In this Perspective, autopsy results and literature are presented supporting the hypothesis that neutrophil extracellular traps (NETs) may contribute to organ damage and mortality in COVID-19. If correct, existing drugs that target NETs, although unspecific, may benefit COVID-19 patients.
Abstract
Coronavirus disease 2019 (COVID-19) is a novel, viral-induced respiratory disease that in ∼10–15% of patients progresses to acute respiratory distress syndrome (ARDS) triggered by a cytokine storm. In this Perspective, autopsy results and literature are presented supporting the hypothesis that a little known yet powerful function of neutrophils—the ability to form neutrophil extracellular traps (NETs)—may contribute to organ damage and mortality in COVID-19. We show lung infiltration of neutrophils in an autopsy specimen from a patient who succumbed to COVID-19. We discuss prior reports linking aberrant NET formation to pulmonary diseases, thrombosis, mucous secretions in the airways, and cytokine production. If our hypothesis is correct, targeting NETs directly and/or indirectly with existing drugs may reduce the clinical severity of COVID-19.
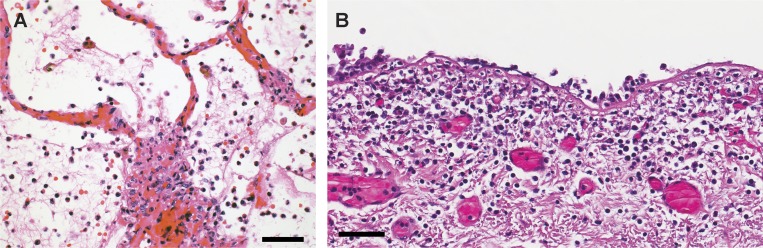
Patients with severe coronavirus disease 2019 (COVID-19)–associated pneumonitis and/or acute respiratory distress syndrome (ARDS) have increased pulmonary inflammation, thick mucous secretions in the airways, elevated levels of serum pro-inflammatory cytokines, extensive lung damage, and microthrombosis. This late stage of the disease is difficult to manage, and a large number of patients die (Chen et al., 2020a Preprint; Wang et al., 2020; Zhao et al., 2020 Preprint; Zheng et al., 2020). The severity of COVID-19, combined with its pandemic spread, has placed unprecedented pressure on our healthcare system, and treatment strategies are urgently needed. Infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causes COVID-19, but it is an exacerbated and poorly understood host response involving a cytokine storm that drives severe COVID-19 (Mehta et al., 2020). It is unclear what initiates and propagates the cytokine storm. We propose that the exacerbated host response in patients with severe COVID-19 centers around the aberrant activation of the most common leukocyte in peripheral blood: the neutrophil. Neutrophilia predicts poor outcomes in patients with COVID-19 (Wang et al., 2020), and the neutrophil-to-lymphocyte ratio is an independent risk factor for severe disease (Liu et al., 2020 Preprint). Furthermore, in autopsy samples from the lungs of three COVID-19 patients at Weill Cornell Medicine, we observed neutrophil infiltration in pulmonary capillaries, acute capillaritis with fibrin deposition, extravasation of neutrophils into the alveolar space, and neutrophilic mucositis (Fig. 1). Neutrophil infiltration was also noted in two recent reports on the pathological findings from autopsied COVID-19 patients (Fox et al., 2020 Preprint; Yao et al., 2020). Although leukocytosis and neutrophilia are hallmarks of acute infection, in the case of COVID-19, we propose that neutrophilia could also be a source of excess neutrophil extracellular traps (NETs).
Figure 1.
Neutrophils in an autopsy specimen from the lungs of a patient who succumbed from COVID-19. (A) Extensive neutrophil infiltration in pulmonary capillaries, with acute capillaritis with fibrin deposition, and extravasation into the alveolar space. An image was chosen to emphasize the capillary lesions. (B) Neutrophilic mucositis of the trachea. The entire airway was affected (images by A. Borczuk, Weill Cornell Medical Center). Both specimens originate from a 64-yr-old male of Hispanic decent with diabetes, end-stage renal disease on hemodialysis, heart failure, and hepatitis C on ledipasvir/sofosbuvir therapy. He declined medical intervention, was therefore not intubated, and died in the emergency room 5 h after presentation, shortly after developing fever. There was no evidence of sepsis in this patient clinically, premortem cultures were negative, and the autopsy was performed within 5 h of death. Similar neutrophil distribution, but with less extensive infiltration, was observed in the two additional autopsies analyzed to date. These other two cases had longer duration of symptoms. Scale bars: 50 µm.
NETs and disease
Neutrophils are recruited early to sites of infection where they kill pathogens (bacteria, fungi, and viruses) by oxidative burst and phagocytosis (Schönrich and Raftery, 2016). However, neutrophils have another much less recognized means of killing pathogens: the formation of NETs (Brinkmann et al., 2004). NETs are web-like structures of DNA and proteins expelled from the neutrophil that ensnare pathogens (Fig. 2). Expelling DNA to the extracellular space is not widely recognized as a critical immune function. Yet, even plants have specialized cells that kill soil pathogens by this mechanism (Wen et al., 2009). NET formation is a regulated process, although the signals involved are incompletely understood. Key enzymes in the formation of NETs are: neutrophil elastase (NE), which degrades intracellular proteins and triggers nuclear disintegration; peptidyl arginine deiminase type 4 (PAD4), which citrullinates histones to facilitate the decondensation and release of the chromosomal DNA; and gasdermin D, which generates pores in the membrane of the neutrophil, thereby facilitating cell membrane rupture and the expulsion of DNA and the associated molecules (Chen et al., 2018; Kaplan and Radic, 2012; Papayannopoulos, 2018; Papayannopoulos et al., 2010; Rohrbach et al., 2012; Sollberger et al., 2018). Although NETs are beneficial in the host defense against pathogens, collateral damage from sustained NET formation also stimulates many disease processes, including those that occur during viral infections (Schönrich and Raftery, 2016). Indeed, excessive NET formation can trigger a cascade of inflammatory reactions that promotes cancer cell metastasis, destroys surrounding tissues, facilitates microthrombosis, and results in permanent organ damage to the pulmonary, cardiovascular, and renal systems (Jorch and Kubes, 2017; Kessenbrock et al., 2009; Papayannopoulos, 2018; Fig. 3). Importantly, these are three commonly affected organ systems in severe COVID-19 (Bonow et al., 2020; Chen et al., 2020b).
Figure 2.
