Abstract
Cases of coronavirus disease 2019 (COVID-19) have been reported in more than 200 countries. Thousands of health workers have been infected, and outbreaks have occurred in hospitals, aged care facilities, and prisons. The World Health Organization (WHO) has issued guidelines for contact and droplet precautions for healthcare workers caring for suspected COVID-19 patients, whereas the US Centers for Disease Control and Prevention (CDC) has initially recommended airborne precautions. The 1- to 2-meter (≈3–6 feet) rule of spatial separation is central to droplet precautions and assumes that large droplets do not travel further than 2 meters (≈6 feet). We aimed to review the evidence for horizontal distance traveled by droplets and the guidelines issued by the WHO, CDC, and European Centre for Disease Prevention and Control on respiratory protection for COVID-19. We found that the evidence base for current guidelines is sparse, and the available data do not support the 1- to 2-meter (≈3–6 feet) rule of spatial separation. Of 10 studies on horizontal droplet distance, 8 showed droplets travel more than 2 meters (≈6 feet), in some cases up to 8 meters (≈26 feet). Several studies of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) support aerosol transmission, and 1 study documented virus at a distance of 4 meters (≈13 feet) from the patient. Moreover, evidence suggests that infections cannot neatly be separated into the dichotomy of droplet versus airborne transmission routes. Available studies also show that SARS-CoV-2 can be detected in the air, and remain viable 3 hours after aerosolization. The weight of combined evidence supports airborne precautions for the occupational health and safety of health workers treating patients with COVID-19.
Keywords: airborne transmission, COVID-19, droplet precautions, mask, respiratory protection
At present, the limited available evidence does not support droplet precautions and 1- to 2-meter (≈3–6 feet) rule of special separation being adequate for occupational health and safety of health workers treating patients with COVID-19.
The epidemic of coronavirus disease 2019 (COVID-19) was reported to the World Health Organization (WHO) on December 31, 2019 [1], with the number of confirmed cases remaining approximately 40–60 until January 20, 2020, when a surge of cases occurred, possibly associated with increased domestic and international travel in China for the Lunar New Year celebration. On January 30, 2020, the number of cases surged to surpass the severe acute respiratory syndrome (SARS) epidemic, with cases spreading to over 28 other countries, mostly through travel from China [2]. On March 11, 2020, with more than 118 000 cases spread across 114 countries and 4291 deaths, it was recognized as a pandemic by the WHO [1].
Coronaviruses (CoVs) are respiratory pathogens, and the SARS-CoV-2 has been identified in both upper and lower respiratory tract samples from patients [3]. Fever, dry cough, malaise, lethargy, shortness of breath, and myalgia are the most common symptoms [2]. Less common symptoms are headache, productive cough, and diarrhea. Mild cases may present with a common cold-like syndrome, whereas severe cases may develop severe acute respiratory distress syndrome and pneumonia. According to the WHO, 21% of cases in China have a severe illness [2]. Early estimates of the reproduction number, R0, give values of approximately 2.2 with a mean incubation period of 5.2 days [4] and a range of up to 24 days. In a recent review, researchers found the average R0 value for COVID-19 to be up to 3.28 and a median value to be approximately 2.79 [5]. In a more recent study, researchers estimated the maximum-likelihood value of R0 to be 2.28 for the Diamond Princess cruise ship [6]. All of these estimates are similar to R0 estimates for SARS [7].
In the past epidemics of SARS and Middle East respiratory syndrome (MERS) coronavirus, healthcare workers (HCWs) have paid a heavy toll. During SARS, HCWs comprised 21% of all cases and in some countries, such as Hong Kong, Singapore, and Canada, more than half of the cases were HCWs, with deaths reported among them [8]. Healthcare worker deaths have already been reported with COVID-19.
The WHO has issued guidelines for protection of HCWs that recommend contact and droplet precautions for HCWs caring for suspected COVID-19 patients [9]. Specifically, a medical mask is recommended for routine care, whereas a respirator (airborne precautions) is recommended if HCWs are conducting an aerosol-generating procedure such as endotracheal intubation, bronchoscopy or airway suctioning, along with droplet precautions [9]. Droplet precautions include the recommendation to maintain spatial separation of 1 meter (≈3 feet) with an infected patient, in the belief that large droplets can only spread horizontally to a maximum of 1 meter (≈3 feet) [10]. The initial guidelines released by the US Centers for Disease Control and Prevention (CDC) recommended a more precautionary approach, which includes the use of a mask by the patient (source control [11]) and airborne precautions for HCWs [12].
