Highlights
-
•
An RT-LAMP method was developed for detection of hMPV A and hMPV B.
-
•
RT-LAMP, RT-PCR and real-time SYBR PCR methods were compared.
-
•
The detection limit of the genotype-specific hMPV RT-LAMP assay was 10 percent of that of conventional RT-PCR.
-
•
It is worthy to recommend this method in clinical application, especially in resource-limited laboratories.
Keywords: Human metapneumovirus, Reverse transcription loop-mediated isothermal amplification, Clinical specimen
Abstract
Human metapneumovirus (hMPV) has been recognized as an important pathogen for acute respiratory infections in children worldwide and classified into genotypes A and B. Reverse transcription loop-mediated isothermal amplification (RT-LAMP) assay is a rapid diagnostic method for detecting nucleic acids with a single step under isothermal conditions in less than 1 h. RT-LAMP targeting the M gene of hMPV was developed for detecting and identifying hMPV genotypes A and B. The detection limit of the genotype-specific hMPV RT-LAMP assay was 10 times greater than that of conventional reverse transcription polymerase chain reaction (RT-PCR). No cross-reactivity was found with respiratory syncytial virus, parainfluenza virus 1–3, adenovirus, human bocavirus, human rhinovirus, influenza virus A and B, human coronaviruses and enteroviruses. One hundred and fifteen clinical specimens were detected for hMPV genotypes A and B with RT-LAMP, RT-PCR and real-time SYBR PCR. Kappa coefficients showed that there was a good agreement among these three methods. Compared with RT-PCR and real-time SYBR PCR, the genotype-specific RT-LAMP showed better specificity, sensitivity and is more convenient to perform with reduced turn-around time.
1. Introduction
Human metapneumovirus (hMPV) has been recognized as an important virus associated with respiratory tract diseases in infants and children. It is the first member of the Metapneumovirus genus in Pneumoviriae, Paramyxoviridae, associated with human diseases (van den Hoogen et al., 2001). Currently, sequence analysis for hMPV isolates indicates that there are two major genetic lineages (genotypes A and B) (Bastien et al., 2003, Biacchesi et al., 2003). As an important viral pathogen causing acute lower respiratory infections in children, hMPV is the second viral pathogen of bronchiolitis during infancy and early childhood next to respiratory syncytial virus (RSV) (Skiadopoulos et al., 2006). HMPV associated acute respiratory infections in infants and young children was reported by the same research group of this study in 2003 (Zhu et al., 2003), which was the first report about hMPV infection in China. Since then, surveillance of hMPV infections in children in Beijing has been conducted by antigen test, virus isolation and reverse transcription-polymerase chain reaction (RT-PCR) (Zhu et al., 2007, Wang et al., 2010).
Due to the difficulty of virus isolation and the shortage of specific antibody for antigen test, the technique currently used for rapid detecting hMPV is RT-PCR (Lopez-Huertas et al., 2005, Zhu et al., 2007). However, RT-PCR has some limitations, such as time-consuming, contamination and need for expensive equipment, especially for application in clinical diagnostic laboratories in hospitals.
With the development of molecular techniques, the methods for virus detection are constantly updated and improved. Loop-mediated isothermal amplification (LAMP) was developed originally as a rapid diagnostic method for the detection of nucleic acids under isothermal conditions (Notomi et al., 2000, Mori and Notomi, 2009). The LAMP assay utilizes four primers, which are specific for six gene sequences on the targeted DNA. The products can be observed as a white precipitant of magnesium pyrophosphate. LAMP and RT-LAMP have been used for detecting various DNA and RNA viruses (Mori et al., 2004, Yoneyama et al., 2007, Curtis et al., 2009, Kubo et al., 2010, Luo et al., 2011). To obtain a rapid and reliable assay to detect hMPV from clinical specimens collected from pediatric patients with acute respiratory infections, a RT-LAMP assay was developed for identifying hMPV A and hMPV B from clinical specimens under isothermal conditions at 63 °C within 60 min and the results can be judged by naked eyes. This assay could be used as a diagnostic method in laboratories, especially in those hospitals or clinics with limited equipment.
2. Materials and methods
2.1. Primer designing
The sequences of M genes from hMPV genotypes A and B were retrieved from GenBank and aligned by DNAStar software. The highly conserved regions of the M genes of hMPV genotypes A (GenBank accession no. DQ843659) and B (GenBank accession no. DQ843658) were chosen as the targets for primer designing. Oligonucleotide genotype-specific primers for hMPV were designed with the online LAMP primer design software PrimerExplorer V4 (http://primerexplorer.jp/elamp4.0.0/index.html) and synthesized by Invitrogen (Shanghai, China). The locations and sequences for the set of four primers including outer primers (F3 and B3), a forward inner primer (FIP) and a backward inner primer (BIP) are shown in Table 1 .
Table 1.
