Abstract
Background
To investigate the genetic and environmental factors responsible for phenotype variability in a family carrying a novel CACNA1A missense mutation. Mutations in the CACNA1A gene were identified as responsible for at least three autosomal dominant disorders: FHM1 (Familial Hemiplegic Migraine), EA2 (Episodic Ataxia type 2), and SCA6 (Spinocerebellar Ataxia type 6). Overlapping clinical features within individuals of some families sharing the same CACNA1A mutation are not infrequent. Conversely, reports with distinct phenotypes within the same family associated with a common CACNA1A mutation are very rare.
Case presentation
A clinical, molecular, neuroradiological, neuropsychological, and neurophysiological study was carried out in proband and his carrier mother. The new heterozygous missense variant c.4262G > A (p.Arg1421Gln) in the CACNA1A gene was detected in the two affected family members. The proband showed a complex clinical presentation characterized by developmental delay, poor motor coordination, hemiplegic migraine attacks, behavioral dysregulation, and EEG abnormalities. The mother showed typical episodic ataxia attacks during infancy with no other comorbidities and mild cerebellar signs at present neurological evaluation.
Conclusions
The proband and his mother exhibit two distinct clinical phenotypes. It can be hypothesized that other unknown modifying genes and/or environmental factors may cooperate to generate the wide intrafamilial variability.
Keywords: CACNA1A gene; Familial hemiplegic migraine type 1; Episodic ataxia type2; Cognitive affective syndrome, neuropsychology.
Background
Hemiplegic Migraine (HM) is a rare migraine variant characterized by specific aura signs including some degree of hemiparesis and/or one-sided weakness. It may include sensory, visual or language impairment during aura. Two forms of HM have been recognized: familial hemiplegic migraine (FHM MIM #301011), in which at least a relative has migraine aura signs with the same clinical features, and sporadic hemiplegic migraine (SHM) with no recognizable familial history of hemiplegic migraine. FHM is an autosomal dominant inherited disorder caused by mutations in ion transporters encoding several genes: CACNA1A mutations cause FHM type 1 (FHM1, MIM:141500), ATP1A2 mutations cause FHM type 2, SCNA1 mutations cause FHM type 3 (ICHD-3beta, 2013) [1]. Episodic ataxia type 2 (EA2, MIM: 108500) is a paroxysmal neurological dysfunction of cerebellum lasting minutes to hours that includes symptoms like ataxia, nausea,vomiting, vertigo, diplopia, nystagmus, dysarthria, tinnitus, headache, and hemiplegia. Age of onset ranges between the first and the second decade of life, prevalence rate is unknown. The EA2 attacks can be triggered by some stressors such as alcohol, caffeine, physical and mental stress and postural changes, and relieved by rest and sleep. Neurological examination between attacks may be normal, but some patients may develop ataxia and nystagmus and progressive cerebellar atrophy on MRI. EA2 is an autosomal dominant disorder caused by mutation in the CACNA1A gene [2]. Mutations of the CACNA1A gene, located on chromosome 19p13.13, coding for the α1 pore –forming subunit of the voltage-gated calcium channel P/Q type (Cav2.1), were identified as responsible for three autosomal dominant disorders with incomplete penetrance: FHM1, EA2, and spinocerebellar ataxia type 6 (SCA6, MIM:183086) [3, 4]. However, overlapping clinical features described within some families with the same CACNA1A mutation are not infrequent [5–8]. Conversely, Pradotto reported distinct phenotypes, ranging from EA2 to SCA6, within the same family associated with a common CACNA1A mutation [9]. We report a child with a clinical phenotype characterized by some hemiplegic migraine attacks and neurodevelopmental disorder associated with a novel heterozygous c.4262G > A CACNA1A (NM_023035.2) variant. The same genetic abnormality was detected only in his mother with clinical history of a distinct infantile EA2 without migraine.
Case presentation
This study was approved by the ethics committee Palermo 1 of the “Paolo Giaccone” University Hospital of Palermo, Italy. Written informed consent for participate and for publication was obtained from each of the family members tested. The proband is a 7 year old child, the first of two siblings born to apparently healthy unrelated parents at the end of a pregnancy undertaken by an emergency caesarean section, following a gestational period complicated with some bleeding events from the seventh month.
