Abstract
Background
Guidelines recommend a restrictive red blood cell transfusion strategy based on hemoglobin (Hb) concentrations in critically ill patients. We hypothesized that the arterial-venous oxygen difference (A-V O2diff), a surrogate for the oxygen delivery to consumption ratio, could provide a more personalized approach to identify patients who may benefit from transfusion.
Methods
A prospective observational study including 177 non-bleeding adult patients with a Hb concentration of 7.0–10.0 g/dL within 72 h after ICU admission. The A-V O2diff, central venous oxygen saturation (ScvO2), and oxygen extraction ratio (O2ER) were noted when a patient’s Hb was first within this range. Transfusion decisions were made by the treating physician according to institutional policy. We used the median A-V O2diff value in the study cohort (3.7 mL) to classify the transfusion strategy in each patient as “appropriate” (patient transfused when the A-V O2diff > 3.7 mL or not transfused when the A-V O2diff ≤ 3.7 mL) or “inappropriate” (patient transfused when the A-V O2diff ≤ 3.7 mL or not transfused when the A-V O2diff > 3.7 mL). The primary outcome was 90-day mortality.
Results
Patients managed with an “appropriate” strategy had lower mortality rates (23/96 [24%] vs. 36/81 [44%]; p = 0.004), and an “appropriate” strategy was independently associated with reduced mortality (hazard ratio [HR] 0.51 [95% CI 0.30–0.89], p = 0.01). There was a trend to less acute kidney injury with the “appropriate” than with the “inappropriate” strategy (13% vs. 26%, p = 0.06), and the Sequential Organ Failure Assessment (SOFA) score decreased more rapidly (p = 0.01). The A-V O2diff, but not the ScvO2, predicted 90-day mortality in transfused (AUROC = 0.656) and non-transfused (AUROC = 0.630) patients with moderate accuracy. Using the ROC curve analysis, the best A-V O2diff cutoffs for predicting mortality were 3.6 mL in transfused and 3.5 mL in non-transfused patients.
Conclusions
In anemic, non-bleeding critically ill patients, transfusion may be associated with lower 90-day mortality and morbidity in patients with higher A-V O2diff.
Trial registration
ClinicalTrials.gov, NCT03767127. Retrospectively registered on 6 December 2018.
Keywords: Red blood cell transfusion, Oxygen consumption, Critical care, Mortality
Background
The decision whether or not to transfuse an anemic patient is a considerable challenge in the intensive care unit (ICU) and should always consider the balance between the risks of anemia and the potential harms of red blood cell (RBC) transfusion in that patient. In critically ill patients, global oxygen consumption (VO2) is often increased, so that anemia can lead to impaired tissue oxygenation and contribute to adverse outcomes, including failed weaning from mechanical ventilation [1], acute kidney injury (AKI) [2], and increased mortality [3, 4]. However, transfusions are also associated with well-described risks, including transfusion-transmitted infections, transfusion-related acute lung injury (TRALI), transfusion-associated circulatory overload (TACO), and acquired immunosuppression [5, 6].
More than one fourth of critically ill patients receive a transfusion during their ICU stay [7], with the primary aim to increase global oxygen delivery (DO2) and decrease the DO2 to VO2 ratio [8, 9]. Clinical guidelines recommend a restrictive RBC transfusion strategy, i.e., transfusions limited to patients with a hemoglobin (Hb) concentration < 7 g/dL [10]. Of note, the guideline recommendations only refer to clinically stable intensive care patients, and there is still equipoise around the optimal transfusion strategy in several subgroups of patients. For example, a higher threshold of 8 or even 9 g/dL may be considered in patients with pre-existing cardiovascular disease [10]. The guidelines are based on results from several randomized clinical studies that showed that liberal transfusion strategies (keeping Hb > 9–10 g/dL) provided no clinical benefit in different populations when compared to a restrictive strategy [11–14]. However, the decision to transfuse should not be based only on Hb concentration [15, 16], because the optimal DO2 can differ from one patient to another [17] and over time in any one patient. These elements have resulted in considerable variability in worldwide transfusion practice, with the average Hb concentrations used to initiate transfusions higher than those recommended [7].
It has been suggested that mixed venous oxygen saturation (SvO2), which reflects the balance between VO2 and DO2, could be a valuable guide for transfusion in critically ill patients [18]. We hypothesized that patients with a greater arterial-venous oxygen difference (A-V O2diff), a surrogate for the ratio between DO2 and VO2, may benefit more from RBC transfusion than patients with lower A-V O2diff, who may benefit from avoiding transfusion. The aim of this observational study was therefore to evaluate whether “appropriate” RBC transfusions, as defined using the A-V O2diff, may be associated with reduced mortality rates and severity of organ dysfunction in non-acutely bleeding critically ill patients.
Methods
Study population
This prospective observational study was conducted in the Department of Intensive Care at the University Hospital of Ferrara (Italy) from 1 January 2017 to 1 October 2018. The study was approved by the local ethics committee (protocol number 160699, date of approval 14 July 2016). We included non-bleeding adult patients who had a Hb concentration between 7.0 and 10.0 g/dL within 72 h after admission to the ICU. All patients with a Hb concentration within the target range (7.0–10.0 g/dL) were included, regardless of whether or not a transfusion was given. Exclusion criteria were as follows: age < 18 years, acute bleeding, pregnancy, and no central venous catheter or central venous catheter placed in a femoral vein. Patients who were readmitted during the study period were not included a second time. Written informed consent was given by the patient or their next of kin. The manuscript is reported according to the STROBE criteria [19].
Transfusion policy
The decision to transfuse was made by the physician in charge of the patient according to our local transfusion policy, which includes [20–22] the following:
Hb target of 7–10 g/dL
A clinical decision that transfusion is appropriate, in addition to the Hb concentration
A single-unit transfusion strategy
A “first-in, first-out” policy (the oldest units are used first), to minimize blood wastage
RBC packs transfused during the study period were stored in saline, adenine, glucose, mannitol (SAGM) additive solution for up to 42 days at a mean temperature of 4 ± 2 °C. The RBC units were all leuko-reduced, according to the policy of our hospital during the study period. Characteristics of the RBC units used (i.e., days of storage, characteristics of donor) were not recorded.
Definitions
Chronic anemia was defined based on a known history of anemia and/or a decreased hematocrit (Hct) and Hb concentration on previous laboratory tests [23]. AKI was defined according to the Kidney Disease: Improving Global Outcomes (KDIGO) guidelines as an increase in serum creatinine of 0.3 mg/dL within 48 h, an increase in serum creatinine to 1.5 times the baseline value present within the previous 7 days, or urine volume < 0.5 mL/kg/h for 6 h [24]. Chronic renal disease was defined according to the same KDIGO guidelines [24].
