Abstract
Eosinophilic granulomatosis with polyangiitis (EGPA) is a vasculitis characterized by an abnormally high number of eosinophils in the peripheral blood and tissues. EGPA is an extremely rare disorder, with an incidence of 0.5 to 3.7 new cases per million people per year and an overall prevalence of 2.4 to 14 per million adults. There is little knowledge about the genetic factors that influence this disease. There are only two reports of familial EGPA: one in Japan and one in Turkey. We herein report a third case of familial EGPA in a brother and sister who were negative for myeloperoxidase-antineutrophil cytoplasmic antibodies.
Keywords: eosinophilic granulomatosis with polyangiitis, ANCA-associated vasculitis, asthma, familial onset, IVIG, mepolizumab
Introduction
Eosinophilic granulomatosis with polyangiitis (EGPA) is a vasculitis characterized by an abnormally high number of eosinophils in the peripheral blood and tissues (1). EGPA is an extremely rare disorder, with an incidence of 0.5 to 3.7 new cases per million people per year (1) and an overall prevalence of 2.4 to 14 per million adults (2,3). Asthma and eosinophilia are highly associated clinical features (1), but there is little knowledge about the genetic factors that influence this disease. There are only two reports of familial EGPA: one in Japan (4) and one in Turkey (5).
We herein report a third case of familial EGPA in a brother and sister who were negative for myeloperoxidase-antineutrophil cytoplasmic antibodies (MPO-ANCA).
Case Reports
Case 1: Brother
Case 1 was the 55-year-old elder brother of Case 2. He had acquired sinusitis at age 13. At age 45, he developed severe non-atopic bronchial asthma, and at age 54, he was hospitalized for asthma 3 times within the year. At age 55, he experienced general malaise followed by 2 months of lumbago, paresthesia and pain in the right lower leg, and numbness in his right hand, accompanied by a weight loss of 7 kg within 3 months. After that, he had difficulty walking because of paresthesia and pain in the left lower leg and arthralgia in both ankles.
Laboratory tests at the onset of EGPA revealed leukocytosis (leukocyte count of 25,400 /μL, 56.2% of which were eosinophils) and a negative MPO-ANCA or C-reactive protein (CRP) result. Both serum rheumatoid factor (RF) (690 IU/mL) and serum interleukin (IL)-2R (1,630 U/mL) were elevated. Lumbar spinal X-ray revealed no degenerative spondylosis. We diagnosed this patient with EGPA because his condition followed the characteristic clinical course outlined in the Japanese diagnostic criteria for “allergic granulomatosis with polyangiitis/Churg-Strauss syndrome (AGA/CSS)” (as EGPA was formerly known), with symptoms due to vasculitis following asthma and elevated levels of eosinophils in peripheral blood (6).
After treatment with methylprednisolone pulse (1,000 mg IV daily for 3 consecutive days) followed by 60 mg/day oral of prednisolone (PSL), mononeuritis multiplex as paresthesia in the right lower leg and numbness in the right hand had some improved, and he was able to walk without using a cane. However, when treatment was tapered to 5 mg/day of PSL, he experienced a relapse of mononeuritis multiplex as paresthesia of the left lower leg and bilateral sensory motor dysfunction. PSL was therefore increased to 7.5 mg/day, with additional treatment of 6 mg methotrexate (MTX) weekly. After treatment with MTX, the leukocyte count decreased, and we decided not to increase the dose of MTX. He was able to walk again after additional treatment with MTX, but sensory and motor neuropathy remained. His motor and sensory neuropathy improved after treatment with 400 mg/kg intravenous immunoglobulin (IVIG) for 5 consecutive days. PSL was reduced to 5.0 mg/day after IVIG treatment, and he remained in remission for more than 9 months (Fig. 1).
Figure 1.
Clinical course of treatment and relapse in Case 1: brother. IVIG: intravenous immunoglobulin, mPSL: methyl prednisolone, MTX: methotrexate, PSL: prednisolone
Case 2: Sister
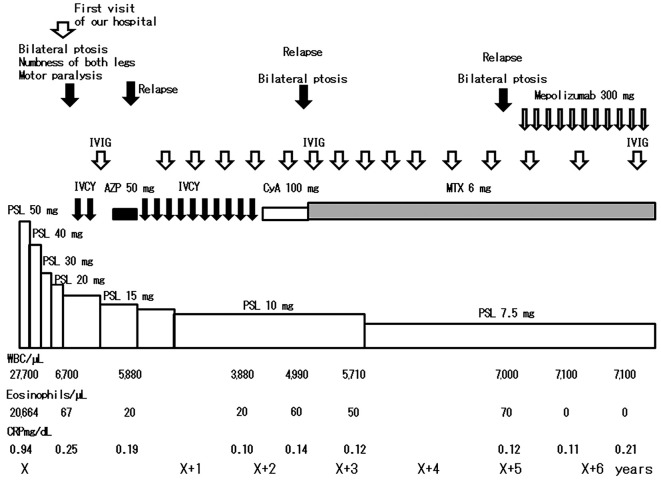
A 53-year-old woman (the younger sister of Case 1) had acquired non-atopic bronchial asthma when she was 20 years old. She experienced asthma exacerbations for several years from age 45. At 53 years old, she experienced general malaise followed by 1 month of abdominal pain, loss of appetite, joint pain in the wrists and fingers of both hands, purpura, and bilateral ptosis.
