Abstract
In this study, the pathogenicity of porcine deltacoronavirus (PDCoV) strain NH (passage 10, P10) was evaluated. We found that PDCoV strain NH is enteropathogenic in 5‐day‐old pigs. Pathogenicity experiments provided a challenge model for studying the protection efficiency of passive immunity. In order to investigate the protective efficacy of passive immunity in newborn piglets, pregnant sows were vaccinated with either a PDCoV‐inactivated vaccine at the Houhai acupoint (n = 5) or DMEM as a negative control (n = 2) using a prime/boost strategy 20 and 40 days before delivery. PDCoV spike (S)‐specific IgG and neutralizing antibody (NA) responses were detected in immunized sows and piglets born to immunized sows. PDCoV spike (S)‐specific sIgA was also detected in the colostrum and milk of immunized sows. Five days post‐farrowing, piglets were orally challenged with PDCoV strain NH (105 TCID50/piglet). Severe diarrhoea, high levels of viral RNA copies and substantial intestinal villus atrophy were detected in piglets born to unimmunized sows. Only 4 of 31 piglets (12.9%) born to immunized sows in the challenge group displayed mild to moderate diarrhoea, lower viral RNA copies and minor intestinal villi damage compared to piglets born to unimmunized sows post‐challenge. Mock piglets exhibited no typical clinical symptoms. The challenge experiment results indicated that the inactivated PDCoV vaccine exhibited 87.1% protective efficacy in the piglets. These findings suggest that the inactivated PDCoV vaccine has the potential to be an effective vaccine, providing protection against virulent PDCoV.
Keywords: inactivated PDCoV vaccine, passive immunity, pathogenicity, piglets, sows
1. INTRODUCTION
Porcine deltacoronavirus (PDCoV) belongs to the genus Deltacoronavirus genus of the Coronaviridae family, which is the causative agent of the contagious gastrointestinal swine disease (Jung, Hu, & Saif, 2016; Song et al., 2015; Zhang, 2016). PDCoV was first reported in 2012 in Hong Kong during an epidemiological investigation conducted to identify new coronaviruses existing in nature (Woo et al., 2012). In early 2014, there was a reported PDCoV outbreak in the pig farms in the United States (Chen et al., 2015; Wang, Byrum, & Zhang, 2014), followed by subsequent outbreaks in Canada (Ojkic et al., 2015), South Korea (Lee et al., 2016), Thailand (Janetanakit et al., 2016; Saeng‐chuto et al., 2017) and mainland China (Dong et al., 2015), exhibiting a global distribution trend. Additionally, clinical reports have indicated that PDCoV exhibits enteropathogenicity, causing severe diarrhoea and vomiting in roughly 5‐ to 10‐day‐old gnotobiotic and conventional piglets (Chen et al., 2015; Jung et al., 2015). Pathological damage to the intestine, primarily in the jejunum and ileum, was characterized by intestinal villi atrophy and shortening and was confirmed by the pathogenicity experiments (Jung et al., 2015; Wang, Hayes, Sarver, Byrum, & Zhang, 2016). Such changes are clinically difficult to distinguish from the pathological changes caused by the porcine epidemic diarrhoea virus (PEDV) and transmissible gastroenteritis virus (TGEV) (Jung et al., 2016; Zhang, 2016). PDCoV infections have resulted in great economic losses for the global swine industry. Therefore, fast and effective preventive measures are essential for the prevention and control of PDCoV. Currently, implementing vaccines remain the most effective means of disease control; however, there are no commercial vaccines available for PDCoV.
Due to their immature immune system, neonatal piglets are highly susceptible to viral infection during their first few weeks of life. Studies suggest that passive immunity is the most effective approach for protecting piglets from viral infection (Langel, Paim, Lager, Vlasova, & Saif, 2016; Leidenberger et al., 2017). Immunized sows can transfer antibodies against enteroviruses (e.g. PEDV, TGEV and porcine rotavirus) to neonatal piglets through their colostrum and milk. The protective efficiency of passive immunization has a high positive correlation with antibody levels in the colostrum and milk (Sestak, Lanza, Park, Weilnau, & Saif, 1996). Therefore, passive immunity of newborn piglets can be achieved by immunizing sows that produce high levels of neutralizing antibodies (NA) and transfer these antibodies through the colostrum and milk to the nursing piglets, which may be an effective means of controlling viral infection.
In this study, a challenge model was established and the results indicated that PDCoV strain NH (P10) is pathogenic to 5‐day‐old specific pathogen‐free (SPF) piglets. Then, an inactivated PDCoV vaccine was prepared and the immune responses and protective efficiency of the inactivated PDCoV vaccine in pregnant sows was evaluated. After two doses of the vaccine, pregnant sows produced strong IgG and NA responses specific to PDCoV S proteins. High levels of IgG antibodies and NA were also detected in the serum of neonatal piglets born to immunized sows, which suggests that the antibodies were successfully transferred through the colostrum and milk. Five‐day‐old piglets were challenged with virulent PDCoV to assess the protective efficacy of the vaccine, and the findings indicated that the inactivated PDCoV vaccine provided 87.1% protective efficacy.
2. MATERIALS AND METHODS
2.1. Virus, cells and inactivated vaccine
PDCoV strain NH was isolated from PDCoV‐positive specimens with LLC‐PK cells (ATCC® CL‐101™), and plaques were purified twice. PDCoV strain NH was continuously passaged in swine testis (ST) cells in DMEM containing 10 μg/ml tosylsulfonyl phenylalanyl chloromethyl ketone (TPCK)‐trypsin (Invitrogen). At 36 hr post‐infection, both the supernatant and cells were harvested, titrated and stored at −70°C for future analyses. The titre of PDCoV strain NH (10th passage, P10) was 105 TCID50/mL, which was used for the challenge experiment. The inactivated PDCoV vaccine was prepared using the 15th generation of viruses due to its low mutation rate and similar antigenicity compared to the parent virus. Viral culture supernatants were inactivated with beta‐propiolactone containing aluminium hydroxide adjuvant at a 1:1 ratio in order to prepare the inactivated PDCoV vaccine.
2.2. Pathogenicity of the PDCoV strain NH
Ten 5‐day‐old SPF piglets were confirmed negative for PDCoV, PEDV, TEGV and RPV by virus‐specific PCR. Pigs were maintained in germ‐free isolation units of the animal facility located at the Harbin Veterinary Research Institute under standard conditions prescribed by the Institutional Guidelines. Piglets were fed suckling piglet formula every 3 hr. Six of the 10 SPF piglets (piglets #245, #246, #248, #241, #242 and #249) were assigned to the PDCoV‐inoculation group, which were orally inoculated with the PDCoV strain NH (P10) (104 TCID50/pig). The remaining 4 piglets (piglets #244, #247, #251 and #252) were orally inoculated with volume‐matched virus‐negative culture medium and served as the negative control group. The clinical signs of vomiting and diarrhoea were evaluated every 12 hr. The level of diarrhoea severity was scored for each piglet using the following criteria: 0 = no vomiting or diarrhoea; 1 = mild; 2 = moderate; 3 = severe. Faecal swabs were collected for detecting viral RNA. Duplicate tissues for the duodenum, jejunum, ileum, caecum, colon and rectum were collected for viral RNA detection and histological examinations.