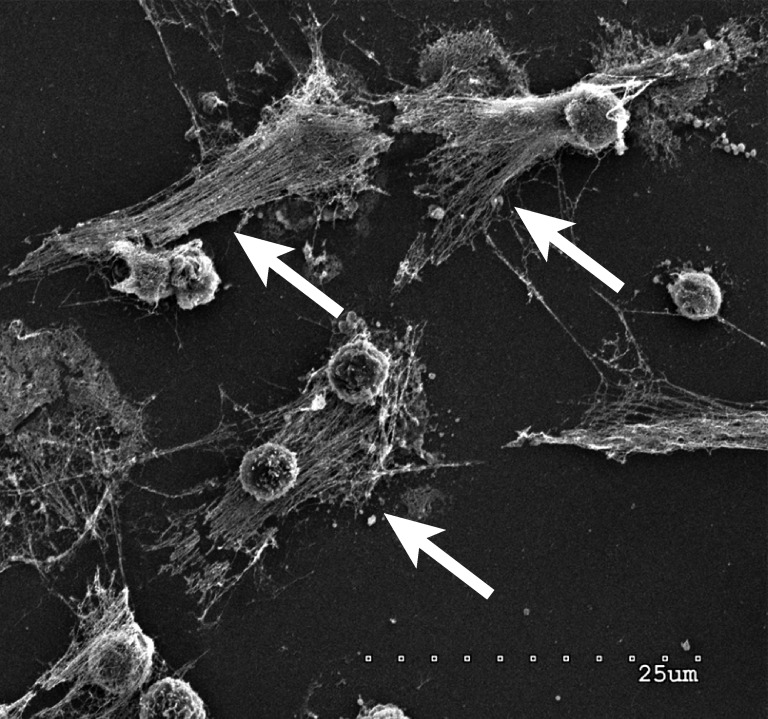
Neutrophils forming NETs in cell culture. Note the expelled DNA strings (arrows). Scanning electron microscopy of neutrophils 3 h after plating and coculturing with 4T1 breast cancer cells. Scale bar: 25 µm.
Figure 3.
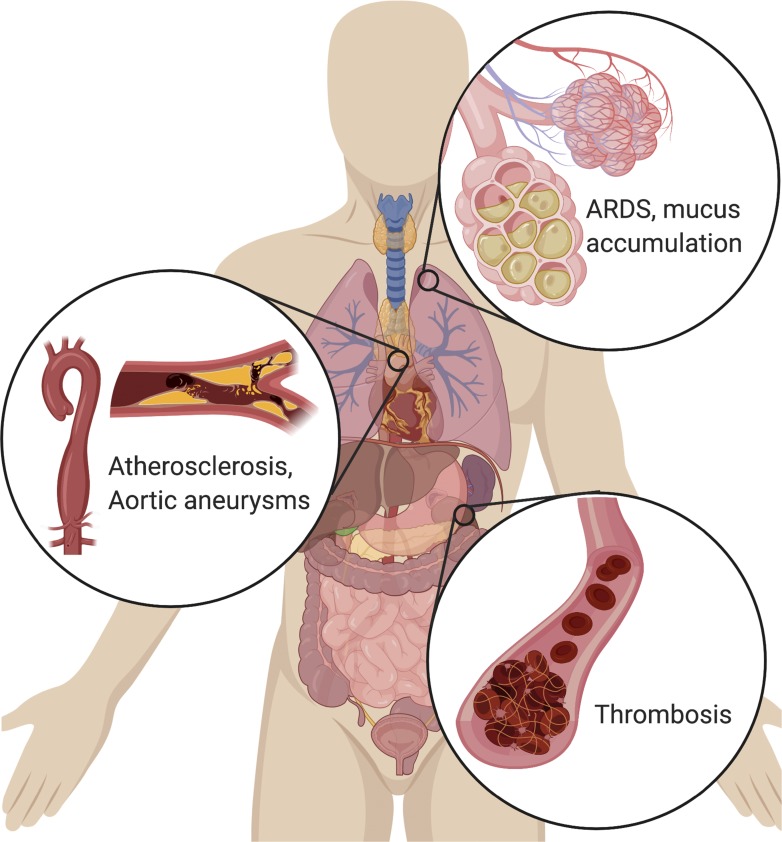
Excess NET formation can drive a variety of severe pathologies. In the lungs, NETs drive the accumulation of mucus in CF patients’ airways. NETs also drive ARDS after a variety of inducers, including influenza. In the vascular system, NETs drive atherosclerosis and aortic aneurysms, as well as thrombosis (particularly microthrombosis), with devastating effects on organ function.
NETs and ARDS
Prior reports extensively link aberrant NET formation to pulmonary diseases, particularly ARDS. Indeed, NET levels in plasma are higher in patients with transfusion-associated ARDS than in subjects without ARDS (Caudrillier et al., 2012). Furthermore, neutrophils from patients with pneumonia-associated ARDS appear “primed” to form NETs, and both the extent of priming and the level of NETs in blood correlate with disease severity and mortality (Adrover et al., 2020; Bendib et al., 2019; Ebrahimi et al., 2018; Lefrançais et al., 2018; Mikacenic et al., 2018). Extracellular histones, likely partly originating from NETs, are elevated in the bronchoalveolar lavage fluid and plasma of ARDS patients (Lv et al., 2017). Naked histones are toxic to cells, and there is strong experimental evidence supporting a role for histones in ARDS and sepsis (Wygrecka et al., 2017; Xu et al., 2015). It is therefore likely that NETs, as a source of extracellular histones, contribute to ARDS and sepsis (Chaput and Zychlinsky, 2009; Lefrançais and Looney, 2017; Xu et al., 2009). In animal models of lung injury, NETs develop in response to a variety of ARDS-inducing stimuli, and preventing or dissolving NETs reduces lung injury and increases survival (Caudrillier et al., 2012; Lefrançais et al., 2018; Liu et al., 2016; Narasaraju et al., 2011).
NETs and cystic fibrosis (CF)
The mucous secretions found in the airways of COVID-19 patients (Mao et al., 2020 Preprint) are reminiscent of those seen in CF patients (Martínez-Alemán et al., 2017). The cause and origin of these secretions are unclear. However, in CF, mucous secretions impair gas exchange and have been shown to contain extracellular DNA, in part originating from NETs released in response to persistent lung infections. Furthermore, the excessive formation of NETs with increased NE makes the mucus thick and viscous (Manzenreiter et al., 2012), not only impairing ventilation but also facilitating the colonization of bacteria. Such colonization further promotes neutrophil recruitment and NET formation, increasing mucus viscosity and consequently lowering the patient’s respiratory function. If the mucous secretions in COVID-19 contain NETs, they may play similar roles as they do in CF: impairing gas exchange and facilitating secondary infections.
