Polymyxin resistance mediated by the mcr-1 gene threatens the last-resort antibiotics. Linear lipopeptide paenipeptin analogues 1 and 15 disrupted the outer membrane of Gram-negative pathogens and potentiated clarithromycin and rifampin against mcr-1-positive Escherichia coli from the FDA-CDC Antimicrobial Resistance Isolate Bank. In the presence of paenipeptin, clarithromycin and rifampin resulted in over 3-log reduction of E. coli in vitro.
KEYWORDS: lipopeptide, paenipeptin, mcr-1, potentiator, combination therapy
ABSTRACT
Polymyxin resistance mediated by the mcr-1 gene threatens the last-resort antibiotics. Linear lipopeptide paenipeptin analogues 1 and 15 disrupted the outer membrane of Gram-negative pathogens and potentiated clarithromycin and rifampin against mcr-1-positive Escherichia coli from the FDA-CDC Antimicrobial Resistance Isolate Bank. In the presence of paenipeptin, clarithromycin and rifampin resulted in over 3-log reduction of E. coli in vitro. Moreover, paenipeptin-antibiotic combinations significantly reduced E. coli in a murine thigh infection model.
INTRODUCTION
Drug-resistant Gram-negative pathogens, including carbapenem-resistant Enterobacteriaceae (CRE), have been widely recognized as a serious threat to public health (1, 2). The cyclic cationic lipopeptides, polymyxin and colistin (polymyxin E), are considered the last-resort therapy for the treatment of infections caused by multidrug-resistant Gram-negative pathogens. However, plasmid-mediated, polymyxin-resistant Escherichia coli harboring the mcr-1 gene has been isolated from farm animals and patients (3, 4), which may compromise the therapeutic efficacy of polymyxins. Therefore, rapidly evolving antibiotic resistance requires a matching effort to develop new and effective antimicrobial agents.
Paenipeptin analogues 1 and 15 are novel synthetic linear lipopeptides which were designed through extensive structure-activity relationship (SAR) studies (5). These two compounds share the same peptide structure (Dab-Ile-Dab-dPhe-Leu-Dab-dVal-Leu-Ser, where Dab represents the positively charged 2,4-diaminobutyric acid) but differ in their N-terminal modifications. Analogue 1 possesses an N-terminal C6 lipid chain, whereas the N terminus of analogue 15 is modified by a hydrophobic carboxybenzyl group. Analogues 1 and 15 are nonhemolytic and showed low cytotoxicity against a human kidney cell line, HEK 293 (6). We previously have shown that these two compounds potentiated clarithromycin and rifampin against carbapenem-resistant Klebsiella pneumoniae and Acinetobacter baumannii in vitro (6). The objective of this study was to evaluate the efficacy of the combination between paenipeptins and clarithromycin/rifampin against mcr-1 mediated, polymyxin-resistant E. coli in vitro and in vivo using a murine thigh infection model.
Paenipeptin analogues 1 and 15 (>95% purity) are synthetic lipopeptides produced by a commercial peptide synthesis company (GenScript Inc., Piscataway, NJ). Clarithromycin, rifampin, and polymyxin B were purchased from Sigma (St. Louis, MO). Polymyxin-resistant E. coli strains harboring the mcr-1 gene were obtained from the FDA-CDC Antimicrobial Resistance Isolate Bank (Table 1). The MIC and minimum bactericidal concentration (MBC) values against E. coli isolates were determined as described previously (5). Polymyxin B MIC values for four mcr-1-positive-E. coli isolates ranged from 4 to 16 μg/ml, which confirmed the polymyxin resistance phenotype. When treated alone by a single antimicrobial agent, these clinical isolates were resistant to clarithromycin and rifampin (MIC, 8 to 32 μg/ml; Table 1), which indicated that clarithromycin and rifampin were excluded by the intact outer membrane permeability barrier in E. coli cells. In the presence of paenipeptin analogue 1 or 15 at subinhibitory concentration (4 μg/ml), the MIC of clarithromycin and rifampin was reduced to 0.0019 to 0.125 μg/ml, whereas the MBC ranged from 0.0019 to 0.25 μg/ml (Table 2). Similarly, other researchers reported that a polymyxin derivative, NAB739, potentiated rifampin and other antibiotics against mcr-positive, polymyxin-resistant strains (7).
