Abstract
Hypoglycaemia is a common, life‐threatening complication that occurs as a component of a wide variety of disease processes. Despite its frequent occurrence, information concerning the aetiology, characteristics and outcomes of hypoglycaemic crises in veterinary medicine is limited. This review summarises the current understanding of the pathophysiology of hypoglycaemia, the body's counter‐regulatory response, underlying aetiologies, diagnosis and treatment. Disease mechanisms are discussed and published evidence in veterinary literature regarding prognostic indicators, prevalence, diagnosis and treatment is examined for hypoglycaemia‐related disease processes including insulinoma, glucose‐lowering toxins and medications.
INTRODUCTION
During homoeostasis, euglycaemia is maintained by a balance between glucose production, storage and release of glucose from its stored forms. Glucose is the most abundant carbohydrate in the body and is the principal fuel for the brain and peripheral tissues. Insulin is secreted from β‐cells in the pancreas and is responsible for transporting glucose into the cells to be utilised immediately or stored as glycogen (Klein 2012). Maintaining euglycaemia depends on the body's ability to secrete the appropriate hormone in response to changing concentrations of blood glucose (BG).
Glucose homoeostasis
In a hypoglycaemic state, glucagon and adrenaline levels rise within minutes to facilitate a transient increase in glucose production (Cryer 2004). Hypoglycaemia causes the hypothalamus to stimulate the sympathetic nervous system to release adrenaline from the adrenal glands. Adrenaline acts quickly to limit further secretion of insulin and increase secretion of glucagon. Glucagon, which is secreted from the α cells of the pancreas, stimulates glycogenolysis and gluconeogenesis, resulting in increased glucose production from hepatic cells (Koenig 2015). Glucagon inhibits glucose uptake by peripheral tissues and stimulates the release of gluconeogenic precursors (Guyton & Hall 2006). However, the effects of glucagon are temporary and are quickly suppressed by increasing levels of insulin. Cortisol and growth hormone are released more slowly, typically a few hours following the onset of hypoglycaemia. Cortisol increases the rate of lipolysis and release of amino acids from muscle that also facilitate gluconeogenesis. Growth hormone works synergistically by decreasing utilisation of glucose by peripheral tissues as well as promoting lipolysis (Guyton & Hall 2006). A study examining these mechanisms in adrenalectomised dogs concluded that even when counter‐regulatory hormones are absent, the body can utilise glucose auto‐regulation and increased neural efferent signalling in order to stimulate hepatic production of glucose (Gregory et al. 2017). There is also evidence that central excitatory amino acids may also contribute to the maintenance of euglycaemia via activation of N‐methyl‐D‐aspartate receptors and subsequent stimulation of the sympathoadrenal and hypothalamic–pituitary adrenal axis (Molina & Abumrad 2001). In addition to increases in concentrations in plasma concentrations of counter‐regulatory hormones, there is evidence of increases in concentrations of immunoreactive β‐endorphins, adrenocorticotropin and cortisol within the cerebrospinal fluid (CSF) in response to hypoglycaemia. However, the significance of these alterations in CSF concentrations remains unknown (Radosevich et al. 1988).
Pathophysiology of hypoglycaemia
Disruption of glucose homoeostasis and development of hypoglycaemia can result from a variety of conditions, including neonatal hypoglycaemia, hepatic insufficiency, sepsis, paraneoplastic syndromes, glycogen storage diseases, deficiencies of counter‐regulatory hormones, exercise‐induced hypoglycaemia, hypoadrenocorticism, pregnancy and iatrogenic insulin overdose (Koenig 2015). Conditions that can that can result in artefactual hypoglycaemia include erythrocytosis (previously often referred to as polycythaemia) and leucocytosis. Table 1 lists conditions that can result in hypoglycaemia.
Table 1.
Causes of hypoglycaemia
| Decreased glucose production |
| Neonatal hypoglycaemia |
| Hypoglycaemia of toy‐breeds |
| Hypoadrenocorticism (primary and secondary) |
| Hepatic dysfunction |
| Portosystemic shunt |
| Cirrhosis |
| Hepatitis |
| Hepatic lipidosis |
| Glycogen storage disease |
| Counterregulatory hormone deficiencies |
| Glucagon, growth hormone deficiency |
| Panhypopituitarism |
| Thyroid hormone deficiency |
| Catecholamine deficiency |
| Cortisol deficiency |
| Glycogenic or gluconeogenic enzyme deficiencies |
| Toxins |
| Xylitol, ethanol |
| β‐blockers |
| Alpha lipoic acid |
| Oral hypoglycaemic medications |
| Oral sulfonylureas |
| Metformin |
| Acarbose |
| Artifactual causes |
| Severe polycythaemia or leukocytosis |
| Collection or storage errors |
| Laboratory error |
| Uncalibrated handheld glucometer |
| Excess glucose utilisation |
| Hunting dog hypoglycaemia |
| Pregnancy hypoglycaemia |
| Starvation/severe malnutrition |
| Infectious causes |
| Babesiosis |
| Sepsis |
| Parvovirus infection |
| Excess insulin or insulin‐like factors |
| Insulinoma |
| Extrapancreatic neoplasia |
| Hepatocellular carcinoma |
| Leiomyosarcoma, leiomyoma |
| Functional β‐cell disorders (nesidioblastosis) |
| Exogenous overdose of insulin or insulin secratagogues |
Clinical signs of hypoglycaemia
Clinical signs of hypoglycaemia can manifest as cerebral dysfunction and include altered mental status, behavioural changes, weakness, ataxia, collapse, blindness or altered vision, seizures, stupor and coma. Clinical signs frequently reported in hypoglycaemic dogs and cats include anxiety, pacing, vocalisation, trembling, vomiting, tachypnoea and diarrhoea (Greene & Bright 2008, Koenig & Verlander 2015). Hypoglycaemia is typically defined as a glucose level of less than 3∙3 mmol/L (Cryer 2012, Koenig & Verlander 2015). The severity of clinical signs typically depends on the duration of hypoglycaemic episode and the rate of decrease of glucose levels. In human patients with chronic hypoglycaemia, clinical signs may be minimal or absent until glucose levels are profoundly decreased, as a result of hypoglycaemic unawareness. The pathophysiology of hypoglycaemic unawareness is not fully understood but is thought to occur in patients whose brains have been conditioned to automatically upregulate cerebral glucose uptake as a result of prolonged, chronic or recurrent hypoglycaemia. This automatic mechanism of cerebral glucose uptake can actually lead to a decreased perception of peripheral hypoglycaemia and blunting of the body's counter‐regulatory responses (Weinstock et al. 2016). While there is little evidence that this occurs in companion animals, it is important to keep in mind when evaluating patients with conditions that may place them at risk for chronic hypoglycaemia, including chronic insulin overdose of diabetes mellitus (DM) patients and insulinomas.
Neuroglycopenia
The brain is particularly vulnerable to the effects of hypoglycaemia because it cannot manufacture its own glucose and is dependent on the constant availability of adequate amounts of glucose for energy (Boyle 1997). While it may briefly utilise ketones, the brain has a limited ability to utilise other forms of energy (Boyle 1997). In order to keep up with the brain's needs, glucose is continuously transported across the blood brain barrier from the peripheral bloodstream via facilitated transport. Glucosensors within the ventromedial nuclei of the hypothalamus, intestinal tract, carotid body and hepatoportal area act to control the amount of glucose that is transported through the blood brain barrier (Loose et al. 2008). If arterial glucose levels fall, the diffusion gradient cannot be adequately maintained and neuroglycopenia may transpire. Prolonged neuroglycopenia can lead to permanent brain injury and neurologic signs that may persist beyond resolution of the hypoglycaemia (Kraje 2003, Loose et al. 2008). While transient hypoglycaemic episodes do not typically cause damage or persistent neurologic deficits, severe or repeated hypoglycaemic states can result in neurologic deficits that may resemble other acute neurological disease processes. This syndrome is referred to as hypoglycaemic encephalopathy (Witsch et al. 2012). The prognosis for human patients is variable and ranges from reversible neurologic deficits (Aoki et al. 2004, Bottcher et al. 2005, Kim et al. 2007, Maruya et al. 2007) to persistent neurologic deficits, persistent vegetative state and death (Mori et al. 2006, Ma et al. 2009, Kang et al. 2010, Yaffe et al. 2013, Mehta et al. 2016). In veterinary literature, a case report of a dog with hypoglycaemia secondary to insulinoma and refractory epilpesy has been described in which brain lesions within the superficial layers of the cerebral cortex and the dentate gyrus of the hippocampus were identified post mortem (Shimada et al. 2000).
Hypoglycaemia and the cardiovascular system
Numerous human case reports document an association between supraventricular, ventricular and bradyarrhythmias in association with hypoglycaemia (Bolognesi et al. 2011, Celebi et al. 2011, Sanon et al. 2014), with atrial fibrillation identified as the most common supraventricular arrhythmia. Bradycardia and circulatory collapse have also been documented in veterinary patients (Little 2005). The occurrence of these arrhythmias has led to concern that hypoglycaemic states may be pro‐arrhythmogenic (Sanon et al. 2014). While hypoglycaemia commonly will lead to the release of catecholamines and secondary tachyarrhythmias, the cause of bradyarrhythmias is unknown.