NETs and excessive thrombosis
Acute cardiac and kidney injuries are common in patients with severe COVID-19 and contribute to the mortality of this disease (Bonow et al., 2020). D-dimer (a fibrin degradation product indicative of hyperactive coagulation) has emerged as a reliable marker of severe COVID-19 (Zhou et al., 2020). High blood levels of NETs may explain these findings: intravascular NETs have been shown to play a vital role in initiating and accreting thrombosis in arteries and veins (Fuchs et al., 2012). For example, in severe coronary artery disease, complexes of NETs are elevated, and NET levels positively associate with thrombin levels, which predict adverse cardiac events (Borissoff et al., 2013). In addition, autopsy samples collected from septic patients show that NETs infiltrate microthrombi (Jiménez-Alcázar et al., 2017). Thus, when NETs circulate at high levels in blood, they can trigger the occlusion of small vessels, leading to damage in the lungs, heart, and kidneys (Cedervall et al., 2015; Fuchs et al., 2010; Laridan et al., 2019; Martinod and Wagner, 2014). In mouse models of septicemia, intravascular NETs form microthrombi that obstruct blood vessels and cause damage to the lungs, liver, and other organs (Jiménez-Alcázar et al., 2017). Mechanistically, NETs activate the contact pathway of coagulation (also called the plasma kallikrein–kinin system) via electrostatic interactions between the NET histones and platelet phospholipids (Oehmcke et al., 2009). Histones can also promote platelet activation by acting as ligands for the Toll-like receptors on platelets (Semeraro et al., 2011). At the same time, NE (which is bound in its active form to NETs) likely also plays an important role by digesting the major coagulation inhibitors antithrombin III and tissue factor pathway inhibitor (Massberg et al., 2010). Furthermore, there is almost surely a feedback loop whereby pro-coagulant activity (e.g., that of thrombin) leads to platelet activation, and activated platelets then further enhance NET formation (Caudrillier et al., 2012; Clark et al., 2007; Fuchs et al., 2010; Massberg et al., 2010; Sreeramkumar et al., 2014; von Brühl et al., 2012). Dissolving NETs with DNase I restores normal perfusion of the heart and kidney microvasculature in animal models (Cedervall et al., 2015; Jansen et al., 2017; Nakazawa et al., 2017; Raup-Konsavage et al., 2018). Based on the above findings, we argue that targeting intravascular NETs may similarly reduce thrombosis in patients with severe COVID-19.
NETs and the cytokine storm
Severe COVID-19 is associated with a cytokine storm characterized by increased plasma concentrations of IL1β, IL2, IL6, IL7, IL8, IL10, IL17, IFNγ, IFNγ-inducible protein 10, monocyte chemoattractant protein 1 (MCP1), G-CSF, macrophage inflammatory protein 1α, and TNFα (Huang et al., 2020; Mehta et al., 2020; Ruan et al., 2020; Wu et al., 2020; Wu and Yang, 2020; Zhang et al., 2020). These inflammatory mediators regulate neutrophil activity and induce the expression of chemoattractants (molecules that increase the trafficking of neutrophils to sites of inflammation). Moreover, cytokine storms lead to acute lung injury, ARDS, and death (Channappanavar and Perlman, 2017; Chousterman et al., 2017). It is especially noteworthy that NETs can induce macrophages to secrete IL1β and that IL1β enhances NET formation in various diseases, including aortic aneurysms and atherosclerosis (Kahlenberg et al., 2013; Meher et al., 2018; Sil et al., 2017; Warnatsch et al., 2015). Together, these data suggest that under conditions in which the normal signals to dampen inflammation are lost, such as during a cytokine storm, a signaling loop between macrophages and neutrophils can lead to uncontrollable, progressive inflammation. Indeed, a correlation between NETs and IL1β exists in severe asthma (Lachowicz-Scroggins et al., 2019). If a NET–IL1β loop is activated in severe COVID-19, the accelerated production of NETs and IL1β could accelerate respiratory decompensation, the formation of microthrombi, and aberrant immune responses. Importantly, IL1β induces IL6 (Dinarello, 2009), and IL6 has emerged as a promising target for COVID-19 treatment (Mehta et al., 2020; Xu et al., 2020 Preprint). IL6 can signal via classic and trans-signaling (Calabrese and Rose-John, 2014). In classic signaling, IL6 binds to a complex of the transmembrane receptor IL6Rα with the common cytokine receptor gp130. In trans-signaling, soluble IL6Rα (sIL6Rα) binds IL6 to initiate signaling via gp130. Trans-signaling is strongly associated with pro-inflammatory states (Calabrese and Rose-John, 2014), and lower levels of sIL6Rα are associated with better lung function in, e.g., asthma (Ferreira et al., 2013; Hawkins et al., 2012). Neutrophils can shed sIL6Rα in response to IL8 (Marin et al., 2002), which is abundant in the COVID-19–associated cytokine storm (Wu and Yang, 2020; Zhang et al., 2020). Together, these findings lead us to speculate that antagonizing IL-6 trans-signaling and/or IL1β could be effective indirect strategies for targeting neutrophils and NETs in severe COVID-19.
NET therapeutics
Due to the clear similarities between the clinical presentation of severe COVID-19 and known NETopathies—ARDS and microthrombosis—we propose that excess NETs may play a major role in the disease. Our understanding of NET formation and function is incomplete, but drugs that target NETs exist or are in development (Fig. 4). These drugs include inhibitors of the molecules required for NET formation: NE, PAD4, and gasdermin D. For example, endogenous inhibitors of NET formation, which may function by inhibiting PAD4, have been isolated from umbilical cord plasma (Yost et al., 2016), and these are in development for the treatment of inflammatory syndromes such as COVID-19. Clinical development of inhibitors against NE is the most advanced, and importantly, they could inhibit both the formation of NETs, where NE activity is part of the signaling mechanism, and the toxic activities of NE on the NETs. The NE inhibitor sivelestat was approved to treat ARDS in Japan and South Korea, but it did not increase survival after ARDS in a meta-analysis of clinical trials (Tagami et al., 2014). However, a new generation of potent NE inhibitors, including lonodelestat (POL6014), alvelestat, CHF6333, and elafin, have undergone Phase I testing. As a result, it may be possible to expedite their development as treatments for COVID-19. Gasdermin D inhibitors remain in preclinical development, but an existing drug—disulfiram, used to treat alcoholism—has been reported to inhibit gasdermin D and limit lung injury in animal models (Hu et al., 2018 Preprint). Finally, colchicine is another existing drug that could inhibit both neutrophil recruitment to sites of inflammation and the secretion of IL1β, and trials using colchicine in COVID-19 are underway (ClinicalTrials.gov identifiers: NCT04326790, NCT04328480, NCT04322565, NCT04322682).
