Abstract
Xenotransplantation using pig cells, tissues and organs may be associated with the transfer of porcine infectious agents, which may infect the human recipient and in the worst case induce a disease (zoonosis). To prevent this, a broad screening program of the donor animals for putative zoonotic microorganisms, including bacteria, viruses, fungi and others, using sensitive and specific detection methods has to be performed. As long as it is still unknown, which microorganism represents a real risk for the recipient, experience from allotransplantation should be brought in. Due to the fact that pigs can be screened long before the date of transplantation, xenotransplantation will become eventually safer compared with allotransplantation.
Screening and selection of animals free of potential zoonotic microorganisms, Caesarean section, vaccination and/or treatment with chemotherapeutics are the strategies of choice to obtain donor animals not transmitting microorganisms. In the case of porcine endogenous retroviruses (PERVs) which are integrated in the genome of all pigs and which cannot be eliminated this way, selection of animals with low virus expression and generation of genetically modified pigs suppressing PERV expressions may be performed.
Keywords: Xenotransplantation, Virus safety, Porcine endogenous retroviruses, Porcine cytomegalovirus, Porcine lymphotropic herpesviruses, Porcine circoviruses, Hepatitis E virus, Elimination programs
Highlights
-
•
PCMV, PLHV, HEV and PERV were identified as main risk factors in xenotransplantation.
-
•
Detection methods and elimination programs for PCMV, PLHV, and HEV were developed.
-
•
PERVs are integrated in the genome of all pigs and infect human cells in vitro.
-
•
Strategies how to prevent PERV transmission are also under development.
1. Introduction
Xenotransplantation using pig cells, tissues and organs has to overcome three hurdles before being applied in the clinic for the treatment of organ failure: immunological rejection, physiological incompatibility and transfer of infectious agents. The microbiological safety of xenotransplantation is an important issue, however it can be managed easily. The risk of infection is also known in allotransplantation. Numerous infectious agents have been transmitted together with human donor transplants, including human cytomegalovirus (HCMV), human immunodeficiency virus-1 (HIV-1) and rabies virus [1]. Since xenotransplantation allows screening the donor animals beforehand, most risks can be excluded by careful testing and xenotransplantation finally will be a microbiologically safer technology compared with allotransplantation.
Like all animals, pigs carry numerous microorganisms in their digestive tract and on their skin, and therefore cells, tissues and organs to be used for transplantation should be removed under aseptic conditions. The number of microorganisms present in the tissues and organs of interest should be zero [2]. In some reviews concerning the microbiological safety of xenotransplantation numerous microorganisms are listed which were thought to induce zoonoses when transmitted to the human recipient [2]. Zoonosis means that the microorganisms not only infect the new host, but cause a disease. In general, bacteria, fungi, parasites and viruses may be transmitted. However, at present it is rather difficult to classify most of the porcine microorganisms into pathogenic and non-pathogenic for human recipients. In addition, when a microorganism is pathogenic in the pig it does not mean that it is also pathogenic in humans and vice versa. The risk of transmission is certainly higher when pharmaceutical immunosuppression to prevent immunological rejection of the transplant will be applied. Therefore it is still unclear which microorganisms should be monitored. The Auckland island pigs which had been used in the first clinical trials performed by the New Zealand company LCT were screened regularly for 10 bacteria, 15 viruses and toxoplasma (Table 1 ) [3]. The Göttingen Minipigs which are used for numerous biomedical investigations are screened regularly for 27 bacteria, 16 viruses, three fungi and four parasites (http//www.minipigs.dk/). An additional screening of the Göttingen Minipigs involved PERV [4], hepatitis E and 89 other microorganisms [5], [6].
Table 1.
Microorganisms tested in Auckland island pigs used for islet cell transplantation [3].
| Bacteria | |
| Leptospira tarrasovi | |
| Leptospira hardjo | |
| Leptospira pomona | |
| Mycoplasma hyopneumoniae | |
| Campylobacter | |
| Isospora | |
| Cryptosporidium | |
| E. coli K88 | |
| Yersinia | |
| Viruses | |
| PCMV | Porcine cytomegalovirus |
| PCV1 | PCV1, porcine circovirus type 1 |
| PCV2 | PCV2, porcine circovirus type 2 |
| PLHV2 | Porcine lymphotrophic herpesvirus type 2 |
| HEV | Hepatitis E virus |
| ReoV | Reovirus (all types) |
| RotaV A-C | Rotavirus A, rotavirus B and rotavirus C |
| PEVB | Porcine enterovirus B |
| PHEV | Porcine hemagglutinating encephalomyelitis virus |
| PTV | Porcine teschovirus |
| BVD | Bovine virus diarrhea |
| AujD | Aujesky's disease |
| PPV | Porcine parvovirus |
| PRRSV | Porcine reproductive and respiratory syndrome virus |
| EMCV | Encephalomyocarditis virus |
| Protozoa | |
| Toxoplasma | |
In general, most microorganisms found in pigs to be used for xenotransplantation may be eliminated by specified pathogen free (spf) or designated pathogen-free (dpf) breeding of the animals. In the case there is a bacterial or fungal infection in the donor pig, treatment with antibiotics or chemotherapeutics may be successful. At the moment hepatitis E virus (HEV), porcine cytomegalovirus (PCMV), porcine circoviruses (PCV), porcine lymphotropic herpes viruses (PLHV), and porcine endogenous retroviruses (PERVs) are thought to pose the main risk for reasons to be discussed below and therefore these microorganisms will be analysed in the next chapters in more details.
