Abstract
Newcastle disease virus (NDV) causes huge economic loss to the poultry industry due to high mortality and morbidity. The present study aimed to assess the protective role of novel phosphorylated analogue ABC‐1 in vivo in NDV‐infected chickens through the inhibition of fusion protein. Both NDV‐induced oxidative damage and protective role of novel phosphorylated ABC‐1 were evaluated in vital organs such as the liver and lung of chickens. Enzyme linked immunosorbent assay (ELISA) results showed that protein oxidation and nitration levels were significantly raised in NDV‐infected tissues compared to healthy controls, whereas these levels were reduced significantly (P < 0.05) in birds treated with phosphorylated compounds compared to the NDV‐infected group alone. Additional investigation with double immunofluorescence showed that the large amount of immuno colocalization and Western blot analysis also confirmed this observation through its band pattern in NDV‐infected birds compared to healthy birds, whereas these alterations were reduced in treatment with novel phosphorylated ABC‐1. The expression of fusion glycoprotein was studied by immuno colocalization, PCR, and flow cytometry, and results demonstrated that the novel phosphorylated analogues reduced the expression of fusion glycoprotein. These results put forth that novel phosphorylated ABC‐1 protects chickens from NDV‐induced pathogenesis, protein oxidation/nitration, and exerts potent antiviral activity.
Keywords: fusion protein, Newcastle disease virus, oxidative stress, phosphorylated compound
Abbreviations
- ABC
abacavir
- ABC‐1
phosphorylated abacavir analogue
- DNA
deoxyribonucleic acid
- F
fusion
- HN
heamgglutinin‐neuraminidase
- ND
newcastle disease
- NDV
newcastle disease virus
- NRTIs
nucleoside reverse transcriptase inhibitors
- RNA
ribonucleic acid
- RNS
reactive nitrogen species
- ROS
reactive oxygen species
- US‐FDA
United States‐food and drug administration
1. Introduction
Newcastle disease (ND) is the most severe avian disease caused by Newcastle disease virus (NDV), affecting economic catastrophe in poultry throughout the world 1. Currently, the existence of NDV in the world chicken population has been extensively reported 2. NDV has an extensive host range that can infect more than 200 avian species. Chicken is the most susceptible host to NDV, which leads to intermittent infections 3, 4. NDV belongs to genus Avulavirus, family Paramyxoviridae, and order Mononegavirales 5. NDV is majorly classified into four pathotypes, such as viscerotropic, neurotropic, mesogenic, and lentogenic. It is an enveloped, negative sense single‐stranded ribonucleic acid (RNA) virus of 15 kb nucleotides length, which codes for six major proteins 6. One very important feature of many viruses is that their surface is covered with either glycoproteins or carbohydrate recognition molecules. It has been recognized for some time that this glycosylation is vital to viral infectivity or replication as well as in the production of active viral particles 7. Fusion (F) and heamgglutinin‐neuraminidase (HN) are important glycoproteins that play a major role in the viral entry. Fusion protein is responsible for fusion of viral envelope and mediates cell membrane conformational changes, whereas HN protein is responsible for cell binding 8. The F protein is synthesized in the beginning as a precursor F0, which is then cleaved to F1 and F2 active precursors by an enzyme furin present in the host cell, which are the key contributors for NDV pathogenicity 9.
It is well established that the viral infections that induce oxidative stress by direct viral effects on cells or by inflammatory responses from host are the common pathways for the damage of tissues 10. The imbalance between pro‐oxidants and endogenous antioxidants in living cell or tissues leads to the development of oxidative damage to the cells. Typically, oxidative stress is initiated by the release of reactive oxygen species (ROS) and reactive nitrogen species (RNS), which play a vital role in tissue damage 11. An endogenously formed reactive nitrogen oxide during viral replication has the potential to mutant both the viruses and host, which results in tissue injury due to oxidative stress, by oxidation and nitration of biomolecules 12. RNS to an extent possess a lower antiviral activity against many DNA viruses such as murine poxvirus, herpesviruses, Epstein‐Barr virus, and some RNA viruses such as coxsackievirus. However, these RNS species do not show antiviral activity against the ortho‐ and paramyxovirus, coronavirus, and others. Increased RNS during the viral replication leads to oxidative stress and damage to the biological molecules such as proteins, DNA, and lipids 13, 14. It is reported that the oxidative stress occupies a major role in pathogenesis in many diseases such as Alzheimer's, diabetes, hepatorenal syndrome, atherosclerosis, and hypertension 15.
Nucleoside reverse transcriptase inhibitors (NRTIs) are synthetic compounds structurally similar to the natural nucleosides, which are building blocks of nucleic acids. In the present scenario, NRTIs are the priority drugs to treat life‐threatening diseases such as human immunodeficiency virus (HIV), Herpes virus, small pox virus, cancer, and hepatitis 16. These NRTIs exert biological activity both by inhibiting viral RNA or DNA or cellular and as terminators of chain elongation or inhibiting enzymes like polymerases 17. Hotard et al. 18 stated that NRTIs shows high antiviral activity against henipaviruses and other paramyxoviruses. Abacavir (ABC) is a carbocyclic nucleoside analogue that terminates the replication of viral gene elongation by incorporating a nucleoside analogue into the new viral gene 19. There are 26 antiretroviral drugs approved by US‐FDA for clinical use; these drugs are effective when used in combinations, even though none of them are considered ideal due to their toxic effects and ascendance of creating drug‐resistant mutants 20. Controlling mechanism of NRTIs against host cell has limitations including low solubility and low bioavailability, toxicity, hypertension, lacking target specificity, short half‐life, cellular toxicities, and unstable at acidic conditions 21. Hence, it is important to design the antiviral drugs with limited toxic effects and more solubility and bioavailability. Youcef et al. 22 reported that these disadvantages of nucleoside drug could be prevailed over by phosphorylated prodrugs.
Hence, the current study aims to evaluate the role of oxidative damage in liver and lung by protein oxidation and nitration during NDV pathogenesis. It is also focused to measure NDV fusion protein expression and to evaluate the protective role of novel phosphorylated compound ABC‐1 against NDV infection.
2. Materials and Methods
2.1. Maintenance of experimental animals
One‐day‐old chickens (Gallus domesticus; BV 300) weighing 35 ± 5g were purchased from Balaji Hatcheris (Chittoor, India). The chickens were kept for 1 day for acclimatization and maintained in isolated cages with free access to water and food during experimental period in 12:12 light/dark cycles. All the animal experiments were approved by IAEC, Sri Venkateswara University, Tirupati (No. 14/2012–2013/(1)/a/CPCSEA/IAEC/SVU/ VL‐WR dt.23.01.2013).
