Summary
Beginning in the fall of 2010, an increasing and alarming number of cases of calves suffering from liver dystrophy were reported in the south of Germany. An epidemiological investigation was carried out by the authors between November 2010 and July 2011, leading to the implication of a commercial dietary supplement as the potential cause for this outbreak. The components of this product were first tested in a cell culture model and two of them (dietary chestnut extract and glycerol monolaurate) showed a cytotoxic effect. The objective of this study was therefore to evaluate the effect of supplemental feeding of both components alone or in combination on liver function in newborn calves on a commercial dairy farm. Ten calves were enrolled in each of the three treatment groups and the control group (group O) following a blocked design. Treatment consisted of supplementation with chestnut extract at 0.02% of birth body mass (BM) (group C), supplementation with glycerol monolaurate at 0.006% of BM (group G) or a combined treatment (group CG) for five consecutive days. The effect of treatments on liver function was evaluated clinically and by measurement of glutamate dehydrogenase (GLDH) and aspartate aminotransferase (AST) activities as well as the determination of the concentrations of glucose, L‐lactate and total bilirubin in serum. There was a significant increase in GLDH and AST activities and a significant decrease in glucose concentration in treatment groups C and CG compared with the control group (p ≤ 0.035), whereas no difference was shown for group G. Survival was significantly decreased in groups C (p = 0.029) and CG (p = 0.001) compared with both group G and the control group. These results suggest that dietary chestnut extract in an amount of 0.02% of BM alone or in combination has a toxic effect on liver function in newborn calves.
Keywords: calf, liver dystrophy, tannin, intoxication
Introduction
An unusually high number of cases of jaundice in young calves were reported in Southern Germany beginning in the fall of 2010. The results of an epidemiological survey carried out in Bavaria between November 2010 and July 2011, followed by a preliminary feeding trial carried out by the authors suggested a causal involvement of a commercially available dietary supplement used for prevention and treatment of diarrhoea in neonatal calves (Metzner et al., 2012). Detailed information about the epidemiological investigations and the preliminary feeding trial with this dietary supplement can be found elsewhere (Metzner et al., 2012). The investigation of acetonitrile–water extracts of each component of the dietary supplemental feed in the MTT assay according to Reubel et al. (1989) [reduction in a yellow tetrazolium salt to violet formazan by cellular enzymes using human epidermoid cancer cells (Hep2‐cells)] revealed a cytotoxic effect of two of the 12 tested components (chestnut extract and glycerol monolaurate). Chestnut extract is obtained by water extraction of the chestnut wood (Castanea sativa) and is mainly composed of tannins, lignin, cellulose, hemicellulose and mineral salt (Graziani and Tosi, 2006). Tannins can be classified into two categories: hydrolysable and non‐hydrolysable (condensed) tannins. Both hydrolysable and condensed tannins exist in sweet chestnut wood, but hydrolysable tannins predominate (Anadón et al., 2005).
As the use of antimicrobial feed additives has been restricted further in the European Union (EU) in 2006, alternative approaches were sought to control or prevent disease and improve growth performance in farm animals. Interest in feed ingredients containing tannins has increased as their potential use as growth promoters in animal feed has been described in the literature (Hooge et al., 2012). The antimicrobial properties (Graziani and Tosi, 2006) of tannins and the resultant beneficial effects on body weight gain and reduction in mortality are documented (Hooge et al., 2012). Furthermore, tannins are suggested to be good candidates in the control of gastrointestinal parasites in ruminants (Min et al., 2005) and broiler chicks (McCann et al., 2006) as well as certain avian virus infections such as avian reovirus and avian metapneumovirus (Lupini et al., 2009).
Paradoxically, tannins can produce dose‐dependent antinutritional effects at the same time. Fed at a higher dose, tannins are reported to cause growth depression, a reduction in feed intake and digestibility in monogastric mammals (Smulikowska et al., 2001) and ruminants (McSweeney et al., 2001).
Glycerol monolaurate is the esterified form of lauric acid (Klevenhusen et al., 2011) and a naturally occurring monoglyceride that is generally recognised as safe for ingestion and has been used extensively in the food industry (Strandberg et al., 2010). Recent in vitro studies documented the effect of glycerol monolaurate as an effective growth inhibitor of Staphylococcus aureus (Kelsey et al., 2006), Candida species (Strandberg et al., 2010) and virulence factor producing Bacillus anthracis (Vetter and Schlievert, 2005).
Recent research has shown that feeding chestnut extract in a concentration of 0.007% and 0.01% of body mass improved average daily gain in feedlot cattle (Barajas et al., 2013; Cervantes et al., 2013), and at a concentration of 0.015% (Liu et al., 2013) had no negative effect on liver function and general condition in transition dairy cows. To the best of our knowledge, there is, however, a lack of information about the effect of chestnut extract as well as glycerol monolaurate on the liver function in newborn (non‐ruminating) calves.
The objective of this study was to investigate whether feeding either one or a combination of the components identified as cytotoxic by MTT assay (chestnut extract and glycerol monolaurate) in a concentration corresponding to that of the commercial product as described by Metzner et al. (2012) would provoke liver disease in calves.
Material and methods
This controlled clinical trial was conducted during the months of March and April 2012 on a commercial dairy farm milking 2500 cows in the southwest of Bavaria.
