Abstract
BACKGROUND: Globally, severe acute respiratory syndrome coronavirus (SARS‐CoV) is a newly emerging virus that causes SARS with high mortality rate in infected people. The nucleocapsid (N) protein of the severe acute respiratory syndrome (SARS)‐associated coronavirus (SARS‐CoV) is an important antigen for the early diagnosis of SARS and the detection of diseases. Here, a new quantum dots (QDs)‐conjugated RNA aptamer with high sensitivity and rapidity is proposed for the detection of SARS‐CoV N protein using an on chip system.
RESULTS: A QDs‐conjugated RNA aptamer can specifically hybridize on the immobilized SARS‐CoV N protein on the surface of a glass chip. Detection is based on the optical signal variation of a QDs‐supported RNA aptamer interacting on an immobilized protein chip. Using an optical QDs‐based RNA aptamer chip, SARS N protein was detected at concentrations as low as 0.1 pg mL−1.
CONCLUSIONS: It was demonstrated that the QDs‐conjugated RNA aptamer could interact on a designed chip specifically and sensitively. This device could form a QDs‐conjugated biosensor prototype chip for SARS‐CoV N protein diagnosis. The proposed visual SARS‐CoV N protein detection technique may avoid the limitations of other reported methods because of its high sensitivity, good specificity, ease of use, and the ability to perform one‐spot monitoring. Copyright © 2011 Society of Chemical Industry
Keywords: SARS coronavirus (CoV), nucleocapsid (N) protein, aptamer, quantum dots (QDs)
INTRODUCTION
Severe acute respiratory syndrome (SARS) is an infectious disease that began in Guandong, China in November 2002, and has caused serious infection in Asia, Europe and Canada.1, 2, 3, 4, 5 The SARS coronavirus (CoV), which is a pathogenic virus of SARS, is a single‐strand RNA virus. It is known that this virus infects animals other than humans.6, 7, 8 According to the World Health Organization (WHO), mortality for patients afflicted with SARS is about 15% on average, and can be 50% or higher in the case of patients aged 65 and over.9 SARS‐CoV is an enveloped and positively single‐stranded RNA virus with a typical genome size 29.7 kb, encoding a RNA directed RNA polymerase and structural proteins including the spike (S), envelope (E), membrane (M) and nucleocapsid (N) proteins.10, 11, 12 The N protein is a 422‐amino‐acid alkaline protein with a short lysine‐rich region suggested to be the nuclear localization signal. It plays an important role in the process of virus particle assembly by enveloping the entire genomic RNA.13 The nucleocapsid (N) protein of the SARS‐associated coronavirus (SARS‐CoV) is an important antigen for the early diagnosis of SARS and the detection of diseases.14 Since SARS broke out in 2003, researchers have made great efforts in the development of fast and accurate analytical methods for its early diagnosis.15, 16, 17, 18, 19
The Nucleocapsid (N) protein of SARS‐CoV is a major pathological determinant in the host that may cause host cell apoptosis, up‐regulate pro‐inflammatory cytokine production, and block innate immune responses. Therefore, SARS‐CoV N protein has long been thought an ideal target for the detection of SARS. The monitoring of SARS‐CoV N protein has been of considerable interest in the development of simple and reliable methods for the detection of SARS for applications in diagnostic medicine. At present, the most widely used method of diagnosing SARS is the detection of anti‐SARS antibodies using a screening enzyme‐linked immunosorbent assay (ELISA), based on recombinant proteins from the genome of SARS.18, 19 Although it is highly specific, this assay has limitations. For example, it cannot detect viruses during the early stage of an infection, at a time when antibodies against SARS antigens are not produced. In addition, the ELISA method sometimes generates false‐positive or false‐negative results. In addition, using antibodies creates problem such as being temperature‐sensitive, specific reactions conditions and requiring secondary antibody conjugated with enzyme and fluorescent dyes.