His birth weight was 3900 g and his clinical postnatal course was uneventful. His developmental milestones have been delayed; he vocalized undifferentiated nasal sounds at 12 months, spoke the first word at 2 years and at 13 months he walked unsupported. He is currently under speech and neuropsychomotor therapy.
At the age of 4 years and 8 months, he was referred to our Child Neuropsychiatric Department for unilateral frontal headache attacks associated with pallor, crying, discomfort in the left lower limb, left-sided weakness, mostly in the left lower limb, leading to unsteady posture. The clinical events, started from 4 years of age, occurred almost every day in both wakefulness and sleep, lasted 10 to30 minutes, and spontaneously remitted after resting.
On admission the neurological examination showed a moderate motor and coordination impairment. The cognitive and behavioral evaluation revealed a severe deficit in behavioral regulation (restlessness, poor control of emotional drives with aggressive behavior, poor waiting and following rules abilities) and impaired language with severe phonetic-phonological failings.
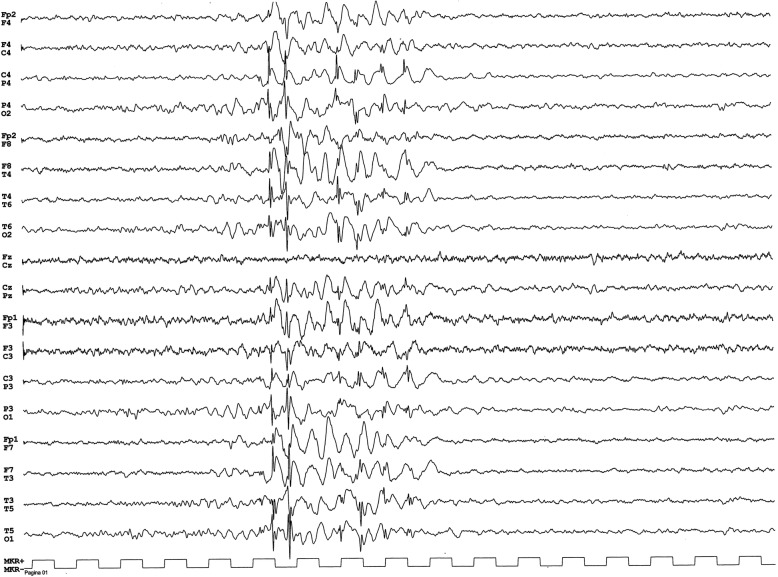
Wakefulness and sleep EEG displayed recurrent generalized 3 Hz regular spike-wave complex discharges lasting 1 to 3 s without associated clinical events (Fig. 1).
Fig. 1.
The EEG displayed recurrent generalized 3 Hz regular spike-wave complex discharges lasting 1to 3 s without associated clinical events
A treatment of valproic acid with increasing doses up to 30 mg∕ kg/die was started. The electroclinical follow-up displayed a striking disappearance of EEG abnormalities associated with a decrease of hemiplegic migraine attacks and a mild improvement of behavioral symptoms.
The latest clinical evaluations confirmed the disappearance of the generalized 3 Hz spike-wave discharges, showed a decrease of hemiplegic migraine attacks, unchanged motor abnormalities and a worsening of behavioral problems that required an antipsychotic drug in add-on to valproic acid.
The child underwent a comprehensive neuropsychological assessment using a battery of tests standardized for the Italian population (WISC-IV, NEPSY-II, Vineland ABS-II; references were reported in “supplementary material”) to evaluate intellectual, cognitive, and behavioral functions. The formal assessment showed an extremely low overall intellectual ability (IQ = 44) with borderline to average conceptual reasoning and problem-solving skills (PRI 72-88). The performance on measures of expressive vocabulary and lexical knowledge were in the borderline average range. The comprehension of instructions was impaired. The performance on motor dexterity and visuomotor precision was impaired; visuospatial and constructional tasks scored in the average- low average range. The memory tasks for faces and for names fell in the average and below average respectively. The performances on memory for designs and a story memory tasks were impaired in both immediate and delayed free recall. Attention was impaired on auditory tasks and in average range on visual tasks. Executive functions were impaired on inhibition, verbal fluency, and planning tasks. Social cognition (ToM and affect recognition from faces tasks) was impaired (Table 1).
