Abstract
Nanoscale materials can improve the efficacy of vaccines. Herein we review latest developments that use nanomaterials for vaccines. By highlighting the relationships between the nanoscale physicochemical characteristics and working mechanisms of nanomaterials, this paper shows the current status of the developments where researchers employ functional nanomaterials as vector and/or immunoregulators for vaccines. It also provides us some clues for improving the design and application of nanomaterials to optimize the efficacy of vaccines.
Keywords: nanomaterials, vaccines, immunoregulators, carriers, immune responses
Nanomaterials, such as peptide‐based materials, non‐peptide polymers, carbon, gold, silver, and calcium salts can act as carriers and/or immunoregulators to optimize the efficacy of vaccines in multiple ways, including enhancing the uptake of antigen‐presenting cells against antigens, stimulating the proliferation of immunologic effector cells, and regulating immunity profiles induced by vaccines.
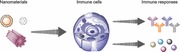
1. Introduction
On the nanoscale, the chemical compositions, physical characteristics, and biochemical modifications of materials all determine their biological effects in vivo. These parameters are important for the applications of these nanomaterials in vaccines.1 Vaccines represent one of the most effective and least costly means for the prevention and control of human diseases. For certain diseases that have no cure because the pathogen integrates its own genetic material into its host, vaccination may represent the only possible means of the fight against these pathogens. One such example is HIV (human immunodeficiency virus). Human beings, however, have only limited access to effective vaccination. Many serious diseases still have no effective vaccines, for instance, HIV, HCV (hepatitis C virus), lung cancer, and mad cow disease.2, 3, 4, 5 Nanomaterials have brought new hopes for people with diseases that currently lack effective vaccines. The unique properties of nanomaterials can improve vaccines in a number of ways, including enhancing the immunity of the vaccine, improving biosafety, and optimizing immunization routes.6 However, the detailed molecular working mechanisms are still largely unknown, and few studies focus on investigating the relationship between the physiochemical characteristics of nanomaterials and their bio‐effects in vivo. Herein, we provide examples of selected studies that show practical applications and working mechanisms of multiple nanomaterials in the field of vaccines. By summarizing six different chemical compositions of nanomaterials—i.e., peptide backbones, carbon, gold, silver, calcium, and polymers—this review shows the diverse biological effects of these materials for regulating immune responses of vaccines based on their various physical characteristics (size, shape, aspect ratio, and so forth) and biochemical modifications (Table 1 and Table 2 ). This review also analyzes how these materials work via targeting and affecting major components in the immune system, such as dendritic cells, macrophages, and T and B cells, to shape the eventual profiles of the immunity of vaccines (Figure 1 ).
Table 1.
Definitions of biological terms used in the current review
| Professional term | Definition |
|---|---|
| Immunoregulator | The factors which can activate or inhibit immune responses. |
| Dendritic cells | A kind of important antigen‐presenting cell that present antigens to active T cells. |
| Macrophage | A kind of important antigen‐presenting cell that present antigens to T helper cells. |
| Nanovector | Nanoscale materials that carry and deliver vaccine to host. |
| Epitopes | An amino acid sequence on an antigen which can be recognized by antibodies. |
| Hapten | A small molecule that can elicit an immune response only when attached to a large carrier such as a protein. |
| Neutralizing antibody | The antibody which can prevent pathogens from entering host cells. |
| Cellular responses | Immune responses that release various antigen‐specific cytokines. |
| Humoral responses | Immune responses that release various antigen‐specific antibodies. |
| CpG | A kind of vaccine adjuvant composed by short single‐stranded unmethlyated DNA molecules. |
| Memory immunity | Immune responses that exist in the long term, and rapidly adapt to a robust level when encountering the same pathogen again. |
This article is being made freely available through PubMed Central as part of the COVID-19 public health emergency response. It can be used for unrestricted research re-use and analysis in any form or by any means with acknowledgement of the original source, for the duration of the public health emergency.
Table 2.
The main characteristics of nanomaterials as vaccine vectors and/or immunoregulators
| Chemical composition | Main characteristics | Vaccines | Ref. | |||
|---|---|---|---|---|---|---|
| Physical characteristics | Modification | |||||
| Size | Shape | |||||
| Peptide | 2 nm (diameter) | Fiber | None | HIV | 6 | |
| 2 nm (diameter) | Fiber | None | Epithelial tumor | 7 | ||
| 10–50 nm | Particle | None | Avian Influenza | 8 | ||
| 10–30 nm | Particle | None | SARS | 9 | ||
| 10–100 nm | Particle | None | Human papillomavirus | 10 | ||
| Non‐peptide polymer | PLGA | 200 nm, 2000 nm | Particle | DC target antibody | Tetanus toxoid | 11 |
| Polystyrene | 20–200 nm, 500–2000 nm | Particle | None | Virus like particle | 12 | |
| Chitosan | None | Particle | CpG | Hepatitis B virus | 13 | |
| Carbon | 30–60 nm | Fullerene particle | Polyhydroxy | HIV | 14 | |
| 8–53 nm | Nanotube | Amino | Foot‐and‐mouth disease | 15 | ||
| diameter 20–30 nm, length 50 nm | Nanotube | Carboxyl | Tumor lysate protein | 16 | ||
| diameter 20–30 nm, length 0.5–2 nm | Nanotube | Maleimido | Plasmodium vivax | 17 | ||
| diameter 50 nm | Nanotube | Carboxyl | Tuberculin | 18 | ||
| Gold | 1–9 nm | Particle | Chitosans | Hepatitis B virus | 19 | |
| 2–50 nm | Particle | Citrate | Foot‐and‐mouth disease | 20 | ||
| 17 ± 2nm | Particle | Citrate | Merozoite surface protein | 21 | ||
| 40 ± 3 nm | Particle | Chitosan | Tetanus toxoid | 22 | ||
| 15 nm | Particle | None | Human nogo‐66 receptor | 23 | ||
| 15.6 nm | Particle | Citrate | Yersinia pestis | 24 | ||
| 2–10 nm | Particle | Carbohydrate‐coat | Tumor | 25 | ||
| 5–20 nm | Particle | Tn‐antigen glycan | Polymerizable Tn‐antigen glycan | 26 | ||
| length 50–60 nm, aspect ratio is 4 | Rod | CTAB‐, PDDAC‐, PEI‐coating | HIV | 27 | ||
| 20 and 40 nm, 40 × 10 nm, 40 × 40 × 40 nm | Particle, Rod, Cubic | Protein coating | West Nile virus | 28 | ||
| Silver | 11 nm | Particle | None | Avian influenza virus | 29 | |
| Calcium | 1000 nm | Calcium Phosphate Particle | None | Herpes simplex virus | 30, 31 | |
| 50–100 nm | Calcium Phosphate Particle | None | Foot‐and‐mouth disease | 32 | ||
| 190–295 nm, 50 nm | Calcium Carbonate Particle, Calcium Phosphate Particle | Poly‐L‐gluta‐mic acid, poly‐L‐lysine | Influenza virus | 33, 34 | ||
| <50 nm | Calcium Phosphate Particle | β‐cyclodextrin, amino propyl, triethoxy silane | Newcastle disease virus | 35 | ||
| 30–45 nm | Calcium Phosphate Particle | Lipid, Mannose | Cancer | 36 | ||
Abbreviations in Table 2: PLGA, poly(lactid‐glycolide acid); HIV, human immunodeficiency virus; SARS, severe acute respiratory syndrome; DC, dendritic cell; CTAB, cetyltrimethylammonium bromide; PDDAC, poly dimethyl diallyl ammonium chloride; PEI, polyetherimide.
This article is being made freely available through PubMed Central as part of the COVID-19 public health emergency response. It can be used for unrestricted research re-use and analysis in any form or by any means with acknowledgement of the original source, for the duration of the public health emergency.
Figure 1.
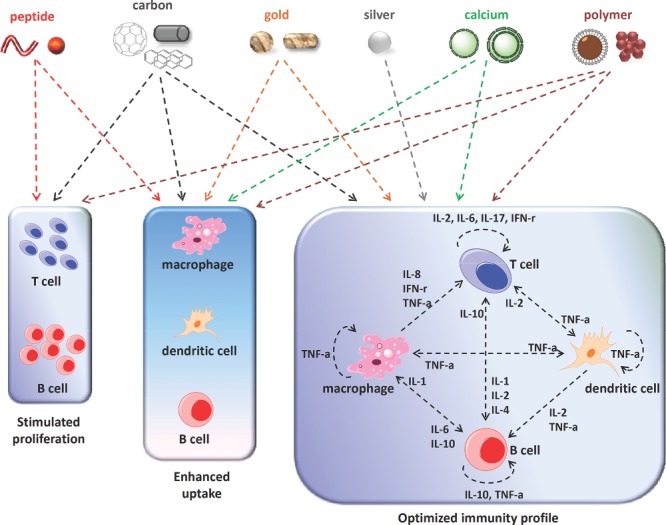
Major ways of immunity regulation mediated by nanomaterials appeared in our current review.
2. Peptide‐based Nanomaterials
Peptide‐based nanomaterials arise from naturally occurring molecules, typically have fairly high levels of biocompatibility, and can trigger robust immune responses. The shapes of peptide‐based nanomaterials mainly determine their bio‐effects, half‐life, and in vivo distribution.37 One potential disadvantage of such a nanomaterial is the instability of its shape. Their shapes might be dynamically modulated along with the changes of pH, salt concentration, and types of solvent,38, 39 resulting in various regulations of immunity over time. Luckily, the stability of their shape can be improved via some modifications, like PEG or its derivative,40 therefore exhibiting potential as an ideal vaccine immunoregulator. As a platform for vaccines, peptide‐based materials display abundant antigen epitopes on their surface. Such an ultra‐strong antigen‐loading capability is a great advantage for an ideal vaccine carrier candidate. Moreover, the peptide backbone of peptide‐based materials allows us to conveniently modify them by covalently linking all kinds of chemical groups or biological markers to their surface. By such artificial operations, peptide nanomaterials can be endowed with more biofunctions for optimizing the immunity of the vaccine. Actually, vaccines composed of either DNA or short peptides, assisted by peptide nanofibers and/or nanoparticles, have indeed triggered significantly enhanced immune responses in animal models, compared with naked vaccines. Herein, we review the promising applications of peptide‐based nanomaterials with regard to improving the efficacy of vaccines.
2.1. Nanofibers
Fibers probably represent the most common type of shape for peptide‐based nanomaterials. Some papers have reported the outstanding property of this kind of nanomaterial for enhancing antigen‐specific humoral and cellular immune responses. For example, in our recent work, the four‐amino acid backbone (glycine–phenylalanine–phenylalanine–tyrosine) peptide (henceforth abbreviated as G‐NMe) modified with naphthalene acetic acid (N‐terminal) and an N‐methyl group (C‐terminal) can rapidly assemble into nanofibers in the presence of alkaline phosphatase (Figure 2 ).
Figure 2.
