Summary
The symptoms associated with COVID-19 are mainly characterized by a triad composed of fever, dry cough and dyspnea. However, digestive symptoms have also been reported. At first considered as infrequent, they in fact seem to affect more than half of patients. The symptoms mainly include anorexia, diarrhea, nausea and/or vomiting and abdominal pain. Even though prognosis is associated with lung injury, digestive symptoms seem significantly more frequent in patients presenting with severe COVID-19 infection. Digestive presentations, which may be isolated or which can precede pulmonary symptoms, have indeed been reported, with diarrhea as a leading clinical sign. The main biological abnormalities that can suggest COVID-19 infection at an early stage are lymphopenia, elevated CRP and heightened ASAT transaminases. Thoraco-abdominal scan seems useful as a means of on the one hand ruling out digestive pathology not connected with coronavirus and on the other hand searching for pulmonary images consistent with COVID-19 infection. No data exist on the value of digestive endoscopy in cases of persistent digestive symptoms. Moreover, the endoscopists may themselves be at significant risk of contamination. Fecal-oral transmission of the infection is possible, especially insofar as viral shedding in stools seems frequent and of longer duration than at the ENT level, including in patients with negative throat swab and without digestive symptoms. In some doubtful cases, virologic assessment of stool samples can yield definitive diagnosis. In the event of prolonged viral shedding in stools, a patient's persistent contagiousness is conceivable but not conclusively established. Upcoming serology should enable identification of the patients having been infected by the COVID-19 epidemic, particularly among previously undetected pauci-symptomatic members of a health care staff. Resumption of medico-surgical activity should be the object of a dedicated strategy preceding deconfinement.
Keywords: COVID-19, Diarrhea, Digestive disorders
Introduction
Coronaviruses are RNA-enveloped viruses transmitted from one human being to another. Among them, three distinct types of coronavirus are responsible for severe lung diseases. SARS-CoV was responsible for an epidemic occurring in Asia from 2002 to 2004 (SARS-CoV-1). MERS-CoV provoked an epidemic in the Middle East in 2012. SARS-CoV-2, which appeared in China and is responsible for the current pandemic, is primarily the virus responsible for the 2019 coronavirus disease, which is why it is known as COVID-19. Over only a few weeks, the medical community has found itself compelled to learn about the semiology and the short-term natural history of the COVID-19 infection This has been done essentially on the basis of the literature from China, initial epicenter of the epidemic. The mortality rate of the infection has been evaluated at 3.6% in China and at 1.5% of the population contaminated outside of China. According to the projections of the World Health Organization, the estimated mortality rate throughout the world will be 5.7% [1]. Up until now, no curative treatment has been validated. Three patient profiles have appeared: pauci-symptomatic infection with initially high viral load; secondary respiratory aggravation on the 10th day notwithstanding reduced viral load suggesting an inadequate host immune response; and rapidly progressive infection with multi-system organ failure and persistence of high viral load [2]. While the general and respiratory signs are known by one and all, the same cannot be said for digestive organ injury. The objective of this paper is to describe COVID-19-related digestive disorders, thereby informing physicians liable to enter into contact with patients presenting these types of symptoms.
COVID-19-related digestive symptoms
The coronavirus spike protein is capable of binding to the receptor of the angiotensin-converting enzyme 2 (ACE2), entering into the infected cell, and interacting with the serine protease TMPRSS2, thereby provoking viral replication in the contaminated tissue. ACE2 plays a role in regulation of inflammatory response and is strongly expressed in the epithelial cells of the enterocytes of the proximal and distal small intestine. This helps to explain the highly elevated incidence of digestive disorders, particularly diarrhea, associated with the COVID-19 infection [3]. In the one reported case of endoscopic biopsy for diarrhea ascribed to COVID-19, note was taken of lymphoplasmocytary infiltration and an edema of the chorion as well as ACE2 and a cytoplasmic viral capsid protein in the stomach, the duodenum and the rectum [4]. As a result, one may hypothesize the ingestion of contaminated food as a possible way of contamination.
