Highlights
-
•
Porcine intestinal epithelial cells could be infected by TGEV successfully, and TGEV propagated in IECs remain infectious for ST cells.
-
•
TGEV entered the target cells by membrane fusion and mature viruses budded into vacuoles, and gradually transported to the cell membrane.
-
•
Proliferative rule of TGEV in target cells is revealed.
Keywords: Transmissible gastroenteritis virus, Morphogenesis, Porcine intestinal epithelial cells
Abstract
To gain a better understanding of the replication, proliferation and infection characteristics of porcine transmissible gastroenteritis virus (TGEV) in porcine intestinal epithelial cells (IECs), this study established a cell model of IECs infected with the Chongqing (CQ) strain of TGEV. The morphogenesis and proliferative rule of TGEV in porcine IECs were investigated using transmission electron microscopy, indirect immunofluorescence assays and real-time fluorescence quantitative PCR. Observations under the TEM indicated that the enveloped viral particles were roughly spherical, with diameters of between 80 and 120 nm. The virions entered porcine IECs by membrane fusion and the mature viruses in the vacuoles were transported to the cell membrane before release. The results also showed that from 0 to 12 h after TGEV infection of porcine IECs, the intracellular viral RNA content did not change significantly. Logarithmic growth occurred from 12 to 36 h, after which it gradually decreased. Moreover, the extracellular RNA content began to rise at 24 h after inoculation and then reduced gradually at approximately 48 h. This study provided a theoretical foundation for further study on the infection characteristics of TGEV in target cells.
1. Introduction
Transmissible gastroenteritis virus (TGEV), a member of the Coronaviridae, is the etiological agent of transmissible gastroenteritis (TGE), which is characterized by severe diarrhea, vomiting and dehydration in approximately 2-week-old suckling piglets (Goodwin and Jennings, 1985, Mullan et al., 1994). TGE results in serious economic losses in swine-producing areas worldwide. TGEV has a large, 28.5 kb single-stranded positive sense RNA genome, which comprises four structural proteins encoded by the spike (S), membrane (M), envelope (E), and nucleoprotein (N) genes (Lai and Cavanagh, 1997, Eleouet et al., 1995). Many cell types are susceptible to TGEV, including pig thyroid cells, swine testis cells (ST), pig kidney cells (PK-15, IBRS-2) and salivary gland cells (Nguyen et al., 1987, Haelterman and Hutchings, 1956). IBRS-2, PK-15 and ST cell lines are used commonly to culture TGEV in the laboratory (Chen et al., 1993). In vivo, TGEV replicates in intestinal epithelial cells of susceptible animals, causing cellular degeneration, villus shortening, malabsorption and diarrhea (Haelterman, 1972, Schwegmaan-wessels and Herrler, 2006).
Most studies concerning TGEV pathogenesis in swine have been performed in animal infection models. Only rarely have in vitro studies been used with cell lines originating from porcine intestines. Cell lines IPEC-1, IPEC-J2 and IPI-2I have been used in in vitro model systems (Fangning et al., 2010, Valentina et al., 2009, Peter et al., 2006). However, data concerning these cell lines, and the morphogenesis and proliferation of the virus in the target cells, are scarce. Thus, we developed an in vitro model based on porcine intestinal epithelial cells (IEC) infected with TGEV, and used transmission electron microscopy (TEM), indirect immunofluorescence assay (IFA) and real-time fluorescence quantitative PCR (FQ-PCR) to investigate the infection mechanism of TGEV. TGEV N genes are highly homologous and are essential for replication. The N gene encodes the nucleocapsid protein, which is a highly basic protein with a molecular mass ranging from 40 to 63 kDa, depending on the species and strain (Escors et al., 2001). The N protein’s function involves cessation of cell division and extension of the cell cycle to provide more suitable conditions for virus assembly (Liu et al., 2001 Motokawa et al., 1996, Kapke and Brian, 1986). This is especially significant when the viral life cycle is longer than that of the host cell cycle (Wurm et al., 2001). Therefore, to analyze the TGEV proliferation in target cells, a one-step growth curve was constructed according to the FQ-PCR data for the transcription dynamics of the N gene.