Primers for the genotype-specific hMPV RT-LAMP assay.
| Genotype | Primera | Sequence (5′–3′) | Genome positionb |
|---|---|---|---|
| A | A-F3 | TTCAGGCCAATACACCAC | 2287–2304 |
| A-B3 | GTCAAGTGCTACAGTCGC | 2445–2462 | |
| A-FIP | ACTTTGTGATGCAGCATACAGA-TTTTCAGTTCTGCTTGATCAGCT | (2348–2369) + TTTT + (2308–2326) | |
| A-BIP | ACTAAAAGTGAATGCATCAGCYC-TTTTACTTCAAACYTTTTGGGAAG | (2378–2400) + TTTT + (2421–2440) | |
| B | B-F3 | GTGTCAAAATTTGTGAGTTCAG | 2545–2566 |
| B-B3 | GGCYTCRCTGCTTATTGC | 2707–2724 | |
| B-FIP | TTCTCTAGGTCCATGAAGTCACA-TTTTCAAATCAGTTGGCAAAAAGACA | (2608–2630) + TTTT + (2568–2589) | |
| B-BIP | TACCTGTGACAATACCAGCATTC-TTTTCAGTRGCTGACTCACTCTCT | (2636–2658) + TTTT + (2679–2698) | |
F: forward outer primer; B: backward outer primer; FIP: forward inner primer; BIP: backward inner primer.
2.2. Clinical specimens and RNA extraction
Clinical specimens including throat swabs from outpatients and nasopharyngeal aspirates from the hospitalized were collected from infants and young children with acute respiratory infections when they visited the Affiliated Children's Hospital of Capital Institute of Pediatrics, Beijing, China. RNA was extracted from the specimens using the QIAamp Viral RNA Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer's instructions and was stored at −80 °C until further processing.
This project was approved by the Institutional Review Board of Capital Institute of Pediatrics (IRB number: SHERLL2011039). This project did not involve personal data of the patients, such as name, age, and gender.
2.3. Genotype-specific RT-LAMP for hMPV
Two RT-LAMP reactions were assembled, including hMPV genotype A specific RT-LAMP and genotype B specific RT-LAMP. Each sample was detected for hMPV genotype A and B by genotype-specific RT-LAMP simultaneously. The RT-LAMP was performed in a one-step reaction in a 25 μL mixture containing 5 μL RNA template, 18 μL reaction solution of Loopamp RNA Amplification Kit (RT-LAMP) (Eiken Chemical, Tokyo, Japan) [12.5 μL 2 × Reaction Mix, 1 μL Enzyme Mix, 4.5 μL distilled water] and 0.5 μL of each of the primers (F3 and B3, 10 μM; FIP and BIP, 80 μM). The template RNA was replaced by distilled water as a negative control. The reaction mixture was incubated at 63 °C for 1.5 h and heated at 80 °C for 5 min to terminate the reaction.
A Loopamp® real-time LA-320C turbidimeter (Eiken Chemical, Tokyo, Japan) was used to monitor the accumulation of magnesium pyrophosphate spectrophotometrically at 650 nm. The amplification of the targeted RNA was indicated by the turbidity real-timely, and the turbidity was shown after the automatically correction of the turbidity of amplification. Each graphic shown by the turbidimeter only includes the results of eight reactions. In the mean while, the result of the reaction could be observed by naked eyes under the natural light. When a white precipitate of magnesium pyrophosphate appeared, the reaction was considered as positive.
In order to make the results easier observed under the natural light or UV light, the Fluorescent Detection Reagent (FDR) (Eiken Chemical, Tokyo, Japan) (1 μL) was added to the reaction tube before amplification started. FDR (calcein) binds with manganese ions and remains quenched. When the LAMP amplifying reaction occurs, a by-product, pyrophosphate, deprives calcein of the manganese ion, which results in the emission of fluorescence. When the free calcein binds to the magnesium ions in the reaction mixture, the fluorescence emission becomes stronger. After amplification, the positive reaction will be indicated by a color change from orange to light green under the natural light or by a green fluorescent change from light to brighter under UV light.
2.4. Sensitivity of genotype-specific RT-LAMP assays for hMPV
The recombinant plasmids with inserted M gene fragments of hMPV genotypes A (pUCm-T-A) or B (pUCm-T-B) amplified from isolated strains A (GenBank accession no. DQ843659) and B (GenBank accession no. DQ843658) were generated previously (Zhu et al., 2005). The recombinant plasmids pUCm-T-A and pUCm-T-B were linearized by vector-specific restriction enzyme Sal I (Promega, Madison, USA) and then transcribed in vitro to obtain RNA using the T7 RiboMAX™ Express Large Scale RNA Production System (Promega, Madison, USA) following the manufacturer's instructions. The RNA was purified and quantified by a Qubit® nucleic acid protein quantitative instrument (Invitrogen, Oregon, USA). The RNA copy numbers of hMPV A and B were 4.33 × 1010 copies/μL and 4.53 × 1010 copies/μL, respectively. The 10-fold serial dilutions of RNA ranging from 10−6 to 10−13 were applied to determine the minimum limit of hMPV genotype-specific RT-LAMP assay. In addition, the RNA of hMPV A and B was mixed by the same concentration (the concentrations of A and B were both 1 × 109 copies/μL), then made a 10-fold serial dilution, and tested by the genotype-specific RT-LAMP assay, respectively.