Aptamers have become increasingly important molecular tools for diagnostics and therapeutics. In particular, Aptamers exhibit advantages as recognition elements in biosensing when compared with antibodies and enzymes, thus making it possible to develop a wide range of aptamer‐based biosensors. Chemically stable and cost‐effective aptamers offer great flexibility in the design of novel biosensors with high detection sensitivity and selectivity.20, 21, 22, 23, 24
Quantum dots (QDs), which are colloidal nanoparticles of semiconductor materials, have attracted much attention in the fields of nanotechnology and biotechnology, especially in biological imaging applications because of their remarkable optical characteristics compared with conventional organic fluorophores in terms of being bright, tunable and having narrow fluorescence emission, and broad absorption spectra.25, 26, 27
In this study, it was established that optical QDs‐conjugated aptamer could be used as a probe for the detection of SARS‐CoV N protein using a chip system. Using an optical QDs‐based RNA aptamer chip, we could detect the SARS‐CoV N protein at concentrations as low as 0.1 pg mL−1. A biosensor assay for SARS‐CoV N protein detection based on an optical QDs‐based aptamer has attracted significant attention. It is a promising method for the specific detection of oligonucleotide because of its high sensitivity, low cost, rapid response, compatibility for miniaturization and low labor requirements.
MATERIALS AND METHODS
Chemicals
EDC (N‐(3‐dimethylaminopropyl)‐N′‐ethylcarbodiimide hydrochloride), bovine serum albumin (BSA), ampicillin and kanamycin were purchased from Sigma–Aldrich Chemical Co. (St Louis, MO, USA). Quantum dots (QDs 605) were purchased from Invitrogen Corporation (Carlsbad, CA, USA). Prolinker™ terminated glass slides from Proteogen (Seoul, Republic of Korea) were used. All other chemicals were of the highest grade.
Preparation of aptamer
The aptamer sequence reported by Oh et al. was used.28 The sequence is 5′‐gggagagcggaagcgugcugggccugucgguucgcuguc uugcuacguuacguuacacgguuggcauaacccagaggucgauggaucccccc‐3'. The production of designed aptamer and primer with T7 promoter sequence was synthesized by BIONEER Co. Ltd (Seoul, Republic of Korea) and amplified by PCR. The template RNA was prepared by in vitro transcription using T7 RNA polymerase (Promega, WI, USA). All RNA transcript product was separated by 8 mol L−1 urea 6% PAGE gel after phenol extraction and ethanol precipitation procedures. The RNA aptamer produced was dissolved in a DEPC solution.
Subcloning, expression and purification of SARS‐CoV N protein
The gene was amplified by a PCR with the primer set, sense: 5′‐ agtggatccatgtctgataatggacccca‐3', antisense: 5′‐gccgtcgacttatgcctgagttgaatcagc‐3', containing restriction enzyme sites of BamHI/SalI. PCR was run with the following conditions on a thermal cycler: denaturation at 94 °C for 1 min, annealing at 60 °C for 30 s, and an extension step at 72 °C for 2 min 30 s. The sequence was repeated 35 times followed by a 7 min final extension step at 72 °C. The PCR product was digested with BamHI/SalI, and then ligated into BamHI/SalI digested expression vector pET 24a+ (Novagen, Madison, WI), and transformed into E. coli DH5α (Stratagene, La Jolla, CA). The transformed colony with inserted gene was transformed into E.coli BL21 (DE3) (Stratagene, La Jolla, CA). Then, it was plated on LB agar containing 50 µg mL−1 kanamycin. GroES/EL expressing plasmid from E.coli and SARS‐CoV N‐expressing plasmid, which possessed ampicillin and kanamycin resistant markers, respectively, were co‐transformed into E. coli BL21 (DE3) according to the biotransformation procedures. The transformant was grown in a 250 mL flask containing 50 mL Luria‐Bertani (LB) medium supplemented with 50 µg mL−1 of kanamycin and ampicillin at 37 °C until the cell concentration reached an OD600nm of 0.6, and isopropyl‐thio‐β‐D‐galactopyranoside (IPTG) of a final concentration of 0.1 mmol L−1, followed by growing for an additional overnight at 25 °C with shaking at 180 rpm. The cells were harvested by centrifugation at 4000 rpm for 30 min at 4 °C and resuspended in 100 mmol L−1 potassium phosphate buffer (pH 7.5) containing 1 mmol L−1 phenylmethylsulfonyl fluoride (PMSF). The cells were lysed by Sonicator (F60 Sonic Dismembrator, Fisher Scientific, USA). The cell debris was removed by centrifugation at 13 000 rpm for 30 min. The supernatant was collected and the recombinant SARS‐CoV N protein was purified with Ni‐nitrilotriacetic acid (Ni‐NTA) affinity chromatography column (Qiagen, Germany). The supernatant was equilibrated with buffer A (10 mmol L−1 Tri‐HCl, 500 mmol L−1 NaCl, 50 mmol L−1 imidazole, 1 mmol L−1 PMSF, pH 8.0). The bound protein was eluted with buffer B (10 mmol L−1 Tris‐HCl, 500 mmol L−1 NaCl, 250 mmol L−1 imidazole, 1 mmol L−1 PMSF, pH 8.0) at 4 °C. The purity of the purified protein was estimated by SDS‐PAGE in the eluted fractions, using 12% polyacrylamide running gels.29 The purity of the enzyme was estimated by SDS‐PAGE. The protein concentration was determined using the Bradford method.30 The purified sample was supplemented with 50% glycerol and stored at − 20 °C until use.
Conjugation of quantum dots and aptamer
The amine group of RNA aptamer was first covalently conjugated onto the surface of the carboxyl terminated QDs605 (10 pmol L−1). That is, 10 pmol L−1 of QDs605 were conjugated with 400 pmol L−1 of aptamer with the coupling reagent EDC (N‐(3‐dimethylaminopropyl)‐N‐ethyl‐carbodiimide hydrochloride, 40 nmol L−1), which was used to activate amide bond formation to produce QDs‐conjugated aptamer (QDs based SARS‐CoV N aptamer) at a QDs:aptamer molar ratio of 1:40 for 1 h at room temperature. Then, QDs‐aptamer conjugate was collected using centrifugal filtration at 15 000 rpm for 30 min, followed by several washing steps with a Tris buffer (50 mmol L−1 Tris‐HCl (pH 7.4), 5 mmol L−1 KCl, 100 mmol L−1 NaCl, 1 mmol L−1 MgCl2, and 0.1% NaN3). After centrifugal filtration and washing, the pellet of QDs‐aptamer was dispersed by brief sonication (22 kHz, amplitude 12 µm, and sonication time 120 s) using a Sonic Dismembrator Model F60 (Fisher Scientific, Fair Lawn, NJ, USA).
Confocal laser scanning microscopy
The recombinant SARS‐CoV N protein was directly immobilized onto the functional ProLinker™‐terminated surface. For the binding of the specific aptamer, the conjugated QDs‐conjugated aptamer was facilitated by spotting on immobilized SARS‐CoV N protein chip. After incubation for 1 h at 25 °C, the chip was then washed three times with phosphate buffer (pH 7.2) for 1 min. The chip was analyzed using a confocal laser scanning microscope LSM510 META (Carl Zeiss, Jena, Germany). The signal intensity was determined by software for the LSM510 (LSM Image Browser). A histogram of the intensity was obtained for the region of the spotted chip. The value of signal intensity was recorded calculating and expressing it as the mean intensity.
RESULTS AND DISCUSSION
A schematic outline of aptamer chip assay
To ensure the feasibility of the targeting and imaging, we designed QDs605 conjugates having RNA aptamer for specific SARS‐CoV N protein. We used a highly sensitive chip using a ProteoChip coated with ProLinker™, novel calixcrown derivatives with a bi‐functional coupling property that permits the efficient immobilization of captured SARS‐CoV N protein on solid matrixes and makes simple analysis of protein–RNA interactions possible.31 Specific detection of the SARS‐CoV N protein was demonstrated by the QDs‐conjugated RNA aptamer on chip. The total procedure for the detection on chip was as follows. First, the SARS‐CoV N protein was spotted on a glass chip with carboxyl as the functional group. Second, the QDs‐conjugated RNA oligonucleotide was bound on an immobilized chip. Third, the bound chip was washed to remove unspecific binding. Fourth, it was analyzed to recognize the specific detection of SARS‐CoV N on chip. The design of targeting for effectively monitoring the SARS‐CoV N protein on chip is illustrated in Fig. 1.