Animals
All calves born to heifers and cows during the month of March 2012 were eligible for enrolment. A total of 40 newborn calves [30 Holstein Friesian (18 male/12 female), two Brown Swiss (female), eight cross‐bred calves (Holstein Friesian × Belgian Blue, four male/four female)] were separated from their dams immediately after birth, weighed and moved into individual calf hutches (Flixbox calf igloos; Mayer, Traunstein, Germany) with concrete flooring and straw bedding. The first colostrum intake was between 1 and 5 h after birth (day 0). All calves received three litres of raw colostrum from their dams, followed by whole milk in the amount of 12% of birth body mass (BM) divided into three daily feedings (6 am, 12 and 6 pm) offered in nipple buckets.
Study design
The order of enrolment in the control and three treatment groups as well as the allocation of the first calf were determined at random. Enrolment thereafter was sequential in blocks of four (10 calves in each group). The treatment consisted of supplementation of the daily ration with chestnut extract (Alce s.r.l., Bagni di Lucca, Italy; Lot no. 26101) at 0.02% of BM in group C (n = 10), supplementation with glycerol monolaurate (BASF, Monheim, Germany, Batch no: GR03490020) at 0.006% of BM in group G (n = 10), supplementation with chestnut extract at 0.02% of BM plus glycerol monolaurate at 0.006% of BM in group CG (n = 10), whereas calves of group O did not receive any supplementation and served as controls (n = 10). The calculated daily amounts of supplement were divided into three equal amounts and mixed into the milk immediately before each feeding. Supplements were fed for a total of 5 days (days 2–6). All calves were observed for a period of 13 days.
Sample size determination was based on the following assumptions: In a preliminary feeding trial (Metzner et al., 2012), liver disease could be observed in four of four experimental calves by feeding the dietary supplemental feed. As the objective of this study was to investigate whether liver disease could be caused by feeding one of the two components alone (chestnut extract, glycerol monolaurate) or the combination of both, the null hypothesis was defined as all treatments being unable to cause liver disease singly or in combination.
Sample size determination was based on a power of 0.80 and a significance level of 0.05 which was subsequently adjusted for familywise error rate with Bonferroni correction (0.05/3), assuming the disease would be caused in at least 70% of the calves, if the component was causal. The resulting number of ten calves per group was inflated by a factor of 0.2 because of an assumed attrition rate of 20%.
Censoring
Animals refusing the supplemented milk for three consecutive feedings were excluded from the study, and their data were censored.
Clinical examination
Daily clinical examination using a standardised score sheet was carried out after morning feeding by the same trained veterinarian who was not blinded to treatment group. Parameters evaluated by the scoring system included suckling reflex, behaviour, posture, degree of dehydration and central nervous system (Table 1). Discolouration of the sclera and oral mucosa, and the consistency of the faeces were recorded. A decrease in suckling reflex and abnormal behaviour was defined as a score of 4 and higher, whereas abnormal posture as well as dehydration was defined as a score of 2 and above. A score of 1 and higher regarding examination of the nervous system was considered abnormal. Rectal temperature was measured at the same time every day with a digital thermometer (8 am).
Table 1.
Score sheet for categorisation of general condition
| Suckling reflex | 0 | Vigorous |
| 1 | Strong | |
| 2 | Strong but intermittent | |
| 3 | Weak | |
| 4 | Weak and intermittent | |
| 5 | Absent or chewing movements | |
| Behaviour | 0 | Adequate reaction to acoustic and optical stimuli, bright and alert |
| 1 | Slightly delayed reaction to acoustic and optical stimuli, but alert | |
| 2 | Delayed reaction to acoustic and optical stimuli, dull | |
| 3 | Delayed reaction to acoustic and optical stimuli, apathetic | |
| 4 | Calf reacts only to painful stimuli | |
| 5 | No reaction to painful stimuli | |
| Posture | 0 | Upright posture, straight back, upright head carriage, normal position of the ears |
| 1 | Straight back line, upright head carriage, slight drooping of ears | |
| 2 | Arched back, lowered head, drooping of ears | |
| 3 | Standing unsteadily after lifting, arched back, lowered, head drooping of ears | |
| 4 | Sternal recumbency, lowered head, drooping of ears | |
| 5 | Unable to stand, lateral recumbency | |
| Degree of dehydration | 0 | Normal skin tent, no space between the eyelids and the eyeball |
| 1 | Decreased skin tent, no space between the eyelids and the eyeball | |
| 2 | Decreased skin tent, small gap (2 mm) between the eyelids and the eyeball | |
| 3 | Decreased skin tent, moderate gap (4 mm) between the eyelids and the eyeball | |
| 4 | Decreased skin tent, large gap (7 mm) between the eyelids and the eyeball | |
| Central nervous symptoms | 0 | None |
| 1 | Partial seizures, tenesmus | |
| 2 | Generalised tonic–clonic seizures, bawling, vehement tenesmus, opisthotonus |
This article is being made freely available through PubMed Central as part of the COVID-19 public health emergency response. It can be used for unrestricted research re-use and analysis in any form or by any means with acknowledgement of the original source, for the duration of the public health emergency.
Treatment
Calves were treated with an anti‐infective (marbofloxacin, 2 mg/kg BM, total of 5 days) and anti‐inflammatory (flunixin meglumine, 1.1 mg/kg BM, total of 3 days) drug in the case of omphalitis and/or bronchopneumonia as diagnosed by the investigator. In cases of diarrhoea (faecal consistency score of soupy/runny), a standardised oral rehydration solution was given. Intravenous (IV) fluids containing 5 l of a 0.15 m (0.9%) sodium chloride solution were administered in cases of severe dehydration as described in a prospective study (Niethammer, 2007). IV fluids containing sodium bicarbonate were administered according to guidelines described by Lorenz and Lorch (2010). Calves were euthanised if they either suffered from severe disturbance of the general condition (>16 points on the score sheet with lack of response to treatment within 24 h or a maximum score of 21 points at any time). All calves that died or were euthanised during the study period were submitted for post‐mortem examination to the Bavarian Health and Food Safety Authority (Oberschleissheim). Surviving male calves were sold 1 week after the end of the study period, whereas all female calves were kept on the farm and raised as replacements.
