Abstract
Objective
To determine the prevalence of human rhinoviruses (HRV) infections in children with lower respiratory disease in Thailand and monitor the association between species of HRV and clinical presentation in hospitalized paediatric patients.
Method
Two hundred and eighty-nine nasopharyngeal (NP) suction specimens were collected from hospitalized paediatric patients admitted to King Chulalongkorn Memorial Hospital, Thailand during February 2006–2007. Nucleic acids were extracted from each sample with subsequent amplification of VP4/2 by semi-nested RT-PCR for HRV detection. Other viral respiratory pathogens were also detected by PCR, RT-PCR or real time PCR. Nucleotide sequences of the VP4 region were used for genotyping and phylogenetic tree construction.
Result
In total, 87 of 289 specimens were positive for HRV indicating an annual prevalence of 30%. Wheezing or asthma exacerbation was the most common clinical presentation observed in infected patients. Sequence analysis and phylogenetic tree showed that 29 (33%) and 8 (9%) specimens belonged to HRV-A and HRV-B, respectively. Most of the HRV positive samples were HRV-C (58%). Moreover, species C was predominantly found in the paediatric population of Thailand in raining season (p < 0.05). The frequency of co-infection of HRV-C with other respiratory viral pathogens was approximately 40%.
Conclusion
HRV-C represents the predominant species and is one of the etiologic agents in acute lower respiratory tract infection, causes of wheezing and asthma exacerbation in infants and young children in Thailand.
Keywords: Human rhinovirus, Prevalence, Children, Respiratory tract diseases
Introduction
Acute viral respiratory tract infections are the major causes of morbidity during early childhood. Among several aetiological pathogens discovered from paediatric patients with either moderate or severe illness, human rhinoviruses (HRV) are the most common. HRV are well recognized as causative agents of upper respiratory tract infections (URTIs),1 generally known as common colds that represent the most frequent illnesses worldwide. They are also significantly associated with the severity of several lower respiratory tract manifestations,2 such as exacerbations of acute wheezing3 and asthma,4, 5, 6, 7 bronchiolitis,8 pneumonia,6, 9 and acute otitis media.10
HRV share the basic properties of viruses in the Picornaviridae family and contain genomic characteristics that closely related to human enteroviruses (HEV). They are small, non-enveloped viruses, comprising an approximately 7.2 kb of positive single-stranded RNA genome of positive polarity surrounded by an icosahedral capsid protein. The respiratory tract epithelial cells are the target of HRV infection. Since the initial discovery of HRV by virus isolation techniques in 1956, HRV have been classified into more than 100 serotypes based on several parameters such as antigen cross-reactivity and nucleotide sequence homology. According to phylogenetic analysis of the VP4/VP2 capsid protein coding regions, HRV were initially classified into 2 distinct species with 75% belonging to the HRV-A and 25% to the HRV-B. In 2006, an as yet unrecognized HRV group, HRV-C, has been detected from influenza-like illness patients by using the MassTag PCR technique.11 Using molecular techniques, the complete genomes of novel species were characterized by Lau et al. in 2007.12 And HRV-C was reported as increasing evidence to a significant cause of wheezing,13 exacerbation of asthma14, 15 and association with lower respiratory tract infection (LRTI) in children.16, 17 However, recent report describing about the epidemiology of HRV among Asian hospitalized paediatric patients with respiratory tract illness has been limited. To demonstrate details of these data, we focused on establishing prevalence and clinical association of HRV among children with LRTI. In addition, the VP4 region of HRV demonstrated from paediatric patients in Thailand was subjected to genetic characterization and phylogenetic analysis.
Materials and methods
Demographic and clinical specimens
The study was retrospectively performed on the stored nasopharyngeal (NP) aspiration. The protocol was approved by the Institutional Review Board, Faculty of Medicine, Chulalongkorn University, Thailand. The studied population comprised all complete inclusion criteria of paediatric patients diagnosed with acute lower respiratory illness and hospitalized at King Chulalongkorn Memorial Hospital, Thailand during February 14, 2006 to February 28, 2007. The study's objectives were explained to the parents and or patients. Their consent, demographic and clinical information of patients were recorded using standardized questionnaire. Admission diagnosis criteria for acute bronchiolitis or community acquired viral pneumonia (CAP) were based on clinical presentations combined with other laboratory results and chest radiography as previously described.18, 19 The criteria recruited for acute bronchiolitis patients were as the following; patient presenting with upper respiratory tract symptoms with or without fever, abnormal lung auscultation including rhonchi or wheezing, and chest radiograph showing hyperinflation. CAP was diagnosed by the following criteria; URT symptoms with tachypnea according to the patient's age as following; less than 2 months old, respiratory rate (RR) > 60/min; 2–12 months old, RR > 50/min; 1–5 years of age, RR > 40/min; more than 5 years of age, RR > 30/min, combined with fever at onset > 37.8 °C, abnormal lung auscultation (e.g., rhonchi, crackles, wheezing) or abnormal chest radiograph consistent with pneumonia (e.g., consolidation, perihilar infiltrates).
