Abstract
OBJECTIVE
The prevalence of type 2 diabetes is increasing among adults under age 45. Onset of type 2 diabetes at a younger age increases an individual’s risk for diabetes-related complications. Given the lasting benefits conferred by early glycemic control, we compared glycemic control and initial care between adults with younger onset (21–44 years) and mid-age onset (45–64 years) of type 2 diabetes.
RESEARCH DESIGN AND METHODS
Using data from a large, integrated health care system, we identified 32,137 adults (aged 21–64 years) with incident diabetes (first HbA1c ≥6.5% [≥48 mmol/mol]). We excluded anyone with evidence of prior type 2 diabetes, gestational diabetes mellitus, or type 1 diabetes. We used generalized linear mixed models, adjusting for demographic and clinical variables, to examine differences in glycemic control and care at 1 year.
RESULTS
Of identified individuals, 26.4% had younger-onset and 73.6% had mid-age–onset type 2 diabetes. Adults with younger onset had higher initial mean HbA1c values (8.9% [74 mmol/mol]) than adults with onset in mid-age (8.4% [68 mmol/mol]) (P < 0.0001) and lower odds of achieving an HbA1c <7% (<53 mmol/mol) 1 year after the diagnosis (adjusted odds ratio [aOR] 0.70 [95% CI 0.66–0.74]), even after accounting for HbA1c at diagnosis. Adults with younger onset had lower odds of in-person primary care contact (aOR 0.82 [95% CI 0.76–0.89]) than those with onset during mid-age, but they did not differ in telephone contact (1.05 [0.99–1.10]). Adults with younger onset had higher odds of starting metformin (aOR 1.20 [95% CI 1.12–1.29]) but lower odds of adhering to that medication (0.74 [0.69–0.80]).
CONCLUSIONS
Adults with onset of type 2 diabetes at a younger age were less likely to achieve glycemic control at 1 year following diagnosis, suggesting the need for tailored care approaches to improve outcomes for this high-risk patient population.
Introduction
Type 2 diabetes is no longer exclusively a disease of middle-aged and older adults. Between 1990 and 2009, the number of U.S. adults diagnosed with type 2 diabetes before age 45 more than doubled (1). Mounting evidence suggests that earlier disease onset significantly increases future risk for diabetes-related micro- and macrovascular complications (2–5). Given this higher risk for future complications, the lasting benefits of establishing early glycemic control may be particularly critical for adults with younger-onset type 2 diabetes.
Findings of randomized controlled trials and retrospective cohort studies support the lasting importance of early glycemic control and its potential role in the diverging long-term trajectories of individuals with younger-onset type 2 diabetes (6,7). However, prior studies of adults with younger-onset type 2 diabetes have not focused on the early period following diagnosis—a critical time filled with new challenges and responsibilities. While individuals’ motivation for change may be high, successful disease self-management may be particularly daunting for younger adults (8–10). Knowledge regarding the earliest experiences of individuals with younger-onset type 2 diabetes would inform optimal initial care and self-management support for this high-risk population. In this study, we addressed this gap in the literature by comparing differences in early diabetes-related outcomes and care between adults newly diagnosed with type 2 diabetes at a younger age (21–44 years) and at mid-age (45–64 years).
Research Design and Methods
Study Design and Setting
We conducted a retrospective, longitudinal cohort study using data from the Kaiser Permanente Northern California (KPNC) electronic health record (EHR) to examine differences in 1) baseline demographic, clinical, and care utilization characteristics; 2) disease management outcomes at 1 year following diagnosis; and 3) care received during the year following diagnosis by adults with newly diagnosed younger-onset type 2 diabetes and those with newly diagnosed mid-age–onset type 2 diabetes. This work was approved by the Kaiser Permanente Northern California Institutional Review Board.
Study Population
KPNC is an integrated health care delivery system serving ∼4.3 million socioeconomically and ethnically/racially diverse members. All members are insured and have within-system access to all types of care, including routine preventive care and laboratory tests (e.g., diabetes screening). We used data from KPNC’s EHR to identify adults between 21 and 64 years of age who had no evidence of prior diabetes but who had a first diabetes-defining HbA1c result (the “index HbA1c,” defined as HbA1c ≥6.5% [≥48 mmol/mol]) between 1 January 2010 and 31 December 2016, and who then received a clinical diagnosis of type 2 diabetes (defined as an ICD-9/10 code for diabetes, or the addition of diabetes to the EHR-based problem list) within 3 months of the index HbA1c result. Any ICD-9/10 diagnostic code, problem list code for diabetes, or prescription for a diabetes-related medication (other than metformin) received before the index HbA1c date was considered evidence of prior diabetes. For KPNC members, nearly all medications are dispensed by KPNC pharmacies. We excluded individuals with gestational diabetes mellitus and those with likely type 1 diabetes (identified by using a validated algorithm) (11–13). To ensure we were correctly identifying KPNC members with newly diagnosed type 2 diabetes, rather than new KPNC members with prevalent type 2 diabetes, we required continuous KPNC membership during the year before the index HbA1c. We also excluded individuals who did not remain as KPNC members throughout the year following their clinical diagnosis of type 2 diabetes.
