Abstract
A novel Rhizobium radiobacter (synonym Agrobacterium tumefaciens)-mediated approach was developed to generate stable infectious clones of plant viruses. This method uses R. radiobacter for both cloning and inoculation of infectious clones, bypassing the requirement of cloning in E. coli to avoid the instability. Only three steps are included in this method: (i) construct viral genome-encoding plasmids in vitro by one-step Gibson assembly; (ii) transform the assembled DNA products into R. radiobacter; (iii) inoculate plants with the R. radiobacter clones containing the viral genome. Stable infectious clones were obtained from two potyviruses papaya ringspot virus (PRSV) and papaya leaf distortion mosaic virus (PLDMV) using this method, whereas attempts utilizing "classical" E. coli cloning system failed repeatedly. This method is simple and efficient, and is promising for a wide application in generation of infectious clones of plant virus, especially for those which are instable in E. coli.
Keywords: Plant viruses, Infectious clone, Rhizobium radiobacter, PRSV, PLDMV, E. coli, Instability
Highlights
-
•
A novel approach was developed to generate infectious clones of plant viruses.
-
•
It uses R. radiobacter for both cloning and inoculation of infectious clones.
-
•
It bypasses the requirement of cloning in E. coli to avoid the instability.
-
•
Stable infectious clones of PRSV and PLDMV were obtained using this method.
1. Introduction
Infectious clone is critical for analysis of virus properties such as viral replication, movement, symptom development, host range and virus-host interactions. However, full-length clones of many viruses have been proved to be difficult or even impossible to perform molecular manipulation in E. coli due to their toxicity to E. coli. The instability in E. coli has been reported for an array of viruses belonging to potyviruses (Bedoya and Daros, 2010, Gao et al., 2012, Johansen, 1996, Lopez-Moya and Garcia, 2000, Olsen and Johansen, 2001, Tuo et al., 2015, Yang et al., 1998), tobraviruses (Constantin et al., 2004, Ratcliff et al., 2001), flaviviruses (Aubry et al., 2015), coronaviruses (Almazan et al., 2000), picornaviruses (Zibert et al., 1990) and pestiviruses (Rasmussen et al., 2010). Several methods have been developed to circumvent this problem, including the in vitro ligation method involving two plasmid, mutating cryptic prokaryotic promoter sites in the viral genome, and using intron insertions, low copy number plasmids, specific E. coli strains and low growth temperature (Aubry et al., 2015, Edmonds et al., 2013, Johansen and Lund, 2008, Pu et al., 2011, Siridechadilok et al., 2013, Yamshchikov et al., 2001). For generation of infectious clones of plant virus that are instable in E. coli, insertion of introns in viral genome to terminate expression of undesired toxic proteins in E. coli is the most extensively used approach so far (Desbiez et al., 2012, Gao et al., 2012, Johansen, 1996, Johansen and Lund, 2008, Lopez-Moya and Garcia, 2000, Tuo et al., 2015, Yang et al., 1998). However, determination of the numbers and locations of intron insertions required to stabilize infectious clones is time-consuming and laborious. Some sequence-independent cloning methods were also developed to assemble infectious clones of plant viruses in vivo or in vitro, such as yeast recombination system (Desbiez et al., 2012, Youssef et al., 2011), Gibson assembly and In-Fusion cloning method (Blawid and Nagata, 2015, Bordat et al., 2015, Tuo et al., 2015), but the assembly products require E. coli transformation for propagation, which means those methods just make easier viral genome assembly, not solving the problem of viral genome instability in E. coli. Thus for plant viruses that cannot maintain stability in E. coli, generation of stable infectious clones by these assembly methods still depends on the using of intron insertions (Desbiez et al., 2012, Tuo et al., 2015), which remains a major hurdle in the research of these viruses.
Here we describe a simple approach for rapid generation of infectious clones of plant viruses, especially for the plant viruses whose genomes are instable in E. coli. This method uses R. radiobacter for both cloning and inoculation of infectious clones of plant viruses, bypassing the requirement for cloning in E. coli to avoid the instability.