Figure 1.
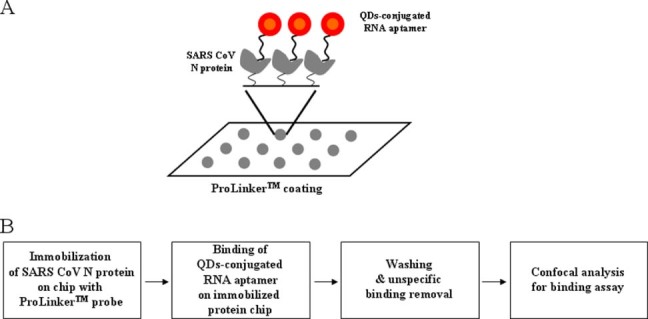
A representative scheme for detection of SARS‐CoV N protein on chip: (A) scheme of SARS‐CoV N protein assay on chip; (B) total procedures of SARS‐CoV N protein assay.
Quantum dots (QDs)‐conjugated aptamer
The QDs‐supported RNA aptamer was conjugated in the reaction for the amide formation from the coupling of 5′‐end‐amine‐modified RNA aptamer at the surface of QDs displaying carboxyl groups via standard EDC coupling. The QDs‐conjugated RNA aptamer was confirmed on a 2.5% agarose gel at 100 V in TAE buffer. Agarose gel electrophoresis showed a band with mobility, showing a different band pattern between the free QDs and the QDs‐conjugated RNA aptamer, confirming the formation of QDs‐conjugated RNA oligonucleotide. The mobility shifts are compared in Fig. 2. On agarose gel, the QDs‐conjugated RNA aptamer showed less mobility shift than free QDs, thus demonstrating the amide formation between QDs and RNA aptamer for conjugation.
Figure 2.

The conjugation pattern on 2.5% agarose gel electrophoresis was examined at UV excitation wavelength 345 nm, showing a different band pattern between free QDs (lane 1) and QDs‐conjugated aptamer (lane 2).
Sensitive detection of SARS‐CoV N protein on chip
To improve the solubility of bacterially overexpressed recombinant N protein, the co‐expression of N protein with E. coli molecular chaperone GroES/EL was performed. The SARS‐CoV N protein was purified by a single chromatography step on a Ni2+ affinity column. The C‐terminally his‐tagged SARS‐CoV N protein was visualized with a molecular mass of approximately 48 kDa on a SDS‐PAGE (Fig. 3(A)). Figure 3(B) shows the sequence and secondary structure of RNA aptamer that binds to SARS‐CoV N protein. The RNA secondary structure of the used aptamer was analyzed using the Mfold program.32
Figure 3.
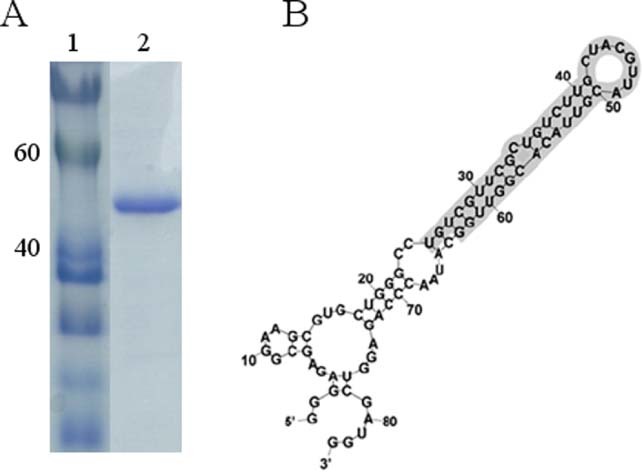
Purification of SARS‐CoV N protein: (A) 12% SDS‐PAGE gel showing SARS‐CoV N protein with his‐tag. lane 1, protein marker; lane 2, his‐tag form of SARS‐CoV N protein; (B) sequence and secondary structure of RNA aptamer that binds to N protein.
