Abstract
Rituximab in combination with chemotherapy has shown efficacy in patients with diffuse large B-cell lymphoma (DLBCL) for more than 15 years. HLX01 was developed as the rituximab biosimilar following a stepwise approach to demonstrate biosimilarity in analytical, pre-clinical, and clinical investigations to reference rituximab. With demonstrated pharmacokinetic similarity, a phase 3 multi-center, randomized, parallel, double-blind study (HLX01-NHL03) was subsequently conducted to compare efficacy and safety between HLX01 plus cyclophosphamide, doxorubicin, vincristine, and prednisone (H-CHOP) and reference rituximab plus CHOP (R-CHOP) in a total of 407 treatment-naïve, CD20-positive DLBCL patients aged 18–80 years. The primary efficacy endpoint was best overall response rate (ORR) within six cycles of treatment in the per-protocol set (PPS). Secondary endpoints included 1-year efficacy outcomes, safety, and immunogenicity profile. The results showed difference in ORRs [H-CHOP 94.1%; R-CHOP 92.8%] between two treatment groups was 1.4% (95% confidence interval [CI], − 3.59 to 6.32, p = 0.608) which falls within the pre-defined equivalence margin of ± 12%. The safety profile was comparable between the treatment groups, with a similar overall incidence of treatment-emergent adverse events (H-CHOP 99.5%, R-CHOP 99.0%, p = 1.000) and serious adverse events (H-CHOP 34.0%, R-CHOP 32.5%, p = 0.752). This study established bioequivalence in efficacy and safety between HLX01 and reference rituximab. The trial was registered at http://www.chinadrugtrials.org.cn on 26 August 2015 [#CTR20150583].
Keywords: Rituximab biosimilar, DLBCL, Efficacy equivalence
Treatment with rituximab, a monoclonal antibody against CD20, in combination with cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) has been used in patients with diffuse large B-cell lymphoma (DLBCL) for more than 15 years with proven efficacy and safety [1]. With demonstrated highly similar analytical characterization and bioequivalence in pharmacokinetics and pharmacodynamics [2], we conducted this phase 3, multi-center, randomized, parallel, double-blind study (HLX01-NHL03) to establish the equivalence in clinical efficacy, safety, and immunogenicity between HLX01 plus CHOP (H-CHOP) and R-CHOP every 21 days for up to six cycles in treatment-naïve patients with CD20-positive DLBCL.
Eligible patients were treatment-naïve adults (≥ 18 to ≤ 80 years) with International Prognostic Index of 0-2, clinical stages I–IV (Ann Arbor Staging) and histologically confirmed CD20-positive DLBCL. The primary efficacy endpoint was best overall response rate (ORR) within six cycles of treatment in the per-protocol set (PPS), and secondary efficacy endpoints included complete response rate, 1-year duration of response, 1-year event-free survival, 1-year progression-free survival, 1-year disease-free survival, 1-year overall survival, and depletion of CD19-positive B-cells in peripheral blood.
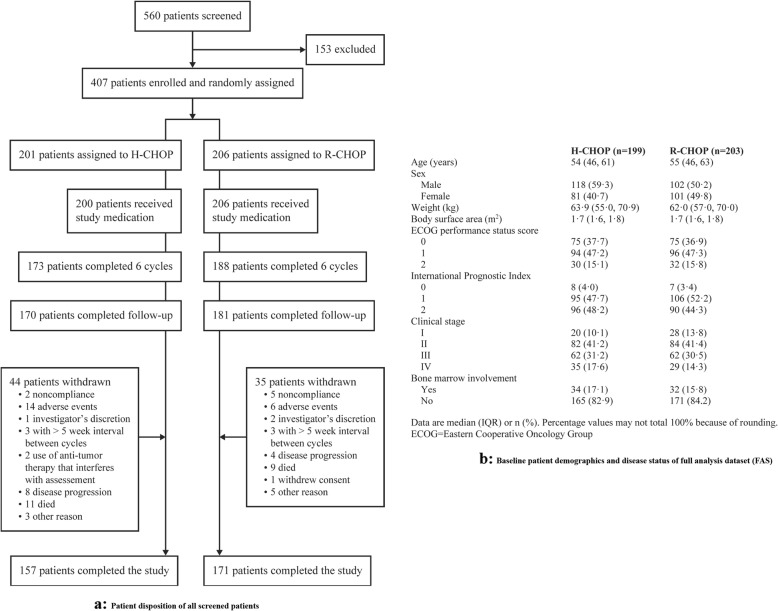
From October 9, 2015 to March 10, 2017, a total of 560 patients were screened, of whom 407 patients were randomized (1:1) at 33 investigational sites; 361 patients (H-CHOP 173; R-CHOP 188) completed six cycles of treatment, and 328 patients (H-CHOP 157; R-CHOP 171) completed the study (Fig. 1a). Baseline characteristics are well balanced between two treatment groups (Fig. 1b). In the PPS, the best ORRs within six cycles of treatment in the PPS were 94·1% (95% confidence interval [CI], 89.77 to 97.04) and 92·8% (95% CI, 88.19 to 96.00) in the H-CHOP and R-CHOP groups, respectively, with an intergroup difference of 1.4% (95% CI, − 3.59 to 6.32, p = 0.608). The efficacy equivalence between HLX01 and reference rituximab was demonstrated with 95% CIs falls entirely within the pre-defined margin of ± 12%. The results of using the full analysis set (FAS) were consistent with the primary efficacy analysis in the PPS. Previous reports of R-CHOP in patients with DLBCL have shown ORRs ranging between 83% and 88% [3, 4], which is comparable with the result from this study. No significant differences were observed in the 1-year analysis of all secondary efficacy endpoints, in either the PPS or the FAS (Table 1).