However, patients were excluded from our study if no inform consent was obtained, or they have history of pre-maturity, pre-existing immunodeficiency, chronic respiratory tract illnesses, or NP suction specimens were not available.
Readmission to the hospital was considered as another separate illness episode. All NP suction samples were collected within 48 h of admission in 1 ml transport medium with antibiotics (2 million units/l of Penicillin G and 200 mg/l of Streptomycin), divided into aliquots, and stored at −70 °C until further tested.
Nucleic acid extraction and reverse transcription
Total DNA and RNA were extracted from NP suction samples using TRI REAGENT LS (Molecular Research Center Inc., Cincinnati, OH). DNA and RNA pellets were suspended in 20 μl of 8 mM NaOH or DEPC-treated water, respectively. cDNA were synthesized from total RNA using the M-MLV reverse-transcription system (Promega, Madison, WI). The reaction mixture comprising 200 units of M-MLV reverse transcriptase, 5 M-MLV reaction buffer, 10 mM of each dNTP, 25 units of RNasin® Ribonuclease inhibitor, 0.5 μM of random hexamer, RNA, nuclease-free water to a final volume of 25 μl was incubated at 37 °C for 2 h. The quality and integrity of cDNA were assessed by conventional PCR amplification of the human glyceraldehyde-3-phosphate dehydrogenase (GAPDH) gene (internal control).
Detection of HRV and other respiratory viruses by PCR amplification
HRV were detected by semi-nested PCR using specific primers including forward primers targeting the 5′ non-coding region (NCR) composed of F484 (position nt484-503): 5′-CGGCCCCTGAATGYGGCTAA-3′, F587 (position nt587-609): 5′-CTACTTTGGGTGTCCGTGTTTC-3′ and a reverse primer specific to the VP2 region, R1126 (position nt1106-1126): 5′-ATCHGGHARYTTCCAMCACCA-3′. These primers were selected and designed from conserved regions based on multiple alignments among several sequences of HRV available at the GenBank Database. The primers F484 and R1126 were used in the first round of PCR yielding the amplified product of 643 bp whereas the primers F587 and R1126 were used in the second round of PCR amplification yielding the expected product of 540 bp. The PCR reaction mixture contained 1 μl of cDNA, 0.5 μM of each primer, 10 μl of 2.5X Eppendorf MasterMix (Eppendorf, Hamburg, Germany), and adjusted to a final volume of 25 μl with nuclease-free water. Each round of PCR amplification was performed in a thermal cycler under the following conditions: initial denaturation at 94 °C for 3 min, followed by 40 amplification cycles consisting of 94 °C for 30 s (denaturation), 55 °C for 30 s (primer annealing), and 72 °C for 1 min (extension), and concluded by a final extension at 72 °C for 7 min. After 2% agarose gel electrophoresis, the expected amplicons were stained with ethidium bromide and then visualized under UV transilluminator.
As for other respiratory viruses such as Parainfluenza virus (PIV), Human Bocavirus (HBoV), Influenza A (IFAV) and B (IFBV) viruses, Adenovirus (HAdV), Respiratory Syncytial Virus (RSV) type A and B, Human Metapneumovirus (HMPV), and WU/KI polyomaviruses, clinical manifestations observed in patients and virus detection methods have been reported elsewhere.20, 21, 22, 23, 24, 25
Nucleotide sequencing
The HRV species for each strain was identified by direct sequencing of PCR products. The PCR products were purified from agarose gel using the Perfectprep Gel Cleanup kit (Eppendorf, Hamburg, Germany) according to the manufacturer's specifications. The resulting purified DNA served as templates for chain termination reaction with the Big Dye Terminator V.3.0 Cycle Sequencing Ready Reaction kit (ABI, Foster City, CA) in the ABI PRISM® 310 automated DNA sequencer.
Genetic characterization and phylogenetic analysis
For genetic characterization, the nucleotide and amino acid sequences of the VP4 region were analyzed using the BLAST program available in GenBank (http://www.ncbi.nlm.gov/BLAST). Sequences were prepared and multiple aligned using Clustal W implemented in the BioEdit program version 7.0.4.1 (www.mbio.ncsu.edu/BioEdit/bioedit.html). Phylogenetic trees were constructed using the neighbor-joining method and Kimura's two-parameter with 1000 boots-trapping method implemented in the MEGA4© version 4.0 program.26
Statistical analysis
Statistical data for seasonal prevalence and clinical symptoms of HRV infected patients within the sample population were analyzed using the SPSS software package (version 13; Chicago, III, USA). The statistic data comparison was performed using ANOVA, Pearson χ 2 and T-test where it's applicable. And the results were considered statistically significant at p < 0.05.