In the resulting cohort, individuals aged 21–44 years at the time of their index HbA1c value were classified as having younger-onset type 2 diabetes, whereas those between 45 and 64 years of age were classified as having mid-age–onset type 2 diabetes. These age thresholds were based on the American Diabetes Association’s recommendation to begin routine diabetes screening at age 45 and on national trends for diabetes incidence (more than half of new diagnoses in the U.S. are given to adults aged 45–64 years) (14,15).
Measures
We examined baseline demographic characteristics (sex, ethnicity/race [those who self-reported Hispanic ethnicity were classified as Latino, and the remaining individuals were classified by self-reported race], insurance type, English proficiency, and neighborhood deprivation index) (16); clinical characteristics (index HbA1c, a preceding diagnosis of prediabetes, prevalence of type 2 diabetes risk factors [hypertension, hyperlipidemia, BMI], and comorbidities [heart disease, renal disease, depression]); prior care utilization (duration of empanelment with a primary care provider [PCP], numbers of encounters with the PCP and missed appointments during the prior year, and activity on the KPNC online patient portal); and diabetes-relevant health behaviors (self-reported weekly exercise and smoking status) (17,18). A preceding prediabetes diagnosis, risk factors, and comorbidities were defined via ICD-9/10 codes.
The primary outcome was glycemic control 1 year after the diagnosis and was assessed by using the HbA1c value obtained closest to the 1-year mark. The American Diabetes Association recommends an HbA1c <7% (<53 mmol/mol) for most nonelderly adults. Failure to achieve this HbA1c target was considered “not at goal,” as was the absence of any follow-up HbA1c measurements during the year after the diagnosis (19). In two sensitivity analyses, we 1) used a goal of HbA1c <8% (<65 mmol/mol) and 2) used the goal of HbA1c <7% (<53 mmol/mol) but included only available (i.e., nonmissing) 1-year values.
Other outcomes included blood pressure (BP) at 1 year, which was determined by using the measurement obtained closest to the 1-year date after diabetes diagnosis; “at goal” was classified as 1) systolic BP <140 mmHg and diastolic BP <90 mmHg, or 2) systolic BP <130 mmHg and diastolic BP <80 mmHg (20). We also assessed measures of diabetes-relevant behavior changes, including weight loss (defined as ≥5% or <5% weight loss, determined by using baseline weight and weight at 1 year), and self-reported weekly exercise (defined as any vs. no physical activity) at 1 year after the diagnosis (17). Finally, we examined differences in care received during the year following diagnosis, specifically 1) interactions with the health care system (in-person visits, telephone visits, and use of the online patient portal), 2) initiation of medication (metformin, sulfonylurea, and insulin), 3) adherence to medication refills (of metformin and sulfonylurea, defined as a ≥80% proportion of days covered), and 4) other diabetes-related care (diabetes-related education, medical nutrition therapy, and retinal exam) (21).
Statistical Analysis
We compared baseline characteristics and outcomes between the groups with younger- and mid-age–onset type 2 diabetes using the χ2 test or an independent two-tailed t test, as appropriate. We compared the prevalence of being at goal (HbA1c <7% [<53 mmol/mol]) at 1 year between the populations with younger and mid-age onset on the basis of their index HbA1c values. Mean index and 1-year HbA1c and BMI values were also described by using more granular (3-year) age groups. Finally, 1-year HbA1c and the secondary outcomes were modeled by using a generalized linear mixed effects regression model that included a logit link and a random intercept for medical facility in order to account for correlation in outcomes among people receiving care at the same KPNC medical facility. All generalized linear mixed effects regression models included as fixed effects sex, ethnicity/race, neighborhood deprivation index, BMI, index HbA1c, and selected preceding diagnoses that may have affected the aggressiveness of diabetes care (hypertension and hyperlipidemia) or individuals’ readiness (prediabetes) or ability (depression) to engage in self-care. All statistical analyses were performed by using SAS software version 9.4.
Results
We identified 32,137 individuals aged 21–64 years with new-onset type 2 diabetes, 26.4% of whom were 21–44 years old at the time of diagnosis (Supplementary Fig. 1). Compared with those with mid-age–onset type 2 diabetes, individuals with younger-onset diabetes were more likely to be men and to be of nonwhite ethnicity/race (Table 1).