2.3. Immunization of pregnant sows
Seven sows were confirmed to be negative for PDCoV, PEDV, TEGV and RPV using virus‐specific PCR and a serum neutralization test. All of the sows were randomly assigned to 2 experimental groups: (1) the immunization group (PDCoV‐inactivated vaccine; n = 5); or (2) the control group (DMEM; n = 2). Sows from the immunization group were immunized with 3 ml inactivated PDCoV vaccine (PDCoV strain NH P15, equivalent to 10 × 105 TCID50) at the Houhai acupoint (a concave part between the anus and tail). Vaccination in the Houhai acupoint was found to be helpful for inducing humoral and mucosal immune responses (Li et al., 2018; Liu, Tan, Wan, Zuo, & Liu, 1998). All of the sows were immunized 40 days prior to delivery, and a booster was administered 20 days before delivery. Sows from the control group were immunized with 3 ml DMEM as described above (Table 1). Serum was collected from sows 0 (prior to the first immunization), 20, 40, 47, 54 and 61 days post‐immunization. Serum samples were inactivated at 56°C for 30 min and stored at −70°C for future analyses. Milk was collected from the sows 1–5 days post‐farrowing.
Table 1.
Immune procedure
| Group | No. | No. of piglets | Treatment | Day of immunization | Route | Dose (TCID50) |
|---|---|---|---|---|---|---|
| Immunization group | #68 | 12 | Inactivated PDCoV vaccine | 40 and 20 days before delivery | Houhai acupoint | 4 × 106 TCID50; 3 ml |
| #72 | 10 | |||||
| #100 | 12 | |||||
| #56 | 14 | |||||
| #2,736 | 10 | |||||
| Control group | #5 | 13 | DMEM | 40 and 20 days before delivery | Houhai acupoint | 3 ml |
| #2,750 | 11 |
This article is being made freely available through PubMed Central as part of the COVID-19 public health emergency response. It can be used for unrestricted research re-use and analysis in any form or by any means with acknowledgement of the original source, for the duration of the public health emergency.
2.4. Challenge and protection assay
Three 5‐day‐old piglets from each PDCoV‐vaccinated gilts or mock gilts were permitted to continue drinking breast milk in order to detect the level of serum antibodies in piglets, while the other piglets were separated from the sows and housed in the experimental animal centre. Separated piglets were fed suckling piglet formula every 3 hr. Newborn piglets (2 or 4 piglets from each sow were set as the control group) derived from PDCoV‐vaccinated gilts or mock gilts were challenged orally with 105 TCID50 of PDCoV strain NH (P10). The clinical signs of vomiting and diarrhoea were evaluated daily. Diarrhoea severity was scored for each piglet using the following criteria: 0 = no vomiting or diarrhoea; 1 = mild; 2 = moderate; 3 = severe. Faecal swabs were collected for the detection of viral RNA. Serum was collected from the breastfed piglets 5, 12, 19 and 26 days post‐birth. The study protocol was approved by the Institutional Animal Care and Use Committee of the Harbin Veterinary Research Institute.
2.5. Viral extraction and RT‐qPCR
Faecal swabs were centrifuged at 5,000×g for 10 min at 4°C, and 140 μl supernatant was collected for viral RNA extraction. Viral RNA was extracted using a QIAamp® Viral RNA mini kit (QIAGEN, Hilden, Germany) following the manufacturer's instructions. One hundred mg tissue samples from each piglet were ground in liquid nitrogen, and the total RNA was extracted using an RNAiso Plus kit (Takara, Kusatsu, Japan) following the manufacturer's instructions. RNA was then used to perform real‐time (RT)‐qPCR using specific primers and probes (PDCoV‐N‐F: CGCTTAACTCCGCCATCAA; PDCoV‐N‐R: TCTGGTGTAACGCAGCCAGTA; PDCoV‐N‐probe: FAM‐CCCGTTGAAAACC‐MGB) as previously described with minor modifications (Ma et al., 2015). Briefly, 2 μl RNA was used in a 20 μl PCR reaction system using a One Step PrimeScript™ RT‐PCR kit (Takara, Kusatsu, Japan) in a LightCycler 480 (Roche Applied Science, IN, USA) under the following conditions: one cycle at 95°C for 5 min and 95°C for 10 s, followed by 40 cycles at 95°C for 5 s and 60°C for 20 s.
2.6. Immunofluorescence staining
The duodenum, jejunum, ileum, caecum, colon and rectum tissues were cut into 10‐μm‐thick sections, mounted onto glass slides and blocked with 5% non‐fat dry milk in PBS for 60 min at 37°C. Then, slides were incubated for 60 min with a mouse polyclonal anti‐PDCoV S antibody followed by incubation with an Alexa Fluor® 680 Donkey Anti‐Mouse IgG (Sigma‐Aldrich, MO, USA) for 60 min. Nuclei were stained with DAPI, and samples were observed with an inverted fluorescence microscope.
2.7. Immunohistochemistry
Tissues from intestinal tissues were fixed in formalin for 48 hr and embedded in paraffin wax following standard methods. IHC detection of PDCoV antigens was performed using an anti‐PDCoV‐N monoclonal antibody prepared in the laboratory followed by an incubation period with horseradish peroxidase (HRP)‐conjugated sheep anti‐mouse IgG (Sigma‐Aldrich) for 40 min at room temperature. Reactions were developed with 3,3'‐diaminobenzidine (DAB). Distilled water was used to finish staining and stained with haematoxylin (HE). Dehydration, clearing and mounting were conducted with neutral gums.
2.8. Enzyme‐linked immunosorbent assay
The extracellular domain of the PDCoV spike gene was amplified and cloned into the pCAGG vector with a C‐terminal flag tag. Recombinant S protein was expressed in 293T cells, purified using anti‐DYKDDDDK G1 Affinity Resin (GenScript: L00432‐1) and used as the coating antigens. PDCoV S‐specific IgG, IgA and sIgA antibody responses elicited by immunization with the inactivated vaccine were assessed by an indirect enzyme‐linked immunosorbent assay (ELISA). Optimal assay conditions (i.e. antigen coating concentration, serum and sow milk dilutions, and secondary dilutions) were determined by a checkerboard titration. Optimal coating concentrations for IgG, IgA and sIgA were 0.28, 0.28 and 0.07 μg/ml, respectively. Next, 96‐well polystyrene microplates were coated with the optimal antigen in a bicarbonate/carbonate coating buffer overnight. Plates were washed three times with PBST (PBS with 0.05% Tween 20) and blocked with 5% non‐fat dry milk at 37°C for 2 hr. Plates were washed 3 times with PBST, diluted in either serum or sow milk (1:100, 100 μl/well) and incubated at 37°C for 1 hr followed by incubation with a streptavidin‐HRP‐conjugated IgG or IgA antibody (1:10,000) at 37°C for 1 hr. After washing 3 times with PBST, a mouse anti‐Fc fragment of sIgA molecule antibody (1:10,000) was added for the detection of sIgA followed by incubation with an HRP‐conjugated anti‐mouse IgG antibody (1:10,000). Reactions were developed with 3,3′,5,5′‐Tetramethylbenzidine (TMB) for 15 min at room temperature and terminated with 2M‐H2SO4. OD values were determined at 450 nm using a microplate reader. PDCoV IgG and IgA negative serum samples and PDCoV‐negative sow milk samples were collected then confirmed by an immunofluorescence assay (IFA) in order to determine the cut‐off value.
