SUMMARY
-
1
Saikosaponins represent a group of oleanane derivatives, usually as glucosides, that are found in a number of plant families. Saikosaponins isolated from medicinal plants such as Bupleurum spp., Heteromorpha spp. and Scrophularia scorodonia have been reported to possess various biological activities, specifically antihepatitis, antinephritis, antihepatoma, anti‐inflammation, immunomodulation and antibacterial effects.
-
2
The aim of the present study was to examine the anticoronaviral activity of saikosaponins (A, B2, C and D) and their mode of action. Using the 2,3‐bis[2‐methoxy‐4‐nitro‐5‐sulfophenyl]‐5‐[(phenylamino) carbonyl‐2H‐tetrazolium hydroxide] (XTT) assay, results showed that all saikosaponins tested demonstrated antiviral activity at concentrations of 0.25–25 µmol/L, with the strongest activity being noted for saikosaponin B2 (IC50 = 1.7 ± 0.1 µmol/L). Interestingly, both saikosaponins A (50% cellular cytotoxicity (CC50) concentration = 228.1 ± 3.8 µmol/L; selectivity index (SI) = 26.6) and B2 (CC50 = 383.3 ± 0.2 µmol/L; SI = 221.9) exhibited no cytotoxic effects on target cells at concentrations that achieved antiviral activity. In the time‐of‐addition studies, saikosaponin B2, at 6 µmol/L, significantly inhibited human coronavirus 229E infection following its addition at various time pre‐infection (−4 to −1 h), coinfection (0 h) and post‐infection (1–4 h). Furthermore, saikosaponin B2 also showed an inhibitory effect on viral attachment and penetration.
-
3
The present results indicate that saikosaponin B2 has potent anticoronaviral activity and that its mode of action possibly involves interference in the early stage of viral replication, such as absorption and penetration of the virus.
Keywords: antiviral activity, saikosaponins, viral replication
INTRODUCTION
Nosocomial respiratory viral infections (NRVI) are increasingly documented in paediatric units. The most common aetiological viral agents that infect the respiratory system are respiratory syncytial virus, influenza virus and rhinovirus. Recently, a novel human coronavirus (HCoV) was identified as a causative agent of the severe acute respiratory syndrome (SARS). 1 Both HCoV‐229E and HCoV‐OC43 are HCoV that have been recognized for the past 30 years; 2 of the two, HCoV‐229E has been recently recognized as an important cause of NRVI in high‐risk infants. 3 , 4
Over the past decades, a number of phytochemicals have been reported to possess potent antiviral activity. 5 , 6 , 7 Saikosaponins represent a group of oleanane derivatives, usually as glucosides, that are found in a number of plant families. Saikosaponins isolated from medicinal plants such as Bupleurum spp., Heteromorpha spp. and Scrophularia scorodonia have been reported to possess various biological activities, namely antihepatitis, antinephritis, antihepatoma, anti‐inflammation and antibacterial effects, 8 , 9 , 10 as well as being able to modulate immune function. 11 , 12 Furthermore, saikosaponins (A, B2, C and D) have been shown to be active against human immunodeficiency virus (HIV), 13 measles, 14 influenza virus, 15 , 16 herpes simplex virus 14 and varicella‐zoster virus. 17 In an attempt to find new anti‐HCoV compounds, we conducted a series of experiments to investigate the antiviral activity and mode of action of saikosaponins against HCoV‐229E in vitro.
METHODS
Chemicals
Actinomycin D, dimethylsulphoxide (DMSO) and saikosaponins A, B2, C and D (Fig. 1) were purchased from Sigma Chemical (St Louis, MO, USA). 2,3‐Bis[2‐methoxy‐4‐nitro‐5‐sulfophenyl]‐5‐[(phenylamino) carbonyl‐2H‐tetrazolium hydroxide] (XTT) kits were purchased from Roche Diagnostics (Mannheim, Germany). Actinomycin D (as a positive control) and saikosaponins of purity greater than 95% were dissolved in DMSO. They were further diluted with RPMI 1640 medium (Sigma Chemical) to the required concentrations with the final concentration of DMSO no greater than 0.1%.
Figure 1.
