Summary
Gram‐positive, nonpathogenic lactic acid bacteria (LAB) are considered to be promising candidates for the development of novel, safe production and delivery systems of heterologous proteins. Recombinant LAB strains were shown to elicit specific systemic and mucosal immune responses against selected antigens. For this reason, this group of bacteria is considered as a potential replacement of classical, often pathogenic, attenuated microbial carriers. Mucosal administration of recombinant LAB, especially via the best explored and universal oral route, offers many advantages in comparison to systemic inoculation, and is attractive from the immunological and practical point of view. Research aimed at designing efficient, mucosally applied vaccines in combination with improved immunization efficiency, monitoring of in vivo antigen production, determination of optimal dose for vaccination, strain selection and characterization is a priority in modern vaccinology. This paper summarizes and organizes the available knowledge on the application of LAB as live oral vaccine vectors. It constitutes a valuable source of general information for researchers interested in mucosal vaccine development and constructing LAB strains with vaccine potential.
Keywords: gut microflora, heterologous gene expression, lactic acid bacteria, oral administration, vaccine vectors
Introduction
Vaccination is the most effective method of preventing and controlling infectious diseases, leading often to eradication of contagious factors from the environment. For this reason, vaccines play a profound role in the improvement of human and animal health. The World Health Organization (WHO) reported 25 licensed vaccines against most common and dangerous maladies available on the market in 2012 (WHO 2013). Induction of an effective immune response to a specific antigen is considered to be the main goal of vaccination. This approach should provide long‐lasting protection against particular infections. Recently, many researchers focused their interest on development of new, safe, mucosally administered vaccines, which production is less time‐ and cost‐consuming, less laborious and which are more easily applied. Additionally, such vaccines are thought to prevent carriage of pathogens in the population with very limited influence on maternal antibodies in infants.
Most often vaccines are based directly on pathogenic antigens. However, when such preparations are administered orally, they induce low or nonexisting immune responses. The reasons of such low efficacy are most probably connected with ineffective microbial adsorption, fast antigen degradation and induction of mucosal tolerance. Yet, although development of efficient oral vaccines may pose many difficulties, advantages of such approach are still predominant. Currently, there are two major trends in development of mucosally applied vaccines. One is based on synthetic systems for oral antigen delivery, such as emulsions, microparticles, immune‐stimulating complexes, liposomes (Saroja et al. 2011). The other strategy relies on live viral or bacterial vectors, enabling antigen production in vivo. In case of pathogenic bacteria, often used for vaccine development, construction of stable recombinant strains that will synthesize sufficient antigen amounts with no adverse effects on the vaccinated host or the environment can be problematic. An innovative solution is the use of lactic acid bacteria (LAB), which possess the so‐called ‘GRAS status’, meaning that they are nonpathogenic and their application is safe for humans and animals. The absence of lipopolysaccharides (LPS) in their cell wall is a great advantage that eliminates the risk of endotoxic shock. Additionally, food industry has long and successful experience in large‐scale production and safe storage of these bacteria, which can simplify preparation and shelving of the potential vaccine (Holzapfel et al. 1998). LAB are mostly known for their widespread use as starter strains in food and feed fermentation technology, but also for the probiotic effect that some species or strains have on humans and animal health (Mercenier et al. 2000). Most LAB are quite acid resistant which enables them to effectively survive passage through the stomach — a vital point in oral vaccine administration. Additionally, many different approaches have been developed to improve the viability of LAB during the gastrointestinal transit (Vidhyalakshmi et al. 2009). Nowadays, the most common technique used to protect bacterial cells against degradation in the gut and/or to target antigens for uptake into the gut‐associated lymphoid tissue (GALT) is encapsulation into microparticles or liposomes. Such approach might also enhance adhesion of the formed particles to mucosal surfaces (Plant and Lapatra 2011) and provide possible adjuvant properties by inducing local inflammatory signals. Encapsulation can be successfully applied for preservation of live vector vaccines with surface‐located antigens anchored in the cell wall. This solution effectively helps to avoid degradation of the externally exposed protein during passage through the gastrointestinal tract (GIT). In case of intracellularly localized antigens, it is assumed that they might not need further encapsulation since the cell wall itself acts as a natural protection barrier against the intestinal environment. Other, more simple methods of preserving the viability of LAB are based on application of appropriate liquid carriers with protective properties, such as trehalose (Jain and Roy 2009; Szatraj et al. 2014).
Some strains, mostly belonging to the Lactobacillus genus, effectively colonize host cavities, where they play a crucial role in maintaining a balance of the natural microbiota. Adhesive properties are considered to be an important quality also for recombinant LAB‐based vaccines, by ensuring long‐lasting persistence in the host and possibly extending the time of antigen presentation to the immune system.
Specific lactic acid bacterial strains can exhibit immunomodulatory effects on human and animal organisms. This observation has been confirmed also for recombinant LAB cells producing various antigens from infectious diseases, allergy promoting proteins and therapeutic antibodies (Capron et al. 1995). Although the precise molecular mechanism of LAB‐induced immunomodulation has not been fully examined, they were determined to affect dendritic cells (DC) maturation and induce cytokine secretion through toll‐like receptors (TLR), including TLR2. A number of studies evidenced the role of LAB in peripheral T‐cell hyporesponsiveness and promotion of regulatory T‐cell development by DC modulation (Dubois et al. 2003; Rigaux et al. 2009; You et al. 2014).
The natural adjuvanticity of LAB is an attractive feature in oral vaccine development. Moreover, LAB have also been shown to be efficient in producing many different heterologous antigens (e.g. Helicobacter pylori urease, LcrV antigen from Yersinia pseudotuberculosis, EP7 antigen of the human type 16 papilloma virus, haemagglutinin (HA) of avian influenza virus) (Bermúdez‐Humarán et al. 2002; Szatraj et al. 2014; Zhou et al. 2015). All mentioned aspects make LAB attractive and promising hosts for oral vaccine production and delivery of many different compounds (Fig. 1).
Figure 1.
Actual LAB application. [Colour figure can be viewed at http://wileyonlinelibrary.com]
This work summarizes and organizes available knowledge about ongoing research on the application of LAB for heterologous protein production and oral mucosal delivery of many different molecules acting as antigens. We hope this article will provide actual and informative data on the new possibilities in vaccine development, especially of orally administered formulations, but also give practical ideas helpful while planning LAB vaccine experiments. The article also discusses the issue of genetic modifications that can lead to development of LAB strains with new, vaccine potential for application in human or animal medicine.
LAB and mucosa‐associated lymphoid tissue
Current research shows that LAB can be efficiently engineered to produce and deliver different proteins, including therapeutics, to mucosal surfaces. The most popular antigen target is the GALT, although nasal‐associated lymphoid tissue (NALT) and genital mucosal surfaces (VALT) are also taken into consideration in vaccine trials. Although nasal immunizations have been proven effective in a number of studies (Falcone et al. 2006; Lee et al. 2006; Oliveira et al. 2006; Hanniffy et al. 2007; Campos et al. 2008), this route of vaccination is chosen primarily against respiratory infectious diseases including tuberculosis, coronavirus, influenza, respiratory syncytial virus, etc. As for VALT, this immunization approach is less sufficient (Ogra et al. 2001) and requires additional stimulation, usually via the oral path, as was shown for cholera vaccine (Kozlowski et al. 1997). Interactions between LAB and LAB‐associated vaccines with GALT are the most extensively explored and will be addressed in the following section.
The GIT is intensively exposed to pathogens and various biologically active factors, among them antigens and carcinogens. Lymphatic tissue associated with the gut plays a crucial role in the local and systemic immune response. Its role relies on mediating the migration and homing of the activated immune cells from the gut to other sites of the body. The mucosal immune system is responsible for about 60% of the daily immunoglobulin production. LAB are among bacteria which occur physiologically in animals and humans, mainly in their digestive tracts. They ensure the balance in the composition of the gut microflora and participate with other factors in regulation of many physiological processes, such as allergies and inflammations (Gonzaga et al. 2009). Although the main role of LAB in dairy food production is milk fermentation, selected strains from this group of bacteria are also known for health‐promoting effects, including enhancement of non and specific immune responses, as well as control of intestinal infections and anti‐tumorigenic activity. Through their actions, based on activation of inflammatory immune responses (IgA antibodies production), production of exopolysaccharides and antibiotic‐like substances, LAB assure the integrity of the gut mucous membrane. They also influence the distribution and the number of lymphoid cells in lymphatic tissues associated with the gut. However, the molecular mechanisms by which LAB affect the immune system are unknown. Probably, LAB alone or products of their metabolism are absorbed by M‐cells and transported to lymphatic follicles where they are analysed by the immune system (Mestecky 1987; Ernst et al. 1988; Lidbeck and Nord 1993; Gibson and Wang 1994). It is possible that LAB can directly impact gut epithelium cells, which contain lymphocytes able to produce a wide scale of cytokines, and influence local immunity (Fig. 2) (Galdeano et al. 2010).
