ABSTRACT
Therapies in oncology have evolved rapidly over the last years. At the same pace, supportive care for patients receiving cancer therapy has also evolved, allowing patients to safely receive the newest advances in treatment in both an inpatient and outpatient basis. The recognition of the role of infection control and prevention (ICP) in the outcomes of patients living with cancer has been such that it is now a requirement for hospitals and involves multidisciplinary groups. Some unique aspects of ICP for patients with cancer that have gained momentum over the past few decades include catheter‐related infections, multidrug‐resistant organisms, community‐acquired viral infections, and the impact of the health care environment on the horizontal transmission of organisms. Furthermore, as the potential for infections to cross international borders has increased, alertness for outbreaks or new infections that occur outside the area have become constant. As the future approaches, ICP in immunocompromised hosts will continue to integrate emerging disciplines, such as antibiotic stewardship and the microbiome, and new techniques for environmental cleaning and for controlling the spread of infections, such as whole‐genome sequencing. CA Cancer J Clin 2018;000:000‐000. © 2018 American Cancer Society.
Keywords: immunosuppression, infection control, infectious disease, prevention
Introduction
Hippocrates (circa 460‐377 BCE) originally recognized the effects of our surroundings on human diseases in his treatise On Airs, Waters and Places, attributing illness to characteristics of climate, water, modes of life, and nutrition.1, 2 Two thousand years later, in the 1800s, Ignaz Semmelweis (1818‐1865 CE) documented the effects of environmental control through hand hygiene on clinical outcomes, achieving a dramatic decrease in puerperal mortality with the widespread use of aseptic techniques, in which practitioners cleaned their hands with chlorine solution in between patients.3, 4
Although it took decades before the medical community accepted this discovery, infection control practices have changed the practice of medicine, have improved patients' outcomes, and have become the law of the land.5, 6 For immunocompromised patients, infection control strategies are a fundamental part of modern oncologic care and comprise a multilevel approach, including the patient, the health care environment, the community, and health care workers (Fig. 1).7, 8, 9 Guidelines for infection control and prevention (ICP) in patients with hematologic or oncologic malignancies are centered on recommendations for hematopoietic cell transplant (HCT) recipients10 and have typically been based on principles of hand hygiene, air quality, barrier isolation (eg, the use of gowns, gloves, masks, and eye protection, depending on the type of exposure), endogenous flora suppression by prophylactic antibiotics, and the prevention of device‐related infections (eg, central venous catheters and urinary catheters).11, 12 An equally relevant aspect of ICP for patients with cancer is the recognition of higher rates of colonization and infections by multidrug‐resistant organisms (MDROs), such as vancomycin‐resistant Enterococcus (VRE), methicillin‐resistant Staphylococcus aureus (MRSA), and multidrug‐resistant Gram‐negative bacilli (MDR‐GNB), and Clostridium difficile compared with the general patient population.11, 13, 14 Well known risk factors associated with the transmission of these MDROs include: hematologic malignancy; neutropenia; frequent contact with the health care environment; multiple and/or prolonged hospitalizations; devices, including urinary catheters and central lines; as well as changes in the microbiome driven by the use of antimicrobial agents and chemotherapy.13, 14, 15, 16 The reported colonization rates by MDROs in various populations of patients with cancer are from 4.7% to 36% for VRE,15, 16 from 5% to 10% for MRSA,17 from 7% to 18% for C. difficile,18, 19 and from 3% to 29% for MDR GNB.14, 20, 21, 22 Moreover, intestinal colonization with VRE and extended‐spectrum β‐lactamases (ESBL) Escherichia coli has been associated with an increased risk ratio (RR) of developing bloodstream infection (BSI) with these organisms (ESBL BSI: RR, 4.5; VRE BSI, RR, 10.2).22
Figure 1.

Important Aspects of Infection Control and Prevention in Patients Living With Cancer.
Emerging disciplines, such as outpatient cancer care and the entailed infection control practices (including up‐to‐date immunizations, community respiratory viruses, and prolonged shedding),23 the role of antibiotic stewardship,24 and the use of whole‐genome sequencing (WGS) for outbreak investigations,25 have great applications in ICP for patients living with cancer. However, there is no clear consensus or guidance on the variety of ICP strategies; recommendations for these practices are mainly center‐adapted.26, 27 In this review, we discuss key aspects of robust, comprehensive ICP programs, not only for hospitals specialized in cancer but for all centers that care for oncological patients, including practical algorithms and new approaches and technologies for enhancing these practices.
Preventing Infection
Hand Hygiene
Washing hands remains the main transmission‐based precaution for preventing the spread of pathogens, and it has been the foundation of every ICP program.28, 29, 30 Hand hygiene must be on the frontline in the care of patients living with cancer, because health care‐associated pathogens can be recovered from infected or draining wounds,31, 32 from colonized areas of a patient's intact skin,33 and from the patient's surrounding environment.34, 35 In 2006, the World Health Organization launched the “My Five Moments for Hand Hygiene” campaign, which defines key moments to perform hand hygiene based on known mechanisms of microbe cross‐transmission between patients, the environment, and health care workers, including: 1) before touching a patient; 2) before cleaning/aseptic procedures; 3) after body fluid exposure/risk of such exposure; 4) after touching a patient; and 5) after touching a patient's surroundings.36
Simple hand washing with soap and water will remove almost all transient Gram‐negative rods in 10 seconds, and most recent evidence does not support the use of antimicrobial soap over regular soap and water.36 Alcohol‐based products have shown superior activity over water and regular soap, both before and after contact with patients, except in the case of exposure to C. difficile or norovirus pathogens.37 The use of alcohol‐based products may be associated with increased compliance of approximately 25%.38 Regardless of the product used, hand washing is an important modality for the prevention of infection and should be performed by patients, visitors, and health care workers.39
Average compliance with hand washing for health care workers in medical and surgical intensive care units of private tertiary care hospitals reportedly ranges from 40% to 50%.40, 41 Reports on compliance with hand washing in oncologic centers are scarce, with a few publications on self‐reported rates between 80% to 90% in a pediatric oncology practice in Italy42 and 90% at a hematology unit in Brazil.43 Research and long‐term quality‐improvement projects are needed to develop reliable and sustainable methods to ensure compliance with hand hygiene.43 A study by Pittet et al showed an association between improved hand washing compliance, increasing from 48% to 66%, and a 40% overall decrease in the rate of nosocomial infections.31
In addition to strict hand hygiene, health care workers in cancer centers should avoid wearing artificial nails or extenders, because these have been associated with the transmission of pathogens, including Gram‐negative organisms.20
Dietary Principles
On the basis of current evidence, following standard principles (such as avoiding unwashed fruits and vegetables as well as undercooked meats, seafood, and eggs) is advised for patients undergoing cancer treatment.44, 45, 46 The US Department of Agriculture recommendations for food safety for patients with cancer include: 1) consumption of only pasteurized juices and dairy products; 2) washing hands in warm, soapy water before handling, preparing, and eating food; 3) consuming food that has not passed the expiration date; and 4) storing raw meat, fish, and chicken carefully in wrapped containers to avoid spillage of juice onto other foods. Notably, these recommendations do not restrict fresh fruits and vegetables.47 Stricter restrictions are usually applied for stem cell transplant (HCT) recipients and neutropenic patients with an absolute neutrophil count below 500 cells/mm3 and include avoidance of raw fruits and vegetables, undercooked meats, unpasteurized milk and cheeses, and well water from private or public wells.48 However, there is no clear evidence that strict dietary restrictions are associated with a lower risk for infectious complications. Two recent studies have questioned the value of a neutropenic diet. A randomized study in 150 pediatric patients who were receiving myelosuppressive chemotherapy in which groups were assigned to either a neutropenic diet or US Food and Drug Administration‐approved food‐safety guidelines found no difference in the prevention of infections.49 In fact, a retrospective evaluation of 726 patients at Northwestern Memorial Hospital reported a higher rate of infections in HCT recipients who followed a neutropenic diet compared with those who consumed a general hospital diet.50
Antibiotic and Antifungal Prophylaxis
Antibiotic prophylaxis is commonly followed in patients with hematologic malignancies and solid tumors who receive myeloablative therapy and develop profound neutropenia as well as in patients in the early post‐transplant period. This approach has previously been shown to reduce the risk of all‐cause mortality (RR, 0.52; 95% confidence interval [95% CI], 0.35‐0.77) in earlier publications,7, 24, 44, 48 and there was an increased rate of GNB infections after quinolone prophylaxis was discontinued.51 However, in a recent literature review from the European Conference on Infections in Leukemia, the authors concluded that, although quinolones were associated with a lower rate of Gram‐negative bacteremia, they did not have an impact on mortality.52 Therefore, the practice of quinolone prophylaxis needs to be constantly reevaluated, especially with regard to balancing antibiotic stewardship versus local susceptibilities to quinolones and an increased risk for drug‐resistant infections, particularly with E. coli and C. difficile.53, 54
Antifungal prophylaxis is used mostly in certain patients with high‐risk cancer, such as patients with hematologic malignancies undergoing induction chemotherapy and high‐risk transplant recipients, to decrease the incidence of Candida and Aspergillus spp. infections.48, 55 This practice has enabled the transfer of care from the inpatient to the outpatient setting for some chemotherapy regimens and low‐risk transplant procedures.56 The type of prophylaxis (eg, fluconazole vs broader triazoles with antimold properties) and the use of preemptive detection strategies (eg, galactomannan‐β‐D‐glucan assays and chest‐sinus computed tomography) are dictated by the host risk factors. High‐risk patients for whom broader antifungal prophylaxis is indicated can include allogeneic stem cell transplant recipients with graft‐versus‐host disease, patients with acute myeloid leukemia who have prolonged neutropenia, or those receiving induction chemotherapy.48, 56, 57
Protective Environment
Protective hospital environments have been integral to preventing infection in patients with hematologic malignancies, especially patients with leukemia or those undergoing HCT, especially in recent decades.58, 59 A 2009 meta‐analysis evaluated protective isolation measures and reported reductions in all‐cause mortality and in infections linked to the use of antimicrobial prophylaxis (antifungals and antibiotics) in combination with barrier isolation and air‐quality control (RR, 0.79; 95% CI, 0.72‐0.87),7 but not when isolation or air‐quality control was the only prevention measured used.
Guidelines for health care facilities that house HCT recipients recommend specialized ventilation systems, including the use of laminar air flow units capable of 12 air exchanges per hour and high‐efficiency particulate air (HEPA) filters. These filters maintain compliant indoor air quality by filtering 99.99% of particulates in the work area. Patient isolation units should use HEPA filtration with the capacity to remove particles greater than 0.3 μm, continuous pressure monitoring with positive air pressure between patients' rooms and the hallway, self‐closing doors, and well sealed rooms.10, 20 A primary purpose of HEPA filters and laminar flow is reducing the risk of aspergillosis in high‐risk patients (those with leukemia and those undergoing HCT); this reduction in risk has been shown in several studies.60 Likewise, patients and health care workers should avoid construction areas because there is an increased risk for mold infections through an airborne route at these locations, and outbreaks of fungal infections because of construction have been reported.61
At our institution, we implement an ICP plan during construction, renovation, and structural repair activities that includes a barrier between construction and patients' areas with the aim of maintaining indoor air quality to prevent Aspergillus and other potentially pathogenic molds from being generated or released into the air. At our center, any activity that disturbs existing building features, possibly causing the release of harmful dust, warrants specific institutional precautions. All construction planning must comply with indoor air‐quality requirements specified by an internal committee, and project contractors are required to provide personnel and equipment to contain and clean up dust and particulates in and around the work area, including dust mops, wet mops, adhesive walk‐off mats, mop buckets, HEPA‐filtered vacuums, and clean rags for removing dust inside and outside the construction site and from equipment.