2.2. Virus maintenance
NDV (Kumarov mesogenic reference strain, India) was obtained from the Department of Microbiology, Sri Venkateswara Veterinary University, Tirupati, Andhra Pradesh. The infectious dose ID50 of the virus is calculated as 109.4 units/mL. The stock of NDV was maintained on embryonated egg passages, and the titer of NDV stock is 1,024 HA units; it was subsequently stored at −40 °C until use.
2.3. Grouping of experimental animals
Chickens were divided into three groups: (1) control, (2) NDV‐infected, 3) NDV + ABC‐1‐treated chickens; each group contained six individuals fed ad libitum water and food in isolated cages. Note that 200 µL of NDV with an infectious dose50 of 109.4 units/mL was administered via intramuscular route to groups 2 and 3 chickens. Group 1 chickens received 200 µL of phosphate buffered saline (PBS) and served as control chicken. Group 3 chickens along with NDV were treated with ABC‐1 compound at the concentration of 2 mg/kg body weight, dissolved in water, and administered by oral route during experiments from the fourth day postinfection.
2.4. Synthesis of novel phosphorylated ABC‐1 compound
Initially, 16 novel derivatives of ABC have been designed and subjected to docking analysis. Based on the docking scores and molecular descriptors analysis, three best ABC analogues (ABC‐1, ‐4, and ‐12) were selected for in vitro analysis. Of the three analogues, ABC‐1 was selected for further in vivo experimental models. The detailed protocol for the synthesis of novel phosphorylated ABC‐1, clinical signs of NDV infection, antiviral activity of novel phosphorylated analogue ABC‐1 compared to parent ABC, and survival rate of animals during infection were already detailed in a previous publication 23.
2.5. Measurement of protein oxidation by ELISA
In brief, 10% w/v tissues of liver and lung of each bird from all three groups, that is, control, NDV infected, and NDV + ABC‐1 treated, were taken and homogenated. Note that 100 µg/mL sample protein in PBS was added to 96‐well plate and allowed to incubate overnight at 4 °C and washed thrice with PBS. Alone PBS was used as control. Note that 200 µL of DNP solution was added and allowed to incubate for 45 Min at room temperature in dark, washed with PBS and ethanol (1:1 v/v), and once only with PBS. Blocking was done using 5% skimmed milk powder for 90 Min followed by three washes with PBS containing 0.1% tween 20 (PBS‐T). Rabbit anti‐DNP antibody (1:500) was added to each well and incubated for 1 H and washed with PBS‐T. Secondary antibody anti‐rabbit (1:2,500) was added and incubated for 1 H and washed with PBS‐T. Tetramethylbenzidine (TMB) substrate was added and kept for 4–5 Min, and the reaction was stopped by 2 N HCl and absorbance was read at 450 nm 14.
2.6. Determination of protein nitration by competitive ELISA
Tissue homogenate samples (100 µg/mL) from the liver and lung of the control group, NDV‐infected, and NDV + ABC‐1‐treated samples were added to 96‐well plate in triplicates coated with nitrated BSA dissolved in 50 mM carbonate buffer (pH 9). Primary antibody anti‐nitrotyrosine (1:200) was added and incubated for 2 H at 37 °C, followed by washing with wash buffer. Secondary antibody goat anti‐mouse (1:5,000) conjugated with HRP was added and incubated for 1 H at room temperature. TMB substrate was added, and the reaction was then stopped by 2 N H2SO4. The intensity/absorbance of the reaction mixture was measured at 492 nm in an ELISA plate reader 24.
2.7. Immuno colocalization studies to detect protein oxidation and nitration
The extent of NDV‐induced oxidative damage in the liver and lung tissue of chickens from the control group, NDV‐infected, and NDV + ABC‐1‐treated groups was analyzed by immunofluorescence using anti‐DNP and anti‐nitrotyrosine primary antibodies, a marker for measuring protein oxidation and nitration 25. The tissue sections were deparaffinized in a sequential step from xylene, ethanol, and distilled water. To assess the protein oxidation levels, 0.1% 2,4‐dinitrophenyl hydrazine (DNPH) in 2 N HCl was added to the sections and kept overnight at 4 °C. For protein nitration, the derivatization by DNPH was not carried out. The tissue was quenched with 0.3% H2O2 for 30 Min and washed with PBS three times. Sections were then blocked by applying 10% normal serum in PBS for 30 Min. Antibody rabbit anti‐DNP (1:100) was added and incubated overnight at 4 °C, washed thrice with PBS. The secondary antibody anti‐rabbit Alexa Flour 488 (1:500) was added to the sections for 60 Min in dark. Second primary antibody mouse anti α‐SMA (1:400; liver) and anti‐clara (1:500; lung) to label the hepatic stellate cells, clara cells respectively for 60 Min at room temperature followed by washing with PBS and second secondary antibody anti‐mouse Alexaflour 594 (1:500) were added and allowed to incubate for 1 H. Sections were then washed with PBS and anti‐profade reagent was added.
2.8. Western blot analysis
Immunoblot analysis was carried out with anti‐DNP and anti‐3‐nitrotyrosine antibodies to measure protein oxidation and nitration in the lung and liver tissues of control, NDV‐infected, and NDV + ABC‐1‐treated groups. To carry out protein oxidation, proteins were derivatized using 20 mM DNP (10 µL) for 15 Min at room temperature followed by the addition of 5 µL of neutralization solution and vortexed. Note that 30 µg of protein samples were then loaded on to 10% SDS‐PAGE and electro‐blotting was made with 0.2‐µm PVDF membrane at 55 V for 45 Min followed by a quick wash with TBS. Blocking was done using 2% BSA in TBST for 2 H and washed three times with TBST for 10 Min of each interval. Rabbit anti‐DNP primary antibody (1:1,000) was added and allowed to incubate for overnight at 4 °C, and after washing with TBST goat anti‐rabbit IgG (1:5,000) horseradish peroxidase conjugate was added and kept for 2 H at room temperature and washed with TBST three times. The bands in the membrane were visualized by developing with chemiluminescence substrate method. In protein nitration, 3‐nitrotyrosine monoclonal antibody (1:1,000) was used as a primary antibody, and immunoblotting was performed with the same experimental conditions without the protein derivation step as for protein carbonyls 26, 27.
