Summary
Checkpoint blockade (CPB) therapy can elicit durable clinical responses by reactivating an exhausted immune response. However, response rates remain limited, likely secondary to a lack of a tumor-reactive immune infiltrate. Chimeric antigen receptor (CAR) T cells may provide the necessary tumor-targeting immune infiltrate and a highly specific antitumor immune response. This can be further amplified by the addition of CPB agents, which serve to counteract the immune inhibitory environment undermining optimal CAR T-cell efficacy. Herein, we review preclinical and clinical combination therapy with CAR T cells and CPB agents, with a focus on solid tumor malignancies.
Keywords: CAR T cell, immunotherapy, chimeric antigen receptor, PD-1, checkpoint blockade
Introduction
Tumor escape from immune elimination is a hallmark of cancer (Dunn et al., 2002). Escape mechanisms include downregulation of major histocompatibility complex (MHC) receptors, low antigen expression and expression of checkpoint ligands (such as programmed death-ligand 1 [PD-L1]). All together, these elements compose a microenvironment that resists infiltration, persistence, and function of immune cells (Sharma et al., 2017).
Therapeutic targeting with checkpoint blockade (CPB) is the most successful immunotherapeutic intervention to date for cancer (Ribas and Wolchok, 2018). The checkpoint signaling that evolved as a means of limiting overactive immune responses and preventing autoimmunity is co-opted by tumor cells to block T-cell immunity, an interaction that CPB targets to reinvigorate antitumor immunity. By reactivating exhausted T cells, CPB has achieved responses in previously untreatable cancers, including long-lasting tumor regressions. However, the enthusiasm generated by the advent of this therapy has been dampened by the recognition that objective response rates to CPB have been limited to <40% for even the most immunogenic malignancies (Zou et al., 2016). Our current understanding of treatment non-responders is that their tumor environment lacks the necessary substrate upon which CPB acts: a preexisting antitumor immune response that can be reactivated by CPB therapy (Huang et al., 2017). Owing to a number of factors, such as decreased antigenicity, MHC downregulation, and resistance to IFN-γ signaling, these “cold” tumor microenvironments do not contain effector T cells to unleash on the tumor. A “hot” tumor microenvironment with T cells that recognize the tumor but have become exhausted may be a prerequisite for response to CPB (Taube et al., 2012). Efforts to combine CPB with another immunotherapy agent that creates an immunogenic tumor microenvironment or directly provides a T-cell infiltrate may therefore provide an ideal combination therapy to increase response rates. This could be accomplished through immunogenic activation (in the form of a cancer vaccine or oncolytic virus, which can release tumor antigens and attract immune effectors) or the direct addition of an antigen-recognizing cellular infiltrate (in the form of adoptive cell therapy). Our focus in this review will be on the adoptive cell therapy approach—in particular, the use of CAR T-cell therapy, which targets T cells directly to tumor cells.
CAR T-cell therapy can achieve remarkably effective—and durable—responses (June et al., 2018). The CAR provides high-avidity binding to cell-surface antigens independent from the MHC receptor, triggering robust T-cell activation and antitumor response (Sadelain et al., 2013). The addition of costimulatory domains to CAR T cells, which enhanced their persistence, catapulted CAR T-cell therapy forward in the clinic. Early trials demonstrated that the CD19targeting CAR could achieve long-lasting remissions in patients with chemotherapy-refractory hematologic malignancies (Park et al., 2018; Schuster et al., 2017). However, this dramatic success has yet to be translated to solid tumors. Solid tumors pose several obstacles, including an immunosuppressive tumor microenvironment that exhausts CAR T cells, making combination therapy with CPB particularly relevant for these malignancies (Joyce and Fearon, 2015). Combining CAR T-cell therapy and CPB may produce a synergistic effect: CAR T cells can provide an infiltrate for immunogenically silent tumors, and CPB can reverse CAR T-cell inhibition and restore functional persistence. In preclinical studies, combination therapy has increased efficacy over each agent alone, supporting the extension to clinical studies in patients (Cherkassky et al., 2016; Hu et al., 2018; John et al., 2013; Strome et al., 2003).
CPB
Checkpoints
Immune system checkpoints play a critical role in balancing T-cell activation and inhibition, as well as preventing autoimmunity. Examples include programmed cell death protein 1 (PD-1; CD279), PD-L1 (CD274; B7-H1), cytotoxic T-lymphocyte–associated protein 4 (CTLA-4; CD154), T-cell immunoglobulin and mucin-domain containing-3 (TIM3), and lymphocyte-activation gene 3 (LAG3; CD223). Downregulation or knockout of CTLA-4 causes rampant lymphoproliferation and death from autoimmunity (Tivol et al., 1995), and in cancer models, CTLA-4 signal blockade leads to tumor regression (Leach et al., 1996). In infection models, repeated signaling through the PD-1 receptor causes T-cell inhibition and chronic infection (Barber et al., 2006), whereas in cancer models, upregulation of PD-L1 leads to tumor persistence and PD-L1 blockade results in tumor regression (Curiel et al., 2003; Iwai et al., 2002).
Cancer cells limit antitumor responses by expressing immune checkpoints. The tumor microenvironment is replete with inhibitory PD-L1 signaling, as PD-L1 can be expressed on both tumor cells and host cells, including fibroblasts (Nazareth et al., 2007), dendritic cells, macrophages, and B and T cells. PD-L1 expression can reflect an adaptive cancer response to immune infiltration or can occur independently of immune cell activation (Pardoll, 2012). In the context of adaptive immune resistance, PD-L1 and PD-L2 overexpression are induced by T cell–secreted immunostimulatory cytokines IFN-γ and TNF-α (Spranger et al., 2013; Tumeh et al., 2014). Cancer cells can also express PD-L1 innately in response to disruption of the regulatory 3′ region of the PD-L1 gene (Kataoka et al., 2016) or activation of oncogenic pathways such as PI3K/Akt, EGFR, STAT3, MYC, and cyclin-dependent kinase 5 (Casey et al., 2016; Marzec et al., 2008; Parsa et al., 2007).
Clinical Success of CPB
The first CPB therapy to emerge and ultimately receive FDA approval was the CTLA-4–blocking agent ipilimumab, a fully human IgG1 antibody that was first approved for melanoma in 2011. Although the objective response rates were low, excitement over the potential promise of CPB came in the form of durability of response, reflecting activation of a long-lasting antitumor immune attack attributable to T cells (Ribas et al., 2005). The most impressive clinical responses were associated with immune cell infiltration and resulting extensive tumor necrosis (Hodi et al., 2003), establishing the ability of CPB to activate an immune response. Phase III studies confirmed that ipilimumab, alone or in combination with other agents, produced stronger anticancer responses than traditional methods such as chemotherapy (Hodi et al., 2010; Robert et al., 2011).
Pembrolizumab, a humanized anti–PD-1 antibody, has since demonstrated even more impressive results and a more manageable toxicity profile. Pembrolizumab achieved an overall objective response rate of 19.4% in patients with non-small cell lung cancer (NSCLC), 45.2% in patients with PD-L1–positive tumors, and 33% in patients with melanoma (Garon et al., 2015; Ribas et al., 2016). Pembrolizumab has also shown efficacy in patients with refractory head and neck cancer (Bauml et al., 2017) and relapsed classic non-Hodgkin lymphoma (Chen et al., 2017), with complete response rates as high as 22%. Nivolumab, another fully human anti–PD-1 antibody, has also produced encouraging results, with response rates of 18%−28% in patients with NSCLC, renal cell carcinoma, and melanoma (Borghaei et al., 2015; Topalian et al., 2012). Of note, a considerable portion of the patients who respond to PD-1 blockade experience long-lasting tumor regression, which suggests the involvement of memory T cells in an ongoing antitumor response (Lipson et al., 2012). CPB is under continued investigation and is now part of the standard therapy for advanced melanoma, NSCLC, Merkel cell carcinoma, head and neck squamous cell carcinoma, urothelial and kidney cancers, refractory Hodgkin lymphoma, hepatocellular carcinoma, gastric cancer, and cancers with high microsatellite instability (MSIH) (Topalian et al., 2012; Zappasodi et al., 2018).
Biomarkers of Response to CPB
Despite the durable responses achieved with CPB, response rates remain under 50%, highlighting the need to identify biomarkers of immune activation that predict response and suggest mechanisms by which we can augment efficacy.
PD-L1.
PD-L1 expression on tumor cells, the first validated predictor of response to CPB, has been investigated in prospective cohorts of patients treated with CPB (Herbst et al., 2014; Tumeh et al., 2014). Higher tumor PD-L1 expression is an established biomarker associated with improved clinical response to CPB (Garon et al., 2015; Robert et al., 2015; Topalian et al., 2012). But in these studies, there were some patients without tumor PD-L1 expression who nevertheless responded, suggesting that PD-L1 expression specifically on tumor cells is neither necessary nor sufficient for response. Instead, immune cells (myeloid-derived suppressor cells [MDSCs], other T cells, antigen-presenting cells) may act as the primary or at least a redundant source of PD-L1, establishing immune resistance (Tang et al., 2018). Additional reasons for why the general correlation between tumor PD-L1 and CPB response is not exact include the following: alternative ligands, such as PD-L2, or other inhibitory pathways may act as immune silencers (Taube et al., 2015); PD-L1 expression may be stimulated de novo following influx of T-cell infiltrate such that pretreatment sampling does not reflect expression over the course of tumor regression (Herbst et al., 2014; Powles et al., 2014) and there may be sampling bias (Taube et al., 2012).
Results of randomized clinical trials also suggest the limitations of screening by PD-L1 tumor staining (Wolchok et al., 2013). Overall, PD-L1 staining of a single specimen may misrepresent what is fundamentally being sought: evidence of an existing immune infiltrate that can be reactivated.
Tumor Mutational Burden (TMB) and Neoantigens.