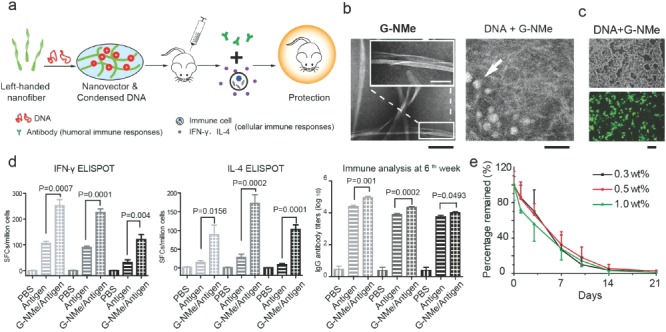
(a) Schematic illustration of the process of peptide‐based nanofibrous hydrogels for enhancing immune responses of HIV DNA vaccines. (b) TEM images of G‐NMe nanovectors with or without DNA. Scale bar: 100 nm (black); 50 nm (white). (c) Fluorescence images of 293 T cells transfected by G‐NMe nanovector/EGFP plasmid. Scale bar: 100 μm. G‐NMe could effectively carry DNA into the cells. (d) Cellular immune responses elicited by different immunization regimens, including antigen‐specific IFN‐γ or IL‐4 producing splenocytes generated by G‐NMe complexed with DNA or DNA alone using ELISPOT assays, and anti‐HIV antibody productions. G‐NMe significantly enhanced the humoral and cellular immune response. (e) Radioactivity remaining (% of activity at day 0) at the injection sites of G‐NMe containing 0.3, 0.5, and 1 wt% precursor at different time points. The data indicated that G‐NMe could be degraded in vivo. Reproduced with permission.6 Copyright 2014, American Chemical Society.
Such nanovectors exhibited the best record reported so far for DNA vaccines against HIV. This vector could result in a balanced immune response (strong antibody secretion, IFN‐γ and IL‐4 secreting lymphocyte production, all of which play crucial roles in the preventiion of the infection and controlling the in vivo spread of HIV). It was rarely seen in other adjuvants or delivery systems, and is considered to be crucial for protecting human beings from HIV infection. A comprehensive analysis of the safety of this nanovector showed that it was non‐toxic in the long‐term study and could completely degrade within 21 days in vivo. Such a property might facilitate its eventual clinical application. The reason for the impressive efficacy of this nanovector is that this hydrogel can form left‐handed nanofibers which can bind with and package DNA, and doing so allows the DNA to enter host cells [this process was characterized and confirmed by transmission electron microscopy (TEM), circular dichroism (CD), and optical microscopy].6 Moreover, such nanomaterials are also convenient for preparation under mild, common laboratory conditions. Such properties further increase the likelihood that this kind of material will be used as the vector for HIV vaccines. More importantly, unlike peptide vaccines which only present linear epitopes to the host, DNA vaccines can eventually express 3D structural proteins in host cells. Nanofibers may therefore be better for presenting the conformational antigen epitopes. It is crucial to induce the production of neutralizing antibodies, which depend on the spatial conformation of the antigen, thus preventing infections from pathogens.41, 42
Similarly, an enhanced antibody response induced by peptide‐based fibers was reported by Yan‐mei Li's group in the field of cancer therapeutic vaccines.7 The sequence of peptide Q11 contains eleven amino acids, which are QQKFQFQFEQQ (Q, K, F and E respectively mean glutamine, lysine, phenylalanine, and glutamic acid). It can self‐assemble into networks of beta‐sheet‐rich nanofibers under mild physiological conditions. Because these nanofibers induce no obvious immunity, negligible tissue responses, and have a large loading capability, they are an ideal vector candidate to enhance the efficacy of vaccines (Figure 3 ).
Figure 3.
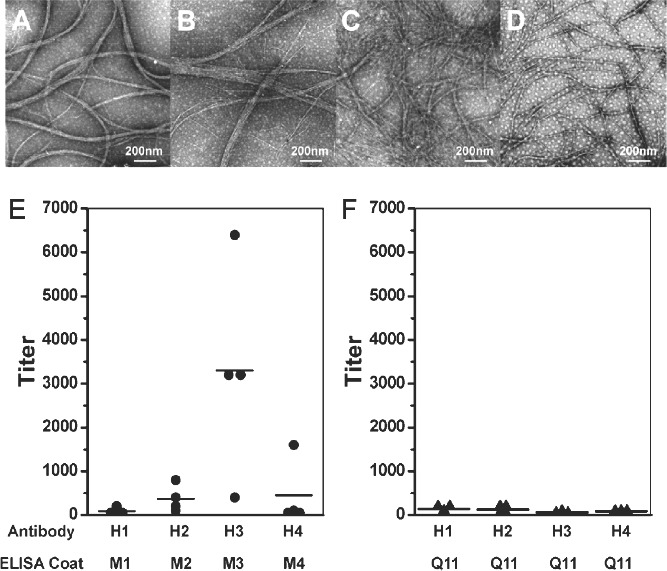
(A) H1, (B) H2, (C) H3, and (D) H4 are names of vaccines. They were prepared in a 400 μM solution, and allowed to aggregate for 8 h at room temperature. The aggregates were negatively stained with tungstophosphoric acid, and imaged with TEM. ELISA results of IgG titers elicited by four kinds of vaccines. Each spot represents the serum of one mouse after the fifth immunization. Black line represents the average value in each group. (E) H1, H2, H3, and H4 elicited antibodies against the B‐cell epitope, and (F) almost no antibody against Q11. Reproduced with permission.7 Copyright 2012, American Chemical Society.
Q11‐based nanofibers carrying multiple antigen epitopes could trigger strong IgG1, IgG2a, IgG3, IgM responses, but undetectable IFN‐γ, IL‐2, IL‐4 responses, therefore suggesting that the Q11 peptide tends to induce humoral responses via a T‐cell independent way.43 Q11 can assemble to present clustered T‐cell epitopes (an artificial peptide that contains only the main antigenic epitopes of the protein) of ovalbumin (OVA, a model antigen) in one nanofiber to elicit strong antigen‐specific primary and memory immune responses, which could protect the host from subsequent infection. This Q11 assembly itself could be degraded rapidly and excreted from the injection site to avoid potential inflammation.44 In another study, nanofibers based on Q11 succeeded in presenting the antigen epitopes and significantly improving antibody responses against malaria.45 These studies show that using peptide‐based nanofibers can significantly improve the efficacy of vaccines.
These studies employing peptide‐based nanofibers have several common mechanisms of action. Generally speaking, increased antigen uptake would lead to an enhanced immune response. This inference is also well supported by published data that nanofibers can carry more linear epitopes, or condense DNA antigens into the compact structure, or significantly improve the attachment between host cells and antigens, all of which help to increase the antigen intake of antigen‐presenting cells (APCs).6, 46, 47, 48 More interestingly, many important findings have provided some novel explanations for enhancing immune responses to vaccines caused by nanofibers. Compared with the naked antigens, those packaged by peptide‐based nanofibers could improve the antigen presentation of DCs and macrophages, both of which are crucial antigen‐presenting cells in vivo. Moreover, peptide‐based nanofibers can significantly promote the differentiation of T and B cells,49, 50 suggesting adjuvant effects of these materials.
2.2. Nanoparticles
Particles are another form of peptide‐based nanoscale material. The strategies for using peptide‐based nanoparticles to optimize the efficacy of vaccines are divided into two broad categories. The first category is the use of peptide‐based nanoparticles as the platform to display and deliver antigen epitopes. We herein call them antigen‐presenting nanoparticles (APNPs). The second category is the assembly of the antigen itself into nanoparticles (antigen as nanoparticles/AANPs) (Figure 4 ).
Figure 4.
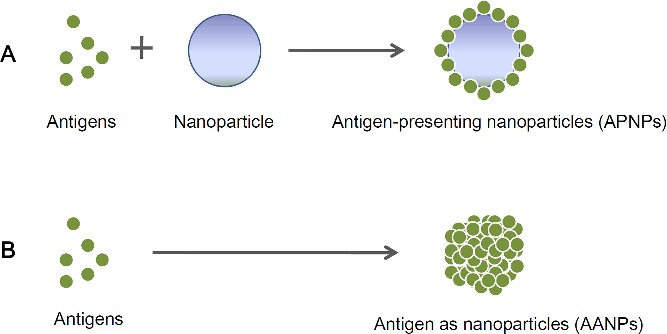
Antigens combined with nanoparticles which are used as the platform for presenting antigens. Such a complex (antigen‐presenting nanoparticles/APNPs) is usually composed of both non‐antigenic carriers and antigenic antigens (Part A). Antigens themselves are assembled into nanoparticles (antigen as nanoparticles/AANPs)
APNPs are usually composed of both a non‐antigenic backbone and antigenic epitope peptides, where antigen epitopes are displayed repetitively on the particle surface. Compared with naked antigens, APNPs can more effectively enter APCs and present antigen epitopes.51, 52 An in vivo experiment indicated that APNPs displaying 4E10 and 2F5 epitopes of HIV on their surface (two crucial targets for inducing neutralizing antibodies) induced potent antibody responses against important neutralizing antibody epitopes, which are crucial to prevent HIV infection.53 Even for the poorly immunogenic antigens (for example, actin), APNPs could induce significant, detectable antigen‐specific immune responses.54 Moreover, APNPs with multiple layers of polypeptides can effectively improve internalization by DCs and promote DC maturation without triggering secretion of inflammatory cytokines, and eventually induced a potent and balanced cellular and humoral immunity profile via cross presenting antigen epitopes to both CD8+ and CD4+ T cells (two main T‐cell subtypes, classified by their surface molecules: either CD8 or CD4). Moreover, epitope peptides carried by peptide‐based nanoparticles protected mice from attacks of Listeria Monocytogenes, indicating the superior performance of peptide‐based nanoparticles as a vaccine platform.33 For mucosal immune responses, jelly peptide particles consisting of a cationic type of cholesteryl‐group‐bearing pullulan significantly improved adhesion to the nasal epithelium. It thus was more effectively taken up by mucosal cells via the intranasal vaccination pathway. Such nanoparticles also induced effective protection via triggering vigorous neutralizing serum IgG and mucosal IgA in mice.52
AANPs can also effectively improve the efficacy of vaccines. For example, the avian influenza antigen M2e, which self‐assembled into a tetrameric form of nanoparticle, induced significantly stronger antibody responses than monomeric M2e proteins. Chickens vaccinated with such tetra‐M2e also gained protection against the infection of H5N2 (a low pathogenicity avian influenza virus).8 The dominant epitope in the HRC elements (C‐terminal heptad repeat region) of severe acute respiratory syndrome (SARS) can self‐assemble into nanoparticles (the trimmer where the structure of each monomer is coiled) via the GCN4 (a self‐assembly peptide) sequence. It induced the highly conformational (3D structured) epitope‐specific antibody in serum, which showed good neutralization against virus in the infection inhibition assay in vitro.9 Human papillomavirus (HPV) is the pathogen that causes cervical cancer. The peptide‐based self‐assembled nanoparticle of HPV antigen was also demonstrated to enhance the frequency of CD8+ memory T‐subtype cells, decrease tumor growth, and maintain a long‐term survival in a mouse model.10 All of the above‐mentioned results indicate advantageous properties of peptide‐based nanoparticles for optimizing the efficacy of vaccines.