2. Materials and methods
2.1. Sources of cells and virus strains
Porcine intestinal epithelial cells (IECs) were developed in our institute, and were isolated from the jejunum of a newborn piglet (unpublished data). TGEV Chongqing (CQ) strain was isolated from sick piglets with symptoms of diarrhea (Song et al., 2015).
2.2. Reagents
An anti-TGEV-NP mouse monoclonal antibody was produced in our laboratory. Anti-Rabbit IgG/Cy3, Anti-mouse IgG/FITC and 4′,6′-diamidino-2-phenylindole (DAPI) were purchased from Sigma (USA). Porcine IECs were grown in Dulbecco’s modified eagle’s medium (DMEM)/F-12 medium (GIBCO) with 10% fetal bovine serum (FBS, GIBCO BRL) and maintained in maintenance medium (DMEM/F-12 supplemented with 2% FBS) in a 5% CO2 incubator.
2.3. Immunofluorescence assay (IFA)
Porcine IECs were seeded on cover slips and grown to 80% confluence. TGEV (2 × 106 TCID50) was then inoculated into the cells. After culture for 3, 6, 12, 24 and 48 h, respectively, cells were fixed with 3% paraformaldehyde for 20 min, the cover slips were washed three times with phosphate buffered saline (PBS), and then permeabilized using 0.1% Triton X-100 for 5 min at room temperature (RT). The primary antibodies, diluted in 1% bovine serum albumin, were incubated with the coverslips for 1 h at RT in a humid chamber, followed by washing three times with PBS. The secondary antibodies were then incubated with coverslips for 45 min at RT. After the last incubation step, the cover slips were washed three times with PBS and twice with distilled water to remove precipitates. The coverslips were embedded in 10% polyvinyl-alcohol (Mowiol) and stored at 4 °C until examination under a fluorescence microscope.
To detect whether TGEV propagated in porcine IECs cells could infect ST cells, ST cells were infected by the supernatant of IECs infected with TGEV for 24 h. A monoclonal antibody against the TGEV nucleoprotein (NP), at a 1:200 dilution, was used to detect infected cells. The secondary antibody comprised FITC-labeled anti-mouse IgG at a 1:200 dilution. An IFA was then performed using the method mentioned above.
To demonstrate apoptosis, IECs were infected with TGEV for 3, 6, 12, 24 and 48 h, and an apoptosis was detected using an anti-caspase-3 antibody. The rabbit anti-caspase-3 antibody was used at a 1:300 dilution, followed by incubation with a Cy3-labeled anti rabbit IgG as the secondary antibody at a dilution of 1:300. Cell nuclei were stained by incubation with DAPI for 20 min 37 °C. An IFA was then performed using the method mentioned above.
2.4. Cell preparation for TEM
At 3, 12, and 24 h post-infection, infected and mock-infected cells were harvested and washed with PBS (0.15 mol L−1, pH 7.2) twice, and fixed with 25 g L−1 glutaraldehyde at 4 °C for 2 h. The cells were scraped from the glass surface and centrifuged at 3000 × g for 10 min. The supernatant was then discarded, and the cell pellet was resuspended in 20 g L−1 low melting agarose at 37 °C and centrifuged at 5000 × g for 10 min. Samples were post-fixed in 10 g L−1 aqueous OsO4. After stepwise dehydration in acetone, the samples were embedded in epoxy resin 618 and polymerized at 80 °C for 3 days. The embedded cells were cut into 50-nm slices and stained with uranyl acetate and lead citrate for subsequent examination using TEM (Hitachi H7500; Hitachi, Tokyo, Japan).
2.5. Proliferation characteristics of TGEV in vitro
2.5.1. Oligonucleotide primers
A pair of primers to amplify the N gene of TGEV was designed according to the published sequences obtained from GenBank at the National Center for Biotechnology Information (NCBI) website. The primer sequences were: sense primer: 5′-TTCAACCCCATAACCCTCCAACAA-3′, and antisense primer: 5′-GGCCCTTCACCATGCGATAGC-3′. Sangon Biotech (Shanghai, China) Co., Ltd synthesized the primers.