2.9. Fusion protein inhibition studies
2.9.1. Immuno colocalization study to detect fusion protein in organs of infected chickens
To estimate whether novel analogue exerts antiviral activity by the inhibition of viral glycoprotein tissue of liver and lung from control, NDV‐infected and NDV + ABC‐1‐treated groups were analyzed by immunofluorescence. The sections were deparaffinized in a sequential step from xylene, ethanol, and distilled water. The tissue was quenched with 0.3% H2O2 for 30 Min and washed with PBS three times. Sections were then blocked by using 10% normal serum in PBS for 30 Min. Anti‐F mAb 8E12A8C3 antibody mouse (1:1,000) was added and incubated overnight at 4 °C and washed three times. The sections were then added secondary antibody anti‐mouse Alexa Flour 594 (1:1,000) for 60 Min at dark. Second primary antibody anti‐clara (1:500; lung) and mouse anti α‐SMA (1:400; liver) to label the hepatic stellate cells, clara cells were added and incubate for 60 Min at room temperature followed by washing with PBS and second secondary antibody anti‐mouse AlexaFlour 488 and 350 (1:1,000) for lung and liver, respectively, were added and kept for 1 H. Sections were then washed with PBS and mounted with anti‐profade reagent 28.
2.9.2. NDV‐fusion mRNA gene expression in lung and liver cells
Total mRNA from liver and lung cells were extracted and amplified to estimate the NDV fusion mRNA expression. In brief, 100 mg of tissue was taken and mRNA was isolated with the trizol reagent method. Reverse transcription of the total mRNA was carried out using Takara prime script first‐strand cDNA synthesis kit (Cat. No. 6110A) and incubated for 5 Min at 65 °C and cooled immediately on ice. The PCR using specific primers for fusion gene of Newcastle disease virus NDV forward primer (5′‐CCTTGGTGACTCTATCCGTA‐3′) and NDV reverse primer (3′‐ACTGCTAGTTGCGATAATCCG‐5′) was carried out using PCR master mix. The cDNA was amplified for 34 cycles with optimized annealing temperature for each primer set. Final extension was performed at 65 °C for 10 Min. Equal amounts of PCR products were run on 1.5% ethidium bromide impregnated agarose gels and visualized using GelDoc Imaging System (Bio‐Rad, Hercules, CA, USA) 29.
2.9.3. Tissue cells homogenation and fluorescence‐activated cell sorting
Lung and liver tissues were cut into pieces and incubated with 5 mg/mL of collagenase in PBS for 45 Min at room temperature on a shaker. To the digested solution, 1 mg/mL of DNase was added and the solution was filtered using 40 µm filter. The solution was centrifuged to obtain clear white colored cell pellet. Primary antibody anti‐F mAb 8E12A8C3 (1:1,000) was added to cell pellet and incubated for 1 H followed by secondary antibody anti‐mouse Alexaflour 350 (1:2,000) was used to stain the viral fusion protein. After three washes in 10% FBS in PBS, cells were fixed in 2% paraformaldehyde in PBS and subjected to flow cytometry 30.
3. Results
3.1. Measurement of protein oxidation by ELISA
The amount of protein oxidation levels in NDV‐infected chickens was assessed by ELISA using anti‐DNP as a marker antibody in lung and liver tissues from control, NDV infected and NDV+ABC‐1 treated tissues in chickens. Protein oxidation levels were significantly (P < 0.05) increased in the NDV‐infected group, when compared to control, whereas the protein oxidation levels were decreased significantly (P < 0.05) during the treatment with NDV + ABC‐1 (Fig. 1) compared with the NDV‐infected group.
Figure 1.
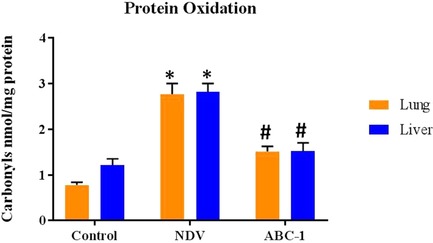
The protein carbonyl levels in lung and liver tissues from control, NDV‐infected, and NDV + ABC‐1‐treated chicken were measured by ELISA. Values expressed are mean and SD of triplicates. *P < 0.05 between the control group and the NDV‐infected group. #P < 0.05 between NDV‐infected and treatment groups.
3.2. Determination of protein nitration by competitive ELISA
NDV‐induced protein nitration levels were measured using antibody 3‐nitrotyrosine in the liver and lung from control, NDV‐infected, and NDV + ABC‐1‐treated groups. Nitrated protein levels were significantly (P < 0.05) elevated in the NDV‐infected group than the healthy chicken group, whereas the nitrated protein levels were significantly (P < 0.05) reduced during the treatment with NDV + ABC‐1 (Fig. 2) in comparison to NDV‐infected chickens.
Figure 2.
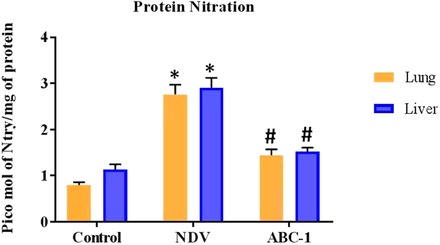
The nitrated protein levels in control, NDV‐infected, and NDV + ABC‐1‐treated chicken were measured in the lung and liver by ELISA. Values expressed are mean and SD of triplicates. *P < 0.05 between the control group and the NDV‐infected group. #P < 0.05 between the NDV‐infected group and NDV + ABC‐1 groups.
3.3. Immuno colocalization and Western blot assay for oxidation and nitration of proteins
ELISA results showed a distinct oxidative damage during NDV pathogenesis and significantly decreased on treatment with ABC‐1. Hence, NDV‐induced oxidative damage was quantified by immuno colocalization and western blot analysis. Immunohistochemical staining was carried out in three groups: control, NDV infected, and NDV + ABC‐1 treated. Immuno colocalization studies of tissue sections indicated that the NDV‐infected chickens have more oxidized protein with large number of hepatic stellate cells in liver (Fig. 3H) and clara cells in lung tissue (Fig. 4H) when compared to healthy control animals (Figs. 3 and 4G). In contrast, cellular alterations were minimal or decreased in the liver and lung of the NDV‐infected group treated with ABC‐1 (NDV + ABC‐1) (Figs. 3 and 4I). The intensity of fluorescence was quantified and represented in bar chart graph (Fig. 10B). Immunofluorescence studies of protein nitration were carried out by anti‐nitrotyrosine antibody. Similar to the protein oxidation, protein nitration is significantly altered in the NDV‐infected animal tissues (Figs. 5 and 6H) compared to control animals (Figs. 5 and 6G), whereas these alterations decreased significantly in the NDV + ABC‐1‐treated group (Figs. 5 and 6I), and the quantification of fluorescence values was presented in bar chart graph (Fig. 10A).