Table 1.
Neuropsychological test results
| WISC-IV | NEPSY II | Vineland ABS-II | ||||||
|---|---|---|---|---|---|---|---|---|
| Score | 95% CI | Classification range | Subtests | score | Classification | Scales | Score | |
| Verbal Comprehension Index | 56 | 52–66 | Extremely low | Memory | Communication | 63 | ||
| Perceptual Reasoning Index | 78 | 72–88 | Borderline - average | Memory for faces total | 8 | average | Daily living skills | 77 |
| Working Memory Index | 46 | 43–61 | Extremely low | Immediate | 9 | average | Socialization | 79 |
| Processing Speed Index | 47 | 45–65 | Extremely low | Delayed | 8 | average | Motor skills | 37 |
| Intelligence Quotient | 44 | 41–53 | Extremely low | Memory for names total | 6 | below average | Adaptive Behavior Composite | 56 |
| Cognitive Proficiency Index | 33 | Extremely low | Delayed | 4 | impaired | |||
| Fluid Reasoning | 63 | 58–74 | Extremely low-borderline | Learning trials | 5 | below average | Subscales | Age (yr.mo) equivalent |
| Lexical knowledge | 74 | 68–85 | Extremely low-average | Memory for designs | ||||
| LTM | 69 | 64–79 | Extremely low-borderline | Immediate | 1 | impaired | Receptive | 3.8 |
| STM | 46 | 43–60 | Extremely low | Delayed | 2 | impaired | Expressive | 4.8 |
| Subtests: | score | Narrative memory | Written | 4.3 | ||||
| Verbal tasks | Free recall | 4 | impaired | Personal | 4.2 | |||
| Similarities | 2 | Impaired | Cued recall | 1 | impaired | Domestic | 5.9 | |
| Vocabulary | 5 | Below average | Recognition | 8 | average | Community | 4.9 | |
| Information | 4 | Impaired | Sentence repetition | 1 | impaired | Interpersonal relationships | 5.6 | |
| Comprehension | 1 | Impaired | Sensorimotor | Play and leasure time | 4.3 | |||
| Word Reasoning | 6 | Below average | Fingertip tapping | 1 | impaired | Coping skills | 4.1 | |
| Perceptual tasks | Imitating hand position | 2 | impaired | Gross motor skills | 2.9 | |||
| Block Design | 9 | Average | Visuomotor precision | 4 | impaired | Fine motor skills | 3.3 | |
| Matrix Reasoning | 8 | Average | Visuospatial | |||||
| Picture Concepts | 3 | Impaired | Design coping general | 5 | below average | |||
| Working Memory Tasks | Design coping specific | 1 | impaired | |||||
| Digit Span | 1 | Impaired | Block construction | 6 | below average | |||
| Forward | 3 | Impaired | Geometric puzzle | 9 | average | |||
| Backward | 3 | Impaired | Language | |||||
| Arithmetic | 2 | Impaired | Phonological processing | 4 | impaired | |||
| Letter-Number sequencing | 1 | Impaired | Speeded naming | 8 | average | |||
| Processing Speed Tasks | Comprehension of instructions | 1 | impaired | |||||
| Coding | 1 | Impaired | Semantic fluency | 1 | impaired | |||
| Symbol Search | 1 | Impaired | Phonological fluency | 2 | impaired | |||
| Attention/Executive | ||||||||
| Auditory attention | <2%ile | impaired | ||||||
| Visual attention | 9 | average | ||||||
| Design fluency | 6 | below average | ||||||
| Inhibition | 1 | impaired | ||||||
| Social perception | ||||||||
| Score: Standard score (mean100, standard deviation 15) | Theory of mind | 4 | impaired | |||||
| Score: Scaled score (mean 10, standard deviation 3) | Affect recognition | 4 | impaired | |||||
The child showed difficulties in emotion regulation including easy frustration, temper tantrums, oppositional behaviors, verbally and physically aggressive behavior.