2. HEV as risk factor
In most cases HEV causes self-limiting hepatitis in humans. Whereas HEV of the genotype (gt) 1 and gt2 are found in people, are transmitted mainly by contaminated water and are causing a high mortality during pregnancy, HEV gt3 and gt4 are swine viruses and do not cause a disease in pigs, however, when they infect humans they may cause in rare cases a zoonotic disease (for review see [5], [7]). A severe hepatitis after infection with HEV gt3 and gt4 was observed only in the case of other underlying liver diseases. Importantly, neurological disorders have also been described for HEV gt3 and gt1. Note, that only HEV gt3/4 may pose a risk when xenotransplantation using pig cells, tissues and organs is performed, not gt1 or gt2.
Usually HEV gt3/4 are transmitted by contaminated meat or direct contact with infected pigs. HEV gt3 RNA was detected in pig liver at grocery stores and infectious virus could be isolated [5], [7]. HEV transmission by shellfish and vegetables possibly contaminated by pig manure as well as by blood transfusion and allotransplantion was also reported. A chronic infection was more likely to develop in immunosuppressed patients, including HIV-1 infected individuals [5], [7]. Sensitive PCR-based methods have been developed to determine a HEV infection and to genotype the virus. Detection of HEV and its elimination from pigs seems not to be easy. First, the virus is heterogeneous, e.g., 10 subtypes of genotype 3 exist, what makes it difficult to design efficient PCR or real-time PCR. Second, the virus load seems to be very low so that even highly sensitive PCRs may be unable to detect the virus. Although HEV gt3/4 are widely distributed, the prevalence in pigs, especially in multitransgenic pigs generated for usage in xenotransplantation, is not well studied.
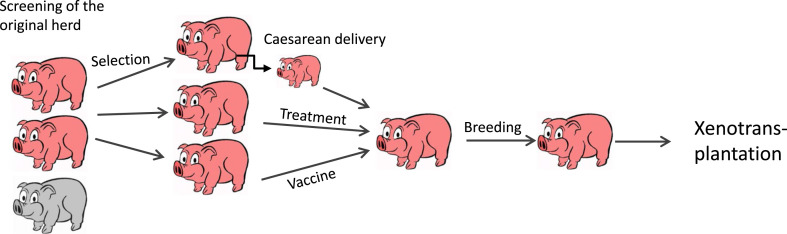
In contrast, the non-transgenic Auckland island pigs, generated by Living Cell Technologies (LCT) in New Zealand are better characterised. Islet cells from these animals have already been used in several clinical trials [3]. Although HEV is very common in pigs in New Zealand, the Auckland island pigs were free of all HEV [8]. Another well investigated breed is the Göttingen minipig produced by Ellegaard in Denmark. These animals are used worldwide for numerous biomedical investigations. The herd was established by entry of animals obtained by Caesarean sectioning and colostrum deprivation. Despite this, HEV was found in one study in 100% of the animals (7 of 7) [9], in another, using real-time PCR and Western blot analysis detecting antibodies against HEV, HEV was found in only very few animals [6]. The result suggested a transplacental mother-to-piglet transmission of the virus. This observation may help to explain how the virus entered the pig herd despite Caesarean sectioning and other precautions. It remains unclear, whether the absence of the virus in all older Göttingen minipigs is due to the elimination of the virus possibly by the immune system or due to the limits of the detection methods. In order to eliminate HEV from a herd, a HEV elimination program was proposed (Fig. 1 ) [5]. Elimination should include selection of HEV negative animals using highly sensitive real-time PCR. Since it is not clear, whether the animals are truly negative, or carry HEV in concentrations below the sensitivity of the detection method, a treatment step should be included using ribavirin, a guanosine analogue used to stop viral RNA synthesis. Although there are no data on the treatment of HEV infection with ribavirin in pigs, ribavirin has been successfully used for the treatment of other virus infections in pigs [10]. Another strategy may include a vaccination step, e.g. using a vaccine based on a recombinant ORF-2 fragment of HEV gt1 that has been approved by the Chinese FDA [11]. Since HEV gt1-4 represents only one serotype, the induced antibodies should be protecting from infection with all genotypes [12]. Immunisation of pigs with ORF-2 of pig HEV gt3 resulted in effective protection [12], indicating that pigs can be immunized and mount an effective antiviral immune response.
Fig. 1.
Schematic presentation of the proposed virus elimination program. The original herd was screened for the presence of a putative zoonotic virus (grey, positive animals; pink, negative animals). Negative animals (that may be actually positive, but below the detection limit) were selected and using Caesarean delivery, and treatment with antiviral drugs and/or vaccines, truly negative animals could be obtained and used for further breeding and xenotransplantation.
3. PCMV and other herpesviruses
As human cytomegalovirus (HCMV) causes severe transplant rejection in allotransplantation [13], [14], considerable concern is warranted on the potential pathogenicity of porcine cytomegalovirus (PCMV) in the setting of xenotransplantation. PCMV is endemic in the world pig population, it is acquired early in life and PCMV infection results in seroconversion and lifelong latent infection [15]. PCMV spreads by both vertical and horizontal transmission [15], [16]. Active infection causes fatal systemic failure in piglets less than 3 weeks of age. The clinical symptoms of infected piglets include pneumonia and inclusion body rhinitis with a high mortality rate. PCMV-infected sows are prone to abortion, with pathological changes including edema in the heart and other organs [15]. PCMV can remain latent in adult pigs. The ubiquitous nature of herpesviruses, including PCMV, means that these viruses should be a major focus in the development of xenotransplantation.