Figure 3.
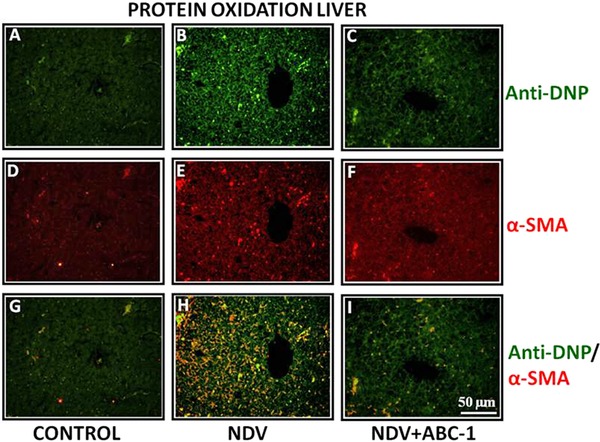
Photomicrographs of double immunofluorescence stained liver tissues with anti‐DNP and α‐SMA to detect oxidized protein. (A–C) Anti‐DNP, (D–F) α‐SMA, and (G–I) colocalized. A considerable amount of immunocolocalized cells in liver was observed (H) when compared to control (G) and treated birds (I). Scale bar ( ) = 50 µm.
) = 50 µm.
Figure 4.
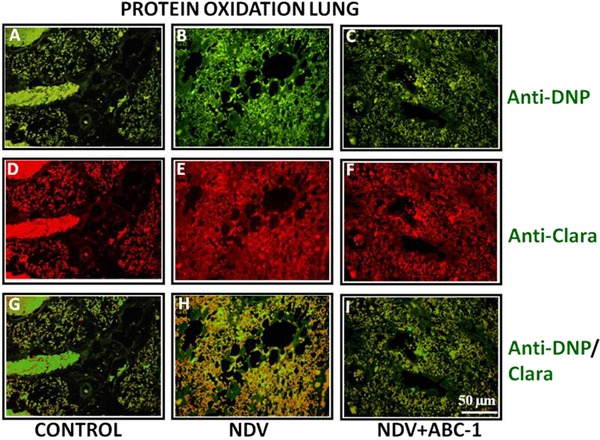
Photomicrographs of double immunofluorescence stained lung tissues with anti‐DNP and anti‐clara primary antibodies. (A–C) Anti‐DNP, (D–F) anti‐clara, and (G–I) colocalized. A considerable amount of immunocolocalized (oxidized protein) cells in lung was observed (H) when compared to control (G) and treated birds (I). Scale bar ( ) = 50 µm.
) = 50 µm.
Figure 10.
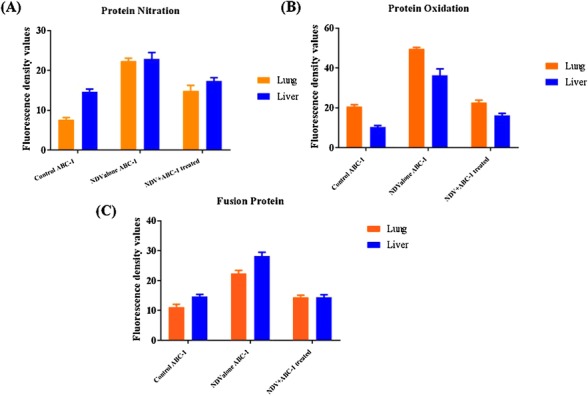
Quantification of immunocolocalized photomicrographs. (A) Protein nitration, (B) protein oxidation, and (C) Fusion protein. Fluorescence density values were raised in the NDV‐infected alone group in comparison to control animals. The levels in treated animals were brought down with novel phosphorylated compounds ABC‐1.
Figure 5.
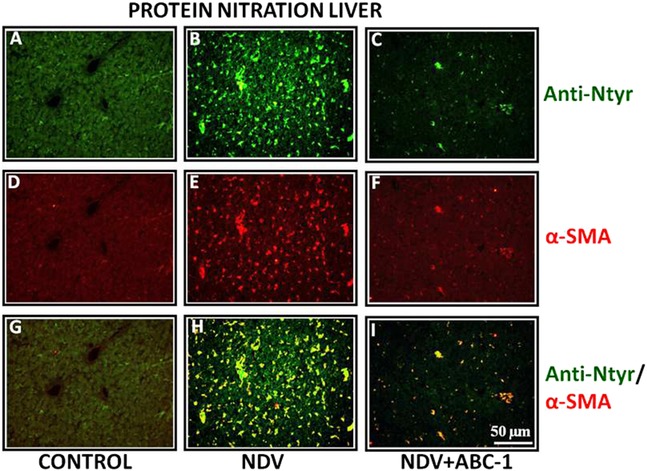
Photomicrographs of double immunofluorescence stained liver tissues with anti‐nitrotyrosine and α‐SMA. (A–C) Anti‐nitrotyrosine, (D–F) α‐SMA, and (G–I) colocalized. (H) A large amount of nitrated protein due to NDV infection, whereas (I) treated bird showed a lesser number of colocalized cells than control bird (G). Scale bar ( ) = 50 µm.
) = 50 µm.
Figure 6.
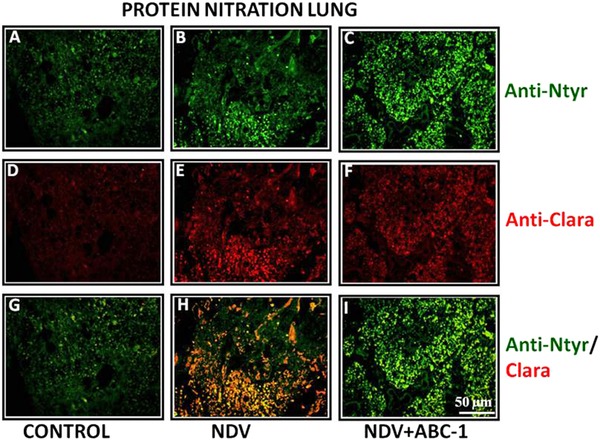
Photomicrographs of double immunofluorescence stained lung tissues with anti‐nitrotyrosine and anti‐clara primary antibodies. (A–C) Anti‐nitrotyrosine, (D–F) anti‐clara, and (G–I) colocalized. (H) Tissue from NDV‐infected animal showed more number of colocalized cells than that of (I) treated bird. (G) Control bird shows only few nitrated cells. Scale bar ( ) = 50 µm.
