Abstract
Background
There is limited experience regarding the safety and efficacy of checkpoint inhibitors (CPI) in patients with autoimmune disorders (AD) and advanced urological cancers as they are generally excluded from clinical trials due to risk of exacerbations.
Methods
This multicenter retrospective cohort analysis of patients with advanced renal cell cancer (RCC) and urothelial cancer (UC) with pre-existing AD treated with CPI catalogued the incidence of AD exacerbations, new immune-related adverse events (irAEs) and clinical outcomes. Competing risk models estimated cumulative incidences of exacerbations and new irAEs at 3 and 6 months.
Results
Of 106 patients with AD (58 RCC, 48 UC) from 10 centers, 35 (33%) had grade 1/2 clinically active AD of whom 10 (9%) required corticosteroids or immunomodulators at baseline. Exacerbations of pre-existing AD occurred in 38 (36%) patients with 17 (45%) requiring corticosteroids and 6 (16%) discontinuing CPI. New onset irAEs occurred in 40 (38%) patients with 22 (55%) requiring corticosteroids and 8 (20%) discontinuing CPI. Grade 3/4 events occurred in 6 (16%) of exacerbations and 13 (33%) of new irAEs. No treatment-related deaths occurred. Median follow-up was 15 months. For RCC, objective response rate (ORR) was 31% (95% CI 20% to 45%), median time to treatment failure (TTF) was 7 months (95% CI 4 to 10) and 12-month overall survival (OS) was 78% (95% CI 63% to 87%). For UC, ORR was 40% (95% CI 26% to 55%), median TTF was 5.0 months (95% CI 2.3 to 9.0) and 12-month OS was 63% (95% CI 47% to 76%).
Conclusions
Patients with RCC and UC with well-controlled AD can benefit from CPI with manageable toxicities that are consistent with what is expected of a non-AD population. Prospective study is warranted to comprehensively evaluate the benefits and safety of CPI in patients with AD.
Keywords: autoimmunity, immunotherapy, kidney neoplasms, urologic neoplasms
Background
Checkpoint inhibitors (CPI) are routinely used across a wide spectrum of cancers types including advanced renal cell cancer (RCC) and urothelial carcinoma (UC).1 2 A distinctive class of side effects, collectively termed immune-related adverse events (irAEs) akin to physiological autoimmune diseases (AD), has been recognized and are inherent to the mechanism of action potentiating T-cell driven immune responses via the programmed death-1 (PD-1) and cytotoxic T-lymphocyte associated protein-4 (CTLA-4) pathways. While the majority of irAEs are manageable and reversible, some episodes can be severe with rare permanent or fatal outcomes.2–4 Generally, patients with pre-existing AD have been excluded from clinical trials evaluating CPI given concerns of exacerbating the underlying AD and obfuscating the toxicity profile of the drug.
AD encompass a broad spectrum of diseases resulting from a misdirected immune system attack on self.3 4 Their prevalence is rising and varies significantly depending on disorder type and geoepidemiological factors; it is estimated that up to 24–50 million North Americans have an AD.5 Associations between AD and cancer have been described6 with upwards of 30% of patients with RCC harboring a comorbid AD in one series.7
No prospective studies have defined strategies for effectively managing CPI in patients with documented AD, and clinical practice is variable. Given the rarity of CPI use in patients with pre-existing AD and safety concerns, clinical experience is relatively limited and the literature consists mostly of retrospective series and case reports.8–15 Recognizing the scarcity of data, we sought to investigate the safety and antitumor activity of CPI in patients with advanced RCC and UC with pre-existing AD across multiple centers to capture real-world evidence.
Methods
Study population
We undertook a multicenter, international retrospective cohort analysis of patients with advanced RCC and UC who had a documented pre-existing AD, received at least one dose of CPI monotherapy or in combination, and who had adequate baseline and on-therapy clinical and imaging data. Each participating center obtained institutional review board approval.
Investigators collected baseline clinicodemographic, pathological, systemic therapy, response and toxicity data via chart review using a uniform database template. AD definitions were based on the American Autoimmune Related Diseases Association; full listing available in online supplementary table 1.5 All AD symptoms and irAEs were investigator assessed using Common Terminology Criteria for Adverse Events version 5 and recorded from the date of first CPI dose to 90 days after last dose. Baseline AD severity was characterized as historical or clinically active and whether on concurrent immunomodulators. Exacerbations were considered flares of symptoms consistent with underlying AD. New irAEs were defined as development of irAEs not related to the underlying AD. Toxicities leading to treatment discontinuation or necessitating therapeutic intervention were captured. Clinical and radiological assessments were not standardized and were performed according to each center’s standard of care. Response was investigator assessed using general Response Evaluation Criteria in Solid Tumors principles.16 Multidisciplinary care and involvement of the AD specialist during CPI treatment were performed per local practice.
jitc-2020-000538supp001.pdf (1MB, pdf)
Statistical analysis
Patient and disease characteristics were described using frequencies (percentages) and medians (ranges). Overall response rate (ORR) was defined as the proportion of patients with complete responses or partial responses and calculated as percentage of patients who achieved ORR along with 95% Clopper-Pearson exact CI. Patients not evaluable for response were conservatively included as non-responders. Time-to-treatment failure (TTF) was determined from CPI initiation until therapy discontinuation for any reason, including progression of disease (PD), toxicity or death. Overall survival (OS) was calculated from CPI initiation until death or last follow-up. Distributions of TTF and OS rates were estimated using the Kaplan-Meier methodology for the overall cohort and by subgroups. No formal comparisons were made for subgroup analyses given the small sample size. Competing risk models estimated cumulative incidences of AD exacerbations and/or new irAEs at 3 months and 6 months, whereas treatment discontinuation due to progression or other reasons without irAE was considered a competing risk. Statistical analyses were performed using SAS V.9.4 (SAS Institute).
Results
Baseline characteristics
Across 10 centers in the USA and Europe, 106 patients with documented pre-existing AD and advanced RCC (n=58) or UC (n=48) were identified who were treated with CPI between 2015 and 2018. Most patients received CPI as the first line or second line (n=92; 87%) and as PD(L)-1 inhibitor monotherapy (n=85; 80%) (table 1).17–19 Dual CPI (anti-PD(L)-1+anti-CTLA-4) were administered in 16% (n=9) of patients with RCC and 2% (n=1) of patients with UC. Eleven (19%) patients with RCC received CPI+vascular endothelial growth factor (VEGF) inhibitor.
