Abstract
Inflammatory diseases of the nose and paranasal sinus are commonly encountered in diagnostic histopathology. This review describes the possible manifestations of the common diseases as well as highlighting some of the uncommon causes of sinonasal inflammation which may have importance for treatment and prognosis. The diagnosis of fungal sinusitis is primarily histological. It is important to distinguish between invasive and non-invasive fungal sinusitis, the latter including allergic fungal sinusitis characterized by ‘allergic mucin’ and scanty fungal hyphae. Nasal eosinophilia is a feature of both allergic and non-allergic rhinosinusitis and a wide range of secondary changes in inflammatory polyps may lead to diagnostic confusion. Nasal biopsies are often taken from perforations or inflammatory masses to confirm or exclude granulomatous diseases. There is a broad differential diagnosis for granulomatous sinonasal disease and pathologists should appreciate the diagnostic histological and clinical features of these conditions.
Keywords: chronic inflammation, diagnosis, fungal diseases, granulomatous inflammation, nose and paranasal sinuses, pathology
The wide range of diseases that may present with sinusitis and/or rhinitis includes some of the most common conditions presenting to head and neck clinicians. The pathologist needs to be familiar with the manifestations of common diseases and be able to recognize those uncommon or rare diseases which require specific treatment. This review considers the normal anatomy and histology of the nasal cavities and sinuses as a prelude to describing the various infective and non-infective causes of sinonasal inflammation (Table 1 ).
Table 1.
Classification of rhinosinusitis
Infectious rhinosinusitis
Non-infectious rhinosinusitis
Rhinosinusitis associated with systemic diseases
|
Surgical anatomy and histology
The nasal cavities are designed to humidify inhaled air and remove particulate matter. The cavities are surrounded by bone and cartilage with a midline septum, floors formed by the hard palate and lateral walls formed by the palatine, maxillary and ethmoid bones which support the three turbinates. The meati beneath the turbinates are the sites into which drain the posterior ethmoidal sinus (superior meatus), other ethmoidal cells, maxillary and frontal sinuses (middle meatus) and the nasolachrymal duct (inferior meatus). The sphenoid sinus drains into the sphenoethmoidal recess behind the superior turbinate. Obstruction to drainage by anatomical variation or mucosal disease around the narrow ostial meati is the cause of much sinus pathology. Functional endoscopic sinus surgery (FESS) with flexible nasendoscopy allows surgeons to visualize sinus disease and has improved the quality of surgery to improvement the drainage of the sinuses and the removal of diseased tissues.
The mucosal lining of the sinonasal tract contains seromucinous glands, vessels and nerves (Figure 1 ). The lining of the sinuses is thinner and less vascular than that of the nasal cavities (Figure 2 ). The sinonasal (Schneiderian) epithelium is derived from ectoderm and, in children, the nasal cavities and sinuses are lined by pseudostratified, ciliated columnar epithelium apart from the olfactory epithelium. Exposure to airborne particles during the first two decades of life leads to squamous metaplasia of the nasal vestibule. The luminal surface of a normal columnar cell has approximately 200 cilia which project into the gel phase of the mucus and beat in a coordinated manner at 10–20 Hz.
Figure 1.

Normal nasal mucosa is covered by respiratory epithelium and contains many vessels.
Figure 2.
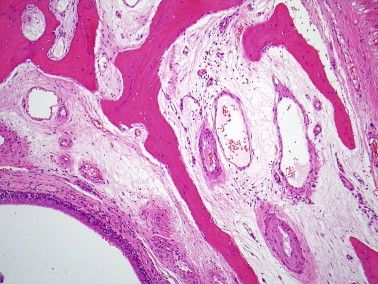
Normal sinus mucosa is thinner and closely apposed to bone.
Infective rhinitis and sinusitis
Acute viral infection, commonly with adenoviruses, echoviruses or coronaviruses, is associated with the outpouring of fluid from the nasal mucosal glands and vessels. Infection by specific bacteria, fungi and other organisms is uncommon in the developed world but may be encountered in biopsies from patients who have travelled through or migrated from countries where such infections are endemic.
Tuberculosis
Tuberculosis of the upper airways is rare, usually associated with pulmonary disease, and results in polyps, ulcers or septal perforation.1 Microscopically, epithelioid and giant cell granulomas are present but caseous necrosis is not prominent. Organisms are rarely seen and the diagnosis relies on clinic–pathological correlation, culture and/or molecular testing. The differential diagnosis includes other granulomatous diseases (Table 2 ).
Table 2.
Differential diagnosis of sinonasal granulomatous inflammation
| Mycobacterial infection – tuberculosis, leprosy |
| Rhinoscleroma |
| Human immunodeficiency virus |
| Fungal infection – Aspergillus flavus |
| Sarcoidosis |
| Wegener's granulomatosis |
| Churg–Strauss angiitis |
| Cocaine-induced midline destructive lesion |
| Intra-nasal narcotic abuse |
| Cholesterol granuloma of the paranasal sinuses (post-haemorrhage) |
| Malakoplakia |
Leprosy
Mycobacterium leprae is transmitted in aerosols that are generated from disease in the upper respiratory tract. The nasal mucosa is a major site of entry of infection and nasal disease may precede skin disease by several years.2 Most nasal lesions occur in patients with anergy who show lepromatous leprosy in which mucosal macrophages, filled with acid fast bacilli, are accompanied by neutrophils, eosinophils, plasma cells and fibrosis. Patients with a good T-cell mediated immune response show tuberculoid leprosy. The diagnosis is made from the clinical features, stains for organisms and/or molecular testing.
