Abstract
Objectives
The airway expression of type III interferons (IFNs) was evaluated in infants hospitalized for respiratory syncytial virus (RSV) or rhinovirus (HRV) bronchiolitis. As an additional objective we sought to determine whether a different expression of IFN lambda 1–3 was associated with different harboring viruses, the clinical course of bronchiolitis or with the levels of well established IFN stimulated genes (ISGs), such as mixovirus resistance A (MxA) and ISG56.
Methods
The analysis was undertaken in 118 infants with RSV or HRV bronchiolitis. Nasopharyngeal washes were collected for virological studies and molecular analysis of type III IFN responses.
Results
RSV elicited higher levels of IFN lambda subtypes when compared with HRV. A similar expression of type III IFN was found in RSVA or RSVB infected infants and in those infected with HRVA or HRVC viruses. Results also indicate that IFN lambda 1 and IFN lambda 2–3 levels were correlated with each other and with MxA and ISG56-mRNAs. In addition, a positive correlation exists between the IFN lambda1 levels and the clinical score index during RSV infection. In particular, higher IFN lambda 1 levels are associated to an increase of respiratory rate.
Conclusions
These findings show that differences in the IFN lambda 1–3 levels in infants with RSV or HRV infections are present and that the expression of IFN lambda 1 correlates with the severity of RSV bronchiolitis.
Keywords: IFN lambda, IL-28, IL-29, RSV, HRV, MxA, ISG56, Viral load, Bronchiolitis
Introduction
Bronchiolitis is a disorder most commonly caused in infants by viral lower respiratory tract infections; it is characterized by acute inflammation, edema and necrosis of epithelial cells lining small airways, increased mucus production, and bronchospasm.1 The most common virus causing bronchiolitis is the respiratory syncytial virus (RSV).2, 3 Other viruses identified as causing bronchiolitis are rhinovirus (HRV), human metapneumovirus, bocavirus, and parainfluenza.4 In particular, HRV has been recently shown to infect the lower airway as well5 and confirmed to be the second most frequent cause of bronchiolitis.6
It has been demonstrated that the combination of both host and viral factors profoundly influence the severity of viral associated bronchiolitis.7, 8, 9 However, it is not yet clear whether the different subtypes of RSV (A and B) or HRV species (A, B, and C) cause different grades of bronchiolitis severity.10, 11 Furthermore, the role of the innate immune response, in the pathogenesis of severe RSV or HRV disease is still to be defined in detail.7, 8, 9
Among the main players of antiviral innate immune response, the type I interferons (IFNs), IFN alpha and beta, are considered cytokines crucial for anti-viral resistance and represent an early antiviral host defense mechanism against viral infections.12 In 2003, a novel class of antiviral cytokines was discovered, characterized and classified as type III IFNs: IFN lambda1/IL-29, IFN lambda 2/IL-28A, and IFN lambda 3/IL-28B.13 At the amino acid level IFN lambda2 and lambda3 are highly similar having 96% sequence identity while IFN lambda1 shares approximately 81% sequence identity with IFN lambda2 and lambda3.13 The Type III IFNs possess antiviral properties similar to those of type I IFNs but appear to be expressed especially by epithelial cells and consequently exert host protection primarily at epithelial surfaces.14, 15, 16
Despite the fact that it is known that IFN lambda contributes to the control of viral infections in epithelial cells of respiratory tract17, 18, 19 and that the presence of single nucleotide polymorphism around IFN lambda 3 (IL-28B) can increase the risk of hospitalization for bronchiolitis at early age,20 the IFN lambda 1–3 expression in the respiratory tracts of hospitalized infants with RSV or HRV infections has never been addressed.
Hence, considering the importance of the IFN lambda in protecting the airway tract from virus infections,17, 18, 19 we hypothesized that the heterogeneity of IFN lambda 1–3 levels could, at least in part, explain the broad clinical spectrum of RSV or HRV bronchiolitis. Therefore, we evaluated whether there was a difference in the gene expression of IFN lambda 1–3 subtypes between infants with a clinical diagnosis of RSV associated acute bronchiolitis and those with HRV infection. The same analysis was also performed between RSV or HRV subtypes. In addition, to characterize the activation of type III IFNs in the airway tract of infants with RSV or HRV infections, we evaluated whether there was a coordinate activation between IFNs lambda and that of MxA and IFN-stimulated gene (ISG) 56, which are well known markers of type I and III IFN antiviral activity.12 Finally, to further characterize the above issues, we also assessed whether a correlation between IFN lambda 1–3 levels and demographic, virological and clinical parameters in RSV and HRV infected infants actually exist.
Patients and methods
Study population
A total of 118 infants with single RSV or HRV infection were retrospectively selected from a total of 250 infants admitted with a clinical diagnosis of acute bronchiolitis during three epidemic seasons (2008–2011) to the Paediatric Department of Policlinico Umberto I Hospital. The study was approved by the ethics committees and informed consent was obtained from the infant's parents.
Bronchiolitis was diagnosed from the presence of a history of upper respiratory tract infection followed by the acute onset of respiratory distress with cough, tachypnea, retraction, and diffuse crackles on auscultation (wheezing alone was not considered sufficient cause for inclusion in the study). The exclusion criteria were underlying chronic disease (such as cystic fibrosis, chronic pulmonary disease, congenital heart disease, and immunodeficiency) and recurrent (more than one) wheezing episodes.4, 21
The severity of the illness was assessed clinically on the following four indications, each of which was assigned a score within the range 0–8.4 In particular, on admission to hospital, the clinical severity was assigned to each infant, based on respiratory rate (<45 breaths/min = 0, 45–60 breaths/min = 1, >60 breaths/min = 2) arterial oxygen saturation in room air (>95% = 0, 95–90% = 1, <90% = 2), presence of retractions (none = 0, present = 1, present + nasal fare = 2), and ability to feed (normal = 0, reduced = 1, endovenous = 2).
Specimen collection
Nasopharyngeal washings were collected in the first 48 h after admission to the hospital from infants suffering from acute bronchiolitis, and an aliquot was tested for viruses as previously described.22 In particular nasopharyngeal washings were obtained with 3 ml of sterile saline physiological solution injected into each nostril and collected with a syringe. All samples were delivered on ice within 1–2 h to the virology laboratory and on arrival, if needed, they were vortexed with beads to solve mucus. They were divided into two aliquots: one was treated for nucleic acid extraction and viral detection; the second was centrifuged at 2000 rpm for 10 min, and each cell pellet was resuspended in 1 ml of phenol and guanidine isothiocyanate reagent (Trizol, Gibco-BRL, NY) and frozen at −80 °C for gene expression analysis.
PCR assays for respiratory viruses
A panel of reverse transcription-PCR (RT-PCR) or nested PCR assays, some in a multiplex format, were used for the detection of 14 respiratory viruses, including RSV; influenza viruses A and B; coronaviruses OC43, 229E, NL63, and HKU1; metapneumovirus; adenovirus; HRV; and parainfluenza virus types 1–3, human bocavirus as previously reported.22, 23 The evaluation of the sensitivity of the RT-PCR or PCR tests for the 14 respiratory viruses was made as described in our previously published papers.22, 23 In particular, the integrity of the extracted nucleic acid was tested by means of the amplification of the cellular gene beta actin.
