Abstract
Platelets aggregation leading to thrombosis plays a pivotal role in the pathophysiology of acute coronary syndrome (ACS) and of stent thrombosis. Antiplatelet therapy with aspirin plus an ADP-receptor inhibitor (ticagrerol, prasugrel or clopidogrel) is recommended to reduce the risk of other platelet-mediated events. Clopidogrel is recommended in patients with Chronic Coronary Syndromes (CCS) or in ACS patients at high bleeding risk. Unfortunately, up to 30% of patients are non-responders to clopidogrel and show residual high platelet reactivity (HPR). Colchicine (COLC) is a drug with cardiovascular effects. We have demonstrated that COLC might exert protective cardiovascular effects by interfering with cytoskeleton rearrangement, a phenomenon involved in platelet aggregation. Here, we investigate in vitro the effects of colchicine on platelet aggregation of patients on DAPT with clopidogrel. Platelets obtained from 35 CCS patients on therapy with clopidogrel were pre-incubated with COLC 10 µM before being stimulated with ADP (20 µM), or TRAP (25 µM) at 0, 30, 60 and 90 min to measure max aggregation by LTA. Platelets not COLC-preincubated served as controls. Seven patients were pre-selected as clopidogrel non-responders. COLC significantly reduced TRAP-induced platelet aggregation in clopidogrel responders and non-responders. Interestingly, COLC inhibited ADP-induced platelet aggregation in clopidogrel non-responders in which ADP still caused activation despite DAPT. We demonstrate that COLC inhibits platelet aggregation in clopidogrel non-responders with HPR despite DAPT with this ADP receptor-inhibitor. Further in vivo studies should be designed to evaluate the opportunity to prescribe colchicine after ACS/CCS to overcome the clopidogrel limitations in the DAPT therapy.
Keywords: Clopidogrel, Colchicine, DAPT, Platelets
Highlights
Clopidogrel is recommended in selected patients to avoid platelet-mediated thrombosis.
Patients non-responders to clopidogrel (NRC) show residual high platelet reactivity (HPR).
Colchicine is a drug with cardiovascular effects with partially unknown mechanisms.
We have previously shown that colchicine inhibits platelet aggregation.
Here we demonstrate that colchicine inhibits platelet aggregation in NRC patients.
Introduction
Dual antiplatelet therapy (DAPT) with aspirin and a P2Y12 receptor inhibitor is mandatory for the prevention of thrombotic events in patients undergoing percutaneous coronary intervention and stenting (PCI-S) [1]. The more potent P2Y12 inhibitors, prasugrel and ticagrelor, are recommended in patients with acute coronary syndromes (ACS). Clopidrogrel is recommended in patients with Chronic Coronary Syndromes (CCS) or in ACS patients treated with PCI-S and at high bleeding risk such as those on therapy with anticoagulants because of atrial fibrillation [1, 2]. Unfortunately, the lack of response to antiplatelet therapy, with residual high platelet reactivity (HPR) has been demonstrated in about 30% of patients on therapy with clopidogrel [3, 4]. Importantly, HPR patients are at greater risk for future major adverse cardiovascular events [4].
Colchicine is a drug extracted from Colchicum autumnale [5]. To date, its use in clinical practice is mainly recommended for the treatment of acute gout, and of its prophylaxis [6]. Several studies recommend colchicine to treat inflammatory cardiovascular diseases such as acute pericarditis and chronic pericardial disease [7]. Finally, different reports have suggested its potential benefits also in acute and chronic cardiovascular disease [8, 9]. Interestingly, the recently published COLCOT Trial has demonstrated that low doses of colchicine significantly reduce the risk of new ischemic cardiovascular events in ACS patients [10]. The observed cardiovascular effects of colchicine have been explained by considering the primary mechanism of action of this drug, represented by tubulin disruption, a phenomenon that causes down regulation of multiple inflammatory pathways and modulation of innate immunity [5]. Thus, it has been suggested that colchicine effects on cardiovascular system might be due to its ability to modulate the immuno-inflammatory pathways involved in athero-thrombotic pathophysiology [10]. However, we have recently suggested another potential mechanism of action for colchicine in the scenario of coronary syndromes since we have demonstrated that this drug reduces platelet aggregation of healthy donors in vitro via inhibition of key proteins involved in cytoskeleton rearrangement [11]. Thus, by considering that in the COLCOT trial, despite use of a P2Y12 inhibitor was around 98% of patients, the percentage of clopidogrel (and of potential residual HPR) use was not specified, we have investigated the effects of colchicine on platelet aggregation of patients on DAPT with clopidogrel.
