Abstract
The aetiology and pathogenesis of bovine respiratory disease (BRD) are complex and involve the interplay of infectious agents, management and environmental factors. Previous studies of BRD focused on ante‐mortem samples from the upper respiratory tract and identified several unconventional viruses. The lung, however, is the primary location where significant BRD lesions are usually found and is a common post‐mortem diagnostic specimen. In this study, results of high‐throughput virome sequencing, bacterial culture, targeted real‐time PCR and histological examination of 130 bovine pneumonic lungs from western Canadian cattle were combined to explore associations of microorganisms with different types of pneumonia. Fibrinous bronchopneumonia (FBP) was the predominant type of pneumonia (46.2%, 60/130) and was associated with the detection of Mannheimia haemolytica. Detection of Histophilus somni and Pasteurella multocida was associated with suppurative bronchopneumonia (SBP) and concurrent bronchopneumonia and bronchointerstitial pneumonia (BP&BIP), respectively. Sixteen viruses were identified, of which bovine parvovirus 2 (BPV2) was the most prevalent (11.5%, 15/130) followed by ungulate tetraparvovirus 1 (UTPV1, 8.5%, 11/130) and bovine respiratory syncytial virus (BRSV, 8.5%, 11/130). None of these viruses, however, were significantly associated with a particular type of pneumonia. Unconventional viruses such as influenza D virus (IDV) and bovine rhinitis B virus (BRBV) were detected, although sparsely, consistent with our previous findings in upper respiratory tract samples. Taken together, our results show that while virus detection in post‐mortem lung samples is of relatively little diagnostic value, the strong associations of H. somni and M. haemolytica with SBP and FBP, respectively, indicate that histopathology can be useful in differentiating bacterial aetiologies.
Keywords: bovine parvovirus 2, bovine pneumonia, influenza D virus, metagenomic sequencing, Mycoplasma bovis, pulmonary virome
1. INTRODUCTION
Bovine respiratory disease (BRD) is one of the most common and costly diseases in both beef and dairy cattle worldwide (Fulton, 2009; Mehinagic, Pilo, Vidondo, & Stokar‐Regenscheit, 2019). Mannheimia haemolytica (M. haemolytica), Pasteurella multocida (P. multocida), Histophilus somni (H. somni), Bibersteinia trehalosi (B. trehalosi) and Mycoplasma bovis (M. bovis) are major bacterial pathogens, and their pathogenicity can be greatly enhanced by viral infections, which can damage the respiratory epithelium (Griffin, Chengappa, Kuszak, & McVey, 2010). Bovine respiratory syncytial virus (BRSV) (Brodersen, 2010), bovine herpesvirus 1 (BHV1) (Jones & Chowdhury, 2010), bovine viral diarrhoea virus (BVDV) (Brodersen, 2014) and bovine parainfluenza virus 3 (BPIV3) (Ellis, 2010) are considered to be the most common and pathogenic viral pathogens associated with BRD. Killed and modified live virus vaccines for BRSV, BVDV, BHV1 and BPIV3 are available for BRD prevention in North America (Fulton et al., 2016). Unfortunately, assessment of the effectiveness of bacterial vaccination for BRD has indicated variable to little benefit (Griffin et al., 2010; Larson & Step, 2012), and excessive anti‐microbial usage for metaphylaxis raises concerns of increasing antibiotic resistance for both animals and humans (Portis, Lindeman, Johansen, & Stoltman, 2012). Bovine respiratory disease morbidity and mortality rates among feedlot cattle are not reduced despite viral vaccination and anti‐microbial usage (Hilton, 2014).
Metagenomic sequencing has been shown to be a powerful tool for the detection of viruses (Buermans & den Dunnen, 2014), and previous studies have applied metagenomic sequencing to identify unconventional viruses associated with BRD (Mitra, Cernicchiaro, Torres, Li, & Hause, 2016; Ng et al., 2015; Zhang et al., 2019). As a result of these work, several unconventional viruses associated with BRD in dairy and beef cattle, such as influenza D virus (IDV) and bovine rhinitis virus A and B (BRAV and BRBV), have been identified (Mitra et al., 2016; Ng et al., 2015; Zhang et al., 2019). These studies have focused on the virome of ante‐mortem samples from the upper respiratory tract (nasal cavity and trachea). To the best of our knowledge, the virome of lungs with BRD has not been investigated, although this is the primary location where significant BRD lesions are usually found, and a common diagnostic specimen.
The complex, multifactorial nature of BRD complicates not only the understanding of disease pathogenesis and development of strategies for disease management, but also making an aetiological diagnosis in cattle affected by the disease. The objective of this study was to characterize the virome of bovine pneumonia lungs collected in western Canada using metagenomic sequencing and to combine this information with bacterial culture and targeted real‐time PCR results from the same samples to determine the relationships between the microorganisms detected and different types of pneumonia defined by histopathological examination.
2. MATERIALS AND METHODS
2.1. Sample collection and histology
Cases of pneumonia (n = 130) submitted to PDS from September 2017 to December 2018 were conveniently selected. The proportion of beef cattle was 90.8% (118/130) compared with 9.2% (12/130) dairy cattle. Histology and H&E staining were performed routinely on formalin‐fixed tissues. Fresh lung samples were stored at −20°C for up to 6 weeks according to routine laboratory practice and then transferred to −80°C for longer term storage prior to metagenomic sequencing.
2.2. Metagenomic sequencing
Lung tissues (about 5 × 5 × 5 mm) were homogenized with silicon beads (0.1 mm diameter, BioSpec Products) in a Tissue Lyser II (30 cycles/s for 5 min, Qiagen). After centrifugation at 13,000 g for 5 min, the supernatant was filtered (Filtropur S 0.2 µm, Sarstedt). Library preparation for metagenomic sequencing was performed as previously described (Zhang et al., 2019). Briefly, filtered supernatants were incubated with 24 units of DNase (Life Technologies) and 20 units of RNase ONE Ribonuclease (Promega) at 37°C for 90 min. Viral nucleic acids were extracted using the QIAamp MinElute virus spin kit (Qiagen) according to the manufacturer's instructions. Reverse transcription was performed using a Superscript III First‐Strand synthesis kit (Life Technologies) with primer FR26RV‐N 5′‐GCC GGA GCT CTG CAG ATA TCN NNN NN‐ 3′ (Allander et al., 2005). Second‐strand synthesis was carried out using Sequenase DNA polymerase (Affymetrix). Double‐stranded DNA was purified using NucleoMag NGS beads (Macherey‐Nagel Inc.) and subsequently amplified using primer FR20RV 5′‐GCC GGA GCT CTG CAG ATA TC‐ 3′ (Allander et al., 2005). cDNA was quantified using Qubit dsDNA BR assay kit (Invitrogen), and 1 ng cDNA from each sample was used for library preparation using the Nextera XT library preparation kit (Illumina) according to manufacturer's instructions. After confirmation of the fragment size distribution of the libraries with Agilent Technology 2100 Bioanalyzer (Agilent Technologies) and normalization of the libraries, the pooled barcoded libraries were sequenced using a MiSeq V2 500 cycle kit (Illumina).
