Abstract
Background
Treatment with fixed orthodontic appliances has been associated with significant biofilm accumulation, thus putting patients at a higher risk of oral health deterioration. The use of probiotics has been proposed to be useful in the prevention or treatment of oral pathologies such as caries and diseases of periodontal tissues. Our aim was to investigate the effects of probiotic use on inflammation of the gingival tissues and the decalcification of the enamel in patients being treated with fixed orthodontic appliances.
Methods
We searched without restrictions 8 databases and performed hand searching until September 2019. We searched for randomized controlled trials (RCTs) evaluating whether individuals with fixed orthodontic appliances benefit from probiotic treatment in terms of the inflammation of the gingivae and decalcification of the enamel. Following the selection of studies and the extraction of pertinent data, we appraised the risk of bias and the confidence in the observed effects based on established methodologies.
Results
From the final qualifying studies, three did not show any statistically significant effect on gingival inflammation after probiotic administration of up to 1 month. Similarly, non-significant differences were noted in another study regarding white spot lesions development (mean administration for 17 months). No adverse effects were reported and the level of evidence was considered moderate.
Conclusions
Supplementation of orthodontic patients with probiotics did not affect the development of inflammation in the gingivae and decalcification in the enamel. Additional RCTs, with longer intervention and follow-up periods, and involving different combinations of probiotic strains are required.
Trial registration
PROSPERO (CRD42018118008)
Keywords: Probiotics, Gingivitis, Caries, White spot lesions, Oral health, Orthodontic treatment
Background
Orthodontic procedures aim to establish a healthy, functional and appealing occlusion that is in balance with facial aesthetics [1]. However, they have been associated with particular oral hygiene challenges when fixed orthodontic appliances are involved, as these act as plaque traps and render satisfactory mechanical oral hygiene laborious [2]. The continued biofilm accumulation could lead to inflammatory changes with concomitant anaerobic shift in the oral microbiota, which can be clinically recognized as gingival bleeding [3, 4]. Moreover, whenever a cariogenic environment is favoured, enamel decalcification can also be observed [5, 6]. As these alterations in the oral environment might be detectable even after 2 years after the removal of orthodontic appliances [7, 8], meticulous oral hygiene, as well as oral health maintenance are considered paramount for the favourable outcome of orthodontic treatment [2].
Probiotics are defined as “live microorganisms which when administered in adequate amounts confer a health benefit on the host” [9]. Since the pathogenesis of caries and the diseases of the periodontal tissues has been associated with alterations in composition of oral microbiome and biofilm formation, the administration of probiotic strains has been proposed to be useful in their prevention and treatment [10–12]. In vitro studies using specific probiotic strains have demonstrated beneficial effects against oral pathogens [13–20]. However, the clinical effectiveness of administering probiotics to positively affect oral health remains undetermined. Whilst findings from an increasing number of studies supports the use of probiotic strains in the prevention or treatment of gingivitis and periodontitis, other trials have failed to show similar effects [21, 22]. Moreover, though the consumption of probiotics has been proposed as a supporting measure for caries prevention based on surrogate markers [23, 24], insufficient information on actual clinical benefits exists [21].
Objective
As treatment with fixed orthodontic appliances has been associated with significant biofilm accumulation, thus putting patients at a greater risk of developing caries and gingivitis [3–6], probiotics could be of benefit. However, studies solely targeting on the clinical effects on orthodontic patients are limited and have not, so far, been reviewed in an evidence-based manner. Our aim was to systematically assess the available evidence from randomized Clinical Trials (RCTs) on whether probiotics reduce gingival inflammation and enamel demineralization development in patients under treatment with fixed orthodontic appliances.
Methods
Protocol development and registration
For the development of the review protocol we adhered to relevant guidelines [25, 26]. The protocol was subsequently listed in the PROSPERO database (CRD42018118008). Being a systematic review, ethical approval was not required for this study.
Eligibility criteria
The criteria were formed on the PICOS basis. For the participants domain we aimed at including studies involving healthy orthodontic patients without age restrictions. Studies on subjects with syndromic or other anomalies of the craniofacial region, individuals with systematic diseases, as well as patients using antibiotics or antimicrobial agents were not considered. Study participants should receive probiotics of any type and be compared to individuals receiving placebo or no intervention at all. The outcomes considered included clinical measurements on gingival inflammation and enamel demineralization development. Plaque measurements were not considered, as reductions in plaque do not always directly reflect benefits in oral health, which is the primary goal of the intervention [27]. Person reported outcomes (preferences, experiences, quality of life, satisfaction, etc.), as well as adverse effects and economic evaluation data were also of interest. Only RCTs were eligible for inclusion. Human studies that did not evaluate clinical outcomes, animal studies, studies without control groups and reviews were excluded (Supplementary Table 1).
Information sources and search strategy
We searched without restrictions the whole content in eight databases, from the beginning of the period covered in each database and up until September 2019 (MEDLINE, CENTRAL, Cochrane Systematic Reviews, Scopus, Web of Science, Arab World Research Source, Clinical Trials registry and ProQuest Dissertations and Theses Global database). We used detailed strategies, developed and customized by one of the researchers (RHH) without placing restrictions on language (Supplementary Table 2). The list of references in the included studies were scanned and we planned to contact their corresponding authors in case we needed additional information.
Study selection of studies and data extraction
The list of records produced by the search was assessed by two researchers (RHH and EGK) independently, in a non-blinded manner, but kappa scores for the extent of agreement were not calculated, as it is not recommended [26]. The same investigators performed data extraction, using pre-designed data collection forms to record the following: duration of follow-up; individual characteristics (inclusion/exclusion criteria, number, age, gender, possible dropouts); interventions (experimental and placebo/control groups; specific details on the probiotic product used, dosage and duration of administration); details on outcomes assessed; and if available, data on patient reported outcomes, adverse effects and economic evaluation data. Finally, additional information was extracted, where possible, concerning a priori sample power analysis and the assessment of reliability. Disagreements were settled by deliberation with a third researcher (AEA).