Tumors often and to varying degrees express neoantigens within the context of MHC as a result of mutations that generate altered proteins. These neoantigens can be recognized as non-self and have binding affinity for MHC to allow representation by antigen-presenting cells (Rizvi et al., 2015; Schreiber et al., 2011). As TMB is correlated with quantity of neoantigens, TMB correlates with response to immunotherapy. Patients whose NSCLC tumors had higher levels of nonsynonymous mutations—namely, mutations that result in the production of a different amino acid and, subsequently, a different protein—were more responsive to PD-1 blockade (Rizvi et al., 2015). Other tumors with high TMB demonstrate high response rates to CPB therapy, including desmoplastic melanoma (Eroglu et al., 2018), virally induced Merkel cell and hepatocellular carcinoma (El-Khoueiry et al., 2017; Nghiem et al., 2016), and carcinogen-induced cancers (Garon et al., 2015).
The clearest demonstration of the association between TMB and response to CPB is seen in MSI-H colorectal cancers, with an overall response rate of 53% in MSI-H tumors (Le et al., 2017). These results led to FDA approval of CPB for any MSI-H tumor in 2017, which marked the first approval of CPB based on a biomarker regardless of tissue histologic profile.
Simply assessing the overall mutational burden misses the nuance of the antigen quality and suggests the potential for a response, as opposed to an actual response (Blank et al., 2016). Clonal antigens, for example, which occur early in tumor development (McGranahan et al., 2016), and neoantigens that are cross-reactive with known microbial epitopes can elicit a stronger immune response (Balachandran et al., 2017), compared with antigens without those qualities. In addition, tumors can be heterogenous in terms of mutational load, which means that a biopsy prone to sampling bias may not determine the actual potential to elicit an immune response (Alexandrov et al., 2013).
Tumor-Infiltrating Lymphocytes (TILs).
Perhaps the most predictive biomarker is the target and end effector of CPB therapy: TILs. TILs are an indicator of a “hot” or immune-inflamed tumor and can indicate whether an immune response is present and directed at the tumor. TIL grade is associated with disease-specific survival in melanoma (Azimi et al., 2012), colorectal cancer (Galon et al., 2006), ovarian cancer (Zhang et al., 2003), and lung adenocarcinoma (Suzuki et al., 2013). Therefore, the presence of an infiltrate is itself a biomarker; furthermore, the characteristics or quality of the infiltrate can also predict response (Melero et al., 2014). The presence of CD8+ T cells has been associated with improved response to chemotherapy and, more recently, CPB (Danilova et al., 2016). A high density of CD8+ T cells on the leading tumor edge has been associated with improved response to immunotherapy (Gajewski et al., 2010; Tumeh et al., 2014).
Need for an Immune Infiltrate
Even the most immunogenic malignancies, such as melanomas, which generally exhibit high levels of both neoantigens and TILs, fail to respond to CPB in significant numbers. One explanation is that these immune features are not prominent in the tumor microenvironment (Obeid et al., 2016). These features need an antigen-sensitive immune infiltrate that can be reactivated. Adoptive cell therapy can serve this need, and its ability to establish an infiltrate has been shown to be feasible.
CAR T Cells
In CAR T-cell therapy, genetically engineered autologous T cells expressing a CAR are administered to the patient. CARs bind antigen in an MHC-independent manner and, therefore, are not susceptible to MHC downregulation, a mechanism often used by malignant cells to evade eradication. The “chimeric” namesake refers to the fusion of two separate protein domains: one that provides the T cell with the antigen recognition to bind to tumor antigen and one that provides signaling for T-cell activation (a CD3ζ intracellular signaling domain with or without costimulatory signaling) (Sadelain et al., 2003). CAR expression is most often achieved using retroviral- or lentiviral-mediated gene transfer. As the T cell is capable of high-fidelity transgene expression and is a potent antitumor effector, it is an ideal host cell for the CAR. The T cell’s rapidly dividing nature facilitates viral integration, its transcriptional machinery promotes high-level transgene expression from viral promoters, and its ability to establish memory serves to reinforce long-lasting transgene expression.
Improved Persistence with Costimulation of CAR T Cells
First-generation CARs designed with only the CD3ζ intracellular signaling domain were prone to cell death and anergy. Second- and third-generation CARs, which were designed with one or multiple costimulatory domains (e.g., 4–1BB, CD28, ICOS, and OX40) in addition to the CD3ζ domain (Sadelain et al., 2013), have demonstrated superior antitumor efficacy, which has been associated with enhanced T-cell accumulation, cytokine secretion, cytotoxicity, and long-term persistence (Adusumilli et al., 2014).
Clinical Success of CAR T Cells in Liquid Tumors
CD19 CAR T-cell therapy has achieved a durable clinical benefit in a subset of patients with chronic lymphocytic leukemia (Brentjens et al., 2011; Kochenderfer et al., 2012; Schuster et al., 2015) and non-Hodgkin lymphoma (Geyer and Brentjens, 2016; Kochenderfer et al., 2012; Kochenderfer et al., 2010; Savoldo et al., 2011), with the most dramatic results—complete response rates of 80%—achieved in patients with acute lymphoblastic leukemia (Davila et al., 2014; Gardner et al., 2017; Lee et al., 2015; Maude et al., 2014). The FDA recently approved CD19 CAR T-cell therapy for the treatment of relapsed or refractory pediatric and young-adult B-cell acute lymphoblastic leukemia (Kymriah [tisagencleucel]; Novartis) and relapsed or refractory adult large B-cell lymphoma (Yescarta [axicabtagene ciloleucel]; Kite Pharma).
Limited Success of CAR T Cells in Solid Tumors
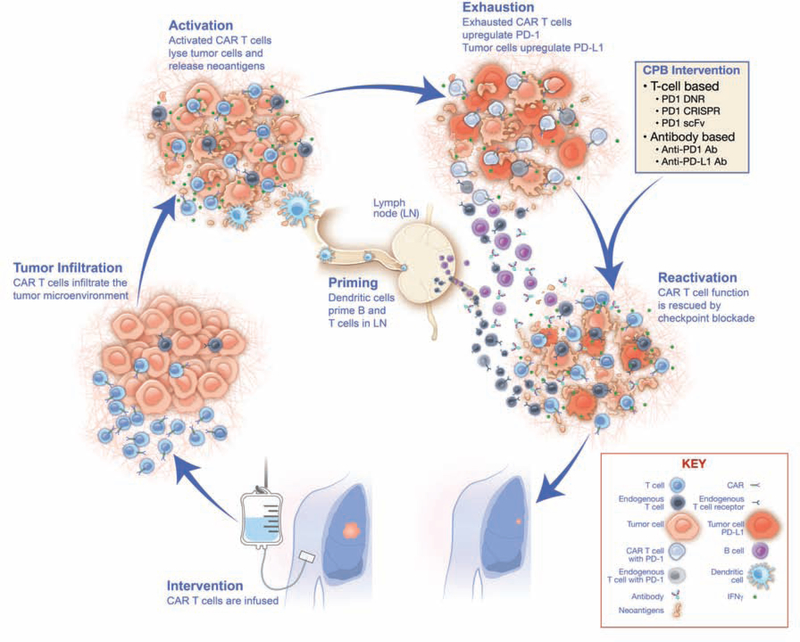
Although hematologic malignancies have a high burden of disease, they are within the same peripheral blood compartment as intravenously administered T cells and do not feature a dense tumor microenvironment that is difficult to infiltrate. In contrast, solid tumors express antigens heterogeneously, are sequestered in organ tissue, and feature an immunosuppressive tumor microenvironment in which T cells function suboptimally (Moon et al., 2014; Morello et al., 2016). In addition, CAR T cells have to overcome several hurdles to infiltrate the tumor and remain activated (Figure 1), as discussed below.
Figure 1. Mechanisms of rescue of CAR T-cell exhaustion with checkpoint blockade.
Intervention: Chimeric antigen receptor (CAR) T cells are administered via intravenous infusion. Tumor Infiltration: CAR T cells infiltrate the tumor microenvironment. Activation: Following major histocompatibility complex (MHC)-independent recognition of surface antigen, CAR T cells are activated and begin releasing cytokines (e.g., IFN-γ) and lyse tumor cells. Priming: Released neoantigens from lysed cancer cells are recognized by dendritic cells and carried to the lymph nodes, where endogenous B and T cells are primed, generating additional adaptive responses. Exhaustion: In response to IFN-γ, tumor cells upregulate programmed death-ligand 1 (PD-L1), which interacts with programmed cell death protein 1 (PD-1) on the surface of exhausted CAR T cells. Checkpoint blockade (CPB) Intervention and Reactivation: Upon administration of CPB, CAR T-cell and endogenous T-cell function is rescued to enable killing of tumor cells. Ab, antibody; CRISPR, clustered regularly interspaced short palindromic repeats; DNR, dominant negative receptor; scFv, single chain variable fragment.
Trafficking of CAR T Cells to the Tumor
The recognition that the efficacy of CPB relies on the reactivation of an existing antitumor T-cell response underscores the importance of achieving high numbers of tumor-infiltrating T cells. With this goal as a priority, our research group employed regional administration of CAR T cells to augment T-cell infiltration into the tumor (Figure 2A), which promoted early intratumoral accumulation and enhanced antitumoral efficacy in a clinically relevant model of mesothelioma (Adusumilli et al., 2014). Furthermore, we demonstrated that regional administration resulted in systemic circulation of CAR T cells that retain their functional activity, establishing a long-term systemic presence capable of eradicating metastatic tumor sites. In our phase I clinical trial in patients with pleural mesothelioma and breast and lung primaries metastatic to the pleura (NCT02414269), we have demonstrated that intrapleural administration results in circulating CAR T cells detectable in systemic circulation up to several months following a single infusion. This ability of locally administered T cells to circulate and persist in the periphery may open new avenues of treatment for other metastatic cancers with accessible tumor sites, which can translate into sustained responses in more inaccessible tumors. Examples include intraperitoneal administration of MUC16-targeted CAR T cells in an animal model of ovarian peritoneal carcinomatosis (Koneru et al., 2015), hepatic vascular infusion of CEA-targeting CARs in a model of CEA-expressing colorectal liver metastases (Burga et al., 2015), and intracranial administration targeting IL13α2-expressing glioblastoma (Brown et al., 2012). Local administration of CAR T cells is being tested in clinical trials for the treatment of other solid tumors (mesothelioma [NCT02414269], glioma [NCT02208362], liver cancer and metastases [NCT03130712], and head and neck cancers [NCT01818323]).