Table 1.
Clinicodemographic characteristics at baseline
| Baseline characteristics | RCC (n=58) | UC (n=48) | Overall (n=106) |
| N (%) | N (%) | N (%) | |
| Age | |||
| Median, years (range) | 66 (25–82) | 72 (47–87) | 68(25-87) |
| Gender | |||
| Male | 40 (69) | 35 (73) | 75 (71) |
| Female | 18 (31) | 13 (27) | 31 (29) |
| Histology | |||
| ccRCC | 46 (79) | NA | NA |
| nccRCC | 12 (21) | NA | NA |
| UC* | NA | 47 (98) | NA |
| Non-UC† | NA | 1 (2) | NA |
| ECOG performance status | |||
| 0 | 21 (36) | 12 (25) | 33 (31) |
| 1 | 29 (50) | 28 (58) | 57 (54) |
| 2–3 | 8 (14) | 8 (17) | 16 (15) |
| RCC IMDC risk group17 | |||
| Favorable | 10 (17) | NA | NA |
| Intermediate | 39 (67) | NA | NA |
| Poor | 9 (16) | NA | NA |
| UC risk group | |||
| Platinum-sensitive group: Bajorin criteria19 | n=21 | ||
| 0 | NA | 5 (24) | NA |
| 1 | NA | 13 (62) | NA |
| 2 | NA | 3 (14) | NA |
| UC risk group | |||
| Platinum-refractory group: Bellmunt criteria18 | n=33 | ||
| 0 | NA | 2 (6) | NA |
| 1 | NA | 8 (24) | NA |
| 2 | NA | 19 (58) | NA |
| 3 | NA | 4 (12) | NA |
| Number of prior systemic therapies | |||
| 0 | 20 (34) | 22 (46) | 42 (40) |
| 1 | 26 (45) | 24 (50) | 50 (47) |
| 2 | 4 (7) | 2 (4) | 6 (6) |
| ≥3 | 8 (14) | 0 (0) | 8 (8) |
| Type of CPI regimen | |||
| PD-1/PD-L1 inhibitor monotherapy | 38 (66) | 47 (98) | 85 (80) |
| PD-1/PD-L1 +CTLA-4 inhibitor | 9 (16) | 1 (2) | 10 (9) |
| PD-1/PD-L1 +VEGF inhibitor | 11 (19) | 0 (0) | 11 (10) |
| Sites of metastases‡ | |||
| Lymph nodes | 46 (79) | 43 (90) | 89 (84) |
| Lung | 42 (72) | 24 (50) | 66 (62) |
| Bone | 16 (28) | 8 (17) | 24 (23) |
| Liver | 12 (21) | 14 (29) | 26 (25) |
| Brain | 5 (9) | 0 (0) | 5 (5) |
*Includes pure urothelial histology and mixed histology with predominant urothelial component.
†Includes one patient with a small cell bladder tumor.
‡Patients may have had more than one metastatic site.
ccRCC, clear cell renal cell carcinoma; CPI, checkpoint inhibitors; CTLA-4, cytotoxic T lymphocyte associated protein-4; ECOG, Eastern Cooperative Oncology Group; IMDC, International Metastatic Renal Cell Carcinoma Database Consortium; NA, not applicable; nccRCC, non-clear cell renal cell carcinoma; PD-1, programmed death-1; PD-L1, programmed death ligand-1; RCC, renal cell carcinoma; UC, urothelial carcinoma; VEGF, vascular endothelial growth factor.
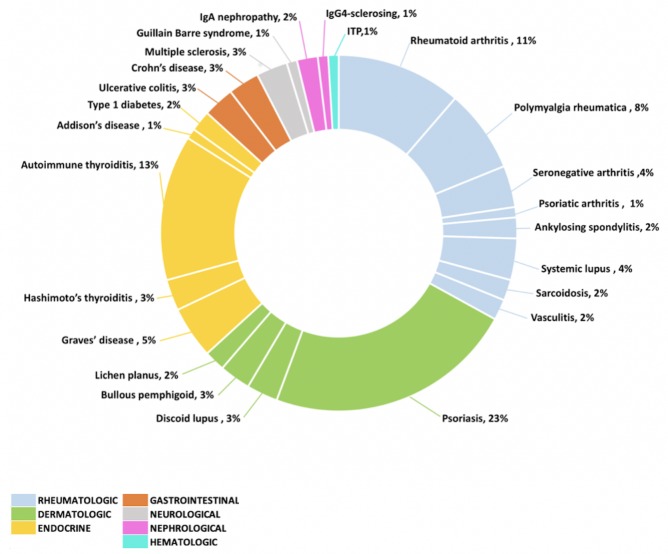
A broad spectrum of baseline AD was captured (figure 1). Psoriasis (n=24, 23%), thyroiditis (n=14, 13%), rheumatoid arthritis (n=12, 11%) and polymyalgia rheumatica (n=8, 8%) were most common. Other clinically relevant disorders were identified such as inflammatory bowel diseases (IBD, n=6, 6%), systemic lupus erythematosus (n=4, 4%), multiple sclerosis (n=3, 3%), sarcoidosis (n=2, 2%) and vasculitis (n=2, 2%) including one case15 of granulomatosis with polyangiitis.
Figure 1.
Autoimmune disorder types at baseline. IgA, Immunoglobulin A; ITP, immune thrombocytopenic purpura.
At CPI initiation, 35 (33%) had baseline grade 1/2 AD symptoms including rheumatological (n=15, 14%), dermatological (n=13, 12%), thyroiditis (n=2, 2%), ulcerative colitis (n=2, 2%), multiple sclerosis (n=2, 2%) and IgG4-related sclerosing kidney disease (n=1, 1%) (table 2, online supplementary figure 1). Ten patients required baseline immunosuppression with systemic corticosteroids (n=5) or other immune-modulating agents (one each: hydroxychloroquine, mesalamine, sulfasalazine, teriflunomide and methotrexate).
Table 2.