Risk of bias in individual studies
Judgements on the risk of bias, on domain and study levels, were completed independently by EGK and ACS, using established methodology [26]. Disagreements were settled as stated previously.
Summary measures and synthesis of results
Although synthesis of the results (using either the Weighted Mean Differences or the Standardized Mean Differences) was planned according to the research protocol, it was not, in the end, carried out due to the lack of adequate data as well as differences in the retrieved studies.
Risk of bias across studies and additional analyses
For the reasons stated above, although planned, we were not able to conduct any exploratory subgroup analyses, in addition to analyses for “small-study effects” and publication bias [26]. Despite the lack of extensive information, the quality of evidence was assessed following Guyatt et al. [28] in order to adopt a structured and transparent approach in formulating an interpretation of the evidence.
Results
Study selection
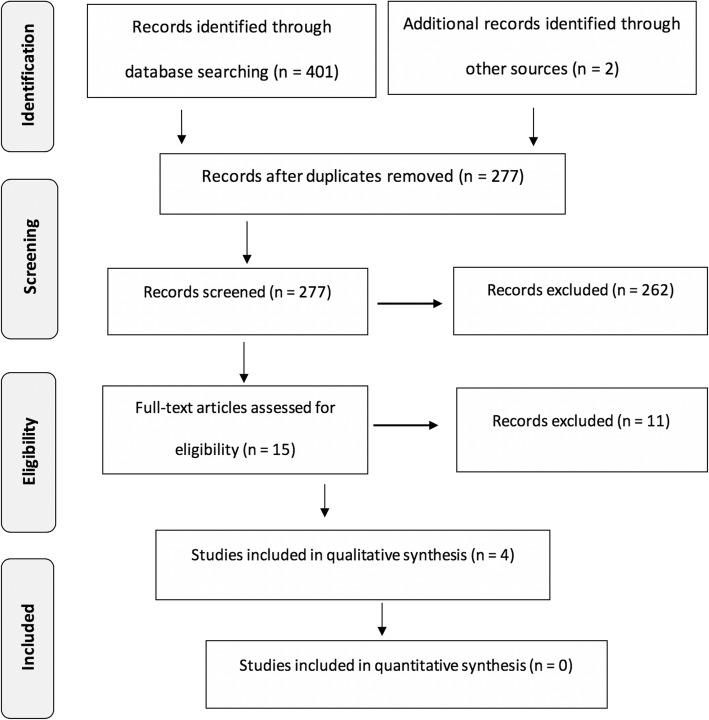
From the 403 records initially identified, we excluded 126 as duplicates, 262 based on title and/or abstract, and 11 more after reading the full paper (Fig. 1) (Supplementary Table 3). Finally, four RCTs were included [29–32] (Tables 1 and 2).
Fig. 1.
Flow diagram of the records through the reviewing process
Table 1.
General characteristics of the studies included in the systematic review
| Study | Intervention characteristics | Outcomes assessed | Others |
|---|---|---|---|
|
Benic [2019] [29] University of Otago New Zealand |
EG: Lozenges [Streptococcus salivarius M18 - 3 × 109 CFU/lozenge] PG: Lozenges without active bacteria Administration: 1 month Dosage: 2 lozenges/d |
Gingival inflammation: Gingival index [33] Adverse effects |
Sample size calculation: Yes, but not for GI Reliability of measurements: Not reported |
|
Gizani et al. [2015] [30] University of Athens Greece |
EG: Lozenges [Lactobacillus reuteri DSM 17938 and Lactobacillus reuteri ATCC PTA 5289–108 bacteria of each strain] PG: Identical lozenges without active bacteria Administration: from the time of enrollment until debonding (mean ± SD: 17.0 ± 6.8 months) Dosage: 1 lozenge/d; after brushing, before bedtime |
Enamel demineralization: Gorelick et al. [34] White Spot Lesion Index assessed photographically Adverse effects |
Sample size calculation: Yes Reliability of measurements: Yes |
|
Habib [2016] [31] University of Toronto Canada |
EG: Lozenges [Streptococcus salivarius K12, Lactobacillus paracasei, Lactobacillus plantarum, Lactobacillus acidophilus, Lactobacillus salivarius, Lactobacillus reuteri - 3 × 109 CFU/lozenge] PG: Identical lozenges without active bacteria Administration: 4 weeks Dosage: loading dose - 2 lozenges, 2 times/d (after breakfast and after dinner) for 1w; maintenance dose; 2 lozenges, 1/d (after breakfast) for next 3 w |
Gingival inflammation: Modified Gingival Index [35] Adverse effects |
Sample size calculation: Yes Reliability of measurements: Yes |
|
Kohar et al. [2015] [32] Trisakti University Indonesia |
EG1: Lozenges [Lactobacillus reuteri - 2 × 108 CFU/lozenge] EG2: Drink [Lactobacillus casei strain Shirota - 6.5 × 106/bottle] CG Administration: 2 weeks Dosage: 1 lozenge or 65 ml/d, at least 1 h after lunch |
Gingival inflammation: Papillary Bleeding Index [36] |
Sample size calculation: Not reported Reliability of measurements: Not reported |
CG Control group, without placebo administration, EG Experimental group, h hour, PG Placebo group, w week
Table 2.