MRI investigation, performed at 5 years of age, did not show structural abnormalities. The more detailed family clinical history highlighted some paroxysmal events in mother childhood with onset at 8 years of age characterized by sudden dramatic gait unsteadiness, vertigo, nausea and sometimes vomiting. These attacks occurred several times a week and lasted for hours, they were triggered by exertion and postural changes and relieved by rest and sleep. At the age of 15 interictal neurological and vestibular evaluation, and a cranial CT scan were not informative regarding the disorder cause. Although no treatment was recommended, the frequency of attacks graduallydecreased until their remission. In addition, the mother firmly denied a family history of FHM1 or EA2 attacks and SCA6. A recent neurological evaluation revealed nystagmus by lateral gaze, moderate fine motor impairment, dysdiadochokinesia. The intellectual assessment showed an extremely low overall intellectual ability (WAIS IV IQ = 49). General adaptive functioning was in the average compared to age matched peers (Vineland II: Adaptive Behavior Composite = 90) and she engages in a work activity and takes care of her family. The clinical and EEgraphic assessment, extended to the sister and the father of proband, showed neither migraine and / or cerebellar symptoms, nor cognitive and behavioral impairment, nor the 3 Hz SW pattern found in the patient.
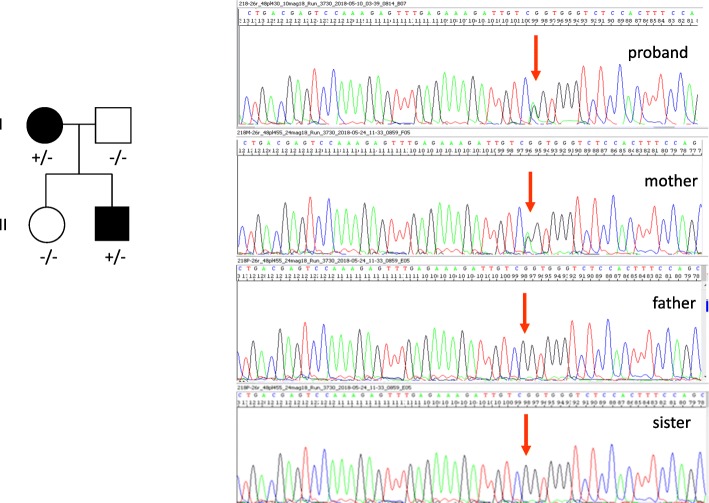
A next generation sequencing panel, exploring 45 genes associated with epileptic encephalopathy (for more details see the “supplementary material”), performed in the proband and his family members, showed a new heterozygous missense variant, c.4262G > A (p.Arg1421Gln) in the CACNA1A gene (NM_023035.2), [Chr19(GRCh37):g.13372264C > T], inherited from his mother, never described in literature and not reported in gnomAD and ExAC database (Fig. 2).
Fig. 2.
The novel mutation at c.4262G > A, p.(Arg1421Gln) in the CACNA1A gene was found in the proband and his mother
This variant was not been found in his healthy father and his healthy sister. This substitution causes the replacement of a positively charged arginine with a neutral not polar glutamine and involves a highly conserved residue located in the extracellular pore loop connecting S5 and S6, likely altering the calcium channel structure at this functionally critical region. A computer-based analysis, able to predict the effect of amino acid change on protein structure and function by using three different algorithms, PolyPhen2(http://genetics.bwh.harvard.edu/pph2/), Mutation taster http://www.mutationtaster.org/, and HSF (https://umd.be › HSF) respectively, indicated a possible deleterious effect on protein function (possibly damaging -score 0,6- for PolyPhen2, disease causing -score 43- for Mutation taster, and potential alteration of splicing for HSF). In addition, the variant, located very close to the exon-intron boundary, could lead to an abnormal splicing resulting in aberrant transcripts (Alamut visual ese predictions: Splicing predictions at nearest natural junction predicted change at donor site 1 bps downstream: − 64.2% MaxEnt: -81.2%, NNSPLICE: -98.9%, HSF: − 12.6%).