Samples and analytical methods
Blood samples were collected daily immediately after the morning feeding from the jugular vein by venipuncture with an 18‐gauge (1.2 × 40 mm) needle on days 0–12 and placed directly into S‐Monovette® tubes (Sarstedt, Nümbrecht‐Rommelsdorf, Germany).
Blood samples were centrifuged within 2 h after sampling at 1892 g for 10 min, and serum was stored at −20 °C until analysis. All parameters including glutamate dehydrogenase (GLDH, U/l), aspartate aminotransferase (AST, U/l), glucose (mm), L‐lactate (mm) and total bilirubin (μ m) were determined using an automatic analysing system (Automatic Analyzer Hitachi 911; Roche Diagnostics, Indianapolis, IN, USA).
Faecal samples of all calves were collected between day 5 and day 12 directly from the rectum of each animal with a clean glove and kept refrigerated. Faeces were transported cooled to the laboratory of Comparative Tropical Medicine and Parasitology, University of Munich, Germany, and processed within 1 day after collection. Samples were screened for the presence of Cryptosporidium species, Escherichia coli F5 (K99), rota‐ and coronavirus using an enzyme‐linked immunoassay (BIO K 348 – Digestive kit; Bio‐X Diagnostics, Jemelle, Belgium).
All observations and interventions were carried out by the same investigator according to study guidelines that were approved before the start of the trial by the regional government of Upper Bavaria, Germany (Ref. 55.2‐1‐54‐2532‐133‐11).
Statistical analysis
Data were analysed using spss (version 20; IBM SPSS Statistics, Armonk, NY, USA). As the assumption of normality was not met, nonparametric tests were applied. Differences between the four groups were assessed using the Kruskal–Wallis test. Mann–Whitney U‐tests were used for comparison of blood values between the groups, using a Bonferroni correction. For comparison of cumulative survival between the three treatment groups and the control group, log‐rank test was used.
Results
The calves remained in the study for an average of 11.4 days. Baseline characteristics of all calves are depicted in Table 2.
Table 2.
Characteristics of experimental calves at time of enrolment
| Gender | Birth body weight (mean; kg) | Breed | ||||
|---|---|---|---|---|---|---|
| Male | Female | Holstein | Cross‐breed | Brown Swiss | ||
| Group | ||||||
| C | 4 | 6 | 37.3 | 5 | 3 | 2 |
| CG | 7 | 3 | 39.5 | 9 | 1 | 0 |
| G | 4 | 6 | 38.5 | 8 | 2 | 0 |
| O | 7 | 3 | 43.4 | 8 | 2 | 0 |
This article is being made freely available through PubMed Central as part of the COVID-19 public health emergency response. It can be used for unrestricted research re-use and analysis in any form or by any means with acknowledgement of the original source, for the duration of the public health emergency.
Total survival experience
During the 13 days of the trial, one calf in group C and another calf in group CG died peracutely. Another three calves in group C, as well as four calves in group CG, were euthanised according to the guidelines. Figure 1 displays the survival curves of the treatment and control calves. On day 2, three calves of group CG were excluded from the study, and their data censored because they refused the supplemented milk for three consecutive feedings. Survival was different between groups C and O (p = 0.029), as well as between groups CG and O (p = 0.001).
Figure 1.
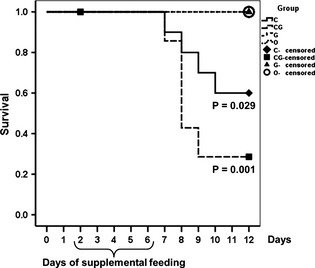
Survival curves. For demonstration of statistical significance between treatment groups and control group, p‐values are displayed below the respective survival curves. Censoring on day 2 is due to withdrawal of three calves of group CG.
Laboratory data
As depicted in Fig. 2, activity of GLDH was significantly higher in group C (day 6–12), as well as in group CG (day 7–10), compared with the control group (p ≤ 0.035). AST activity was significantly higher than in control calves on days 5, 7, 8, 9, 10, 11 in group C (p ≤ 0.009) and on day 8 in group CG (p = 0.007, Fig. 3).
Figure 2.
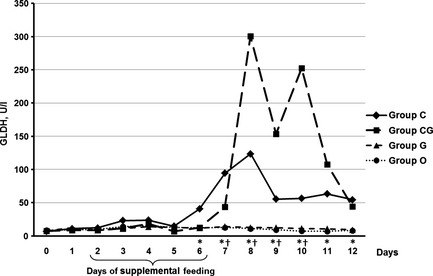
Median of GLDH (U/l) in calves fed for five consecutive days (2–6) with chestnut extract (group C), chestnut extract plus glycerol monolaurate (group CG), glycerol monolaurate (group G) and control calves (group O). Statistically significant differences between groups C and O are indicated by * and between groups CG and O by †.
Figure 3.
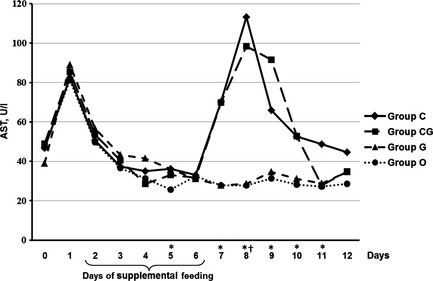
Median of AST (U/l) in calves fed for five consecutive days (2–6) with chestnut extract (group C), chestnut extract plus glycerol monolaurate (group CG), glycerol monolaurate (group G) and control calves (group O). Statistically significant differences between group C and O are indicated by * and between group CG and O by †.