2.9. Neutralization antibodies
The levels of sow and piglet serum neutralizing antibodies were determined using PDCoV strain NH (P10) with a virus neutralization test (VNT). In order to perform the assay, serum was heated at 56°C for 30 min for complement inactivation. Next, 100 μL of twofold serially diluted serum was mixed with 100 μl DMEM containing 100 TCID50 PDCoV virus and incubated at 37°C for 1 hr. Then, 200 μl mixture was added to the ST cells in four wells of a 96‐well cell culture plate. VNT was determined by the highest dilution of serum that exhibited at least 50% cytopathic effect when compared to the negative control.
2.10. Statistical analyses
Data were analysed by Student's t test. A threshold of p < .05 was considered to be significant. The antibody levels of the piglets are presented as box and whisker plots created with GraphPad Prism v7. Values are reported as the mean ± standard deviation (SD).
3. RESULTS
3.1. Pathogenicity of PDCoV strain NH
In order to confirm the pathogenicity of PDCoV strain NH, the cell culture‐adapted PDCoV strain NH (P10) in 5‐day‐old SPF piglets was investigated. At 28 hr post‐infection, piglets exhibited a range of mild to moderate degrees of diarrhoea (Table 2) (Figure 1a(A)). No vomiting or mortality was observed in the PDCoV‐inoculated pigs, which differs from other reports. Dilated and thin intestinal walls were observed in piglets terminated at 48 hr post‐inoculation (hpi) (Figure 1a(B)).
Table 2.
Pathogenesis and clinical signs of 5‐day‐old SPF pigs inoculated with the PDCoV strain NH
| Group | Pig no. | Diarrhoea scorea | Gross pathology scorec | |||||
|---|---|---|---|---|---|---|---|---|
| 12 hr | 24 hr | 36 hr | 48 hr | 60 hr | 72 hr | |||
| Infected | 245 | 0 | 1 | 3 | 3 | 3 | 3 | 3 |
| 246 | 0 | 2 | 3 | 2 | 3 | 3 | 3 | |
| 248 | 0 | 2 | 3 | 3 | NA | NA | 3 | |
| 241 | 0 | 2 | NAb | NA | NA | NA | 1 | |
| 242 | 0 | 1 | NA | NA | NA | NA | 1 | |
| 249 | 0 | 2 | 3 | 3 | NA | NA | 3 | |
| Mock | 244 | 0 | NA | NA | NA | NA | NA | 0 |
| 247 | 0 | 0 | 0 | NA | NA | NA | 0 | |
| 251 | 0 | 0 | 0 | 0 | 0 | NA | 0 | |
| 252 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
Diarrhoea was scored for each piglet. 0 = no diarrhoea; 1 = mild; 2 = moderate; 3 = severe. Vomiting was not observed.
NA, not applicable. Pigs were euthanized for pathologic examination at 24, 48 and 72 hpi.
Goss pathology was scored for each piglet. 0 = no change; 1 = mild; 2 = moderate; 3 = severe.
This article is being made freely available through PubMed Central as part of the COVID-19 public health emergency response. It can be used for unrestricted research re-use and analysis in any form or by any means with acknowledgement of the original source, for the duration of the public health emergency.
Figure 1.
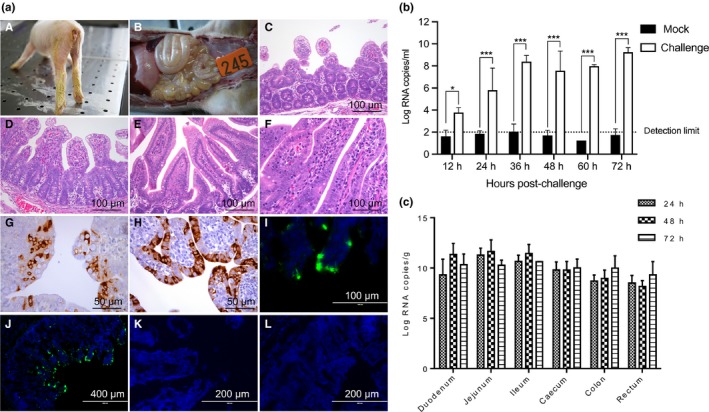
Pathogenicity of PDCoV strain NH (P10). (a) (A) At 28 hr post‐infection, piglets exhibited diarrhoea and yellow faeces were observed behind the legs. (B) Dilated and thin intestinal walls were observed in the piglets terminated at 48 hpi. (C) Pigs were euthanized for pathological examinations at 72 hpi. (D) Severe villous atrophy of the jejunum and ileum was observed. (E) Negative pigs displayed no obvious lesions or pathological changes in the (E) jejunum or (F) ileum. (G, H) PDCoV antigen was detected using IHC and IFA. The jejunum and ileum stained (I) IHC and (J) IFA positive. No PDCoV antigens were detected in the (K) jejunum or (L) ileum in the mock group. (b) Viral RNA copies in faecal swabs and intestine tissues were detected by RT‐qPCR. (c) Viral levels in the jejunum and ileum were higher than in other intestinal sections. Data are expressed as the mean ± SD of triplicate samples. *p < .05; ***p < .001
Pigs were euthanized for pathological examinations at 24, 48 and 72 hpi. Severe villous atrophy of the jejunum and ileum was observed (Figure 1a(C) and a(D)). Negative pigs displayed no obvious lesions or pathological changes (Figure 1a(E) and a(F)). PDCoV antigens were only detected in the jejunum and ileum, not in other intestinal sections (Figure 1a(G)–a(J)). No PDCoV antigens were detected in the mock group (Figure 1a(K) and a(L)). These results suggest that the jejunum and ileum are the replication sites in the intestines, which is consistent with the findings of previous studies (Jung et al., 2015; Jung & Saif, 2015).
Viral RNA copies in faecal swabs and intestinal tissues were detected by RT‐qPCR. Viral RNA in faecal swabs increased to the maximum concentration recorded (~9.6 log10 PDCoV RNA copies/ml) at 72 hpi (Figure 1b). The viral levels in the jejunum and ileum were higher than in other intestinal sections (Figure 1c). These results indicate that PDCoV strain NH (P10) was enteropathogenic in 5‐day‐old SPF pigs and primarily replicated within small intestinal tissues.
