Abstract
This article describes several aspects of oxygen (O2) deficiency with an emphasis on respirator programs and respirator selection. The Occupational Safety and Health Administration’s (OSHA) 29 CFR 1910.1341 and ANSI/ASSE Z88.2-2015 (Z88.2) have much in common. However, their exposure criteria and terminology used for describing levels of O2-deficiency and the approaches to assessing O2-deficiency differ. These differences can have a significant impact on an employer’s respirator program and respirator selections for workplaces at altitudes above sea level.
Under certain circumstances, Z88.2 leads to a more conservative respirator selection than OSHA because its O2-deficiency criteria and hazard assessment approach relies directly on partial pressure of oxygen (PO2)2 at all altitudes. Z88.2 defines an O2-deficient atmosphere as either immediately dangerous to life or health (IDLH), or non-IDLH based on the atmosphere’s PO2 and defines respirator selection for these two O2-deficient atmospheres. Unlike Z88.2, OSHA does not directly access the biologically significant aspect of an atmosphere’s PO2 in its hazard assessment. OSHA defines an O2-deficient atmosphere based upon a percentage of oxygen. OSHA does not use the term “O2-deficient IDLH”; however, OSHA considers any atmosphere with less than 19.5% O2 as IDLH and defines respirator selection for IDLH atmospheres. Although OSHA does not use the term “PO2” in their respirator standard, OSHA’s exceptions to O2-deficient IDLH respirator selection policy are based on PO2 altitude-adjusted, O2 percentage criteria.
This article provides descriptions of OSHA and Z88.2 requirements to evaluate workplace oxygen deficiency, their approaches to O2-deficiency hazard assessment, and describes their significance on respirator programs and selections. Alternative solutions to wearing respirators for protection against O2-deficiency resulting solely from high altitudes are also discussed. Selection and implementation of alternative solutions by the employer and their Physician or other Licensed Health Care Professional (PLHCP) are not covered by either respirator standard. Appendix A provides information about the physiological effect of wearing respirators and the mechanics of respiration, which is an important consideration in lower O2 atmospheres.
Keywords: Respiratory protection, Oxygen Deficient Atmospheres, Respirator program, Z88.2 standards
Understanding Oxygen Deficiency
Each year, many workers are injured or die due to O2-deficiency. Bureau of Labor Statistics (BLS, 2011) reported 62 and 60 occupational fatalities, respectively during the years 2009 and 2010 due to O2-deficiency. One of the problems with O2-deficient atmospheres is that at lowered concentrations, starting at approximately 16% O2 by volume, attentiveness and thinking becomes impaired. This of course, varies based on the individual’s fitness and acclimatization. Individuals impaired by exposure to deficient levels of O2 have died simply because they did not recognize the peril they were in, even though they should have been able to easily escape from this hazard. There have been reports of workers who have opened a hatch to an O2-deficient atmosphere and died with only their head inside the hazard. The low level of O2 resulted in a feeling of euphoria and the workers could not comprehend that they only needed to lean back out of the hatch to save their lives. From discussion with one of the original human test subjects in an early study of hypoxia (Reinert, 2016), he knew that he was about to die and would have allowed himself to die if he had not been in a controlled experiment. He explained that he was not concerned about his imminent death because of the intense euphoric feeling experienced from breathing low concentrations of oxygen.
The oxygen concentration in atmospheric air is 20.95% by volume throughout the atmosphere, from sea level to the highest mountain top. Up to 70,000 feet (21,336 meters), the percentage of oxygen and other gaseous components of air remains the same as at sea level. However, the partial pressure of air components, including oxygen PO2, decreases with increasing altitude because as atmospheric pressure decreases fewer molecules exist in a given volume of air. The concept that percentage of oxygen remains the same at high altitudes has nothing to do with the amount of oxygen available for biological processes. The more significant concept to human performance and survival at high altitudes and O2-deficient atmospheres at sea level is that of biologically available oxygen based on PO2. It is the PO2 level that determines the amount of oxygen that is available for respiration and biological metabolism. The amount of biologically available oxygen for respiration decreases as the PO2 decreases. Another consideration for respirator selection air-purifying or demand- and pressure-demand supplied-air respirators (not continuous flow), which is a consideration when working in atmospheric oxygen concentrations below the ambient oxygen level of PO2 <159 (<20.95% at sea level) because the volume inside the facepiece can affect the oxygen content breathed by the wearer when these respirators are used in reduced oxygen atmospheres.
The percent oxygen and PO2 can also be decreased when oxygen is consumed (oxidation) or displaced by other gases or vapors including water vapor. In other cases, there can be combined effects due to an increase in altitude plus displacement of oxygen. Atmospheric pressure is also affected by temperature, relative humidity, and other factors. Average atmospheric pressure even differs with latitude above and below the equator. An example of the impact water vapor has on the atmospheric oxygen level is described in Sidebar 1. Sidebar 1 also illustrates reduction in the percentage of atmospheric O2 and PO2 from the combined effects of oxygen displacement plus an increase in altitude.
Sidebar 1. Role of Water Vapor in Atmospheric Oxygen Levels.
Moisture, in the form of water vapor, is the most variable atmospheric component. Atmospheric water vapor ranges from 0 to 4%. In dry air with 0% water vapor, the oxygen concentration is approximately 20.95%. However, atmospheres with 4% water vapor contain only 20.06% oxygen.
X = (0.2095) (0.96)/1 = 0.2011
X = (0.2011) (100) = 20.11%
At Sea Level Atmospheric PO2 = 760 mmHg
At 7,000 Feet Atmospheric PO2 = 543 mmHg
For the conditions faced by the respirator wearer, respirator program administrators are encouraged to measure their actual atmospheric pressure and related parameters and not estimate it. Additionally, for the purposes of Z88.2, the atmospheric PO2 level must be known.
Annex A.3 of Z88.2, explains that PO2, not the percentage of O2 by volume is of greater biological significance. We will primarily use the term PO2 in this article when discussing the Z88.2 standard; however, discussions concerning the OSHA policy will, by necessity, require using percentages of oxygen because that is their terminology. Although discussed in the 1998 Preamble, nowhere in the OSHA respirator standard is the term PO2 used.
OSHA’s Requirements to Evaluate Workplace Oxygen Deficiency
The 1998 Preamble provides an informative discussion on the treacherous uncertainties of O2-deficient environments in which employers may mistakenly believe that they have a location’s oxygen content under control. The 1998 Preamble described how accurate oxygen level monitoring can be difficult, since testing is limited to a finite number of areas, allowing for the possibility that untested pockets of lower oxygen levels may exists. The 1998 Preamble further explains that it is difficult to ensure oxygen levels will not drop suddenly with little or no warning with oxygen being displaced by other gases, vapors, or by work that consumes oxygen.