Data collection
The Simplified Acute Physiology Score (SAPS) was recorded on admission. Demographic data and clinical variables, including heart rate, arterial pressure Glasgow coma scale score, and the Richmond Agitation-Sedation Scale, were recorded at the time of study inclusion (i.e., the first point at which the Hb concentration was measured as being between 7.0 and 10.0 g/dL). The Sequential Organ Failure Assessment (SOFA) score was calculated at the time of study inclusion and on the subsequent 5 days. Results of routine arterial and central venous blood sampling for blood gas analysis (GEM Premier™ 4000, Werfen, Le Pré-Saint-Gervais, France) and hematologic variables (Sysmex K-1000 auto-analyzer, Block Scientific, Bohemia, NY, USA) were recorded at the time of study inclusion. Decisions to transfuse on the day of study inclusion were also noted. Hb concentrations were measured at least daily as part of the routine practice and recorded 5 days after study inclusion. The number of RBC units given was also recorded for the first 5 days after study inclusion.
The A-V O2diff was calculated on the day of study inclusion as the difference between arterial oxygen content (CaO2) and central venous oxygen content (CcvO2) where:
and
We also calculated the oxygen extraction ratio (O2ER), defined as [(CaO2 − CcvO2)/CaO2]. Using the median A-V O2diff of our patients as the cutoff, we retrospectively categorized patients as having high A-V O2diff (greater than the median value) or low A-V O2diff (less than or equal to the median value). We then grouped patients into those who had been managed using an “appropriate” (transfused if A-V O2diff was high, not transfused if A-V O2diff was low) or an “inappropriate” (transfused if A-V O2diff was low, not transfused if A-V O2diff was high) transfusion strategy (Supplemental Figure S1). A patient was considered “non-transfused” if he/she did not receive RBC units on the day of study inclusion.
Treating physicians were not blinded to the A-V O2diff results, but were not supposed to use them to influence decisions to transfuse.
Outcomes
The primary outcome of the study was 90-day mortality. Secondary outcomes were the occurrence of AKI, the ICU length of stay, the mechanical ventilation- and vasopressor-free days over the 28 days post-study inclusion, the time course of organ dysfunction as assessed by the SOFA score over the 5 days post-study inclusion (the worst daily value was recorded for analysis), and the daily Hb concentrations.
Sample size
The sample size was calculated from a pilot study, presented as a congress abstract [25], in which the ratio of “appropriate” to “inappropriate” transfusion strategy was 1.38, with a mortality of 35% in the “inappropriate” group and 17% in the “appropriate” group. We therefore calculated that 148 patients should be enrolled to have 80% power to detect an 18% difference in 90-day mortality between the groups with an alfa error of 5%. Taking into account up to 20% loss to follow-up and a variation in the “appropriate”/“inappropriate” transfusion ratio, we planned to enroll at least 175 patients.
Statistical analysis
Normal distribution of data was tested using the Shapiro–Wilk normality test. Data are reported as mean ± standard deviation or median [interquartile range] when appropriate. Unpaired Student’s t tests or Mann–Whitney U tests were used for data with normal and non-normal distributions, respectively. ROC curves were used to analyze the ability of A-V O2diff to predict 90-day mortality in each group. Optimal cutoff scores for A-V O2diff were determined using Youden’s index. Differences in repeated measurements in the two groups were analyzed using repeated measure ANOVA or Friedman’s rank analysis for normally and not normally distributed variables, respectively. When multiple comparisons were made, p values were adjusted using the Bonferroni post hoc procedure. Mortality was also analyzed by quartiles of A-V O2diff in the “appropriate” and “inappropriate” groups. Multivariable logistic regression models were performed to investigate predictors of mortality; we included as covariates in the model all variables with p ≤ 0.10 at univariate analysis. Finally, a Cox proportional hazard regression model was used to obtain hazard ratios (HRs) for 90-day mortality. The analysis of ICU length of stay was only performed on patients discharged alive.
Four post hoc analyses were also performed. The first was to describe the time course of non-renal SOFA score over the first 5 days after study inclusion. The second was to identify the A-V O2diff cutoffs that were the best predictors of mortality in the whole population and in transfused and non-transfused patients. The third was to compare the results obtained using the A-V O2diff as a physiological marker of an “appropriate” strategy with those obtained using ScvO2 and O2ER as other oxygen-derived parameters. For this purpose, the area under the A-V O2diff receiver operating characteristic (AUROC) curve for 90-day mortality was compared with the AUROCs for ScvO2 and O2ER, using the median ScvO2 and O2ER values to define ScvO2-based and O2ER-based “appropriate” and “inappropriate” strategies. Finally, for patients who had a Hb concentration ≤ 10 g/dL at any time during the first 5 days after study inclusion, we calculated whether the transfusion strategy was “appropriate” or not on each day. We compared the rates of “appropriate” transfusion strategy in survivors and non-survivors, and mortality rates in patients in whom all transfusions were “appropriate” and all were “inappropriate.”
A p value < 0.05 was considered statistically significant. Statistical analyses were performed using SPSS Statistics for Windows, version 25.0 (IBM, Armonk, NY, USA).
Results
Study population
During the study period, 212 patients were screened for eligibility; 177 met the inclusion criteria and were enrolled (Supplemental Figure S2). The most common reasons for ICU admission were sepsis/septic shock (n = 58 [33%]) and respiratory failure (n = 50 [28%]) (Table 1).
Table 1.