In the original series by Huang et al. reporting a series of 41 patients hospitalized in a hospital center in Wuhan, the incidence of digestive disorders seemed anecdotal, limited to diarrhea occurrence in 3% of the patients [5]. Another, large-scale Chinese study (1099 patients) seemed to confirm these findings, reporting incidence of nausea and/or vomiting (5%) and diarrhea (3.8%) [6]. The study also provided interesting epidemiological data including median age (47 years), comorbidities (23.7%), median incubation time (4 days), medical staff affected (3.5% of the cohort), normal pulmonary imagery in non-severe patient (17.9%), utilization of oxygen therapy (41.3%), need for mechanical ventilation (6.1%) and risk factors for severe forms (age and comorbidities).
While possible, a purely digestive form was initially considered as rare. At the outset of the epidemic, there was a reported case of a 22-year-old female patient presenting with isolated febrile diarrhea along with normal blood test and negative fecal culture; on the other hand, chest scan revealed bilateral pneumopathy suggesting COVID-19 [7]. Throat swab confirmed the diagnosis, and during an 18-day hospitalization the diarrhea progressively improved, with no development of extra-digestive signs.
However, recent and consistent data suggest that in point of fact, digestive disorders are more frequent. In a recent retrospective study involving 1141 patients presenting with documented infection, the frequency of initially isolated digestive disorders came to 16%, whereas in 96% of cases, injured lungs appeared on CT-scan [8]. In these patients, the symptoms observed were the following: anorexia (98%), nausea (73%), diarrhea (37%), diffuse abdominal pain (25%), nausea and vomiting (20%), abdominal pain and diarrhea (9%), all of these symptoms (7%). Mortality (3.8%) was exclusively associated with acute respiratory insufficiency.
It bears mentioning that the diarrhea reported in the different studies was invariably fluid, that the severe hydroelectrolytic disorders secondary to the diarrhea were not clearly described, and that no case of mucohemorrhagic diarrhea secondary to viral infection has been reported. Moreover, up until now abdominal pains have not been clearly characterized in the literature, and do not appear specific.
The most recent Chinese epidemiological data report that the incidence of digestive disorders during infection progression is 79%, including (in descending order): anorexia, diarrhea, nausea/vomiting, abdominal pain and bleeding in the digestive tract [9]. In a cohort study of 305 patients from Wuhan, diarrhea with median duration of 4.1 days ± 2.5 days (1 to 14 days) was observed in half of the patients while they were hospitalized. Vomiting seemed more frequent in children than in adults.
The study by Jin et al. compared the evolution of a group of 577 patients without digestive symptoms with that of a group of 74 patients having presented with at least one of the three following digestive symptoms: diarrhea (defined as the passage of more than 3 stools a day) with negative fecal culture, nausea and vomiting[10]. Familial contamination (31% vs. 20%) and a more severe form of the disease (23% vs. 8%) entailing a need for in-hospital resuscitation (6.8% vs. 2.1%) were significantly more frequent in the “digestive symptoms” group. From a clinical standpoint, fever ≥ 38.5°C, asthenia, dyspnea and headaches were significantly more frequent in the “digestive symptoms” group. From a biological standpoint, lymphopenia and elevated ASAT and CRP were likewise significantly more frequent in the “digestive symptoms” group. Productive cough and elevated LDH were the two main predictive factors for evolution toward a severe form of COVID-19 in patients presenting with digestive symptoms.
Schematically, one may distinguish two clinical presentations in an endemic zone. Some patients without any known infection have aspecific, isolated, acute inaugural digestive symptoms suggestive of COVID-19 infection. Other patients have had COVID-19 infection revealed and documented by respiratory signs, and present secondarily with digestive symptoms. In these cases, it is important not to be misled by digestive illness unrelated to COVID-19, especially in the event of persistent fever. Particular attention should be paid to diarrhea so as to avoid overlooking another possible cause (medication, Clostridium Difficile-associated disease….). It also bears mentioning that infected patients tend to have a large number of digestive hemorrhages with usual causes; this may stem from a frequently observed need for effective anticoagulation, which is related to a heightened and documented risk of thromboembolic complications associated with COVID-19.
The main data in the literature are summarized in Table 1 .
Table 1.