2.3. Bioinformatic analysis
Bioinformatic analysis was performed as previously described (Zhang et al., 2019). In brief, quality trimming was performed using Trimmomatic‐0.32 (Bolger, Lohse, & Usadel, 2014) with minimum length of 50 and Phred score of 20. Bowtie2 (Langmead & Salzberg, 2012) was used to map the trimmed reads to the host (Bos taurus) reference genome (PRJNA33843, PRJNA32899), and SAMtools (Li et al., 2009) was implemented to identify the unmapped reads, which were subsequently extracted from the original fastq files using cdbyank. The extracted unmapped reads were assembled into contigs with default parameters using Trinity (Grabherr et al., 2011). The assembled reads were compared with the virus Reference Sequence (RefSeq) database (Brister, Ako‐Adjei, Bao, & Blinkova, 2015) using BLASTn. Virus‐like contigs aligning over a minimum length of 100 base pairs with expect value < 10–3 to the RefSeq database were further examined manually by BLASTx comparison to the NCBI non‐redundant protein database to identify any spurious matches. The total number of reads corresponding to each identified virus was determined from the Bowtie2 SAM results files.
2.4. Histological examination
Cases were classified as suppurative bronchopneumonia (SBP), fibrinous bronchopneumonia (FBP), interstitial pneumonia (IP), bronchointerstitial pneumonia (BIP), bronchopneumonia + bronchointerstitial pneumonia (BP&BIP) and bronchiolitis using features described in Table 1, which were adapted from Caswell (Caswell, 2016a).
Table 1.
Definition of histopathological criteria for different types of pneumonia
| Types of pneumonia | Features |
|---|---|
| Suppurative bronchopneumonia (SBP) | The exudate predominantly composed of neutrophils within the lumen of bronchi, bronchioles and alveoli with hyperaemia, cellular debris or hyperplasia of BALT or fibrosis |
| Fibrinous bronchopneumonia (FBP) | The exudate predominantly composed of fibrin within the lumen of bronchi, bronchioles and alveoli with oedema of inter‐lobular septa or coagulative necrosis or fibrosis |
| Interstitial pneumonia (IP) | Thickening of the alveolar septum by oedema, congestion or hypercellularity or hyperplasia of type II pneumocytes with hyaline membranes, interstitial fibrosis or BALT hyperplasia |
| Bronchointerstitial pneumonia (BIP) | The presence of features of both bronchiolar necrosis and IP |
| Bronchopneumonia + bronchointerstitial pneumonia (BP&BIP) | The presence of features of both BP (SBP or FBP) and BIP |
| Bronchiolitis | Necrosis of epithelial cells in bronchi or bronchioles with purulent or fibrinous exudates or accumulation of inflammatory cells, epithelial hyperplasia (squamous metaplasia) |
Abbreviation: BALT, bronchus‐associated lymphoid tissue.
This article is being made freely available through PubMed Central as part of the COVID-19 public health emergency response. It can be used for unrestricted research re-use and analysis in any form or by any means with acknowledgement of the original source, for the duration of the public health emergency.
2.5. Routine diagnostic tests for conventional pathogens associated with BRD
Various adjunct tests were performed by PDS according to submitting veterinarians' requests as part of the diagnostic investigation in individual cases.
DNA and RNA were extracted using DNeasy Blood & Tissue Kit (Qiagen) and RNeasy Mini kit (Qiagen) according to the manufacturer's instructions. Viral real‐time PCR was performed on 34 lung samples targeting common viruses including BHV1, BRSV, BVDV, BPIV3 and BCV following standardized protocols (Decaro et al., 2008; Hakhverdyan, Hagglund, Larsen, & Belak, 2005; Horwood & Mahony, 2011; Mahlum et al., 2002; Wang et al., 2008). Real‐time PCR for M. bovis was performed on 120 lung samples (Clothier et al., 2010).
To isolate M. haemolytica, H. somni, P. multocida and B. trehalosi, lung samples were cultured on 5% Columbia sheep blood and Chocolate agar plates, incubated at 35°C with 5% CO2 for 18 hr. Characteristics of bacterial colonies were observed such as yellow pigment for H. somni, β‐hemolysis for M. haemolytica and mucoid appearance for P. multocida. Matrix‐Assisted Laser Desorption and Ionization Time‐of‐Flight (MALDI‐TOF) Mass Spectrometry (MALDI Biotyper 3.1.66) was used for bacterial identification, with scores equal or greater than 2.0 considered positive identification (Karunarathna et al., 2017).
2.6. Statistical analysis
Binary logistic regressions using IBM SPSS Statistics (version 25) were implemented to analyse the association between microorganisms and types of pneumonia. Linear regressions using IBM SPSS Statistics (Version 25) were used to evaluate associations between the presence of the numbers of viruses and the presence of different bacteria. Specificity, sensitivity and agreement of detection of BHV1, BRSV, BVDV, BCV and BPIV3 between metagenomic sequencing and qPCR were determined using 2 × 2 tables. p < .05 is defined as statistical significance.
Samples were clustered according to the prevalence of pathogens using UPGMA (Unweighted Pair Group Method with Arithmetic Mean), and relationships visualized as a heatmap generated using heatmap.plus 1.3 in R (Williams et al., 2019). The distance matrix was created for hierarchical clustering using hclust function in R. The optimal number of clusters was determined by the elbow method and further confirmed with gap statistics using fviz_nbclust and combining clustering methods using NbClust function in R.
3. RESULTS
3.1. Cases description and histopathological patterns
A total of 130 cases were included in our study. The age of the cattle included ranged from 1 week to 8 years with a median age of 10 months. About 26.7% (35/130) of the cases were collected in November, the month with most cases, followed by December with 9.9% (13/130). Most (62.3%, 81/130) of the cases were from Alberta and 31.5% (41/130) from Saskatchewan, with 3.9% (5/130) from Manitoba and 2.3% (3/130) from British Columbia. Typical examples of the histopathological features corresponding to the classification system described in Table 1 are shown in Figure 1. Fibrinous bronchopneumonia (46.2%, 60/130) was the most frequent type of pneumonia, followed by SBP (16.9%, 22/130), BP&BIP (13.8%, 18/130), BIP (11.5%, 15/130) and IP (6.2%, 8/130). Cases showing only bronchiolitis (5.4%, 7/130) without pneumonia were least frequently diagnosed.
Figure 1.