TABLE 1.
MIC values of polymyxin B, clarithromycin, rifampin, and paenipeptin analogues 1 and 15 against polymyxin-resistant pathogens from the FDA-CDC Antimicrobial Resistance Isolate Bank
| E. coli strain | MIC (μg/ml) values for:a |
||||
|---|---|---|---|---|---|
| Polymyxin B | Clarithromycin | Rifampin | Analogue 1 | Analogue 15 | |
| FDA-CDC AR 0350 | 4–8 | >32 | 16 | 16–32 | 16 |
| FDA-CDC AR 0493 | 8–16 | >32 | 16 | 32 | 16 |
| FDA-CDC AR 0494 | 8 | ≥32 | 16–32 | 16–32 | 8–16 |
| FDA–CDC AR 0495 | 4–16 | >32 | 8–16 | 16–32 | 16 |
| ATCC 25922 | ≤0.5 | >32 | 8–16 | 8 | 8–16 |
The MIC represents the range of individual concentrations measured from at least three independent experiments.
TABLE 2.
MIC (μg/ml) and MBC (μg/ml) values for clarithromycin and rifampin in the presence of paenipeptin analogue 1 or analogue 15 at 4 μg/ml against polymyxin-resistant isolates from the FDA-CDC Antimicrobial Resistance Isolate Banka
| E. coli strain | Data (μg/ml) for the indicated strain in the presence of: |
|||||||
|---|---|---|---|---|---|---|---|---|
| Analogue 1 |
Analogue 15 |
|||||||
| Clarithromycin |
Rifampin |
Clarithromycin |
Rifampin |
|||||
| MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | |
| FDA-CDC AR 0350 | 0.0313–0.0625 | 0.125 | ≤0.0019 | 0.0078 | 0.0313–0.0625 | 0.0313 | ≤0.0019 | 0.0156 |
| FDA-CDC AR 0493 | 0.0625 | 0.125 | ≤0.0019 | 0.0019 | 0.0625 | 0.0625 | ≤0.0019 | 0.0019 |
| FDA-CDC AR 0494 | 0.0313 | 0.0625 | ≤0.0019 | 0.0039 | 0.0156–0.0313 | 0.0313 | ≤0.0019 | 0.0019 |
| FDA-CDC AR 0495 | 0.0625–0.125 | 0.125 | ≤0.0019 | 0.0019 | 0.125 | 0.25 | ≤0.0019 | 0.0019 |
| ATCC 25922 | 0.0313 | 0.0625 | ≤0.0019 | 0.0019 | 0.0078 | 0.0625 | ≤0.0019 | 0.0019 |
The MIC represents the range of individual concentrations measured from at least three independent experiments.
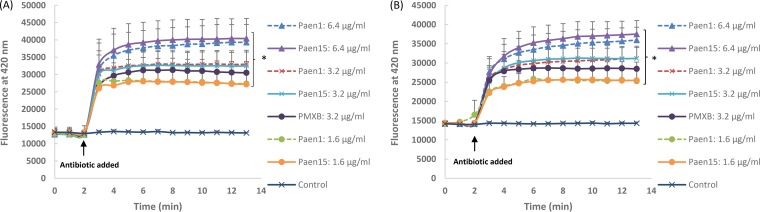
The enhanced antimicrobial activity in the presence of paenipeptin analogues is presumably due to the compromised outer membrane induced by the cationic paenipeptin molecules. To test the mechanism of potentiation, we determined the change of outer membrane permeabilization in the presence of paenipeptin analogue using a fluorescent probe, 1-N-phenylnaphthylamine (NPN) (8, 9). The fluorescence of NPN was measured using a cell imaging multimode reader (Cytation 3; BioTek, Winooski, VT) with an excitation at 350 nm and emission at 420 nm. Polymyxin B, a known outer membrane active compound (10), was used as a positive control. Paenipeptin analogues 1 and 15, at a final concentration of 1.6 to 6.4 μg/ml, significantly increased the uptake of the NPN probe in a concentration-dependent manner, suggesting disruption of the outer membrane in E. coli ATCC 25922 (Fig. 1A) and in the mcr-1-positive strain, E. coli FDA-CDC AR 0494 (Fig. 1B). Paenipeptin treatment may promote the uptake of clarithromycin and rifampin by disrupting the outer membrane of Gram-negative pathogens.