2. Results
2.1. The feasibility and efficiency of this method
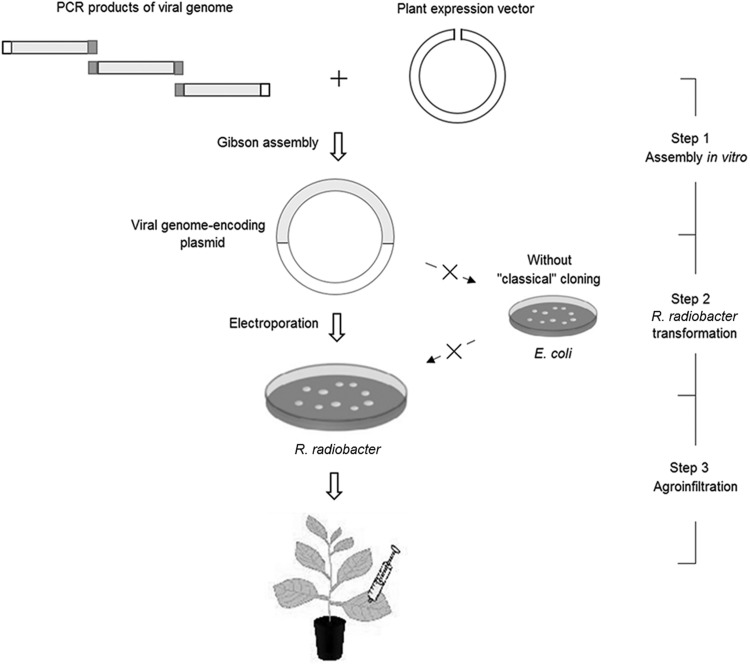
The schematic representation of the novel approach to generate stable infectious clones of plant viruses was showed in Fig. 1. This method uses R. radiobacter both for cloning and inoculation, which completely eliminates the need for plasmid propagation in E. coli. Two viruses, PRSV and PLDMV, were chosen to generate infectious clones by using this method. Full-length cDNAs of both PRSV and PLDMV were divided into two fragments for easier PCR amplification. The two fragments were cloned into pGreen-35S vector by one-step Gibson assembly. Then the assembled DNA products were transformed into R. radiobacter and E. coli, respectively, with the same growth temperature 28 °C. As shown in Table 1, the R. radiobacter transformation of PRSV or PLDMV had more than 80 colonies (average for three experiments), while the E. coli transformation of PRSV and PLDMV had 2 and 3 colonies (total for three experiments), respectively. All the five E. coli colonies were sequenced and found to be incorrect genome of the viruses with deletion or insertion mainly in the region of P3 or CI. Seventeen PCR-positive R. radiobacter colonies were sequenced and 15 colonies were found to be correct. The R. radiobacter colonies with correct viral genome were used for inoculation, and 96% of inoculated papaya plants showed system infections with similar symptoms to those caused by the wild-type viruses (Table 1 and Fig. 2). The systemically infected plants showed visibly mosaic on leaves and water-soaking streaks on petioles at 20 days post inoculation (dpi), and developed severely distorted leaves at 45 dpi, whereas the plants inoculated with the infiltrating buffer were of no symptoms (Fig. 2).
Fig. 1.
Schematic representation of the novel approach to generate stable infectious clones of plant viruses by using R. radiobacter for both cloning and inoculation.
Table 1.