Participant characteristics in included studies
| Study | Inclusion and exclusion criteria | Analyzed sample [age; gender] |
|---|---|---|
|
Benic [2019] [29] University of Otago New Zealand |
Inclusion Criteria: Presence of at least 20 natural teeth; stainless steel brackets Exclusion Criteria: Systemic disease; lingual braces; living in a non-fluoridated area; periodontal disease; taking antibiotics; using non-fluoride/antibacterial toothpaste; dental fluorosis; smoking; using powered toothbrushes; lactose intolerance; allergy to dairy products; physical inability to brush |
Age [range]: 10-30y EG: 32 [20F, 12 M] PG: 32 [21F, 11 M] Missing data: No |
|
Gizani et al. [2016] [30] University of Athens Greece |
Inclusion Criteria: Fixed appliances on at least eight maxillary front teeth (incisors, cuspids, and premolars); expected duration of Tx 7–24 m Exclusion Criteria: Individuals under treatment with systemic or local antibiotics up to two weeks before starting the study |
Age [mean ± SD]: 15.9 ± 3.9y EG: 42 (23F, 19 M); PG: 43 (33F, 10 M) Missing data: 9 [errors in follow-up photos] |
|
Habib [2016] [31] University of Toronto Canada |
Inclusion Criteria: Healthy; aged 11–18 y; mild to moderate gingivitis; fixed appliances on at least 20 teeth for at least 5 m; complete eruption of teeth #16, 21, 23, 36, 41, 43; no active caries; no use of antimicrobial mouth rinses, probiotics, antibiotics or anti-inflammatory drugs within 1 m before the trial; undergone standard orthodontic bonding procedure Exclusion Criteria: Inability to consent or communicate fluently in English; allergies or sensitivity to lozenge ingredients; immunocompromised; major underlying medical condition or ENT problems; pregnancy; smoking, alcohol consumption; oral diseases or conditions; surgery within the past 45d or the next 90d; use of antibiotics, anti-inflammatory drugs, ongoing or use of probiotics within the past 1 m; nausea, fever, vomiting, bloody diarrhea or severe abdominal pain within the past 1 m; molar bands |
Age [mean ± SD; range]: 15.69 ± 1.70y; 11-18y EG: 29 [15.75 ± 1.67y; 13F, 16 M] PG: 29 [15.64 ± 1.75y; 20F, 9 M] Missing data: 1/group [lost from to follow-up] |
|
Kohar et al. (2015) [32] Trisakti University Indonesia |
Inclusion Criteria: Healthy; no medication; aged 18-25y; fixed appliances Tx for at least 1y Exclusion Criteria: Using xylitol gums, mouthwashes, systemic antibiotics; smokers; pregnancy; topical fluoride treatment |
Age [range]: 18-25y EG1: 10; EG2: 10; CG: 10 Missing data: No |
CG Control group, without placebo administration, d days, EG Experimental group, F Females, M Males, m months, PG Placebo group, Tx treatment, y years
Study characteristics
Gizani et al. [30] assessed the development of white spot lesions (WSL) on photographs [34], following the administration of lozenges containing Lactobacillus reuteri for a mean period of 17 months. The other three studies investigated gingival inflammation for a maximum period of 1 month, using gingivitis [33, 35] or bleeding [36] indices. The interventions assessed were lozenges with Streptococcus salivarius M18 only [29], lozenges containing Streptococcus salivarius K12, Lactobacillus paracasei, Lactobacillus plantarum, Lactobacillus acidophilus and Lactobacillus reuteri [31], as well as lozenges with Lactobacillus reuteri and a drink with Lactobacillus casei strain Shirota [32]. Three of the retrieved studies assessed adverse effects [29–31].
Risk of bias within studies
Three studies were assessed to be of low risk of bias [29–31], while for Kohar et al. [32] most domains were considered to be of unclear risk (Table 3).
Table 3.
Summary of the risk of bias assessment
| Domain | Benic [2019] [29] | Gizani et al. [2016] [30] | Habib [2016] [31] | Kohar et al. [2015] [32] |
|---|---|---|---|---|
| 1 | Low | Low | Low | Unclear |
| 2 | Low | Unclear | Unclear | Unclear |
| 3 | Low | Low | Low | Unclear |
| 4 | Low | Low | Low | Unclear |
| 5 | Low | Low | Low | Low |
| 6 | Low | Low | Low | Low |
| 7 | Unclear | Unclear | Unclear | Unclear |
| Summary | Low | Low | Low | Unclear |
Domains examined: 1: Random sequence generation 2: Allocation concealment, 3: Blinding of participants and personnel, 4: Blinding of outcome assessment, 5: Incomplete outcome data, 6: Selective outcome reporting, 7: Other potential threats to validity
Results of individual studies
No statistically significant benefit was noted regarding the presence (p = 0.515) or the mean number of new WSL (p = 0.423) [30]. In addition, no statistically significant effect was demonstrated in terms of gingival inflammation (Benic et al. [29], p = 0.867; Habib [31], p = 0.797; Kohar et al. [32], p = 0.053). Finally, no adverse effects were noted in any of the included studies. Gizani et al. [30] reported that 8 participants could not tolerate the taste of the lozenges. Habib [31] reported one participant with gastrointestinal pain and diarrhoea, but it was later shown that this individual had received the placebo.
Risk of bias across studies and additional analyses
Overall, the confidence in the obtained information was moderate (Table 4).
Table 4.
Quality of available evidence
| Quality assessment | No of patients | Effect | Quality | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Studies | Risk of bias | Inconsistency | Indirectness | Imprecision | Other | Probiotic | Control | ||
| Enamel demineralization development | |||||||||
| 1 | Not serious | Not serious | Not serious | Serious1 | None | 42 | 43 | No difference |
⨁⨁⨁◯ Moderate |
| Gingival inflammation development | |||||||||
| 3 | Not serious | Not serious | Not serious | Seriousa | None | 81 | 71 | No difference |
⨁⨁⨁◯ Moderate |
aThe number of patients analyzed was limited
Discussion
Summary of available evidence
Patients undergoing orthodontic treatment are linked with significant biofilm accumulation, thus exposing them to a greater risk of caries and gingivitis [37–39]. Nowadays, several reports have investigated probiotic effects in enhancing oral health in the general dental population, but, up to date, their effectiveness remains inconclusive [21, 22]. To the best of our knowledge, studies solely focusing on the clinical effects on orthodontic patients have not been previously summarized in an evidence-based manner.
Based on the data presented in this systematic review, there is a moderate level of evidence that administering probiotics to orthodontic patients does not have an effect on WSL development and gingival inflammation in the short-term, while no marked adverse effects were noted. Studies investigating the effect of probiotics on various microbiological parameters during orthodontic treatment have been conflicting [29–31, 40–43]. The lack of significant results presented in the current systematic review might be attributed to various causes, including the use of inappropriate and ineffective bacterial strains, ineffective concentrations of bacteria and administration protocols, ineffectiveness of the selected probiotic strain to colonize effectively the oral environment or strain inability to compete with the bacteria and biofilm accumulation present in the oral cavity [31].