3.2. Immunogenicity of the inactivated vaccine in gilts
Gilts were immunized twice with the inactivated PDCoV vaccine. After eating colostrum for 5 days, piglets were divided into the challenge group, mock group and monitoring group in each sow (Figure 2a). The immune response in the serum and milk after two doses of the vaccine using a prime/boost strategy were detected with a spike‐specific ELISA. IgG antibodies in the sow serum were first detected 20 days post‐vaccination and after the second immunization, the antibody levels increased rapidly (Figure 2b). Antibody levels were monitored 21 days post‐farrowing. Results revealed that S‐specific antibodies in the sow serum remained at a high level and persisted longer (~21 days). IgG antibody responses were also detected in sow colostrum and milk, and unlike the long‐term persistence of high antibody levels in serum, the S‐specific IgG antibodies in milk only lasted 5 days, although a high antibody level was detected 1 days post‐farrowing (Figure 2c). Similar to the IgG response, a higher S‐specific sIgA antibody response was observed in the colostrum and milk after 2 immunizations with the inactivated vaccine at the Houhai acupoint. However, the sIgA antibodies lasted only a few days (~5 days) and decreased rapidly to negative levels (Figure 2d). No S‐specific IgG or sIgA antibody response was detected in control sows.
Figure 2.
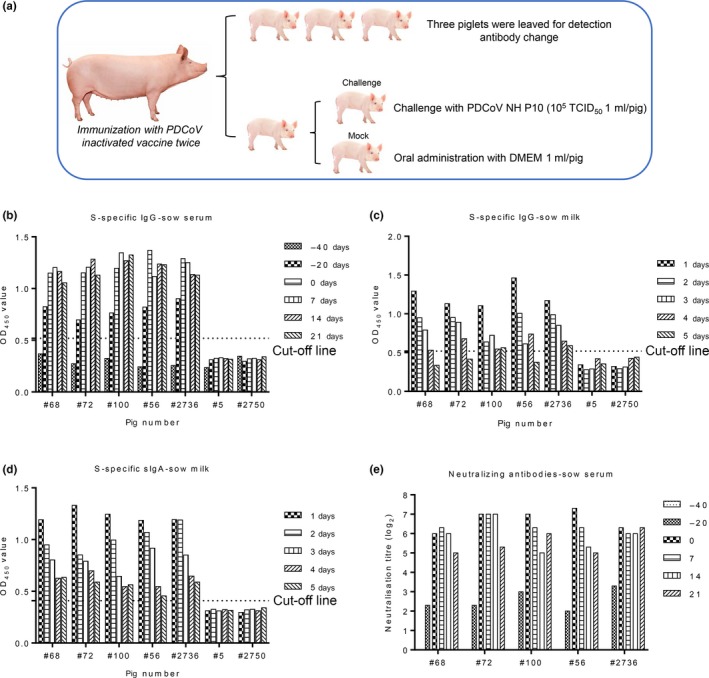
Immune response in the serum and milk of sows. (a) Gilts were immunized twice with an inactivated PDCoV vaccine. After eating colostrum for 5 days, piglets were divided into a challenge, mock or monitoring group for each sow. (b) S‐specific IgG antibody responses in sow serums. (c) S‐specific IgG antibody responses in sow milk. (d) S‐specific sIgA antibody responses in sow milk. (e) NA responses in sow serums. Dotted lines represent the cut‐off [Colour figure can be viewed at http://wileyonlinelibrary.com]
The ability of the inactivated vaccine to induce an NA response against PDCoV was determined using VNT. NAs were detected 20 days after the first immunization (log2 2–3.3) with very low NA responses (Figure 2e). An increase in the NA response was observed in the serum of immunized sows after the boost immunization (log2 6–7.3) and remained at a high level for a longer period of time (~21 days post‐farrowing). These results indicate that the inactivated PDCoV vaccine induced an immune response in the serum of immunized sows with high levels of NA and S‐specific IgG. Moreover, high levels of IgG and sIgA antibodies were detected in the colostrum and milk, although these only persisted for a short period of time.
3.3. Passive immunization
The passive transfer of antibodies from immunized sows to piglets through their colostrum and milk was detected with an S‐specific ELISA. A high level of S‐specific IgG was detected in the serum of piglets born to immunized sows 5 days post‐farrowing (Figure 3a). S‐specific IgG antibodies persisted long‐term in the serum of piglets (~12 days post‐farrowing). No S‐specific sIgA antibodies were detected in the serum of piglets, although high levels of antibodies were detected in the colostrum and milk of their dams (Figure 3b). NA levels were also detected in the serum of piglets 5 days post‐birth, such that high levels of NA responses were detected in the serum of piglets and gradually decreased. NA responses remained positive until the end of the experiment 26 days post‐birth (Figure 3c). These results indicate that the antibodies were successfully transferred from the sows to piglets by suckling the colostrum and milk.
Figure 3.
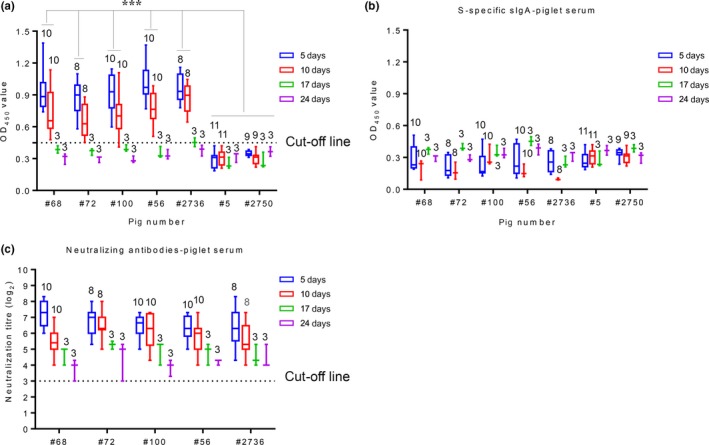
Antibodies in the piglets’ serum. (a) S‐specific IgG antibody in piglet serum. (b) S‐specific sIgA antibody in piglet serum. (c) NA in piglet serum. The number of piglets is located above the box and whisker plots. Dotted lines represent the cut‐off. The antibody levels of the piglets are presented as box and whisker plots created with GraphPad Prism v7. ***p < .001
3.4. Challenge with PDCoV strain NH P10
3.4.1. Clinical signs post‐challenge
In order to assess the protective effect of the vaccine, 5‐day‐old piglets (31 total) born to immunized sows and 5‐day‐old piglets (14 total) born to unimmunized sows were challenged with a high dose (105 TCID50) of PDCoV strain NH (P10). Piglets (12 total) born to immunized sows and 5‐day‐old piglets born to unimmunized sows (4 total) were used as controls. Piglets were monitored, and clinical symptoms were scored according to their level of diarrhoea (Table 3). Six piglets exhibited mild to moderate diarrhoea, in which the symptoms of four piglets from the challenge group and 2 piglets from the mock group born to immunized group were due to indigestion. Following oral inoculation with PDCoV, two piglets born to immunized sows #72 (1/5) and #56 (1/7) exhibited mild diarrhoea 2 days post‐infection. One day later, the piglet from sow #72 continued to display mild diarrhoea, but the diarrhoea conditions disappeared 3 days post‐infection. The piglet from sow #56 exhibited moderate diarrhoea 3 days post‐infection, which reverted to mild diarrhoea 5 days post‐infection. Two piglets in the mock group born to immunized sows #56 and #2,736 exhibited mild to moderate diarrhoea due to indigestion, and undigested curd was present in their faeces; no severe diarrhoea was observed in piglets born to immunized sows in the mock group. The diarrhoea rate of piglets born to immunized sows was 4 of 31 pigs (12.9%) 3 days post‐challenge. Piglets born to unimmunized sows #5 and #2756 were used as controls and orally challenged with virulent PDCoV. At 2 days post‐infection, piglets displayed moderate diarrhoea and quickly worsened, exhibiting severe diarrhoea until the end of the experiment. The diarrhoea rate of the piglets born to unimmunized sows was 100% 4 days post‐challenge. No obvious clinical symptoms were observed in the mock piglets from each group.