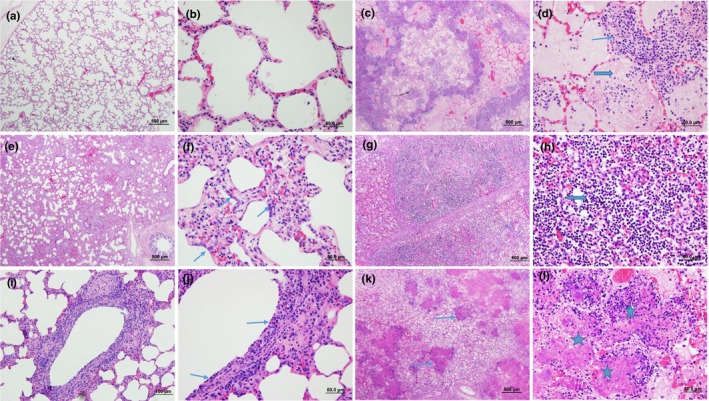
Histopathological features of bovine lungs with different types of pneumonia. (a) Normal lung in low magnification (LM) and (b) in high magnification (HM). (c) FBP with multifocal areas of necrosis, congestion and oedema and (d) infiltration of large numbers of oat cells (thin arrow) and fibrin (thick arrow) in alveoli. (e) IP with thickening of alveolar septa and infiltration of inflammatory cells and (f) type II pneumocytes hyperplasia (arrows). (g) and (h) SBP with predominate infiltration by large numbers of neutrophils (thick arrow). (i) and (j) bronchiolitis with necrotic epithelial cells, loss of cilia and inflammatory cells infiltrated mucosa and submucosa. (k) and (l) showed multifocal areas of coagulative necrosis characterized by hypereosinophilic materials and necrotic cellular debris (thin arrows and stars). Haematoxylin and eosin stain. (a), (c), (e), (g), (k): low magnification, 500 μm; (i): low magnification, 100 μm; (b), (d), (f), (j), (l): high magnification, 50 μm
3.2. Metagenomic identification of viruses
A total of 30.9 million sequence reads were generated. After removing low‐quality and host‐derived reads, 8.9 million reads remained. Viral reads of high quality (0.19 million) accounted for 0.61% of the total reads generated and 2.13% of the total high‐quality reads. A total of 16 viruses were identified. The largest viral contigs assembled from individual samples varied from 234 to 6,126 bp (Table 2). Similarity of the assembled contigs to reference sequences from the RefSeq virus database based on BLASTx were all above 93.7% amino acid identity, and most (9/16) were >97% identical to their closest match in the database. The numbers of reads aligned to each virus ranged from 2 to 77,240 reads, and there was a corresponding range of genome coverage observed for each virus in each individual sample with the proportion of the genome covered ranging from as low as 0.54% to almost complete coverage (>98%, UTPV1). Only viruses identified in at least two samples were included in statistical analyses.
Table 2.
Viruses identified by metagenomics
| Virus | Family | Genome size (bp) | Largest contig size (bp) from any individual sample | Largest contig % AA identity (protein) | Total number of reads from all samples |
|---|---|---|---|---|---|
| FAdV‐A | Adenoviridae | 43,804 | 235 | 100 (short fibre) | 2 |
| BAV | Astroviridae | 6,233 | 6,126 | 96.2 (nsp1a) | 23,350 |
| PeSV | Betaflexiviridae | 8,041 | 344 | 95.6 (replicase) | 8 |
| BCV | Coronaviridae | 308,845 | 4,952 | 98.4 (polyprotein) | 77,240 |
| BVDV1 | Flaviviridae | 12,258 | 1,125 | 94.9 (polyprotein) | 1,452 |
| BVDV2 | Flaviviridae | 12,476 | 1,113 | 99.5 (polyprotein) | 158 |
| IDV | Orthomyxoviridae | 12,546 | 251 | 98.8 (PB2) | 2 |
| BRSV | Paramyxoviridae | 15,140 | 2,244 | 100 (nucleocapsid) | 13,167 |
| BPIV3 | Paramyxoviridae | 15,537 | 2,151 | 95.7 (phosphoprotein) | 312 |
| BPV2 | Parvoviridae | 5,610 | 1,670 | 94.2 (capsid) | 262 |
| BPV3 | Parvoviridae | 5,100 | 612 | 99.4 (non‐structural) | 20 |
| UTPV1 | Parvoviridae | 5,108 | 5,010 | 99.9 (minor capsid) | 71,761 |
| AAAV | Parvoviridae | 4,684 | 203 | 96 (rep) | 4 |
| EVE | Picornaviridae | 7,414 | 251 | 98.8 (polyprotein) | 4 |
| BRBV | Picornaviridae | 7,556 | 234 | 93.7 (polyprotein) | 9 |
| BPyV | Polyomaviridae | 4,697 | 285 | 99.6 (agnoprotein) | 8 |
Abbreviations: AA, amino acid; AAAV, avian adeno‐associated virus; BAV, bovine astrovirus; BCV, bovine coronavirus; bp, base pairs; BPIV3, bovine parainfluenza virus 3; BPV 2 and 3, bovine parvovirus 2 and 3; BPyV, bovine polyomavirus; BRBV, bovine rhinitis virus B; BRSV, bovine respiratory syncytial virus; BVDV 1 and 2, bovine viral diarrhoea virus 1 and 2; EVE, Enterovirus E; FAdV‐A, fowl aviadenovirus; IDV, influenza D virus; PeSV, pea streak virus; UTPV1, ungulate tetraparvovirus 1.
This article is being made freely available through PubMed Central as part of the COVID-19 public health emergency response. It can be used for unrestricted research re-use and analysis in any form or by any means with acknowledgement of the original source, for the duration of the public health emergency.
For additional confirmation of virome sequencing results, available real‐time PCR results for BVDV, BPIV3, BRSV, BHV1 and BCV for 34 samples were compared with metagenomic sequencing results (Table S1). In all cases where one or more of these viruses was detected by metagenomic sequencing, the detection was confirmed by real‐time PCR, and however in four cases, a virus was detected by real‐time PCR that was not detected in the metagenomic data from that sample. When real‐time PCR was considered as the gold standard, the specificity of metagenomic sequencing was 100% and sensitivity was 60%. The agreement of identification of viruses between MiSeq and real‐time PCR was 91%.
3.3. Prevalence of microorganisms and their associations with BRD
The prevalence of viruses was generally low, with at least one virus detected in only 36.9% (48/130) of samples. Overall, bovine parvovirus 2 (BPV2) was the most prevalent virus (11.5%, 15/130), followed by ungulate tetraparvovirus 1 (UTPV1) and BRSV (both 8.5%, 11/130) (Figure 2). The conventional BRD viruses BRSV (8.5%, 11/130), BVDV1 and BVDV2 (2.3%, 3/130 and 3.8%, 5/130, respectively) and BPIV3 (2.3%, 3/130) were identified infrequently. Six animal viruses (bovine rhinitis B virus [BRBV], IDV, fowl aviadenovirus [FAdV‐A], avian adeno‐associated virus [AAAV] and bovine polyomavirus [BPyV]) were only identified in one sample each (Figure 2). The most prevalent virus in each type of pneumonia was BPV2 (6.9%, 9/130) in FBP, bovine astrovirus (BAV) (3.1%, 4/130) in SBP, BRSV (1.5%, 2/130) in IP, UTPV1 (1.5%, 2/130) in BIP, BVDV (1.5%, 2/130) in bronchiolitis, and BRSV and UTPV1 (1.5%, 2/130) in BP&BIP (Figure 3a). However, for every type of pneumonia, samples in which no virus was detected were the largest category (Figure 3a). Only BPV2 and BRSV were detected in all pneumonia categories with BPV2 detection rates varying from 0.8% to 6.9% and those of BRSV varying from 0.8% to 3.1% (Figure 3a).