Figure 4.
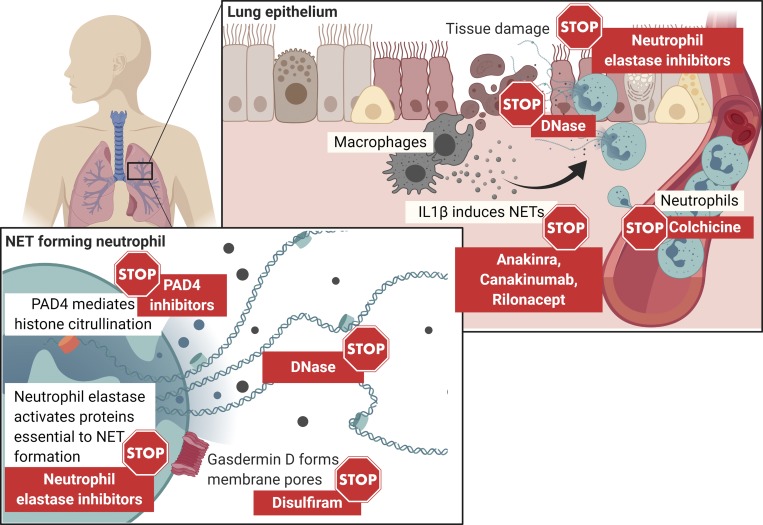
Approaches to targeting NETs. NETs can be targeted by existing drugs through several means. NE, PAD4, and gasdermin D inhibitors will prevent NET formation. DNase has been used safely to digest NETs in the mucous secretions of the airways of CF patients. Colchicine inhibits neutrophil migration and infiltration into sites of inflammation. IL1β blockers will prevent an inflammatory loop between NETs and IL1β. Of these approaches, trials to treat COVID-19 with colchicine and anakinra are already ongoing or being launched (ClinicalTrials.gov identifiers: NCT04324021, NCT04330638, NCT02735707, NCT04326790, NCT04328480, NCT04322565, NCT04322682).
A recombinant DNase I (dornase alfa), delivered by inhalation, is approved to dissolve NETs in the airways of patients with CF to clear mucus and improve symptoms (Papayannopoulos et al., 2011). Additionally, an actin-resistant DNase (PRX-110/alidornase alfa) has been tested in CF patients in Phase I and II trials with encouraging results (ClinicalTrials.gov identifiers: NCT02605590, NCT02722122) and could potentially be more potent than dornase alfa. Other engineered DNase proteins, such as DNase 1–like 3, which is being developed to dissolve NETs (Fuchs et al., 2019), could enter clinical development soon. We propose that DNases may help dissolve the mucous secretions of COVID-19 patients as they do in CF patients, improving ventilation and reducing the risk of secondary infections. Dornase alfa is normally administered through nebulizers, but in many medical centers, these are avoided in COVID-19 due to the risk of aerosolizing SARS-CoV-2 and endangering healthcare workers. However, approaches exist that deliver aerosols in closed circuits for mechanically ventilated patients (Dhand, 2017). For nonintubated patients, therapies can be safely nebulized in negative pressure rooms. In addition to their possible effects on mucous secretions, DNase treatments may also prevent the further progression to ARDS, as DNase I delivered through the airways increases survival in relevant animal models (Lefrançais et al., 2018; Thomas et al., 2012; Zou et al., 2018).
In addition to directly targeting NETs, the NET–IL1β loop could be antagonized with approved drugs against IL1β, such as anakinra, canakinumab, and rilonacept. Trials are now being launched to test the efficacy of anakinra in COVID-19 (ClinicalTrials.gov identifiers: NCT04324021, NCT04330638, NCT02735707). Dornase alfa, sivelestat, and anakinra have excellent safety profiles. None of the drugs that currently are available to target NETs are specific. Nevertheless, collectively, there are multiple individual or combinatorial—and likely safe—therapeutic strategies available to antagonize NETs in COVID-19 patients today, and NETs themselves may be an appropriate biomarker to follow studies to test their efficacy.
Opportunities to target NETs in COVID-19
NETs can be detected in tissues by immunohistochemistry and in blood by sandwich ELISA (Caudrillier et al., 2012; Jiménez-Alcázar et al., 2017; Lachowicz-Scroggins et al., 2019; Park et al., 2016). As samples from patients become available, it will therefore be possible to determine whether the presence of NETs is associated with severe COVID-19. If it is, this result would provide rationale for using the above-mentioned NET-targeting approaches in the treatment for COVID-19. Though treatments targeting NETs would not directly target the SARS-CoV-2 virus, they could dampen the out-of-control host response, thereby reducing the number of patients who need invasive mechanical ventilation, and importantly, reducing mortality. NETs were identified in 2004 (Brinkmann et al., 2004), and they are often overlooked as drivers of severe pathogenic inflammation. Indeed, we posit here that excess NETs may elicit the severe multi-organ consequences of COVID-19 via their known effects on tissues and the immune, vascular, and coagulation systems. Targeting NETs in COVID-19 patients should therefore be considered by the biomedical community.
Acknowledgments
The authors, members of “The NETwork to Target Neutrophils in COVID-19,” thank Andrew Whiteley for organizing the discussions that are summarized in this paper; Laura Maiorino for generating the illustrations; Laura Maiorino and Stephen Hearn for the photomicrograph of NETs; and Bruce Stillman and David Tuveson for discussions and critical review of the manuscript. We acknowledge the Cold Spring Harbor Laboratory CCSG P30CA45508 and Microscopy shared resource. BioRender was used to generate the illustrations in the figures. We apologize to those with relevant work that was not included in the discussion due to space limitations.