Clinical and demographic characteristics
| Characteristic | All patients (n = 177) | “Appropriate” strategy (n = 96) | “Inappropriate” strategy (n = 81) | p value |
|---|---|---|---|---|
| Age, years | 72 ± 12 | 71 ± 14 | 72 ± 10 | 0.73 |
| BMI, kg/m2 | 28 ± 5 | 27 ± 4 | 29 ± 6 | 0.08 |
| SAPS II score at admission | 42 [29–50] | 42 [30–48] | 45 [33–55] | 0.06 |
| SOFA score at admission | 5 [2–6] | 5 [3–6] | 5 [2–6] | 0.81 |
| SOFA score at study inclusion | 5 [3–7] | 5 [3–7] | 5 [3–7] | 0.46 |
| RASS score at study inclusion | − 1 [− 2; 1] | − 2 [− 2; 1] | − 1 [− 2; 1] | 0.33 |
| ICU days before study inclusion | 1 [0–2] | 1 [0–2] | 1 [0–2] | 0.91 |
| Transfused, n (%) | 95 (54) | 48 (50) | 47 (58) | 0.36 |
| Comorbidity | ||||
| Heart disease, n (%) | 102 (58) | 58 (60) | 44 (54) | 0.50 |
| Hypertension, n (%) | 118 (67) | 62 (65) | 56 (69) | 0.63 |
| Diabetes, n (%) | 53 (30) | 26 (27) | 27 (33) | 0.46 |
| Chronic anemia, n (%) | 28 (16) | 15 (16) | 13 (16) | 0.99 |
| COPD/asthma, n (%) | 30 (17) | 11 (11) | 19 (23) | 0.06 |
| History of smoking, n (%) | 45 (26) | 25 (26) | 14 (17) | 0.22 |
| Chronic renal disease, n (%) | 49 (28) | 22 (23) | 27 (33) | 0.22 |
| Reason for admission | ||||
| Sepsis/septic shock, n (%) | 58 (33) | 30 (31) | 28 (35) | 0.76 |
| Respiratory failure, n (%) | 50 (28) | 30 (31) | 20 (25) | 0.42 |
| Hypovolemic shock, n (%) | 28 (16) | 15 (16) | 13 (16) | 0.94 |
| Cardiogenic shock, n (%) | 19 (11) | 6 (6) | 13 (16) | 0.06 |
| Trauma, n (%) | 8 (4) | 4 (4) | 4 (5) | 0.80 |
| Others | 13 (7) | 11 (11) | 3 (1) | 0.10 |
| Interventions on admission | ||||
| Mechanical ventilation, n (%) | 157 (88) | 88 (93) | 69 (85) | 0.18 |
| Vasopressors, n (%) | 81 (46) | 47 (49) | 34 (42) | 0.40 |
| Laboratory values on inclusion | ||||
| Hemoglobin, g/dL | 8.7 ± 0.7 | 8.8 ± 0.6 | 8.6 ± 0.8 | 0.06 |
| MCV, fL | 88 ± 9 | 90 ± 6 | 87 ± 11 | 0.07 |
| RDW, % | 15.9 ± 3.0 | 15.4 ± 2.5 | 16.4 ± 3.3 | 0.02 |
| Platelets, 103/μL | 179 [129–266] | 178 [132–282] | 189 [126–244] | 0.72 |
| INR | 1.27 ± 0.3 | 1.26 ± 0.3 | 1.32 ± 0.3 | 0.12 |
| Creatinine, mg/dL | 1.09 [0.89–2.01] | 1.08 [0.77–1.96] | 1.10 [0.98–2.40] | 0.15 |
| Bilirubin, mg/dL | 0.72 [0.41–1.10] | 0.85 [0.45–1.00] | 0.63 [0.41–1.00] | 0.26 |
| Lactate, mmol/L | 1.6 [1.1–2.0] | 1.4 [1.0–2.0] | 2.0 [1.2–2.0] | 0.04 |
| CaO2, mL | 12.4 ± 1.6 | 12.5 ± 1.1 | 12.2 ± 2.1 | 0.26 |
| ScvO2, % | 71 ± 9 | 71 ± 10 | 73 ± 9 | 0.16 |
BMI body mass index, SAPS Simplified Acute Physiology Score, RASS Richmond Agitation-Sedation Scale, COPD chronic obstructive pulmonary disease, RDW red blood cell distribution width, MCV mean corpuscular volume, INR international normalized ratio, CaO2 arterial oxygen content, ScvO2 central venous oxygen saturation
The median A-V O2diff of the whole population was 3.7 mL, and using this value, 96 patients (54%) were considered to have been managed using an “appropriate” transfusion strategy and 81 (46%) using an “inappropriate” strategy. The transfusion rates in the “appropriate” and “inappropriate” groups were 50% (48/96) and 58% (47/81), respectively (p = 0.36). The mean lowest Hb concentration before transfusion was 8.3 ± 0.7 (range 7.0–9.8) g/dL, and the mean first Hb after transfusion was 9.0 ± 0.7 g/dL (range 7.7–11.3). There were no significant differences in Hb concentrations between the groups during the study period (Supplemental Figure S3). The CaO2 at study inclusion and the proportion of patients with a history of chronic anemia did not differ between the groups (Table 1).
Ninety-day mortality
The 90-day mortality was 33% (59/177) and was similar in transfused and non-transfused patients (31/95 [33%] vs. 28/82 [34%]; p = 0.87). The RBC transfusion rate was not significantly different in survivors and non-survivors (Supplemental Table 1). Patients in the “appropriate” strategy group had a lower mortality rate than the other patients (23/96 [24%] vs. 36/81 [44%]; odds ratio [OR] = 0.39 [95% CI 0.21–0.75], p = 0.004); this was mainly evident in the patients who received a transfusion (8/48 [17%] vs. 23/47 [49%]; p = 0.001) (Fig. 1). The association between “appropriate” strategy and lower mortality rate was confirmed in a subgroup analysis taking into account different Hb levels (Online Supplement).
Fig. 1.
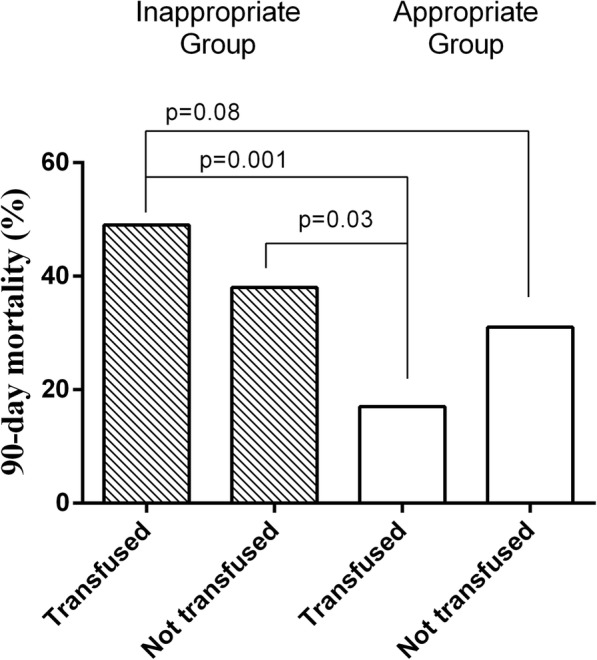
Ninety-day mortality in transfused and non-transfused patients in each group
Higher SAPS II score, RBC distribution width (RDW) values, and lactate concentrations on admission were associated with higher 90-day mortality in univariate analysis, but these were not significant after multivariable adjustment (Table 2). The use of an “appropriate” transfusion strategy was independently associated with lower 90-day mortality in a multivariable-adjusted Cox regression (HR 0.51 [95% CI 0.30–0.89], p = 0.01) (Table 2, Fig. 2). A-V O2diff as a continuous variable was associated with 90-day mortality in non-transfused patients (OR = 1.50 [95% CI 1.02–2.20], p = 0.04); this association was confirmed in the multivariable analysis (Supplemental Table 2).