Figure 2.
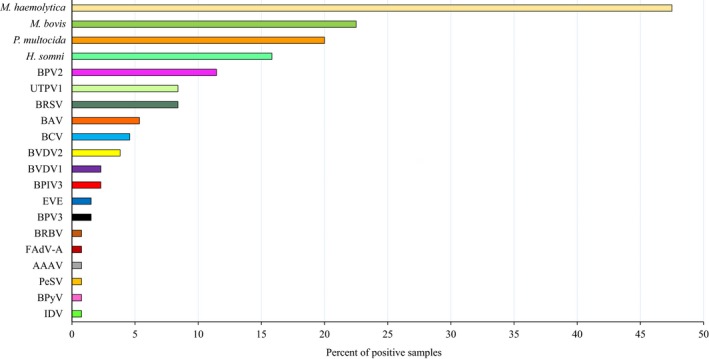
Prevalence of viruses and bacteria in bovine pneumonia lung samples. Viruses were investigated in 130 samples but bacterial results were only available for 120. AAAV, avian adeno‐associated virus; BAV, bovine astrovirus; BCV, bovine coronavirus; BPIV3, bovine parainfluenza virus 3; BPV 2 and 3, bovine parvovirus 2 and 3; BPyV, bovine polyomavirus; BRBV, bovine rhinitis virus B; BRSV, bovine respiratory syncytial virus; BVDV 1 and 2, bovine viral diarrhoea virus 1 and 2; EVE, Enterovirus E; FAdV‐A, fowl aviadenovirus; H. somni, Histophilus somni; IDV, influenza D virus; M. bovis, Mycoplasma bovis; M. haemolytica, Mannheimia haemolytica; P. multocida, Pasteurella multocida; PeSV, pea streak virus; UTPV1, ungulate tetraparvovirus 1
Figure 3.
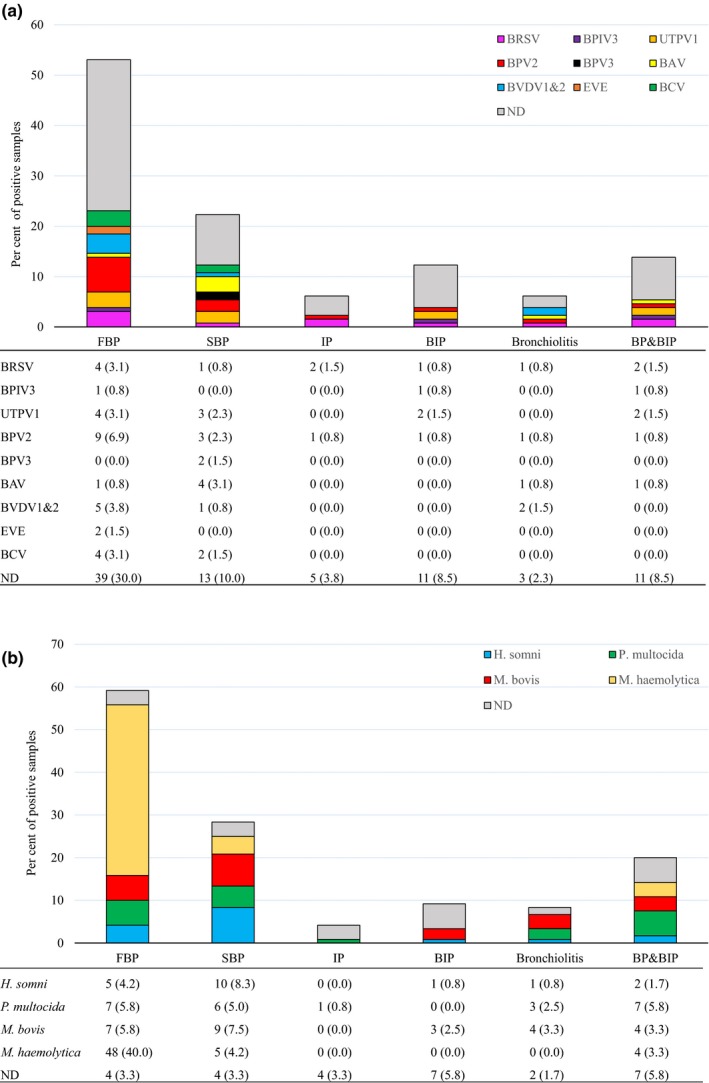
(a) The prevalence of identified viruses in each type of pneumonia (n = 130). The bar charts show the proportion of virus positive and ND samples assigned to each type of pneumonia by colour as illustrated in the legend. The lower table shows the number (per cent) of virus positive and ND samples out of 130 total samples. BAV, bovine astrovirus; BCV, bovine coronavirus; BPIV3, bovine parainfluenza virus 3; BPV 2 and 3, bovine parvovirus 2 and 3; BRSV, bovine respiratory syncytial virus; BVDV 1 and 2, bovine viral diarrhoea virus 1 and 2; EVE, Enterovirus E; ND, viruses not detected; UTPV1, ungulate tetraparvovirus 1. Only viruses identified in at least two samples are shown. (b) The prevalence of bacteria in each type of pneumonia (n = 120). The bar charts show the proportion of bacteria positive and ND samples assigned to each type of pneumonia by colour as illustrated in the legend. The lower table shows the number (per cent) of bacteria positive and ND samples out of the total cultured. H. somni, Histophilus somni; M. bovis, Mycoplasma bovis; M. haemolytica, Mannheimia haemolytica; ND, bacteria not detected; P. multocida, Pasteurella multocida
Complete bacteriological results including culture and real‐time PCR for M. bovis were only available for 120/130 lung samples. Among these samples, M. haemolytica (47.5%, 57/120) was the most prevalent bacterial species, followed by M. bovis (22.5%, 27/120), P. multocida (20.0%, 24/120) and H. somni (15.8%, 19/120) (Figure 2). The most prevalent bacterium in each type of pneumonia was M. haemolytica (40.0%, 48/120) in FBP, H. somni (8.3%, 10/120) in SBP, M. bovis (2.5%, 3/120) in BIP, M. bovis (3.3%, 4/120) in bronchiolitis and P. multocida (5.8%, 7/120) in BP&BIP (Figure 3b). In contrast to the virus findings, samples in which no bacteria were detected were the smallest category for each type of pneumonia except IP and BIP (Figure 3b).
The association between viruses and bacteria and different types of pneumonia was analysed using binary logistic regression. None of the viruses detected were significantly associated with any type of pulmonary pathology. M. haemolytica was positively associated with FBP (p = .001), H. somni with SBP (p = .003) and P. multocida with BP&BIP (p = .01) (Table S2).
The results from linear regression analysis showed that the number of virus species detected in an individual sample was positively associated with the presence of M. bovis (p = .002) but not with the presence of other BRD bacteria (data not shown). Furthermore, the presence of M. bovis showed statistically significant association with formation of bronchiolitis obliterans (p = .0001, data not shown).
