Abstract
Human coronaviruses (CoVs) mostly cause a common cold that is mild and self‐limiting. Zoonotic transmission of CoVs such as the recently identified Middle East respiratory syndrome (MERS)‐CoV and severe acute respiratory syndrome (SARS)‐CoV, on the other hand, may be associated with severe lower respiratory tract infection. This article reviews the clinical and pathological data available on MERS and compares it to SARS. Most importantly, chest radiographs and imaging results of patients with MERS show features that resemble the findings of organizing pneumonia, different from the lesions in SARS patients, which show fibrocellular intra‐alveolar organization with a bronchiolitis obliterans organizing pneumonia‐like pattern. These findings are in line with differences in the induction of cytopathological changes, induction of host gene responses and sensitivity to the antiviral effect of interferons in vitro when comparing both MERS‐CoV and SARS‐CoV. The challenge will be to translate these findings into an integrated picture of MERS pathogenesis in humans and to develop intervention strategies that will eventually allow the effective control of this newly emerging infectious disease. Copyright © 2014 Pathological Society of Great Britain and Ireland. Published by John Wiley & Sons, Ltd.
Keywords: MERS, SARS, coronavirus, virology, respiratory tract
Introduction
Coronaviruses (CoVs) are large, enveloped, positive‐sense RNA viruses that infect birds and a wide range of mammals, including humans. These viruses are composed of a few structural proteins that hold a relatively long (around 30 kb) positive‐stranded genome (Figure 1). They occur worldwide and can cause diseases of medical and veterinary significance. Generally, infections are localized to the respiratory, enteric and/or nervous systems, although systemic disease has been observed in a number of host species 1, 2. At present, six CoVs have been identified that infect humans. Human CoVs HKU1, NL63, 229E and OC43 predominantly cause a mild respiratory tract infection, characterized by upper respiratory tract disease that includes coryza, cough and sore throat. These viruses only occasionally induce lower respiratory tract disease, including bronchitis, bronchiolitis and pneumonia 2, 3, 4, 5, 6, 7, 8, 9. In contrast, two recently emerged CoVs induce a more severe lower respiratory infection that may be fatal, Middle East respiratory syndrome (MERS‐CoV) and severe acute respiratory syndrome (SARS‐CoV) 10, 11. The SARS outbreak started in 2002 in China and, after rapid global spread through human‐to‐human transmission, was halted in 2004. The number of cases reported to the World Health Organization (WHO) was 8096, including 774 deaths 12. Ten years later, the MERS outbreak started in the Middle East and is still ongoing. A total number of 837 laboratory‐confirmed cases have been reported to the WHO, including 291 fatalities 13.
Figure 1.

Schematic diagram of a MERS‐CoV particle and MERS‐CoV genome organization: S, spike protein; M, membrane protein, E, envelope protein; N, nucleocapsid protein.
So far, reports describing autopsies of fatal MERS‐CoV cases have not been published. Therefore, at this stage one can only speculate about the pathology of MERS‐CoV in humans. However, further insight into the pathogenesis and pathological potential of MERS‐CoV may be obtained by comparing and contrasting the epidemiology, clinical manifestations and host cell response of MERS‐CoV to infection with SARS‐CoV, which may also cause a life‐threatening lower respiratory tract disease.
MERS‐CoV transmission
All human CoVs are thought to originate from animal reservoirs, with SARS‐CoV and MERS‐CoV being the most recent examples, emerging from bats via masked palm civet cats on Chinese wet‐markets and dromedary camels in the Middle East, respectively 2, 14, 15, 16, 17. Given the fact that MERS‐CoV seems to be widely present in dromedary camels in the Middle East and some parts of Africa 18, 19, zoonotic transmission is likely to have originated from this animal species and is expected to continue for a long period of time in these regions. Through usage of a common entry receptor, dipeptidyl peptidase 4 (DPP4), the emergence of MERS‐CoV in humans from dromedary camels, and potentially earlier in time from bats, is facilitated (Figure 2).
Figure 2.
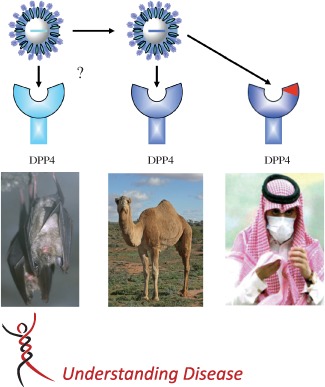
Zoonotic transmission of MERS‐CoV. The emergence of MERS‐CoV from dromedary camels is facilitated by the presence of a highly similar viral receptor (DPP4) in humans. Hypothetically, MERS‐CoV present in dromedary camels may have emerged from CoVs in bats that also use DPP4 as an entry receptor
Human‐to‐human spread of MERS‐CoV does not seem to be efficient but is reported in hospital outbreaks and travellers returning from the Middle East and their close contacts 20, 21, 22, 23, 24. The hospital outbreaks are mostly due to person‐to‐person transmission in haemodialysis units, intensive care units or in‐patient units, where patients are infected with MERS‐CoV of a single monophyletic clade 23. An outbreak among healthcare workers in a hospital was due to overcrowding and inadequate infection control measures 25. It is still unclear whether the transmission through person‐to‐person contact occurs via large respiratory droplets, due to coughing and sneezing, as in SARS, or via fomites 23, 25, 26. Also, the episodes of transmission are not clearly defined but are reported to take place during both the symptomatic and the incubation phases 20. Repeated testing of sputum, nasopharyngeal swabs or bronchoalveolar lavages (BALs) at different time points will be needed to provide a better understanding 20. Some of the larger clusters of patients observed in Al Hasa and Jeddah may be primarily related to human‐to‐human transmission of MERS‐CoV, but super‐spreader events, as described during the SARS outbreak, have not been noted thus far. This may be due to an overall lower level of virus shedding from the upper respiratory tract contributing to lower transmissibility than seen in SARS 26. As a result, the outbreak is more restricted when compared to SARS, although future adaptations of MERS‐CoV in humans may potentially increase human‐to‐human transmission or may alter the virulence of the virus, causing more severe disease.