Rhinoscleroma
Rhinoscleroma is endemic in parts of Central and South America, North and Central Africa and some areas of Eastern Europe. The disease is caused by the gram-negative bacillus, Klebsiella rhinoscleromatis, and commonly affects the nasal cavities, nasopharynx, lip and oropharynx.3, 4 The disease has three phases. The rhinitic phase shows ulceration, necrosis, acute inflammation and granulation tissue. The florid or granulomatous phase shows pseudoepitheliomatous hyperplasia and an intense mixed inflammatory infiltrate of lymphocytes, plasma cells and macrophages. Russell bodies are prominent in the plasma cells and the large, vacuolated macrophages (Mickulicz cells) (Figure 3 ) contain numerous bacilli which are seen on Warthin–Starry, gram or Giemsa stains and confirmed by culture. The final, fibrotic phase shows variable fibrosis with few Mickulicz cells.
Figure 3.
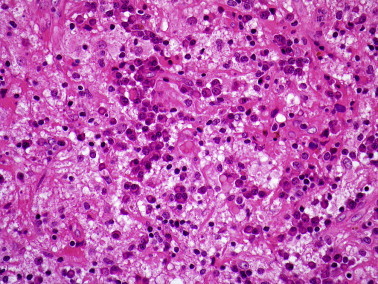
Rhinoscleroma showing large vacuolated macrophages, plasma cells and lymphocytes.
Rhinosporidiosis
Although rhinosporidiosis was described by Seeber in 1900, the causative organism, Rhinosporidium seeberi, has been difficult to classify. Molecular taxonomy using 18S rRNA sequencing places the organism in a clade of protists (Ichthyosporea) that usually infect fish and amphibian.5 The disease is endemic in India and Sri Lanka where sinonasal and ocular disease are common manifestations.6 Nasal infection presents as bulky mucosal tissue covered by hyperplastic squamous or respiratory epithelium and containing an intense infiltrate of lymphocytes and plasma cells associated with numerous cysts (sporangia) that are 100–300 μm in diameter, have thick, PAS-positive walls and contain numerous endospores (Figure 4 ).
Figure 4.
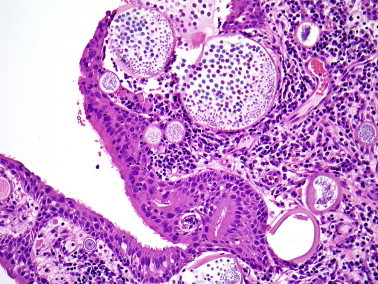
Rhinosporidiosis with large sporangia containing endospores in the superficial mucosal tissue.
Fungal diseases
The categorization of fungus-related diseases of the nose and paranasal sinuses is important for prognosis and treatment (Table 3 ), although the complexities of the interactions between different fungal species and the host immune system are not fully understood. Fungi are common airborne allergens and can be cultured from the nasal secretions of most healthy individuals7; consequently, the diagnosis of fungal sinusitis relies on the histological recognition of fungi in sinus tissue. As the density of fungal elements varies from sparse to extensive, special stains (Grocott and diastase-PAS) should be used routinely when examining sinonasal biopsies, particularly those from immunocompromised individuals. The morphology of fungal hyphae is of limited value in distinguishing between fungal species and cultures should always be performed. The irregular broad hyphae of Mucor sp. are characteristic and dichotomously branching septate hyphae are seen in diseases associated with Aspergillus sp. and the dematiaceous fungi (Figure 5, Figure 6 ).
Table 3.
Classification of fungal rhinosinusitis (adapted from ref. 9).
| Category | Host immune status | Role of fungus |
|---|---|---|
| A. Invasive | ||
| 1. Granulomatous invasive | Immunocompetent | Pathogen |
| 2. Chronic invasive | Impaired (diabetes, steroids) | Pathogen |
| 3. Acute invasive | Immunocompromised | Pathogen |
| B. Non-invasive | ||
| 1. Localized colonization | Immunocompetent | Saprophyte |
| 2. Fungus ball | Immunocompetent | Saprophyte |
| 3. Allergic | Atopic | Allergen |
| 4. Eosinophilic | Mostly non-atopic | Uncertain |
Figure 5.
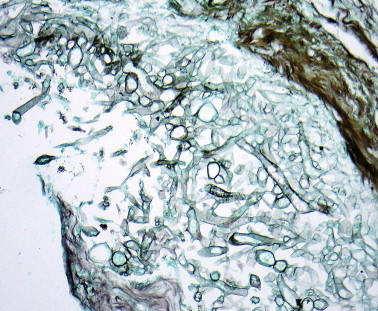
Broad irregular hyphae of Mucor sp. (Grocott silver stain).
Figure 6.

Dichotomously branching septate hyphae of Aspergillus sp.
Invasive fungal sinusitis: invasive fungal sinusitis is almost always encountered in patients whose immune system is compromised by disease (malignancy, autoimmune disease, malnutrition, HIV infection or diabetes) or immunosuppressive treatment. Biopsies show acute inflammation with extensive necrosis and fungal hyphae invading the mucosa, blood vessels and bone.7, 8 Acute fulminant invasive sinusitis results from the rapid, vascular spread of fungi into the sinus walls, orbit and brain and has a high mortality unless treated early. Infection by normally saprophytic fungi of the order Mucorales (including Rhizopus and Mucor) is known as zygomyocosis, and other fungi including Aspergillus fumigatus, have also been implicated. Chronic invasive fungal sinusitis usually affects diabetic patients and is a slowly progressive infection, eroding bone with thick mucus in the sinuses and a mixed inflammatory infiltrate of the tissues containing masses of fungal hyphae, typically those of A. fumigatus. Granulomatous invasive fungal sinusitis has been described in immunocompetent patients in Sudan and the Indian subcontinent; there is profuse growth of A. flavus associated with a non-caseating granulomatous inflammatory infiltrate.