FIG 1.
Time course of increase in 1-N-phenylnaphthylamine (NPN) fluorescence intensity in the presence of intact Escherichia coli cells at different concentrations of paenipeptin analogues 1 and 15. (A) E. coli ATCC 25922; (B) E. coli FDA-CDC AR 0494. Paen1, analogue 1; Paen15, analogue 15; PMXB, polymyxin B. The final antibiotic concentrations after mixing with NPN and cells were reported. Values are expressed as means (three independent repeats), and error bars represent standard deviations. An asterisk indicates statistical significance (P < 0.05) at the final data points between the control and antimicrobial treatment groups.
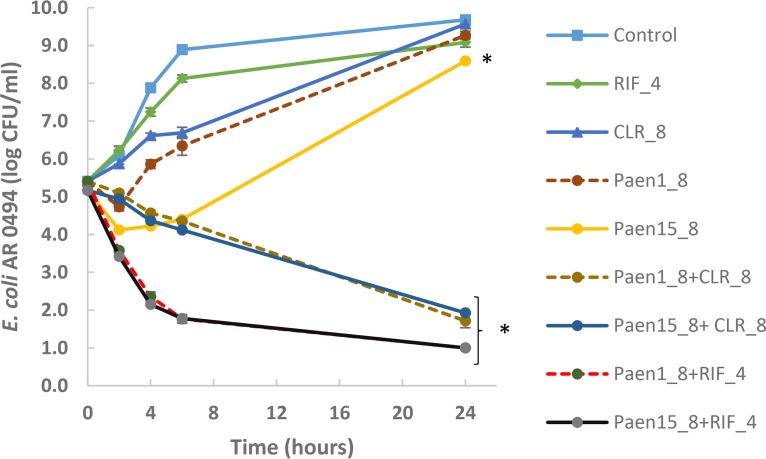
Time-kill kinetics were used to study the bactericidal effect of the combined treatments against a selected polymyxin-resistant strain, E. coli FDA-CDC AR 0494, in a microbiological medium, tryptic soy broth. A single treatment by paenipeptin analogue 1 (8 μg/ml), analogue 15 (8 μg/ml), clarithromycin (8 μg/ml), or rifampin (4 μg/ml) alone reduced the growth rate of E. coli by 6 h but failed to inhibit pathogen growth for 24 h. At the same tested concentrations, paenipeptin analogue 1, in combination with clarithromycin or rifampin, resulted in 3.7- and 4.4-log reduction within 24 h, respectively (Fig. 2). Analogue 15 in the presence of clarithromycin or rifampin exhibited very similar inactivation kinetics. These results are consistent with our previous report in which paenipeptin analogues potentiated antibiotics against carbapenem-resistant K. pneumoniae and A. baumannii in vitro (6). Because of the high similarity in time-kill kinetics for both analogues, we selected analogue 1 for further acute toxicity and therapeutic efficacy studies in mice.
FIG 2.