Herpesviruses are able to infect other species. Porcine cells can be infected by HCMV [17], indicating that the pig transplants may be infected when the recipient is HCMV positive, and PCMV can infect human cells [18]. Entry of HCMV into porcine endothelial cells depends on both the cellular vascular origin and the viral strain [19]. When PCMV was transmitted by the pig transplant into baboons, the baboon CMV was activated causing invasive disease and consumptive coagulopathy, the PCMV was mainly replicating in the pig transplant causing ureteric necrosis in one transplant [20], [21], [22]. When baboons received pig kidneys from PCMV-infected pigs, the survival time was 14.1 days in comparison to 48.3 or 53 days when organs from uninfected animals were transplanted [23]. In a similar experiment with cynomolgus monkeys the difference was 9.2 days versus 28.7 days [24]. Alone the presence of the virus had such an important influence on transplant survival.
When CD55-transgenic Large White pigs were analyzed, all animals were found positive for PCMV, however under specified pathogen free (spf) or designated pathogen-free (dpf) conditions PCMV-free animals were obtained [25], indicating that selection of PCMV-free animals by Caesarean delivery and spf breeding is possible. On the other hand, due to reactivation of the baboon CMV after pig cardiac xenotransplantation with pharmaceutical immunosuppression a lethal outcome in some cases was observed despite prophylactic treatment with the antiviral drugs ganciclovir or valganciclovir [26]. The Auckland island pigs, already used in clinical islet cell transplantation were shown to be free of PCMV [27].
As mentioned above, PCMV can be eliminated easily by Caesarean delivery and dpf or spf breeding of the herd [23], [25]. In addition, early weaning of the piglet from the sow can eradicate CMV [28]. To be on the safer site, a treatment with the antiviral drugs ganciclovir, cidofovir, foscarnet, acyclovir, valaciclovir, a prodrug of acyclovir, or valganciclovir can be included into the elimination protocol (Fig. 1) [29]. Concerning vaccination against HCMV, despite the urgent need for allotransplant recipients, no success was reported, although first attempts to use the major envelope glycoprotein gB have demonstrated efficacy against HCMV infection and on HCMV-induced disease [30], [31], [32]. Immunization studies with the gB protein of PCMV should be performed in pigs.
Porcine lymphotropic herpesvirus (PLHV) 1, 2, and 3 are common porcine viruses, however the prevalence and importance of these viruses for xenotransplantation is not well studied [33]. Phylogenetic analyses showed that all three PHLV clustered together with ruminant gammaherpesviruses, but the PLHV-3 is more distantly related to PLHV-1 and PLHV-2 [34]. The transmission of PHLV in pigs is not well understood. PLHV may be transmitted by pre-partum cross-placental vertical transfer and post-partum horizontal transmission, however, cross-placental transfer is not the common way [35]. Between 26% up to 88% of animals in different herds in Germany, Ireland, France, Spain and the United States were infected with one of the PLHV [34], [35], [36], [37], [38]. In contrast to PCMV, early weaning cannot eradicate PHLV [39].
4. Circoviruses
Circoviruses belong to the smallest viruses replicating autonomously in mammalian cells [40]. Porcine circovirus 1 (PCV1) has not been linked with any disease, whereas PCV2 is the causing agent of post-weaning multisystemic wasting syndrome (PMWS), a multifactorial disease in pigs [41]. This means that the presence of the virus is necessary for the disease but requires additional factors. The onset of the disease and the severity of the symptoms are influenced by the status of the immune system and genetic predisposition [41]. Characteristic clinical signs of PMWS are wasting, respiratory dysfunction, enlargement of inguinal lymph nodes, diarrhoea and a generalised depletion of lymphocytes. Although humans ingest PCV-contaminated foods and are exposed to PCV through other sources, serological evidence indicated that no transmission of PCV2 to humans took place [42]. Also, a contamination of a human vaccine with PCV was shown not to transmit PCV to humans [43], [44]. Retrospective testing confirmed the presence of PCV1 DNA in Rotarix, an oral live-attenuated human rotavirus vaccine for children, beginning with the initial stages of its development and in vaccine lots used in clinical studies conducted pre- and post-licensure [44]. When human cell lines have been infected with PCV1 and PCV2, PCV1 persisted in most cell lines without causing any visible changes, while PCV2-transfected cells showed a cytopathogenic effect [45]. Most importantly, the infection was non-productive [44], [45].