3. Non‐peptide Polymeric Nanomaterials
Non‐peptide polymer nanomaterials herein are defined as nanoscale complexes which contain large non‐peptide molecules made of many smaller molecules of the same kind. The shape of this kind of material plays a crucial role in determining its biological functions. For example, different shapes of polymeric nanomaterials exhibit significantly different adhesion capabilities, which is an important parameter to evaluate antigen‐uptake efficacy.55 Similarly, larger aspect ratio (AR) particles have a greater impact on multiple physiological behaviors of cells, such as proliferation, apoptosis, cytoskeleton formation, adhesion, and migration. For example, silica nanoparticles with large ARs were taken up in remarkably large amounts and more quickly than those with smaller ARs.56 These results suggest a real shape effect of polymer nanomaterials on regulating immune responses induced by vaccines.
Size is another crucial physical feature to affect the biofunction of non‐peptide polymeric nanomaterials on regulating the immunity of vaccines. Polymer micelle assemblies can persist in circulation up to one week (ten times longer than monomers) after intravenous injection. Circulation time is an important indicator for estimating the duration and strength of immune responses. This result therefore suggests that the size of the polymer could affect the immune response via controlling the circulation cycle in vivo.57 Various sizes of nanobeads (from 20 to 123 nm) carrying antigen epitopes also exhibit different IFN‐γ and IL‐4 immune responses. The IFN‐γ response was significantly stronger for both 40 and 49 nm beads than for larger ones. In contrast, a higher IL‐4 response was triggered by large beads (93, 101, and 123 nm). It indicates that precise selection of nanobead size for vaccination can regulate the type of immune responses.58 In another study, among three sizes of poly(propylene sulfide) nanoparticles (20 nm, 45 nm, 100 nm), only 20 nm and 45 nm could significantly improve nanoparticles to enter into antigen presenting cells.59 As such, for two types of PLGA core particles with remarkably different sizes (200 nm and 2 μm), only the 200 nm particle could effectively target human DCs.11 Moreover, small size (20–200 nm) polystyrene nanoparticles can freely transport to lymph nodes from the injection site. By contrast, large nanoparticles (500–2000 nm) cannot migrate to lymph nodes.12
Besides the physical features of nanomaterials like shape and size, chemical or biological surface modifications are also excellent options to optimize the bio‐effects of nanomaterials. Generally speaking, the antigen uptake by cell is the first step for recognition by the immune system of pathogens. It is crucial for the form of the eventual immune response. The charge quantity on the particle surface can affect the capture of host cells against positively charged nanopolymer particles, suggesting that charge modification is a method for regulating immune response.60 CpG is a common vaccine adjuvant. CpG‐modified chitosan‐based polymer nanoparticles coated with HBV antigen can induce an enhanced IFN‐γ secretion from the spleen in mice, and achieve balanced cellular and humoral responses.13 Mild tri‐methylation modification on the chitosan surface can significantly optimize the solution of chitosan over a broad pH range, and also improve its bio‐adhesive properties. Chitosan as the vector of mucosal vaccines can effectively trigger potent humoral and mucosal immune responses.61 Moreover, studies have investigated the release ratio of DNA vaccine encapsulated by chitosan or loaded by PLGA, which is highly related to immune responses. The results indicate that a reasonable modification could regulate release: acidic conditions (pH 2) triggered 50–60% DNA release, whereas basic solutions (pH 10) triggered less release (21–25%).62, 63 PLGA surfaces modified with targeting antibodies to DCs or negative charges could effectively improve the antigen uptake and activation of DCs, and induced a stronger immune response.64, 65
In most cases, vaccine‐specific immune responses are influenced by factors such as shape, size, and modification of polymeric nanomaterials. Such views have been supported by an increasing number of studies. Among unmodified silica beads with various shapes (spheres, quasi‐hemispheres, cylinders, discoids) and diameters (from 700 nm to 3 μm), only small‐and‐spherical nanosilica can reduce their accumulation in the organs in non‐reticuloendothelial system, and such in vivo distribution is considered a crucial factor affecting vaccine efficacy.66, 67 For nanoparticles based on polypropylene sulfide (PPS) which is modified with polyhydroxylate, both surface chemistry (polyhydroxylate) and size synergistically contributed to modulate the reaction of a complement cascade. In particular, 25 nm polypropylene particles modified with polyhydroxylate exhibited the best activation for the complement cascade.68 A similar phenomenon was also found for carboxylated polystyrene core nanoparticles. Immune responses induced by OVA‐conjugated carboxylated nanoparticles are dependent on the size of particle. Nanoparticles 40∼50 nm in diameter exhibited optimized effects: bead‐linked vaccines can protect mice from developing tumors in two different model challenges (OVA and HPV tumor models), and/or clear those established tumors more rapidly.69
In general, non‐peptide polymeric nanomaterials as vaccine carriers or immunoregulators are probably closest to clinical application because of their good biological safety and effectiveness in vivo. Typically, PLGA (a kind of polymeric nanomaterial) is one of few drug carriers that has been licensed for use in clinical treatments by the Food and Drug Adminstration (FDA) of the US. However, the complex correlations between biological effects and physiochemical characteristics present a major obstacle when using polymeric nanomaterials for vaccines. To exclude the unpredictable hidden dangers, large‐scale screening is still the most dependable means to comprehensively evaluate the biological effects of polymeric nanomaterials.
4. Carbon Nanomaterials
This section will discuss vaccines based on zero‐dimensional (fullenene and its derivatives), one‐dimensional (carbon nanotubes), and two‐dimensional (graphene) carbon nanomaterials. There have been extensive studies on the use of carbon nanomaterials as adjuvants or carriers for various kinds of vaccine. Their immune effects are significantly affected by their physicochemical properties, especially surface modifications.
4.1. Fullerenes
Fullerene C60 and its derivatives can up‐regulate the expression of certain cytokines and activate innate or adaptive immunity. For example, the conjugates of fullerene C60 and immunomodulating peptide tuftsin (Thr‐Lys‐Pro‐Arg) could enhance the phagocytosis, chemotaxis, and MHC II molecule expression of murine peritoneal macrophages as compared to natural tuftsin.70 Polyhydroxylated fullerene C60, C60(OH)x, could activate an innate immunity to inhibit the growth of murine H22 hepatocarcinoma; pointing to the possibility that cancers such as carcinomae can be treated using nanoparticle‐activated immunity.71 Similarly, we found that C60(OH)20 nanoparticles significantly inhibited the growth of Lewis lung carcinoma, and such an effect was associated with an increased TNF‐α production and CD4 +/CD8 + lymphocyte ratio.72 Moreover, our research on a C82 derivative, Gd@C82(OH)22 nanoparticles, also revealed a remarkable anti‐tumor effect as a result of immunity activation.72 Further investigation showed the activation effect of Gd@C82(OH)22 on inducing phenotypic maturation of dendritic cells, and mice immunized with a mixture of ovalbumin and Gd@C82(OH)22 exhibited enhanced ovalbumin‐specific cellular immune responses.73 These results suggest that the Gd@C82(OH)22 nanoparticle can promote immune responses against tumors and is a promising nanoadjuvant for cancer vaccination.
Recently, we designed a fullerenol (polyhydroxy fullerenes) nanoparticle that can assemble into virus‐like particles with an average size of 40 nm, which as a dual‐functional nanoadjuvant brought forth both strong immune responses and a high delivery capacity against the HIV‐1 DNA antigen in mice.14 Virus‐like particles (VLPs) can usually self‐assemble by viral capsid subunits and can induce strong immune responses. The self‐assembly property of fullerenol allows the encapsulation of DNA antigen encoding the HIV‐1 envelope protein gp145 (Env) and formulation of fullerenol‐Env complex. In vitro, these VLPs significantly enhanced DNA transfection of enhanced green fluorescent protein (EGFP) DNA plasmid in human embryonic kidney 293 (HEK293) cells compared with DNA alone. The complex enhanced both innate and cellular immunity via various immunization routes. These fullerenol‐based VLPs also induced DC maturation and triggered polyvalent immunities via the activation of multiple Toll‐like receptor (TLR) signaling pathways. The fullerenol‐based VLPs up‐regulated many genes involved in the TLR signaling pathways. This is the first report showing that fullerenols designed as virus‐like particles can be used as an effective vaccine nanoadjuvant (Figure 5 ).
Figure 5.

(a) Schematic illustration of HIV‐1 envelope protein gp145 (Env) plasmid DNA encapsulated during the self‐assembly of fullerenol into a virus‐like particle. (b) TEM image of fullerenol‐Env complex. (c) Compared to naked Env immunization group, IFN‐γ production was significantly enhanced when mice were immunized with fullerenol‐Env complex via various immunization routes, including intradermal (i.d.), intramuscular (i.m.), subcutaneous (s.c.) and intranasal (i.n.) injections. (d) Fullerenol significantly promoted the maturation of DCs. (e,f) Fullerenol‐Env complex significantly up‐regulated almost all TLRs except TLR7. (e) and adaptive immunity pathways. (f) compared with DCs treated with naked Env. Reproduced with permission.14 Copyright 2013, John Wiley & Sons, Ltd.
The immune effects of fullerene derivatives and their potential use as vaccine adjuvants are most probably determined by their surface functionalization. For instance, C60 conjugated to the N‐ and C‐terminals of immunomodulatory peptide tuftsin have different biological activity.70 Also, TNF‐α secretions in RAW264.7 macrophages exposed to fullerene derivatives with different surface modifications were significantly different.74 Thus, appropriate modification on the surface may modulate the immune adjuvant effect of fullerene derivatives.
4.2. Carbon Nanotubes (CNTs)
Interactions between CNTs and the immune system are investigated and their potential uses as vaccine carriers and adjuvants have been reported in a number of studies. Dumortier et al. found that modified water‐soluble CNTs had excellent biocompatibility. They could be internalized quickly by B, T lymphocytes and macrophages, and had a negligible effect on cell viability and immune function.75 Carboxyl‐ and taurine‐modified multiwalled CNTs (MWCNTs) are also reported to be engulfed by immature DCs without affecting cell viability or causing phenotype changes.76 These results indicated that MWCNTs have good compatibility to immune cells. Pantarotto et al. covalently linked a peptide antigen from the foot‐and‐mouth disease virus (FMDV) to single‐walled CNTs (SWCNTs). This conjugation not only retains the structural integrity of the peptide, but also elicits a strong peptide‐specific antibody response in mice with undetectable cross‐reactivity to SWCNTs.15, 77 The conjugation of tumor lysate proteins and MWCNTs improved the cure rate for H22 hepatoma in mice and enhanced the anti‐tumor immune response.16 Another conjugation involving N‐terminus Plasmodium vivax apical membrane antigen‐1 (AMA‐1) peptide and MWCNTs was able to preserve the correct peptide conformation to generate high levels of antibodies.17 Due to their larger surface areas, CNTs were used to absorb T cell‐stimulating antibodies such as anti‐CD3 to induce potent activation of T cells, which can stimulate T cells more effectively than soluble anti‐CD3.78 Moreover, CNTs linked with CpG exerted increased immunostimulatory activity in vitro.79 The conjugation of synthesized tuberculin purified protein derivative (PPD) and carboxylated SWCNTs generated a preferential cellular response in the absence of potential cytotoxic effects in mice.18 For some weak immunogens (for example, human tumor antigens), SWCNTs as a carrier could be rapidly internalized into professional antigen‐presenting cells within minutes, and induced specific IgG responses against the antigen, while the same antigen alone or that mixed with an adjuvant failed to induce a similarly potent response.80 Other data indicated that simultaneous immunization with embryonic stem cells (ESC) and MWCNTs could lead to enhanced tumor rejection and significant antitumor responses in mice.81
Intrinsic physicochemical properties such as size and length can influence the effect of CNTs on regulating the immune responses induced by vaccines. This view was clarified in the experiment of Parra et al., in which complexes of short and thick CNTs, coupled covalently with the azoxystrobin derivative hapten AZc6 and bovine serum albumin (BSA), CNT‐BSA‐AZc6, produced the best antibody response under all tested conditions, thus proving that the adjuvant capability of CNTs might be dependent on shape and size.82 The conjugation method also influences the immune effect of the delivered antigens. For instance, the monoconjugate of SWCNTs and the FMDV peptide antigen induced higher antibody titers than their bis‐conjugate.77 Therefore, with appropriate functionalization method, it is possible to improve the vaccine efficiency of antigens by using CNTs as carriers.