2.5.2. Amplification of the N gene
Total RNA was isolated from infected porcine IECs using the TRIZOL regent (Takara Biotechnology (Dalian) Co., Ltd.), according to the manufacturer’s protocol. The following reaction mixture was used to generate cDNAs: MgCl2 2 μL, 10 × RT Buffer 1 μL, RNase Free dH2O 4.75 μL, dNTP Mixture 1 μL, RNase Inhibitor 0.25 μL, AMV Reverse Transcriptase 0.5 μL, Random 9-mer 0.5 μL, antisense primer 0.5 μL and RNA 1 μL. The reaction conditions were: 30 °C for 10 min, 42 °C for 30 min, 95 °C for 5 min, 5 °C for 5 min. The PCR amplification reaction comprised forward primer 1.0 μL, reverse primer 1.0 μL, Ex Taq HS 0.5 μL, 5 × PCR buffer 10 μL, cDNA template 0.5 μL and 37 μL of distilled water for a total volume of 50 μL. The PCR conditions were: 94 °C for 2 min, followed by 30 cycles of DNA amplification (30 s at 94 °C, 30 s at 55 °C, and 1 min at 72 °C) and a 10 min incubation at 72 °C. The PCR products were cloned into the pMD18-T vector (Takara Biotechnology) and sequenced using the classical Sanger dideoxy sequencing method (Takara Biotechnology).
2.5.3. Preparation of a standard sample
The concentration of the recombinant plasmid was determined using a micro-spectrophotometer. The copy number of the plasmid was calculated and the plasmid was then subjected to a 10-fold serial dilution (from 10−1 to 10−7 copies/μL) for use as the standard template in real-time PCR method.
2.5.4. Real-time PCR amplification and establishment of a standard curve
The standard samples were amplified using real-time PCR using the following reaction mixture: ddH2O 8 μL, SYBR Premix Ex Taq II 10 μL, specific primers 0.5 μL and DNA 1 μL. The reaction conditions were: 39 cycles of 95 °C for 30s, 95 °C for 5 s and 54 °C 30s. One high-concentration and one low-concentration plasmid template were selected for three repeats. The average value was then calculated, which permitted the construction of the kinetic curve and the standard curve.
2.5.5. The one-step growth curve of TGEV
Porcine IECs were infected with TGEV CQ strain, and the supernatant and cells were collected separately at different time points (0, 2, 4, 6, 8, 12, 24, 36, 48 and 72 h). The total RNA in the different samples was extracted and used to produce cDNA as a template for quantitative PCR. The PCR reaction comprised: ddH2O 8 μL, SYBR Premix Ex Taq II 10 μL, specific upstream primer 0.5 μL, specific downstream primer 0.5 μL and cDNA template 1 μL. The reaction conditions were: 39 cycles of 95 °C for 30 s, 95 °C for 5 s and 54 °C for 30 s. The reaction for each sample was repeated three times and a negative control was included. The copy numbers of the gene were obtained from the test sample and used to construct a one-step growth curve for TGEV.
3. Results
3.1. Detection of TGEV replication in porcine IECs
An indirect immunofluorescence technique was used to observe the distribution of TGEV in porcine IECs at different time points. Cell nuclei stained by DAPI appeared blue, and the cytoplasm of the control group did not generate specific fluorescence compared with the treated cells (Fig. 1 A). After 3 h, 6 h and 12 h of infection, some green fluorescence was observed (Fig. 1B, 1C, 1D). At 24 h after treatment, a large amount of green fluorescence was noted close to the DAPI-stained cell nuclei (Fig. 1E). At 48 h, the green fluorescence had decreased, although the nuclear fluorescence remained clearly visible (Fig. 1F).
Fig. 1.