Molecular typing of RSV and HRV
RSV or HRV-positive samples were typed as RSVA-B or as HRV A-C respectively. The RSV fragment to be sequenced was obtained by re-extracting RNA from RSV-positive samples and directly amplifying it using SuperScript One-Step RT-PCR for Long Templates kit (Life Technologies, Monza, Italy) with two expressly designed forward primers, A-Fseq 5′-AAT GAT TTT CAC TTT GAA-3′; B-Fseq 5′-GAT GAT TAC CAT TTT GAA-3′, corresponding to G gene position 481–498 of RSV-A2 and to position 475–492 of the RSV-BA reference strains, respectively, and with one reverse primer targeting the fusion protein gene 5′end.24 HRV-positive samples were retrospectively amplified with primers widely used for genotyping targeting 390 bases of the 5′Untranslated Region (5′UTR) central portion.25
TaqMan-based real-time RT-PCR for type III IFN and ISGs
The mRNA copy content of IFN lambda 1–3, MxA and ISG56 was measured by a real-time 5′ exonuclease RT-PCR assay using the Light Cycler 480 sequence detector (Roche, Monza, Italy). Briefly, the total cellular RNA was extracted from the cells collected from nasopharyngeal washings as described, using phenol and guanidine isothiocyanate reagent (TRIzol, Gibco-BRL, NY), by following the manufacturer's instructions, and was retro-transcribed as previously specified.26 Primer pairs and probes for IFN lambda 1 [forward primer, 5′-GGACGCCTTGGAAGAGTCACT-3′; reverse primer, 5′-AGAAGCCTCAGGTCCCAATTC-3′; probe, 5′FAM-AGTTGCAGCTCTCCTGTCTTCCCCG-3’TAMRA27], IFN lambda 2–3 [forward primer, 5′-CTGCCACATAGCCCAGTTCA-3′; reverse primer, 5′-AGAAGCGACTCTTCTAAGGCATCTT-3; probe, 5′ FAM-TCTCCACAGGAGCTGCAGGCCTTTA-3’TAMRA27], MxA (forward primer, 5′-CTGCCTGGCAGAAAAACTTAC-3; reverse primer, 5′-CTCTGTTATTCTCTGGTGAGTCTCCTT-3′; probe, 5′FAM CATCACACATATCTGTAAATCTCTGCCCCTGTT-3’TAMRA); ISG56 [forward primer, 5′-TGAAGAAGCTCTAGCCAACATGTC-3′; reverse primer, 5′-GAGCTTTATCCACAGAGCCTTTTC-3′; probe: 6’FAM 5′-TATGTCTTTCGATATGCAGCCAAGTTTTACCG-3′ TAMRA28] were added to the universal PCR master mix (Roche) at 600 and 400 nM, respectively, in a final volume of 20 μl.
The coamplification of the beta-glucuronidase gene (forward primer, 5′-TCTGTCAAGGGCAGTAACCTG-3; reverse primer, 5′-GCCCACGACTTTGTTTTCTG-3′; probe, 5′FAM-TCAAGTTGGAAGTGCGTCTTTTGGATGC-3’TAMRA) was used to normalize the amount of total RNA present using the threshold cycle relative quantification [the 2–(delta) Ct] method according to the supplier's guidelines. All the determinations were performed in duplicate.
TaqMan-based real-time RT-PCR technique for RSV or HRV detection
A TaqMan-based real-time PCR technique for RSV28 or HRV29 RNA quantification was performed on all nasopharyngeal washing specimens with positive RT-PCR results for RSV or HRV respectively. Briefly, viral RNA was extracted from nasopharyngeal washings NPW that were positive for RSV or HRV, using a QIAamp Viral RNA Mini Kit (Qiagen, Milan, Italy). The RNA was dissolved in RNase-free water and the RSV quantification was performed by Taqman assay after generation of cDNA using a High Capacity cDNA Archive Kit (Applied Biosystems, Monza, Italy). Type-specific primers and probes for N gene of both RSV A and B30 or 5′UTR region of HRV A-C strains29 were added to the universal PCR master mix (Roche) at 600 and 400 nM, respectively, in a final volume of 20 μl.
The standards for RSV or HRV were obtained respectively by cloning the 82 bp of RSV N gene or 203 bp of the 5′ UTR HRV region into the pCR2.1 plasmid using a TOPO TA cloning kit (Invitrogen Corporation, San Diego, CA, USA). A linear distribution (r = 0.99) was obtained between 101 and 108 copies of RSV or HRV-DNA. Viral load values were Log transformed for analysis and data was expressed as the Log number of RSV or HRV copies per ml of nasopharyngeal washings. All the determinations were performed in duplicate.
Statistical analysis
All measurements are expressed median (range) or frequency (percentage), unless otherwise indicated. The demographic and clinical characteristics of infants suffering from RSV or HRV associated bronchiolits were compared using the Mann–Whitney test. Differences in the clinical score index values were analyzed using Student's t test.
Differences between infants with RSV or HRV infections and between RSV (A and B) or HRV (A and C) strains, in terms of the level of IFN lambda 1–3 measured in cells from nasopharyngeal washings, were compared using the Mann–Whitney test. Spearman's rho coefficient was calculated in order to assess the correlation between the level of IFN lambda 1 and IFN lambda 2–3 and between IFN lambda 1–3 and ISGs, demographic, clinical and RSV or HRV viral load. Differences in the IFN lambda1 levels in RSV infected infants divided into 3 groups on the basis of the respiratory rate were evaluated by using Kruskal–Wallis test. The significance was fixed at the 5% level. Analysis was performed with SPSS v.17.0 for Windows.
Results
Demographic, clinical and virological characteristics of infants suffering from RSV or HRV bronchiolitis
One hundred and eighteen infants, admitted over a period of 3 years to the Paediatric Department of Policlinico Umberto I University Hospital with a diagnosis of single RSV or HRV associated bronchiolitis were included (Table 1 ).
Table 1.