Diagnosis of hypoglycaemia
In order to demonstrate that clinical signs are secondary to hypoglycaemia Whipple's triad must be observed: (1) clinical signs of hypoglycaemia; (2) documentation of a low blood glucose level at the time of clinical signs and (3) resolution of clinical signs with correction of hypoglycaemia (Cryer 2013). Although the gold standard for the measurement of blood glucose is analysis by isotope dilution mass spectrometry, this is not available in a clinical setting (Tonyushkina & Nichols 2009). Therefore, most glucose levels are measured with handheld point‐of‐care glucometers (POCG). POCGs that are typically designed for humans can demonstrate inconsistencies regarding accuracy in veterinary species (Cohen et al. 2009). Other POCGs marketed to veterinarians utilise internal algorithms that attempt to quantify species differences between free and haemoglobin‐bound glucose (Zini et al. 2009). The accuracy of handheld glucometers can be vulnerable to delays in separation of the serum from the red blood cells longer that 30 minutes after collection, leading to pseudohypoglycaemia as a result of continued consumption of glucose by the red blood cells in vitro (Koenig & Verlander 2015). This effect can be prevented by collecting blood samples in sodium fluoride tubes if they cannot be processed via centrifugation within that time period. Haemodilution and haemoconcentration can also affect the accuracy of glucose readings (Solnica et al. 2012). The mechanism behind this is believed to be a result of a reduction of the volume of plasma due to the presence of increased erythrocytes in a haemoconcentrated sample. This causes the volume of plasma that is able to penetrate the reagent layer to be reduced, resulting in a falsely reduced glucose measurement (Tang et al. 2000). Conversely, a haemodiluted sample allows an abnormally large amount of plasma to contact the test strip reagent layer, which falsely elevates the glucose measurement (Tang et al. 2000). Lane et al. (2015) proposed a formula to correct the glucose level given a known packed cell volume, but additional algorithms would need to be developed and validated for other glucometer models.
Continuous glucose monitoring
Less invasive alternatives to traditional monitoring of blood glucose for veterinary patients include the continuous glucose monitoring system (CGMS; Guardian REAL‐Time continuous glucose monitoring), in which measurements of interstitial glucose concentrations are monitored via a sensor implanted within the subcutaneous (sc) space (Reineke et al. 2010). The sensor is comprised of a glucose diffusion limiting membrane which contains glucose oxidase. When the membrane is exposed to glucose in the presence of oxygen, it undergoes an oxidation reaction to produce gluconic acid and hydrogen peroxide. The hydrogen peroxide creates a current that reflects the glucose level within the interstitium via a reaction with the platinum electrode (Reineke et al. 2010). Over the last 10 years, studies in experimental non‐diabetic dogs, stable diabetic dogs and cats and those with diabetic ketoacidosis have, for the most part, demonstrated accuracy and agreement between glucose values obtained via CGMS compared with those obtained via traditional glucometers (Davison et al. 2003, Ristic et al. 2005, Wiedmeyer et al. 2005, Reineke et al. 2010).
Causes of Hypoglycaemia
The causes of hypoglycaemia are multi‐factorial and can be divided into groups: (1) excess insulin or insulin analogues; (2) inadequate glucose production; and (3) increased glucose consumption. Fig. 1 shows a diagnostic algorithm that can be helpful in guiding the evaluation of a hypoglycaemic patient.
Figure 1.
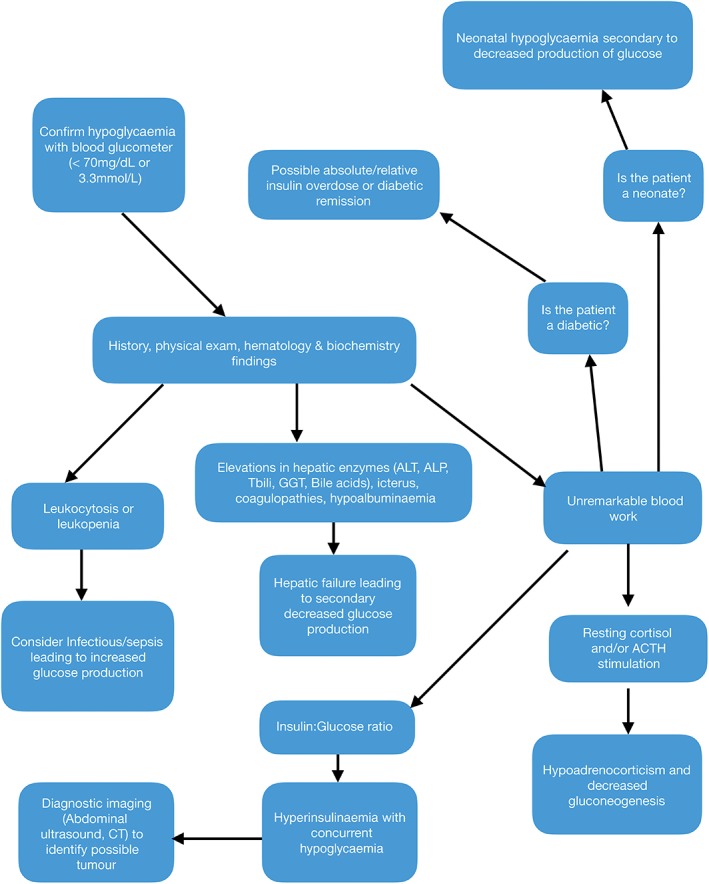
Diagnostic algorithm for hypoglycaemia
Excess insulin or insulin analogues
Exogenous insulin overdose
Both veterinary and human patients with DM are at risk for exogenous insulin overdoses, either secondary to insulin mishandling and administration errors or because of concurrent illness and transient remission. Information regarding insulin overdoses in diabetic animals is relatively scarce, but a previous retrospective article identified that cats were more likely to experience overdose compared to dogs. Furthermore, obese animals and cats that regularly received more than 6 units of insulin per injection were at increased risk (Whitley et al. 1997). Diabetic animals that are experiencing gastrointestinal upset secondary to concurrent underlying illness were also reported to be at increased risk for experiencing hypoglycaemia secondary to concurrent administration of exogenous insulin (Whitley et al. 1997). In 2017, the authors of a second retrospective article reported that in a review of 28 cats with DM that had experienced hypoglycaemia, vomiting or reduced intake was reported in association with hypoglycaemia (Viebrock and Dennis 2018). This study revealed that patients experiencing clinical improvement within 12 hours of presentation were more likely to survive, and that mental status and severity of hypoglycaemia on presentation were not negative prognostic indicators (Viebrock and Dennis 2018).
Insulinomas
Insulinomas are malignant insulin‐secreting tumours of the pancreas which are most common in middle‐aged to older dogs. These patients commonly present with weakness, collapse or seizures secondary to severe hypoglycaemia (Polton et al. 2007).
Diagnostic modalities include an amended or standard insulin glucose ratio, fructosamine levels, glucagon tolerance testing and oral glucose tolerance tests (Greene & Bright 2008, Melano & Peterson 2013). Abdominal ultrasound has low sensitivity to detect these tumours and, in some cases, CT or abdominal exploratory is recommended for definitive identification (Robben et al. 2005). Medical treatment options include stabilisation of refractory and life‐threatening hypoglycaemia via administration of glucagon continuous infusions if indicated, frequent feeding and oral administration of glucocorticoids as well as other adjunctive medications such as diazoxide and somatostatin therapy to help alleviate clinical signs and maintain normoglycaemia. Both diazoxide and somatostatin result in membrane hyperpolarisation and inhibition of normal insulin release (Dunne & Peterson 1991). Diazoxide may increase hepatic glucose production and reduce peripheral glucose uptake (Altszuler et al. 1977). Somatostatin has inhibitory effects on several other hormones including glucagon, secretin and vasoactive inhibitory peptide. Somatostatin analogues like octreotide are used more commonly due to their longer half‐life (Lamberts et al. 1996). Octreotide has been evaluated in healthy dogs and dogs with insulinomas (Robben et al. 2006): following administration, plasma insulin and glucagon concentrations decreased in healthy dogs but only insulin decreased in dogs with insulinomas. None of the dogs experienced significant adverse effects from the medication. Evaluation of somatostatin receptors in canine insulinomas revealed that dogs only express one type of somatostatin receptor, in contrast to humans who can express several types (Robben et al. 1997). This can make it difficult to extrapolate information from human studies of somatostatin use in insulinomas to canine patients. There have been rare reports of insulinomas diagnosed in cats (McMillan et al. 1985, Hawks et al. 1992, Elie & Zerbe 1995, Kraje 2003, Greene & Bright 2008), but there are no reports of using somatostatins or diazoxide in cats. Other medications include streptozocin, which selectively destroys pancreatic β cells.
Surgical removal and a partial pancreatectomy are required for more definitive control (Moore et al. 2002). Median survival times for dogs following surgical resection alone has been reported as 785 days versus 196 days for medical management alone (Polton et al. 2007).