Figure 2.
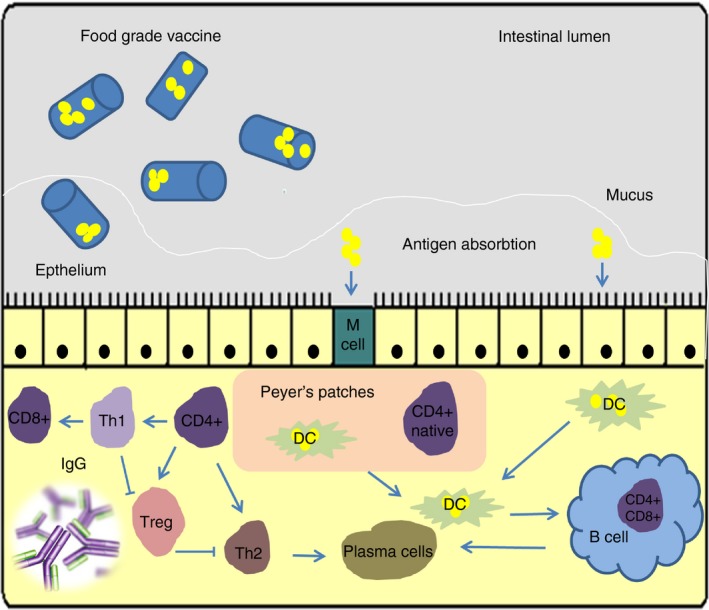
Food‐grade vaccines based on lactic acid bacteria. After oral administration of food‐grade vaccine, the mucosal and systemic responses are stimulated after antigen presentation by M‐cells (M) and dendritic cells (DC) in the intestine. In consequence, lymphocytes induce adaptive reactions that involve IgA, IgG production and recruitment of cytotoxic cells. [Colour figure can be viewed at http://wileyonlinelibrary.com]
Interactions of LAB and products of their metabolism with immunocompetent cells, such as macrophages and T‐cells, can stimulate production of cytokines. It is hypothesized that cytokines can be induced by LAB through two possible pathways: (i) via antigen presentation to T‐lymphocytes or (ii) after direct interaction between LAB and immunocompetent cells. These speculations are reinforced by the fact that lymphocytes and macrophages are equipped with specific receptors for LAB peptidoglycan (Fuller 1997). Two different scientific groups proved that the peptidoglycan of LAB can induce the secretion of interleukin (IL)‐1 (stimulation of T‐ and B‐cell proliferation), IL‐6 (induction of B‐cell differentiation) and TNF‐α (anti‐tumorigenic activity) by monocytes (Fernandes and Shahani 1990; Bhakdi and Tranumjensen 1991; Dziarski 1991). TNF‐α interferon induces expression of MHC class I and class II antigens, stimulates T‐helper lymphocytes, activates macrophages which can all enhance vaccine immunogenicity (Heumann et al. 1994). The most important factors that influence the immunomodulative character of LAB are connected with:
the ability to modulate the immune system by particular strains;
doses which are dependent on the applied strain and the vaccination scheme (Murray 1988; Perdigón and Alvarez 1992; de Petrino et al. 1995)
age and physiological state of the host.
In regards to the immune modulation, an important feature of LAB is that they can be internalized in the gut via different pathways, which enables them to induce different immune responses. The interaction of LAB with M‐cells activates mainly specific immune responses, while interaction with FAE cells (follicle‐associated epithelial cells) induces a nonspecific or inflammatory response. Contact of LAB with epithelial cells can lead to enhancement of local immunity (Perdigón et al. 2002). It has been shown that Lactobacillus casei, Lactobacillus delbrueckii ssp. bulgaricus and Lactobacillus acidophilus affect the systemic humoral immune response, which can be an attribute during selection of appropriate lactobacilli strains as effective antigen carriers. Studies show that the choice of specific LAB for oral administration is crucial for directed modulation of the systemic immune response according to the observed cytokine profiles. A significant increase of interleukins IL‐10 and IL‐4 levels was observed in mice fed with Lact. delbrueckii ssp. bulgaricus or Lact. casei. In turn, induction of IL‐2 and IL‐12 was observed in case of Lact. acidophilus. The role of produced cytokines in the balance Th1/Th2 was determined. It was reported that Lact. casei, Lact. delbrueckii ssp. bulgaricus all increased the IgG1 response in favour of Th2. Lactobacillus acidophilus induced the IgG2a response, with the advantage for Th1. In turn, Streptococcus thermophilus did not affect the Th1/Th2 balance. Other studies also confirmed the effect of various LAB strains in stimulating different mucosal cytokine profiles (Martin and Lew 1998; Hershberg and Mayer 2000). Thus, for vaccine development, selection of strains with specific immunological properties must be well defined to induce the desired immune response and cytokine expression.
Lactic acid bacteria strains can induce also nonspecific immune responses (the first line of defence), which are connected with monocytes, macrophages, neutrophils and NK cells. Phagocytosis leads to intracellular reactions, and production of reactive oxygen and nitrogen radicals, as well as additional factors, like TNF‐α and IL‐1 (Moineau and Goulet 1991; Balasubramanya et al. 1995; Tizard 2000). Results from various animal studies show that the level of phagocytic cell functions, as in case of systemic immune response, is strain‐dependent. Secretion of lysosomal enzymes by macrophages in mice fed fermented milk containing Lact. casei was more effective compared to animals supplemented with Lact. acidophilus or Strep. thermophilus. Tortuero and Fernandez (1995) reported an increase in IL‐2 concentration in ileal tissues in piglets treated with Streptococcus faecium M‐74 and Lact. casei spp. Histological examination indicated an increased phagocytic activity of these cells. The differences in cell‐wall composition were suggested to be responsible for the different immunomodulative activities of the tested lactobacilli strains (Tortuero and Fernandez 1995). Moreover, strains able to survive in the GIT and adhere to the intestine mucous membrane were reported to induce a far more effective immune response (Perdigón and Alvarez 1992). Stimulation of the nonspecific immune system by fermented products is probably due to immune‐active peptides produced during fermentation (Fiat et al. 1993). As reported by Perdigón et al. (1999), some LAB strains are able to induce specific secretory immunity, while others enhance the gut inflammatory immune response. Lactobacillus casei and Lact. plantarum were shown to interact with Peyer's patch cells which resulted in an increase in IgA, CD4+ cells, and specific antibodies directed against the stimulating strain. In turn, Lactococcus lactis and Lact. delbrueckii ssp. bulgaricus strains induced an increase in IgA+ cells entering the IgA cycle, but not CD4+ cells (Perdigón et al. 1999).
Lactic acid bacteria strains were also determined to have a positive role in eradicating intestinal infections. In the study by Herias et al. (1999), gnotobiotic rats fed with Lact. plantarum in combination with Escherichia coli showed lower amounts of E. coli in the small intestine and caecum 1 week after colonization, compared with a group colonized with E. coli alone. It appeared that colonized rats had a significantly higher total serum IgA levels and slightly higher IgM and IgA antibody levels against E. coli than those colonized with E. coli alone (Herias et al. 1999).
Health‐promoting effects of LAB‐based milk products, enhancement of nonspecific and specific immune responses, as well as control of intestinal infections have been confirmed in a large number of different publications. Nader de Macías et al. (1992) reported increased resistance to Shigella infection mediated by high titres of anti‐Shigella antibodies in serum and in intestine secretions in mice fed fermented milk (Nader de Macías et al. 1992). Also, Paubert‐Braquet et al. (1995) showed a correlation between an increased immune response and resistance to Salmonella Typhimurium infection in mice fed milk cultures (Paubert‐Braquet et al. 1995). In another study, the application of Lact. casei and Lact. bulgaricus was reported to terminate corticoid‐induced immunosuppression in mice with Candida albicans infection (de Petrino et al. 1995). Animals inoculated with lactobacilli showed a significant increase in faecal secretory IgA levels compared with control animals (Lee et al. 2016). According to Alvarez et al. (1998), cultures of Lact. casei protect the host against Salm. Typhimurium infection (not only) after the first application, but the effect is maintained after a simple revaccination (on day 15 or 30). A protective effect was observed when the number of IgA‐secreting cells in the lamina propria and the level of secretory IgA in the gut fluid increased (Alvarez et al. 1998). This correlated with an elevated number of polymorph nuclear cells that induced inflammatory immune response and was noted to influence the mucous membrane integrity. It was discussed that the population of CD4+ and CD8+ T‐lymphocytes as a whole can be increased, but the balance between them should be maintained. Higher level of cells in the CD8+ T‐cell population could induce an inflammatory response through the cytotoxicity effect. On the other hand, the elevated number of CD4+ T‐lymphocytes, peculiarly the Th1 population, could through the cytokine pathway, stimulate an enhanced expression of HLA class II. This event could be associated with a higher capture of antigens and overstimulation of the mucus.