Demographic and clinical characteristics of infants suffering from RSV or HRV bronchiolitis.
| Item | RSV n = 78 |
HRV n = 40 |
p | RSVA n = 63 |
RSVB n = 15 |
p | HRVA n = 18 |
HRVC n = 22 |
p | |
|---|---|---|---|---|---|---|---|---|---|---|
| Age (months) | 2 (1–12) | 2.5 (1–12) | 0.562 | 2 (1–12) | 2 (1–5) | 0.185 | 2 (1–12) | 1.5 (1–5) | 0.059 | |
| Birth weight (kg) | 3.25 (2.4–4.5) | 3.11 (1.94–4.06) | 0.316 | 3.16 (2.4–4.4) | 3.29 (2.45–4.5) | 0.876 | 3.14 (2.08–3.79) | 2.96 (1.94–4.06) | 0.939 | |
| Weight on admission (kg) | 5 (2.8–8.6) | 4.9 (2.84–7.7) | 0.401 | 5 (2.8–8.6) | 5.425 (3–6.46) | 0.584 | 5.28 (2.8–7.7) | 4.54 (2.97–7.6) | 0.243 | |
| Male gender | 40/78 (51.28%) | 21/40 (52%) | 0.851 | 32/63 (50%) | 8/15 (53%) | 0.912 | 6/18 (32%) | 15/22 (68%) | 0.060 | |
| Caucasian race | 75/78 (96%) | 40/40 (100%) | 0.523 | 60/63 (95%) | 15/15 (100%) | 0.909 | 20/18 (90%) | 20/22 (91%) | 0.670 | |
| Hemoglobin (g/dl) | 11.05 (8.2–15.7) | 11.65 (9.3–16.6) | 0.062 | 11.05 (8.2–15.7) | 11.05 (9.7–13.7) | 0.675 | 11.6 (9.8–16.6) | 11.7 (9.3–14.3) | 0.778 | |
| C-reactive protein (mg/dl) | 0.35 (0–19.86) | 0.4 (0.03–3.97) | 0.530 | 0.22 (0.02–19.86) | 0.575 (0–3.26) | 0.617 | 0.26 (0.03–0.34) | 0.63 (0.04–3.97) | 0.494 | |
| White blood cells (cells/μl) | 10,820 (4770–29,250) | 11,465 (4170–18,100) | 0.742 | 10,815 (6770–20,440) | 12,600 (7730–29,250) | 0.220 | 10,000 (4170–14,700) | 12,600 (7240–18,100) | 0.418 | |
| Neutrophils (cells/μl) | 3449 (510–17,790) | 4025 (820–9720) | 0.661 | 4387 (3510–11,916) | 5209 (2460–17,790) | 0.167 | 3810 (820–2640) | 4706 (2614–9720) | 0.263 | |
| Lymphocytes (cells/μl) | 4440 (1803–10,136) | 4935.5 (2772–10,136) | 0.094 | 4342.5 (1803–10,136) | 4653.5 (4330–7880) | 0.263 | 4951 (4140–7890) | 4920 (2772–10,136) | 0.778 | |
| Eosinophils (cells/μl) | 70 (4–1081) | 169.5 (60–1316) | 0.004 | 81 (4–1081) | 52.5 (10–143.5) | 0.285 | 150 (60–1316) | 189 (119–1081) | 0.801 | |
| Platelets (106 cells/μl) | 0.49 (0.25–1.12) | 0.58 (0.27–0.83) | 0.701 | 0.486 (0.25–1.12) | 0.542 (0.25–0.71) | 0.928 | 0.525 (0.32–0.68) | 0.546 (0.27–0.83) | 0.264 | |
| Glycemia (mg/dl) (mean ± SD) | 106.83 ± 24.51 | 100.88 ± 26.17 | 0.229 | 104.95 ± 24.10 | 113.14 ± 25.84 | 0.655 | 110.76 ± 32.07 | 91 ± 13.65 | 0.064 | |
| Sodium (mEq/l) (mean ± SD) | 137.36 ± 3.09 | 138.07 ± 3.18 | 0.303 | 137.76 ± 3.08 | 135.91 ± 2.77 | 0.075 | 137 ± 2.58 | 139.5 ± 3.46 | 0.101 | |
| Presence of fever | 32/78 (41%) | 12/40 (30%) | 0.011 | 24/63 (38%) | 8/15 (53%) | 0.432 | 3/18 (17%) | 9/22 (40%) | 0.188 | |
| Clinical score index (mean ± SD)a,b | 4.4 ± 1.8 | 3.5 ± 1.5 | 0.008 | 4.35 ± 2.03 | 4.3 ± 2.18 | 0.930 | 3.76 ± 2.00 | 3.38 ± 1.98 | 0.554 | |
| Days in hospital (days) | 5 (1–15) | 5.5 (3–16) | 0.134 | 5 (1–13) | 4 (2–15) | 0.624 | 6 (3–7) | 5 (4–16) | 0.749 | |
Data are expressed as median (range) or frequency (percentage), unless otherwise indicated.
Differences between infants with RSV or HRV infection and between those with different RSV or HRV strains in terms of demographic and clinical parameters were compared using the Mann–Whitney test or Chi-Square test.
Differences in the clinical score index values were analyzed using Student's t test. The presence of statistical significances are highlighted in bold-face. Analysis was performed with SPSS v.17.0 for Windows.
The clinical severity was assigned to each infant with the range 0–8, based on respiratory rate (<45 breaths/min = 0, 45–60 breaths/min = 1, >60 breaths/min = 2), arterial oxygen saturation in room air (>95% = 0, 95–90% = 1, <90% = 2), presence of retractions (none = 0, present = 1, present + nasal fare = 2), and ability to feed (normal = 0, reduced = 1, endovenous = 2).
As far as virological characteristics are concerned, a total of 78 (66%) infants carried a single RSV infection whereas 40 (34%) had an HRV single infection. In particular 81% (63/78) of the RSV positive infants had a RSVA infection and the remaining had a RSVB infection. Among HRV infected infants, 45% (18/40) had an infection with HRVA and 55% (22/40) had an HRVC infection. Interestingly, no infants had an HRVB infection.
When we analyzed the demographic and clinical parameters of infants with RSV or HRV infection, there were no significant differences between infants with RSV or HRV infection and between infants with different RSV (A vs B) or HRV (A vs C) strains (Table 1). On the contrary the differences between infants with RSV or HRV infection were statistical significant when the clinical score index, and the percentage of infants with fever were analyzed (Table 1). Moreover, the number of eosinophils was lower in infants with RSV infection compared to those with HRV infection (Table 1).
Activation of type III IFN response in infants hospitalized for RSV or HRV associated bronchiolitis
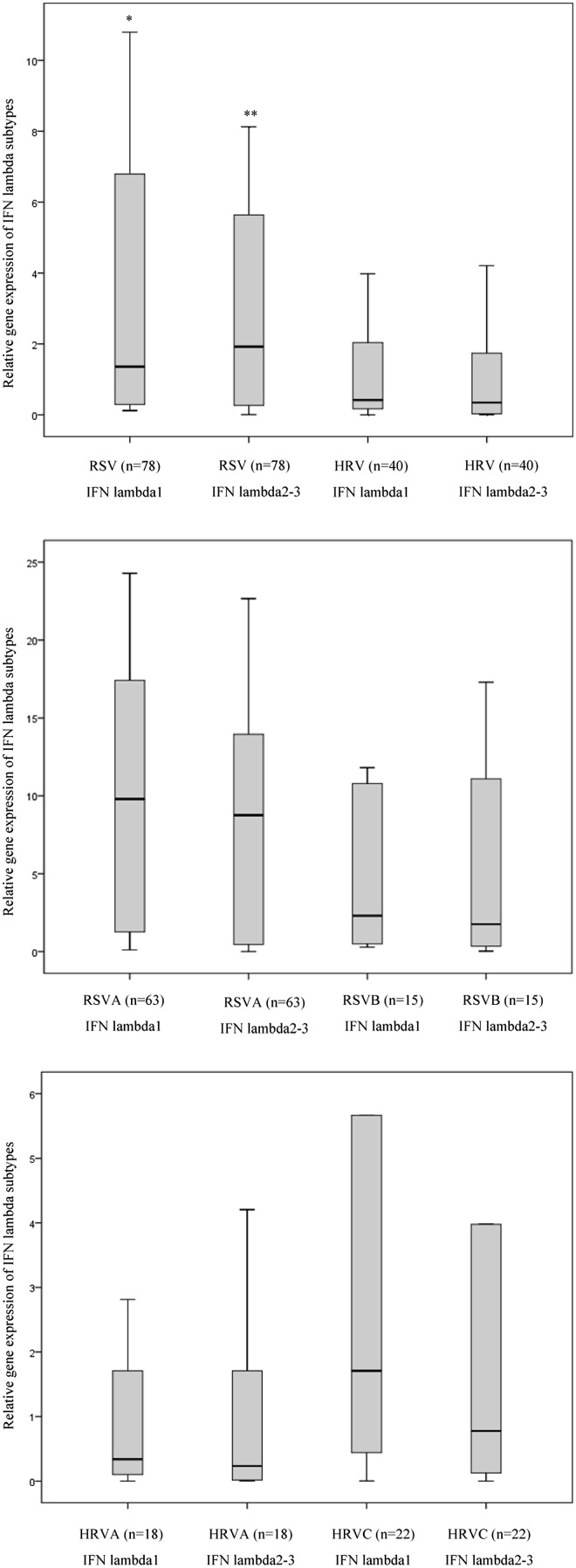
The airway transcription levels of IFN lambda 1 and IFN lambda 2–3 were evaluated using real-time RT-PCR in cells from nasopharyngeal washings collected from infants with RSV infection (n = 78) or HRV infections (n = 40). The gene expression level of IFN lambda 1 and IFN lambda 2–3 showed high variability between infants with RSV or HRV infection [(coefficient of variation >100%), Fig. 1 panel (A–C)].
Figure 1.