The wording in paragraph (d) of 29 CFR 1910.134 is very succinct, it “requires the employer to evaluate respiratory hazard(s) in the workplace, identify relevant workplace and user factors, and base respirator selection on these factors.” This includes evaluating the workplace for O2-deficiency. Paragraph (d)(1)(iii) further states, “The employer shall identify and evaluate the respiratory hazard(s) in the workplace; this evaluation shall include a reasonable estimate of employee exposures to respiratory hazard(s) and an identification of the contaminant’s chemical state and physical form. Where the employer cannot identify or reasonably estimate the employee exposure, the employer shall consider the atmosphere to be IDLH.”
OSHA Oxygen Exposure Limits and Hazard Assessment Approach
OSHA evaluates an atmosphere’s percent oxygen. It does not directly rely upon the biologically significant aspect of an atmosphere based on PO2 as does Z88.2. The OSHA respirator standard defines O2-deficient atmospheres as having an oxygen content below 19.5% by volume, which it considers IDLH. The standard requires that the most protective and reliable respirators be used for IDLH atmospheres. Per paragraphs (d)(2)(i)(A) and (B), for entry into IDLH atmospheres, OSHA requires the use of a full facepiece pressure-demand SCBA or a combination full facepiece pressure-demand supplied-air respirator (SAR) with auxiliary self-contained air supply.
OSHA allows wearing air-purifying respirators provided the respirator is adequate to protect the health of employees and is in compliance with all other OSHA statutory and regulatory requirements under routine and foreseeable emergency situations (paragraph (d) (3)). Although OSHA does not prohibit wearing air-purifying respirators in O2-reduced atmospheres, the standard’s preamble clearly explains that employees are not permitted to work in atmospheres containing <19.5 % oxygen without an atmosphere supplying respirator, which rules out the use of air-purifying respirators (APR). Also, 1910.134(d)(1)(ii) requires that only National Institute for Occupational Safety and Health (NIOSH) approved respirators can be worn and used in compliance with the conditions of its certificate of approval. One of the limiting conditions of NIOSH APR approval is they must be used in atmospheres containing at least 19.5% oxygen (NIOSH, 2004)
As described in the 1998 Preamble, OSHA “…will allow acclimated workers to continue to perform their work without oxygen-supplying respirators, at any altitude up to 14,000 feet altitude, as long as the ambient oxygen content remains above 19.5% and the employee has no medical condition that would require the use of supplemental oxygen.” Additionally, above the altitude of 14,000 feet OSHA requires oxygen-enriched breathing air.
Logic behind the OSHA Oxygen Exposure Limits and Hazard Assessment Approach
OSHA believes that the 19.5% oxygen cutoff is appropriate for safety and health issues and is consistent with the O2-deficiency policy in its confined space standard (OSHA 1994) and the requirement that Grade D (CGA, 2011) breathing air must contain a minimum of 19.5% oxygen for atmosphere-suppling respirators.
OSHA Exceptions to <19.5% O2 by Volume IDLH and Respirator Selection Requirement
The Final Rule defines altitude-adjusted, non-IDLH oxygen percentages, and allows exceptions to IDLH respirator selections at altitudes up to 8,000 feet, provided the employer can demonstrate that under all foreseeable conditions, the oxygen concentration can be maintained within the ranges specified in Table II (reproduced in Table I), of paragraph (d)(2)(iii).
Table I.
Table II of 29 CFR 1910.134
| Table II
| |
|---|---|
| Altitude (ft.) | Oxygen deficient Atmospheres (% 02) for which the employer may rely or atmosphere-supplying respirators |
|
| |
| Less than 3,001 | 16.0–19.5 |
| 3,001–4,000 | 16.4–19.5 |
| 4,001–5,000 | 17.1–19.5 |
| 5,001–6,000 | 17.8–19.5 |
| 6,001–7,000 | 18.5–19.5 |
| 7,001–8,0001 | 19.3–19.5. |
Above 8,000 feet the exception does not apply. Oxygen-enriched breathing air must be supplied above 14,000 feet.
Logic behind OSHA Exceptions to <19.5% O2 by Volume IDLH and Respirator Selection Requirement
Per the 1998 Preamble, the exceptions are permitted “if the employer can meet the difficult evidentiary burden of showing that the oxygen content can be controlled reliably enough to remain within the ranges specified in Table II, the atmosphere is not considered IDLH under this standard, and the employer may provide any SAR.”
OSHA’s Table II (reproduced in Table 1) is specified in paragraph (d)(2)(iii). However according to the 1998 Preamble,” OSHA believes that employers will only rarely have occasion to avail themselves of the exception in paragraph (d)(2)(iii) … Most atmospheres with oxygen content between 16 and 19.5 percent are not well-controlled, and a drop in oxygen content could have severe consequences.”
OSHA O2-deficient policy defines O2 content below 19.5% by volume as IDLH. Per the 1998 Preamble, OSHA’s logic for allowing any atmosphere-supplying respirator (e.g., SAR) to be worn when employers understand, control, and demonstrate that workplace oxygen is strictly controlled within O2 concentrations of 29 CFR 1910.134, Table II is largely based on the ANSI Z88.2-1980 (ANSI, 1980) definition for O2-deficient IDLH (See Sidebar 2). The Z88.2-1980 O2-deficient IDLH atmosphere cutoff point, is, “an oxygen partial pressure of 100 mmHg or less in the freshly inspired air in the upper portion of the lungs [trachea], which is saturated with water vapor.” PO2 ≤ 100 mmHg in the trachea equates to an estimated alveolar blood having only 90% oxygenated hemoglobin, which is a dangerously low level of oxygen for humans. O2-deficiency symptoms become very noticeable below this blood oxygenation level.
Sidebar 2. OSHA Exception to 19.5% O2–Deficient IDLH Limit Based on Z88.2-1980 O2–Deficient IDLH Equation For Altitudes Between 3,001 to 8,000 Feet.
The ANSI Z88.2-1980 O2-deficient IDLH atmosphere cutoff point equation is reproduced below:
*Note: 47 mmHg is the partial pressure of water vapor of the air in the trachea which is saturated with water vapor.