Up to the present time, no consensus has been reached about which bacterial strain is most appropriate and effective. Some reports have found that the Lactobacillus species have shown positive effects in the treatment of periodontal diseases, including Lactobacillus reuteri strains [44–46], Lactobacillus paracasei [47], Lactobacillus salivarius, Lactobacillus plantarum and Lactobacillus rhamnosus [48–52]. Lactobacillus brevis has also been suggested to be potentially beneficial in view of its anti-inflammatory characteristics [53]. Bifidobacterium is another species that has been found to exert a positive impact on periodontal disease [54]. Based on our knowledge, the Streptococcus salivarius K12 strain used by Habib [31], has not been previously assessed for its effect in treating gingivitis, but has been tested for changes in oral malodor parameters [55]. The Streptococcus salivarius M18 strain has been mainly tested for anti-caries activity [56, 57].
In principle, multi-strain probiotics products could possess synergistic and symbiotic properties because of the interactions of each strain with the others. However, some very limited data suggests that probiotic strains may also exhibit inhibitory properties against each other. For instance, hydrogen peroxide and bacteriocin production may induce the desired effect when inhibiting endogenous strains such as Streptococcus mutans, while, simultaneously they might also disable other probiotic strains in the same formulation, thus reducing its effectiveness [58].
The concentrations required for producing the desired outcomes from oral probiotic formulations have not been widely investigated. It is critical to be certain about the exact dose required to initiate a dose-response reaction during the administration of probiotics. In the field of medicine, the industry standard for the counts of viable bacteria should range from 1 × 106 to 1 × 109 CFU [59]. However, when using oral probiotics, it is logical to assume that a lower dose or concentration would be required, since it does not have to pass through the gastrointestinal system. The vast majority of probiotic studies evaluating various oral health parameters have used concentrations of 1 × 108 CFU. Moreover, it is important to remember that each individual strain has a different potential for oral colonization [31]. All doses should be selected according to the specific strain used.
In addition, the administration method and duration may also modulate the effect of a probiotic product. Various vehicles for oral probiotics have been employed, including gums, lozenges, tablets, drops and drinks [21, 22]. It has been suggested, for example, that the use of vehicles derived from milk that contain calcium, could potentially increase the anti-cariogenic effect [24]. Milk derived products produce also ammonia that helps increase pH and delay biofilm formation, by preventing bacterial adhesion on teeth [60].
Furthermore, effective probiotic activity, necessitates first adherence and subsequently colonization of oral surfaces [40]. These processes could be compromised in the case of a mature biofilm that is difficult to penetrate, or in the existence of an oral pH that is not compatible with bacterial viability [47]. Moreover, the capacity of a probiotic strain to colonize might vary between members of the same species, as it has been demonstrated for Lactobacilli [61–63]. Finally, there is the possibility that the administered strains are unable to compete with the quantity of the bacteria and plaque accumulation present in the oral cavity, as is possibly the case with orthodontic patients [47]. In such cases, higher concentrations of probiotics or administration for a longer duration may be required to demonstrate any potential for clinical improvements [31]. Supplementation with probiotics for periods 1 month or less as reported in the located studies may not be sufficient for the strain to colonize and establish a stable microbiome. Recently, alterations in bacterial composition was only detected after 6 weeks administration of an oral probiotic preparation [64].
Apart from factors associated with specific probiotic characteristics or the mode/duration of administration, patients’ compliance could also affect results. Although in the retrieved studies compliance was found to vary from good to excellent, these assessments were based on patient self-report [29–31]. Finally, the diet of participants during the interventions, the potential use of antibacterial or antiseptic products, changes in brushing/flossing technique and swallowing or chewing the lozenge rather than sucking it, thereby washing-out the probiotic from the mouth, could have affected the reported changes [31, 40].
Strengths and limitations
For this review we adhered to well-established guidelines and focused on RCTs that provide the highest level of evidence in health care interventions. As far as we can know there has been no other review conducted on the possible effectiveness of probiotics on different clinical parameters in orthodontic patients. The search was extensive, comprehensive, without restrictions and every effort to reduce methodological bias was made.
The characteristics of the information located gave rise to some limitations as well. Due to insufficient information, it was not possible to conduct analyses for ‘small study effects’, publication bias or subgroup analyses. Moreover, the small number of individuals analysed could pose a threat to the precision of the results. Finally, the short duration of most interventions may confound the results and the use of specific strains, concentrations, dose regimens or modes of administration, might diminish the generalizability of the retrieved information.
Recommendations for future research
The use of probiotics has been widely accepted by the general population by virtue of their natural source, as well as their beneficial effects on conditions pertaining to oral and overall health, like caries, periodontal tissues, nutrition status, immune response and respiratory infections [65–71]. However, further research is needed, in order to optimize probiotic use and quantify the extent of clinical benefit. In order to take full advantage of using oral probiotics, a more complete understanding regarding their mechanism of action in the area of adhesion and colonization and the capabilities of the different strains is required. Although nowadays, more and more research focus on the use of probiotics, the literature is still unable to reach a consensus on the optimum duration required or the ideal concentration or dose regimen and mode of administration for each probiotic strain. It is essential to understand the efficacy of each strain when used alone, as well as to evaluate the potential synergistic effects of combining probiotic strains into a single entity.
As orthodontic patients require continuous and rigorous oral hygiene control, caries prevention and maintenance of gingival health, more high-quality studies, involving different combinations of probiotic strains and of longer durations of intervention and follow-up are warranted. Moreover, instead of testing the use of probiotics to combat established gingivitis, research could be conducted on the possibility of preventing gingivitis using probiotics prior to the bonding of orthodontic brackets. Although much is known about probiotics in the gastrointestinal field, there is a great deal of knowledge to be learned pertaining to probiotics for oral health.
Conclusions
Overall, probiotic administration does not seem to have an effect on the gingival inflammation and enamel decalcification development in patients under treatment with fixed orthodontic appliances. Further RCTs with particular focus on controlling the various possible sources of data, involving different combinations of probiotic strains and of longer duration of intervention and follow-up are required.