Gene expression of IFN lambda 1–3 during RSV or HRV bronchiolitis. The levels of type III IFNs were evaluated by using RT-Taqman based real time PCR assays in cells from nasopharyngeal washings collected from 118 infants suffering from RSV (n = 78) or HRV (n = 40) infections. The analysis of IFN lambda 1–3 –mRNAs levels was undertaken in infants suffering from RSV or HRV infection, independently of which type of viral strain was detected (Panel A), or considering the specific strain of RSV [A vs B, panel B] or HRV [A vs C, panel C]. Panel A: *p < 0.05 IFN lambda 1 (RSV) vs IFN lambda1 (HRV); **p < 0.05 IFN lambda 2–3 (RSV) vs IFN lambda 2–3 (HRV) by using the Mann–Whitney test. Panel B and C: p > 0.05 IFN lambda 1–3 (RSVA or HRVA) vs IFN lambda 1–3 (RSVB or HRVC) by using the Mann–Whitney test.
As reported in Fig. 1 (panel A) the transcript levels of IFN lambda 1and IFN lambda 2–3 in infants suffering from RSV infection were higher than in those with HRV infection [IFN lambda 1 (RSV vs HRV): p = 0.029; IFN lambda 2–3(RSV vs HRV): p = 0.015]. Furthermore, we found no differences between mRNA levels of IFN lambda 1 and those of the IFN lambda 2–3 in RSV or HRV infections (Fig. 1, panel A). No significant differences in transcript levels of type III IFN were observed between infants with different RSV (A vs B) or HRV (A vs C) strains (Fig. 1, panel B–C). In order to characterize the activation of type III IFN response during bronchiolitis, we also evaluated whether there was a coordinate activation of IFN lambda subtypes in the airway tract of infants suffering from bronchiolitis. Results indicate that in the respiratory tract of infants with RSV or HRV the transcript levels of IFN lambda 1 were significantly correlated with those of IFN lambda 2–3 (Table 2 ). Furthermore, considering that IFN lambda induces the expression of IFN-stimulated genes (ISGs), we evaluated whether there was a correlation between the expression of IFN lambda subtypes and that of well established ISGs, namely MxA and ISG56. Results indicate that type III IFN mRNA levels were significantly correlated with the transcript expression of MxA and ISG56 in infants with RSV or HRV bronchiolitis (Table 2).
Table 2.
Correlation between IFN lambda 1 and IFN lambda 2–3 and between IFN lambda 1–3 and ISGs (MxA and ISG56) in infants suffering RSV or HRV bronchiolitis.
| RSV |
HRV |
|||||||
|---|---|---|---|---|---|---|---|---|
| IFN lambda 1 |
IFN lambda 2–3 |
IFN lambda 1 |
IFN lambda 2–3 |
|||||
| p | r | p | r | p | r | p | r | |
| IFN lambda 1 | NAa | NA | 0.000 | 0.855 | NA | NA | 0.000 | 0.863 |
| MxA | 0.000 | 0.657 | 0.000 | 0.621 | 0.000 | 0.619 | 0.000 | 0.725 |
| ISG56 | 0.021 | 0.421 | 0.032 | 0.349 | 0.003 | 0.552 | 0.023 | 0.552 |
Spearman's rho coefficient was calculated in order to assess the correlation between the level of IFN lambda 1 and IFN lambda 2–3 and between IFN lambda 1–3 and ISGs (MxA and ISG56). Significant correlations are highlighted in bold-face. Analysis was performed with SPSS v.17.0 for Windows.
NA, not applicable.
Relationship between IFN lambda 1–3 levels and clinical course of RSV or HRV bronchiolitis
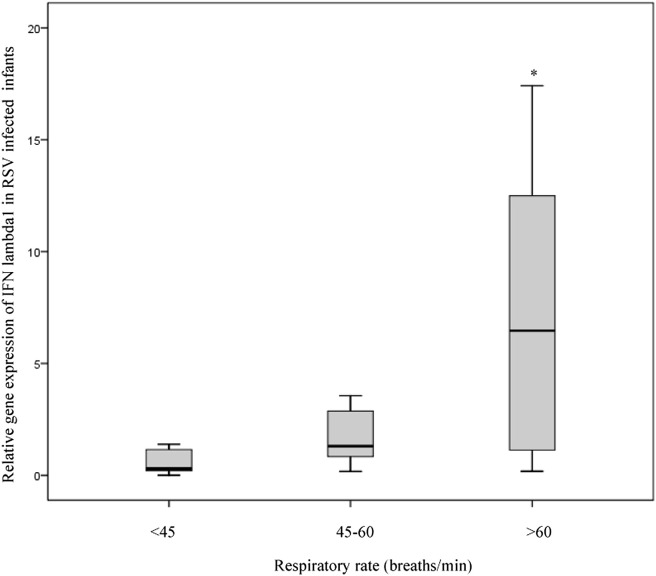
The relationship between patient data as independent variables and IFN lambda 1–3 mRNA levels measured in nasopharyngeal washings in infants with RSV or HRV bronchiolitis was analyzed (Table 3 ). We found a significant positive correlation between the transcript level of IFN lambda 1 and the clinical score index in infants with RSV infection (r = 0.350, p = 0.025) but not in those HRV infected (Table 3). In particular, IFN lambda 1 seems to be associated to an increase in the respiratory rate during RSV infection. Indeed, as showed in Fig. 2 , when RSV infected infants were divided into 3 groups on the basis of the respiratory rate (<45 breaths/min, 45–60 breaths/min, and >60 breaths/min), there was a significant difference between the groups (p = 0.022). Specifically, infants with >60 respiratory breaths per minute showed higher gene expression of IFN lambda 1 compared with infants with <45 or 45–60 respiratory breaths per minute. In contrast, we failed to detect any correlation between the IFN lambda 1–3 gene expression levels and age, weight, number of days of hospitalization, and several immunological and biochemical parameters (numbers of neutrophils, lymphocytes, eosinophils or platelets, and levels of glycemia, sodium, C-reactive protein and hemoglobin). In addition, no differences were detected in IFN lambda 1–3 gene expression for male and female and between infants with fever or without fever (data not shown). In an attempt to determine whether RSV or HRV load influences the gene expression of type III IFN, levels of RSV- or HRV-RNA were analyzed respect to the expression of IFN lambda subtypes. No significant correlations were observed between RSV or HRV load and the levels of IFN lambda 1–3(Table 3).
Table 3.
Correlation between IFN lambda 1–3 subtypes and host and virological parameters in infants suffering from RSV or HRV associated bronchiolitis.