We aimed to review the evidence supporting the rule of 1-meter (≈3 feet) spatial separation for droplet precautions in the context of guidelines issued by the WHO, CDC, and European Centre for Disease Prevention and Control (ECDC) for HCWs on respiratory protection for COVID-19.
METHODS
A systematic review was conducted for evidence of horizontal distance traveled by respiratory droplets, using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) criteria [13]. We used an open date strategy up to March 2020 for searching the literature. The search was made on PubMed and Scopus database, and the search terms used for literature search are as follows: (cough OR sneeze AND droplet AND spread) OR (cough OR sneeze AND droplet AND distance).
There are few studies on horizontal spread of droplets in medical journals, so we included original research studies from various science and engineering disciplines, including mathematical, numerical, and experimental studies, published in English language journals. We searched the Scopus database with the same keywords and date strategy for studies published in nonmedical journals. Editorials and reviews were excluded from the review.
Initial screening of articles was done by 1 reviewer (P.B.). For initial screening, the title and abstract of all the articles were reviewed. Articles were excluded if there is no information on droplet spread. All the articles that were potentially relevant after initial screening were procured in full text. Articles were included for the final review only if it specifically measured the horizontal distance of droplet spread. References of the papers were also included for screening if they fit the inclusion criteria. Four reviewers with expertise in fluid dynamics (P.B., C.D., C.d.S., and L.B.) reviewed the selected articles.
For the review, we focused on the following 4 variables among the studies included: (1) type of study, ie, experimental or modeling; (2) methodology used for modeling; (3) use of human subjects for data; and (4) data on extent of horizontal spread. Separate to review of original research evidence for horizontal spread of droplets, the guidelines for respiratory protection issued by the WHO, CDC, and ECDC for SARS, MERS, and COVID-19 coronaviruses were reviewed.
RESULTS
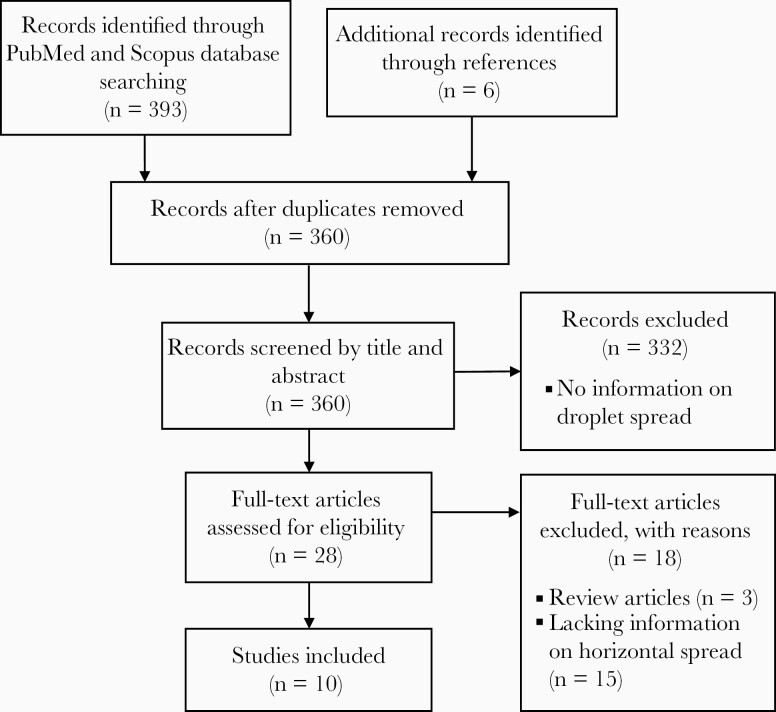
We found 393 papers in the initial search. After reviewing the titles and abstracts, 28 papers were selected for full text review. Finally, 10 papers were included in the review (Figure 1).
Figure 1.
Flow diagram of literature search.