Interestingly, administration of antigens in a certain mucosal region may generate S‐IgA antibodies at distant mucosal sites (Kozlowski et al. 1997). For example, oral immunization was shown to allow antigen uptake at inductive sites of the oral cavity and upper intestine and can elicit antibodies not only in saliva and intestine, but also in mammary glands and vaginal secretions. Therefore, it seems that oral vaccination can induce immune responses locally in the gut and at other mucosal sites, as well as systemic humoral and cellular immune responses (De Moreno de LeBlanc et al. 2005). Concluding, the effect of local mucosal immunization with LAB can have a broader range and elicit a global immune response.
Antigen dose and vaccine protection are also crucial issues for live vaccine development. Firstly, it seems that some proteins (e.g. the hepatitis B surface antigen) are easily recognized by the immune system (Yum et al. 2012). Other compounds are less effective, and the immune response may not provide efficient protection. The type of strain used as antigen vehicle (the producer) is also an important determinant. Different strains possess various adjuvant properties, as well as different adhesive abilities, which can have a significant impact on the stimulation of the immune system. Another concern is designing a proper vaccination scheme. In case of orally administered vaccines, prime‐boost strategy is required. One must take into account that overdosage of the antigen can lead to oral tolerance. Therefore, appropriate doses and vaccination schemes need to be separately optimized for each chosen vaccine system. In general, the dose of the produced antigen should be relatively high to be recognized as ‘nonself’ by the immune system of the host and elicit a protective response, but still remain low to avoid immunosuppression.
In regards to age and physiological state of the host, in immunocompromised patients, less often infants or elderly, certain LAB can cause opportunistic infections. Those rare cases are connected most often with probiotic preparations containing Lactobacillus sp. However, the risk of lactobacillemia was considered as ‘unequivocally negligible’ (Sanders et al. 2010). Moreover, there is no published evidence that consumption of orally administered LAB vaccines carries the risk of bacteraemia. This is probably due to significant difference in doses as well as application schemes between probiotics and live vector vaccines. In case of the latter, administration is not long‐lasting, which decreases the risk of bacteraemia.
Choice of appropriate LAB strain
The choice of appropriate strain for the live vector vaccine development seems to be crucial in order to provide efficient protection. Until now, in frame of the LABVAC European research network, three different species of LAB (i.e. L. lactis, Streptococcus gordonii and Lactobacillus sp.) have been quite well characterized, not only based on the physiological properties, but also according to the available molecular tools and developed experimental approaches.
Lactococcus lactis
Lactococcus lactis strains do not colonize the digestive tracts of animals and humans. In mice, these microbes reside for about 24 h and only passive transit is observed. In humans, lactococci persist in the gut for about 3 days (Klijn et al. 1995; Chamberlain et al. 1997; Havenith et al. 2002). Due to these facts, attention has been focused on expressing various antigens in L. lactis cells, both intra‐ and extracellularly. For this, efficient, well‐controlled expression systems were developed. The first high‐level inducible expression systems developed for L. lactis were based on the properties of the E. coli T7 bacteriophage RNA polymerase (pLET vectors). These systems were used for successful intracellular production of many heterologous antigens in L. lactis (e.g. diphtheria toxin fragment B, immunogen of Shistosoma manson, tetanus toxin fragment C (TTFC) Wells et al. 1996; Robinson et al. 1997; Mercenier et al. 2000). Further on, modified pLET vectors were developed to secrete antigens (up to 3 mg l−1) into the extracellular matrix or anchor them to the cell surface. Also, a variety of TTFC fusion proteins (e.g. TTFCHIV‐gp120V3 loop), were produced with quite good efficiency, ranging between 2 and 20% of the total soluble cell proteins. An analogous study was performed with the glutathione S‐transferase (P28) of the parasite Schistosoma mansoni (Capron et al. 1995). The P28 antigen has been efficiently expressed in L. lactis, separately and also in fusion with TTFC. The immunogenicity of these antigens has been proven by in vivo experiments (Klijn et al. 1995; Chamberlain et al. 1997).
Lactococci were also shown to be efficient cytokine delivery vehicles. Constitutive expression strains of L. lactis that accumulate a TTFC, within the cytoplasmic compartment and also secrete either murine IL‐2 or IL‐6 were tested (Steidler et al. 1995). Recombinant L. lactis strains were shown to produce active interleukins in the culture supernatants at a level of 0·9 mg l−1. Immunization trials with these interleukin‐producing strains confirmed strong adjuvant effect only for the modified live strains. The anti‐TTFC serum IgG titres were 10‐ to 15‐fold higher and increased more rapidly as compared to the response generated by immunization with strains producing TTFC alone. In contrast, the level of anti‐lactococcal immune responses was not enhanced by co‐expression of the cytokines (Walker 1994; Miettinen et al. 1996). Cell localization of the expressed heterologous protein, and the route of administration are also crucial, and often correlate with immune response levels. A study showed that recombinant L. lactis strains producing three different types of TTFC protein (intracellular, membrane‐anchored, extracellular) were able after subcutaneous administration into mice, without any adjuvant, to evoke protection against lethal toxin challenge (Michon et al. 2016). All three types of localization allowed development of specific immune responses. However, the highest IgG serum antibody titres were obtained with the strain producing TTFC in the cytoplasm. In experiments conducted by Robinson et al. (1997), mice were immunized by oral and intranasal routes. Nasal inoculation of mice with the strain expressing TTFC intracellularly using the lactococcal T7 system led to significant IgG serum antibody response, and 75% protection from lethal challenge with tetanus toxin (20 × LD50). The antibody titres were similar whether live or inactivated (mitomycin C‐ or formalin‐treated) recombinant L. lactis cells were used. It was shown that effective immune response can be obtained both in mice immunized orally with L. lactis strains producing TTFC at high levels (T7 expression system) or at a 10‐fold lower level (pTREX1) (Robinson et al. 1997; Mercenier et al. 2000). TTFC‐specific serum antibody responses of both IgG1 and IgG2a isotypes were induced and significantly elevated levels of anti‐TTFC IgA antibodies were detected in faeces and gut secretions. Even though the antibody titres were lower than those following nasal immunization, the protective efficacy was similar. Often, high antibody titres detected in ELISA do not find confirmation in neutralizing assays that are based only on specific neutralizing antibodies (Nabs) and reflect the actual protective efficacy of the vaccine.
Apart from pLET, different vectors with constitutively active promoters of low to medium strength have been applied for heterologous gene expression in L. lactis. For example, the pTREX1 vector has been used to express TTFC and P28 proteins with efficiency of 1–3% of total cell proteins (Capron et al. 1995). These kind of vectors are suitable for the expression of membrane‐associated antigens which exhibit a certain degree of insolubility or toxicity in bacterial cells. Other examples of expression systems in L. lactis include the regulated ptcB promoter inserted in the pIL253 vector which was successfully used that in our previous work (Szatraj et al. 2014) for expression of different kinds of viral HA genes. The same vector was applied by Kasarello et al. (2015) during experiments connected with oral administration of L. lactis expressing synthetic genes of myelin antigens. Many researchers rely on the use of the commercially available nisin‐inducible expression system (NICE®), which includes a variety of different vectors, adapted for intra‐ or extracellular protein expression. It is probably the most often used expression system in Gram‐positive bacteria nowadays (Kleerebezem et al. 1997).
Certain L. lactis strains exhibit quite good persistence in the GIT of animals, which is an important aspect in oral vaccine development as it can prolong the exposure of particular antigens to specific receptors of GALT. Boguslawska et al. (2009) reported on the use of a L. lactis strain which exhibited the ability to persist efficiently in the GIT of rats (Boguslawska et al. 2009). Further examination of the identified L. lactis IBB 477 strain by Radziwill‐Bienkowska et al. (2014, 2016) led to determining that its persistence is inextricably connected with adhesive properties. In L. lactis IBB 477, three surface proteins connected with adhesion have been described to date: the chromosomally encoded sex factor aggregation protein CluA, and two plasmid‐encoded proteins, serine proteinase PrtP (Radziwill‐Bienkowska et al. 2014, 2016) and protein YghE2 displaying pilin characteristics (Meyrand et al. 2013). It seems that the persistence of live bacterial vaccines depends also on the host. For example, some strains have been suggested to be adapted specifically to the GIT of humans, but not to those of other animals (Ozawa et al. 2012). Strains that possess increased adhesive abilities and thus exhibit high persistence (e.g. IBB477) are very attractive in view of mucosally applied vaccines. Nonetheless, it seems that bacterial persistence in the gut is a feature that should be determined for each strain and host separately.