Table 1.
Population characteristics at baseline by age at time of type 2 diabetes onset
| Younger onset (21–44 years) (n = 8,496) | Mid-age onset (45–64 years) (n = 23,641) | P value | |
|---|---|---|---|
| Men | 5,336 (62.8) | 12,760 (54.0) | <0.0001 |
| Ethnicity/race* | <0.0001 | ||
| White | 1,848 (22.5) | 8,821 (38.2) | |
| Latino | 2,864 (34.9) | 5,098 (22.1) | |
| Asian | 2,302 (28.0) | 5,483 (23.7) | |
| Black | 739 (9.0) | 2,413 (10.4) | |
| Other | 454 (5.5) | 1,286 (5.6) | |
| Neighborhood deprivation index (quartiles) | <0.0001 | ||
| 1 (Residing in the least deprived neighborhoods in Northern California) | 1,452 (17.3) | 4,704 (20.2) | |
| 2 | 2,136 (25.4) | 6,649 (28.5) | |
| 3 | 2,543 (30.3) | 6,995 (30.0) | |
| 4 (Residing in the most deprived neighborhoods in Northern California) | 2,270 (27.0) | 4,981 (21.4) | |
| Proficient in English | 7,493 (88.4) | 20,834 (88.3) | <0.0001 |
| Insurance plan type | <0.0001 | ||
| Commercial | 8,045 (94.7) | 22,144 (93.7) | |
| Medicaid/Medicare | 401 (4.8) | 1,342 (5.7) | |
| Other | 50 (0.6) | 155 (0.7) | |
| Index HbA1c (%), mean (SD) | 8.9 (2.3) | 8.4 (2.2) | <0.0001 |
| Index HbA1c | <0.0001 | ||
| 6.5 to <7% (48 to <53 mmol/mol) | 2,202 (25.9) | 9,241 (39.1) | |
| 7 to <8% (53 to <64 mmol/mol) | 1,852 (21.8) | 5,431 (23.0) | |
| 8 to <9% (64 to <75 mmol/mol) | 887 (10.4) | 2,127 (9.0) | |
| 9 to <10% (75 to <86 mmol/mol) | 842 (9.9) | 1,498 (6.3) | |
| ≥10% (≥86 mmol/mol) | 2,713 (31.9) | 5,344 (22.6) | |
| BMI (kg/m2), mean (SD) | 36.1 (8.3) | 33.4 (7.1) | <0.0001 |
| BMI strata | <0.0001 | ||
| Underweight/normal (BMI <25 kg/m2) | 408 (5.0) | 1,833 (8.0) | |
| Overweight (BMI 25 to <30 kg/m2) | 1,527 (18.5) | 6,155 (27.0) | |
| Obesity class I (BMI 30–34.9 kg/m2) | 2,175 (26.4) | 6,734 (29.5) | |
| Obesity class II (BMI 35–39.9 kg/m2) | 1,736 (21.1) | 4,203 (18.4) | |
| Obesity class III (BMI ≥40 kg/m2) | 2,388 (29.0) | 3,870 (17.0) | |
| Preceding diagnosis of prediabetes | 2,174 (25.6) | 8,011 (33.9) | <0.0001 |
| Depression | 641 (7.5) | 2,081 (8.8) | <0.0001 |
| Heart disease | 93 (1.1) | 1,027 (4.3) | <0.0001 |
| Chronic kidney disease | 84 (1.0) | 486 (2.1) | <0.0001 |
| Hypertension | 2,209 (26.0) | 11,103 (47.0) | <0.0001 |
| Taking hypertension medication | 1,957 (23.0) | 11,050 (46.7) | <0.0001 |
| Hyperlipidemia | 1,751 (20.6) | 9,121 (38.6) | <0.0001 |
| Taking statin medication | 386 (4.5) | 4,428 (18.7) | <0.0001 |
| Current smoker | 911 (12.5) | 2,267 (11.3) | <0.0001 |
| Any physical activity | 2,848 (50.3) | 7,835 (51.2) | <0.0001 |
| Active on online patient portal | 4,863 (57.2) | 13,134 (55.6) | <0.0001 |
| Empanelment with PCP (years), mean (SD) | 3.9 (3.7) | 5.1 (4.8) | <0.0001 |
| Any visit to the PCP in the prior year | 4,811 (56.6) | 14,683 (62.1) | <0.0001 |
Data are n (%) unless otherwise indicated.
Composite variable was defined by self-reported ethnicity and race, whereby all those responding as having Hispanic ethnicity were categorized as Latino and the remaining individuals were categorized by reported race.