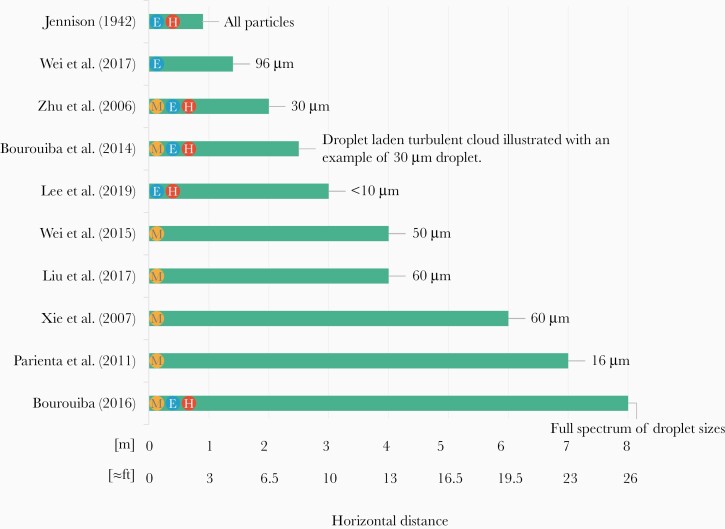
Eight of the 10 studies discussed a horizontal trajectory greater than 2 meters (≈6 feet) for a range of droplet sizes of less than 60 µm [14–21]. Seven of 10 studies are based on modeling, and among them the extent of horizontal spread of droplets vary between 2 and 8 meters (≈6–26 feet) [14–20], highlighting the different findings between them, which can be partially attributed to the methodologies used. Specifically, 4 of these studies rely on computational fluid dynamics approaches that do not accurately account for the multiphase particle-flow interaction physics [14, 15, 18, 20], and 3 of them model cough as a turbulent jet (continuous ejection with conservation of momentum flux) instead of a turbulent puff (short sudden ejection with conservation of momentum) [15, 18, 20]. The fourth study used Lagrangian modeling for the droplet dispersion, and it was acknowledged that this approach assigns a larger momentum to air hence, making it difficult to translate the results into relevant settings for hospital infection control [14].
Two studies used analogous water tank experiments to validate the mathematical modeling developed and reported distances up to 1.4 meters (≈4.5 feet) and 2.5 meters (≈8.2 feet) [17, 22]. One of these 2 studies modeled coughs as turbulent jets (continuous emission) [22] despite published contrary evidence showing that the physics of violent exhalations is captured by puffs, sudden high momentum emission of moist and hot air [17].
In 5 studies, experiments were performed on human subjects [14, 17, 19, 21, 23], 4 of them generated undisturbed/natural sneezes and coughs, without injestion of fluid or powders by the human subjects [17, 19, 21, 23]. Of 5 studies, 2 used the human subject measurements to develop and validate the mathematical modeling of the droplet dispersal and showed the importance of the exhaled gas cloud of hot and moist air in trapping and extending the range of all droplets [17, 19]. One involved injection of powder in the mouth of the human subject potentially shifting the natural droplet sizes ejected [14]. The other 2 used still photographs [23] and particle counters [21], and the distance reported among these 2 vary from 1 to 3 meters (≈3–10 feet). Table 1 summarizes all the findings, and Figure 2 shows the horizontal distance of droplet spread reported by all the studies.
Table 1.
Summary of Studies on Horizontal Spread of Droplets
| Author (Year) | Type of Study | Type of Experiments | Type of Modeling | Use of Human Subjects (Number of Subjects) | Main Findings Regarding Horizontal Distance |
|---|---|---|---|---|---|
| Jennison (1942) | Experimental | High-speed illumination for still photography | NA | Yes (not specified) | Majority of respiratory droplets, generated during sneezing, coughing, and talking, are expelled within 1 m (≈3 ft), the size of the filed of observation. |
| Zhu et al (2006) | Experimental and Modeling | Particle image velocimetry | Numerical Modeling | Yes (3) | More than 6.7 mg of saliva was expelled during coughing, at a maximum velocity of 22 m/s during each cough, affecting even area more than 2 m (≈6.5 ft) away from source. |
| Xie et al (2007) | Modeling | NA | Numerical Modeling | No | Expelled large droplets (>60 µm) can travel more than 6 m (≈20 ft) for sneezing with an exhalation velocity of 50 m/s and more than 2 m (≈6.5 ft) for coughing at an exhalation velocity of 10 m/s. |
| Parienta et al (2011) | Modeling | NA | Mathematical Modeling | No | With a coughing velocity of 11.7 m/s droplets with a diameter of 16 µm can travel a distance more than 7 m (≈23 ft). |
| Bourouiba et al (2014) | Experimental and Modeling | High-speed videography of human subject exhalations; Water tank physical experiments for model validation | Mathematical Modeling | Yes (not specified) | Droplets expelled during sneezing and coughing travel within a turbulent gas cloud and examples of ranges, such as that of particle with 30-µm diameter, which can have a horizontal range of 2.5 m (≈8 ft). |
| Wei and Li (2015) | Modeling | NA | Numerical Modeling | No | Relative humidity (RH) plays an important role in the evaporation of the droplets and the distance a droplet can travel. At a RH of 80% and expiration velocity of 10 m/s, 95% of medium droplets (50 μm) were able to travel 4 m (≈13 ft). |
| Bourouiba (2016) | Experimental and Modeling | High-speed imaging | Mathematical Modeling | Yes (not specified) | The smaller and evaporating droplets are trapped in the turbulent cloud, remain suspended, and can travel up to 6 to 8 m (≈20–26 ft). Based on modeling validated in Bourouiba et al [17]. |
| Wei and Li (2017) | Experimental and Modeling | Water tank experiments | Mathematical Modeling | No | Scaling relationships were used to scale the results of experiments in water with that of air. With mouth opening of 2 cm, large particles (96 μm) can travel a distance up to 1.4 m (≈4.5 ft). |
| Liu et al (2017) | Modeling | NA | Numerical Modeling | No | At 0% RH, 60-μm droplets would dry out and become droplet nuclei with a diameter of 19 μm and could fall out of the jet to reach a distance more than 4 m (≈13 ft). |
| Lee et al (2019) | Experimental | Optical particle spectrometer | NA | Yes (10) | Particle sizer and optical particle spectrometer were used to measure cough particle concentration of 10 patients with cold symptoms in real time. Results showed that transmission can spread more than 3 m (≈10 ft) from the patient. |
Abbreviations: ft, feet; m, meter; NA, not applicable; s, second.