Non-invasive fungal disease: non-invasive fungal disease includes colonization of mucus crusts by saprophytic fungi. This is often seen in patients after endoscopic sinus surgery and may be asymptomatic or lead to a foul smell.9
Sinus fungal ball: sinus fungal ball (previously known as mycetoma or aspergilloma) is defined as radiological sinus opacification due to mucopurulent material containing a dense mass of fungal hyphae; the sinus mucosa shows non-specific inflammation without histological evidence of fungal invasion.8, 9, 10 Fungus balls may coexist with other forms of fungal rhinosinusitis particularly allergic fungal rhinosinusitis. The fungi involved are those whose spores normally contaminate the upper aerodigestive tract, typically A. fumigatus. The fungal hyphae are tightly packed and form a green, yellow, brown or black laminated mass surrounded by neutrophils and which may calcify (Figure 7 ).
Figure 7.

Laminated masses of fungal hyphae in a sinus fungal ball.
Allergic fungal sinusitis (AFS): AFS is a predominantly type 1 hypersensitivity reaction to extramucosal fungal hyphae, although the immunological processes are complex. Patients with AFS present with chronic rhinosinusitis and nasal polyps. The diagnosis of AFS is histological and depends on identifying so-called ‘allergic’ mucin (Figure 8 ). ‘Allergic’ mucin is inspissated mucin with strongly eosinophilic concretions of pyknotic eosinophils surrounded by paler areas that contain Charcot–Leyden crystals; silver stains show scattered fungal hyphae.8, 11 The mucosal tissue is oedematous and lacks fungal hyphae. Culture is required to reliably identify the fungi which are commonly dematiaceous fungi (Bipolaris and Curvularia sp.) and various species of Aspergillus. Patients with AFS are usually atopic, with allergies to multiple aeroallergens on skin testing, asthma and sensitivity to non-steroidal anti-inflammatory drugs.11
Figure 8.

‘Allergic mucin’ composed of inspissated eosinophilic mucin containing masses of degenerate eosinophils and epithelial cells.
Eosinophilic mucin rhinosinusitis (EMRS): EMRS may be diagnosed when the sinuses contain ‘allergic’ mucin but where no fungal hyphae are identified. This may be a clinically distinct disorder of systemic immunological deregulation associated with asthma and aspirin sensitivity,12 but others have found considerable overlap in clinical, radiological and immunological parameters between AFS and EMRS.9
Non-infective chronic rhinosinusitis and nasal polyposis
Chronic rhinosinusitis is sinonasal inflammation lasting for more than 12 weeks with or without nasal polyps. Nasal polyposis affects approximately 1% of the population, presenting with bilateral nasal obstruction, rhinorrhoea and headaches. Imaging studies show soft tissue masses in the nasal cavities and/or sinuses that can erode bone and extend into the nasopharynx, orbit and cranial cavity.
Aetiology and pathogenesis
Allergic rhinitis is a common cause of rhinitis in children. Seasonal allergens include tree pollen (spring), grass pollen (summer), weed pollen (late summer) and fungal spores (autumn and winter). Perennial rhinitis is typically associated with allergens found in the faeces of the house dust mite. Individual susceptibility is influenced by genetic and environmental factors including cigarette smoke, infection and airborne pollutants which depress the responses of T suppressor cells to allergens.
Non-allergic rhinitis with eosinophilia syndrome (NARES) is a clinical syndrome that is most prevalent in middle-aged adults who have symptoms of allergic rhinitis with polyposis in the absence of atopy on skin testing and in whom the nasal mucosa and fluid contains eosinophils.13 IgE levels are not elevated and anosmia is a prominent feature. The pathophysiology is poorly understood but mast cells appear to be important in recruiting eosinophils to the mucosa.
Non-allergic non-eosinophilic rhinitis is most prevalent in middle-aged and elderly adults and often related to mechanical obstruction to drainage and secondary bacterial infection. Symptoms are triggered by changes in temperature, light intensity and emotion, probably due to hyperreactivity of the autonomic nervous system leading to the release of vasoactive neurotransmitters.
The pathogenesis of the inflammatory changes in chronic rhinosinusitis is complex, involving an interplay between genetically-influenced immune responses and environmental factors.14, 15 Activated mast cells and fibroblasts produce pro-inflammatory cytokines (Il-1, Il-6, interferon-γ and TNF-α) which attract and activate neutrophils and eosinophils.16 Lymphocyte subpopulations differ between patients without polyposis (Th-1 predominant with high levels of interferon-γ) and those with polyposis (Th-2 predominant with high IL-5 and IgE levels).14 Mucosal tissue remodelling is manifested by breakdown of the extracellular matrix by proteases (MMP-9) leading to pseudocysts, basement membrane thickening due to the accumulation of fibronectin and types I, III and V collagens, and mucosal gland hyperplasia (possibly related to EGF and EGFR expression).16 TGF-β produced by eosinophils stimulates fibroblasts, as well as upregulating VEGF expression which induces angiogenesis and oedema.15 Tissue eosinophilia is found in up to 90% of mucosal biopsies but the role of eosinophils is uncertain. While eosinophils are characteristically seen in patients with allergies to pollen, fungi, bacterial proteins such as Staphylococcus aureus superantigen or aspirin, they are also seen in patients with no demonstrable allergies (NARES).