Table 3.
Pathogenicity of PDCoV in piglets
| Group | No. | No. of piglets | No. of sick piglets/total (avg. score)a | Histology scoreb | No. of piglets with viral RNA | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Challenge/mock | Day 2 | Day 3 | Day 4 | Day 5 | Jejunum | Ileum | Jejunum | Ileum | ||
| Inactivated PDCoV vaccine | #68 | 7/2 | 0/9 (0) | 1/9 (2) | 1/9 (2) | 1/9 (1) | 0 | 0 | 2 | 2 |
| #72 | 5/2 | 1/7 (1) | 1/7 (1) | 0/7 (0) | 0/7 (0) | 0 | 0 | 0 | 0 | |
| #100 | 7/2 | 0/9 (0) | 1/9 (2) | 1/9 (3) | 1/9 (3) | 1 | 1 | 1 | 1 | |
| #56c | 7/4 | 1/11 (1) | 2/11 (1) | 2/11 (2) | 1/11 (1) | 1 | 1 | 1 | 1 | |
| #2,736c | 5/2 | 0/7 (0) | 1/7 (2) | 1/7 (1) | 0/7 (0) | 0 | 0 | 0 | 0 | |
| DMEM | #5 | 8/2 | 4/10 (2) | 5/10 (3) | 8/10 (3) | 8/10 (3) | 3 | 3 | 8 | 8 |
| #2,750 | 6/2 | 3/8 (2) | 6/8 (3) | 6/8 (3) | 6/8 (3) | 3 | 3 | 6 | 6 | |
Score of the severity of symptoms after 2, 3, 4 and 5 days: 0 = no vomiting or diarrhoea; 1 = mild; 2 = moderate; 3 = severe. The average score for each group is shown in parentheses.
The severity of histological changes was scored based on the length of the small intestine villi and changes in the intestinal wall: 0 = no lesion; 1 = mild; 2 = moderate; 3 = severe.
Two piglets from sows #56 and #2,736 in the mock group exhibited mild to moderate diarrhoea 3 days post‐challenge.
This article is being made freely available through PubMed Central as part of the COVID-19 public health emergency response. It can be used for unrestricted research re-use and analysis in any form or by any means with acknowledgement of the original source, for the duration of the public health emergency.
3.4.2. Viral RNA copies in faecal and intestinal tissues samples
The piglet faecal specimens were collected daily, and RNA was extracted in order to detect viral RNA copies by RT‐qPCR. PDCoV RNA copies were detected in four piglets born to sows #68, #100 and #56, and low levels of viral RNA were detected in 1 piglet (2.8 log10 RNA copies/ml) 24 hr post‐infection (Figure 4a). With the prolonged infection time, a high level of viral RNA was detected in the 4 piglets (6–6.5 log10 RNA copies/ml) (Figure 4a, c and d). No RNA copies were detected in piglets born to sows #72 and #2,736 (Figure 4b and e). A high level of RNA was detected in piglets born to unimmunized sows #5 and #2,736 24 hr post‐infection (6.2–8 log10 RNA copies/ml), and several RNA copies were detected 48 hr post‐infection (8.2–12 log10 RNA copies/ml) (Figure 4f and g). At the end of the experiment, all of the piglets were euthanized and viral RNA copies from the intestinal tissues (i.e. duodenum, jejunum, ileum, caecum, colon and rectum) were detected by RT‐qPCR. Viral RNA from the intestinal tissues was detected from the 4 piglets described above (Figure 4h). No viral RNA was detected in other piglets born to immunized sows post‐challenge with PDCoV. A high number of viral RNA copies were detected in the intestinal tissues of piglets born to unimmunized sows (Figure 4i and j).
Figure 4.