Figure 2.
Extent of horizontal spread of droplets. Note that direct visualization of 8 meters also appears in [35]. E, experimental studies; H, human subjects; M, modeling (mathematical or numerical) studies.
Table 2 summarizes the respiratory protection guidelines by the WHO, CDC, and ECDC for SARS, MERS CoV, and COVID-19. Guidelines differentiate between high-risk and low-risk situations. High-risk is categorized as situations involving an aerosol-generating procedure, ie, endotracheal intubation, bronchoscopy, open suctioning, administration of nebulized treatment, manual ventilation before intubation, turning the patient to the prone position, disconnecting the patient from the ventilator, noninvasive positive-pressure ventilation, tracheostomy, and cardiopulmonary resuscitation. All other situations are considered low risk. The WHO and CDC recommend respirators to protect from SARS in both low- and high-risk situations [24, 25]. For MERS, WHO recommends masks in low-risk situations and respirators in high-risk situations, CDC recommends respirators in both situations, and ECDC recommends a preassessment of workplace to decide between mask and respirator in low-risk situations and respirators for high-risk situations [26–28]. For COVID-19, the WHO recommends masks in low-risk situations and respirators in high-risk situations. The CDC and ECDC initially recommended respirators in both situations, but after personal protective equipment shortages, the CDC downgraded to use of masks in low-risk situations and the ECDC recommended use of mask in case of nonavailability of respirators [29–31]. The interim guidelines for COVID-19 appear to assume only droplet and contact spread, and the general risk limit defined for HCWs is 1 meter (≈3 feet) from the patient [10, 31].
Table 2.
The Use of Masks/Respirators for Coronaviruses: Recommendations From WHO, CDC, and ECDC
| Pathogen | WHO | CDC | ECDC | |||
|---|---|---|---|---|---|---|
| Low Risk | High Riska | Low Risk | High Risk | Low Risk | High Risk | |
| Severe acute respiratory syndrome coronavirus (SARS-CoV) | Respiratorb | Respirator | Respirator | Respirator | - | - |
| Middle East respiratory syndrome coronavirus (MERS-CoV) | Mask | Respirator | Respirator | Respirator | Mask/Respiratorc | Respirator |
| Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) | Mask | Respirator | Mask | Respirator | Mask/Respiratord | Respirator |
Abbreviations: CDC, Centers for Disease Control and Prevention; ECDC, European Centre for Disease Control and Prevention; WHO, World Health Organization.
aHigh risk are the situations involving an aerosol-generating procedure, ie, endotracheal intubation, bronchoscopy, open suctioning, administration of nebulized treatment, manual ventilation before intubation, turning the patient to the prone position, disconnecting the patient from the ventilator, noninvasive positive-pressure ventilation, tracheostomy, and cardiopulmonary resuscitation.
bN/R/P 95/99/100 or FFP 2/3 or an equivalent national manufacturing standard (NIOSH [N, R, P 95, 99, 100] or European CE EN149:2001 [FFP2, FFP3] and EN143:2000 [P2] or comparable).
cNo clear recommendation. Choice is based on the type of exposure risk defined after preassessment of workplace.
dHealthcare workers in contact with a suspected or confirmed coronavirus disease 2019 (COVID-19) case should wear a surgical mask or, if available, an FFP2 respirator tested for fitting.