Author contributions: B.J. Barnes and M. Egeblad outlined the manuscript. A. Baxter-Stoltzfus, A. Borczuk, and S. Salvatore performed the COVID autopsies. A. Baxter-Stoltzfus, A. Borczuk, S. Salvatore, and M. Loda reviewed the autopsy pathology. B.J. Barnes, J.M. Adrover, A. Baxter-Stoltzfus, A. Borczuk, J. Cools-Lartigue, J.M. Crawford, J. Daßler-Plenker, P. Guerci, C. Huynh, J.S. Knight, M. Loda, M.R. Looney, F. McAllister, R. Rayes, S. Renaud, S. Rousseau, S. Salvatore, R.E. Schwartz, J.D. Spicer, C.C. Yost, A. Weber, Y. Zuo, and M. Egeblad wrote and/or edited the manuscript.
References
- Adrover J.M., Aroca-Crevillén A., Crainiciuc G., Ostos F., Rojas-Vega Y., Rubio-Ponce A., Cilloniz C., Bonzón-Kulichenko E., Calvo E., Rico D., et al. 2020. Programmed ‘disarming’ of the neutrophil proteome reduces the magnitude of inflammation. Nat. Immunol. 21:135–144. 10.1038/s41590-019-0571-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bendib I., de Chaisemartin L., Granger V., Schlemmer F., Maitre B., Hüe S., Surenaud M., Beldi-Ferchiou A., Carteaux G., Razazi K., et al. 2019. Neutrophil Extracellular Traps Are Elevated in Patients with Pneumonia-related Acute Respiratory Distress Syndrome. Anesthesiology. 130:581–591. 10.1097/ALN.0000000000002619 [DOI] [PubMed] [Google Scholar]
- Bonow R.O., Fonarow G.C., O’Gara P.T., and Yancy C.W.. 2020. Association of Coronavirus Disease 2019 (COVID-19) With Myocardial Injury and Mortality. JAMA Cardiol. 10.1001/jamacardio.2020.1105 [DOI] [PubMed] [Google Scholar]
- Borissoff J.I., Joosen I.A., Versteylen M.O., Brill A., Fuchs T.A., Savchenko A.S., Gallant M., Martinod K., Ten Cate H., Hofstra L., et al. 2013. Elevated levels of circulating DNA and chromatin are independently associated with severe coronary atherosclerosis and a prothrombotic state. Arterioscler. Thromb. Vasc. Biol. 33:2032–2040. 10.1161/ATVBAHA.113.301627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brinkmann V., Reichard U., Goosmann C., Fauler B., Uhlemann Y., Weiss D.S., Weinrauch Y., and Zychlinsky A.. 2004. Neutrophil extracellular traps kill bacteria. Science. 303:1532–1535. 10.1126/science.1092385 [DOI] [PubMed] [Google Scholar]
- Calabrese L.H., and Rose-John S.. 2014. IL-6 biology: implications for clinical targeting in rheumatic disease. Nat. Rev. Rheumatol. 10:720–727. 10.1038/nrrheum.2014.127 [DOI] [PubMed] [Google Scholar]
- Caudrillier A., Kessenbrock K., Gilliss B.M., Nguyen J.X., Marques M.B., Monestier M., Toy P., Werb Z., and Looney M.R.. 2012. Platelets induce neutrophil extracellular traps in transfusion-related acute lung injury. J. Clin. Invest. 122:2661–2671. 10.1172/JCI61303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cedervall J., Zhang Y., Huang H., Zhang L., Femel J., Dimberg A., and Olsson A.K.. 2015. Neutrophil Extracellular Traps Accumulate in Peripheral Blood Vessels and Compromise Organ Function in Tumor-Bearing Animals. Cancer Res. 75:2653–2662. 10.1158/0008-5472.CAN-14-3299 [DOI] [PubMed] [Google Scholar]
- Channappanavar R., and Perlman S.. 2017. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Semin. Immunopathol. 39:529–539. 10.1007/s00281-017-0629-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaput C., and Zychlinsky A.. 2009. Sepsis: the dark side of histones. Nat. Med. 15:1245–1246. 10.1038/nm1109-1245 [DOI] [PubMed] [Google Scholar]
- Chen G., Wu D., Guo W., Cao Y., Huang D., Wang H., Wang T., Zhang X., Chen H., Yu H., et al. 2020a Clinical and immunologic features in severe and moderate forms of Coronavirus Disease 2019. MedRxiv. 10.1101/2020.02.16.20023903 (Preprint posted February 19, 2020) [DOI]
- Chen K.W., Monteleone M., Boucher D., Sollberger G., Ramnath D., Condon N.D., von Pein J.B., Broz P., Sweet M.J., and Schroder K.. 2018. Noncanonical inflammasome signaling elicits gasdermin D-dependent neutrophil extracellular traps. Sci. Immunol. 3:eaar6676 10.1126/sciimmunol.aar6676 [DOI] [PubMed] [Google Scholar]
- Chen N., Zhou M., Dong X., Qu J., Gong F., Han Y., Qiu Y., Wang J., Liu Y., Wei Y., et al. 2020b Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 395:507–513. 10.1016/S0140-6736(20)30211-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chousterman B.G., Swirski F.K., and Weber G.F.. 2017. Cytokine storm and sepsis disease pathogenesis. Semin. Immunopathol. 39:517–528. 10.1007/s00281-017-0639-8 [DOI] [PubMed] [Google Scholar]
- Clark S.R., Ma A.C., Tavener S.A., McDonald B., Goodarzi Z., Kelly M.M., Patel K.D., Chakrabarti S., McAvoy E., Sinclair G.D., et al. 2007. Platelet TLR4 activates neutrophil extracellular traps to ensnare bacteria in septic blood. Nat. Med. 13:463–469. 10.1038/nm1565 [DOI] [PubMed] [Google Scholar]
- Dhand R. 2017. How Should Aerosols Be Delivered During Invasive Mechanical Ventilation? Respir. Care. 62:1343–1367. 10.4187/respcare.05803 [DOI] [PubMed] [Google Scholar]
- Dinarello C.A. 2009. Targeting the pathogenic role of interleukin 1beta in the progression of smoldering/indolent myeloma to active disease. Mayo Clin. Proc. 84:105–107. 10.4065/84.2.105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ebrahimi F., Giaglis S., Hahn S., Blum C.A., Baumgartner C., Kutz A., van Breda S.V., Mueller B., Schuetz P., Christ-Crain M., and Hasler P.. 2018. Markers of neutrophil extracellular traps predict adverse outcome in community-acquired pneumonia: secondary analysis of a randomised controlled trial. Eur. Respir. J. 51:1701389 10.1183/13993003.01389-2017 [DOI] [PubMed] [Google Scholar]
- Ferreira R.C., Freitag D.F., Cutler A.J., Howson J.M., Rainbow D.B., Smyth D.J., Kaptoge S., Clarke P., Boreham C., Coulson R.M., et al. 2013. Functional IL6R 358Ala allele impairs classical IL-6 receptor signaling and influences risk of diverse inflammatory diseases. PLoS Genet. 9:e1003444 10.1371/journal.pgen.1003444 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox S.E., Akmatbekov A., Harbert J.L., Li G., Brown J.Q., and Vander Heide R.S.. 2020. Pulmonary and Cardiac Pathology in Covid-19: The First Autopsy Series from New Orleans. MedRxiv 10.1101/2020.04.06.20050575 (Preprint posted April 10, 2020). [DOI]
- Fuchs T.A., Brill A., Duerschmied D., Schatzberg D., Monestier M., Myers D.D. Jr., Wrobleski S.K., Wakefield T.W., Hartwig J.H., and Wagner D.D.. 2010. Extracellular DNA traps promote thrombosis. Proc. Natl. Acad. Sci. USA. 107:15880–15885. 10.1073/pnas.1005743107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuchs T.A., Brill A., and Wagner D.D.. 2012. Neutrophil extracellular trap (NET) impact on deep vein thrombosis. Arterioscler. Thromb. Vasc. Biol. 32:1777–1783. 10.1161/ATVBAHA.111.242859 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuchs T.A., Jiménez-Alcázar M., Göbel J., and Englert H.. 2019. Neutrolis, Inc. US patent application US 2020/0024585 A1, filed August 20, 2019.