Transformation of E. coli and A. tumefaciens with viral genome-encoding plasmid and infectivity of clones.
| Virus | Rep |
E. coli transformation |
A. tumefaciens transformation |
|||||
|---|---|---|---|---|---|---|---|---|
| Colonies | PCR (positive/tested) | Sequencing (correct/tested) | Colonies | PCR (positive/tested) | Sequencing (correct/tested) | Inoculation (symptomatic/tested) | ||
| PRSV | 1 | 0 | 106 | 17/20 | 3/3 | 30/31 | ||
| 2 | 2 | 2/2 | 0/2 | 52 | 12/14 | 2/3 | 30/32 | |
| 3 | 0 | 98 | 20/24 | 2/2 | 30/30 | |||
| PLDMV | 1 | 1 | 1/1 | 0/1 | 197 | 14/14 | 4/4 | 20/22 |
| 2 | 2 | 1/2 | 0/2 | 103 | 10/12 | 2/2 | 20/20 | |
| 3 | 0 | 54 | 8/12 | 2/3 | 21/22 | |||
Fig. 2.
Symptoms derived from infectious clones of PRSV and PLDMV. These photos were taken at 45 dpi. Papaya plants inoculated with the infectious clones of PRSV and PLDMV showed system infections with similar symptoms to those caused by the wild-type viruses, whereas the plants inoculated with the infiltrating buffer (mock) were symptomless.
To test whether other R. radiobacter strains and binary vectors can be used to generate infectious clones by this method, pGreen or pCambia-derived pCXSN (Chen et al., 2009) was used to generate infectious clones of PLDMV by transforming R. radiobacter strains GV3101 and LBA4404, respectively, and the infectious clones were obtained successfully by using each of these combinations of vectors and strains (data not shown).
2.2. Generation of GFP-tagged infectious clone by this method
A GFP-tagged infectious clone of PRSV was also generated by this method. The green fluorescent protein gene gfp was inserted into PRSV genome between the Nib- and CP-encoding regions ( Fig. 3A). Two fragments of PRSV and the gfp were cloned into pGreen-35S vector by one-step Gibson assembly, and then the assembled DNA product was transformed to R. radiobacter. The R. radiobacter colony with PRSV-GFP was used to inoculate papaya plants, and typical symptoms as well as green fluorescence were detected in the systemically infected leaves at 30 dpi (Fig. 3B), indicating that GFP can be expressed stably and GFP insertion did not affect virus infection and movement.
Fig. 3.
Generation of GFP-tagged infectious clone of PRSV. (A) Assembly schemes of infectious clone of PRSV-GFP. (B) Expression of GFP was observed under UV lamp and fluorescence microscope in the papaya leaves inoculated with infectious clone of PRSV-GFP at 30 dpi.
3. Discussion
Full-length cDNA clones of many RNA viruses, especially (+) RNA viruses are difficult to maintain stably in E. coli. In this study, two potyviruses PRSV and PLDMV (family Potyviridae, positive-sense RNA virus), were demonstrated to be impossible to clone in E. coli. None colony or a few colonies with incorrect viral genome were obtained in E. coli transformation. The infectious clone of Taiwan PRSV isolate has been reported to be generated by E. coli transformation (Chen et al., 2008), but the Hainan PRSV isolate was proved to be impossible to propagate correctly in E. coli in this study. The reasons may be the different sequences of viral genomes between the two isolates, or the use of different vectors. Several other potyviruses also have proved to be instable in E. coli (Bedoya and Daros, 2010, Gao et al., 2012, Johansen, 1996, Lopez-Moya and Garcia, 2000, Olsen and Johansen, 2001, Tuo et al., 2015, Yang et al., 1998). When the viral genome-encoding plasmids assembled in vitro were transformed into R. radiobacter, the colonies with correct viral genome were obtained with high efficiency. We speculate two possibilities for the stability of infectious clones in R. radiobacter: (i) low copy number of infectious clones in R. radiobacter causes less toxicity. (ii) R. radiobacter is more tolerant to toxicity than E. coli. In fact, using low copy number vectors is an approach to get stable infectious clones in E. coli for some viruses (Almazan et al., 2000, Choi et al., 1999, Gualano et al., 1998, Hurrelbrink et al., 1999). However, this does not always resolve the problem. Many viral genomes are instable in E. coli even using low copy number vectors, so more sophisticated procedures have been used, such as ligation in vitro and insertion of introns. In this study, we used R. radiobacter for both cloning and inoculation of infectious clones, and found that even a high copy number plasmid (pGreen) containing viral genome without intron insertions can be stable in R. radiobacter.