In order to explore qualitatively whether any particular combinations of pathogens were associated with pneumonia types, a pathogen ‘profile’ for each sample was created based on positive or negative detection of the 13 viruses or bacteria that were detected in the 120 samples that were screened for both viruses and bacteria. Profiles were clustered using UPGMA, and clustering of the distribution patterns was visualized as a heatmap (Figure 4). The optimal number of clusters determined based on clustering algorithm was 5. Cluster 1 contained samples representing all pneumonia types in which none of the agents were detected. Cluster 3 (BRSV) and Cluster 5 (mostly mixed profiles including all agents except BRSV) also contained a mixture of samples from all pneumonia types. Cluster 2 consisted of a singleton sample in which only BAV was detected. Consistent with the analysis of associations of individual agents with pneumonia type, most (60/130) FBP profiles were found in Cluster 5, which was characterized by the detection of M. haemolytica. Interestingly, M. haemolytica was frequently observed in the absence of any other agents, unlike H. somni, P. multocida and M. bovis, which were more frequently detected along with at least one other virus or bacterium (Figure 4).
Figure 4.
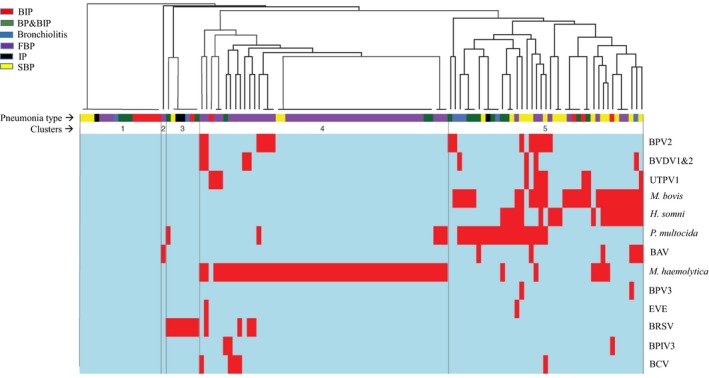
Clustering of 120 samples for which both virome and bacteriology data were available based on microbial profiles. The coloured blocks under the dendrogram indicate pneumonia type according to the legend. The five clustered supported by elbow analysis are indicated. The heatmap below the dendrogram shows pathogen profiles of individual samples including positive (red) or negative (blue) for each microorganism. BAV, bovine astrovirus; BCV, bovine coronavirus; BIP, bronchointerstitial pneumonia; BP&BIP, bronchopneumonia + bronchointerstitial pneumonia; BPIV3, bovine parainfluenza virus 3; BPV 2 and 3, bovine parvovirus 2 and 3; BRSV, bovine respiratory syncytial virus; BVDV 1 and 2, Bovine viral diarrhoea virus 1 and 2; EVE, Enterovirus E; FBP, fibrinous bronchopneumonia; H. somni, Histophilus somni; IP, interstitial pneumonia; M. bovis, Mycoplasma bovis; M. haemolytica, Mannheimia haemolytica; P. multocida, Pasteurella multocida; SBP, suppurative bronchopneumonia; UTPV1, ungulate tetraparvovirus 1
4. DISCUSSION
Advances in BRD epidemiology, pathogen genomics and proteomics, characterization of the respiratory tract microbiome and the development of experimental challenge models (Fulton, 2009) have led to an evolution of our understanding of the relationship of pneumonic lesions with infectious pathogens originally reported in the 1970s (Jensen et al., 1976b, 1976a). Most recently, application of advanced technologies for virus identification has resulted in further insight into the relationships between well‐recognized pathogens and BRD (Booker et al., 2008; Mehinagic et al., 2019; Oliveira et al., 2019). Previously, we detected 21 viruses in bovine nasal swabs and tracheal washes from western Canadian beef cattle, among which IDV, BRBV, BRSV and BCV were significantly associated with BRD (Zhang et al., 2019). In dairy calves, metagenomic sequencing showed a significant association between the presence of BRAV, IDV and bovine adenovirus 3 (BAdV3) in nasopharyngeal and pharyngeal samples with BRD (Ng et al., 2015). Furthermore, another study of characterization of virome of BRD in nasal swabs from feedlot cattle suggested IDV as an aetiology (Mitra et al., 2016). In order to investigate whether these viruses play direct roles in pulmonary lesions, the metagenomic virome of pneumonic lungs was characterized.
The viruses detected in lung tissue were similar to those previously detected in upper respiratory tract samples; however, the prevalence of viruses in the lungs in this study was lower than that in nasal swabs and tracheal washes. There are three possible explanations for the lower viral prevalence in lungs. Firstly, viruses may have already been cleared by the host by the time the animal died or was euthanized (Fulton et al., 2009). Cattle were usually treated repeatedly before death or the owner decided to euthanize for diagnostic purposes. At that point, viruses, if any, might have already been cleared, leaving only the pulmonary lesions. Secondly, some viruses, such as IDV (Ferguson et al., 2016), preferentially infect the upper respiratory tract, not the lung. In the current study, IDV was detected in the lung of only 1/130 cattle, compared with 16/116 upper respiratory tract samples in our previous study (Zhang et al., 2019). Similarly, UTPV1 was only detected in 8.5% (11/130) of pneumonic lungs, but was the most prevalent virus in nasal swabs and tracheal washes (36.2%, 42/116) (Zhang et al., 2019). Thirdly, the method used to deplete host DNA may not be as effective in tissues samples compared with nasal swabs and tracheal washes, where there is less host material. In fact, 38.8% of sequence reads from the lung samples were host‐derived, compared with only 27.7% of reads from nasal swabs and tracheal washes (Zhang et al., 2019). The relatively larger amounts of contaminating host DNA could contribute to a reduction in the analytical sensitivity for viruses. The accuracy of the virome sequencing results was supported by targeted real‐time PCR assays with 91% agreement between the methods.
Apart from the well‐documented conventional BRD viruses (BRSV, BVDV, BPIV3 and BHV1), several parvoviruses were identified in lungs including BPV2 (genus Copiparvovirus), UTPV1 (genus Tetraparvovirus), BPV3 (genus Erythroparvovirus) and avian adeno‐associated virus (AAAV, genus Dependoparvovirus), all of which have been identified and reported in cattle except AAAV (Cibulski et al., 2016). Bovine parvovirus 2 was first reported as a contaminant of commercial bovine sera, and BPV3 was frequently detected in common laboratory reagents (Allander, Emerson, Engle, Purcell, & Bukh, 2001). Although various parvoviruses have been detected in cattle, whether these viruses cause disease in cattle remain to be investigated. In our previous study, parvoviruses were not associated with BRD (Zhang et al., 2019).
BAV (5%) was the fourth most prevalent virus in the pneumonia lung virome. Although BAV showed no statistically significant association with any types of pneumonia, recent studies have demonstrated association of BAV with encephalitis in cattle (Selimovic‐Hamza, Sanchez, Philibert, Clark, & Seuberlich, 2017) and it is commonly identified in animal faeces (Li et al., 2011; Shan et al., 2011). Taken together, these findings indicate the potential importance of BAV in other body systems. Because all BAV‐positive cases also contained parvoviruses, M. haemolytica, P. multocida, H. somni or M. bovis, it remains possible that BAV synergizes with other viruses and bacteria to aggravate symptoms.