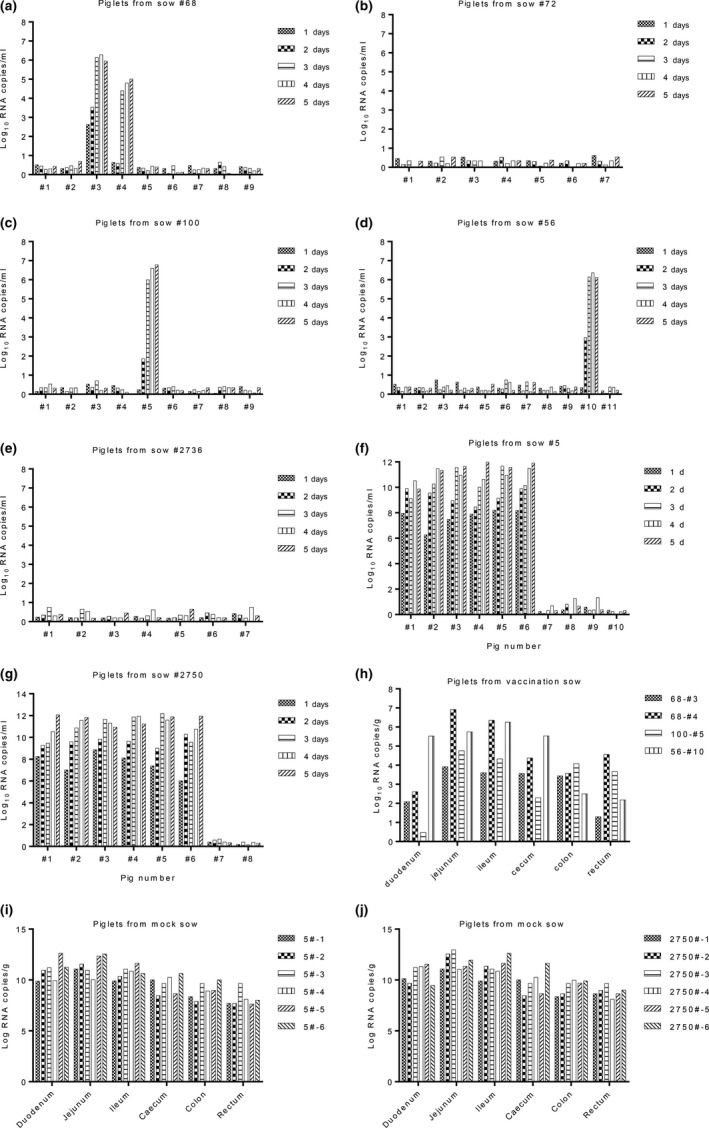
Detection of viral RNA with RT‐qPCR. The number of viral RNA copies from the faecal samples of the piglets born to sows (a) #68, (b) #72, (c) #100, (d) #56, (e) #2,736, (f) #5 and (g) #2,750. (h) Detected viral RNA copies from the intestinal tissues of 4 piglets born to immunized sows. A high number of viral RNA copies detected in the intestinal tissues of piglets born to unimmunized sows (i) #5 and (j) #2,750
3.4.3. Pathology and viral antigen detection
Piglets were euthanized for pathological examinations 5 days post‐challenge. Pathological damage was scored based on the length of the small intestine villi and changes in the intestinal wall. Two piglets from sow #68 exhibited virus shedding in faecal samples and viral RNA was detected in the intestinal tissues; only 1 piglet exhibited mild diarrhoea. At 5 days post‐challenge, viral antigens were not detected in the jejunum or ileum and no obvious lesions or pathological changes were observed in the intestinal tissues of 2 piglets. One piglet from sow #100 (#100‐5) exhibited severe diarrhoea and virus shedding, and lesions in the jejunum and ileum displayed mild villus atrophy (Figure 5a and b). The pathology score in the jejunum and ileum was 1. Few viral antigens were detected using anti‐PDCoV‐N monoclonal antibodies with IHC (Figure 5c). Two piglets born to immunized sows #56 and #2,736 in the mock group exhibited no virus shedding 5 days post‐challenge and no PDCoV antigens were detected (data not show). Piglets born to unimmunized sows and challenged with PDCoV, exhibited lesions in the jejunum and ileum, which were the most obvious compared to other intestinal tissues with severe villous atrophy, followed by extensive intestinal epithelial degeneration, swelling and necrosis. The pathology score in the jejunum and ileum was 3. Several viral antigens were detected in the mock group born to unimmunized sows post‐challenge (Figure 5a–c). Based on PCR data from the faeces and intestinal tissues, 4 of 31 pigs from the vaccinated sows were evidently infected after virus challenge. Therefore, the passive immunity obtained from immunized sows induced 87.1% protection against highly pathogenic PDCoV challenge.
Figure 5.
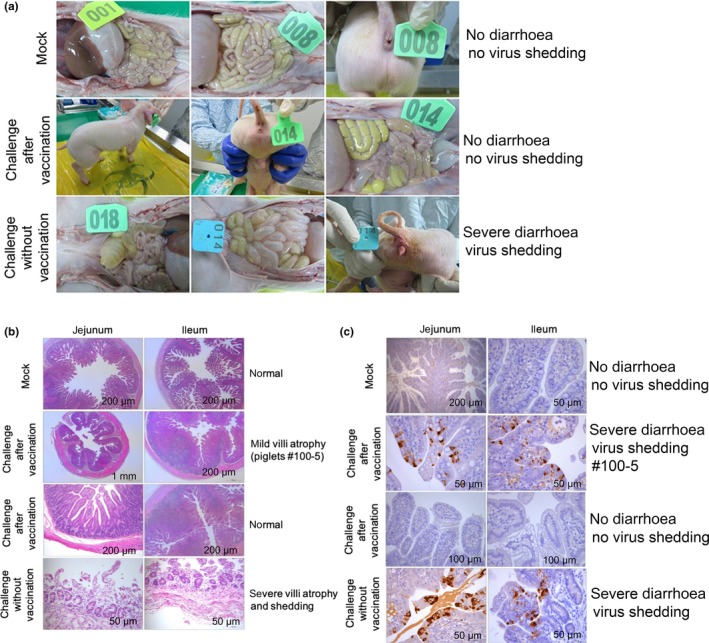
Clinical signs and histopathology changes post‐challenge. (a) Clinical signs post‐challenge with virulent PDCoV. No severe diarrhoea was observed in the mock group or piglets born to immunized sows post‐challenge. Severe diarrhoea was observed in piglets born to unimmunized sows. (b) HE staining. One piglet from sow #100 (#100‐5) was born to immunized sows and exhibited mild villus atrophy in the jejunum and ileum post‐challenge. No obvious lesions were observed in the mock group or piglets born to immunized sows post‐challenge. Severe villus atrophy in the jejunum and ileum was observed in piglets born to unimmunized sows post‐challenge. (c) IHC analysis. Few viral antigens were detected in the aforementioned piglet (#100‐5). Several viral antigens were detected in the mock group born to unimmunized sows post‐challenge with PDCoV. No PDCoV antigens were detected in the mock group or piglets born to immunized sows post‐challenge
4. DISCUSSION
In the present study, the pathogenicity of PDCoV strain NH isolated in 2014 was assessed. The results suggest that the virus was enteropathogenic in 5‐day‐old SPF pigs. A challenge model to study the protective passive immunity in neonatal piglets was conducted. The protective efficacy of passive immunity elicited by the inactivated PDCoV vaccine against challenge with a highly pathogenic virulent strain in neonatal piglets born to immunized sows was investigated. Results revealed that immunization with an inactivated PDCoV vaccine could produce a strong antibody response in pregnant sows after the second vaccination 20 days before delivery. High S‐specific IgG antibody and NA responses were observed in the serum of sows post‐farrowing. Interestingly, a high level of S‐specific sIgA was detected in colostrum and milk, although it lasted for only a short period of time. Previous studies have indicated that sIgA was produced in the mammary gland by antibody‐secreting cells and the recruitment of antibody‐secreting cells into the mammary gland contribute to the production of sIgA (Wilson & Butcher, 2004). Therefore, it has been suggested that the oral route appears to be the most effective method for eliciting a strong sIgA response when vaccinating sows (Gerdts & Zakhartchouk, 2017). However, the findings of this study suggest that vaccination with an inactivated vaccine by injection at the Houhai acupoint in pregnant sows could also induce a strong sIgA response in the colostrum, which may subsequently provide protection for piglets against virulent challenge. Results also confirmed that vaccination at the Houhai acupoint could induce a mucosal immune response (Liu et al., 1998). The duration of antibody persistence until the end of the experiment was monitored, and the results indicated that a strong positive correlation existed between the IgG antibody and NA responses in the serum of immunized sows. The S‐specific IgA antibody response was also detected with an ELISA; however, the results indicated that the serum, colostrum and milk of sows failed to produce an IgA response after immunization with an inactivated PDCoV vaccine.