Using plasmids prepared from E. coli to transform R. radiobacter is the routine procedure for a long time. So many approaches have been developed to solve the problem of instability of infectious clones in E. coli, such as using intron insertions, low copy number plasmids and low growth temperature. In this study, the viral genome-encoding plasmids assembled in vitro were used to transform R. radiobacter, which eliminates the need for plasmid propagation in E. coli. The high efficiency of R. radiobacter transformation with in vitro assembled plasmids may be due to the newly developed DNA assembly methods, such as Gibson assembly (Gibson, 2011, Gibson et al., 2009) and In-Fusion (Sleight et al., 2010). Infectious clones can be constructed and reconstructed in vitro easily with these assembly methods.
Infectious RNA can be produced via in vitro or in vivo transcription. Performing in vitro transcription is inconvenient and expensive, so more and more infectious clones of plant viruses have been generated using R. radiobacter -mediated transfection (Ambros et al., 2011, Park et al., 2017, Shi et al., 2016, Wang et al., 2015, Wieczorek et al., 2015, Zheng et al., 2015). We developed the method for generation of infectious clones of plant viruses by using R. radiobacter for cloning and inoculation, which is convenient and not expensive.
In conclusion, a simple and efficient method was developed for the generation of stable infectious clones of plant viruses by using R. radiobacter for both cloning and inoculation, bypassing the propagation in E. coli. This method is promising for a wide application in the generation of infectious clones of plant virus, especially those which are instable in E. coli.
4. Methods
4.1. Virus source
PRSV and PLDMV were originally isolated from commercialized papaya in Hainan province, China. The complete genomic sequence of PRSV (GenBank Accession no. KF791028) (Zhao et al., 2015) and PLDMV (GenBank Accession no. JX974555) (Tuo et al., 2013) have been reported previously.
4.2. cDNA synthesis and PCR amplification
The total RNA was extracted from 100 mg of symptomatic papaya leaves with TRIzol reagent according to the manufacturer's protocol. The first-strand cDNA was synthesized from 0.5 µg of total RNA with the Takara RNA PCR Kit (AMV) Ver. 3.0 (TaKaRa, Dalian, China) using random 9 mers and oligo dT-Adaptor primers. Both the full-length PRSV and PLDMV cDNA were divided into two overlapping amplified fragments using specific primers with 25-base overlap (Table S1). A 35 S promoter and a CaMV polyA terminator were inserted into the multiple cloning sites of binary expression vector pGreenII 0000 to form pGreen-35S (Hellens et al., 2000). The pGreen-35S was amplified by primers with 25–33 homologous bases to the viral genome fragments. The PCR amplification reactions were performed with Phusion® High-Fidelity DNA Polymerases (NEB). The amplicons of the expected sizes were purified with the MiniBEST Agarose Gel DNA Extraction Kit (TaKaRa, Dalian, China). The viral genome fragments were cloned to the linearized pGreen-35S by Gibson Assembly. The reaction was performed in a total volume of 10 µL, containing 100–200 ng of each purified PCR fragments and 5 µL of 2×Gibson Mix (NEB). The reaction mix was incubated at 50 °C for 1 h, and then placed on ice for R. radiobacter transformation.
4.3. E. coli and R. radiobacter transformation
The reaction mix (5 µL) was transformed to 100 µL E. coli strain DH5α chemically competent cells (Transgen Biotech, Beijing, China) by heat shock according to the standard protocol. The transformants were selected on LB medium containing 50 mg/L kanamycin for 3 days at 28 °C to keep the same incubation temperature as that of R. radiobacter transformation, and then screened with colony PCR using the primers shown in Table S1. The plasmids were extracted from the PCR positive colonies for sequencing the full-length of PRSV and PLDMV in infectious viral clones using Sanger's method by primer walking.