All the negative controls presented the turbidities below 0.05 within 60 min in the preliminary experiments. The threshold value of amplification was recommended by the manufacturer's instructions at 0.1 or 0.2 of the turbidity of judgment. According to the results of genotype-specific RT-LAMP using serial diluted transcribed RNA from recombinant plasmids (pUCm-T-A and pUCm-T-B), the threshold value of the RT-LAMP was defined at 0.2 of the turbidity of judgment within 60 min.
2.5. Specificity of genotype-specific RT-LAMP assays
To evaluate the specificity of the genotype-specific RT-LAMP assays for hMPV, viral nucleic acids were extracted from clinical respiratory samples which were positive for respiratory syncytial virus (RSV), parainfluenza virus 1–3 (PIV1-3), adenovirus (ADV), human bocavirus (HBOV), human rhinovirus (HRV), influenza virus A and B, human coronaviruses (HCoV, including 229E, OC43, HKU1 and NL63) and enteroviruses (EV), and then tested by the genotype-specific RT-LAMP assays for hMPV. These positive samples mentioned above had been tested by PCR or RT-PCR described previously (Zhao et al., 2003, van der Hoek et al., 2004, Moes et al., 2005, Woo et al., 2005, Zhao et al., 2006a, Zhao et al., 2006b, Zhao et al., 2007, Deng et al., 2010) and xTAG Respiratory Viral panel Fast (RVP Fast) assay (submitted for publication). The primers targeting 5′UTR used for EV detection by RT-PCR were as follows: EVf 5′-TCCGGCCCCTGAATGCGGC-3′, EVr 5′-CACCGGATGGCCAATCCAATAG-3′. The positive samples with influenza viruses were confirmed by real-time RT-PCR recommended by the National Influenza Center, China, Center for Disease Prevention and Control. The specific primers and probe for influenza A were as follows (Flu A, targeting M gene; Flu B, targeting NS1 gene): FluA-For 5′-GAC CRA TCC TGT CAC CTC TGA C-3′, FluA-Rev 5′-GGG CAT TYT GGA CAA AKC GTC TAC G-3′, Flu A-Probe 5′-FAM TGC AGT CCT CGC TCA CTG GGC ACG-BHQ1; FluB-For 5′-TCC TCA ACT CAC TCT TCG AGC G-3′, FluB-Rev 5′-CGG TGC TCT TGA CCA AAT TGG-3′, Flu B-Probe 5′-FAM CCA ATT CGA GCA GCT GAA ACT GCG GTG-BHQ1.
2.6. Genotype-specific RT-PCR and real-time SYBR PCR for hMPV detection
The in house genotype-specific RT-PCR and real-time SYBR PCR methods were developed previously for detecting hMPV from clinical samples.
2.6.1. Genotype-specific RT-PCR
Primers for genotype-specific RT-PCR were designed according to the sequences of M genes of hMPVs (hMPV-M-f 5′-CAATGTCTGTACTTCCCAAA-3′, hMPV-M-A-r 5′-TACTTATGCTTTCAGCCTGG-3′, hMPV-M-B-r 5′-TTTTCTCTAGGTCCATGAAG-3′,). The RT-PCR was carried out in a 25 μL reaction mixture volume (One-step RT-PCR Kit, Qiagen, Hilden, Germany) containing 5.0 μL 5 × buffer, 1.0 μL dNTP, 1.0 μL Enzyme Mix, 0.1 μL RNase inhibitor, 1.0 μL of each primer (10 μM), 9.9 μL distilled H2O, 5.0 μL RNA template. Amplification was performed as follows: 60 °C for 1 min, 42 °C for 10 min, 50 °C for 30 min, 95 °C for 15 min, and then 45 cycles consisting of 95 °C for 30 s, 49 °C for 30 s and 72 °C for 45 s and final extension at 72 for 10 min. After the RT-PCR was completed, 5 μL of the product was analyzed by 2% agarose gel electrophoresis with ethidium bromide staining. The products from RT-PCR for hMPV A and B were 471 bp and 225 bp in length, respectively.
2.6.2. Real-time SYBR PCR
First-strand cDNA was synthesized using the M-MLV reverse transcriptase (Invitrogen, CA, USA). The reaction included the following components: 8.0 μL of 5× reverse transcriptase buffer, 2.0 μL dNTP mix (10 mM), 4.0 μL random primer (50 ng/μL), 0.4 μL M-MLV reverse transcriptase, 0.1 μL RNase inhibitor (50 U/μL), 4.0 μL DTT, and 21.5 μL of total RNA. The reaction mixture was incubated sequentially at 25 °C for 10 min, 37 °C for 60 min and 72 °C for 15 min. The synthesized cDNA was stored at −25 °C before use.