With respect to controlling MERS, rapid isolation and rigorous infection control practices may in the end suffice to limit the outbreak. These include standard contact and airborne precautions, such as wearing a surgical mask, gloves and a gown on entering the room of infected or suspect patients, and removing them on leaving 25. Dromedary camels infected with MERS‐CoV may not show disease but still may excrete MERS‐CoV through nasal fluids, faeces and, potentially, in their milk and urine 27. Therefore, the WHO recommends avoiding contact with camels, not drinking raw camel milk or camel urine and not eating meat that has not been thoroughly cooked. Also, people who work, or come into contact with, dromedary camels, such as people working at camel farms, slaughterhouses, markets and camel‐racing facilities, and also veterinarians, are at risk and should practise good personal hygiene and wear facial protection and protective clothing where feasible 28.
Clinical aspects of MERS‐CoV infection in humans
Most MERS patients acquired the infection in the Middle East, which subsequently led to limited human‐to‐human transmission in clusters, in healthcare workers and in travel‐related cases outside the region, with mild to severe or even fatal respiratory disease. The median incubation period of a MERS‐CoV infection is 5 days 20. Current data indicate that, overall, more men than women have become infected, with a median age of 47 years (range 9 months–94 years), and most fatalities are observed in patients over 60 years 20, 22. Clinical symptoms observed include fever, cough, sore throat, shortness of breath, myalgia, chest pain, malaise and gastro‐intestinal symptoms, such as diarrhoea, vomiting and abdominal pain. Less common symptoms include chills, wheezing, palpitations and confusion 20, 21, 22, 29. Respiratory symptoms are mainly related to lower respiratory tract disease (dyspnoea, cough and fever), while upper respiratory tract disease is reported infrequently. A large proportion of the severely ill patients required mechanical ventilation 30. Interestingly, many of the reported secondary cases showed mild respiratory symptoms or were asymptomatic 31.
Radiology of MERS patients revealed mild to severe pulmonary consolidation. Chest radiographs of a large percentage of the patients admitted to hospital showed airspace and interstitial opacities, with subtle to extensive, unilateral to bilateral, and focal to diffuse distribution. Air space opacities are variable in their distribution, described as reticular or reticulonodular, and demonstrate thickening of bronchovascular areas 20, 21, 32. Computed tomography (CT) examination of hospitalized patients with MERS revealed bilateral, mostly subpleural and basilar, airspace involvement, with ground‐glass opacities and limited consolidation. The fact that most lesions were found in the subpleural and peribronchovascular region is suggestive of organizing pneumonia 32.
During the course of the infection, MERS‐CoV is mainly detected in the lower respiratory tract, while earlier in the infection virus is detected in the upper, as opposed to the lower, respiratory tract 16, 26, 33, 34, 35, 36, 37. Although virus is detected in urine and blood of some MERS patients, this is not a consistent finding 26, 35, 36, but indicates that systemic infection can occur. Potential risk factors for the development of severe disease are obesity, diabetes mellitus, end‐stage renal disease, cardiac disease, hypertension, lung disease, including asthma and cystic fibrosis, and any immunosuppressive condition 20, 21, 22, 23, 29, 30. Complications described in fatal cases are hyperkalaemia with associated ventricular tachycardia, disseminated intravascular coagulation leading to cardiac arrest, pericarditis and multi‐organ failure 21. One report demonstrated stillbirth during MERS‐CoV infection 38. When compared to a case‐control group, MERS patients were more likely to be admitted to the intensive care unit and had a higher mortality rate 22. The survival rate in patients detected via the active surveillance system was higher than in the clinically identified cases 23. Co‐infections with other pathogens are also described frequently, but their relevance as a critical factor for disease progression is uncertain 20, 29, 32.
Laboratory analyses of blood from MERS patients have revealed mild to severe abnormalities. Haematological abnormalities included elevated leukocyte counts and lymphopenia, while a few cases showed lymphocytosis, thrombocytopenia and coagulopathy 20, 21, 32. Other laboratory findings included elevated creatinine, lactate dehydrogenase, alanine aminotransferase and aspartate aminotransferase levels, suggestive of renal and liver disease or failure 20, 21, 30, 32.
When MERS is compared with SARS, many similarities in the clinical symptoms and respiratory disease become apparent. There are only small differences in the median incubation period, a slightly longer time for MERS (5.2 days) than for SARS (4 days). In SARS, a wide spectrum of clinical symptoms is described, similar to that seen in MERS; fever, chills, diarrhoea and pneumonia 39. In both conditions severely ill patients presented with acute hypoxic respiratory failure that often required mechanical ventilation. Although both viruses are considered respiratory viruses, gastro‐intestinal infections and symptoms have been described in both SARS and MERS patients. The case‐fatality rate may seem higher for MERS‐CoV (around 30%) than for SARS‐CoV (9.6%), but whether this is the result of a higher virulence of MERS‐CoV is not clear, since many MERS‐CoV infections may have gone unnoticed. Similar co‐morbidities have been described for both MERS and SARS. In MERS when compared to SARS, predominantly diabetes type 2 and chronic renal disease are important co‐morbidities 40, 41, 42, 43, 44, 45, 46, 47. In SARS, age is a significant risk factor for the development of severe disease; similarly, for MERS, most fatalities occur in patients aged > 60 years 20, 22. The laboratory findings more often showed higher levels of parameters associated with liver and kidney dysfunction in SARS than in the described cases of MERS, although acute renal failure is often seen in MERS. Complications during pregnancy, such as maternal mortality and stillbirth, have been described in SARS and thus far only rarely seen in MERS cases, but that may be attributable to decreased maternal tolerance to hypoxia and reduced fetal oxygen flow due to respiratory disease 38.
Most importantly, chest radiographs and imaging results of patients with MERS showed opacities and distribution of lesions that resemble the findings of organizing pneumonia seen in patients with pandemic influenza virus A(H1N1)pdm09 32, 48, 49. This is different from the lesions in SARS patients, which show fibrocellular intra‐alveolar organization with a bronchiolitis obliterans organizing pneumonia (BOOP)‐like pattern 50, 51. Thus, although clinical symptoms may be relatively similar between SARS and MERS patients, the mechanisms leading to disease may actually be different.
Pathology of MERS‐CoV infection in humans
So far, no reports describing autopsies of fatal MERS‐CoV cases have been published. From the clinical data and X‐rays of the severe cases of MERS that have been described in humans, a severe and progressive pneumonia may be suspected, as described for SARS, with diffuse alveolar damage (DAD) in the acute phase and more proliferative change in the later phase of the disease. Pulmonary fibrosis was seen frequently in SARS, including in patients who survived the infection. In the absence of accurate follow‐up of MERS patients, there is limited information on the exact course of the disease in the long term. Comparison of the epidemiology, clinical manifestations and host cell response in MERS‐CoV infection to infection with SARS‐CoV may provide further insight into the pathogenesis and pathological potential of MERS‐CoV.