Supplementary information
Additional file 1: Table S1. Eligibility criteria for the present systematic review. Table S2. Strategy for database search (up to September 1st, 2019). Table S3. Studies excluded during full-text eligibility assessment with reasons.
Acknowledgements
Not applicable.
Abbreviations
- PICOS
Participants, Interventions, Comparisons, Study design
- PROSPERO
International prospective register of systematic reviews
- RCT
Randomized controlled clinical trials
- WSL
white spot lesions
Authors’ contributions
EGK conceived the study and initiated the study design. RHH, EGK, ACS, AEA, contributed to data collection, data analysis, data interpretation and manuscript draft preparation. All authors approved the final manuscript. Data.
Funding
None.
Availability of data and materials
All data and materials are available upon request.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
Nothing to declare.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information accompanies this paper at 10.1186/s12903-020-01109-3.
References
- 1.Proffit WR, Fields HW, Larson BE, Sarver DM. Malocclusion and dentofacial deformity in contemporary society. In: Proffit WR, Fields HW, Larson BE, Sarver DM, editors. Contemporary orthodontics. 6. Philadelphia: Elsevier; 2019. pp. 2–17. [Google Scholar]
- 2.Heintze SD, Jost-Brinkman P, Finke C, Miethke R-R. Effects of orthodontic treatment on oral health. In: Heintze SD, Jost-Brinkman P, Finke C, Miethke R-R, editors. Oral health for the orthodontic patient. Quintessence Publishing: Berlin; 1999. pp. 1–25. [Google Scholar]
- 3.Davis SM, Plonka AB, Fulks BA, Taylor KL, Bashuktski J. Consequences of orthodontic treatment on periodontal health: clinical and microbial effects. Semin Orthod. 2014;20:139–149. doi: 10.1053/j.sodo.2014.06.002. [DOI] [Google Scholar]
- 4.Justus R. Prevention of periodontal deterioration/damage. In: Justus R, editor. Iatrogenic effects of orthodontic treatment: decision-making in prevention, diagnosis, and treatment. Springer: Heidelberg; 2015. pp. 37–84. [Google Scholar]
- 5.Øgaard B, Bishara SE, Duschner H. Enamel effects during bonding-debonding and treatment with fixed appliances. In: Graber TM, Eliades T, Athanasiou AE, editors. Risk management in orthodontics: experts’ guide to malpractice. Quintessence: Berlin; 2004. pp. 19–46. [Google Scholar]
- 6.Ata-Ali F, Ata-Ali J, Ferrer-Molina M, Cobo T, De Carlos F, Cobo J. Adverse effects of lingual and buccal orthodontic techniques: a systematic review and meta-analysis. Am J Orthod Dentofac Orthop. 2016;149:820–829. doi: 10.1016/j.ajodo.2015.11.031. [DOI] [PubMed] [Google Scholar]
- 7.Ghijselings E, Coucke W, Verdonck A, Teughels W, Quirynen M, Pauwels M, Carels C, van Gastel J. Long-term changes in microbiology and clinical periodontal variables after completion of fixed orthodontic appliances. Orthod Craniofacial Res. 2014;17:49–59. doi: 10.1111/ocr.12031. [DOI] [PubMed] [Google Scholar]
- 8.Kim K, Jung W-S, Cho S, Ahn SJ. Changes in salivary periodontal pathogens after orthodontic treatment: an in vivo prospective study. Angle Orthod. 2016;86:998–1003. doi: 10.2319/070615-450.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.World Health Organization / Food and Agriculture Organization of the United Nations . Probiotics in food. Health and nutritional properties and guidelines for evaluation. Rome: World Health Organization / Food and Agriculture Organization of the United Nations; 2006. [Google Scholar]
- 10.Meurman JH, Stamatova I. Probiotics: contributions to oral health. Oral Dis. 2007;13:443–451. doi: 10.1111/j.1601-0825.2007.01386.x. [DOI] [PubMed] [Google Scholar]
- 11.Meurman JH. Probiotics: do they have a role in oral medicine and dentistry? Eur J Oral Sci. 2005;113:188–196. doi: 10.1111/j.1600-0722.2005.00191.x. [DOI] [PubMed] [Google Scholar]
- 12.Twetman S, Keller MK. Probiotics for caries prevention and control. Adv Dent Res. 2012;24:98–102. doi: 10.1177/0022034512449465. [DOI] [PubMed] [Google Scholar]
- 13.Dierksen KP, Moore CJ, Inglis M, Wescombe PA, Tagg JR. The effect of ingestion of milk supplemented with salivaricin a-producing Streptococcus salivarius on the bacteriocin-like inhibitory activity of streptococcal populations on the tongue. FEMS Microbiol Ecol. 2007;59:584–591. doi: 10.1111/j.1574-6941.2006.00228.x. [DOI] [PubMed] [Google Scholar]
- 14.Hedberg M, Hasslöf P, Sjöström I, Twetman S, Stecksén-Blicks C. Sugar fermentation in probiotic bacteria - an in vitro study. Oral Microbiol Immunol. 2008;23:482–485. doi: 10.1111/j.1399-302X.2008.00457.x. [DOI] [PubMed] [Google Scholar]
- 15.Haukioja A, Söderling E, Tenovuo J. Acid production from sugars and sugar alcohols by probiotic lactobacilli and bifidobacteria in vitro. Caries Res. 2008;42:449–453. doi: 10.1159/000163020. [DOI] [PubMed] [Google Scholar]
- 16.Twetman L, Larsen U, Fiehn NE, Stecksén-Blicks C, Twetman S. Coaggregation between probiotic bacteria and caries-associated strains: An in vitro study. Acta Odontol Scand. 2009;67:284–288. doi: 10.1080/00016350902984237. [DOI] [PubMed] [Google Scholar]