- Hawkins G.A., Robinson M.B., Hastie A.T., Li X., Li H., Moore W.C., Howard T.D., Busse W.W., Erzurum S.C., Wenzel S.E., et al. National Heart, Lung, and Blood Institute–sponsored Severe Asthma Research Program (SARP) . 2012. The IL6R variation Asp(358)Ala is a potential modifier of lung function in subjects with asthma. J. Allergy Clin. Immunol. 130:510–5.e1. 10.1016/j.jaci.2012.03.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu J.J., Liu X., Zhao J., Xia S., Ruan J., Luo X., Kim J., Lieberman J., and Wu H.. 2018. Identification of pyroptosis inhibitors that target a reactive cysteine in gasdermin D. bioRxiv 10.1101/365908 (Preprint posted July 10, 2018) [DOI]
- Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X., et al. 2020. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 395:497–506. 10.1016/S0140-6736(20)30183-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jansen M.P., Emal D., Teske G.J., Dessing M.C., Florquin S., and Roelofs J.J.. 2017. Release of extracellular DNA influences renal ischemia reperfusion injury by platelet activation and formation of neutrophil extracellular traps. Kidney Int. 91:352–364. 10.1016/j.kint.2016.08.006 [DOI] [PubMed] [Google Scholar]
- Jiménez-Alcázar M., Rangaswamy C., Panda R., Bitterling J., Simsek Y.J., Long A.T., Bilyy R., Krenn V., Renné C., Renné T., et al. 2017. Host DNases prevent vascular occlusion by neutrophil extracellular traps. Science. 358:1202–1206. 10.1126/science.aam8897 [DOI] [PubMed] [Google Scholar]
- Jorch S.K., and Kubes P.. 2017. An emerging role for neutrophil extracellular traps in noninfectious disease. Nat. Med. 23:279–287. 10.1038/nm.4294 [DOI] [PubMed] [Google Scholar]
- Kahlenberg J.M., Carmona-Rivera C., Smith C.K., and Kaplan M.J.. 2013. Neutrophil extracellular trap-associated protein activation of the NLRP3 inflammasome is enhanced in lupus macrophages. J. Immunol. 190:1217–1226. 10.4049/jimmunol.1202388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaplan M.J., and Radic M.. 2012. Neutrophil extracellular traps: double-edged swords of innate immunity. J. Immunol. 189:2689–2695. 10.4049/jimmunol.1201719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kessenbrock K., Krumbholz M., Schönermarck U., Back W., Gross W.L., Werb Z., Gröne H.J., Brinkmann V., and Jenne D.E.. 2009. Netting neutrophils in autoimmune small-vessel vasculitis. Nat. Med. 15:623–625. 10.1038/nm.1959 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lachowicz-Scroggins M.E., Dunican E.M., Charbit A.R., Raymond W., Looney M.R., Peters M.C., Gordon E.D., Woodruff P.G., Lefrançais E., Phillips B.R., et al. 2019. Extracellular DNA, Neutrophil Extracellular Traps, and Inflammasome Activation in Severe Asthma. Am. J. Respir. Crit. Care Med. 199:1076–1085. 10.1164/rccm.201810-1869OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laridan E., Martinod K., and De Meyer S.F.. 2019. Neutrophil Extracellular Traps in Arterial and Venous Thrombosis. Semin. Thromb. Hemost. 45:86–93. 10.1055/s-0038-1677040 [DOI] [PubMed] [Google Scholar]
- Lefrançais E., and Looney M.R.. 2017. Neutralizing Extracellular Histones in Acute Respiratory Distress Syndrome. A New Role for an Endogenous Pathway. Am. J. Respir. Crit. Care Med. 196:122–124. 10.1164/rccm.201701-0095ED [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lefrançais E., Mallavia B., Zhuo H., Calfee C.S., and Looney M.R.. 2018. Maladaptive role of neutrophil extracellular traps in pathogen-induced lung injury. JCI Insight. 3:e98178 10.1172/jci.insight.98178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J., Liu Y., Xiang P., Pu L., Xiong H., Li C., Zhang M., Tan J., Xu Y., Song R., et al. 2020. Neutrophil-to-Lymphocyte Ratio Predicts Severe Illness Patients with 2019 Novel Coronavirus in the Early Stage. MedRxiv 10.1101/2020.02.10.20021584 (Preprint posted February 12, 2020) [DOI] [PMC free article] [PubMed]
- Liu S., Su X., Pan P., Zhang L., Hu Y., Tan H., Wu D., Liu B., Li H., Li H., et al. 2016. Neutrophil extracellular traps are indirectly triggered by lipopolysaccharide and contribute to acute lung injury. Sci. Rep. 6:37252 10.1038/srep37252 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lv X., Wen T., Song J., Xie D., Wu L., Jiang X., Jiang P., and Wen Z.. 2017. Extracellular histones are clinically relevant mediators in the pathogenesis of acute respiratory distress syndrome. Respir. Res. 18:165 10.1186/s12931-017-0651-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manzenreiter R., Kienberger F., Marcos V., Schilcher K., Krautgartner W.D., Obermayer A., Huml M., Stoiber W., Hector A., Griese M., et al. 2012. Ultrastructural characterization of cystic fibrosis sputum using atomic force and scanning electron microscopy. J. Cyst. Fibros. 11:84–92. 10.1016/j.jcf.2011.09.008 [DOI] [PubMed] [Google Scholar]
- Mao Y., Lin W., Wen J., and Chen G.. 2020. Clinical and pathological characteristics of 2019 novel coronavirus disease (COVID-19): a systematic reviews. MedRxiv 10.1101/2020.02.20.20025601 (Preprint posted March 19, 2020) [DOI]
- Marin V., Montero-Julian F., Grès S., Bongrand P., Farnarier C., and Kaplanski G.. 2002. Chemotactic agents induce IL-6Ralpha shedding from polymorphonuclear cells: involvement of a metalloproteinase of the TNF-alpha-converting enzyme (TACE) type. Eur. J. Immunol. 32:2965–2970. [DOI] [PubMed] [Google Scholar]