Discussion
The transmission of COVID-19 is not well characterized, but it is likely to be similar to SARS, which was spread by contact, droplet, and airborne routes [32]. Given the presence of SARS-CoV-2 viral loads in both the lower and upper respiratory tract [3], as well as the persistence of the virus in the air 3 hours after aerosolization in laboratory settings [33], airborne transmission is possible. A recent study showed that seasonal CoVs were more commonly emitted in aerosols than in droplets, even through normal tidal breathing [34]. It is timely to review the evidence informing the 1- to 2-meter (≈3–6 feet) rule of infection control, which drives guidelines for droplet precautions. Most studies of horizontal transmission of droplets show distances of greater than 2 meters (≈6 feet). The maximum distance recorded in the few available studies is 8 meters (≈26 feet) [19, 35]. We note that although the studies used very different methodologies and should be interpreted cautiously, they still confirm that the spatial separation limit of 1 meter (≈3 feet) prescribed for droplet precautions, and associated recommendations for staff at ports of entry [10], are not based on current scientific evidence.
The horizontal distance of droplet spread depends on various factors such as viscoelasticity of the expiration fluid, type of ventilation, velocity of expiration, rate of evaporation, and the dynamics of turbulent cloud generated during exhalations, sneezing, or coughing [15, 17–19]. The 1- to 2-meter (≈3–6 feet) limit is based on very limited epidemiologic and simulated studies of some selected infections [36]. Some studies cite Jennison [23] as the evidence in support of the 1- to 2-meter (≈3–6 feet) risk limit. This study used high-speed exposure to capture still photographs of the atomizing secretions generated by human sneezing, coughing, and talking, imaged very close to the mouth. It was concluded that the distance to which the majority of droplets were expelled is 2–3 feet (≈1 meter), but no details were provided about how they reached this conclusion. The study acknowledges that the motion picture film used for the experiments was not sensitive enough to capture all the droplets. The lighting technique used inherently selects for the largest sizes of droplets and fluid ligaments, not capturing the rest of the emissions and gas cloud carrying them. The author used still photographs, in which many droplets move out of focus and become unrecordable very quickly, especially using photographic technology from the 1940s. More recent studies have shown the extent of droplet spread to be greater than 2 meters (≈6 feet) [16–21, 35], and that infection risk exists well beyond the recommended range of spatial separation.
Furthermore, there is no agreement on the definition of “droplet” route of transmission. There is some agreement that particles with diameters less than 5 µm are airborne particles, but there is significant variation in the literature when it comes to the classification of the lower size limit of droplets. Wells [37] considered 100 µm as the cutoff limit for the droplet route. However, later studies considered a cutoff particle diameter of more than 10 µm to more than 100 µm [14, 15, 20]. The WHO uses a cutoff limit of 5 µm to differentiate between aerosols (≤5 µm) and droplet (>5 µm) [38] transmission routes. However, even particles with a diameter of more than 10 µm can remain airborne long enough to not fall under the framework of classification of “droplet” route [39]. In addition, the size of a droplet is dynamic and changes within seconds during the transit from the respiratory tract to the environment due to evaporation [39]. A large droplet expelled during coughing or sneezing can become an airborne particle in less than 1 second [39], and that timescale changes depending on the cloud dynamics of exhalation [17, 19]. Hence, it is not possible to characterize droplet and airborne spread as separate, mutually exclusive modes of transmission, and further studies of the risks accounting for combined ambient conditions and patient exhaled cloud are needed.
Indeed, another important consideration is the effect of temperature, relative humidity, ventilation, etc on the extent of droplet spread, which has been examined by only a few studies. To summarize, they have shown that relative humidity plays an important role in the evaporation of the droplets and the distance a droplet can travel. They report that as the relative humidity increases, the extent of droplet spread decreases [18, 20], yet the horizontal range of the cloud propelling the drops was found to increase with an increase in relative humidity, due to the role of buoyancy of the exhaled cloud [17]. For droplets less than 20 µm in diameter, local airflow field due to body heat is an important factor in determining the extent of spread because it can lift the droplets upwards into the breathing zone [40]. Studies have also shown that depending on the flow direction and airflow pattern, increasing ventilation rate can effectively reduce the risk of long-range, airborne transmission [41]. Most patients spend the majority of time in normal breathing and can saturate the room air with airborne particles expelled during breathing. Moreover, despite negative pressure isolation conditions, airflow due to door motion can cause breakdown in isolation conditions and as a result pathogen can escape the room, and there is a probability of infection spread outside the room [42]. In general, recent studies show distances reached by potentially pathogen-laden droplets of a continuum of sizes to be far greater than 2 meters (≈6 feet) [16–20]; therefore, the probability of infection well beyond the defined risk limit can be significant. For example, SARS was classified as predominantly transmitted through contact and droplet modes, but aerosolized transmission well beyond 2 meters (≈6 feet) was reported in the Amoy Gardens outbreak [32].
