Abstract
Concurrently, several porcine epidemic diarrhea virus (PEDV) variants are circulating in US swine farms, including the original US and the spike insertion-deletion (S-INDEL) strains. In this study, reverse transcription (RT)-PCR assays for the detection and differentiation of different US PEDV variants were developed based on the differences in the S1 domain of the spike (S) gene. This assay successfully differentiated three PEDV strains: PC22A (the original US virulent), Iowa106 (S-INDEL), and PC177 (S-197DEL) that was derived from cell culture adaptation and has a 197 amino acid-deletion in the S1 domain. The assays did not amplify the porcine deltacoronavirus OH-FD22 strain or transmissible gastroenteritis virus Miller strain. It is the first report on the development of RT-PCR assays allowing the detection and differentiation of all major types of US PEDV variants.
Keywords: Porcine epidemic diarrhea virus, Reverse transcription-PCR, Spike gene
Porcine epidemic diarrhea (PED) is a highly contagious viral disease and was first observed in farm pigs in England in 1971 (Wood, 1977). The disease is caused by porcine epidemic diarrhea virus (PEDV), an Alphacoronavirus within the family Coronaviridae and order Nidovirales (Song and Park, 2012). To date, PEDV has spread worldwide and become a critical threat to the swine industry.
PEDV first emerged in the US in April, 2013 and then the virus spread rapidly nationwide and led to enormous economic losses (Chen et al., 2014, Stevenson et al., 2013). To date, three major types of PEDV have been reported in the US: (1) the original US PEDV strains that are highly virulent causing high mortality in suckling piglets (Jung et al., 2014, Stevenson et al., 2013); (2) the spike (S) insertion-deletion (S-INDEL) PEDV strains that are less virulent compared with the original US PEDV strains, but can cause variable (none-to-high) mortality in suckling piglets (Lin et al., 2015, Wang et al., 2014a); and (3) the PC177 strain that has a 197 aa-deletion in the S1 region (S-197DEL) and was detected after Vero cell culture adaptation (Oka et al., 2014). A diagnostic assay to differentiate those PEDV variants is urgently needed in the laboratory and in clinical practice because they cannot be differentiated based on cytopathic effects (CPE) in Vero cell cultures or by clinical signs in pigs. Recently a real-time reverse transcription (RT)-PCR assay for the differentiation of the original US and S-INDEL PEDV strains was developed (Wang et al., 2014b). In this study we developed RT-PCR assays to differentiate the three major types of US PEDV strains. These assays are specific and cost-effective, and could serve as a valuable diagnostic tool for the rapid differentiation of PEDV variants and for molecular epidemiological studies.
PEDV strains used in this study were isolated in Vero cell cultures in our laboratory (Oka et al., 2014). The infectious titer and RNA titer of each strain were determined by plaque assay and real-time reverse transcription-PCR (RT-qPCR), respectively, as described previously (Oka et al., 2014). The titers and GenBank accession no. are in the parentheses after each strain: S-197DEL strain PC177 (5.1 log10 plaque-forming units (PFU)/mL, 10.6 log10 genomic equivalents (GE)/mL, KM392229), original US strain PC22A (6.1 log10 PFU/mL, 11.2 log10 GE/mL, KM392224) and S-INDEL strain Iowa106 (5.6 log10 PFU/mL and 12.9 log10 GE/mL, KM392232).
Four primers, three forward and one reverse, (Fig. 1 ) (F1: 5′-ATGGTACGTTGCTAGTGCGTA-3′, F2: 5′-TGGTGAAATCCAGAGTGTCAAT-3′, F3: 5′-CCAGCTTATATGCAGGATGGAA-3′, and UR: 5′-TACCATGCACCAAAGTGGAATCAT-3′) were designed using Primer 3 V.4.0.0 (http://bioinfo.ut.ee) based on the sequence alignment of the complete genomic sequences of the three types of PEDV strains using BioEdit 7.2.1 (Hall 1999). The primary primer pair F1 and UR was used in the first PCR assay and amplified different size fragments for each strain: PC177 (293 bp), PC22A (884 bp) and Iowa106 (875 bp). Two primer pairs F2/UR and F3/UR were used in the secondary PCR to amplify strains PC22A (650 bp) and Iowa106 (358 bp), respectively. The primers were synthesized by Integrated DNA Technologies Inc (Coralville, IA).