- 17.Chuang LC, Huang CS, Ou-Yang LW, Lion SY. Probiotic Lactobacillus paracasei effect on cariogenic bacterial flora. Clin Oral Investig. 2001;15:471–476. doi: 10.1007/s00784-010-0423-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lee DK, Park SY, An HM, Kim JR, Kim MJ, Lee SW, Cha MK, Kim SA, Chung MJ, Lee KO, Ha NJ. Antimicrobial activity of Bifidobacterium spp. isolated from healthy adult Koreans against cariogenic microflora. Arch Oral Biol. 2011;56:1047–1054. doi: 10.1016/j.archoralbio.2011.03.002. [DOI] [PubMed] [Google Scholar]
- 19.Lee H, Kim J. A comparative study of the effect of probiotics on cariogenic biofilm model for preventing dental caries. Arch Microbiol. 2014;196:601–609. doi: 10.1007/s00203-014-0998-7. [DOI] [PubMed] [Google Scholar]
- 20.Han N, Jia L, Su Y, Du J, Guo L, Luo Z, Liu Y. Lactobacillus reuteri extracts promoted wound healing via PI3K/AKT/β-catenin/TGFβ1 pathway. Stem Cell Res Ther. 2019;10:243. doi: 10.1186/s13287-019-1324-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gruner D, Paris S, Schwendicke F. Probiotics for managing caries and periodontitis: systematic review and meta-analysis. J Dent. 2016;48:16–25. doi: 10.1016/j.jdent.2016.03.002. [DOI] [PubMed] [Google Scholar]
- 22.Jayaram P, Chatterjee A, Raghunathan V. Probiotics in the treatment of periodontal disease: a systematic review. J Indian Soc Periodontol. 2016;20:488–495. doi: 10.4103/0972-124X.207053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Laleman I, Detailleur V, Slot DE, Slomka V, Quirynen M, Teughels W. Probiotics reduce mutans streptococci counts in humans: a systematic review and meta-analysis. Clin Oral Investig. 2014;18:1539–1552. doi: 10.1007/s00784-014-1228-z. [DOI] [PubMed] [Google Scholar]
- 24.Nadelman P, Magno MB, Masterson D, da Cruz AG, Maia LC. Are dairy products containing probiotics beneficial for oral health? A systematic review and meta-analysis. Clin Oral Investig. 2018;22:2763–2785. doi: 10.1007/s00784-018-2682-9. [DOI] [PubMed] [Google Scholar]
- 25.Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA group Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol. 2009;62:1006–1012. doi: 10.1016/j.jclinepi.2009.06.005. [DOI] [PubMed] [Google Scholar]
- 26.Higgins JPT, Green S. Cochrane handbook for systematic reviews of interventions version 5.1.0. [Updated March 2011] 2011. [Google Scholar]
- 27.American Dental Association . Acceptance program guidelines: adjunctive dental therapies for the reduction of plaque and gingivitis. Chicago: American Dental Association, Council on Scientific Affairs; 2011. [Google Scholar]
- 28.Guyatt GH, Oxman AD, Schünemann HJ, Tugwell P, Knottnerus A. GRADE guidelines: a new series of articles in the the journal of clinical epidemiology. J Clin Epidemiol. 2011;64:380–382. doi: 10.1016/j.jclinepi.2010.09.011. [DOI] [PubMed] [Google Scholar]
- 29.Benic GZ, Farella M, Morgan XC, Viswam J, Heng NC, Cannon RD, Mei L. Oral probiotics reduce halitosis in patients wearing orthodontic braces: a randomized, triple-blind, placebo-controlled trial. J Breath Res. 2019;13:036010. doi: 10.1088/1752-7163/ab1c81. [DOI] [PubMed] [Google Scholar]
- 30.Gizani S, Petsi S, Twetman S, Caroni C, Makou M, Papagianoulis L. Effect of the probiotic bacterium Lactobacillus reuteri on white spot lesion development in orthodontic patients. Eur J Orthod. 2016;38:85–89. doi: 10.1093/ejo/cjv015. [DOI] [PubMed] [Google Scholar]
- 31.Habib S. Assessment of the therapeutic potential of a dental probiotic in orthodontic patients affected by gingivitis: a randomized controlled trial. Toronto: Thesis. University of Toronto; 2016. [Google Scholar]
- 32.Kohar M, Emmanuel V, Astuti L. Comparison between probiotic lozenges and drinks towards periodontal status improvement of orthodontic patients. Dent J (Majalah Kedokteran Gigi) 2015;48:126–129. doi: 10.20473/j.djmkg.v48.i3.p126-129. [DOI] [Google Scholar]
- 33.Löe H, Silness J. Periodontal disease in pregnancy. I. Prevalence and severity. Acta Odontol Scand. 1963;21:533–551. doi: 10.3109/00016356309011240. [DOI] [PubMed] [Google Scholar]
- 34.Gorelick L, Geiger AM, Gwinnett AJ. Incidence of white spot formation after bonding and banding. Am J Orthod Dentofac Orthop. 1982;81:93–98. doi: 10.1016/0002-9416(82)90032-X. [DOI] [PubMed] [Google Scholar]
- 35.Lobene R, Weatherford T, Ross N, Lamm R, Menaker L. A modified gingival index for use in clinical trials. Clin Prev Dent. 1986;8:3–6. [PubMed] [Google Scholar]
- 36.Saxer UP, Mühlemann HR. Motivation and education. SSO Schweiz Monatsschr Zahnheilkd. 1975;85:905–919. [PubMed] [Google Scholar]
- 37.Al-Jewair T, Suri S, Tompson B. Predictors of adolescent compliance with oral hygiene instructions during two-arch multi bracket fixed orthodontic treatment. Angle Orthod. 2011;81:525–531. doi: 10.2319/092010-547.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Al Makhmari SA, Kaklamanos EG, Athanasiou AE. Short-term and long-term effectiveness of powered toothbrushes in promoting periodontal health during orthodontic treatment: a systematic review and meta-analysis. Am J Orthod Dentofac Orthop. 2017;152:753–766. doi: 10.1016/j.ajodo.2017.09.003. [DOI] [PubMed] [Google Scholar]