Pathology
Inflammatory polyps are smooth-surfaced, pink or pale brown masses up to 3–4 cm in diameter (Figure 9 ) and usually have a broad stalk which is attached to the mucosa of the lateral nasal cavity close to the sinus ostia. Polyps are covered by ciliated respiratory epithelium which often shows immature squamous metaplasia (Figure 10 ) and sometimes ulceration. The epithelial basement membrane is often thickened and the stroma shows marked oedema and myxoid change with pseudocystic degeneration. Variable numbers of blood vessels and myofibroblasts are present. The intensity and type of inflammatory infiltrate varies markedly both within and between polyps; eosinophils, lymphocytes and plasma cells are seen and eosinophils often predominate (Figure 11 ). Polyps from patients with asthma show more basement membrane thickening, goblet cell hyperplasia and eosinophilic infiltration than polyps from patients without asthma,17 but this is not of practical importance.
Figure 9.

Inflammatory nasal polyp formed by oedematous, pale brown mucosal tissue.
Figure 10.
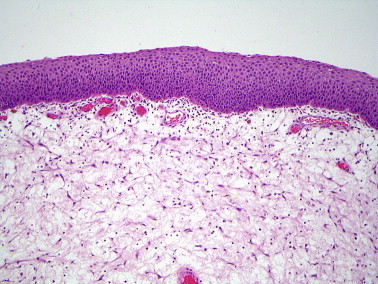
Immature squamous metaplasia on the surface of an inflammatory polyp.
Figure 11.

Inflammatory nasal polyp covered by respiratory epithelium with basement membrane thickening and an eosinophil-rich inflammatory infiltrate.
Secondary changes that may create problems in diagnosis include ulceration and infection with a predominantly neutrophilic infiltrate, fibrosis, infarction, granulation tissue, the deposition of dense fibrinous material resembling amyloid (Figure 12 ), cartilaginous and osseous metaplasia, glandular hyperplasia (Figure 13 ) and cholesterol granuloma formation (Figure 14 ).
Figure 12.

Inflammatory polyp showing extensive fibrinous exudation that superficially resembles amyloid.
Figure 13.

Inflammatory polyp with marked glandular hyperplasia.
Figure 14.

Cholesterol granuloma in an inflammatory polyp associated with haemorrhage.
Differential diagnosis
Myxoid neoplasms: inflammatory polyps often contain atypical stromal myofibroblasts which express α-smooth muscle actin and lack myogenin expression and mitotic activity, distinguishing them from the spindle cells seen in embryonal rhabdomyosarcoma or myxoma.
Nasopharyngeal angiofibromas: angiofibromas contain spindle and stellate myofibroblasts associated with morphologically abnormal, thick-walled vessels (Figure 15 ) and are not often a diagnostic problem.
Figure 15.

Nasopharyngeal angiofibroma is formed from plump spindle cells which surround thick-walled vessels.
Schneiderian papillomas: inverted, transitional type papilloma often have an oedematous stroma and may coexist with inflammatory polyps, but are typically covered by a thick transitional epithelium with infolding of the surface creating a more solid neoplastic structure. The microcystic intra-epithelial changes in the columnar type of papilloma are not features of inflammatory polyps.
Sinonasal NK/T-cell lymphoma: nasal lymphomas may be clinically indistinguishable from allergic rhinitis and superficial biopsies may show only non-specific chronic inflammation. A high clinical index of suspicion and repeated deep biopsies may be necessary to reach a definitive diagnosis in patients who do not respond to rhinitis treatment.18
Respiratory epithelial adenomatoid hamartoma (REAH): REAH is a rare, adenomatoid glandular proliferation that presents as a polypoid lesion arising from the posterior septum or the lateral wall of the nasal cavity. The appearances vary from large glands lined by ciliated columnar epithelium to clusters of small glands lined by simple cuboidal epithelium (seromucinous hamartoma) (Figure 16 ). A myoepithelial layer is identified by expression of smooth muscle actin. The stroma is usually oedematous and contains a mixture of inflammatory cells including eosinophils. The lack of the lobulated clustering of glands seen in rhinosinusitis also distinguishes this disease from REAH.
Figure 16.

Seromucinous hamartoma showing small glands that lack the normal lobularity of mucosal glands.
Other localized types of chronic rhinosinusitis
Rhinitis medicamentosa
Rhinitis medicamentosa is drug-induced sinonasal inflammation that associated with the use of topical nasal decongestants. The pathophysiology is unclear but is likely to involve loss of sensitivity of the mucosal vessels to the vasoconstrictor effects of decongestants so that, on cessation of treatment, there is a rebound increase in blood flow and mucosal congestion. Histological changes are non-specific.19
Atrophic rhinosinusitis
Atrophic rhinosinusitis is a chronic disease related to the progressive loss and replacement of the normal columnar epithelium by squamous epithelium accompanied by resorption of bone and excessive, viscid nasal secretion. This may occur in previously healthy patients or secondary to sinus surgery, nasal trauma, radiation and other inflammatory disease of the sinonasal tract. The primary form is associated with a foul odour (ozaena) due to mucosal colonization with K. ozenae.20
Nasal septal perforation
The causes of septal perforation include local trauma (‘nose-pickers’ perforation), inhalation of toxic substances such as cocaine or intra-nasal steroid and decongestant sprays, infective and non-infective granulomatous diseases and nasal malignancies.21 Occupational exposure to corrosive chemicals such as strong acids and alkalis, chromic acid mists, nickel, arsenic and copper may lead to perforation.22 Biopsies are taken from the mucosal edge of a perforation to exclude specific pathologies, and usually show non-specific acute inflammatory debris, inflamed granulation tissue and reactive changes in the adjacent epithelium.23
Cocaine-induced midline destructive lesions (CIMDL)
CIMDL are a result of cocaine inhalation leading to intense vasoconstriction and extensive destruction of the cartilaginous tissues of the nose.24 Biopsies tend to show non-specific chronic inflammation and necrosis but may include a granulomatous reaction to foreign material (Figure 17 ). The clinical and histological features may suggest a diagnosis of atypical Wegener's granulomatosis if the history of drug abuse is not provided. CIMDL are associated with cytoplasmic antineutrophil cytoplasmic antibodies (C-ANCA) directed against neutrophil elastase.25
Figure 17.