Ferrets can commonly also present with hypoglycaemia secondary to insulinomas. Most ferrets develop clinical signs secondary to this condition around 4 to 5 years of age (Caplan et al. 1996, Ehrhart et al. 1996). Normal fasting blood glucose in a ferret is 4∙9 to 6∙8 mmol/L, and in one case series evaluating 57 ferrets with a confirmed insulinoma, the mean blood glucose was less than 3∙3 mmol/L (Caplan et al. 1996). Ferrets can be treated in a similar way to dogs and glucocorticoids may aid in maintaining normoglycaemia. The recommended oral starting dose of 0∙25 mg/kg prednisone is every 24 hours, which can then be titrated as needed (Melano & Peterson 2013). Diazoxide can also be prescribed, but this medication can be cost‐prohibitive. Surgery can also be pursued with published mean survival times of 668 days with a partial pancreatectomy versus 186 days with medical management alone (Weiss et al. 1998). One case report details the successful dose of 15 ng/kg/min glucagon constant rate infusion at in a 3‐year‐old female domestic ferret with an insulinoma (Bennett et al. 2015). In addition to numerous case reports in ferrets, there is one report of a pre mortem diagnosis and treatment of an insulinoma in a 5‐year‐old male guinea pig that was treated medically with a starting dose of 5 mg/kg diazoxide orally every 12 hours, eventually increasing to 25 mg/kg orally every 12 hours. The clinical signs were managed medically for 3 weeks before the patient presented with signs of constipation and abdominal distension and died the following day (Hess et al. 2013).
Paraneoplastic hypoglycaemia
Any neoplasia can potentially cause a secondary hypoglycaemia. The most common tumours associated with this syndrome include hepatocellular carcinoma, leiomyomas and leiomyosarcomas (Beaudry et al. 1995, Battaglia et al. 2005, Bergman 2007). Although the exact cause is unknown, it is suspected that the cancerous cells have accelerated glucose consumption or can secrete insulin or insulin‐like peptides (Finotello et al. 2009, 2014, Singhal et al. 2017). In the case of hepatic neoplasia in particular, the tumour may affect the liver's endogenous ability to perform glycogenolysis or gluceoneogenesis. Treatment of the hypoglycaemia is symptomatic with definitive treatment of the specific cancer required for adequate control.
Hyperinsulinaemic hypoglycaemic syndrome
Hyperinsulinaemic hypoglycaemic syndrome (HHS) is the most common cause of hypoglycaemia in human infants but has rarely been recognised in veterinary medicine. This syndrome is defined as excessive production of insulin due to hyperplasia within the β cells of the pancreas (Senniappan et al. 2013). There is a case report proposing that HHS may have been the cause of refractory hypoglycaemia in two dogs, both of which were under 1 year of age: there was persistent hyperinsulinaemia in the face of hypoglycaemia and elevated insulin glucose ratios without evidence of any insulin‐secreting tumours (Breitschwerdt et al. 2014). A recent report also documents for the first time HHS in a 6‐year‐old female spayed British shorthair cat secondary to suspect nesidioblastosis, a term which describes non‐neoplastic β‐cell hyperplasia within the pancreas (Hambrook et al. 2016).
Toxins and medications
Xylitol
One of the most common toxins associated with hypoglycaemia in dogs is xylitol, an artificial sweetener that is commonly found in sugar‐free gum, sweets, baked goods, nutritional supplements and dental care products, many of which are designed as sugar‐free alternatives for humans with DM (Dunayer 2004). Once ingested, peak plasma levels of xylitol in dogs are reached within 30 minutes. The majority of xylitol is metabolised in the liver and transformed to d‐xylulose, which then becomes d‐xylulose‐5‐phosphate (Froesch & Jakob 1974, Piscitelli et al. 2010). Xylitol is then converted to glucose, glycogen or lactate via the pentose phosphate pathway (Piscitelli et al. 2010). Unlike humans, dogs experience a marked dose‐dependent insulin surge following xylitol ingestion, leading to hypoglycaemia (Xia et al. 2009). Hepatic failure in dogs secondary to xylitol has also been described (Dunayer & Gwaltney‐Brant 2006, Todd & Powell 2007). A recent retrospective article found the most common clinical signs associated with xylitol ingestion in dogs were vomiting and lethargy, and 15% of dogs became hypoglycaemic during hospitalisation (DuHadway et al. 2015). None of the dogs developed hepatic failure and all survived to discharge. Historically, 0∙1 g/kg is the minimal dose believed to result in hypoglycaemia, while dogs ingesting 0∙5 g/kg or greater are believed to be at increased risk of developing acute liver failure (Dunayer & Gwaltney‐Brant 2006). There is also evidence that the development of hypoglycaemia and, especially, acute liver failure may be idiosyncratic rather than strictly dose‐dependent, because previous cases series have documented dogs recovering after ingesting up to 16 g/kg of xylitol, while other dogs have died after ingesting far lower doses (Dunayer & Gwaltney‐Brant 2006). There are no reports documenting xylitol toxicity in cats to our knowledge.
Alcohol
Ingestion of various types of alcohol compounds can also result in hypoglycaemia when it is. metabolised in the liver via alcohol dehydrogenase and aldehyde dehydrogenase to acetate. The oxidation of ethanol raises levels of nicotinamide adenine dinucleotide hydride (NADH), which then inhibits enzymes that regulate gluconeogenesis, leading to a decreased ability to release glucose from the liver (Kandi et al. 2014). The most common types of alcohols encountered include methanol, ethanol and isopropanol (also known as rubbing alcohol). Previous case reports of poisoning from exposure to ethanol in veterinary medicine have included alcoholic beverages (Ratclife & Zuber 1977, Houston & Head 1993, Keno & Langston 2011), various types of uncooked dough (Thrall et al. 1984, Suter 1992, Means 2003) and uncooked apples (Kammerer et al. 2001). Though ethanol administration has been documented to induce hypoglycaemia in dogs in the laboratory (Lochner et al. 1967), none of the cited case reports documented hypoglycaemia in the affected patients. However, hypoglycaemia is also intermittently described in human and veterinary patients that have ingested ethylene glycol, and is thought to occur secondary to depletion of glycogen stores and reduction of gluconeogenesis (Brent et al. 1999, Garcia‐Ortuno et al. 2006, Kraut & Kurtz 2008).
Alpha lipoic acid
Alpha lipoic acid (ALA) is an acid that is found as a natural compound and acts as a biological cofactor (Loftin & Herold 2009). It has been investigated as a possible therapy in the treatment of a number of conditions in humans, including cancer (Wenzel et al. 2005), heavy metal intoxication (Grunert 1960), human immunodeficiency virus (Baur et al. 1991) and multiple sclerosis (Packer et al. 1995, Khamaisi et al. 1999, Yadav et al. 2005, Ziegler et al. 2006). It has been utilised in veterinary medicine by alternative medicine practitioners as a potential therapy for patients with DM and cognitive dysfunction in dogs (Means 2008). The reported therapeutic dose in cats is 1 to 5 mg/kg with a maximum dose of 25 mg/day, and up to 80 to 200 mg/day in dogs. However, due to limited information on safety, its use in veterinary medicine remains controversial. However, ALA has been reported to be a concerning potential toxicity in companion animals. There are three existing case reports of toxicity in dogs (Means 2008, Loftin & Herold 2009), and ALA has been noted to be 10 times more toxic in cats as compared to dogs, humans and rats (Hill et al. 2004). Cats with ALA toxicity have been noted to exhibit anorexia, ataxia, hypersalivation, vomiting and elevated hepatic enzymes with single doses of 60 mg/kg (Loftin & Herold 2009). Dogs can exhibit similar signs as well as hypoglycaemia, hepatic and renal failure (Loftin & Herold 2009). Proposed mechanisms for hypoglycaemia include increased uptake of glucose, increased number of glucose transport proteins and inhibition of gluconeogenesis, among others (Jacob et al. 1999, Loftin & Herold 2009).
Oleander
There is one case report of oleander toxicity resulting in the development of hypoglycaemia in a 7‐year‐old female spayed Maltese that presented after known ingestion of oleander leaves. Other causes of hypoglycaemia were excluded and the patient recovered with supportive care (Page & Murtaugh 2015). Nerium oleander is a flowering shrub in the family Apocyanaceae, and all parts of the plant contain cardenolides, which are cardiac glycosides (Langford & Boor 1996). Proposed mechanisms of action regarding oleander's ability to cause hypoglycaemia include disruption of the sodium gradient necessary for the transport of glucose via disruption of the Na/K ATPase pump, or inhibition of α‐glucosidases within the small intestine responsible for hydrolysing saccharides to glucose (Mwafy & Yassin 2011, Page & Murtaugh 2015).
Inadequate glucose production
Neonatal hypoglycaemia
Neonatal and paediatric animals are susceptible to hypoglycaemia due to a combination of poor glycogen stores with which to initiate glycolysis and gluconeogenesis, and an underdeveloped liver (McMichael 2005). Other factors that may predispose neonatal patients to the development of hypoglycaemia include decreased birth weight, premature birth and being born to a bitch with systemic illness or diabetes. There is some evidence to support neonatal hypoglycaemia as a predictor of higher mortality between 1 and 21 days of age (Mila et al. 2017). Once weaned, paediatric patients can experience hypoglycaemia secondary to concurrent infections, immune system stimulation, exercise, decreased quality of nutrition, hypothermia, ongoing gastrointestinal losses and fasting (McMichael 2015). Parvovirus is one of the most common causes of hypoglycaemia in young animals. A study by Castro et al. (2013) evaluating puppies with either coronavirus or parvovirus showed that both groups commonly experienced hypoglycaemia, although they did not evaluate whether hypoglycaemia was a prognostic factor. In cats with parvovirus (feline panleukopenia virus), hypoglycaemia was much less common and not associated with prognosis (Kruse et al. 2010). A 2017 article comparing inpatient versus outpatient treatment protocols found that 50% of the outpatient group required dextrose supplementation, while 80% of the inpatient group required dextrose supplementation (Venn et al. 2017).