Real-time SYBR PCR genotype-specific primers for hMPV F gene were designed (hMPV-A-f 5′-AGGAGCAATAGCACTCGGTGTT-3′, hMPV-A-r 5′-TTGAGGGCATTCTTAATTGCTGT-3′, hMPV-B-f 5′-GCAATAGCCAAAACCATAAGGC-3′ and hMPV-B-r 5′-TTGAGAGCACCTTTAATTGCATTC-3′). The real-time SYBR PCR was carried out in a total of 25 μL reaction mixture containing A or B primer set (0.5 μL, 40 μM for each), 12.5 μL 2× Power SYBR Green PCR Master Mix (Applied Biosystems, Warrington, UK), 6.5 μL distilled H2O, 5.0 μL cDNA template. Amplification was performed as follows: 95 °C for 1 min, and then 45 cycles consisting of 95 °C for 15 s and 60 °C for 1 min, and finally the dissociation stage (95 °C 15 s, 60 °C 1 min, 95 °C 15 s, 60 °C 15 s). The mean melting temperatures for hMPV A and B were 80.3 ± 0.5 °C and 73.5 ± 0.5 °C, respectively. The results were judged by the amplification curve and melting curve. Amplification, detection and data analysis were performed with the ABI 7500 system software.
2.7. Detection of hMPV from clinical samples by genotype-specific RT-LAMP, RT-PCR and real-time SYBR PCR
One hundred and fifteen clinical specimens were tested for hMPV with the genotype-specific RT-LAMP, RT-PCR and real-time SYBR PCR as described above and the results were compared. Coincidence rate was evaluated by the same results from these three methods. SPSS software (v16.0) was used for statistical analysis of the data. The correlation among the results of three methods was evaluated by the Kappa test. A P < 0.05 was considered statistically significant.
3. Results
3.1. Sensitivity of the hMPV genotype-specific RT-LAMP
To evaluate the sensitivity of the genotype-specific RT-LAMP, 10-fold serial dilutions of quantitative in vitro transcribed RNA from the recombinant plasmid pUCm-T-A and pUCm-T-B containing the full M genes from the isolated viruses were used as the templates. The results indicated that the minimum limits of genotype-specific RT-LAMP for detecting hMPV genotypes A and B were 4.33 copies/μL and 4.53 copies/μL of the temple, respectively (Fig. 1 ). The minimum limit of the genotype-specific RT-PCR was analyzed in the same condition, which showed that the sensitivities for detecting hMPV genotypes A and B were 43.3 copies/μL and 45.3 copies/μL of RNA temples (Fig. 2 ). The reaction was repeated three times for each template with the same concentration. The results showed that the sensitivity of the genotype-specific RT-LAMP was 10 times greater than that of conventional RT-PCR. In addition, the transcribed RNAs of hMPV A and B were mixed by the same concentration and the minimum limits of A and B detection were both 5 copies/μL, indicating that coexistence of nucleic acids of hMPV A and B would not affect the sensitivity of the hMPV genotype-specific RT-LAMP.
Fig. 1.
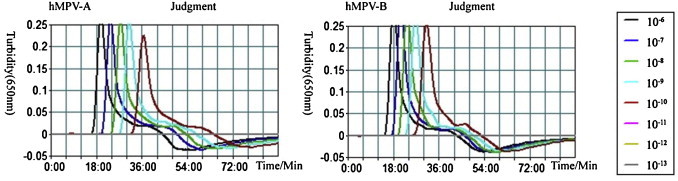
Sensitivity of the hMPV genotype-specific RT-LAMP assay. Serial diluted RNAs ranging from 10−6 to 10−13 were used to determine the detection limits of the hMPV genotype-specific RT-LAMP. The results of the genotype-specific RT-LAMP were assessed by the Loopamp® real-time LA-320C turbidimeter.
Fig. 2.
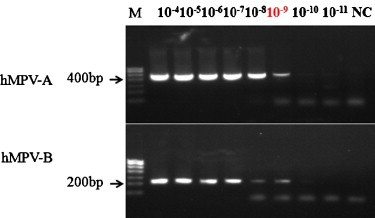
Sensitivity of the hMPV genotype-specific RT-PCR with serial diluted RNAs ranging from 10−4 to 10−11. Products were visualized by 2% agarose gel electrophoresis.