Methods
Venous blood was collected from 35 patients with CCS treated with PCI-S. All patients started DAPT (clopidogrel plus aspirin) at least 7 days before and were currently on DAPT therapy. All patients had taken the last clopidogrel dose (75 mg) 4 h before collection of blood samples. Informed consent was obtained from each patient and the study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution's human research committee. Blood was collected into vacutainer tubes containing 3.8% sodium citrate (9:1 v/v) and centrifuged at 120×g for 20 min at room temperature to obtain platelet-rich plasma (PRP) as previously described [11]. The level of platelet aggregation was evaluated by Light Transmission Aggregometry (LTA) and expressed as maximal aggregation (AGGmax) in PRP after activation at baseline, 30, 60, and 90 min, in response to Adenosine Diphosphate (ADP, 20 µM, Sigma-Aldrich, Milan, Italy), and Thrombin Receptor Activating Peptide (TRAP 25 µM, Sigma Aldrich, Milan, Italy). To investigate the effects of colchicine on platelet aggregation, PRP was pre-incubated 1 h at 37 °C with colchicine 10 μM [11], and then platelets were stimulated with ADP or TRAP as above. Platelets aggregometry was performed at 37 °C following standard procedure [12]. Twenty milliliters of Platelet Rich Plasma (PRP) were used for platelets activation under continuous stirring at 37 °C.
Statistical analysis
Results are expressed as means ± SD. All data from in vitro experiments were analyzed by one-way ANOVA followed by the Tukey test for multiple comparison test or by a non-parametric test (Kruskal–Wallis) when data were not normally distributed. All analyses were performed using the GraphPad Prism 4.00 for Windows software (GraphPad Software, San Diego California, USA).
Results
In 28 patients, platelets stimulated with ADP showed the following mean AGGmax values: 26 ± 2% at baseline; 25 ± 4% at 30 min, 26 ± 2% at 60 min and 21 ± 3% at 90 min. These patients were classified as clopidogrel-responders. On the contrary, in other 7 patients, platelets stimulated with ADP showed the mean AGGmax values of 83 ± 4% at baseline, 80 ± 3% at 30 min, 79 ± 4% at 60 min and 75 ± 4% at 90 min. These patients were classified as clopidogrel non-responders (Figs. 1 and 2, Panel A). When platelets were stimulated with TRAP, that does not cause platelet aggregation binding to ADP receptor, a significant platelet aggregation was observed with similar elevated AGGmax values in clopidogrel-responders as well as non-responders throughout 90 min (Figs. 1 and 2, Panel B). Preincubation with colchicine significantly reduced AGGmax after stimulation with TRAP in clopidogrel responders (22 ± 7; 19 ± 4; 15 ± 1 p < 0.05) and non-responders (20 ± 9; 24 ± 8; 22 ± 1 p < 0.05) as compared to TRAP-stimulated platelets but not preincubated with colchicine (Figs. 1 and 2, Panel B). In clopidogrel responders, preincubation with colchicine did not cause any significant additional effect on AGGmax after ADP stimulation as compared with platelets not treated with colchicine (20 ± 3% at 30 min, 19 ± 2% at 60 min and 20 ± 3% at 90 min, p:NS) (Fig. 1, Panel A). Interestingly, in clopidogrel non-responders, in whom HPR was still observed despite DAPT, preincubation with colchicine significantly reduced AGGmax in response to ADP (22 ± 12%, 19 ± 11%, 21 ± 8%, p < 0.05) (Fig. 2, Panel A).
Fig. 1.