This calculation as shown below is rearranged to calculate the allowable lower oxygen limit for each altitude range between 3,001 to 8,000 feet in OSHA’s Table II (reproduced in Table 1), which is the percent oxygen required to keep the atmosphere above the Z88.2-1980 oxygen deficient-IDLH level (i.e., above PO2 ≤ 100 mmHg in the trachea saturated with water vapor):
The Final Rule preamble explains “that the language of paragraph (d)(2)(iii), along with the exception, reflects the same requirement as that proposed [1994 Proposed Rule](OSHA, 1994B), “but avoids the potential confusion associated with having separate definitions and requirements for oxygen-deficient, and oxygen-deficient IDLH, atmospheres, as originally proposed.”
According to the 1994 Proposed Rule, OSHA decided,” … up to 8,000 feet the oxygen concentration equivalent of an oxygen partial pressure of 100 mm of Hg in the upper portion of the lungs [trachea] is appropriate for a threshold IDLH level. This is equivalent at sea level to an oxygen concentration of 14%. However, NIOSH has pointed out (Ex. 25-4) that in the presence of an oxygen concentration of less than 16% at sea level one can experience impaired attention, thinking and coordination. At 14% or below, NIOSH states the possibility of faulty judgment, poor muscular coordination, rapid fatigue that could cause permanent heart damage, and intermittent respiration. In an IDLH or escape situation all of the described effects could place a worker in serious jeopardy. Therefore, OSHA believes that an oxygen concentration of 16% or below at sea level should require the extra precautions that go with IDLH atmospheres [Bolding is authors’ emphasis].”
This is the reason why OSHA chose 16% O2 as the IDLH limit for atmospheres from sea level up to 3,000 feet instead of allowing 14% O2 as calculated by the O2-deficient IDLH equation. Paradoxically, OSHA considers PO2 in the trachea equivalent to 14% as being too low of an oxygen exposure limit at sea level, but allows the PO2 in the trachea equivalent to 14% at sea level as the acceptable oxygen exposure limit for altitudes from 3,001 to 8,000 feet.
The best description of OSHA’s exception to O2-deficiency IDLH policy is in the original 1994 Proposed Rule succinct summary of paragraph (d)(2)(iii) and the original table of paragraph (d)(2)(iii) reproduced below:
“OSHA is proposing a value of 16% oxygen by volume as the level below which an oxygen deficient IDLH atmosphere exists for altitudes from sea level to 3,000 feet. For altitudes from 3,001 feet up to 8,000 feet [Bolding is authors’ emphasis], percent oxygen levels have been calculated that correspond to a value of 100 mmHg oxygen partial pressure [in upper portion of the lungs]. At altitudes above 8,000 feet and up to 14,000 feet, OSHA is proposing that an oxygen level below 19.5% would be considered an oxygen deficient IDLH atmosphere. This agrees with the ANSI Z 88.2-1980 oxygen deficiency-IDLH level of 100 mmHg [in the trachea], which corresponds to the point where the oxygen content of the alveolar blood is 90% saturated with oxygen and below which symptoms of hypoxia occur. …To avoid possible confusion, OSHA has not used a formula for calculating the oxygen deficient IDLH levels as ANSI did, but instead presents in Column 3 of Table I in paragraph (d) a list of the percent oxygen levels for altitudes from sea level to 14,000 feet.”
Table I.
Oxygen Percentages Constituting Oxygen Deficient and Oxygen Deficient IDLH Atmospheres
| Column 1 altitude above sea level (in feet) | Column 2 percent oxygen below which an oxygen deficient atmosphere exists | Column 3 percent oxygen below which an oxygen deficient IDLH atmosphere exists |
|---|---|---|
| 0 to 3000 | 19.5 | 16.0 |
| 3001 to 4000 | 19.5 | 16.4 |
| 4001 to 5000 | 19.5 | 17.1 |
| 5001 to 6000 | 19.5 | 17.8 |
| 6001 to 7000 | 19.5 | 18.5 |
| 7001 to 8000 | 19.5 | 19.3 |
| Above 8000 to 14,000 | (1) | 19.5 |
Footnote (1) For altitudes above 8000 feet, an oxygen deficient IDLH atmosphere exists when the oxygen level falls below 19.5%”
Z88.2 Requirements to Evaluate Workplace Oxygen Deficiency
Z88.2 requires performing a hazard assessment that includes evaluating potential respiratory hazards including both airborne contaminants and O2-deficiency. Z88.2 augments OSHA requirements by providing detailed information to make an oxygen assessment. Per Section 7.1.1.4, where the potential for an O2-deficient atmosphere exists, the workplace oxygen shall be measured:
Wherever PO2 <159 mmHg (<20.95% at sea level), the cause of the deficiency shall be identified and understood and/or monitored if appropriate.
Wherever the workplace oxygen concentration is confirmed as PO2 <148 mmHg (<19.5% at sea level) the cause shall be determined and the location’s oxygen concentration shall be followed with ongoing monitoring. Otherwise, the atmosphere shall be assumed to be IDLH until the cause of the deficiency is understood and controlled to PO2 >122 mmHg) (>16% oxygen at sea level).
Please see Sidebar 3 (McKay, 2007), which provides additional information on how oxygen can also be displaced by other gases/vapors. It shows an example calculation.
Sidebar 3. on Oxygen Assessment.
The best way to determine oxygen concentration as a partial pressure, is by direct measurement. However, according to Roy T. McKay, Ph.D., if the contaminant concentration is known, then the oxygen concentration can be estimated when no other factors influence the ambient O2 level. When a gas or vapor displaces air, it displaces nitrogen, oxygen, and other trace gases. Since the composition of air is approximately 4/5ths nitrogen and 1/5ths oxygen, the reduction in oxygen concentration is not calculated by simple subtraction of contaminant concentration from oxygen concentration. Instead, the gas/vapor displaces approximately four times more nitrogen than oxygen. Therefore, when a single contaminant concentration is known, it is possible to estimate the reduction in oxygen concentration using the following approach.
- Step 1: Convert contaminant concentration from ppm to a percent:
Step 2: Divide “% contaminant” by 5 (this represents the fact that the gas/vapor displaces nitrogen & oxygen)
Step 3: Subtract this value from the normal oxygen concentration (20.95%)
Example: What is the approximate oxygen concentration when displaced by toluene at a measured concentration of 2,500 ppm?