Results
Molecular characterization and phylogenetic analysis within the VP4 gene of HRV
All common respiratory viral pathogens were identified, including RSV in 15%, IFAV in 31%, HMPV in 24%, HBoV in 20%, WU polyomavirus in 19%, HAdV in 17%, PIV 13%, and KI polyomavirus in 6%. Unfortunately, we did not perform the test for Human Coronaviruses (HCoVs). According to our previous observation, the prevalence of HCoVs infection in children who had acute LRT diseases in Thailand was found to be extremely low.27
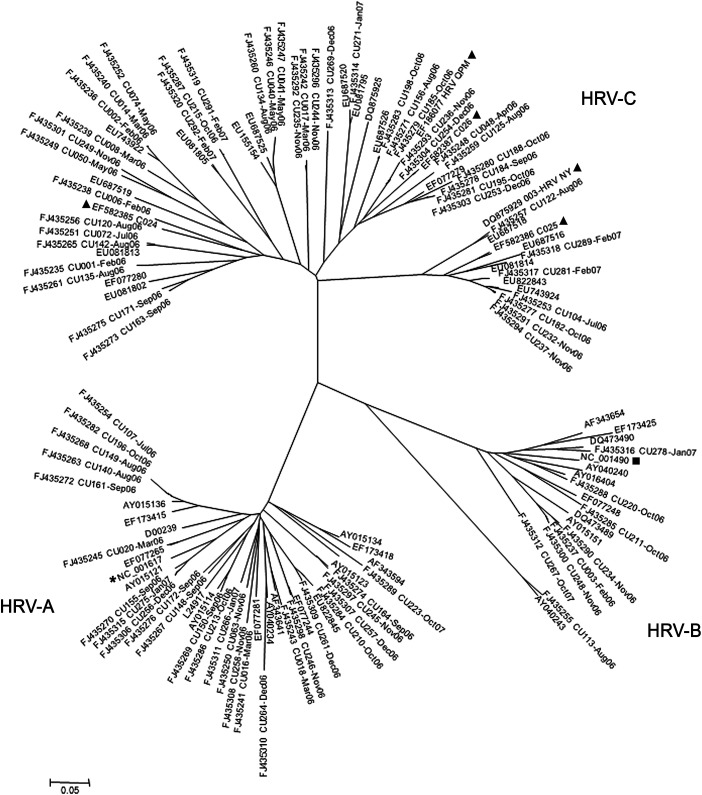
HRV was the most common respiratory pathogen found in 87 samples from 84 patients indicating a prevalence of 30% in this sample population. Two patients had a total of 5 HRV infections between them. Among of HRV positive paediatric patients, 52 and 32 were males and females (1.6:1), respectively. The relationship between HRV characterized in our study and the closest related-prototype strains was investigated based on phylogenetic tree constructions of the VP4 regions (Fig. 1 ) and showed that species A and B accounted for 29 (33%) and 8 (9%) specimens, respectively. Most of the HRV positive specimens belonged to a genetically distinct species, HRV-C (58%). The results revealed that HRV-C was the most predominant species responsible for HRV infection and that HRV-B represented a minor species in Thailand.
Figure 1.
Phylogenetic analysis of the nucleotide sequences of the virus capsid protein (VP4) region of 87 HRV strains identified from the 289 NP suction specimens. Phylogenetic tree was constructed by using the neighbor-joining method and Kimura's two-parameter with bootstrap replicated from 1000 trees using Molecular Evolutionary Genetics Analysis version 4.0 (MEGA4) program (www.megasoftware.net). Genetic distance (nucleotide substitutions per site) was represented by the scale bar. The HRV strain name in this study referred to number of specimen and patients' admission month and year. For prototype strains, (*), (■), (▴) were designated for HRV-A, HRV-B, and HRV-C, respectively.
All VP4 nucleotide sequences of HRV obtained from this study were submitted to the GenBank database and assigned accession numbers FJ435235–FJ435337FJ435235FJ435236FJ435237FJ435238FJ435239FJ435240FJ435241FJ435242FJ435243FJ435244FJ435245FJ435246FJ435247FJ435248FJ435249FJ435250FJ435251FJ435252FJ435253FJ435254FJ435255FJ435256FJ435257FJ435258FJ435259FJ435260FJ435261FJ435262FJ435263FJ435264FJ435265FJ435266FJ435267FJ435268FJ435269FJ435270FJ435271FJ435272FJ435273FJ435274FJ435275FJ435276FJ435277FJ435278FJ435279FJ435280FJ435281FJ435282FJ435283FJ435284FJ435285FJ435286FJ435287FJ435288FJ435289FJ435290FJ435291FJ435292FJ435293FJ435294FJ435295FJ435296FJ435297FJ435298FJ435299FJ435300FJ435301FJ435302FJ435303FJ435304FJ435305FJ435306FJ435307FJ435308FJ435309FJ435310FJ435311FJ435312FJ435313FJ435314FJ435315FJ435316FJ435317FJ435318FJ435319FJ435320FJ435321FJ435322FJ435323FJ435324FJ435325FJ435326FJ435327FJ435328FJ435329FJ435330FJ435331FJ435332FJ435333FJ435334FJ435335FJ435336FJ435337. Pairwise nucleotide identity within the VP4 regions of HRV-C was determined among HRV prototype strains including species A (NC_001617), B (NC_001490), and previous reported species C (EF582385–EF582387EF582385EF582386EF582387, DQ875929, and EF186077). Our results revealed that the HRV-C detected from Thai paediatric patients displayed lower sequence identity scores, ranging from 51–62% for nucleotide identity and 49–64% for deduced amino acid identity when compared to species A. In Comparison with species B, HRV-C from our study represented 50–61% for nucleotide identity and 46–54% for predicted amino acid identity. Within intra HRV-C species, identity ranged from 63–99% and 78–94% for nucleotide sequences and predicted amino acid sequences, respectively. Interestingly, we found that some HRV-C strains (FJ435242: CU017-Mar06, FJ435292: CU235-Nov06, and FJ435296: CU244-Nov06) were distinctly clustered in phylogenetic tree (Fig. 1). Their average nucleotide identity when compared with prior prototype strains represented 73% of nucleotide and 80% of predicted amino acid identity with HRV-C 024 (EF582385), 72% of nucleotide and 80% of predicted amino acid identity with HRV-QPM (EF186077), and 69% of nucleotide and 77% of predicted amino acid identity with 003-HRV NY (DQ875929).
