H, Du K, Zhu Y, Meng H, Gong Z, Zong Y, Huang L, Lu M, Tang J, Li Y, Zhai X, Wang X, Ye S, Chen D, Yuan L, Qi L, Yang L, Phase 1 clinical trial demonstrated that MUC1 positive metastatic seminal vesicle cancer can be effectively eradicated by modified Anti-MUC1 chimeric antigen receptor transduced T cells, Sci. China Life Sci 59 (2016) 386–397. doi: 10.1007/s11427-016-5024-7. [DOI] [PubMed] [Google Scholar]
- [50].Maher J, Wilkie S, CAR mechanics: Driving T cells into the MUC of cancer, Cancer Res. 69 (2009) 4559–4562. doi: 10.1158/0008-5472.CAN-09-0564. [DOI] [PubMed] [Google Scholar]
- [51].Gheybi E, Amani J, Salmanian AH, Mashayekhi F, Khodi S, Designing a recombinant chimeric construct contain MUC1 and HER2 extracellular domain for prediagnostic breast cancer, Tumor Biol. 35 (2014) 11489–11497. doi: 10.1007/s13277-014-2483-y. [DOI] [PubMed] [Google Scholar]
- [52].Sørensen AL, Reis CA, Tarp MA, Mandel U, Ramachandran K, Sankaranarayanan V, Schwientek T, Graham R, Taylor-Papadimitriou J, Hollingsworth MA, Burchell J, Clausen H, Chemoenzymatically synthesized multimeric Tn/STn MUC1 glycopeptides elicit cancer-specific anti-MUC1 antibody responses and override tolerance, Glycobiology. 16 (2006) 96–107. doi: 10.1093/glycob/cwj044. [DOI] [PubMed] [Google Scholar]
- [53].Posey AD, Schwab RD, Boesteanu AC, Steentoft C, Mandel U, Engels B, Stone JD, Madsen TD, Schreiber K, Haines KM, Cogdill AP, Chen TJ, Song D, Scholler J, Kranz DM, Feldman MD, Young R, Keith B, Schreiber H, Clausen H, Johnson LA, June CH, Engineered CAR T cells targeting the cancer-associated Tn-glycoform of the membrane mucin MUC1 control adenocarcinoma, Immunity. 44 (2016) 1444–1454. doi: 10.1016/j.immuni.2016.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [54].Zhou R, Yazdanifar M, Das Roy L, Whilding LM, Gavrill A, Maher J, Mukherjee P, CAR T cells targeting the tumor MUC1 glycoprotein reduce triple-negative breast cancer growth, Front. Immunol 10 (2019) 1–12. doi: 10.3389/fimmu.2019.01149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [55].Wei X, Lai Y, Li J, Qin L, Xu Y, Zhao R, Li B, Lin S, Wang S, Wu Q, Liang Q, Peng M, Yu F, Li Y, Zhang X, Wu Y, Liu P, Pei D, Yao Y, Li P, PSCA and MUC1 in non-small-cell lung cancer as targets of chimeric antigen receptor T cells, Oncoimmunology. 6 (2017) e1284722. doi: 10.1080/2162402X.2017.1284722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [56].Dotti G, Savoldo B, Brenner M, Fifteen years of gene therapy based on chimeric antigen receptors: Are we nearly there yet?, Hum. Gene Ther 20 (2009) 1229–1239. doi: 10.1089/hum.2009.142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [57].Büning H, Uckert W, Cichutek K, Hawkins RE, Abken H, Do CARs need a driver’s license? Adoptive cell therapy with chimeric antigen receptor-redirected T cells has caused serious adverse events, Hum. Gene Ther 21 (2010) 1039–1042. doi: 10.1089/hum.2010.131. [DOI] [PubMed] [Google Scholar]
- [58].Szymczak AL, Workman CJ, Wang Y, Vignali KM, Dilioglou S, Vanin EF, Vignali DAA, Correction of multi-gene deficiency in vivo using a single “self-cleaving” 2A peptide-based retroviral vector, Nat. Biotechnol 22 (2004) 589–594. doi: 10.1038/nbt957. [DOI] [PubMed] [Google Scholar]
- [59].Wilkie S, Van Schalkwyk MCI, Hobbs S, Davies DM, Van Der Stegen SJC, Pereira ACP, Burbridge SE, Box C, Eccles SA, Maher J, Dual targeting of ErbB2 and MUC1 in breast cancer using chimeric antigen receptors engineered to provide complementary signaling, J. Clin. Immunol 32 (2012) 1059–1070. doi: 10.1007/s10875-012-9689-9. [DOI] [PubMed] [Google Scholar]
- [60].Kloss CC, Condomines M, Cartellieri M, Bachmann M, Sadelain M, Combinatorial antigen recognition with balanced signaling promotes selective tumor eradication by engineered T cells, Nat. Biotechnol 31 (2013) 71–75. doi: 10.1038/nbt.2459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [61].Lanitis E, Poussin M, Klattenhoff AW, Song D, Sandaltzopoulos R, June CH, Powell DJ, Chimeric antigen receptor T Cells with dissociated signaling domains exhibit focused antitumor activity with reduced potential for toxicity in vivo, Cancer Immunol. Res 1 (2013) 43–53. doi: 10.1158/2326-6066.CIR-13-0008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [62].Fan G, Wang Z, Hao M, Li J, Bispecific antibodies and their applications, J. Hematol. Oncol 8 (2015) 130. doi: 10.1186/s13045-015-0227-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [63].Kontermann RE, Brinkmann U, Bispecific antibodies; different formats, Drug Discov. Today 20 (2015) 838–847. doi: 10.1016/j.drudis.2015.02.008. [DOI] [PubMed] [Google Scholar]