Younger-onset type 2 diabetes was associated with higher mean (SD) index HbA1c values (8.9% [2.3%] [74 mmol/mol (25 mmol/mol)]) than was mid-age–onset diabetes (8.4% [2.2%] [68 mmol/mol (24 mmol/mol)]) (P < 0.0001); 31.9% of individuals with younger-onset diabetes had an index HbA1c ≥10% (86 mmol/mol), whereas 22.6% of those with mid-age–onset diabetes had such an index value (P < 0.0001). The mean (SD) BMI was also higher among individuals with younger-onset diabetes (36.1 [8.3] kg/m2) than among those with mid-age–onset diabetes (33.4 [7.1] kg/m2) (P < 0.0001).
Significantly fewer individuals with younger-onset type 2 diabetes (48.9%) than with mid-age–onset diabetes (61.9%) achieved glycemic control at 1 year (P < 0.0001) (Table 2). The number of individuals with younger-onset diabetes who had a 1-year HbA1c value ≥10% (86 mmol/mol) (7.5%) was more than two times higher than the number of individuals with mid-age–onset diabetes who had such an HbA1c value at 1 year (3.0%) (P < 0.0001). When stratified by index HbA1c value, each category of individuals with younger-onset diabetes had lower rates of achieving glycemic control at 1 year (Fig. 1). Among individuals whose index HbA1c values were <7% (<53 mmol/mol), significantly fewer individuals with younger onset (62.7%) than with mid-age onset (75.0%) remained at goal at the 1-year mark (P < 0.0001).
Table 2.
Early outcomes and initial care received 1 year after type 2 diabetes diagnosis, by age at onset
| Younger onset (21–44 years) | Mid-age onset (45–64 years) | aOR (95% CI)* | P value | |
|---|---|---|---|---|
| Clinical outcomes | ||||
| HbA1c at goal (<7% [<53 mmol/mol]) at 1 year | 48.9 | 61.9 | 0.70 (0.66–0.74) | <0.0001 |
| BP at goal | ||||
| <130/80 mmHg | 47.9 | 48.1 | 0.93 (0.87–0.98) | 0.013 |
| <140/90 mmHg | 87.5 | 86.7 | 0.96 (0.88–1.05) | 0.338 |
| Loss of ≥5% body weight | 25.1 | 27.3 | 0.92 (0.86–0.99) | 0.020 |
| Any physical activity | 59.9 | 59.3 | 1.06 (0.99–1.14) | 0.111 |
| Care interactions | ||||
| Any outpatient encounter with PCP | 80.6 | 84.3 | 0.82 (0.76–0.89) | <0.0001 |
| Any telephone encounter with PCP | 39.9 | 39.5 | 1.05 (0.99–1.11) | 0.113 |
| Active on online patient portal | 75.1 | 70.1 | 1.68 (1.57–1.80) | <0.0001 |
| Received retinal screening | 34.0 | 36.8 | 0.81 (0.76–0.86) | <0.0001 |
| Received diabetes education | 40.0 | 43.8 | 0.85 (0.80–0.90) | <0.0001 |
| Received nutrition therapy | 22.9 | 24.0 | 0.96 (0.90–1.03) | 0.248 |
| Medications | ||||
| Insulin initiated | 12.4 | 11.7 | 1.05 (0.96–1.14) | 0.281 |
| Sulfonylurea initiated | 25.0 | 18.8 | 1.08 (1.00–1.16) | 0.051 |
| Metformin initiated | 70.6 | 59.7 | 1.20 (1.12–1.29) | <0.0001 |
| Metformin adherence (≥80 PDC) | 56.7 | 66.1 | 0.74 (0.69–0.80) | <0.0001 |
| Sulfonylurea adherence (≥80 PDC) | 48.9 | 58.8 | 0.74 (0.66–0.84) | <0.0001 |
Data are percentages unless otherwise indicated. PDC, proportion of days covered.
Multilevel logistic model adjusting for onset group; sex; ethnicity/race; index HbA1c; neighborhood deprivation index; BMI; and diagnosed hypertension, hyperlipidemia, prediabetes, and depression, with a random intercept for medical facility. Mid-age onset is the reference category.
Figure 1.
The proportions of individuals with younger-onset and with mid-age–onset type 2 diabetes from among the total analytic cohort who achieved goal glycemic control (HbA1c <7% [<53 mmol/mol]) at 1 year after diagnosis, by index HbA1c value.
The mean (SD) 1-year HbA1c was 7.2% (1.6%) (55 mmol/mol [18 mmol/mol]) for individuals with younger-onset diabetes and 6.9% (1.2%) (52 mmol/mol [13 mmol/mol]) for the mid‐age–onset group (P < 0.0001). The mean 1-year BMI was also higher among individuals with a younger onset (35.2 [8.0] kg/m2) than among those with mid‐age onset (32.5 [6.9] kg/m2) (P < 0.0001).