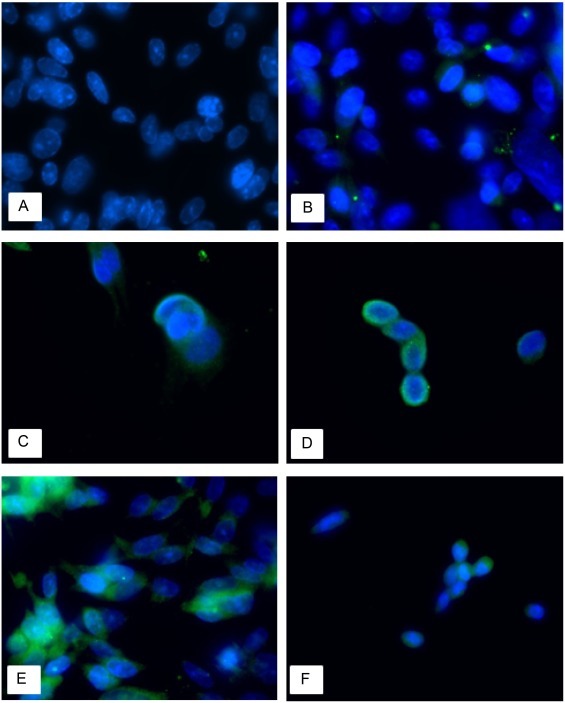
The replication of TGEV in porcine IECs (1000×).
To determine whether ST cells could be infected by TGEV propagated in IECs, the growth of TGEV in ST cells infected by the culture supernatant of IECs was determined. The IFA results showed that ST cells were susceptible to infection by TGEV propagated in IECs (Fig. 2 ).
Fig. 2.
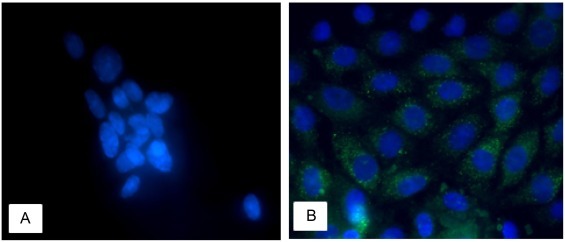
Fluorescence microscopy showing ST cells infected with TGEV (1000×).
In addition, the apoptotic effect of TGEV from IECs was assessed using an anti-caspase-3 antibody. As time increased, more caspase-3-positive cells were observed (Fig. 3 ), indicating that TGEV induced apoptosis in IECs.
Fig. 3.
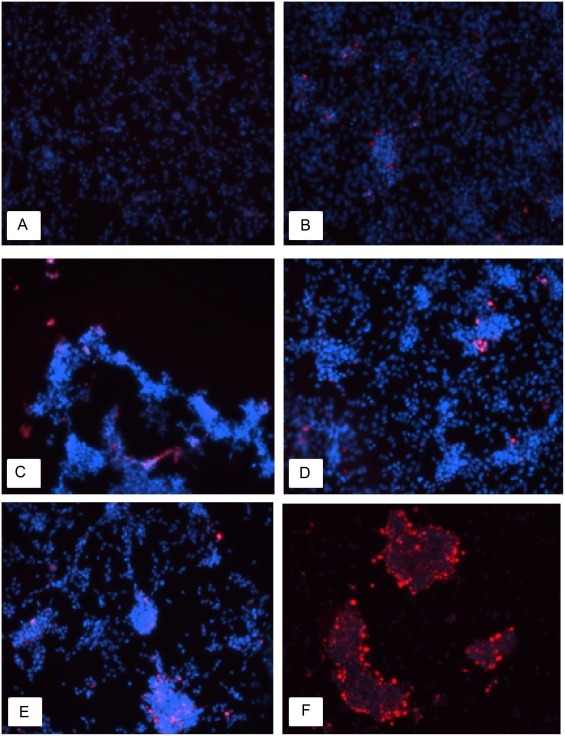
Immunofluorescent staining of apoptotic cells (200×).