In SARS patients, gross pathology of the respiratory tract demonstrates variable consolidation, with pulmonary oedema, haemorrhage and congestion, and pleural effusion (as reviewed in 52). Histopathology of SARS shows DAD with an exudative phase, a proliferative phase and a fibrotic phase. The exudative phase is seen in patients in the initial 10 days of the disease, and is characterized by necrosis of alveolar, bronchiolar and bronchial epithelial cells, intraluminal oedema, fibrin exudation, hyaline membrane formation, haemorrhage and infiltration of inflammatory cells, such as monocytes or macrophages, lymphocytes and neutrophils, into the alveolar wall and lumina 50, 53, 54, 55. The proliferative phase, after 10–14 days, shows interstitial and alveolar fibrosis, bronchiolitis obliterans organizing pneumonia (BOOP), regeneration with type II pneumocyte hyperplasia and multinucleated giant cells 50, 51, 55, 56, 57. The fibrotic phase, after 14 days, shows interstitial thickening, with fibrosis and a BOOP‐like pattern and few inflammatory cells (mainly histiocytes and lymphocytes) 51, 57. So far, the clinical data of MERS patients demonstrate a similar disease in MERS. However, there is limited evidence for the development of fibrosis in the end‐stage acute respiratory distress syndrome (ARDS) induced by MERS‐CoV.
Pathology of experimental MERS‐CoV infection in animals
Several animal species, such as rhesus macaques (Macaca mulatta), cynomolgus macaques (Macaca fascicularis), marmosets (Callithrix jacchus), ferrets (Mustela putorius), mice (Mus muris), Syrian hamsters (Mesocricetus auratus), rabbits (Oryctolagus cuniculus), guinea‐pigs (Cavia porcellus) and dromedary camels (Camelus dromedarius) have been experimentally infected with MERS‐CoV to study the pathological changes as a result of the viral infection 58, 59, 60, 61, 62, 63, 64. However, in the absence of any descriptive study on the pathological changes in the lungs of human MERS patients, it is very difficult to interpret data from experimental MERS‐CoV animal infection experiments. Overall, the outcome of MERS‐CoV infection and the subsequent development of lower respiratory disease seems variable in different animal species inoculated with MERS‐CoV.
Rhesus macaques can be infected with MERS‐CoV 62 intratracheally or by a combination of inoculation routes 58. The rhesus macaques developed increased body temperatures at 1–2 days post‐infection (dpi) and transient clinical signs, such as increased respiration rate and cough. Haematology showed an increase in total white blood cells and neutrophils from 1–2 dpi and a decrease in lymphocytes from 1–2 dpi 60. Radiographic imaging revealed localized infiltration and interstitial markings. By gross pathology on 3 dpi, there was congestion and little indication of acute pneumonia. By histopathology on 3 dpi, there was a multifocal mild‐to‐moderate interstitial pneumonia characterized by thickened alveolar septa with oedema, fibrin and few macrophages and neutrophils, intraluminal alveolar macrophages, neutrophils and multinucleated giant cells, fibrin and sloughed epithelial cells; perivascular inflammatory infiltrates were present in the interstitium 62. At 6 dpi there was type II pneumocyte hyperplasia with alveolar oedema, fibrin deposition and hyaline membranes 60. In situ hybridization (ISH) and immunohistochemistry (IHC) demonstrated the presence of virus RNA and antigen expression in type I and II pneumocytes and alveolar macrophages. In rhesus macaques inoculated intratracheally, virus was present in the lungs at 3 dpi, but not in nasal, oropharyngeal or rectal swabs or organs (kidney, trachea, brain, heart, liver, spleen, intestine) by RT–PCR and virus titration 62. In rhesus macaques with a combined inoculation route, virus was present predominantly in nasal swabs at 1 and 3 dpi, but not in urogenital and rectal swabs. Virus was also present in the bronchoalveolar lavage (BAL) and in the respiratory tract, but not in the kidney. Up‐regulation of expression levels of proinflammatory cytokines and chemokines for chemotaxis and neutrophil activation, such as IL‐6, CXCL1 and matrix metalloproteinase (MMP)4, was seen in the serum 60. Similarly, cynomolgus macaques can be infected with MERS‐CoV and show virus replication in the lower respiratory tract but, in the lungs, only limited infiltration was observed at different days after infection (van den Brand et al, unpublished observations; Figure 3). Thus, macaque animal models do not fully depict the severe and sometimes fatal pneumonia seen in human patients.
Figure 3.
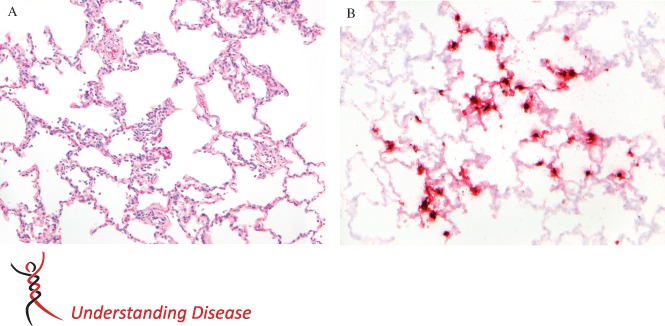
Histopathology and in situ detection of MERS‐CoV in the lungs of a cynomolgus macaque after intratracheal inoculation with MERS‐CoV. (A) The alveolar septa are mildly thickened, with infiltration of a few neutrophils and monocytes. There are also increased numbers of intraluminal alveolar macrophages and a few neutrophils; H&E, magnification = ×10. (B). MERS‐CoV specific RNA by in situ hybridization (ISH), targeting the MERS‐CoV nucleocapsid, is present in lung tissue of of a cynomolgus macaque after intratracheal inoculation with MERS‐CoV. RNA is present predominantly in type II alveolar epithelial cells, with less in type I epithelial cells; ISH, magnification = ×10.
Marmosets infected with a high dose of MERS‐CoV [5.2 × 106 median tissue culture infective dose (TCID50) via the intratracheal, intranasal, oral and ocular route] develop a more severe disease than rhesus macaques 61. From 1 dpi the respiration rate increases with loss of appetite, developing into open‐mouth breathing and laboured breathing at 3–6 dpi. A few animals had to be euthanized before the end of the experiment. No significant alterations in the blood were demonstrated, except for hypoproteinaemia in the severely ill animals. Radiology by X‐rays showed progressive mild to severe interstitial infiltration starting at 1 dpi that was resolved at 13 dpi. Gross pathology showed multifocal consolidation with increased relative lung weights. By histopathology, there was multifocal to coalescing, moderate to marked broncho‐interstitial pneumonia centred around the terminal bronchioles and adjacent alveoli.