Discussion and conclusions
The neuronal CaV channels control several cellular functions and are involved in some neurological and psychiatric disorders [10].
The Cav2.1 channels are localized at presynaptic nerve terminals where the depolarization induces voltage-dependent calcium influx mediating the neurotransmitter release. They are expressed in all brain regions including the thalamocortical system, where P/Q-type VGCCs support the production of characteristic γ-band oscillations considered to be a functional prerequisite to cognitive states, but they are particularly expressed in Purkinje and granule cells [11, 12]. The identification of the pathogenic role of CACNA1A mutations in some disorders such as EA2, FHM1, and SCA6 suggested that these may be considered allelic disorders [3, 4]. Earlier it was hypothesized that the mutations inducing loss-of-function were associated with EA2, missense mutations resulting in gain-of-function caused FHM1, and abnormal CAG expansions were related with SCA6 [3, 4]. But overlapping features, including typical and uncommon symptoms of mentioned disorders, have been reported among the members of the same family [5–8]. Recently, a common CACNA1A mutation with distinct phenotypes, ranging from epileptic encephalopathy, EA2 to SCA6, was also found within the same family, suggesting that several factors could play a role in the phenotypic variability [9, 13].
Our affected family members share the same heterozygous c.4262G > A CACNA1A nucleotide exchange, located very close to the exon-intron boundary, which leads to the p.Arg1421Gln missense mutation. This variant is not included in the gnomAD browser and never identified in patients with FHM1, EA2, SCA6 with whom, therefore, a comparison is impossible. Nevertheless, it has been reported that most FHM1 and EA2 missense mutations produce substitutions of conserved amino acids (frequently arginine) clustering in important functional regions of the Cav2.1 channel including the pore lining (FHM1), the voltage sensors (FHM1), and the S5–S6 linkers (EA2) [12, 14, 15]. In addition, the p.Arg1421Gln variant segregating in all our affected family members and sparing the asymptomatic father and sister enhances its pathogenic relevance. The two patients showed two distinct phenotypes, the mother exhibited the typical EA2 without other comorbidities, and her son developed a more complex clinical feature characterized by hemiplegic migraine attacks, developmental delay, poor developmental movement coordination, and behavioral dysregulation. Since no relative has migraine aura symptoms, our child’s clinical feature could be classified as SHM with early onset. This co-occurrence in CACNA1A mutant patients has been associated with more severe outcome [16]. Many studies remarked in FHM1, EA2,and SCA6 patients the co-occurrence of behavioral and neuropsychological disorders but the investigations carried out with formal neuropsychological testing are scarce [17–19]. Recently, a large case series of 23/44 patients (11/23 FHM1,10/23 EA2, 2/23 SCA6), investigated by a formal neuropsychological testing, predominantly showed figural memory, visuoconstructive abilities, and verbal fluency impairment. Furthermore, the study revealed that neuropsychological deficits were consistent with history of development delay of patients [19]. A case study carried out using a formal neuropsychological assessment reported an impairment of semantic fluency and of processing speed, and a mild executive dysfunction [18]. A careful evaluation of our patient using a comprehensive neuropsychological battery revealed a partial agreement with the results of Indelicato and Trahan. Indeed, we pointed out intellectual disability and motor skill disorders but the lack of visual constructive disturbances. This discrepancy could depend more on the higher sensitivity of the WISC IV and NEPSY II than on the diagnostic tools used in above studies. We assume that the behavioral symptoms of our patient, irrespective of the lack of cerebellum atrophy, may depend on cerebellar dysfunction, being the Cav2.1 channel greatly expressed in the cerebellum. These behavioral symptoms could be comparable to the clinical features characterizing the well-known cerebellar cognitive affective syndrome [20–22]. Development delay of the child suggests also an impairment of cognitive networks due, besides the cerebellar dysfunction, also to an abnormal hippocampal neurotransmission as documented in knock-in mice model expressing the FHM1 mutation [23].