3.2. Specificity of the hMPV genotype-specific RT-LAMP
The specificity of the primers for amplifying hMPV A and B was evaluated by performing hMPV genotype-specific RT-LAMP with nucleic acids extracted from positive respiratory samples for RSV, PIV, ADV, HBoV, HRV, influenza virus A and B, HCoV (229E, OC43, HKU1 and NL63) and EV. In addition, ten of the samples tested by RVP Fast assay were positive for more than one viruses mentioned above. One of them was positive for both hMPV and HRV, and the others were dual positive for different types and subtypes of respiratory viruses mentioned above except hMPV. Only the sample positive for hMPV and HRV was positive for hMPV B as detected by the hMPV genotype-specific RT-LAMP assay. No cross-reactivity was shown with the above respiratory viruses, indicating that the hMPV genotype-specific RT-LAMP assay developed in this study is of high specificity. Furthermore, no amplification signal was presented from hMPV B positive samples when these samples were tested by genotype A-specific RT-LAMP primers, and vice versa.
3.3. Identifying and genotyping for hMPV in clinical specimens
A total of 115 clinical specimens collected from infants and children with acute respiratory infections were tested by the genotype-specific RT-LAMP, RT-PCR and real-time SYBR PCR, respectively. Comparison of genotyping results from these three methods is shown in Table 2 . The hMPV-positive samples detected by RT-LAMP, RT-PCR and real-time SYBR PCR were 61, 56 and 53, respectively. The coincidence rate of hMPV A and B tested by the three methods was 89.6% (103/115, A 17, B 32 and negative 54). The results of the genotype-specific RT-LAMP agreed with that of the genotype-specific RT-PCR and real-time SYBR PCR both in 92.2% (106/115). The Kappa coefficients for both RT-LAMP/RT-PCR and RT-LAMP/real-time SYBR PCR were 0.897 (P = 0.000), indicating a good agreement among the three methods. The genotype-specific RT-LAMP detected more positive samples than the RT-PCR and the real-time SYBR PCR (Table 2). However, one of the samples was genotype A only detected by RT-PCR, but mixed genotype A and B by the RT-LAMP and real-time SYBR PCR. Another sample was genotype A only detected by RT-PCR and real-time SYBR PCR, but mixed genotype A and B by the RT-LAMP assay. The average reaction time for all positive reactions of the RT-LAMP assay was 24.8 min (13.5–58.2 min) reported by the Loopamp® real-time LA-320C turbidimeter.
Table 2.
Comparison for hMPV detection from clinical samples by the genotype-specific RT-LAMP, RT-PCR and real-time PCR.
| RT-LAMP | RT-PCR | Real-time SYBR PCR | No. |
|---|---|---|---|
| A | A | A | 17 |
| B | B | B | 32 |
| – | – | – | 54 |
| A | A | – | 2 |
| B | – | – | 5 |
| B | – | B | 2 |
| B | B | – | 1 |
| AB | A | A | 1 |
| AB | A | AB | 1 |
The Kappa coefficients of RT-LAMP/RT-PCR and RT-LAMP/real-time SYBR PCR were 0.897 (P = 0.000).
3.4. Visual observation of the results for genotype-specific RT-LAMP
The by-products of amplified hMPV RNA with a white precipitate of magnesium pyrophosphate produced by RT-LAMP could be visualized by naked eyes under the natural daylight (Fig. 3A). When FDR was added to the reaction tube before amplification started, the positive reaction was indicated by a color change from faint orange to faint yellowish green by daylight or by a light green fluorescent change to stronger one under the ultraviolet (UV) light (Fig. 3B). It was easy to differentiate the positive reaction from negative one under the daylight or UV light. Although the time for positive-reaction of the RT-LAMP was longer by 7.5 min (3–15 min) on average due to adding FDR, there was no disagreement in the final qualitative judgment of sample detection. The results judged by the Loopamp® real-time LA-320C turbidimeter or visual observation with naked eyes under the natural light or under UV Lamp when FDR was added were consistent.
Fig. 3.
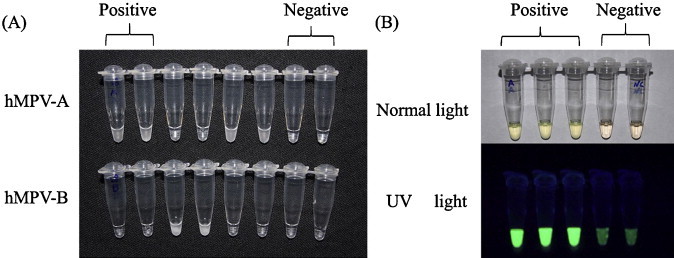
Results of the hMPV genotype-specific RT-LAMP assay observed by daylight and UV light. (A) The results of the RT-LAMP assays visualized by daylight with a white precipitate of magnesium pyrophosphate. 1, 2, 5 and 6, positive for hMPV-A; 3 and 4, positive for hMPV-B; 7, negative reaction; 8, negative control. (B) The positive results of the RT-LAMP assays with FDR visualized by naked eyes with faint yellowish green under daylight and with strong green fluorescence under UV light.