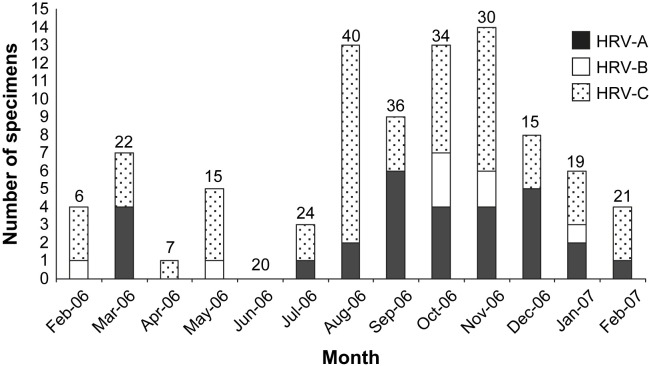
Seasonal prevalence
A seasonal pattern in our study was categorized into 3 seasonal periods according to tropical country style as the following; February to April (summer), May to October (rainy), and November to January (winter). As for seasonal prevalence, HRV-C was detected throughout the year except for June. Of the 50 samples from children with HRV-C infections, 52% (26/50), 28% (14/50) and 20% (10/50) were collected during the rainy (May–October), winter (November–January) and summer (February–April) seasons in Thailand, respectively (Fig. 2 ). HRV-A was detected in specimens collected during the rainy (45% (13/29)) and winter (36.7% (11/29)) season with only 5 cases during the summer (17%). The result indicated that the predominance of HRV-C and HRV-A infection were detected during the raining and early winter seasons (p < 0.05). HRV-B was observed during 5 months of the year including February, May, October, November, and January.
Figure 2.
Seasonal prevalence of HRV in paediatric patients studied from February 2006–2007 (the number on top of the bar is the total number of patients).
HRV infection in paediatric patients with different ages
The subjects were categorized into 4 groups based on their age as the following; 5 months of age or less, 6–23 months, 24–59 months and 60 months or older. We detected HRV infection as a sole pathogen in 62% (54/87), whereas approximately 38% (33/87) were co-infected with other respiratory viruses except for IFBV as the result showed in Table 1 . Among these co-infected samples, 15% with HBoV, 18% with HAdV, 18% with IFAV, 18% with WU/KI polyomaviruses, 6% with PIV, 9% with HMPV, and found a higher frequency with RSV (36%) but without statistically significant (p > 0.05). Of all the patients with HRV infection alone, 30 cases (34%) were diagnosed with HRV-C, 19 (22%) with HRV-A and 5 (6%) with HRV-B. Although HRV could be detected in paediatric patients ranging from 11 days to 10 years 5 months, the majority of cases comprised children of approximately 6–23 months of age. The proportion of newborn to 5 months who infected with each HRV species was not significantly difference (p > 0.05). There were 3 cases with HRV-A, 2 cases with HRV-B and 5 cases with HRV-C. Most of HRV-C infections were found in children of 6–23 months of age (31/55) and a similar proportion in children of 24–59 months of age (12/16). In contrast, HRV-A (21/55 and 3/16, respectively) and HRV-B (3/55 and 1/16, respectively) were diagnosed at a much lower frequency.
Table 1.
Characteristics of HRV infected pediatric patients in 2006–2007.