Streptococcus gordonii
Streptococcus gordonii is a member of the endogenic human microflora. It colonizes the oral and vaginal cavities, but persists only transiently in the digestive tract. The naturally competent genetic expression system of heterologous proteins on the surface of Strep. gordonii strain CH1 is called ‘Challis’ (Medaglini et al. 1995, 1997; Pouwels et al. 1998). Its basis relies on the chromosomal integration of recombinant DNA encoding the vaccine antigen fused with the M6 surface protein of Streptococcus pyogenes. The system allows for release of the antigen to the medium or its presentation in the anchored form. Recombinant proteins ranging in size from 15 to 441 amino acids have been effectively produced on the surface of Strep. gordonii cells, including the E7 protein of human papilloma virus type 16, the V3 domain of HIV‐1 gp120, allergen from the hornet venom, ovalbumin, surface proteins (F and H) of the measles virus, the B subunit of the heat labile toxin (LTB) of E. coli (Salminen et al. 1996). An interesting feature of the system is that a recombinant strain can express at the cell surface two different antigens. Conducted experiments revealed that use of such strain in single dose (in a range of 107–109 CFU) led to stable colonization in the oropharyngeal mucosa of animals. Moreover, antigen expression was stable in vitro and antigen‐specific local (IgA) and systemic (IgG) antibodies were produced. The effectiveness of Strep. gordonii colonization was shown to be related with the observed immune response (Moineau and Goulet 1991; Tizard 2000). Intragastric immunization with strains producing LTB was the only one described so far (Moineau and Goulet 1991; Havenith et al. 2002). In contrast to oropharyngeal mucosa, it was shown that Strep. gordonii cells did not colonize the intestinal mucosa, but they did induce serum IgG and faecal IgA. Subsequent results indicated that phagocytosis of Strep. gordonii activates DC. This is a great advantage as those cells represent efficient antigen‐presenting cells, and are responsible for generating primary T‐cell responses. Despite promising results, indicating the potential role of Strep. gordonii‐based system for live vaccine vector development, there are still some serious safety issues to be solved before licensing it for human/animal use. Firstly, the vector was formerly classified as Streptococcus sanguis and the M6 protein originating from Strep. pyogenes for a long time has been considered as a virulence determinant. Moreover, the system can evoke chromosomal integration of a gene fusion together with an associated drug resistance marker what constitutes an additional disadvantage (Mercenier et al. 2000). In spite of that, this is still the most common system used for designing live vaccines based on Strep. gordonii (Ricci et al. 2000; Sharma et al. 2001; Kotloff et al. 2005; Wang et al. 2013). Other selection markers, such as the cadmium resistance gene, were also investigated in Strep. thermophilus. Yet, the usefulness of this system can be doubted since it necessitates the use of a harmful and toxic compound (cadmium) (Peterbauer et al. 2011). It is without doubts that a variety of safety issues need to be resolved before Strep. gordonii‐based vaccines can be licensed for human or animal use. For sure, other less controversial solutions, lacking unwarranted markers or sequences, need to be developed.
Lactobacillus
Lactobacilli are known as safe bacteria possessing a number of properties that render them highly suitable for delivering many different compounds to the mucosa. The immunomodulating capacity of lactobacilli together with the possibility of targeted antigen production at specific sites of the bacterial cell seems to be an attractive feature. Two types of Lactobacillus strains: ‘commensal’ and ‘dietary’, may be taken into account as a potential vaccine vehicles. Commensal strains are expected to combine health‐promoting properties with the ability to adhere in the area of the oral cavity, stomach, intestine, vagina and urethra. Dietary strains are mainly used in the milk industry as starters for production of fermented milk, meat or vegetable products. It should be taken for consideration that different strains vary in their immunomodulatory characteristic, what can have a great impact on their intrinsic vaccine potential. Especially, their capacity to adhere to the relevant epithelial surfaces is an important property. Adhesion can be mediated through direct adherence to the antigen sampling cells (M‐cells, epithelial cells, mucus), or through aggregation with resident bacteria. The second strategy leads to intense competition with the endogenous bacterial community, what can result in prolonged antigen exposure. Both in vitro and in vivo models have been used to select or screen for adherent Lactobacillus strains (Pouwels et al. 1998). Gathered data indicate that colonization is host‐ and tissue‐ or even site‐specific. It is unlikely that one particular strain will constitute an ideal vector to deliver antigens to different hosts or to different mucosal cavities within the same host (Medaglini et al. 1997). A study performed on nonrecombinant, chromosomally marked (Rifr, Smr) lactic acid bacterial strains: Lactobacillus fermentum KLD, Lact. plantarum NCIMB 8826 and Lact. salivarius UCC 433118, given orally to human volunteers as a fermented milk product, showed that the two latter strains were far more superior in their ability to survive the passage through the stomach. These strains were able to reach quite high viable counts in the ileum, what is advantageous in regards to the localization of Peyer's patches (Mercenier et al. 2000). It seems that the best strategy of selecting the most efficient colonizer strain is by direct isolation of the candidate strain from the targeted host. Some strains of human or murine origin were shown to be effective in colonizing at least two body cavities of the mouse animal model. Lactobacillus paracasei LbTGS1.4 (vaginal murine isolate) and Lact. plantarum NCIMB 8826 (human saliva isolate) were determined to persist in the gut or the vagina for over a week (Salminen et al. 1996). Choice of the most favourable Lactobacillus strain is also linked with strong strain‐specificity of the selected expression system (Havenaar et al. 1992; Mercenier et al. 1996). Optimal translation, transcription and targeting sequences can differ significantly between species and might even be strain‐dependent (Wells et al. 1995). In lactobacilli, various antigens were produced and targeted to different localizations: (i) the cytoplasm (up to the level of few percent of the total protein content), (ii) the culture medium (up to 13 mg l−1) or (iii) the cell surface (Gonzaga et al. 2009). Recent improvements in gene expression included development of plasmid expression vectors with increased stability and chromosomal integration systems targeting specific or random loci. Those systems are mostly based on different nonreplicative or, alternatively, on recombinant conjugative transposons (Wells et al. 1995). The latter, offer the possibility to rapidly test the expression of the specific antigen in a variety of recipient strains. However, this approach may lead to integrants which carry an antibiotic resistance marker, with inactivated chromosomal gene(s) essential for persistence or immunomodulation. A system, based on a nonreplicative plasmid, with nondisruptive integration achieved in the tRNA‐Ser locus has been described by Dupont et al. (1995). Another integration vector, especially suited for Lact. plantarum, that can lead to insertion either at the tRNA‐Ser or at the l‐LDH (l‐lactate dehydrogenase) locus was constructed (Mercenier et al. 2000). Inactivation of the l‐LDH was shown not to impair the growth of Lact. plantarum in vitro or in vivo. The recombinant plasmid carries the antigen‐encoding DNA in a translational fusion with the l‐LDH gene. Integration of the plasmid occurs via a double crossing‐over, where the second homologous recombination leads to resections containing only the heterologous gene and no antibiotic marker. Systems exploiting both loci (tRNA‐Ser and l‐LDH) were used successfully to produce antigens in lactobacilli. For example, recombinant transposons allowed Rush et al. (1997) to express the E. coli LTB at the cell surface of different lactobacilli. Integration into the l‐LDH gene allowed for higher production levels in comparison to insertion at the tRNA‐Ser locus. The chromosomal integrants produced comparable antigen amounts to the recombinant strains carrying multicopy plasmids. It should be noted that improvement of expression levels often relied on modification of the translation initiation region or a translational fusion with well‐expressed endogenous genes. Few regulated promoters are available for lactobacilli. The nisin‐inducible expression system originally designed for L. lactis was also implemented in Lact. plantarum NCIMB 8826 (Kleerebezem et al. 1997). This required integration of the sensor and regulatory genes, nisK and nisR, into the chromosome of the host and optimization of the induction conditions. The nisin system was determined to be very efficient and allowed for high expression levels of gp50, TTFC and GFP (Green Fluorescent Protein from Aequorea victoria). Since the antigen production level can be controlled by conditions of induction, it is possible to investigate the effect of antigen quantity on the level and duration of the specific immune response. The GFP+ strains that do not require addition of exogenous substrate or co‐factor to emit fluorescence represent an ideal tool to perform such studies in vivo. Their usefulness has been confirmed in vitro (phagocytosis by macrophages) and in vivo (intragastric and intranasal administration to mice) (Geoffroy et al. 2000). Use of GFP markers is also applied to track the survival of strains in the environment. Although the function of lactobacilli as adjuvants or carriers was established early on, the immunogenicity of recombinant strains by the intraperitoneal route was demonstrated relatively recently. Latest experiments carried within the LABVAC network confirmed that after nasal administration all lactobacilli producing TTFC at the level of a few percent of the total cellular protein content, including the Lact. plantarum NCIMB 8826 integrant, induced production of serum IgG and local IgA (Mercenier et al. 2000). A controlled comparison of TTFC‐producing LAB strains by the oral route has been undertaken as well. The ongoing works include selection of strains appropriate for human use, identification of adhesion factors and use of S‐layers for antigen presentation (Mercenier et al. 1996).