Lastly, health care facilities are required to perform routine environmental (water) controls to prevent HAIs from water sources as hospital water systems are frequently identified as sources of health care‐associated infections (HAIs), especially in immunocompromised individuals. These controls include the upkeep of a quality‐managed water system, routine sampling and testing of water and surveillance for HAIs from water sources, and actions required in health care facilities if HAIs from water sources (eg, Pseudomonas infections, legionellosis, cryptosporidiosis, and atypical mycobacterial infections) are suspected.62, 63
Isolation Precautions
Other contact precautions include the use of gowns or gloves, placing patients in a private room, or cohorting patients, which have been used within health care facilities to help contain pathogens to a restricted location. Contact precautions are recommended for patients who have viral infections and MDROs identified either by screening or from evidence of active infection.11, 20, 64 The appropriate use of a mask and/or eye and face protection varies by type of pathogen, exposure, and other risks.
Because these interventions come with financial and social costs,65, 66 each center must delineate their own policies and periodically analyze the risks and benefits. There is growing knowledge regarding the role of the hospital environment and risk for nosocomial infections, which are discussed below.
Multidrug‐Resistant Organisms and Clostridium difficile
The main risk factors for the acquisition of MDROs in patients with cancer are admission to an intensive care unit in the past 3 months, previous receipt of antibiotic therapy, and the use of a urinary catheter.67 Patients with hematologic malignancies (acute leukemia in particular) or HCT recipients, who experience extended periods of neutropenia, are at the highest risk for complications because of MDROs and have a high subsequent mortality rate of up to 80%.68 Outbreaks because of MDROs in cancer centers have been reported, mainly from Enterococci and drug‐resistant Gram‐negative organisms.69 Surveillance studies have demonstrated how being admitted to a hospital room that has been occupied by a preceding patient colonized with antibiotic‐resistant bacteria could be a risk factor for acquisition of such organisms,70 including MRSA,71 VRE,72 and C. difficile.73
Currently, there are not clear guidelines for the detection of MDRO colonization during hospitalization. The type and frequency of screening MDROs and the audience to which screening is directed varies among institutions owing to differences in patient populations, including high‐risk inpatient groups, prevalence, and these measures also can change in an outbreak setting.13, 74 How often patients need to be screened is a matter of debate and depends on the type of organism and the center (Fig. 2). Other unanswered questions include the screening modality and the target organisms. The recent Society for Health Care Epidemiology of America (SHEA) guidelines highlight the benefits of screening for VRE colonization75; consequently, several institutions such as ours perform rectal screening for VRE in the hematologic services and intensive care units, both on admission and once a week.11 Data are less clear between ESBL carriage and subsequent infections or outcomes, or the benefit of screening for MDR Gram‐negative bacilli (MDR‐GNB).76 Recent publications suggest the need to screen patients or travelers from regions with high endemicity of MDR‐GNB, such as the Middle East and Asia, or regions with recent outbreaks.68 Recommendations are clear, however, that, once MDROs are detected, hospitalized patients should be on contact precautions to prevent horizontal transmission or hospital spread.11, 20 Most recommendations to reduce the transmission of MDROs in hospitalized patients involve a bundle of best practices, including hand hygiene, active screening of patients with swabs for cultures, contact barrier precautions, enhanced environmental cleaning, decolonization in the case of MRSA, and antimicrobial stewardship.20, 68
Figure 2.

Suggested Infection‐Control Practices for Multidrug‐Resistant Organisms (MDROs) in Cancer Centers.
Some practices, such as antiseptic whole‐body washing or bathing of patients, have been shown by some researchers to reduce VRE and MRSA colonization and infections in hematologic units77, 78, 79 and have been proposed by some for selected patients with recurrent MRSA soft tissue infections.80 However, this impact has not been reproduced in other studies.81 Interestingly, in a recent report, the use of a chlorhexidine wash was associated with an increased risk of infections caused by MDR‐GNB, including carbapenem‐resistant Enterobacteriaceae,79 as well as the development of reduced susceptibility to chlorhexidine and the presence of efflux‐mediated resistance genes in staphylococci.82
Multidrug‐resistant gram‐negative bacteria
Gram‐negative organisms can develop resistance to β‐lactams or carbapenems and many other antimicrobials, including quinolones, aminoglycosides, and polymyxins. The most common MDR‐GNBs are ESBL‐producing organisms, MDR‐Pseudomonas aeruginosa, MDR‐Acinetobacter spp., and carbapenem‐resistant Enterobacteriaceae. A recent systematic review on the transmission of MDR‐GNB showed that the hospital water environment was a key factor for transmission of MDR‐GNB and for nosocomial infections, including clonal spread of MDR‐Klebsiella spp.,83 which has been linked to several nosocomial outbreaks.35, 84
Active surveillance for MDR‐GNB in patients without signs or symptoms of such infections is not recommended, except in the situation of an outbreak or infections with high prevalence at a specific ward or unit. However, in cases of outbreaks or a high prevalence of MDR‐GNB, the application of bundle interventions can be effective as part of infection control measures in intensive care units.85 A multifaceted intervention in an oncological intensive care unit, including hand hygiene, contact precautions, and patient screening upon intensive care unit admission and weekly thereafter, along with environmental cleaning, resulting in a sustained decrease of MDR‐Pseudomonas infection and colonization86; however, the effectiveness and sustainability of these best practices in the long term are still undetermined.86
Vancomycin‐resistant enterococci
VREs usually are not highly pathogenic and tend to cause more colonization than infection. However, in a recent meta‐analysis that included adult and pediatric patients diagnosed with VRE infection, VRE was associated with an increase in hospital mortality, even in a nonimmunocompromised population.87 VRE colonization rates in HCT recipients are 6% to 40% at admission, and VRE colonization has been associated with an increased risk for VRE BSIs.88, 89 In patients with hematologic malignancies, active surveillance for VRE by rectal swabs on admission and weekly thereafter, with subsequent isolation if the patient is VRE‐positive, has been shown to decrease the incidence of VRE nosocomial infections by decreasing rates of nosocomial transmission.15, 16, 75, 89
Clostridium difficile
Collateral damage from antimicrobial use includes alterations in the normal intestinal microbiota, creating the right environment for C. difficile infection (CDI).41, 42 Several studies have demonstrated that the risk for C. difficile is higher in HCT and solid organ transplant recipients than for other hospitalized or surgical patients, particularly in the setting of graft‐versus‐host disease, given the potential for damage to the gut luminal mucosa and the need for additional immunosuppression.14, 18 Incidence has been estimated between 5% and 27%, with higher rates in patient after HCT.14 Environmental contamination by C. difficile spores plays a major role in horizontal transmission to patients and subsequent infections.90 Certain strains of C. difficile that are known to hypersporulate have been linked to several outbreaks.91 Therefore, patients with C. difficile should be placed under contact precautions; all personnel should wear gowns and gloves, whether or not they anticipate touching the patient's environment. Hand washing with soap and water and thorough cleaning of all potentially contaminated surfaces with a 1:10 dilution of concentrated sodium hypochlorite are recommended and may reduce the environmental burden of C. difficile.92 A multicenter survey to determine the rates of CDI in patients with cancer and in HCT recipients assessed isolation practices and found great variations across all centers. Most centers kept patients on isolation until the resolution of gastrointestinal symptoms, but few centers did so for the entire duration of hospitalization.93
Viral Infections
Respiratory viruses
Respiratory viral infections can cause high morbidity and mortality in patients with cancer. Respiratory viruses spread primarily through exposure to respiratory droplets expelled during coughing or sneezing from infectious individuals. Droplet transmission is associated with particles greater than 5 μm in diameter that do not remain suspended in the air, and airborne transmission is associated with particles 5 μm or less in diameter that remain suspended in the air for a long time.94 Current ICP measures for respiratory viruses are intended to prevent droplet, contact and, for some viruses, airborne transmission.95 Furthermore, outbreak of respiratory viral infections can be fatal in some patients receiving cancer therapy.96, 97, 98 Therefore, preventing the exposure and spread of respiratory viruses is of utmost importance and involves not only patients but also visitors and hospital personnel. Health care workers and visitors with respiratory viral infections should abstain from direct contact with immunocompromised patients until symptoms of infection resolve.
A lack of data precludes recommendations regarding the routine testing of asymptomatic patients for respiratory virus infections. However, active surveillance of patients living with cancer who have signs and symptoms of respiratory viral infection is strongly indicated.23, 99 Some respiratory viruses, including respiratory syncytial virus, influenza viruses, and human metapneumovirus, are seasonal and occur most commonly in winter; however, perennial infections have been reported with other viruses, such as parainfluenza viruses, adenovirus, rhinovirus, and coronavirus.99, 100 Patients who have symptoms compatible with respiratory viral infection should be placed on contact precautions while diagnosis is underway.99, 101, 102, 103
A recent meta‐analysis by Cochrane showed that the spread of respiratory viruses can be prevented by hygienic measures, such as barrier precautions and hand washing.101 Different modalities of isolation by viruses are portrayed in Table 1,94, 102, 104, 105, 106, 107, 108, 109, 110, 111, 112, 113, 114, 115, 116, 117, 118, 119, 120, 121, 122, 123 including current recommendations by the American Society of Blood and Marrow Transplantation guidelines and standard practices at our own institution. Emerging data regarding enhanced isolation precautions, including contact and droplet precautions for all respiratory viruses, showed a 39% reduction in nosocomial infections.124 Furthermore, the importance of health care workers' compliance with masking was evident in a recent study from Duke University Medical Center, in which universal masking was associated with a reduction in respiratory viral infections.125 While this practice has several drawbacks, including long‐term compliance and the possible perception of a barrier between patient and provider, it does bring up the importance of health care workers' compliance with ICP, especially during the active respiratory season and during outbreaks.
Table 1.