3.2. Morphogenesis of the TGEV virion
To confirm TGEV infection of the target cells, the morphogenesis of the TGEV virions was observed under TEM. At 3 h after infection, virions with high electron density were observed attached to the membrane surface (Fig. 4 B); however, very few viruses appeared in the cytoplasm or vacuoles (Fig. 4C). At 12 h after inoculation, the cytoplasm was filled with vacuoles containing TGEV particles (Fig. 4D, E). Fig. 4F shows that the viral envelope was obtained by budding into vacuoles. Virions in vacuoles were transported to the cell membrane (Fig. 4G), with which they fused and then released into the extracellular space via exocytosis (Fig. 4H). At the later stage (24 h post-infection), the Golgi complex became disaggregated (Fig. 4I).
Fig. 4.
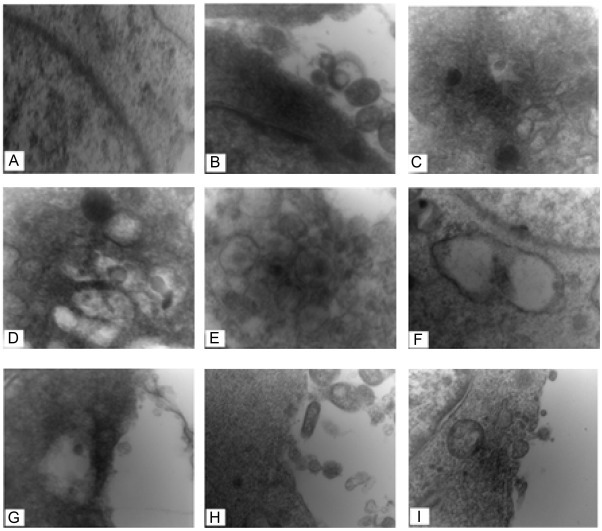
Morphogenesis of TGEV in experimentally infected porcine IECs (80000×).
A. Uninfected porcine IECs B. Virions attached to the cytoplasmic membrane (3 h post-infection); C. A few progeny virions were observed in the vacuole (3 h post-infection); D, E. Most of viruses had entered the vacuole (12 h post-infection); F. Progeny virions budding into the cytoplasmic vacuole (24 h post-infection); G. Progeny mature virions preparing to be released from porcine IECs by exocytosis (24 h post-infection); H. Mature virions released into the extracellular space (24 h post-infection); I. The Golgi complex was disaggregated because of the virus infection (24 h post-infection).
3.3. The proliferative rule of TGEV in porcine IECs
TGEV CQ strain was used to infect porcine IECs, and the supernatant and cells were collected at different time points for FQ-PCR. The one-step growth curve of TGEV was plotted based on amount of RNA for the TGEV N gene in different samples. The resulting growth curve (Fig. 5 ) showed that the amount of intracellular TGEV RNA did not change substantially during the first 12 h post-infection. From 12 h–36 h after infection, intracellular viral RNA increased logarithmically, after which its proliferation slowed down. From 0h–24 h post-infection, the extracellular TGEV RNA content did not change significantly. Thereafter, the viral RNA level exhibited logarithmic growth, reaching a maximum at 36 h.
Fig. 5.
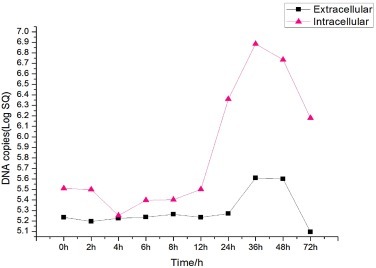
The one step growth curve of TGEV in porcine IECs.
4. Discussion
TGEV can proliferate in many kinds cultured cells, especially in ST cells and PK-15 cells (Ren et al., 2008). After infection by TGEV for 36 h, ST cells showed a typical cytopathic effect, characterized by shrinkage and rounding, and a gradual weakening of their adherence ability: ultimately, the majority of the cells were floating in the medium (Yin et al., 2004). Sirinarumitr et al. (1998) found that TGEV could induce apoptosis during infection of ST cells. Nuclear chromatin condensation and fragmentation, as well as cell shrinkage and cytoplasmic vacuolation, were observed in PK-15 cells infected by TGEV.