Other proposed causes of inadequate glucose production include what is often referred to as toy‐breed hypoglycaemia, which is anecdotally reported either as a form of transient juvenile hypoglycaemia or to occur even in adults within this breed grouping. However, a thorough search of available literature reveals little evidence to describe and classify such a syndrome as distinct cause of hypoglycaemia.
Refeeding syndrome
A recent case report by DeAvilla & Leech (2016) described hypoglycaemia as a complication of refeeding syndrome in a 2‐year‐old male neutered domestic shorthair that was treated for starvation after being missing for 12 weeks. Despite beginning with 24% of resting energy requirement on day 1 of hospitalisation, it developed hypoglycaemia 12 hours after refeeding was initiated and required supplementation via a dextrose constant‐rate infusion. The cat went on to make a full recovery and theories regarding the cause of hypoglycaemia included reduced glycogen stores, depleted body fat secondary to starvation and exacerbation of inappropriate release of insulin secondary to dextrose supplementation (DeAvilla & Leech 2016).
Excess glucose utilisation
Infection
Sepsis
Hypoglycaemia that accompanies any septic focus, including pyothorax, pyometra, septic peritonitis or septic arthritis is caused by non‐insulin‐mediated increased consumption of glucose and decreased intake. Increases in anaerobic glycolysis secondary to upregulation of inflammatory mediators such as tumour necrosis factor and other cytokines may be exacerbated by hypotension or hypoxia, further driving excess glucose consumption. Although glucose can aid diagnosis of sepsis in patients with septic effusion, it may not be as helpful in those without effusion (Koenig & Verlander 2015). Other studies have found no significant difference in glucose between septic and non‐septic dogs and no difference when comparing glucose in survivors versus non‐survivors with sepsis (Hauptman et al. 1997). Interestingly, in a recent retrospective evaluating septic cats, hypoglycaemia was not commonly seen or associated with outcome (Brady et al. 2000). In this cohort, hyperglycaemia was more common than hypoglycaemia.
Parasitic infections
Babesia is an example of a specific infection in which 20% of infected patients are hypoglycaemic. Similar to sepsis, hypoglycaemia is associated with consumption of glucose by the parasite, depletion of hepatic stores or hepatic dysfunction, increased anaerobic glycolysis and hyperinsulinaemia (Rees & Schoeman 2010, Keller et al. 2004). A prospective study examining the pituitary–adrenal and pituitary‐thyroid axes in dogs with babesiosis found elevations in basal and post ACTH serum cortisol concentrations as well as decreases in body temperature, serum thyroxine and free thyroxine in those patients with hypoglycaemia, suggesting that adrenal insufficiency was not responsible for the alterations in glucose levels and that adrenal and thyroidal hormones respond differently to hypoglycaemia (Schoeman & Herrtage 2007). Some studies have reported that hypoglycaemia may be associated with more pathologic strains of babesia and that hypoglycaemia at presentation is a poor prognostic indicator (Keller et al. 2004, Jacobson & Lobetti 2005, Ayoob et al. 2010). According to one study, risk factors associated with the development of hypoglycaemia in dogs with babesia included collapsed state, youth, severe anaemia, vomiting and icterus (Keller et al. 2004). Other parasitic infections also have the potential to cause hypoglycaemia in their hosts. A recent case report from France (Deschamps et al. 2016) details refractory hypoglycaemia in a 2‐year‐old female intact mixed breed dog infected with Trypanosoma congolense. The primary presenting complaint was seizure activity secondary to persistent hypoglycaemia. Trypanosomes were visible in a routine blood smear and infection was confirmed by PCR. The infection responded to therapy with intramuscular injections of pentamidine at 48‐hour intervals, the hypoglycaemia initially failed to improve despite aggressive therapy that included both intravenous supplementation of 30% glucose and administration of 70% sucrose via feeding tube. The authors speculated that glucose administration may have served as a food source for the trypanosomes as potential explanation for the persistent hypoglycaemia. A previous case report of a 6‐year‐old male intact dog with trypanosomiasis also documented a profound hypoglycaemia (Gunaseelan et al. 2009).
Hepatic dysfunction
Glucose homoeostasis is typically maintained by the liver's ability to facilitate gluconeogenesis and glycogenolysis. Hepatic diseases such as hepatitis, hepatic lipidosis, hepatic neoplasia, cirrhosis, portosystemic shunt, glycogen storage disease and hepatotoxins can all induce hepatic dysfunction (Walton 2002). Even if there is hepatic dysfunction, hypoglycaemia does not occur until more than 70% of hepatic function is lost (Center et al. 1996). Once there is depletion of glycogen stores, the body utilises protein catabolism to supply amino acids from gluconeogenesis (Weingarten & Sande 2015). A study by Lester et al. (2016) evaluating dogs with acute liver failure from multiple aetiologies revealed that on presentation, 20% of these patients were hypoglycaemic. Studies evaluating feline hepatic failure are sparse but in an evaluation of seven cats with benzodiazepine‐associated liver failure, three were hypoglycaemic but the remaining four had blood glucose levels at the lower end of the reference interval (Hughes et al. 1996).
Hypocortisolism
Hypoadrenocorticism may cause hypoglycaemia due to cortisol depletion since glucocorticoids stimulate hepatic gluconeogenesis. Studies evaluating hypoadrenocorticism in dogs showed hypoglycaemia in 3 to 37% of cases (Rakich & Lorenz 1984, Melian & Peterson 1996, Peterson et al. 1996). The percentage of dogs that go on to develop symptomatic neuroglycopenia, including seizures, appears to be quite low (Levy 1994, Melian & Peterson 1996). Apart from hypoglycaemia, other clinicopathologic abnormalities that may be noted included azotaemia, hyponatraemia, hyperkalaemia, hypochloraemia and the absence of a stress leukogram (Syme & Scott‐Moncrieff 1998, Feldman & Nelson 2004). Clinical signs may include gastrointestinal signs such as vomiting, diarrhoea and anorexia in addition to polyuria, polydipsia, bradycardia and dehydration (Feldman & Nelson 2004). Animals may be asymptomatic until confronted with the stress of concurrent illness or other physical sources of stress, including anaesthetic episodes (Lane et al. 1999). Hypoadrenocorticism is less common in cats but appears to rarely cause hypoglycaemia (Peterson et al. 1989, Stonehewer & Tasker 2001).
Therapy
Emergency treatment of the hypoglycaemic crisis
Dextrose administration
The mainstay of therapy for the patient experiencing hypoglycaemic crisis is intravenous administration of dextrose, typically using an initial bolus of 0∙5 to 1 g/kg bolus of 50% dextrose diluted 1:1 with 0∙9% NaCl followed by a constant‐rate infusion of 1∙25 to 10% dextrose diluted with an isotonic crystalloid fluid (Loose et al. 2008). Complications of intravenous dextrose include phlebitis secondary to its hypertonicity and so placement of a central line is recommended for administration of dextrose solutions with concentrations greater than 5%. Additional complications of dextrose administration include rebound hypoglycaemia, as the influx of dextrose can cause pancreatic β cells to release insulin in response to the acute hyperglycaemia and may induce recurrence of hypoglycaemia (Goutal et al. 2012). Rebound hypoglycaemia is particularly a risk during treatment of insulinomas, prompting recommendations to avoid aggressive dextrose bolusing in favour of small frequent meals and medical management, as discussed previously (Table 2).
Table 2.
Therapeutics for hypoglycaemia
| Therapy | Dose | Route/comments | Side effects |
|---|---|---|---|
| Dextrose bolus | 0∙5 to 1 mL/kg (0∙25 to 0∙5 g/kg) of 50% dextrose diluted 1:2 | iv, administered over 5 minutes | Phlebitis |
| Dextrose continuous rate infusion | 2∙5 to 5% dextrose diluted with lactated Ringers solution or 0∙9% saline | iv | |
| Glucagon bolus Glucagon CRI | 50 ng/kg iv followed by CRI of 5 to 10 ng/kg/min with a maximum dose of 40 ng/kg/min | iv | Hyperglycaemia, sedation, nausea, vomiting, hypokalaemia (rare), hypersensitivity, reactions (rare) |
| Streptozocin* | Reconstituted in 5% dextrose or 0∙9% saline | iv | Renal toxicity, GI effects (vomiting/nausea), increased liver enzymes, acute transient hypoglycaemia |
| Diazoxide* | 5 mg/kg orally every 12 hours initial dose and maximum dose of 30 mg/kg | Oral | Hypersalivation GI effects (vomiting/nausea) |
| Corticosteroids: Dexamethasone | 0∙1 to 0∙2 mg/kg iv initial dose then 0∙05 to 0∙1 mg/kg iv every 12 hours | iv | Vomiting, diarrhoea, polydipsia, polyuria, polyphagia |
| Prednisone/ prednisolone |
For treatment of hypoadrenocorticism: 0∙1 to 0∙22 mg/kg after patient stabilisation, higher doses may be needed initially. For treatment of insulinomas: 0∙25 to 0∙5 mg/kg |
Oral |
iv intravenously
Medications intended for treatment of insulinoma
Glucagon
Glucagon is a 29‐amino acid polypeptide hormone that is produced by the α cells of the pancreas in order to facilitate glycogenolysis and gluconeogenesis (Zeugswetter et al. 2012, Datte et al. 2016). In cases of hypoglycaemia that are refractory to dextrose administration, glucagon is also utilised as a therapy in human and veterinary medicine. In a recent retrospective study of nine dogs with hypoglycaemia due to a variety of causes including insulinoma and atypical hypoadrenocorticism, all patients demonstrated a statistically significant increase in blood glucose with administration of glucagon infusions compared to levels at intake (Datte et al. 2016). The only adverse effect reported in this study attributable to glucagon was mild hyperglycaemia. A recent prospective study by Zeugswetter et al. (2012) investigated the potential for use of sc glucagon as part of an emergency kit intended for patients with DM that experience hypoglycaemic crises. The authors administered 1 mg synthetic glucagon intravenously in one group of healthy beagles and sc in another group. After treatment the levels of insulin‐like immunoreactivity (Insulin‐imr), glucose, ACTH and cortisol were compared between the two groups. The authors concluded that sc glucagon did increase the glucose and Insulin‐imr levels, with peak glucose concentrations reached in 20 minutes. Administration of sc glucagon did not increase cortisol or ACTH levels. Although the peak concentration in the sc glucagon group was lower than those reached in the intravenous group, the authors concluded that it could potentially be beneficial in the initial at‐home treatment of hypoglycaemic crises in dogs with DM, although it would not be useful in the treatment of animals with suspect hypocortisolism (Zeugswetter et al. 2012). Apart from mild sedation, no other potential adverse effects were observed (Graf et al. 1999, Fall et al. 2008).