The reaction mix (5 µL) was transformed to 100 µL R. radiobacter strain C58C1 (provided by Thierry Candresse, INRA, France) (Youssef et al., 2011) competent cells (containing a helper plasmid pSoup) by electroporation according to the standard protocol. The transformants were screened on LB medium containing 50 mg/L kanamycin and 50 mg/L rifampicin for 3 days at 28 °C, and then screened with colony PCR using the primers shown in Table S1. The plasmids (final 50 µL) were extracted from 100 mL culture of the positive colonies for sequencing. Or the plasmids (final 30 µL) were extracted from 4 mL culture of the positive colonies, and then used as the template to amplify two overlap fragments of the viral genomes using Q5 High-Fidelity DNA polymerase (NEB). The PCR products were used for sequencing the full-length cDNAs of infectious clones using Sanger's method by primer walking.
4.4. R. radiobacter-mediated transfection
Agroinfiltration was performed as previously reported (Sparkes et al., 2006, Yan et al., 2012). A single colony of agrobacterium for each infectious clone was picked to inoculate 10 mL YEP medium (Bacto-Trypton, 10 g/L; yeast extract, 10 g/L; NaCl, 5 g/L; pH 7.0) supplemented with 50 mg/L kanamycin and 50 mg/L rifampicin. Bacteria were grown overnight at 28 °C to obtain an OD600 of 1.0–1.5. The cultures were pelleted and washed one time using infiltration buffer (10 mM MES pH 5.6, 10 mM MgCl2, and 100 μM acetosyringone). Then cells were diluted with infiltration buffer to a final OD600 of 0.6–0.8, and incubated for 3 h at room temperature in dark before the agroinfiltration of papaya plants using 1 mL syringes without needle. The inoculated papaya seedlings were grown in a controlled environment with a 16 h light cycle at 28 °C and then for 8 h in the dark at 25 °C.
4.5. RT-PCR and fluorescence assays
Total RNA was extracted from the upper non-inoculated leaves of inoculated papaya plants and reverse transcribed as described above. The primers for the RT-PCR of each infectious viral clone were shown in Table S1. The fluorescence of the leaves from the GFP-tagged virus was detected using a long-wave length UV lamp (Black Ray model B 100 A; UV products; Upland, CA, USA) and a fluorescence microscope.
Acknowledgments
We thank Thierry Candresse (INRA, France) for kindly providing R. radiobacter strain C58C1. We are grateful to Gang Yu (Shanghai Center for Plant Stress Biology, CAS) for critical reading of the manuscript. This work was supported by grants from the National Natural Science Foundation of China (31301639), the Natural Science Foundation of Hainan Province (No. 20163121) and the Program of Hainan Association for Science and Technology Plans to Youth R & D Innovation (No. 201511).
Footnotes
Supplementary data associated with this article can be found in the online version at doi:10.1016/j.virol.2017.07.012.