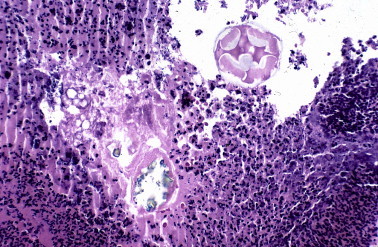
Foreign material in the inflammatory exudate from a patient with septal perforation associated cocaine inhalation.
Mucocele
The accumulation of large amounts of mucus in the maxillary sinus is most commonly seen in children and young adults with a long history of chronic rhinosinusitis and an obstructed sinus ostium. Mucoceles of the frontal sinuses may present with orbital swelling and proptosis while those of the posterior ethmoid or sphenoid can compress the optic nerve. The grey/pink, gelatinous mass of mucus contains a mixture of desquamated epithelial cells, neutrophils, lymphocytes and plasma cells. The differential diagnosis includes fungal sinusitis, myxoma, mucinous adenocarcinoma and embryonal rhabdomyosarcoma.
Nasal manifestation of systemic diseases
Cystic fibrosis
Nasal polyps in children are uncommon and are often associated with cystic fibrosis, an autosomal recessive condition resulting from mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) gene. CFTR is both a cyclic-AMP regulated chloride channel in epithelial cells and a reduced glutathione transporter. Mutated CFTR is sequestered in the endoplasmic reticulum where it induces a stress response including pro-inflammatory cytokines.26 Carriers (parents) with one abnormal CFTR gene have an increased incidence of sinusitis (36%) compared with the normal population (14–16%), and all patients with cystic fibrosis have sinonasal disease but this is only symptomatic in a minority.27 There is an increased number of goblet cells in the nasal mucosa and the mucus is abnormally viscous leading to recurrent infections. The mucus is more acidic than that seen in normal seromucinous glands and polyps in cystic fibrosis contain fewer eosinophils and show less basement membrane thickening than inflammatory polyps in adults.
Primary ciliary dyskinesia
Primary ciliary dyskinesia (immotile cilia syndrome) is an autosomal recessive condition arising from the absence of the inner and/or outer dynein arms of the ciliary axoneme. Patients suffer from recurrent upper and lower respiratory tract infections and bronchiectasis, and about 50% have Kartagener's syndrome with dextrocardia, bronchiectasis, chronic rhinosinusitis and infertility. The diagnosis of ciliopathies has traditionally relied on ultrastructural examination of cilia and clinical analysis of ciliary motility but is now facilitated by mutational analysis of ciliary proteins.28, 29 Secondary ciliary dysfunction may result from bacterial or viral infections and is usually temporary, although severe infections may lead to squamous metaplasia and permanent loss of cilia.
Immunodeficiency
Nasal infections are common in patients with primary and secondary immunodeficiencies. Common variable immunodeficiency most frequently presents in adult life and includes IgG subclass and specific antibody deficiencies, although the number of B lymphocytes is usually normal. IgG subclass deficiency usually presents with recurrent infections such as sinusitis, while IgA deficiency is usually asymptomatic.30 Fungal infections are common (see above). HIV infection is associated with rhinitis and crusting and an increased susceptibility to a range of atypical infections including Cytomegalovirus, Cryptosporidium and Acanthamoeba.31, 32
Sarcoidosis
Sarcoidosis is a multisystem, granulomatous disease of unknown aetiology that affects young and middle-aged adults.33 1–6% of patients with sarcoidosis have symptomatic nasal obstruction due to swelling of the inferior turbinates. The mucosa may be dry, crusted or nodular, but ulceration is uncommon. Microscopy reveals non-caseating epithelioid and giant cell granulomas (Figure 18 ) associated with CD4+ T lymphocytes.34 The association with cutaneous or lung disease, hilar lymphadenopathy and raised serum ACE levels usually allows diagnosis but skin or nasal biopsies might be required for confirmation.
Figure 18.

Granulomatous inflammation due to sarcoidosis has well defined epithelioid cell granulomas.