There was an increase in the concentration of total bilirubin on day 1 in all four groups (Fig. 4). On days 9 and 10, concentrations of total bilirubin were significantly (p ≤ 0.022) higher in group C compared with group O. The comparison between group CG and control group revealed no statistically significant differences in concentration of total bilirubin at any time; however, there was a moderate increase in group CG on days 8 and 9.
Figure 4.
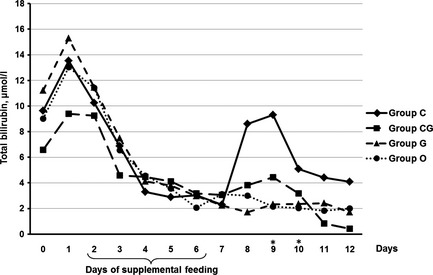
Median of total bilirubin (μ m) in calves fed for five consecutive days (2–6) with chestnut extract (group C), chestnut extract plus glycerol monolaurate (group CG), glycerol monolaurate (group G) and control calves (group O). Statistically significant differences between groups C and O are indicated by *.
Glucose concentrations were significantly lower in group C on day 7 (p = 0.002), as well as on day 8 and 12 in group CG (p ≤ 0.049) compared with the control group (Fig. 5).
Figure 5.
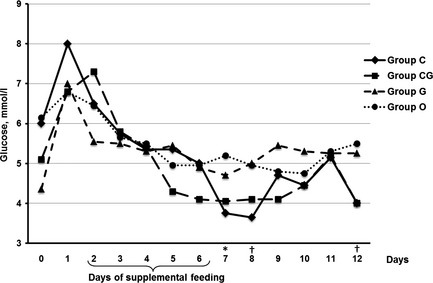
Median of glucose concentration (mm) in calves fed for five consecutive days (2–6) with chestnut extract (group C), chestnut extract plus glycerol monolaurate (group CG), glycerol monolaurate (group G) and control calves (group O). Statistically significant differences between groups C and O are indicated by * and between groups CG and O by †.
Significant differences in GLDH and AST activities, and total bilirubin and glucose concentrations were not detected at any point between group G and the control group (Figs 2, 3, 4 and 5).
The median L‐lactate concentration in all four groups was above the respective reference range of 2.5 mm for the majority of sampling days (Fig. 6). There was a significant difference in concentrations of L‐lactate on day 7 between all three treatment groups and the control group (p ≤ 0.035), as well as between groups C and O on day 8 (p = 0.008).
Figure 6.
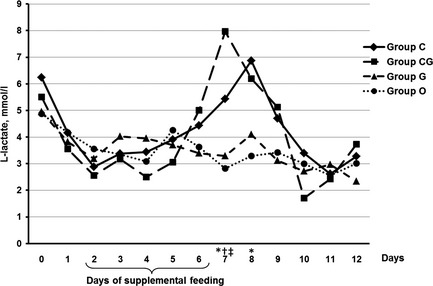
Median of L‐lactate concentration (mm) in calves fed for five consecutive days (2–6) with chestnut extract (group C), chestnut extract plus glycerol monolaurate (group CG), glycerol monolaurate (group G) and control calves (group O). Statistically significant differences between groups C and O are indicated by *, between groups CG and O by †, and between groups G and O by ‡.
Clinical findings
Abnormal posture (score 2, Table 1) was recorded in three (group C), two (group CG) and four cases (group G) across the 13 days of the trial, whereas lateral recumbency (score 5, Table 1) was detected in three animals in group C and another four calves in group CG between days 7 and 9.
Abnormal behaviour was detected in two calves in group C and another five calves in group CG between days 6 and 9.
Involvement of the central nervous system between days 7 and 9 was documented in two calves in group C and another three calves in group CG.
Dehydration was recorded in one calf in group C and another two calves in group CG during the 13 days of the trial.
A decrease in suckling reflex was observed in ten cases in group C, nine calves in group CG and another two calves in group G during the 13 days of the trial.
Between days 5 and 9, severe hypothermia (rectal temperature ≤37.5 °C) was recorded in three calves in group C, five calves in group CG and another two calves in group G.
In four calves of group C, two calves of group G and another calf of group O, rectal temperature exceeded 39.5 °C. In three cases (two calves of group C and another calf of group O), fever was detected concurrently with pneumonia (2) and omphalitis (1). Omphalitis was diagnosed in two calves of group C and O and another calf of group CG and G. Pneumonia was recorded in two calves of group C and one calf in each of the groups CG, G and O.
None of the calves showed any obvious yellow discolouration of the mucosa. A dark grey discolouration of the gingiva around the incisors between days 4 and 12 was recorded in seven calves in group C and another six calves in group CG, whereas no alteration of this kind was detected in any calf of groups G or O.
Faeces were reddish brown in all calves of groups C and CG between days 3 and 10 for up to 6 days, whereas no discolouration was detected in any calf of groups G and O.
All calves suffered from diarrhoea for up to 9 days (average 4.7 days). Table 3 lists the results of the investigation of faecal samples.
Table 3.
Results of faecal examination of experimental calves between days 5 and 12 using an enzyme‐linked immunoassay (BIO K 348 – Digestive kit; Bio‐X Diagnostics)
| Pathogen | ||||
|---|---|---|---|---|
| Escherichia coli F5 (K99) | Rotavirus | Coronavirus | Cryptosporidium | |
| Group | ||||
| C (n = 10) | 1 | 3 | 0 | 9 |
| CG (n = 7) | 0 | 3 | 1 | 5 |
| G (n = 10) | 0 | 4 | 0 | 9 |
| O (n = 10) | 0 | 3 | 1 | 9 |
This article is being made freely available through PubMed Central as part of the COVID-19 public health emergency response. It can be used for unrestricted research re-use and analysis in any form or by any means with acknowledgement of the original source, for the duration of the public health emergency.