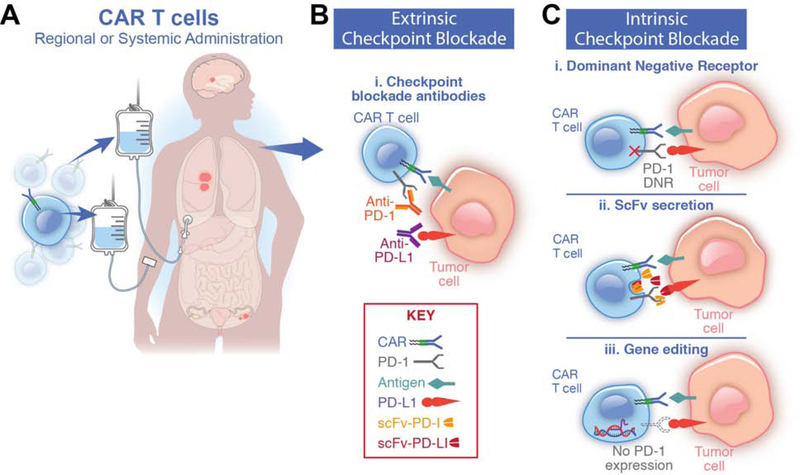
Figure 2. Strategies to combine CAR T cells and checkpoint blockade.
(A) Chimeric antigen receptor (CAR) T cells are delivered either systemically via intravenous infusion or by regional administration, which enhances primary tumor elimination. Both methods establish systemic immunosurveillance. Combination therapy with programmed death-ligand 1 checkpoint blockade (PD-1 CPB) is provided using either cell-extrinsic or cell-intrinsic strategies.
(B) Extrinsic CPB relies on programmed cell death protein 1 (PD-1) receptor/ligand blocking antibodies. A potential advantage of cell-extrinsic CPB is additional enhancement of endogenous antitumor T-cell immunity.
(C) Cell-intrinsic PD-1 CPB uses genetic engineering to express nucleic acids or proteins that interfere with PD-1/PD-L1 signaling. A possible advantage to cell-intrinsic CPB is efficacy with a single administration lasting the lifetime of the T cell. (i) PD-1 dominant negative receptor (DNR) is constructed lacking the intracellular signaling domain. It acts as a decoy receptor that competes with the native PD-1 receptor, thereby decreasing inhibitory signaling via the native wild-type receptor. (ii) CAR T cells can be employed to produce PD-1/PD-L1 blocking single chain variable fragments (scFvs), providing regional antibody blockade of PD-1 receptor/ligand binding. (iii) Gene-editing techniques such as CRISPR/Cas9 or TALEN eliminate PD-1 expression by editing the PDCD1 gene locus.
Since some tumors (such as retroperitoneal tumors including sarcoma) may not be as amenable to regional administration and because local administration may still not be sufficient to penetrate the dense solid tumor stromal network or effectively perfuse the distorted and immature blood vessels that constitute many solid tumors, other strategies may be necessary to further augment T-cell infiltration into primary and metastatic tumor sites. Promoting a cellular infiltrate can be achieved by employing traditional cancer treatments such as radiotherapy and chemotherapy to induce a local inflammatory state that increases cellular presence within the tumor bed (Ariyan et al., 2018; DeSelm et al., 2018). Achieving the correct chemokine receptor and ligand pairing to effectively target homing to tumor sites is another strategy to achieve infiltration, either by the use of virus to deliver chemokine ligand expression to tumor (Moon et al., 2018) or, conversely, by expressing a chemokine receptor on CAR T cells that is complementary to the most abundant chemokine ligand within the tumor environment (Moon et al., 2011). Other groups have employed strategies targeting the stromal matrix by administering stromal degradation enzymes such as heparanase (Caruana et al., 2015) to enhance infiltration or employing FAP (fibroblast activating protein)–directed CARs to directly target the dense stromal matrix that serves as a barrier to infiltration in many solid tumors (Schuberth et al., 2013; Wang et al., 2014). Furthermore, using T-cell receptors with the highest affinity for target antigen can achieve infiltration and efficacy in tumors known for their high-density stromal matrix (pancreatic ductal adenocarcinoma) (Stromnes et al., 2015), and anti-angiogenic therapy targeting VEGF, D276, or endothelin B receptor may normalize tumor vasculature and promote T cell delivery (Daenen et al., 2009; Yang et al., 2018). Furthermore, we must recognize that metastatic sites are often different from the primary tumor with respect to ease of infiltration, sensitivity to therapy, and degree of immunosuppression. Attention to efficacy at both primary tumor and metastases is therefore needed to optimize long-term oncologic outcomes.
Limited Functional Persistence of CAR T Cells in the Tumor Microenvironment
Even upon achieving an adequate T-cell infiltrate, we and many others have demonstrated the relevance of an additional profound hurdle to effective therapy—overcoming the immunosuppressive tumor environment. An understanding of this profoundly T-cell–inhibiting environment justifies the rational use of CAR T-cell and CPB combination therapy. Immunologically, large tumor burdens are similar to chronic viral infections in the large antigenic burden stressing T-cell function, requiring persistent function upon repeated antigen stimulation in an immunosuppressive environment to achieve successful elimination. Chronic viral infection models were the first to characterize endogenous T-cell exhaustion resulting from repeated antigen stimulation, which is characterized by loss of cytokine secretion and lytic function with simultaneous expression of inhibitory receptors and exhaustion-related transcription factors. Chronic antigen stimulation by tumor can similarly lead to CAR T-cell exhaustion, which we have characterized as an environment replete with CAR T cells that are nonetheless hypofunctional (Cherkassky et al., 2016). Therefore, T-cell persistence alone may not reflect the more relevant need of functional persistence for solid tumor regression and costimulation alone still does not prevent long-term exhaustion resulting from the expression of inhibitory ligands.
Evidence for Checkpoint-Mediated CAR T-Cell Inhibition
Overexpression of PD-L1 and PD-L2 on tumor cells directly inhibits CAR T-cell effector functions, especially after chronic antigen stimulation (Cherkassky et al., 2016). This effect is seen regardless of the CAR costimulation domain. Both 4–1BB and CD28 CAR T cells appear to be sensitive to PD-L1–mediated inhibition, even though 4–1BB CAR T cells express lower levels of PD-1, compared with CD28 (Cherkassky et al., 2016) and other markers of exhaustion, such as TIM-3 and LAG-3 (Long et al., 2015), following chronic antigen stimulation.
Combination Therapy
T cells naturally undergo activation-induced upregulation of coinhibitory pathways, which can limit antitumor immune responses when the T cells bind their complementary ligands. Tumor cells upregulate the expression of coinhibitory ligands, such as PD-L1, following exposure to T cell–secreted Th1 cytokines. Expression of these ligands can then limit the antitumor response in favor of tumor survival. Strategies that combine adoptive T-cell therapy with CPB hold promise to achieve functional persistence of CAR T cells (Figure 1).
Methods of Combination: Cell Extrinsic vs. Cell Intrinsic
Cell-Extrinsic Strategies (Figure 2B).
In a breast cancer model, the use of HER2-targeted CAR T cells upregulated PD-1 in vitro after incubation with target cells. PD-1 blockade enhanced in vitro T-cell proliferation, IFN-γ production, granzyme B expression, and in vivo activity of CAR T cells (John et al., 2013). A study of an anti-GD2 CAR showed that the use of pembrolizumab improved T-cell function and survival following repeated antigen stimulation against PD-L1–positive tumor cells (Gargett et al., 2016). Our group demonstrated that PD-1 CPB antibodies can rescue the effector function of exhausted CAR T cells (Cherkassky et al., 2016). However, tumor relapse was observed following cessation of treatment, which suggests that the efficacy of the PD-1 antibody relies on repeated antibody administration.
PD-L1 CPB was also effective in enhancing CAR T-cell therapy by blocking PD-L1 expression on MDSCs. The combined use of CAR T cells and PD-L1 antibody with MDSC-depleting antibodies (anti-Gr1 or anti–GM-CSF) further enhanced the efficacy of the therapy (Burga et al., 2015) and highlights the immunosuppressive role that PD-L1–positive MDSCs can play in CAR T-cell therapy. To avoid the possible toxicities associated with systemic administration of CPB, Tanoue et al. combined anti-HER2 CAR T-cell therapy with an oncolytic adenovirus expressing a PD-L1–blocking mini-antibody that allows for local production of PDL1 antibodies within the tumor microenvironment (Tanoue et al., 2017). In a subcutaneous mouse model of prostate cancer, this PD-L1 oncolytic strategy was more efficient at reinvigorating CAR T cells than systemic injection of PD-L1 antibodies.
Two phase I clinical trials have investigated the efficacy of combining CAR T cells and PD-1/PD-L1 CPB (Chong et al., 2017; Heczey et al., 2017). In the study by Chong et al., 1 patient with refractory diffuse large B-cell lymphoma received cyclophosphamide lymphodepleting chemotherapy followed by autologous CD19 CAR T cells. Following treatment, the tumor continued to progress and strongly expressed PD-L1. Beginning on day 26 after T-cell infusion, pembrolizumab was administered every 3 weeks (2 mg/kg), after which the percentage of T cells expressing PD-1/EOMES decreased, expansion of CART19 cells and TCRβ T-cell clones was observed, and regression of multiple lesions occurred by day 45 (Chong et al., 2017).