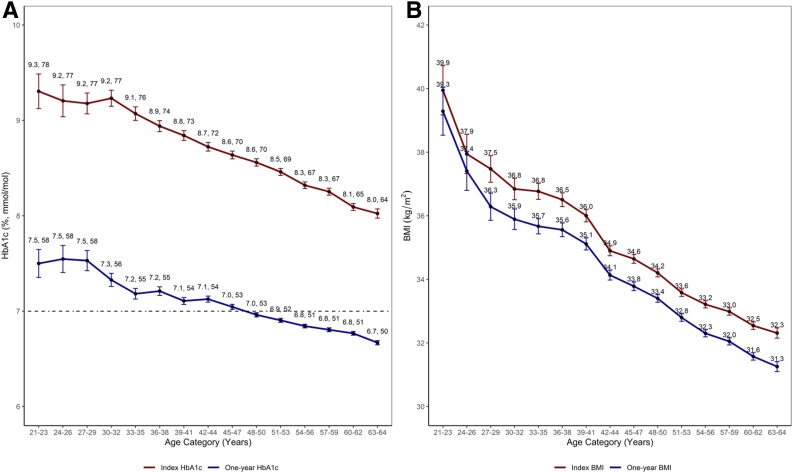
Examining unadjusted trends for index and 1-year HbA1c and BMI values by 3-year age groups, we noted an inverse linear relationship. Increasing age category was associated with lower HbA1c and BMI measurements at both time points (Fig. 2).
Figure 2.
A: Mean index and 1-year HbA1c values by age at diabetes diagnosis (grouped by 3-year age categories). The dashed line indicates an HbA1c value of 7% (53 mmol/mol). B: Mean index and 1-year BMI values by age at diabetes diagnosis (grouped by 3-year age categories).
After adjusting for demographic and clinical characteristics, including index HbA1c, individuals with younger-onset type 2 diabetes had substantially lower odds of being at goal (HbA1c <7% [<53 mmol/mol]) at 1 year after the diagnosis (adjusted odds ratio [aOR] 0.70 [95% CI 0.66–0.74]; P < 0.0001) (Table 2; results of the full model are included in Supplementary Table 1). More individuals with younger-onset diabetes (15.5%) than with mid-age–onset diabetes (11.1%) were missing 1-year follow-up HbA1c results (P < 0.0001). In sensitivity analyses, the odds of achieving glycemic control remained significantly lower for individuals with younger onset than for those with mid-age onset when using an HbA1c threshold of <8% (<64 mmol/mol) (67.9% vs. 79.3%, respectively) (aOR 0.68 [95% CI 0.63–0.72]; P < 0.0001) and when analyzing only nonmissing 1-year HbA1c values (57.9% vs. 69.6%, respectively) (aOR 0.73 [95% CI 0.68–0.77]; P < 0.0001).
Individuals with younger-onset diabetes had lower odds (25.1%) than those with mid-age–onset diabetes (27.3%) of losing at least 5% body weight during the 1st year (aOR 0.92 [95% CI 0.86–0.99]; P = 0.020) (Table 2). Regarding PCP contact during early care, individuals with younger-onset diabetes had lower odds (80.6%) than those with mid-age–onset diabetes (84.3%) of having at least one in-person PCP visit after the diagnosis (aOR 0.82 [95% CI 0.76–0.89]; P < 0.0001). The two groups had a similar likelihood of having at least one telephone visit with the PCP, but those with younger onset had higher odds of being active on the online patient portal after diagnosis (Table 2).
Individuals with younger-onset type 2 diabetes were more likely than those with mid-age–onset type 2 diabetes to begin taking metformin (70.6% vs. 59.7%, respectively) (aOR 1.20 [95% CI 1.12–1.29]; P < 0.0001). However, the odds of adherence to metformin were significantly lower for individuals with younger onset (56.7%) than for those with mid-age onset (66.1%) (aOR 0.74 [95% CI 0.69–0.80]; P < 0.0001).
Conclusions
We found that among members of a large, integrated health care delivery system, adults diagnosed with type 2 diabetes between the ages of 21 and 44 (younger onset) had higher initial HbA1c levels and a much lower likelihood of achieving glycemic control at 1 year following diagnosis than those diagnosed between 45 and 64 years of age (mid-age onset). Although mean HbA1c values decreased after diagnosis for all of the examined 3-year age groups, the mean 1-year HbA1c values remained >7% (>53 mmol/mol) for all groups younger than 45 years (Fig. 2). Individuals with younger-onset type 2 diabetes also had less in-person contact with their PCP and lower adherence to metformin during the year after their type 2 diabetes diagnosis. Tailored treatment strategies that address the more severe hyperglycemia and obesity seen at diagnosis and the unique treatment barriers present among this higher-risk younger-onset population are needed in order to support timely achievement of recommended treatment goals that are associated with improved long-term outcomes (4,22–24).