In addition, the child showed an electroencephalographic pattern characterized by frequent but short generalized 3 Hz regular spike-wave discharges without evidence of ictal clinical events. Nevertheless, their disappearance after valproic acid treatment associated with a mild improvement of attention and behavioral symptoms suggested that the child for an indefinite time may have suffered from “micro absence” and/or a mild cognitive impairment. This finding agrees with the electroclinical pattern often highlighted in EA2 patients carrying a truncating mutation of CACNA1A, suggesting a role of calcium channels in absence epilepsy although the electroclinical pattern in our patient is associated with the FHM1 phenotype [24, 25].
The impressive behavioral feature found in our patient and in many subjects previously reported in literature suggests the possibility to further expand the phenotype of CACNA1A mutations beyond ataxic and hemiplegic symptoms including cognitive and behavioral symptoms.
The careful analysis of our phenotypes does not provide useful data to elucidate the molecular mechanisms underlying the different clinical expressions of the disorder in mother and son.
Although the CACNA1A gene can generate different Cav channels by alternative pre-mRNA splicing, some of which have been associated with SCA6 and FHM1 phenotypes, to our knowledge no alternative splicing has been identified to be the cause of intrafamilial phenotypic variability [10, 26]. Furthermore, it has been suggested that mutations involving the splice site regions are often associated with higher variability of the phenotype and with the overlap of the common symptoms of the allelic disorders (FHM1 and SCA6) [17]; in addition, the different expression of Cav2.1 in the several neural networks could account for the variability of symptoms among the family members carrying the same mutation. Finally, as the emotional stress, minor head trauma, physical exercise, alcohol or caffeine are the most common triggers of FHM1 and EA2 attacks, other unknown individual modifier genes and / or environmental factors could play a regulator role in the phenotypic variability.
Acknowledgements
We thank the child and his parents who participated in this study. We acknowledge Dr. Vincenzo Antona for his advice and support in genetic counselling. We are grateful to Mr. Brinley Thomas for his assistance in checking the translation.
Abbreviations
- HM
Hemiplegic migraine
- SHM
Sporadic hemiplegic migraine
- FHM
Familial hemiplegic migraine
- CACNA1A
Calcium channel, voltage-dependent, p/q type, alpha-1a subunit
- ATP1A2
ATPase Na+/K+ transporting subunit alpha 2
- SCNA1
Sodium channel, neuronal type i, alpha subunit
- EA2
Episodic ataxia type 2
- SCA6
Spinocerebellar ataxia type 6
- EEG
Electroencephalogram
- WISC-IV
Wechsler intelligence scale IQ score
- NEPSY-II
Developmental NEuroPSYchological Assessment
- ToM
Theory of Mind
- ExAC
Exome Aggregation Consortium
- gnomAD
Genome Aggregation Database
- PolyPhen2
Polymorphism phenotyping v2
- HSF
Human splicing finder
- CT
Computed tomography
- WAIS
Wechsler Adult intelligence scale IQ score
- Cav2.1
P/Q voltage-dependent calcium channel
- Vineland ABS-II
Vineland adaptive behavior scales-ii
- VGCCs
Voltage-gated calcium channels
- CAG
Repeated codon CAG
Authors’ contributions
RN participated in the conceptualization, in writing the study and in revising and redrafting the manuscript. GP participated in the acquisition of the clinical data. GDM participated in the acquisition of EEG data. EG participated in the acquisition of genetic data. SM participated in revising the manuscript and in the interpretation of EEG data. FB participated in the acquisition and in the interpretation of the clinical data. VR participated in the acquisition and in the interpretation of the clinical data. AF participated in the conceptualization of the study and in the interpretation of the neuropsychological data. All authors have read and approved the manuscript.
Funding
The publication of the paper was paid thanks to funds of the grant: PSN Sicilia 2015 "Azione- 5.22: "Implementation of care pathways for continuity of care through the management of integrated hospital-territory services in patients with rare neuromuscular diseases ALS."
Availability of data and materials
All data generated or analyzed during current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
This study was approved by the ethics committee Palermo 1 of the “Paolo Giaccone” University Hospital of Palermo, Italy. Written informed consent for participate was obtained from each of the family members tested.
Consent for publication
Written informed consent for publication was obtained from each of the family members tested.