4.3. Graphenes
Graphenes are two‐dimensional nanomaterials. Several studies have revealed immune effects of graphenes and have utilized them as vaccine carriers and adjuvants. Chen et al. uncovered simultaneous induction of autophagy and TLR4/TLR9 regulated inflammatory responses in macrophages by graphene oxide (GO), and the autophagy was considered at least partly regulated by the TLR pathway.83 Zhou et al. reported an interaction between pristine graphene and macrophages. They revealed that graphene stimulates the expression of cytokines, including IL‐1, IL‐6, IL‐10, TNF‐α, GM‐CSF, as well as chemokines such as MCP‐1, MIP‐1, MIP‐1 and RANTES. These responses were induced probably by TLR activation and nuclear factor‐kappa B‐dependent transcription.84
An in vivo study indicated that graphene nanosheets actuate the humoral immune response one day after intravenous administration, and this response is mediated by the IL‐33/ST2 pathway.85 Recently, Ni et al. utilized antibody‐absorbed GO as a potent vaccine adjuvant.86 Anti‐IL10R antibody‐absorbed GO can enhance LPS‐induced immune responses both in vitro and in vivo. This is the only report on the adjuvant effect of graphene, which suggests the potential use of modified GO as an adjuvant for vaccination.
As mentioned above, surface modification can modulate the immune activity of graphene, which was also verified by Zhi et al. They found that polyvinylpyrrolidone (PVP)‐coated GO had a lower immunogenicity than unmodified GO.87 Sasidharan et al. also reported that the carboxyl‐functionalization of graphene significantly reduces the active interaction with macrophages and ROS‐mediated toxicity effects.88 Therefore, it is possible to modulate the immune activity of graphene and develop graphene‐based nanovaccines by rational modification.
Fullerene, CNTs, and graphene have all been investigated for their adjuvant effects for various vaccines. These carbon nanomaterials exhibit relatively good biocompatibility and therefore are promising candidates for new vaccine adjuvants. Although their sizes and shapes are different, the adjuvant effects of all these carbon nanomaterials are affected by the surface modification. However, its might be interesting to compare the influence of the shape on the adjuvant effects of these three nanomaterials. Further work should also be done to elucidate the mechanisms for their adjuvant effects.
5. Gold Nanomaterials
Multiple gold‐based nanomaterials, including gold nanoparticles (AuNPs), gold nanorods (AuNRs), gold nanoshells (AuNSs), and gold nanocages (AuNCs), can stimulate the immune system. By investigating the immune responses of macrophages to different sizes in vitro, researchers found that AuNPs up‐regulated expressions of pro‐inflammatory genes IL‐1, IL‐6, and TNF‐α, and this effect was more potent with small particles (<10 nm).89 AuNRs functinalized with carboxyl groups induce expression of pro‐inflammatory mRNA, while amino groups induce expression of anti‐inflammatory mRNA. However, incubation of AuNPs with dendritic cells did not change the phenotype of the cells, while the secretion of cytokines was significantly modified.90 Enhanced cellular uptake and cytokine secretion by N9 cells when CpG was modified by the AuNPs were also demonstrated.91
The use of gold nanomaterials as vaccine adjuvants has been reported since 2006. Dykman et al. reported that colloidal gold complexed with either haptens or complete antigens could induce highly specific antibodies even without using Complete Freund's adjuvant (a common adjuvant made up of inactivated and dried mycobacteria).92 The delivering of DNA vaccine by AuNPs with covalently attached low molecular weight chitosans induced an enhanced serum antibody response 10 times more potent than that induced by naked DNA vaccine, as well as potent cellular responses via intramuscular immunization in mice.19 FMDV peptide antigen conjugated with AuNPs of different sizes initiated a size‐dependent antibody response, with the maximal antibody production in the range of 8–17 nm size of AuNPs. A three‐fold increase in the antibody response was observed when 8 nm diameter AuNPs were administered, compared to a pFMDV‐keyhole limpet hemocyanin positive control.20 Parween et al. investigated the adjuvant effect of AuNPs on a weak immunogen C‐terminal 19 kDa fragment of merozoite surface protein 1 (MSP‐1(19)).93 They found that recombinant PfMSP‐1(19) or PvMSP‐1(19) coated on AuNPs or in alum was poorly immunogenic in mice. However, a strong antibody response was observed when PfMSP‐1(19)/PvMSP‐1(19)‐coated AuNPs were immunized with alum. Therefore, antigen‐coated AuNPs with conventional adjuvants may be useful in the development of an alternate adjuvant formulation. Tetanus toxoid (TT)‐loaded AuNPs elicited significantly higher mucosal responses following oral administration, while TT alone did not elicit any mucosal response.22 Also, the formulation containing AuNPs, TT antigen, and immunostimulant Quillaja saponaria (QS, a normal immune stimulator) elicited enhanced immune responses after oral administration in mice.94 The increased titer of antibodies in animals immunized with transmissible gastroenteritis virus‐conjugated AuNRs was also reported.95 Wang et al. conjugated a recombinant protein vaccine targeting human Nogo‐66 receptor (hNgR‐Fc) to 15 nm AuNPs to develop a therapeutic vaccine for spinal cord injury therapy. They found that the conjugate produced higher titers of anti‐NgR antibody than that with Freund's adjuvant, and improved the repair of spinal cord‐injured rats.23 Yersinia pestis F1‐antigen coated on the surface of 15 nm AuNPs generated a greater IgG antibody response in mice compared to F1‐antigen alone.24
The immune effects of AuNRs have been extensively investigated. The surface chemistry of AuNRs had also strong effects on the activation state of human macrophages. For instance, AuNRs functionalized with carboxyl groups induced expressions of pro‐inflammatory mRNAs, whereas amino group modifications induced expressions of anti‐inflammatory mRNA.96 Recently, for the first time, we reported that poly(diallydimethylammonium chloride) (PDDAC)‐ and polyethyleneimine (PEI)‐modified AuNRs can act as adjuvants for HIV DNA vaccine. The two cationic polyelectrolytes PDDAC‐ and PEI‐AuNRs exhibited good transfection capabilities, while cetyltrimethylammonium bromide (CTAB)‐modified AuNRs did not. Both PDDAC‐AuNR‐Env and PEI‐AuNR‐Env significantly enhanced cellular and humoral immunity.27 They also could promote the maturation of dendritic cells, which might be the mechanism for the adjuvant effects of PDDAC‐AuNRs and PEI‐AuNRs. This is the first report on the use of AuNRs as DNA vaccine adjuvants and it provides insight into the rational design of nanoadjuvants by surface‐engineering (Figure 6 ).
Figure 6.
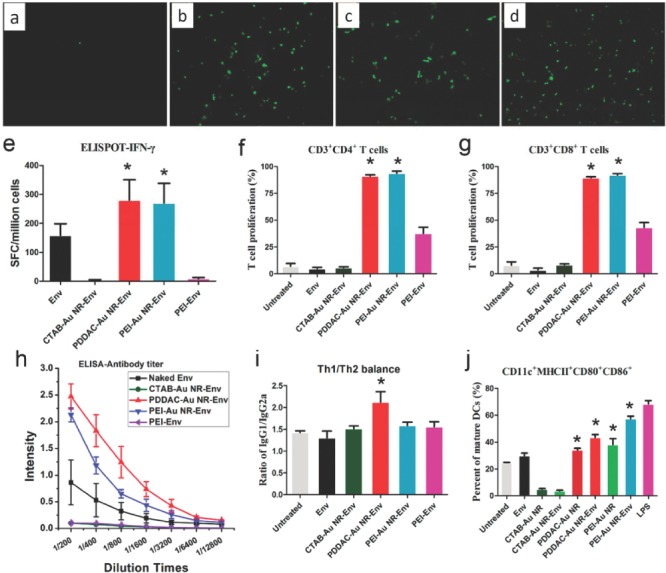
(a–d) HEK293 cells transfection capability by CTAB‐ (a), PDDAC‐ (b), and PEI‐AuNRs (c). PEI serves as a positive control (d). (e–j) AuNRs influence immune responses and dendritic cell maturation. (e) IFN‐γ analyzed by ELISPOT. (f) CD3+CD4+ T cell proliferation. (g) CD3+CD8+ T cells proliferation. (h) Env specific antibody titer. (i) Determination of the type of immune response after mice immunized with AuNR‐Env complex. (j) The effect of AuNRs and the AuNR‐Env complex on DC maturation. Reproduced with permission.27 Copyright 2012, American Chemical Society.
Moreover, AuNPs have been used for cancer vaccines. Brinas et al. reported tumor‐associated glycopeptide antigens conjugated to AuNPs.25 To construct a local lymph node (LN)‐targeted vaccine for cancer therapy, researchers conjugated the model antigen red fluorescent protein (RFP) and CpG ODN as a TLR9 ligand to 7 nm AuNPs. This complex elicited antibody production and cellular response, and also exhibited a significant antitumor effect in a RFP‐expressing melanoma tumor model.97 In another work, Parry et al. developed ‘multicopy multivalent’ nanoscale glycoconjugates by conjugation of a polymerizable Tn‐antigen glycan to AuNPs for cancer immunotherapy, which generated strong and long‐lasting production of Tn‐antigen glycan‐specific antibodies.26
Immune effects of gold nanostructures are mainly influenced by their size and shape. Niikura et al. investigated the influence of the shape and size of AuNPs on immune responses in vivo and in vitro.28 Spherical, rod, and cubic Au nanostructures coated with West Nile virus envelope (WNVE) protein were injected into mice. Spherical Au nanostructures (diameter 40 nm) induced the highest level of antibodies. Mechanism studies indicated that Au nanostructures enhanced the immune response via different cytokine pathways depending on their size and shape. The surface chemistry is also an important determinant for immune effects of Au nanomaterials, which has been demonstrated by Bartneck et al.96 and also by us.27 Therefore, rational modification of the surface of the Au nanostructure can modulate the immune responses of delivered antigens. Furthermore, it has been proven that gold nanostructures exhibit promising adjuvant effects on various kinds of vaccines, including those for infectious diseases and cancer. The potential application of gold nanostructures in vaccine development should be paid more attention.