4. Discussion
As an important pathogen related to acute respiratory infections, hMPV has attracted world-wide attention and research interest. Up to date, the virus has been classified into two major genotypes, designated as genotype A and genotype B. Several studies focused on the relatedness between genotypes and disease severity (Skiadopoulos et al., 2004, Agapov et al., 2006). Although most of the published data suggested that disease severity was not related with a particular genotype (Agapov et al., 2006, Pitoiset et al., 2010), the extensive investigation on pathogenicity, cross-protective effectiveness and epidemiology for different hMPV genotypes has an important role in the vaccine development. A previous work from China reported that hMPV was detected by RT-LAMP assay (Wang et al., 2012), however, genotypes A and B could not be differentiated by that assay. Therefore, the genotype-specific RT-LAMP assay targeting the M gene of hMPV described in this study would have a better application prospects.
LAMP assay is based on autocycling strand-displacement DNA synthesis performed by Bst DNA polymerase. RNA can be amplified effectively using reverse transcriptase (RTase) together with DNA polymerase. LAMP employs a set of four specifically designed primers (not including two loop primers, which only for time saving) that recognize six distinct sequences of the target. Only when two pairs of specific primers match the target sequences exactly can the gene amplification starts. Therefore, RT-LAMP offers a high level of specificity for reverse transcription and cDNA amplification reactions of RNA viruses under isothermal environments plus the advantages of saving time and reducing experimental error. It is not necessary to open the reaction tubes in the detecting process, which can reduce the risk of contamination. In this study, the detecting limits for both hMPV A and B by genotype-specific RT-LAMP were 10 times greater than conventional RT-PCR. No cross-reaction was shown when templates of hMPV A and B were mixed, indicating that this assay was of highly specificity for detecting and differentiating hMPV A and B. According to the reaction time spent for testing the 63 positive clinical samples (13.5–58.2 min, average 24.8 min), the entire testing process can be completed within 1 h under isothermal conditions at 63 °C. Therefore, the genotype-specific RT-LAMP assay developed in this study can be used for identifying and genotyping for hMPV efficiently and accurately, especially in those laboratories lack of thermocycling equipments.
In the process of LAMP reaction, pyrophosphate ions as by-product of reaction bind to magnesium ions and form a white precipitate of magnesium pyrophosphate. Mori (Mori et al., 2004) designed a real-time monitoring turbidimeter for keeping stable and consistent temperature and measure the turbidities from amplification in multiple tubes from different clinical samples real-timely and simultaneously. Using this apparatus, the efficiency of the reactions can be observed real-timely and the exact reaction peak time can be determined, which is useful and helpful for the developing of a new LAMP assay for a specific target. Additionally, results can be readily observed by the white precipitate of magnesium pyrophosphate generated during the reaction or by color change by the daylight or UV light via adding FDR in the reaction mixture. Besides thermostatic equipment, no more laboratory equipments are required for the result determination. Therefore, RT-LAMP assay is easy to perform and efficient to determine the positive results. In this study, it was found that the reaction time was extended due to adding FDR, suggesting that the shorter reaction time might lead to a false negative result despite no disagreement in the final qualitative judgment of sample detection. Although the results were consistent when judged by turbidimeter and visual observation with naked eyes under the natural light or under UV Lamp when FDR was added, it could not be ruled out the possibility of false negative determination if the clinical samples contain less RNA copies which are close to the detection limit when judged by naked eyes. Therefore, result judged by turbidimeter or addition fluorescent FDR could be more reliable than by observing precipitate only.
The coincidence rate for detection of hMPV genotypes A and B from 115 clinical specimens was high by the genotype-specific RT-LAMP, RT-PCR and real-time SYBR PCR. The data suggested that the genotype-specific RT-LAMP assay has advantages for detecting hMPV B over the conventional RT-PCR and real-time SYBR PCR. Moreover, there were more mixed-genotype positive samples detected by RT-LAMP, which makes it possible to find out more mixed-genotype hMPV infections from clinical samples.
In summary, the genotype-specific RT-LAMP assay for hMPV detection from clinical respiratory specimens is a convenient, rapid, specific and sensitive method to identify hMPV genotypes A and B. It is worthy to recommend this method in clinical application, especially in resource-limited laboratories.
Acknowledgements
The authors of this paper would like to thank all doctors and nurses in the Affiliated Children's Hospital to Capital Institute of Pediatrics for collecting samples from patients and information from their parents. This work was supported by the grant No. Z111107056811041 from the Beijing Municipal Science & Technology Commission and the grant No. 30872153 from the Natural Science Foundation of China.
Contributor Information
Qinwei Song, Email: songtsinwei@163.com.
Runan Zhu, Email: runanzhu@163.com, runan@sina.com.
Yu Sun, Email: sunyu780312@163.com.
Linqing Zhao, Email: linqingzhao@gmail.com.
Fang Wang, Email: fangwang1128@sina.com.
Jie Deng, Email: jie_deng@sina.com.
Yuan Qian, Email: yqianbjc@263.net.