Autoimmune disorders (AD) symptoms and management at baseline
| Characteristic | RCC (n=58) N (%) |
UC (n=48) N (%) |
Overall (n=106) N (%) |
| AD symptoms | |||
| Asymptomatic | 41 (71) | 30 (63) | 71 (67) |
| Symptomatic | 17 (29) | 18 (38) | 35 (33) |
| Severity of baseline AD symptoms* | |||
| Grade 0 (asymptomatic) | 41 (71) | 30 (63) | 71 (67) |
| Grade 1 | 13 (22) | 13 (27) | 26 (25) |
| Grade 2 | 3 (5) | 3 (6) | 6 (6) |
| Grade 3–4 | 0 (0) | 0 (0) | 0 (0) |
| Unknown | 1 (2) | 2 (4) | 3 (3) |
| Concurrent AD treatment at CPI initiation | |||
| Topical corticosteroids | 1 (2) | 2 (4) | 3 (3) |
| Systemic corticosteroids | 2 (3) | 3 (6) | 5 (5) |
| Immunomodulatory agents† | 4 (7) | 1 (2) | 5 (5) |
*Only the worst grade for the same symptom is captured.
†Five patients received one of the following treatments: hydroxychloroquine (rheumatoid arthritis), mesalamine (ulcerative colitis), sulfasalazine (ulcerative colitis), teriflunomide (multiple sclerosis) and methotrexate (psoriatic arthritis).
CPI, checkpoint inhibitor; RCC, renal cell carcinoma; UC, urothelial carcinoma.
AD exacerbations
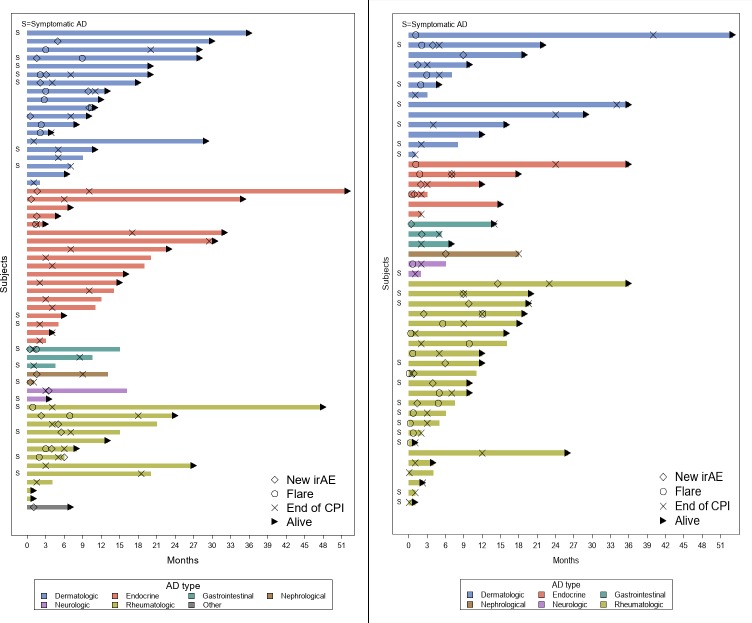
Globally, 38 (36%) patients experienced an exacerbation of their underlying AD. Median time to exacerbation was 76 days (range: 25–315) for RCC (n=18, 31%) and 33 days (range: 1–368) for UC (n=20, 42%) (figure 2, table 3). The cumulative incidence at 3 and 6 months in the overall cohort was 29% (95% CI 20% to 38%) and 32% (95% CI 23% to 41%), respectively.
Figure 2.
Swimmers plot denoting time on checkpoint inhibitors (CPI) treatment in patients with (A) renal cell carcinoma and (B) urothelial carcinoma with pre-existing autoimmune disorder (AD) with time to onset of AD exacerbation and/or new immune-related adverse event (irAE) and time of CPI discontinuation.
Table 3.
Characteristics and management of autoimmune disorder (AD) exacerbations and new immune-related adverse events (irAEs) on checkpoint inhibitors (CPI)
| RCC | UC | Overall | ||||
| AD flare | New irAE |
AD flare | New irAE |
AD flare | New irAE |
|
| In all patients | (n=58) | (n=48) | (n=106) | |||
| Total events, N (%) | 18 (31%) | 22 (38%) | 20 (42%) | 18 (38%) | 38 (36%) | 40 (38%) |
| Cumulative incidence, % (95% CI) | ||||||
| At 3 months | 21 (11 to 33) | 22 (12 to 34) | 31 (19 to 45) | 17 (8 to 29) | 29 (20 to 38) | 22 (15 to 30) |
| At 6 months | 21 (11 to 33) | 34 (21 to 47) | 38 (24 to 52) | 26 (14 to 39) | 32 (23 to 41) | 32 (23 to 41) |
| In patients with irAE | (n=18) | (n=22) | (n=20) | (n=18) | (n=38) | (n=40) |
| Median time from CPI start to event, days (range) | 76 (25–315) | 56 (2–305) | 33 (1–368) | 120 (12–443) | 61 (1–368) | 68 (2–443) |
| Severity of symptoms*, N (%) | ||||||
| Grade 1 | 6 (33%) | 7 (32%) | 2 (10%) | 3 (17%) | 8 (21%) | 10 (25%) |
| Grade 2 | 7 (39%) | 8 (36%) | 12 (60%) | 7 (39%) | 19 (50%) | 15 (38%) |
| Grade 3–4 | 2 (11%) | 7 (32%) | 4 (20%) | 6 (33%) | 6 (16%) | 13 (33%) |
| Unknown | 3 (17%) | 0 (0%) | 2 (10%) | 2 (11%) | 5 (13%) | 2 (5%) |
| Received topical corticosteroids, N (%) | ||||||
| Yes | 9 (50%) | 0 (0%) | 3 (15%) | 2 (6%) | 12 (32%) | 2 (5%) |
| No | 9 (50%) | 22 (100%) | 17 (85%) | 16 (94%) | 26 (68%) | 38 (95%) |
| Received systemic corticosteroids, N (%) | ||||||
| Yes | 5 (28%) | 11 (50%) | 12 (60%) | 11 (61%) | 17 (45%) | 22 (55%) |
| No | 13 (72%) | 11 (50%) | 8 (40%) | 7 (39%) | 21 (55%) | 18 (45%) |
| Received immunomodulatory agents†, N (%) | ||||||
| Yes | 1 (6%) | 0 (0%) | 1 (5%) | 0 (0%) | 2 (5%) | 0 (0%) |
| No | 17 (94%) | 22 (100%) | 19 (95%) | 18 (100%) | 36 (95%) | 40 (100%) |
| CPI management, N (%) | ||||||
| Continued | 13 (72%) | 11 (50%) | 11 (55%) | 7 (39%) | 24 (63%) | 18 (45%) |
| Temporarily discontinued | 3 (17%) | 8 (36%) | 5 (25%) | 6 (33%) | 8 (21%) | 14 (35%) |
| Permanently discontinued | 2 (11%) | 3 (14%) | 4 (20%) | 5 (28%) | 6 (16%) | 8 (20%) |
| irAE outcome‡, N (%) | ||||||
| Ongoing but controlled | 7 (39%) | 6 (27%) | 12 (60%) | 5 (28%) | 19 (50%) | 11 (28%) |
| Ongoing but uncontrolled | 3 (17%) | 0 (0%) | 3 (15%) | 0 (0%) | 6 (16%) | 0 (0%) |
| Resolved | 8 (44%) | 16 (73%) | 5 (25%) | 11 (61%) | 13 (34%) | 27 (68%) |
| Unknown | 0 (0%) | 0 (0%) | 0 (0%) | 2 (11%) | 0 (0%) | 2 (5%) |
*Only the worst grade for the same symptom is captured. No grade 5 events occurred.