Appendix A. Supplementary material
Supplementary material
References
- Almazan F., Gonzalez J.M., Penzes Z., Izeta A., Calvo E., Plana-Duran J., Enjuanes L. Engineering the largest RNA virus genome as an infectious bacterial artificial chromosome. Proc. Natl. Acad. Sci. USA. 2000;97:5516–5521. doi: 10.1073/pnas.97.10.5516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ambros S., El-Mohtar C., Ruiz-Ruiz S., Pena L., Guerri J., Dawson W.O., Moreno P. Agroinoculation of Citrus tristeza virus causes systemic infection and symptoms in the presumed nonhost Nicotiana benthamiana. Mol. Plant Microbe Interact. 2011;24:1119–1131. doi: 10.1094/MPMI-05-11-0110. [DOI] [PubMed] [Google Scholar]
- Aubry F., Nougairede A., Gould E.A., de Lamballerie X. Flavivirus reverse genetic systems, construction techniques and applications: a historical perspective. Antivir. Res. 2015;114:67–85. doi: 10.1016/j.antiviral.2014.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bedoya L.C., Daros J.A. Stability of tobacco etch virus infectious clones in plasmid vectors. Virus Res. 2010;149:234–240. doi: 10.1016/j.virusres.2010.02.004. [DOI] [PubMed] [Google Scholar]
- Blawid R., Nagata T. Construction of an infectious clone of a plant RNA virus in a binary vector using one-step Gibson Assembly. J. Virol. Methods. 2015;222:11–15. doi: 10.1016/j.jviromet.2015.05.003. [DOI] [PubMed] [Google Scholar]
- Bordat A., Houvenaghel M.C., German-Retana S. Gibson assembly: an easy way to clone potyviral full-length infectious cDNA clones expressing an ectopic VPg. Virol. J. 2015;12:89. doi: 10.1186/s12985-015-0315-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen K.C., Chiang C.H., Raja J.A., Liu F.L., Tai C.H., Yeh S.D. A single amino acid of niapro of papaya ringspot virus determines host specificity for infection of papaya. Mol. Plant Microbe Interact. 2008;21:1046–1057. doi: 10.1094/MPMI-21-8-1046. [DOI] [PubMed] [Google Scholar]
- Chen S., Songkumarn P., Liu J., Wang G.L. A versatile zero background T-vector system for gene cloning and functional genomics. Plant Physiol. 2009;150:1111–1121. doi: 10.1104/pp.109.137125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi I.R., French R., Hein G.L., Stenger D.C. Fully biologically active in vitro transcripts of the eriophyid mite-transmitted wheat streak mosaic tritimovirus. Phytopathology. 1999;89:1182–1185. doi: 10.1094/PHYTO.1999.89.12.1182. [DOI] [PubMed] [Google Scholar]
- Constantin G.D., Krath B.N., MacFarlane S.A., Nicolaisen M., Johansen I.E., Lund O.S. Virus-induced gene silencing as a tool for functional genomics in a legume species. Plant J.: Cell Mol. Biol. 2004;40:622–631. doi: 10.1111/j.1365-313X.2004.02233.x. [DOI] [PubMed] [Google Scholar]
- Desbiez C., Chandeysson C., Lecoq H., Moury B. A simple, rapid and efficient way to obtain infectious clones of potyviruses. J. Virol. Methods. 2012;183:94–97. doi: 10.1016/j.jviromet.2012.03.035. [DOI] [PubMed] [Google Scholar]
- Edmonds J., van Grinsven E., Prow N., Bosco-Lauth A., Brault A.C., Bowen R.A., Hall R.A., Khromykh A.A. A novel bacterium-free method for generation of flavivirus infectious DNA by circular polymerase extension reaction allows accurate recapitulation of viral heterogeneity. J. Virol. 2013;87:2367–2372. doi: 10.1128/JVI.03162-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao R., Tian Y.P., Wang J., Yin X., Li X.D., Valkonen J.P. Construction of an infectious cDNA clone and gene expression vector of Tobacco vein banding mosaic virus (genus Potyvirus) Virus Res. 2012;169:276–281. doi: 10.1016/j.virusres.2012.07.010. [DOI] [PubMed] [Google Scholar]