In other animal species, including dromedary camels that act as the reservoir species, the virus is suggested to induce only limited clinical symptoms. In most cases reported so far, overt clinical disease is absent in dromedary camels positive for MERS‐CoV. Dromedary camels from the Middle East, Africa and Spain have been found to harbour MERS‐CoV‐specific antibodies 16, 17, 18, 65. In dromedary camels inoculated intratracheally, intranasally and intraconjunctivally with MERS‐CoV, nasal discharge was seen at 2–14 dpi with excretion of virus in the nose, while no virus was found in fecal samples or urine. One dromedary camel at 5 dpi had mild to moderate inflammation and necrosis in the nose, trachea, bronchi and bronchioles, but not in the alveoli. Virus antigen was present, with subsequent presence of infectious virus, in nasal, laryngeal, trachea, bronchial and bronchiolar epithelium, the tonsils and draining lymph nodes, but not in the alveoli 66. Similar observations have been made in rabbits experimentally infected with MERS‐CoV (van den Brand et al, unpublished). Virus was excreted from the upper respiratory tract and detected in the lungs, although no clinical signs were observed and there were limited histopathological changes. Ferrets, guinea‐pigs and Syrian hamsters cannot be infected; there is no virus replication and no seroconversion 59, 62, 64.
Since wild‐type and innately immune‐deficient mice cannot become infected with MERS‐CoV 63, an infectable mouse model has been generated by transducing mice with a recombinant non‐replicating adenovirus expressing the human host cell receptor DPP4. These mice show widespread hDDP4 expression in airway and alveolar epithelial cells. After MERS‐CoV infection, there is no mortality but aged mice lose weight and have abundant virus in the lungs that is cleared by 6–8 dpi. In the lungs there is antigen expression with perivascular and peribronchiolar lymphocytic infiltrates, progressing to interstitial pneumonia. The disease is more severe when there is no type‐I IFN signalling, and the T cell response is necessary for virus clearance 67.
Pathogenesis of MERS‐CoV
Although MERS and SARS resemble each other clinically, in vitro studies have highlighted remarkable differences between these viruses with respect to their growth characteristics, receptor usage and host responses, suggesting that their pathogenesis may be quite different. One way to predict the changes in the lungs after MERS‐CoV infection is to use human tissue that has been infected ex vivo 68. Hocke et al 69 demonstrated widespread MERS‐CoV antigen expression in type I and II alveolar cells, ciliated bronchial epithelium and unciliated cuboid cells of terminal bronchioles, using spectral confocal microscopy. Virus antigen was also found in endothelial cells of pulmonary vessels and rarely in alveolar macrophages. Electron microscopy revealed alveolar epithelial damage, consisting of detachment of type II alveolar epithelial cells and associated disruption of tight junctions, chromatin condensation, nuclear fragmentation and membrane blebbing, the latter suggesting apoptosis 69. Although this ex vivo model does not fully mimic the situation in vivo, these changes are in line with observations in cell lines infected with MERS‐CoV. Severe cytopathic effects were observed in human hepatoma cells infected with MERS‐CoV; these were more severe than those due to SARS‐CoV infection 70, although the in vivo relevance of this observation remains unclear.
DPP4 (also named CD26) has been identified as the receptor for MERS‐CoV 71. All HCoV receptors identified to date are exopeptidases, although their proteolytic activity is not necessary for the virus to bind to the receptors, nor for them to enter the host cell 69, 72, 73. A comparative analysis of HCoV receptor expression across the respiratory tract of humans may provide clues regarding differences in pathobiology between HCoVs. In cell lines and ex vivo lung cultures, DPP4 is expressed in type I and II alveolar cells, ciliated and non‐ciliated bronchial epithelium, bronchial submucosal glands, endothelium, alveolar macrophages and leukocytes 68, 71. This largely corresponds with viral tropism in ex vivo human lung cultures, which show infection of non‐ciliated cells in bronchi, bronchioles, endothelial cells and type I and II pneumocytes, but rarely in alveolar macrophages 68, 71, 75, 76, 77, 78. Remarkably, the binding site of DPP4 is different in different species, explaining why not all animals can be infected with MERS‐CoV 64.
SARS‐CoV and NL63 use a different receptor for entry into cells, angiotensin‐converting enzyme 2 (ACE2) 72, 79, which shows in part a similar cell‐type tropism as is seen for DPP4. In humans, ACE2 is expressed in ciliated bronchial epithelial cells, type I and II pneumocytes and arterial and venous endothelial cells, but not in tissues of the upper respiratory tract, suggesting that these tissues are not the primary site of entrance for SARS‐CoV or NL63 80, 81. SARS‐CoV infects some ACE2‐positive cells – ciliated bronchial epithelial cells, bronchioles and type I and II pneumocytes, but not endothelial cells 82.
Comparative genomics provides a way to study the molecular basis for the host response against different but related viral pathogens, as was demonstrated previously in SARS‐CoV infection of different primate host species 83, 84, 85, 86. At present, only one comparative in vitro study using MERS‐CoV has been performed 87. Calu3 cells, differentiated into polarized ciliated cells, permit replication of both SARS‐CoV and MERS‐CoV at similar levels. However, MERS‐CoV induces a substantial cytopathic effect, starting 18–24 h after infection, whereas SARS‐CoV maintains steady replication and cell viability until 72 h after infection 70, 77, 87. On the other hand, MERS‐CoV seems more sensitive to prophylactic and therapeutic treatment of infection in vitro than SARS‐CoV 70. In addition, MERS‐CoV induces much greater dysregulation of the host response to infection than SARS‐CoV. MERS‐CoV specifically down‐regulates genes involved in the antigen‐presentation pathway, which could have substantial implications for the development of adaptive immune responses 87. With respect to the analysis of the immune response in humans infected with MERS‐CoV, limited data from two patients with different disease outcomes are available to date. In BAL and serum from the patient with a poor outcome, there was a decrease in interferon (IFN)α as well as decreased expression of retinoic inducible‐acid gene (RIG)‐1, melanoma differentiation‐associated protein 5 (MDA5) and interferon regulatory factors (IRF)3 and IRF7, which are involved in the recognition of viruses by the innate immune system. In the patient with poor outcome there were also high levels of CXC‐motif chemokines ligand (CXCL)10 and interleukin (IL)‐10, which may have resulted in lower IFNγ expression and higher levels of IL‐17A and IL‐23 88. In the patient who survived, rapid clearance of the virus with increased levels of IL‐12 and IFNγ was noted 88.