Answer:
- Step 1: Convert contaminant concentration from ppm to a percent:
-
Step 2: Divide “% contaminant” by 5
0.25%/5 = 0.05%
-
Step 3: Subtract this value from the normal oxygen concentration (20.95%)
20.95% - 0.05% = 20.90% oxygen
Although this atmosphere is not oxygen deficient, the IDLH concentration for toluene (2,000 ppm) is exceeded. Note: According to a personal communication (Johnson, 2016), a rule of thumb is, assuming oxygen is displaced rather than consumed, for every 0.1% drop in O2 concentration there is 5,000 ppm of another gas or vapor present.
Z88.2 Oxygen Exposure Limits and Hazard Assessment Approach
In contrast to OSHA, the Z88.2 approach to O2-deficient hazard assessment is based on PO2 at all altitudes which corresponds to more conservative policy regarding O2-deficiency atmospheres. These differences may lead to a more conservative respirator program and respirator selection for unacclimatized individuals especially at high altitudes with their lower PO2 levels. Z88.2 is also more conservative by classifying all workplace atmospheres containing PO2 <159 mmHg (<20.95% oxygen at sea level) as O2-deficient IDLH unless the source of the oxygen reduction is understood and controlled - that is, the source of O2 reduction has been determined through a hazard evaluation and O2 levels are controlled through engineering methods (e.g. ventilation). Reduced oxygen concentrations are rarely stable and it is difficult to ensure that oxygen levels will not drop precipitously with little or no warning.
Using PO2 enables employers to assess O2-deficient atmospheres at sea level, at increased altitudes, and combinations of increased altitude combined with decreased O2 concentrations regardless of the cause. PO2 can decrease when oxygen is consumed during natural processes or displaced by other gases or vapors including water vapor. In other cases, there can be combined effects due to an increase in altitude plus displacement of oxygen. For example, Sidebar 1 illustrates the reduction in PO2 resulting from the combination of O2 displacement by 4% water vapor plus decreased atmospheric pressure at 7,000 feet (PO2 = 584 mmHg). The atmospheric PO2 changes from an O2-deficient non-IDLH atmosphere at 7,000 feet in dry air to an O2-deficient IDLH atmosphere when combined with O2 displacement by 4% water vapor. This example illustrates why respirator program administrators are encouraged to measure their actual atmospheric pressure and related parameters for the conditions faced by the respirator wearer, and not estimate it.
Unlike OSHA, Z88.2 classifies O2-deficient atmospheres as either IDLH or non-IDLH and primarily discusses O2 deficiency in terms of PO2.
Section 7.1.1.4.1 of Z88.2 defines atmospheres with PO2 less than 122 mmHg (<16% oxygen at sea level) as being O2-deficient IDLH.
Section 7.1.1.4.2 states that O2-deficient non-IDLH atmospheres have a PO2 range between greater than or equal to 122 mmHg (≥16% oxygen at sea level) and PO2 less than 148 mmHg (<19.5% oxygen at sea level).
The Z88.2 respirator selection for O2-deficient IDLH atmospheres requires either a full facepiece pressure-demand SCBA or a combination full facepiece pressure-demand SAR with auxiliary self-contained air supply.
Similar to OSHA’s exception for allowing SAR in controlled O2-deficient atmospheres, Z88.2 respirator selection for O2-deficient non-IDLH atmospheres between PO2 ≥122 mmHg and PO2 <148 mmHg (<19.5% and ≥16% oxygen at sea level) are any atmosphere-supplying respirators (e.g., SARs), only if the source of the oxygen reduction is understood and controlled (Z88.2 Section 7.2.4).
Z88.2 requires at least 23% oxygen at 10,000 feet (PO2 <523 mmHg) and 27% oxygen at 14,000 feet (PO2 <450 mmHg) to provide workers with the same amount of oxygen in the tracheal region of their lungs that is available for biological processes as they would receive by breathing ambient air at sea level.
Additionally, Z88.2 only permits the use of APRs for atmospheres with an oxygen concentration range between PO2 159 to 148 mmHg (20.95% to 19.5% oxygen at sea level) if the source of the oxygen reduction is understood and controlled and the type of other inhalation hazards and their concentrations are such that the protection provided by APRs are adequate.
Table 1 of Z88.2 provides a generalized guide of the types of respirators required to work safely in increasingly dangerous O2-deficient environments. As shown in Appendix B, Table 1 from Z88.2 combines increasingly stringent types of respirators based upon oxygen concentration, altitude, and PO2. The three columns with oxygen concentrations of 21%, <19.5 %, and <16% oxygen were chosen because 21% is ambient atmospheric oxygen concentration; <19.5% is the oxygen concentration below which OSHA and Z88.2 consider to be O2-deficient; and the <16% was chosen because that is what Z88.2 defines as O2-deficient IDLH. This table works equally well both at sea level and at increased altitude for evaluating O2-deficient atmospheres.
Although providing readers with an approximate respirator selection for increasingly dangerous levels of O2-deficient atmospheres, Table 1 of Z88.2 must be used with an understanding of all the O2-deficiency related topics explained in the standard, including Table 1 footnotes. A brief summary of some but not all of these O2-deficiency related topics is provided in Appendix C. Appendix C also includes comments and recommendations to the next Z88.2 Subcommittee concerning Z88.2 Table 1. These comments will also be helpful for program managers in understanding Table 1.
Logic behind the Z88.2 Oxygen Hazard Assessment Approach
The following discussion addresses the reasoning behind Z88.2’s approach to O2-deficient hazard assessments based on evaluating both % O2 and PO2. At high altitudes, the air still contains 21% oxygen but at a much lower total pressure and PO2. NIOSH guidance document (NIOSH, 1979) explains the importance of evaluating biologically available oxygen based on workplace PO2, which supports the Z88.2 approach for assessing O2-deficient atmospheres. The document also supports following the more conservative Z88.2 approach as compared to following OSHA’s approach by stating, “If the, oxygen deficiency level as legally defined is less than the O2 concentration you believe safe for human exposure, you must consider raising your minimum O2 level above the legal limit.” This is exactly what Z88.2 did with its approach to assessing O2-deficiency in workplace hazard assessments by requiring evaluation of not only the legally required % O2, but also requiring evaluation of the more biologically significant PO2.