Recommendations for Contact Precautions for Respiratory Viral Infections
| PRECAUTIONS RECOMMENDED | ||||
|---|---|---|---|---|
| VIRUS | MODE OF TRANSMISSION | ASBMT | MD ANDERSON CANCER CENTER | REPORTS OF OUTBREAKS IN HEMATO‐ONCOLOGY UNITS |
| Respiratory syncytial virus | Small and large droplets and fomites102 | Contact | Droplet and contact | Stem cell transplant units104, 105, 106 |
| Parainfluenza viruses | Large droplets and fomites107 | Contact | Droplet and contact | Pediatric108 and adult hematology109 |
| Influenza viruses | Large and small droplets and fomites94, 110 | Droplet | Droplet and contact | Pediatric hematologic unit111 and pediatric oncology112 |
| Adenovirus | Large and small droplets and fomites113 | Droplet and contact | Droplet and contact | Stem cell transplant units114, 115 |
| Coronavirus | Large droplets and fomites116, 117 ,a | Contact | Droplet and contact | No reports in patients with cancera |
| Rhinovirus | Large droplets and fomites, with recent data indicating small droplets117, 118 | Contact | Droplet and contact | Hemato‐oncology wards119 |
| Human metapneumovirus | Small droplets, close contacts, and fomites120 | No recommendation | Droplet and contact | Hematology unit121 |
Abbreviation: ASBMT, American Society of Bone Marrow Transplantation. aReports on Middle East respiratory syndrome in the nonimmunocompromised population indicate that the virus can be transmitted on large droplets and fomites, and there is potential for animal‐to‐human transmission. Adapted from: Ho KY, Singh KS, Habib AG, et al. Mild illness associated with severe acute respiratory syndrome coronavirus infection: lessons from a prospective seroepidemiologic study of health‐care workers in a teaching hospital in Singapore. J Infect Dis. 2004;189:642‐647122; and Patrick DM, Petric M, Skowronski DM, et al. An outbreak of human coronavirus OC43 infection and serological cross‐reactivity with SARS coronavirus. Can J Infect Dis Med Microbiol. 2006;17:330‐336.123
In addition, in immunocompromised patients, shedding of respiratory viruses can be prolonged to more than 30 days and, in some instances, up to 160 days117, 126, 127 or even a period of years, especially in transplant recipients on steroids.126 Prolonged shedding of respiratory viruses in an immunocompromised host needs to be considered when establishing ICP in the inpatient and outpatient setting to limit horizontal transmissions.126
Gastrointestinal Viruses
Gastrointestinal viruses are most commonly transmitted by the fecal‐oral route, but reports of airborne transmission also have been established.128 Several nosocomial outbreaks have been linked to norovirus and rotavirus in pediatric oncology units in association with shared toys.129, 130, 131 These organisms can survive on nonporous surfaces for several days and require strict infection control precautions, including contact precautions and environmental cleaning.131 In the case of norovirus, soap and water are needed for hand washing, and sodium hypochlorite is required for environmental cleaning.132 As with respiratory viruses, prolonged shedding from the gastrointestinal tract is common and has been linked to nosocomial infections.133
Catheter‐Related Infections
Important risk factors associated with hospital‐acquired infections in patients living with cancer are the use of invasive medical devices, especially central lines, indwelling urinary catheters, and intratracheal tubes. In this section, we discuss central line‐associated BSIs (CLABSIs).
In patients receiving oncological treatments, central venous catheters are commonly used for venous access for chemotherapy, blood transfusions, and intravenous fluid administration. The presence of these catheters is associated with increased risk for BSIs.134 CLABSI is a surveillance definition used by the Centers for Disease Control and Prevention (CDC) and the National Health Safety Network as follows: recovery of a pathogen from a blood culture (a single blood culture for an organism not commonly present on the skin and 2 or more blood cultures for an organism commonly present on the skin) in a patient who had a central line at the time of infection or within 48 hours before the development of infection. CLABSIs also must meet the following criteria: 1) the patient has a recognized pathogen culture from one or more blood cultures, and the organisms cultured are not related to an infection at another site; and 2) the patient has at least one of the following signs or symptoms within 24 hours: fever, chills, and hypotension.135 Furthermore, in 2013, the National Health Safety Network and the CDC added a definition for CLABSI that applies to patients with hematologic malignancies and/or HCT recipients: mucosal barrier injury laboratory‐confirmed BSI, which is defined by either only an intestinal organism or only viridans group streptococcus; by allogeneic transplant within the past year with grade 3 or 4 graft‐versus‐host disease, or more than a single liter of diarrhea, or neutropenia with an absolute neutrophil count below 500 cells/mm3 within 7 days of the positive cultures.136, 137
In recent studies, the reported catheter‐related infection rate in adults with cancers was from 0.02 to 3 per 1000 catheter‐days,138 and the incidence of exit‐site infection ranged from 1.9% to 60.9%.139, 140 Risk factors reported for CLABSI in patients with cancer include thrombosis, difficulty during the insertion procedure, total parenteral nutrition, neutropenia, age, hematologic malignancies, and HCT.138, 141, 142
The central venous port has grown in importance in cancer centers because of its advantages over central venous catheters, including the reduction of contamination of the device by external or skin pathogens, with an incidence of port‐related infection that varies between 0.9% and 5.4%.143, 144 Compared with the incidence of CLABSI for tunneled lines (hazard ratio, 1.77; P ≤ .011), nontunneled central venous catheters have a higher incidence of infection (hazard ratio, 3.50; P < .0001),142, 145, 146 with the exception of peripherally inserted central catheters, in which the incidence rate of infection has been low, between 0.5 and 0.95 per 1000 catheter‐days.147, 148
In terms of site, CLABSI is generally more common when catheters are inserted in the femoral veins and, probably to a lesser degree, in the internal jugular veins compared with the subclavian veins.149, 150
The most commonly reported causative pathogens in catheter‐related infections remain coagulase‐negative staphylococci, S. aureus, Enterococcus spp., and Candida spp. Gram‐negative organisms accounted for 19% of CLABSIs reported to the CDC and for 21% of CLABSIs reported to the Prevention's National Healthcare Safety Network (NHSN).151, 152 Because bacteria can enter the catheter through migration of skin organisms, the most common etiology of CLABSI is the contamination of the hub by hand manipulation or blood products.134, 153, 154 Thus aseptic techniques during catheter insertion, specialized “intravenous teams,” and postinsertion care bundles are best practices that have been shown to decrease the rates of CLABSI, especially for short‐term catheters.155, 156, 157 In addition, recognized postinsertion practices that reduce the rates of CLABSI include the use of chlorhexidine gluconate‐impregnated dressings,158 assessment of the insertion site, change of dressings weekly or as needed, scrubbing the hub for 15 seconds or more before access, clot prevention strategies, continuous reevaluation of the need for the catheter,134, 159, 160 and the use of an antiseptic barrier cap using alcohol‐impregnated port protectors. Regarding the barrier cap, 2 recent studies demonstrated a 34% reduction in hospital‐wide CLABSI rates.161, 162
Further modalities for the prevention of CLABSI, especially for long‐term catheters, include: 1) catheters impregnated with minocycline‐rifampin134, 163; and other antimicrobial and antiseptic preparations164 and 2) lock solutions containing minocycline and ethylene diamine tetraacetate (M‐EDTA),165 or ethanol locks.166 The use of ethanol locks lessens concern for antibiotic resistance compared with antibiotic‐coated catheters or antibiotic‐based lock solutions165, 167; however, concerns about protein precipitation with ethanol locks warrant further studies before these locks can be routinely recommended. Continuous education and training of health care workers, regular audits of bundle implementation, and engagement of patients and caregivers are key for the long‐term prevention of CLABSI (Fig. 3).
Figure 3.
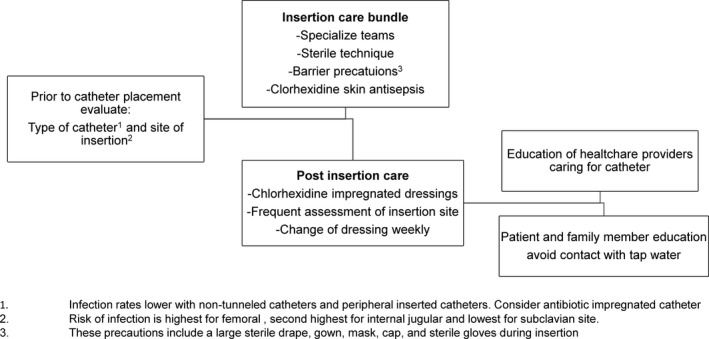
Algorithm for the Prevention of Central Line‐Associated Blood Stream Infection (CLABSI).
Surgical Site Infections
The incidence of surgical site infections (SSIs) in patients with oncological diagnosis has been reported to be between 3.2% and 7.9%168 and is similar to that of patients without malignancies. The most frequent organisms found at surgical sites in patients with malignancies are Gram‐positive bacteria, specifically S. aureus, as in the general population. However, the rates of MRSA infections can be higher in patients with cancer than in those without cancer (up to 40%),169 and, more recently, different authors have reported higher rates of GNB, P. aeruginosa, ESBL‐producing organisms, and other MDROs, such as VRE.169, 170, 171 In general, however, the isolated organisms depend on the type of surgery, as shown in a retrospective review from The University of Texas MD Anderson Cancer Center (MD Anderson), where S. aureus was the predominant organism in SSIs after breast cancer surgery, thoracotomy, craniotomy, and abdominal/pelvic surgery; however, 42% of SSIs were polymicrobial, with P. aeruginosa and E. coli being the predominant organisms among the GNBs.169 Prevention strategies for SSIs include enhanced nutritional support; preoperative bathing; decolonization with mupirocin ointment with or without chlorhexidine body wash for MRSA nasal carriers; surgical site preparation with alcohol‐based, antiseptic solutions that contain chlorhexidine gluconate; and, mainly, timing and stopping at 24 hours of the appropriate perioperative antimicrobial prophylaxis.169, 172
Environmental Cleaning
There is growing knowledge about the role of the health care environment as a reservoir of and vehicle for the transmission of various pathogens and how environmental cleaning and sterilization practices can help decrease the transmission of HAIs.35 For patients with cancer who are in constant contact with the health care environment, this role is of paramount importance. Survival times of organisms on dry, inanimate objects vary according to temperature, humidity, and surface type173, 174 and have been reported to be several days (>12 days) for viruses,175 2 months for MRSA, and up to 36 months for VRE.84, 176 These organisms can thereby be a continuous source of hospital‐associated transmission of infections.