Hypoglycaemia is a common presenting problem in small animal practice and has a large variety of possible underlying causes. It is important that veterinarians are able to recognise the vast array of symptoms that a hypoglycaemic patient may exhibit. Fortunately, hypoglycaemic patients commonly respond well to initial symptomatic treatment although the prognosis for the underlying diseases vary considerably and it is important that owners are adequately informed on the prognosis of their individual animal. Advances in at‐home monitoring for patients with DM in the form of CGMS and potential future development of sc glucagon emergency kits may help owners to recognise hypoglycaemic events in the early stages and provide initial therapy before the episode becomes prolonged. Additional novel methods of early detection that may one day be extrapolated from human medicine include development of a method to analyse breath samples to detect the presence of volatile organic compounds which may signal hypoglycaemia in patients with DM (Siegel et al. 2017).
Conflict of interest
None of the authors of this article has a financial or personal relationship with other people or organisations that could inappropriately influence or bias the content of the paper.
Acknowledgements
None of the authors has a financial or personal relationship with other people or organisations that could inappropriately influence or bias the content of the paper.
References
- Altszuler, N. , Moraru, E. & Hampshire, J. (1977) On the mechanism of diazoxide‐induced hyperglycemia. Diabetes 26, 931‐935 [DOI] [PubMed] [Google Scholar]
- Aoki, T. , Sato, T. , Hasegawa, K. , et al (2004) Reversible hyperintensity lesion on diffusion‐weighted MRI in hypoglycemic coma. Neurology 63, 392‐393 [DOI] [PubMed] [Google Scholar]
- Ayoob, A. L. , Hackner, S. G. & Prittie, J. (2010) Clinical management of canine babesiosis. Journal of Veterinary Emergency and Critical Care 20, 77‐89 [DOI] [PubMed] [Google Scholar]
- Battaglia, L. , Petterino, C. , Zappulli, V. , et al (2005) Hypoglycaemia as a paraneoplastic syndrome associated with renal adenocarcinoma in a dog. Veterinary Research Communications 29, 671‐675 [DOI] [PubMed] [Google Scholar]
- Baur, A. , Harrer, T. , Peukert, M. , et al (1991) Alpha‐lipoic acid is an effective inhibitor of human immuno‐deficiency virus (HIV‐1) replication. Klinische Wochenschrift 69, 722‐724 [DOI] [PubMed] [Google Scholar]
- Beaudry, D. , Knapp, D. B. , Montgomery, T. , et al (1995) Hypoglycemia in four dogs with smooth muscle tumors. Journal of Veterinary Internal Medicine 9, 415‐418 [DOI] [PubMed] [Google Scholar]
- Bennett, K. R. , Gaunt, C. & Parker, D. (2015) Constant rate infusion of glucagon as an emergency treatment for hypoglycemia in a domestic ferret (Mustela putorius furo). Journal of the American Veterinary Medical Association 246, 451‐454 [DOI] [PubMed] [Google Scholar]
- Bergman, P. J. (2007) Paraneoplastic syndromes In: Small Animal Clinical Oncology. 4th edn. Eds Withrow S. J. and Vail D. M. Saunders, Philadelphia, PA, USA: pp 77‐94 [Google Scholar]
- Bolognesi, R. , Tsialtas, D. , Bolognesi, M. G. , et al (2011) Marked sinus bradycardia and QT prolongation in a diabetic patient with severe hypoglycaemia. Journal of Diabetes and its Complications 25, 349‐351 [DOI] [PubMed] [Google Scholar]
- Bottcher, J. , Kunze, A. , Kurrat, C. , et al (2005) Localized reversible reduction of apparent diffusion coefficient in transient hypoglycemia‐induced hemiparesis. Stroke 36, 20‐e22 [DOI] [PubMed] [Google Scholar]
- Boyle, P. J. (1997) Alteration in brain glucose metabolism induced by hypoglycaemia in man. Diabetologia 40, S69‐S74 [DOI] [PubMed] [Google Scholar]
- Brady, C. A. , Otto, C. M. , Van Winkle, T. J. , et al (2000) Severe sepsis in cats: 29 cases (1986– 1998). Journal of the American Veterinary Medical Association 217, 531‐535 [DOI] [PubMed] [Google Scholar]
- Breitschwerdt, E. B. , Goldkamp, C. , Castleman, W. L. , et al (2014) Hyperinsulinemic hypoglycemia syndrome in 2 dogs with Bartonellosis. Journal of Veterinary Internal Medicine 28, 1331‐1335 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brent, J. , McMartin, K. , Phillips, S. P. , et al (1999) Fomepizole for the treatment of ethylene glycol poisoning. New England Journal of Medicine 340, 832‐838 [DOI] [PubMed] [Google Scholar]
- Caplan, E. R. , Peterson, M. E. , Mullen, H. S. , et al (1996) Diagnosis and treatment of insulin‐secreting pancreatic islet cell tumors in ferrets: 57 cases (1986‐1994). Journal of the American Veterinary Medical Association 209, 1741‐1745 [PubMed] [Google Scholar]
- Castro, T. X. , Rita de Cassia, N. , Goncalves, L. P. , et al (2013) Clinical, hematological, and biochemical findings in puppies with coronavirus and parvovirus enteritis. The Canadian Veterinary Journal 54, 885 [PMC free article] [PubMed] [Google Scholar]
- Celebi, S. , Celebi, O. , Aydogdu, S. , et al (2011) A peculiar medical cardioversion of atrial fibrillation with glucose infusion—a rare cause of atrial fibrillation: hypoglycemia. The American Journal of Emergency Medicine 29, 134‐e1 [DOI] [PubMed] [Google Scholar]
- Center, S. A. , Guilford, W. G. & Strombeck, D. R. (1996) Acute hepatic injury: hepatic necrosis and fulminant hepatic failure Strombeck's . Small Animal Gastroenterology 3, 654‐704 [Google Scholar]
- Cohen, T. A. , Nelson, R. W. , Kass, P. H. , et al (2009) Evaluation of six portable blood glucose meters for measuring blood glucose concentration in dogs. Journal of the American Veterinary Medical Association 235, 276‐280 [DOI] [PubMed] [Google Scholar]
- Cryer, P. (2004) Diverse causes of hypoglycemia‐associated autonomic failure in diabetes. New England Journal of Medicine 350, 2272‐2279 [DOI] [PubMed] [Google Scholar]
- Cryer, P. (2012) Hypoglycemia In: Williams Textbook of Endocrinology. 12th edn. Eds Melmed S., Polonsky K. S., Larsen P. R. and Kronenberg H. M. Saunders, Philadelphia, PA, USA: pp 1582‐1607 [Google Scholar]
- Cryer, P. (2013) Mechanisms of hypoglycemia‐associated autonomic failure in diabetes. New England Journal of Medicine 369, 362‐372 [DOI] [PubMed] [Google Scholar]
- Datte, K. , Guillaumin, J. , Barrett, S. , et al (2016) Retrospective evaluation of the use of glucagon infusion as adjunctive therapy for hypoglycemia in dogs: 9 cases (2005‐2014). Journal of Veterinary Emergency and Critical Care 26, 775‐781 [DOI] [PubMed] [Google Scholar]
- Davison, L. J. , Slater, L. A. , Herrtage, M. E. , et al (2003) Evaluation of a continuous glucose monitoring system in diabetic dogs. Journal of Small Animal Practice 44, 435‐442 [DOI] [PubMed] [Google Scholar]
- DeAvilla, M. & Leech, E. (2016) Hypoglycemia associated with refeeding syndrome in a cat. Journal of Veterinary Emergency and Critical Care 26, 708‐803 [DOI] [PubMed] [Google Scholar]
- Deschamps, J. , Desquesnes, M. , Laetitia, D. , et al (2016) Refractory hypoglycaemia in a dog infected with Trypanosoma congolense. Parasite 23, 1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- DuHadway, M. R. , Sharp, C. R. , Meyers, K. E. , et al (2015) Retrospective evaluation of xylitol ingestion in dogs: 192 cases (2007‐2012). Journal of Veterinary Emergency and Critical Care 25, 646‐654 [DOI] [PubMed] [Google Scholar]
- Dunayer, E. K. (2004) Hypoglycemia following canine ingestion of xylitol‐containing gum. Veterinary and Human Toxicology 46, 87‐88 [PubMed] [Google Scholar]
- Dunayer, E. K. & Gwaltney‐Brant, S. M. (2006) Acute hepatic failure and coagulopathy associated with xylitol ingestion in eight dogs. Journal of the American Veterinary Medical Association 229, 1113‐1117 [DOI] [PubMed] [Google Scholar]