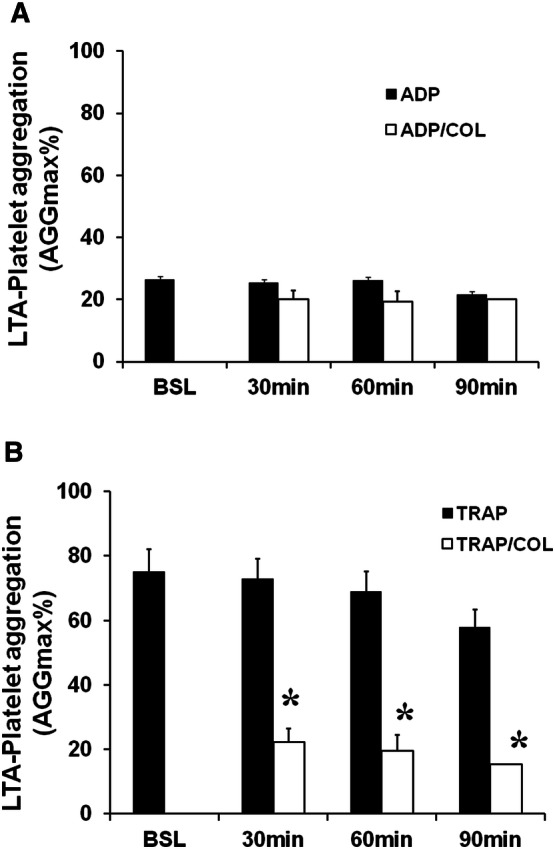
Platelet aggregation in patients’ clopidogrel responders. Panel A, Stimulation with ADP did not cause significant platelet aggregation. Any significant difference was observed after colchicine (COL) preincubation. Panel B, Stimulation with TRAP caused significant platelet aggregation. Preincubation with COL significantly reduced TRAP-induced platelet aggregation. Data are expressed as mean ± SD; *p < 0.05 vs TRAP alone
Fig. 2.
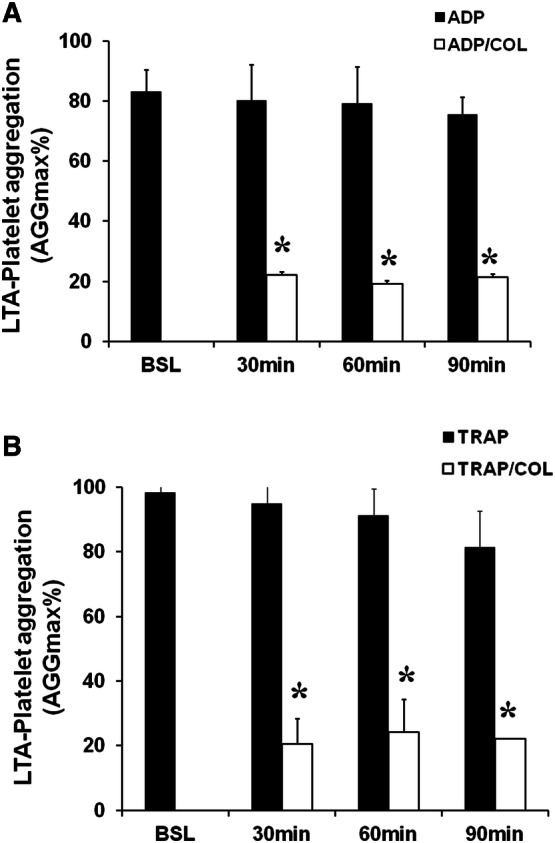
Platelet aggregation in patients’ clopidogrel non-responders. Panel A, Platelets stimulated with ADP showed significant activation. Colchicine (COL) reduced ADP-induced platelet aggregation. Panels B, Stimulation with TRAP caused significant platelet aggregation. Preincubation with COL significantly reduced TRAP-induced platelet aggregation. Data are expressed as mean ± SD; *p < 0.05 vs ADP or TRAP alone
Discussion
Platelets play a critical role in the maintenance of physiological hemostasis and are actively involved in thrombosis observed in ACS and in CCS after stent implantation [13]. Once activated, platelets undergo dramatic changes in their shape due to reorganization of cytoskeletal components, including microtubules. Several stimuli including ADP or thrombin activate platelets by binding to their specific surface receptors [14, 15]. To inhibit platelet mediated thrombosis, DAPT in selected patients includes aspirin and an ADP-receptor inhibitor such as clopidogrel [1, 2]. However, high on-clopidogrel platelet reactivity (HPR) defined as an AGGmax > 46% despite DAPT [4], still persists in a substantial proportion of clopidogrel non-responder patients. Of note, HPR-patients have an increased risk of new cardiovascular thrombotic events [16]. Here, we demonstrate that colchicine significantly reduces HPR observed in patients on DAPT with aspirin plus clopidogrel but not-responders to this P2Y12 inhibitors, witnessed by AGGmax > 46%. Our results are in line with those of a previous study in which colchicine attenuated platelet activation but in healthy donors not on DAPT [17]. On the contrary, our data seem to be only apparently in contrast with those by Raju et al. who showed that colchicine did not reduce platelet aggregation in ACS patients on DAPT with clopidogrel [18]. Indeed, in the present study colchicine does not reduce platelet aggregation in clopidogrel responders, as in the Raju's study, but significantly reduces this phenomenon in clopidogrel non-responders, not specifically investigated in that study. Results of our study, with all the limitations of an in vitro study, might give another pathophysiological point of view to explain the beneficial effects observed in COLCOT trial for ACS patients treated with colchicine: we cannot exclude that colchicine effects might be due not only to its role in modulating inflammation as suggested by the Authors, but also to its proprieties as antiplatelet drug. However, it should be kept in mind that in the COLCOT trial, the percentage of patients treated with prasugrel or ticagrelor rather than clopidogrel was not specified, representing a major limitation of our hypothesis. Moreover, another missing piece in this puzzle is that bleeding outcomes were not assessed in the COLCOT trial. Thus, we do not have data about a possible increase of bleeding risk that could be found in patients treated with P2Y12 inhibitors and concomitant colchicine.