Gross pathology and histopathology
Nine calves (four calves of group C and another five calves of group CG) were necropsied at the Bavarian Health and Food Safety Authority, and any organ showing obvious macroscopic changes was examined histopathologically.
Findings in calves of group C comprised of single‐cell necrosis with mild multifocal periacinar fatty liver cell degeneration (n = 1), moderate fatty liver cell degeneration with periportal mononuclear infiltrates (n = 1) and severe liver cell necrosis (n = 2, combined with proliferation of bile duct epithelial cells in the periportal zone in one case). In group CG, the changes in the liver varied from single‐cell necrosis with mild multifocal periacinar fatty liver cell degeneration (n = 2) to severe liver cell necrosis (n = 3), once combined with proliferation of bile duct epithelial cells in the periportal zone.
Further macroscopic findings included seven cases of catarrhal enteritis (group CG n = 3, group C n = 4) and volvulus of the small intestine and partial caecal necrosis in one calf in group CG. Of those cases with enteritis, five tested positive for rotavirus and/or Cryptosporidium parvum and in one case for bovine viral diarrhoea virus. Furthermore, presence of several abomasal ulcers was documented in one calf (group CG).
Discussion
The aim of this study was to investigate the influence of dietary chestnut extract and glycerol monolaurate on liver function in newborn calves on the basis of clinical and laboratory findings. GLDH is considered to be a liver‐specific enzyme and effective biomarker of acute hepatic injury with activity in hepatocytes ten times higher than in other tissues. It is released in cases of hepatocellular necrosis (Thomas, 2005) and therefore of great value for indication of liver disease in adult cattle as well of liver dystrophy in calves (Hunter et al., 2013). The significant increase in serum GLDH activity in calves from group C and CG indicates acute hepatic injury.
Despite the fact that elevation of AST may be due to conditions not involving the liver, it is considered to be a sensitive biomarker of liver cell injury in cattle (Sattler and Fuerll, 2004). The significant difference of activity of AST between groups C and O on days 5, 7, 8, 9, 10, 11, as well as between groups CG and O on day 8 indicates hepatocellular damage in the two treatment groups that included feeding of chestnut extract. Although total serum bilirubin is not considered to be a sensitive indicator of hepatic dysfunction (Pratt and Kaplan, 2007), elevation of total bilirubin concentration is frequently documented in cases of hepatocellular necrosis in cattle (Raposo et al., 1998). Hence, the significant increase in concentration of total bilirubin in group C on day 9 and 10 indicates a liver dysfunction which supports the evidence that chestnut extract alone or in combination might be the causal factor.
Elevated concentration of L‐lactate within the first 48 h of life followed by a decrease as shown in Fig. 5 has been documented in cattle by several authors (Egli and Blum, 1998). According to Steinhardt et al. (1995), elevation of L‐lactate concentration can be a consequence of an increase in production due to hypoxia and enhanced sympathetic activity due to stress around the time of birth. More importantly, elevation of concentration of L‐lactate is known to be a result of inhibition of mitochondrial function by certain drugs which impair both beta‐oxidation and the respiratory chain enzymes, and is a hallmark of mitochondrial impairment (Scatena et al., 2007) and hepatocellular dysfunction (Pratt and Kaplan, 2007). Elevation of the concentration of L‐lactate is also seen in severely dehydrated calves suffering from acute diarrhoea (Tennant et al., 1968). We cannot determine with certainty whether the increase in concentration of L‐lactate in treatment groups C and CG was a direct consequence of hepatic failure or if it was due to dehydration as a consequence of acute diarrhoea. However, as only three calves were severely dehydrated, it seems likely that the substantial increase in the concentration of L‐lactate in group C and CG was a direct consequence of hepatocellular dysfunction. Neonatal diarrhoea can lead to abnormal posture and behaviour in calves. In several cases, this may be due to septicaemia (Fecteau et al., 1997) or D‐lactataemia (Lorenz, 2004). However, similar abnormalities have also been documented in cases of hepatic encephalopathy (Buczinski et al., 2007). As all calves in this study suffered from diarrhoea to some extent, the cause for abnormal posture and behaviour scores cannot be determined. Abnormalities in the central nervous system of calves can be due to hypoglycaemia (Leifer and Peterson, 1984) as well as hepatic failure among other reasons (Mayhew, 2009). Several authors highlight the effect of low ambient temperature and the resulting hypothermia on glucose level in newborn calves (Diesch et al., 2004). Temperatures during the study period ranged from −8.5 to +18.8 °C, with lower temperatures possibly resulting in an excessive cold stress and a more rapid depletion of carbohydrate reserves (Todd et al., 2000). Hypoglycaemia as depicted in Fig. 6 in calves of groups C and CG may be due to loss of hepatic mass and a consecutive failure of gluconeogenesis (Scatena et al., 2007).
The dark grey discolouration of the gingiva around the incisors as well as the reddish brown discolouration of the manure are probably a direct effect of the colouring properties of chestnut extract as described by Liukkonen‐Anttila et al. (2001).