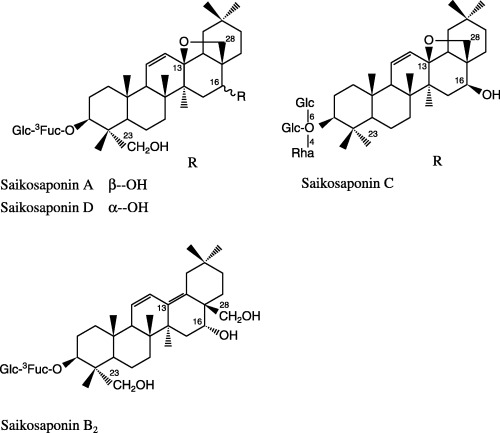
Structure of saikosaponins.
Virus and cells
The HCoV‐229E was obtained from American Type Culture Collection (ATCC; VR‐740; Rockville, MD, USA). Human fetal lung fibroblasts (MRC‐5; ATCC CCL‐171) were used as target cells for viral infection. Cells was grown in RPMI 1640 medium supplemented with 3% fetal calf serum (FCS), 100 units/mL penicillin G, 100 µg/mL streptomycin and 0.25 µg/mL amphotericin B at 37°C in a humidified atmosphere of 5% CO2. Virus titres were determined by cytopathic effects and were expressed as 50% tissue culture infective dose (TCID50) per mL. All viruses were stored at −70°C until use.
Cytotoxicity assay
The MRC‐5 cells were seeded onto a 96‐well plate at a concentration of 9 × 103 cells. A volume of 10 µL/well, with different concentrations of saikosaponins (A, B2, C and D), was applied to culture wells in triplicate. Dimethylsulphoxide (0.1%) and actinomycin D (0.2, 0.02 and 0.002 µmol/L) were used as negative and positive controls, respectively. After incubation at 37°C with 5% CO2 for 4 days, a mixture of 0.1 mL phenazine methosulphate (PMS; electron‐coupling reagent) and 50 µL XTT was added to each well. The trays were further incubated for 3 h to allow the production of XTT formazan. Absorbance was determined with an ELISA reader (Multiskan EX; Labsystems, Helsinki, Finland) at a test wavelength of 492 nm and a reference wavelength of 690 nm. Data were calculated as percentage inhibition using the following formula:
| Inhibition (%) = (100 − (At/As)) × 100 |
where At and As refer to the absorbance of the test substances and the solvent control, respectively. The 50% cellular cytotoxicity (CC50) concentration of test substances was calculated according to Chiang et al. 18 The selectivity index (SI) was calculated as the ratio of the CC50 to the EC50.
Antiviral assay using the XTT method
The antiviral activity of pure compounds against HCoV‐229E was evaluated using the XTT method. 18 The MRC‐5 cells, treated with trypsin, were seeded onto 96‐well plates at a concentration of 7 × 103 cells/mL and a volume of 70 µL/well. After incubation at 34°C with 5% CO2 overnight, 20 µL test virus (100 TCID50) was added and cells were incubated for a further 2 h. The XTT test was performed as described above. The percentage protection was calculated as:
| Protection (%) = (Atv − Acv)/(Acd − Acv) × 100 |
where Atv is the absorbance of the viral infected cells in the presence of test compounds and Acv and Acd are the absorbance of the virus control and cell control, respectively.
The antiviral concentration of 50% effectiveness (EC50) was defined as the concentration that achieved 50% inhibition of virus‐induced cytopathic effects. The number of viruses used in each experiment was based on infection target cells by 100 TCID50 of virus to produce 50% XTT formazan products as in uninfected control cells.
Time‐course and dose–response effects of saikosaponin B2 on HCoV‐229E
Twenty microlitres of 100 TCID50 HCoV‐229E per well was absorbed onto confluent monolayers of MRC‐5 cells for 2 h, followed by the addition of different concentrations of saikosaponin B2 (0, 0.2, 1 and 6 µmol/L) to culture cells at various times pre‐infection (−4 to −1 h), coinfection (0 h) or post‐infection (1–4 h) of the virus to MRC‐5 cells at 34°C. After 4 days incubation, the XTT test was performed and antiviral activity was determined as described above.