Fig. 1.
a Patient disposition of all screened patients. b Baseline patient demographics and disease status of full analysis dataset (FAS)
Table 1.
Efficacy outcomes
| Per-protocol dataset | Full analysis dataset | |||||
|---|---|---|---|---|---|---|
| H-CHOP (n = 188) | R-CHOP (n = 194) | P value | H-CHOP (n = 199) | R-CHOP (n = 203) | P value | |
| Best overall response rate | 177 (94·1) | 180 (92·8) | 0·608 | 184 (92·5) | 187 (92·1) | 0·839 |
| Complete response | 88 (46·8) | 101 (52·1) | 0·231 | 90 (45·2) | 104 (51·2) | 0·190 |
| Partial response | 89 (47·3) | 79 (40·7) | 94 (47·2) | 83 (40·9) | ||
| Stable disease | 8 (4·3) | 13 (6·7) | 11 (5·5) | 15 (7·4) | ||
| Disease progression | 2 (1·1) | 1 (0·5) | 2 (1·0) | 1 (0·5) | ||
| No evidence of disease | 1 (0·5) | 0 | 2 (1·0) | 0 | ||
| Duration of response | ||||||
| Patients experiencing events | 25 (13·8) | 21 (11·5) | 0·424 | 26 (13·7) | 21 (11·1) | 0·355 |
| Patients censored | 156 (86·2) | 161 (88·5) | 164 (86·3) | 168 (88·9) | ||
| Event-free survival | ||||||
| Patients experiencing events | 80 (42·6) | 67 (34·5) | 0·125 | 88 (44·2) | 71 (35·0) | 0·087 |
| Patients censored | 108 (57·4) | 127 (65·5) | 111 (55·8) | 132 (65·0) | ||
| 1-year event-free survival rate | 55·4 (47·9, 63·0) | 64·5 (57·6, 71·4) | 53·7 (46·4, 61·0) | 63·4 (56·6, 70·2) | ||
| Progression-free survival | ||||||
| Patients experiencing events | 31 (16·5) | 29 (14·9) | 0·534 | 33 (16·6) | 30 (14·8) | 0·473 |
| Patients censored | 157 (83·5) | 165 (85·1) | 166 (83·4) | 173 (85·2) | ||
| 1-year progression-free survival rate | 75·0 (66·5, 83·6) | 80·1 (73·5, 86·7) | 74·1 (65·6, 82·7) | 79·7 (73·1, 86·3) | ||
| Overall survival | ||||||
| Patient deaths | 15 (8·0) | 13 (6·7) | 0·661 | 16 (8·0) | 14 (6·9) | 0·701 |
| Patients censored | 173 (92·0) | 181 (93·3) | 183 (92·0) | 189 (93·1) | ||
| 1-year overall survival rate | 91·8 (87·8, 95·8) | 92·4 (88·3, 96·6) | 91·6 (87·6, 95·5) | 92·1 (88·0, 96·3) | ||
| Disease-free survival | ||||||
| Patients experiencing events | 27 (14·4) | 24 (12·4) | 0·462 | 28 (14·1) | 25 (12·3) | 0·477 |
| Patients censored | 161 (85·6) | 170 (87·6) | 171 (85·9) | 178 (87·7) | ||
| 1-year disease-free survival rate | 77·4 (68·9, 85·9) | 83·0 (76·7, 89·3) | 76·9 (68·4, 85·4) | 82·6 (76·2, 88·9) | ||
Data are n (%) or %, (95% CI). Percentage values may not total 100% because of rounding
The safety analysis set (Table 2) comprised 406 patients who received at least one treatment. 199/200 in H-CHOP group and 204/206 in R-CHOP group (H-CHOP 99.5%, R-CHOP 99.0%, p = 1.000) experienced at least one treatment-emergent adverse event; 68/200 in H-CHOP and 67/206 in R-CHOP (H-CHOP 34.0%, R-CHOP 32.5%, p = 0.752) experienced at least one serious adverse event; 14/200 in H-CHOP and 9/206 in R-CHOP (H-CHOP 7.0%, R-CHOP 4.4%, p = 0.252) discontinued treatment because of adverse events (AEs). The most common AEs were hematological events such as decreased white blood cell count (H-CHOP 85.5%; R-CHOP 85.9%), decreased neutrophil count (H-CHOP 79.0%; R-CHOP 81.6%), and anemia (H-CHOP 38.5%; R-CHOP 35.0%).