| Host and virological parameters | RSV |
RSV |
HRV |
HRV |
||||
|---|---|---|---|---|---|---|---|---|
| IFN lambda 1 |
IFN lambda 2–3 |
IFN lambda 1 |
IFN lambda 2–3 |
|||||
| p | r | p | r | p | r | p | r | |
| Age (days) | 0.476 | 0.115 | 0.444 | 0.138 | 0.121 | −0.340 | 0.131 | −0.359 |
| Birth Weight (kg) | 0.061 | 0.295 | 0.05 | 0.394 | 0.905 | 0.027 | 0.991 | 0.003 |
| Weight on admission (kg) | 0.484 | 0.121 | 0.475 | 0.080 | 0.710 | −0.083 | 0.811 | −0.161 |
| Neutrophils (cells/μl) | 0.394 | 0.138 | 0.597 | 0.097 | 0.640 | −0.106 | 0.983 | −0.005 |
| Lymphocytes (cells/μl) | 0.759 | −0.050 | 0.122 | −0.279 | 0.743 | 0.074 | 0.764 | −0.074 |
| Eosinophils (cells/μl) | 0.525 | −0.103 | 0.394 | −0.156 | 0.954 | −0.013 | 0.197 | −0.310 |
| Platelets (cells/μl) | 0.515 | 0.106 | 0.694 | −0.072 | 0.440 | −0.173 | 0.557 | −0.144 |
| Glycemia (mg/dl) | 0.788 | 0.045 | 0.157 | 0.256 | 0.259 | −0.251 | 0.303 | −0.249 |
| Sodium (mEq/l) | 0.212 | −0.204 | 0.279 | −0.201 | 0.286 | 0.248 | 0.619 | −0.066 |
| C-reactive protein (mg/dl) | 0.387 | −0.142 | 0.707 | −0.070 | 0.715 | −0.083 | 0.895 | 0.010 |
| Hemoglobin (g/dl) | 0.811 | 0.039 | 0.558 | −0.108 | 0.601 | 0.118 | 0.764 | −0.166 |
| Clinical Score index | 0.025 | 0.350 | 0.204 | 0.227 | 0.308 | 0.228 | 0.352 | 0.226 |
| Days in hospital (days) | 0.832 | −0.034 | 0.352 | 0.167 | 0.345 | 0.211 | 0.988 | −0.004 |
| Viral load (number of copies/ml) | 0.800 | 0.041 | 0.723 | 0.064 | 0.697 | −0.090 | 0.918 | 0.028 |
Spearman's rho coefficient was calculated in order to assess the correlation between IFN lambda 1–3 mRNA levels and host and virological parameters. Significant correlations are highlighted in bold-face. Analysis was performed with SPSS v.17.0 for Windows. * The clinical severity was assigned to each infant with the range 0–8, based on respiratory rate (<45 breaths/m = 0, 45–60 breaths/m = 1, >60 breaths/m = 2), arterial oxygen saturation in room air (>95% = 0, 95–90% = 1, <90% = 2), presence of retractions (none = 0, present = 1, present + nasal fare = 2), and ability to feed (normal = 0, reduced = 1, endovenous = 2).
Figure 2.
Influence of IFN lambda 1 mRNA levels on the severity of respiratory rate in RSV infected infants. Gene expression of IFN lambda1 was examined in RSV positive infants (n = 78) divided into 3 groups on the basis of the respiratory rate (<45 breath/m, 45–60 breath/m, and >60 breath/m). A significant difference (p = 0.022) was observed among the groups by using Kruskal–Wallis test.
Discussion
It has been proposed that IFN lambdas are likely one of the main IFN produced during innate responses to respiratory viruses in the airway tract,14, 17, 19, 31 in line with the observations that the expression of the IFN lambda receptor is limited primarily to epithelial surfaces including that of the lung.14, 32 However the in vivo role of these cytokines in the host innate immune response to RSV or HRV infection is yet to be defined.
Our study gave some significant new insights into this complex issue. In particular, we found that in the respiratory tract of infants infected with RSV or HRV, there is a coordinate expression of different subtypes of IFN lambda: such an expression parallel also with those of MxA and ISG56, well established antiviral proteins induced by type I and III IFNs.12 This data could suggest that the airway tract is responsive to type III IFNs and that RSV or HRV caused a coordinate induction of IFN lambda subtypes that in turn can regulate antiviral pathways.12 However since both ISGs analyzed are also regulated by IFN type I which is known to be produced during viral infections and to share the same pathways with IFN lambda, it remains unclear to which extent IFN lambda might specifically contribute to antiviral immunity against RSV or HRV in infants suffering from bronchiolitis.
In this study we also compared the activation of IFN lambda 1–3 in the respiratory tract of infants suffering from RSV or HRV associated bronchiolitis. Results demonstrated that in cells collected from nasopharyngeal washings of RSV positive infants there are higher mRNA levels of type III IFNs compared to those observed in infants with HRV infection. This is particularly interesting considering that it is known that the RSV NS1 and NS2 proteins can suppress in vitro the induction of IFN lambda.33 However, whether such response reflects host reactions for counteracting the NS1/NS2 viral interference on IFN lambda induction is actually unknown. In our previous studies, we also observed a higher transcript level of ISGs as well as of most pattern recognition receptors in infants with RSV compared to those with HRV infections.26, 34 All these findings may suggest that RSV infection are generally associate to a more robust innate immune response compared to those caused by HRV. In agreement, Garcia et al. reported that concentrations of several cytokines in nasal wash tend to be higher in children with RSV than in those with HRV.35 In contrast, Jartii et al. found that children with HRV-associated wheezing episodes show increased concentrations of Th1 and Th2 cytokines compared with those with RSV.36 However they measured cytokines concentrations in serum rather than respiratory tract secretions during wheezing episodes.
As far as HRV is concerned, there are no studies on the evaluation of IFN lambda expression during HRV bronchiolitis: however wheezing during the first HRV infections is considered a risk factor for subsequent asthma development.37 Interestingly, a deficient IFN lambda response to HRV infection has been reported in childhood in asthmatic subjects irrespective of their atopic status and in atopic patients without asthma.38 Furthermore, Contoli et al. reported a deficient induction of IFN lambda by HRV in asthmatic primary bronchial epithelial cells and alveolar macrophages, which was highly correlated with severity of HRV-induced asthma exacerbation and virus load in experimentally infected human volunteers.27
In apparent contrast, the presence of higher IFN lambda levels were recently associated with worsening illness of HRV associated asthmatic exacerbations in children.39
Undoubtedly, the complex correlation between cytokine responses and viral infections deserves more studies to be performed carefully monitoring the various components of innate immunity during the natural course of respiratory viral infections.
In this study, we also found no differences in the expression of type III IFNs between infants infected with RSVA or RSVB subtypes and between those infected with HRVA or HRVC strains, suggesting that the specific strain of RSV or HRV would not affect diversely the rate of activation of antiviral response. As far as the influence of specific RSV or HRV strains on the clinical course of bronchiolitis is concerned, no significant differences in the clinical characteristics between RSV (A vs B) or HRV (A vs C) infected infants were also found. However it must be considered that the samples size of infants analyzed in this study when divided according to the specific RSV or HRV strain was too small. Conflicting results have been published on RSV (A or B) and HRV (A-C) association with recurrent gravity of respiratory diseases.10, 11, 40, 41, 42, 43 In particular, many, but not all, of the published studies11, 40, 41, 42, 43 found that HRV-C is associated with more severe lower respiratory disease and with wheezing, compared with HRV-A. Nonetheless, because of the small size of HRV-positive specimens within each individual diagnosis category, most studies suffered from low power and were unable to detect significant differences among specific diagnoses, as discussed in one of the largest study ever.43 Moreover, few data are available on the comparison of the ability of RSV A or B to modulate innate immune responses44 and no studies were performed on this issue for HRV A-C. Therefore we retain that no definite conclusions can be drawn on such issues.
Interestingly, this study also demonstrated that levels of IFN lambda 1 seem to be associated with severity of respiratory disease in RSV but not in HRV infected infants. This observation is not unusual for RSV pathogenesis. In fact, several studies have described severe RSV disease in children who have high levels of inflammatory cytokines and chemokines produced by innate immunity in respiratory secretions.45, 46 Whether or not IFN lambda contributes to augmentation of inflammation in our RSV positive infants is currently unknown. However, in addition to their antiviral effects, type III IFNs have been shown to play a critical role in regulating the adaptive immune response by acting directly on Th1/2 polarization and cytokine production.47, 48 In particular, IFN lambda 1 has been shown to increase production of IL-6, IL-8, and IL-10, with no concomitant increase in IL-1 or TNF, suggesting it may not directly engender local tissue destruction, but could contribute to the inflammatory process during bronchiolitis.47 A reciprocal control of IFN lambda 1 and Th2-associated cytokines seems also to exist.49 Indeed, it has been shown that there is a reciprocal regulation between IFN lambda1 and IL-4 or IL-13 which are well established Th2 cytokines associated with increased RSV disease severity.49 In addition, although type III IFNs seems to be activated during in vivo RSV infection, we observed that enhancement was not related to viral loads. Indirectly the lack of a direct effect of IFN lambdas on viral replication may reflect a more relevant role of the cross-talk between inflammatory citokines and type III IFN subtypes in determining the bronchiolitis clinical course.