Table 2.
Univariate and multivariable analyses with 90-day mortality as the dependent variable
| Variables | Unadjusted odds ratio | p value | Adjusted odds ratio | p value |
|---|---|---|---|---|
| Age, years | 1.02 [0.98–1.03] | 0.85 | ||
| BMI, kg/m2 | 1.09 [0.99–1.20] | 0.73 | ||
| SAPS II | 1.02 [1.00–1.05] | 0.05 | 1.01 [0.99–1.04] | 0.39 |
| Transfused, n (%) | 0.93 [0.49–1.74] | 0.83 | ||
| “Appropriate” group | 0.39 [0.21–0.75] | 0.004 | 0.48 [0.25–0.92] | 0.03 |
| SOFA score | 1.07 [0.95–1.22] | 0.26 | ||
| Comorbidity | ||||
| Heart disease, n (%) | 1.17 [0.56–2.42] | 0.57 | ||
| Hypertension, n (%) | 1.18 [0.55–2.55] | 0.67 | ||
| Diabetes, n (%) | 1.74 [0.81–3.75] | 0.16 | ||
| COPD/asthma, n (%) | 0.61 [0.23–1.65] | 0.33 | ||
| History of smoking, n (%) | 1.02 [0.57–2.52] | 0.98 | ||
| Chronic renal disease, n (%) | 1.10 [0.49–2.44] | 0.81 | ||
| Laboratory values on inclusion | ||||
| Hemoglobin, g/dL | 1.16 [0.74–1.81] | 0.51 | ||
| Platelets, 103/μL | 0.99 [0.99–1.01] | 0.20 | ||
| INR | 1.57 [0.42–5.79] | 0.49 | ||
| RDW, % | 1.12 [1.01–1.25] | 0.04 | 1.11 [0.99–1.24] | 0.07 |
| Creatinine, mg/dL | 1.03 [0.92–1.29] | 0.68 | ||
| Bilirubin, mg/dL | 1.35 [0.84–2.18] | 0.21 | ||
| Lactate, mmol/L | 1.36 [1.04–1.78] | 0.02 | 1.22 [0.95–1.59] | 0.12 |
| PaO2/FiO2 ratio | 0.99 [0.99–1.13] | 0.62 | ||
BMI body mass index, SAPS Simplified Acute Physiology Score, COPD chronic obstructive pulmonary disease, RDW red blood cell distribution width, INR international normalized ratio, PaO2 partial pressure of oxygen, FiO2 fraction of inspired oxygen
Fig. 2.
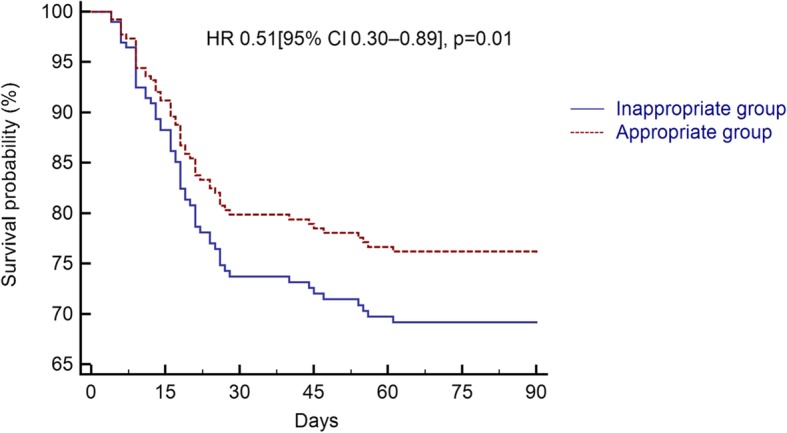
Cox regression analysis for 90-day mortality in the “appropriate” and “inappropriate” groups
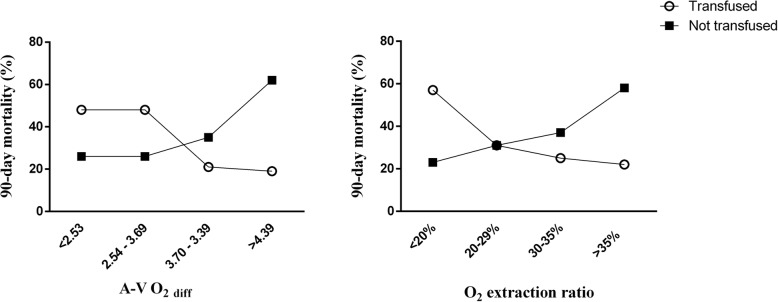
In all non-transfused patients, mortality increased across increasing A-V O2diff quartiles; in all transfused patients, mortality decreased over A-V O2diff quartiles (Fig. 3). The ROC analysis showed that the A-V O2diff was a moderate independent predictor of 90-day mortality in transfused (AUROC = 0.656, best cutoff = 3.6 mL) and non-transfused (AUROC = 0.630, best cutoff = 3.5 mL) patients.
Fig. 3.
Ninety-day mortality in transfused and non-transfused patients according to quartiles of arterial-venous oxygen difference (A-V O2diff) and O2 extraction ratio
Secondary outcomes and post hoc analyses
Fewer patients in the “appropriate” than in the “inappropriate” strategy group developed AKI (21/81 [26%] vs. 13/96 [13%]; p = 0.06) (Supplemental Table 3). The number of patients transfused during the first 5 days after study inclusion was similar in the two groups as was the median number of RBC units given (Supplemental Table 3). The SOFA score decreased more rapidly in patients managed with the “appropriate” strategy than in other patients (Supplemental Figure S4; p = 0.019). To avoid cross-interactions between the occurrence of AKI and the SOFA score evaluation, we also analyzed the non-renal SOFA scores, which gave similar results (p = 0.009 for comparison between “appropriate” and “inappropriate” strategies; data not shown).