6. Silver Nanoparticles
Silver nanoparticles (AgNPs) are reported to possess an anti‐inflammatory ability, which is useful for accelerating wound healing.98, 99 Carlson et al. found that hydrocarbon‐coated AgNPs increased the secretion of cytokines in rat alveolar macrophages in a size‐independent manner.100 Other reports also evidenced the size‐dependent inflammatory and toxicological effects of AgNPs on macrophages.101, 102 In vivo, orally administrated AgNPs induced inflammatory responses, showing increased production of cytokines, B cell distribution, and inflammatory cell infiltration.103 In both healthy and asthmatic mice, AgNP inhalation induced allergic and infammatory responses indicated by bronchiolocentric infammatory infiltration.104 The above‐mentioned studies suggest a potential biological regulation on the whole immune system in vivo.
One report on directly applying AgNPs as antigen carriers or adjuvants for vaccination showed that intraperitoneal administration with montmorillonite‐entrapped AgNPs and antigen derived from sheep red blood cells had no effect on modulating the function of plasma cells, which seems to imply a poor capability of AgNPs to improve antigen‐specific immune responses.105 Jazayeri et al. developed an oral DNA vaccine by formulating the plasmid‐encoding hemagglutinin (HA) gene of avian influenza virus H5N1(pcDNA3.1/H5) with PEG‐coated AgNPs.29 The AgNP/H5 plasmid was detected in the duodenum of the inoculated chicken 1 h post immunization. Oral immunization of chickens with AgNP/H5 increased cytokine production, as well as inducing humoral and cellular immune responses. Recently, Xu et al. evaluated the adjuvant effects of AgNPs both in vitro and in vivo. They found that AgNPs significantly enhanced the immune responses of model antigen OVA and bovine serum albuminin in mice by both intraperitoneal and subcutaneous immunization. AgNPs elicited a humoral immune response, indicated by the increase of the IgG1/IgG2a ratio and antigen‐specific IgE.106 These results imply adjuvant effects of AgNPs and the potential use of AgNPs as vaccine adjuvants.
According to the literature, both cytotoxicity and immune effects of AgNPs are affected by their size and concentration. Smaller sized AgNPs increased the production of IL‐8, while 4 nm AgNPs at high concentrations (>6.25 g/mL) decreased the production due to nanoparticle‐induced cell death.101 An in vivo study also demonstrated size‐dependent biodistribution and inflammatory induction of AgNPs by oral administration.103 Other physicochemical properties such as the surface properties and shape also need investigation for their influence on immune effects.
7. Calcium Nanomaterials
Calcium‐based inorganic nanomaterials, especially calcium phosphate nanoparticles (CaP‐NPs), are stable in physiological conditions and have been widely used as non‐viral DNA vectors.107, 108 In these nanomaterials, the cations Ca2+ or calcium phosphate can form complexes with the backbone of nucleic acids.109 DNA vaccines encapsulated by CaP‐NPs would not only be immediately taken up by cells, but would also prevent DNA from degradation by lysosomes.110 Moreover, calcium phosphates are biocompatible and easily biodegradable.111, 112, 113 Therefore, CaP‐NPs are considered biocompatible and safe systems by the FDA and have gained wide clinical application.114, 115, 116, 117, 118, 119 Considering the release of encapsulated materials in CaP‐NPs is mainly related to its pH‐dependent solubility,120 the pH environment changes from neutral to acidic during cellular uptake processes would dissolve endocytosed CaP‐NPs to release encapsulated materials into the cytoplasm.114 CaP‐NPs therefore can naturally release incorporated contents without any external trigger. Previous studies reported that surface‐modified CaP‐NPs of 80 nm size could target genes in the liver, protect the inner DNA from external DNase both in vitro and in vivo, and exhibit more effective transfection to mammalian cells.107, 120, 121
Calcium nanomaterials are also investigated as an alternative to aluminium as an adjuvant for many vaccines. For example, CaP‐NP injection elicited little inflammation at the site of administration and a little IgE response in animal models, which shows an obvious advantage over the injection of commercial alum compounds.31 Moreover, other in vivo experiments have indicated that micrometer‐sized CaP aggregates could induce high titers of neutralizing antibody, and showed high protection against viral infection, which is more potent than the aluminium adjuvant.30, 31 The same method can also be used to encapsulate DNA vaccines. The resulting complex (Cap/DNA, a size of 50–100 nm diameter) induced significant cellular and humoral immune responses, and protected animals against virus challenges.32
CaP‐NPs can also be prepared in a multishell way and functionalized with targeting molecules or biological macromolecules. The finding that CD11c (one kind of antibody)‐functionalized calcium phosphate nanoparticles could specifically target DCs resulted in many clinical applications in delivering biological molecules into cells.122 By modifying biodegradable calcium phosphate nanoparticles with B cell antigens on their surfaces, these nanoparticles could be preferentially bound and internalized by B cells. In addition, they could increase the surface expression of B cell activation markers, and promote the activation of B cells (100‐fold more efficient than soluble antigen).123 Since B cells are crucial for antibody production, these CaP‐NPs are promising as a vaccine adjuvant for inducing humoral immunity.
Meanwhile, the structure of CaP‐NPs can also be used to enhance the immune responses of other adjuvants. For example, flagellin functionalized with calcium phosphate nanoparticles could produce stronger proinflammatory cytokines (IL‐8, IL‐1β, IL‐6) than naked flagellin.124 Additionally, antigens such as viruses, proteins, or peptides can be linked onto calcium nanomaterials by a continuous flow method, calcium binding domain, or a layer‐by‐layer method. These calcium particles composed of vaccines are remarkably biocompatible,35 and could induce DC maturation,33 balanced cellular and humoral immune responses,125 and robust antigen‐specific cellular memory responses.34 Another mannose‐modified lipid‐calcium‐phosphate LCP nanoparticle delivered both tumor antigen (Trp 2 peptide) and adjuvant (CpG oligonucleotide) to dendritic cells, resulting in an increased level of tumor infiltrating CD8+ T cells and an decreased level of regulatory T cells, which significantly inhibits melanoma tumor growth in mice.36 Calcium phosphate possesses an excellent biocompatibility due to its chemical similarity to human hard tissue such as bone. It has potential for use in the development of single‐dose vaccines due to its sustained‐release capabilities. Moreover, combination of CaP with other delivery systems offers a flexible and powerful platform for novel immunotherapeutic strategy development. However, although interactions between CaP‐NPs and cells have been investigated for a long time, the exact working mechanism of CaP‐NPs has not been defined clearly. In the future, more research should be directed to revealing the CaP–bio interactions and to increase the lifespan of CaP‐based nanomaterials.
8. Biological Fates of these Nanomaterials
The biological fatea of nanoparticles is one major consideration for their potential use as vectors and/or immunoregulators for vaccines. An ideal vector/immunoregulator should be biocompatible, biodegradable, cheap to produce, not antigenic, and be able to promote an appropriate immune response.126 Among the six kinds of nanomaterials introduced in this review, except fullerene, gold and silver, all the other materials could be degraded and cleared from the body. All of the introduced nanomaterials have good biocompatibility except silver nanoparticles, which exhibit toxicity at high doses due to the released silver ions from the nanoparticles.127
Generally, nanomaterials can cause oxidative stress and inflammation on cells and organisms.128 In particular, they can induce the production of reactive oxygen species (ROS), which may cause toxic effects to the cells.129 These toxic effects are generally affected by the intrinsic physicochemical properties of the nanomaterial, including size, shape, surface charge, surface modification, chemical composition, degradation, and the formation of a “protein corona”.130 However, appropriate modulation of the physicochemical properties such as size and surface functionalization can effectively reduce toxicity and improve the clearance efficiency of nanomaterials from the body. For instance, the cytotoxicity of CTAB‐coated AuNRs could be avoided by polyelectrolyte functionalization.131 This result indicates that reasonable surface chemical modifications can significantly reduce the toxicity of metal nanoparticles. It provides us with a guide as to how to avoid serious side effects caused by nanomaterials in the process of their practical application. Therefore, a rational design for the nanomaterials is still hopeful to develop safe and efficient vaccine vectors and/or immunoregulators.
9. Summary and Outlook
At present, vaccines still face many difficulties for application worldwide because of their modest efficacy. Nanomaterials as carriers or immunoregulators are promising for optimizing the efficacy of vaccines (Table 3 ).132, 133, 134, 135 In this review, we have summarized the major working modes of multiple nanomaterials used for vaccines (Figure 1).
Table 3.
Nanomaterials might improve the efficacy of vaccines
| Vaccines | Current status | Main disadvantages | Reference |
|---|---|---|---|
| HIV | Clinical trials (Phase I, II and III) | Low efficacy of vaccine, virus variation | 132 |
| HCV | Clinical trials | Low efficacy of vaccine, virus variation | 133 |
| Rabies | Licensed | Need to receive multiple immunization | 134 |
| Malaria | Clinical trials | Low efficacy of vaccine | 135 |
| Lung cancer | Clinical trials | No efficacy of vaccine | 136 |
| Tuberculosis | Licensed | Low efficacy of vaccine, age limit | 137 |
| HBV | Licensed | Need to receive multiple immunization | 138 |
| SARS | Clinical trials | Low efficacy of vaccine, virus variation | 139 |
| Prostate cancer | Licensed | Non long‐term remission | 140 |
| Flu | Licensed | Modest efficacy of vaccine, virus variation, age limit (under the age of 50) | 141 |
This article is being made freely available through PubMed Central as part of the COVID-19 public health emergency response. It can be used for unrestricted research re-use and analysis in any form or by any means with acknowledgement of the original source, for the duration of the public health emergency.
Actually, the bioeffects of nanomaterials for regulating immune responses are affected by multiple factors, including chemical composition, physical characteristics, and biochemical modifications. In particular, the bioeffects of peptide‐based nanomaterials are mainly influenced by the shape characteristics, when they are used as vectors to optimize the efficacy of vaccines. Fibers are the most common form. Such a structure is very helpful for enhancing the uptake of antigen‐presentation cells against their targets loaded on fibers. Compared with peptide‐based nanomaterials, non‐peptide polymer and carbon nanomaterials not only possess obviously more possible shapes (ball, tube, stick, sheet, and others) and sizes within the nanoscale scope, but also are easier to chemically or biologically modify (such as adjusting charge and CpG) on the nanomaterial surface. They also therefore exhibit even more varied and complicated functions. Metal materials, such as gold and silver, are convenient to be prepared into various controllable shapes and sizes. By some surface modification, like poly(diallydimethylammonium chloride) (PDDAC) and polyethyleneimine (PEI), these materials are endowed with novel immunoregulation effects, including promoting the maturation of dendritic cells, increasing cytokine secretion, and enhancing antibody titers. The antigens encapsulated by calcium‐based nanoparticles are not only more effectively taken up by the host, but they also prevent antigens from degradation and damage by the in vivo conditions. Meanwhile, such carriers also can slowly release antigens as a harbor, producing long‐term immunostimulation and inducing potent memory immunity. Although the biological effects of other kinds of nanomaterials are not mentioned in this review, their underlying immunregulation actions are still worth deeper investigation. Such studies on uncovering the relationships between physiochemical characteristics and biological effects of nanomaterials would bring us many amazing guidelines for the design of vaccines. At present, in view of the fact that immunoregulation effects of nanomaterials are so far difficult to reproducibly predict based on their physicochemical characteristics, large‐scale screening is still a necessary means to comprehensively evaluate the biological function of nanomaterials in the next studies.