5. PERVs, the enemies in the pig genome
Porcine endogenous retroviruses are the result of a transspecies transmission of a retrovirus and integration into the genome of all pigs. PERV-A and PERV-B are polytropic viruses, infecting also human cells (for review see [46]). PERV-C infects only pig cells, it is present in most, but not all pigs. Whereas transformed human cells lacking some intracellular restrictions factors such as ABOPEC can be infected easily, human primary cells can be infected only by high-titre PERV-A and recombinant PERV-A/C after adaptation on human cells leading to an increased number of transcription factor binding sites in their long terminal repeats [47], [48], [49], [50]. Since PERV-C are present in many pigs, but not all, the selection of pigs not carrying PERV-C proviruses automatically prevents generation of high-titre recombinant PERV-A/C. Until now, no transmission of PERVs was observed, neither in first clinical trials enrolling more than 200 patients, nor in preclinical pig to non-human primate transplantation, nor in infection experiments in small animals or non-human primates with or without pharmaceutical immunosuppression (for review see [46]). However, most of the patients in the clinical trials were not exposed for a long time to the xenotransplant and with some exceptions associated with parallel kidney allotransplantations, no pharmaceutical immunosuppression was applied. In addition, no transmission of PERV was observed in numerous pig to non-human primate transplantations [51]. However, it is meanwhile clear that non-human primates are not a suitable model to study this question since non-human primates carry – in contrast to humans – a mutated receptor for PERV allowing only infection at a low efficiency [52]. Therefore, the question, whether PERVs may be transmitted during xenotransplantation is still open and an elimination of infectious proviruses is advised, since retroviruses may induce tumours and immunodeficiencies. PERVs are closely related to other gammaretroviruses such as the feline leukaemia virus (FeLV), the murine leukaemia virus (MuLV) and the Koala retrovirus (KoRV) [53]. All these related viruses induce severe immunodeficiencies and tumours in the infected host. Gammaretroviruses used as vectors in gene therapy for the treatment of a severe combined immunodeficiency in children were shown to induce leukaemia in the patients [54], indicating that transspecies transmission of gammaretroviruses into human may induce tumour development.
Highly sensitive and specific methods have been developed to screen for the prevalence and expression of PERV in donor pigs. Whereas expression of PERV in German landrace pigs is relatively low, expression in Yucatan Minipigs is high [55]. In these animals viral protein may be detected in different tissues of the animals [56] and infectious virus was found released from stimulated peripheral blood mononuclear cells (PBMCs) [55]. Crossing of Landrace pigs with Minipigs increased the expression rate of PERV [55]. When Göttingen Minipigs were screened, a relatively low expression and no release of virus particles were observed [4].
6. Elimination programs: screening, selection, treatment and isolation of donor pigs
The elimination programs for HEV and PCMV were described in detail above [5], [29]. The elimination programs for other microorganisms (with exception of PERV, see below) will be similarly, including screening of the donor pigs using specific and sensitive methods, treatment with antiviral drugs and immunisation (Fig. 1). For some porcine microorganisms effective vaccines are available, for example for circoviruses (Ingelvac CircoFLEX®) and mycoplasma (Ingelvac CircoFLEX®). Several other vaccines are available to treat pigs and prevent transmission of the infectious agents (Table 2 ) [57]. However, vaccination is only necessary, when the infectious agent is present in the pig herd and pose a risk for the transplant recipient. In the case of bacteria it has to be considered whether the donor pig (and in some cases also the recipient) should be treated with antibiotics.
Table 2.
Vaccines available against viral and bacterial infection [57].
| Viral diseases | Causing virus |
|---|---|
| Aujeszky's disease (pseudorabies) | Suid herpesvirus 1 (SuHV1) |
| Food-and-mouth disease | Food-and-mouth disease virus (aphthovirus) |
| SMEDI (stillbirth, mummification, embryonic death, and infertility) syndrome | Porcine parvovirus |
| Porcine reproductive and respiratory syndrome (PRRS) | Porcine reproductive and respiratory syndrome virus (arterivirus) |
| Swine fever | Classical swine fever virus (pestivirus) |
| Swine influenza | Swine influenza virus |
| Transmissible Gastro-Enteritis (TGE) | TGE virus (coronavirus) |
|
Bacterial diseases |
Causing bacterium |
| Necrotic pleuropneumonia | Actinobacillus pleuropneuminia |
| Atrophic rhinitis | Pasteurella multocida |
| Clostridial disease | Clostridium |
| E. coli diarrhoea | E. coli |
| Enzootic pneumonia | Mycoplasma hyopneumoniae |
| Erysipelas | Erysipelothrix rhusiopathiae |
| Glässers disease | Hemophilus parasuis |
| Leptospirosis | Leptospira pomona, L. tarassovi, L. bratislava and L. muenchen |
| Pasteurellosis | Pasteurella multocidia |
| Streptococcal meningitis | Streptococcus suis, haemophilus parasuis |
7. Strategies to prevent transfer of PERVs
Since PERVs are present in the genome of all pigs and cannot be eliminated by the way other microorganisms can be eliminated easily, other strategies were developed to prevent transmission of these infectious agents. First, animals with a low copy number of PERV and a low expression rate should be selected. Second, animals not carrying PERV-C should be selected in order to prevent PERV-A/C recombination. Third, transgenic pigs have been generated expressing small interfering RNA, specifically inhibiting the expression of PERV and therefore lowering the probability of PERV transmission [58], [59], [60], [61].Since in the genome of pigs multiple PERV proviruses were found, sometimes more than 150, an elimination of all PERV copies by genome editing using zinc finger nucleases (ZFNs) may be difficult. We selected ZFN specific for a sequence of the highly conserved pol region of the virus. However first attempts to eliminate PERVs in the pig genome failed, obviously due to the strong toxic effect when ZFNs were cutting the genome at multiple sites and therefore destabilising the genome [62]. A lower concentration of the ZFN and a gradual elimination may be alternative strategies. In addition, new attempts using CRISPR/Cas9 (Clustered Regularly Interspaced Short Palindromic Repeats/CRISPR-associated) for gene editing should be undertaken with the goal to eliminate at least the replication competent proviruses. Fourth, and last but not least, transmission of PERVs may be prevented by a vaccine. When immunising different animal species (goats, rats, mice, hamster, guinea pigs) with the recombinant transmembrane envelope protein p15E and the surface envelope protein gp70 of PERV, always effective neutralising antibodies were induced [63], [64], [65], [66]. The results of the immunisation with the transmembrane envelope protein are of great interest. In all cases neutralising antibodies were obtained and one epitope recognised was localised in the membrane proximal external region (MPER), the other in the fusion peptide proximal region [63], [64]. The epitope in the MPER was not only localised similarly as a corresponding epitope recognised by antibodies broadly neutralising HIV-1 isolated from HIV-1 infected individuals in the transmembrane envelope protein gp41 of HIV-1, but despite the evolutionary difference between PERV and HIV-1 three identical amino acids were detected in the epitopes [63], [64]. Immunisation with the surface envelope protein gp70 induced higher titres of neutralising antibodies compared with immunisation with the transmembrane envelope protein p15E [65]. Since there is no animal model which allows analysing the efficiency of the PERV vaccine in vivo, the related FeLV was studied. Cats were immunised with recombinant p15E and gp70 of FeLV. Neutralising antibodies were detected and the epitopes recognised were very similarly located compared with the epitopes recognised by the antibodies neutralising PERV [67], [68]. When the immunised cats were challenged with infectious FeLV, the animals were protected, suggesting that in the case of PERV the vaccine may also work [69]. These data indicate that immunisation of the transplant recipient may be successful in the case other prevention strategies do not work.