Environmental cleaning is typically performed manually using disinfectant agents, including hypochlorous acid, hydrogen peroxide, and paracetic acid; however, studies have shown that manual cleaning is not predictable and depends on the education of cleaning personnel and nurses and the appropriate amounts of disinfectant solutions,177 with reports indicating that only about 47% of surfaces are appropriately disinfected.41, 178
Environmental cleaning also can be supplemented by using automated technologies (also known as no‐touch disinfection methods), including aerosol and hydrogen peroxide vapor systems such as microcondensation systems, which have shown effectiveness in cleaning MRSA, C. difficile, and Mycobacterium tuberculosis; and, although several studies have shown that these are effective decontamination methods (especially microcondensation systems), the costs and room turn‐around times have hampered adoption of this technology in health care settings,41 and mobile devices that emit continuous ultraviolet (UV‐C) light have become more popular. In particular, a portable UV light germicidal device using pulsed xenon lamps (PX‐UV) has been shown to be a safe, easy‐to‐operate, and effective system for decreasing the number of pathogens in the environment. PX‐UV uses a xenon flash lamp to generate broad‐spectrum, high‐intensity UV light to deactivate and kill bacteria, spores, and viruses on high‐touch surfaces in 5 minutes or less.179 PX‐UV was evaluated in clinical hematologic and bone marrow units, and it provided an overall reduction of 90% in total aerobic colony counts compared with a 76% reduction with manual cleaning.180 At MD Anderson, we observed that PX‐UV light was noninferior compared with bleach for decreasing C. difficile spores from patients' rooms,181 as described in a recent systematic review of over 20 studies in which the use of automated UV‐C light devices achieved a significant reduction in C. difficile infections (RR, 0.64; 95% CI, 0.49‐0.84) and VRE infections (RR, 0.42; 95% CI, 0.28‐0.65); however, there was no significant impact on the rates of infection with MRSA or MDR‐GNB.182 In a recent cluster‐randomized, multicenter, crossover study, there was a decrease in the target organisms (mainly C. difficile) in exposed patients after adding UV‐C light (using mercury devices) to standard cleaning, but this decreased rate was not associated with a change in the incidence of C. difficile infection.183 Therefore, further data are needed to clearly determine the benefits of UV light, but enhanced terminal cleaning may be part of the solution. Some of the drawbacks for the use of automated methods has been cost‐effectiveness and logistical problems, including the ability of the systems to reach certain areas, the need for furniture rearrangement in the rooms for its use, and longer cleaning times of an average 1 hour per room.184 More recently, we were able to demonstrate equivalent efficiency in reduction of colony counts (approximately 73%) on high‐touch surface areas between cycles of 2 and 8 minutes using the PX‐UV system at a single position in the operating rooms.185
Outbreak Management
The future of outbreak investigation will include the integration of WGS, which allows strain characterization and epidemiologic investigation and will likely replace traditional methods of identification, such as pulsed‐field gradient electrophoresis and other sequence‐based methods.186 WGS has yielded important insights into transmission pathways for several significant pathogens and has revealed outbreaks in situations in which standard infection control surveillance and definitions showed no indications of causative pathogens.104, 186 In a recent study at MD Anderson using WGS of VRE isolates, we demonstrated potential transmission networks between the patient and the environment within and between rooms as well as between patients within and between floors.25 The limitations of WGS include the need for effective semiautomated pipelines, standardized quality control and data interpretation, bioinformatics expertise, and the infrastructure's cost.187
Outpatient Infection Control
Because cancer centers deliver most of their care on an outpatient basis,188 ICP programs for this setting are of particular importance. Guidance for prevention in outpatient oncology settings has been published by the CDC. Key recommendations comprise the development of an outpatient infection prevention program that includes at least one individual with training in infection prevention, the establishment of infection prevention policies and procedures (ie, hand hygiene and standard precautions, use of personal protective equipment, injection safety, and environmental cleaning), as well as provision of the appropriate supplies necessary for adherence to standard precautions.23
Other important considerations beyond those of the CDC guidance include recommendations regarding diet; outdoor activities; hobbies; pet care; and immunizations of patients, family members, and health care personnel (Table 2).8, 23, 46, 189, 190 In addition, managing the access of visitors during the respiratory viral season (usually during winter time) by screening for respiratory illnesses at the point of entrance, alerting about hand and cough hygiene, and encouraging influenza vaccination are of utmost importance.23 Similar practices should be upheld for health care workers,125 particularly the receipt of influenza vaccination, because we recently demonstrated the relation between health care workers' vaccination and decreased rates of nosocomial influenza infection.191
Table 2.
Recommendations for Infection‐Control Practices in an Outpatient Settingsa
| ASPECT OF CARE | RECOMMENDATIONS MUST INCLUDE GUIDANCE ON |
|---|---|
| Standard precautions | Hand hygiene, use of personal protective equipment (including gloves, masks, and gowns) |
| Respiratory hygiene and cough etiquette | Identification of potential respiratory infections, including asking about and separation of persons with respiratory symptoms |
| Injection safety and central venous catheter care | Safe practices for procedures |
| Cleaning and disinfection of devices and environmental surfaces | Patient areas, bathrooms, waste disposal, and cleaning spills of blood and body substances |
| Transmission precautions | Contact, droplet, and airborne transmission precautions according to facility protocol |
| Medication storage and handling | Both outpatient centers and patients at home |
| Dietary recommendations | In general, avoid nonpasteurized milk products; cheese with molds; raw or undercooked meat, poultry, fish, seafood, game, and tofu; undercooked eggs; unwashed fruits and vegetables; fresh store bought salsa or salads; unroasted nuts |
| Immunizations | Follow recommendations by the CDC, the IDSA on immunizations, the risk for transmission, and receipt of live‐attenuated vaccines |
Abbreviations: CDC, Centers for Disease Control and Prevention; IDSA, Infectious Diseases Society of America. aAdapted from: Division of Healthcare Quality Promotion, National Center for Emerging and Zoonotic Infectious Diseases, Centers for Disease Control and Prevention (CDC). Basic Infection Control and Prevention Plan for Outpatient Oncology Settings. Atlanta, GA: CDC; 2011. http://cdc.gov/hai/pdfs/guidelines/basic-infection-control-prevention-plan-2011.pdf. Accessed October 17, 201723; Ariza‐Heredia EJ, Kontoyiannis DP. Our recommendations for avoiding exposure to fungi outside the hospital for patients with haematological cancers. Mycoses. 2014;57:336‐34146; and Sipsas NV, Kontoyiannis DP. Occupation, lifestyle, diet, and invasive fungal infections. Infection. 2008;36:515‐525.190
Antibiotic Stewardship
The main goal of antimicrobial stewardship is to help optimize the use of antibiotic therapy through several strategies (including education and the development of guidelines), to increase drug safety, and to avoid antibiotic overuse and consequent antimicrobial resistance.24, 192 Because of the high risk of infectious complications in patients receiving cancer care, as well as the increased risk for MDROs and C. diff, there is increased interest in the role and importance of antimicrobial stewardship in cancer centers.192 Several strategies used by different centers include antimicrobial cycling, antimicrobial restrictions, and computer‐based programs.24, 192 According to a recent survey in US transplant centers, some of the tests perceived as useful for guiding antibiotic therapy include respiratory viral panels, testing for azole levels, and serum/bronchoalveolar lavage galactomannan.27, 193
Future
The future of ICP practices in centers caring for patients with cancer will involve further work on environmental control, modern technologies that improve and facilitate cleaning of hospital surfaces, the role of microbiota‐fecal transplant in the control of MDROs, and the use of lytic bacteriophages as part of controlling antibiotic‐resistant bacteria not only in the clinical setting but also in applications to control bacterial food contamination.
Conclusions
The growth of the infection control discipline has played a vital role in the progress of cancer treatments, allowing patients to safely undergo new therapies. The application of current recommendations to cancer care and other health care environments must follow local patterns of infections; must continuously be reevaluated; and requires a multidisciplinary team, including infection control practitioners, physicians, nurses, and administrators, as well as a space for patients to voice their concerns. A good ICP program depends on current and open communication within the institution to ensure constant guidance on evolving infection control ICP practices, especially those that cover the needs of the immunosuppressed patient.
DISCLOSURES: Roy F. Chemaly reports grants and personal fees from Xenex, outside the submitted work. Ella J. Ariza‐Heredia reports no conflicts of interest.
References
- 1. Gourevitch D. Hippocratic medicine and the treatise Airs, Waters and Places. A short history of the beginnings and influence of a scientific error [article in Italian]. Med Secoli. 1995;7:425‐433. [PubMed] [Google Scholar]
- 2. Pappas G, Kiriaze IJ, Falagas ME. Insights into infectious disease in the era of Hippocrates. Int J Infect Dis. 2008;12:347‐350. [DOI] [PubMed] [Google Scholar]
- 3. Breido IS. The discovery and tragedy of Ignaz Semmelweis (on the 140th anniversary of his discovery) [article in Russian]. Akush Ginekol (Mosk). 1987;4:73‐74. [PubMed] [Google Scholar]
- 4. Jumaa PA. Hand hygiene: simple and complex. Int J Infect Dis. 2005;9:3‐14. [DOI] [PubMed] [Google Scholar]
- 5. Gostin LO. Public health law in a new century. Part I: law as a tool to advance the community's health. JAMA. 2000;283:2837‐2841. [DOI] [PubMed] [Google Scholar]
- 6. Stone PW, Pogorzelska‐Maziarz M, Reagan J, et al. Impact of laws aimed at healthcare‐associated infection reduction: a qualitative study [serial online]. BMJ Qual Saf. 2015;24:637‐644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Schlesinger A, Paul M, Gafter‐Gvili A, Rubinovitch B, Leibovici L. Infection‐control interventions for cancer patients after chemotherapy: a systematic review and meta‐analysis. Lancet Infect Dis. 2009;9:97‐107. [DOI] [PubMed] [Google Scholar]