Concerns about IDV have been raised since its discovery, and it has now been found worldwide, particularly in cattle (Hause et al., 2014; Su, Fu, Li, Kerlin, & Veit, 2017). We first reported the identification of IDV in western Canada, and the prevalence was 13.8% in nasal swabs and tracheal washes from cattle with BRD (Zhang et al., 2019). In this study, the single IDV‐positive lung had bronchiolitis and was co‐infected by BRSV. It has been shown that IDV primarily infects the upper respiratory tract (Ferguson et al., 2016), and thus, the low prevalence of IDV in this study is not surprising and does not disprove its potential significance in BRD.
Considering the low rate of virus detection and the lack of association of any of the viruses detected with lung lesion type, post‐mortem virus detection in lung tissue is unlikely to be diagnostically informative and provides low analytical sensitivity relative to ante‐mortem sampling of the upper respiratory tract for virus surveillance.
Bacteria associated with BRD are mostly considered opportunistic pathogens (Caswell, 2016a). They are frequently isolated from the nasopharynx of healthy cattle, but can cause disease under some circumstances when host defences have been compromised by stress or viral infection (Caswell, 2014). Experimental models of bacterial pneumonia have been successfully established by infection with BHV1 or BVDV followed by M. haemolytica (Burciaga‐Robles et al., 2010; Jericho & Langford, 1978). We observed significant associations of bacteria with particular types of pneumonia. M. haemolytica was associated with FBP, consistent with well‐established knowledge (Caswell, 2016a). We also found that H. somni was associated with SBP (but not FBP), and P. multocida was associated with BP&BIP. The strong associations of H. somni and M. haemolytica with SBP and FBP, respectively, indicate that histopathology can be useful in differentiating different bacterial aetiologies. A recent study showed that aerosolized challenge of M. haemolytica consistently resulted in development of FBP, which may indicate that M. haemolytica can also be a primary pathogen (Bassel et al., 2019) although the study design did not rule out the infection of unconventional viruses such as IDV or BRBV as a contributing factor. Interestingly, in most cases where M. haemolytica was detected, no viruses or rare bacteria were detected (Figure 4), which was consistent with results of a previous study (Fulton et al., 2009). The association of P. multocida with BP&BIP suggests an opportunistic infection from a primary or concurrent viral infection; because the pathogenesis of BIP is thought to be initiated by epithelial damage by viruses or toxic gases (Autio et al., 2007; Caswell, 2016b), consistent with the idea, in case where P. multocida was detected, other viruses were also present (Figure 4).
Analysis of the relationship between the number of viruses and infection with different bacteria was conducted to investigate whether infection with multiple viruses predisposes a host to bacterial infection, or vice versa. We found that M. bovis infection was associated with the detection of higher numbers of viruses (p = .002). Further, bronchiolitis obliterans, which is regarded as a chronic lesion, was associated with M. bovis infection, consistent with results of a recent study using immunohistochemistry (IHC) to analyse association of histological features with infectious agents in dairy cows (Oliveira et al., 2019). It is possible that chronic M. bovis infection can compromise host immunity and lead to infection with multiple viruses, or alternatively, multiple viral infections could predispose cattle to M. bovis infection.
Our results demonstrate that there are strong associations between different types of histopathologic lesions and the presence of particular BRD‐associated bacteria. While the relatively low prevalence of viruses in pneumonic lungs limits the diagnostic utility of virus detection in this specimen, our results do confirm the effectiveness of the metagenomic sequencing technique itself and illustrate how its non‐targeted nature allows accurate and simultaneous detection of multiple viruses, which presents a distinct advantage over application of multiple targeted assays. Future development of robust methods for simultaneous detection of viruses, bacteria and fungi with metagenomic sequencing will further enable the application of this approach to both pathogenesis research and diagnostic investigations of complex clinical syndromes like BRD.
ETHICAL APPROVAL
Ethical statement is not applicable in this study because the bovine lung samples were submitted to Prairie Diagnostic Services Inc. (PDS, Saskatoon, Canada) for routine diagnostic purposes.
Supporting information
ACKNOWLEDGEMENTS
We are grateful for the help of Anju Tumber (Prairie Diagnostic Service Inc. PDS) with reagent purchasing and other logistics. We thank Dr. Sarah Parker, Scott Dos Santos, Salahuddin Khan and Dr. Maarten J. Voordouw (Western College of Veterinary Medicine, University of Saskatchewan) for support on statistical analyses. We also thank all the pathologists (PDS and Department of Veterinary Pathology, University of Saskatchewan) for helping to collect the lung samples.
Zhang M, Hill JE, Godson DL, Ngeleka M, Fernando C, Huang Y. The pulmonary virome, bacteriological and histopathological findings in bovine respiratory disease from western Canada. Transbound Emerg Dis. 2020;67:924–934. 10.1111/tbed.13419
REFERENCES
- Allander, T. , Emerson, S. U. , Engle, R. E. , Purcell, R. H. , & Bukh, J. (2001). A virus discovery method incorporating DNase treatment and its application to the identification of two bovine parvovirus species. Proceedings of the National Academy of Sciences of the United States of America, 98, 11609–11614. 10.1073/pnas.211424698 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allander, T. , Tammi, M. T. , Eriksson, M. , Bjerkner, A. , Tiveljung‐Lindell, A. , & Andersson, B. (2005). Cloning of a human parvovirus by molecular screening of respiratory tract samples. Proceedings of the National Academy of Sciences of the United States of America, 102, 12891–12896. 10.1073/pnas.0504666102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Autio, T. , Pohjanvirta, T. , Holopainen, R. , Rikula, U. , Pentikainen, J. , Huovilainen, A. , … Pelkonen, S. (2007). Etiology of respiratory disease in non‐vaccinated, non‐medicated calves in rearing herds. Veterinary Microbiology, 119, 256–265. 10.1016/j.vetmic.2006.10.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bassel, L. L. , Kaufman, E. I. , Alsop, S. N. A. , Stinson, K. J. , Hewson, J. , Sharif, S. , … Caswell, J. L. (2019). Development of an aerosolized Mannheimia haemolytica experimental pneumonia model in clean‐catch colostrum‐deprived calves. Veterinary Microbiology, 234, 34–43. 10.1016/j.vetmic.2019.05.012 [DOI] [PubMed] [Google Scholar]