Time-kill curves of Escherichia coli FDA-CDC AR 0494 with exposure to paenipeptin analogues alone or in combination with clarithromycin/rifampin in tryptic soy broth. RIF_4, 4 μg/ml rifampin; CLR_8, 8 μg/ml clarithromycin; Paen1_8, 8 μg/ml analogue 1; Paen15_8, 8 μg/ml analogue 15; Paen1_8+CLR_8, 8 μg/ml analogue 1 plus 8 μg/ml clarithromycin; Paen15_8+CLR_8, 8 μg/ml analogue 15 plus 8 μg/ml clarithromycin; Paen1_8+RIF_4, 8 μg/ml analogue 1 plus 4 μg/ml rifampin; Paen15_8+RIF_4, 8 μg/ml analogue 15 plus 4 μg/ml rifampin. Values are expressed as means (four independent repeats), and error bars represent standard deviations. An asterisk indicates statistical significance (P < 0.05) at the final data points between the control and antimicrobial treatment groups.
The animal experiments have been approved by the Institutional Animal Care and Use Committee (IACUC) at University of Arkansas for Medical Sciences (approval numbers 3760 and 3922). An acute toxicity study was conducted to evaluate the tolerance of paenipeptin analogue 1 in female CD-1 mice (n = 4; 6 weeks old). Briefly, mice were treated by subcutaneous injection of a single dose of paenipeptin analogue 1 at 40, 60, 80, 100, or 120 mg/kg. The treated mice were monitored for 10 days, and the survival rate was recorded. All mice in the 40- to 80-mg/kg groups showed normal eating and drinking activity and survived the whole 10-day period. However, analogue 1 at 100 and 120 mg/kg resulted in one and two deaths out of four animals, respectively. Therefore, analogue 1 was tested at 40 and 60 mg/kg for its therapeutic activity in mice.
A neutropenic murine thigh infection model was used to evaluate the therapeutic efficacy of the combined treatments against E. coli FDA-CDC AR 0494 in vivo. Female CD-1 mice (6 weeks old) were rendered neutropenic by intraperitoneal injection of cyclophosphamide on 4 days (150 mg/kg) and 1 day (100 mg/kg) prior to bacterial inoculation. On the day of inoculation, E. coli cells (3.4 × 105 CFU in 100 μl saline) were intramuscularly injected into each posterior thigh. At 2 and 5 h postinfection, mice were treated with single or combined antibiotics in phosphate-buffered saline (PBS) by subcutaneous injection. At 8 h postinfection, mice were euthanized by CO2, and tissues from each thigh were collected and homogenized for bacterial count determination. Mice showed normal eating and drinking activity in all treatment groups.
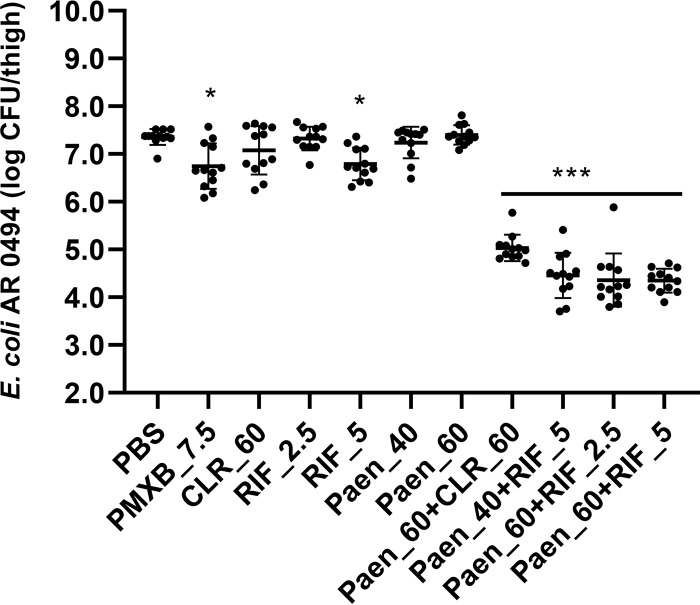
As showed in Fig. 3, polymyxin B at 7.5 mg/kg and rifampin at 5 mg/kg reduced 0.6 log of the polymyxin-resistant E. coli strain. All other single treatments, including clarithromycin (60 mg/kg), rifampin (2.5 mg/kg), and paenipeptin analogue 1 (40 and 60 mg/kg), didn’t significantly inhibit the growth of the bacterium in thigh tissues. In contrast, all combined treatments significantly reduced bacterial counts in thigh tissues. For example, paenipeptin analogue 1 at 60 mg/kg, in combination with clarithromycin at 60 mg/kg, showed 2.3-log reduction compared with the PBS control. Moreover, paenipeptin-rifampin combinations displayed 2.9- to 3.0-log reductions in comparison with the PBS control group (Fig. 3). Therefore, paenipeptin analogue 1 potentiated clarithromycin and rifampin in vivo against polymyxin-resistant E. coli. Combination therapy has been proved as a promising strategy to overcome antibiotic resistance. Colistin and clarithromycin combination therapy resulted in 2.9-log reduction of mcr-1-positive K. pneumoniae in a mouse thigh infection model (11). Similarly, combination therapy of colistin plus rifampin or minocycline was efficacious against NDM- and MCR-1 coproducing E. coli in a murine thigh infection model (12).