- 8. Thom KA, Kleinberg M, Roghmann MC. Infection prevention in the cancer center. Clin Infect Dis. 2013;57:579‐585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Ramanathan T. Law as a tool to promote healthcare safety. Clin Gov. 2014;19:172‐180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Yokoe D, Casper C, Dubberke E, et al. Infection prevention and control in health‐care facilities in which hematopoietic cell transplant recipients are treated. Bone Marrow Transplant. 2009;44:495‐507. [DOI] [PubMed] [Google Scholar]
- 11. Ariza‐Heredia EJ, Chemaly RF. Infection control practices in patients with hematological malignancies and multidrug‐resistant organisms: special considerations and challenges. Clin Lymphoma Myeloma Leuk. 2014;14(suppl):S104‐S110. [DOI] [PubMed] [Google Scholar]
- 12. Tomblyn M, Chiller T, Einsele H, et al. Guidelines for preventing infectious complications among hematopoietic cell transplantation recipients: a global perspective. Biol Blood Marrow Transplant. 2009;15:1143‐1238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Heidenreich D, Kreil S, Nolte F, Hofmann WK, Miethke T, Klein SA. Multidrug‐resistant organisms in allogeneic hematopoietic cell transplantation. Eur J Haematol. 2017;98:485‐492. [DOI] [PubMed] [Google Scholar]
- 14. Trubiano JA, Worth LJ, Thursky KA, Slavin MA. The prevention and management of infections due to multidrug resistant organisms in haematology patients. Br J Clin Pharmacol. 2015;79:195‐207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Matar MJ, Tarrand J, Raad I, Rolston KV. Colonization and infection with vancomycin‐resistant Enterococcus among patients with cancer. Am J Infect Control. 2006;34:534‐536. [DOI] [PubMed] [Google Scholar]
- 16. Hefazi M, Damlaj M, Alkhateeb HB, et al. Vancomycin‐resistant Enterococcus colonization and bloodstream infection: prevalence, risk factors, and the impact on early outcomes after allogeneic hematopoietic cell transplantation in patients with acute myeloid leukemia. Transplant Infect Dis. 2016;18:913‐920. [DOI] [PubMed] [Google Scholar]
- 17. Schaefer AM, McMullen KM, Mayfield JL, Richmond A, Warren DK, Dubberke ER. Risk factors associated with methicillin‐resistant Staphylococcus aureus colonization on hospital admission among oncology patients. Am J Infect Control. 2009;37:603‐605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Gorschluter M, Glasmacher A, Hahn C, et al. Clostridium difficile infection in patients with neutropenia. Clin Infect Dis. 2001;33:786‐791. [DOI] [PubMed] [Google Scholar]
- 19. Neemann K, Freifeld A. Clostridium difficile‐associated diarrhea in the oncology patient. J Oncol Pract. 2017;13:25‐30. [DOI] [PubMed] [Google Scholar]
- 20. Sehulster L, Chinn RY; CDC; HICPAC . Guidelines for environmental infection control in health‐care facilities. Recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee (HICPAC). MMWR Recomm Rep. 2003;52:1‐42. [PubMed] [Google Scholar]
- 21. Han SB, Jung SW, Bae EY, et al. Extended‐spectrum β‐lactamase‐producing Escherichia coli and Klebsiella pneumoniae bacteremia in febrile neutropenic children. Microb Drug Resist. 2015;21:244‐251. [DOI] [PubMed] [Google Scholar]
- 22. Liss BJ, Vehreschild JJ, Cornely OA, et al. Intestinal colonisation and blood stream infections due to vancomycin‐resistant enterococci (VRE) and extended‐spectrum beta‐lactamase‐producing Enterobacteriaceae (ESBLE) in patients with haematological and oncological malignancies. Infection. 2012;40:613‐619. [DOI] [PubMed] [Google Scholar]
- 23. Division of Healthcare Quality Promotion, National Center for Emerging and Zoonotic Infectious Diseases, Centers for Disease Control and Prevention . Basic Infection Control and Prevention Plan for Outpatient Oncology Settings. Atlanta, GA: Centers for Disease Control and Prevention; 2011. http://cdc.gov/hai/pdfs/guidelines/basic-infection-control-prevention-plan-2011.pdf. Accessed October 17, 2017. [Google Scholar]
- 24. Abbo LM, Ariza‐Heredia EJ. Antimicrobial stewardship in immunocompromised hosts. Infect Dis Clin North Am. 2014;28:263‐279. [DOI] [PubMed] [Google Scholar]
- 25. El Haddad L, Ghantoji SS, Scarpino S, et al. Single nucleotide polymorphism (SNP) analyses reveal potential vancomycin-resistant enterococci (VRE) transmission networks between rooms and patients on stem cell transplant (SCT) and leukemia units. Oral Presentation at the 2018 BMT Meeting, Salt Lake City, UT (USA). Biology of Blood and Marrow Transplantation. 2018;24:S99‐S100. [Google Scholar]
- 26. Kruger WH, Hornung RJ, Hertenstein B, et al. Practices of infectious disease prevention and management during hematopoietic stem cell transplantation: a survey from the European Group for Blood and Marrow Transplantation. J Hematother Stem Cell Res. 2001;10:895‐903. [DOI] [PubMed] [Google Scholar]
- 27. Beam E, Keating MR, Razonable RR. A Survey of infection prevention and control practices among hematopoietic stem cell transplant centers. Infect Control Hosp Epidemiol. 2016;37:348‐351. [DOI] [PubMed] [Google Scholar]
- 28. Maki DG. The use of antiseptics for handwashing by medical personnel. J Chemother. 1989;1(suppl 1):3‐11. [DOI] [PubMed] [Google Scholar]
- 29. Doebbeling BN, Stanley GL, Sheetz CT, et al. Comparative efficacy of alternative hand‐washing agents in reducing nosocomial infections in intensive care units. N Engl J Med. 1992;327:88‐93. [DOI] [PubMed] [Google Scholar]
- 30. Siegel JD, Rhinehart E, Jackson M, Chiarello L; Health Care Infection Control Practices Advisory Committee . 2007 Guideline for isolation precautions: preventing transmission of infectious agents in health care settings. Am J Infect Control. 2007;35(10 suppl 2):S65‐S164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Pittet D, Allegranzi B, Sax H, et al. Evidence‐based model for hand transmission during patient care and the role of improved practices. Lancet Infect Dis. 2006;6:641‐652. [DOI] [PubMed] [Google Scholar]
- 32. Pittet D, Dharan S, Touveneau S, Sauvan V, Perneger TV. Bacterial contamination of the hands of hospital staff during routine patient care. Arch Intern Med. 1999;159:821‐826. [DOI] [PubMed] [Google Scholar]
- 33. Casewell M, Phillips I. Hands as route of transmission for Klebsiella species. BMJ. 1977;2:1315‐1317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Maki DG, Alvarado CJ, Hassemer CA, Zilz MA. Relation of the inanimate hospital environment to endemic nosocomial infection. N Engl J Med. 1982;307:1562‐1566. [DOI] [PubMed] [Google Scholar]
- 35. Bhalla A, Pultz NJ, Gries DM, et al. Acquisition of nosocomial pathogens on hands after contact with environmental surfaces near hospitalized patients. Infect Control Hosp Epidemiol. 2004;25:164‐167. [DOI] [PubMed] [Google Scholar]
- 36. World Health Organization . SAVE LIVES: Clean Your Hands: WHO's global annual call to action for health workers. Geneva, Switzerland: World Health Organization; 2017. http://who.int/gpsc/5may/en. Accessed October 5, 2017. [Google Scholar]
- 37. Boyce JM. Update on hand hygiene. Am J Infect Control. 2013;41(5 suppl):S94‐S96. [DOI] [PubMed] [Google Scholar]
- 38. Bissett L. Can alcohol hand rubs increase compliance with hand hygiene? Br J Nurs. 2002;11:1072, 1074‐1077. [DOI] [PubMed] [Google Scholar]
- 39. Siegel JH, Korniewicz DM. Keeping patients safe: an interventional hand hygiene study at an oncology center. Clin J Oncol Nurs. 2007;11:643‐646. [DOI] [PubMed] [Google Scholar]
- 40. Marra AR, Moura DF Jr, Paes AT, dos Santos OF, Edmond MB. Measuring rates of hand hygiene adherence in the intensive care setting: a comparative study of direct observation, product usage, and electronic counting devices. Infect Control Hosp Epidemiol. 2010;31:796‐801. [DOI] [PubMed] [Google Scholar]
- 41. Boyce JM. Modern technologies for improving cleaning and disinfection of environmental surfaces in hospitals [serial online]. Antimicrob Resist Infect Control. 2016;5:10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Livadiotti S, Milano GM, Serra A, Folgori L, Jenkner A, Castagnola E, Cesaro S, Rossi MR, Barone A, Zanazzo G, Nesi F, Licciardello M, De Santis R, Ziino O, Cellini M, Porta F, Caselli D, Pontrelli G. Infectious Diseases Working Group of the Associazione Italiana Ematologia Oncologia Pediatrica. A survey on hematology-oncology pediatric AIEOP centers; prophylaxis, empirical therapy and nursing prevention procedures of infectious complications. Haematologica. 2012;97:147‐50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Sodre da Costa LS, Neves VM, Marra AR, et al. Measuring hand hygiene compliance in a hematology‐oncology unit: a comparative study of methodologies. Am J Infect Control. 2013;41:997‐1000. [DOI] [PubMed] [Google Scholar]
- 44. Centers for Disease Control and Prevention; Infectious Disease Society of America; American Society of Blood and Marrow Transplantation . Guidelines for preventing opportunistic infections among hematopoietic stem cell transplant recipients. MMWR Recomm Rep. 2000;49:1‐125, CE121‐127. [PubMed] [Google Scholar]
- 45. Gardner A, Mattiuzzi G, Faderl S, et al. Randomized comparison of cooked and noncooked diets in patients undergoing remission induction therapy for acute myeloid leukemia. J Clin Oncol. 2008;26:5684‐5688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Ariza‐Heredia EJ, Kontoyiannis DP. Our recommendations for avoiding exposure to fungi outside the hospital for patients with haematological cancers. Mycoses. 2014;57:336‐341. [DOI] [PubMed] [Google Scholar]
- 47. Food Safety and Inspection Service, US Department of Agriculture . Food Safety for People with Cancer. Washington, DC: US Department of Agriculture; 2006. http://www.fsis.usda.gov/PDF/Food_Safety_for_People_with_Cancer.pdf. Accessed August 21, 2017. [Google Scholar]
- 48. Tomblyn M, Chiller T, Einsele H, et al. Guidelines for preventing infectious complications among hematopoietic cell transplant recipients: a global perspective. Preface Bone Marrow Transplant. 2009;44:453‐455. [DOI] [PubMed] [Google Scholar]
- 49. Moody K, Finlay J, Mancuso C, Charlson M. Feasibility and safety of a pilot randomized trial of infection rate: neutropenic diet versus standard food safety guidelines. J Pediatr Hematol Oncol. 2006;28:126‐133. [DOI] [PubMed] [Google Scholar]
- 50. Trifilio S, Helenowski I, Giel M, et al. Questioning the role of a neutropenic diet following hematopoetic stem cell transplantation. Biol Blood Marrow Transplant. 2012;18:1385‐1390. [DOI] [PubMed] [Google Scholar]
- 51. Chong Y, Yakushiji H, Ito Y, Kamimura T. Clinical impact of fluoroquinolone prophylaxis in neutropenic patients with hematological malignancies. Int J Infect Dis. 2011;15:e277‐e281. [DOI] [PubMed] [Google Scholar]
- 52. Mikulska M, Averbuch D, Tissot F, et al. Fluoroquinolone prophylaxis in haematological cancer patients with neutropenia: ECIL critical appraisal of previous guidelines. J Infect. 2018;76:20‐37. [DOI] [PubMed] [Google Scholar]
- 53. Cattaneo C, Quaresmini G, Casari S, et al. Recent changes in bacterial epidemiology and the emergence of fluoroquinolone‐resistant Escherichia coli among patients with haematological malignancies: results of a prospective study on 823 patients at a single institution. J Antimicrob Chemother. 2008;61:721‐728. [DOI] [PubMed] [Google Scholar]