It cannot be determined from this study to which extent depression of the suckling reflex was due to D‐lactataemia (Lorenz, 2004) or due to hepatic dysfunction. Although decrease in the suckling reflex could have been due to diarrhoea associated with D‐lactataemia, we speculate that hepatic failure was a major cause of disturbance of the general condition in the two calves of group C and the five calves of group CG with scores 4 and 5 respectively. Even though clinical examination was carried out according to a standardised protocol, the lack of blinding might have led to the introduction of an undetermined degree of observer bias.
The significant increases in GLDH and AST activities, and total bilirubin concentration, as well as the significant decrease in glucose concentration in groups C and CG compared with group O indicate a hepatotoxic effect of both chestnut extract and the combination of chestnut extract and glycerol monolaurate. By contrast, changes in those parameters could not be seen by feeding glycerol monolaurate alone.
When considering the chemical composition of chestnut extract, this hepatotoxic effect was likely due to its high content of tannins, albeit the toxic principles of tannins have not been fully ascertained (Radostits et al., 2007). Toxic effects associated with tannins differ between species. Monogastric animals predominantly develop liver and gastrointestinal lesions (Zhu et al., 1992), whereas in ruminants, renal damage predominates (Zhu et al., 1995). This was not confirmed in this study. This may have been due to the functional monogastric status of newborn calves and a consecutive absence of transformation of tannins in the rumen and a lack of nephrotoxic metabolites (Zhu et al., 1995).
The clinical course and severity of disease as well as mortality in this study were unexpected as the experimental calves in the preliminary feeding trial showed less pronounced abnormalities (Metzner et al., 2012). Yet the findings of this study are strikingly similar to descriptions and findings of those calves described in the preceding epidemiological investigation of commercial dairy farms carried out between November 2010 and July 2011. Several causes have to be discussed when trying to explain this difference between both feeding trials. In contrast to the preliminary feeding trial under hospital conditions, the present study was carried out under field conditions on a commercial dairy farm. We assume that differences due to location, management and pathogen load played a substantial role and were more consistent with conditions described in the preliminary study. Despite the fact that each calf received chestnut extract according to its individual body weight, some calves recovered from clinical disease or were only affected subclinically (showing abnormal blood parameters only), while others died or had to be euthanised. Concurrent infections (diarrhoea, pneumonia, etc.) could have influenced the development and severity of the disease caused by the treatment. It has to be mentioned that none of the experimental calves in the preliminary feeding trial suffered from diarrhoea, whereas in the present study, all experimental calves did so to some extent during the 13 days of the trial. Apart from this, temperatures during the preliminary trial (May and June, 2011) were between +0.4 and +33.9 °C and therefore decreased the risk of hypothermia in newborns. The striking difference concerning the clinical outcome in the present study in comparison with the experimental calves in the preliminary feeding trial as reported by Metzner et al. (2012) may also have been due to the fact that feeding a single component (chestnut extract) or two components (chestnut extract plus glycerol monolaurate) had a stronger impact on liver function compared with feeding the complete feed supplement. We cannot rule out that the hepatotoxic effect of chestnut extract as shown in this study was attenuated by one or more components in the commercial product. The question whether there is a potentiation in the hepatotoxic effect when both components are fed at the same time could not fully resolved due to an unexpectedly high mortality leading to a premature termination of enrolment according to the stopping guidelines defined prior to the start of the study.
In conclusion, supplementation of both chestnut extract at 0.02% of BM and the combination of chestnut extract at 0.02% of BM and glycerol monolaurate at 0.006% of BM have a major impact on liver function in newborn calves. Further investigations have to be carried out to investigate the toxic principle and predisposing factors leading to the harmful effect of chestnut extract alone or in combination before this supplement can be considered as a safe feed ingredient for newborn calves. Further evaluation of possible beneficial effects should be proceeded only after this can be guaranteed.
Acknowledgements
We greatly acknowledge the technical assistance of Ingrid Hartmann, Monika Altmann and Christina Beyer, the work of the laboratory technicians at the Bavarian Authority for Health and Food Safety and of Monika Held at the Institute of Technical Hygiene. We also acknowledge the assistance of Miriam Scheuerle at the Institute of Comparative Tropical Medicine and Parasitology. We would like to thank Wolfgang Klee and Sabine Mann for the critical review of the manuscript. We are grateful to the farm owner for the provision of the technical support and for providing the calves. The study was financially supported by Josera Tierernährung, Germany, and Alce s.r.l., Bagni di Lucca, Italy.