Comparison of OSHA and Z88.2 Oxygen-Deficiency Policies
Z88.2 O2-deficiency policy is more conservative than OSHA’s. In their exception to the 19.5% O2 IDLH policy OSHA established three different criteria for O2-deficient non-IDLH levels, depending on altitude, from sea level to up to 14,000 feet (see our discussion on Logic behind OSHA exceptions to <19.5% O2 by volume IDLH and respirator selection requirement). In contrast, Z88.2 established atmospheric PO2 <122 mmHg (<16% O2 at sea level) as the O2-deficient IDLH cutoff point from sea level up to 14,000 feet, which is consistent with NIOSH evidence in OSHA proposed rule (OSHA 1994B), reproduced below:
“… that in the presence of an oxygen concentration of less than 16% at sea level one can experience impaired attention, thinking and coordination. At 14% or below, NIOSH states the possibility of faulty judgment, poor muscular coordination, rapid fatigue that could cause permanent heart damage, and intermittent respiration. In an IDLH or escape situation all of the described effects could place a worker in serious jeopardy. Therefore, OSHA believes that an oxygen concentration of 16% or below at sea level should require the extra precautions that go with IDLH atmospheres.”
The following lists reasons why Z88.2 policy allows SARs to be worn in atmospheres containing PO2 between <148 and ≥122 mmHg (between <19.5% and ≥16% O2 at sea level) no matter what altitude this O2-deficiency is occurring (up to 14,000 feet):
Because of the NIOSH evidence cited in OSHA proposed rule (OSHA 1994B), OSHA decided, “Therefore, OSHA believes that an oxygen concentration of 16% or below at sea level should require the extra precautions that go with IDLH atmospheres.” Therefore, Z88.2 is in agreement with OSHA that oxygen concentrations equivalent to <16% O2 at sea level do require extra precautions that go with IDLH atmospheres. However, Z88.2 set PO2 <122 mmHg (<16% O2 at sea level) as workplace O2-deficient IDLH limit not just at sea level but up to 14,000 feet.
Un-acclimated individuals working at high altitudes in atmospheres with PO2 <122 mmHg (equivalent to <16% O2 at sea level) will experience the same severe symptoms from oxygen deprivation described above by NIOSH as workers at sea level to 3,000 feet who are exposed to O2-deficient atmospheres with PO2 <122 mmHg.
Engineering controls designed to maintain oxygen deficient atmospheres below 16 % O2 are difficult at any altitude.
Employees who are not protected risk their lives if an employer mistakenly believes workplace oxygen content can be controlled.
Besides having higher O2-deficient IDLH cutoff criteria than OSHA, Z88.2 approach to hazard assessment is also more conservative, in that OSHA only assesses % O2 while the Z88.2 approach assesses both % O2 and PO2. Of the two approaches to hazard assessment, the Z88.2 approach leads to a more conservative respirator selection than OSHA. The following scenario illustrates the differences between both approaches.
Consider a workplace at the elevation of 9,000 feet in which there are no other airborne contaminants and the oxygen concentration is 20.95%.
In OSHA’s assessment, no respirator is required because the workplace oxygen concentration is not below 19.5% oxygen.
In contrast, Z88.2 assesses both % O2 and PO2. Because of the increased altitude at 9,000 feet, the atmospheric PO2 is 113 mmHg, which is below the Z88.2 O2-deficiency IDLH limit of <122 mmHg, which is equivalent to breathing the atmospheric PO2 of <16% oxygen at sea level.
The Z88.2 approach to assessing O2-deficiency requires the following respirator selection:
Individuals who are acclimatized at this altitude are not required to wear respirators up to 14,000 feet as long as the ambient oxygen content remains above 19.5% and the wearer has no medical condition that would require the use of supplemental oxygen.
-
For individuals un-acclimatized to this altitude, a full face pressure-demand SCBA or full face pressure-demand combination SAR/SCBA is required. The PLHCP will determine if the employee has any medical conditions that would preclude the use of respirators, limitations on use, or other restrictions.
Thus using Z88.2 to assess O2-deficiency, respirator selection is more conservative than OSHA because in Z88.2, the same precautions that apply for workers at sea level also apply for unacclimatized workers at high altitudes. In other words, it is Z88.2 policy to provide the same protection for un-acclimatized workers at high altitudes as is provided for workers with identical levels of PO2 at sea level.However, it is important to recognize that the “cause” for a reduction in PO2 plays an important role in health outcomes. Reduction in PO2 can result by increase in altitude, displacement of oxygen, or consumption of oxygen. With regard to displacement of oxygen, this can occur with introduction of simple asphyxiants, chemical asphyxiants, or volatile chemical contaminants. Oxygen consumption can result in O2-deficiency due to the process of oxidation such as rust. Fatalities have occurred from inert gases and chemical contaminants displacing oxygen.For example, two causes of reduced PO2 are displacement of oxygen due to the introduction of an airborne contaminant (such as a solvent) or increasing altitude without a contaminant. If both scenarios have an identical PO2 (let’s assume 113 mmHg), the health effects at sea level will likely be of far greater magnitude despite the same partial pressure of oxygen. In addition to greater magnitude, additional health effects may be present. This occurs because at sea level, (except for oxygen consumption and inert gases) the reduction in PO2 can only be caused by the introduction of airborne contaminants which contribute to health effects (at least additive). It is these contaminants that cause health outcomes to be of greater magnitude and/or effect. For example, solvents, especially at high concentrations such as those necessary to reduce oxygen concentration to levels near 16% (i.e., approximately 247,500 ppm), have significant neurotoxic properties resulting in health effects far greater than those caused by equivalent reductions in PO2 with increasing altitude. Therefore, whenever oxygen is displaced by an airborne contaminant, the resulting health effects are due to the combination of reductions in PO2 as well as the toxic properties of the airborne contaminants. If the reported symptoms are impaired attention, thinking, coordination, it is impossible to distinguish if these symptoms are caused by a low PO2 alone, toxicity of the contaminant, or a combination of the two. At sea level the concentrations necessary to achieve 16% O2, require very high concentrations of airborne contaminants. As a result, the health effects will differ in both severity and appearance. Consequently, as discussed in the following section, respirator program administrator in collaboration with the PLHCP may determine that a worker would be better served with supplemental oxygen rather than a respirator in the absence of airborne contaminants when PO2 is reduced solely by altitude.