Transferring antibodies (i.e. sIgA, IgG and IgM) from the sows to the piglets through the colostrum and milk has been considered to be the primary mechanism of protection mediated by passive immunity (Poonsuk et al., 2016; Saif & Bohl, 1983; Salmon, Berri, Gerdts, & Meurens, 2009). The IgG antibodies were absorbed by the piglets within the first 24–48 hr of life through the colostrum. In this study, a high level of IgG antibodies was detected in the serum of piglets and, more importantly, such levels displayed long‐term persistence (~10 days) although piglets stopped consuming milk from immunized sows. These results suggest that IgG from the serum of sows could be absorbed into the serum of piglets through colostrum. The level of IgG antibodies in the serum of piglets (31 piglets, 10 days post‐challenge) did not increase, which was due to the existing maternal antibodies in the serum of piglets interfering with an active immune response to infection. No S‐specific sIgA antibodies were observed in the serum of piglets, which indicated that the function of sIgA was limited to the enteric cavity rather than the serum. The challenge assay suggested that antibodies transferred from sows through their colostrum and milk could provide protection for piglets. Four piglets born to immunized sows displayed diarrhoea post‐inoculation with PDCoV strain NH, and viral RNA copies were detected in the faeces. However, compared to the mock group, the four piglets exhibited mild to moderate diarrhoea (no severe diarrhoea) and lower viral RNA copies in the faeces and intestinal tissues. Low PDCoV antigen levels were also detected in the jejunum and ileum by IHC, and minor intestinal villi damage was observed by HE staining. Collectively, these results suggest that the antibodies from sows could provide the 4 piglets with partial, but not complete, protection.
Previous studies on the passive immunity of TGEV suggest that viral inoculation by different routes induce different immunoglobulin isotypes (Bohl & Saif, 1975). The production of IgA in the milk of TGEV antibodies was associated with an intestinal infection, while the production of IgG was associated with parenteral antigenic stimulation. This study also indicated that the IgG response in serum, colostrum and milk was predominantly detected after pregnant sows were inoculated with virulent TGEV by intramammary injections. Moreover, the passive immunity protection was good (0% mortality and morbidity) in 3‐ and 4‐day‐old newborn piglets born to immunized sows post‐challenge. However, morbidity was 100% 11 days post‐farrowing with 0% mortality after TGEV challenge. These results suggest that within the first week, IgG antibodies in colostrum and milk of immunized sows could provide protection for piglets against TGEV virulent challenge. In this study, IgA antibodies were not detected in serum, colostrum or milk of immunized sows. The immunoglobulin in the antibodies was predominantly IgG, and although the piglets were no longer receiving anti‐PDCoV IgG antibodies from the milk, IgG lasted ~10 days in the serum of piglets. Because of this, the immune protection response observed in PDCoV‐challenged piglets born to inactivated PDCoV‐vaccinated mothers was likely due to circulating anti‐PDCoV IgG antibodies that were passively transferred through colostrum rather than anti‐PDCoV IgA antibodies from milk, as observed previously in PEDV research (Poonsuk et al., 2016).
The sIgA and IgG in colostrum and milk consumed by piglets for 1–2 days post‐birth survived and were maintained until challenged to some extent in the gut. In the intestinal environment of piglets born to immunized sows, VNT antibodies might neutralize PDCoV in gut and only a small amount of the virus might remain under the minimal infectious doses that could infect piglets (Poonsuk et al., 2016). Another possible reason for this result may be due to the antibody‐dependent cell‐mediated cytotoxicity (ADCC) effect, which promotes cell‐mediated immune responses against PDCoV, as previously studied in PEDV (Casadevall & Pirofski, 2003).
In summary, the findings of the present study demonstrate that immunizing sows twice with an inactivated PDCoV vaccine could induce IgG, sIgA and NA responses. Piglets born to immunized sows could obtain protective antibodies by ingesting colostrum and milk. Moreover, high levels of IgG antibodies and NA responses were detected in serum, which protected the piglets against virulent PDCoV challenge.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
ETHICAL STATEMENT
The study's protocol was approved by the Institutional Animal Care and Use Committee of the Harbin Veterinary Research Institute. This study does not contain research with human participants performed by any of the authors.
ACKNOWLEDGEMENTS
This work was supported by the National Key Technology R&D Program of China (No. 2016YFD0500103), National Natural Science Foundation of China (Nos. 31602072 and 31572541), Natural Science Foundation of Heilongjiang Province of China (No. C2017079), State Key Laboratory of Veterinary Biotechnology Foundation (No. SKLVBP2018002), and "Twelfth Five‐Year Plan” National Science and Technology Plan in Rural Areas Research mission contract (No. 2015BAD12B02‐6).
Zhang J, Chen J, Liu Y, et al. Pathogenicity of porcine deltacoronavirus (PDCoV) strain NH and immunization of pregnant sows with an inactivated PDCoV vaccine protects 5‐day‐old neonatal piglets from virulent challenge. Transbound Emerg Dis. 2020;67:572–583. 10.1111/tbed.13369
Zhang and Chen contributed equally to this study.