- 54. Gafter‐Gvili A, Fraser A, Paul M, et al. Antibiotic prophylaxis for bacterial infections in afebrile neutropenic patients following chemotherapy [serial online]. Cochrane Database Syst Rev. 2012;1:CD004386. [DOI] [PubMed] [Google Scholar]
- 55. Akan A, H., Antia VP, Kouba M, et al. Preventing invasive fungal disease in patients with haematological malignancies and the recipients of haematopoietic stem cell transplantation: practical aspects. J Antimicrob Chemother. 2013;68(suppl 3):iii5‐iii16. [DOI] [PubMed] [Google Scholar]
- 56. Kruger WH, Bohlius J, Cornely OA, et al. Antimicrobial prophylaxis in allogeneic bone marrow transplantation. Guidelines of the Infectious Diseases Working Party (AGIHO) of the German Society of Haematology and Oncology. Ann Oncol. 2005;16:1381‐1390. [DOI] [PubMed] [Google Scholar]
- 57. Baden LR, Swaminathan S, Angarone M, et al. Prevention and Treatment of Cancer‐Related Infections, Version 22016, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2016;14:882‐913. [DOI] [PubMed] [Google Scholar]
- 58. Dietrich M, Gaus W, Vossen J, van der Waaij D, Wendt F. Protective isolation and antimicrobial decontamination in patients with high susceptibility to infection. A prospective cooperative study of gnotobiotic care in acute leukemia patients. I: clinical results Infection. 1977;5:107‐114. [DOI] [PubMed] [Google Scholar]
- 59. Levine AS, Siegel SE, Schreiber AD, et al. Protected environments and prophylactic antibiotics. A prospective controlled study of their utility in the therapy of acute leukemia. N Engl J Med. 1973;288:477‐483. [DOI] [PubMed] [Google Scholar]
- 60. Benet T, Nicolle MC, Thiebaut A, et al. Reduction of invasive aspergillosis incidence among immunocompromised patients after control of environmental exposure. Clin Infect Dis. 2007;45:682‐686. [DOI] [PubMed] [Google Scholar]
- 61. Vonberg RP, Gastmeier P. Nosocomial aspergillosis in outbreak settings. J Hosp Infect. 2006;63:246‐254. [DOI] [PubMed] [Google Scholar]
- 62. Anaissie EJ, Penzak SR, Dignani MC. The hospital water supply as a source of nosocomial infections: a plea for action. Arch Intern Med. 2002;162:1483‐1492. [DOI] [PubMed] [Google Scholar]
- 63. Exner M, Kramer A, Lajoie L, Gebel J, Engelhart S, Hartemann P. Prevention and control of health care‐associated waterborne infections in health care facilities. Am J Infect Control. 2005;33(5 suppl 1):S26‐S40. [DOI] [PubMed] [Google Scholar]
- 64. Boyce JM, Pittet D; Healthcare Infection Control Practices Advisory Committee; HIPAC/SHEA/APIC/IDSA Hand Hygiene Task Force . Guideline for hand hygiene in health‐care settings. Recommendations of the Healthcare Infection Control Practices Advisory Committee and the HIPAC/SHEA/APIC/IDSA Hand Hygiene Task Force. Am J Infect Control. 2002;30:S1‐S46. [DOI] [PubMed] [Google Scholar]
- 65. Gasink LB, Brennan PJ. Isolation precautions for antibiotic‐resistant bacteria in healthcare settings. Curr Opin Infect Dis. 2009;22:339‐344. [DOI] [PubMed] [Google Scholar]
- 66. Abad C, Fearday A, Safdar N. Adverse effects of isolation in hospitalised patients: a systematic review. J Hosp Infect. 2010;76:97‐102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Gudiol C, Tubau F, Calatayud L, et al. Bacteraemia due to multidrug‐resistant Gram‐negative bacilli in cancer patients: risk factors, antibiotic therapy and outcomes. J Antimicrob Chemother. 2011;66:657‐663. [DOI] [PubMed] [Google Scholar]
- 68. Tacconelli E, Cataldo MA, Dancer SJ, et al. ESCMID guidelines for the management of the infection control measures to reduce transmission of multidrug‐resistant Gram‐negative bacteria in hospitalized patients. Clin Microbiol Infect. 2014;20(suppl 1):1‐55. [DOI] [PubMed] [Google Scholar]
- 69. Ruhnke M, Arnold R, Gastmeier P. Infection control issues in patients with haematological malignancies in the era of multidrug‐resistant bacteria. Lancet Oncol. 2014;15:e606‐e619. [DOI] [PubMed] [Google Scholar]
- 70. Huang SS, Datta R, Platt R. Risk of acquiring antibiotic‐resistant bacteria from prior room occupants. Arch Intern Med. 2006;166:1945‐1951. [DOI] [PubMed] [Google Scholar]
- 71. Huang R, Mehta S, Weed D, Price CS. Methicillin‐resistant Staphylococcus aureus survival on hospital fomites. Infect Control Hosp Epidemiol. 2006;27:1267‐1269. [DOI] [PubMed] [Google Scholar]
- 72. Drees M, Snydman DR, Schmid CH, et al. Prior environmental contamination increases the risk of acquisition of vancomycin‐resistant enterococci. Clin Infect Dis. 2008;46:678‐685. [DOI] [PubMed] [Google Scholar]
- 73. Shaughnessy MK, Micielli RL, DePestel DD, et al. Evaluation of hospital room assignment and acquisition of Clostridium difficile infection. Infect Control Hosp Epidemiol. 2011;32:201‐206. [DOI] [PubMed] [Google Scholar]
- 74. Centers for Disease Control and Prevention . Management of Multidrug‐Resistant Organisms in Healthcare Settings. Updated: 2016. Atlanta, GA: Centers for Disease Control and Prevention; 2016. http://cdc.gov/infectioncontrol/guidelines/mdro/prevention-control.html. Accessed March 29, 2018. [Google Scholar]
- 75. Muto CA, Jernigan JA, Ostrowsky BE, et al. SHEA guideline for preventing nosocomial transmission of multidrug‐resistant strains of Staphylococcus aureus and enterococcus. Infect Control Hosp Epidemiol. 2003;24:362‐386. [DOI] [PubMed] [Google Scholar]
- 76. Arnan M, Gudiol C, Calatayud L, et al. Risk factors for, and clinical relevance of, faecal extended‐spectrum beta‐lactamase producing Escherichia coli (ESBL‐EC) carriage in neutropenic patients with haematological malignancies. Eur J Clin Microbiol Infect Dis. 2011;30:355‐360. [DOI] [PubMed] [Google Scholar]
- 77. Buehlmann M, Frei R, Fenner L, Dangel M, Fluckiger U, Widmer AF. Highly effective regimen for decolonization of methicillin‐resistant Staphylococcus aureus carriers. Infect Control Hosp Epidemiol. 2008;29:510‐516. [DOI] [PubMed] [Google Scholar]
- 78. Decker BK, Palmore TN. Universal decolonization was better than MRSA screening and isolation for preventing nosocomial ICU infections [serial online]. Ann Intern Med. 2013;159:JC4. [DOI] [PubMed] [Google Scholar]
- 79. Mendes ET, Ranzani OT, Marchi AP, et al. Chlorhexidine bathing for the prevention of colonization and infection with multidrug‐resistant microorganisms in a hematopoietic stem cell transplantation unit over a 9‐year period: impact on chlorhexidine susceptibility [serial online]. Medicine (Baltimore). 2016;95:e5271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin‐resistant Staphylococcus aureus infections in adults and children: executive summary. Clin Infect Dis. 2011;52:285‐292. [DOI] [PubMed] [Google Scholar]
- 81. Bass P, Karki S, Rhodes D, et al. Impact of chlorhexidine‐impregnated washcloths on reducing incidence of vancomycin‐resistant enterococci colonization in hematology‐oncology patients. Am J Infect Control. 2013;41:345‐348. [DOI] [PubMed] [Google Scholar]
- 82. Horner C, Mawer D, Wilcox M. Reduced susceptibility to chlorhexidine in staphylococci: is it increasing and does it matter? J Antimicrob Chemother. 2012;67:2547‐2559. [DOI] [PubMed] [Google Scholar]
- 83. Kizny Gordon AE, Mathers AJ, Cheong EYL, et al. The hospital water environment as a reservoir for Carbapenem‐resistant organisms causing hospital‐acquired infections—a systematic review of the literature. Clin Infect Dis. 2017;64:1435‐1444. [DOI] [PubMed] [Google Scholar]
- 84. Chemaly RF, Simmons S, Dale C Jr, et al. The role of the healthcare environment in the spread of multidrug‐resistant organisms: update on current best practices for containment. Ther Adv Infect Dis. 2014;2:79‐90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Huskins WC, Huckabee CM, O'Grady NP, et al. Intervention to reduce transmission of resistant bacteria in intensive care. N Engl J Med. 2011;364:1407‐1418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Adachi JA, Perego C, Graviss L, et al. The role of interventional molecular epidemiology in controlling clonal clusters of multidrug resistant Pseudomonas aeruginosa in critically ill cancer patients. Am J Infect Control. 2009;37:442‐446. [DOI] [PubMed] [Google Scholar]
- 87. Prematunge C, MacDougall C, Johnstone J, et al. VRE and VSE bacteremia outcomes in the era of effective VRE therapy: a systematic review and meta‐analysis. Infect Control Hosp Epidemiol. 2016;37:26‐35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. DiazGranados CA, Zimmer SM, Klein M, Jernigan JA. Comparison of mortality associated with vancomycin‐resistant and vancomycin‐susceptible enterococcal bloodstream infections: a meta‐analysis. Clin Infect Dis. 2005;41:327‐333. [DOI] [PubMed] [Google Scholar]
- 89. Hachem R, Graviss L, Hanna H, et al. Impact of surveillance for vancomycin‐resistant enterococci on controlling a bloodstream outbreak among patients with hematologic malignancy. Infect Control Hosp Epidemiol. 2004;25:391‐394. [DOI] [PubMed] [Google Scholar]
- 90. McFarland LV, Mulligan ME, Kwok RY, Stamm WE. Nosocomial acquisition of Clostridium difficile infection. N Engl J Med. 1989;320:204‐210. [DOI] [PubMed] [Google Scholar]
- 91. O'Connor JR, Johnson S, Gerding DN. Clostridium difficile infection caused by the epidemic BI/NAP1/027 strain. Gastroenterology. 2009;136:1913‐1924. [DOI] [PubMed] [Google Scholar]
- 92. Gerding DN, Muto CA, Owens RC Jr. Measures to control and prevent Clostridium difficile infection. Clin Infect Dis. 2008;46(suppl 1):S43‐S49. [DOI] [PubMed] [Google Scholar]
- 93. Kamboj M, Son C, Cantu S, et al. Hospital‐onset Clostridium difficile infection rates in persons with cancer or hematopoietic stem cell transplant: a C3IC Network report. Infect Control Hosp Epidemiol. 2012;33:1162‐1165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Cowling BJ, Ip DK, Fang VJ, et al. Aerosol transmission is an important mode of influenza A virus spread [serial online]. Nat Commun. 2013;4:1935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Gralton J, Tovey ER, McLaws ML, Rawlinson WD. Respiratory virus RNA is detectable in airborne and droplet particles. J Med Virol. 2013;85:2151‐2159. [DOI] [PubMed] [Google Scholar]
- 96. Shah DP, Ghantoji SS, Mulanovich VE, Ariza‐Heredia EJ, Chemaly RF. Management of respiratory viral infections in hematopoietic cell transplant recipients. Am J Blood Res. 2012;2:203‐218. [PMC free article] [PubMed] [Google Scholar]
- 97. Englund J, Feuchtinger T, Ljungman P. Viral infections in immunocompromised patients. Biol Blood Marrow Transplant. 2011;17(1 suppl):S2‐S5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Raad I, Abbas J, Whimbey E. Infection control of nosocomial respiratory viral disease in the immunocompromised host. Am J Med. 1997;102:48‐52; discussion 53‐44. [DOI] [PubMed] [Google Scholar]
- 99. von Lilienfeld‐Toal M, Berger A, Christopeit M, et al. Community acquired respiratory virus infections in cancer patients—guideline on diagnosis and management by the Infectious Diseases Working Party of the German Society for Haematology and Medical Oncology. Eur J Cancer. 2016;67:200‐212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Chemaly RF, Shah DP, Boeckh MJ. Management of respiratory viral infections in hematopoietic cell transplant recipients and patients with hematologic malignancies. Clin Infect Dis. 2014;59(suppl 5):S344‐S351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Jefferson T, Del Mar CB, Dooley L, et al. Physical interventions to interrupt or reduce the spread of respiratory viruses [serial online]. Cochrane Database Syst Rev. 2011;7:CD006207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Hall CB. Nosocomial respiratory syncytial virus infections: the “Cold War” has not ended. Clin Infect Dis. 2000;31:590‐596. [DOI] [PubMed] [Google Scholar]