†Two patients received one of the following treatments: methotrexate (polymyalgia rheumatica), rituximab (granulomatosis with polyangiitis).
‡Controlled was defined as a now asymptomatic adverse event still requiring immunosuppression agents. Uncontrolled was defined as an irAE that was still symptomatic at the time of the analysis.
RCC, renal cell carcinoma; UC, urothelial carcinoma.
Exacerbations occurred most frequently among patients with rheumatological disorders (n=18/36, 50%), followed by dermatological (n=14/32, 44%), neurological (n=1/4, 25%), endocrine (n=5/25, 20%) and gastrointestinal (n=1/6, 17%; online supplementary tables 2–3) and were generally low grade (grade 1/2: n=27, 71%). Six patients (16%) experienced grade 3 exacerbations including arthralgias (n=4), neurological events (neuromuscular weakness, loss of sensation) (n=2), myalgia (n=1), and colitis (n=1); 33% had more than one symptom. No grade 4/5 events were observed.
Regarding the impact of baseline symptom status, 40% (n=14/35) of symptomatic and 34% (n=24/71) of asymptomatic patients experienced AD exacerbations. There was a higher cumulative incidence at 3 months among symptomatic patients compared with non-symptomatic patients at baseline: 30% (95% CI 16% to 46%) and 24% (95% CI 14% to 34%), respectively (online supplementary figure 2). A lower exacerbation rate was described among patients treated with single agent CPI: 32% (n=27/85) compared with 52% (n=11/21) treated with combinations. Similar frequencies were observed between dual CPI (n=5/10, 50%) and CPI+VEGF inhibitors (n=6/11, 55%). Of 11 patients who flared on combination therapy, only one patient treated with CPI+VEGF discontinued treatment (both agents). More exacerbations were described among patients receiving baseline immunomodulators (n=5/10, 50%) compared with patients who were not (n=33/96, 35%). Details of the 13 patients with AD of clinical interest such as IBD, neurological and renal disorders, of whom five were symptomatic and three required baseline chronic immunomodulators, are described (online supplementary table 4). Of these, only two patients with ulcerative colitis and Guillain-Barre syndrome experienced exacerbations and both discontinued CPI.
Among the 38 AD exacerbations, immunotherapy was continued in 24 patients (63%) and discontinued in 14 patients (37%): eight (21%) temporarily, six (16%) permanently. AE types leading to discontinuation were arthralgia (n=3), myalgia (n=1), diarrhoea (n=1) and neuropathy (n=1); all were grade 3 except for neuropathy (unknown grade). Corticosteroids were required in 17/38 (45%) patients, of whom 3 (8%) became steroid refractory: 1 patient was on mesalamine at baseline for ulcerative colitis and 2 required rituximab and methotrexate for granulomatosis with polyangiitis and polymyalgia rheumatica exacerbations, respectively. On analysis, six patients had ongoing exacerbations: one was receiving systemic corticosteroids, two topical corticosteroids, two supportive care and one had discontinued CPI. Ten patients received subsequent non-CPI systemic therapy (online supplementary table 5).
New onset irAEs (unrelated to primary AD)
New onset irAEs occurred in 40 (38%) patients with pre-existing AD with similar frequencies among the RCC (n=22, 38%) and UC cohorts (n=18, 38%). Median time after CPI initiation was 56 days (range: 2–305) and 120 days (range: 12–443), respectively (table 3, figure 2). The cumulative incidence was 22% (95% CI 15% to 30%) at 3 months and 32% (95% CI 23% to 41%) at 6 months in the overall cohort.
The most frequent new irAEs were colitis (n=9, 8%), rash (n=8, 8%) and hypothyroidism (n=7, 7%) (online supplementary table 3). Grade 3 events occurred in 12 patients (30%) including colitis (n=4), nephritis (n=2), adrenal insufficiency (n=1), hypophysitis (n=1), arthritis (n=1), pneumonitis (n=1), rash (n=1) and hepatitis (n=1). One patient developed grade 4 hepatitis. No grade 5 irAEs occurred.
Of the 40 patients who developed new irAEs, 22 (55%) received corticosteroids with only one steroid-refractory case of grade 3 colitis requiring infliximab. CPI was discontinued in 22 (55%) patients: 14 (35%) temporarily and 8 (20%) permanently. The AEs types necessitating permanent discontinuation were colitis (n=3), pneumonitis (n=3), adrenal insufficiency (n=1) and rash (n=1); all events were grade 3 with the exception of one unknown grade pneumonitis. On analysis, 11 (28%) patients had an ongoing but resolving irAE and 27 (68%) had experienced complete resolution.