- Martínez-Alemán S.R., Campos-García L., Palma-Nicolas J.P., Hernández-Bello R., González G.M., and Sánchez-González A.. 2017. Understanding the Entanglement: Neutrophil Extracellular Traps (NETs) in Cystic Fibrosis. Front. Cell. Infect. Microbiol. 7:104 10.3389/fcimb.2017.00104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinod K., and Wagner D.D.. 2014. Thrombosis: tangled up in NETs. Blood. 123:2768–2776. 10.1182/blood-2013-10-463646 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Massberg S., Grahl L., von Bruehl M.L., Manukyan D., Pfeiler S., Goosmann C., Brinkmann V., Lorenz M., Bidzhekov K., Khandagale A.B., et al. 2010. Reciprocal coupling of coagulation and innate immunity via neutrophil serine proteases. Nat. Med. 16:887–896. 10.1038/nm.2184 [DOI] [PubMed] [Google Scholar]
- Meher A.K., Spinosa M., Davis J.P., Pope N., Laubach V.E., Su G., Serbulea V., Leitinger N., Ailawadi G., and Upchurch G.R. Jr. 2018. Novel Role of IL (Interleukin)-1β in Neutrophil Extracellular Trap Formation and Abdominal Aortic Aneurysms. Arterioscler. Thromb. Vasc. Biol. 38:843–853. 10.1161/ATVBAHA.117.309897 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., and Manson J.J.. HLH Across Speciality Collaboration, UK . 2020. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 395:1033–1034. 10.1016/S0140-6736(20)30628-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikacenic C., Moore R., Dmyterko V., West T.E., Altemeier W.A., Liles W.C., and Lood C.. 2018. Neutrophil extracellular traps (NETs) are increased in the alveolar spaces of patients with ventilator-associated pneumonia. Crit. Care. 22:358 10.1186/s13054-018-2290-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakazawa D., Kumar S.V., Marschner J., Desai J., Holderied A., Rath L., Kraft F., Lei Y., Fukasawa Y., Moeckel G.W., et al. 2017. Histones and Neutrophil Extracellular Traps Enhance Tubular Necrosis and Remote Organ Injury in Ischemic AKI. J. Am. Soc. Nephrol. 28:1753–1768. 10.1681/ASN.2016080925 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Narasaraju T., Yang E., Samy R.P., Ng H.H., Poh W.P., Liew A.A., Phoon M.C., van Rooijen N., and Chow V.T.. 2011. Excessive neutrophils and neutrophil extracellular traps contribute to acute lung injury of influenza pneumonitis. Am. J. Pathol. 179:199–210. 10.1016/j.ajpath.2011.03.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oehmcke S., Mörgelin M., and Herwald H.. 2009. Activation of the human contact system on neutrophil extracellular traps. J. Innate Immun. 1:225–230. 10.1159/000203700 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papayannopoulos V. 2018. Neutrophil extracellular traps in immunity and disease. Nat. Rev. Immunol. 18:134–147. 10.1038/nri.2017.105 [DOI] [PubMed] [Google Scholar]
- Papayannopoulos V., Metzler K.D., Hakkim A., and Zychlinsky A.. 2010. Neutrophil elastase and myeloperoxidase regulate the formation of neutrophil extracellular traps. J. Cell Biol. 191:677–691. 10.1083/jcb.201006052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papayannopoulos V., Staab D., and Zychlinsky A.. 2011. Neutrophil elastase enhances sputum solubilization in cystic fibrosis patients receiving DNase therapy. PLoS One. 6:e28526 10.1371/journal.pone.0028526 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park J., Wysocki R.W., Amoozgar Z., Maiorino L., Fein M.R., Jorns J., Schott A.F., Kinugasa-Katayama Y., Lee Y., Won N.H., et al. 2016. Cancer cells induce metastasis-supporting neutrophil extracellular DNA traps. Sci. Transl. Med. 8:361ra138 10.1126/scitranslmed.aag1711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raup-Konsavage W.M., Wang Y., Wang W.W., Feliers D., Ruan H., and Reeves W.B.. 2018. Neutrophil peptidyl arginine deiminase-4 has a pivotal role in ischemia/reperfusion-induced acute kidney injury. Kidney Int. 93:365–374. 10.1016/j.kint.2017.08.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rohrbach A.S., Slade D.J., Thompson P.R., and Mowen K.A.. 2012. Activation of PAD4 in NET formation. Front. Immunol. 3:360 10.3389/fimmu.2012.00360 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruan Q., Yang K., Wang W., Jiang L., and Song J.. 2020. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 10.1007/s00134-020-05991-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schönrich G., and Raftery M.J.. 2016. Neutrophil Extracellular Traps Go Viral. Front. Immunol. 7:366 10.3389/fimmu.2016.00366 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Semeraro F., Ammollo C.T., Morrissey J.H., Dale G.L., Friese P., Esmon N.L., and Esmon C.T.. 2011. Extracellular histones promote thrombin generation through platelet-dependent mechanisms: involvement of platelet TLR2 and TLR4. Blood. 118:1952–1961. 10.1182/blood-2011-03-343061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sil P., Wicklum H., Surell C., and Rada B.. 2017. Macrophage-derived IL-1β enhances monosodium urate crystal-triggered NET formation. Inflamm. Res. 66:227–237. 10.1007/s00011-016-1008-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sollberger G., Choidas A., Burn G.L., Habenberger P., Di Lucrezia R., Kordes S., Menninger S., Eickhoff J., Nussbaumer P., Klebl B., et al. 2018. Gasdermin D plays a vital role in the generation of neutrophil extracellular traps. Sci. Immunol. 3:eaar6689 10.1126/sciimmunol.aar6689 [DOI] [PubMed] [Google Scholar]