The ability of countries to respond effectively depends on the safety and confidence of the health workforce, especially in low-income countries with low ratios of HCWs per head of population, and protective measures are crucial to ensure a functional health workforce. We have previously shown that masks do not have clinical efficacy against respiratory infections [43, 44], and that intermittent use of respirators (which depends on HCWs to assess their own risk and use the device when they judge they are at risk) is as equally ineffective as mask use [44]. A recent trial confirmed there is no difference between targeted respirator use and surgical mask use, but it did not have a control arm and so it may have shown equal efficacy or inefficacy [45]. Proven efficacy of a respirator is seen when the device is worn continually during the shift [43]. The SARS-CoV-2 has been found in both upper and lower respiratory tract specimens, often early in the upper and later in the lower respiratory tract [3], which means it can potentially be dispersed in fine, airborne particles. Influenza studies show that in a busy emergency department or hospital ward, airborne particles with viable virus can persist for hours in the air [46]. A study of SARS-CoV-2 in a hospital in Wuhan found virus at least 4 meters (≈13 feet) within a hospital ward, and virus was identified in air samples and on multiple air outlet vents [47]. Other studies have also found SARS-CoV-2 on air vents in a patient room [48]. Another study found virus to be viable in air samples 3 hours after aerosolization in laboratory settings [33]. We have also shown that airborne precautions are more efficacious in protecting HCWs even against infections assumed to be spread by the droplet route [49]. This further supports the conclusion that transmission cannot be neatly separated into droplet versus airborne routes, and that it is likely driven by both airborne, and large droplets, carried by the respiratory gas cloud. In light of the lack of definitive transmission data for SARS-CoV-2, as well as persistence of the virus in the air 3 hours after aerosolization in laboratory settings [33], the precautionary principle in the initial CDC guidance was justified. This includes use of a mask by the patient, for which the limited evidence is supportive [11]. Guidelines should be precautionary in ensuring protection of the occupational health and safety of health workers treating COVID-19 [50]. Although the majority of the studies reviewed point towards horizontal spread of more than 2 meters (≈6 feet), these results cannot be translated directly to hospital settings, because the studies used a varying range of assumptions. The recent data on SARS-CoV-2 in a hospital ward shows a distance traveled by the virus of at least 4 meters (≈13 feet), double the assumed safe distance [47].
Conclusions
This review reveals the limited scientific data to inform spatial separation guidelines and a growing body of evidence that droplet precautions are not appropriate for SARS-CoV-2. Hence, future work on carefully documenting and studying the mechanisms shaping transmission distances are warranted, particularly with experiments over a large number of subjects and a variety of conditions, to update current spatial separation guidelines and the current paradigm of droplet and airborne respiratory transmission routes.
Notes
Financial support. This research was funded by NHMRC Centre for Research Excellence (Grant Number APP1107393), Integrated Systems for Epidemic Response. C. R. MacIntyre is supported by a NHMRC Principal Research Fellowship, grant number 1137582. L. B. acknowledges support from the Smith Family Foundation and MIT Policy Lab.
Potential conflicts of interest. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1. World Health Organization. Rolling updates on coronavirus disease (COVID-19). [cited 2020 April 14] Available at: https://www.who.int/emergencies/diseases/novel-coronavirus- 2019/events-as-they-happen. Accessed 14 April 2020.
- 2. World Health Organization. Novel coronavirus (2019-nCoV) situation reports. [cited 2020 January 31] Available at: https://www.who.int/emergencies/diseases/novel-coronavirus- 2019/situation-reports. Accessed 31 January 2020.
- 3. Wölfel R, Corman VM, Guggemos W, et al. Virological assessment of hospitalized patients with COVID-2019. Nature 2020. doi:10.1038/s41586-020-2196-x [DOI] [PubMed] [Google Scholar]
- 4. Li Q, Guan X, Wu P, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N Engl J Med 2020. doi:10.1056/NEJMoa2001316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Liu Y, Gayle AA, Wilder-Smith A, Rocklöv J. The reproductive number of COVID-19 is higher compared to SARS coronavirus. J Travel Med 2020; 27. doi:10.1093/jtm/taaa021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Zhang S, Diao M, Yu W, Pei L, Lin Z, Chen D. Estimation of the reproductive number of novel coronavirus (COVID-19) and the probable outbreak size on the Diamond Princess cruise ship: a data-driven analysis. Int J Infect Dis 2020; 93:201–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Lipsitch M, Cohen T, Cooper B, et al. Transmission dynamics and control of severe acute respiratory syndrome. Science 2003; 300:1966–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Sepkowitz KA, Eisenberg L. Occupational deaths among healthcare workers. Emerg Infect Dis 2005; 11:1003–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. World Health Organization. Infection prevention and control during health care when novel coronavirus (nCoV) infection is suspected. Interim guidance. [cited 2020 January 26] Available at: https://www.who.int/publications-detail/infection-prevention-and-control-during-health-care-when-novel-coronavirus-(ncov)-infection-is-suspected. Accessed 26 January 2020.