Considerations for Alternative Solutions
Z88.2 requirements include a medical evaluation (Section 4.5.3) by a PLHCP. In collaboration with the respirator program administrator, the PLHCP will determine if the employee has any medical conditions that would preclude the use of respirators, limitations on use, or other restrictions. In some high altitude workplace scenarios, the PLHCP may determine that an un-acclimatized worker would be better served with supplemental oxygen (e.g. nasal cannula) rather than a respirator that may introduce additional safety hazards (e.g., tangling of airline hoses) and physiologic stress on the wearer. Supplemental oxygen may sometimes be provided in lieu of a respirator depending on the industrial complexity of the high altitude workplace, wearer’s medical condition, other equipment that may be worn, and the operations performed. Considering these factors, employers may decide to provide unacclimatized workers with supplemental oxygen as an alternative to a respirator when a hazard assessment determines the absence of other inhalation hazards. However, there must be no potential of flammability hazards caused by the supplemental oxygen. The decision to equip the worker with supplementary oxygen should be made by the employer and facility medical staff on a case-by-case basis. This is a choice for the employer in consultation with the PLHCP.
Providing employees supplemental oxygen is under cognizance of the medical department’s program and is not covered by the Z88.2 Standard respirator programs. The Scope (Section 1.1) of Z88.2 states, “The following are not covered by this standard:… medical inhalators and resuscitators.” Another reasonable alternative for inclusion in the respirator program is to protect un-acclimatized individuals from high altitude illness with appropriate training to recognize early warning signs of high altitude illness, avoid alcohol, drink plenty of water, etc. These actions will gradually acclimatize the worker to working at high elevations.
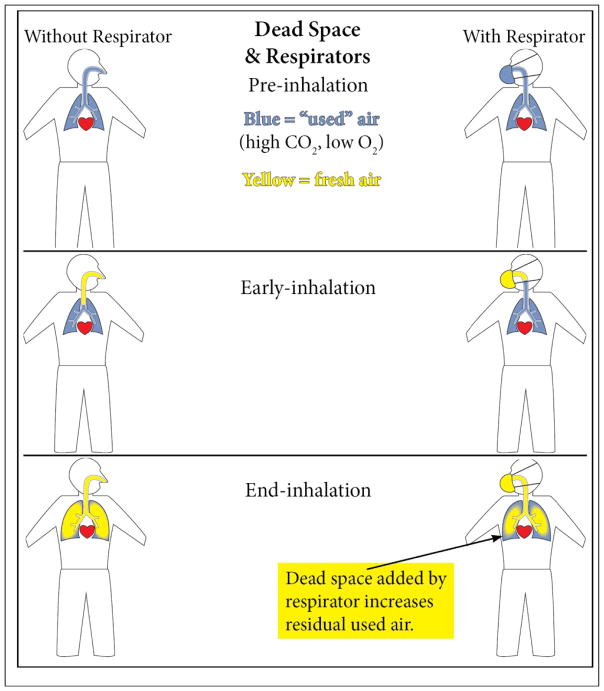
Appendix A3. Effect of Respiratory Protection on Anatomical Dead Air Space in the Lungs
Anatomical dead air space (“dead space”) is that part of the respiratory tract not involved with gas exchange: the nasal pharyngeal area, trachea and bronchi, which serve to conduct air. ANSI Z88.2 standard (2015) discusses why the increased dead air space added by wearing respirators lowers oxygen intake during inhalation:
When someone breathes in normal air at 21% oxygen, part of the oxygen is absorbed to be used by the body. However, on exhalation, the breath will at first consist of this same inspired air, since there is little oxygen/carbon dioxide exchange in the airways (trachea and bronchi)4 of the lung. As a person continues to exhale, the last portion of the breath is from the alveoli (where exchange of oxygen and carbon dioxide occur) and may contain 5% carbon dioxide and 16% oxygen. When a worker wears a respirator, a portion of the worker’s exhaled breath remains in the respirator. This exhaled air has a lowered oxygen percentage because of the oxygen removed by the lungs, and the similar amount of carbon dioxide added. Thus, on [the subsequent] inhalation, the percentage of oxygen inhaled is reduced by including this rebreathed air.
For example, if we assume the volume of the respiratory tract anatomical dead air space is about 150 cc and assume an inhalation of 500 cc air, then 150 cc remains in the anatomic dead air space, and 350 cc of fresh air reaches the alveoli (for gas exchange). When exhaling, some air stays in these air passageways and fills the anatomical dead air space. Wearing a respirator increases the total dead air space because some exhaled air is held inside the respirator, effectively increasing dead air space within the respiratory tract as illustrated in Figure A-1.
Respirator dead space varies from one respirator to another, but in general a half-mask adds about 260 cc of dead air space to the respiratory system and a full-facepiece respirator adds 815 cc dead air space. Besides reduction in oxygen, another result of increasing dead air space is the buildup of carbon dioxide (CO2), which is a respiratory stimulant. Inside the facepiece, CO2 ranges in concentration from 2% to 5%. In contrast, CO2 in normal fresh air is 0.04%.
Increasing dead air space (and, thus, “used” air) results in increased alveolar partial pressure of CO2 (PCO2) and decreases alveolar partial pressure of oxygen (PO2). Increased CO2 concentration stimulates breathing more than the lack of O2, at least initially for controlling respiration. Figure A-2 shows the location of peripheral chemoreceptor sensors. The peripheral chemoreceptor sensors in the carotid sinus of the carotid arteries supply blood to the brain. According to the NIOSH guide to industrial respiratory protection (NIOSH, 1979), the peripheral chemoreceptors detect changes in both PO2 and PCO2. However, PO2 must be reduced by about half before the carotid artery sensors send a message to the respiratory control center in the medulla to breathe harder. In contrast, an increase in PCO2 signals an increase in breathing much more quickly than the lack of oxygen.
Figure A-1. Respirator Effect on Respiratory System Dead Air Space.
Figure A-1 was created by John Muller, MD, MPH, while in official capacity as a Department Of Navy employee.
The central chemoreceptors in the medulla (Figure A-3) detect changes in cerebrospinal fluid surrounding the brain and are much more sensitive to changes in respiration than the peripheral chemoreceptors. The central chemoreceptors monitor PCO2 levels in the blood indirectly by monitoring hydrogen ion (H+) concentrations.
Figure A-2. Peripheral Chemoreceptors.
Figures A-2 and A-3 were created by John Muller, MD, MPH, while in official capacity as a Department Of Navy employee.
Figure A-3. Central Chemoreceptors.
Figures A-2 and A-3 were created by John Muller, MD, MPH, while in official capacity as a Department Of Navy employee.
Figure A-3 shows that most CO2 in the blood is in the ionized form as bicarbonate and a hydrogen ion (HCO3− plus H+). The central chemoreceptors do not measure the CO2 directly, but instead monitor the concentration of hydrogen ions associated with the ionized carbonic acid (H2CO3) in the cerebrospinal fluid. When the hydrogen ion concentration changes, the central chemoreceptor sensors send messages to the respiratory control center almost instantaneously, so proper respiration is restored immediately. Therefore, the CO2 concentration is more crucial than the lack of O2 for controlling respiration.