The main data in the literature on digestive disorders associated with COVID-19.
| Author | Patients (N) | Patients with digestive disorders (N or %) | Isolated or inaugural digestive disorders | Diarrhea | Diarrhea in case of severe COVID-19 infection | Frequency of diarrhea (N/day) | Nauseas/vomiting | Abdominal pain | Viral excretion in stools |
|---|---|---|---|---|---|---|---|---|---|
| Huang | 41 | 3% | |||||||
| Guan | 1099 | 4% | 16% | 5% | |||||
| Luo | 1141 | 16% | 37% | 73% | 25% | ||||
| Tian | 295 | 79% | 22% | 50% | 3.3 ± 1.6 | ||||
| Jin | 651 | 74 (11%) | 23% vs. 8% (P < 0.001) | ||||||
| Xiao | 73 | 44% | 53% |
Biological abnormalities associated with COVID-19
Viral detection
COVID-19 viremia seems anecdotal. For example, a study published in Nature reports no case of viremia in patients having undergone iterative testing [11]; these findings are in agreement with previously observed data on Middle East respiratory syndrome coronavirus (MERS-CoV) and severe acute syndrome-related coronavirus SARS-CoV, which were responsible for other recent epidemics. As concerns the safety of blood donations in patients having been affected by the disease, these data are reassuring.
Serology seems to be a reliable means of detecting infected patients. A recent study showed that 100% of the infected patients tested presented with positive ELISA Ig G [12]. Even though no validated kit presently exists, this method is clearly likely to emerge in routine practice, particularly as a means of detection in caregivers who may have unknowingly contracted the disease.
Fecal-oral transmission of the corona virus has been a known fact for a number of years. In a study published in 2003, 100% of patients presented with viral excretion in stool specimens and 67% still had a detectable virus 3 weeks after the onset of clinical symptoms [13]. This has also been reported in COVID-19, with a well-documented case of isolated positive RT-PCR results in stools (while in hospital there were also 4 other negative RT-PCR test results, 2 on throat swabs, and the other 2 on sputum) in a patient presenting with non-severe bilateral pneumopathy [14]. Moreover, it appears that viral excretion in stool specimens from coronavirus patients is a frequent occurrence. In a study involving 73 patients, RNA virus was found in the stools of 53% of the population, with viral clearance lasting from 1 to 12 days [4]. However, only 44% of the patients with positive results in stools had diarrhea. It bears mentioning that while stool analysis showed persistent viral RNA, in one quarter of cases the initially positive throat swab became negative. That is one reason why the criterion for healing currently utilized in China (negativization of 2 RT-PCR at an interval of least 24 hours on a throat swab) may soon be reevaluated [15]. On this subject, we do not presently know whether the quantity of residual virus in a patient's stools is associated with persistent contagiousness. For the protection of family members and others residing in the same home, it is nonetheless recommended to proceed to daily cleaning and disinfection (applying ready-to-use concentrated household bleach tablets or an equivalent household disinfectant) of the toilets used by infected patients for as many as fourteen days after disappearance of respiratory symptoms.
Standard biology
A number of biological abnormalities have been consistently reported in several studies and need to be better known insofar as they may draw attention to infection in a patient with scant symptomatology.
In the series by Guan et al. [6], the frequency of the main biological abnormalities was: lymphopenia < 1500/mm3 (median value 1000/mm3): 83%, thrombopenia < 150 000/mm3: 36%, leucopenia < 4000/mm3: 34%, ASAT > 40 UI/l: 22%, ALAT > 40 UI/l: 21%, total bilirubin > 17.1 μmol/l: 11%, CRP ≥ 10 mg/l: 61%, LDH ≥ 250 UI/l: 41%, CPK ≥ 200 UI/l: 14%, D Dimers ≥ 0.5 mg/l: 46%.
In the series by Luo et al. [8] involving 183 patients with initially isolated digestive disorders, the authors observed leucopenia (mean value 2700/mm3), lymphopenia (mean value 530/mm3), elevated CRP (mean value 18.7 ± 6.8 mg/l), minimal hepatic cytolysis (ASAT 65.8 ± 12.7 and ALAT 66.4 ± 13.2) and normal renal function.