Younger adults may experience distinct psychosocial barriers to optimal early type 2 diabetes management. For example, earlier age at diagnosis has been associated with a higher prevalence of depression, greater stress, poorer diet, lower medication adherence, lower diabetes self-efficacy, and less time to spend on self-care because of work and family demands (25–29). Previous evidence suggests that age-related differences in glycemic control are likely to persist beyond the initial period following diagnosis (22,30). While inherent differences in the physiology of type 2 diabetes and longer exposure to the disease contribute to the increased risk of disease-related complications among people with younger-onset type 2 diabetes, suboptimal glycemic control starting from the time of diagnosis may also fuel this population’s vulnerability to adverse type 2 diabetes–related outcomes.
We found differences in the modality of primary care contact following diagnosis (e.g., in person, by phone, or via the online patient portal) that suggest that traditional approaches and resources for delivering care may not be appropriately designed to meet the needs and preferences of younger adults with type 2 diabetes. Adults with younger onset in our study were more active users of the online patient portal and had less in-person contact with their PCP during the year after their type 2 diabetes diagnosis. These results suggest that technology-based tools may provide more optimal opportunities for PCP interaction and diabetes self-management support that fit better with younger adults’ schedules (i.e., not restricted to times during the workday). Studies have demonstrated that patients from ethnic/racial minorities frequently use smartphone technology and the internet for gathering health-related information or interacting with health systems (31). This trend may be particularly relevant to individuals with younger-onset type 2 diabetes, who were more likely to identify as Latino or Asian than individuals with mid-age–onset diabetes.
Although people in the younger-onset type 2 diabetes group had higher odds of initiating metformin, 29% were still not prescribed this medication during the year after diagnosis. Even among those who did start metformin, almost half inadequately adhered to this medication. Optimizing early metformin treatment in this younger-onset population could help many newly diagnosed individuals achieve the level of early glycemic control associated with lasting reductions in risks for macrovascular complications and mortality (6). Indeed, initiation of metformin early, when HbA1c values may still be low, may help preserve β-cell function, prolonging the effectiveness of metformin and decreasing the risk of future disease-related complications (32). There are likely unique, incompletely understood barriers to metformin initiation and early adherence for younger adults, including greater difficulty coping with a type 2 diabetes diagnosis (e.g., it does not fit with their personal model of health), an aversion toward medications or a preference for “natural approaches,” or present-future bias (the natural tendency to value the present “costs” of a task [i.e., starting a new daily medication] over future or long-term benefits). A better understanding of these barriers to early metformin treatment can help inform approaches for early care for type 2 diabetes in this population.
Our results must be interpreted within the context of the study design. First, eligible individuals were all members of a single health care system, potentially limiting the generalizability of the findings to other settings. Past work has demonstrated that the demographic characteristics of and prevalence of type 2 diabetes among KPNC members are largely representative of the general population and insured populations in Northern California (33). Further, given national trends showing that younger onset of type 2 diabetes is associated with a higher likelihood of not meeting type 2 diabetes treatment goals (34), and given KPNC’s record of above-average type 2 diabetes care outcomes (35), it is unlikely that our findings can be simply attributed to poor care provided to this population within KPNC. Second, we focused on individuals who received a “timely” diagnosis of type 2 diabetes (within 3 months of the index HbA1c result) and therefore excluded individuals whose clinical diagnosis may have been substantially delayed despite the HbA1c result. Future research will be needed in order to address the factors related to delayed or missed diagnoses. Third, we were unable to examine insulin adherence. However, only 12.4% of people with younger-onset diabetes and 11.7% of people with mid-age–onset diabetes were prescribed insulin during the 1st year. Finally, this study cannot address causation. We can only comment on observed associations between age at type 2 diabetes onset and the examined outcomes.
There are myriad potential contributors to the observed differences in early glycemic control between individuals with younger onset and those with mid-age onset of type 2 diabetes, including some that are not modifiable. Still, the higher likelihood of inadequate early control among younger adults suggests that our current approaches to caring for this population are inadequate. Our findings speak to the need for novel, tailored strategies to better support this distinct, high-risk population of patients. Given that achieving early glycemic control has been linked to better health and lower mortality years into the future, such care approaches should be proactive and designed to achieve HbA1c goals early, rather than to react to persistently high HbA1c values or—even worse—the onset of disease-related complications. Important next steps for this line of research are to gain further insights into why individuals with younger-onset type 2 diabetes are at higher risk for poor early control, to define barriers to and facilitators of successful early type 2 diabetes management, and to develop and test novel care strategies for tailoring initial care and improving outcomes for this growing population.