Fig. 1.
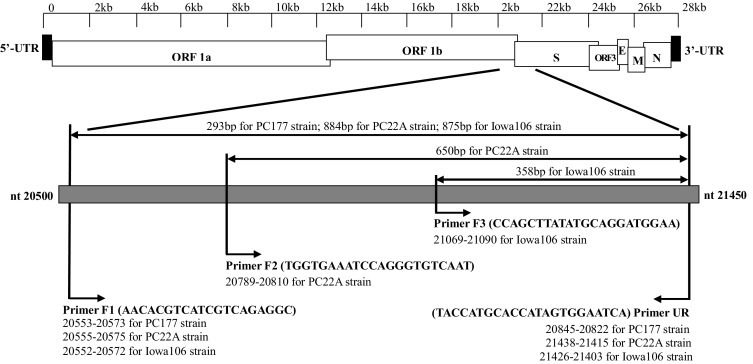
Location of external and internal primers and their combinations to generate five different sizes of product.
Total RNA was extracted from the cell culture supernatants containing each PEDV strain using RNeasy Mini Kit (Qiagen, Valencia, CA, USA). All reagents for RT and PCR were from Promega (Madsion, WI, USA). For the cDNA synthesis, a 25 μL RT reaction contained 5 μL AMV reverse transcriptase 5 × reaction buffer, 3 μL dNTP mix (10 mM), 0.5 μL AMV reverse transcriptase, 0.5 μL Rnasin ribonuclease inhibitor, 10 μL RNA, 2 μL primer UR (10 pmol/μL) and 4 μL RNase-free water. The RT reaction was performed at 42 °C for 60 min.
For the primary PCR, a 25 μL reaction volume contained 5 μL 5 × GoTaq® Flexi PCR buffer, 2.5 μL MgCl2 (25 mM), 0.5 μL dNTPs mix (10 mM), 0.5 μL GoTaq® Flexi DNA Polymerase, 1 μL of each primer F1 and UR (10 pmol/μL), 3 μL cDNA and 11.5 μL RNase-free water. PCR was performed using Applied Biosystems® 2720 Thermal Cycler (Thermo Fisher Scientific Inc, Waltham, MA, USA) with an initial denaturation step at 95 °C for 2 min, followed by 30 cycles of 95 °C for 35 s, 60 °C for 35 s, and 72 °C for 30 s, and a final extension step at 72 °C for 1 min.
The secondary PCR was carried out using primer pair F2/UR or F3/UR (10 pmol/μL), respectively, in a 25 μL reaction similar to the first PCR. The 1:100 diluted PCR products of the primary PCR were used as templates. The secondary PCR was performed with an initial denaturation at 95 °C for 2 min, followed by 25 cycles of 95 °C for 20 s, 55 °C for 20 s, and 72 °C for 15 s, and a final extension step at 72 °C for 1 min. The PCR products were subjected to electrophoresis on a 1.5% agarose gel.
To determine how the PCR assays worked for samples containing a mixed infection, the individual original virus and a mixture of the three PEDV strains with the proportions of 1:1:1, 10:1:1, 1:10:1, and 1:1:10 were tested, respectively (Fig. 2 ). In addition, porcine transmissible gastroenteritis virus (TGEV) (Miller strain, GenBank accession No. DQ811786) and porcine deltacoronavirus (PDCoV) (OH-FD22 strain, GenBank accession No. KP995364) were used to test specificity.
Fig. 2.