) = 50 µm.
Detection of oxidative and nitrated protein damage was also confirmed by western blot analysis. Tissue homogenates of control, NDV‐infected, and NDV+ABC‐1 groups were subjected to blotting using anti‐DNP and 3‐nitrotyrosine antibodies for protein oxidation and nitration, respectively. Blotting analysis showed reduced band intensity in treatment with ABC‐1 compound. The intensity of the bands in the NDV group is more compared to control, which is an indication of presence of oxidized and nitrated protein in a large amount. These alterations in the protein oxidation and nitration levels were brought down with the treatment of ABC‐1, as shown in Fig. 7.
Figure 7.
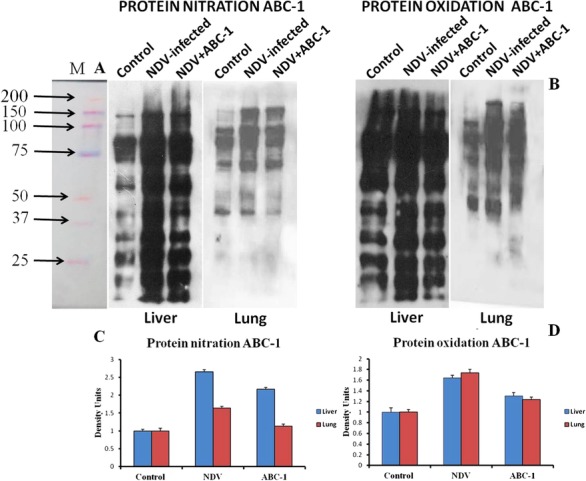
Western blot analysis of protein nitration and oxidation in control, NDV‐infected, and NDV + ABC‐1‐treated chickens. The antibodies used to measure nitrated proteins are primary antibody (mouse anti‐nitrotyrosine, 1:1,000) and secondary antibody (rabbit antimouse, 1:5,000), respectively, and protein oxidation was detected using the antibodies: primary antibody (rabbit anti‐DNP, 1:1,000 dilution) and secondary antibody (goat anti‐rabbit, 1:5,000), respectively. The NDV‐infected group animal shows an increased intensity of bands compared to control, whereas the band intensity was decreased in the treated animals. (A) Protein nitration, (B) protein oxidation, and (C and D) quantification of band density by image j software.
3.4. Immuno colocalization study to detect fusion protein in organs of infected chickens
NDV‐induced tissue damage was studied by performing immuno colocalization technique. The study revealed that the NDV‐infected chicken liver (Fig. 8) and lung (Fig. 9) sections showed numerous colocalized hepatic and clara cells, indicating the severity of the viral pathogenesis, whereas these colocalized cells were reduced in liver treated with ABC‐1 (Fig. 8I) and clara cells in lung tissue treated with ABC‐1 (Fig. 9I) when compared to NDV control animals. The quantification of fluorescence values was presented in bar chart graph (Fig. 10C).
Figure 8.
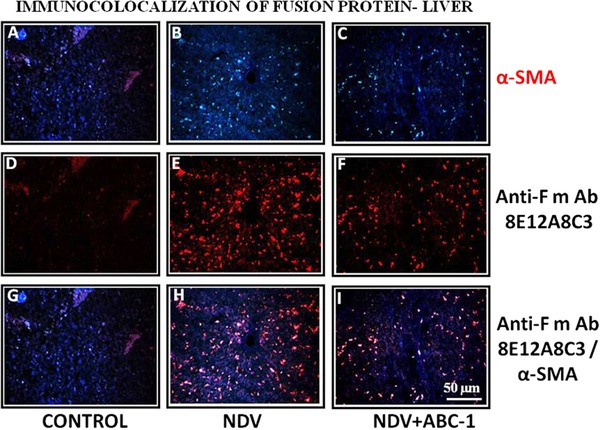
Photomicrographs of immunocolocalization of fusion protein in liver tissues. (A–C) α‐SMA, (D–F) anti‐F mAb, and (G–I) colocalized. A considerable amount of immunocolocalized cells in liver was observed (H) when compared to control (G) and treated birds (I). Scale bar ( ) = 50 µm.
) = 50 µm.
Figure 9.
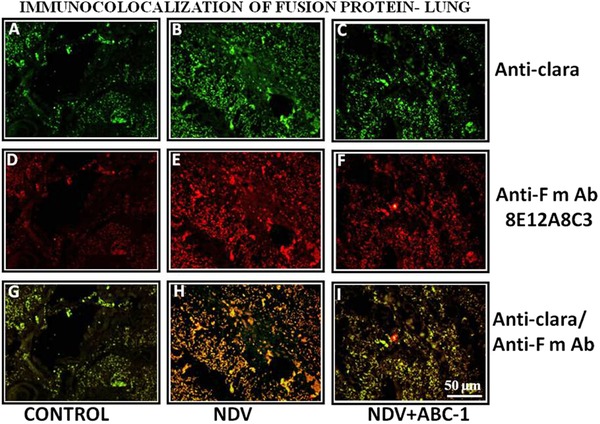
Photomicrographs of immunocolocalization of fusion protein in lung tissues. (A–C) Anti‐clara, (D–F) anti‐F mAb, and (G–I) colocalized. A considerable amount of immunocolocalized cells in lung was observed (H) when compared to control (G) and treated birds (I). Scale bar ( ) = 50 µm.
) = 50 µm.
3.5. NDV‐fusion mRNA gene expression in lung and liver cells
The NDV fusion gene expression was tested in different groups such as healthy control, NDV‐infected alone, and NDV+ABC‐1‐treated group tissue of liver and lung. The obtained PCR product was resolved on the 1% agarose gel and bands were visualized by chemidoc systems (Bio‐Rad). The visualization of gel has revealed no band in control, which indicated that there was no presence of viral fusion gene. NDV‐infected tissues have shown very intensified bands, which are a measure of higher viral replication during infection period. In contrast, the band intensity was reduced in the ABC‐1‐treated groups (Fig. 11), which implies that these novel phosphorylated compounds had the potential to reduce the expression of fusion protein gene, therefore viral replication is reduced and viral pathogenesis has been controlled.