Mortality prediction was better for A-V O2diff than for ScvO2 (AUROC = 0.489 in transfused and AUROC = 0.440 in non-transfused) and O2ER (AUROC = 0.623 in transfused and AUROC = 0.619 in non-transfused). An O2ER-based “appropriate” strategy (using the median value of 29%) was also independently associated with a lower 90-day mortality (OR 0.44 [95% CI 0.23–0.86]; p = 0.02) (Supplemental Table 4), but a ScvO2-based “appropriate” strategy (using the median value of 71.5%) was not (OR 0.60 [95% CI 0.32–1.13]; p = 0.11).
When we divided the population using the best A-V O2diff cutoffs for predicting mortality determined by ROC curve analysis (i.e., 3.6 mL in transfused and 3.5 mL in non-transfused patients) rather than the median value, we identified 94 patients who were “appropriately” transfused. There was a significant association between 90-day mortality and “appropriateness” of transfusion even when these cutoffs were used (HR 0.46 [95% CI 0.27–0.77]; p = 0.004).
During the first 5 days after study inclusion, 100 patients had a Hb concentration ≤ 10 g/dL on day 2, 97 patients on day 3, 64 on day 4, and 64 on day 5. During the study period, the transfusion strategy when the Hb was ≤ 10 g/dL was always “appropriate” in 76 of the patients and always “inappropriate” in 50 patients; the mortality rates were 20% (15/76) and 56% (28/50), respectively (p < 0.001).
Discussion
The main result of this study is that the evaluation of the A-V O2diff can identify critically ill patients who seem to benefit from RBC transfusion. Indeed, patients included in the “appropriate” transfusion group, defined according to the A-V O2diff on study inclusion (i.e., transfused when the A-V O2diff was > 3.7 mL and not transfused when the A-V O2diff was ≤ 3.7 mL), had lower 90-day mortality, and organ function improved more rapidly in those in the “inappropriate” group. Moreover, RBC transfusions in patients with an A-V O2diff ≤ 3.7 mL (“inappropriate” strategy) were associated with higher mortality, raising the hypothesis that transfusion may have harmful effects in patients without impaired oxygen reserve; this finding should be a starting point for future studies investigating whether avoiding RBC transfusion in patients without impaired oxygen reserve may not only be cost-saving but also clinically relevant [26].
Although some studies and many experts endorse the opinion that decisions to transfuse should not be based on Hb concentrations alone [7, 15, 16, 27–30], the latest guidelines based their recommendations only on this variable, suggesting that the Hb should be kept > 7 g/dL [10]. However, there is an individual response to low Hb concentrations, and patients with less physiological reserve are more vulnerable to anemia [15]. Differences in individual oxygen demand could partially explain the individual tolerance to anemia and, consequently, some of the conflicting results reported in the literature. A liberal transfusion strategy aimed at keeping Hb values > 9 g/dL was recently shown to be superior to a restrictive strategy in critically ill oncologic patients [31], and a recent clinical trial in vascular surgery suggested potential harm with a restrictive strategy, including lower cerebral tissue oxygenation and higher mortality rate [32]. However, a liberal strategy was associated with greater mortality in younger ICU patients (< 55 years) and those with an APACHE II score < 20 [28]. The A-V O2diff may represent a good marker of a patient’s oxygen reserve, influenced by the patient’s comorbidities and oxygen demand. Indeed, we showed a reduction in mortality in the “appropriate” strategy group but no statistically significant difference in Hb level between the two groups; accordingly, within the same Hb range (7–10 g/dL), we observed great individual variability in the A-V O2diff.
The concept of a physiological transfusion trigger was introduced by Adamczyk et al., who proposed the use of ScvO2 as a tool to assess tolerance to anemia [33]. These authors showed that, in patients undergoing general anesthesia, RBC transfusion could result in a 5% increase in SvO2 only when pre-transfusion ScvO2 values were < 70%. We showed that basing the transfusion strategy on the difference between arterial and venous oxygen content, a physiological principle that reflects the balance between VO2 and DO2 can improve clinical outcomes. Additionally, we showed that the A-V O2diff was a better prognostic factor than ScvO2, probably because the A-V O2diff provides a more accurate estimation of the DO2 to VO2 ratio than does ScvO2 [34]. In support of this hypothesis, we also showed that O2ER, another surrogate of the DO2 to VO2 ratio, could be used to identify the appropriateness of a transfusion strategy, with an O2ER-based “inappropriate” strategy being independently associated with higher mortality. These results have some physiological background: the A-V O2diff has been shown to be more accurate than ScvO2 in predicting tissue hypoxia [35]; further, the A-V O2diff showed a strong inverse relationship with hemodilution [36], a well-known cause of unnecessary transfusion, whereas ScvO2 did not [37]. Finally, ScvO2 is considered a good estimator of oxygen extraction only when assuming that SaO2 = 1, but in hypoxemic patients, a low SaO2 can result in low ScvO2 values with normal A-V O2diff.
There was a trend towards more AKI with the “inappropriate” than with the “appropriate” strategy. Renal oxygen consumption is known to be high [38], so this organ may be particularly sensitive to an imbalance between DO2 and VO2. Moreover, RBCs can have beneficial effects on renal microvascular oxygenation [39]. Because the oxygen consumption of the kidney is so high, it is reasonable that an imbalance between DO2 and VO2 may affect this organ first. We also showed better organ function recovery in patients managed with an “appropriate” transfusion strategy, as shown by a more rapid decrease in SOFA score. RBC transfusions have a greater benefit in patients with severe organ failure, as reflected by a high SOFA score [7], but the average SOFA score at study inclusion did not differ in our two groups of patients. Therefore, in the presence of high A-V O2diff, RBC transfusion may be beneficial regardless of the baseline SOFA score. When transfused, patients with a high A-V O2diff may also benefit from some relief of the heart when cardiac function is compromised [27]; of note, previous studies in animal models showed that treatments focused on improving myocardial VO2/efficiency were associated with improved ventricular function [40]. We did not explore cardiac function well enough to be able to evaluate this effect.