References
- Anadón, A. ; Arzo, M. A. ; Bories, G. ; Brantom, P. ; Brufau de Barbera, J. ; Chesson, A. ; Cocconcelli, P. S. ; de Knecht, J. ; Dierick, N. ; Flachowsky, G. ; Franklin, A. ; Gropp, J. ; Lundebye Haldorsen, A.‐K. ; Halle, I. ; Mantovani, A. ; Peltonen, K. ; Rychen, G. ; Sanders, P. ; Soares, A. ; Wester, P. ; Windisch, W. , 2005: Opinion of the scientific panel on additives and products or substances used in animal feed on a request from the commission on the safety and efficacy of the product Farmatan for rabbits and piglets. The EFSA Journal 222, 1–20. [Google Scholar]
- Barajas, R. ; Cervantes, B. J. ; Espino, M. A. ; Camacho, A. ; Verdugo, M. ; Flores, L. R. ; Romo, J. A. , 2013: Interaction of tannin extract and zilpaterol hydrochloride supplementation on feedlot performance of bulls. Journal of Animal Science 91 (E‐Suppl. 2), 8. [Google Scholar]
- Buczinski, S. ; Duval, J. ; D'Anjou, M. A. ; Francoz, D. ; Fecteau, G. , 2007: Portacaval shunt in a calf: clinical, pathologic, and ultrasonographic findings. The Canadian Veterinary Journal 48, 407–410. [PMC free article] [PubMed] [Google Scholar]
- Cervantes, B. J. ; Camacho, A. ; Vazquez, J. A. ; Espino, M. A. ; Heras, T. J. ; Flores, L. R. ; Lomeli, J. J. ; Barajas, R. , 2013: Influence of tannins extract supplementation on feedlot performance and plasma urea nitrogen of non implanted growing heifers. Journal of Animal Science 91 (E‐Suppl. 2), 7. [Google Scholar]
- Diesch, T. J. ; Mellor, D. J. ; Stafford, K. J. ; Ward, R. N. , 2004: The physiological and physical status of single calves at birth in a dairy herd in New Zealand. New Zealand Veterinary Journal 52, 250–255. [DOI] [PubMed] [Google Scholar]
- Egli, C. P. ; Blum, J. W. , 1998: Clinical, haematological, metabolic and endocrine traits during the first three months of life of suckling simmentaler calves held in a cow‐calf operation. Zentralblatt für Veterinärmedizin Reihe A 45, 99–118. [DOI] [PubMed] [Google Scholar]
- Fecteau, G. ; Pare, J. ; Van Metre, D. C. ; Smith, B. P. ; Holmberg, C. A. ; Guterbock, W. ; Jang, S. , 1997: Use of a clinical sepsis score for predicting bacteremia in neonatal dairy calves on a calf rearing farm. The Canadian Veterinary Journal 38, 101–104. [PMC free article] [PubMed] [Google Scholar]
- Graziani, R. ; Tosi, G. , 2006: In vitro antimicrobial activity of SILVA FEED ENC® tannin on bacterial strains of poultry origin. World's Poultry Science Journal 62, 384–385. [Google Scholar]
- Hooge, D. M. ; Mathis, G. F. ; Lumpkins, B. ; Ponebsek, J. ; Moran, D. , 2012: Dose‐responses of broiler chicks, given live coccidia vaccine on day of hatch, to diets supplemented with various levels of Farmatan® (sweet chestnut wood tannins) or BMD®/Stafac® in a 42‐day pen trial on built‐up litter. International Journal of Poultry Science 11, 474–481. [Google Scholar]
- Hunter, A. G. ; Suttle, N. ; Martineau, H. M. ; Spence, M. A. ; Thomson, J. R. ; Macrae, A. I. ; Brown, S. , 2013: Mortality, hepatopathy and liver copper concentrations in artificially reared Jersey calves before and after reductions in copper supplementation. The Veterinary Record 172, 46. [DOI] [PubMed] [Google Scholar]
- Kelsey, J. A. ; Bayles, K. W. ; Shafii, B. ; McGuire, M. A. , 2006: Fatty acids and monoacylglycerols inhibit growth of Staphylococcus aureus . Lipids 41, 951–961. [DOI] [PubMed] [Google Scholar]
- Klevenhusen, F. ; Meile, L. ; Kreuzer, M. ; Soliva, C. R. , 2011: Effects of monolaurin on ruminal methanogens and selected bacterial species from cattle, as determined with the rumen simulation technique. Anaerobe 17, 232–238. [DOI] [PubMed] [Google Scholar]
- Leifer, C. E. ; Peterson, M. E. , 1984: Hypoglycemia. The Veterinary Clinics of North America Small Animal Practice 14, 873–889. [DOI] [PubMed] [Google Scholar]
- Liu, H. W. ; Zhou, D. W. ; Li, K. , 2013: Effects of chestnut tannins on performance and antioxidative status of transition dairy cows. Journal of Dairy Science 96, 5901–5907. [DOI] [PubMed] [Google Scholar]
- Liukkonen‐Anttila, T. ; Kentala, A. ; Hissa, R. , 2001: Tannins ‐ a dietary problem for hand‐reared grey partridge Perdix perdix after release?. Comparative Biochemistry and Physiology. Toxicology & Pharmacology 130, 237–248. [DOI] [PubMed] [Google Scholar]
- Lorenz, I. , 2004: Investigations on the influence of serum D‐lactate levels on clinical signs in calves with metabolic acidosis. The Veterinary Journal 168, 323–327. [DOI] [PubMed] [Google Scholar]
- Lorenz, I. ; Lorch, A. , 2010: Klinische Erscheinungen der Azidose beim Durchfallkalb. Neubetrachtung unter dem Gesichtspunkt der D‐Laktatazidose. In: Müller K. E. (ed.) Proceedings of the 8 Berlin‐Brandenburgischer Rindertag, 2010 October 7–9. Cuvillier Verlag Göttingen, Germany, pp. 51–53 [Google Scholar]
- Lupini, C. ; Cecchinato, M. ; Scagliarini, A. ; Graziani, R. ; Catelli, E. , 2009: In vitro antiviral activity of chestnut and quebracho woods extracts against avian reovirus and metapneumovirus. Research in Veterinary Science 87, 482–487. [DOI] [PubMed] [Google Scholar]