Abnormal liver function tests have been reported in most of the studies. In the series by Shi et al., it was suggested that patients presenting with symptoms for less than a week showed less elevated transaminases levels than those with symptoms from 1 to 3 weeks[16]. Moreover, the abnormalities observed in hepatic tests were more pronounced in patients presenting with a severe infection, particularly those hospitalized in intensive care [17]. In the absence of available histological data, the presence of viral RNA in the liver has yet to be demonstrated. One may suppose that liver dysfunction is multifactorial (drug toxicity, inflammatory cascade, hypoxemia). It should nonetheless be noted that notwithstanding the presence of the ACE2 receptor in the cholangiocytes, the patients did not present with intrahepatic cholestasis, which is associated with the virus (absence of elevated Gamma GT and PAL in study patients without preexisting liver disease). As regards intensive care patients, up until now acute liver failure has not been reported as a complicating factor in severe COVID-19 infections. Even though it is not documented in the literature yet, cirrhotic patients may be particularly at risk of severe COVID-19 infection. The AFEF (Société Française d’Hépatologie) recommends sick leave for cirrhotic patients unable to telework and systematic thoracic CT-scan in the event of any decompensated cirrhosis, the objective being to avoid overlooking possible COVID-19 infection.
Digestive imaging abnormalities associated with COVID-19
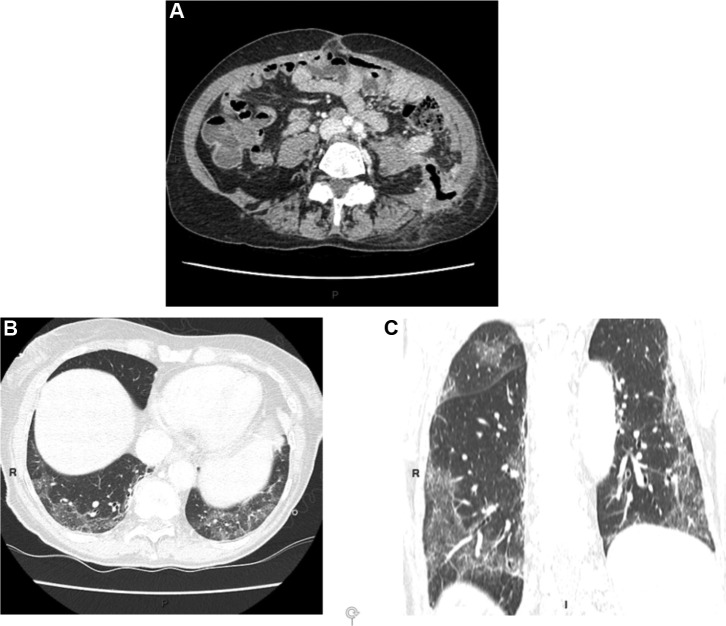
Up until now, no relevant data has been reported in the literature. Even though frequency of occurrence remains unknown, a clinical picture of "ordinary" gastroenteritis can contain scanographic abnormalities such as thickening and/or parietal contrast. In one series, it was estimated that in a systematic analysis of 446 abnormal CT-scans, thickness of the jejunum and/or the ileum was probably infectious in 25% of cases [18]. A recent retrospective analysis of abdominal imagery from patients admitted to Hôpital Saint-Louis in Paris with documented COVID-19 infection did not seem to show parietal abnormalities (personal data). On the other hand, in some patients presenting with digestive symptoms we observed unusual small-intestinal and/or colic fluidic stasis (Figure 1, Figure 2 ). It also bears mentioning that a clinical presentation highlighting digestive disorder may in some cases preclude recognition of a progressive viral infection. For example, a 67-year-old woman monitored in our unit for metastatic colon cancer who had been undergoing chemotherapy was hospitalized for fever and abdominal pain related to streptococcal septicemia that was complicating the evolution of sigmoid fistula with probable loco-regional recurrence. Systematic thoracic slices revealed an aspect typical of COVID-19 infection (Fig. 3 ). Thoracic CT scan fostered suspicion of the infection, which was confirmed by throat swab. During questioning, the patient reported a dry cough (2 days) and anosmia with ageusia (4 days). Notwithstanding rapid apyrexia (48 hours) under antibiotic treatment, her respiratory state quickly deteriorated, leading finally to her death on day 9 of hospitalization. This case illustrates the potential interest of systematic thoracic CT scan in a patient presenting with digestive disorders accompanied by fever.