Doubts
The Food and Drug Administration acknowledges LAB as food‐grade bacteria with GRAS status, meaning they are safe for use in humans and animals. Nonetheless, live vaccine strains based on these micro‐organisms should not be considered as avirulent. That is why an increasing number of projects aim to assess the biosafety of LAB. After polyphasic taxonomic identification, biosafety of these microorganisms for human or animal application should be generally assessed by: detection of antibiotic resistance and horizontal transfer of antibiotic resistance genes, detection of known and new virulence properties, evaluation of immunological adverse effects, and the survival, colonization, and genetic stability in the human gut. Lactococcus lactis and lactobacilli are generally classified to Biosafety Level 1 (BSL‐1), while Strep. gordonii is classified to BL‐2 class. LAB strains used as live vector vaccines should be handled according to official guidelines and procedures available, for example, on the CDC (Centers for Disease Control and Prevention) Website.
The use of genetically modified organisms always raises concerns about their uncontrolled persistence and spread in the environment. Thus, transfer of antibiotic selection markers or other genetic sequences between microbes cannot be ignored, especially when many efficient methods that improve adaptation skills, such as conjugation, transformation, retromobilization or transduction have been identified. Toomey et al. (2010) examined the involvement of Enterococcus faecalis and L. lactis, in spreading of resistance determinants between other LAB and such pathogenic strains as E. coli, Listeria spp., Staphylococcus aureus and Salmonella spp. While no resistance transfer was noted for the first two strains, transfer of erythromycin resistance to Listeria spp. from the donor strains was determined. Additionally, a high frequency of erythromycin and tetracycline‐resistance transfer was observed between LAB species. Also, many strains of lactobacilli are naturally resistant to vancomycin, which may evoke questions about the potential transfer. However, vancomycin resistance genes of Lactobacillus species are chromosomally located and cannot be easily transmitted to other genera (Tynkkynen et al. 1998). Additionally, vancomycin is excluded from treatment of lactobacillemia to minimize the occurrence of nonsusceptible strains to this antibiotic and further spread of resistance onto other bacteria.
Much attention is paid to the development of different selection markers that meet the requirements specified by food‐relevant definitions. The two main groups of selection markers replacing the routinely used selection for antibiotic resistance can be classified into dominant (active) and complementation (passive) selection approaches (Peterbauer et al. 2011; He et al. 2012). Active containment is based on the conditional genetic control of either activation of a compound or repression of an essential gene. Passive containment is based mainly on complementation of an auxotrophy or other gene defect by supplementation with either the intact gene or the essential metabolite. Dominant markers do not rely on host‐specific genes and therefore they are widely applicable and can often be adapted in several different LAB species. For example, the nsr gene, encoding a hydrophobic protein that provides resistance to nisin, was one of the first genes used for alternative dominant selection. The nisin immunity gene nisI and the lafI gene, encoding immunity to class II bacteriocin – lactacin F, were also successfully used as selection markers in heterologous hosts. More recently, a dominant selection marker based on the bile salt hydrolase gene, bsh, from Lact. plantarum was reported (Jarocki et al. 2014). Another example of dominant selection exploit genes involved in the utilization of rare and unusual sugars like: d‐xylose catabolism encoding genes (xylRAB), α‐galactosidase gene, sucrose genes scrA and scrB.
The second type of food‐grade markers is based on complementation selection that involves the selectable host/vector system. The system is based on complementation of specific mutations in chromosomal genes that are essential in a particular metabolic pathway. The most known mutations are: nonsense mutation in the purine biosynthetic pathway combined with a nonsense suppressor (ochre suppressor gene supB), deletion of an internal fragment of the chromosomal alr gene, mutation in the thyA gene, mutations in the chromosomal lactose operon of various LAB. Complementation systems are often bacteriostatic rather than bactericidal. Among them, two systems with potential human application can be distinguished. The first one is based on alanine racemase mutants that require d‐Ala, and can be generated in a large number of LAB strains. The second one relies on a thymidine synthase (thyA) mutant of L. lactis. In a potential situation when the genetically modified strain would be released into the environment, the transgene will be eliminated from the genome and the strain will revert to the wild‐type. In L. lactis for example, Steidler et al. (2000), Steidler (2003) replaced thyA with a synthetic human IL‐10 transgene, what resulted in creation of the Thy12 strain. After validation studies in pigs, this strain has been approved in the Netherlands for experimental therapy in humans with inflammatory bowel disease (IBD). It would be of great interest to determine the specificities of double mutants that could offer additional benefits over a single thyA mutation, such as increased antigen expression in the presence of two transgene copies, redundancy with respect to biological containment, and the applicability of the mutant using different kind of transgenes (Steidler et al. 2000; Steidler 2003).
A special subgroup of food‐grade host/vector systems are inducible gene expression systems like: the NICE (NIsin‐Controlled Expression) system, systems based on quorum sensing, pSIP vectors developed based on promoters and regulatory genes of the class II bacteriocin – sakacin A (sap gene cluster), or sakacin P (spp gene cluster). The possibilities of selecting an adequate expression vector and selection marker are diverse; however, the final steps towards food‐grade systems, such as the removal or replacement of undesired parts of the expression vectors, have not yet been reported (Peterbauer et al. 2011; He et al. 2012).
Another issue that should be addressed in designing administration schemes of live vaccines (especially via the oral route) is the phenomenon of mucosal tolerance (Mowat and Weiner 1999). Oral tolerance is the state of local and systemic immune unresponsiveness that is induced by oral administration of an antigen. Systemic hyporesponsiveness has also been induced by administration of antigens via the nasal route. At the level of molecular mechanisms, the proper balance between CD4+ and CD8+ should be maintained in order to elicit an effective immune response (Dubois et al. 2003). Rise solely in CD8+ can lead to inflammation, while increase of CD4+ will result in overstimulation and appearance of the mucosal tolerance.
It appears that induction of mucosal tolerance and mucosal immunity occurs at the same site in the gut and other mucosal lymphoid sites. In some cases, mucosal tolerance can serve as a very beneficial function (Ogra et al. 2001). There are some diseases for which oral immunization has been shown to suppress autoimmune responses, e.g. rheumatoid arthritis, multiple sclerosis, experimental autoimmune encephalitis, myelitis, uveoretinitis and diabetes mellitus.
Information on clinical trials connected with LAB vaccines is limited. A biological containment strategy is a prerequisite for marketing live bacterial vaccine vectors and its lack is the major reason for restrictions in development of this field. However, the system established in L. lactis used for therapeutic delivery of human IL‐10 has already undergone phase II clinical trials for the treatment of IBD (Steidler 2003). This recombinant L. lactis strain was termed ‘Actobiotics™’ and is being developed for clinical use by ‘ActoGenix’ company. In spite of some serious safety issues, also Strep. gordonii has been used in a phase I clinical trial to demonstrate that the organism can be safely administered to humans by the nasal/oral route (Kotloff et al. 2005). Still, development of new stable auxotrophic LAB strains that are not able to replicate in the human body and can safely be used even in immune compromised individuals is highly desirable.
Research networking
The selection of colonizing vs noncolonizing vaccine species or strains is a crucial step in designing vaccines. On one side, in vivo synthesis of the antigen by bacteria persisting at the desired mucosal surface should efficiently stimulate the immune system due to prolonged exposure to the antigen. On the other hand, noncolonizing LAB can act as live microparticles preloaded with the antigen, which release it during transit in the gut. It is not possible to univocally predict which system is better according to optimal antigen presentation, a parameter which is known to affect its immunogenicity. Strains able to efficiently colonize humans need to be selected on the basis of safety, physiological and metabolic criteria. Vaccine strains should be genetically stable to allow expression of protective epitopes in different cellular locations. From the immunological point of view, analysis of the immune response, the nature and the intensity according to the mode of antigen presentation, the immunization route and the nature of the bacterial vector should be well characterized as it is essential for the final immune‐protective effect.
Two research networks (contracts BIO2‐CT94‐3055 and BIO4‐ CT96‐0542) focused on a common model system have been organized to examine the potential of three LAB as vaccine vehicles: L. lactis (a prototype of a noncolonizing strain), lactobacilli (colonizing bacteria) and Strep. gordonii (an oral commensal bacterium with a stable antigen presentation system) (Mercenier et al. 2000). The mentioned project concentrated on examining production of antigens of bacterial and viral origin by these three bacteria. The first antigen used was the TTFC, a 47 kDa nontoxic polypeptide with the ganglioside‐binding domain. It is a useful model antigen due to its well‐established immunogenicity and the availability of a lethal mouse challenge model (Fairweather et al. 1987). It allows to effectively evaluate the capability of a mucosal delivery system to elicit systemic humoral immune response. The second examined antigen was the gp50 protein of the porcine pseudorabies virus (Aujeszky's disease virus, ADV). This pathogen infects animals through the respiratory tract. The best way for its inactivation is induction of mucosal immune responses. This approach was designed to evaluate the potential of the three selected LAB to target viral mucosa‐associated diseases. Additionally, challenging mice or pigs with ADV will enable evaluating the bioactivity of the induced antibody responses (Mercenier et al. 2000). Thanks to the information gathered in frame of the project, knowledge about characteristics and properties of the three examined LAB species for mucosal vaccination has been updated, unified and organized.