Ethical approval
Not applicable.
Funding
German Science Foundation, Transregio Collaborative Research Centre 127 (J.D.)
Swiss National Science Foundation (N.M.)
Author contribution
J.D. and N. J. M. contributed equally.
Conflict of interest
None.
Guarantor
J.D. and N.J.M. are the Guarantors of the work, they accept full responsibility for the work and/or the conduct of the study, had access to the data, and controlled the decision to publish.
Acknowledgment
This work was supported by the German Science Foundation, Transregio Collaborative Research Centre 127 (TRR 127) (J.D.) and the Swiss National Science Foundation (grant number 310030-130705) (N.M.).
Footnotes
Supplementary data related to this article can be found at http://dx.doi.org/10.1016/j.ijsu.2015.08.032.
Appendix A. Supplementary data
The following is the supplementary data related to this article:
References
- 1.Fishman J.A., Greenwald M.A., Grossi P.A. Transmission of infection with human allografts: essential considerations in donor screening. Clin. Infect. Dis. 2012;55:720–727. doi: 10.1093/cid/cis519. [DOI] [PubMed] [Google Scholar]
- 2.Schuurman H.J. The International Xenotransplantation Association consensus statement on conditions for undertaking clinical trials of porcine islet products in type 1 diabetes–chapter 2: source pigs. Xenotransplantation. 2009;16:215–222. doi: 10.1111/j.1399-3089.2009.00541.x. [DOI] [PubMed] [Google Scholar]
- 3.Wynyard S., Nathu D., Garkavenko O. Microbiological safety of the first clinical pig islet xenotransplantation trial in New Zealand. Xenotransplantation. 2014;21:309–323. doi: 10.1111/xen.12102. [DOI] [PubMed] [Google Scholar]
- 4.Semaan M., Rotem A., Barkai U. Screening pigs for xenotransplantation: prevalence and expression of porcine endogenous retroviruses in Göttingen minipigs. Xenotransplantation. 2013;20(3):148–156. doi: 10.1111/xen.12032. [DOI] [PubMed] [Google Scholar]
- 5.Denner J. Xenotransplantation and hepatitis E virus. Xenotransplantation. 2015 doi: 10.1111/xen.12156. [DOI] [PubMed] [Google Scholar]
- 6.V.A. Morozov, A.V. Morozov, A. Rotem, et al., Extended microbiological characterization of Göttingen minipigs in the context of xenotransplantation: vertical transmission of hepatitis E virus. PLOS One, submitted. [DOI] [PMC free article] [PubMed]
- 7.Meng X.J. Hepatitis E virus: animal reservoirs and zoonotic risk. Vet. Microbiol. 2010;140:256–265. doi: 10.1016/j.vetmic.2009.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Garkavenko O., Muzina M., Muzina Z. Monitoring for potentially xenozoonotic viruses in New Zealand pigs. J. Med. Virol. 2004;72:338–344. doi: 10.1002/jmv.10575. [DOI] [PubMed] [Google Scholar]
- 9.Busby S.A., Crossan C., Godwin J. Suggestions for the diagnosis and elimination of hepatitis E virus in pigs used for xenotransplantation. Xenotransplantation. 2013;20:188–192. doi: 10.1111/xen.12038. [DOI] [PubMed] [Google Scholar]
- 10.Kim Y., Lee C. Ribavirin efficiently suppresses porcine nidovirus replication. Virus Res. 2013;171:44–53. doi: 10.1016/j.virusres.2012.10.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wu T., Li S.W., Zhang J. Hepatitis E vaccine development: a 14 year odyssey. Hum. Vaccin. Immunother. 2012;8:823–827. doi: 10.4161/hv.20042. [DOI] [PubMed] [Google Scholar]
- 12.Sanford B.J., Opriessnig T., Kenney S.P. Assessment of the cross-protective capability of recombinant capsid proteins derived from pig, rat, and avian hepatitis E viruses (HEV) against challenge with a genotype 3 HEV in pigs. Vaccine. 2012;30:6249–6255. doi: 10.1016/j.vaccine.2012.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fishman J.A. Overview: cytomegalovirus and the herpesviruses in transplantation. Am. J. Transpl. 2013;13(Suppl. 3):1–8. doi: 10.1111/ajt.12002. quiz 8. [DOI] [PubMed] [Google Scholar]
- 14.Ramanan P., Razonable R.R. Cytomegalovirus infections in solid organ transplantation: a review. Infect. Chemother. 2013;45:260–271. doi: 10.3947/ic.2013.45.3.260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Edington N. Porcine cytomegalovirus. In: Straw B.E., Allaire S.D., Mengeling W.L., Taylor D.J., editors. Diseases of Swine. Iowa State University Press; Ames, IA: 1999. [Google Scholar]