In SARS, the development of severe lower respiratory tract disease correlates partly with aberrant immune responses, with unbalanced cytokine and chemokine profiles 39, 50, 89, 90. The levels of both cytokines and chemokines in the blood are elevated: IL‐1, IL‐6, IL‐8, IL‐12, IFNγ, monocyte chemotactic protein (MCP)‐1 (or CC‐motif ligand 2, CCL2), monokine induced by IFNγ (MIG), IFN‐inducible protein (IP‐10, or CXCL10), and transforming growth factor (TGF)β 89, 90, 91, 92, 93, 94. Some of these chemokines are important for chemotaxis and activation of neutrophils and monocytes 95, 96, 97, which corresponds with the infiltration of these cells in the respiratory tract of human SARS cases 83, 91, 92, 98. It remains to be determined which host responses dominate MERS‐CoV infection in vivo but, based on the results obtained thus far, MERS‐CoV and SARS‐CoV may induce different pathways.
Conclusion
At this time it is difficult to describe or predict the pathology of severe respiratory disease from MERS‐CoV infection. First, there are no reports on autopsies of human fatal MERS cases. This is partly related to the religious backgrounds of the patients, which exclude autopsy. Although limited numbers of human fatal SARS patients have been described, these studies indicate an immunopathological component that may dominate the pathogenesis of SARS. Second, to gain more knowledge of fatal MERS in humans, further studies are needed in animal models that could provide information on replication dynamics, clinical disease, histological lesions and cellular tropism. Third, the patients with severe MERS have very diverse pre‐existing conditions, and therefore it may be difficult to obtain definite answers from these studies. Virus‐induced histological changes may be obscured by clinical treatments or pre‐existing disease. Thus, it may be very difficult to know the full scope of this disease, including the histological lesions, viral cellular tropism and pathogenesis of MERS‐CoV. On the other hand, although there are better descriptive studies of SARS regarding the pathology induced in the lower respiratory tract, these provided limited benefit for the development of intervention strategies. Therapeutically, corticosteroids were used for the treatment of SARS, producing variable improvement with less fever, reduced inflammatory infiltrates and better oxygenation 99. More promising results were obtained with the administration of IFNs in SARS patients. In fact, in vitro studies have demonstrated that MERS‐CoV is more sensitive to type I IFN than SARS‐CoV. Overall, the current available data on MERS‐CoV suggest that patients at risk for severe exacerbations after infection are elderly patients or patients who have underlying co‐morbid conditions. These patients may not be able to combat the viral infection with type I IFN host responses as efficiently as other patient groups, as aging and co‐morbid conditions have been described to have a negative effect on the ability to mount strong type I IFN responses 84, 100. The sensitivity of MERS‐CoV in vitro to type I IFN may indicate that type I IFN can be used as a prophylactic/therapeutic intervention strategy in vivo.
In general, human CoV infections cause a relatively mild respiratory disease in children and many healthy adults. Severe exacerbations of CoV disease, with lower respiratory tract involvement, seem to occur in the elderly and/or patients with underlying co‐morbidities. Possibly, the lack of pre‐existing immunity against SARS‐CoV and MERS‐CoV in the human population resulted in a higher infection rate in adults and the elderly during the SARS outbreak than normally observed for the other non‐SARS human CoVs, leading to a higher morbidity and mortality. Alternatively, viral factors, including those encoded by different accessory viral genes, may play a decisive role in determining pathogenesis.
Overall, there are a number of potential key in vivo and in vitro differences between MERS‐CoV and SARS‐CoV infections. These include radiographic differences suggesting a different type of pneumonia, a large difference in cytopathic effect, differences in host response to infection, and differences in sensitivity to type I IFN in vitro. These observations point to different disease‐causing mechanisms and warrant further studies into the effect of these two viruses on different cell populations in vitro, ex vivo and in vivo, the outcome of which may have significant implications for the development of intervention strategies.
Author contributions
All authors contributed to the writing and editing of the manuscript and the design of the figures.
Acknowledgements
This work was partially funded by ZonMW TOP (Project Nos 91213058 and 40‐00812‐98‐13066). We thank S. Getu, P. van Run, and F. van der Panne for preparation of Figure 2.
Conflict of interest: BL Haagmans has a patent filed on MERS‐CoV; SL Smits is employed part‐time by Viroclinics Biosciences.