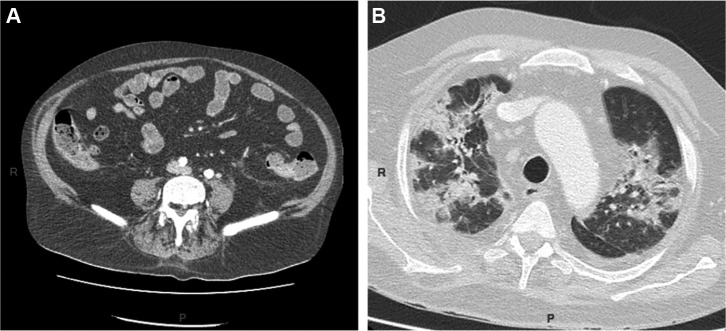
Figure 1.
A: diffuse small intestinal stasis; B: moderate lung injury with ground glass opacification and mixed (sub-pleural and central) condensation associated with COVID-19.
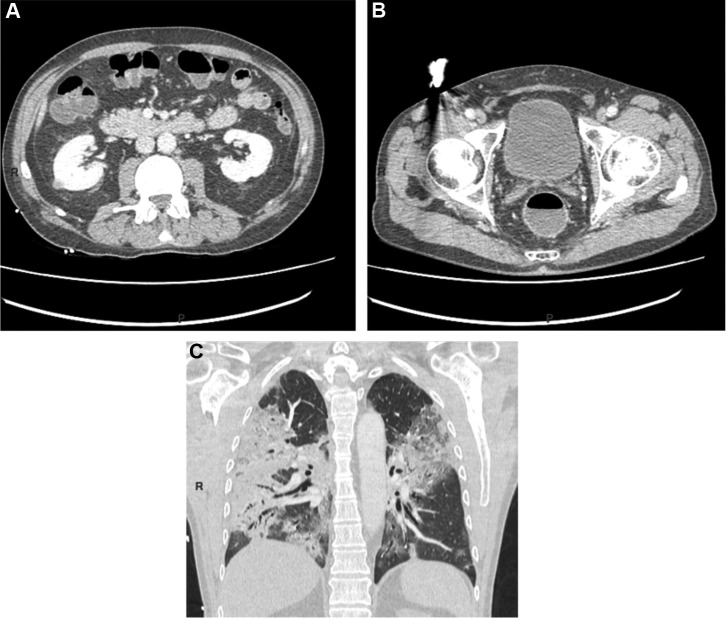
Figure 2.
A: colonic fluid stasis; B: rectal fluid stasis; C: severe pulmonary injury with large ground glass opacification and predominantly sub-pleural bilateral condensation.
Figure 3.
A: sigmoid fistula in contact with the left psoas muscle; B and C: fortuitous discovery on thoracic slices of bilateral predominantly sub-pleural and lower lobar ground glass opacities typical of COVID-19.
Endoscopic abnormalities associated with COVID-19
Up until now, no data has been reported in the literature. The extreme contagiousness induced by the aerosolization of saliva droplets or coughing during upper gastrointestinal endoscopy and the potential contagiousness of stools in lower gastrointestinal endoscopy are such that these procedures should be limited to emergency endoscopy [19]. In clinical practice, the indications are as follows: digestive hemorrhage with deglobulization, impaction of foreign bodies, symptomatic digestive stenosis and biliary obstruction, necrosectomy or drainage of pancreatic fluid collection, sigmoid volvulus. Even though the Société française d’endoscopie digestive recommends continuation (to the greatest possible extent) of colonoscopy in the event of positive fecal blood test and as a means of exploring iron deficiency anemia, it seems illusory to hope to apply these recommendations in highly endemic areas. In Ile de France, for example, virtually all public and private structures limit implementation of these procedures to dire, life-threatening emergencies.
Specific gastroenterology populations
Chronic inflammatory bowel disease (IBD)
With our current level of knowledge, IBD patients are not more at risk of this infection than the general population [20]. Up until now, no case of severe COVID-19-related infection has been reported in IBD patients exposed to either immunosuppressive therapy (azathioprine, methotrexate) or a monoclonal antibody (anti-TNF, ustekinumab, vedolizumab). It is consequently recommended not to suspend immunomodulatory treatments to reduce the risk of COVID-19 infection.Discontinuation of treatment would increases the risk of relapse with an additional loss of opportunity due to the difficulties of obtaining emergency care during the sanitary crisis. A worldwide register of IBD patients with COVID-19 infection has been set up by the IOIBD (International Organization for the Study of Inflammatory Bowel Diseases), which should be providing supplementary details over the months to come.