Risk of any AD exacerbation and/or new irAE
The cumulative incidence of AD exacerbation and/or new irAE was 45% (95% CI 35% to 54%) and 53% (95% CI 43% to 62%) at 3 and 6 months, respectively (online supplementary figure 2) with 16 (15%) patients developing both event types (AD exacerbation and new irAE; figure 2). There was a slightly higher rate of AD exacerbations in patients who also developed a new irAE (n=16/40, 40%) compared with those who experienced only AD exacerbation (n=24/68, 35%). Similarly, risk for a new irAE was higher in patients who also experienced an AD exacerbation (n=16/38, 42%) compared with patients who had a new irAE but no AD exacerbation (24/68, 35%). At time of analysis, the new irAE had resolved or was controlled in all 16 patients and the AD exacerbation in 14 patients. Two patients with symptomatic AD exacerbations (arthralgias and hypothyroidism) were still receiving CPI and supportive treatment for grade 1 events. CPI was permanently discontinued in five patients due to the new irAE and in two due to AD exacerbation.
Clinical outcomes
For the RCC cohort, ORR was 31% (95% CI 20% to 45%) including four CRs. Five non-evaluable patients were included as non-responders. Median follow-up was 13 months (range: 1–52). Forty-one (71%) discontinued treatment because of radiological/clinical progression (n=30, 73%), toxicity (n=6, 15%) or physician choice (n=5, 12%). Median TTF was 7 months (95% CI 4 to 10) with 1-year OS of 78% (95% CI 63% to 87%; online supplementary figure 3).
For the UC cohort, ORR was 40% (95% CI 26% to 55%) with six CRs. The eight non-evaluable patients were included as non-responders. Median follow-up was 15 months (range: 1–53). CPI was discontinued in 41 (85%) patients due to PD (n=18, 44%), toxicity (n=10, 24%), physician choice (n=7, 17%), patient choice (n=3, 7%), and therapy completion (n=3, 7%). Median TTF was 5 months (95% CI 2 to 9) with 1-year OS of 63% (95% CI 47% to 76%; online supplementary figure 3).
Subgroup analysis across RCC and UC by baseline symptom status, treatment lines and types of treatment was analyzed with no significant differences (table 4).
Table 4.
Efficacy outcomes: subset analyses based on autoimmune disorder (AD) baseline symptom status, treatment line and type of treatment
| ORR | TTF | OS | |||||
| Total | N | % (95% CI) | No of events | Median TTF, months (95% CI) | No of events | 12-month OS rate, % (95% CI) | |
| RCC | |||||||
| Overall | 58 | 18 | 31 (20 to 45) | 41 | 7 (4 to 10) | 21 | 78 (63 to 87) |
| Baseline symptomatic | |||||||
| Yes | 17 | 7 | 41 (18 to 67) | 12 | 7 (2 to 19) | 8 | 68 (40 to 86) |
| No | 41 | 11 | 27 (14 to 43) | 29 | 7 (4 to 10) | 13 | 82 (63 to 91) |
| Treatment line | |||||||
| First line | 20 | 10 | 50 (27 to 73) | 14 | 7 (4 to 20) | 3 | 86 (54 to 96) |
| Second line or more | 38 | 8 | 21 (10 to 37) | 27 | 6 (3 to 9) | 18 | 73 (55 to 85) |
| Type of treatment | |||||||
| Anti-PD-1/PD-L1 | 38 | 8 | 21 (10 to 37) | 25 | 7 (4 to 11) | 16 | 74 (55 to 85) |
| Anti-PD-1/PD-L1 +anti-CTLA-4 | 9 | 2 | 22 (3 to 60) | 6 | 6 (2 to 10) | 1 | 100 |
| Anti-PD-1/PD-L1 +anti-VEGF | 11 | 8 | 73 (39 to 94) | 10 | 5 (4 to 18)* | 4 | 82 (45 to 95) |
| UC | |||||||
| Overall | 48 | 19 | 40 (26 to 55) | 41 | 5 (2 to 9) | 18 | 63 (47 to 76) |
| Baseline symptomatic | |||||||
| Yes | 18 | 5 | 28 (10 to 53) | 14 | 4 (1 to 20) | 8 | 49 (23 to 71) |
| No | 30 | 14 | 47 (28 to 66) | 27 | 5 (2 to 12) | 10 | 71 (51 to 85) |
| Treatment line | |||||||
| First line | 22 | 8 | 36 (17 to 59) | 17 | 5 (2 to 9) | 6 | 69 (43 to 85) |
| Second line or more | 26 | 11 | 42 (23 to 63) | 24 | 3 (2 to 12) | 12 | 59 (37 to 76) |
*Reasons of discontinuation among the eight patients who achieved complete response or partial response: toxicity (n=3), physician choice (n=2), progressive disease (n=2) and unknown (n=1).
CTLA-4, cytotoxic T lymphocyte associated protein-4; irAE, immune-related adverse event; ORR, overall response rate; OS, overall survival; PD-1, programmed death-1; PD-L1, programmed death ligand-1; RCC, renal cell carcinoma; TTF, time-to-treatment failure; UC, urothelial carcinoma; VEGF, vascular endothelial growth factor.
Discussion
Leveraging a large international collaboration, we captured real-world evidence of the safety profile and efficacy of CPI in RCC and UC patients with pre-existing AD. Based on pivotal trials showing significant improvements in ORR and OS, CPI are now broadly employed in advanced RCC and UC to treat both treatment-naïve and previously treated disease.20–24 To minimize the risk of heightened treatment-related toxicity, most prospective studies excluded patients with pre-existing AD. There is a meager although growing literature mostly comprised cases series and retrospective experiences of CPI in patients with AD with melanoma or non-small cell lung cancer (NSCLC) which describe rates of irAEs ranging from 23% to 42%.8–12 To our knowledge, we report the largest series of the CPI administration in patients with pre-existing AD and specifically evaluate patients with urological cancer. In our study, the rates of AD exacerbations and new irAEs were similar at 36% and 38%, respectively, and in line with the reported melanoma and NSCLC series.