References
- Occupational Safety and Health Administration (OSHA) Federal Register. 5. Vol. 63. Washington, D.C: U.S. Government Printing Office, Office of the Federal Register; Jan 8, 1998. 29 CFR Parts 1910 and 1926 Respiratory Protection: Final Rule; pp. 1152–1300. http://www.gpo.gov/fdsys/pkg/FR-1998-01-08/pdf/97-33843.pdf . [Google Scholar]
- National Standards Institute (ANSI) American National Standard for Respiratory Protection (ANSI Z88.2) New York: ANSI; 2015. [Google Scholar]
- NIOSH – A Guide to Industrial Respiratory Protection, NIOSH Pub No. 76–189 (reprinted April 1979).
Appendix B. Table 1 of Z88.2-2015
Table 1.
Respirator Selection for Combined Effect of Altitude and Reduced Percentage of Oxygen for Un-Acclimatized Individuals
Acclimatized workers can continue to perform their work without atmosphere-supplying respirators, at altitudes up to 14,000 feet, as long as the ambient oxygen content remains above 19.5% and the wearer has no medical condition that would require the use of supplemental oxygen.
- Oxygen partial pressures <122 mmHg dictate the need for an SCBA or a combination airline respirator with auxiliary air cylinder and assumes a normal healthy un-acclimatized worker. Also, see Annex A.3 for other considerations in using respirators in reduced oxygen atmospheres.
- For oxygen partial pressures between 159 and 148 mmHg, air-purifying respirators may be worn if the source of the oxygen reduction is understood and controlled and the type of other inhalation hazards and their concentrations are such that the protection provided by air-purifying respirators is adequate.
- For oxygen partial pressure equal to or greater than 122 and less than 148 mmHg, airline respirators may be worn if the source of the oxygen reduction is understood and controlled.
- At 10,000 feet or higher or in any space where the total ambient pressure is less than 523 mmHg, specially designed and approved SCBA supplying enriched oxygen or a closed-circuit SCBA shall be used. At least 23% oxygen is required at 10,000 feet or at a total ambient pressure of less than 523 mmHg and 27% oxygen at 14,000 feet or at a total ambient pressure of less than 450 mmHg.
Appendix C. Selected Z88.2 O2 Deficiency Related Topics
Table 1 of Z88.2 (reproduced in Appendix B) must be used with an understanding of all the O2-deficiency related topics explained in the standard. A brief summary of some but not all of these O2 deficiency related topics is provided below:
In selecting appropriate respirators, first perform a hazard assessment including evaluating the workplace environment for potential oxygen deficiency.
All atmospheres with unknown oxygen concentrations are considered O2 deficient-IDLH.
PO2 <122 mmHg (<16% oxygen at sea level) requires either a full facepiece pressure-demand SCBA or a combination full facepiece pressure-demand SAR with auxiliary self-contained air supply.
Per Section 7.2.5.1 of Z88.2-2015, respirators worn for protection in IDLH atmospheres, including O2-deficient IDLH atmospheres shall provide a minimum flow rate of 100 liters per minute.
Also, SCBA that operate in the demand mode shall not be worn for entering IDLH atmospheres.
SCBA or a combination SAR with auxiliary air cylinders that operate in the demand mode shall not be worn for entering IDLH atmospheres. Respirators operating in demand mode are under negative pressure during inhalation, allowing for greater chance of facepiece leakage, thus they provide lower protection to the wearer. However, pressure-demand respirators are designed to maintain positive pressure during inhalation and exhalation. This may not always be true during periods with very high inspiratory peak flows and/or in combination with facepieces that do not maintain a satisfactory face to facepiece seal. Pressure-demand respirators are required for entry into IDLH atmospheres because they provide a much greater level of protection for the respirator wearer against inhalation hazards.
When combination airline/SCBA respirators are to be worn, they shall be selected to ensure that the auxiliary-cylinder capacity is sufficient for the anticipated egress from the IDLH atmosphere (Section 7.2.5.1 of Z88.2-2015).
The supplied air to the respirator must meet Grade D quality air specified in the CGA G7.1-2011 (CGA, 2011), which requires 19.5–23.5% oxygen.
Acclimatized workers can work without atmosphere-supplying respirators at altitudes up to 14,000 feet, if the ambient oxygen content remains above 19.5% and workers have no medical condition that requires using supplemental oxygen. As explained in the 1998 Preamble (OSHA, 1998), OSHA also adopts these provisions for acclimatized individuals. According to Annex A.3 of Z88.2-2015, people can live and work at high altitudes with little or no physiological ill effects because they become acclimatized to reduced PO2 levels. Acclimatized individuals’ respiratory, cardiovascular, and hematopoietic systems have adapted to less available oxygen. Complete acclimatization requires about four weeks. Therefore, acclimatized workers entering a confined space at high altitude should not be required to wear a SCBA for protection against O2-deficiency, if the PO2 inside the confined space is the same as the PO2 outside the confined space. Although not addressed in Z88.2, this same logic applies to acclimatized people not being prevented from wearing air-purifying respirators in ambient atmospheric conditions at the altitudes for which they are used to living and working as long as the ambient oxygen content remains above 19.5% oxygen and the wearer has no medical condition that would require the use of supplemental oxygen.
Comments and Recommendations to the next Z88.2 Subcommittee
This article presents a number of topics that provide an opportunity for the next Z88.2 Subcommittee to address, including the following.
Normal atmospheric PO2 should be expressed to two decimal places (20.95%) to be consistent with nearly all authoritative references. This maintains the appropriate number of significant digits for calculation purposes. This is necessary when taking into account the concentrations of other substances that may be introduced in the atmosphere. Otherwise, 500 ppm of oxygen is not accounted for.
The PO2 values for Table 1 were calculated at 1,000 feet increments. At 5,000 feet, the atmospheric pressure is ~656 mmHg with a PO2 value of 123 mmHg and at 6,000 feet, the atmospheric pressure is ~609 mmHg with a PO2 value of 119 mmHg. To be conservative the O2-deficient IDLH cutoff line was drawn at the PO2 value of 123 mmHg instead of 119 mmHg to remain above the O2-deficient IDLH cutoff point of <122 mmHg. Drawing the cutoff line for the O2-deficient IDLH just below the cutoff <122 mmHg, say at 121.9 mmHg, the atmospheric pressure would be 625 mmHg at the approximate altitude of ~5,500 feet. Remember that Table 1 includes the “average” atmospheric pressure at various altitudes above sea level. We remind program managers on a case by case basis to measure the actual atmospheric pressure and related parameters for the conditions faced by the respirator wearer.