This study showed that TGEV’s infection of porcine IECs has a partial cytopathic phase, characterized by cells changing to a fusiform shape, pyknosis, refraction and activation of caspase 3. However, the cytopathic effect in IECs was not as obvious as that in ST or PK15 cells. TGEV could induce porcine IECs to undergo apoptosis, which is consistent with the results of Tong et al. (2011). TGEV-induced apoptosis of porcine IECs might function via blocking cell-cycle transfer from G1 to S phase; however, its specific mechanism requires further exploration.
Indirect IFA extends the application range of an antibody and further improves the detection sensitivity by labeling the specific secondary antibodies with fluorescein for signal amplification. In this study, both infected and uninfected cell cultures were detected by IFA using TGEV antiserum. The results revealed the distribution of TGEV in porcine IECs clearly. At 6 h post-infection, many TGEV particles bound to goat anti-rabbit IgG-FITC showed green fluorescence when exposed to laser light and were located in the cytoplasm. At 24 h, the fluorescence transferred to the perinuclear area, before decreasing gradually at 48 h. Porcine aminopeptidase N (pAPN), a cellular receptor for TGEV, is abundantly expressed in the membrane of porcine IECs (data not shown), which suggested that pAPN plays a role in mediating TGEV infection. In addition, we demonstrated that ST cells could be infected with the culture supernatant of IECs infected TGEV at 24 h. Taken together, these data indicated that IECs, which are derived from a target tissue for porcine coronaviruses, could be infected by TGEV successfully, and that TGEV propagated in IECs remains infectious for ST cells.
The TGEV S protein is involved in the viral invasion process (Trincone and Schwegmaan-wessels, 2015). Combined with pAPN, the S protein mediates the fusion of TGEV and the cell membrane, such that nuclear capsid is released into the cell (Gelhaus et al., 2014). A previous report also suggested that viral genome and structural proteins were assembled into virus particles in the area between the endoplasmic reticulum and the Golgi, and that the particles were transported to the extracellular space through budding of vesicles (Corse and Machamer, 2000, Lili and Masters, 2010). Until now, the morphology of TGEV in target cells had not explored in detail. In this study, TGEV particles in porcine epithelial cells were observed to have a diameter ranging from 80 to 120 nm, which was consistent with previous reports. The infectious virions entered the target cells by membrane fusion and mature viruses budded into vacuoles, which were gradually transported to the cell membrane before being released.
FQ-PCR was used to quantify TGEV inside porcine IECs in comparison with the SC-TGEV virus inside PK-15 cells (Dai et al., 2012). The one-step growth curve of TGEV in porcine IECs showed that the amount of viral RNA did not change significantly from 0 to 12 h post-infection, whereas Dai et al. showed that in PK-15 cells, the RNA increased rapidly from 2 to 8 h. The viral RNA displayed a logarithmic increase in porcine IECs from 12 to 36 h, while Dai et al. showed that the viral RNA was maintained at high level and entered into stationary phase gradually. Extracellular TGEV RNA levels were unchanged at 0 to 24 h post-infection of porcine IECs, and increased logarithmically until reaching maximum at 36 h, before decreasing gradually. In PK-15 cells, Dai et al. demonstrated that extracellular TGEV RNA levels increased logarithmically at 6–28 h post-infection, and maintained relatively stable and high level at 28–32 h. The differences between the curves indicated that differences in virulence and cell types might lead to changes in virus replication. The one-step growth curve of TGEV revealed the rule of virus proliferation, and thus will provide further guidance for clarifying the infection mechanism of TGEV.
In conclusion, this is the first study to report the proliferative rule and morphogenesis of TGEV in porcine IECs. These results lay the foundation for further study of the infection characteristics between TGEV and its target cells.
Acknowledgements
This work was supported by the Graduate Scientific Research in Chongqing (CYS14057, CYS2015076), and the Chongqing Basic Research Program (cstc2014jcyjA80015, cstc2016jcyA0235), and the Fundamental Research Funds for the Central Universities (XDJK2014B039).