Wegener's granulomatosis
Wegener's granulomatosis is a multisystem disorder characterized by necrotizing granulomatous inflammation and pauci-immune vasculitis. The prevalence in Europe is approximately five per 100,000 with more cases described in more northern latitudes. The cause of Wegener's granulomatosis is unknown but neutrophils are regarded as key to the pathogenesis of the acute injury causing endothelial damage and being targets for the autoimmune response.35, 36 Genetic factors and complex alterations in T helper cell responses promote granuloma formation.37 Most patients present with sinusitis, nasal discharge or bleeding and the disease can destroy the nasal septum.38 Seventy percent of patients will develop crescentic glomerulonephritis and many will have systemic symptoms reflecting small-vessel inflammation in the lungs, skin, joints and nerves. The diagnosis is facilitated by testing for C-ANCA which is directed against neutrophil proteinase-3 (PR3-ANCA) in 90% patients, or myeloperoxidase (MPO-ANCA). For entry into European clinical trials, a diagnosis of Wegener's granulomatosis requires a history of chronic inflammation for at least four weeks which is not attributable to another cause, supported by characteristic biopsy histology and/or detectable C-ANCA and/or detectable PR3-ANCA.39 Confirmatory histology in non-renal biopsies requires an inflammatory infiltrate that is dominated by neutrophils accompanied by at least one of necrotizing small-vessel vasculitis, granulomas or giant cells (Figure 19 ). The differential diagnosis includes other small-vessel vasculitides associated with ANCA, especially microscopic polyangiitis and Churg–Strauss syndrome.
Figure 19.

Granulomatous inflammation in Wegener's granulomatosis shows poorly defined epithelial cell granulomas, multinucleate cells and lymphocytes.
Churg–Strauss syndrome
Churg–Strauss syndrome was first described in patients who had previously been diagnosed as polyarteritis nodosa with necrotizing vasculitis, but who showed tissue eosinophilia and extravascular granulomas.40 Patients present with symptoms of allergic rhinitis, polyposis and sinusitis and a history of asthma. Clinically significant disease affects the lungs, skin and kidneys, although severe renal disease is uncommon. C-ANCA are present in about 40% patients and are usually P-ANCA or MPO-ANCA.41 Biopsies typically show an eosinophil-rich inflammatory infiltrate and may also show vasculitis and granulomatous inflammation.41
Relapsing polychondritis
Relapsing polychondritis is a rare disease presenting in young and middle-aged adults with recurrent episodes of painful acute inflammation resulting in destruction of the cartilages of the nose and ears.42 Tracheal chondritis leads to airway obstruction and pneumonia. Other manifestations include migratory arthritis, ocular inflammation, cardiac and neurological disease. About one third of patients have other autoimmune diseases and there is overlap with Behcet's disease – the so-called MAGIC syndrome of mouth and genital ulcers with inflamed cartilage.43 Circulating antibodies to type II collagen is found in many patients.42 Biopsies of the involved cartilage show loss of mucopolysaccharide matrix and erosion by lymphocytes (Figure 20 ). Neutrophils may be present in the early stages and there is usually perichondrial granulation tissue and fibrosis.42 The diagnosis is primarily clinical and immunological.
Figure 20.
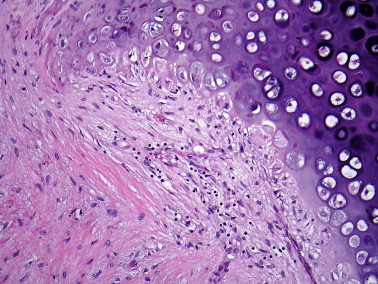
Inflammatory destruction of cartilage in relapsing polychondritis.
Eosinophilic angiocentric fibrosis
Eosinophilic angiocentric fibrosis is an uncommon, idiopathic inflammatory condition of the nose which tends towards healing by fibrosis leading to nasal obstruction. About 30% patients also have granuloma faciale. In the early stages, there is an eosinophil-rich inflammatory exudate around vessels. In later stages the inflammatory infiltrate is more patchy and mixed (eosinophils, neutrophils, lymphocytes and plasma cells) and progresses to onion-skin perivascular fibrosis (Figure 21 ). The clinical and imaging features are non-specific and the diagnosis is histological.44 The differential diagnosis includes most of the other chronic inflammatory conditions mentioned in this review.
Practice points.
Fungal rhinosinusitis
-
•
The manifestations of fungal sinusitis depend on the interplay between the host immune system and various species of fungi.
-
•
Histology has an important role in diagnosis and classification.
-
•
Histology identifies the presence of fungi and microbiology determines the type of fungus.
Chronic rhinosinusitis and polyposis
-
•
Polyposis is an inflammatory reaction of the nasal mucosa to damage by allergy, infection, airborne pollutants or the release of neurotransmitters.
-
•
Nasal polyps in children are often associated with cystic fibrosis.
-
•
Tissue eosinophilia may result from allergic or non-allergic processes.
Figure 21.

a Eosinophilic angiocentric fibrosis showing nodular fibrosis around vessels. b Very few eosinophils are present at this stage of the disease.
Acknowledgements
I would like to thank the head and neck clinicians and pathologists from various countries who have contributed to my knowledge and understanding of sinonasal diseases.