The phase I study reported by Heczey et al. enrolled 11 patients with relapsed or refractory neuroblastoma. The patients were treated with CAR T cells alone (n=4), CAR T cells plus cyclophosphamide and fludarabine (Cy/Flu; n=4), or CAR T cells, Cy/Flu, and pembrolizumab n=3). All infusions were well tolerated, and no dose-limiting toxicities were observed. Cy/Flu lymphodepletion significantly increased levels of IL-15 and CAR T-cell expansion, but the addition of pembrolizumab did not further enhance this accumulation or persistence. Although a complete response was observed in 1 patient in the pembrolizumab-treated cohort, no conclusions can be drawn from this small group of patients (Heczey et al., 2017).
Overall, the clinical trials exploring CAR T-cell therapy to date have yielded encouraging results, with several studies ongoing (Table 1). The use of PD-1/PD-L1 described above is FDA approved, which can simplify logistics; however, several factors can limit this approach, including the short half-life of PD-1 antibodies, requiring multiple administrations; variable tumor penetration and pharmacokinetics inherent to antibodies; and the risk of systemic nontargeted therapy and its associated toxicity. T cell–intrinsic strategies centered on genetically modifying CAR T cells to induce sustained resistance to PD-1 signaling can address some of these limitations.
Table 1.
Clinical Trials Exploring Combination Therapy with CAR T Cells and Checkpoint Blockade
| Trial | Launch | Phase | Center(s) | CPB Agent | CAR Target/Design | Cancer Diagnosis |
|---|---|---|---|---|---|---|
| NCT00586391 | 2009 | I | Baylor | Ipilimumab | CD19/CD19CAR-28-zeta T cells | B-cell lymphoma, chronic lymphocytic leukemia, acute lymphocytic leukemia |
| NCT01822652 | 2013 | I | Baylor | Pembrolizumab | GD2/iC9-GD2-CD28–OX40 (iC9-GD2) T cells | Neuroblastoma |
| NCT02650999 | 2016 | I/II | University of Pennsylvania | Pembrolizumab | Anti-CD19 CARs | CD19+ diffuse large B-cell lymphoma, follicular lymphoma, mantle cell lymphoma |
| NCT02706405 | 2016 | I | Fred Hutchinson | Durvalumab | Autologous anti-CD 19CAR-4-1BB-CD3ζ-EGFRt-expressing CD4+/CD8+ central memory T-lymphocytes JCAR014 | Diffuse large B-cell lymphoma |
| NCT02926833 | 2016 | I/II | City of Hope, Stanford, Moffitt, Dana Farber, MD Anderson | Atezolizumab | CD19/KTE-C19 | Diffuse large B-cell lymphoma |
| NCT03310619 | 2017 | I/II | City of Hope, Northwestern University, Massachusetts General, University of Nebraska, University of Pennsylvania, MD Anderson | Durvalumab | JCAR017 | Lymphoma, non-Hodgkin lymphoma, diffuse large B-cell lymphoma, follicular lymphoma |
| NCT03726515 | 2018 | I | University of Pennsylvania | Pembrolizumab | CART-EGFRvIII T cells | Glioblastoma |
Cell-Intrinsic Strategies (Figure 2C).
A cell-intrinsic model could eliminate the need for separate dosing (Table 2). It would also equip the patient with a self-replicating and constitutive biological treatment that could continuously provide CPB. One cell-intrinsic strategy that has been investigated is the use of a bicistronic lentiviral vector to induce expression of an anticarbonic anhydrase IX CAR and anti–PD-L1 scFv antibodies in primary human T cells. This promoted accumulation of anti–PD-L1 antibodies in the tumor microenvironment and blocked PD-1 signaling. The CAR T cells had decreased expression of inhibitory receptors, such as PD-1, TIM-3, and LAG-3, and enhanced effector activity in an orthotopic model of human renal cell carcinoma (Suarez et al., 2016). Similarly, CART19 cells engineered to locally secrete an anti–PD-1–blocking scFv were more efficacious in vivo (Li et al., 2017; Rafiq et al., 2018b).
Table 2.
Comparison of Characteristics of Therapy with PD-1 DNR CAR T Cells vs. Checkpoint Blockade Agents with CAR T cells
| Characteristic | PD-1 DNR CAR T cells | CPB Agents with CAR T cells |
|---|---|---|
| Mechanism | Cell-intrinsic | Cell-extrinsic |
| Potential doses | Single | Multiple doses of CPB |
| Targeted therapy | Yes | No |
| Potential toxicity | Localized to tumor | Systemic |
| Limits to tumor penetration | Unlikely | Limits of antibody penetration |
| Antigen responsiveness | Multiple | Multiple |
Our group developed a dominant negative receptor of PD-1 that consists of the extracellular domain of PD-1 without the intracellular signaling domain. When expressed in combination with an anti-mesothelin CAR that harbors CD28 and CD3ζ domains, PD-1 dominant negative receptor competes for PD-L1/PD-L2 with the endogenous PD-1 receptor and significantly enhances CAR T-cell antitumoral activity in mesothelioma and lung cancer mouse models (Cherkassky et al., 2016). In the “switch receptor” approach, the extracellular domain of PD-1 is fused with the transmembrane and intracellular domains of CD28 to create an artificial receptor that, upon PD-L1 recognition, provides costimulation. When co-expressed with an anti-mesothelin CAR T cell, PD-1:CD28 chimera enhanced CAR T-cell effector functions and persistence in vivo, thus leading to durable efficacy and tumor eradication of established tumors (Liu et al., 2016). More recently, we compared intrinsic and extrinsic strategies and found similar efficacy between the dominant negative receptor and multiple doses of anti–PD-1 antibody; these findings led to the development of our upcoming clinical trial investigating a mesothelin-targeting CAR featuring a PD-1 dominant negative receptor (Dozier et al., 2018).
Genome-editing approaches are also being used to induce resistance to PD-1 signaling, taking advantage of the CRISPR/Cas9 (Rupp et al., 2017) and TALEN (Gautron et al., 2017) systems. PD-1–knockout CAR T cells demonstrate improved cytotoxic activity in vitro and tumor clearance in vivo. A similar approach combined multiplex genome engineering of three loci (TCR, β−2 microglobulin, and PD-1) and CAR expression to generate universal CAR T cells resistant to PD-1 signaling and with enhanced effector function in vivo (Ren et al., 2017). There are some limitations specific to the gene-editing approach: In a mouse model of chronic viral infection, complete absence of PD-1 signaling may counterintuitively produce terminal differentiated exhausted T cells (Odorizzi et al., 2015), and so it may be best for T cells to upregulate some amount of PD-1 that is then blocked by cell-intrinsic or cell-extrinsic CPB. Also, off-target cleavage must be identified and limited (Kleinstiver et al., 2016; Slaymaker et al., 2016; Tsai et al., 2015), and added PD-1 gene editing, in addition to CAR transgene, may produce genotoxicity, with negative effects on T-cell proliferation and effector functions.
CAR T cells have been modified to secrete PD-1–blocking scFvs, which may avoid the need to administer two treatments separately but would achieve similar levels of each in vivo (Chong et al., 2017; Rafiq et al., 2018a). The safety and effectiveness of PD-1 cell-intrinsic therapy using CAR T cells that secrete PD-1 antibodies are being investigated for the treatment of EGFR-positive advanced solid malignancies, including lung cancer, gastric cancer, and liver cancer (NCT02862028). Similarly, PD-1–knockout genome-editing strategies to knock out the PD-1 receptor in adoptively transferred T cells are also being investigated (NCT03081715, NCT02867332, NCT02867345, NCT02793856, NCT03044743).
The cell-intrinsic approach is attractive for its simplicity in drug administration: the repeated administrations required for persistence of CPB effect when the antibody/cell-extrinsic strategy is used can be replaced by a single administration of CPB-enabled T cells bolstered by the long-term transgene expression achieved by viral vector integration and reinforced by the long-lasting persistence of T-cell memory. However, when considering the limitations, it is unclear which might provide the most efficacious and least toxic of the two approaches. The cell-intrinsic approach may limit recruitment of the endogenous T-cell response, which may provide the polyclonal antitumor immunity most resistant to antigen escape and optimal for what are often highly heterogeneous tumors. Whereas the cell-intrinsic strategy may be better in limiting autoimmunity to off-target epitopes, in the end, it may be that antibody-mediated CPB should be used in tandem with the cell-intrinsic strategy to both prevent CAR T-cell exhaustion and reactivate what is an initially quiescent endogenous immune response but which is then recruited via epitope spreading.
Current Trials of Combination Therapy
At present, several trials are exploring combination therapy with CPB and CAR T cells. These trials vary in terms of agents used, dosing, and schedules. Models to explore these elements in the preclinical setting have yet to be developed.
Our group investigated the safety of anti–PD-1 CPB (off trial) following treatment with mesothelin CAR T cells (on trial) in patients with malignant pleural mesothelioma (Adusumilli et al., Cancer Res 2019; 79(13 Suppl): abstract CT036). In a subcohort of 11 patients with mesothelioma who received cyclophosphamide preconditioning followed by a single dose of CAR T cells and subsequent anti–PD-1 agent (at least 3 doses) with a minimum of 3 months of follow-up, 8 patients showed response (response rate, 72%), including complete metabolic responses in 2 patients. The observation of a complete metabolic response in tumors of which only 40% expressed the targeted antigen raises the intriguing possibility of combination therapy–induced neoantigen response and epitope spreading (Figure 1)—possibilities that have not yet been investigated.