However, compared with some existing methodologies, nanomaterials possess many unique advantages. For example, the conventional vaccine vector, virus‐based vector, can express genes of antigens in a high‐efficacy way and trigger potent antigen‐specific immune responses. However, the applications of these vectors are limited greatly (usually only can be immunized once) because of anti‐vector antibody responses induced by the vectors themselves. Moreover, the complexity of element genes within virus vectors which would modulate immune responses probably brings people some unpredictable consequences. For example, Merck II phage clinical trials of HIV vaccine, in which one kind of adenovirus vector (Adv5) was used as vector, showed a higher infection ratio of HIV in group which received vaccine immunization, suggesting the unsafe aspects of application of virus vectors.142 For traditional emulsions or waters which are used as vaccine adjuvants (Freund's adjuvant, LPS), their serious side‐effects hinder their further applications in clinical trials.143 In contrast, nanomaterials hardly induce antibodies against themselves because of their poor immunogenicity. Their small size and appropriate surface functionalization allow them to be excreted easily, therefore ensuring their good biosafety for in vivo applications.
Of course, there are still some obstacles needing to be overcome for the application of nanomaterials. For example, bindings between nanomaterials and proteins in vivo would not only change both the shapes and sizes of the nanomaterials, but also might mask surface modifications of the materials when they are delivered into the body, therefore affecting the initial regulatory functions of these materials to trigger unpredictable and unwanted complications.
Considering the obvious advantages of nanomaterials for optimizing the efficacy of vaccines, uncovering the inner relations between their physicochemical characteristics and biological effects would be an important direction in their following research and development. It is certainly useful for broadening vaccine‐based prophylaxis and therapy against diseases in the future. Moreover, the structure and surface modification of the nanomaterials might be changed when they enter a body because of their bindings with proteins in vivo. Such changes should be the subject of intense investigations in the future. Thus, how to maintain the stability of nanomaterials should become another important issue in the field of developing functional nanomaterials for vaccines.
Acknowledgements
Y. Liu and Y. Xu contributed equally to this work. This work was financially supported by MOST (2012AA030308, 2012AA022703, 2012CB934000, 2011CB933400, 2010CB934004), NSFC (21025520, 21222502, 51373043, 21320102003), CAS (XDA09030305, 81361140345), MoH (2012ZX10001–008).
Biographies
Chunying Chen is a Professor at the Key Laboratory for Biological Effects of Nanomaterials and Nanosafety in the National Center for Nanoscience and Technology of China. Her research interests include: the potential toxicity of nanoparticles used for nanotechnology applications, the mechanism of toxicity, and the key properties of nanoparticles that make them toxic; therapies for malignant tumors using nanoparticles for their immunomodulatory effects and their drug delivery and tumor targeting abilities, and improving vaccines using nanomaterials as potential nonviral vectors.

Xingyu Jiang obtained his BS at the University of Chicago (1999), followed by an AM (2001) and a PhD (2004) from Harvard University, working with Prof. George Whitesides. He joined the NCNST in 2005 as a Professor in the National Center for Nanoscience and Technology of China. Xingyu's research interests include nanomaterials as vector/immunoregulators for vaccines and drugs, surface chemistry, and microfluidics.

References
- 1. Smith D. M., Simon J. K., Baker Jr., J. R. , Nat. Rev. Immunol. 2013, 13 (8), 592–605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. McMichael A. J., Haynes B. F., Nat. Immunol. 2012, 13 (5), 423–427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Kong L., Giang E., Nieusma T., Kadam R. U., Cogburn K. E., Hua Y., Dai X., Stanfield R. L., Burton D. R., Ward A. B., Wilson I. A., Law M., Science 2013, 342 (6162), 1090–1094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Drake C. G., Lipson E. J., Brahmer J. R., Nat. Rev. Clin. Oncol. 2014, 11 (1), 24–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Wisniewski T., Goni F., Expert Rev. Vaccines 2010, 9 (12), 1441–1452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Tian Y., Wang H., Liu Y., Mao L., Chen W., Zhu Z., Liu W., Zheng W., Zhao Y., Kong D., Yang Z., Zhang W., Shao Y., Jiang X., Nano Lett. 2014, 14 (3), 1439–1445. [DOI] [PubMed] [Google Scholar]
- 7. Huang Z. H., Shi L., Ma J. W., Sun Z. Y., Cai H., Chen Y. X., Zhao Y. F., Li Y. M., J. Am. Chem. Soc. 2012, 134 (21), 8730–8733. [DOI] [PubMed] [Google Scholar]
- 8. Babapoor S., Neef T., Mittelholzer C., Girshick T., Garmendia A., Shang H., Khan M. I., Burkhard P., Influenza. Res. Treat. 2011, 2011, 126794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Pimentel T. A., Yan Z., Jeffers S. A., Holmes K. V., Hodges R. S., Burkhard P., Chem. Biol. Drug Des. 2009, 73 (1), 53–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Tang J., Yin R., Tian Y., Huang Z., Shi J., Fu X., Wang L., Wu Y., Hao F., Ni B., Vaccine 2012, 30 (6), 1071–1082. [DOI] [PubMed] [Google Scholar]
- 11. Cruz L. J., Tacken P. J., Fokkink R., Joosten B., Stuart M. C., Albericio F., Torensma R., Figdor C. G., J. Control. Release 2010, 144 (2), 118–126. [DOI] [PubMed] [Google Scholar]
- 12. Manolova V., Flace A., Bauer M., Schwarz K., Saudan P., Bachmann M. F., Eur. J. Immunol. 2008, 38 (5), 1404–1413. [DOI] [PubMed] [Google Scholar]
- 13. Borges O., Silva M., de Sousa A., Borchard G., Junginger H. E., Cordeiro‐da‐Silva A., Int. Immunopharmacol. 2008, 8 (13–14), 1773–1780. [DOI] [PubMed] [Google Scholar]
- 14. Xu L. G., Liu Y., Chen Z. Y., Li W., Liu Y., Wang L. M., Ma L. Y., Shao Y. M., Zhao Y. L., Chen C. Y., Adv. Mater. 2013, 25 (41), 5928–5936. [DOI] [PubMed] [Google Scholar]
- 15. Pantarotto D., Partidos C. D., Graff R., Hoebeke J., Briand J. P., Prato M., Bianco A., J. Am. Chem. Soc. 2003, 125 (20), 6160–6164. [DOI] [PubMed] [Google Scholar]
- 16. Meng J., Duan J. H., Kong H., Li L., Wang C., Xie S. S., Chen S. C., Gu N., Xu H. Y., Yang X. D., Small 2008, 4 (9), 1364–1370. [DOI] [PubMed] [Google Scholar]
- 17. Yandara N., Pastorin G., Prato M., Bianco A., Patarroyo M. E., Lozano J. M., Vaccine 2008, 26 (46), 5864–5873. [DOI] [PubMed] [Google Scholar]
- 18. Zeinali M., Jammalan M., Ardestani S. K., Mosaveri N., Immunol. Lett. 2009, 126 (1–2), 48–53. [DOI] [PubMed] [Google Scholar]
- 19. Zhou X. F., Zhang X. H., Yu X. H., Zha X., Fu Q., Liu B., Wan X., Chen Y., Chen Y., Shan Y. M., Jin Y. H., Wu Y. G., Liu J. Q., Kong W., Shen J. C., Biomaterials 2008, 29 (1), 111–117. [DOI] [PubMed] [Google Scholar]
- 20. Chen Y. S., Hung Y. C., Lin W. H., Huang G. S., Nanotechnology 2010, 21 (19). [DOI] [PubMed] [Google Scholar]
- 21. Parween S., Gupta P. K., Chauhan V. S., Vaccine 2011, 29 (13), 2451–2460. [DOI] [PubMed] [Google Scholar]
- 22. Pokharkar V., Bhumkar D., Suresh K., Shinde Y., Gairola S., Jadhav S. S., J. Biomed. Nanotechnol. 2011, 7 (1), 57–59. [DOI] [PubMed] [Google Scholar]
- 23. Wang Y. T., Lu X. M., Zhu F., Huang P., Yu Y., Zeng L., Long Z. Y., Wu Y. M., Biomaterials 2011, 32 (31), 7988–7998. [DOI] [PubMed] [Google Scholar]
- 24. Gregory A. E., Williamson E. D., Prior J. L., Butcher W. A., Thompson I. J., Shaw A. M., Titball R. W., Vaccine 2012, 30 (48), 6777–6782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Brinas R. P., Sundgren A., Sahoo P., Morey S., Rittenhouse‐Olson K., Wilding G. E., Deng W., Barchi J. J., Bioconjugate Chem. 2012, 23 (8), 1513–1523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Parry A. L., Clemson N. A., Ellis J., Bernhard S. S. R., Davis B. G., Cameron N. R., J. Am. Chem. Soc. 2013, 135 (25), 9362–9365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Xu L. G., Liu Y., Chen Z. Y., Li W., Liu Y., Wang L. M., Liu Y., Wu X. C., Ji Y. L., Zhao Y. L., Ma L. Y., Shao Y. M., Chen C. Y., Nano Lett. 2012, 12 (4), 2003–2012. [DOI] [PubMed] [Google Scholar]
- 28. Niikura K., Matsunaga T., Suzuki T., Kobayashi S., Yamaguchi H., Orba Y., Kawaguchi A., Hasegawa H., Kajino K., Ninomiya T., Ijiro K., Sawa H., ACS Nano 2013, 7 (5), 3926–3938. [DOI] [PubMed] [Google Scholar]
- 29. Jazayeri S. D., Ideris A., Zakaria Z., Shameli K., Moeini H., Omar A. R., J. Control. Release 2012, 161 (1), 116–123. [DOI] [PubMed] [Google Scholar]
- 30. He Q., Mitchell A., Morcol T., Bell S. J. D., Clin. Diagn. Lab. Immunol. 2002, 9 (5), 1021–1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. He Q., Mitchell A. R., Johnson S. L., Wagner‐Bartak C., Morcol T., Bell S. J. D., Clin. Diagn. Lab. Immunol. 2000, 7 (6), 899–903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Joyappa D. H., Ashok Kumar C., Banumathi N., Reddy G. R., Suryanarayana V. V. S., Vet. Microbiol. 2009, 139 (1–2), 58–66. [DOI] [PubMed] [Google Scholar]