| No. of positive NPAs | HRV-A |
HRV-B |
HRV-C |
Total |
|---|---|---|---|---|
| 29 | 8 | 50 | 87 | |
| Age | ||||
| 0–5 months | 3 | 2 | 5 | 10 |
| 6–23 months | 21 | 3 | 31 | 55 |
| 24–59 months | 3 | 1 | 12 | 16 |
| More than 60 months | 1 | 2 | 3 | 6 |
| Single infection | 19 | 5 | 30 | 54 |
| Co-infection with | ||||
| Human Bocavirus | 0 | 0 | 5 | 5 |
| Adenovirus | 1 | 0 | 5 | 6 |
| WU polyomavirus | 1 | 0 | 2 | 3 |
| KI polyomavirus | 2 | 0 | 1 | 3 |
| Influenza A virus | 1 | 0 | 5 | 6 |
| Influenza B virus | 0 | 0 | 0 | 0 |
| Parainfluenza virus | 0 | 0 | 2 | 2 |
| Respiratory syncytial virus | 4 | 3 | 5 | 12 |
| Human Metapneumovirus | 2 | 0 | 1 | 3 |
Clinical manifestations of HRV positive patients
HRV infection was associated with several of clinical characteristics contributing from moderate and severe lower respiratory illness. The most common clinical presentations of hospitalized children positive for HRV were acute bronchiolitis and pneumonia exhibited by more than 50% of the patients however their symptoms do not cause more severe diseases than other respiratory viruses including RSV. We detected HRV-C in 11/50 (22%) of the first time wheezers (age ranged from 4 to 23 months) and their symptoms were consistent with acute bronchiolitis (Table 2 ). Also, 9 of 50 (18%) patients were recurrent wheezers who responded well to the bronchodilators with or without corticosteroids. Additional, we also detected 2 patients who had been admitted with 5 different episodes of acute RTIs and subsequent recurrent wheezing associated with HRV-A and C.28 Exacerbation of asthma or RAD was also commonly found especially with the HRV-C positive group. Additionally, there was a trend to detect HRV-C species more often than HRV-A or HRV-B species in children with underlying disease of asthma or reactive airway disease (p = 0.09). Fever and cough were the common presenting symptoms. Initial body temperature of positive patients were showed in mean ± SD (°C) as following; with species A 37.5 ± 0.83 °C in, with species B 37.8 ± 0.57 °C, and with species C 37.3 ± 0.7 °C. Initial fever represented not different among HRV species (p = 0.2). The median length of hospitalization was 4 days for all children with HRV (ranging from 1 to 80 days with HRV-A, 3 to 121 with HRV-B and 1 to 47 with HRV-C infection). Comparison among groups did not show significant difference (p = 0.4). Comparison with HRV negative patients (control group), they had fever, rhinorrhea, and cough as the common clinical presentation similar to those of HRV positive individuals.
Table 2.
Comparison of clinical characteristics among different HRV subgroups.
| Clinical characteristics | HRV-A (N = 29) | HRV-B (N = 8) | HRV-C (N = 50) |
|---|---|---|---|
| Initial BT (C) | 37.5 ± 0.83 | 37.8 ± 0.57 | 37.3 ± 0.7 |
| Underlying disease | |||
| Asthma or RAD | 8 (27%) | 2 (25%) | 25 (50%) |
| Diagnosis | |||
| Acute bronchiolitis | 8 (27.5%) | 1 (12.5%) | 11 (22%) |
| Pneumonia | 17 (58%) | 7 (87.5%) | 30 (60%) |
| Wheezing | 17 (58%) | 3 (37.5%) | 36 (72%) |
Discussion
Our retrospective study had investigated HRV prevalence among the Thai paediatric population between February 2006 and 2007 along with clinical manifestations which represented the possible association between novel HRV-C infection and exacerbations of wheezing and asthma. Overall, HRV (30%) were more detected than RSV (16%) suggested that HRV are predominant pathogens found in Thai paediatric patients diagnosed with acute LRI. Of those 87 positive cases, 54 (62%) episodes were due to single infection and more than half of them (30/54, 55%) were caused by a novel HRV-C. Phylogenetic analysis of the VP4 region showed that novel HRV strain was more closely related to HRV-A than HRV-B. The nucleotide identity scores compared with previously classified HRV-C strains, HRV-QPM identified from an infant with bronchiolitis specimen from Australia16 were 64–97%, with HRV-C 024–026 (EF582385–EF582387EF582385EF582386EF582387) detected in Hong Kong12 they amounted to 66–81%, 63–88%, and 65–99%, respectively. And compared with an unclassified HRV species, 003-HRV NY, investigated from patients with influenza-like symptoms from the United States29 were 62–96% identity and 64–77% identity to the BCH200Oct07-343 Dec07 (EU687515–EU687528EU687515EU687516EU687517EU687518EU687519EU687520EU687521EU687522EU687523EU687524EU687525EU687526EU687527EU687528) detected from children with lower acute RTIs in Beijing, People's Republic of China.17 This demonstrates that relative to all previous reports, the HRV-C strains identified in our study were closely related to the proposed novel species, HRV-C. Interestingly, based on its intra-group diversity, some of HRV-C strains from our study (FJ435242: CU017-Mar06, FJ435292: CU235-Nov06, and FJ435296: CU244-Nov06) showed identical clustered group with approximately 26–37% of nucleotide divergence from the HRV-C reference strains.