References
- 1. Perlman S. Pathogenesis of coronavirus‐induced infections – review of pathological and immunological aspects. Coronavirus Arterivirus 1998; 440: 503–513. [PubMed] [Google Scholar]
- 2. Weiss SR, Navas‐Martin S. Coronavirus pathogenesis and the emerging pathogen severe acute respiratory syndrome coronavirus. Microbiol Mol Biol Rev 2005; 69: 635–664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Bradburne AF, Bynoe ML, Tyrrell DA. Effects of a 'new' human respiratory virus in volunteers. Br Med J 1967; 3: 767–769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Hamre D, Procknow JJ. A new virus isolated from the human respiratory tract. Proc Soc Exp Biol Med 1966; 121: 190–193. [DOI] [PubMed] [Google Scholar]
- 5. Fouchier RA, Hartwig NG, Bestebroer TM, et al. A previously undescribed coronavirus associated with respiratory disease in humans. Proc Natl Acad Sci USA 2004; 101: 6212–6216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. van der Hoek L, Sure K, Ihorst G, et al. Croup is associated with the novel coronavirus NL63. PLoS Med 2005; 2: e240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Woo PC, Lau SK, Tsoi HW, et al. Clinical and molecular epidemiological features of coronavirus HKU1‐associated community‐acquired pneumonia. J Infect Dis 2005; 192: 1898–1907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Gaunt ER, Hardie A, Claas EC, et al. Epidemiology and clinical presentations of the four human coronaviruses 229E, HKU1, NL63, and OC43 detected over 3 years using a novel multiplex real‐time PCR method. J Clin Microbiol 2010; 48: 2940–2947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Wevers BA, van der Hoek L. Recently discovered human coronaviruses. Clin Lab Med 2009; 29: 715–724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. van Boheemen S, de Graaf M, Lauber C, et al. Genomic characterization of a newly discovered coronavirus associated with acute respiratory distress syndrome in humans. MBio 2012; 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Xu RH, He JF, Evans MR, et al. Epidemiologic clues to SARS origin in China. Emerg Infect Dis 2004; 10: 1030–1037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. WHO . Summary of probable SARS cases with onset of illness from 1 November 2002 to 31 July 2003 [cited 25 September 2014]: http://www.who.int/csr/sars/country/table2004_04_21/en/
- 13. WHO . Middle East respiratory syndrome coronavirus (MERS‐CoV) – update, 23 July 2014. [cited 25 September 2014]: http://www.who.int/csr/don/2014_07_23_mers/en/
- 14. Sawicki SG, Sawicki DL. Coronaviruses use discontinuous extension for synthesis of subgenome‐length negative strands. Coronavirus Arterivirus 1995; 380: 499–506. [DOI] [PubMed] [Google Scholar]
- 15. Shi Z, Hu Z. A review of studies on animal reservoirs of the SARS coronavirus. Virus Res 2008; 133: 74–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Azhar EI, El‐Kafrawy SA, Farraj SA, et al. Evidence for camel‐to‐human transmission of MERS coronavirus. N Engl J Med 2014; 370: 2499–2505. [DOI] [PubMed] [Google Scholar]
- 17. Reusken CB, Haagmans BL, Muller MA, et al. Middle East respiratory syndrome coronavirus neutralising serum antibodies in dromedary camels: a comparative serological study. Lancet Infect Dis 2013; 13: 859–866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Haagmans BL, Al Dhahiry SH, Reusken CB, et al. Middle East respiratory syndrome coronavirus in dromedary camels: an outbreak investigation. Lancet Infect Dis 2014; 14: 140–145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Reusken CB, Messadi L, Feyisa A, et al. Geographic Distribution of MERS coronavirus among dromedary camels, Africa. Emerg Infect Dis 2014; 20: 1370–1374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Assiri A, Al‐Tawfiq JA, Al‐Rabeeah AA, et al. Epidemiological, demographic, and clinical characteristics of 47 cases of Middle East respiratory syndrome coronavirus disease from Saudi Arabia: a descriptive study. Lancet Infect Dis 2013; 13: 752–761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Al‐Abdallat MM, Payne DC, Alqasrawi S, et al. Hospital‐associated outbreak of Middle East respiratory syndrome coronavirus: a serologic, epidemiologic, and clinical description. Clin Infect Dis 2014; 59: 1225–1233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Al‐Tawfiq JA, Hinedi K, Ghandour J, et al. Middle East respiratory syndrome coronavirus: a case‐control study of hospitalized patients. Clin Infect Dis 2014; 59: 160–165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Assiri A, McGeer A, Perl TM, et al. Hospital outbreak of Middle East respiratory syndrome coronavirus. N Engl J Med 2013; 369: 407–416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Memish ZA, Al‐Tawfiq JA, Makhdoom HQ, et al. Screening for Middle East respiratory syndrome coronavirus infection in hospital patients and their healthcare worker and family contacts: a prospective descriptive study. Clin Microbiol Infect 2014; 20: 469–474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Zumla A, Hui DS. Infection control and MERS‐CoV in health‐care workers. Lancet 2014; 383: 1869–1871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Guery B, Poissy J, el Mansouf L, et al. Clinical features and viral diagnosis of two cases of infection with Middle East respiratory syndrome coronavirus: a report of nosocomial transmission. Lancet 2013; 381: 2265–2272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Reusken CB, Farag EA, Jonges M, et al. Middle East respiratory syndrome coronavirus (MERS‐CoV) RNA and neutralising antibodies in milk collected according to local customs from dromedary camels, Qatar, April 2014. Eur Surveill 2014; 19: 20829. [DOI] [PubMed] [Google Scholar]
- 28. WHO . Update on MERS‐CoV transmission from animals to humans, and interim recommendations for at‐risk groups [cited 30 September 2014]: http://www.who.int/csr/disease/coronavirus_infections/MERS_CoV_RA_20140613.pdf?ua=1
- 29. Memish ZA, Al‐Tawfiq JA, Assiri A, et al. Middle East Respiratory syndrome coronavirus disease in children. Pediatr Infect Dis J 2014; 33: 904–906. [DOI] [PubMed] [Google Scholar]
- 30. Arabi YM, Arifi AA, Balkhy HH, et al. Clinical course and outcomes of critically ill patients with Middle East respiratory syndrome coronavirus infection. Ann Intern Med 2014; 160: 389–397. [DOI] [PubMed] [Google Scholar]
- 31. WHO . Middle East respiratory syndrome coronavirus (MERS‐CoV) summary and literature update – as of 11 June 2014 [cited 25 september 2014]: http://www.who.int/csr/disease/coronavirus_infections/MERS-CoV_summary_update_20140611.pdf?ua=1
- 32. Ajlan AM, Ahyad RA, Jamjoom LG, et al. Middle East respiratory syndrome coronavirus (MERS‐CoV) infection: chest CT findings. Am J Roentgenol 2014; 203: 782–787. [DOI] [PubMed] [Google Scholar]
- 33. Bermingham A, Chand MA, Brown CS, et al. Severe respiratory illness caused by a novel coronavirus, in a patient transferred to the United Kingdom from the Middle East, September 2012. Eur Surveill 2012; 17: 20290. [PubMed] [Google Scholar]
- 34. Buchholz U, Muller MA, Nitsche A, et al. Contact investigation of a case of human novel coronavirus infection treated in a German hospital, October–November 2012. Eur Surveill 2013; 18: 20406. [PubMed] [Google Scholar]