- Bolger, A. M. , Lohse, M. , & Usadel, B. (2014). Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics, 30, 2114–2120. 10.1093/bioinformatics/btu170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Booker, C. W. , Abutarbush, S. M. , Morley, P. S. , Jim, G. K. , Pittman, T. J. , Schunicht, O. C. , … Janzen, E. D. (2008). Microbiological and histopathological findings in cases of fatal bovine respiratory disease of feedlot cattle in Western Canada. Canadian Veterinary Journal, 49, 473–481. [PMC free article] [PubMed] [Google Scholar]
- Brister, J. R. , Ako‐Adjei, D. , Bao, Y. , & Blinkova, O. (2015). NCBI viral genomes resource. Nucleic Acids Research, 43, D571–D577. 10.1093/nar/gku1207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brodersen, B. W. (2010). Bovine respiratory syncytial virus. The Veterinary Clinics of North America: Food Animal Practice, 26, 323–333. 10.1016/j.cvfa.2010.04.010 [DOI] [PubMed] [Google Scholar]
- Brodersen, B. W. (2014). Bovine viral diarrhea virus infections: Manifestations of infection and recent advances in understanding pathogenesis and control. Veterinary Pathology, 51, 453–464. 10.1177/0300985813520250 [DOI] [PubMed] [Google Scholar]
- Buermans, H. P. , & den Dunnen, J. T. (2014). Next generation sequencing technology: Advances and applications. Biochimica et Biophysica Acta, 1842, 1932–1941. 10.1016/j.bbadis.2014.06.015 [DOI] [PubMed] [Google Scholar]
- Burciaga‐Robles, L. O. , Step, D. L. , Krehbiel, C. R. , Holland, B. P. , Richards, C. J. , Montelongo, M. A. , … Fulton, R. W. (2010). Effects of exposure to calves persistently infected with bovine viral diarrhea virus type 1b and subsequent infection with Mannheima haemolytica on clinical signs and immune variables: Model for bovine respiratory disease via viral and bacterial interaction. Journal of Animal Science, 88, 2166–2178. 10.2527/jas.2009-2005 [DOI] [PubMed] [Google Scholar]
- Caswell, J. L. (2014). Failure of respiratory defenses in the pathogenesis of bacterial pneumonia of cattle. Veterinary Pathology, 51, 393–409. 10.1177/0300985813502821 [DOI] [PubMed] [Google Scholar]
- Caswell, J. L. e. a. (2016a). Respiratory system In Maxie M. G. (Ed.), Jubb, Kennedy, and Palmer's pathology of domestic animals (6th ed., Vol. 2, pp. 465–591). St. Louis, MO: Elsevier. [Google Scholar]
- Caswell, J. L. e. a. R. s. I. M. M. (Eds.) (2016b). Jubb, Kennedy, and Palmer's pathology of domestic animals (6th ed., Vol. 2, pp. 465–591). St. Louis, MO: Elsevier. [Google Scholar]
- Cibulski, S. P. , Teixeira, T. F. , dos Santos, H. F. , de Sales Lima, F. E. , Scheffer, C. M. , Varela, A. P. M. , … Roehe, P. M. (2016). Ungulate copiparvovirus 1 (bovine parvovirus 2): Characterization of a new genotype and associated viremia in different bovine age groups. Virus Genes, 52, 134–137. 10.1007/s11262-015-1266-x [DOI] [PubMed] [Google Scholar]
- Clothier, K. A. , Jordan, D. M. , Thompson, C. J. , Kinyon, J. M. , Frana, T. S. , & Strait, E. L. (2010). Mycoplasma bovis real‐time polymerase chain reaction assay validation and diagnostic performance. Journal of Veterinary Diagnostic Investigation, 22, 956–960. 10.1177/104063871002200618 [DOI] [PubMed] [Google Scholar]
- Decaro, N. , Elia, G. , Campolo, M. , Desario, C. , Mari, V. , Radogna, A. , … Buonavoglia, C. (2008). Detection of bovine coronavirus using a TaqMan‐based real‐time RT‐PCR assay. Journal of Virological Methods, 151, 167–171. 10.1016/j.jviromet.2008.05.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellis, J. A. (2010). Bovine parainfluenza‐3 virus. The Veterinary Clinics of North America: Food Animal Practice, 26, 575–593. 10.1016/j.cvfa.2010.08.002 [DOI] [PubMed] [Google Scholar]
- Ferguson, L. , Olivier, A. K. , Genova, S. , Epperson, W. B. , Smith, D. R. , Schneider, L. , … Wan, X. F. (2016). Pathogenesis of influenza D virus in cattle. Journal of Virology, 90, 5636–5642. 10.1128/JVI.03122-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fulton, R. W. (2009). Bovine respiratory disease research (1983–2009). Animal Health Research Reviews, 10, 131–139. 10.1017/S146625230999017X [DOI] [PubMed] [Google Scholar]
- Fulton, R. W. , Blood, K. S. , Panciera, R. J. , Payton, M. E. , Ridpath, J. F. , Confer, A. W. , … Reck, A. (2009). Lung pathology and infectious agents in fatal feedlot pneumonias and relationship with mortality, disease onset, and treatments. Journal of Veterinary Diagnostic Investigation, 21, 464–477. 10.1177/104063870902100407 [DOI] [PubMed] [Google Scholar]
- Fulton, R. W. , d'Offay, J. M. , Landis, C. , Miles, D. G. , Smith, R. A. , Saliki, J. T. , … Payton, M. E. (2016). Detection and characterization of viruses as field and vaccine strains in feedlot cattle with bovine respiratory disease. Vaccine, 34, 3478–3492. 10.1016/j.vaccine.2016.04.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grabherr, M. G. , Haas, B. J. , Yassour, M. , Levin, J. Z. , Thompson, D. A. , Amit, I. , … Regev, A. (2011). Full‐length transcriptome assembly from RNA‐Seq data without a reference genome. Nature Biotechnology, 29, 644–652. 10.1038/nbt.1883 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffin, D. , Chengappa, M. M. , Kuszak, J. , & McVey, D. S. (2010). Bacterial pathogens of the bovine respiratory disease complex. The Veterinary Clinics of North America: Food Animal Practice, 26, 381–394. 10.1016/j.cvfa.2010.04.004 [DOI] [PubMed] [Google Scholar]
- Hakhverdyan, M. , Hagglund, S. , Larsen, L. E. , & Belak, S. (2005). Evaluation of a single‐tube fluorogenic RT‐PCR assay for detection of bovine respiratory syncytial virus in clinical samples. Journal of Virological Methods, 123, 195–202. 10.1016/j.jviromet.2004.09.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hause, B. M. , Collin, E. A. , Liu, R. , Huang, B. , Sheng, Z. , Lu, W. , … Li, F. (2014). Characterization of a novel influenza virus in cattle and Swine: Proposal for a new genus in the Orthomyxoviridae family. MBio, 5, e00031‐00014 10.1128/mBio.00031-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hilton, W. M. (2014). BRD in 2014: Where have we been, where are we now, and where do we want to go? Animal Health Research Reviews, 15, 120–122. 10.1017/s1466252314000115 [DOI] [PubMed] [Google Scholar]
- Horwood, P. F. , & Mahony, T. J. (2011). Multiplex real‐time RT‐PCR detection of three viruses associated with the bovine respiratory disease complex. Journal of Virological Methods, 171, 360–363. 10.1016/j.jviromet.2010.11.020 [DOI] [PubMed] [Google Scholar]