Conclusions
Lactic acid bacteria‐based delivery and expression systems can be considered as promising tools for efficient production of antigens and many other, biologically active compounds. Crucial aspects that should be taken into account when designing an effective mucosal vaccine are manifold. Despite numerous studies using LAB as potential vaccine vehicles, it is necessary to broaden the knowledge about the immune effects of local mucosal application of this group of bacteria and the antigen levels they may produce. Development of effective mucosally administrated vaccines is also inextricably linked to the understanding of the immune response mechanisms and the cellular and molecular pathways involved in its control. More comprehensive knowledge about the type of induced cells and immune responses, the role of cytokines or phagocytic functions would be helpful. Currently, hopes are directed at new systems for production of immunogenic antigens that are targeted to specific areas, cells or even receptors. The development of recombinant LAB vaccines is in its early stages. Fortunately, specific immunological experiments, new recombinant strains and vectors continue to be constructed and described in detail what can lead in the near future to standardization of LAB vectors in vaccine production.
Conflict of Interest
The authors declare no conflict of interest.
Acknowledgements
This work was funded by grant no. PBS2/A7/14/2014 ‘Anti flu vaccines – innovative production of subunit antigens’.
References
- Alvarez, S. , Gobbato, N. , Bru, E. , de Ruiz Holgado, A.P. and Perdigón, G. (1998) Specific immunity induction at the mucosal level by viable Lactobacillus casei: a perspective for oral vaccine development. Food Agric Immunol 10, 79–87. [Google Scholar]
- Balasubramanya, N.N. , Lokesh, B.R. , Ramesh, H.P. and Krishnakantha, T.P. (1995) Effect of lactic microbes on superoxide anion generating ability of peritoneal macrophages and tissue histopathology of murines. Indian J Dairy Biosci 6, 28–33. [Google Scholar]
- Bermúdez‐Humarán, L.G. , Langella, P. , Miyoshi, A. , Gruss, A. , Tamez Guerra, R. , Montes de Oca‐Luna, R. and Le Loir, Y. (2002) Production of human papillomavirus type 16 E7 protein in Lactococcus lactis . Appl Environ Microbiol 68, 917–922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhakdi, S. and Tranumjensen, J. (1991) Alpha‐toxin of Staphylococcus aureus . Microbiol Rev 55, 733–751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boguslawska, J. , Zycka‐Krzesinska, J. , Wilcks, A. and Bardowski, J. (2009) Intra‐ and interspecies conjugal transfer of Tn916‐like elements from Lactococcus lactis in vitro and in vivo. Appl Environ Microbiol 75, 6352–6360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campos, I.B. , Darriuex, M. , Ferreira, D.M. , Miyaji, E.N. , Silva, D.A. , Areas, A.P. , Aires, K.A. , Leite, L.C. et al (2008) Nasal immunization if mice with Lactobacillus casei expressing the pneumococcal surface protein A: induction of antibodies, complement deposition and partial protection against Streptococcus pneumoniae challenge on the mouse respiratory tract. Microbes Infect 10, 481–488. [DOI] [PubMed] [Google Scholar]
- Capron, A. , Riveau, G. , Grzych, J.M. , Boulanger, D. , Capron, M. and Pierce, R. (1995) Development of a vaccine strategy against human and bovine schistosomiasis. Background and update. Mem Inst Oswaldo Cruz 90, 235–240. [DOI] [PubMed] [Google Scholar]
- Chamberlain, L.M. , Wells, J.M. , Robinson, K. , Schofield, K.M. and Le Page., R.W.F . (1997) Mucosal immunisation with recombinant Lactococcus lactis In Gram‐Positive Bacteria as Vaccine Vehicles for Mucosal Immunization ed. Pozzi G. and Wells J.M. pp. 83–106. Austin, TX: Landes Bioscience. [Google Scholar]
- De Moreno de LeBlanc, A. , Maldonado Galdeano, C. , Chaves, S. and Perdigon, G. (2005) Oral administration of Lactobacillus casei CRL 431 increases immunity in bronchus and mammary glands. Eur J Inflamm 3, 23–28. [Google Scholar]
- Dubois, B. , Chapat, L. , Goubier, A. , Papiernik, M. , Nicolas, J.F. and Kaiserlian, D. (2003) Innate CD4CD25 regulatory T cells are required for oral tolerance and inhibition of CD8 T cells mediating skin inflammation. Blood 102, 3295–3301. [DOI] [PubMed] [Google Scholar]
- Dupont, L. , Boizet‐Bonhoure, B. , Coddeville, M. , Auvray, F. and Ritzenthaler, P. (1995) Characterisation of genetic elements required for site‐specific integration of Lactobacillus delbrueckii subsp. bulgaricus bacteriophage mv4 and construction of an integration proficient vector for Lactobacillus plantarum . J Bacteriol 177, 586–595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dziarski, R. (1991) Demonstration of peptidoglycan‐binding sites on lymphocytes and macrophages by photoaffinity cross‐linking. J Biol Chem 266, 4713–4718. [PubMed] [Google Scholar]
- Ernst, P.B. , Scicchitano, R. , Underdown, B.J. and Bienenstock, J. (1988) Oral immunization and tolerance In Immunology of the gastrointestinal tract and liver eds. Heyworth M.F. and Jones A.L., pp 125–144. New York, N.Y: Raven. [Google Scholar]
- Fairweather, N.F. , Lyness, V.A. and Maskell, D.J. (1987) Immunisation of mice against tetanus toxin with fragments of tetanus toxin synthesised in Escherichia coli . Infect Immun 55, 2541–2545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falcone, V. , Mihm, D. , Neumann‐Haefelin, D. , Costa, C. , Nguyen, T. , Pozzi, G. and Ricci, S. (2006) Systemic and mucosal immunity to respiratory sincitial virus induced by recombinant Streptococcus gordonii surface displaying a domain of viral glycoprotein G. FEMS Immunol Med Microbiol 48, 116–122. [DOI] [PubMed] [Google Scholar]
- Fernandes, C.F. and Shahani, K.M. (1990) Anticarcinogenic and immunological properties of dietary lactobacilli. J Food Prot 53, 704–710. [DOI] [PubMed] [Google Scholar]
- Fiat, A.M. , Migliore‐Samour, D. , Jollès, P. , Drouet, L. , Sollier, C.B. and Caen, J. (1993) Biologically active peptides from milk proteins with emphasis on two examples concerning antithrombotic and immunomodulating activities. J Dairy Sci 76, 301–310. [DOI] [PubMed] [Google Scholar]
- Fuller, R. (1997) An introduction In Probiotics 2: Applications and Practical Aspects ed. Fuller R., pp 1–9. London, England: Chapman & Hall. [Google Scholar]
- Galdeano C.M., de Moreno de LeBlanc A., Dogi C. and Perdigon G. (2010) Lactic acid bacteria as immunomodulators of the gut ‐ associated immune system In Biotechnology of lactic acid bacteria: novel applications eds. Mozzi F, Raya R. R. and Vignolo G.M. pp. 124–140. Ames: Wiley‐Blackwell. [Google Scholar]
- Geoffroy, M.C. , Guyard, C. , Quatannens, B. , Pavan, S. , Lange, M. and Mercenier, A. (2000) Use of GreenFluorescent protein to tag lactic acid bacterium strains under development as live vaccine vectors. Appl Environ Microbiol 66, 383–391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson, G.R. and Wang, X. (1994) Regulatory effects of bifidobacteria on the growth of other colonic bacteria. J Appl Bacteriol 77, 412–420. [DOI] [PubMed] [Google Scholar]
- Gonzaga, V.E. , Lescano, A.G. , Huaman, A.A. , Salmon‐Mulanovich, G. and Blazes, D.I. (2009) Histamine levels in fish from markets in Lima, Peru. J Food Protect 72, 1112–1115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanniffy, S.B. , Carter, A.T. , Hitchin, E. and Wells, J.M. (2007) Mucosal delivery of a pneumococcal vaccine using Lactococcus lactis affords protection against respiratory infection. J Infect Dis 195, 185–193. [DOI] [PubMed] [Google Scholar]
- Havenaar, R. , Brink, B.T. and Huis in't Veld, J.H.J . (1992) Selection of strains for probiotic use In Probiotics, the Scientific Basis ed. Fuller R. pp. 209–224. London: Chapman and Hall. [Google Scholar]
- Havenith, C.E.G. , Seegers, J.F.M.L. and Pouwels, P.H. (2002) Gut‐associated lactobacilli for oral immunisation. Food Res Int 35, 151–163. [Google Scholar]
- He, S. , Gong, F. , Guo, Y. and Zhang, D. (2012) Food‐grade selection markers in lactic acid bacteria. TAF Prev Med Bull 11(4), 499–510. [Google Scholar]
- Herias, M.V. , Hessle, C. , Telemo, E. , Midtvedt, T. , Hanson, L.A. and Wold, A.E. (1999) Immunomodulatory effects of Lactobacillus plantarum colonizing the intestine of gnotobiotic rats. Clin Exp Immunol 116, 283–290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hershberg, R.M. and Mayer, L.F. (2000) Antigen processing and presentation by intestinal epithelial cells – polarity and complexity. Immunol Today 21, 123–128. [DOI] [PubMed] [Google Scholar]
- Heumann, K.G. , Rottman, L. and Vogl, J. (1994) Elemental speciation with liquid chromatography‐inductively coupled plasma isotope dilution mass spectrometry. J Anal At Spectrom 9, 1351–1355. [Google Scholar]
- Holzapfel, W.H. , Haberer, P. , Snel, J. , Schillinger, U. and Huis in't Veld, J.H.J . (1998) Overview of gut flora and probiotics. Int J Food Microbiol 41, 85–101. [DOI] [PubMed] [Google Scholar]
- Jain, N.K. and Roy, I. (2009) Effect of trehalose on protein structure. Protein Sci 18, 24–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jarocki, P. , Podleśny, M. , Glibowski, P. and Targoński, Z.A. (2014) New insight into the physiological role of bile salt hydrolase among intestinal bacteria from the genus Bifidobacterium . PLoS ONE 9(12), e114379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasarello, K. , Kwiatkowska‐Patzer, B. , Lipkowski, A.W. , Bardowski, J.K. and Szczepankowska, A.K. (2015) Oral administration of Lactococcus lactis expressing synthetic genes of myelin antigens in decreasing experimental autoimmune encephalomyelitis in rats. Med Sci Monit 21, 1587–1597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleerebezem, M. , Beerthuyzen, M.M. , Vaughan, E.E. , de Vos, W.M. and Kuipers, O.P. (1997) Controlled gene expression systems for lactic acid bacteria: transferable nisin‐inducible expression cassettes for Lactococcus, Leuconostoc, and Lactobacillus spp. Appl Environ Microbiol 63, 4581–4584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klijn, N. , Weerkamp, A.H. and de Vos, W.M. (1995) Genetic marking of Lactococcus lactis shows its survival in the human gastrointestinal tract. Appl Environ Microbiol 61, 2771–2774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kotloff, K.L. , Wasserman, S.S. , Jones, K.F. , Livio, S. , Hruby, D.E. , Franke, C.A. and Fischetti, V.A. (2005) Clinical and microbiological responses of volunteers to combined intranasal and oral inoculation with a Streptococcus gordonii carrier strain intended for future use as a group A Streptococcus vaccine. Infect Immun 3, 2360–2366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozlowski, P.A. , Cu‐Uvin, S. , Neutra, M.R. and Flanigan, T.P. (1997) Comparison of the oral, rectal, and vaginal immunization routes for induction of antibodies in rectal and genital tract secretions of women. Infect Immun 65, 1387–1394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee, J.S. , Poo, H. , Han, D.P. , Hong, S.P. , Kim, K. , Cho, M.W. , Kim, E. , Sung, M.H. et al (2006) Mucosal immunization with surface‐display severe acute respiratory syndrome Coronavirus spike protein on Lactobaclillus casei induces neutralizing antibodies in mice. J Virol 80, 4079–4087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee, H.A. , Kim, H. , Lee, K.W. and Park, K.Y. (2016) Dead Lactobacillus plantarum stimulates and skews immune responses toward T helper 1 and 17 polarizations in RAW 264.7 cells and mouse splenocytes. J Microbiol Biotechnol 26, 469–476. [DOI] [PubMed] [Google Scholar]
- Lidbeck, A. and Nord, C.E. (1993) Lactobacilli and the normal human anaerobic microflora. Clin Infect Dis 16, S181–S187. [DOI] [PubMed] [Google Scholar]
- Martin, R.M. and Lew, A.M. (1998) Is IgG2a a good Th1 marker in mice. Immunol Today 19, 49. [DOI] [PubMed] [Google Scholar]
- Medaglini, D. , Pozzi, G. , King, T.P. and Fischetti, V.A. (1995) Mucosal and systemic immune responses to a recombinant protein expressed on the surface of the oral commensal bacterium Streptococcus gordonii after oral colonisation. Proc Natl Acad Sci USA 92, 6868–6872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medaglini, D. , Ricci, S. , Maggi, T. , Rush, C.M. , Manganelli, R. , Oggioni, M.R. and Pozzi, G. (1997) Recombinant gram‐positive bacteria as vehicles of vaccine antigens. Biotechnol Ann Rev 3, 297–312. [Google Scholar]
- Mercenier, A. , Dutot, P. , Kleinpeter, P. , Aguirre, M. , Paris, P. , Reymund, J. and Slos, P. (1996) Development of lactic acid bacteria as live vectors for oral or local vaccines. Adv Food Sci 18, 73–77. [Google Scholar]
- Mercenier, A. , Müller‐Alouf, H. and Grangette, C. (2000) Lactic acid bacteria as live vaccines. Curr Issues Mol Biol 2, 17–25. [PubMed] [Google Scholar]
- Mestecky, J. (1987) The common mucosal immune system and current strategies for induction of immune responses in external secretions. J Clin Immunol 7, 265. [DOI] [PubMed] [Google Scholar]
- Meyrand, M. , Guillot, A. , Goin, M. , Furlan, S. , Armalyte, J. , Kulakauskas, S. , Cortes‐Perez, N.G. , Thomas, G. et al (2013) Surface proteome analysis of a natural isolate of Lactococcus lctis reveals the presence of pili able to bind human intestinal epithelial cells. Mol Cell Proteomics 12, 3935–3947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michon, C. , Langella, P. , Eijsink, V.G.H. , Mathiesen, G. and Chatel, J.M. (2016) Display of recombinant proteins at the surface of lactic acid bacteria: strategies and applications. Microb Cell Fact 15, 70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miettinen, M. , Vuopio‐Varkila, J. and Varkila, K. (1996) Production of human tumor necrosis factor alpha, interleukin‐6, and interleukin‐10 is induced by lactic acid bacteria. Infect Immun 64, 5403–5405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moineau, S. and Goulet, J. (1991) Effect of feeding fermented milks on the pulmonary macrophage activity in mice. Milchwissenschaft 46, 551–554. [Google Scholar]
- Mowat, A.M. and Weiner, H.L. (1999) Oral tolerance: physiological basis and clinical applications In Mucosal Immunology ed. Ogra P.L., Mestecky J., Lamm M.E., Strober W., Bienenstock J. and McGhee J.R., 2nd edn pp. 587–618. New York, NY: Academic Press. [Google Scholar]
- Murray, H.W. (1988) Interferon‐gamma, the activated macrophage, and host defense against microbial challenge. Ann Intern Med 108, 595–608. [DOI] [PubMed] [Google Scholar]
- Nader de Macías, M.E. , Apella, M.C. , Romero, N.C. , González, S.N. and Oliver, G. (1992) Inhibition of Shigella sonnei by Lactobacillus casei and Lactobacillus acidophilus . J Appl Bacteriol 73, 407–411. [DOI] [PubMed] [Google Scholar]
- Ogra, P.L. , Faden, H. and Welliver, R.C. (2001) Vaccination strategies for mucosal immune responses. Clin Microbiol Rev 14, 430–445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliveira, M.L. , Areas, A.P. , Campos, I.B. , Monedero, V. , Perez‐Martinez, G. , Miyaji, E.N. , Leite, L.C. , Aire, K.S. et al (2006) Induction of systemic and mucosal immune response and decrease in Streptococcus pneumoniae colonization by nasal inoculation of mice with recombinant lactic acid bacteria expressing pneumococcal surface antigen A. Microbes Infect 8, 1016–1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozawa, K. , Fujiwara, R. , Watanbe, K. and Sonoyama, K. (2012) Persistence of orally administered Lactobacillus strains in the gut of infant mice. Biosci Microbiota Food Health 31, 85–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paubert‐Braquet, M. , Gan, X.H. , Gaudichon, C. , Hedef, N. , Serikoff, A. , Bouley, C. , Bonavida, B. and Braquet, P. (1995) Enhancement of host resistance against Salmonella typhimurium in mice fed a diet supplemented with yoghurt or milks fermented with various Lactobacillus casei strains. Int J Immunother 11, 153–161. [Google Scholar]
- Perdigón, G. and Alvarez, S. (1992) Probiotics and the immune state In Probiotics, the Scientific Basis ed. Fuller R., pp. 145–180. London: Chapman and Hall. [Google Scholar]