References
- 1.Kim Y.M., Kim A.Y., Park Y.H., Kim D.H., Rha K.S. Eight cases of nasal tuberculosis. Otolaryngol Head Neck Surg. 2007;137:500–504. doi: 10.1016/j.otohns.2007.04.009. [DOI] [PubMed] [Google Scholar]
- 2.Melo Naves M., Gomes Patrocinio L., Patrocinio J.A. Contribution of nasal biopsy to leprosy diagnosis. Am J Rhinol Allergy. 2009;23:177–180. doi: 10.2500/ajra.2009.23.3301. [DOI] [PubMed] [Google Scholar]
- 3.de Pontual L., Ovetchkine P., Rodriguez D. Rhinoscleroma: a French national retrospective study of epidemiological and clinical features. Clin Infect Dis. 2008;47:1396–1402. doi: 10.1086/592966. [DOI] [PubMed] [Google Scholar]
- 4.Hart C.A., Rao S.K. Rhinoscleroma. J Med Microbiol. 2000;49:395–396. doi: 10.1099/0022-1317-49-5-395. [DOI] [PubMed] [Google Scholar]
- 5.Fredricks D.N., Jolley J.A., Lepp P.W., Kosek J.C., Relman D.A. Rhinosporidium seeberi: a human pathogen from a novel group of aquatic protistan parasites. Emerg Infect Dis. 2000;6:273–282. doi: 10.3201/eid0603.000307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sudarshan V., Goel N.K., Gahine R., Krishnani C. Rhinosporidiosis in Raipur, Chhattisgarh: a report of 462 cases. Indian J Pathol Microbiol. 2007;50:718–721. [PubMed] [Google Scholar]
- 7.Deshazo R.D. Syndromes of invasive fungal sinusitis. Med Mycol. 2009;47(suppl 1):S309–S314. doi: 10.1080/13693780802213399. [DOI] [PubMed] [Google Scholar]
- 8.Taxy J.B. Paranasal fungal sinusitis: contributions of histopathology to diagnosis: a report of 60 cases and literature review. Am J Surg Pathol. 2006;30:713–720. doi: 10.1097/00000478-200606000-00006. [DOI] [PubMed] [Google Scholar]
- 9.Chakrabarti A., Das A., Panda N.K. Controversies surrounding the categorization of fungal sinusitis. Med Mycol. 2009;47(suppl 1):S299–S308. doi: 10.1080/13693780802213357. [DOI] [PubMed] [Google Scholar]
- 10.Grosjean P., Weber R. Fungus balls of the paranasal sinuses: a review. Eur Arch Otorhinolaryngol. 2007;264:461–470. doi: 10.1007/s00405-007-0281-5. [DOI] [PubMed] [Google Scholar]
- 11.Schubert M.S. Allergic fungal sinusitis: pathophysiology, diagnosis and management. Med Mycol. 2009;47(suppl 1):S324–S330. doi: 10.1080/13693780802314809. [DOI] [PubMed] [Google Scholar]
- 12.Ferguson B. J. Eosinophilic mucin rhinosinusitis: a distinct clinicopathological entity. Laryngoscope. 2000;110:799–813. doi: 10.1097/00005537-200005000-00010. [DOI] [PubMed] [Google Scholar]
- 13.Ellis A.K., Keith P.K. Nonallergic rhinitis with eosinophilia syndrome. Curr Allergy Asthma Rep. 2006;6:215–220. doi: 10.1007/s11882-006-0037-0. [DOI] [PubMed] [Google Scholar]
- 14.Bachert C., Van Bruaene N., Toskala E. Important research questions in allergy and related diseases: 3-chronic rhinosinusitis and nasal polyposis – a GALEN study. Allergy. 2009;64:520–533. doi: 10.1111/j.1398-9995.2009.01964.x. [DOI] [PubMed] [Google Scholar]
- 15.Huvenne W., van Bruaene N., Zhang N. Chronic rhinosinusitis with and without nasal polyps: what is the difference? Curr Allergy Asthma Rep. 2009;9:213–220. doi: 10.1007/s11882-009-0031-4. [DOI] [PubMed] [Google Scholar]
- 16.Pawankar R., Nonaka M. Inflammatory mechanisms and remodeling in chronic rhinosinusitis and nasal polyps. Curr Allergy Asthma Rep. 2007;7:202–208. doi: 10.1007/s11882-007-0073-4. [DOI] [PubMed] [Google Scholar]
- 17.Ardehali M.M., Amali A., Bakhshaee M., Madani Z., Amiri M. The comparison of histopathological characteristics of polyps in asthmatic and nonasthmatic patients. Otolaryngol Head Neck Surg. 2009;140:748–751. doi: 10.1016/j.otohns.2009.01.027. [DOI] [PubMed] [Google Scholar]
- 18.Sands N.B., Tewfik M.A., Hwang S.Y., Desrosiers M. Extranodal T-cell lymphoma of the sinonasal tract presenting as severe rhinitis: case series. J Otolaryngol Head Neck Surg. 2008;37:528–533. [PubMed] [Google Scholar]
- 19.Doshi J. Rhinitis medicamentosa: what an otolaryngologist needs to know. Eur Arch Otorhinolaryngol. 2009;266:623–625. doi: 10.1007/s00405-008-0896-1. [DOI] [PubMed] [Google Scholar]
- 20.Ly T.H., deShazo R.D., Olivier J., Stringer S.P., Daley W., Stodard C.M. Diagnostic criteria for atrophic rhinosinusitis. Am J Med. 2009;122:747–753. doi: 10.1016/j.amjmed.2008.12.025. [DOI] [PubMed] [Google Scholar]
- 21.Dosen L.K., Haye R. Nasal septal perforation 1981–2005: changes in etiology, gender and size. BMC Ear Nose Throat Disord. 2007;7:1. doi: 10.1186/1472-6815-7-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Castano R., Theriault G., Gautrin D. Categorizing nasal septal perforations of occupational origin as cases of corrosive rhinitis. Am J Ind Med. 2007;50:150–153. doi: 10.1002/ajim.20419. [DOI] [PubMed] [Google Scholar]