Intriguingly, there are also indications that IFN lambda signaling may be resistant to feedback mechanisms targeting IFN alpha50 allowing it to be a more effective and durable antiviral and immunomodulator cytokine, at least under some circumstances, that could cause a prolonged, diffuse inflammatory response in the airway tract of RSV infected infants. The presence of this strong inflammatory response is consistent with the observation of the presence of string respiratory rates in RSV positive infants with increased IFN lambda1 levels. On the other hand, the reduced expression of IFN lambda subtypes observed in the airway tract of HRV infected infants compared to those with RSV infection, may not be enough to activate an excessive inflammatory response affecting the clinical course of bronchiolitis. In agreement with this hypothesis, Wang Q et al. found that MDA5 deficient mice with reduced IFN lambda productions show less HRV induced airway inflammation.51 A greater number of eosinophils was also observed in HRV infected infants than in those with RSV accordingly to our previous study.52 In this regards it has been recently demonstrated that eosinophils may contribute to antiviral immunity and play a beneficial role in limiting viral respiratory lung dysfunction.53 However, it is also believed that eosinophilia is one of the atopic features that may contribute to the higher risk to develop recurrent wheezing and asthma.37
Furthermore, in this study in line with previous studies4, 34, 52 a greater clinical severity in RSV infected infants than in those infected with HRV has been observed. Therefore it is conceivable that the presence of reduced airway IFN lambda response in HRV infected infants than in those with RSV infections could reflect the presence of a milder bronchiolititis clinical course caused by HRV compared to that associated to RSV. Indeed, Miller et al. reported that IFN lambda1 levels were higher in wheezing children infected with HRV compared with no-wheezing and increased with worsening symptoms.39 Alternatively, our data could indicate that an impaired IFN lambda production during HRV infection is present, not only in asthmatic subjects,27, 54 but also in infants with bronchiolitis which can be associated with the inability to control early virus replication and to mount an adequate Th1/Th2 immune response which in turn may impact on recurrent wheezing predisposition and an exacerbation pathogenesis.
These results on the presence of a positive correlation between IFN lambda1 levels and the clinical score of bronchiolitis in RSV infected infants does not seem to be in agreement with those obtained previously on the evaluation of ISGs levels in infants with bronchiolitis.34 This discrepancy, although unexpected, might be explained considering that several host and viral factors independently from IFN lambda can regulate, directly or indirectly, the ISGs production.12, 55 In addition it has been shown that the signaling pathways which lead to the activation of IFN regulatory factor can induce transcription of IFN-stimulated response elements without the involvement of IFNs.56, 57, 58 Thus it remains plausible that two different players of the same biological system might exert an opposite, complementary or simply additive effect on the clinical outcome of RSV bronchiolitis.
All these findings would be greatly strengthened by comparing the level of expression of mRNAs encoding type III IFNs with the level of expression of other cytokines or antiviral responses (e.g. type I IFNs) in the cells from nasopharyngeal washings derived from infants with RSV or HRV associated bronchiolitis. This analysis could allow us to deep understand the complex picture of the airway IFN lambda response as well as the intensity and the dynamic nature of the antiviral and inflammatory pathways associated to type III IFN response during pediatric lower respiratory tract infections. Unfortunately the collected material was just enough to perform the experiments shown in the present study and the above issues could not be addressed. Another important analysis which should be, but for the above reason have not been made, is the separate analysis of IFN lambda 2 and IFN lambda 3 subtypes expression in order to characterize the distinct contribute of these subtypes in RSV or HRV bronchiolitis. Further studies specifically aimed to address these important issues are needed.
In conclusion, we have shown for the first time to our knowledge that IFN lambda 1–3 are expressed in infants suffering from bronchiolitis although their levels may be different on the basis of which virus, RSV or HRV, has been detected in the respiratory tract. In addition, we have demonstrated that IFN lambda 1 can influence the severity of bronchiolitis caused by RSV, but not by HRV, suggesting that the rate of activation of type III IFN response may act as a double-edged sword in some circumstance during pediatric respiratory viral infections. However, it must be underlined that several factors associated or not with the IFN system may influence the clinical severity of bronchiolitis.59 The latter issue is exemplified by our observations about the opposite effect exerted by ISGs34 and IFN lambda (this study) on the clinical outcome of bronchiolitis.
All these findings highlight the importance of studying the interplay between the pathways of IFN lambda subtypes and those of other inflammatory cytokines or chemokines in order to deeply understand the influence of type III IFN response on the clinical course of respiratory diseases.
Acknowledgments
This work was supported by a grant from “Sapienza” University of Rome to Carolina Scagnolari (“Ricerche Universitarie”, Anno 2012, prot C26A12X2HP).
References
- 1.Garibaldi B.T., Illei P., Danoff S.K. Bronchiolitis. Immunol Allergy Clin North Am. 2012;4:601–619. doi: 10.1016/j.iac.2012.08.002. [DOI] [PubMed] [Google Scholar]
- 2.Mullins J.A., Lamonte A.C., Bresee J.S., Anderson L.J. Substantial variability in community respiratory syncytial virus season timing. Pediatr Infect Dis J. 2003;22:857–862. doi: 10.1097/01.inf.0000090921.21313.d3. [DOI] [PubMed] [Google Scholar]
- 3.Fitzgerald D.A. Viral bronchiolitis for the clinician. J Paediatr Child Health. 2011;4:160–166. doi: 10.1111/j.1440-1754.2010.01735.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Midulla F., Scagnolari C., Bonci E., Pierangeli A., Antonelli G., De Angelis D. Respiratory syncytial virus, human bocavirus and rhinovirus bronchiolitis in infants. Arch Dis Child. 2010;95:35–41. doi: 10.1136/adc.2008.153361. [DOI] [PubMed] [Google Scholar]
- 5.Friedlander S.L., Busse W.W. The role of rhinovirus in asthma exacerbations. J Allergy Clin Immunol. 2005;116:267–273. doi: 10.1016/j.jaci.2005.06.003. [DOI] [PubMed] [Google Scholar]
- 6.Arden K.E., Mackay I.M. Newly identified human rhinoviruses: molecular methods heat up the cold viruses. Rev Med Virol. 2010;20:156–176. doi: 10.1002/rmv.644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bont L. Current concepts of the pathogenesis of RSV bronchiolitis. Adv Exp Med Biol. 2009;634:31–40. doi: 10.1007/978-0-387-79838-7_3. [DOI] [PubMed] [Google Scholar]
- 8.Bont L., Ramilo O. The relationship between RSV bronchiolitis and recurrent wheeze: the chicken and the egg. Early Hum Dev. 2011;87(Suppl. 1):S51–S54. doi: 10.1016/j.earlhumdev.2011.01.011. [DOI] [PubMed] [Google Scholar]