- Dunne, M. J. & Peterson, O. H. (1991) Potassium selective ion channels in insulin‐secreting cells: physiology, pharmacology and their role in stimulus secretion coupling. Biochimica et Biophysica Acta 1071, 67‐82 [DOI] [PubMed] [Google Scholar]
- Ehrhart, N. , Withrow, S. J. , Ehrhart, J. , et al (1996) Pancreatic beta cell tumor in ferrets: 20 cases (1986‐1994). Journal of the American Veterinary Medical Association 209, 1737‐1740 [PubMed] [Google Scholar]
- Elie, M. S. & Zerbe, C. A. (1995) Insulinoma in dogs, cats, and ferrets. Compendium of Continuing Education for the Practicing Veterinarian 17, 51‐59 [Google Scholar]
- Fall, T. , Holm, B. , Karlsson, A. , et al (2008) Glucagon stimulation test for estimating endogenous insulin secretion in dogs. Veterinary Record 163, 266‐270 [DOI] [PubMed] [Google Scholar]
- Feldman, E. C. & Nelson, R. W. (2004) Hypoadrenocorticism (Addison's disease) In: Canine and Feline Endocrinology and Reproduction. 3rd edn. Eds Feldman E. C. and Nelson R. W. Saunders, St Louis, MO, USA: pp 394‐439 [Google Scholar]
- Finotello, R. , Marchetti, V. , Nesi, G. , et al (2009) Pancreatic islet cell tumor secreting insulin‐like growth factor type‐II in a dog. Journal of Veterinary Internal Medicine 23, 1289‐1292 [DOI] [PubMed] [Google Scholar]
- Finotello, R. , Ressel, L. , Arvigo, M. , et al (2014) Canine pancreatic islet cell tumours secreting insulin‐like growth factor type 2: a rare entity. Veterinary and Comparative Oncology 14, 170‐180 [DOI] [PubMed] [Google Scholar]
- Froesch, E. R. & Jakob, A. (1974) The metabolism of xylitol In: Sugars in Nutrition. Eds Sipple H. L. and Mc‐Nutt K. W. Academic Press Inc, New York, NY, USA: pp 241‐258 [Google Scholar]
- Garcia‐Ortuno, L. E. , Bouda, J. , Jardon Herrera, G. , et al (2006) Clinical‐pathological diagnosis of ethylene glycol poisoning: a case report. Veterinaria México 37, 503‐512 [Google Scholar]
- Goutal, C. M. , Brugmann, B. L. & Ryan, K. A. (2012) Insulinoma in dogs: a review. Journal of the American Animal Hospital Association 48, 151‐163 [DOI] [PubMed] [Google Scholar]
- Graf, C. J. , Woodworth, J. R. , Seger, M. E. , et al (1999) Pharmacokinetic and glucodynamic comparisons of recombinant and animal‐source glucagon after IV, IM, and SC injection in healthy volunteers. Journal of Pharmacological Sciences 88, 991‐995 [DOI] [PubMed] [Google Scholar]
- Greene, S. N. & Bright, R. M. (2008) Insulinoma in a cat. Journal of Small Animal Practice 49, 38‐40 [DOI] [PubMed] [Google Scholar]
- Gregory, J. M. , Rivera, N. , Kraft, G. , et al (2017) Glucose autoregulation is the dominant component of the hormone‐independent counterregulatory response to hypoglycemia in the conscious dog. American Journal of Physiology. Endocrinology and Metabolism 313, E273‐E283 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grunert, R. R. (1960) The effect of DL‐a‐lipoic acid on heavy‐metal intoxication in mice and dogs. Archives of Biochemistry and Biophysics 86, 190‐194 [DOI] [PubMed] [Google Scholar]
- Gunaseelan, L. , Sentil Kumar, K. , Selvaraj, P. , Kathiresan, D. (2009) Haemato Biochemical Changes in a Case of Canine Trypanosomiasis. Tamilnadu Journal of Veterinary & Animal Sciences 5, 122‐123 [Google Scholar]
- Guyton, A. C. & Hall, J. E. (2006) Insulin, glucagon and diabetes mellitus In: Textbook of Medical Physiology. 11th edn. Eds Guyton A. C. and Hall J. E. Elsevier Saunders, Philadelphia, PA, USA: pp 997‐1013 [Google Scholar]
- Hambrook, L. E. , Ciavarella, A. A. , Nimmo, J. S. , et al (2016) Hyperinsulinaemic, hypoglycaemic syndrome due to acquired nesidioblastosis in a cat. Journal of Feline Medicine and Surgery Open Reports, 2, 1‐5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hauptman, J. G. , Walshaw, R. & Olivier, N. B. (1997) Evaluation of the sensitivity and specificity of diagnostic criteria for sepsis in dogs. Veterinary Surgery 26, 393‐397 [DOI] [PubMed] [Google Scholar]
- Hawks, D. , Peterson, M. E. , Hawkins, K. L. , et al (1992) Insulin‐secreting pancreatic (islet cell) carcinoma in a cat. Journal of the American Veterinary Medical Association 6, 193‐196 [DOI] [PubMed] [Google Scholar]
- Hess, L. R. , Ravich, M. L. & Reavill, D. R. (2013) Diagnosis and treatment of an insulinoma in a Guinea pig (Cavia porcellus). Journal of the American Veterinary Medical Association 242, 522‐526 [DOI] [PubMed] [Google Scholar]
- Hill, A. S. , Werner, J. A. , Rogers, Q. R. , et al (2004) Lipoic acid is 10 times more toxic in cats than reported in humans, dogs or rats. Journal of Animal Physiology and Animal Nutrition 88, 150‐156 [DOI] [PubMed] [Google Scholar]
- Houston, D. M. & Head, L. (1993) Acute alcohol intoxication in a dog. Canadian Veterinary Journal 732, 34, 41‐42 [PMC free article] [PubMed] [Google Scholar]
- Hughes, D. , Moreau, R. E. , Overall, K. L. , et al (1996) Acute hepatic necrosis and liver failure associated with benzodiazepine therapy in six cats, 1986‐1995. Journal of Veterinary Emergency and Critical Care 6, 13‐20 [Google Scholar]
- Jacob, S. , Rett, K. , Henriksen, E. J. , et al (1999) Thioctic acid—effects on insulin sensitivity and glucose metabolism. BioFactors 10, 169‐174 [DOI] [PubMed] [Google Scholar]
- Jacobson, L. S. & Lobetti, R. G. (2005) Glucose, lactate, and pyruvate concentrations in dogs with babesiosis. American Journal of Veterinary Research 66, 244‐250 [DOI] [PubMed] [Google Scholar]
- Kammerer, M. , Sachot, E. & Blanchot, D. (2001) Ethanol toxicosis from the ingestion of rotten apples by a dog. Veterinary and Human Toxicology 43, 349‐350 [PubMed] [Google Scholar]
- Kandi, S. , Desphande, N. , Rao, P. , et al (2014) Alcoholism and its relation to hypoglycemia—an overview. American Journal of Medical Studies 2, 46‐49 [Google Scholar]
- Kang, E. G. , Jeon, S. J. , Choi, S. S. , et al (2010) Diffusion MR imaging of hypoglycemic encephalopathy. American Journal of Neuroradiology 31, 559‐564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keller, N. , Jacobson, L. S. , Mirinda, N. , de Clerq, M. , Thompson, P. N. , & Schoeman, J. P. (2004) Prevalence and Risk Factors of Hypoglycemia in Virulent Canine Babesiosis. Journal of Veterinary Internal Medicine 18, 265‐270 [DOI] [PubMed] [Google Scholar]
- Keno, L. A. & Langston, C. E. (2011) Treatment of accidental ethanol intoxication with hemodialysis in a dog. Journal of Veterinary Emergency and Critical Care 21, 363‐368 [DOI] [PubMed] [Google Scholar]
- Khamaisi, M. , Rudich, A. , Potashnik, R. , et al (1999) Lipoic acid induces hypoglycemia in fasting nondiabetic and diabetic rats. Metabolism 48, 504‐510 [DOI] [PubMed] [Google Scholar]
- Kim, J. H. , Choi, J. Y. , Koh, S. B. , et al (2007) Reversible splenial abnormality in hypoglycemic encephalopathy. Neuroradiology 49, 217‐222 [DOI] [PubMed] [Google Scholar]
- Klein, B. G. (2012) Cunningham's Textbook of Veterinary Physiology. 5th edn. Saunders, Philadelphia, PA, USA: [Google Scholar]
- Koenig, A. (2015) Hypoglycemia In: Small Animal Critical Care Medicine. 2nd edn. Eds Silverstein D. C. and Hopper K. Elsevier Saunders, St. Louis, MO, USA: pp 352‐357 [Google Scholar]
- Koenig, A. & Verlander, L. L. (2015) Usefulness of whole blood, plasma, peritoneal fluid, and peritoneal fluid supernatant glucose concentrations obtained by a veterinary point‐of‐care glucometer to identify septic peritonitis in dogs with peritoneal effusion. Journal of the American Veterinary Medical Association 247, 1027‐1032 [DOI] [PubMed] [Google Scholar]
- Kraje, A. C. (2003) Hypoglycemia and irreversible neurologic complications in a cat with insulinoma. Journal of the American Veterinary Medical Association 223, 812‐814 [DOI] [PubMed] [Google Scholar]
- Kraut, J. A. & Kurtz, I. (2008) Toxic alcohol ingestions: clinical features, diagnosis, and management. Clinical Journal of the American Society of Nephrology 3, 208‐225 [DOI] [PubMed] [Google Scholar]