Laboratory tests at the onset of EGPA revealed leukocytosis (leukocyte count of 27,700 /μL, 74.6% of which were eosinophils), a negative MPO-ANCA or CRP result, serum IgE radioimmunosorbebt test (RIST) level of 993 IU/mL, elevated RF level of 80 IU/mL, and increased levels of aspartate aminotransferase (AST) (147 IU/L), alanine aminotransferase (ALT) (356 IU/L), alkaline phosphatase (ALP) (1,339 IU/L), and glutamyl transpeptidase (GTP) (122 IU/L). Computed tomography showed bilateral ground-glass opacity and consolidation continuing from the pleura in the upper lobes of the lungs and bilateral bronchial thickening in the lower lobes (indicating eosinophilic pneumonia), ethmoid sinusitis, and gallbladder swelling. The bronchoalveolar lavage fluid contained a high percentage of eosinophils (88.0%). She had a large number of eosinophils infiltrating the mucosa of the colon, and cardiac scintigraphy with iodine-123-labeled metaiodobenzylguanidine (MIBG) revealed cardiac involvement, appearing as spotty deficit accumulation of MIBG in the anterior region, the inferior region or apex of the heart. She was diagnosed by the Japanese diagnostic criteria for AGA/CSS as having EGPA (6).
She was treated with 50 mg/day (tapered to 20 mg/day) PSL. Her eosinophilic pneumonitis, abdominal pain, and gallbladder swelling improved. However, mononeuritis multiplex, such as muscle weakness and numbness in the lower legs and toes and bilateral ptosis, rapidly deteriorated. She was unable to walk because of sensory impairment, and she could not open her eyes. In addition to treatment with steroids, she was treated with intravenous cyclophosphamide (IVCY) (600 mg/m2) two times with a 3-week interval and 400 mg/kg of IVIG. Her mononeuritis multiplex, eosinophilic pneumonia abdominal pain, and gallbladder swelling improved. After that, she was treated with 50 mg/day azathioprine (AZP) as maintenance therapy in addition to 15 mg PSL. Four months after starting AZP, she relapsed again with bilateral ptosis, heart failure, and motor and sensory disturbance. After the discontinuation of AZP, she was treated with IVCY 10 times with a 4-week interval in addition to 10 to 12.5 mg/day of PSL and IVIG (4 times for 5 consecutive days at 400 mg/kg/day with a 3- or 4-month interval). She achieved remission of vasculitis and was treated with 100 mg/day of cyclosporine A as maintenance therapy.
However, after 7 months on this maintenance therapy, she again relapsed with bilateral ptosis and sensory motor disturbance. She started 6 mg/week MTX, but because she experienced general fatigue and nausea, we could not treat her with an increased dose of MTX. She was treated with IVIG (400 mg/kg for 5 consecutive days) 7 times with a 3- or 4-month interval in addition to the 6 mg/week of MTX and was able to have her PSL dose tapered to 7.5 mg/day. Almost all vasculitis symptoms improved, but ptosis and heart failure did not. She then began treatment with 300 mg/month mepolizumab in addition to 7.5 mg/day prednisolone and 6 mg MTX weekly. At 8 months after starting mepolizumab and after 2 courses of IVIG (400 mg/kg for 5 consecutive days), her ptosis had gradually improved (Fig. 2).
Figure 2.
Clinical course of treatment and relapse in Case 2: sister. AZP: azathioprine, CyA: cyclosporine A, IVCY: intravenous cyclophosphamide, IVIG: intravenous immunoglobulin, MTX: methotrexate, PSL: prednisolone
These were the only cases of EGPA in this family. Two other brothers had bronchial asthma, and another sister had allergic rhinitis. In addition, their mother, maternal grandmother, and maternal grandfather each had bronchial asthma, but there were no other relatives with allergic disease, vasculitis, or autoimmune diseases.
The ethics committee at our hospital approved the study, and written informed consent was obtained from the two patients.