Our study has some limitations. First, the observational design does not enable us to establish a cause-effect relationship, and we cannot exclude the possibility that other clinical variables could have contributed to the difference in mortality described; a prospective randomized study would be needed for this purpose, and this study should therefore be considered as hypothesis generating. Second, because of the lack of published data in this field, we chose to separate patients into two groups based on median values, rather than trying to generate an optimal cutoff value; however, the Youden index analysis showed that the “best” A-V O2diff cutoff identified, a value we could potentially have used to interpret the data, was very similar to the median value. Furthermore, the post hoc analysis performed using the “best” A-V O2diff cutoff seemed to confirm our results. Third, the lack of hemodynamic monitoring did not enable us to separately investigate patients with and without preload dependency. We also did not systematically assess the clinical signs of cardiac congestion and thus cannot compare the occurrence of heart failure in the two groups. Future studies should address the reproducibility of our results in different hemodynamic settings. In this connection, we cannot exclude that a transfusion algorithm which considers clinical variables other than oxygen consumption (hemodynamic setting, lactate, SOFA score, and others) would improve our results; however, our study was not designed, and not powered enough, to develop such clinical tool. Similarly, we defined the occurrence of AKI according to the KDIGO guidelines, without including measurement of markers of kidney injury, such as cystatin C or neutrophil gelatinase-associated lipocalin; the addition of more specific markers may have strengthened our findings. Fourth, a recent meta-analysis showed that RBC transfusion can decrease O2ER in critically ill patients [41]. We did not systematically record A-V O2diff and O2ER after transfusion and, therefore, cannot describe the A-V O2diff and O2ER changes before and after RBC transfusion in the “appropriate” and “inappropriate” groups. In addition, although we showed a trend to an association of higher RDW values and mortality, as recently described [42], this was not confirmed in the multivariable analysis; nonetheless, there is a complex relationship between RDW values, iron deficiency, and RBC transfusion [43, 44], and our study was not designed to investigate this issue. Fifth, A-V O2diff measurement requires a correctly placed central venous catheter; although widely used in critically ill patients, the central venous catheter is not always available or not placed in the superior vena cava. Finally, our transfusion rate (54%) was higher than that described in other studies [7], and we considered only patients who had a Hb concentration between 7.0 and 10.0 g/dL within 72 h after admission. External validation of our results and further analysis on patients with anemia later during the ICU stay are therefore needed.
Conclusions
In anemic, non-bleeding critically ill patients, transfusion may be associated with lower 90-day mortality and morbidity in patients with higher A-V O2diff. These results support the hypothesis that transfusion therapy should be individualized according to the oxygen reserve of the patient.
Supplementary information
Additional file 1: Fig. S1. Study definitions of “inappropriate” and “appropriate” transfusion strategies.
Additional file 2: Fig. S2. Flowchart of the study.
Additional file 3; Fig. S3. Daily changes in hemoglobin levels after study inclusion. Data are shown as mean ± standard deviation.
Additional file 4: Fig. S4. Daily assessment of SOFA score after study inclusion. + indicates p=0.048, * indicates p=0.002, # indicates p=0.001 comparing “inappropriate” with “appropriate” transfusion strategy
Additional file 5: Table S1. Clinical and demographic characteristics in survivors and non-survivors. Table S2. Univariate and multivariate analysis with 90-day mortality as dependent variable in non-transfused patients. Table S3. Secondary outcomes in the two groups. Table S4. Univariate and multivariate analysis with 90-day mortality as dependent variable. Appropriate group was defined using an oxygen extraction ratio (O2ER)-based strategy.
Abbreviations
- ICU
Intensive care unit
- RBC
Red blood cell
- VO2
Oxygen consumption
- AKI
Acute kidney injury
- TRALI
Transfusion-related acute lung injury
- TACO
Transfusion-associated circulatory overload
- DO2
Oxygen delivery
- Hb
Hemoglobin
- SvO2
Mixed venous oxygen saturation
- A-V O2diff
Arterial-venous oxygen difference
- SAPS
Simplified Acute Physiology Score
- SOFA
Sequential Organ Failure Assessment
- SAGM
Saline, adenine, glucose, mannitol
- CaO2
Arterial oxygen content
- CcvO2
Central venous oxygen content
- O2ER
Oxygen extraction ratio
Authors’ contributions
FA, FST, VCA, and SS were involved in the conception and the design of the study, analyzed the data, and wrote the paper. FA, BG, ME, and RR collected the data. FA performed the statistical work. FST, JLV, CE, and JC contributed to the analysis of the data and critical revision of the manuscript for important intellectual content. All authors reviewed the manuscript and agreed with the final version.
Funding
The study was supported by the University of Ferrara, Ferrara, Italy.
Availability of data and materials
The datasets generated during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
The study was approved by the local ethics committee (Comitato etico unico della Provincia di Ferrara, protocol number 160699, date of approval 14 July 2016).
Consent for publication
Not applicable
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Alberto Fogagnolo and Fabio Silvio Taccone were joint first authors.
Supplementary information
Supplementary information accompanies this paper at 10.1186/s13054-020-2827-5.
References
- 1.Khamiees M, Raju P, DeGirolamo A, Amoateng-Adjepong Y, Manthous CA. Predictors of extubation outcome in patients who have successfully completed a spontaneous breathing trial. Chest. 2001;120(4):1262–1270. doi: 10.1378/chest.120.4.1262. [DOI] [PubMed] [Google Scholar]
- 2.Malhotra R, Kashani KB, Macedo E, Kim J, Bouchard J, Wynn S, Li G, Ohno-Machado L, Mehta R. A risk prediction score for acute kidney injury in the intensive care unit. Nephrol Dial Transplant. 2017;32(5):814–822. doi: 10.1093/ndt/gfx026. [DOI] [PubMed] [Google Scholar]