- Mayhew, I. G. , 2009: Large Animal Neurology, 2nd edn Wiley‐Blackwell Publishing, Ames, IA. [Google Scholar]
- McCann, M. E. E. ; Newell, E. ; Preston, C. ; Forbes, K. , 2006: The use of mannan‐oligosaccharides and/or tannin in broiler diets. International Journal of Poultry Science 5, 873–879. [Google Scholar]
- McSweeney, C. S. ; Palmer, B. ; McNeill, D. M. ; Krause, D. O. , 2001: Microbial interactions with tannins: nutritional consequences for ruminants. Animal Feed Science and Technology 91, 83–93. [Google Scholar]
- Metzner, M. ; Wieland, M. ; Rademacher, G. ; Weber, B. K. ; Hafner‐Marx, A. ; Langenmayer, M. C. ; Ammer, H. ; Klee, W. , 2012: High incidence of jaundice in young calves in Southern Germany. Tierärztliche Praxis Ausgabe G, Grosstiere/Nutztiere 40, 283–292. [PubMed] [Google Scholar]
- Min, B. R. ; Hart, S. P. ; Miller, D. ; Tomita, G. M. ; Loetz, E. ; Sahlu, T. , 2005: The effect of grazing forage containing condensed tannins on gastro‐intestinal parasite infection and milk composition in Angora does. Veterinary Parasitology 130, 105–113. [DOI] [PubMed] [Google Scholar]
- Niethammer, F. M. , 2007: Untersuchungen zur Dehydratation bei Kälbern mit akuter Diarrhoe unter Berücksichtigung ausgewählter klinischer und labordiagnostischer Parameter. Diss, Ludwig‐Maximilians‐University, Munich. [Google Scholar]
- Pratt, D. S. ; Kaplan, M. M. , 2007: Laboratory tests In: Schiff E. R., Maddrey W. C., Sorrell M. F. (eds), Schiff's Diseases of the Liver. Lippincott Williams and Wilkins, Philadelphia, pp. 19–60. [Google Scholar]
- Radostits, O. M. ; Hichcliff, K. W. ; Constable, P. D. , 2007: Diseases caused by toxins in plants, fungi, cyanobacteria, plant‐associated bacteria, and venoms in ticks and vertebrate animals In: Radostits O. M., Gay C. C., Hichcliff K. W., Constable P. D. (eds), Veterinary Medicine. Saunders‐Elsevier, Edinburgh, pp. 1851–1920. [Google Scholar]
- Raposo, J. B. ; Mendez, M. C. ; de Andrade, G. B. ; Riet‐Correa, F. , 1998: Experimental intoxication by Myoporum laetum in cattle. Veterinary and Human Toxicology 40, 275–277. [PubMed] [Google Scholar]
- Reubel, G. H. ; Gareis, M. ; Amselgruber, W. M. , 1989: Effects of the Fusarium mycotoxins zearalenone and deoxynivalenol on the mitochondrial methylthiazol tetrazolium‐cleavage activity of monolayer cells. Toxicology in Vitro 3, 311–316. [DOI] [PubMed] [Google Scholar]
- Sattler, T. ; Fuerll, M. , 2004: Creatine kinase and aspartate aminotransferase in cows as indicators for endometritis. Journal of Veterinary Medicine. A, Physiology, Pathology, Clinical Medicine 51, 132–137. [DOI] [PubMed] [Google Scholar]
- Scatena, R. ; Bottoni, P. ; Botta, G. ; Martorana, G. E. ; Giardina, B. , 2007: The role of mitochondria in pharmacotoxicology: a reevaluation of an old, newly emerging topic. American Journal of Physiology. Cell Physiology 293, C12–C21. [DOI] [PubMed] [Google Scholar]
- Smulikowska, S. ; Pastuszewska, B. ; Swiech, E. ; Ochtabinska, A. ; Mieczkowska, A. ; Nguyen, V. C. ; Buraczewska, L. , 2001: Tannin content affects negatively nutritive value of pea for monogastrics. Journal of Animal and Feed Sciences 10, 511–523. [Google Scholar]
- Steinhardt, M. ; Thielscher, H. H. ; von Horn, R. ; von Horn, T. ; Ermgassen, K. ; Ladewig, J. ; Smidt, D. , 1995: Adaptation reaction of dairy calves in the first days of life. Effects of type of delivery and individuality of the newborn. Tierärztliche Praxis Ausgabe G, Grosstiere/Nutztiere 23, 243–249. [PubMed] [Google Scholar]
- Strandberg, K. L. ; Peterson, M. L. ; Lin, Y. C. ; Pack, M. C. ; Chase, D. J. ; Schlievert, P. M. , 2010: Glycerol monolaurate inhibits Candida and Gardnerella vaginalis in vitro and in vivo but not Lactobacillus. Antimicrobial Agents and Chemotherapy 54, 597–601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tennant, B. ; Harrold, D. ; Reina‐Guerra, M. , 1968: Hypoglycemia in neonatal calves associated with acute diarrhea. The Cornell Veterinarian 58, 136–146. [PubMed] [Google Scholar]
- Thomas, L. , 2005: Enzyme In: Thomas L. (ed.), Labor und Diagnose. Th‐Books Verlagsgesellschaft mbH, Frankfurt am Main, pp. 29–120. [Google Scholar]
- Todd, S. E. ; Mellor, D. J. ; Stafford, K. J. ; Gregory, N. G. ; Bruce, R. A. ; Ward, R. N. , 2000: Effects of food withdrawal and transport on 5‐ to 10‐day‐old calves. Research in Veterinary Science 68, 125–134. [DOI] [PubMed] [Google Scholar]
- Vetter, S. M. ; Schlievert, P. M. , 2005: Glycerol monolaurate inhibits virulence factor production in Bacillus anthracis . Antimicrobial Agents and Chemotherapy 49, 1302–1305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu, J. ; Filippich, L. J. ; ALsalam, M. T. , 1992: Tannic acid intoxication in sheep and mice. Research in Veterinary Science 53, 280–292. [DOI] [PubMed] [Google Scholar]
- Zhu, J. ; Filippich, L. J. ; Ng, J. , 1995: Rumen involvement in sheep tannic acid metabolism. Veterinary and Human Toxicology 37, 436–440. [PubMed] [Google Scholar]