Digestive cancers
Two studies seem to show that cancer patients have a higher risk of developing a COVID-19 infection with a more advanced age at diagnosis and a less favorable prognosis than for non-cancer patients[21], [22]. Nevertheless, among the 3114 patients analyzed in the two studies, only 30 presented with cancer, including 11 undergoing treatment (chemotherapy ± immunotherapy N = 5, chemotherapy and/or surgery during the previous month N = 4, radiotherapy N = 2) and not a single reported case of digestive cancer. Even though these data remain highly fragmentary, digestive cancer patients are probably significantly at risk of COVID-19 infection, particularly those currently undergoing intravenous chemotherapy, who risk contamination during their care pathway (repeated hospitalizations, imaging examinations and blood tests in medicalized structures…). Moreover, given that the mean age of cancer diagnosis in the most frequent tumor locations approximates 70 years, whether undergoing treatment or being monitored this population is clearly particularly at risk of developing a severe infection. For example, among the 6 infected patients in our cohort (mean age 62.6 years, ongoing chemotherapy N = 5, palliative home treatment N = 1, colorectal cancer N = 4, pancreatic cancer N = 1, esophageal cancer N = 1), 3 died of respiratory failure and 2 required oxygen therapy. It is mentioned in the French national thesaurus of digestive cancerology that existing chemotherapy modalities ought to be revised, taking into closer account the risk/benefit ratio (expert agreement). This is singularly relevant, especially insofar as intensive care admission criteria for patients presenting with severe COVID-19 infection are exceedingly strict. That is one reason why patients presenting with incurable metastatic cancer have limited access to mechanical ventilation. It is also necessary to customize treatments according to the level of contamination in the health care structure accommodating the patient.
As regards these two types of “at risk” population, in the event of COVID-19 infection the GETAID (Groupe d’Etude Thérapeutique des Affections Inflammatoires du Tube Digestif) and the TNCD (Thésaurus National de Cancérologie Digestive) recommend suspension of all treatment, until full resolution of symptoms. Even though this approach has yet to be validated, it would seem judicious immediately prior to resumption of treatment to ascertain negativation of at least a throat swab either at the conclusion of quarantine (ambulatory patient) or following resolution of symptoms (hospitalized patient).
In the overwhelming majority of cases involving any type of digestive pathology, teleconsultation is preeminent. Even though there have been no dedicated studies on the subject, its acceptability by actively monitored patients seems satisfactory, and use of the telephone appears to suffice (personal data). Among patients necessitating hospitalization, in some centers systematic search for COVID-19 infection is carried out, even in the absence of symptoms, to avoid potential risk of dissemination in a hospital structure.
Conclusion
The COVID-19 epidemic necessitates continual updating of our knowledge pertaining to the infection. A multiplicity of publications and their free availability online illustrates the intense activity of the medical community on this subject. Even though they are not clinically highlighted, digestive disorders are in all likelihood more frequent than initially reported. They can precede other symptoms or be present in isolation. Biological abnormalities, particularly lymphopenia and moderate elevation of CRP and transaminases, can suggest clinical diagnosis. Digestive imaging is only marginally contributory. However, pulmonary injury can be fortuitously discovered on low thoracic slices in abdominal CT-scan. That is one reason why some authors have discussed the possible value of systematic thoracic CT-scan when abdominal injury is indicated, especially in the event of fever. Resumption of medical-surgical activity as the epidemic recedes will be difficult, given the multiplicity of endoscopic procedures and surgical interventions requiring rescheduling and the delays in treatment of confined non-COVID patients
Following resolution of the first epidemic wave, a screening strategy aimed at limiting the risk of new contaminations will call for discussion on patients suffering from digestive diseases necessitating hospital treatment.
Disclosure of interest
The authors declare that they have no competing interest.