Attachment assay
The attachment assay was conducted according to the procedures described by Albin et al. 19 and De Logu et al. 20 with minor modification. Briefly, the MRC‐5 cell monolayer was grown in a 24‐well culture plate and then prechilled at 4°C for 1 h. After the medium had been aspirated, the cell monolayer was infected with 100 TCID50 HCoV‐229E in the absence or presence of various concentrations of saikosaponin B2 (0.75–25 µmol/L). After further incubation of the infected cell monolayer at 4°C for 3 h, the medium was aspirated to remove unabsorbed virus. The cell monolayer was then washed with phosphate‐buffered saline (PBS) three times and overlaid with 2% FCS medium. After 4 days of incubation, the XTT test was performed as described above. Absorbance was measured at a test wavelength of 450 nm and a reference wavelength of 690 nm using an ELISA reader. The percentage inhibition of attachment was calculated as:
| Inhibition (%) = (100 − (ODt/ODs)) × 100 |
where ODt and ODs are the optical density of the test substances and the solvent control, respectively.
Penetration assay
The penetration assay of HCoV‐229E into MRC‐5 cells was performed according to published procedures with minor modifications. 19 , 20 The MRC‐5 cell monolayer was grown in a 24‐well culture plate and prechilled at 4°C for 1 h. After the cell monolayer had been infected with 100 TCID50 HCoV‐229E and incubated at 4°C for 3 h to allow the attachment of HCoV‐229E, 6 µmol/L saikosaponin B2 or solvent control (0.1% DMSO) was added to the culture medium. The infected cell monolayer was incubated at 37°C to maximize the penetration of the virus. At 10 min intervals, the infected cell monolayer was treated with acidic PBS (pH 3) for 1 min to inactivate non‐penetrating virus. Immediately after the addition of PBS at pH 7 to neutralize acidic PBS (pH 3), the neutral PBS was removed and the cell monolayer was overlaid with 2% FCS medium. After 4 days of incubation, the XTT test was performed as described above. Absorbance was measured at a test wavelength of 450 nm and a reference wavelength of 690 nm using an ELISA reader. The percentage inhibition of penetration was calculated as:
| Inhibition (%) = (100 − (ODt/ODs)) × 100 |
where ODt and ODs indicate the optical density of the test substances and the solvent control, respectively.
Statistical analysis
Results are expressed as the mean±SD from three independent experiments. Statistical analysis was performed with one‐way anova, followed by the least significant difference (LSD) multiple range test. Differences between test compounds and control were evaluated using Student's t‐test. P < 0.05 was considered statistically different.
RESULTS
Cytotoxic effects of saikosaponins (A, B2, C, D) and actinomycin D on MRC‐5 cells
We first evaluated the cytotoxicity of saikosaponins A, B2, C, D and actinomycin D on MRC‐5 cells using the XTT assay. The results show that saikosaponins A, B2, C and D have no cytotoxic effect on the cells at concentrations of 2.5 µmol/L (Fig. 2). Interestingly, more than 60% cell viability was noted when saikosaponins A, B2, C, and D were used at a concentration of 25 µmol/L. Furthermore, no visible changes in cell morphology or cell density were observed at the concentrations tested. The concentrations estimated to reduce cell viability by 50% or the CC50 of the various saikosaponins were in the range 121.5–383.3 µmol/L, which was better than for actinomycin D (positive control; CC50 = 2.8 mol/L).
Figure 2.
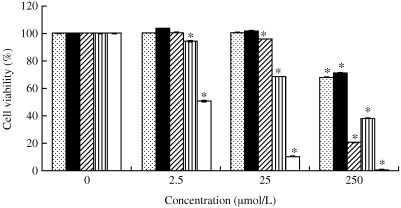
Efects of saikosaponin A (), B2 (▪), C (▨) and D (▥) and actinomycin D (□) on the viability of the human fetal lung fibroblast (MRC‐5) cell line. Cells were treated with various concentrations (0, 2.5, 25 and 250 µmol/L) of saikosaponins A, B2, C and D for 96 h. Cell viability was determined by the XTT assay. Data are the mean±SD of three independent experiments. *P < 0.05 compared with control (Student's t‐test).
Inhibitory effect of saikosaponins (A, B2, C and D) on HCoV‐229E
Figure 3 shows the antiviral activity of saikosaponins A, B2, C and D. The results show that saikosaponins A, B2, C and D, at concentrations of 25 µmol/L or less, significantly inhibited HCoV‐229E viral infection (P < 0.05), with saikosaponin B2 exhibiting the strongest potency. The percentage viral inhibition for 0.25, 2.5 and 25 µmol/L saikosaponin B2 was 35.7 ± 0.7, 63.0 ± 0.8 and 100.0 ± 0.2%, respectively.
Figure 3.