Future Directions of Combination Therapy
Combination immunotherapy with CAR T cells and CPB is poised to be the next frontier in immunotherapy as it addresses the two elements necessary for a robust immune attack: the presence of immune cells and functional persistence. CAR T cells can provide an infiltrate, and PD-1/PD-L1 blockade can ensure sustained persistence and function. In this section, we recognize that even this combination may not be sufficient to attain the T-cell infiltration and effector function necessary to optimally combat solid tumors. In turn, we describe some additional barriers to effective combination therapy. Also, we address the innate problem that arises whenever a targeted therapy’s efficacy is enhanced—that of toxicity to normal tissues expressing the same target as tumor, or on-target off-tumor toxicity.
Enhancing Functional Persistence and Other Obstacles.
Other inhibitory receptors, such as TIM-3 and LAG-3, are upregulated in T cells after chronic antigen stimulation (Schildberg et al., 2016) and PD-1 blockade (Huang et al., 2017; Koyama et al., 2016), which suggests that additional combinations of blocking antibodies may optimize function. In order to offset the immunosuppression imposed by multiple inhibitory signaling pathways (Huang et al., 2017), cell-extrinsic (infusions of multiple checkpoint targeting antibodies) or cell-intrinsic (delivery of multicistronic elements targeting multiple pathways) strategies may be necessary to offset redundant inhibitory signaling. Other newly described inhibitory receptors, such as B7-H3, VISTA, and B7S1, provide alternative pathways to modulate T-cell responses (Ni and Dong, 2017), and recruiting other elements of the immune system—both innate and adaptive—has great potential to induce a more robust antitumor response (Moynihan et al., 2016).
Still, other barriers to effective therapy exist, such as antigen heterogeneity—an obstacle that must be addressed to successfully target solid tumors often characterized by heterogenous antigen expression. Overcoming this obstacle begins with the selection of a target antigen that is as homogenously and highly expressed as possible on tumor tissue and can be further countered by infusing CAR T cells with the ability to target multiple antigens (Majzner and Mackall, 2018) or by adding radiation therapy that results in TRAIL-mediated death or even targeting of antigen negative tumour cells (DeSelm et al., 2018).
The requirement for a functionally persistent effector response, the principle underlying combination CPB and T-cell therapy, will guide the next steps in the preclinical and clinical settings. More-accurate and clinically relevant models are needed for investigation of CAR T-cell efficacy. To address the current shortcomings in the currently available models, our lab has investigated CAR T-cell persistence and function in malignant pleural effusions (Tano et al., 2018).
Limiting On-Target Off-Tumor Toxicity.
Most CARs targeting solid tumors are aimed at antigens shared by normal tissues, carrying the risk of on-target off-tumor toxicity (Lamers et al., 2006; Morgan et al., 2010). And as with all functional enhancements to a targeted therapy, combination CPB enhances efficacy but risks increasing toxicity. In addition to the methods already in clinical practice such as use of steroids to reduce the number of CAR T cells, incorporating suicide genes (iCaspase-9, epidermal growth factor receptor mutation) can be used to mediate T-cell elimination upon detection of side effects but may squander T-cell efficacy. An optimal solid-tumor antigen target is one whose expression is restricted to expendable cells or, better yet, tumor cells only, such as the mutated form of EGFR expressed on glioblastoma multiforme tumors (Johnson et al., 2015; Sampson et al., 2014), or neoantigen peptide: MHC complexes, which have yet to be developed but theoretically could selectively target unique tumor mutations. Other carefully selected antigens are those occurring at high densities on tumor but very low levels in normal tissues, an example being our target, mesothelin—chosen for its overexpression in mesothelioma, triple-negative breast cancer, and pancreatic cancer, and with only low levels of expression by the pleural and pericardial lining. With this selection strategy, we have demonstrated anti-tumor responses but have not as of yet experienced any on-target off-tumor toxicities in our ongoing phase I clinical trial. As scientists and clinicians are faced with a limited availability of efficiently targetable antigens, other strategies can be used to improve the specificity and safety of CAR T cells, even while targeting tumor-associated antigens that are shared by normal cells, such as a trans-signaling approach that splits CD3ζ signaling from costimulatory signaling (Kloss et al., 2013; Wilkie et al., 2012), engineering a requirement for dual antigen activation (Roybal et al., 2016), or activating inhibitory signaling upon binding of a normal tissue antigen (Fedorov et al., 2013).
As we take the next steps in combining CAR T-cell therapy and CPB agents, the current clinical trials will provide the best insights into the future of this approach for the treatment of solid tumors and will shine light on strategies to develop new methods of studying combination therapy and providing these groundbreaking treatments to our patients.
Acknowledgments
The authors’ laboratory work is supported by grants from the National Institutes of Health (P30 CA008748, R01 CA236615-01 and R01 CA235667), the U.S. Department of Defense (BC132124, LC160212, CA170630, and CA180889), the Emerson Collective Cancer Research Fund, Mr. William H. Goodwin and Alice Goodwin, the Commonwealth Foundation for Cancer Research, the Experimental Therapeutics Center of Memorial Sloan Kettering Cancer Center, and the Baker Street Foundation.
Declarations of Interest
P.S.A. has received research funding from ATARA Biotherapeutics and OSE Immunotherapies and has patents on mesothelin chimeric antigen receptor and PD-1 dominant negative receptor. All other authors have no interests to declare.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Adusumilli PS, Cherkassky L, Villena-Vargas J, Colovos C, Servais E, Plotkin J, Jones DR, and Sadelain M. (2014). Regional delivery of mesothelin-targeted CAR T cell therapy generates potent and long-lasting CD4-dependent tumor immunity. Sci Transl Med 6, 261ra151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexandrov LB, Nik-Zainal S, Wedge DC, Aparicio SA, Behjati S, Biankin AV, Bignell GR, Bolli N, Borg A, Borresen-Dale AL. et al. (2013). Signatures of mutational processes in human cancer. Nature 500, 415–421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ariyan CE, Brady MS, Siegelbaum RH, Hu J, Bello DM, Rand J, Fisher C, Lefkowitz RA, Panageas KS, Pulitzer M. et al. (2018). Robust antitumor responses result from local chemotherapy and CTLA-4 blockade. Cancer Immunol Res 6, 189–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azimi F, Scolyer RA, Rumcheva P, Moncrieff M, Murali R, McCarthy SW, Saw RP, and Thompson JF (2012). Tumor-infiltrating lymphocyte grade is an independent predictor of sentinel lymph node status and survival in patients with cutaneous melanoma. J Clin Oncol 30, 2678–2683. [DOI] [PubMed] [Google Scholar]
- Balachandran VP, Luksza M, Zhao JN, Makarov V, Moral JA, Remark R, Herbst B, Askan G, Bhanot U, Senbabaoglu Y. et al. (2017). Identification of unique neoantigen qualities in long-term survivors of pancreatic cancer. Nature 551, 512–516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barber DL, Wherry EJ, Masopust D, Zhu B, Allison JP, Sharpe AH, Freeman GJ, and Ahmed R. (2006). Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 439, 682–687. [DOI] [PubMed] [Google Scholar]
- Bauml J, Seiwert TY, Pfister DG, Worden F, Liu SV, Gilbert J, Saba NF, Weiss J, Wirth L, Sukari A. et al. (2017). Pembrolizumab for platinum- and cetuximab-refractory dead and neck cancer: Results from a single-arm, phase II study. J Clin Oncol 35, 1542–1549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blank CU, Haanen JB, Ribas A, and Schumacher TN (2016). The “cancer immunogram”. Science 352, 658–660. [DOI] [PubMed] [Google Scholar]
- Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, Ready NE, Chow LQ, Vokes EE, Felip E, Holgado E. et al. (2015). Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med 373, 1627–1639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brentjens RJ, Rivière I, Park JH, Davila ML, Wang X, Stefanski J, Taylor C, Yeh R, Bartido S, Borquez-Ojeda O. et al. (2011). Safety and persistence of adoptively transferred autologous CD19-targeted T cells in patients with relapsed or chemotherapy refractory B-cell leukemias. Blood 118, 4817–4828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown CE, Starr R, Aguilar B, Shami AF, Martinez C, D’Apuzzo M, Barish ME, Forman SJ, and Jensen MC (2012). Stem-like tumor-initiating cells isolated from IL13Ralpha2 expressing gliomas are targeted and killed by IL13-zetakine-redirected T cells. Clin Cancer Res 18, 2199–2209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burga RA, Thorn M, Point GR, Guha P, Nguyen CT, Licata LA, DeMatteo RP, Ayala A, Joseph Espat N, Junghans RP, and Katz SC (2015). Liver myeloid-derived suppressor cells expand in response to liver metastases in mice and inhibit the anti-tumor efficacy of anti-CEA CAR-T. Cancer Immunol Immunother 64, 817–829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caruana I, Savoldo B, Hoyos V, Weber G, Liu H, Kim ES, Ittmann MM, Marchetti D, and Dotti G. (2015). Heparanase promotes tumor infiltration and antitumor activity of CAR-redirected T lymphocytes. Nat Med 21, 524–529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casey SC, Tong L, Li Y, Do R, Walz S, Fitzgerald KN, Gouw AM, Baylot V, Gütgemann I, Eilers M, and Felsher DW (2016). MYC regulates the antitumor immune response through CD47 and PD-L1. Science 352, 227–231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen R, Zinzani PL, Fanale MA, Armand P, Johnson NA, Brice P, Radford J, Ribrag V, Molin D, Vassilakopoulos TP. et al. (2017). Phase II study of the efficacy and safety of pembrolizumab for relapsed/refractory classic Hodgkin lymphoma. J Clin Oncol 35, 2125–2132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cherkassky L, Morello A, Villena-Vargas J, Feng Y, Dimitrov DS, Jones DR, Sadelain M, and Adusumilli PS (2016). Human CAR T cells with cell-intrinsic PD-1 checkpoint blockade resist tumor-mediated inhibition. J Clin Invest 126, 3130–3144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chong EA, Melenhorst JJ, Lacey SF, Ambrose DE, Gonzalez V, Levine BL, June CH, and Schuster SJ (2017). PD-1 blockade modulates chimeric antigen receptor (CAR)-modified T cells: refueling the CAR. Blood 129, 1039–1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curiel TJ, Wei S, Dong H, Alvarez X, Cheng P, Mottram P, Krzysiek R, Knutson KL, Daniel B, Zimmermann MC. et al. (2003). Blockade of B7-H1 improves myeloid dendritic cell-mediated antitumor immunity. Nat Med 9, 562–567. [DOI] [PubMed] [Google Scholar]
- Daenen LG, Shaked Y, Man S, Xu P, Voest EE, Hoffman RM, Chaplin DJ, and Kerbel RS (2009). Low-dose metronomic cyclophosphamide combined with vascular disrupting therapy induces potent antitumor activity in preclinical human tumor xenograft models. Mol Cancer Ther 8, 2872–2881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danilova L, Wang H, Sunshine J, Kaunitz GJ, Cottrell TR, Xu H, Esandrio J, Anders RA, Cope L, Pardoll DM. et al. (2016). Association of PD-1/PD-L axis expression with cytolytic activity, mutational load, and prognosis in melanoma and other solid tumors. Proc Natl Acad Sci U S A 113, E7769–E7777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davila ML, Riviere I, Wang X, Bartido S, Park J, Curran K, Chung SS, Stefanski J, Borquez-Ojeda O, Olszewska M. et al. (2014). Efficacy and toxicity management of 19–28z CAR T cell therapy in B cell acute lymphoblastic leukemia. Sci Transl Med 6, 224ra225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeSelm C, Palomba ML, Yahalom J, Hamieh M, Eyquem J, Rajasekhar VK, and Sadelain M. (2018). Low-dose radiation conditioning enables CAR T Cells to mitigate antigen escape. Mol Ther 26, 2542–2552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dozier J, Chen N, Saini J, Chintala N, and Adusumilli PS (2018). Comparative efficacy of T-Cell intrinsic versus extrinsic PD-1 blockade to overcome PD-L1+ tumor-cediated exhaustion. J Thorac Oncol 13, S392–S392. [Google Scholar]
- Dunn GP, Bruce AT, Ikeda H, Old LJ, and Schreiber RD (2002). Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol 3, 991–998. [DOI] [PubMed] [Google Scholar]
- El-Khoueiry AB, Sangro B, Yau T, Crocenzi TS, Kudo M, Hsu C, Kim TY, Choo SP, Trojan J, Welling THR. et al. (2017). Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase ½ dose escalation and expansion trial. Lancet 389, 2492–2502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eroglu Z, Zaretsky JM, Hu-Lieskovan S, Kim DW, Algazi A, Johnson DB, Liniker E, Ben K, Munhoz R, Rapisuwon S. et al. (2018). High response rate to PD-1 blockade in desmoplastic melanomas. Nature 553, 347–350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fedorov VD, Themeli M, and Sadelain M. (2013). PD-1- and CTLA-4-based inhibitory chimeric antigen receptors (iCARs) divert off-target immunotherapy responses. Sci Transl Med 5, 215ra172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gajewski TF, Louahed J, and Brichard VG (2010). Gene signature in melanoma associated with clinical activity: a potential clue to unlock cancer immunotherapy. Cancer J 16, 399–403. [DOI] [PubMed] [Google Scholar]
- Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pages C, Tosolini M, Camus M, Berger A, Wind P. et al. (2006). Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313, 1960–1964. [DOI] [PubMed] [Google Scholar]
- Gardner RA, Finney O, Annesley C, Brakke H, Summers C, Leger K, Bleakley M, Brown C, Mgebroff S, Spratt K. et al. (2017). Intent to treat leukemia remission by CD19CAR T cells of defined formulation and dose in children and young adults. Blood 129, 3322–3331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gargett T, Yu W, Dotti G, Yvon ES, Christo SN, Hayball JD, Lewis ID, Brenner MK, and Brown MP (2016). GD2-specific CAR T cells undergo potent activation and deletion following antigen encounter but can be protected from activation-induced cell eeath by PD-1 blockade. Mol Ther 24, 1135–1149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garon EB, Rizvi NA, Hui R, Leighl N, Balmanoukian AS, Eder JP, Patnaik A, Aggarwal C, Gubens M, Horn L. et al. (2015). Pembrolizumab for the treatment of nonsmall-cell lung cancer. N Engl J Med 372, 2018–2028. [DOI] [PubMed] [Google Scholar]
- Gautron A-S, Juillerat A, Guyot V, Filhol J-M, Dessez E, Duclert A, Duchateau P, and Poirot L. (2017). Fine and predictable tuning of TALEN gene editing targeting for improved T Cell adoptive immunotherapy. Molecular Therapy - Nucleic Acids 9, 312–321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geyer MB, and Brentjens RJ (2016). Review: Current clinical applications of chimeric antigen receptor (CAR) modified T cells. Cytotherapy 18, 1393–1409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heczey A, Louis CU, Savoldo B, Dakhova O, Durett A, Grilley B, Liu H, Wu MF, Mei Z, Gee A. et al. (2017). CAR T cells administered in combination with lymphodepletion and PD-1 inhibition to patients with neuroblastoma. Mol Ther 25, 2214–2224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herbst RS, Soria J-C, Kowanetz M, Fine GD, Hamid O, Gordon MS, Sosman JA, McDermott DF, Powderly JD, Gettinger SN. et al. (2014). Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 515, 563–567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodi FS, Mihm MC, Soiffer RJ, Haluska FG, Butler M, Seiden MV, Davis T, Henry-Spires R, MacRae S, Willman A. et al. (2003). Biologic activity of cytotoxic T lymphocyte-associated antigen 4 antibody blockade in previously vaccinated metastatic melanoma and ovarian carcinoma patients. Proc Natl Acad Sci U S A 100, 4712–4717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, Gonzalez R, Robert C, Schadendorf D, Hassel JC. et al. (2010). Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363, 711–723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu W, Zi Z, Jin Y, Li G, Shao K, Cai Q, Ma X, and Wei F. (2019). CRISPR/Cas9-mediated PD-1 disruption enhances human mesothelin-targeted CAR T cell effector functions. Cancer immunology, immunotherapy : Cancer Immunol Immunother 68, 365–377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang AC, Postow MA, Orlowski RJ, Mick R, Bengsch B, Manne S, Xu W, Harmon S, Giles JR, Wenz B. et al. (2017). T-cell invigoration to tumour burden ratio associated with anti-PD-1 response. Nature 545, 60–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwai Y, Ishida M, Tanaka Y, Okazaki T, Honjo T, and Minato N. (2002). Involvement of PD-L1 on tumor cells in the escape from host immune system and tumor immunotherapy by PDL1 blockade. Proc Natl Acad Sci U S A 99, 12293–12297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- John LB, Devaud C, Duong CPM, Yong CS, Beavis PA, Haynes NM, Chow MT, Smyth MJ, Kershaw MH, and Darcy PK (2013). Anti-PD-1 antibody therapy potently enhances the eradication of established tumors by gene-modified T cells. Clin Cancer Res 19, 5636–5646. [DOI] [PubMed] [Google Scholar]
- Johnson LA, Scholler J, Ohkuri T, Kosaka A, Patel PR, McGettigan SE, Nace AK, Dentchev T, Thekkat P, Loew A. et al. (2015). Rational development and characterization of humanized anti-EGFR variant III chimeric antigen receptor T cells for glioblastoma. Sci Transl Med 7, 275ra222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joyce JA, and Fearon DT (2015). T cell exclusion, immune privilege, and the tumor microenvironment. Science 348, 74–80. [DOI] [PubMed] [Google Scholar]
- June CH, O’Connor RS, Kawalekar OU, Ghassemi S, and Milone MC (2018). CAR T cell immunotherapy for human cancer. Science 359, 1361–1365. [DOI] [PubMed] [Google Scholar]
- Kataoka K, Shiraishi Y, Takeda Y, Sakata S, Matsumoto M, Nagano S, Maeda T, Nagata Y, Kitanaka A, Mizuno S. et al. (2016). Aberrant PD-L1 expression through 3’-UTR disruption in multiple cancers. Nature 534, 402–406. [DOI] [PubMed] [Google Scholar]