According to research data reported from New York and Atlanta between 2001 and 2003,13 HRV species were identified in 16% (167/1052) of children below 5 years of age hospitalized with acute LRI with 44% identified as species A, 4% as B and 52% as C. Research carried out in Hong Kong from 2004 to 200512, discovered HRV infection in 31% (26/83) of the population studied, with 4% (1/26) HRV-A and 96% (25/26) HRV-C related whereas HRV-B was not detected. Taken together, HRV-C has a seasonal occurrence and appears to represent the dominant strain in Southeast Asian, having circulated globally since at least 2002, whereas species B plays only a minor role. It also seems likely that HRV-C has not emerged de novo. HRV-C does not show seasonality in Southeast Asia appears to be contradicted by the prior discussion (rainy season and winter in the current study, Oct to Feb in Hong Kong12 and Oct to Dec in China17).
Acute LRT infections contribute to significant morbidity and mortality especially resulting from respiratory virus infections. The importance of HRV infection in LRT morbidity during the first year of life is underappreciated.30 As reported by Miller et al.,13 HRV-C infection appears to be significantly age related, more than infection with any other HRV species. Both Infants between 6–23 months and 24–59 months of age groups seem to be specifically targeted by HRV-C. On the contrary, HRV-A and HRV-B were diagnosed with less prevalence but the difference did not show any statistical significance.
Acute wheezing that resulted from lower respiratory tract infections and exacerbation of asthma episodes are significant manifestations of disease associated by HRV infection in young children (e.g. acute bronchiolitis, asthma or RAD exacerbation).3, 4, 5, 6, 15, 31, 32, 33, 34, 35, 36, 37, 38 In agreement of recent publication, most of our HRV positive patients presented with acute wheezing episodes. Moreover, we mainly detected the novel HRV-C in children with underlying asthma or RAD. This is in agreement with the recent publication. Compare to the Atlanta study,13 HRVs were detected in 37% (53/142) of paediatric patients with exacerbation of asthma and the majority of HRV strains (62%) identified as species A whereas 28% and 10% were identified as HRV-C and B, respectively. Acute exacerbation of RAD or asthma is frequently the only evidence of infection in our study. This finding is also in agreement with several studies.3, 4, 5, 6, 15, 31, 32, 33, 34, 35, 36, 37, 38 We also detected RSV as a common co-infection. This may mimic the clinical presentations of HRV infection and may cause more severe disease. Nevertheless, the recent data from a large clinical cohort study (COAST) performed on asthmatic children, indicated that rather than RSV, HRVs infection of lower respiratory tract is the most important predictor of subsequent wheezing in early childhood.3 Compare to our data, we found both different HRV species and the same species caused different episodes of wheezing-LRI which might indicate a possible association between HRV infection and potential development of recurrent wheezing.28
In brief, HRV-C infections were the most prevalent HRV infection identified in this study population, and demonstrated a genetically diverse population of HRV-C viruses that circulated in Thailand during the study period. However, further studies would be required in order to understand more clearly about the role of HRV infection as a cause of recurrent wheezing together with requirement of future population-based viral surveillance.
Conflict of interest
None declared.
Acknowledgements
This study was supported by the Higher Commission of Education, Ministry of Education, The Center of Excellence Research Fund, CU Centernary Academic Development Project, Chulalongkorn University and King Chulalongkorn Memorial Hospital. We would like to express our gratitude to the entire staffs of the Center of Excellence in Clinical Virology, Paediatric Pulmonary and Critical Care, Faculty of Medicine, Chulalongkorn University, and all paediatric pulmonary fellows as well as paediatric residents who have made this study possible. We also would like to thank Ms Petra Hirsch for reviewing the manuscript and Mr. Wasan Punyasang for statistical analysis.
References
- 1.Brownlee J.W., Turner R.B. New developments in the epidemiology and clinical spectrum of rhinovirus infections. Curr Opin Pediatr. 2008;20:67–71. doi: 10.1097/MOP.0b013e3282f41cb6. [DOI] [PubMed] [Google Scholar]
- 2.Leonard K., Lauren P., Evelyn K., Kevin M., David W., Martin H. The association of rhinoviruses with lower respiratory tract disease in hospitalized patients. J Med Virol. 2005;19:345–352. doi: 10.1002/jmv.1890190407. [DOI] [PubMed] [Google Scholar]
- 3.Lemanske R.F., Jackson D.J., Gangnon R.E., Evans M.D., Li Z., Shult P.A. Rhinovirus illnesses during infancy predict subsequent childhood wheezing. J Allergy Clin Immunol. 2005;116:571–577. doi: 10.1016/j.jaci.2005.06.024. [DOI] [PubMed] [Google Scholar]