We demonstrated the specific interaction between QDs‐conjugated RNA aptamer and immobilized SARS‐CoV N protein on a chip. Preferential binding of QDs‐conjugated RNA aptamer for the detection of SARS‐CoV N protein was determined by analyzing the fluorescence intensity measured by confocal microscopy. As illustrated in Fig. 4(B), the QDs‐supported RNA aptamer conjugates showed high fluorescence signals on the chip. No fluorescence signal was detected with BSA protein, due to its lack of affinity (Fig. 4(A)). The signal of QDs‐conjugated RNA oligonucleotide for BSA protein binding affinity was very similar to the background signal. Figure 4(C) and 4(D) shows that QDs‐conjugated RNA aptamer is selective against SARS‐CoV N protein due to protein concentrations. The binding affinity was examined by confocal assay with varying concentrations of the SARS‐CoV N protein. It was observed that the signal intensity of the QDs‐conjugated RNA aptamer could be detected even at the lowest concentration (0.1 pg mL−1). The signal intensity was found to increase gradually by up to 0.1 pg mL−1 of SARS‐CoV N protein concentration. These results suggest the QDs‐conjugated RNA aptamer could specifically target the SARS‐CoV N protein on an immobilized chip. The detection limit was improved at least 10‐fold compared with the aptamer‐based chemiluminescence immunosorbent assay.28 Furthermore, this system may be useful for rapid detection of SARS‐CoV N protein within 1 h. These results suggest that the QDs‐conjugated RNA oligonucleotide could be effective for the quantification of SARS‐CoV N protein. The immobilized BSA protein chip showed no binding to RNA aptamer, thus demonstrating selectivity for SARS‐CoV N protein. The remarkable imaging by semiconductor nanocrystals QDs allows for monitoring with the intense light emission and accuracy.
Figure 4.
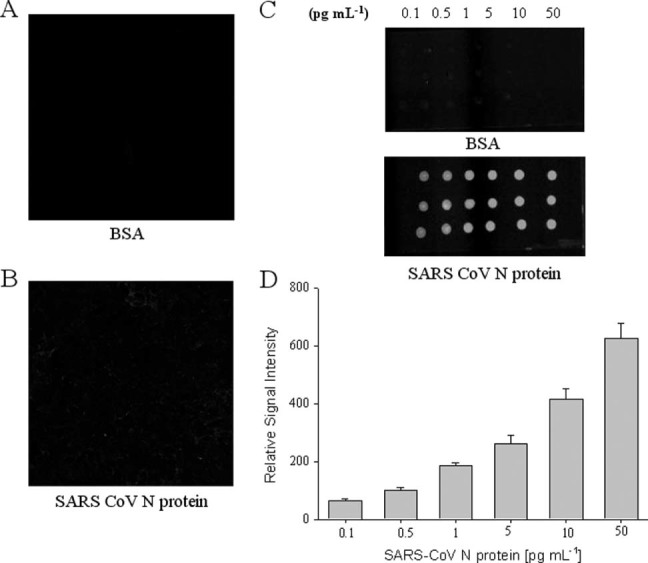
Detection of SARS‐CoV N protein by QDs‐conjugated aptamer assay. Difference in fluorescent emission was measured: (A) BSA; (B) SARS‐CoV N protein. Effect of SARS‐CoV N protein concentrations on binding assay: (C) spotted chip; (D) normalized signal intensity.
In summary, we showed that QDs‐conjugated RNA oligonucleotide can specifically recognize the SARS‐CoV N protein. We demonstrated that this QDs‐conjugated RNA aptamer could interact on a designed chip specifically and sensitively. This device could form a QDs‐conjugated biosensor prototype chip for SARS‐CoV N protein diagnosis. The proposed visual SARS‐CoV N protein detection technique may avoid the limitations of other reported methods because of its high sensitivity, good specificity, simplicity, and speed.