- Kleinstiver BP, Pattanayak V, Prew MS, Tsai SQ, Nguyen NT, and Joung JK (2016). High-fidelity CRISPR-Cas9 nucleases with no detectable genome-wide off-target effects. Nature 529, 490–495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kloss CC, Condomines M, Cartellieri M, Bachmann M, and Sadelain M. (2013). Combinatorial antigen recognition with balanced signaling promotes selective tumor eradication by engineered T cells. Nat Biotechnol 31, 71–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kochenderfer JN, Dudley ME, Feldman SA, Wilson WH, Spaner DE, Maric I, Stetler-Stevenson M, Phan GQ, Hughes MS, Sherry RM. et al. (2012). B-cell depletion and remissions of malignancy along with cytokine-associated toxicity in a clinical trial of antiCD19 chimeric-antigen-receptor-transduced T cells. Blood 119, 2709–2720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kochenderfer JN, Wilson WH, Janik JE, Dudley ME, Stetler-Stevenson M, Feldman SA, Maric I, Raffeld M, Nathan D-AN, Lanier BJ. et al. (2010). Eradication of B-lineage cells and regression of lymphoma in a patient treated with autologous T cells genetically engineered to recognize CD19. Blood 116, 4099–4102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koneru M, Purdon TJ, Spriggs D, Koneru S, and Brentjens RJ (2015). IL-12 secreting tumor-targeted chimeric antigen receptor T cells eradicate ovarian tumors in vivo. Oncoimmunology 4, e994446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koyama S, Akbay EA, Li YY, Herter-Sprie GS, Buczkowski KA, Richards WG, Gandhi L, Redig AJ, Rodig SJ, Asahina H. et al. (2016). Adaptive resistance to therapeutic PD-1 blockade is associated with upregulation of alternative immune checkpoints. Nat Commun 7, 10501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamers CH, Sleijfer S, Vulto AG, Kruit WH, Kliffen M, Debets R, Gratama JW, Stoter G, and Oosterwijk E. (2006). Treatment of metastatic renal cell carcinoma with autologous T-lymphocytes genetically retargeted against carbonic anhydrase IX: first clinical experience. J Clin Oncol 24, e20–22. [DOI] [PubMed] [Google Scholar]
- Le DT, Durham JN, Smith KN, Wang H, Bartlett BR, Aulakh LK, Lu S, Kemberling H, Wilt C, Luber BS. et al. (2017). Mismatch-repair deficiency predicts response of solid tumors to PD-1 blockade. Science 357, 409–413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leach DR, Krummel MF, and Allison JP (1996). Enhancement of antitumor immunity by CTLA-4 blockade. Science 271, 1734–1736. [DOI] [PubMed] [Google Scholar]
- Lee DW, Kochenderfer JN, Stetler-Stevenson M, Cui YK, Delbrook C, Feldman SA, Fry TJ, Orentas R, Sabatino M, Shah NN. et al. (2015). T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet 385, 517–528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li S, Siriwon N, Zhang X, Yang S, Jin T, He F, Kim YJ, Mac J, Lu Z, Wang S. et al. (2017). Enhanced cancer immunotherapy by chimeric antigen receptor-modified T cells engineered to secrete checkpoint inhibitors. Clin Cancer Res 23, 6982–6992. [DOI] [PubMed] [Google Scholar]
- Lipson EJ, Sharfman WH, Drake CG, Wollner I, Taube JM, Anders RA, Xu H, Yao S, Pons A, Chen L. et al. (2012). Durable cancer regression off-treatment and effective reinduction therapy with an anti-PD-1 antibody. Clin Cancer Res 19, 462–468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X, Ranganathan R, Jiang S, Fang C, Sun J, Kim S, Newick K, Lo A, June CH, Zhao Y, and Moon EK (2016). A chimeric switch-receptor targeting PD1 augments the efficacy of second-generation CAR T cells in advanced solid tumors. Cancer Res 76, 1578–1590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Long AH, Haso WM, Shern JF, Wanhainen KM, Murgai M, Ingaramo M, Smith JP, Walker AJ, Kohler ME, Venkateshwara VR. et al. (2015). 4–1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors. Nat Med 21, 581–590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Majzner RG, and Mackall CL (2018). Tumor antigen escape from CAR T-cell therapy. Cancer Discov 8, 1219–1226. [DOI] [PubMed] [Google Scholar]
- Marzec M, Zhang Q, Goradia A, Raghunath PN, Liu X, Paessler M, Wang HY, Wysocka M, Cheng M, Ruggeri BA, and Wasik MA (2008). Oncogenic kinase NPM/ALK induces through STAT3 expression of immunosuppressive protein CD274 (PD-L1, B7-H1). Proc Natl Acad Sci U S A 105, 20852–20857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maude SL, Frey N, Shaw PA, Aplenc R, Barrett DM, Bunin NJ, Chew A, Gonzalez VE, Zheng Z, Lacey SF. et al. (2014). Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med 371, 1507–1517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGranahan N, Furness AJ, Rosenthal R, Ramskov S, Lyngaa R, Saini SK, Jamal-Hanjani M, Wilson GA, Birkbak NJ, Hiley CT. et al. (2016). Clonal neoantigens elicit T cell immunoreactivity and sensitivity to immune checkpoint blockade. Science 351, 1463–1469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melero I, Rouzaut A, Motz GT, and Coukos G. (2014). T-cell and NK-cell infiltration into solid tumors: a key limiting factor for efficacious cancer immunotherapy. Cancer Discov 4, 522526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moon EK, Carpenito C, Sun J, Wang LC, Kapoor V, Predina J, Powell DJ Jr., Riley JL, June CH, and Albelda SM (2011). Expression of a functional CCR2 receptor enhances tumor localization and tumor eradication by retargeted human T cells expressing a mesothelin-specific chimeric antibody receptor. Clin Cancer Res 17, 4719–4730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moon EK, Wang LC, Dolfi DV, Wilson CB, Ranganathan R, Sun J, Kapoor V, Scholler J, Pure E, Milone MC. et al. (2014). Multifactorial T-cell hypofunction that is reversible can limit the efficacy of chimeric antigen receptor-transduced human T cells in solid tumors. Clin Cancer Res 20, 4262–4273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moon EK, Wang LS, Bekdache K, Lynn RC, Lo A, Thorne SH, and Albelda SM (2018). Intra-tumoral delivery of CXCL11 via a vaccinia virus, but not by modified T cells, enhances the efficacy of adoptive T cell therapy and vaccines. Oncoimmunology 7, e1395997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morello A, Sadelain M, and Adusumilli PS (2016). Mesothelin-targeted CARs: Driving T cells to solid tumors. Cancer Discov 6, 133–146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan RA, Yang JC, Kitano M, Dudley ME, Laurencot CM, and Rosenberg SA (2010). Case report of a serious adverse event following the administration of T cells transduced with a chimeric antigen receptor recognizing ERBB2. Mol Ther 18, 843–851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moynihan KD, Opel CF, Szeto GL, Tzeng A, Zhu EF, Engreitz JM, Williams RT, Rakhra K, Zhang MH, Rothschilds AM. et al. (2016). Eradication of large established tumors in mice by combination immunotherapy that engages innate and adaptive immune responses. Nat Med 22, 1402–1410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nazareth MR, Broderick L, Simpson-Abelson MR, Kelleher RJ Jr., Yokota SJ, and Bankert RB (2007). Characterization of human lung tumor-associated fibroblasts and their ability to modulate the activation of tumor-associated T cells. J Immunol 178, 5552–5562. [DOI] [PubMed] [Google Scholar]
- Nghiem PT, Bhatia S, Lipson EJ, Kudchadkar RR, Miller NJ, Annamalai L, Berry S, Chartash EK, Daud A, Fling SP. et al. (2016). PD-1 blockade with pembrolizumab in advanced Merkel-cell carcinoma. N Engl J Med 374, 2542–2552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ni L, and Dong C. (2017). New checkpoints in cancer immunotherapy. Immunol Rev 276, 5265. [DOI] [PubMed] [Google Scholar]
- Obeid JM, Erdag G, Smolkin ME, Deacon DH, Patterson JW, Chen L, Bullock TN, and Slingluff CL (2016). PD-L1, PD-L2 and PD-1 expression in metastatic melanoma: Correlation with tumor-infiltrating immune cells and clinical outcome. Oncoimmunology 5, e1235107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Odorizzi PM, Pauken KE, Paley MA, Sharpe A, and Wherry EJ (2015). Genetic absence of PD-1 promotes accumulation of terminally differentiated exhausted CD8+ T cells. J Exp Med 212, 1125–1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardoll DM (2012). The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12, 252–264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park JH, Riviere I, Gonen M, Wang X, Senechal B, Curran KJ, Sauter C, Wang Y, Santomasso B, Mead E. et al. (2018). Long-term follow-up of CD19 CAR therapy in acute lymphoblastic leukemia. N Engl J Med 378, 449–459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parsa AT, Waldron JS, Panner A, Crane CA, Parney IF, Barry JJ, Cachola KE, Murray JC, Tihan T, Jensen MC. et al. (2007). Loss of tumor suppressor PTEN function increases B7-H1 expression and immunoresistance in glioma. Nat Med 13, 84–88. [DOI] [PubMed] [Google Scholar]
- Powles T, Eder JP, Fine GD, Braiteh FS, Loriot Y, Cruz C, Bellmunt J, Burris HA, Petrylak DP, Teng SL. et al. (2014). MPDL3280A (anti-PD-L1) treatment leads to clinical activity in metastatic bladder cancer. Nature 515, 558–562. [DOI] [PubMed] [Google Scholar]
- Rafiq S, Yeku OO, Jackson HJ, Purdon TJ, van Leeuwen DG, Drakes DJ, Song M, Miele MM, Li Z, Wang P. et al. (2018b). Targeted delivery of a PD-1-blocking scFv by CAR-T cells enhances anti-tumor efficacy in vivo. Nat Biotechnol 36, 847–856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ren J, Liu X, Fang C, Jiang S, June CH, and Zhao Y. (2017). Multiplex genome editing to generate universal CAR T cells resistant to PD1 inhibition. Clin Cancer Res 23, 2255–2266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ribas A, Camacho LH, Lopez-Berestein G, Pavlov D, Bulanhagui CA, Millham R, Comin-Anduix B, Reuben JM, Seja E, Parker CA. et al. (2005). Antitumor activity in melanoma and anti-self responses in a phase I trial with the anti-cytotoxic T lymphocyte-associated antigen 4 monoclonal antibody CP-675,206. J Clin Oncol 23, 8968–8977. [DOI] [PubMed] [Google Scholar]
- Ribas A, Hamid O, Daud A, Hodi FS, Wolchok JD, Kefford R, Joshua AM, Patnaik A, Hwu WJ, Weber JS. et al. (2016). Association of pembrolizumab with tumor response and survival among patients with advanced melanoma. JAMA 315, 1600–1609. [DOI] [PubMed] [Google Scholar]