Table 2.
Safety profiles in the safety analysis dataset
| H-CHOP (n = 200) | R-CHOP (n = 206) | |
|---|---|---|
| Patients with ≥1 TEAE | 199 (99·5) | 204 (99·0) |
| Patients with ≥1 SAE | 68 (34·0) | 67 (32·5) |
| Patients with ≥1 AE leading to treatment discontinuation | 14 (7) | 9 (4·4) |
| Patients deaths due to AE | 5 (2·5) | 3 (1·5) |
| Adverse events with an incidence ≥10% | ||
| Hematological | ||
| Decreased white blood cell count | 171 (85·5) | 177 (85·9) |
| Decreased neutrophil count | 158 (79·0) | 168 (81·6) |
| Anemia | 77 (38·5) | 72 (35·0) |
| Decreased platelet count | 34 (17·0) | 19 (9·2) |
| Decreased lymphocyte count | 24 (12·0) | 34 (16·5) |
| Decreased hemoglobin concentration | 23 (11·5) | 20 (9·7) |
| Non-hematological | ||
| Nausea | 46 (23·0) | 49 (23·8) |
| Increased alanine aminotransferase | 49 (24·5) | 38 (18·4) |
| Fever | 47 (23·5) | 34 (16·5) |
| Decreased appetite | 32 (16·0) | 42 (20·4) |
| Increased lactate dehydrogenase | 30 (15·0) | 40 (19·4) |
| Debilitation | 38 (19·0) | 31 (15·0) |
| Alopecia | 35 (17·5) | 34 (16·5) |
| Increased aspartate aminotransferase | 34 (17·0) | 30 (14·6) |
| Cough | 31 (15·5) | 26 (12·6) |
| Vomiting | 22 (11·0) | 30 (14·6) |
| Upper respiratory tract infection | 19 (9·5) | 29 (14·1) |
| Hypokalemia | 28 (14·0) | 17 (8·3) |
| Constipation | 27 (13·5) | 25 (12·1) |
| Non-infectious pneumonia | 19 (9·5) | 24 (11·7) |
| Pulmonary infection | 19 (9·5) | 24 (11·7) |
| Diarrhea | 16 (8·0) | 22 (10·7) |
| Chills | 20 (10·0) | 14 (6·8) |
| Adverse events by CTCAE Grade | ||
| Grade 1 | 8 (4·0) | 6 (2·9) |
| Grade 2 | 35 (17·5) | 35 (17·0) |
| Grade 3 | 54 (27·0) | 75 (36·4) |
| Grade 4 | 98 (49·0) | 85 (41·3) |
| Grade 5 | 4 (2·0) | 3 (1·5) |
| Grade 4 adverse events with an incidence ≥2·5% | ||
| Decreased neutrophil count | 85 (42·5) | 75 (36·4) |
| Decreased white blood cell count | 44 (22·0) | 42 (20·4) |
| Febrile neutropenia | 5 (2·5) | 6 (2·9) |
| Bone marrow failure | 5 (2·5) | 5 (2·4) |
Data are n (%). Percentage values may not total 100% because of rounding
Among the patients observed with infusion-related reactions (IRRs), 61/200 in H-CHOP group and 61/206 in R-CHOP group (H-CHOP 30.5%; R-CHOP 29.6%), the most common reactions were those affecting skin and subcutaneous tissues. Most IRRs were grade 1 or 2, and no grade 4 or 5 IRRs were reported. Increases in hepatitis B virus (HBV) DNA titer were observed in five patients in H-CHOP group and eight patients in R-CHOP group, and nine of whom were receiving antiviral therapy for chronic HBV; however, no patients developed signs or symptoms of fulminant hepatitis.
Anti-drug antibodies (ADAs) were detected in one patient (< 1%) in each treatment group at baseline and immediately before administration of the second treatment cycle. After 6 months of follow-up, ADAs were detected in one patient in H-CHOP group and two patients in R-CHOP group (H-CHOP 1.0%, R-CHOP 1.7%, p = 1.000), and after 8 months of follow-up in seven patients in H-CHOP group and six patients in R-CHOP group (H-CHOP 7.1%, R-CHOP 5.5%, p = 0.629). During the entire study, only one patient in R-CHOP group had both ADAs and neutralizing antibodies.