- 103. Sokol KA, De la Vega‐Diaz I, Edmondson‐Martin K, et al. Masks for prevention of respiratory viruses on the BMT unit: results of a quality initiative. Transplant Infect Dis. 2016;18:965‐967. [DOI] [PubMed] [Google Scholar]
- 104. Zhu Y, Zembower TR, Metzger KE, Lei Z, Green SJ, Qi C. Investigation of respiratory syncytial virus outbreak on an adult stem cell transplant unit by use of whole‐genome sequencing. J Clin Microbiol. 2017;55:2956‐2963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Kassis C, Champlin RE, Hachem RY, et al. Detection and control of a nosocomial respiratory syncytial virus outbreak in a stem cell transplantation unit: the role of palivizumab. Biol Blood Marrow Transpl. 2010;16:1265‐1271. [DOI] [PubMed] [Google Scholar]
- 106. Nabeya D, Kinjo T, Parrott GL, et al. The clinical and phylogenetic investigation for a nosocomial outbreak of respiratory syncytial virus infection in an adult hemato‐oncology unit. J Med Virol. 2017;89:1364‐1372. [DOI] [PubMed] [Google Scholar]
- 107. Burke CW, Bridges O, Brown S, Rahija R, Russell CJ. Mode of parainfluenza virus transmission determines the dynamics of primary infection and protection from reinfection [serial online]. PLoS Pathog. 2013;9:e1003786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Piralla A, Percivalle E, Di Cesare‐Merlone A, Locatelli F, Gerna G. Multicluster nosocomial outbreak of parainfluenza virus type 3 infection in a pediatric oncohematology unit: a phylogenetic study. Haematologica. 2009;94:833‐839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Harvala H, Gaunt E, McIntyre C, et al. Epidemiology and clinical characteristics of parainfluenza virus 3 outbreak in a haemato‐oncology unit. J Infect. 2012;65:246‐254. [DOI] [PubMed] [Google Scholar]
- 110. Wong BC, Lee N, Li Y, et al. Possible role of aerosol transmission in a hospital outbreak of influenza. Clin Infect Dis. 2010;51:1176‐1183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Buchbinder N, Dumesnil C, Pinquier D, et al. Pandemic A/H1N1/2009 influenza in a paediatric haematology and oncology unit: successful management of a sudden outbreak. J Hosp Infect. 2011;79:155‐160. [DOI] [PubMed] [Google Scholar]
- 112. Chironna M, Tafuri S, Santoro N, Prato R, Quarto M, Germinario CA. A nosocomial outbreak of 2009 pandemic influenza A(H1N1) in a paediatric oncology ward in Italy, October‐November 2009 [serial online]. Euro Surveill. 2010;15:pii:19.454. [DOI] [PubMed] [Google Scholar]
- 113. Yolken RH, Bishop CA, Townsend TR, et al. Infectious gastroenteritis in bone‐marrow‐transplant recipients. N Engl J Med. 1982;306:1010‐1012. [DOI] [PubMed] [Google Scholar]
- 114. Venard V, Carret A, Corsaro D, Bordigoni P, Le Faou A. Genotyping of adenoviruses isolated in an outbreak in a bone marrow transplant unit shows that diverse strains are involved. J Hosp Infect. 2000;44:71‐74. [DOI] [PubMed] [Google Scholar]
- 115. Swartling L, Allard A, Torlen J, Ljungman P, Mattsson J, Sparrelid E. Prolonged outbreak of adenovirus A31 in allogeneic stem cell transplant recipients. Transpl Infect Dis. 2015;17:785‐794. [DOI] [PubMed] [Google Scholar]
- 116. Durai P, Batool M, Shah M, Choi S. Middle East respiratory syndrome coronavirus: transmission, virology and therapeutic targeting to aid in outbreak control [serial online]. Exp Mol Med. 2015;47:e181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Milano F, Campbell AP, Guthrie KA, et al. Human rhinovirus and coronavirus detection among allogeneic hematopoietic stem cell transplantation recipients. Blood. 2010;115:2088‐2094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Myatt TA, Johnston SL, Zuo Z, et al. Detection of airborne rhinovirus and its relation to outdoor air supply in office environments. Am J Resp Crit Care Med. 2004;169:1187‐1190. [DOI] [PubMed] [Google Scholar]
- 119. Cutino‐Moguel T, Lauinger IL, Srivastava S, Zuckerman M, Tong CY, Devereux S. Analysis of a potential cluster of rhinovirus infections in patients and staff on two haemato‐oncology wards. J Clin Virol. 2014;60:57‐59. [DOI] [PubMed] [Google Scholar]
- 120. Panda S, Mohakud NK, Pena L, Kumar S. Human metapneumovirus: review of an important respiratory pathogen. Int J Infect Dis. 2014;25:45‐52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Hoellein A, Hecker J, Hoffmann D, et al. Serious outbreak of human metapneumovirus in patients with hematologic malignancies. Leuk Lymphoma. 2016;57:623‐627. [DOI] [PubMed] [Google Scholar]
- 122. Ho KY, Singh KS, Habib AG, et al. Mild illness associated with severe acute respiratory syndrome coronavirus infection: lessons from a prospective seroepidemiologic study of health‐care workers in a teaching hospital in Singapore. J Infect Dis. 2004;189:642‐647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. Patrick DM, Petric M, Skowronski DM, et al. An outbreak of human coronavirus OC43 infection and serological cross‐reactivity with SARS coronavirus. Can J Infect Dis Med Microbiol. 2006;17:330‐336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Rubin LG, Kohn N, Nullet S, Hill M. Reduction in rate of nosocomial respiratory virus infections in a children's hospital associated with enhanced isolation precautions. Infect Control Hosp Epidemiol. 2018;39:152‐156. [DOI] [PubMed] [Google Scholar]
- 125. Sung AD, Sung JAM, Thomas S, et al. Universal mask usage for reduction of respiratory viral infections after stem cell transplant: a prospective trial. Clin Infect Dis. 2016;63:999‐1006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Lehners N, Tabatabai J, Prifert C, et al. Long‐term shedding of influenza virus, parainfluenza virus, respiratory syncytial virus and nosocomial epidemiology in patients with hematological disorders [serial online]. PLoS One. 2016;11:e0148258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Lehners N, Schnitzler P, Geis S, et al. Risk factors and containment of respiratory syncytial virus outbreak in a hematology and transplant unit. Bone Marrow Transplant. 2013;48:1548‐1553. [DOI] [PubMed] [Google Scholar]
- 128. Marks PJ, Vipond IB, Carlisle D, Deakin D, Fey RE, Caul EO. Evidence for airborne transmission of Norwalk‐like virus (NLV) in a hotel restaurant. Epidemiol Infect. 2000;124:481‐487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Sheahan A, Copeland G, Richardson L, et al. Control of norovirus outbreak on a pediatric oncology unit. Am J Infect Control. 2015;43:1066‐1069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130. Simon A, Schildgen O, Maria Eis‐Hubinger A, et al. Norovirus outbreak in a pediatric oncology unit. Scand J Gastroenterol. 2006;41:693‐699. [DOI] [PubMed] [Google Scholar]
- 131. Rogers M, Weinstock DM, Eagan J, Kiehn T, Armstrong D, Sepkowitz KA. Rotavirus outbreak on a pediatric oncology floor: possible association with toys. Am J Infect Control. 2000;28:378‐380. [DOI] [PubMed] [Google Scholar]
- 132. Green KY. Norovirus infection in immunocompromised hosts. Clin Microbiol Infect. 2014;20:717‐723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Sukhrie FH, Siebenga JJ, Beersma MF, Koopmans M. Chronic shedders as reservoir for nosocomial transmission of norovirus. J Clin Microbiol. 2010;48:4303‐4305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134. O'Grady NP, Alexander M, Burns LA, et al. Guidelines for the prevention of intravascular catheter‐related infections. Am J Infect Control. 2011;39:S1‐34. [DOI] [PubMed] [Google Scholar]
- 135. Mermel LA, Allon M, Bouza E, et al. Clinical practice guidelines for the diagnosis and management of intravascular catheter‐related infection: 2009 update by the Infectious Diseases Society of America. Clin Infect Dis. 2009;49:1‐45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136. Steinberg JP, Robichaux C, Tejedor SC, Reyes MD, Jacob JT. Distribution of pathogens in central line‐associated bloodstream infections among patients with and without neutropenia following chemotherapy: evidence for a proposed modification to the current surveillance definition. Infect Control Hosp Epidemiol. 2013;34:171‐175. [DOI] [PubMed] [Google Scholar]
- 137. Centers for Disease Control and Prevention . Bloodstream Infection Event (Central Line‐Associated Bloodstream Infection and Non‐Central Line Associated Bloodstream Infection). Atlanta, GA: Centers for Disease Control and Prevention; 2018. http://cdc.gov/nhsn/PDFs/pscManual/4PSC_CLABScurrent.pdf. Accessed March 23, 2018. [Google Scholar]
- 138. Zakhour R, Chaftari AM, Raad II. Catheter‐related infections in patients with haematological malignancies: novel preventive and therapeutic strategies. Lancet Infect Dis. 2016;16:e241‐e250. [DOI] [PubMed] [Google Scholar]
- 139. Mueller BU, Skelton J, Callender DP, et al. A prospective randomized trial comparing the infectious and noninfectious complications of an externalized catheter versus a subcutaneously implanted device in cancer patients. J Clin Oncol. 1992;10:1943‐1948. [DOI] [PubMed] [Google Scholar]
- 140. Lim MY, Al‐Kali A, Ashrani AA, et al. Comparison of complication rates of Hickman catheters versus peripherally inserted central catheters in patients with acute myeloid leukemia undergoing induction chemotherapy. Leuk Lymphoma. 2013;54:1263‐1267. [DOI] [PubMed] [Google Scholar]
- 141. Richters A, van Vliet M, Peer PG, et al. Incidence of and risk factors for persistent Gram‐positive bacteraemia and catheter‐related thrombosis in haematopoietic stem cell transplantation. Bone Marrow Transplant. 2014;49:264‐269. [DOI] [PubMed] [Google Scholar]
- 142. Groeger JS, Lucas AB, Thaler HT, et al. Infectious morbidity associated with long‐term use of venous access devices in patients with cancer. Ann Intern Med. 1993;119:1168‐1174. [DOI] [PubMed] [Google Scholar]
- 143. Akahane A, Sone M, Ehara S, et al. Central venous port‐related infection in patients with malignant tumors: an observational study. Ups J Med Sci. 2012;117:300‐308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Biffi R, Pozzi S, Agazzi A, et al. Use of totally implantable central venous access ports for high‐dose chemotherapy and peripheral blood stem cell transplantation: results of a monocentre series of 376 patients. Ann Oncol. 2004;15:296‐300. [DOI] [PubMed] [Google Scholar]
- 145. Mollee P, Jones M, Stackelroth J, et al. Catheter‐associated bloodstream infection incidence and risk factors in adults with cancer: a prospective cohort study. J Hosp Infect. 2011;78:26‐30. [DOI] [PubMed] [Google Scholar]
- 146. Goltz JP, Scholl A, Ritter CO, Wittenberg G, Hahn D, Kickuth R. Peripherally placed totally implantable venous‐access port systems of the forearm: clinical experience in 763 consecutive patients. Cardiovasc Intervent Radiol. 2010;33:1159‐1167. [DOI] [PubMed] [Google Scholar]