- 35. Zaki AM, van Boheemen S, Bestebroer TM, et al. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med 2012; 367: 1814–1820. [DOI] [PubMed] [Google Scholar]
- 36. Drosten C, Seilmaier M, Corman VM, et al. Clinical features and virological analysis of a case of Middle East respiratory syndrome coronavirus infection. Lancet Infect Dis 2013; 13: 745–751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Memish ZA, Assiri A, Almasri M, et al. Prevalence of MERS‐CoV nasal carriage and compliance with the Saudi health recommendations among pilgrims attending the 2013 Hajj. J Infect Dis 2014. 210: 1067–1072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Payne DC, Iblan I, Alqasrawi S, et al. Stillbirth during infection with Middle East respiratory syndrome coronavirus. J Infect Dis 2014; 209: 1870–1872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Peiris JS, Guan Y, Yuen KY. Severe acute respiratory syndrome. Nat Med 2004; 10: S88–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Chan‐Yeung M, Xu RH. SARS: epidemiology. Respirology 2003; 8(suppl): S9–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Donnelly CA, Ghani AC, Leung GM, et al. Epidemiological determinants of spread of causal agent of severe acute respiratory syndrome in Hong Kong. Lancet 2003; 361: 1761–1766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Lee N, Hui D, Wu A, et al. A major outbreak of severe acute respiratory syndrome in Hong Kong. N Engl J Med 2003; 348: 1986–1994. [DOI] [PubMed] [Google Scholar]
- 43. Manocha S, Walley KR, Russell JA. Severe acute respiratory distress syndrome (SARS): a critical care perspective. Crit Care Med 2003; 31: 2684–2692. [DOI] [PubMed] [Google Scholar]
- 44. Karlberg J, Chong DS, Lai WY. Do men have a higher case fatality rate of severe acute respiratory syndrome than women do? Am J Epidemiol 2004; 159: 229–231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Leung GM, Hedley AJ, Ho LM, et al. The epidemiology of severe acute respiratory syndrome in the 2003 Hong Kong epidemic: an analysis of all 1755 patients. Ann Intern Med 2004; 141: 662–673. [DOI] [PubMed] [Google Scholar]
- 46. Lau EH, Hsiung CA, Cowling BJ, et al. A comparative epidemiologic analysis of SARS in Hong Kong, Beijing and Taiwan. BMC Infect Dis 2010; 10: 50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Liu JW, Lu SN, Chen SS, et al. Epidemiologic study and containment of a nosocomial outbreak of severe acute respiratory syndrome in a medical center in Kaohsiung, Taiwan. Infect Control Hosp Epidemiol 2006; 27: 466–472. [DOI] [PubMed] [Google Scholar]
- 48. Bai L, Gu L, Cao B, et al. Clinical features of pneumonia caused by 2009 influenza A(H1N1) virus in Beijing, China. Chest 2011; 139: 1156–1164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Mineo G, Ciccarese F, Modolon C, et al. Post‐ARDS pulmonary fibrosis in patients with H1N1 pneumonia: role of follow‐up CT. Radiol Med 2012; 117: 185–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Nicholls JM, Poon LL, Lee KC, et al. Lung pathology of fatal severe acute respiratory syndrome. Lancet 2003; 361: 1773–1778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Tse GM, To KF, Chan PK, et al. Pulmonary pathological features in coronavirus associated severe acute respiratory syndrome (SARS). J Clin Pathol 2004; 57: 260–265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. van den Brand JM, Haagmans BL, van Riel D, et al. The pathology and pathogenesis of experimental severe acute respiratory syndrome and influenza in animal models. J Comp Pathol 2014; 151: 83–112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Ding Y, Wang H, Shen H, et al. The clinical pathology of severe acute respiratory syndrome (SARS): a report from China. J Pathol 2003; 200: 282–289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Hsueh PR, Chen PJ, Hsiao CH, et al. Patient data, early SARS epidemic, Taiwan. Emerg Infect Dis 2004; 10: 489–493. [DOI] [PubMed] [Google Scholar]
- 55. Shieh WJ, Hsiao CH, Paddock CD, et al. Immunohistochemical, in situ hybridization, and ultrastructural localization of SARS‐associated coronavirus in lung of a fatal case of severe acute respiratory syndrome in Taiwan. Hum Pathol 2005; 36: 303–309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Franks TJ, Chong PY, Chui P, et al. Lung pathology of severe acute respiratory syndrome (SARS): a study of 8 autopsy cases from Singapore. Hum Pathol 2003; 34: 743–748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Cheung OY, Chan JW, Ng CK, et al. The spectrum of pathological changes in severe acute respiratory syndrome (SARS). Histopathology 2004; 45: 119–124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Munster VJ, de Wit E, Feldmann H. Pneumonia from human coronavirus in a macaque model. N Engl J Med 2013; 368: 1560–1562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. de Wit E, Prescott J, Baseler L, et al. The Middle East respiratory syndrome coronavirus (MERS‐CoV) does not replicate in Syrian hamsters. PLoS One 2013; 8: e69127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. de Wit E, Rasmussen AL, Falzarano D, et al. Middle East respiratory syndrome coronavirus (MERS‐CoV) causes transient lower respiratory tract infection in rhesus macaques. Proc Natl Acad Sci USA 2013; 110: 16598–16603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Falzarano D, de Wit E, Feldmann F, et al. Infection with MERS‐CoV causes lethal pneumonia in the common m. PLoS Pathog 2014; 10: e1004250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Yao Y, Bao L, Deng W, et al. An animal model of MERS produced by infection of rhesus macaques with MERS coronavirus. J Infect Dis 2014; 209: 236–242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Coleman CM, Matthews KL, Goicochea L, et al. Wild‐type and innate immune‐deficient mice are not susceptible to the Middle East respiratory syndrome coronavirus. J Gen Virol 2014; 95: 408–412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Raj VS, Smits SL, Provacia LB, et al. Adenosine deaminase acts as a natural antagonist for dipeptidyl peptidase 4‐mediated entry of the Middle East respiratory syndrome coronavirus. J Virol 2014; 88: 1834–1838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Alagaili AN, Briese T, Mishra N, et al. Middle East respiratory syndrome coronavirus infection in dromedary camels in Saudi Arabia. MBio 2014; 5: e00884–00814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Adney DR, Van Doremalen N, Brown VR, et al. Replication and shedding of MERS‐CoV in upper respiratory tract of inoculated dromedary camels. Emerg Infect Dis 2014; doi: 10.3201/eid2012.141280, (in press). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Zhao J, Li K, Wohlford‐Lenane C, et al. Rapid generation of a mouse model for Middle East respiratory syndrome. Proc Natl Acad Sci USA 2014; 111: 4970–4975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Chan RW, Chan MC, Agnihothram S, et al. Tropism of and innate immune responses to the novel human betacoronavirus lineage C virus in human ex vivo respiratory organ cultures. J Virol 2013; 87: 6604–6614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Hocke AC, Becher A, Knepper J, et al. Emerging human middle East respiratory syndrome coronavirus causes widespread infection and alveolar damage in human lungs. Am J Respir Crit Care Med 2013; 188: 882–886. [DOI] [PubMed] [Google Scholar]