- Sreeramkumar V., Adrover J.M., Ballesteros I., Cuartero M.I., Rossaint J., Bilbao I., Nácher M., Pitaval C., Radovanovic I., Fukui Y., et al. 2014. Neutrophils scan for activated platelets to initiate inflammation. Science. 346:1234–1238. 10.1126/science.1256478 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tagami T., Tosa R., Omura M., Fukushima H., Kaneko T., Endo T., Rinka H., Murai A., Yamaguchi J., Yoshikawa K., et al. 2014. Effect of a selective neutrophil elastase inhibitor on mortality and ventilator-free days in patients with increased extravascular lung water: a post hoc analysis of the PiCCO Pulmonary Edema Study. J. Intensive Care. 2:67 10.1186/s40560-014-0067-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas G.M., Carbo C., Curtis B.R., Martinod K., Mazo I.B., Schatzberg D., Cifuni S.M., Fuchs T.A., von Andrian U.H., Hartwig J.H., et al. 2012. Extracellular DNA traps are associated with the pathogenesis of TRALI in humans and mice. Blood. 119:6335–6343. 10.1182/blood-2012-01-405183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Brühl M.L., Stark K., Steinhart A., Chandraratne S., Konrad I., Lorenz M., Khandoga A., Tirniceriu A., Coletti R., Köllnberger M., et al. 2012. Monocytes, neutrophils, and platelets cooperate to initiate and propagate venous thrombosis in mice in vivo. J. Exp. Med. 209:819–835. 10.1084/jem.20112322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang D., Hu B., Hu C., Zhu F., Liu X., Zhang J., Wang B., Xiang H., Cheng Z., Xiong Y., et al. 2020. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA. 323:1061 10.1001/jama.2020.1585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warnatsch A., Ioannou M., Wang Q., and Papayannopoulos V.. 2015. Inflammation. Neutrophil extracellular traps license macrophages for cytokine production in atherosclerosis. Science. 349:316–320. 10.1126/science.aaa8064 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wen F., White G.J., VanEtten H.D., Xiong Z., and Hawes M.C.. 2009. Extracellular DNA is required for root tip resistance to fungal infection. Plant Physiol. 151:820–829. 10.1104/pp.109.142067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu C., Chen X., Cai Y., Xia J., Zhou X., Xu S., Huang H., Zhang L., Zhou X., Du C., et al. 2020. Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern. Med. 10.1001/jamainternmed.2020.0994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu D., and Yang X.O.. 2020. TH17 responses in cytokine storm of COVID-19: An emerging target of JAK2 inhibitor Fedratinib. J. Microbiol. Immunol. Infect.:S1684-1182(20)30065-7 10.1016/j.jmii.2020.03.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wygrecka M., Kosanovic D., Wujak L., Reppe K., Henneke I., Frey H., Didiasova M., Kwapiszewska G., Marsh L.M., Baal N., et al. 2017. Antihistone Properties of C1 Esterase Inhibitor Protect against Lung Injury. Am. J. Respir. Crit. Care Med. 196:186–199. 10.1164/rccm.201604-0712OC [DOI] [PubMed] [Google Scholar]
- Xu J., Zhang X., Pelayo R., Monestier M., Ammollo C.T., Semeraro F., Taylor F.B., Esmon N.L., Lupu F., and Esmon C.T.. 2009. Extracellular histones are major mediators of death in sepsis. Nat. Med. 15:1318–1321. 10.1038/nm.2053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu X., Han M., Li T., Sun W., Wang D., Fu B., Zhou Y., Zheng X., Yang Y., Li X., et al. 2020. Effective Treatment of Severe COVID-19 Patients with Tocilizumab. ChinaXiv. http://chinaxiv.org/abs/202003.00026 (Preprint posted March 5, 2020) [DOI] [PMC free article] [PubMed]
- Xu Z., Huang Y., Mao P., Zhang J., and Li Y.. 2015. Sepsis and ARDS: The Dark Side of Histones. Mediators Inflamm. 2015:205054 10.1155/2015/205054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yao X.H., Li T.Y., He Z.C., Ping Y.F., Liu H.W., Yu S.C., Mou H.M., Wang L.H., Zhang H.R., Fu W.J., et al. 2020. [A pathological report of three COVID-19 cases by minimally invasive autopsies]. Zhonghua Bing Li Xue Za Zhi. 49:E009 10.3760/cma.j.cn112151-20200312-00193 [DOI] [PubMed] [Google Scholar]
- Yost C.C., Schwertz H., Cody M.J., Wallace J.A., Campbell R.A., Vieira-de-Abreu A., Araujo C.V., Schubert S., Harris E.S., Rowley J.W., et al. 2016. Neonatal NET-inhibitory factor and related peptides inhibit neutrophil extracellular trap formation. J. Clin. Invest. 126:3783–3798. 10.1172/JCI83873 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang W., Zhao Y., Zhang F., Wang Q., Li T., Liu Z., Wang J., Qin Y., Zhang X., Yan X., et al. 2020. The use of anti-inflammatory drugs in the treatment of people with severe coronavirus disease 2019 (COVID-19): The Perspectives of clinical immunologists from China. Clin. Immunol. 214:108393 10.1016/j.clim.2020.108393 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Z., Xie J., Yin M., Yang Y., He H., Jin T., Li W., Zhu X., Xu J., Zhao C., et al. 2020. Clinical and Laboratory Profiles of 75 Hospitalized Patients with Novel Coronavirus Disease 2019 in Hefei. MedRxiv, 10.1101/2020.03.01.20029785 (Preprint posted March 6, 2020) [DOI]
- Zheng M., Gao Y., Wang G., Song G., Liu S., Sun D., Xu Y., and Tian Z.. 2020. Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cell. Mol. Immunol. 10.1038/s41423-020-0402-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z., Xiang J., Wang Y., Song B., Gu X., et al. 2020. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 395:1054–1062. 10.1016/S0140-6736(20)30566-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zou Y., Chen X., Xiao J., Bo Zhou D., Xiao Lu X., Li W., Xie B., Kuang X., and Chen Q.. 2018. Neutrophil extracellular traps promote lipopolysaccharide-induced airway inflammation and mucus hypersecretion in mice. Oncotarget. 9:13276–13286. 10.18632/oncotarget.24022 [DOI] [PMC free article] [PubMed] [Google Scholar]