FIG 3.
Paenipeptin analogue 1 in combination with clarithromycin or rifampin was efficacious against Escherichia coli FDA-CDC AR 0494 in a murine thigh infection model. Treatment groups (antimicrobial injection time, hours postinfection): PBS, phosphate-buffered saline; PMXB_7.5, 7.5 mg/kg polymyxin B (2 h); CLR_60, 60 mg/kg clarithromycin (2 and 5 h); RIF_2.5, 2.5 mg/kg rifampin (2 and 5 h); RIF_5, 5 mg/kg rifampin (2 and 5 h); Paen_40: 40 mg/kg analogue 1 (2 h); Paen_60, 60 mg/kg analogue 1 (2 h); Paen_60+CLR_60, 60 mg/kg analogue 1 (2 h) plus 60 mg/kg clarithromycin (2 h and 5 h); Paen_40+RIF_5, 40 mg/kg analogue 1 (2 h) plus 5 mg/kg rifampin (2 h and 5 h); Paen_60+RIF_2.5, 60 mg/kg analogue 1 (2 h) plus 2.5 mg/kg rifampin (2 h and 5 h); Paen_60+RIF_5, 60 mg/kg analogue 1 (2 h) plus 5 mg/kg rifampin (2 h and 5 h). Statistical significance between the antimicrobial treatment groups and the PBS control group is indicated by an asterisk(s) (*, P < 0.05; ***, P < 0.001).
In conclusion, cationic paenipeptin analogues 1 and 15 disrupted the outer membrane of Gram-negative bacteria and thus may promote the entry of hydrophobic antibiotics into bacterial cells. Paenipeptin analogue 1 potentiated clarithromycin and rifampin against mcr-1-positive E. coli in vitro and in vivo. This is the first report on the combination effect between potentiator paenipeptin and other antibiotics in vivo. Pharmacokinetics and pharmacodynamics studies of paenipeptin analogues will be needed in the future to develop optimal treatment strategies and to prevent the emergence of resistance (13).
ACKNOWLEDGMENTS
This research project was supported by National Institutes of Health (NIH)/National Institute of Allergy and Infectious Diseases (NIAID) award R21AI146693. This work was supported by the Center for Microbial Pathogenesis and Host Inflammatory Responses through the Center of Biomedical Research Excellence Award P20GM103625 from National Institute of General Medical Sciences (NIH/NIGMS). This project was also supported in part by the Arkansas Biosciences Institute, the major research component of the Arkansas Tobacco Settlement Proceeds Act of 2000.