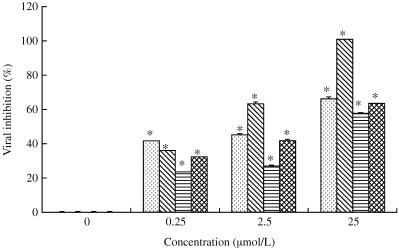
Antiviral activity of saikosaponin A (), B2 (▧), C (▤) and D (▩) against HCoV‐229E. The virus was treated with various concentrations (0, 0.25, 2.5 and 25 µmol/L) of saikosaponin A, B2, C and D for 96 h, followed by determination of viral inhibition using the XTT assay. Data are the mean±SD of three independent experiments. *P < 0.05 compared with control (Student's t‐test).
Table 1 gives the EC50 and SI values of saikosaponins A, B2, C and D in inhibiting infection by HCoV‐229E. Because saikosaponin B2 showed the strongest anticoronaviral activity and the best SI value, we investigated the mechanistic effects of this drug on viral infection further.
Table 1.
Assessement of anticoronaviral activity of saikosaponins
| Test drug | CC50 (µmol/L) | EC50 (µmol/L) | SI |
|---|---|---|---|
| Actinomycin D | 2.8 ± 0.3 | 0.02 ± 0.0 | 140 |
| Saikosaponin A | 228.1 ± 3.8 | 8.6 ± 0.3 | 26.6 |
| Saikosaponin B2 | 383.3 ± 0.2 | 1.7 ± 0.1* | 221.9 |
| Saikosaponin C | 121.5 ± 0.1 | 19.9 ± 0.1[Link], [Link] | 19.2 |
| Saikosaponin D | 176.2 ± 0.2 | 13.2 ± 0.3[Link], [Link], [Link] | 13.3 |
Data are the mean±SD of three independent experiments. *P < 0.05 compared with saikosaponin A; † P < 0.05 compared with saikosaponin B2; ‡ P < 0.05 compared with saikosaponin C (Student's t‐test).
CC50, the concentration that reduces the growth of target (MRC‐5) cells by 50%; EC50, the inhibitory concentration of compound that produces 50% inhibition of virus‐induced cytopathic effects; SI, selectivity index (= CC50/EC50).
Actinomycin D was used as a positive control.
Time‐of‐addition studies of saikosaponin B2 on viral replication
In order to investigate the mechanism as to how saikosaponin B2 inhibits infection by the HCoV‐229E virus, a study was conducted to investigate the time‐course of effects of various concentrations of saikosaponin B2 (0.2, 1.0 and 6 µmol/L) at 4 h before and 0–4 h after viral infection. The results show that saikosaponin B2 decreases infection by HCoV‐229E in a dose‐ and time‐dependent manner (Fig. 4). In addition, saikosaponin B2 was noted to inhibit viral infection more efficiently when it was added before viral adsorption (45.0–60.0, 67.6–74.9 and 91.8–99.6% at 0.2, 1.0 and 6 µmol/L saikosaponin B2, respectively) than if it was added after viral adsorption (19.3–34.6, 37.2–58.7 and 69.8–88.4% at 0.2, 1.0 and 6 µmol/L saikosaponin B2, respectively).
Figure 4.
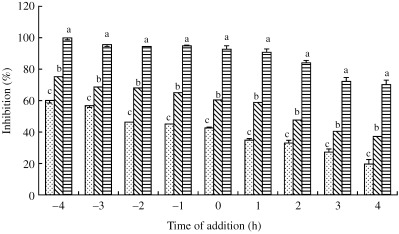
Time‐course of effects of saikosaponin B2 on HCoV‐229E. Different concentrations of saikosaponin B2 (0, 0.2 (), 1 (▧) and 6 µmol/L (▤)) were added at various times pre‐infection (−4 to −1 h), coinfection (0 h) or post‐infection (1–4 h) of HCoV‐229E to MRC‐5 cells at 34°C. After incubation for 4 days, the percentage inhibition was evaluated by the XTT method. Data are the mean±SD of three independent experiments. a,b,c P < 0.05 compared with control (one‐way anova followed by the LSD multiple range test).