CONCLUSIONS
The results of this study suggest that the QDs‐conjugated RNA aptamer can recognize the SARS‐CoV N protein with a detection limit of 0.1 pg mL−1 on a designed chip. Thus, it could be developed as a sensitive diagnostic tool. Furthermore, this QDs‐conjugated aptamer chip might have application potential as a simple and rapid diagnosis tool. This technique has potential applications in many fields, especially in multiple diseases detection.
Acknowledgements
We thank Professor Dr Jong‐Won Oh for his kind donation of SARS‐CoV N gene. This work was supported by SNU‐KAERI Degree and Research Center for Radiation Convergence Sciences from Korea Research Council of Foundation (KRCF) Science & Technology, Republic of Korea. Also, this research was in part supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (grant no. 2011‐0010634).
REFERENCES
- 1. Cleri DJ, Ricketti AJ and Vernaleo JR, Severe acute respiratory syndrome (SARS). Infect Dis Clin North Am 24: 175–202 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Denison MR, Graham RL, Donaldson EF, Eckerle LD and Baric RS, Coronaviruses: an RNA proofreading machine regulates replication fidelity and diversity. RNA Biol 8: 270–279 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Du L, He Y, Zhou Y, Liu S, Zheng BJ and Jiang S, The spike protein of SARS‐CoV—a target for vaccine and therapeutic development. Nat Rev Microbiol 7: 226–236 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Peiris JSM, Lai ST, Poon LLM, Guan Y, Yam LYC, Lim W et al, Coronavirus as a possible cause of severe acute respiratory syndrome. Lancet 361: 1319–1325 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Drosten C, Gunther S, Preiser W, van der Werf S, Brodt HR, Becker S et al, Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med 348: 1967–1976 (2003). [DOI] [PubMed] [Google Scholar]
- 6. Marra MA, Jones SJ, Astell CR, Holt RA, A Brooks‐Wilson YSN, Butterfield JK et al, The Genome sequence of the SARS‐associated coronavirus. Science 300: 1399–1404 (2003). [DOI] [PubMed] [Google Scholar]
- 7. Rota PA, Oberste MS, Monroe SS, Nix WA, Campagnoli R, Icenogle JP et al, Characterization of a novel coronavirus associated with severe acute respiratory syndrome. Science 300: 1394–1399 (2003). [DOI] [PubMed] [Google Scholar]
- 8. Smits SL, van den Brand JM, de Lang A, Leijten LM, van Ijcken WF, van Amerongen G et al, Distinct severe acute respiratory syndrome coronavirus‐induced acute lung injury pathways in two different nonhuman primate species. J Virol 85: 4234–4245 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.(2003). World Health Organization. Severe acute respiratory syndrome (SARS): multi‐country outbreak. http://www.who.int/csr/don/2003_03‐16/en/.
- 10. Marra MA, Jones SJ, Astell CR, Holt RA, Brooks‐Wilson A, Butterfield YS et al, The Genome sequence of the SARS‐associated coronavirus. Science 300: 1399–1404 (2003). [DOI] [PubMed] [Google Scholar]
- 11. Rota PA, Oberste MS, Monroe SS, Nix WA, Campagnoli R, Icenogle JP et al, Characterization of a novel coronavirus associated with severe acute respiratory syndrome. Science 300: 1394–1399 (2003). [DOI] [PubMed] [Google Scholar]
- 12. Ruan YJ, Wei CL, Ee AL, Vega VB, Thoreau H, Su ST et al, Comparative full‐length genome sequence analysis of 14 SARS coronavirus isolates and common mutations associated with putative origins of infection. Lancet 361: 1779–1785 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Hatakeyama S, Matsuoka Y, Ueshiba H, Komatsu N, Itoh K, Shichijo S et al, Dissection and identification of regions required to form pseudoparticles by the interaction between the nucleocapsid (N) and membrane (M) proteins of SARS coronavirus. Virology 380: 99–108 