- Kruse, B. D. , Unterer, S. , Horlacher, K. , Sauter‐Louis, C. and Hartmann, K. (2010) Prognostic factors in cats with feline panleukopenia. Journal of veterinary internal medicine 24, 1271‐1276 [DOI] [PubMed] [Google Scholar]
- Lamberts, S. W. , van der Lely, A. J. , de Herder, W. W. , et al (1996) Octreotide. New England Journal of Medicine 334, 246‐254 [DOI] [PubMed] [Google Scholar]
- Lane, I. F. , Matwichuk, C. L. , Carpenter, L. G. , et al (1999) Profound postanesthetic hypoglycemia attributable to glucocorticoid deficiency in 2 dogs. Canadian Veterinary Journal 40, 497‐500 [PMC free article] [PubMed] [Google Scholar]
- Lane, S. L. , Koenig, A. & Brainard, B. M. (2015) Formulation and validation of a predictive model to correct blood glucose concentrations obtained with a veterinary point‐of‐care glucometer in hemodiluted and hemoconcentrated canine blood samples. Journal of the American Veterinary Medical Association 246, 307‐312 [DOI] [PubMed] [Google Scholar]
- Langford, S. D. & Boor, P. J. (1996) Oleander toxicity: an examination of human and animal toxic exposures. Toxicology 109, 1‐13 [DOI] [PubMed] [Google Scholar]
- Lester, C. , Cooper, J. , Peters, R. M. , et al (2016) Retrospective evaluation of acute liver failure in dogs (1995‐2012): 49 cases. Journal of Veterinary Emergency and Critical Care 26, 559‐567 [DOI] [PubMed] [Google Scholar]
- Levy, J. K. (1994) Hypoglycemic seizures attributable to hypoadrenocorticism in a dog. Journal of the American Veterinary Medical Association 204, 526‐528 [PubMed] [Google Scholar]
- Little, C. J. L. (2005) Hypoglycaemic bradycardia and circulatory collapse in a dog and a cat. Journal of Small Animal Practice 46, 445‐448 [DOI] [PubMed] [Google Scholar]
- Lochner, A. , Wulff, J. & Madison, L. L. (1967) Ethanol‐induced hypoglycemia: I. The acute effects of ethanol on hepatic glucose output and peripheral glucose utilization in fasted dogs. Metabolism 16, 1‐18 [DOI] [PubMed] [Google Scholar]
- Loftin, E. G. & Herold, L. V. (2009) Therapy and outcome of suspected alpha lipoic acid 818 toxicity in two dogs. Journal of Veterinary Emergency and Critical Care 19, 501‐506 [DOI] [PubMed] [Google Scholar]
- Loose, N. L. , Rudloff, E. & Kirby, R. (2008) Hypoglycemia and its effect on the brain. Journal of Veterinary Emergency and Critical Care 18, 223‐234 [Google Scholar]
- Ma, J. H. , Kim, Y. J. , Yoo, W. J. , et al (2009) MR imaging of hypoglycemic encephalopathy: lesion distribution and prognosis prediction by diffusion‐weighted imaging. Neuroradiology 51, 641‐649 [DOI] [PubMed] [Google Scholar]
- Maruya, J. , Endoh, H. , Watanabe, H. , et al (2007) Rapid improvement of diffusion weighted imaging abnormalities after glucose infusion in hypoglycaemic coma. Journal of Neurology, Neurosurgery, and Psychiatry 78, 102‐103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMichael, M. (2005) Pediatric emergencies. Veterinary Clinics of North America: Small Animal Practice 35, 421‐434 [DOI] [PubMed] [Google Scholar]
- McMichael, M. (2015) Critically ill neonatal and pediatric patients In: Small Animal Critical Care Medicine. 2nd edn. Eds Silverstein D. C. and Hopper K. Elsevier Saunders, St. Louis, MO, USA: pp 860‐865 [Google Scholar]
- McMillan, F. D. , Barr, T. & Feldman, H. C. (1985) Functional pancreatic islet cell tumor in a cat. Journal of the American Animal Association 21, 741‐746 [Google Scholar]
- Means, C. (2003) Bread dough toxicosis in dogs. Journal of Veterinary Emergency and Critical Care 13, 39‐41 [Google Scholar]
- Means, C. (2008) Ataxia and vomiting in a German Shepherd. North American Veterinary Community Clinician's Brief 6, 31‐33 [Google Scholar]
- Mehta, H. B. , Mehta, V. & Goodwin, J. S. (2016) Association of hypoglycemia with subsequent dementia in older patients with type 2 diabetes mellitus. The Journals of Gerontology. Series A, Biological Sciences and Medical Sciences 72, 1110‐1116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melano, K. A. & Peterson, M. E. (2013) Treatment of insulinoma in the dog, cat, and ferret In: Kirk's Current Veterinary Therapy, Vol. XV Eds Bonagura J. D. and Twedt D. C. Saunders Elsevier, Philadelphia, PA, USA: pp 130‐134 [Google Scholar]
- Melian, C. & Peterson, M. E. (1996) Diagnosis and treatment of naturally occurring hypoadrenocorticism in 42 dogs. Journal of Small Animal Practice 37, 268‐275 [DOI] [PubMed] [Google Scholar]
- Mila, H. , Grellet, A. , Delebarre, M. , et al (2017) Monitoring of the newborn dog and prediction of neonatal mortality. Preventive Veterinary Medicine 143, 11‐20 [DOI] [PubMed] [Google Scholar]
- Molina, P. E. & Abumrad, N. N. (2001) Contribution of excitatory amino acids to hypoglycemic counter‐regulation. Brain Research 899, 201‐208 [DOI] [PubMed] [Google Scholar]
- Moore, A. S. , Nelson, R. W. , Henry, C. J. , et al (2002) Streptozocin for treatment of pancreatic islet cell tumors in dogs: 17 cases(1989‐1999). Journal of the American Veterinary Medicine Association 221, 811‐818 [DOI] [PubMed] [Google Scholar]
- Mori, F. , Nishie, M. , Houzen, H. , et al (2006) Hypoglycemic encephalopathy with extensive lesions in the cerebral white matter. Neuropathology 26, 147‐152 [DOI] [PubMed] [Google Scholar]
- Mwafy, S. N. & Yassin, M. M. (2011) Antidiabetic activity evaluation of glimepiride and Nerium oleander extract on insulin, glucose levels and some liver enzyme activities in experimental diabetic rat model. Pakistan Journal of Biological Sciences 14, 984‐990 [DOI] [PubMed] [Google Scholar]
- Packer, L. , Witt, E. H. & Tritschler, H. J. (1995) Alpha‐lipoic acid as a biological antioxidant. Free Radical Biology and Medicine 19, 227‐250 [DOI] [PubMed] [Google Scholar]
- Page, C. & Murtaugh, R. J. (2015) Hypoglycemia associated with oleander toxicity in a dog. Journal of Medical Toxicology 11, 145‐143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson, M. E. , Greco, D. S. & Orth, D. N. (1989) Primary hypoadrenocorticism in ten cats. Journal of Veterinary Internal Medicine 3, 55‐58 [DOI] [PubMed] [Google Scholar]
- Peterson, M. E. , Kintzer, P. P. & Kass, P. H. (1996) Pretreatment clinical and laboratory findings in dogs with hypoadrenocorticism: 225 cases (1973‐1993). Journal of the American Veterinary Medicine Association 208, 85‐91 [PubMed] [Google Scholar]
- Piscitelli, C. M. , Dunayer, E. K. & Aumann, M. (2010) Xylitol toxicity in dogs. Compendium on Continuing Education for the Practising Veterinarian 32, E1‐E4 [PubMed] [Google Scholar]
- Polton, G. , White, R. N. , Brearley, M. J. , et al (2007) Improved survival in a retrospective cohort of 28 dogs with insulinoma. Journal of Small Animal Practice 48, 151‐156 [DOI] [PubMed] [Google Scholar]
- Radosevich, P. M. , Lacy, D. B. , Brown, L. L. , et al (1988) Effects of insulin‐induced hypoglycemia on plasma and cerebrospinal fluid levels of ir‐beta‐endorphins, ACTH, cortisol, norepinephrine, insulin and glucose in the conscious dog. Brain Research 458, 325‐338 [DOI] [PubMed] [Google Scholar]
- Rakich, P. M. & Lorenz, M. D. (1984) Clinical signs and laboratory abnormalities 902 in 23 dogs with spontaneous hypoadrenocorticism. Journal of the American Animal Hospital Association 20, 647‐649 [Google Scholar]
- Ratclife, R. C. & Zuber, R. M. (1977) Acute ethyl alcohol poisoning in dogs. Australian Veterinary Journal 53, 48‐49 [DOI] [PubMed] [Google Scholar]
- Reineke, E. L. , Fletcher, D. J. , King, L. G. , et al (2010) Accuracy of a continuous glucose monitoring systemin dogs and cats with diabetic ketoacidosis. Journal of Veterinary Emergency and Critical Care 20, 303‐312 [DOI] [PubMed] [Google Scholar]