- 10. World Health Organization. Management of ill travellers at points of entry - international airports, seaports and ground crossings - in the context of COVID-19 outbreak: interim guidance. [cited 16 February 2020 ] Available at: https://apps.who.int/iris/handle/10665/331003. Accessed 16 February 2020.
- 11. MacIntyre CR, Zhang Y, Chughtai AA, et al. Cluster randomised controlled trial to examine medical mask use as source control for people with respiratory illness. BMJ Open 2016; 6:e012330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Centers for Disease Control and Prevention. Interim healthcare infection prevention and control recommendations for patients under investigation for 2019 novel coronavirus. [cited 2020 January 31] Available at: https://www.cdc.gov/coronavirus/2019-nCoV/infection-control.html. Accessed 31 January 2020.
- 13. Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 2009; 339:b2535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Zhu SW, Kato S, Yang JH. Study on transport characteristics of saliva droplets produced by coughing in a calm indoor environment. Build Environ 2006; 41:1691–702. [Google Scholar]
- 15. Xie X, Li Y, Chwang AT, Ho PL, Seto WH. How far droplets can move in indoor environments–revisiting the Wells evaporation-falling curve. Indoor Air 2007; 17:211–25. [DOI] [PubMed] [Google Scholar]
- 16. Parienta D, Morawska L, Johnson GR, et al. Theoretical analysis of the motion and evaporation of exhaled respiratory droplets of mixed composition. J Aerosol Sci 2011; 42:1–10. [Google Scholar]
- 17. Bourouiba L, Dehandschoewercker E, Bush JWM. Violent expiratory events: on coughing and sneezing. J Fluid Mech 2014; 745:537–63. [Google Scholar]
- 18. Wei J, Li Y. Enhanced spread of expiratory droplets by turbulence in a cough jet. Build Environ 2015; 93:86–96. [Google Scholar]
- 19. Bourouiba L. A sneeze. N Engl J Med 2016; 375:e15. [DOI] [PubMed] [Google Scholar]
- 20. Liu L, Wei J, Li Y, Ooi A. Evaporation and dispersion of respiratory droplets from coughing. Indoor Air 2017; 27:179–90. [DOI] [PubMed] [Google Scholar]
- 21. Lee J, Yoo D, Ryu S, et al. Quantity, size distribution, and characteristics of cough-generated aerosol produced by patients with an upper respiratory tract infection. Aerosol Air Qual Res 2019; 19:840–53. [Google Scholar]
- 22. Wei J, Li Y. Human cough as a two-stage jet and its role in particle transport. PLoS One 2017; 12:1–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Jennison MW. Atomizing of mouth and nose secretions into the air as revealed by high-speed photography. Aerobiology. 17th ed. Washington: American Association for the Advancement of Science, 1942: pp 106–28. [Google Scholar]
- 24. Centers for Disease Control and Prevention. Interim domestic guidance on the use of respirators to prevent transmission of SARS. Available at: https://www.cdc.gov/sars/clinical/respirators.html. Accessed 11 April 2020.
- 25. World Health Organization. Hospital infection control guidance for severe acute respiratory syndrome (SARS). Available at: https://www.who.int/ihr/lyon/surveillance/infectioncontrol/en/. Accessed 11 April 2020.
- 26. World Health Organization. Infection prevention and control during health care for probable or confirmed cases of Middle East respiratory syndrome coronavirus (MERS-CoV) infection: interim guidance: updated October 2019. Available at: https://apps.who.int/iris/handle/10665/174652. Accessed 11 April 2020.
- 27. Centers for Disease Control and Prevention. Interim infection prevention and control recommendations for hospitalized patients with Middle East respiratory syndrome coronavirus (MERS-CoV). Available at: https://www.cdc.gov/coronavirus/mers/infection-prevention-control.html. Accessed 11 April 2020.
- 28. European Centre for Disease Control and Prevention. Rapid risk assessment: severe respiratory disease associated with Middle East respiratory syndrome coronavirus (MERS-CoV). Available at: https://www.ecdc.europa.eu/en/publications-data/rapid-risk-assessment-severe-respiratory-disease-associated-middle-east-8. Accessed 11 April 2020.
- 29. Jenco M. CDC updates guidance on PPE for health care personnel; COVID-19 declared a pandemic. Available at: https://www.aappublications.org/news/2020/03/11/coronavirus031120. Accessed 11 April 2020.