- 23.Murray A., McGarry G.W. The clinical value of septal perforation biopsy. Clin Otolaryngol Allied Sci. 2000;25:107–109. doi: 10.1046/j.1365-2273.2000.00332.x. [DOI] [PubMed] [Google Scholar]
- 24.Rachapalli S.M., Kiely P.D. Cocaine-induced midline destructive lesions mimicking ENT-limited Wegener's granulomatosis. Scand J Rheumatol. 2008;37:477–480. doi: 10.1080/03009740802192043. [DOI] [PubMed] [Google Scholar]
- 25.Wiesner O., Russell K.A., Lee A.S.G. Antineutrophil cytoplasmic antibodies reacting with human neutrophil elastase as a diagnostic marker for cocaine-induced midline destructive lesions but not autoimmune vasculitis. Arthritis Rheum. 2004;50:2954–2965. doi: 10.1002/art.20479. [DOI] [PubMed] [Google Scholar]
- 26.Rottner M., Freyssinet J.M., Martinez M.C. Mechanisms of the noxious inflammatory cycle in cystic fibrosis. Respir Res. 2009;10:23. doi: 10.1186/1465-9921-10-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Robertson J.M., Friedman E.M., Rubin B.K. Nasal and sinus disease in cystic fibrosis. Paediatr Respir Rev. 2008;9:213–219. doi: 10.1016/j.prrv.2008.04.003. [DOI] [PubMed] [Google Scholar]
- 28.Escudier E., Duquesnoy P., Papon J.F., Amselem S. Ciliary defects and genetics of primary ciliary dyskinesia. Paediatr Respir Rev. 2009;10:51–54. doi: 10.1016/j.prrv.2009.02.001. [DOI] [PubMed] [Google Scholar]
- 29.Leigh M.W., Zariwala M.A., Knowles M.R. Primary ciliary dyskinesia: improving the diagnostic approach. Curr Opin Pediatr. 2009;21:320–325. doi: 10.1097/MOP.0b013e328329cddb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Vanlerberghe L., Joniau S., Jorissen M. The prevalence of humoral immunodeficiency in refractory rhinosinusitis: a retrospective analysis. B-ENT. 2006;2:161–166. [PubMed] [Google Scholar]
- 31.Dunand V.A., Hammer S.M., Rossi R. Parasitic sinusitis and otitis in patients infected with human immunodeficiency virus: report of five cases and review. Clin Infect Dis. 1997;25:267–272. doi: 10.1086/514536. [DOI] [PubMed] [Google Scholar]
- 32.Youngs R. Human immunodeficiency virus in otolaryngology. J Laryngol Otol. 1997;111:209–211. doi: 10.1017/s0022215100136928. [DOI] [PubMed] [Google Scholar]
- 33.Dempsey O.J., Paterson E.W., Kerr K.M., Denison A.R. Sarcoidosis. BMJ. 2009;339:b3206. doi: 10.1136/bmj.b3206. [DOI] [PubMed] [Google Scholar]
- 34.Noor A., Knox K.S. Immunopathogenesis of sarcoidosis. Clin Dermatol. 2007;25:250–258. doi: 10.1016/j.clindermatol.2007.03.002. [DOI] [PubMed] [Google Scholar]
- 35.Savage C.O.S. Vascular biology and vasculitis. APMIS Suppl. 2009:37–40. doi: 10.1111/j.1600-0463.2009.02475.x. [DOI] [PubMed] [Google Scholar]
- 36.Witko-Sarsat V., Daniel S., Noel L.H., Mouthon L. Neutrophils and B lymphocytes in ANCA-associated vasculitis. APMIS Suppl. 2009:27–31. doi: 10.1111/j.1600-0463.2009.02473.x. [DOI] [PubMed] [Google Scholar]
- 37.Lamprecht P., Wieczorek S., Epplen J.T., Ambrosch P., Kallenberg C.G. Granuloma formation in ANCA-associated vasculitides. APMIS Suppl. 2009:32–36. doi: 10.1111/j.1600-0463.2009.02474.x. [DOI] [PubMed] [Google Scholar]
- 38.Cannady S.B., Batra P.S., Koening C. Sinonasal Wegener granulomatosis: a single-institution experience with 120 cases. Laryngoscope. 2009;119:757–761. doi: 10.1002/lary.20161. [DOI] [PubMed] [Google Scholar]
- 39.Jayne D. The diagnosis of vasculitis. Best Pract Res Clin Rheumatol. 2009;23:445–453. doi: 10.1016/j.berh.2009.03.001. [DOI] [PubMed] [Google Scholar]
- 40.Churg J., Strauss L. Allergic granulomatosis, allergic angiitis, and periarteritis nodosa. Am J Pathol. 1951;27:277–301. [PMC free article] [PubMed] [Google Scholar]
- 41.Sinico R.A., Bottero P. Churg–Strauss angiitis. Best Pract Res Clin Rheumatol. 2009;23:355–366. doi: 10.1016/j.berh.2009.02.004. [DOI] [PubMed] [Google Scholar]
- 42.Rapini R.P., Warner N.B. Relapsing polychondritis. Clin Dermatol. 2006;24:482–485. doi: 10.1016/j.clindermatol.2006.07.018. [DOI] [PubMed] [Google Scholar]
- 43.Kotter I., Deuter C., Gunaydin I., Zierhut M. MAGIC or not MAGIC – does the MAGIC (mouth and genital ulcers with inflamed cartilage) syndrome really exist? A case report and review of the literature. Clin Exp Rheumatol. 2006;24:S108–S112. [PubMed] [Google Scholar]
- 44.Kosarac O., Luna M.A., Ro J.Y., Ayala A.G. Eosinophilic angiocentric fibrosis of the sinonasal tract. Ann Diagn Pathol. 2008;12:267–270. doi: 10.1016/j.anndiagpath.2007.02.002. [DOI] [PubMed] [Google Scholar]