Figure 11.
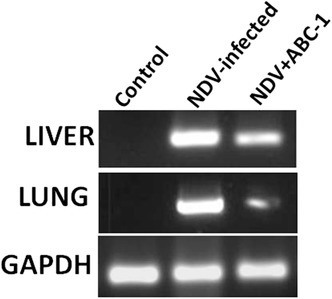
Viral detection in liver and lung tissue of experimentally infected and treated animals by PCR. Control chicken was uninfected and represents a healthy group, whereas the NDV‐infected group is experimentally infected and NDV + ABC‐1 represents the treatment group. The intensity of bands decreased upon treating with ABC‐1 compared to NDV‐infected liver and lung tissue. GAPDH was used as internal control for the experiment.
3.6. Tissue cells homogenation and fluorescence‐activated cell sorting
Fluorescence‐activated cell sorting (FACS) analysis was performed in different groups such as control, NDV infected, and ABC‐1‐treated organs (liver and lung). Liver and lung cells were sorted based on labeling with anti‐F mAb 8E12A8C3 primary antibody (1:5,000; CVI, Lelystad) and AF‐350 secondary antibody. The results obtained demonstrated that viral fusion protein levels were as high as 17.8% (Fig. 12B) and 23.5% (Fig. 12E), respectively, in liver and lung tissues in the NDV‐infected group. The amount of viral fusion protein levels was distinctly decreased in the chicken group treated with the ABC‐1, measured as 10.3% (Fig. 12C) and 7.8% (Fig. 12F), respectively, in liver and lung tissues. These results signify that these novel phosphorylated compounds have shown potent antiviral activity by inhibiting the imperative viral protein, which is important in viral replication cycle. Hence, these findings demonstrated that ABC‐1 might be more potent antiviral drug against NDV‐infected chickens.
Figure 12.
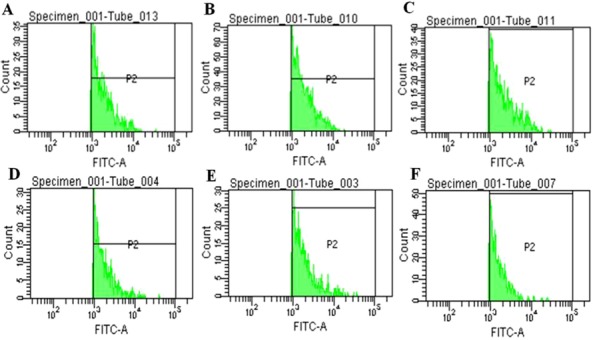
Characterization of NDV viral fusion protein in liver and lung cell population. Cells were sorted on the basis of labeling with anti‐F mAb 8E12A8C3 (1:5,000; CVI, Lelystad) primary antibody and AF‐350 secondary antibody. Liver: (A) Control and (B) NDV infected, and (C) ABC‐1 treated. NDV fusion protein level make up 17.8% in infected and 10.3% ABC‐1 based on population values of fusion protein. Lung: (D) Control, (E) NDV infected, and (F) ABC‐1 treated. NDV fusion protein level make up 23.5% in infected, and 7.8% in ABC‐1 based on population values of fusion protein.
4. Discussion
Poultry industry is one of the major food‐producing sectors all over the world. The major constraint faced by poultry is the microbial diseases, along with viral infections. Due to the lack of early diagnosis and treatment, viral infections deleteriously affect the poultry sector causing huge economic loss to farmers 31. Of the poultry viruses, NDV is the devastating one, which causes 100% mortality by killing entire flocks. NDV infection is an airborne disease, which is transmitted by air from infected birds to healthy chickens 32. Oxidative stress is implicated as a pathogenic factor in several viral infections. It is well known that RNA viruses such as influenza virus, hepatitis c virus (HCV), and HIV induce oxidative damage, which contributes to various aspects of viral pathogenesis 33. First evidence of oxidative stress induced during viral infections was noticed during paramyxovirus where Sendai virus increased the production of reactive species during viral infection 34. It is well known that NDV mesogenic and velogenic strains lead to oxidative stress, resulting in the increased lipid peroxidation and decreased antioxidant enzymes such as superoxide dismutase and catalase in vital organs of chickens 35. Recently, we had shown that the NDV‐infected group shows increased lipid peroxidation and decreased superoxide dismutase and catalase. These perturbations were reduced in treatment with novel phosphorylated ABC‐1 than native ABC 23. Similar to the other RNA viruses, RNA from NDV virus induces oxidative damage, a condition of imbalance between ROS and host antioxidant mechanism leads to increased lipid peroxidation and DNA damage in tissues 36. Oxidative stress is caused when cells are exposed to more amount of ROS like H2O2 or O2−. In the present study, NDV‐induced pathogenesis and oxidative damage in the vital organs of chicken liver and lung were measured by oxidation and nitration of tissue proteins and also evaluated the protective role of novel phosphorylated ABC‐1.
Protein oxidation is the covalent modification of proteins either by ROS directly or indirectly by its reactive secondary metabolites. These oxidative modifications are induced in various diseased conditions in vivo; protein oxidation serves as the marker for measuring the oxidative stress in vivo 37. Protein nitration is an imperative oxidative damage caused by peroxynitrile radicals and has significant role in disease pathogenesis, which regulates the phosphorylation of proteins 38. The by‐products produced during oxidative damage are more stable than the ROS or RNS and can be measured by their products such as lipid peroxidation. Our earlier studies have shown an upsurge in the lipid peroxidation levels while chickens were infected with NDV and decreased upon treatment, which indicates that NDV induced oxidative stress 23. In the present study, we have measured oxidative damage in chickens infected with NDV using distinct techniques such as ELISA, Western blot analysis, and immuno colocalization studies. DNPH and 3‐nitrotyrosine primary antibodies that are specific for detecting protein oxidation and nitration were used. As a preliminary test, ELISA test was performed and results demonstrated that protein carbonyls and nitrated protein levels were significantly increased in the NDV‐infected group than that of control chicken, whereas these levels were significantly reduced in the ABC‐1‐treated group (Figs. 1 and 2). After ELISA results being positive for oxidative damage, western blot analysis was carried out in control, NDV‐infected, and ABC‐1‐treated groups in liver and lung tissues. Blotting analysis of the tissue showed a distinct intensified band patterns in the NDV‐infected group compared to control and the treated group (Fig. 7). The extent of tissue damage caused due to viral pathogenesis was also determined by immuno colocalization assay, and the results correlated well with ELISA and western blot analysis; the NDV‐infected group tissue showed large number of immuno colocalized stellate hepatic cells and clara cells in liver and lung, respectively, than control chicken tissue. These colocalized cells were decreased during treatment with novel phosphorylated compounds. Similar type of result was observed previously during NDV infection and treatment with vitamin E 14. A large body of evidence suggested that most of the viruses cause oxidative damage by protein oxidation and nitration. Antiviral compounds were used to protect from these viral infections and their consequently produced oxidative damage. P‐icariin, a phosphorylated compound, has produced protection in oxidative damage and improved survival of animals during Duck viral hepatitis infection 37.