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Hurst KR, Kuo L, Koetzner CA, Ye R, Hsue B and Masters PS, A major determinant for membrane protein interaction localizes to the carboxy‐terminal domain of the mouse coronavirus nucleocapsid protein. J Virol 79: 13285–13297 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Peiris JS and Poon LL, Detection of SARS coronavirus. Methods Mol Biol 665: 369–382 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Grant PR, Garson JA, Tedder RS, Chan PK, Tam JS and Sung JJ, Detection of SARS coronavirus in plasma by real‐time RT‐PCR. N Engl J Med 349: 2468–2469 (2003). [DOI] [PubMed] [Google Scholar]
- 17. Poon LLM, Leung CSW, Tashiro M, Chan KH, Wong BWY, Yuen KY et al, Rapid detection of severe acute respiratory syndrome (SARS) coronavirus by a loop‐mediated isothermal amplification assay. Clin Chem 50: 1050–1052 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Woo PCY, Lau SKP, Wong BHL, Tsoi HW, Fung AMY, Chan KH et al, Detection of specific antibodies to severe acute respiratory syndrome (SARS) coronavirus nucleocapsid protein for serodiagnosis of SARS coronavirus pneumonia. J Clin Microbiol 42: 2306–2309 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Lau SK, Woo PC, Wong BH, Tsoi HW, Woo GK, Poon RW et al, Detection of SARS coronavirus nucleocapsid protein in SARS patients by enzyme‐linked immunosorbent assay. J Clin Microbiol 42: 2884–2889 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Song S, Wang L, Li J, Zhao J and Fan C, Aptamer‐based biosensors. Trends in Anal Chem 27: 108–117 (2008). [Google Scholar]
- 21. Cook PD, Medicinal chemistry of oligonucleotides in Antisense Drug Technology: Principles, Strategies and Applications, ed by Crooke ST. Marcel Dekker, pp. 9–58 (2001). [Google Scholar]
- 22. Cload ST, McCauley TG, Keefe AD, Healy JM and Wilson C, Properties of therapeutic aptamers in The Aptamer Handbook, ed by Klussmann S. Wiley–VCH Verlag GmbH & Co. KGaA, pp. 363–416 (2006). [Google Scholar]
- 23. Ellington AD and Szostak JW, In vitro selection of RNA molecules that bind specific ligands. Nature 346: 818–822 (1990). [DOI] [PubMed] [Google Scholar]
- 24. Iliuk AB, Hu L and Tao WA, Aptamer in bioanalytical applications. Anal Chem 83: 4440–4452 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Smith AM, Duan H, Mohs AM and Nie S, Bioconjugated quantum dots for in vivo molecular and cellular imaging. Adv Drug Deliv Rev 60: 1226–1240 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Yu WW, Chang E, Drezek R and Colvin VL, Water‐soluble quantum dots for biomedical applications. Biochem Biophys Res Comm 348: 781–786 (2006). [DOI] [PubMed] [Google Scholar]
- 27. Smith AM and Nie S, Next‐generation quantum dots. Nat Biotechnol 27: 732–733 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Ahn DG, Jeon IJ, Kim JD, Song MS, Han SR, Lee SW et al, RNA aptamer‐based sensitive detection of SARS coronavirus nucleocapsid protein. Analyst 134: 1896–1901 (2009). [DOI] [PubMed] [Google Scholar]
- 29. Laemmli UK, Cleavage of structural proteins during the assembly of the head of Bacteriophage T4. Nature 227: 680–685 (1970). [DOI] [PubMed] [Google Scholar]
- 30. Bradford M, A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein‐dye binding. Anal Biochem 72: 248–254 (1976). [DOI] [PubMed] [Google Scholar]
- 31. Lee Y, Lee EK, Cho YW, Matsui T, Kang IC, Kim TS et al, ProteoChip: a highly sensitive protein microarray prepared by a novel method of protein immobilization for application of protein‐protein interaction studies. Proteomics 3: 2289–2304 (2003). [DOI] [PubMed] [Google Scholar]
- 32. Jacobson AB and Zuker M, Structural analysis by energy dot plot of a large mRNA. J Mol Biol 233: 261–269 (1993). [DOI] [PubMed] [Google Scholar]