While irAEs can develop at any time, including after CPI cessation, they generally appear within the first few weeks to months.3 In our AD cohort, the median time to development of an irAE was <3 months from CPI initiation and similar in time frame, 61 and 68 days, across the two cancers. In phase 3 RCC and UC studies evaluating single agent CPIs, irAE were generally reversible with grade 3/4 events ranging from 15% to 19% and treatment discontinuation due to toxicity in 6%–8%.20 21 In our analyses, toxicity severity was comparable for AD exacerbations with 16% being grade 3/4. However, we observed higher rates of new irAEs (33% grade 3/4) and CPI interruption (16% for AD exacerbations, 20% for new irAEs) than the phase 3 studies. Corticosteroid use was similar for exacerbations and new irAEs (45% vs 55%) in our series.
Our intensive chart review permitted evaluation of detailed information on subset populations. CPI combinations are standard of care in treatment-naïve patients with RCC resulting in higher rates of irAEs, but also higher efficacy than monotherapy. In our series, while combinations induced more AD exacerbations or new irAEs, only one patient permanently discontinued anti-PD-L1+VEGF due to polymyalgia rheumatica exacerbation. More frequent exacerbations were seen among those with clinically active AD at baseline and in patients receiving chronic immunosuppressants. With respect to ADs of clinical concern, such as neurological (eg, multiple sclerosis, Guillain-Barré syndrome) or IBD, exacerbations did not appear more frequent but perhaps were more aggressive as most resulted in CPI discontinuation (online supplementary table 4),15 although this latter finding could be biased by physician comfort level and experience.
Robust clinical activity was observed in our AD cohort. One hypothesis is that CPI may have greater efficacy in patients with AD (especially those not on immune suppression) due to a propensity for immune stimulation.1 2 However, in the Dana-Farber single institution experience evaluating CPI in 52 patients with AD (11%) compared with a control cohort of 442 patients without AD, we did not find statistically significant differences in the cumulative incidence of new irAEs at 6 months (55% vs 37%, p=0.69 for RCC; 33% vs 27%, p=0.64 for UC, respectively, for AD and non-AD) or in efficacy outcomes.25 Toxicity was generally mild and manageable in this asymptomatic or mildly symptomatic AD population at baseline. Another retrospective study of patients with different solid tumors, mostly NSCLC and melanoma, treated with PD-1 inhibitors compared the incidence of irAEs in patients with pre-existing AD (n=85) to a control cohort without AD (n=666).14 While incidence of any grade irAEs was higher in patients with AD (66% vs 40%) with a significant rate of AD exacerbations (47%), there was no difference in survival outcomes. However, multiple pooled analyses of non-AD patients with a variety of solid tumors support enhanced checkpoint blockade efficacy in patients who experience irAEs.26–28
Clinical experiences capturing real-world evidence in patients who are under-represented in clinical trials are critical to optimize CPI management in these populations. The SAUL (NCT02928406) study prospectively evaluated atezolizumab in patients with metastatic urinary tract tumors and complex comorbidities who are often excluded from the pivotal trials.29 Only 35 patients with AD were included, and degree of AD severity was limited (most frequent was psoriasis, n=15). Investigators described consistent efficacy, more common treatment-related AEs (69%) but low rates of treatment discontinuation (9%). To our knowledge, our series of 106 patients is the largest to provide real-world evidence highlighting the potential benefit and tolerability of CPI among patients with urological cancer with well-controlled, pre-existing AD. It highlights the moderate risk of AD exacerbations and new irAEs, especially in patients with active conditions at baseline or in those receiving CPI combinations. However, irAEs tended to be low grade, manageable with corticosteroids and <20% required treatment discontinuation. Prospective efforts to elucidate the interplay between AD and enhanced risk of CPI toxicity are underway such as studies evaluating nivolumab in patients with pre-existing AD (NCT03656627, NCT03816345).
In the absence of consensus guidelines for the relatively large population with pre-existing AD and as we await prospective results, retrospective real-world evidence can provide reassurance that CPI generally can be administered safely and that patients with well-controlled AD should not be denied the potential significant clinical benefit. Further study is needed to confirm these findings and extend the experience to more symptomatic or severe cases or disorders not captured in our study that may be more clinically risky. Until then, we recommend carefully weighing the risk/benefit ratio with the patient, designing a thoughtful multidisciplinary monitoring strategy, and developing a proactive treatment plan in concert with the AD subspecialist in anticipation of exacerbations.
Limitations of our study included the retrospective nature with potential selection bias of patients with generally well-controlled, non-life-threatening AD. However, inclusion of a heterogeneous population with different types of immunotherapy and lines of treatment also enhances the generalizability of our results. Patients with more severe and rare types of AD were under-represented. Most AD patients at CPI initiation were asymptomatic or mildly symptomatic, with good performance status and not on immunosuppression. Nevertheless, the population included is representative of that commonly seen in clinical practice, and patients with severe AD treated with multiple biological therapies or who have life-threatening diseases would need a highly personalized multidisciplinary approach that is unlikely to be captured in even in the ongoing prospective studies. Despite this being the largest series yet reported, relatively short follow-up and small numbers limited the assessment of delayed AEs as well as safety and efficacy by prognostic risk and treatment subgroups. The study lacked central radiographic review, which may have impacted response assessment. TTF was employed rather than progression free survival as a metric that better reflects real-world practice given that treatment discontinuation generally encompasses toxicity, tolerability and subjective physician judgment of clinical benefit in addition to progressive disease.
Conclusions
Patients with RCC and UC with well-controlled AD can benefit from CPI and experience manageable toxicities that are consistent with what is expected of a non-AD population. Research collaborations and support of prospective clinical trials including more severe AD types are warranted to evaluate clinical outcomes and the risk–benefit profile in this understudied population. In the absence of available prospective data, our real-world evidence study supports the cautious use of CPI across the AD spectrum with close monitoring and proactive multidisciplinary care in concert with the AD specialist.
Acknowledgments
The authors thank the patients and the study staff at each center for their contributions to research.
Footnotes
Twitter: @AbhiTrip87, @AminNassarMD, @PierVitaleNuzzo, @DrChoueiri
Presented at: Presented at the Annual ASCO Meeting 2019 in Chicago, IL.