Effect of saikosaponin B2 on viral attachment and penetration
According to the results of time‐of‐addition studies, saikosaponin B2 appeared to affect the early stage of HCoV‐229E infection. This observation suggests that saikosaponin B2 may disturb the early events of HCV‐229E infection, including viral attachment and penetration. Thus, the effect of saikosaponin B2 on viral attachment and penetration was investigated. The results show that HCoV‐229E attachment is significantly inhibited by saikosaponin B2 in a dose‐dependent manner (Fig. 5). The percentage attachment inhibition at concentrations of 0, 0.75, 1.5, 3, 6, 12 and 25 µmol/L saikosaponin B2 was 0, 10.4, 12.2, 20, 70.8, 81.8 and 89.3%, respectively. These results suggest that saikosaponin B2 prevents the attachment of HCoV‐229E into host cells (Fig. 5).
Figure 5.
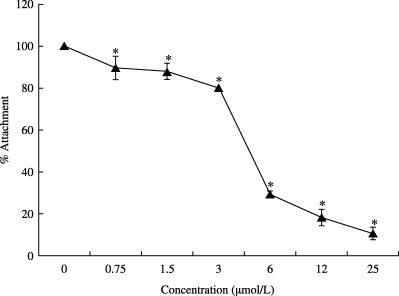
Effect of saikosaponin B2 on HCoV‐229E attachment. Cells were prechilled at 4°C for 1 h, followed by inoculation with HCoV‐229E (100 TCID50) of the cell monolayer in the absence or presence of test compound. After 3 h incubation, percentage attachment of the test compound‐treated group was evaluated using the XTT assay. Data are the mean±SD of three independent experiments. *P < 0.05 between different doses of saikosaponin B2 (one‐way anova).
Penetration of HCoV‐229E into host cells was inhibited by 6 µmol/L saikosaponin B2 in a time‐dependent manner, with an inhibition rate of 0, 17.3, 19, 21.9, 24.8, 31, 35, 38.9, 53.3 and 81.3% at 0, 10, 20, 30, 40, 50, 60, 70, 80 and 90 min, respectively (Fig. 6). This observation suggests that saikosaponin B2 also prevents the penetration of HCoV‐229E into host cells.
Figure 6.
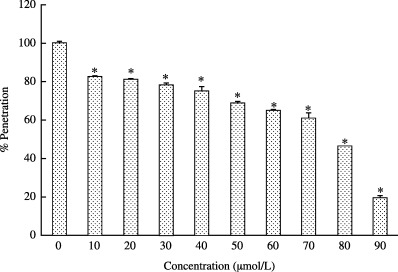
Effect of saikosaponin B2 on the penetration of HCoV‐229E to MRC‐5 cells. The cell monolayer was prechilled at 4°C for 1 h and then infected with 100 TCID50 HCoV‐229E at 4°C for 3 h. After 3 h incubation, 6 µmol/L saikosaponin B2 was added to the culture. At 10 min intervals, extracellular virus was inactivated by acidic phosphate‐buffered saline (PBS; pH 3) for 1 min. Then, PBS pH 7 was added to neutralize the acidic PBS. The neutral PBS was removed and 3% fetal calf serum was added. The percentage penetration of virus was evaluated using the XTT assay. Data are the mean±SD of three independent experiments. *P < 0.05 between different time points (one‐way anova).
DISCUSSION
In the present study, we demonstrated that saikosaponins (A, B2, C and D) possess anti‐HCoV‐229E activity, with saikosaponin B2 exhibiting the strongest potency. Furthermore, saikosaponins A and B2 showed no cytotoxic effect on cell viability. The time‐course study revealed that the anti‐HCoV‐229E activity of saikosaponin B2 was higher when it was added before the virus entered the host cells than if it was added after the virus infected host cells. These results suggest that saikosaponin B2 may interfere with the early stage of viral replication, such as the adsorption and penetration of the virus.
Natural products from plants and micro‐organisms have traditionally provided the pharmaceutical industry with many important leads in the search for new drugs. Over the past decades, many compounds isolated from medicinal plants have been found to possess inhibitory activity against viruses. 5 , 6 , 7 Viral infection remains a serious problem in many countries, coupled with the recent outbreak of SARS, for which no effective drug is available, and scientists worldwide are searching for new cures for this disease.