Competing interests
No author has any conflict of interest to declare except Filippo Brighina who is a member of the editorial board (Associate Editor) of this Journal.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Rosaria Nardello, Email: rosaria.nardello@unipa.it.
Giorgia Plicato, Email: giorgia.plicato@gmail.com.
Giuseppe Donato Mangano, Email: giuseppedonato.mangano@unipa.it.
Elena Gennaro, Email: elena.gennaro@galliera.it.
Salvatore Mangano, Email: salvatore.mangano@unipa.it.
Filippo Brighina, Email: filippo.brighina@unipa.it.
Vincenzo Raieli, Email: v.raieli@alice.it.
Antonina Fontana, Email: antonina.fontana@unipa.it.
References
- 1.The International Classification of Headache Disorders 3rd edition (beta version).Cephalalgia. 2013; 33:629-808. [DOI] [PubMed]
- 2.Jen J, Kim GW, Baloh RW. Clinical spectrum of episodic ataxia type 2. Neurology. 2004;62:17–22. doi: 10.1212/01.WNL.0000101675.61074.50. [DOI] [PubMed] [Google Scholar]
- 3.Ophoff RA, Terwindt GM, Vergouwe MN, et al. Familial hemiplegic migraine and episodic Ataxia Type-2 are caused by mutations in the Ca21 channel gene CACNL1A4. Cell. 1996;87:543–552. doi: 10.1016/S0092-8674(00)81373-2. [DOI] [PubMed] [Google Scholar]
- 4.Zhuchenko O, Bailey J, Bonnen P, et al. Autosomal dominant cerebellar ataxia (SCA6) associated with small polyglutamine expansions in the alpha 1A-voltagedependent calcium channel. Nat Genet. 1997;15:62–69. doi: 10.1038/ng0197-62. [DOI] [PubMed] [Google Scholar]
- 5.Jodice C, Mantuano E, Veneziano L, et al. Episodic Ataxia type 2 (EA2) and spinocerebellar ataxia type 6 (SCA6) due to CAG repeat expansion in the CACNA1A gene on chromosome 19p. Hum Mol Genet. 1997;6:1973–1978. doi: 10.1093/hmg/6.11.1973. [DOI] [PubMed] [Google Scholar]
- 6.Denier C, Ducros A, Vahedi K, et al. High prevalence of CACNA1A truncations and broader clinical spectrum in episodic ataxia type 2. Neurology. 1999;52:1816–1821. doi: 10.1212/WNL.52.9.1816. [DOI] [PubMed] [Google Scholar]
- 7.Alonso I, Barros J, Tuna A, et al. Phenotypes of spinocerebellar ataxia type 6 and familial hemiplegic migraine caused by a unique CACNA1A missense mutation in patients from a large family. Arch Neurol. 2003;60:610–614. doi: 10.1001/archneur.60.4.610. [DOI] [PubMed] [Google Scholar]
- 8.Romaniello R, Zucca C, Tonelli A, et al. A wide spectrum of clinical, neurophysiological and neuroradiological abnormalieties in a family with a novel CACNA1A mutation. J Neurol Neurosurg Psychiatry. 2010;8:840–843. doi: 10.1136/jnnp.2008.163402. [DOI] [PubMed] [Google Scholar]
- 9.Pradotto L, Mencarelli M, Bigoni M, et al. Episodic ataxia and SCA6 within the same family due to the D302N CACNA1A gene mutation. J Neurol Sci. 2016;371:81–84. doi: 10.1016/j.jns.2016.10.029. [DOI] [PubMed] [Google Scholar]
- 10.Lipscombe D, Andrade A, Allen S. Alternative splicing: functional diversity among voltage-gated calcium channels and behavioral consequences. Biochim Biophys Acta. 1828;2013:1522–1529. doi: 10.1016/j.bbamem.2012.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Llinás RR, Choi S, Urbano FJ, Shin HS. Gamma-band deficiency and abnormal thalamocortical activity in P/Q-type channel mutant mice. Proc Natl Acad Sci U S A. 2007;104:17819–17824. doi: 10.1073/pnas.0707945104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pietrobon D. CaV2.1 channelopathies. Pflugers Arch. 2010;460:375–393. doi: 10.1007/s00424-010-0802-8. [DOI] [PubMed] [Google Scholar]