REFERENCES
- 1.Boucher HW, Talbot GH, Bradley JS, Edwards JE, Gilbert D, Rice LB, Scheld M, Spellberg B, Bartlett J. 2009. Bad bugs, no drugs: no ESKAPE! An update from the Infectious Diseases Society of America. Clin Infect Dis 48:1–12. doi: 10.1086/595011. [DOI] [PubMed] [Google Scholar]
- 2.Bonomo RA. 2011. New Delhi metallo-β-lactamase and multidrug resistance: a global SOS? Clin Infect Dis 52:485–487. doi: 10.1093/cid/ciq179. [DOI] [PubMed] [Google Scholar]
- 3.Liu Y-Y, Wang Y, Walsh TR, Yi L-X, Zhang R, Spencer J, Doi Y, Tian G, Dong B, Huang X, Yu L-F, Gu D, Ren H, Chen X, Lv L, He D, Zhou H, Liang Z, Liu J-H, Shen J. 2016. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect Dis 16:161–168. doi: 10.1016/S1473-3099(15)00424-7. [DOI] [PubMed] [Google Scholar]
- 4.Macesic N, Khan S, Giddins MJ, Freedberg DE, Whittier S, Green DA, Furuya EY, Verna EC, Annavajhala MK, Gomez-Simmonds A, Uhlemann AC. 2019. Escherichia coli harboring mcr-1 in a cluster of liver transplant recipients: detection through active surveillance and whole-genome sequencing. Antimicrob Agents Chemother 63:e02680-18. doi: 10.1128/AAC.02680-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Moon SH, Zhang X, Zheng G, Meeker DG, Smeltzer MS, Huang E. 2017. Novel linear lipopeptide paenipeptins with potential for eradicating biofilms and sensitizing Gram-negative bacteria to rifampicin and clarithromycin. J Med Chem 60:9630–9640. doi: 10.1021/acs.jmedchem.7b01064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Moon SH, Huang E. 2018. Lipopeptide paenipeptin analogues potentiate clarithromycin and rifampicin against carbapenem-resistant pathogens. Antimicrob Agents Chemother 62:e00329-18. doi: 10.1128/AAC.00329-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tyrrell JM, Aboklaish AF, Walsh TR, Vaara T, Vaara M. 2019. The polymyxin derivative NAB739 is synergistic with several antibiotics against polymyxin-resistant strains of Escherichia coli, Klebsiella pneumoniae and Acinetobacter baumannii. Peptides 112:149–153. doi: 10.1016/j.peptides.2018.12.006. [DOI] [PubMed] [Google Scholar]
- 8.Loh B, Grant C, Hancock RE. 1984. Use of the fluorescent probe 1-N-phenylnaphthylamine to study the interactions of aminoglycoside antibiotics with the outer membrane of Pseudomonas aeruginosa. Antimicrob Agents Chemother 26:546–551. doi: 10.1128/aac.26.4.546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jangra M, Kaur M, Tambat R, Rana R, Maurya SK, Khatri N, Ghafur A, Nandanwar H. 2019. Tridecaptin M, a new variant discovered in mud bacterium, shows activity against colistin-and extremely drug-resistant Enterobacteriaceae. Antimicrob Agents Chemother 63:e00338-19. doi: 10.1128/AAC.00338-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vaara M. 1992. Agents that increase the permeability of the outer membrane. Microbiol Rev 56:395–411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.MacNair CR, Stokes JM, Carfrae LA, Fiebig-Comyn AA, Coombes BK, Mulvey MR, Brown ED. 2018. Overcoming mcr-1 mediated colistin resistance with colistin in combination with other antibiotics. Nat Commun 9:458. doi: 10.1038/s41467-018-02875-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yu Y, Walsh TR, Yang RS, Zheng M, Wei MC, Tyrrell JM, Wang Y, Liao XP, Sun J, Liu YH. 2019. Novel partners with colistin to increase its in vivo therapeutic effectiveness and prevent the occurrence of colistin resistance in NDM- and MCR-co-producing Escherichia coli in a murine infection model. J Antimicrob Chemother 74:87–95. doi: 10.1093/jac/dky413. [DOI] [PubMed] [Google Scholar]
- 13.Levison ME, Levison JH. 2009. Pharmacokinetics and pharmacodynamics of antibacterial agents. Infect Dis Clin North Am 23:791–815. doi: 10.1016/j.idc.2009.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]