In conclusion, this study demonstrated therapeutic equivalence between HLX01 and reference rituximab. The analysis of the primary and secondary efficacy endpoints did not reveal any statistically significant differences between two treatment groups. The safety and immunogenicity profiles of HLX01 were comparable with reference rituximab with no clinically meaningful differences observed between two treatment groups.
Acknowledgements
We thank all the patients and families who were involved in the HLX01-NHL03 study and the clinical study teams and Hangzhou Tigermed Consulting Co., Ltd. for providing support for the study.
Abbreviations
- R-CHOP
Rituximab plus cyclophosphamide, hydroxydaunorubicin, oncovin (vincristine), and prednisone
- DLBCL
Diffuse large B-Cell lymphoma
- H-CHOP
HLX01 plus cyclophosphamide, hydroxydaunorubicin, oncovin (vincristine), and prednisone
- ORR
Best overall response rate
- PPS
Per-protocol set
- CI
Confidence interval
- FAS
Full analysis set
- AE
Adverse event
- IRR
Infusion-related reaction
- HBV
Hepatitis B virus
- ADA
Anti-drug antibody
Authors’ contributions
YKS was the lead principal investigator and contributed to the design and conception, interpretation data, and writing of the manuscript. YKS, YPS, YQ, QYZ, XHH, XNH, DW, WL, YZ, JFF, JMY, HLZ, CJ, YY, JDH, ZW, ZMJ, HS, HQW, HYY, WJF, MZZ, XHZ, YC, XYK, LL, DY, GAC, XLW, JJ, TS, XD, YC, PYY, and XLZ contributed to data collection. XHH contributed to detecting CD19- and CD20-positive B-cells in peripheral blood. JCC contributed to statistical data analysis and interpretation of the data. EL contributed to the design and conception of the research. CMM, AL, EL, and XZ contributed to the interpretation of data and the completeness and accuracy of the data. KC and AL provided writing assistance for the initial manuscript and editorial assistance for reviewing and editing subsequent versions of the manuscript. The authors read and approved the final manuscript.
Funding
The study was supported by the sponsor, Shanghai Henlius Biotech, Inc., and by grants from the China National Major Project for New Drug Innovation (Grant No. 2015ZX09501008, 2017ZX09304015) and the Chinese Academy of Medical Sciences (CAMS) Innovation Fund for Medical Sciences (CIFMS) (Grant No. 2016-I2M-1-001).
Availability of data and materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Ethics approval and consent to participate
The protocol was developed by the sponsor of the study, Shanghai Henlius Biotech, Inc. The study protocol was reviewed and approved by the relevant independent ethics committee at each participating study center. Written informed consent was obtained from all study participants prior to screening. Data were collected by the site investigators who vouch for the completeness and accuracy of the data and the fidelity of the trial to the protocol. The sponsor analyzed the data. The study was conducted in accordance with the Declaration of Helsinki, Guideline for Good Clinical Practice, and applicable national and local regulations for clinical trials.
Consent for publication
Not applicable.
Competing interests
CMM, JCC, KC, AL, EL, and XZ are employees of Shanghai Henlius Biotech, Inc. All other authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Coiffier B, Lepage E, Brière J, Herbrecht R, Tilly H, Bouabdallah R, et al. CHOP Chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-Cell lymphoma. The N Engl J Med. 2002;346(4):235–242. doi: 10.1056/NEJMoa011795. [DOI] [PubMed] [Google Scholar]
- 2.Xu Y, Xie L, Zhang E, Gao W, Wang L, Cao Y, et al. Physicochemical and functional assessments demonstrating analytical similarity between rituximab biosimilar HLX01 and the MabThera®. MAbs. 2019;11(3):606–620. doi: 10.1080/19420862.2019.1578147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pfreundschuh M, Kuhnt E, Trümper L, Österborg A, Trneny M, Shepherd L, et al. CHOP-like chemotherapy with or without rituximab in young patients with good-prognosis diffuse large-B-cell lymphoma: 6-year results of an open-label randomised study of the MabThera International Trial (MInT) Group. Lancet Oncol. 2011;12(11):1013–1022. doi: 10.1016/S1470-2045(11)70235-2. [DOI] [PubMed] [Google Scholar]
- 4.Pfreundschuh M, Truemper L, Gill D, Osterborg A, Pettengell R, Trneny M, et al. First analysis of the Completed Mabthera International (Mint) Trial in young patients with low-risk diffuse large B-cell lymphoma (DLBCL): addition of rituximab to a CHOP-like regimen significantly improves outcome of all patients with the identification of a very favorable subgroup with IPI=O and no bulky disease. Blood. 2004;104(11):157. doi: 10.1182/blood.V104.11.157.157. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.