- Ribas A, and Wolchok JD (2018). Cancer immunotherapy using checkpoint blockade. Science 359, 1350–1355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rizvi NA, Hellmann MD, Snyder A, Kvistborg P, Makarov V, Havel JJ, Lee W, Yuan J, Wong P, Ho TS. et al. (2015). Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 348, 124–128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robert C, Long GV, Brady B, Dutriaux C, Maio M, Mortier L, Hassel JC, Rutkowski P, McNeil C, Kalinka-Warzocha E. et al. (2015). Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med 372, 320–330. [DOI] [PubMed] [Google Scholar]
- Robert C, Thomas L, Bondarenko I, O’Day S, Weber J, Garbe C, Lebbe C, Baurain JF, Testori A, Grob JJ. et al. (2011). Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N Engl J Med 364, 2517–2526. [DOI] [PubMed] [Google Scholar]
- Roybal KT, Rupp LJ, Morsut L, Walker WJ, McNally KA, Park JS, and Lim WA (2016). Precision tumor recognition by T cells with combinatorial antigen-sensing circuits. Cell 164, 770–779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rupp LJ, Schumann K, Roybal KT, Gate RE, Ye CJ, Lim WA, and Marson A. (2017). CRISPR/Cas9-mediated PD-1 disruption enhances anti-tumor efficacy of human chimeric antigen receptor T cells. Sci Rep 7, 737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sadelain M, Brentjens R, and Riviere I. (2013). The basic principles of chimeric antigen receptor design. Cancer Discov 3, 388–398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sadelain M, Riviere I, and Brentjens R. (2003). Targeting tumours with genetically enhanced T lymphocytes. Nat Rev Cancer 3, 35–45. [DOI] [PubMed] [Google Scholar]
- Sampson JH, Choi BD, Sanchez-Perez L, Suryadevara CM, Snyder DJ, Flores CT, Schmittling RJ, Nair SK, Reap EA, Norberg PK. et al. (2014). EGFRvIII mCAR-modified T-cell therapy cures mice with established intracerebral glioma and generates host immunity against tumor-antigen loss. Clin Cancer Res 20, 972–984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savoldo B, Ramos CA, Liu E, Mims MP, Keating MJ, Carrum G, Kamble RT, Bollard CM, Gee AP, Mei Z. et al. (2011). CD28 costimulation improves expansion and persistence of chimeric antigen receptor-modified T cells in lymphoma patients. J Clin Invest 121, 1822–1826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schildberg FA, Klein SR, Freeman GJ, and Sharpe AH (2016). Coinhibitory pathways in the B7-CD28 ligand-receptor family. Immunity 44, 955–972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreiber RD, Old LJ, and Smyth MJ (2011). Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science 331, 1565–1570. [DOI] [PubMed] [Google Scholar]
- Schuberth PC, Hagedorn C, Jensen SM, Gulati P, van den Broek M, Mischo A, Soltermann A, Jungel A, Marroquin Belaunzaran O, Stahel R. et al. (2013). Treatment of malignant pleural mesothelioma by fibroblast activation protein-specific re-directed T cells. J Transl Med 11, 187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schuster SJ, Svoboda J, Chong EA, Nasta SD, Mato AR, Anak O, Brogdon JL, Pruteanu-Malinici I, Bhoj V, Landsburg D. et al. (2017). Chimeric antigen receptor T cells in refractory B-Cell lymphomas. N Engl J Med 377, 2545–2554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schuster SJ, Svoboda J, Nasta SD, Porter DL, Chong EA, Landsburg DJ, Mato AR, Lacey SF, Melenhorst JJ, Chew A. et al. (2015). Sustained remissions following chimeric antigen receptor modified T cells directed against CD19 (CTL019) in patients with relapsed or refractory CD19+ lymphomas. Blood 126, 183–183. [Google Scholar]
- Sharma P, Hu-Lieskovan S, Wargo JA, and Ribas A. (2017). Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell 168, 707–723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slaymaker IM, Gao L, Zetsche B, Scott DA, Yan WX, and Zhang F. (2016). Rationally engineered Cas9 nucleases with improved specificity. Science 351, 84–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spranger S, Spaapen RM, Zha Y, Williams J, Meng Y, Ha TT, and Gajewski TF (2013). Up-regulation of PD-L1, IDO, and T(regs) in the melanoma tumor microenvironment is driven by CD8+ T cells. Sci Transl Med 5, 200ra116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strome SE, Dong H, Tamura H, Voss SG, Flies DB, Tamada K, Salomao D, Cheville J, Hirano F, Lin W. et al. (2003). B7-H1 blockade augments adoptive T-cell immunotherapy for squamous cell carcinoma. Cancer Res 63, 6501–6505. [PubMed] [Google Scholar]
- Stromnes IM, Schmitt TM, Hulbert A, Brockenbrough JS, Nguyen H, Cuevas C, Dotson AM, Tan X, Hotes JL, Greenberg PD, and Hingorani SR (2015). T cells engineered against a native antigen can surmount immunologic and physical barriers to treat pancreatic ductal adenocarcinoma. Cancer Cell 28, 638–652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suarez ER, Chang DK, Sun J, Sui J, Freeman GJ, Signoretti S, Zhu Q, and Marasco WA (2016). Chimeric antigen receptor T cells secreting anti-PD-L1 antibodies more effectively regress renal cell carcinoma in a humanized mouse model. Oncotarget 7, 3434134355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki K, Kadota K, Sima CS, Nitadori J, Rusch VW, Travis WD, Sadelain M, and Adusumilli PS (2013). Clinical impact of immune microenvironment in stage I lung adenocarcinoma: tumor interleukin-12 receptor beta2 (IL-12Rbeta2), IL-7R, and stromal FoxP3/CD3 ratio are independent predictors of recurrence. J Clin Oncol 31, 490–498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang H, Liang Y, Anders RA, Taube JM, Qiu X, Mulgaonkar A, Liu X, Harrington SM, Guo J, Xin Y. et al. (2018). PD-L1 on host cells is essential for PD-L1 blockade-mediated tumor regression. J Clin Invest 128, 580–588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tano Z, Kiesgen S, Chintala N, Dozier J, Messinger J, Tan KS, and Adusumilli P. (2018). An ex-vivo patient-derived, immunocompetent (PDI) culture system to evaluate immunotherapeutic agents’ anti-tumor efficacy. J Thorac Oncol 13, S376. [Google Scholar]
- Tanoue K, Rosewell Shaw A, Watanabe N, Porter C, Rana B, Gottschalk S, Brenner M, and Suzuki M. (2017). Armed oncolytic adenovirus-expressing PD-L1 mini-body enhances antitumor effects of chimeric antigen receptor T cells in solid tumors. Cancer Res 77, 2040–2051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taube JM, Anders RA, Young GD, Xu H, Sharma R, McMiller TL, Chen S, Klein AP, Pardoll DM, Topalian SL, and Chen L. (2012). Colocalization of inflammatory response with B7-h1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Sci Transl Med 4, 127ra137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taube JM, Young GD, McMiller TL, Chen S, Salas JT, Pritchard TS, Xu H, Meeker AK, Fan J, Cheadle C. et al. (2015). Differential expression of immune-regulatory genes associated with PD-L1 display in melanoma: Implications for PD-1 pathway blockade. Clin Cancer Res 21, 3969–3976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tivol EA, Borriello F, Schweitzer AN, Lynch WP, Bluestone JA, and Sharpe AH (1995). Loss of Ctla-4 leads to massive lymphoproliferation and fatal multiorgan tissue destruction, revealing a critical negative regulatory role of Ctla-4. Immunity 3, 541–547. [DOI] [PubMed] [Google Scholar]
- Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF, Powderly JD, Carvajal RD, Sosman JA, Atkins MB. et al. (2012). Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med 366, 2443–2454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai SQ, Zheng Z, Nguyen NT, Liebers M, Topkar VV, Thapar V, Wyvekens N, Khayter C, Iafrate AJ, Le LP. et al. (2015). GUIDE-seq enables genome-wide profiling of off-target cleavage by CRISPR-Cas nucleases. Nat Biotechnol 33, 187–197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tumeh PC, Harview CL, Yearley JH, Shintaku IP, Taylor EJM, Robert L, Chmielowski B, Spasic M, Henry G, Ciobanu V. et al. (2014). PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 515, 568–571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang LC, Lo A, Scholler J, Sun J, Majumdar RS, Kapoor V, Antzis M, Cotner CE, Johnson LA, Durham AC. et al. (2014). Targeting fibroblast activation protein in tumor stroma with chimeric antigen receptor T cells can inhibit tumor growth and augment host immunity without severe toxicity. Cancer Immunol Res 2, 154–166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilkie S, van Schalkwyk MC, Hobbs S, Davies DM, van der Stegen SJ, Pereira AC, Burbridge SE, Box C, Eccles SA, and Maher J. (2012). Dual targeting of ErbB2 and MUC1 in breast cancer using chimeric antigen receptors engineered to provide complementary signaling. J Clin Immunol 32, 1059–1070. [DOI] [PubMed] [Google Scholar]
- Wolchok JD, Kluger H, Callahan MK, Postow MA, Rizvi NA, Lesokhin AM, Segal NH, Ariyan CE, Gordon RA, Reed K. et al. (2013). Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med 369, 122–133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang J, Yan J, and Liu B. (2018). Targeting VEGF/VEGFR to modulate antitumor immunity. Front Immunol 9, 978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zappasodi R, Merghoub T, and Wolchok JD (2018). Emerging concepts for immune checkpoint blockade-based combination therapies (vol 33, pg 581, 2018). Cancer Cell 34, 690690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L, Conejo-Garcia JR, Katsaros D, Gimotty PA, Massobrio M, Regnani G, Makrigiannakis A, Gray H, Schlienger K, Liebman MN. et al. (2003). Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N Engl J Med 348, 203–213. [DOI] [PubMed] [Google Scholar]
- Zou W, Wolchok JD, and Chen L. (2016). PD-L1 (B7-H1) and PD-1 pathway blockade for cancer therapy: Mechanisms, response biomarkers, and combinations. Sci Transl Med 8, 328rv324. [DOI] [PMC free article] [PubMed] [Google Scholar]