REFERENCES
- Bohl, E. H. , & Saif, L. J. (1975). Passive immunity in transmissible gastroenteritis of swine: Immunoglobulin characteristics of antibodies in milk after inoculating virus by different routes. Infection and Immunity, 11(1), 23–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casadevall, A. , & Pirofski, L. A. (2003). Antibody‐mediated regulation of cellular immunity and the inflammatory response. Trends in Immunology, 24(9), 474–478. 10.1016/S1471-4906(03)00228-X [DOI] [PubMed] [Google Scholar]
- Chen, Q. I. , Gauger, P. , Stafne, M. , Thomas, J. , Arruda, P. , Burrough, E. , … Zhang, J. (2015). Pathogenicity and pathogenesis of a United States porcine deltacoronavirus cell culture isolate in 5‐day‐old neonatal piglets. Virology, 482, 51–59. 10.1016/j.virol.2015.03.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong, N. , Fang, L. , Zeng, S. , Sun, Q. , Chen, H. , & Xiao, S. (2015). Porcine deltacoronavirus in mainland China. Emerging Infectious Diseases, 21(12), 2254 10.3201/eid2112.150283 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerdts, V. , & Zakhartchouk, A. (2017). Vaccines for porcine epidemic diarrhea virus and other swine coronaviruses. Veterinary Microbiology, 206, 45–51. 10.1016/j.vetmic.2016.11.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janetanakit, T. , Lumyai, M. , Bunpapong, N. , Boonyapisitsopa, S. , Chaiyawong, S. , Nonthabenjawan, N. , … Amonsin, A. (2016). Porcine deltacoronavirus, Thailand, 2015. Emerging Infectious Diseases, 22(4), 757 10.3201/eid2204.151852 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung, K. , Hu, H. , Eyerly, B. , Lu, Z. , Chepngeno, J. , & Saif, L. J. (2015). Pathogenicity of 2 porcine deltacoronavirus strains in gnotobiotic pigs. Emerging Infectious Diseases, 21(4), 650–654. 10.3201/eid2104.141859 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung, K. , Hu, H. , & Saif, L. J. (2016). Porcine deltacoronavirus infection: Etiology, cell culture for virus isolation and propagation, molecular epidemiology and pathogenesis. Virus Research, 226, 50–59. 10.1016/j.virusres.2016.04.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung, K. , & Saif, L. J. (2015). Porcine epidemic diarrhea virus infection: Etiology, epidemiology, pathogenesis and immunoprophylaxis. The Veterinary Journal, 204(2), 134–143. 10.1016/j.tvjl.2015.02.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langel, S. N. , Paim, F. C. , Lager, K. M. , Vlasova, A. N. , & Saif, L. J. (2016). Lactogenic immunity and vaccines for porcine epidemic diarrhea virus (PEDV): Historical and current concepts. Virus Research, 226, 93–107. 10.1016/j.virusres.2016.05.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee, J. H. , Chung, H. C. , Nguyen, V. G. , Moon, H. J. , Kim, H. K. , Park, S. J. , … Park, B. K. (2016). Detection and phylogenetic analysis of porcine deltacoronavirus in Korean swine farms, 2015. Transboundary and Emerging Diseases, 63(3), 248–252. 10.1111/tbed.12490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leidenberger, S. , Schröder, C. H. , Zani, L. , Auste, A. , Pinette, M. , Ambagala, A. , … Blome, S. (2017). Virulence of current German PEDV strains in suckling pigs and investigation of protective effects of maternally derived antibodies. Scientific Reports, 7(1), 10825 10.1038/s41598-017-11160-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li, Q. , Xu, Z. , Wu, T. , Peng, O. , Huang, L. , Zhang, Y. , … Cao, Y. (2018). A flagellin‐adjuvanted PED subunit vaccine improved protective efficiency against PEDV variant challenge in pigs. Vaccine, 36(29), 4228–4235. 10.1016/j.vaccine.2018.05.124 [DOI] [PubMed] [Google Scholar]
- Liu, Y. , Tan, H. , Wan, X. , Zuo, Z. , & Liu, K. (1998). Spinal segment distribution of neural innervation related to houhai acupoint and compared with zusanli and dazhui acupoints in domestic chicken. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. Acta Academiae Medicinae Sinicae, 20(2), 154–160. [PubMed] [Google Scholar]
- Ma, Y. , Zhang, Y. , Liang, X. , Lou, F. , Oglesbee, M. , Krakowka, S. , & Li, J. (2015). Origin, evolution, and virulence of porcine deltacoronaviruses in the United States. MBio, 6(2), e00064 10.1128/mBio.00064-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ojkic, D. , Hazlett, M. , Fairles, J. , Marom, A. , Slavic, D. , Maxie, G. , … Burlatschenko, S. (2015). The first case of porcine epidemic diarrhea in Canada. The Canadian Veterinary Journal, 56(2), 149. [PMC free article] [PubMed] [Google Scholar]
- Poonsuk, K. , Gimenez‐Lirola, L. G. , Zhang, J. Q. , Arruda, P. , Chen, Q. , Carrion, L. C. D. , … Main, R. (2016). Does Circulating Antibody Play a Role in the Protection of Piglets against Porcine Epidemic Diarrhea Virus? PLoS ONE, 11(4), e0153041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saeng‐chuto, K. , Lorsirigool, A. , Temeeyasen, G. , Vui, D. T. , Stott, C. J. , Madapong, A. , … Nilubol, D. (2017). Different lineage of porcine deltacoronavirus in Thailand, Vietnam and Lao PDR in 2015. Transboundary and Emerging Diseases, 64(1), 3–10. 10.1111/tbed.12585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saif, L. J. , & Bohl, E. H. (1983). Passive immunity to transmissible gastroenteritis virus: Intramammary viral inoculation of sows. Annals of the New York Academy of Sciences, 409, 708–723. 10.1111/j.1749-6632.1983.tb26910.x [DOI] [PubMed] [Google Scholar]
- Salmon, H. , Berri, M. , Gerdts, V. , & Meurens, F. (2009). Humoral and cellular factors of maternal immunity in swine. Developmental and Comparative Immunology, 33(3), 384–393. 10.1016/j.dci.2008.07.007 [DOI] [PubMed] [Google Scholar]
- Sestak, K. , Lanza, I. , Park, S. K. , Weilnau, P. A. , & Saif, L. J. (1996). Contribution of passive immunity to porcine respiratory coronavirus to protection against transmissible gastroenteritis virus challenge exposure in suckling pigs. American Journal of Veterinary Research, 57(5), 664–671. [PubMed] [Google Scholar]
- Song, D. , Zhou, X. , Peng, Q. , Chen, Y. , Zhang, F. , Huang, T. , … Wu, Q. (2015). Newly emerged porcine deltacoronavirus associated with diarrhoea in swine in China: Identification, prevalence and full‐length genome sequence analysis. Transboundary and Emerging Diseases, 62(6), 575–580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, L. , Byrum, B. , & Zhang, Y. (2014). Detection and genetic characterization of deltacoronavirus in pigs, Ohio, USA, 2014. Emerging Infectious Diseases, 20(7), 1227–1230. 10.3201/eid2007.140296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, L. , Hayes, J. , Sarver, C. , Byrum, B. , & Zhang, Y. (2016). Porcine deltacoronavirus: Histological lesions and genetic characterization. Archives of Virology, 161(1), 171–175. 10.1007/s00705-015-2627-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson, E. , & Butcher, E. C. (2004). CCL28 controls immunoglobulin (Ig)A plasma cell accumulation in the lactating mammary gland and IgA antibody transfer to the neonate. Journal of Experimental Medicine, 200(6), 805–809. 10.1084/jem.20041069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woo, P. C. , Lau, S. K. , Lam, C. S. , Lau, C. C. , Tsang, A. K. , Lau, J. H. , … Wang, M. (2012). Discovery of seven novel mammalian and avian coronaviruses in Deltacoronavirus supports bat coronaviruses as the gene source of Alphacoronavirus and Betacoronavirus and avian coronaviruses as the gene source of Gammacoronavirus and Deltacoronavirus. Journal of Virology, 86, 3995–4008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, J. (2016). Porcine deltacoronavirus: Overview of infection dynamics, diagnostic methods, prevalence and genetic evolution. Virus Research, 226, 71–84. 10.1016/j.virusres.2016.05.028 [DOI] [PMC free article] [PubMed] [Google Scholar]