- 16.Edington N., Watt R.G., Plowright W. Experimental transplacental transmission of porcine cytomegalovirus. J. Hyg. Lond. 1977;78:243–251. doi: 10.1017/s0022172400056138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Degre M., Ranneberg-Nilsen T., Beck S. Human cytomegalovirus productively infects porcine endothelial cells in vitro. Transplantation. 2001;72(7):1334–1337. doi: 10.1097/00007890-200110150-00028. [DOI] [PubMed] [Google Scholar]
- 18.Whitteker J.L., Dudani A.K., Tackaberry E.S. Human fibroblasts are permissive for porcine cytomegalovirus in vitro. Transplantation. 2008;86(1):155–162. doi: 10.1097/TP.0b013e31817d4823. [DOI] [PubMed] [Google Scholar]
- 19.Taveira A., Ponroy N., Mueller N.J. Entry of human cytomegalovirus into porcine endothelial cells depends on both the cellular vascular origin and the viral strain. Xenotransplantation. 2014;21(4):324–340. doi: 10.1111/xen.12097. Epub 2014. [DOI] [PubMed] [Google Scholar]
- 20.Michaels M.G., Jenkins F.J., ST George K. Detection of infectious baboon cytomegalovirus after baboon-to-human liver xenotransplantation. J. Virol. 2001;75:2825–2828. doi: 10.1128/JVI.75.6.2825-2828.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gollacker B., Mueller N.J., Houser S. Porcine cytomegalovirus and coagulopathy in pig-to-primate xenotransplantation. Transplantation. 2003;75(11):1841–1847. doi: 10.1097/01.TP.0000065806.90840.C1. [DOI] [PubMed] [Google Scholar]
- 22.Mueller N.J., Barth R.N., Yamamoto S. Activation of cytomegalovirus in pig-to-primate organ xenotransplantation. J. Virol. 2002;76:4734–4740. doi: 10.1128/JVI.76.10.4734-4740.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yamada K., Tasaki M., Sekijima M. Porcine cytomegalovirus infection is associated with early rejection of kidney grafts in a pig to baboon xenotransplantation model. Transplantation. 2014;98:411–418. doi: 10.1097/TP.0000000000000232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sekijima M., Waki S., Sahara H. Results of life-supporting galactosyltransferase knockout kidneys in cynomolgus monkeys using two different sources of galactosyltransferase knockout swine. Transplantation. 2014;98:419–426. doi: 10.1097/TP.0000000000000314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mueller N.J., Fishman J.A. Herpesvirus infections in xenotransplantation: pathogenesis and approaches. Xenotransplantation. 2004;11:486–490. doi: 10.1111/j.1399-3089.2004.00180.x. [DOI] [PubMed] [Google Scholar]
- 26.Byrne G.W., Logan J.S., McGregor C.G. Prevention, detection, and management of early bacterial and fungal infections in a preclinical cardiac xenotransplantation model that achieves prolonged survival. Xenotransplantation. 2005;12(2):127–133. doi: 10.1111/j.1399-3089.2005.00205.x. [DOI] [PubMed] [Google Scholar]
- 27.Wynyard S., Nathu D., Garkavenko O. Microbiological safety of the first clinical pig islet xenotransplantation trial in New Zealand. Xenotransplantation. 2014;4:309–323. doi: 10.1111/xen.12102. Epub 2014. [DOI] [PubMed] [Google Scholar]
- 28.Mueller N.J., Kuwaki K., Dor F.J. Reduction of consumptive coagulopathy using porcine cytomegalovirus-free cardiac porcine grafts in pig-to-primate xenotransplantation. Transplantation. 2004;78(10):1449–1453. doi: 10.1097/01.tp.0000141361.68446.1f. [DOI] [PubMed] [Google Scholar]
- 29.J. Denner. Xenotransplantation and porcine cytomegalovirus (PCMV). Xenotransplantation, submitted. [DOI] [PubMed]
- 30.Kharfan-Dabaja M.A., Boeck H.M., Wilck M.B. Reanalysis of TransVax immunogenicity. Lancet Infect. Dis. 2013;13(1):18. doi: 10.1016/S1473-3099(12)70296-7. [DOI] [PubMed] [Google Scholar]
- 31.Schleiss M.R. Developing a vaccine against congenital cytomegalovirus (CMV) infection: what have we learned from animal models? Where should we go next? Future Virol. 2013;8(12):1161–1182. doi: 10.2217/fvl.13.106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pass R.F., Zhang C., Evans A. Vaccine prevention of maternal cytomegalovirus infection. N. Engl. J. Med. 2009;360(12):1191–1199. doi: 10.1056/NEJMoa0804749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Doucette K., Dor F.J., Wilkinson R.A. Gene expression of porcine lymphotrophic herpesvirus-1 in miniature swine with posttransplant lymphoproliferative disorder. Transplantation. 2007;83(1):87–90. doi: 10.1097/01.tp.0000228237.32549.16. [DOI] [PubMed] [Google Scholar]
- 34.Chmielewicz B., Goltz M., Franz T. A novel porcine gammaherpesvirus. Virology. 2003;308(2):317–329. doi: 10.1016/s0042-6822(03)00006-0. [DOI] [PubMed] [Google Scholar]