- 33. Powell T. J., Palath N., DeRome M. E., Tang J., Jacobs A., Boyd J. G., Vaccine 2011, 29 (3), 558–569. [DOI] [PubMed] [Google Scholar]
- 34. Zhou W., Moguche A. O., Chiu D., Murali‐Krishna K., Baneyx F., Nanomedicine 2014, 10 (3), 571–578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Viswanathan K., Gopinath V. P., Raj G. D., Colloids Surf. B Biointerfaces 2014, 116 (0), 9–16. [DOI] [PubMed] [Google Scholar]
- 36. Xu Z., Wang Y., Zhang L., Huang L., ACS Nano 2014, 8 (4), 3636–3645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. TJ M., SW J., KP H., FR K., AR S., M N., JC L., H W., WC Z., AZ W., JE B., JM D., Proc. Natl. Acad. Sci. USA 2011, 108 (2), 586–591.21220299 [Google Scholar]
- 38. Yan X., He Q., Wang K., Duan L., Cui Y., Li J., Angew. Chem. 2007, 46 (14), 2431–2434. [DOI] [PubMed] [Google Scholar]
- 39. Madine J., Davies H. A., Shaw C., Hamley I. W., Middleton D. A., Chem. Commun. 2012, 48 (24), 2976–2978. [DOI] [PubMed] [Google Scholar]
- 40. Sadatmousavi P., Mamo T., Chen P., Acta Biomater. 2012, 8 (9), 3241–3250. [DOI] [PubMed] [Google Scholar]
- 41. Kuwata T., Takaki K., Yoshimura K., Enomoto I., Wu F., Ourmanov I., Hirsch V. M., Yokoyama M., Sato H., Matsushita S., J. Virol. 2013, 87 (10), 5424–5436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Goo L., Milligan C., Simonich C. A., Nduati R., Overbaugh J., J. Virol. 2012, 86 (18), 9566–9582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Rudra J. S., Tian Y. F., Jung J. P., Collier J. H., Proc. Natl. Acad. Sci. USA 2010, 107 (2), 622–627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Chesson C. B., Huelsmann E. J., Lacek A. T., Kohlhapp F. J., Webb M. F., Nabatiyan A., Zloza A., Rudra J. S., Vaccine 2014, 32 (10), 1174–1180. [DOI] [PubMed] [Google Scholar]
- 45. Rudra J. S., Mishra S., Chong A. S., Mitchell R. A., Nardin E. H., Nussenzweig V., Collier J. H., Biomaterials 2012, 33 (27), 6476–6484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Cai H., Chen M. S., Sun Z. Y., Zhao Y. F., Kunz H., Li Y. M., Angew. Chem. 2013, 52 (23), 6106–6110. [DOI] [PubMed] [Google Scholar]
- 47. Jung J. P., Nagaraj A. K., Fox E. K., Rudra J. S., Devgun J. M., Collier J. H., Biomaterials 2009, 30 (12), 2400–2410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Banwell E. F., Abelardo E. S., Adams D. J., Birchall M. A., Corrigan A., Donald A. M., Kirkland M., Serpell L. C., Butler M. F., Woolfson D. N., Nat. Mater. 2009, 8 (7), 596–600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Chen J., Pompano R. R., Santiago F. W., Maillat L., Sciammas R., Sun T., Han H., Topham D. J., Chong A. S., Collier J. H., Biomaterials 2013, 34 (34), 8776–8785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Rudra J. S., Sun T., Bird K. C., Daniels M. D., Gasiorowski J. Z., Chong A. S., Collier J. H., ACS Nano 2012, 6 (2), 1557–1564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Yang Y., Neef T., Mittelholzer C., Garcia Garayoa E., Blauenstein P., Schibli R., Aebi U., Burkhard P., J. Nanobiotechnology 2013, 11, 36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Nochi T., Yuki Y., Takahashi H., Sawada S.‐i., Mejima M., Kohda T., Harada N., Kong I. G., Sato A., Kataoka N., Tokuhara D., Kurokawa S., Takahashi Y., Tsukada H., Kozaki S., Akiyoshi K., Kiyono H., Nat. Mater. 2010, 9 (7), 572–578. [DOI] [PubMed] [Google Scholar]
- 53. Wahome N., Pfeiffer T., Ambiel I., Yang Y., Keppler O. T., Bosch V., Burkhard P., Chem. Biol. Drug Des. 2012, 80 (3), 349–357. [DOI] [PubMed] [Google Scholar]
- 54. Schroeder U., Graff A., Buchmeier S., Rigler P., Silvan U., Tropel D., Jockusch B. M., Aebi U., Burkhard P., Schoenenberger C. A., J. Mol. Biol. 2009, 386 (5), 1368–1381. [DOI] [PubMed] [Google Scholar]
- 55. Doshi N., Prabhakarpandian B., Rea‐Ramsey A., Pant K., Sundaram S., Mitragotri S., J. Control. Release 2010, 146 (2), 196–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Huang X., Teng X., Chen D., Tang F., He J., Biomaterials 2010, 31 (3), 438–448. [DOI] [PubMed] [Google Scholar]
- 57. Geng Y., Dalhaimer P., Cai S., Tsai R., Tewari M., Minko T., Discher D. E., Nat. Nanotechnol. 2007, 2 (4), 249–255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Mottram P. L., Leong D., Crimeen‐Irwin B., loster S. G., Xiang S. D., Meanger J., Ghildyal R., Vardaxis N., Plebanski M., Mol. Pharm. 2007, 4(1), 73–84. [DOI] [PubMed] [Google Scholar]
- 59. Reddy S. T., Rehor A., Schmoekel H. G., Hubbell J. A., Swartz M. A., J. Control. Release 2006, 112 (1), 26–34. [DOI] [PubMed] [Google Scholar]
- 60. Lorenz M. R., Holzapfel V., Musyanovych A., Nothelfer K., Walther P., Frank H., Landfester K., Schrezenmeier H., Mailander V., Biomaterials 2006, 27 (14), 2820–2828. [DOI] [PubMed] [Google Scholar]
- 61. Mangal S., Pawar D., Garg N. K., Jain A. K., Vyas S. P., Rao D. S., Jaganathan K. S., Vaccine 2011, 29 (31), 4953–4962. [DOI] [PubMed] [Google Scholar]
- 62. Silva A. L., Rosalia R. A., Sazak A., Carstens M. G., Ossendorp F., Oostendorp J., Jiskoot W., Eur. J. Pharm. Biopharm. 2013, 83 (3), 338–345. [DOI] [PubMed] [Google Scholar]
- 63. Boyoglu S., Vig K., Pillai S., Rangari V., Dennis V. A., Khazi F., Singh S. R., Nanomedicine 2009, 5 (4), 463–472. [DOI] [PubMed] [Google Scholar]
- 64. Li N., Peng L. H., Chen X., Nakagawa S., Gao J. Q., Vaccine 2011, 29 (37), 6179–6190. [DOI] [PubMed] [Google Scholar]
- 65. Bandyopadhyay A., Fine R. L., Demento S., Bockenstedt L. K., Fahmy T. M., Biomaterials 2011, 32 (11), 3094–3105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Champion J. A., Mitragotri S., Proc. Natl. Acad. Sci. USA 2006, 103 (13), 4930–4934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Doshi N., Zahr A. S., Bhaskar S., Lahann J., Mitragotri S., Proc. Natl. Acad. Sci. USA 2009, 106 (51), 21495–2149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Reddy S. T., van der Vlies A. J., Simeoni E., Angeli V., Randolph G. J., O'Neil C. P., Lee L. K., Swartz M. A., Hubbell J. A., Nat. Biotechnol. 2007, 25 (10), 1159–1164. [DOI] [PubMed] [Google Scholar]
- 69. Fifis T., Gamvrellis A., Crimeen‐Irwin B., Pietersz G. A., Li J., Mottram P. L., McKenzie I. F. C., Plebanski M., J. Immunol. 2004, 173 (5), 3148–3154. [DOI] [PubMed] [Google Scholar]
- 70. Xu Y. Y., Zhu J. D., Xiang K., Li Y. K., Sun R. H., Ma J., Sun H. F., Liu Y. F., Biomaterials 2011, 32 (36), 9940–9949. [DOI] [PubMed] [Google Scholar]
- 71. Zhu J., Ji Z., Wang J., Sun R., Zhang X., Gao Y., Sun H., Liu Y., Wang Z., Li A., Ma J., Wang T., Jia G., Gu Y., Small 2008, 4 (8), 1168. [DOI] [PubMed] [Google Scholar]
- 72. Liu Y., Jiao F., Qiu Y., Li W., Qu Y., Tian C., Li Y., Bai R., Lao F., Zhao Y., Chai Z., Chen C., Nanotechnology 2009, 20 (41), 415102. [DOI] [PubMed] [Google Scholar]
- 73. Yang D., Zhao Y., Guo H., Li Y., Tewary P., Xing G., Hou W., Oppenheim J. J., Zhang N., ACS Nano 2010, 4 (2), 1178–1186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Xiang K., Dou Z., Li Y., Xu Y., Zhu J., Yang S., Sun H., Liu Y., J. Nanosci. Nanotechnol. 2012, 12 (3), 2169–2178. [DOI] [PubMed] [Google Scholar]
- 75. Dumortier H., Lacotte S., Pastorin G., Marega R., Wu W., Bonifazi D., Briand J. P., Prato M., Muller S., Bianco A., Nano Lett. 2006, 6 (12), 3003–3003. [DOI] [PubMed] [Google Scholar]
- 76. Wang J., Sun R. H., Zhang N., Nie H., Liu J. H., Wang J. N., Wang H., Liu Y., Carbon 2009, 47 (7), 1752–1760. [Google Scholar]
- 77. Pantarotto D., Partidos C. D., Hoebeke J., Brown F., Kramer E., Briand J. P., Muller S., Prato M., Bianco A., Chem. Biol. 2003, 10 (10), 961–966. [DOI] [PubMed] [Google Scholar]
- 78. Fadel T. R., Steenblock E. R., Stern E., Li N., Wang X. M., Haller G. L., Pfefferle L. D., Fahmy T. M., Nano Lett. 2008, 8 (7), 2070–2076. [DOI] [PubMed] [Google Scholar]
- 79. Bianco A., Hoebeke J., Godefroy S., Chaloin O., Pantarotto D., Briand J. P., Muller S., Prato M., Partidos C. D., J. Am. Chem. Soc. 2005, 127 (1), 58–59. [DOI] [PubMed] [Google Scholar]
- 80. Villa C. H., Dao T., Ahearn I., Fehrenbacher N., Casey E., Rey D. A., Korontsvit T., Zakhaleva V., Batt C. A., Philips M. R., Scheinberg D. A., ACS Nano 2011, 5 (7), 5300–5311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Mocan T., Iancu C., Int. J. Nanomed. 2011, 6, 1945–1954. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 82. Parra J., Abad‐Somovilla A., Mercader J. V., Taton T. A., Abad‐Fuentes A., J. Control. Release 2013, 170 (2), 242–251. [DOI] [PubMed] [Google Scholar]
- 83. Chen G. Y., Yang H. J., Lu C. H., Chao Y. C., Hwang S. M., Chen C. L., Lo K. W., Sung L. Y., Luo W. Y., Tuan H. Y., Hu Y. C., Biomaterials 2012, 33 (27), 6559–6569. [DOI] [PubMed] [Google Scholar]
- 84. Zhou H. J., Zhao K., Li W., Yang N., Liu Y., Chen C. Y., Wei T. T., Biomaterials 2012, 33 (29), 6933–6942. [DOI] [PubMed] [Google Scholar]