Contributor Information
Zhenhui Song, Email: szh7678@126.com.
Xianjin Dai, Email: daixianjin0301@163.com.
Cuifang Ye, Email: 1512358773@qq.com.
Yuntian Li, Email: 57034412@qq.com.
Li Wang, Email: wang7232015@163.com.
Yang Hu, Email: hyeason1024@163.com.
References
- Chen C.M., Pocock D.H., Britton P. Genomic organisation of a virulent Taiwanese strain of transmissible gastroenteritis virus. Adv. Exp. Med. Biol. 1993;342:23–28. doi: 10.1007/978-1-4615-2996-5_4. [DOI] [PubMed] [Google Scholar]
- Corse E., Machamer C.E. Infectious bronchitis virus E protein is targeted to the Golgi complex and directs release of virus-like particles. J. Virol. 2000;74(9):4319–4326. doi: 10.1128/jvi.74.9.4319-4326.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai H.B., Chen L., Zhu L. Isolation and identification of porcine transmissible gastroenteritis virus in Sichuan Province and determination of one-step growth curve (In Chinese) Chin. Vet. Sci. 2012;42(12):1224–1229. [Google Scholar]
- Eleouet J.F., Rasschaert D., Lambert P., Levy L., Vende P., Laude H. Complete sequence (20 kilobases) of the polyprotein-encoding gene 1 of transmissible gastroenteritis virus. Virology. 1995;206:817–822. doi: 10.1006/viro.1995.1004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Escors D., Camafeita E., Ortego J., Laude H., Enjuanes L. Organization of two transmissible gastroenteritis coronavirus membrane protein topologies within the virion and core. J. Virol. 2001;75(24):12228–12240. doi: 10.1128/JVI.75.24.12228-12240.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fangning L., Guohua L., Ke W., Tammy B., Dianjun C., Yanming Z., Lijuan Y. Porcine small intestinal epithelial cell line (IPEC-J2) of rotavirus infection as a new model for the study of innate immune responses to rotaviruses and probiotics. Virus Immunol. 2010;23(2):135–149. doi: 10.1089/vim.2009.0088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gelhaus S., Thaa B., Eschke K., Veit M., Schwegmaan-wessels C. Palmitoylation of the Alphacoronavirus TGEV spike protein S is essential for incorporation into virus-like particles but dispensable for S-M interaction. Virology. 2014;464-465:397–405. doi: 10.1016/j.virol.2014.07.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodwin R.F., Jennings A.R. A highly infectious gastroenteritis of pig. Vet. Rec. 1985;70:271–272. [Google Scholar]
- Haelterman E.O., Hutchings L.M. Epidemic diarrheal disease of viral origin in newborn swine. Ann. N.Y. Acad. Sci. 1956;66(1):186–190. doi: 10.1111/j.1749-6632.1956.tb40119.x. [DOI] [PubMed] [Google Scholar]
- Haelterman E.O. On the pathogenesis of transmissible gastroenteritis of swine. J. Am. Vet. Med. Assoc. 1972;160(40):534–540. [PubMed] [Google Scholar]
- Kapke P.A., Brian D.A. Sequence analysis of the porcine transmissible gastroenteritis coronavirus nucleocapsid protein gene. Virology. 1986;151:41–49. doi: 10.1016/0042-6822(86)90102-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai M.M.C., Cavanagh D. The molecular biology of coronaviruses. Adv. Virus Res. 1997;48:1–100. doi: 10.1016/S0065-3527(08)60286-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lili K., Masters P.S. Evolved variants of the membrane protein can partially replace the envelope protein in murine coronavirus assembly. J. Virol. 2010;84(24):12872–12885. doi: 10.1128/JVI.01850-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu C., Kokuho T., Kubota T., Watanabe S., Inumaru S., Yokomizo Y., Onodera T. DNA mediated immunization with encoding the nucleoprotein gene of porcine transmissible gastroenteritis virus. Virus Res. 2001;80:75–82. doi: 10.1016/s0168-1702(01)00333-1. [DOI] [PubMed] [Google Scholar]