The present study indicates that saikosaponin B2 has potent activity against HCoV‐229E. With further investigation and a better understanding of its molecular mode of action, saikosaponin B2 could be a novel lead for the develoment of a potential chemopreventive agent for this viral infection. In previous studies, triterpenoid saponins have been shown to possess marked antiviral activity at concentrations at which the saponin showed no cytotoxic effect on Vero cells. 13 , 15 The mechanism of the antiviral activity was suggested to be associated with an early event in the viral infectious cycle, through the inhibition of virus–host cell attachment based on a direct interaction of saikosaponin D with the surface membrane of host cells. 16 , 17 In the present study, we postulated that the antiviral activity of saikosaponin B2 on anti‐HCoV‐229E could be through a direct inactivation of the virus or by direct interaction with the host cell membrane, resulting in morphological changes in the cell membrane. These events could consequently lead to an arrest in viral adsorption and penetration.
In previous studies, although saikosaponins have been shown to have inhibitory effect against herpes simplex type I (HSV‐1) in vitro, the mechanism of action was not elucidated. 21 In another study, saikosaponin C showed activity in inhibiting hepatitis B virus DNA replication, 12 whereas saikosaponin D was able to inactivate the measles virus and herpes simplex virus in vitro. 14 In other phytochemical studies, 22 baicalein has been reported to functionally block the epidermal growth factor receptor tyrosine kinase activity and human cytomegalovirus (HCMV) nuclear translocation, whereas genistein does not. The antiviral activity of genistein against HCMV infection was shown to inhibit early and late HCMV gene expression. 22 Quercetin and two 3‐substituted derivatives of kaempferol appeared to inhibit HIV infection by preventing binding of gp120 to CD4. 23 Allophycocyanin was able to delay viral RNA synthesis in infected cells. 24 To our knowledge, the present study is the first to demonstrate that saikosaponin B2 inhibits HCoV‐229E viral absorption and penetration.
Saikosaponin B2 inhibited HCoV‐229E attachment to the host cells and consequently led to the arrest of viral absorption and eventual viral penetration. In addition to affecting viral attachment, saikosaponin B2 also appears to prevent HCoV‐229E from penetrating into host cells, as demonstrated in the penetration experiment, in which HCoV‐229E failed to attach to host cells. The addition of saikosaponins was observed to block HCoV‐229E penetration, suggesting that saikosaponin B2 affects the viral penetration process, possibly through detaching virus that has already bound to the cell, perhaps by disturbing viral glycoproteins. 6
In conclusion, in the present study, saikosaponin B2 was found to: (i) inhibit HCoV‐229E viral infection at concentrations of 25 µmol/L or less; (ii) inhibit viral attachment to cells in a dose‐dependent manner; (iii) block viral penetration into cells; and (iv) interfere with the early stage of viral replication, such as virus absorption and penetration. The multiple modes of action of saikosaponin B2 merit further investigation.
REFERENCES
- 1. Ksiazek TG, Erdman D, Goldsmith CS et al. A novel coronavirus associated with severe acute respiratory syndrome. N. Engl. J. Med. 2003; 348: 1953–66. [DOI] [PubMed] [Google Scholar]
- 2. Hamre D, Procknow JJ. A new virus isolated from the human respiratory tract. Proc. Soc. Exp. Biol. Med. 1966; 121: 190–3. [DOI] [PubMed] [Google Scholar]
- 3. Sizun J, Gagneur A, Legrand MC, Baron R. Respiratory coronavirus infections in children. Pediatr. Infect. Dis. J. 2001; 20: 555–6. [DOI] [PubMed] [Google Scholar]
- 4. Gagneur A, Legrand MC, Picard B et al. Nosocomial infections due to human coronaviruses in the newborn. Arch. Pediatr. 2002; 9: 61–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Lin YM, Flavin MT, Schure R et al. Antiviral activities of biflavonoids. Planta Med. 1999; 65: 120–5. [DOI] [PubMed] [Google Scholar]