- Gibson D.G. Enzymatic assembly of overlapping DNA fragments. Methods Enzymol. 2011;498:349–361. doi: 10.1016/B978-0-12-385120-8.00015-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson D.G., Young L., Chuang R.Y., Venter J.C., Hutchison C.A., 3rd, Smith H.O. Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat. Methods. 2009;6:343–345. doi: 10.1038/nmeth.1318. [DOI] [PubMed] [Google Scholar]
- Gualano R.C., Pryor M.J., Cauchi M.R., Wright P.J., Davidson A.D. Identification of a major determinant of mouse neurovirulence of dengue virus type 2 using stably cloned genomic-length cDNA. J. General. Virol. 1998;79(Pt 3):437–446. doi: 10.1099/0022-1317-79-3-437. [DOI] [PubMed] [Google Scholar]
- Hellens R.P., Edwards E.A., Leyland N.R., Bean S., Mullineaux P.M. pGreen: a versatile and flexible binary Ti vector for Agrobacterium-mediated plant transformation. Plant Mol. Biol. 2000;42:819–832. doi: 10.1023/a:1006496308160. [DOI] [PubMed] [Google Scholar]
- Hurrelbrink R.J., Nestorowicz A., McMinn P.C. Characterization of infectious Murray Valley encephalitis virus derived from a stably cloned genome-length cDNA. J. General. Virol. 1999;80(Pt 12):3115–3125. doi: 10.1099/0022-1317-80-12-3115. [DOI] [PubMed] [Google Scholar]
- Johansen I.E. Intron insertion facilitates amplification of cloned virus cDNA in Escherichia coli while biological activity is reestablished after transcription in vivo. Proc. Natl. Acad. Sci. USA. 1996;93:12400–12405. doi: 10.1073/pnas.93.22.12400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johansen I.E., Lund O.S. Insertion of introns: a strategy to facilitate assembly of infectious full length clones. Methods Mol. Biol. 2008;451:535–544. doi: 10.1007/978-1-59745-102-4_36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez-Moya J.J., Garcia J.A. Construction of a stable and highly infectious intron-containing cDNA clone of plum pox potyvirus and its use to infect plants by particle bombardment. Virus Res. 2000;68:99–107. doi: 10.1016/s0168-1702(00)00161-1. [DOI] [PubMed] [Google Scholar]
- Olsen B.S., Johansen I.E. Nucleotide sequence and infectious cDNA clone of the L1 isolate of Pea seed-borne mosaic potyvirus. Arch. Virol. 2001;146:15–25. doi: 10.1007/s007050170187. [DOI] [PubMed] [Google Scholar]
- Park C.H., Ju H.K., Han J.Y., Park J.S., Kim I.H., Seo E.Y., Kim J.K., Hammond J., Lim H.S. Complete nucleotide sequences and construction of full-length infectious cDNA clones of cucumber green mottle mosaic virus (CGMMV) in a versatile newly developed binary vector including both 35S and T7 promoters. Virus Genes. 2017;53:286–299. doi: 10.1007/s11262-016-1415-x. [DOI] [PubMed] [Google Scholar]
- Pu S.Y., Wu R.H., Yang C.C., Jao T.M., Tsai M.H., Wang J.C., Lin H.M., Chao Y.S., Yueh A. Successful propagation of flavivirus infectious cDNAs by a novel method to reduce the cryptic bacterial promoter activity of virus genomes. J. Virol. 2011;85:2927–2941. doi: 10.1128/JVI.01986-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasmussen T.B., Reimann I., Uttenthal A., Leifer I., Depner K., Schirrmeier H., Beer M. Generation of recombinant pestiviruses using a full-genome amplification strategy. Vet. Microbiol. 2010;142:13–17. doi: 10.1016/j.vetmic.2009.09.037. [DOI] [PubMed] [Google Scholar]
- Ratcliff F., Martin-Hernandez A.M., Baulcombe D.C. Technical advance. Tobacco rattle virus as a vector for analysis of gene function by silencing. Plant J.: Cell Mol. Biol. 2001;25:237–245. doi: 10.1046/j.0960-7412.2000.00942.x. [DOI] [PubMed] [Google Scholar]
- Shi Y., Shi Y., Gu Q., Yan F., Sun X., Li H., Chen L., Sun B., Wang Z. Infectious clones of the crinivirus cucurbit chlorotic yellows virus are competent for plant systemic infection and vector transmission. J. General. Virol. 2016;97:1458–1461. doi: 10.1099/jgv.0.000453. [DOI] [PubMed] [Google Scholar]