- 4.Leigh R., Oyelusi W., Wiehler S., Koetzler R., Zaheer R.S., Oyelusi W. Human rhinovirus infection enhances airway epithelial cell production of growth factors involved in airway remodeling. J Allergy Clin Immunol. 2008;121:1238–1245. doi: 10.1016/j.jaci.2008.01.067. [DOI] [PubMed] [Google Scholar]
- 5.Psarras S., Volonaki E., Skevaki C.L., Xatzipsalti M., Bossios A., Pratsinis H. Vascular endothelial growth factor-mediated induction of angiogenesis by human rhinoviruses. J Allergy Clin Immunol. 2006;117:291–297. doi: 10.1016/j.jaci.2005.11.005. [DOI] [PubMed] [Google Scholar]
- 6.De Silva D., Dagher H., Ghildyal R., Lindsay M., Li X., Freezer N.J. Vascular endothelial growth factor induction by rhinovirus infection. J Med Virol. 2006;78:666–672. doi: 10.1002/jmv.20591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Papadopoulos N.G., Papi A., Psarras S., Johnston S.L. Mechanisms of rhinovirus-induced asthma. Paediatr Respir Rev. 2004;5:255–260. doi: 10.1016/j.prrv.2004.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jartti T., Lehtinen P., Vuorinen T., Österback R., van den Hoogen B., Osterhaus A.D. Respiratory picornaviruses and respiratory syncytial virus as causative agents of acute expiratory wheezing in children. Emerg Infect Dis. 2004;10:1095–1101. doi: 10.3201/eid1006.030629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Imakita M., Shiraki K., Yutani C., Ishibashi-Ueda H. Pneumonia caused by rhinovirus. Clin Infect Dis. 2000;30:611–612. doi: 10.1086/313723. [DOI] [PubMed] [Google Scholar]
- 10.Chantzi F.M., Papadopoulos N.G., Bairamis T., Tsiakou M., Bournousouzis N., Constantopoulos A.G. Human rhinoviruses in otitis media with effusion. Pediatr Allergy Immunol. 2006;17:514–518. doi: 10.1111/j.1399-3038.2006.00448.x. [DOI] [PubMed] [Google Scholar]
- 11.Lamson D., Renwick N., Kapoor V., Liu Z., Palacios G., Ju J. MassTag polymerase-chain reaction detection of respiratory pathogens, including a new rhinovirus genotype, that caused influenza-like illness in New York State during 2004–2005. J Infect Dis. 2006;194:1398–1402. doi: 10.1086/508551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Susanna K.P.L., Cyril C.Y.Y., Hoi-wah T., Rodney A.L., Lok-yee S., Yu-lung L. Clinical features and complete genome characterization of a distinct human rhinovirus (hrv) genetic cluster, probably representing a previously undetected HRV species, HRV-C, associated with acute respiratory illness in children. J Clin Microbiol. 2007;45:3655–3664. doi: 10.1128/JCM.01254-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Miller E.K., Edwards K.M., Weinberg G.A., Iwane M.K., Griffin M.R., Hall C.B. A novel group of rhinoviruses is associated with asthma hospitalizations. J Allergy Clin Immunol. 2009;123:98–104. doi: 10.1016/j.jaci.2008.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Khetsuriani N., Lu X., Teague G., Kazerouni N., Anderson L.J., Erdman D.D. Novel human rhinoviruses and exacerbation of asthma in children. Emerg Infect Dis. 2008;14:1793–1796. doi: 10.3201/eid1411.080386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Miller E.K., Lu X., Erdman D.D., Poehling K.A., Yuwei Zhu, Griffin M.R. Rhinovirus-associated hospitalizations in young children. J Infect Dis. 2007;195:773–781. doi: 10.1086/511821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lee W.M., Kiesner C., Pappas T., Lee I., Grindle K., Jartti T. A diverse group of previously unrecognized human rhinoviruses are common causes of respiratory illnesses in infants. PLoS ONE. 2007;2(10):e966. doi: 10.1371/journal.pone.0000966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Xiang Z., Gonzalez R., Xie Z., Xiao Y., Chen L., Li Y. Human rhinovirus group C infection in children with lower respiratory tract infection. Emerg Infect Dis. 2008;14:1665–1667. doi: 10.3201/eid1410.080545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Samransamruajkit R., Moonviriyakit K., Vanapongtipagorn P., Prapphat N., Deerojanawong J., Poovorawan Y. Plasma endothelin-1 in infants and young children with acute bronchiolitis and viral pneumonia. Asian Pac J Allergy Immunol. 2002;20:229–234. [PubMed] [Google Scholar]
- 19.Samransamruajkit R., Hiranrat T., Chieochansin T., Sritippayawan S., Deerojanawong J., Papphal N. Prevalence, clinical presentations and complications among hospitalized children with influenza pneumonia. J Infect Dis. 2008;61:446–449. [PubMed] [Google Scholar]
- 20.Chieochansin T., Samransamruajkit R., Chutinimitkul S., Payungporn S., Hiranras T., Theamboonlers A. Human Bocavirus (HBoV) in Thailand; clinical manifestations in a hospitalized pediatric patients and molecular virus characterization. J Infect. 2008;56:138–142. doi: 10.1016/j.jinf.2007.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chutinimitkul S., Chieochansin T., Payungporn S., Samransamruajkit R., Hiranras T., Theamboonlers A. Molecular characterization and phylogenetic analysis of H1N1 and H3N2 human influenza A viruses among infants and children in Thailand. Virus Res. 2008;132:122–131. doi: 10.1016/j.virusres.2007.11.007. [DOI] [PubMed] [Google Scholar]