- 9.Halfhide C., Smyth R.L. Innate immune response and bronchiolitis and preschool recurrent wheeze. Paediatr Respir Rev. 2008;9:251–262. doi: 10.1016/j.prrv.2008.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.González P.A., Bueno S.M., Carreño L.J., Riedel C.A., Kalergis A.M. Respiratory syncytial virus infection and immunity. Rev Med Virol. 2012;22:230–244. doi: 10.1002/rmv.1704. [DOI] [PubMed] [Google Scholar]
- 11.Jacobs S.E., Lamson D.M., St George K., Walsh T.J. Human rhinoviruses. Clin Microbiol Rev. 2013;26:135–162. doi: 10.1128/CMR.00077-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Scagnolari C., Antonelli G. Antiviral activity of the interferon α family: biological and pharmacological aspects of the treatment of chronic hepatitis C. Expert Opin Biol Ther. 2013;5:693–711. doi: 10.1517/14712598.2013.764409. [DOI] [PubMed] [Google Scholar]
- 13.Sheppard P., Kindsvogel W., Xu W., Henderson K., Schlutsmeyer S., Whitmore T.E. IL-28, IL-29 and their class II cytokine receptor IL-28R. Nat Immunol. 2003;4:63–68. doi: 10.1038/ni873. [DOI] [PubMed] [Google Scholar]
- 14.Sommereyns C., Paul S., Staeheli P., Michiels T. IFN-lambda (IFN-lambda) is expressed in a tissue-dependent fashion and primarily acts on epithelial cells in vivo. PLoS Pathog. 2008;4:e1000017. doi: 10.1371/journal.ppat.1000017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ank N., Iversen M.B., Bartholdy C., Staeheli P., Hartmann R., Jensen U.B. An important role for type III interferon (IFN-lambda/IL-28) in TLR-induced antiviral activity. J Immunol. 2008;180:2474–2485. doi: 10.4049/jimmunol.180.4.2474. [DOI] [PubMed] [Google Scholar]
- 16.Mordstein M., Kochs G., Dumoutier L., Renauld J.C., Paludan S.R., Klucher K. Interferon-lambda contributes to innate immunity of mice against influenza A virus but not against hepatotropic viruses. PLoS Pathog. 2008;4:e1000151. doi: 10.1371/journal.ppat.1000151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Khaitov M.R., Laza-Stanca V., Edwards M.R., Walton R.P., Rohde G., Contoli M. Respiratory virus induction of alpha-, beta- and lambda-interferons in bronchial epithelial cells and peripheral blood mononuclear cells. Allergy. 2009;64:375–386. doi: 10.1111/j.1398-9995.2008.01826.x. [DOI] [PubMed] [Google Scholar]
- 18.Okabayashi T., Kojima T., Masaki T., Yokota S., Imaizumi T., Tsutsumi H. Type-III interferon, not type-I, is the predominant interferon induced by respiratory viruses in nasal epithelial cells. Virus Res. 2011;160:360–366. doi: 10.1016/j.virusres.2011.07.011. [DOI] [PubMed] [Google Scholar]
- 19.Mordstein M., Neugebauer E., Ditt V., Jessen B., Rieger T., Falcone V. Lambda interferon renders epithelial cells of the respiratory and gastrointestinal tracts resistant to viral infections. J Virol. 2010;84:5670–5677. doi: 10.1128/JVI.00272-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Scagnolari C., Midulla F., Riva E., Monteleone K., Solimini A., Bonci E. Evaluation of interleukin 28B single nucleotide polymorphisms in infants suffering from bronchiolitis. Virus Res. 2012;165:236–240. doi: 10.1016/j.virusres.2012.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wilson J., Rowlands K., Rockett K., Moore C., Lockhart E., Sharland M. Genetic variation at the IL 10 gene locus is associated with severity of respiratory syncytial virus bronchiolitis. J Infect Dis. 2005;191:1705–1709. doi: 10.1086/429636. [DOI] [PubMed] [Google Scholar]
- 22.Pierangeli A., Gentile M., Di Marco P., Pagnotti P., Scagnolari C., Trombetti S. Detection and typing by molecular techniques of respiratory viruses in children hospitalized for acute respiratory infection in Rome, Italy. J Med Virol. 2007;79:463–468. doi: 10.1002/jmv.20832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pierangeli A., Scagnolari C., Trombetti S., Grossi R., Battaglia M., Moretti C. Human bocavirus infection in hospitalized children in Italy. Infuenza Other Respi Viruses. 2008;2:175–179. doi: 10.1111/j.1750-2659.2008.00057.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Peret T.C., Hall C.B., Schnabel K.C., Golub J.A., Anderson L.J. Circulation patterns of genetically distinct group A and B strains of human respiratory syncytial virus in a community. J Gen Virol. 1998;79:2221–2229. doi: 10.1099/0022-1317-79-9-2221. [DOI] [PubMed] [Google Scholar]
- 25.Kiang D., Kalra I., Yagi S., Louie J.K., Boushey H., Boothby J. Assay for 5' noncoding region analysis of all human rhinovirus prototype strains. J Clin Microbiol. 2008;46:3736–3745. doi: 10.1128/JCM.00674-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Scagnolari C., Midulla F., Pierangeli A., Moretti C., Bonci E., Berardi R. Gene expression of nucleic acid-sensing pattern recognition receptors in children hospitalized for respiratory syncytial virus-associated acute bronchiolitis. Clin Vaccine Immunol. 2009;16:816–823. doi: 10.1128/CVI.00445-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Contoli M., Message S.D., Laza-Stanca V., Edwards M.R., Wark P.A.B., Bartlett N.W. Role of deficient type III interferon-λ production in asthma exacerbations. Nat Med. 2006;12:1023–1026. doi: 10.1038/nm1462. [DOI] [PubMed] [Google Scholar]
- 28.Scagnolari C., Midulla F., Selvaggi C., Monteleone K., Bonci E., Papoff P. Evaluation of viral load in infants hospitalized with bronchiolitis caused by respiratory syncytial virus. Med Microbiol Immunol. 2012;201:311–317. doi: 10.1007/s00430-012-0233-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tapparel C., Cordey S., Van Belle S., Turin L., Lee W.-M., Regamey N. New molecular detection tools adapted to emerging rhinoviruses and enteroviruses. J Clin Microbiol. 2009;47:1742–1749. doi: 10.1128/JCM.02339-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hu A., Colella M., Tam J.S., Rappaport R., Cheng S.M. Simultaneous detection, subgrouping, and quantitation of respiratory syncytial virus A and B by real-time PCR. J Clin Microbiol. 2003;41:149–154. doi: 10.1128/JCM.41.1.149-154.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Jewell N.A., Cline T., Mertz S.E., Smirnov S.V., Flaño E., Schindler C. Lambda interferon is the predominant interferon induced by influenza A virus infection in vivo. J Virol. 2010;84:11515–11522. doi: 10.1128/JVI.01703-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Brand S., Beigel F., Olszak T., Zitzmann K., Eichhorst S.T., Otte J.M. IL-28A and IL-29 mediate antiproliferative and antiviral signals in intestinal epithelial cells and murine CMV infection increases colonic IL-28A expression. Am J Physiol Gastrointest Liver Physiol. 2005;289:G960–G968. doi: 10.1152/ajpgi.00126.2005. [DOI] [PubMed] [Google Scholar]
- 33.Spann K.M., Tran K.C., Chi B., Rabin R.L., Collins P.L. Suppression of the induction of alpha, beta, and lambda interferons by the NS1 and NS2 proteins of human respiratory syncytial virus in human epithelial cells and macrophages. J Virol. 2004;78:4363–4369. doi: 10.1128/JVI.78.8.4363-4369.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Scagnolari C., Midulla F., Trombetti S., Pierangeli A., Tromba V., Grossi R. Upregulation of interferon-induced genes in infants with virus-associated acute bronchiolitis. Exp Biol Med. 2007;232:1355–1359. doi: 10.3181/0705-BC-124. [DOI] [PubMed] [Google Scholar]