NDV fusion protein was the major one in replication/propagation of virus. Fusion protein was initially synthesized as F0 inactive precursor, thereafter cleaved by host to biologically active F1 and F2 units. The cleavage of the fusion protein is important for viral entry and cell‐to‐cell fusion 39. These novel phosphorylated compounds were effective against NDV both in vitro and in vivo; in the present study, fusion protein was targeted to identify whether these compounds inhibit its development. To evaluate the expression levels of fusion protein in the vital organs of NDV‐infected tissues, immuno fluorescence, PCR, and flow cytometry assays were performed. Immuno colocalization of viral F‐protein was hypothesized as a conformational change of fusion protein that is associated with the membrane‐bound fusion protein; immunofluorescence of cells infected with NDV predominantly expresses the cleaved fusion protein 28. Ailing et al. 40 experimentally infected geese with NDV, and NDV fusion protein was detected in tissues by immunohistochemical assay. Emetine dihydrochloride, an ipecac alkaloid, has inhibitory activity against dengue virus evaluated by immuno fluorescence, which is a protein and nucleic acid inhibitor. Similarly, the present study demonstrated that NDV‐infected chicken liver and lung sections had uncovered numerous colocalized hepatic and clara cells, signifying the relentlessness of viral pathogenesis (Figs. 8 and 9). Although immuno colocalization studies have shown a clear positive signal for fusion protein, further to evaluate and quantify the extent of fusion protein levels, which was also detected using PCR and FACS analysis. The mRNA expression of NDV fusion gene was measured by PCR in comparison with control–infected and infected–treated. The results express that mRNA expression of fusion gene is relatively lower in treatment with ABC‐1 compound compared to the NDV infection group. The increasing intensity of band in the NDV‐infected group indicates more amount of viral replication, whereas in treated groups the band intensity decreased, which might be due to the inhibition of fusion gene by novel phosphorylated compound (Fig. 11). Previous studies with matrix gene of NDV in murine macrophage cells infected with NDV virus had increasing intensity of bands, which decreased during the treatment with HM‐γ‐PGA 41. A fluorescence‐activated cell sorter was used to detect and quantify the virus‐infected cells in tissues. Anti‐F mAb 8E12A8C3 that specifically binds to the NDV fusion protein present in tissues was tagged with fluorescent secondary antibody AF‐350 anti‐mouse that was used to capture the signal in FACS assay. The FACS assay showed that in NDV infection alone the fusion protein levels were higher than that in comparison with ABC‐1‐treated group tissues (Fig. 12). Previously cytomegalovirus antigen was detected with a monoclonal antibody by fluorescent flow cytometry to quantify the viral antigen content in infected and uninfected cells 30. Even though the two novel phosphorylated compounds had potential antiviral properties, fusion protein is major determinant of the NDV virus pathogenicity and its replication. Based on the above results, it was clear that ABC‐1 has inhibited the fusion protein levels, hence these results put forth that the novel compound might be potent inhibitors of NDV disease virus, as detected using various assays.
5. Conclusion
In conclusion, NDV pathogenesis‐induced protein oxidation and nitration were higher in infected group compared to control. These perturbations were brought down to normal in the NDV‐infected group treated with novel phosphorylated compounds. Further examinations of immuno colocalization, PCR, and flow cytometry analysis show that the novel phosphorylated compounds are effective in inhibiting the fusion protein expression, which is important in replication of NDV virus. The significant finding of this study is that novel phosphorylated antiviral compounds ABC‐1 protect the NDV‐infected chicken through its antiviral properties. In summary, the novel phosphorylated abacavir (ABC‐1) potentially reduces the NDV‐induced tissue oxidative stress and inhibits the expression of fusion protein.
The authors declare no conflict of interest.
6. Acknowledgements
Dr. Lokanatha Valluru is thankful to SERB (No. SR/SO/AS‐021/2013 dt.29.11.2013), New Delhi, for providing financial assistance in the form of research grant. We are also thankful to Lisette Cornelissen DVM, PhD, Central Veterinary Institute, Wageningen UR, Edelhertweg, The Netherlands, for generous gift of fusion protein mAb anti‐F mAb 8E12A8C3. I am thankful to Dr. Bindu Madhava Reddy Armati for his technical support.