- 35.Tucker A.W., McNeilly F., Meehan B. Methods for the exclusion of circoviruses and gammaherpesviruses from pigs. Xenotransplantation. 2003;10(4):343–348. doi: 10.1034/j.1399-3089.2003.02048.x. [DOI] [PubMed] [Google Scholar]
- 36.Chmielewicz B., Goltz M., Lahrmann K.H. Approaching virus safety in xenotransplantation: a search for unrecognized herpesviruses in pigs. Xenotransplantation. 2003;10(4):349–356. doi: 10.1034/j.1399-3089.2003.02074.x. [DOI] [PubMed] [Google Scholar]
- 37.McMahon K.J., Minihan D., Campion E.M. Infection of pigs in Ireland with lymphotropic gamma-herpesviruses and relationship to postweaning multisystemic wasting syndrome. Vet. Microbiol. 2006;116(1–3):60–68. doi: 10.1016/j.vetmic.2006.03.022. Epub 2006. [DOI] [PubMed] [Google Scholar]
- 38.Ulrich S., Goltz M., Ehlers B. Characterization of the DNA polymerase loci of the novel porcine lymphotropic herpesviruses 1 and 2 in domestic and feral pigs. J. Gen. Virol. 1999;80(12):3199–3205. doi: 10.1099/0022-1317-80-12-3199. [DOI] [PubMed] [Google Scholar]
- 39.Mueller N.J., Kuwaki K., Knosalla C. Early weaning of piglets fails to exclude porcine lymphotropic herpesvirus. Xenotransplantation. 2005;12(1):59–62. doi: 10.1111/j.1399-3089.2004.00196.x. [DOI] [PubMed] [Google Scholar]
- 40.Mankertz A., Caliskan R., Hattermann K. Molecular biology of Porcine circovirus: analyses of gene expression and viral replication. Vet. Microbiol. 2004;98(2):81–88. doi: 10.1016/j.vetmic.2003.10.014. [DOI] [PubMed] [Google Scholar]
- 41.Burbelo P.D., Ragheb J.A., Kapoor A. The serological evidence in humans supports a negligible risk of zoonotic infection from porcine circovirus type 2. Biologicals. 2013;41(6):430–434. doi: 10.1016/j.biologicals.2013.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Segalés J., Allan G.M., Domingo M. Porcine circovirus diseases. Anim. Health. Res. Rev. 2005;6(2):119–142. doi: 10.1079/ahr2005106. J. Clin. Invest. 118(9) (2008) 3132–3142. [DOI] [PubMed] [Google Scholar]
- 43.Quintana J., Segalés J., Calsamiglia M. Detection of porcine circovirus type 1 in commercial pig vaccines using polymerase chain reaction. Vet. J. 2006;171(3):570–573. doi: 10.1016/j.tvjl.2004.12.008. [DOI] [PubMed] [Google Scholar]
- 44.Dubin G., Toussaint J.F., Cassart J.P. Investigation of a regulatory agency enquiry into potential porcine circovirus type 1 contamination of the human rotavirus vaccine, Rotarix: approach and outcome. Hum. Vaccin. Immunother. 2013;9(11):2398–2408. doi: 10.4161/hv.25973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hattermann K., Roedner C., Schmitt C. Infection studies on human cell lines with porcine circovirus type 1 and porcine circovirus type 2. Xenotransplantation. 2004;11(3):284–294. doi: 10.1111/j.1399-3089.2004.00134.x. [DOI] [PubMed] [Google Scholar]
- 46.Denner J., Tönjes R.R. Infection barriers to successful xenotransplantation focusing on porcine endogenous retroviruses. Clin. Microbiol. Rev. 2012;25(2):318–343. doi: 10.1128/CMR.05011-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Denner J., Specke V., Thiesen U. Genetic alterations of the long terminal repeat of an ecotropic porcine endogenous retrovirus during passage in human cells. Virology. 2003;314(1):125–133. doi: 10.1016/s0042-6822(03)00428-8. [DOI] [PubMed] [Google Scholar]
- 48.Rodrigues Costa M., Fischer N., Gulich B. Comparison of porcine endogenous retroviruses infectious potential in supernatants of producer cells and in cocultures. Xenotransplantation. 2014 doi: 10.1111/xen.12081. (Epub ahead of print) [DOI] [PubMed] [Google Scholar]
- 49.Denner J. PERV infection of human peripheral blood mononuclear cells (PBMCs) Xenotransplantation. 2014 doi: 10.1111/xen.12150. [DOI] [PubMed] [Google Scholar]
- 50.Scheef G., Fischer N., Krach U. The number of a U3 repeat box acting as an enhancer in long terminal repeats of polytropic replication-competent porcine endogenous retroviruses dynamically fluctuates during serial virus passages in human cells. J. Virol. 2001;75:6933–6940. doi: 10.1128/JVI.75.15.6933-6940.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Denner J., Graham M. Xenotransplantation of islet cells: what can the non-human primate model bring for the evaluation of efficacy and safety? Xenotransplantation. 2015;22:231–235. doi: 10.1111/xen.12169. [DOI] [PubMed] [Google Scholar]