- [64].Bhutani D, Lum LG, Activated T cells armed with bispecific antibodies kill tumor targets, Curr. Opin. Hematol 22 (2015) 476–483. doi: 10.1097/MOH.0000000000000176. [DOI] [PubMed] [Google Scholar]
- [65].Husain B, Ellerman D, Expanding the Boundaries of Biotherapeutics with Bispecific Antibodies, BioDrugs. 32 (2018) 441–464. doi: 10.1007/s40259-018-0299-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [66].Holmes D, Buy buy bispecific antibodies, Nat. Rev. Drug Discov 10 (2011) 798–800. doi: 10.1038/nrd3581. [DOI] [PubMed] [Google Scholar]
- [67].Yankelevich M, Kondadasula SV, Thakur A, Buck S, Cheung NKV, Lum LG, Anti-CD3×anti-GD2 bispecific antibody redirects T-cell cytolytic activity to neuroblastoma targets, Pediatr. Blood Cancer 59 (2012) 1198–1205. doi: 10.1002/pbc.24237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [68].Xu H, Cheng M, Guo H, Chen Y, Huse M, Cheung NKV, Retargeting T cells to GD2 pentasaccharide on human tumors using bispecific humanized antibody, Cancer Immunol. Res 3 (2015) 266–277. doi: 10.1158/2326-6066.CIR-14-0230-T. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [69].Cheng M, Santich BH, Xu H, Ahmed M, Huse M, Cheung NKV, Successful engineering of a highly potent single-chain variable-fragment (scFv) bispecific antibody to target disialoganglioside (GD2) positive tumors, Oncoimmunology. 5 (2016) 1–9. doi: 10.1080/2162402X.2016.1168557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [70].Deppisch N, Ruf P, Eißler N, Lindhofer H, Mocikat R, Potent CD4+ T cell-associated antitumor memory responses induced by trifunctional bispecific antibodies in combination with immune checkpoint inhibition, Oncotarget. 8 (2017) 4520–4529. doi: 10.18632/oncotarget.13888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [71].Katayose Y, Kudo T, Suzuki M, Shinoda M, Saijyo S, Sakurai N, Saeki H, Fukuhara K, Imai K, Matsuno S, MUC1-specific targeting immunotherapy with bispecific antibodies: Inhibition of xenografted human bile duct carcinoma growth, Cancer Res. 56 (1996) 4205–4212. http://www.ncbi.nlm.nih.gov/pubmed/8797593 (accessed October 31, 2019). [PubMed] [Google Scholar]
- [72].Modak S, Le Luduec JB, Cheung IY, Goldman DA, Ostrovnaya I, Doubrovina E, Basu E, Kushner BH, Kramer K, Roberts SS, O’Reilly RJ, Cheung NKV, Hsu KC, Adoptive immunotherapy with haploidentical natural killer cells and Anti-GD2 monoclonal antibody m3F8 for resistant neuroblastoma: Results of a phase I study, Oncoimmunology. 7 (2018) e1461305. doi: 10.1080/2162402X.2018.1461305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [73].Tarek N, Le Luduec JB, Gallagher MM, Zheng J, Venstrom JM, Chamberlain E, Modak S, Heller G, Dupont B, Cheung NKV, Hsu KC, Unlicensed NK cells target neuroblastoma following anti-GD2 antibody treatment, J. Clin. Invest 122 (2012) 3260–3270. doi: 10.1172/JCI62749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [74].Schettini J, Kidiyoor A, Besmer DM, Tinder TL, Das Roy L, Lustgarten J, Gendler SJ, Mukherjee P, Intratumoral delivery of CpG-conjugated anti-MUC1 antibody enhances NK cell anti-tumor activity, Cancer Immunol. Immunother 61 (2012) 2055–2065. doi: 10.1007/s00262-012-1264-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [75].Li Y, Zhou C, Li J, Liu J, Lin L, Li L, Cao D, Li Q, Wang Z, Single domain based bispecific antibody, Muc1-Bi-1, and its humanized form, Muc1-Bi-2, induce potent cancer cell killing in muc1 positive tumor cells, PLoS One. 13 (2018) 1–14. doi: 10.1371/journal.pone.0191024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [76].Hoseini SS, Dobrenkov K, Pankov D, Xu XL, Cheung NKV, Bispecific antibody does not induce T-cell death mediated by chimeric antigen receptor against disialoganglioside GD2, Oncoimmunology. 6 (2017) e1320625. doi: 10.1080/2162402X.2017.1320625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [77].Alvarez-Rueda N, Desselle A, Cochonneau D, Chaumette T, Clemenceau B, Leprieur S, Bougras G, Supiot S, Mussini JM, Barbet J, Saba J, Paris F, Aubry J, Birklé S, A monoclonal antibody to O-Acetyl-GD2 ganglioside and not to GD2 shows potent anti-tumor activity without peripheral nervous system cross-reactivity, PLoS One. 6 (2011) e25220. doi: 10.1371/journal.pone.0025220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [78].Samraj AN, Läubli H, Varki N, Varki A, Involvement of a non-human sialic acid in human cancer, Front. Oncol 4 MAR (2014) 33. doi: 10.3389/fonc.2014.00033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [79].Inoue S, Sato C, Kitajima K, Extensive enrichment of N-glycolylneuraminic acid in extracellular sialoglycoproteins abundantly synthesized and secreted by human cancer cells, Glycobiology. 20 (2010) 752–762. doi: 10.1093/glycob/cwq030. [DOI] [PubMed] [Google Scholar]