- 3.Rasmussen L, Christensen S, Lenler-Petersen P, Johnsen SP. Anemia and 90-day mortality in COPD patients requiring invasive mechanical ventilation. Clin Epidemiol. 2010;3:1–5. doi: 10.2147/CLEP.S12885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Carson JL, Noveck H, Berlin JA, Gould SA. Mortality and morbidity in patients with very low postoperative Hb levels who decline blood transfusion. Transfusion. 2002;42(7):812–818. doi: 10.1046/j.1537-2995.2002.00123.x. [DOI] [PubMed] [Google Scholar]
- 5.Vlaar AP, Wolthuis EK, Hofstra JJ, Roelofs JJ, Boon L, Schultz MJ, Lutter R, Juffermans NP. Mechanical ventilation aggravates transfusion-related acute lung injury induced by MHC-I class antibodies. Intensive Care Med. 2010;36(5):879–887. doi: 10.1007/s00134-010-1802-z. [DOI] [PubMed] [Google Scholar]
- 6.Bosboom JJ, Klanderman RB, Zijp M, Hollmann MW, Veelo DP, Binnekade JM, Geerts BF, Vlaar APJ. Incidence, risk factors, and outcome of transfusion-associated circulatory overload in a mixed intensive care unit population: a nested case-control study. Transfusion. 2018;58(2):498–506. doi: 10.1111/trf.14432. [DOI] [PubMed] [Google Scholar]
- 7.Vincent JL, Jaschinski U, Wittebole X, Lefrant JY, Jakob SM, Almekhlafi GA, Pellis T, Tripathy S, Rubatto Birri PN, et al. Worldwide audit of blood transfusion practice in critically ill patients. Crit Care. 2018;22(1):102. doi: 10.1186/s13054-018-2018-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hayden SJ, Albert TJ, Watkins TR, Swenson ER. Anemia in critical illness: insights into etiology, consequences, and management. Am J Respir Crit Care Med. 2012;185(10):1049–1057. doi: 10.1164/rccm.201110-1915CI. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Van der Linden P, Rausin I, Deltell A, Bekrar Y, Gilbart E, Bakker J, Vincent JL. Detection of tissue hypoxia by arteriovenous gradient for PCO2 and pH in anesthetized dogs during progressive hemorrhage. Anesth Analg. 1995;80(2):269–275. doi: 10.1097/00000539-199502000-00012. [DOI] [PubMed] [Google Scholar]
- 10.Mueller MM, Van RH, Meybohm P, Aranko K, Aubron C, Burger R, Carson JL, Cichutek K, De BE, et al. Patient blood management: recommendations from the 2018 Frankfurt Consensus Conference. JAMA. 2019;321(10):983–997. doi: 10.1001/jama.2019.0554. [DOI] [PubMed] [Google Scholar]
- 11.Carson JL, Terrin ML, Noveck H, Sanders DW, Chaitman BR, Rhoads GG, Nemo G, Dragert K, Beaupre L, et al. Liberal or restrictive transfusion in highrisk patients after hip surgery. N Engl J Med. 2011;365(26):2453–2462. doi: 10.1056/NEJMoa1012452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hebert PC, Wells G, Blajchman MA, Marshall J, Martin C, Pagliarello G, Tweeddale M, Schweitzer I, Yetisir E. Transfusion Requirements in Critical Care Investigators, Canadian Critical Care Trials Group. A multicenter, randomized, controlled clinical trial of transfusion requirements in critical care. N Engl J Med. 1999;340(6):409–417. doi: 10.1056/NEJM199902113400601. [DOI] [PubMed] [Google Scholar]
- 13.Villanueva C, Colomo A, Bosch A, Concepcion M, Hernandez-Gea V, Aracil C, Graupera I, Poca M, Alvarez-Urturi C, et al. Transfusion strategies for acute upper gastrointestinal bleeding. N Engl J Med. 2013;368(1):11–21. doi: 10.1056/NEJMoa1211801. [DOI] [PubMed] [Google Scholar]
- 14.Murphy GJ, Pike K, Rogers CA, Wordsworth S, Stokes EA, Angelini GD, Reeves BC. Liberal or restrictive transfusion after cardiac surgery. N Engl J Med. 2015;372(11):997–1008. doi: 10.1056/NEJMoa1403612. [DOI] [PubMed] [Google Scholar]
- 15.Vincent JL. Indications for blood transfusions: too complex to base on a single number? Ann Intern Med. 2012;157(1):71–72. doi: 10.7326/0003-4819-156-12-201206190-00431. [DOI] [PubMed] [Google Scholar]
- 16.Sakr Y, Vincent JL. Should red cell transfusion be individualized? Yes Intensive Care Med. 2015;41(11):1973–1976. doi: 10.1007/s00134-015-3950-7. [DOI] [PubMed] [Google Scholar]
- 17.Du Pont-Thibodeau G, Harrington K, Lacroix J. Anemia and red blood cell transfusion in critically ill cardiac patients. Ann Intensive Care. 2014;4:16. doi: 10.1186/2110-5820-4-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Vallet B, Robin E, Lebuffe G. Venous oxygen saturation as a physiologic transfusion trigger. Crit Care. 2010;14(2):213. doi: 10.1186/cc8854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol. 2008;61(4):344–349. doi: 10.1016/j.jclinepi.2007.11.008. [DOI] [PubMed] [Google Scholar]
- 20.Kozek-Langenecker SA, Afshari A, Albaladejo P, Santullano CA, De RE, Filipescu DC, Fries D, Gorlinger K, Haas T, et al. Management of severe perioperative bleeding: guidelines from the European Society of Anaesthesiology. Eur J Anaesthesiol. 2013;30(6):270–382. doi: 10.1097/EJA.0b013e32835f4d5b. [DOI] [PubMed] [Google Scholar]
- 21.Yang WW, Thakkar RN, Gehrie EA, Chen W, Frank SM. Single-unit transfusions and hemoglobin trigger: relative impact on red cell utilization. Transfusion. 2017;57(5):1163–1170. doi: 10.1111/trf.14000. [DOI] [PubMed] [Google Scholar]
- 22.Spadaro S, Taccone FS, Fogagnolo A, Fontana V, Ragazzi R, Verri M, Valpiani G, Greco P, Bianconi M, et al. The effects of storage of red blood cells on the development of postoperative infections after noncardiac surgery. Transfusion. 2017;57(11):2727–2737. doi: 10.1111/trf.14249. [DOI] [PubMed] [Google Scholar]
- 23.Cullis JO. Diagnosis and management of anaemia of chronic disease: current status. Br J Haematol. 2011;154(3):289–300. doi: 10.1111/j.1365-2141.2011.08741.x. [DOI] [PubMed] [Google Scholar]
- 24.Okusa MD, Davenport A. Reading between the (guide)lines--the KDIGO practice guideline on acute kidney injury in the individual patient. Kidney Int. 2014;85(1):39–48. doi: 10.1038/ki.2013.378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fogagnolo A, Spadaro S, Creteur J, Cavalcante E, Taccone FS, Volta CA. Can arterio-venous-oxygen content difference be a target to guide transfusion in critically ill patients? Intensive Care Med Exp. 2017;5(Suppl 2):44. [Google Scholar]
- 26.Carson JL, Guyatt G, Heddle NM, Grossman BJ, Cohn CS, Fung MK, Gernsheimer T, Holcomb JB, Kaplan LJ, et al. Clinical practice guidelines from the AABB: red blood cell transfusion thresholds and storage. JAMA. 2016;316(19):2025–2035. doi: 10.1001/jama.2016.9185. [DOI] [PubMed] [Google Scholar]