Supplementary Material
Article Information
Funding. A.G. has been supported by a grant from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) (K23DK116968) and a Kaiser Permanente Northern California Community Benefit Award. R.W.G. has received support from an NIDDK grant (K24DK109114).
The funders had no role in the design and conduct of the study, the completion of the analysis, the interpretation of the data, or the content and preparation of the manuscript.
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. All authors have met the requirements for authorship. A.G. oversaw the study design, data analysis, and results interpretation and wrote the manuscript. P.M. designed the study, performed all programming and data analysis, and wrote and reviewed the manuscript. S.E.A. planned and directed the statistical analysis and interpretation of the results, and wrote and reviewed the manuscript. M.A.B. reviewed the manuscript and constructed the tables and figures. E.K. and A.M. contributed clinical expertise and reviewed the manuscript. A.J.K. contributed scientific expertise during the review and editing of the manuscript. R.W.G. designed the study and reviewed and edited the manuscript. A.G. is the guarantor of this work and, as such, has full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Prior Presentation. Parts of this study were presented in poster form at the 79th Scientific Sessions of the American Diabetes Association, San Francisco, CA, 7–11 June 2019.
Footnotes
This article contains Supplementary Data online at https://care.diabetesjournals.org/lookup/suppl/doi:10.2337/dc19-1380/-/DC1.
This article is featured in a podcast available at https://www.diabetesjournals.org/content/diabetes-core-update-podcasts.
References
- 1.Centers for Disease Control and Prevention United States Diabetes Surveillance System: diagnosed diabetes [Internet]. Available from https://gis.cdc.gov/grasp/diabetes/DiabetesAtlas.html. Accessed 22 October 2019.
- 2.Hillier TA, Pedula KL. Complications in young adults with early-onset type 2 diabetes: losing the relative protection of youth. Diabetes Care 2003;26:2999–3005 [DOI] [PubMed] [Google Scholar]
- 3.Eppens MC, Craig ME, Cusumano J, et al. Prevalence of diabetes complications in adolescents with type 2 compared with type 1 diabetes. Diabetes Care 2006;29:1300–1306 [DOI] [PubMed] [Google Scholar]
- 4.Wilmot E, Idris I. Early onset type 2 diabetes: risk factors, clinical impact and management. Ther Adv Chronic Dis 2014;5:234–244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wong J, Molyneaux L, Constantino M, Twigg SM, Yue DK. Timing is everything: age of onset influences long-term retinopathy risk in type 2 diabetes, independent of traditional risk factors. Diabetes Care 2008;31:1985–1990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 2008;359:1577–1589 [DOI] [PubMed] [Google Scholar]
- 7.Laiteerapong N, Ham SA, Gao Y, et al. The legacy effect in type 2 diabetes: impact of early glycemic control on future complications (the Diabetes & Aging Study). Diabetes Care 2019;42:416–426 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schneider KL, Andrews C, Hovey KM, et al. Change in physical activity after a diabetes diagnosis: opportunity for intervention. Med Sci Sports Exerc 2014;46:84–91 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Keenan PS. Smoking and weight change after new health diagnoses in older adults. Arch Intern Med 2009;169:237–242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gorin AA, Phelan S, Hill JO, Wing RR. Medical triggers are associated with better short- and long-term weight loss outcomes. Prev Med 2004;39:612–616 [DOI] [PubMed] [Google Scholar]
- 11.Makam AN, Nguyen OK, Moore B, Ma Y, Amarasingham R. Identifying patients with diabetes and the earliest date of diagnosis in real time: an electronic health record case-finding algorithm. BMC Med Inform Decis Mak 2013;13:81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Klompas M, Eggleston E, McVetta J, Lazarus R, Li L, Platt R. Automated detection and classification of type 1 versus type 2 diabetes using electronic health record data. Diabetes Care 2013;36:914–921 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schroeder EB, Donahoo WT, Goodrich GK, Raebel MA. Validation of an algorithm for identifying type 1 diabetes in adults based on electronic health record data. Pharmacoepidemiol Drug Saf 2018;27:1053–1059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Centers for Disease Control and Prevention National Diabetes Statistics Report, 2017 [Internet], 2017. Atlanta, GA, US Department of Health and Human Services, CDC. Available from https://www.cdc.gov/diabetes/data/statistics-report/index.html. Accessed 22 October 2019.