- Perdigón, G. , Vintiñi, E. , Alvarez, S. , Medina, M. and Medici, M. (1999) Study of the possible mechanisms involved in the mucosal immune system activation by lactic acid bacteria. J Dairy Sci 82, 1108–1114. [DOI] [PubMed] [Google Scholar]
- Perdigón, G. , Maldonado Galdeano, C. , Valdez, J.C. and Medici, M. (2002) Interaction of lactic acid bacteria with the gut immune system. Eur J Clin Nutr 56(Suppl. 4), S21–S26. [DOI] [PubMed] [Google Scholar]
- Peterbauer, C. , Meichsberger, T. and Haltrich, D. (2011) Food‐grade gene expression in lactic acid bacteria. Biotechnol J 6, 1147–1161. [DOI] [PubMed] [Google Scholar]
- de Petrino, S.F. , de Jorrrat, M.E.B.B. , Meson, O. and Perdigón, G. (1995) Protective ability of certain lactic acid bacteria against an infection with Candida albicans in a mouse immunosuppression model by corticoid. Food Agric Immunol 7, 365–373. [Google Scholar]
- Plant, K.P. and Lapatra, S.E. (2011) Advances in fish vaccine delivery. Dev Comp Immunol 35, 1256–1262. [DOI] [PubMed] [Google Scholar]
- Pouwels, P.H. , Leer, R.J. , Shaw, M. , Heijne den Bak‐Glashouwer, M.J. , Tielen, F.D. , Smit, E. , Martinez, B. , Jore, J. et al (1998) Lactic acid bacteria as antigen delivery vehicles for oral immunization purposes. Int J Food Microbiol 41, 155–167. [DOI] [PubMed] [Google Scholar]
- Radziwill‐Bienkowska, J.M. , Zochowska, D. , Bardowski, J.K. , Mercier‐Bonin, M. and Kowalczyk, M. (2014) Lactococcus lactis IBB477 presenting adhesive and muco‐adhesive properties as a candidate carrier strain for oral vaccination against influenza virus. Acta Biochim Pol 61, 603–607. [PubMed] [Google Scholar]
- Radziwill‐Bienkowska, J.M. , Le, D.T.L. , Szczesny, P. , Duviau, M.P. , Aleksandrzak‐Piekarczyk, T. , Loubière, P. , Mercier‐Bonin, M. , Bardowski, J.K. et al (2016) Adhesion of the genome‐sequenced Lactococcus lactis subsp. cremoris IBB477 strain is mediated by specific molecular determinants. Appl Microbiol Biotechnol 100, 9605–9617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ricci, S. , Medalgini, D. , Rush, C.M. , Marcello, A. , Peppeloni, S. , Manganelli, R. , Palu, G. and Pozzi, G . (2000) Immunogenicity of the B Monomer of Escherichia coli HeatLabile toxin expressed on the surface of Streptococcus gordonii . Infect Immun 68, 760–766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rigaux, P. , Daniel, C. , Hisbergues, M. , Muraille, E. , Hols, P. , Pot, B. , Pestel, J. and Jacquet, A. (2009) Immunomodulatory properties of Lactobacillus plantarum and its use as a recombinant vaccine against mite allergy. Allergy 64, 406–414. [DOI] [PubMed] [Google Scholar]
- Robinson, K. , Chamberlain, L.M. , Schofield, K.M. , Wells, J.M. and Le Page, R.W.F . (1997) Oral vaccination of mice against tetanus with recombinant Lactococcus lactis . Nature Biotechnol 15, 653–657. [DOI] [PubMed] [Google Scholar]
- Rush, C.M. , Mercenier, A. and Pozzi, G . (1997) Expression of vaccine antigens in Lactobacillus In Gram‐Positive Bacteria. Vaccine Vehicles for Mucosal Immunisation. ed. Pozzi G. and Wells J.M. pp. 107–144. Landes, Austin, TX: Biotechnology Intelligence Unit. [Google Scholar]
- Salminen, S. , Isolauri, E. and Salminen, E. (1996) Clinical uses of probiotics for stabilising the gut mucosal barrier: successful strains and future challenges. Anton van Leeuwen 70, 347–358. [DOI] [PubMed] [Google Scholar]
- Sanders, M.E. , Akkermans, L.M. , Haller, D. , Hammerman, C. , Heimbach, J. , Hormanseprger, G. , Huys, G. , Levy, D.D. et al (2010) Safety assessment of probiotics for human use. Gut Microbes 1, 164–185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saroja, C.H. , Lakshmi, P.K. and Bhaskaran, S. (2011) Recent trends in vaccine delivery systems: a review. Int J Pharm Investig 1, 64–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma, A. , Honma, K. , Evans, R.T. , Hruby, D.E. and Genco, R.J. (2001) Oral Immunization with Recombinant Streptococcus gordonii expressing porphyromonas gingivalis FimA domains. Infect Immun 69, 2928–2934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steidler, L. (2003) Gene exchange of ThyA for interleukin‐10 eecures live GMO bacterial therapeutics. Discov Med 3, 49–51. [PubMed] [Google Scholar]
- Steidler, L. , Wells, J.M. , Raeymaekers, J. , Vandekerckhove, J. , Fiers, W. and Remaut, E. (1995) Secretion of biologically active murine interleukin‐2 by Lactococcus lactis subsp. lactis. Appl Environ Microbiol 61, 1627–1629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steidler, L. , Hans, W. , Schotte, L. , Neirynck, S. , Obermeier, F. , Falk, W. , Fiers, W. and Remaut, E. (2000) Treatment of murine colitis by Lactococcus lactis secreting interleukin‐10. Science 289, 1352–1355. [DOI] [PubMed] [Google Scholar]
- Szatraj, K. , Szczepankowska, A.K. , Sączyńska, V. , Florys, K. , Gromadzka, B. , Łepek, K. , Płucienniczak, G. , Szewczyk, B. et al (2014) Expression of avian influenza haemagglutinin (H5) and chicken interleukin 2 (chIL‐2) under control of the ptcB promoter in Lactococcus lactis . Acta Biochim Pol 61, 609–614. [PubMed] [Google Scholar]
- Tizard, I.R. (2000) An introduction In: Veterinary immunology ed. Tizard I.R. 6th edn pp. 210–221. Philadelphia, PA: Saunders W.B. Company. [Google Scholar]
- Toomey, N. , Bolton, D. and Fanning, S. (2010) Characterisation and transferability of antibiotic resistance genes from lactic acid bacteria isolated from Irish pork and beef abattoirs. Res Microbiol 161, 127–135. [DOI] [PubMed] [Google Scholar]
- Tortuero, F. and Fernandez, E. (1995) Effect of inclusion of microbial culture in barley‐based diets fed to laying hens. Anim Feed Sci Tec 53, 255–265. [Google Scholar]
- Tynkkynen, S. , Singh, K.V. and Varmanen, P. (1998) Vancomycin resistance factor of Lactobacillus rhamnosus GG in relation to enterococcal vancomycin resistance (van) genes. Int J Food Microbiol 41, 195–204. [DOI] [PubMed] [Google Scholar]
- Vidhyalakshmi, R. , Parantham, R. and Indhumathi, J. (2009) Amylase production on submerged fermentationby Bacillus spp. World J Chem 4, 89–91. [Google Scholar]
- Walker, R.I. (1994) New strategies for using mucosal vaccination to achieve more effective immunisation. Vaccine 12, 387–400. [DOI] [PubMed] [Google Scholar]
- Wang, L. , Liu, W. , Yang, M. , Peng, D. and Chen, L. (2013) Development of a Streptococcus gordonii vaccine strain expressing Schisostoma japonicum Sj‐F1 and evaluation of using this strain for intranasal immunization in mice. Parasitol Res 112, 1701–1708. [DOI] [PubMed] [Google Scholar]
- Wells, J.M. , Norton, P.M. and le Page, R.W.F. (1995) Progress in the development of mucosal vaccines based on Lactococcus lactis . Int Dairy J 5, 1071–1079. [Google Scholar]
- Wells, J.M. , Robinson, K. , Chamberlain, L.M. , Schofield, K.M. and Le Page, R.W.F . (1996) Lactic acid bacteria as vaccine delivery vehicles. Anton van Leeuwen 70, 317–330. [DOI] [PubMed] [Google Scholar]
- World Health Organization . Global Vaccine Action Plan: 2011–2020. Geneva, Switzerland: WHO Press; 2013. http://www.who.int/ immunization/global_vaccine_action_plan/en/ [Google Scholar]
- You, J. , Dong, H. , Mann, E.R. , Knight, S.C. and Yaqoob, P. (2014) Probiotic modulation of dendritic cell function is influenced by ageing. Immunobiology 219, 138–148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yum, J.S. , Ahn, B.C. , Jin Jo, H.J. , Kim, D.Y. , Kim, K.H. , Kim, H.S. , Chul Sung, Y.C. , Yoon, J. et al (2012) Use of pre‐S protein‐containing hepatitis B virus surface antigens and a powerful adjuvant to develop an immune therapy for chronic hepatitis B virus infection. Clin Vaccine Immunol 19, 120–127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou, Z. , Gong, S. , Li, X.M. , Yangm, Y. , Guan, R. , Zhou, S. , Yao, S. , Xie, Y. et al (2015) Expression of Helicobacter pylori urease B on the surface of Bacillus subtilis spores. J Med Microbiol 64, 104–110. [DOI] [PubMed] [Google Scholar]