References
- Agapov E., Sumino K.C., Gaudreault-Keener M., Storch G.A., Holtzman M.J. Genetic variability of human metapneumovirus infection: evidence of a shift in viral genotype without a change in illness. J. Infect. Dis. 2006;193:396–403. doi: 10.1086/499310. [DOI] [PubMed] [Google Scholar]
- Bastien N., Normand S., Taylor T., Ward D., Peret T.C., Boivin G., Anderson L.J., Li Y. Sequence analysis of the N, P, M and F genes of Canadian human metapneumovirus strains. Virus Res. 2003;93:51–62. doi: 10.1016/S0168-1702(03)00065-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biacchesi S., Skiadopoulos M.H., Boivin G., Hanson C.T., Murphy B.R., Collins P.L., Buchholz U.J. Genetic diversity between human metapneumovirus subgroups. Virology. 2003;315:1–9. doi: 10.1016/s0042-6822(03)00528-2. [DOI] [PubMed] [Google Scholar]
- Curtis K.A., Rudolph D.L., Owen S.M. Sequence-specific detection method for reverse transcription, loop-mediated isothermal amplification of HIV-1. J. Med. Virol. 2009;81:966–972. doi: 10.1002/jmv.21490. [DOI] [PubMed] [Google Scholar]
- Deng J., Qian Y., Zhao L.Q., Zhu R.N., Wang F., Sun Y., Liao B., Huang R.Y., Yuan Y., Qu D., Ren X.X. Identification and typing of adenoviruses from pediatric patients with acute respiratory infections in Beijing from 2003 to 2008. Chin. J. Pediatr.Zhonghua er ke za zhi. 2010;48:739–743. [PubMed] [Google Scholar]
- Kubo T., Agoh M., Mai le Q., Fukushima K., Nishimura H., Yamaguchi A., Hirano M., Yoshikawa A., Hasebe F., Kohno S., Morita K. Development of a reverse transcription-loop-mediated isothermal amplification assay for detection of pandemic (H1N1) 2009 virus as a novel molecular method for diagnosis of pandemic influenza in resource-limited settings. J. Clin. Microbiol. 2010;48:728–735. doi: 10.1128/JCM.01481-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez-Huertas M.R., Casas I., Acosta-Herrera B., Garcia M.L., Coiras M.T., Perez-Brena P. Two RT-PCR based assays to detect human metapneumovirus in nasopharyngeal aspirates. J. Virol. Methods. 2005;129:1–7. doi: 10.1016/j.jviromet.2005.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo L., Nie K., Yang M.J., Wang M., Li J., Zhang C., Liu H.T., Ma X.J. Visual detection of high-risk human papillomavirus genotypes 16, 18, 45, 52, and 58 by loop-mediated isothermal amplification with hydroxynaphthol blue dye. J. Clin. Microbiol. 2011;49:3545–3550. doi: 10.1128/JCM.00930-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moes E., Vijgen L., Keyaerts E., Zlateva K., Li S., Maes P., Pyrc K., Berkhout B., van der Hoek L., Van Ranst M. A novel pancoronavirus RT-PCR assay: frequent detection of human coronavirus NL63 in children hospitalized with respiratory tract infections in Belgium. BMC Infect. Dis. 2005;5:6. doi: 10.1186/1471-2334-5-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mori Y., Kitao M., Tomita N., Notomi T. Real-time turbidimetry of LAMP reaction for quantifying template DNA. J. Biochem. Biophys. Methods. 2004;59:145–157. doi: 10.1016/j.jbbm.2003.12.005. [DOI] [PubMed] [Google Scholar]
- Mori Y., Notomi T. Loop-mediated isothermal amplification (LAMP): a rapid, accurate, and cost-effective diagnostic method for infectious diseases. J. Infect. Chemother. 2009;15:62–69. doi: 10.1007/s10156-009-0669-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Notomi T., Okayama H., Masubuchi H., Yonekawa T., Watanabe K., Amino N., Hase T. Loop-mediated isothermal amplification of DNA. Nucleic acids Res. 2000;28:E63. doi: 10.1093/nar/28.12.e63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pitoiset C., Darniot M., Huet F., Aho S.L., Pothier P., Manoha C. Human metapneumovirus genotypes and severity of disease in young children (n = 100) during a 7-year study in Dijon hospital, France. J. Med. Virol. 2010;82:1782–1789. doi: 10.1002/jmv.21884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skiadopoulos M.H., Biacchesi S., Buchholz U.J., Amaro-Carambot E., Surman S.R., Collins P.L., Murphy B.R. Individual contributions of the human metapneumovirus F, G, and SH surface glycoproteins to the induction of neutralizing antibodies and protective immunity. Virology. 2006;345:492–501. doi: 10.1016/j.virol.2005.10.016. [DOI] [PubMed] [Google Scholar]