- 15.American Diabetes Association 2. Classification and diagnosis of diabetes: Standards of Medical Care in Diabetes—2019. Diabetes Care 2019;42(Suppl. 1):S13–S28 [DOI] [PubMed] [Google Scholar]
- 16.Messer LC, Laraia BA, Kaufman JS, et al. The development of a standardized neighborhood deprivation index. J Urban Health 2006;83:1041–1062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Grant RW, Schmittdiel JA, Neugebauer RS, Uratsu CS, Sternfeld B. Exercise as a vital sign: a quasi-experimental analysis of a health system intervention to collect patient-reported exercise levels. J Gen Intern Med 2014;29:341–348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Karter AJ, Parker MM, Moffet HH, et al. Missed appointments and poor glycemic control: an opportunity to identify high-risk diabetic patients. Med Care 2004;42:110–115 [DOI] [PubMed] [Google Scholar]
- 19.American Diabetes Association 6. Glycemic targets: Standards of Medical Care in Diabetes—2019. Diabetes Care 2019;42(Suppl. 1):S61–S70 [DOI] [PubMed] [Google Scholar]
- 20.American Diabetes Association 9. Cardiovascular disease and risk management: Standards of Medical Care in Diabetes—2018. Diabetes Care 2018;41(Suppl. 1):S86–S104 [DOI] [PubMed] [Google Scholar]
- 21.Hofmeyer BA, Look KA, Hager DR. Refill-based medication use quality measures in kidney transplant recipients: examination of proportion of days covered and medication possession ratio. J Manag Care Spec Pharm 2018;24:367–372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Berkowitz SA, Meigs JB, Wexler DJ. Age at type 2 diabetes onset and glycaemic control: results from the National Health and Nutrition Examination Survey (NHANES) 2005-2010. Diabetologia 2013;56:2593–2600 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Naranjo DM, Jacobs EA, Fisher L, Hessler D, Fernandez A. Age and glycemic control among low-income Latinos. J Immigr Minor Health 2013;15:898–902 [DOI] [PubMed] [Google Scholar]
- 24.El-Kebbi IM, Cook CB, Ziemer DC, Miller CD, Gallina DL, Phillips LS. Association of younger age with poor glycemic control and obesity in urban african americans with type 2 diabetes. Arch Intern Med 2003;163:69–75 [DOI] [PubMed] [Google Scholar]
- 25.Toobert DJ, Hampson SE, Glasgow RE. The summary of diabetes self-care activities measure: results from 7 studies and a revised scale. Diabetes Care 2000;23:943–950 [DOI] [PubMed] [Google Scholar]
- 26.Zhao W, Chen Y, Lin M, Sigal RJ. Association between diabetes and depression: sex and age differences. Public Health 2006;120:696–704 [DOI] [PubMed] [Google Scholar]
- 27.Hilliard ME, Yi-Frazier JP, Hessler D, Butler AM, Anderson BJ, Jaser S. Stress and A1c among people with diabetes across the lifespan. Curr Diab Rep 2016;16:67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.American Diabetes Association; Professional Practice Committee Professional practice committee for the Standards of Medical Care in Diabetes—2016. Diabetes Care 2016;39(Suppl. 1):S107–S108 [DOI] [PubMed] [Google Scholar]
- 29.Hessler DM, Fisher L, Mullan JT, Glasgow RE, Masharani U. Patient age: a neglected factor when considering disease management in adults with type 2 diabetes. Patient Educ Couns 2011;85:154–159 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hillier TA, Pedula KL. Characteristics of an adult population with newly diagnosed type 2 diabetes: the relation of obesity and age of onset. Diabetes Care 2001;24:1522–1527 [DOI] [PubMed] [Google Scholar]
- 31.Khoong EC, Le GM, Hoskote M, Rivadeneira NA, Hiatt RA, Sarkar U. Health information-seeking behaviors and preferences of a diverse, multilingual urban cohort. Med Care 2019;57(Suppl. 6 Suppl. 2):S176–S183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Brown JB, Conner C, Nichols GA. Secondary failure of metformin monotherapy in clinical practice. Diabetes Care 2010;33:501–506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gordon NP. Similarity of the adult Kaiser Permanente membership in Northern California to the insured and general population in Northern California: statistics from the 2011 California Health Interview Survey [Internet], 2015. Available from https://divisionofresearch.kaiserpermanente.org/projects/memberhealthsurvey/SiteCollectionDocuments/chis_non_kp_2011.pdf. Accessed 21 April 2017.
- 34.Kazemian P, Shebl FM, McCann N, Walensky RP, Wexler DJ. Evaluation of the cascade of diabetes care in the United States, 2005-2016. JAMA Intern Med. 12 August 2019 [Epub ahead of print]. DOI: 10.1001/jamainternmed.2019.2396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rana JS, Karter AJ, Liu JY, Moffet HH, Jaffe MG. Improved cardiovascular risk factors control associated with a large-scale population management program among diabetes patients. Am J Med 2018;131:661–668 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.