- 39.Cozzani M, Ragazzini G, Delucchi A, Mutinelli S, Barreca C, Rinchuse DJ, Servetto R, Piras V. Oral hygiene compliance in orthodontic patients: a randomized controlled study on the effects of a post-treatment communication. Prog Orthod. 2016;17:41. doi: 10.1186/s40510-016-0154-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jivraj FE. Effectiveness of the Lorodent probiotic lozenge in reducing plaque and S. mutans levels in orthodontic patients: A randomized, double-blind, placebo controlled trial. Toronto: Thesis. University of Toronto; 2015. [Google Scholar]
- 41.Jose JE, Padmanabhan S, Chitharanjan AB. Systemic consumption of probiotic curd and use of probiotic toothpaste to reduce Streptococcus mutans in plaque around orthodontic brackets. Am J Orthod Dentofac Orthop. 2013;144:67–72. doi: 10.1016/j.ajodo.2013.02.023. [DOI] [PubMed] [Google Scholar]
- 42.Alp S, Baka ZM. Effects of probiotics on salivary Streptecoccus mutans and Lactobacillus levels in orthodontic patients. Am J Orthod Dentofac Orthop. 2018;154:517–523. doi: 10.1016/j.ajodo.2018.01.010. [DOI] [PubMed] [Google Scholar]
- 43.Pinto GS, Cenci MS, Azevedo MS, Epifanio M, Jones MH. Effect of yogurt containing Bifidobacterium animalis subsp. lactis DN-173010 probiotic on dental plaque and saliva in orthodontic patients. Caries Res. 2014;48:63–68. doi: 10.1159/000353467. [DOI] [PubMed] [Google Scholar]
- 44.Twetman S, Derawi B, Keller M, Ekstrand K, Yucel-Lindberg T, Stecksén-Blicks C. Short-term effect of chewing gums containing probiotic Lactobacillus reuteri on the levels of inflammatory mediators in gingival crevicular fluid. Acta Odontol Scand. 2009;67:19–24. doi: 10.1080/00016350802516170. [DOI] [PubMed] [Google Scholar]
- 45.Vivekananda MR, Vandana KL, Bhat KG. Effect of the probiotic lactobacilli reuteri (Prodentis) in the management of periodontal disease: a preliminary randomized clinical trial. J Oral Microbiol. 2010;2:105. doi: 10.3402/jom.v2i0.5344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Vicario M, Santos A, Violant D, Nart J, Giner L. Clinical changes in periodontal subjects with the probiotic Lactobacillus reuteri Prodentis: a preliminary randomized clinical trial. Acta Odontol Scand. 2013;71:813–819. doi: 10.3109/00016357.2012.734404. [DOI] [PubMed] [Google Scholar]
- 47.Sookkhee S, Chulasiri M, Prachyabrued W. Lactic acid bacteria from healthy oral cavity of Thai volunteers: inhibition of oral pathogens. J Appl Microbiol. 2001;90:172–179. doi: 10.1046/j.1365-2672.2001.01229.x. [DOI] [PubMed] [Google Scholar]
- 48.Lozo J, Vukasinovic M, Strahinic I, Topisirovic A. Characterization and antimicrobial activity of bacteriocin 217 produced by natural isolate Lactobacillus paracasei subsp. paracasei BGBUK2-16. J Food Prot. 2004;67:2727–2734. doi: 10.4315/0362-028X-67.12.2727. [DOI] [PubMed] [Google Scholar]
- 49.Shimauchi H, Mayanagi G, Nakaya S, Minamibuchi M, Ito Y, Yamaki K, Hirata H. Improvement of periodontal condition by probiotics with Lactobacillus salivarius WB21: a randomized, double-blind, placebo-controlled study. J Clin Periodontol. 2008;35:897–905. doi: 10.1111/j.1600-051X.2008.01306.x. [DOI] [PubMed] [Google Scholar]
- 50.Simark-Mattsson C, Emilson CG, Håkansson EG, Jacobsson C, Roos K, Holm S. Lactobacillus-mediated interference of mutans streptococci in caries-free vs. caries active subjects. Eur J Oral Sci. 2007;115:308–314. doi: 10.1111/j.1600-0722.2007.00458.x. [DOI] [PubMed] [Google Scholar]
- 51.Mayanagi G, Kimura M, Nakaya S, Hirata H, Sakamoto M, Benno Y, Shimauchi H. Probiotic effects of orally administered Lactobacillus salivarius WB21-containing tablets on periodontopathic bacteria: a double-blinded, placebo controlled randomized clinical trial. J Clin Periodontol. 2009;36:506–513. doi: 10.1111/j.1600-051X.2009.01392.x. [DOI] [PubMed] [Google Scholar]
- 52.Ahmed S, Hoon J, Mun H, Yang C. Evaluation of Lactobacillus and bacillus-based probiotics as alternatives to antibiotics in enteric microbial challenged weaned piglets. Afr J Microbiol Res. 2014;8:96–104. doi: 10.5897/AJMR2013.6355. [DOI] [Google Scholar]
- 53.Riccia DN, Bizzini F, Perilli MG, Polimeni A, Trinchieri V, Amicosante G, Gifone MG. Anti-inflammatory effects of Lactobacillus brevis (CD2) on periodontal disease. Oral Dis. 2007;13:376–385. doi: 10.1111/j.1601-0825.2006.01291.x. [DOI] [PubMed] [Google Scholar]
- 54.Hojo K, Mizoguchi C, Taketomo N, Ohshima T, Gomi K, Arai T, Maeda N. Distribution of salivary Lactobacillus and Bifidobacterium species in periodontal health and disease. Biosci Biotechnol Biochem. 2007;71:152–157. doi: 10.1271/bbb.60420. [DOI] [PubMed] [Google Scholar]
- 55.Burton JP, Chilcott CN, Moore CJ, Speiser G, Tagg JR. A preliminary study of the effect of probiotic Streptococcus salivarius K12 on oral malodour parameters. J Appl Microbiol. 2006;100:754–764. doi: 10.1111/j.1365-2672.2006.02837.x. [DOI] [PubMed] [Google Scholar]