Discussion
Whether the two patients had eosinophilic necrotizing vasculitis or granulomatosis could not be proven. However, both patients developed typical clinical signs of vasculitis following asthma and showed an elevated number of eosinophils in the peripheral blood (6), allowing us to distinguish their condition from chronic eosinophilic pneumonitis, hypereosinophilic syndrome (HES), and other types of vasculitis. In short, EGPA was able to be distinguished from HES in these siblings because it was preceded by an allergic disease, such as asthma or rhinitis.
A state of relapse was defined as the relapse, after remission, of vasculitis symptoms and signs, excluding the exacerbation of asthma or sinusitis (with or without an increase in the proportion of eosinophils among white blood cells), that required the resumption of immunosuppressive therapy or an increased dose of immunosuppressant (7). An increase in the number of eosinophils or an increase in the CRP level is not often found at relapse of EGPA with or without ANCA.
EGPA is a rare ANCA-associated vasculitis (2,3) with a poorly understood etiopathogenesis. The HLA-DRB1 (8), HLA-DRB4 (9), and IL-10 (10) genes are reported to play a role in EGPA pathogenesis. However, those reports are from studies in European populations, and no studies have analyzed the relationship with genetic factors in non-European populations. There are some reports of familial occurrences of systemic vasculitis, such as Goodpasture syndrome, microscopic polyangiitis, rapidly progressive glomerulonephritis, polyarteritis nodosa, and granulomatosis with polyangiitis (11). However, apart from this case report, there are only two reports of familial EGPA. In 2007, Tsurikisawa et al. reported the first case of familial EGPA in two sisters in Japan (4), and in 2014, Harmanci et al. reported the second case of familial EGPA in a mother and daughter in Turkey (5).
All six known patients with familial EGPA (Table) were negative for MPO-ANCA, and the two patients reported here were the only ones without house dust mite (HDM) sensitization. There was no marked difference in the types or ratios of organ involvement between cases of familial EGPA and the previously reported non-familial EGPA cases (7,12-14).
Table.
Comparison of Reported Cases and These Cases.
| Reference | Nationality | Family relationship | Age at onset of EGPA | WBC/mL | Eosinophils/mL | MPO-ANCA | IgE RAST/skin pric test |
|---|---|---|---|---|---|---|---|
| (4) | Japan | my sister | 69 | 20,900 | 18,622 | negative | HDM |
| sister | 62 | 13,510 | 7,863 | negative | HDM | ||
| (5) | Turkey | daughter | 17 | 19,800 | 13,682 | negative | HDM, grass pollen |
| mother | 45 | 18,500 | 5384 | negative | HDM | ||
| These cases | Japan | brother | 55 | 25,400 | 14,275 | negative | JC |
| sister | 53 | 27,700 | 20,664 | negative | all negative |
| Bronchial asthma | Sinusitis | Lung involvement | Mononeuritis multiplex | Cardiac involvement | Skin involvement | |
|---|---|---|---|---|---|---|
| (4) | 〇 | 〇 | 〇 | 〇 | 〇 | |
| 〇 | 〇 | 〇 | ||||
| (5) | 〇 | 〇 | 〇 | |||
| 〇 | 〇 | 〇 | 〇 | 〇 | ||
| These cases | 〇 | 〇 | 〇 | |||
| 〇 | 〇 | 〇 | 〇 | 〇 | 〇 |
HDM: house dust mite, JC: japanese cedar, circle: organ involve
EGPA patients with mononeuritis multiplex or heart failure occasionally respond poorly to standard therapy, such as combined therapy with corticosteroids and cyclophosphamide (15,16). We previously showed that IVIG therapy was effective against severe mononeuritis multiplex and heart failure in patients with EGPA who did not respond to combined corticosteroid-cyclophosphamide treatment (17). In both patients reported in the present study, sensory and motor dysfunction remained after treatment with corticosteroids and immunosuppressants. Even after the addition of several courses of IVIG, the treatment was not sufficient to improve the vasculitis of the central nervous system, as evidenced by continued ptosis.
Mepolizumab, a humanized anti-IL-5 monoclonal antibody, reduces the blood eosinophil count and may be of clinical benefit for the treatment of vasculitis symptoms in EGPA (18). The ptosis and heart failure in Case 2 improved gradually after starting treatment with mepolizumab. We consider mepolizumab to be a new treatment for EGPA.
We here report a rare case of familial EGPA. Further studies are needed in order to clarify the etiology and pathogenesis of EGPA.
The authors state that they have no Conflict of Interest (COI).