- Siridechadilok B., Gomutsukhavadee M., Sawaengpol T., Sangiambut S., Puttikhunt C., Chin-inmanu K., Suriyaphol P., Malasit P., Screaton G., Mongkolsapaya J. A simplified positive-sense-RNA virus construction approach that enhances analysis throughput. J. Virol. 2013;87:12667–12674. doi: 10.1128/JVI.02261-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sleight S.C., Bartley B.A., Lieviant J.A., Sauro H.M. In-fusion BioBrick assembly and re-engineering. Nucleic Acids Res. 2010;38:2624–2636. doi: 10.1093/nar/gkq179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sparkes I.A., Runions J., Kearns A., Hawes C. Rapid, transient expression of fluorescent fusion proteins in tobacco plants and generation of stably transformed plants. Nat. Protoc. 2006;1:2019–2025. doi: 10.1038/nprot.2006.286. [DOI] [PubMed] [Google Scholar]
- Tuo D., Shen W., Yan P., Li C., Gao L., Li X., Li H., Zhou P. Complete genome sequence of an isolate of papaya leaf distortion mosaic virus from commercialized PRSV-resistant transgenic papaya in China. Acta Virol. 2013;57:452–455. doi: 10.4149/av_2013_04_452. [DOI] [PubMed] [Google Scholar]
- Tuo D., Shen W., Yan P., Li X., Zhou P. Rapid construction of stable infectious full-length cDNA clone of papaya leaf distortion mosaic virus using n-fusion cloning. Viruses. 2015;7:6241–6250. doi: 10.3390/v7122935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Q., Ma X., Qian S., Zhou X., Sun K., Chen X., Zhou X., Jackson A.O., Li Z. Rescue of a plant negative-strand RNA virus from cloned cDNA: insights into enveloped plant virus movement and morphogenesis. PLoS Pathog. 2015;11:e1005223. doi: 10.1371/journal.ppat.1005223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wieczorek P., Budziszewska M., Obrepalska-Steplowska A. Construction of infectious clones of tomato torrado virus and their delivery by agroinfiltration. Arch. Virol. 2015;160:517–521. doi: 10.1007/s00705-014-2266-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamshchikov V., Mishin V., Cominelli F. A new strategy in design of +RNA virus infectious clones enabling their stable propagation in E. coli. Virology. 2001;281:272–280. doi: 10.1006/viro.2000.0793. [DOI] [PubMed] [Google Scholar]
- Yan P., Shen W., Gao X., Li X., Zhou P., Duan J. High-throughput construction of intron-containing hairpin RNA vectors for RNAi in plants. PloS One. 2012;7:e38186. doi: 10.1371/journal.pone.0038186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang S.J., Revers F., Souche S., Lot H., Le Gall O., Candresse T., Dunez J. Construction of full-length cDNA clones of lettuce mosaic virus (LMV) and the effects of intron-insertion on their viability in Escherichia coli and on their infectivity to plants. Arch. Virol. 1998;143:2443–2451. doi: 10.1007/s007050050474. [DOI] [PubMed] [Google Scholar]
- Youssef F., Marais A., Faure C., Gentit P., Candresse T. Strategies to facilitate the development of uncloned or cloned infectious full-length viral cDNAs: apple chlorotic leaf spot virus as a case study. Virol. J. 2011;8:488. doi: 10.1186/1743-422X-8-488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao G., Yan P., Shen W., Tuo D., Li X., Zhou P. Complete genome sequence of papaya ringspot virus isolated from genetically modified papaya in Hainan Island, China. Genome Announc. 2015:3. doi: 10.1128/genomeA.01056-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng H., Xiao C., Han K., Peng J., Lin L., Lu Y., Xie L., Wu X., Xu P., Li G., Chen J., Yan F. Development of an agroinoculation system for full-length and GFP-tagged cDNA clones of cucumber green mottle mosaic virus. Arch. Virol. 2015;160:2867–2872. doi: 10.1007/s00705-015-2584-y. [DOI] [PubMed] [Google Scholar]
- Zibert A., Maass G., Strebel K., Falk M.M., Beck E. Infectious foot-and-mouth disease virus derived from a cloned full-length cDNA. J. Virol. 1990;64:2467–2473. doi: 10.1128/jvi.64.6.2467-2473.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material