Contributors: Conception and design: NM-C and LH. Acquisition of data: NM-C, MI, HD, AT, BB, EL, YZ, RM, SS, AM, MH, SS, MDK, SAS, AHN, PVN and AH. Analysis and interpretation of data: NM-C, LH and TC. Drafting of the manuscript: NM-C, WX and LH. Critical revision of the manuscript for important intellectual content: MI, HD, AT, BB, EL, YZ, RM, SS, AM, MH, SS, MDK, SAS, AHN, PVN, AH and TC. Statistical analysis: WX. Supervision: LH.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: NM-C reports support for research travel from Pfizer and Ipsen, and consulting fees for BMS and Bayer. AT reports advisory fee from Foundation medicine and research funding to institution from EMD Serono, Aravive, Bayer, Corvus, WindMil Therapeutics. BB received an unrestricted research grant from Bristol-Myers Squib and speakers' fee from Bristol-Myers Squib, Pfizer, Ipsen and Merck. EL reports research clinical trial funding from Bristol-Myers Squib, Exelixis, Pfizer, Merck, Calithera, Peloton, Genentech, Roche; and consultant fees from Calithera. YZ reports advisory board: Amgen, Roche Diagnostics, Novartis, Jansen, Eisai, Exelixis, Castle Bioscience, Array, Bayer and Pfizer. RM reports consulting fees for Bristol-Myers Squib, Dendreon, Exelixis, Jannsen, Novartis, Pfizer, Tempus; RM receives research funding from Pfizer and Bayer. SS reports consulting fees from Jannsen, and receives research funding from Genentech. AM reports advisory board fees from Seattle Genetics and Debiopharm Group and institution research funding from Acerta Pharma, Genentech, Roche, Merck, Novartis, Seattle Genetics, Acerta Pharma, Mirati Therapeutics and Bristol-Myers Squibb. MH reports consulting fees from AstraZeneca, Bayer, BMS, Exelixis FujiFilm, Genentech and Pfizer; speaking fees from Exelixis; and research funding from BMS, Clovis, Exelixis, Genentech, Merck and Pfizer. TC reports honoraria from AstraZeneca, Alexion, Sanofi/Aventis, Bayer, BMS, Cerulean, Eisai, Foundation Medicine, Heron Therapeutics, Exelixis, Genentech, Roche, Lilly, GlaxoSmithKline, Merck, Novartis, Peloton, Pfizer, EMD Serono, Prometheus Labs, Corvus, Ipsen, Up-to-Date, NCCN, Analysis Group, NCCN, Michael J. Hennessy (MJH) Associates (Healthcare Communications Company with several brands such as OnClive and PER), L-path, Kidney Cancer Journal, Clinical Care Options, Platform Q, Navinata Healthcare, Harborside Press, American Society of Medical Oncology, NEJM, Lancet Oncology; Consulting fees from AstraZeneca, Alexion, Sanofi/Aventis, Bayer, BMS, Cerulean, Eisai, Foundation Medicine, Exelixis, Heron therapeutics, Genentech, Roche, GlaxoSmithKline, Lilly, Merck, Novartis, Peloton, Pfizer, EMD Serono, Prometheus Labs, Corvus, Ipsen, Up-to-Date, NCCN, Analysis Group; support for research travel from Part of Consulting, Advisory role and Honoraria; research funding (Institutional and personal) from AstraZeneca, Bayer, BMS, Cerulean, Eisai, Foundation Medicine, Exelixis, Ipsen, Tracon, Genentech, Roche, Roche Products Limited, GlaxoSmithKline, Lilly, Merck, Novartis, Peloton, Pfizer, Prometheus Labs, Corvus, Calithera, Analysis Group, Takeda. LH reports consulting fees from Genentech, Dendreon, Pfizer, Medivation/Astellas, Exelixis, Bayer, Kew Group, Corvus, Merck, Novartis, Michael J Hennessy Associates (Healthcare Communications Company and several brands such as OncLive and PER), Jounce, EMD Serono, Ology Medical Education; research funding from Bayer, Sotio, Bristol-Myers Squib, Merck, Takeda, Dendreon/Valient, Jannsen, Medivation/Astellas, Genentech, Pfizer, Endocyte (Novartis), and support for research travel from Bayer and Genentech.
Patient consent for publication: Not required.
Ethics approval: This study was approved by the Institutional Review Board at the coordinating center Dana-Farber Cancer Institute and each participating center. Patient consent was not required given retrospective nature and use of deidentified data.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: Data are available upon reasonable request. All data relevant to the study are included in the article or uploaded as supplementary information. All data and materials relevant to this article are available to referees at submission and to readers promptly upon request.
References
- 1.Ribas A, Wolchok JD. Cancer immunotherapy using checkpoint blockade. Science 2018;359:1350–5. 10.1126/science.aar4060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Seidel JA, Otsuka A, Kabashima K. Anti-PD-1 and anti-CTLA-4 therapies in cancer: mechanisms of action, efficacy, and limitations. Front Oncol 2018;8:86 10.3389/fonc.2018.00086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Postow MA, Sidlow R, Hellmann MD. Immune-related adverse events associated with immune checkpoint blockade. N Engl J Med 2018;378:158–68. 10.1056/NEJMra1703481 [DOI] [PubMed] [Google Scholar]
- 4.Wang Y, Zhou S, Yang F, et al. Treatment-related adverse events of PD-1 and PD-L1 inhibitors in clinical trials: a systematic review and meta-analysis. JAMA Oncol 2019;5:1008–19. 10.1001/jamaoncol.2019.0393 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.AARDA American autoimmune related diseases association website. Available: https://www.aarda.org/news-information/statistics/ [Accessed 6 Apr 2019].