- 6. Cheng HY, Lin TC, Yang CM, Wang KC, Lin LT, Lin CC. Putranjivain A from Euphorbia jolkini inhibits both virus entry and late stage replication of herpes simplex virus type 2 in vitro . J. Antimicrob. Chemother. 2004; 53: 577–83. [DOI] [PubMed] [Google Scholar]
- 7. Chang JS, Liu HW, Wang KC et al. Ethanol extract of Polygonum cuspidatum inhibits hepatitis B virus in a stable HBV‐producing cell line. Antiviral Res. 2005; 66: 29–34. [DOI] [PubMed] [Google Scholar]
- 8. Guinea MC, Parellada J, Lacaille‐Dubois MA, Wagner H. Biologically active triterpene saponins from Bupleurum fruticosum . Planta Med. 1994; 60: 163–7. [DOI] [PubMed] [Google Scholar]
- 9. Recio MC, Just MJ, Giner RM, Manez S, Rios JL, Hostettmann K. Anti‐inflammatory activity of saikosaponins from Heteromorpha trifoliata . J. Nat. Prod 1995; 58: 140–4. [DOI] [PubMed] [Google Scholar]
- 10. Bermejo Benito P, Abad Martinez MJ, Silvan Sen AM et al. In vivo and in vitro antiinflammatory activity of saikosaponins. Life Sci. 1998; 63: 1147–56. [DOI] [PubMed] [Google Scholar]
- 11. Ushio Y, Abe H. The effects of saikosaponin on macrophage functions and lymphocyte proliferation. Planta Med. 1991; 57: 511–14. [DOI] [PubMed] [Google Scholar]
- 12. Chiang LC, Ng LT, Liu LT, Shieh DE, Lin CC. Cytotoxicity and anti‐hepatitis B virus activities of saikosaponins from Bupleurum species. Planta Med. 2003; 69: 705–9. [DOI] [PubMed] [Google Scholar]
- 13. Ito M, Nakashima H, Baba M et al. Inhibitory effect of glycyrrhizin on the in vitro infectivity and cytopathic activity of the human immunodeficiency virus [HIV (HTLV‐III/LAV)]. Antiviral Res. 1987; 7: 127–37. [DOI] [PubMed] [Google Scholar]
- 14. Ushio Y, Abe H. Inactivation of measles virus and herpes simplex virus by saikosaponin d. Planta Med. 1992; 58: 171–3. [DOI] [PubMed] [Google Scholar]
- 15. Rao GS, Sinsheimer JE. Antiviral activity of triterpenoid saponins containing acylated beta‐amyrin aglycones. J. Pharm. Sci. 1974; 63: 471–3. [DOI] [PubMed] [Google Scholar]
- 16. Amoros M, Fauconnier B, Girre RL. In vitro antiviral activity of a saponin from Anagallis arvensis, Primulaceae, against herpes simplex virus and poliovirus. Antiviral Res. 1987; 8: 13–25. [DOI] [PubMed] [Google Scholar]
- 17. Baba M, Shigeta S. Antiviral activity of glycyrrhizin against varicella‐zoster virus in vitro . Antiviral Res. 1987; 7: 99–107. [DOI] [PubMed] [Google Scholar]
- 18. Chiang LC, Chiang W, Chang MY, Ng LT, Lin CC. Antiviral activity of Plantago major extracts and related compounds in vitro . Antiviral Res. 2002; 55: 53–62. [DOI] [PubMed] [Google Scholar]
- 19. Albin R, Chase R, Risano C et al. SCH 43478 and analogs: In vitro activity and in vivo efficacy of novel agents for herpesvirus type 2. Antiviral Res. 1997; 35: 139–46. [DOI] [PubMed] [Google Scholar]
- 20. De Logu A, Loy G, Pellerano ML, Bonsignore L, Schivo ML. Inactivation of HSV‐1 and HSV‐2 and prevention of cell‐to‐cell virus spread by Santolina insularis essential oil. Antiviral Res. 2000; 48: 177–85. [DOI] [PubMed] [Google Scholar]
- 21. Bermejo P, Abad MJ, Diaz AM et al. Antiviral activity of seven Iridoids, three saikosaponins and one phenylpropanoid glycoside extracted from Bupleurum rigidum and Scrophularia scorodonia . Planta Med. 2002; 68: 106–10. [DOI] [PubMed] [Google Scholar]
- 22. David LE, Chao CF, Wang X, Zhang Z, Huong SM, Huang ES. Human cytomegalovirus‐inhibitory flavonoids: Studies on antiviral activity and mechanism of action. Antiviral Res. 2005; 68: 124–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Mahmood N, Piacente S, Pizza C, Burke A, Khan AI, Hay AJ. The anti‐HIV activity and mechanisms of action of pure compounds isolated from Rosa damascena . Biochem. Biophys. Res. Commun. 1996; 229: 73–9. [DOI] [PubMed] [Google Scholar]
- 24. Shih SR, Tsai KN, Li YS, Chueh CC, Chan EC. Inhibition of enterovirus 71‐induced apoptosis by allophycocyanin isolated from a blue‐green alga Spirulina platensis . J. Med. Virol. 2003; 70: 119–25. [DOI] [PubMed] [Google Scholar]