References
- 1. Jennette JC, Falk RJ, Bacon PA, et al. 2012 revised International Chapel Hill Consensus Conference nomenclature of vasculitides. Arthritis Rheum 65: 1-11, 2013. [DOI] [PubMed] [Google Scholar]
- 2. Gibelin A, Maldini C, Mahr A. Epidemiology and etiology of Wegener granulomatosis, microscopic polyangiitis, Churg-Strauss syndrome and Goodpasture syndrome: vasculitides with frequent lung involvement. Semin Respir Crit Care Med 32: 264-273, 2011. [DOI] [PubMed] [Google Scholar]
- 3. Watts RA, Carruthers DM, Scott DG. Epidemiology of systemic vasculitis: changing incidence or definition? Semin Arthritis Rheum 25: 28-34, 1995. [DOI] [PubMed] [Google Scholar]
- 4. Tsurikisawa N, Morita S, Tsuburai T, et al. Familial Churg-Strauss syndrome in two sisters. Chest 131: 592-594, 2007. [DOI] [PubMed] [Google Scholar]
- 5. Harmanci K, Anil H, Kocak A, Dinleyici EC. Familial eosinophilic granulomatosis with polyangiitis in a mother and daughter. BMJ Case Rep: 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Ozaki S. ANCA-associated vasculitis: diagnostic and therapeutic strategy. Allergol Int 56: 87-96, 2007. [DOI] [PubMed] [Google Scholar]
- 7. Guillevin L, Cohen P, Gayraud M, Lhote F, Jarrousse B, Casassus P. Churg-Strauss syndrome. Clinical study and long-term follow-up of 96 patients. Medicine 78: 26-37, 1999. [DOI] [PubMed] [Google Scholar]
- 8. Wieczorek S, Hellmich B, Gross WL, Epplen JT. Associations of Churg-Strauss syndrome with the HLA-DRB1 locus, and relationship to the genetics of antineutrophil cytoplasmic antibody-associated vasculitides. Arthritis Rheum 58: 329-330, 2008. [DOI] [PubMed] [Google Scholar]
- 9. Vaglio A, Martorana D, Maggiore U, et al. HLA-DRB4 as a genetic risk factor for Churg-Strauss syndrome. Arthritis Rheum 56: 3159-3166, 2007. [DOI] [PubMed] [Google Scholar]
- 10. Wieczorek S, Hellmich B, Arning L, et al. Functionally relevant variations of the interleukin-10 gene associated with antineutrophil cytoplasmic antibody-negative Churg-Strauss syndrome, but not with Wegener's granulomatosis. Arthritis Rheum 58: 1839-1848, 2008. [DOI] [PubMed] [Google Scholar]
- 11. Nowack R, Lehmann H, Flores-Suárez LF, Nanhou A, van der Woude FJ. Familial occurrence of systemic vasculitis and rapidly progressive glomerulonephritis. Am J Kidney Dis 34: 364-373, 1999. [DOI] [PubMed] [Google Scholar]
- 12. Sinco RA, Di Toma L, Maggiore U, et al. Prevalence and clinical significance of antineutrophil cytoplasmic antibodies in Churg-Strauss syndrome. Arthritis Rheum 52: 2926-2935, 2005. [DOI] [PubMed] [Google Scholar]
- 13. Tsurikisawa N, Oshikata C, Kinoshita A, Tsuburai T, Saito H. Long-term prognosis of 121 patients with eosinophilic granulomatosis with polyangiitis in Japan. J Rheumatol 44: 1206-1215, 2017. [DOI] [PubMed] [Google Scholar]
- 14. Moosig F, Bremer JP, Hellmich B, et al. A vasculitis centre based management strategy leads to improved outcome in eosinophilic granulomatosis and polyangiitis (Churg-Strauss, EGPA): monocentric experiences in 150 patients. Ann Rheum Dis 72: 1011-1017, 2013. [DOI] [PubMed] [Google Scholar]
- 15. Richter C, Schnabel A, Csernok E, De Groot K, Reinhold-Keller E, Gross WL. Treatment of anti-neutrophil cytoplasmic antibody (ANCA)-associated systemic vasculitis with high-dose intravenous immunoglobulin. Clin Exp Immunol 101: 2-7, 1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Renaldini E, Spandrio S, Cerudelli B, Affatato A, Balestrieri GP. Cardiac involvement in Churg-Strauss syndrome: a follow-up of three cases. Eur Heart J 14: 1712-1716, 1993. [DOI] [PubMed] [Google Scholar]
- 17. Tsurikisawa N, Taniguchi T, Saito H, et al. Treatment of Churg-Strauss syndrome with high-dose intravenous immunoglobulin. Ann Allerg Asthma Immunol 92: 80-87, 2004. [DOI] [PubMed] [Google Scholar]
- 18. Wechsler ME, Akuthota P, Jayne D, et al. Mepolizumab or placebo for eosinophilic granulomatosis with polyangiitis. N Engl J Med 376: 1921-1932, 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]