- Rees, P. & Schoeman, J.P. (2010) Plasma insulin concentrations in hypoglycaemic dogs with Babesia canis rossi infection. Veterinary Parasitology 152, 60‐66 [DOI] [PubMed] [Google Scholar]
- Ristic, J. M. , Herrtage, M. E. , Walti‐Lauger, S. M. , et al (2005) Evaluation of a continuous glucose monitoring system in cats with diabetes mellitus. Journal of Feline Medicine and Surgery 7, 153‐162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robben, J. H. , Visser‐Wisselaar, H. A. , Rutteman, G. R. , et al (1997) In vitro and in vivo detection of functional somatostatin receptors in canine insulinomas. Journal of Nuclear Medicine 38, 1036‐1042 [PubMed] [Google Scholar]
- Robben, J. H. , Pollak, Y. W. , Kirpensteijn, J. , et al (2005) Comparison of ultrasonography, computed tomography, and single‐photon emission computed tomography for the detection and localization of canine insulinoma. Journal of Veterinary Internal Medicine 19, 15‐22 [DOI] [PubMed] [Google Scholar]
- Robben, J. H. , van den Brom, W. E. , Mol, J. A. , et al (2006) Effect of octreotide on plasma concentrations of glucose, insulin, glucagon, growth hormone and cortisol in healthy dogs and dogs with insulinoma. Research in Veterinary Science 80, 23‐32 [DOI] [PubMed] [Google Scholar]
- Sanon, V. P. , Sanon, S. , Kanakia, R. , et al (2014) Hypoglycemia from a Cardiologist's perspective. Clinical Cardiology 37, 499‐504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoeman, J. P. & Herrtage, M. E. (2007) The response of the pituitary‐adrenal and pituitary‐thyroidal axes to the plasma glucose perturbations in Babesia canis rossi babesiosis. Journal of the South African Veterinary Association 78, 215‐220 [DOI] [PubMed] [Google Scholar]
- Senniappan, S. , Arya, V. B. & Hussain, K. (2013) The molecular mechanisms, diagnosis and management of congenital hyperinsulinism. Indian Journal of Endocrinology and Metabolism 17, 19‐30 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimada, A. , Morita, T. , Ikeda, N. , et al (2000) Hypoglycaemic 946 brain lesions in a dog with Insulinoma. Journal of Comparative Pathology 122, 67‐71 [DOI] [PubMed] [Google Scholar]
- Siegel, A.P. , Daneshkah, A. , Shrestha, S. , et al (2017) Analyzing breath samples of hypoglycemic events in type 1 diabetes patients: towards developing an alternative to diabetes alert dogs. Journal of Breath Research 11, 026007 [DOI] [PubMed] [Google Scholar]
- Singhal, A. , Hadi, R. , Mehrotra, K. , et al (2017) Paraneoplastic Hypoglycaemia: a rare manifestation of pelvic gastrointestinal stromal tumour. Journal of Clinical and Diagnostic Research 11, XD01‐XD02 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solnica, B. , Skupien, J. , Kusnierz‐Cabala, B. , et al (2012) The effect of hematocrit on the results of measurements using glucose meters based on different techniques. Clinical Chemistry and Laboratory Medicine 50, 361‐365 [DOI] [PubMed] [Google Scholar]
- Stonehewer, J. & Tasker, S. (2001) Hypoadrenocorticism in a cat. Journal of Small Animal Practice 42, 186‐190 [DOI] [PubMed] [Google Scholar]
- Suter, R. J. (1992) Presumed ethanol intoxication in sheep dogs fed uncooked pizza dough. Australian Veterinary Journal 69, 20 [DOI] [PubMed] [Google Scholar]
- Syme, H. M. & Scott‐Moncrieff, J. C. (1998) Chronic hypoglycaemia in a hunting dog clue to secondary hypoadrenocorticism. Journal of Small Animal Practice 39, 348‐351 [DOI] [PubMed] [Google Scholar]
- Tang, Z. , Lee, J. H. , Louie, R. F. , et al (2000) Effects of different hematocrit levels on glucose measurements with handheld meters for point‐of‐care testing. Archives of Pathology & Laboratory Medicine 124, 1135‐1140 [DOI] [PubMed] [Google Scholar]
- Thrall, M. A. , Freemyer, F. G. , Hamar, D. W. , et al (1984) Ethanol toxicosis secondary to sourdough ingestion in a dog. Journal of the American Veterinary Medicine Association 184, 1513‐1514 [PubMed] [Google Scholar]
- Todd, J. M. & Powell, L. L. (2007) Xylitol intoxication associated with fulminant hepatic failure in a dog. Journal of Veterinary Emergency and Critical Care 17, 286‐289 [Google Scholar]
- Tonyushkina, K. & Nichols, J. H. (2009) Glucose meters: a review of technical challenges to obtaining accurate results. Journal of Diabetes Science and Technology 3, 971‐980 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venn, E. C. , Preisner, K. , Boscan, P. L. , et al (2017) Evaluation of an outpatient protocol in the treatment of canine parvoviral enteritis. Journal of Veterinary Emergency and Critical Care 27, 52‐65 [DOI] [PubMed] [Google Scholar]
- Viebrock, K. A. & Dennis, J. (2018) Hypoglycemic episodes in cats with diabetes mellitus: 30 cases (2013‐2015). Journal of Feline Medicine and Surgery 20(6), 563‐570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walton, R. S. (2002) Severe liver disease In: The Veterinary ICU. Book edn. Eds Wingfield W. E. and Raffe M. R. Teton New Media, Jackson, WY, USA: pp 785‐803 [Google Scholar]
- Weingarten, M. A. & Sande, A. A. (2015) Acute liver failure in dogs and cats. Journal of Veterinary Emergency and Critical Care 25, 455‐473 [DOI] [PubMed] [Google Scholar]
- Weinstock, R. S. , DuBose, S. N. , Bergenstal, R. M. , et al (2016) Risk factors associated with severe hypoglycemia in older adults with type 1 diabetes. Diabetes Care 39, 603‐661 [DOI] [PubMed] [Google Scholar]
- Weiss, C. A. , Williams, B. H. & Scott, M. V. (1998) Insulinoma in the ferret: clinical findings and treatment comparison of 66 cases. Journal of the American Animal Hospital Association 34, 471‐475 [DOI] [PubMed] [Google Scholar]
- Wenzel, U. , Nickel, A. & Daniel, H. (2005) A‐lipoic acid induces apoptosis in human colon cancer cells by increasing mitochondrial respiration with a concomitant O2 ‐generation. Apoptosis 10, 359‐368 [DOI] [PubMed] [Google Scholar]
- Whitley, N. T. , Drobatz, K. J. & Panciera, D. L. (1997) Insulin overdose in dogs and cats: 28 cases (1986‐1993). Journal of the American Veterinary Medical Association 211, 326‐330 [PubMed] [Google Scholar]
- Wiedmeyer, C. E. , Johnson, P. J. , Cohn, L. A. , et al (2005) Evaluation of a continuous glucose monitoring system for use in veterinary medicine. Diabetes Technology & Therapeutics 7, 885‐895 [DOI] [PubMed] [Google Scholar]
- Witsch, J. , Neugebauer, H. , Flechsenhar, J. , et al (2012) Hypoglycemic encephalopathy: a case series and literature review on outcome determination. Journal of Neurology 259, 2172‐2181 [DOI] [PubMed] [Google Scholar]
- Xia, Z. , He, Y. & Yu, J. (2009) Experimental acute toxicity of xylitol in dogs. Journal of Veterinary Pharmacology and Therapeutics 32, 465‐469 [DOI] [PubMed] [Google Scholar]
- Yadav, V. , Marracci, G. , Lovera, J. , et al (2005) Lipoic acid in multiple sclerosis: a pilot study. Multiple Sclerosis 11, 159‐165 [DOI] [PubMed] [Google Scholar]
- Yaffe, K. , Falvey, C. M. & Hamilton, N. (2013) Association between hypoglycemia and dementia in a biracial cohort of older adults with diabetes mellitus. Journal of American Medical Association Internal Medicine 173, 1300‐1306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeugswetter, F. K. , Schornsteiner, E. , Haimel, G. , et al (2012) Metabolic and hormonal responses to subcutaneous glucagon in healthy beagles. Journal of Veterinary Emergency and Critical Care 22, 558‐563 [DOI] [PubMed] [Google Scholar]
- Ziegler, D. , Ametov, A. , Barinov, A. , et al (2006) Oral treatment with α‐lipoic acid improves symptomatic diabetic polyneuropathy: the SYDNEY 2 trial. Diabetes Care 29, 2365‐2370 [DOI] [PubMed] [Google Scholar]
- Zini, E. , Moretti, S. , Tschuor, F. , et al (2009) Evaluation of a new portable glucose meter designed for the use in cats. Schweizer Archiv für Tierheilkunde 151, 448‐451 [DOI] [PubMed] [Google Scholar]