- 52.Mattiuzzo G., Takeuchi Y. Suboptimal porcine endogenous retrovirus infection in non-human primate cells: implication for preclinical xenotransplantation. PLoS One. 2010;5(10):e13203. doi: 10.1371/journal.pone.0013203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Denner J., Young P.R. Koala retroviruses: characterization and impact on the life of koalas. Retrovirology. 2013;10(1):108. doi: 10.1186/1742-4690-10-108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hacein-Bey-Abina S., Garrigue A., Wang G.P. Insertional oncogenesis in 4 patients after retrovirus-mediated gene therapy of SCID-X1. J. Clin. Invest. 2008;118(9):3132–3142. doi: 10.1172/JCI35700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Dieckhoff B., Kessler B., Jobst D. Distribution and expression of porcine endogenous retroviruses in multi-transgenic pigs generated for xenotransplantation. Xenotransplantation. 2009;16(2):64–73. doi: 10.1111/j.1399-3089.2009.00515.x. [DOI] [PubMed] [Google Scholar]
- 56.Bittmann I., Mihica D., Plesker R. Expression of porcine endogenous retroviruses (PERV) in different organs of a pig. Virology. 2012;433(2):329–336. doi: 10.1016/j.virol.2012.08.030. [DOI] [PubMed] [Google Scholar]
- 57.http://www.thepigsite.com/pighealth/article/41/vaccination/.
- 58.Ramsoondar J., Vaught T., Ball S. Production of transgenic pigs that express porcine endogenous retrovirus small interfering RNAs. Xenotransplantation. 2009;16:164–180. doi: 10.1111/j.1399-3089.2009.00525.x. [DOI] [PubMed] [Google Scholar]
- 59.Dieckhoff B., Petersen B., Kues W.A. Knockdown of porcine endogenous retrovirus (PERV) expression by PERV-specific shRNA in transgenic pigs. Xenotransplantation. 2008;15:36–45. doi: 10.1111/j.1399-3089.2008.00442.x. [DOI] [PubMed] [Google Scholar]
- 60.Semaan M., Kaulitz D., Petersen B. Long-term effects of PERV-specific RNA interference in transgenic pigs. Xenotransplantation. 2012;19(2):112–121. doi: 10.1111/j.1399-3089.2012.00683.x. [DOI] [PubMed] [Google Scholar]
- 61.Karlas A., Kurth R., Denner J. Inhibition of porcine endogenous retroviruses by RNA interference: increasing the safety of xenotransplantation. Virology. 2004;325(1):18–23. doi: 10.1016/j.virol.2004.04.022. [DOI] [PubMed] [Google Scholar]
- 62.Semaan M., Ivanusic D., Denner J. Attempts to knock out multiple porcine endogenous retrovirus (PERV) sequences in the pig genome by zinc finger nucleases (ZFN) PLoS One. 2015 doi: 10.1371/journal.pone.0122059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Fiebig U., Stephan O., Kurth R. Neutralizing antibodies against conserved domains of p15E of porcine endogenous retroviruses (PERVs): basis for a vaccine for xenotransplantation? Virology. 2003;307:406–413. doi: 10.1016/s0042-6822(02)00140-x. [DOI] [PubMed] [Google Scholar]
- 64.Kaulitz D., Mihica D., Dorna J. Development of sensitive methods for detection of porcine endogenous retrovirus-C (PERV-C) in the genome of pigs. J. Virol. Methods. 2011;175:60–65. doi: 10.1016/j.jviromet.2011.04.017. [DOI] [PubMed] [Google Scholar]
- 65.Denner J., Mihica D., Kaulitz D. Increased titers of neutralizing antibodies after immunization with both envelope proteins of the porcine endogenous retroviruses (PERVs) Virol. J. 2012;9:260. doi: 10.1186/1743-422X-9-260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Waechter A., Denner J. Novel neutralising antibodies targeting the N-terminal helical region of the transmembrane envelope protein p15E of the porcine endogenous retrovirus (PERV) Immunol. Res. 2014;58(1):9–19. doi: 10.1007/s12026-013-8430-y. [DOI] [PubMed] [Google Scholar]
- 67.Langhammer S., Fiebig U., Kurth R. Neutralising antibodies against the transmembrane protein of feline leukaemia virus (FeLV) Vaccine. 2005;23(25):3341–3348. doi: 10.1016/j.vaccine.2005.01.091. [DOI] [PubMed] [Google Scholar]
- 68.Langhammer S., Fiebig U., Kurth R. Increased neutralizing antibody response after simultaneous immunization with leucogen and the feline leukemia virus transmembrane protein. Intervirology. 2011;54(2):78–86. doi: 10.1159/000318892. [DOI] [PubMed] [Google Scholar]
- 69.Langhammer S., Hübner J., Jarrett O. Immunization with the transmembrane protein of a retrovirus, feline leukemia virus: absence of antigenemia following challenge. Antivir. Res. 2011;89(1):119–123. doi: 10.1016/j.antiviral.2010.11.011. Epub 2010. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.