- 27.Vincent JL, Van der Linden P. Restrictive versus more liberal blood transfusions? The answer is in your heart. Minerva Anestesiol. 2016;82(5):511–513. [PubMed] [Google Scholar]
- 28.Deans KJ, Minneci PC, Suffredini AF, Danner RL, Hoffman WD, Ciu X, Klein HG, Schechter AN, Banks SM, et al. Randomization in clinical trials of titrated therapies: unintended consequences of using fixed treatment protocols. Crit Care Med. 2007;35(6):1509–1516. doi: 10.1097/01.CCM.0000266584.40715.A6. [DOI] [PubMed] [Google Scholar]
- 29.Lelubre C, Vincent JL, Taccone FS. Red blood cell transfusion strategies in critically ill patients: lessons from recent randomized clinical studies. Minerva Anestesiol. 2016;82(9):1010–1016. [PubMed] [Google Scholar]
- 30.Chandra S, Kulkarni H, Westphal M. The bloody mess of red blood cell transfusion. Crit Care. 2017;21:310. doi: 10.1186/s13054-017-1912-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bergamin FS, Almeida JP, Landoni G, Galas FRBG, Fukushima JT, Fominskiy E, Park CHL, Osawa EA, Diz MPE, et al. Liberal versus restrictive transfusion strategy in critically ill oncologic patients: the transfusion requirements in critically ill oncologic patients randomized controlled trial. Crit Care Med. 2017;45(5):766–773. doi: 10.1097/CCM.0000000000002283. [DOI] [PubMed] [Google Scholar]
- 32.Moller A, Nielsen HB, Wetterslev J, Pedersen OB, Hellemann D, Winkel P, Marcussen KV, Ramsing BGU, Mortensen A, et al. Low vs high hemoglobin trigger for transfusion in vascular surgery: a randomized clinical feasibility trial. Blood. 2019;133(25):2639–2650. doi: 10.1182/blood-2018-10-877530. [DOI] [PubMed] [Google Scholar]
- 33.Adamczyk S, Robin E, Barreau O, Fleyfel M, Tavernier B, Lebuffe G, Vallet B. Contribution of central venous oxygen saturation in postoperative blood transfusion decision. Ann Fr Anesth Reanim. 2009;28(6):522–530. doi: 10.1016/j.annfar.2009.03.013. [DOI] [PubMed] [Google Scholar]
- 34.Squara P. Central venous oxygenation: when physiology explains apparent discrepancies. Crit Care. 2014;18(5):579. doi: 10.1186/s13054-014-0579-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mekontso-Dessap A, Castelain V, Anguel N, Bahloul M, Schauvliege F, Richard C, Teboul JL. Combination of venoarterial PCO2 difference with arteriovenous O2 content difference to detect anaerobic metabolism in patients. Intensive Care Med. 2002;28(3):272–277. doi: 10.1007/s00134-002-1215-8. [DOI] [PubMed] [Google Scholar]
- 36.Dubin A, Ferrara G, Kanoore Edul VS, Martins E, Canales HS, Canullan C, Murias G, Pozo MO, Estenssoro E. Venoarterial PCO2-to-arteriovenous oxygen content difference ratio is a poor surrogate for anaerobic metabolism in hemodilution: an experimental study. Ann Intensive Care. 2017;7:65. doi: 10.1186/s13613-017-0288-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Krantz T, Warberg J, Secher NH. Venous oxygen saturation during normovolaemic haemodilution in the pig. Acta Anaesthesiol Scand. 2005;49(8):1149–1156. doi: 10.1111/j.1399-6576.2005.00778.x. [DOI] [PubMed] [Google Scholar]
- 38.Ricksten SE, Bragadottir G, Redfors B. Renal oxygenation in clinical acute kidney injury. Crit Care. 2013;17:221. doi: 10.1186/cc12530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zafrani L, Ergin B, Kapucu A, Ince C. Blood transfusion improves renal oxygenation and renal function in sepsis-induced acute kidney injury in rats. Crit Care. 2016;20:406. doi: 10.1186/s13054-016-1581-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Santos-Gallego CG, Requena-Ibanez JA, San Antonio R, Ishikawa K, Watanabe S, Picatoste B, Flores E, Garcia-Ropero A, et al. Empagliflozin ameliorates adverse left ventricular remodeling in nondiabetic heart failure by enhancing myocardial energetics. J Am Coll Cardiol. 2019;73(15):1931–1944. doi: 10.1016/j.jacc.2019.01.056. [DOI] [PubMed] [Google Scholar]
- 41.Cavalcante Dos Santos E, Orbegozo D, Mongkolpun W, Galfo V, Nan W, Gouvêa Bogossian E, Taccone FS, Vallet B, Creteur J, Vincent JL. Systematic review and meta-analysis of effects of transfusion on hemodynamic and oxygenation variables. Crit Care Med. 2019;48(2):241–248. doi: 10.1097/CCM.0000000000004115. [DOI] [PubMed] [Google Scholar]
- 42.Fogagnolo A, Spadaro S, Taccone FS, Ragazzi R, Romanello A, Fanni A, Marangoni E, Franchi F, Scolletta S, Volta CA. The prognostic role of red blood cell distribution width in transfused and non-transfused critically ill patients. Minerva Anestesiol. 2019;85(11):1159–1167. doi: 10.23736/S0375-9393.19.13522-5. [DOI] [PubMed] [Google Scholar]
- 43.Lasocki S, Lefebvre T, Mayeur C, Puy H, Mebazaa A, Gayat E, FROG-ICU study group Iron deficiency diagnosed using hepcidin on critical care discharge is an independent risk factor for death and poor quality of life at one year: an observational prospective study on 1161 patients. Crit Care. 2018;22(1):314. doi: 10.1186/s13054-018-2253-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Spadaro S, Taccone FS, Fogagnolo A, Franchi F, Scolletta S, Ragazzi R, Fanni A, Marangoni E, Govoni M, et al. The effects of blood transfusion on red blood cell distribution width in critically ill patients: a pilot study. Transfusion. 2018;58(8):1863–1869. doi: 10.1111/trf.14759. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Fig. S1. Study definitions of “inappropriate” and “appropriate” transfusion strategies.
Additional file 2: Fig. S2. Flowchart of the study.
Additional file 3; Fig. S3. Daily changes in hemoglobin levels after study inclusion. Data are shown as mean ± standard deviation.
Additional file 4: Fig. S4. Daily assessment of SOFA score after study inclusion. + indicates p=0.048, * indicates p=0.002, # indicates p=0.001 comparing “inappropriate” with “appropriate” transfusion strategy
Additional file 5: Table S1. Clinical and demographic characteristics in survivors and non-survivors. Table S2. Univariate and multivariate analysis with 90-day mortality as dependent variable in non-transfused patients. Table S3. Secondary outcomes in the two groups. Table S4. Univariate and multivariate analysis with 90-day mortality as dependent variable. Appropriate group was defined using an oxygen extraction ratio (O2ER)-based strategy.
Data Availability Statement
The datasets generated during the current study are available from the corresponding author on reasonable request.