- Motokawa K., Hohdatsu T., Hashimoto H., Koyama H. Comparison of the amino acid sequence and phylogenetic analysis of the peplomer, internal membrane and nucleocapsid protein of feline, canine and porcine coronavirus. Microbiol. Immunol. 1996;40(6):425–433. doi: 10.1111/j.1348-0421.1996.tb01089.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mullan B.P., Davies G.T., Cutler R.S. Simulation of the economic impact of transmissible gastroenteritis on commercial pig production in Australia. Aust. Vet. J. 1994;71:151–154. doi: 10.1111/j.1751-0813.1994.tb03370.x. [DOI] [PubMed] [Google Scholar]
- Nguyen T.D., Bottreau E., Aynaud J.M. Transmissible gastroenteritis (TGE) of swine: in vitro virus attachment and effects of polyanions and polycations. Vet. Microbiol. 1987;14(4):343–354. doi: 10.1016/0378-1135(87)90026-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peter S., Marcel N., Marion P., Kark D.W., Salah A., Ulrike L. Characterization of a porcine intestinal epithelial cell line for in vitro studies of microbial pathogenesis in swine. Histochem. Cell Biol. 2006;125:293–305. doi: 10.1007/s00418-005-0067-z. [DOI] [PubMed] [Google Scholar]
- Ren X., Glende J., Yin J., Schwegmaan-wessels C., Herrler G. Importance of cholesterol for infection of cells by transmissible gastroenteritis virus. Virus Res. 2008;137(2):220–224. doi: 10.1016/j.virusres.2008.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwegmaan-wessels C., Herrler G. Sialic acids as receptor determinants for coronaviruses. Glycoconj. J. 2006;23:51–58. doi: 10.1007/s10719-006-5437-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sirinarumitr T., Kluge J.P., Paul P.S. Transmissible gastroenteritis virus induced apoptosis in swine testes cell cultures. Arch. Virol. 1998;143(12):2471–2485. doi: 10.1007/s007050050477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song Z.H., Dai X.J., Cao X.Z., Zhou Y., Li Y.T., Long Y. Isolation and identification of porcine transmissible gastroenteritis virus from chongqing, southwestern China. Isr. J. Vet. Med. 2015;70(4):22–30. [Google Scholar]
- Tong G., Zhang Y.M., Tang Q.H., Wang J.J. Influence of the transmissible gastroenteritis virus on activities of swine intestinal epithelial cells. Acta Agric. Boreali-occidentalis Sin. 2011;20(1):40–44. (in Chinese) [Google Scholar]
- Trincone A., Schwegmaan-wessels C. Looking for a needle in a haystack: cellular proteins that may interact with the tyrosine-based sorting signal of the TGEV S protein. Virus Res. 2015;202:3–11. doi: 10.1016/j.virusres.2014.11.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valentina M., Simona P., Silvia F., Alessandra L., Elisabetta G. Gene expression study of two widely used pig intestinal epithelial cell lines: IPEC-J2 and IPI-2I. Vet. Immunol. Immunopathol. 2009;131:278–284. doi: 10.1016/j.vetimm.2009.04.006. [DOI] [PubMed] [Google Scholar]
- Wurm T., Chen H., Hoddgson T., Britton P., Brooks G., Hiscox J.A. Localization to the nucleolus is a common feature of coronavirus nucleoproteins and the protein may disrupt host cell division. J. Virol. 2001;75(19):9345–9356. doi: 10.1128/JVI.75.19.9345-9356.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yin J.C., Ren X.F., Li Y.J. Molecular cloning and phylogenetic analysis of ORF7 region of chinese isolate TH-98 from transmissible gastroenteritis virus. Virus Genes. 2004;30(3):395–401. doi: 10.1007/s11262-004-6783-y. [DOI] [PMC free article] [PubMed] [Google Scholar]