- 147. Chopra V, O'Horo JC, Rogers MA, Maki DG, Safdar N. The risk of bloodstream infection associated with peripherally inserted central catheters compared with central venous catheters in adults: a systematic review and meta‐analysis. Infect Control Hosp Epidemiol. 2013;34:908‐918. [DOI] [PubMed] [Google Scholar]
- 148. Bertoglio S, Faccini B, Lalli L, Cafiero F, Bruzzi P. Peripherally inserted central catheters (PICCs) in cancer patients under chemotherapy: a prospective study on the incidence of complications and overall failures. J Surg Oncol. 2016;113:708‐714. [DOI] [PubMed] [Google Scholar]
- 149. Lorente L, Henry C, Martin MM, Jimenez A, Mora ML. Central venous catheter‐related infection in a prospective and observational study of 2,595 catheters. Crit Care. 2005;9:R631‐R635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150. Ge X, Cavallazzi R, Li C, Pan SM, Wang YW, Wang FL. Central venous access sites for the prevention of venous thrombosis, stenosis and infection [serial online]. Cochrane Database Syst Rev. 2012;3:CD004084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151. Weiner LM, Webb AK, Limbago B, et al. Antimicrobial‐resistant pathogens associated with healthcare‐associated infections: summary of data reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2011‐2014. Infect Control Hosp Epidemiol. 2016;37:1288‐1301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152. See I, Freifeld AG, Magill SS. Causative organisms and associated antimicrobial resistance in healthcare‐associated, central line‐associated bloodstream infections from oncology settings, 2009‐2012. Clin Infect Dis. 2016;62:1203‐1209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153. Raad I, Costerton W, Sabharwal U, Sacilowski M, Anaissie E, Bodey GP. Ultrastructural analysis of indwelling vascular catheters: a quantitative relationship between luminal colonization and duration of placement. J Infect Dis. 1993;168:400‐407. [DOI] [PubMed] [Google Scholar]
- 154. Anaissie E, Samonis G, Kontoyiannis D, et al. Role of catheter colonization and infrequent hematogenous seeding in catheter‐related infections. Eur J Clin Microbiol Infect Dis. 1995;14:134‐137. [DOI] [PubMed] [Google Scholar]
- 155. Raad II, Hohn DC, Gilbreath BJ, et al. Prevention of central venous catheter‐related infections by using maximal sterile barrier precautions during insertion. Infect Control Hosp Epidemiol. 1994;15:231‐238. [PubMed] [Google Scholar]
- 156. Miller JM, Goetz AM, Squier C, Muder RR. Reduction in nosocomial intravenous device‐related bacteremias after institution of an intravenous therapy team. J Intraven Nurs. 1996;19:103‐106. [PubMed] [Google Scholar]
- 157. Hunter MR. Development of a vascular access team in an acute care setting. J Infus Nurs. 2003;26:86‐91. [DOI] [PubMed] [Google Scholar]
- 158. Ruschulte H, Franke M, Gastmeier P, et al. Prevention of central venous catheter related infections with chlorhexidine gluconate impregnated wound dressings: a randomized controlled trial. Ann Hematol. 2009;88:267‐272. [DOI] [PubMed] [Google Scholar]
- 159. Carrer S, Bocchi A, Bortolotti M, et al. Effect of different sterile barrier precautions and central venous catheter dressing on the skin colonization around the insertion site. Minerva Anesthesiol. 2005;71:197‐206. [PubMed] [Google Scholar]
- 160. Rinke ML, Chen AR, Bundy DG, et al. Implementation of a central line maintenance care bundle in hospitalized pediatric oncology patients. Pediatrics. 2012;130:e996‐e1004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161. Kamboj M, Blair R, Bell N, et al. Use of disinfection cap to reduce central‐line‐associated bloodstream infection and blood culture contamination among hematology‐oncology patients. Infect Control Hosp Epidemiol. 2015;36:1401‐1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162. Sweet MA, Cumpston A, Briggs F, Craig M, Hamadani M. Impact of alcohol‐impregnated port protectors and needleless neutral pressure connectors on central line‐associated bloodstream infections and contamination of blood cultures in an inpatient oncology unit. Am J Infect Control. 2012;40:931‐934. [DOI] [PubMed] [Google Scholar]
- 163. Raad I, Reitzel R, Jiang Y, Chemaly RF, Dvorak T, Hachem R. Anti‐adherence activity and antimicrobial durability of anti‐infective‐coated catheters against multidrug‐resistant bacteria. J Antimicrob Chemother. 2008;62:746‐750. [DOI] [PubMed] [Google Scholar]
- 164.Viola GM, Rosenblat J, Raad II. Drug eluting antimicrobial vascular catheters: Progress and Promise. [DOI] [PubMed]
- 165. Chatzinikolaou I, Zipf TF, Hanna H, et al. Minocycline‐ethylenediaminetetraacetate lock solution for the prevention of implantable port infections in children with cancer. Clin Infect Dis. 2003;36:116‐119. [DOI] [PubMed] [Google Scholar]
- 166. Schoot RA, van Ommen CH, Stijnen T, et al. Prevention of central venous catheter‐associated bloodstream infections in paediatric oncology patients using 70% ethanol locks: a randomised controlled multi‐centre trial. Eur J Cancer. 2015;51:2031‐2038. [DOI] [PubMed] [Google Scholar]
- 167. Ramos ER, Reitzel R, Jiang Y, et al. Clinical effectiveness and risk of emerging resistance associated with prolonged use of antibiotic‐impregnated catheters: more than 0.5 million catheter days and 7 years of clinical experience. Crit Care Med. 2011;39:245‐251. [DOI] [PubMed] [Google Scholar]
- 168. Sammon J, Trinh VQ, Ravi P, et al. Health care‐associated infections after major cancer surgery: temporal trends, patterns of care, and effect on mortality. Cancer. 2013;119:2317‐2324. [DOI] [PubMed] [Google Scholar]
- 169. Rolston KV, Nesher L, Tarrand JT. Current microbiology of surgical site infections in patients with cancer: a retrospective review. Infect Dis Ther. 2014;3:245‐256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170. Hernaiz‐Leonardo JC, Golzarri MF, Cornejo‐Juarez P, et al. Microbiology of surgical site infections in patients with cancer: a 7‐year review. Am J Infect Control. 2017;45:761‐766. [DOI] [PubMed] [Google Scholar]
- 171. Bhattacharya S, Goel G, Mukherjee S, Bhaumik J, Chandy M. Epidemiology of antimicrobial resistance in an oncology center in eastern India. Infect Control Hosp Epidemiol. 2015;36:864‐866. [DOI] [PubMed] [Google Scholar]
- 172. Allegranzi B, Bischoff P, de Jonge S, et al. New WHO recommendations on preoperative measures for surgical site infection prevention: an evidence‐based global perspective. Lancet Infect Dis. 2016;16:e276‐e287. [DOI] [PubMed] [Google Scholar]
- 173. Otter JA, French GL. Survival of nosocomial bacteria and spores on surfaces and inactivation by hydrogen peroxide vapor. J Clin Microbiol. 2009;47:205‐207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174. Otter JA, Yezli S, Salkeld JA, French GL. Evidence that contaminated surfaces contribute to the transmission of hospital pathogens and an overview of strategies to address contaminated surfaces in hospital settings. Am J Infect Control. 2013;41:S6‐S11. [DOI] [PubMed] [Google Scholar]
- 175. Rzezutka A, Cook N. Survival of human enteric viruses in the environment and food. FEMS Microbiol Rev. 2004;28:441‐453. [DOI] [PubMed] [Google Scholar]
- 176. Kramer A, Schwebke I, Kampf G. How long do nosocomial pathogens persist on inanimate surfaces? A systematic review [serial online], BMC Infect Dis. 2006;6:130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177. Havill NL, Havill HL, Mangione E, Dumigan DG, Boyce JM. Cleanliness of portable medical equipment disinfected by nursing staff. Am J Infect Control. 2011;39:602‐604. [DOI] [PubMed] [Google Scholar]
- 178. Munoz‐Price LS, Birnbach DJ, Lubarsky DA, et al. Decreasing operating room environmental pathogen contamination through improved cleaning practice. Infect Control Hosp Epidemiol. 2012;33:897‐904. [DOI] [PubMed] [Google Scholar]
- 179. Haas JP, Menz J, Dusza S, Montecalvo MA. Implementation and impact of ultraviolet environmental disinfection in an acute care setting. Am J Infect Control. 2014;42:586‐590. [DOI] [PubMed] [Google Scholar]
- 180. Beal A, Mahida N, Staniforth K, Vaughan N, Clarke M, Boswell T. First UK trial of Xenex PX‐UV, an automated ultraviolet room decontamination device in a clinical haematology and bone marrow transplantation unit. J Hosp Infect. 2016;93:164‐168. [DOI] [PubMed] [Google Scholar]
- 181. Ghantoji SS, Stibich M, Stachowiak J, et al. Non‐inferiority of pulsed xenon UV light versus bleach for reducing environmental Clostridium difficile contamination on high‐touch surfaces in Clostridium difficile infection isolation rooms. J Med Microbiol. 2015;64:191‐194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182. Marra AR, Schweizer ML, Edmond MB. No‐touch disinfection methods to decrease multidrug‐resistant organism infections: a systematic review and meta‐analysis. Infect Control Hosp Epidemiol. 2018;39:20‐31. [DOI] [PubMed] [Google Scholar]
- 183. Anderson DJ, Chen LF, Weber DJ, et al. Enhanced terminal room disinfection and acquisition and infection caused by multidrug‐resistant organisms and Clostridium difficile (the Benefits of Enhanced Terminal Room Disinfection study): a cluster‐randomised, multicentre, crossover study. Lancet. 2017;389:805‐814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184. Hosein I, Madeloso R, Nagaratnam W, Villamaria F, Stock E, Jinadatha C. Evaluation of a pulsed xenon ultraviolet light device for isolation room disinfection in a United Kingdom hospital. Am J Infect Control. 2016;44:e157‐e161. [DOI] [PubMed] [Google Scholar]
- 185. El Haddad L, Ghantoji SS, Stibich M, et al. Evaluation of a pulsed xenon ultraviolet disinfection system to decrease bacterial contamination in operating rooms [serial online]. BMC Infect Dis. 2017;17:672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186. Robilotti E, Kamboj M. Integration of whole‐genome sequencing into infection control practices: the potential and the hurdles. J Clin Microbiol. 2015;53:1054‐1055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187. Kwong JC, Mercoulia K, Tomita T, et al. Prospective whole‐genome sequencing enhances national surveillance of Listeria monocytogenes. J Clin Microbiol. 2016;54:333‐342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188. Halpern MT, Yabroff KR. Prevalence of outpatient cancer treatment in the United States: estimates from the Medical Panel Expenditures Survey (MEPS). Cancer Invest. 2008;26:647‐651. [DOI] [PubMed] [Google Scholar]
- 189. Ariza‐Heredia EJ, Chemaly RF. Practical review of immunizations in adult patients with cancer. Hum Vaccin Immunother. 2015;11:2606‐2614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190. Sipsas NV, Kontoyiannis DP. Occupation, lifestyle, diet, and invasive fungal infections. Infection. 2008;36:515‐525. [DOI] [PubMed] [Google Scholar]
- 191. Frenzel E, Chemaly RF, Ariza‐Heredia E, et al. Association of increased influenza vaccination in health care workers with a reduction in nosocomial influenza infections in cancer patients. Am J Infect Control. 2016;44:1016‐1021. [DOI] [PubMed] [Google Scholar]
- 192. Tverdek FP, Rolston KV, Chemaly RF. Antimicrobial stewardship in patients with cancer. Pharmacotherapy. 2012;32:722‐734. [DOI] [PubMed] [Google Scholar]
- 193. Robilotti E, Holubar M, Seo SK, Deresinski S. Feasibility and applicability of antimicrobial stewardship in immunocompromised patients. Curr Opin Infect Dis. 2017;30:346‐353. [DOI] [PubMed] [Google Scholar]