- 13.Angelini C, Van Gils J, Bigourdan A, et al. Major intra-familial phenotypic heterogeneity and incomplete penetrance due to a CACNA1A pathogenic variant. Eur J Med Genet. 2019;62:103530. doi: 10.1016/j.ejmg.2018.08.011. [DOI] [PubMed] [Google Scholar]
- 14.Mantuano E, Veneziano L, Spadaro M, et al. Clusters of non-truncating mutations of P/Q type Ca2+ channel subunit ca(v)2.1 causing episodic ataxia 2. J Med Genet. 2004;41:e82. doi: 10.1136/jmg.2003.015396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sintas C, Carreño O, Fernàndez-Castillo N, et al. Mutation Spectrum in the CACNA1A gene in 49 patients with episodic Ataxia. Sci Rep. 2017;31:2514. doi: 10.1038/s41598-017-02554-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Riant F, Ducros A, Ploton C, et al. De novo mutations in ATP1A2 and CACNA1A are frequent in early-onset sporadic hemiplegic migraine. Neurology. 2010;75:967–972. doi: 10.1212/WNL.0b013e3181f25e8f. [DOI] [PubMed] [Google Scholar]
- 17.Nachbauer W, Nocker M, Karner E, et al. Episodic ataxia type 2: phenotype characteristics of a novel CACNA1A mutation and review of the literature. J Neurol. 2014;261:983–991. doi: 10.1007/s00415-014-7310-2. [DOI] [PubMed] [Google Scholar]
- 18.Trahan EM, Mercado JM. Familial hemiplegic migraines and baseline neuropsychological testing: a case report. Headache. 2019;59:917–923. doi: 10.1111/head.13505. [DOI] [PubMed] [Google Scholar]
- 19.Indelicato E, Nachbauer W, Karner E, et al. The neuropsychiatric phenotype in CACNA1A mutations: a retrospective single center study and review of the literature. Eur J Neurol. 2019;26:66–73. doi: 10.1111/ene.13765. [DOI] [PubMed] [Google Scholar]
- 20.Schmahmann JD, Sherman JC. The cerebellar cognitive affective syndrome. Brain. 1998;121:561–579. doi: 10.1093/brain/121.4.561. [DOI] [PubMed] [Google Scholar]
- 21.Levisohn L, Cronin-Golomb A, Schmahmann JD. Neuropsychological consequences of cerebellar tumour resection in children: cerebellar cognitive affective syndrome in a paediatric population. Brain. 2000;123:1041–1050. doi: 10.1093/brain/123.5.1041. [DOI] [PubMed] [Google Scholar]
- 22.Riva D, Giorgi C. The cerebellum contributes to higher functions during development: evidence from a series of children surgically treated for posterior fossa tumours. Brain. 2000;123:1051–1061. doi: 10.1093/brain/123.5.1051. [DOI] [PubMed] [Google Scholar]
- 23.Dilekoz E, Houben T, Eikermann-Haerter K, et al. Migraine mutations impair ippocampal learning despite enhanced long-term potentiation. J Neurosci. 2015;35:3397–3402. doi: 10.1523/JNEUROSCI.2630-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jouvenceau A, Eunson LH, Spauschus A, et al. Human epilepsy associated with dysfunction of the brain P/Q-type calcium channel. Lancet. 2001;358:801–807. doi: 10.1016/S0140-6736(01)05971-2. [DOI] [PubMed] [Google Scholar]
- 25.Imbrici P, Jaffe SL, Eunson LH, et al. Dysfunction of the brain calcium channel CaV2.1 in absence epilepsy and episodic ataxia. Brain. 2004;127:2682–2692. doi: 10.1093/brain/awh301. [DOI] [PubMed] [Google Scholar]
- 26.Chang SY, Yong TF, Yu CY, et al. Age and gender-dependent alternative splicing of P/Q-type calcium channel EF-hand. Neuroscience. 2007;145:1026–1036. doi: 10.1016/j.neuroscience.2006.12.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during current study are available from the corresponding author on reasonable request.