Further in vivo studies should be designed to evaluate the opportunity to prescribe colchicine after ACS/CCS to overcome the clopidogrel limitations in the DAPT therapy.
Acknowledgements
This paper is dedicated to our families. In these days of COVID-19 crisis, their constant support has permitted to overcome our daily difficulties in research/hospital centers and to conclude our work.
Funding
No specific grant supported the present article.
Compliance with ethical standards
Conflicts of interest
All Authors declare that they have no conflict of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Valgimigli M, Bueno H, Byrne RA, Collet JP, Costa F, Jeppsson A, Jüni P, Kastrati A, Kolh P, Mauri L, Montalescot G, Neumann FJ, Petricevic M, Roffi M, Steg PG, Windecker S, Zamorano JL, Levine GN. 2017 ESC focused update on dual antiplatelet therapy in coronary artery disease developed in collaboration with EACTS: the task force for dual antiplatelet therapy in coronary artery disease of the European society of cardiology (ESC) and of the European association for cardio-thoracic surgery (EACTS) Eur Heart J. 2018;39(3):213–260. doi: 10.1093/eurheartj/ehx419. [DOI] [PubMed] [Google Scholar]
- 2.Neumann FJ, Sousa-Uva M, Ahlsson A, Alfonso F, Banning AP, Benedetto U, Byrne RA, Collet JP, Falk V, Head SJ, Jüni P, Kastrati A, Koller A, Kristensen SD, Niebauer J, Richter DJ, Seferovic PM, Sibbing D, Stefanini GG, Windecker S, Yadav R, Zembala MO. ESC Scientific Document Group. 2018 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J. 2019;40(2):87–165. doi: 10.1093/eurheartj/ehy394. [DOI] [PubMed] [Google Scholar]
- 3.Warlo EMK, Arnesen H, Seljeflot I. A brief review on resistance to P2Y12 receptor antagonism in coronary artery disease. Thromb J. 2019;17:11. doi: 10.1186/s12959-019-0197-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bonello L, Tantry US, Marcucci R, Blindt R, Angiolillo DJ, Becker R, Bhatt DL, Cattaneo M, Collet JP, Cuisset T, Gachet C, Montalescot G, Jennings LK, Kereiakes D, Sibbing D, Trenk D, Van Werkum JW, Paganelli F, Price MJ, Waksman R, Gurbel PA, Working Group on High On-Treatment Platelet Reactivity Consensus and future directions on the definition of high on-treatment platelet reactivity to adenosine diphosphate. J Am Coll Cardiol. 2010;56(12):919–933. doi: 10.1016/j.jacc.2010.04.047. [DOI] [PubMed] [Google Scholar]
- 5.Slobodnick A, Shah B, Krasnokutsky S, Pillinger MH. Update on colchicine, 2017. Rheumatology (Oxford) 2018;57(suppl_1):i4–i11. doi: 10.1093/rheumatology/kex453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Leung YY, Yao Hui LL, Kraus VB. Colchicine-update on mechanisms of action and therapeutic uses. Semin Arthritis Rheum. 2015;3:341–350. doi: 10.1016/j.semarthrit.2015.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Adler Y, Charron P, Imazio M, Badano L, Barón-Esquivias G, Bogaert J, Brucato A, Gueret P, Klingel K, Lionis C, Maisch B, Mayosi B, Pavie A, Ristic AD, Sabaté Tenas M, Seferovic P, Swedberg K, Tomkowski W, ESC Scientific Document Group European Society of, ESC guidelines for the diagnosis and management of pericardial diseases: the task force for the diagnosis and management of pericardial diseases of the european society of cardiology (ESC) endorsed by: the European association for cardio-thoracic surgery (EACTS) Eur Heart J. 2015;36(42):2921–2964. doi: 10.1093/eurheartj/ehv318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Martínez GJ, Robertson S, Barraclough J, Xia Q, Mallat Z, Bursill C, Celermajer DS, Patel S. Colchicine acutely suppresses local cardiac production of inflammatory cytokines in patients with an acute coronary syndrome. J Am Heart Assoc. 