- 30. Centers for Disease Control and Prevention. Interim infection prevention and control recommendations for patients with suspected or confirmed coronavirus disease 2019 (COVID-19) in healthcare settings. Available at: https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html. Accessed 11 April 2020.
- 31. European Centre for Disease Control and Prevention. Infection prevention and control and preparedness for COVID-19 in healthcare settings - second update. [cited 2020 April 11] Available at: https://www.ecdc.europa.eu/en/publications-data/infection-prevention-and-control-and-preparedness-covid-19-healthcare-settings. Accessed 11 April 2020.
- 32. Yu ITSS, Li Y, Wong TW, et al. Evidence of airborne transmission of the severe acute respiratory syndrome virus. N Engl J Med 2004; 350:1731–9. [DOI] [PubMed] [Google Scholar]
- 33. Doremalen N van, Bushmaker T, Morris DH, et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N Engl J Med 2020. doi:10.1056/NEJMc2004973 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Leung NHL, Chu DKW, Shiu EYC, et al. Respiratory virus shedding in exhaled breath and efficacy of face masks. Nat Med 2020. doi:10.1038/s41591-020-0843-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Bourouiba L. Turbulent gas clouds and respiratory pathogen emissions. JAMA 2020. doi:10.1001/jama.2020.4756 [DOI] [PubMed] [Google Scholar]
- 36. Siegel JD, Rhinehart E, Jackson M, Chiarello L. 2007 Guideline for isolation precautions: preventing transmission of infectious agents in health care settings. Am J Infect Control 2007; 35:S65–S164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Wells WF. On air-borne infection. II. Droplets and droplet nuclei. Am J Hyg 1934; 20:611–8. [Google Scholar]
- 38. World Health Organization. Infection prevention and control of epidemic- and pandemic-prone acute respiratory diseases in health care. WHO Guidelines [Internet] 2014:1–156. Available from: https://apps.who.int/iris/bitstream/10665/112656/1/9789241507134_eng.pdf. Accessed 14 April 2020. [PubMed] [Google Scholar]
- 39. Hinds WC. Aerosol Technology: Properties, Behavior, and Measurement of Airborne Particles. New York: John; Wiley & Sons; 2012. [Google Scholar]
- 40. Yan Y, Li X, Tu J. Thermal effect of human body on cough droplets evaporation and dispersion in an enclosed space. Build Environ 2019; 148:96–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Qian H, Zheng X. Ventilation control for airborne transmission of human exhaled bio-aerosols in buildings. J Thorac Dis 2018; 10:2295–304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Tang JW, Eames I, Li Y, et al. Door-opening motion can potentially lead to a transient breakdown in negative-pressure isolation conditions: the importance of vorticity and buoyancy airflows. J Hosp Infect 2005; 61:283–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. MacIntyre CR, Wang Q, Cauchemez S, et al. A cluster randomized clinical trial comparing fit-tested and non-fit-tested N95 respirators to medical masks to prevent respiratory virus infection in health care workers. Influenza Other Respir Viruses 2011; 5:170–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. MacIntyre CR, Wang Q, Seale H, et al. A randomized clinical trial of three options for N95 respirators and medical masks in health workers. Am J Respir Crit Care Med 2013; 187:960–6. [DOI] [PubMed] [Google Scholar]
- 45. Radonovich LJ, Simberkoff MS, Bessesen MT, et al. N95 respirators vs medical masks for preventing influenza among health care personnel. JAMA 2019; 322:824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Blachere FM, Lindsley WG, Pearce TA, et al. Measurement of airborne influenza virus in a hospital emergency department. Clin Infect Dis 2009; 48:438–40. [DOI] [PubMed] [Google Scholar]
- 47. Guo Z-D, Wang Z-Y, Zhang S-F, et al. Aerosol and surface distribution of severe acute respiratory syndrome coronavirus 2 in hospital wards, Wuhan, China, 2020. Emerg Infect Dis 2020; 26. doi:10.3201/eid2607.200885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Ong SWX, Tan YK, Chia PY, et al. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2) from a symptomatic patient. JAMA 2020. doi:10.1001/jama.2020.3227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. MacIntyre CRCR, Chughtai AAAA, Rahman B, et al. The efficacy of medical masks and respirators against respiratory infection in healthcare workers. Influenza Other Respi Viruses 2017; 11:511–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. MacIntyre CR, Chughtai AA, Seale H, Richards GA, Davidson PM. Uncertainty, risk analysis and change for Ebola personal protective equipment guidelines. Int J Nurs Stud 2015; 52:899–903. [DOI] [PMC free article] [PubMed] [Google Scholar]