- 22.Chi X.S., Hu A., Bolar T.V., Al-Rimawi W., Zhao P., Tam J.S. Detection and characterization of new influenza B virus variants in 2002. J Clin Microbiol. 2005;43:2345–2349. doi: 10.1128/JCM.43.5.2345-2349.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Krafft A.E., Russell K.L., Hawkswort A.W., McCall S., Irvine M., Daum L.T. Evaluation of PCR testing of ethanol-fixed nasal swab specimens as an augmented surveillance strategy for influenza virus and adenovirus identification. J Clin Microbiol. 2005;43:1768–1775. doi: 10.1128/JCM.43.4.1768-1775.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Samransamruajkit R., Thanasugarn W., Prapphal N., Theamboonlers A., Poovorawan Y. Human metapneumovirus in infants and young children in Thailand with lower respiratory tract infections; molecular characteristics and clinical presentations. J Infect. 2006;52:254–263. doi: 10.1016/j.jinf.2005.07.001. [DOI] [PubMed] [Google Scholar]
- 25.Payungporn S., Chieochansin T., Thongmee C., Samransamruajkit R., Theamboolers A., Poovorawan Y. Prevalence and molecular characterization of WU/KI polyomaviruses isolated from pediatric patients with respiratory disease in Thailand. Virus Res. 2008;135:230–236. doi: 10.1016/j.virusres.2008.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tamura K., Dudley J., Nei M., Kumar S. MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol. 2007;24:1596–1599. doi: 10.1093/molbev/msm092. [DOI] [PubMed] [Google Scholar]
- 27.Theamboonlers A., Samransamruajkit R., Thongme C., Amonsin A., Chongsrisawat V., Poovorawan Y. Human coronavirus infection among children with acute lower respiratory tract infection in Thailand. Intervirology. 2007;50:71–77. doi: 10.1159/000097392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Linsuwanon P., Payungporn S., Samransamruajkit R., Theamboonlers A., Poovorawan Y. Recurrent Human Rhinovirus (A and C) infection in infants with refractory wheezing. Emerg Infect Dis. 2009;15:978–980. doi: 10.3201/eid1506.081558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Domingueza S.R., Brieseb T., Palacios G., Huib J., Villari J., Kapoor V. Multiplex MassTag-PCR for respiratory pathogens in paediatric nasopharyngeal washes negative by conventional diagnostic testing shows a high prevalence of viruses belonging to a newly recognized rhinovirus clade. J Clin Virol. 2008;43:219–222. doi: 10.1016/j.jcv.2008.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kusel M.M.H., De Klerk N.H., Holt P.G., Kebadze T., Johnston S.L., Sly P.D. Role of respiratory viruses in acute upper and lower respiratory tract illness in the first year of life: a birth cohort study. Pediatr Infect Dis J. 2006;25:680–686. doi: 10.1097/01.inf.0000226912.88900.a3. [DOI] [PubMed] [Google Scholar]
- 31.Hayden F.G. Rhinovirus and the lower respiratory tract. Rev Med Virol. 2004;14:17–31. doi: 10.1002/rmv.406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tan W.C. Viruses in asthma exacerbations. Curr Opin Pulm Med. 2005;11:21–26. doi: 10.1097/01.mcp.0000146781.11092.0d. [DOI] [PubMed] [Google Scholar]
- 33.Thumerelle C., Deschildre A., Bouquillon C., Santos C., Sardet A., Scalbert M. Role of viruses and atypical bacteria in exacerbations of asthma in hospitalized children: a prospective study in the Nord-Pas de Calais region (France) Pediatr Pulmonol. 2003;35:75–82. doi: 10.1002/ppul.10191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Singh A.M., Moore P.E., Gern J.E., Lemanske R.F., Jr., Hartert T.V. Bronchiolitis to asthma: a review and call for studies of gene–virus interactions in asthma causation. Am J Respir Crit Care Med. 2007;175:108–119. doi: 10.1164/rccm.200603-435PP. [DOI] [PubMed] [Google Scholar]
- 35.Arden K.E., McErlean P., Nissen M.D., Sloots T.P., Mackay I.M. Frequent detection of human rhinoviruses, paramyxoviruses, coronaviruses, and bocavirus during acute respiratory tract infections. J Med Virol. 2006;78:1232–1240. doi: 10.1002/jmv.20689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kistler A., Avila P., Rouskin S., Wang D., Ward T., Yagi S. Pan-viral screening of respiratory tract infections in adults with and without asthma reveals unexpected human coronavirus and human rhinovirus diversity. J Infect Dis. 2007;196:817–825. doi: 10.1086/520816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.El-Sahly H.M., Atmar R.L., Glezen W.P., Greenberg S.B. Spectrum of clinical illness in hospitalized patients with “Common cold” virus infections. Clin Infect Dis. 2000;31:96–100. doi: 10.1086/313937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Silva M.J., Ferraz C., Pissarra S., Cardoso M.J., Simoes J., Bonito V.A. Role of virus and atypical bacteria in asthma exacerbations among children in Portugal. Allergol Immunopathol. 2007;35:4–9. doi: 10.1157/13099088. [DOI] [PubMed] [Google Scholar]