- 35.García C., Soriano-Fallas A., Lozano J., Leos N., Gomez A.M., Ramilo O. Decreased innate immune cytokine responses correlate with disease severity in children with respiratory syncytial virus and human rhinovirus bronchiolitis. Pediatr Infect Dis J. 2012;3:86–89. doi: 10.1097/INF.0b013e31822dc8c1. [DOI] [PubMed] [Google Scholar]
- 36.Jartti T., Paul-Anttila M., Lehtinen P., Parikka V., Vuorinen T., Simell O. Systemic T-helper and T-regulatory cell type cytokine responses in rhinovirus vs. respiratory syncytial virus induced early wheezing: an observational study. Respir Res. 2009;10:85. doi: 10.1186/1465-9921-10-85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Midulla F., Pierangeli A., Cangiano G., Bonci E., Salvadei S., Scagnolari C. Rhinovirus bronchiolitis and recurrent wheezing: 1-year follow-up. Eur Respir J. 2012;39:396–402. doi: 10.1183/09031936.00188210. [DOI] [PubMed] [Google Scholar]
- 38.Baraldo S., Contoli M., Bazzan E., Turato G., Padovani A., Marku B. Deficient antiviral immune responses in childhood: distinct roles of atopy and asthma. J Allergy Clin Immunol. 2012;130:1307–1314. doi: 10.1016/j.jaci.2012.08.005. [DOI] [PubMed] [Google Scholar]
- 39.Miller E.K., Hernandez J.Z., Wimmenauer V., Shepherd B.E., Hijano D., Libster R. A mechanistic role for type III IFN-λ1 in asthma exacerbations mediated by human rhinoviruses. Am J Respir Crit Care Med. 2012;185:508–516. doi: 10.1164/rccm.201108-1462OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Piralla A., Lilleri D., Sarasini A., Marchi A., Zecca M., Stronati M. Human rhinovirus and human respiratory enterovirus (EV68 and EV104) infections in hospitalized patients in Italy, 2008–2009. Diagn Microbiol Infect Dis. 2012;73:162–167. doi: 10.1016/j.diagmicrobio.2012.02.019. [DOI] [PubMed] [Google Scholar]
- 41.Iwane M.K., Prill M.M., Lu X., Miller E.K., Edwards K.M., Hall C.B. Human rhinovirus species associated with hospitalizations for acute respiratory illness in young US children. J Infect Dis. 2011;204:1702–1710. doi: 10.1093/infdis/jir634. [DOI] [PubMed] [Google Scholar]
- 42.Pierangeli A., Ciccozzi M., Chiavelli S., Concato C., Giovanetti M., Cella E. Molecular epidemiology and genetic diversity of human rhinovirus affecting hospitalized children in Rome. Med Microbiol Immunol. 2013;202(4):303–311. doi: 10.1007/s00430-013-0296-z. [DOI] [PubMed] [Google Scholar]
- 43.Linder J.E., Kraft D.C., Mohamed Y., Lu Z., Heil L., Tollefson S. Human rhinovirus C: age, season, and lower respiratory illness over the past 3 decades. J Allergy Clin Immunol. 2012;131:69–77. doi: 10.1016/j.jaci.2012.09.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bermejo-Martin J.F., Tenorio A., Ortiz de Lejarazu R., Eiros J.M., Matías V., Dominguez G.M. Similar cytokine profiles in response to infection with respiratory syncytial virus type A and type B in the upper respiratory tract in infants. Intervirology. 2008;51:112–115. doi: 10.1159/000134268. [DOI] [PubMed] [Google Scholar]
- 45.Sheeran P., Jafri H., Carubelli C., Saavedra J., Johnson C., Krisher K. Elevated cytokine concentrations in the nasopharyngeal and tracheal secretions of children with respiratory syncytial virus disease. Pediatr Infect Dis J. 1999;18:115–122. doi: 10.1097/00006454-199902000-00007. [DOI] [PubMed] [Google Scholar]
- 46.Noah T.L., Ivins S.S., Murphy P., Kazachkova I., Moats-Staats B., Henderson F.W. Chemokines and inflammation in the nasal passages of infants with respiratory syncytial virus bronchiolitis. Clin Immunol. 2002;104:86–95. doi: 10.1006/clim.2002.5248. [DOI] [PubMed] [Google Scholar]
- 47.Jordan W.J., Eskdale J., Boniotto M., Rodia M., Kellner D., Gallagher G. Modulation of the human cytokine response by interferon lambda-1 (IFN-lambda1/IL-29) Genes Immun. 2007;8:13–21. doi: 10.1038/sj.gene.6364348. [DOI] [PubMed] [Google Scholar]
- 48.Pekarek V., Srinivas S., Eskdale J., Gallagher G. Interferon lambda-1 (IFN-lambda1/IL-29) induces ELR(-) CXC chemokine mRNA in human peripheral blood mononuclear cells, in an IFN-gamma-independent manner. Genes Immun. 2007;8:177–180. doi: 10.1038/sj.gene.6364372. [DOI] [PubMed] [Google Scholar]
- 49.Dai J., Megjugorac N.J., Gallagher G.E., Yu R.Y., Gallagher G. IFN-lambda1 (IL-29) inhibits GATA3 expression and suppresses Th2 responses in human naive and memory T cells. Blood. 2009;113:5829–5838. doi: 10.1182/blood-2008-09-179507. [DOI] [PubMed] [Google Scholar]
- 50.Francois-Newton V., Magno de Freitas A.G., Payelle-Brogard B., Monneron D., Pichard-Garcia L., Piehler J. USP18-based negative feedback control is induced by type I and type III interferons and specifically inactivates interferon alpha response. PLoS One. 2011;6:e22200. doi: 10.1371/journal.pone.0022200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wang Q., Miller D.J., Bowman E.R., Nagarkar D.R., Schneider D., Zhao Y. MDA5 and TLR3 initiate pro-inflammatory signaling pathways leading to rhinovirus-induced airways inflammation and hyperresponsiveness. PLoS Pathog. 2011;7(5):e1002070. doi: 10.1371/journal.ppat.1002070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Papoff P., Moretti C., Cangiano G., Bonci E., Roggini M., Pierangeli A. Incidence and predisposing factors for severe disease in previously healthy term infants experiencing their first episode of bronchiolitis. Acta Paediatr. 2011;100:17–23. doi: 10.1111/j.1651-2227.2011.02181.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Phipps S., Lam C.E., Mahalingam S., Newhouse M., Ramirez R., Rosenberg H.F. Eosinophils contribute to innate antiviral immunity and promote clearance of respiratory syncytial virus. Blood. 2007;110:1578–1786. doi: 10.1182/blood-2007-01-071340. [DOI] [PubMed] [Google Scholar]
- 54.Edwards M.R., Regamey N., Vareille M., Kieninger E., Gupta A., Shoemark A. Impaired innate interferon induction in severe therapy resistant atopic asthmatic children. Mucosal Immunol. 2013;6:797–806. doi: 10.1038/mi.2012.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lo M.S., Brazas R.M., Holtzman M.J. Respiratory syncytial virus nonstructural proteins NS1 and NS2 mediate inhibition of Stat2 expression and alpha/beta interferon responsiveness. J Virol. 2005;79(14):9315–9319. doi: 10.1128/JVI.79.14.9315-9319.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Grandvaux N., Servant M.J., TenOever B., Sen G.C., Balachandran S., Barber G.N. Transcriptional profiling of interferon regulatory factor 3 target genes: direct involvement in the regulation of interferon-stimulated genes. J Virol. 2002;76:5532–5539. doi: 10.1128/JVI.76.11.5532-5539.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Sato M., Suemori H., Hata N., Asagiri M., Ogasawara K., Nakao K. Distinct and essential roles of transcription factors IRF-3 and IRF-7 in response to viruses for IFN-alpha/beta gene induction. Immunity. 2000;13:539–548. doi: 10.1016/s1074-7613(00)00053-4. [DOI] [PubMed] [Google Scholar]
- 58.Servant M.J., Grandvaux N., Hiscott J. Multiple signaling pathways leading to the activation of interferon regulatory factor 3. Biochem Pharmacol. 2002;64:985–992. doi: 10.1016/s0006-2952(02)01165-6. [DOI] [PubMed] [Google Scholar]
- 59.Tregoning J.S., Schwarze J. Respiratory viral infections in infants: causes, clinical symptoms, virology, and immunology. Clin Microbiol Rev. 2010;23(1):74–98. doi: 10.1128/CMR.00032-09. [DOI] [PMC free article] [PubMed] [Google Scholar]