- Jensen, R. , Pierson, R. E. , Braddy, P. M. , Saari, D. A. , Lauerman, L. H. , England, J. J. , … McChesney, A. E. (1976a). Diseases of yearling feedlot cattle in Colorado. Journal of the American Veterinary Medical Association, 169, 497–499. [PubMed] [Google Scholar]
- Jensen, R. , Pierson, R. E. , Braddy, P. M. , Saari, D. A. , Lauerman, L. H. , England, J. J. , … Christie, R. M. (1976b). Shipping fever pneumonia in yearling feedlot cattle. Journal of the American Veterinary Medical Association, 169, 500–506. [PubMed] [Google Scholar]
- Jericho, K. W. , & Langford, E. V. (1978). Pneumonia in calves produced with aerosols of bovine herpesvirus 1 and Pasteurella haemolytica . Canadian Journal of Comparative Medicine, 42, 269–277. [PMC free article] [PubMed] [Google Scholar]
- Jones, C. , & Chowdhury, S. (2010). Bovine herpesvirus type 1 (BHV‐1) is an important cofactor in the bovine respiratory disease complex. The Veterinary Clinics of North America: Food Animal Practice, 26, 303–321. 10.1016/j.cvfa.2010.04.007 [DOI] [PubMed] [Google Scholar]
- Karunarathna, R. , Popowich, S. , Wawryk, M. , Chow‐Lockerbie, B. , Ahmed, K. A. , Yu, C. , … Gomis, S. (2017). Increased incidence of enterococcal infection in nonviable broiler chicken embryos in Western Canadian hatcheries as detected by matrix‐assisted laser desorption/ionization‐time‐of‐flight mass spectrometry. Avian Diseases, 61, 472–480. 10.1637/11678-052317-Reg.1 [DOI] [PubMed] [Google Scholar]
- Langmead, B. , & Salzberg, S. L. (2012). Fast gapped‐read alignment with Bowtie 2. Nature Methods, 9, 357–359. 10.1038/nmeth.1923 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larson, R. L. , & Step, D. L. (2012). Evidence‐based effectiveness of vaccination against Mannheimia haemolytica, Pasteurella multocida, and Histophilus somni in feedlot cattle for mitigating the incidence and effect of bovine respiratory disease complex. The Veterinary Clinics of North America. Food Animal Practice, 28, 97–106.e7, 106e101‐107, ix. 10.1016/j.cvfa.2011.12.005 [DOI] [PubMed] [Google Scholar]
- Li, H. , Handsaker, B. , Wysoker, A. , Fennell, T. , Ruan, J. , Homer, N. , … Durbin, R. (2009). The sequence alignment/map format and SAMtools. Bioinformatics, 25, 2078–2079. 10.1093/bioinformatics/btp352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li, L. , Shan, T. , Wang, C. , Côté, C. , Kolman, J. , Onions, D. , … Delwart, E. (2011). The fecal viral flora of California Sea lions. Journal of Virology, 85, 9909–9917. 10.1128/jvi.05026-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahlum, C. E. , Haugerud, S. , Shivers, J. L. , Rossow, K. D. , Goyal, S. M. , Collins, J. E. , & Faaberg, K. S. (2002). Detection of bovine viral diarrhea virus by TaqMan reverse transcription polymerase chain reaction. Journal of Veterinary Diagnostic Investigation, 14, 120–125. 10.1177/104063870201400205 [DOI] [PubMed] [Google Scholar]
- Mehinagic, K. , Pilo, P. , Vidondo, B. , & Stokar‐Regenscheit, N. (2019). Coinfection of Swiss cattle with bovine parainfluenza virus 3 and Mycoplasma bovis at acute and chronic stages of bovine respiratory disease complex. Journal of Veterinary Diagnostic Investigation, 31, 674–680. 10.1177/1040638719861686 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitra, N. , Cernicchiaro, N. , Torres, S. , Li, F. , & Hause, B. M. (2016). Metagenomic characterization of the virome associated with bovine respiratory disease in feedlot cattle identified novel viruses and suggests an etiologic role for influenza D virus. Journal of General Virology, 97, 1771–1784. 10.1099/jgv.0.000492 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng, T. F. , Kondov, N. O. , Deng, X. , Van Eenennaam, A. , Neibergs, H. L. , & Delwart, E. (2015). A metagenomics and case‐control study to identify viruses associated with bovine respiratory disease. Journal of Virology, 89, 5340–5349. 10.1128/JVI.00064-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliveira, T. E. S. , Pelaquim, I. F. , Flores, E. F. , Massi, R. P. , Valdiviezo, M. J. J. , Pretto‐Giordano, L. G. , … Headley, S. A. (2019). Mycoplasma bovis and viral agents associated with the development of bovine respiratory disease in adult dairy cows. Transboundary and Emerging Diseases, 10.1111/tbed.13223 [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Portis, E. , Lindeman, C. , Johansen, L. , & Stoltman, G. (2012). A ten‐year (2000–2009) study of antimicrobial susceptibility of bacteria that cause bovine respiratory disease complex–Mannheimia haemolytica, Pasteurella multocida, and Histophilus somni–in the United States and Canada. Journal of Veterinary Diagnostic Investigation, 24, 932–944. 10.1177/1040638712457559 [DOI] [PubMed] [Google Scholar]
- Selimovic‐Hamza, S. , Sanchez, S. , Philibert, H. , Clark, E. G. , & Seuberlich, T. (2017). Bovine astrovirus infection in feedlot cattle with neurological disease in western Canada. Canadian Veterinary Journal, 58, 601–603. [PMC free article] [PubMed] [Google Scholar]
- Shan, T. , Li, L. , Simmonds, P. , Wang, C. , Moeser, A. , & Delwart, E. (2011). The fecal virome of pigs on a high‐density farm. Journal of Virology, 85, 11697–11708. 10.1128/jvi.05217-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su, S. , Fu, X. , Li, G. , Kerlin, F. , & Veit, M. (2017). Novel Influenza D virus: Epidemiology, pathology, evolution and biological characteristics. Virulence, 8, 1580–1591. 10.1080/21505594.2017.1365216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, J. , O'Keefe, J. , Orr, D. , Loth, L. , Banks, M. , Wakeley, P. , … Kerkhofs, P. (2008). An international inter‐laboratory ring trial to evaluate a real‐time PCR assay for the detection of bovine herpesvirus 1 in extended bovine semen. Veterinary Microbiology, 126, 11–19. 10.1016/j.vetmic.2007.06.005 [DOI] [PubMed] [Google Scholar]
- Williams, J. R. , Yang, R. , Clifford, J. L. , Watson, D. , Campbell, R. , Getnet, D. , … Jett, M. (2019). Functional Heatmap: An automated and interactive pattern recognition tool to integrate time with multi‐omics assays. BMC Bioinformatics, 20, 81 10.1186/s12859-019-2657-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, M. , Hill, J. E. , Fernando, C. , Alexander, T. W. , Timsit, E. , van der Meer, F. , & Huang, Y. (2019). Respiratory viruses identified in western Canadian beef cattle by metagenomic sequencing and their association with bovine respiratory disease. Transboundary and Emerging Diseases, 66, 1379–1386. 10.1111/tbed.13172 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials