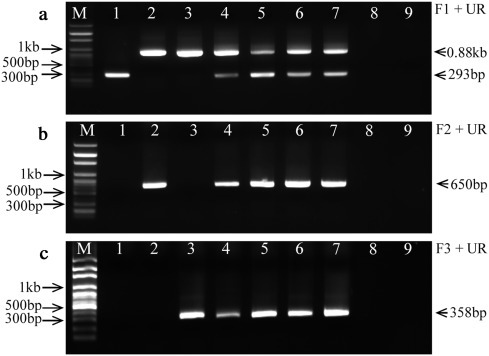
Specificity of reverse transcription-PCR assays. PCR products were analyzed by electrophoresis on a 1.5% agarose gel stained with EZ-VISION® THREE 6× loading buffer. (a) PCR products of primary amplification with primer pair F1/UR and using cDNA as templates; (b) PCR products of secondary amplification with primer pair F2/UR using 10−2 diluted first round PCR products as template; (c) PCR products of secondary amplification with primer pair F3/UR and using 10−2 diluted first round PCR products as templates. M: 100 bp ladder DNA marker (PHENIX Research Products, NC, USA); Lane 1: PEDV strain PC177; Lane 2: PEDV strain PC22A; Lane 3: PEDV strain Iowa106; Lane 4: a mixture of PC177, PC22A and Iowa106 with 1:1:1 ratio; Line 5: a mixture of PC177, PC22A and Iowa106 with 10:1:1 ratio; Line 6: a mixture of PC177, PC22A and Iowa106 with 1:10:1 ratio; Line 7: a mixture of PC177, PC22A and Iowa106 with 1:1:10 ratio; Line 8: porcine transmissible gastroenteritis virus (TGEV); Line 9: porcine deltacoronavirus (PDCoV).
The sensitivity of the primary PCR assays using individual primer set (F1/UR, F2/UR or F3/UR) was evaluated using 10-fold serial dilutions (from original cell culture supernatants to 1:105 in RNase-free water) of each strain (Fig. 3 ).
Fig. 3.
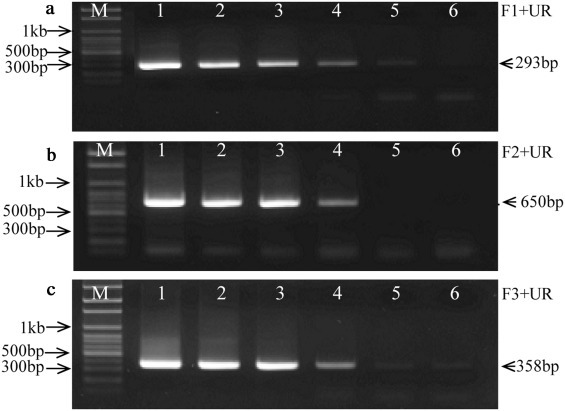
Sensitivity of the primary PCR assays with the individual primer set. The sensitivity of each PCR assay was determined using 10-fold serial dilutions (from original cell culture supernatants to 1:105 in RNase-free water) of each strain. PCR products were analyzed by electrophoresis on a 1.5% agarose gel stained with EZ-VISION® THREE 6× loading buffer. M: 100 bp ladder DNA marker (PHENIX Research Products, NC, USA); Lines 1–6: 100–10−5 diluted virus stocks. (a) PCR products of PC177 strain with primer pair F1/UR; (b) PCR products of PC22A strain with primer pair F2/UR; (c) PCR products of Iowa106 strain with primer pair F3/UR.
The primary PCR with primer pair F1/UR generated 293 bp, 884 bp and 875 bp products for PC177, PC22A and Iowa106, respectively (Fig. 2a). All the mixtures with different proportions of the three PEDV strains generated 293 bp and 0.88 kb products (mixed fragment of 884 bp and 875 bp) (Fig. 2a), but were negative for TGEV and PDCoV (Fig. 2a). In the secondary PCR, strain PC22A and Iowa106 generated 650 bp (F2/UR) and 358 bp (F3/UR) products, respectively, while strain PC177, TGEV and PDCoV were negative (Fig. 2b and c).
The primer sets F2/UR and F3/UR also detected PC22A and Iowa106 successfully in primary PCR assays, respectively. We evaluated the sensitivity of the primary PCR assays with the three primer sets for strains PC177, PC22A and Iowa106, respectively (Fig. 3). The detection limits were 1.1 log10 PFU/mL for PC177 strain (original 5.1 log10 PFU/mL, 10−4 diluted) (Fig. 3a), 3.1 log10 PFU/mL for PC22A strain (original 6.1 log10 PFU/mL, 10−3 diluted) (Fig. 3b) and 0.6 log10 PFU/mL for Iowa106 strain (original 5.6 log10 PFU/mL, 10−5 diluted), respectively (Fig. 3c).
To effectively detect the three major types of US PEDV strains, including the original US, S-INDEL and S-197DEL strains, specific and sensitive assays are required. To date, many techniques have been developed to detect PEDV strains, including conventional RT-PCR (Ishikawa et al., 1997), real-time RT-PCR (Wang et al., 2014b), immunofluorescence (IF) tests (Debouck et al., 1981), immunohistochemical techniques (Guscetti et al., 1998), immunoelectron microscopy (Pensaert and De Bouck, 1978), enzyme-linked immunosorbent assays (ELISA) (Wang et al., 2015) and a nanoparticle-assisted PCR assay (Yuan et al., 2015). However, all of these techniques can detect and differentiate only one or two types of PEDV strains. Therefore, a method that can simultaneously detect the three types of PEDV strains is urgently needed. In this study, we developed simple and economic RT-PCR assays for the detection and differentiation of different US PEDV strains.
For primer design, we optimized the original primer sequence to reduce non-specific amplifications as follows: “5′-TGGTGAAAACCAGGGTGTCAAT-3′” of the original PC22A-specific forward primer (F2) was optimized as “5′-TGGTGAAATCCAGAGTGTCAAT-3′” (nt 9 A to T and nt 14 G to A). When the primer contained the original nucleotide sequence including tri- and quad- nucleotide repeats, non-specific binding occurred and several non-specific bands were seen after agarose electrophoresis (data not shown).
The reliability of PCR assays for the detection of a virus depends on the specificity and sensitivity of primers to the target virus. In this study, the specificity of the primer pairs was evaluated using the cell culture supernatants of three types of US PEDV strains and two other porcine coronaviruses (TGEV and PDCoV). We found that our assays were specific for the PEDV strains. A previous study showed that the sensitivity of conventional RT-PCR was 104 TCID50/mL (Kweon et al., 1997). In this study, the detection limits of the primary PCR assays for strain PC177, PC22A and Iowa106 varied. In general, nested PCR could increase the sensitivity about 100–1000 fold (Cowley et al., 2004). It was our observation that the sensitivity of the nested PCR assays for the detection of PC22A (primary and the secondary PCR assays with primer sets F1/UR and F2/UR, respectively) and Iowa106 (primary and the secondary PCR assays with primer sets F1/UR and F3/UR, respectively) was much higher than the primary PCR with primer set F1/UR (for both strains) or the strain-specific primer sets (F2/UR for PC22A, and F3/UR for PC177) (data not shown). A total of 12 clinical fecal and intestinal samples (9 US original and 3 S-INDEL PEDV strains) were tested by these assays and the PCR products were sequenced to verify the specificity. We found that these assays were strain-specific (data not shown).
Currently, several PEDV variants circulate in US swine farms: original US PEDV, S-INDEL PEDV, and recombinant strains, such as Minnesota211 strain (Jarvis et al., 2015). We performed sequence analysis of the representative PEDV strains from the United States, European and Asian countries. Fig. 4 shows the sequence alignment in the internal primers F2 and F3 regions. It suggests that our assays can detect and differentiate those PEDV strains. However, this should be verified by experiments with additional PEDV strains.
Fig. 4.
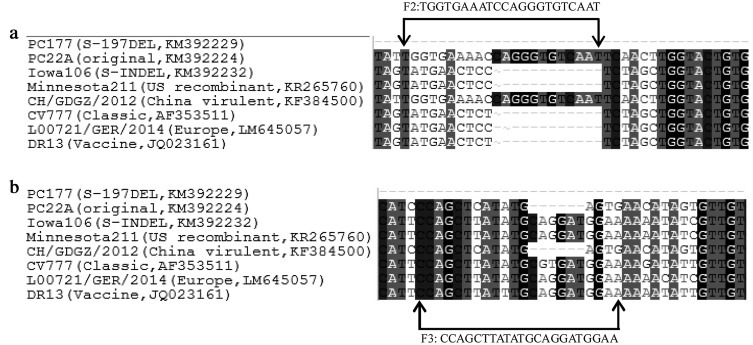
Nucleotide alignment of representative PEDV strains in the F2 and F3 primer regions. (a) Sequence and location of primer F2; and (b) Sequence and location of primer F3.
In conclusion, we have developed RT-PCR and semi-nested PCR assays that are specific and sensitive for the detection and differentiation of the three major types of US PEDV strains. These assays are urgently needed and can be used in future molecular epidemiological surveillance of various PEDV strains.
Conflict of interest
There are no conflicts of interest for any of the authors.
Acknowledgements
We thank Dr. Linda Saif for providing porcine transmissible gastroenteritis virus and porcine deltacoronavirus. We thank Drs. A. Vlasova and K. Jung for their critical review, and Ms. Susan Sommer-Wagner for her grammatical editing of this manuscript. This work was partially supported by a grant from the Ohio Agricultural Research and Development Center (OARDC) SEEDS Grant # OHOA1564 (Q. Wang, PI). We thank State Key Laboratory of Veterinary Etiological Biology, Lanzhou Veterinary Research Institute, Chinese Academy of Agricultural Sciences for scholarship support towards X. Liu. Salaries and research support were provided by state and federal funds appropriated to OARDC, The Ohio State University.
Contributor Information
Xinsheng Liu, Email: liuxinsheng@caas.cn.
Qiuhong Wang, Email: wang.655@osu.edu.
References
- Chen Q., Li G., Stasko J., Thomas J.T., Stensland W.R., Pillatzki A.E., Gauger P.C., Schwartz K.J., Madson D., Yoon K.J., Stevenson G.W., Burrough E.R., Harmon K.M., Main R.G., Zhang J. Isolation and characterization of porcine epidemic diarrhea viruses associated with the 2013 disease outbreak among swine in the United States. J. Clin. Microbiol. 2014;52:234–243. doi: 10.1128/JCM.02820-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cowley J.A., Cadogan L.C., Wongteerasupaya C., Hodgson R.A., Boonsaeng V., Walker P.J. Multiplex RT-nested PCR differentiation of gill-associated virus (Australia) from yellow head virus (Thailand) of Penaeus monodon. J. Virol. Methods. 2004;117:49–59. doi: 10.1016/j.jviromet.2003.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debouck P., Pensaert M., Coussement W. The pathogenesis of an enteric infection in pigs, experimentally induced by the coronavirus-like agent, CV777. Vet. Microbiol. 1981;6:157–165. [Google Scholar]
- Guscetti F., Bernasconi C., Tobler K., Van Reeth K., Pospischil A., Ackermann M. Immunohistochemical detection of porcine epidemic diarrhea virus compared to other methods. Clin. Diagn. Lab. Immunol. 1998;5:412–414. doi: 10.1128/cdli.5.3.412-414.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hall T.A. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999:95–98. [Google Scholar]
- Ishikawa K., Sekiguchi H., Ogino T., Suzuki S. Direct and rapid detection of porcine epidemic diarrhea virus by RT-PCR. J. Virol. Methods. 1997;69:191–195. doi: 10.1016/s0166-0934(97)00157-2. [DOI] [PubMed] [Google Scholar]
- Jarvis M.C., Lam H.C., Zhang Y., Wang L., Hesse R.A., Hause B.M., Vlasova A., Wang Q., Zhang J., Nelson M.I. Genomic and evolutionary inferences between American and global strains of porcine epidemic diarrhea virus. Prev. Vet. Med. 2015;123:175–184. doi: 10.1016/j.prevetmed.2015.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung K., Wang Q., Scheuer K.A., Lu Z., Zhang Y., Saif L.J. Pathology of US porcine epidemic diarrhea virus strain PC21A in gnotobiotic pigs. Emerg. Infect. Dis. 2014;20:662. doi: 10.3201/eid2004.131685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kweon C.H., Lee J.G., Han M.G., Kang Y.B. Rapid diagnosis of porcine epidemic diarrhea virus infection by polymerase chain reaction. J. Vet. Med. Sci. 1997;59:231–232. doi: 10.1292/jvms.59.231. [DOI] [PubMed] [Google Scholar]
- Lin C.-M., Gao X., Oka T., Vlasova A.N., Esseili M.A., Wang Q., Saif L.J. Antigenic relationships among porcine epidemic diarrhea virus and transmissible gastroenteritis virus strains. J. Virol. 2015;89:3332–3342. doi: 10.1128/JVI.03196-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oka T., Saif L.J., Marthaler D., Esseili M.A., Meulia T., Lin C.-M., Vlasova A.N., Jung K., Zhang Y., Wang Q. Cell culture isolation and sequence analysis of genetically diverse US porcine epidemic diarrhea virus strains including a novel strain with a large deletion in the spike gene. Vet. Microbiol. 2014;173:258–269. doi: 10.1016/j.vetmic.2014.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pensaert M., De Bouck P. A new coronavirus-like particle associated with diarrhea in swine. Arch. Virol. 1978;58:243–247. doi: 10.1007/BF01317606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song D., Park B. Porcine epidemic diarrhoea virus: a comprehensive review of molecular epidemiology, diagnosis, and vaccines. Virus Genes. 2012;44:167–175. doi: 10.1007/s11262-012-0713-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevenson G.W., Hoang H., Schwartz K.J., Burrough E.B., Sun D., Madson D., Cooper V.L., Pillatzki A., Gauger P., Schmitt B.J. Emergence of porcine epidemic diarrhea virus in the United States: clinical signs, lesions, and viral genomic sequences. J. Vet. Diagn. Invest. 2013;25:649–654. doi: 10.1177/1040638713501675. [DOI] [PubMed] [Google Scholar]
- Wang L., Byrum B., Zhang Y. New variant of porcine epidemic diarrhea virus, United States, 2014. Emerg. Infect. Dis. 2014;20:917–919. doi: 10.3201/eid2005.140195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L., Zhang Y., Byrum B. Development and evaluation of a duplex real-time RT-PCR for detection and differentiation of virulent and variant strains of porcine epidemic diarrhea viruses from the United States. J. Virol. Methods. 2014;207:154–157. doi: 10.1016/j.jviromet.2014.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Z., Jiyuan Y., Su C., Xinyuan Q., Lijie T., Yijing L. Development of an antigen capture enzyme-linked immunosorbent assay for virus detection based on porcine epidemic diarrhea virus monoclonal antibodies. Viral Immunol. 2015;28:184–189. doi: 10.1089/vim.2014.0065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wood E. An apparently new syndrome of porcine epidemic diarrhoea. Vet. Rec. 1977:243–244. doi: 10.1136/vr.100.12.243. [DOI] [PubMed] [Google Scholar]
- Yuan W., Li Y., Li P., Song Q., Li L., Sun J. Development of a nanoparticle-assisted PCR assay for detection of porcine epidemic diarrhea virus. J. Virol. Methods. 2015;220:18–20. doi: 10.1016/j.jviromet.2015.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]