- 6.Franks AL, Slansky JE. Multiple associations between a broad spectrum of autoimmune diseases, chronic inflammatory diseases and cancer. Anticancer Res 2012;32:1119–36. [PMC free article] [PubMed] [Google Scholar]
- 7.El-Refai SM, Brown JD, Black EP, et al. Immune checkpoint inhibition and the prevalence of autoimmune disorders among patients with lung and renal cancer. Cancer Inform 2017;16:1176935117712520 10.1177/1176935117712520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Johnson DB, Sullivan RJ, Ott PA, et al. Ipilimumab therapy in patients with advanced melanoma and preexisting autoimmune disorders. JAMA Oncol 2016;2:234–40. 10.1001/jamaoncol.2015.4368 [DOI] [PubMed] [Google Scholar]
- 9.Menzies AM, Johnson DB, Ramanujam S, et al. Anti-PD-1 therapy in patients with advanced melanoma and preexisting autoimmune disorders or major toxicity with ipilimumab. Ann Oncol 2017;28:368–76. 10.1093/annonc/mdw443 [DOI] [PubMed] [Google Scholar]
- 10.Gutzmer R, Koop A, Meier F, et al. Programmed cell death protein-1 (PD-1) inhibitor therapy in patients with advanced melanoma and preexisting autoimmunity or ipilimumab-triggered autoimmunity. Eur J Cancer 2017;75:24–32. 10.1016/j.ejca.2016.12.038 [DOI] [PubMed] [Google Scholar]
- 11.Kähler KC, Eigentler TK, Gesierich A, et al. Ipilimumab in metastatic melanoma patients with pre-existing autoimmune disorders. Cancer Immunol Immunother 2018;67:825–34. 10.1007/s00262-018-2134-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Leonardi GC, Gainor JF, Altan M, et al. Safety of programmed death-1 pathway inhibitors among patients with non-small-cell lung cancer and preexisting autoimmune disorders. J Clin Oncol 2018;36:1905–12. 10.1200/JCO.2017.77.0305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Danlos F-X, Voisin A-L, Dyevre V, et al. Safety and efficacy of anti-programmed death 1 antibodies in patients with cancer and pre-existing autoimmune or inflammatory disease. Eur J Cancer 2018;91:21–9. 10.1016/j.ejca.2017.12.008 [DOI] [PubMed] [Google Scholar]
- 14.Cortellini A, Buti S, Santini D, et al. Clinical outcomes of patients with advanced cancer and pre-existing autoimmune diseases treated with Anti-Programmed death-1 immunotherapy: a real-world transverse study. Oncologist 2019;24:e327–37. 10.1634/theoncologist.2018-0618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nabel CS, Severgnini M, Hung YP, et al. Anti-Pd-1 Immunotherapy-Induced flare of a known underlying relapsing vasculitis mimicking recurrent cancer. Oncologist 2019;24:1013–21. 10.1634/theoncologist.2018-0633 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 2009;45:228–47. 10.1016/j.ejca.2008.10.026 [DOI] [PubMed] [Google Scholar]
- 17.Heng DYC, Xie W, Regan MM, et al. Prognostic factors for overall survival in patients with metastatic renal cell carcinoma treated with vascular endothelial growth factor-targeted agents: results from a large, multicenter study. J Clin Oncol 2009;27:5794–9. 10.1200/JCO.2008.21.4809 [DOI] [PubMed] [Google Scholar]
- 18.Bellmunt J, Choueiri TK, Fougeray R, et al. Prognostic factors in patients with advanced transitional cell carcinoma of the urothelial tract experiencing treatment failure with platinum-containing regimens. J Clin Oncol 2010;28:1850–5. 10.1200/JCO.2009.25.4599 [DOI] [PubMed] [Google Scholar]
- 19.Bajorin DF, Dodd PM, Mazumdar M, et al. Long-term survival in metastatic transitional-cell carcinoma and prognostic factors predicting outcome of therapy. J Clin Oncol 1999;17:3173–81. 10.1200/JCO.1999.17.10.3173 [DOI] [PubMed] [Google Scholar]
- 20.Motzer RJ, Escudier B, McDermott DF, et al. Nivolumab versus everolimus in advanced renal-cell carcinoma. N Engl J Med 2015;373:1803–13. 10.1056/NEJMoa1510665 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bellmunt J, de Wit R, Vaughn DJ, et al. Pembrolizumab as second-line therapy for advanced urothelial carcinoma. N Engl J Med 2017;376:1015–26. 10.1056/NEJMoa1613683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Motzer RJ, Tannir NM, McDermott DF, et al. Nivolumab plus ipilimumab versus sunitinib in advanced renal-cell carcinoma. N Engl J Med 2018;378:1277–90. 10.1056/NEJMoa1712126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rini BI, Plimack ER, Stus V, et al. Pembrolizumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med 2019;380:1116–27. 10.1056/NEJMoa1816714 [DOI] [PubMed] [Google Scholar]
- 24.Powles T, Durán I, van der Heijden MS, et al. Atezolizumab versus chemotherapy in patients with platinum-treated locally advanced or metastatic urothelial carcinoma (IMvigor211): a multicentre, open-label, phase 3 randomised controlled trial. Lancet 2018;391:748–57. 10.1016/S0140-6736(17)33297-X [DOI] [PubMed] [Google Scholar]
- 25.Martinez Chanza N, Xie W, Kaymakcalan MD, et al. Safety and efficacy of immune checkpoint inhibitors (CPI) in metastatic renal cell cancer (RCC) and urothelial cancer (UC) patients (pts) with pre-existing autoimmune disorders (AD). J Clin Oncol 2019;37:653 10.1200/JCO.2019.37.7_suppl.653 [DOI] [Google Scholar]
- 26.Maher VE, Fernandes LL, Weinstock C, et al. Analysis of the association between adverse events and outcome in patients receiving a programmed death protein 1 or programmed death ligand 1 antibody. J Clin Oncol 2019;37:2730–7. 10.1200/JCO.19.00318 [DOI] [PubMed] [Google Scholar]
- 27.Horvat TZ, Adel NG, Dang T-O, et al. Immune-Related adverse events, need for systemic immunosuppression, and effects on survival and time to treatment failure in patients with melanoma treated with ipilimumab at Memorial Sloan Kettering cancer center. J Clin Oncol 2015;33:3193–8. 10.1200/JCO.2015.60.8448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Weber JS, Hodi FS, Wolchok JD, et al. Safety profile of nivolumab monotherapy: a pooled analysis of patients with advanced melanoma. J Clin Oncol 2017;35:785–92. 10.1200/JCO.2015.66.1389 [DOI] [PubMed] [Google Scholar]
- 29.Sternberg CN, Loriot Y, James N, et al. Primary results from SAUL, a multinational single-arm safety study of Atezolizumab therapy for locally advanced or metastatic urothelial or Nonurothelial carcinoma of the urinary tract. Eur Urol 2019;76:73–81. 10.1016/j.eururo.2019.03.015 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
jitc-2020-000538supp001.pdf (1MB, pdf)