- 85. Wang X. J., Podila R., Shannahan J. H., Rao A. M., Brown J. M., Int. J. Nanomed. 2013, 8, 1733–1748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Ni G. Y., Wang Y. J., Wu X. L., Wang X. F., Chen S., Liu X. S., Immunol. Lett. 2012, 148 (2), 126–132. [DOI] [PubMed] [Google Scholar]
- 87. Zhi X., Fang H. L., Bao C. C., Shen G. X., Zhang J. L., Wang K., Guo S. W., Wan T., Cui D. X., Biomaterials 2013, 34 (21), 5254–5261. [DOI] [PubMed] [Google Scholar]
- 88. Sasidharan A., Panchakarla L. S., Chandran P., Menon D., Nair S., Rao C. N. R., Koyakutty M., Nanoscale 2011, 3 (6), 2461–2464. [DOI] [PubMed] [Google Scholar]
- 89. Yen H. J., Hsu S. H., Tsai C. L., Small 2009, 5 (13), 1553–1561. [DOI] [PubMed] [Google Scholar]
- 90. Villiers C., Freitas H., Couderc R., Villiers M. B., Marche P., J Nanopart. Res. 2010, 12 (1), 55–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Zhou Y., Chen T., Zhang L., Li Y. Y., Zhang H. L., J. Wuhan. Univ. Technol. 2011, 26 (5), 832–836. [Google Scholar]
- 92. Dykman L. A., BogatyreV V. A., Staroverov S. A., Pristensky D. V., Shchyogolev S. Y., Khlebtsov N. G., The adjuvanticity of gold nanoparticles – art. no. 616404iIn Saratov Fall Meeting 2005: Coherent Optics of Ordered and Random Media VI (Eds: Zimnyakov D. A., Khlebtsov N. G.), 2006, 6164, 16404–16404. [Google Scholar]
- 93. Parween S., Gupta P. K., Chauhan V. S., Vaccine 2011, 29 (13), 2451–2460. [DOI] [PubMed] [Google Scholar]
- 94. Barhate G., Gautam M., Gairola S., Jadhav S., Pokharkar V., Int. J. Pharm. 2013, 441 (1–2), 636–642. [DOI] [PubMed] [Google Scholar]
- 95. Staroverov S. A., Vidyasheva I. V., Gabalov K. P., Vasilenko O. A., Laskavyi V. N., Dykman L. A., Bull. Exp. Biol. Med. 2011, 151 (4), 436–439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Bartneck M., Keul H. A., Singh S., Czaja K., Bornemann J., Bockstaller M., Moeller M., Zwadlo‐Klarwasser G., Groll J., ACS Nano 2010, 4 (6), 3073–3086. [DOI] [PubMed] [Google Scholar]
- 97. Lee I. H., Kwon H. K., An S., Kim D., Kim S., Yu M. K., Lee J. H., Lee T. S., Im S. H., Jon S., Angew. Chem. 2012, 51 (35), 8800–8805. [DOI] [PubMed] [Google Scholar]
- 98. Nadworny P. L., Wang J. F., Tredget E. E., Burrell R. E., Nanomed‐Nanotechnol 2008, 4 (3), 241–251. [DOI] [PubMed] [Google Scholar]
- 99. Sibbald R. G., Contreras‐Ruiz J., Coutts P., Fierheller M., Rothman A., Woo K., Adv. Skin Wound Care 2007, 20 (10), 549–558. [DOI] [PubMed] [Google Scholar]
- 100. Carlson C., Hussain S. M., Schrand A. M., Braydich‐Stolle L. K., Hess K. L., Jones R. L., Schlager J. J., J. Phys. Chem. B 2008, 112 (43), 13608–13619. [DOI] [PubMed] [Google Scholar]
- 101. Park J., Lim D. H., Lim H. J., Kwon T., Choi J. S., Jeong S., Choi I. H., Cheon J., Chem. Commun. 2011, 47 (15), 4382–4384. [DOI] [PubMed] [Google Scholar]
- 102. Martinez‐Gutierrez F., Thi E. P., Silverman J. M., de Oliveira C. C., Svensson S. L., Hoek A. V., Sanchez E. M., Reiner N. E., Gaynor E. C., Pryzdial E. L. G., Conway E. M., Orrantia E., Ruiz F., Av‐Gay Y., Bach H., Nanomed‐Nanotechnol 2012, 8 (3), 328–336. [DOI] [PubMed] [Google Scholar]
- 103. Park E. J., Bae E., Yi J., Kim Y., Choi K., Lee S. H., Yoon J., Lee B. C., Park K., Environ. Toxicol. Phar. 2010, 30 (2), 162–168. [DOI] [PubMed] [Google Scholar]
- 104. Chuang H. C., Hsiao T. C., Wu C. K., Chang H. H., Lee C. H., Chang C. C., Cheng T. J., Int. J. Nanomed. 2013, 8, 4495–4506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Daniel S., Tharmaraj V., Sironmani T. A., Pitchumani K., Appl. Clay. Sci. 2010, 48 (4), 547–551. [Google Scholar]
- 106. Xu Y. Y., Tang H., Liu J. H., Wang H. F., Liu Y. F., Toxicol. Lett. 2013, 219 (1), 42–48. [DOI] [PubMed] [Google Scholar]
- 107. Altınogˇlu E. I., Russin T. J., Kaiser J. M., Barth B. M., Eklund P. C., Kester M., Adair J. H., ACS Nano 2008, 2 (10), 2075–2084. [DOI] [PubMed] [Google Scholar]
- 108. Graham F., van der Eb A., Virology 1973, 52 (2), 456–467. [DOI] [PubMed] [Google Scholar]
- 109. Truong‐Le V. L., Walsh S. M., Schweibert E., Mao H.‐Q., Guggino W. B., August J. T., Leong K. W., Arch. Biochem. Biophys. 1999, 361 (1), 47–56. [DOI] [PubMed] [Google Scholar]
- 110. Giger E. V., Puigmartí‐Luis J., Schlatter R., Castagner B., Dittrich P. S., Leroux J.‐C., J. Control. Release 2011, 150 (1), 87–93. [DOI] [PubMed] [Google Scholar]
- 111. Dorozhkin S. V., Epple M., Angew. Chem. 2002, 41 (17), 3130–3146. [DOI] [PubMed] [Google Scholar]
- 112. Sokolova V., Epple M., Angew. Chem. 2008, 47 (8), 1382–1395. [DOI] [PubMed] [Google Scholar]
- 113. Dobrovolskaia M. A., McNeil S. E., Nat. Nanotechnol. 2007, 2 (8), 469–478. [DOI] [PubMed] [Google Scholar]
- 114. Morgan T. T., Muddana H. S., Altinoglu E. I., Rouse S. M., Tabakovic A., Tabouillot T., Russin T. J., Shanmugavelandy S. S., Butler P. J., Eklund P. C., Nano Lett. 2008, 8 (12), 4108–4115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Epple M., Ganesan K., Heumann R., Klesing J., Kovtun A., Neumann S., Sokolova V., J. Mater. Chem. 2010, 20 (1), 18–23. [Google Scholar]
- 116. Pittella F., Zhang M., Lee Y., Kim H. J., Tockary T., Osada K., Ishii T., Miyata K., Nishiyama N., Kataoka K., Biomaterials 2011, 32 (11), 3106–3114. [DOI] [PubMed] [Google Scholar]
- 117. Bose S., Tarafder S., Acta Biomater. 2012, 8 (4), 1401–1421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Chiu D., Zhou W., Kitayaporn S., Schwartz D. T., Murali‐Krishna K., Kavanagh T. J., Baneyx F., Bioconjugate Chem. 2012, 23 (3), 610–617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Shadanbaz S., Dias G. J., Acta Biomater. 2012, 8 (1), 20–30. [DOI] [PubMed] [Google Scholar]
- 120. Roy I., Mitra S., Maitra A., Mozumdar S., Int. J. Pharm. 2003, 250 (1), 25–33. [DOI] [PubMed] [Google Scholar]
- 121. Sokolova V. V., Radtke I., Heumann R., Epple M., Biomaterials 2006, 27 (16), 3147–3153. [DOI] [PubMed] [Google Scholar]
- 122. Kozlova D., Chernousova S., Knuschke T., Buer J., Westendorf A. M., Epple M., J. Mater. Chem. 2012, 22 (2), 396–404. [Google Scholar]
- 123. Temchura V. V., Kozlova D., Sokolova V., Überla K., Epple M., Biomaterials 2014, 35 (23), 6098–6105. [DOI] [PubMed] [Google Scholar]
- 124. Kozlova D., Sokolova V., Zhong M., Zhang E., Yang J., Li W., Yang Y., Buer J., Westendorf A., Epple M., Yan H., Virol. Sin. 2014, 29 (1), 33–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Jones S., Asokanathan C., Kmiec D., Irvine J., Fleck R., Xing D., Moore B., Parton R., Coote J., Vaccine 2013, 32 (33), 4234–4242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Aguilar J. C., Rodriguez E. G., Vaccine 2007, 25, 3752–3762. [DOI] [PubMed] [Google Scholar]
- 127. Kittler S., Greulich C., Diendorf J., Koller M., Epple M., Chem. Mater. 2010. 22(16), 4548–4554. [Google Scholar]
- 128. Li Y. F., Chen C., Small 2011, 7, 2965–2980. [DOI] [PubMed] [Google Scholar]
- 129. Xia T., Kovochich M., Brant J., Hotze M., Sempf J., Oberley T., Sioutas C., Yeh JI., Wiesner MR., Nel AE., Nano Lett. 2006, 6, 1794–1807. [DOI] [PubMed] [Google Scholar]
- 130. Xu Y. Y., Lin X. Y., Chen C. Y., Chin. Sci. Bull. (Chin. Ver.) 2013, 58(24), 2466–2478. [Google Scholar]
- 131. Qiu Y., Liu Y., Wang L. M., Xu L., Bai R., Ji Y., Wu X., Zhao Y., Li Y., Chen C., Biomaterials. 2010, 31, 7606–7619. [DOI] [PubMed] [Google Scholar]
- 132. Rerks‐Ngarm S., Pitisuttithum P., Ganguly N., Zhang L., Tamashiro H., Cooper D. A., Vun M. C., Bela B., Ditangco R., Van Kinh N., Bernstein A., Osmanov S., Mathieson B., Kent S. J., Shao Y., Curr. Op. HIV AIDS 2010, 5 (5), 435–452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Man John Law L., Landi A., Magee W. C., Lorne Tyrrell D., Houghton M., Emerg. Microbes Infect. 2013, 2, e79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134. McGettigan J. P., Expert Rev. Vaccines 2010, 9 (10), 1177–1186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135. Schwartz L., Brown G. V., Genton B., Moorthy V. S., Malar J. 2012, 11, 11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136. Kelly R. J., Giaccone G., Cancer J. 2011, 17 (5), 302–308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137. Ottenhoff T. H., Kaufmann S. H., PLoS Pathog. 2012, 8 (5), e1002607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Abushady E. A., Gameel M. M., Klena J. D., Ahmed S. F., Abdel‐Wahab K. S., Fahmy S. M., World J. Hepatol. 2011, 3 (6), 147–156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139. Du L., He Y., Zhou Y., Liu S., Zheng B. J., Jiang S., Nat. Rev. Microbiol. 2009, 7 (3), 226–236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Drake C. G., Nat. Rev. Immunol. 2010, 10 (8), 580–593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141. Lee Y. T., Kim K. H., Ko E. J., Lee Y. N., Kim M. C., Kwon Y. M., Tang Y., Cho M. K., Lee Y. J., Kang S. M., Clin. Exp. Vaccine Res. 2014, 3 (1), 12–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142. Robb M. L., Lancet. 2008, 372 (9653), 1881–1893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143. Reed S. G., Nat Med. 2013, 19 (12), 1597–1608. [DOI] [PubMed] [Google Scholar]