2015;4(8):e002128. doi: 10.1161/JAHA.115.002128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nidorf SM, Eikelboom JW, Budgeon CA, Thompson PL. Low-dose colchicine for secondary prevention of cardiovascular disease. J Am Coll Cardiol. 2013;61(4):404–410. doi: 10.1016/j.jacc.2012.10.027. [DOI] [PubMed] [Google Scholar]
- 10.Tardif JC, Kouz S, Waters DD, Bertrand OF, Diaz R, Maggioni AP, Pinto FJ, Ibrahim R, Gamra H, Kiwan GS, Berry C, López-Sendón J, Ostadal P, Koenig W, Angoulvant D, Grégoire JC, Lavoie MA, Dubé MP, Rhainds D, Provencher M, Blondeau L, Orfanos A, L'Allier PL, Guertin MC, Roubille F. Efficacy and safety of low-dose colchicine after myocardial infarction. N Engl J Med. 2019;381(26):2497–2505. doi: 10.1056/NEJMoa1912388. [DOI] [PubMed] [Google Scholar]
- 11.Cimmino G, Tarallo R, Conte S, Morello A, Pellegrino G, Loffredo FS, Calì G, De Luca N, Golino P, Trimarco B, Cirillo P. Colchicine reduces platelet aggregation by modulating cytoskeleton rearrangement via inhibition of cofilin and LIM domain kinase 1. Vascul Pharmacol. 2018;111:62–70. doi: 10.1016/j.vph.2018.09.004. [DOI] [PubMed] [Google Scholar]
- 12.Cimmino G, Tarallo R, Nassa G, De Filippo MR, Giurato G, Ravo M, Rizzo F, Conte S, Pellegrino G, Cirillo P, Calabro P, Öhman T, Nyman TA, Weisz A, Golino P. Activating stimuli induce platelet microRNA modulation and proteome reorganisation. Thromb Haemost. 2015;114(1):96–108. doi: 10.1160/TH14-09-0726. [DOI] [PubMed] [Google Scholar]
- 13.Freynhofer MK, Bruno V, Wojta J, Huber K. The role of platelets in atherothrombotic events. Curr Pharm Des. 2012;18(33):5197–5214. doi: 10.2174/138161212803251899. [DOI] [PubMed] [Google Scholar]
- 14.Cimmino G, Golino P. Platelet biology and receptor pathways. J Cardiovasc Transl Res. 2013;6(3):299–309. doi: 10.1007/s12265-012-9445-9. [DOI] [PubMed] [Google Scholar]
- 15.Cerecedo D. Platelet cytoskeleton and its hemostatic role. Blood Coagul Fibrinolysis. 2013;24(8):798–808. doi: 10.1097/MBC.0b013e328364c379. [DOI] [PubMed] [Google Scholar]
- 16.Aradi D, Komócsi A, Vorobcsuk A, Rideg O, Tokés-Füzesi M, Magyarlaki T, Horváth IG, Serebruany VL. Prognostic significance of high on-clopidogrel platelet reactivity after percutaneous coronary intervention: systematic review and meta-analysis. Am Heart J. 2010;160(3):543–551. doi: 10.1016/j.ahj.2010.06.004. [DOI] [PubMed] [Google Scholar]
- 17.Reddel CJ, Pennings GJ, Curnow JL, Chen VM, Kritharides L. Procoagulant effects of low-level platelet activation and its inhibition by colchicine. Thromb Haemost. 2018;118(4):723–733. doi: 10.1055/s-0038-1636915. [DOI] [PubMed] [Google Scholar]
- 18.Raju NC, Yi Q, Nidorf M, Fagel ND, Hiralal R, Eikelboom JW. Effect of colchicine compared with placebo on high sensitivity C-reactive protein in patients with acute coronary syndrome or acute stroke: a pilot randomized controlled trial. J Thromb Thrombolysis. 2012;33(1):88–94. doi: 10.1007/s11239-011-0637-y. [DOI] [PubMed] [Google Scholar]