References
- 1.Baud D., Qi X., Nielsen-Saines K., Musso D., Pomar L., Favre G. Real estimates of mortality following COVID-19 infection. Lancet Infect Dis. 2020 doi: 10.1016/S1473-3099(20)30195-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lescure F.-X., Bouadma L., Nguyen D. Clinical and virological data of the first cases of COVID-19 in Europe: a case series. Lancet Infect Dis. 2020 doi: 10.1016/S1473-3099(20)30200-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Liang W., Feng Z., Rao S. Diarrhoea may be underestimated: a missing link in 2019 novel coronavirus. Gut. 2020 doi: 10.1136/gutjnl-2020-320832. [DOI] [PubMed] [Google Scholar]
- 4.Xiao F., Tang M., Zheng X., Liu Y., Li X., Shan H. Evidence for gastrointestinal infection of SARS-CoV-2. Gastroenterology. 2020 doi: 10.1053/j.gastro.2020.02.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Huang C., Wang Y., Li X. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Guan W.-J., Ni Z.-Y., Hu Y. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020 doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Song Y., Liu P., Shi X.L. SARS-CoV-2 induced diarrhoea as onset symptom in patient with COVID-19. Gut. 2020 doi: 10.1136/gutjnl-2020-320891. [DOI] [PubMed] [Google Scholar]
- 8.Luo S., Zhang X., Xu H. Don’t overlook digestive symptoms in patients with 2019 novel coronavirus disease (COVID-19) Clinical Gastroenterology and Hepatology. 2020 doi: 10.1016/j.cgh.2020.03.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tian Y., Rong L., Nian W., He Y. Review article: gastrointestinal features in COVID-19 and the possibility of faecal transmission. Aliment Pharmacol Ther. 2020 doi: 10.1111/apt.15731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jin X., Lian J.-S., Hu J.-H. Epidemiological, clinical and virological characteristics of 74 cases of coronavirus-infected disease 2019 (COVID-19) with gastrointestinal symptoms. Gut. 2020 doi: 10.1136/gutjnl-2020-320926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wölfel R., Corman V.M., Guggemos W. Virological assessment of hospitalized patients with COVID-2019. Nature. 2020 doi: 10.1038/s41586-020-2196-x. [DOI] [PubMed] [Google Scholar]
- 12.Zhang W., Du R.-H., Li B. Molecular and serological investigation of 2019-nCoV infected patients: implication of multiple shedding routes. Emerg Microbes Infect. 2020;9:386–389. doi: 10.1080/22221751.2020.1729071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Peiris J., Chu C., Cheng V. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet. 2003;361:1767–1772. doi: 10.1016/S0140-6736(03)13412-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen L., Lou J., Bai Y., Wang M. COVID-19 Disease With Positive Fecal and Negative Pharyngeal and Sputum Viral Tests. Am J Gastroenterol. 2020 doi: 10.14309/ajg.0000000000000610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mao R., Liang J., Wu K.-C., Chen M.-H. Responding to COVID-19: Perspectives from the Chinese Society of Gastroenterology. Gastroenterology. 2020 doi: 10.1053/j.gastro.2020.03.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shi H., Han X., Jiang N. Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: a descriptive study. Lancet Infect Dis. 2020;20:425–434. doi: 10.1016/S1473-3099(20)30086-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang C., Shi L., Wang F.-S. Liver injury in COVID-19: management and challenges. Lancet Gastroenterol Hepatol. 2020 doi: 10.1016/S2468-1253(20)30057-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Finkelstone L., Wolf E.L., Stein M.W. Etiology of small bowel thickening on computed tomography. Can J Gastroenterol. 2012;26:897–901. doi: 10.1155/2012/282603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ong J., Cross G.B., Dan Y.Y. Prevention of nosocomial SARS-CoV-2 transmission in endoscopy: international recommendations and the need for a gold standard. Gut. 2020 doi: 10.1136/gutjnl-2020-321154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ungaro R.C., Sullivan T., Colombel J.-F., Patel G. What Should Gastroenterologists and Patients Know About COVID-19? Clin Gastroenterol Hepatol. 2020 doi: 10.1016/j.cgh.2020.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yu J., Ouyang W., Chua M.L.K., Xie C. SARS-CoV-2 Transmission in Patients With Cancer at a Tertiary Care Hospital in Wuhan, China. JAMA Oncol. 2020 doi: 10.1001/jamaoncol.2020.0980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liang W., Guan W., Chen R. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol. 2020;21:335–337. doi: 10.1016/S1470-2045(20)30096-6. [DOI] [PMC free article] [PubMed] [Google Scholar]