Atmospheric pressures in Table 1 are not at expressed at room temperature conditions. Expressing atmospheric pressure at room temperature conditions would be more representative. Although direct measurement is preferred, a formula for calculating atmospheric pressures at other temperatures would be of value to some users.
Z88.2, Table 1 should actually have been entitled “Respirator Selection for Combined Effect of Altitude and Reduced Percentage of Oxygen,” leaving out the “for Un-Acclimatized Individuals” at the end of the title. Also, instead of including asterisks from 9,000 – 14,000 feet to inform readers of the footnote exempting acclimatized workers from wearing respirators, the title should have included an asterisk pointing readers to the acclimatized worker exemption footnote that applies to the entire Table 1.
Note – The top second column of the top row in Z88.2, Table 1 should be entitled “% O2” not “% O2 at Sea Level.”
Z88.2 does not address using ambient air breathing apparatus (AABA) at increasing altitudes. Consider the following discussion for addressing AABA use at higher altitudes in the next edition of Z88.2.
Z88.2 oxygen assessment is based on both percent O2 and PO2. To comply with Z88.2 policy, respirators supplied with breathing air by AABA cannot be worn at higher altitudes because although the percentage of O2 remains the same with increasing altitude, the PO2 of supplied breathing air must be between 178.6 to 148 mmHg to meet Grade D air requirements for 19.5 – 23.5% oxygen at sea level. For compliance with Z88.2, respirators supplied by AABA cannot be worn at high altitudes because they deliver ambient air, which contains the same PO2 as the higher altitude workplace air.
For example: Assume a workplace at 6,000 feet, with an atmosphere containing 20.9 % O2. The PO2 at this altitude is 127 mmHg. In this scenario, the AABA delivers 20.9 % O2, but the PO2 delivered is 127 mmHg. Breathing air with a PO2 of 127 mmHg does not comply with the PO2 equivalent to meet Grade D air requirements for 19.5 – 23.5% O2 at sea level, which is a between 178.6 to 148 mmHg.
References
- Compressed Gas Association (CGA) Commodity Specification For Air. CGA; Arlington, VA: 2011. CGA G-7.1-2004. [Google Scholar]
- Occupational Safety and Health Administration (OSHA) Federal Register. 5. Vol. 63. Washington, D.C: U.S. Government Printing Office, Office of the Federal Register; Jan 8, 1998. 29 CFR Parts 1910 and 1926 Respiratory Protection: Final Rule; pp. 1152–1300. https://www.gpo.gov/fdsys/pkg/FR-1998-01-08/pdf/97-33843.pdf . [Google Scholar]
Footnotes
The Federal Register, which includes the final rule is referenced instead of 29 CFR 1910.134 because the Federal Register includes the preamble to the OSHA Respirator Standard, which will be referred to as the 1998 Preamble.
Partial pressure (e.g., PO2), measured in millimeters of mercury (mmHg), refers to the pressure a particular gas exerts in a mixture of gases.
his appendix is modified from previous work by David L. Spelce, MS, CIH and John Muller, MD, MPH while in official capacity as Department Of Navy employees.
Note that the non-gas exchanging region is not just limited to the trachea and bronchi.
Disclaimer
The findings and conclusions in this paper are those of the authors and do not necessarily represent the views of the National Personal Protective Technology Laboratory, the Centers for Disease Control and Prevention, or the National Institute for Occupational Safety and Health. Mention of commercial products or trade names does not constitute endorsement. The authors do not have any financial interests in the present research.
References
- American National Standards Institute (ANSI) American National Standard for Respiratory Protection (ANSI Z88.2) New York: ANSI; 1980. [Google Scholar]
- Bureau of Labor Statistics. News Release, USDL-11-1247, NATIONAL CENSUS OF FATAL OCCUPATIONAL INJURIES IN 2010 (PRELIMINARY RESULTS) 2011 August 25; [Google Scholar]
- Compressed Gas Association (CGA) Commodity Specification For Air. CGA; Arlington, VA: 2011. CGA G-7.1-2004. [Google Scholar]
- National Standards Institute (ANSI) American National Standard for Respiratory Protection (ANSI Z88.2) New York: ANSI; 2015. [Google Scholar]
- NIOSH – A Guide to Industrial Respiratory Protection, NIOSH Pub No. 76–189 (reprinted April 1979).
- Bollinger Nancy., editor. NIOSH. NIOSH Respirator Selection Logic 2004. Cincinnati, OH: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, DHHS; 2004. (NIOSH Publication No. 2005-100) http://www.cdc.gov/niosh/docs/2005-100/pdfs/2005-100.pdf. [Google Scholar]
- Occupational Safety and Health Administration (OSHA) Federal Register. 5. Vol. 63. Washington, D.C: U.S. Government Printing Office, Office of the Federal Register; Jan 8, 1998. 29 CFR Parts 1910 and 1926 Respiratory Protection: Final Rule; pp. 1152–1300. http://www.gpo.gov/fdsys/pkg/FR-1998-01-08/pdf/97-33843.pdf . [Google Scholar]
- Occupational Safety and Health Administration (OSHA) 29 CFR Part 1910.146 Permit-Required Confined Spaces Federal Register. Vol. 59. Washington, D.C: U.S. Government Printing Office, Office of the Federal Register; Feb 4, 1994. pp. 55208–55209. [Google Scholar]
- Occupational Safety and Health Administration (OSHA) Federal Register 59 FR 58921: 1218-0099. Washington, D.C: U.S. Government Printing Office, Office of the Federal Register; Nov 15, 1994. 29 CFR Parts 1910 and 1926 Respiratory Protection; Proposed Rule. https://www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=FEDERAL_REGISTER&p_id=13426 . [Google Scholar]
- Personal Communication with Jim Johnson (JSJ and Associates in Pleasanton, Calif., and Chair of the ANSI/ASSE Z88 Committee)/Chris Wrenn (Vice President of Americas Sales for AEssense Corp.) of February 29, 2016
- Personal Communication with Bruce Reinert (Los Alamos National Laboratory)/David Spelce (Navy Environmental Health Center) of circa 1998.
- Respirator Selection and Development of Cartridge Change Out Schedules Course, Roy T. McKay, Ph.D. Hampton VA, of May 2007.