7. References
- 1. Zhu, W. , Dong, J. , Xie, Z. , Liu, Q. , and Khan, M. I. (2010) Virus Genes 40, 231–235. [DOI] [PubMed] [Google Scholar]
- 2. Wentao, F. , Yang, W. , Shenghua, W. , Ziqiang, C. , Huijun, G. , Xiaona, Z. , and Jianzhu, L. (2017) Res. Vet. Sci. 111, 49–54.27951521 [Google Scholar]
- 3. Kuiken, T. , Heckert, R. A. R. J. , Leighton, F. A. , and Wobeser, G. (1998) Avian Pathol. 27, 541–546. [DOI] [PubMed] [Google Scholar]
- 4. Alexander, D. J. (2000) Avian Pathol. 29, 95–100. [DOI] [PubMed] [Google Scholar]
- 5. King A. M. Q., Adams M. J., Carstens E. B., and Lefkowitz E. J. (eds.). (2011) In Virus Taxonomy—Ninth Report of the International Committee on Taxonomy of Viruses. Pp. 1273–1277, Elsevier/Academic Press, London. [Google Scholar]
- 6. Steward, M. , Vipond, I. B. , Millar, N. S. , and Emmerson, P. T. (1993) J. Gen. Virol. 74, 2539–2547. [DOI] [PubMed] [Google Scholar]
- 7. Tony, M. , and Sviatlana, A. (2010) Biotechnol. Appl. Biochem. 56, 103–109.20649513 [Google Scholar]
- 8. Lamb, R. A. (1993) Virology 197, 1–11. [DOI] [PubMed] [Google Scholar]
- 9. Regina, E. G. , Elizabeth, C. S. L. , Denis, R. M. , Mendoza, E. G. , Cristina, R. P. , and Laura, M. T. A. (2012) Virol. J. 9, 307.23234372 [Google Scholar]
- 10. Valyi‐Nagy, T. , and Dermody, T. S. (2005) Histol. Histopathol. 20, 957–967. [DOI] [PubMed] [Google Scholar]
- 11. Estevez, M. (2015) Poult. Sci. 94, 1368–1378. [DOI] [PubMed] [Google Scholar]
- 12. Takaaki, A. (2001) Rev. Med. Virol. 11, 87–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Akaike, T. , Noguchi, Y. , Ijiri, S. , Setoguchi, K. , Suga, M. , Zheng, Y. M. , Dietzschold, B. , and Maeda, H. (1996) Proc. Natl. Acad. Sci. USA 93, 2448–2453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Venkata Subbaiah, K. C. , Lokanatha, V. , Rajendra, W. , Ramamurthy, C. , Thirunavukkarusu, C. , and Subramanyam, R. (2015) Int. J. Biochem. Cell Biol. 64, 97–106. [DOI] [PubMed] [Google Scholar]
- 15. Terry, L. , Sprinz, E. , Stein, R. , Medeiros, N. B. , Oliveira, J. , and Ribeiro, J. P. (2006) Med. Sci. Sports Exerc. 38, 411–417. [DOI] [PubMed] [Google Scholar]
- 16. Lalanne, M. , Andrieux, K. , and Couvreur, P. (2009) Curr. Med. Chem. 16, 1391–1399. [DOI] [PubMed] [Google Scholar]
- 17. Zhang, Y. , Gao, Y. , Wen, X. , and Ma, H. (2014) Asian J. Pharm. Sci. 9, 65–74 [Google Scholar]
- 18. Hotard, A. L. , He, B. , Nichol, S. T. , Spiropoulou, C. F. , and Lo, M. K. (2017) Antiviral Res. 144, 147–152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Daluge, S. M. , Good, S. S. , and Faletto, M. B. (1997) Antimicrob. Agents Chemother. 41, 1082–1093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Pang, W. , Tam, S. C. , and Zheng, Y. T. A . (2009) Chem. Chemother. 20, 1–18. [DOI] [PubMed] [Google Scholar]
- 21. Brittain, H. G. (2002) Analytical Profiles of Drug Substances and Excipients. Academic Press, New York. [Google Scholar]
- 22. Youcef, M. , Jan, B. , and Christopher, M. (2009) Chem. Med. Chem. 4, 1779–1791.19760699 [Google Scholar]
- 23. Suresh, K. A. , Venkata Subbaiah, K. C. , Lavanya, R. , Chandrasekhar, K. , Naga Raju, C. , Suresh, M. K. , Rajendra, W. , and Lokanatha, V. (2016) Appl. Biochem. Biotechnol. 180, 361–381. [DOI] [PubMed] [Google Scholar]
- 24. Khan, J. , David, M. B. , Bradley, N. , Gao, B. , Bruckdorfer, R. , and Jacob, M. (1998) Biochem. J. 330, 795–801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Smith, M. A. , Sayre, L. M. , Anderson, V. E. , Harris, P. L. R. , Beal, M. F. , Kowall, N. , and Perry, G. (1998) J. Histochem. Cytochem. 46, 731–735. [DOI] [PubMed] [Google Scholar]
- 26. Laemmli, U. K. (1970) Nature 227, 680–685. [DOI] [PubMed] [Google Scholar]
- 27. Greenacre, S. A. , and Ischiropoulos, H. (2001) Free Radic. Res. 34, 541–581. [DOI] [PubMed] [Google Scholar]
- 28. Lori, W. M. , Kathryn, G. , and Trudy, G. M. (2002) J. Virol. 76, 12622–12633.12438588 [Google Scholar]
- 29. Ilaria, C. , and Dennis, J. A. (2001) Avian Influenza and Newcastle Disease. A Field and Laboratory Manual. Springer, Milan, Italy. [Google Scholar]
- 30. Sarah, E. , James, M. , Judith, L. , David, F. , and John, M. L. (1988) Cytometry 9, 9264–9260. [Google Scholar]
- 31. Vetrivel, S. C. , and Chandrakumarmangalam S. (2013) Braz. J. Poultry Sci. 15, 287–294. [Google Scholar]
- 32. Alexander, D. J. (2001) Br. Poult. Sci. 42, 5–22. [DOI] [PubMed] [Google Scholar]
- 33. Mohammad, L. R. , Yi‐Che, S. , and Jiann, R. , Hong, R. (2014) Int. J. Cell. Biol. 467452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Peterhans, E. (1979) Biochem. Biophys. Res. Commun. 91, 383–392. [DOI] [PubMed] [Google Scholar]
- 35. Rehman, Z. U. , Meng, C. , Sun, Y. , Safdar, A. , Pasha, R. H. , Munir, M. , and Ding, C. (2018) Oxid. Med. Cell. Longev. 2018, 5123147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Xiong, W. , Chen, Y. , Yu, W. , and Jiaguo, L. (2014) BMC Vet. Res. 10, 226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Emily, S. (2000) Drug Metab. Rev. 32, 307–326. [DOI] [PubMed] [Google Scholar]
- 38. Butterfield, D. A. , Marzia, P. , Tanea, R. , Tasneem, M. , Christopher, P. H. , Robinson, R. A. , and Sultana, R. (2012) Antioxid. Redox Signal. 17, 1610–1655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Shin‐Hee, K. , Nanchaya, W. , Anandan, P. , Sa, X. , Peter, L. C. , and Siba, K. S. (2013) PLoS One 8, 1–10. [Google Scholar]
- 40. Ailing, L. , Youxiang, D. , Hao, C. , Jiao, W. , Pingping, G. , Xiaoyan, S. , and Dongmin, H. (2014) Avian Pathol. 43, 325–332. [DOI] [PubMed] [Google Scholar]
- 41. Melbourne, T. , Jong‐Soo, L. , Hojin, M. , Mohammed, Y. E. C. , and Chul, J. K. (2014) Adv. Virol. 9, 301386. [Google Scholar]