- 70. de Wilde AH, Raj VS, Oudshoorn D, et al. MERS‐coronavirus replication induces severe in vitro cytopathology and is strongly inhibited by cyclosporin A or interferon‐α treatment. J Gen Virol 2013; 94: 1749–1760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Raj VS, Mou H, Smits SL, et al. Dipeptidyl peptidase 4 is a functional receptor for the emerging human coronavirus‐EMC. Nature 2013; 495: 251–254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Li W, Moore MJ, Vasilieva N, et al. Angiotensin‐converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 2003; 426: 450–454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Delmas B, Gelfi J, L'Haridon R, et al. Aminopeptidase N is a major receptor for the entero‐pathogenic coronavirus TGEV. Nature 1992; 357: 417–420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. van der Velden VH, Wierenga‐Wolf AF, Adriaansen‐Soeting PW, et al. Expression of aminopeptidase N and dipeptidyl peptidase IV in the healthy and asthmatic bronchus. Clin Exp Allergy 1998; 28: 110–120. [DOI] [PubMed] [Google Scholar]
- 75. Chan JF, Chan KH, Choi GK, et al. Differential cell line susceptibility to the emerging novel human betacoronavirus 2c EMC/2012: implications for disease pathogenesis and clinical manifestation. J Infect Dis 2013; 207: 1743–1752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Dijkman R, Jebbink MF, Koekkoek SM, et al. Isolation and characterization of current human coronavirus strains in primary human epithelial cell cultures reveal differences in target cell tropism. J Virol 2013; 87: 6081–6090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Chan RW, Hemida MG, Kayali G, et al. Tropism and replication of Middle East respiratory syndrome coronavirus from dromedary camels in the human respiratory tract: an in vitro and ex vivo study. Lancet Respir Med 2014; 2: 813–822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Kindler E, Jonsdottir HR, Muth D, et al. Efficient replication of the novel human betacoronavirus EMC on primary human epithelium highlights its zoonotic potential. MBio 2013; 4: e00611–00612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Hofmann H, Pyrc K, van der Hoek L, et al. Human coronavirus NL63 employs the severe acute respiratory syndrome coronavirus receptor for cellular entry. Proc Natl Acad Sci USA 2005; 102: 7988–7993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Hamming I, Timens W, Bulthuis ML, et al. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol 2004; 203: 631–637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Jia HP, Look DC, Shi L, et al. ACE2 receptor expression and severe acute respiratory syndrome coronavirus infection depend on differentiation of human airway epithelia. J Virol 2005; 79: 14614–14621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Nicholls JM, Butany J, Poon LL, et al. Time course and cellular localization of SARS‐CoV nucleoprotein and RNA in lungs from fatal cases of SARS. PLoS Med 2006; 3: e27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Smits SL, de Lang A, van den Brand JM, et al. Exacerbated innate host response to SARS‐CoV in aged non‐human primates. PLoS Pathog 2010; 6: e1000756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Smits SL, van den Brand JM, de Lang A, et al. Distinct severe acute respiratory syndrome coronavirus‐induced acute lung injury pathways in two different nonhuman primate species. J Virol 2011; 85: 4234–4245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Lederer S, Favre D, Walters KA, et al. Transcriptional profiling in pathogenic and non‐pathogenic SIV infections reveals significant distinctions in kinetics and tissue compartmentalization. PLoS Pathog 2009; 5: e1000296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Mandl JN, Barry AP, Vanderford TH, et al. Divergent TLR7 and TLR9 signaling and type I interferon production distinguish pathogenic and nonpathogenic AIDS virus infections. Nat Med 2008; 14: 1077–1087. [DOI] [PubMed] [Google Scholar]
- 87. Josset L, Menachery VD, Gralinski LE, et al. Cell host response to infection with novel human coronavirus EMC predicts potential antivirals and important differences with SARS coronavirus. MBio 2013; 4: e00165–00113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Faure E, Poissy J, Goffard A, et al. Distinct immune response in two MERS‐CoV‐infected patients: can we go from bench to bedside? PLoS One 2014; 9: e88716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Wong CK, Lam CW, Wu AK, et al. Plasma inflammatory cytokines and chemokines in severe acute respiratory syndrome. Clin Exp Immunol 2004; 136: 95–103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Zhang Y, Li J, Zhan Y, et al. Analysis of serum cytokines in patients with severe acute respiratory syndrome. Infect Immun 2004; 72: 4410–4415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Huang KJ, Su IJ, Theron M, et al. An interferon‐γ‐related cytokine storm in SARS patients. J Med Virol 2005; 75: 185–194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Jiang Y, Xu J, Zhou C, et al. Characterization of cytokine/chemokine profiles of severe acute respiratory syndrome. Am J Respir Crit Care Med 2005; 171: 850–857. [DOI] [PubMed] [Google Scholar]
- 93. Tang NL, Chan PK, Wong CK, et al. Early enhanced expression of interferon‐inducible protein‐10 (CXCL‐10) and other chemokines predicts adverse outcome in severe acute respiratory syndrome. Clin Chem 2005; 51: 2333–2340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Baas T, Roberts A, Teal TH, et al. Genomic analysis reveals age‐dependent innate immune responses to severe acute respiratory syndrome coronavirus. J Virol 2008; 82: 9465–9476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Ware LB, Matthay MA. The acute respiratory distress syndrome. N Engl J Med 2000; 342: 1334–1349. [DOI] [PubMed] [Google Scholar]
- 96. Fan J, Ye RD, Malik AB. Transcriptional mechanisms of acute lung injury. Am J Physiol Lung Cell Mol Physiol 2001; 281: L1037–1050. [DOI] [PubMed] [Google Scholar]
- 97. Tsushima K, King LS, Aggarwal NR, et al. Acute lung injury review. Intern Med 2009; 48: 621–630. [DOI] [PubMed] [Google Scholar]
- 98. Cameron MJ, Ran L, Xu L, et al. Interferon‐mediated immunopathological events are associated with atypical innate and adaptive immune responses in patients with severe acute respiratory syndrome. J Virol 2007; 81: 8692–8706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Chen RC, Tang XP, Tan SY, et al. Treatment of severe acute respiratory syndrome with glucosteroids: the Guangzhou experience. Chest 2006; 129: 1441–1452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Stout‐Delgado HW, Yang X, Walker WE, et al. Aging impairs IFN regulatory factor 7 up‐regulation in plasmacytoid dendritic cells during TLR9 activation. J Immunol 2008; 181: 6747–6756. [DOI] [PMC free article] [PubMed] [Google Scholar]