- Skiadopoulos M.H., Biacchesi S., Buchholz U.J., Riggs J.M., Surman S.R., Amaro-Carambot E., McAuliffe J.M., Elkins W.R., St Claire M., Collins P.L., Murphy B.R. The two major human metapneumovirus genetic lineages are highly related antigenically, and the fusion (F) protein is a major contributor to this antigenic relatedness. J. Virol. 2004;78:6927–6937. doi: 10.1128/JVI.78.13.6927-6937.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van den Hoogen B.G., de Jong J.C., Groen J., Kuiken T., de Groot R., Fouchier R.A., Osterhaus A.D. A newly discovered human pneumovirus isolated from young children with respiratory tract disease. Nat. Med. 2001;7:719–724. doi: 10.1038/89098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Hoek L., Pyrc K., Jebbink M.F., Vermeulen-Oost W., Berkhout R.J., Wolthers K.C., Wertheim-van Dillen P.M., Kaandorp J., Spaargaren J., Berkhout B. Identification of a new human coronavirus. Nat. Med. 2004;10:368–673. doi: 10.1038/nm1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang F., Zhu R.N., Qian Y., Deng J., Zhao L.Q., Sun Y., Sha L., Liao B., Huang R.Y. Human metapneumoviruses were isolated from infants and children with acute respiratory infections in Beijing. Chin. J. Pediatr.Zhonghua er ke za zhi. 2010;48:820–823. [PubMed] [Google Scholar]
- Wang X., Zhang Q., Zhang F., Ma F., Zheng W., Zhao Z., Bai Y., Zheng L. Visual detection of the human metapneumovirus using reverse transcription loop-mediated isothermal amplification with hydroxynaphthol blue dye. Virol. J. 2012;9:138. doi: 10.1186/1743-422X-9-138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woo P.C., Lau S.K., Tsoi H.W., Huang Y., Poon R.W., Chu C.M., Lee R.A., Luk W.K., Wong G.K., Wong B.H., Cheng V.C., Tang B.S., Wu A.K., Yung R.W., Chen H., Guan Y., Chan K.H., Yuen K.Y. Clinical and molecular epidemiological features of coronavirus HKU1-associated community-acquired pneumonia. J. Infect. Dis. 2005;192:1898–1907. doi: 10.1086/497151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoneyama T., Kiyohara T., Shimasaki N., Kobayashi G., Ota Y., Notomi T., Totsuka A., Wakita T. Rapid and real-time detection of hepatitis A virus by reverse transcription loop-mediated isothermal amplification assay. J. Virol. Methods. 2007;145:162–168. doi: 10.1016/j.jviromet.2007.05.023. [DOI] [PubMed] [Google Scholar]
- Zhao L.Q., Qian Y., Wang F., Deng J. Comparison of four methods used in detection of the respiratory syncytial virus in nasopharyngeal aspirates from children with the acute lower respiratory tract infection. J. Clin. Pediatr. 2003;21:20–24. [Google Scholar]
- Zhao L.Q., Qian Y., Wang F., Zhu R.N., Deng J. Human parainfluenza virus infections in infants and young children with acute respiratory infections in Beijing. Chin. J. Pediatr.Zhonghua er ke za zhi. 2007;45:91–95. [PubMed] [Google Scholar]
- Zhao L.Q., Qian Y., Zhu R.N., Deng J., Wang F. Human rhinovirus detection from infants and young children with acute respiratory infections by nested-polymerase chain reaction. Zhonghua liu xing bing xue za zhi=Zhonghua liuxingbingxue zazhi. 2006;27:154–156. [PubMed] [Google Scholar]
- Zhao L.Q., Qian Y., Zhu R.N., Deng J., Wang F., Li Y. Preliminary studies suggest that a novel parvovirus called human bocavirus (HBoV), is related to acute respiratory infections in pediatric patients in Beijing. Chin. J. Microbiol. Immunol. 2006;26:385–388. [Google Scholar]
- Zhu R.N., Qian Y., Deng J., Wang F., Hu A.Z., Lu J., Cao L., Yuan Y., Cheng H.Z. Human metapneumovirus may associate with acute respiratory infections in hospitalized pediatric patients in Beijing, China. Chin. J. Pediatr.Zhonghua er ke za zhi. 2003;41:441–444. [PubMed] [Google Scholar]
- Zhu R.N., Qian Y., Zhao L.Q., Deng J., Wang F. Sequence analysis of the P and M genes of human metapneumovirus circulating in Beijing. Chin. J. Microbiol. Immunol. 2005;25:5–9. [Google Scholar]
- Zhu R.N., Qian Y., Zhao L.Q., Deng J., Wang F., Sun Y., Liao B. Surveillance on human metapneumovirus in infants and children with acute respiratory infections in Beijing, from 2004 to 2006. Zhonghua liu xing bing xue za zhi=Zhonghua liuxingbingxue zazhi. 2007;28:679–682. [PubMed] [Google Scholar]