- 56.Burton JP, Drummond BK, Chilcott CN, Tagg JR, Thomson WM, Hale JD, Wescombe PA. Influence of the probiotic Streptococcus salivarius strain M18 on indices of dental health in children: a randomized double-blind, placebo-controlled trial. J Med Microbiol. 2013;62:875–884. doi: 10.1099/jmm.0.056663-0. [DOI] [PubMed] [Google Scholar]
- 57.Di Pierro F, Zanvit A, Nobili P, Risso P, Fornaini C. Cariogram outcome after 90 days of oral treatment with Streptococcus salivarius M18 in children at high risk for dental caries: results of a randomized, controlled study. Clin Cosmet Investig Dent. 2015;7:107–113. doi: 10.2147/CCIDE.S93066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kailasapathy K. Chin JSurvival and therapeutic potential of probiotic organisms with reference to Lactobacillus acidophilus and Bifidobacterium spp. Immunol Cell Biol. 2000;78:80–88. doi: 10.1046/j.1440-1711.2000.00886.x. [DOI] [PubMed] [Google Scholar]
- 59.Imran F, Das S, Padmanabhan S, Rao R, Suresh A, Bharath D. Evaluation of the efficacy of a probiotic drink containing Lactobacillus casei on the levels of periodontopathic bacteria in periodontitis: a clinico-microbiologic study. Indian J Dent Res. 2015;26:462–468. doi: 10.4103/0970-9290.172033. [DOI] [PubMed] [Google Scholar]
- 60.Cochrane N, Cai F, Huq N, Burrow M, Reynolds E. New approaches to enhanced remineralization of tooth enamel. J Dent Res. 2010;89:1187–1197. doi: 10.1177/0022034510376046. [DOI] [PubMed] [Google Scholar]
- 61.Caglar E, Cildir K, Ergeneli S, Sandalli N, Twetman S. Salivary mutans streptococci and lactobacilli levels after ingestion of the probiotic bacterium Lactobacillus reuteri ATCC 55730 by straws or tablets. Acta Odontol Scand. 2006;64:314–318. doi: 10.1080/00016350600801709. [DOI] [PubMed] [Google Scholar]
- 62.Krasse P, Carlsson B, Dahl C, Paulsson A, Nilsson A, Sinkiewicz G. Decreased gum bleeding and reduced gingivitis by the probiotic Lactobacillus reuteri. Swed Dent J. 2006;30:55–60. [PubMed] [Google Scholar]
- 63.Yli-Knuuttila H, Snäll J, Kari K, Meurman H. Colonization of Lactobacillus rhamnosus GG in the oral cavity. Oral Microbiol Immunol. 2006;21:129–131. doi: 10.1111/j.1399-302X.2006.00258.x. [DOI] [PubMed] [Google Scholar]
- 64.Montero E, Iniesta M, Rodrigo M, Marín MJ, Figuero E, Herrera D, Sanz M. Clinical and microbiological effects of the adjunctive use of probiotics in the treatment of gingivitis: a randomized controlled clinical trial. J Clin Periodontol. 2017;44:708–716. doi: 10.1111/jcpe.12752. [DOI] [PubMed] [Google Scholar]
- 65.Inchingolo F, Dipalma G, Cirulli N, Cantore S, Saini RS, Altini V, Santacroce L, Ballini A, Saini R. Microbiological results of improvement in periodontal condition by administration of oral probiotics. J Biol Regul Homeost Agents. 2018;32:1323–1328. [PubMed] [Google Scholar]
- 66.Cantore S, Ballini A, De Vito D, Abbinante A, Altini V, Dipalma G, Inchingolo F, Saini R. Clinical results of improvement in periodontal condition by administration of oral probiotics. J Biol Regul Homeost Agents. 2018;32:1329–1334. [PubMed] [Google Scholar]
- 67.Ballini A, Gnoni A, De Vito D, Dipalma G, Cantore S, Gargiulo Isacco C, Saini R, Santacroce L, Topi S, Scarano A, Scacco S, Inchingolo F. Effect of probiotics on the occurrence of nutrition absorption capacities in healthy children: a randomized double-blinded placebo-controlled pilot study. Eur Rev Med Pharmacol Sci. 2019;23:8645–8657. doi: 10.26355/eurrev_201910_19182. [DOI] [PubMed] [Google Scholar]
- 68.Inchingolo F, Santacroce L, Cantore S, Ballini A, Del Prete R, Topi S, Saini R, Dipalma G, Arrigoni R. Probiotics and EpiCor® in human health. J Biol Regul Homeost Agents. 2019;33:1973–1979. doi: 10.23812/19-543-L. [DOI] [PubMed] [Google Scholar]
- 69.Campanella V, Syed J, Santacroce L, Saini R, Ballini A, Inchingolo F. Oral probiotics influence oral and respiratory tract infections in pediatric population: a randomized double-blinded placebo-controlled pilot study. Eur Rev Med Pharmacol Sci. 2018;22:8034–8041. doi: 10.26355/eurrev_201811_16433. [DOI] [PubMed] [Google Scholar]
- 70.Ballini A, Santacroce L, Cantore S, Bottalico L, Dipalma G, Topi S, Saini R, De Vito D, Inchingolo F. Probiotics efficacy on oxidative stress values in inflammatory bowel disease: a randomized double-blinded placebo-controlled pilot study. Endocr Metab Immune Disord Drug Targets. 2019;19:373–381. doi: 10.2174/1871530319666181221150352. [DOI] [PubMed] [Google Scholar]
- 71.Ballini A, Cantore S, Saini R, Pettini F, Fotopoulou EA, Saini SR, Georgakopoulos IP, Dipalma G, Gargiulo Isacco C, Inchingolo F. Effect of activated charcoal probiotic toothpaste containing Lactobacillus paracasei and xylitol on dental caries: a randomized and controlled clinical trial. J Biol Regul Homeost Agents. 2019;33:977–981. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. Eligibility criteria for the present systematic review. Table S2. Strategy for database search (up to September 1st, 2019). Table S3. Studies excluded during full-text eligibility assessment with reasons.
Data Availability Statement
All data and materials are available upon request.